Bio-Based Poly(Ether Imide)s from Isohexide-Derived Isomeric Dianhydrides
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Monomers
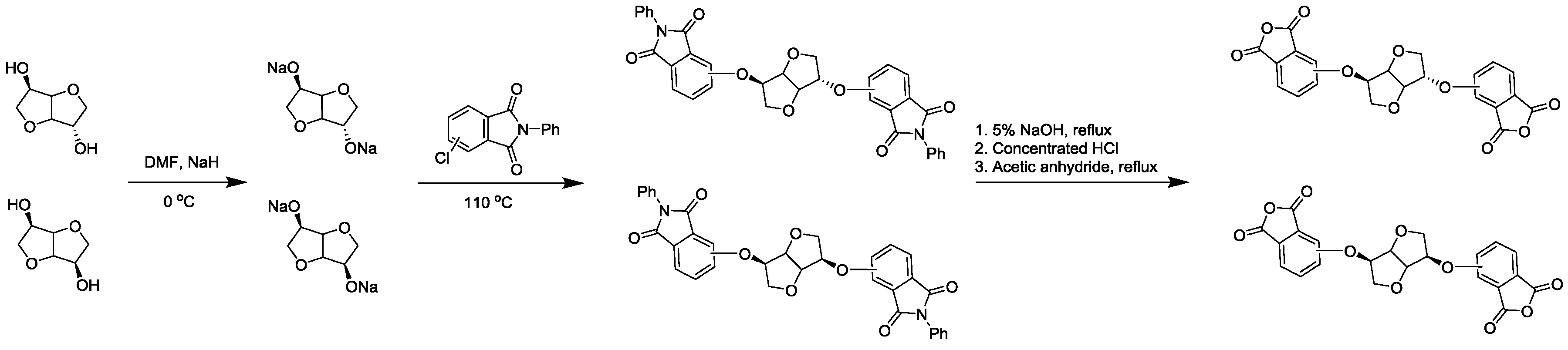
2.2.1. 1,4:3,6-Dianhydro-2,5-di-O-(3-(N-phenyl-phthalimido))-d-sorbitol
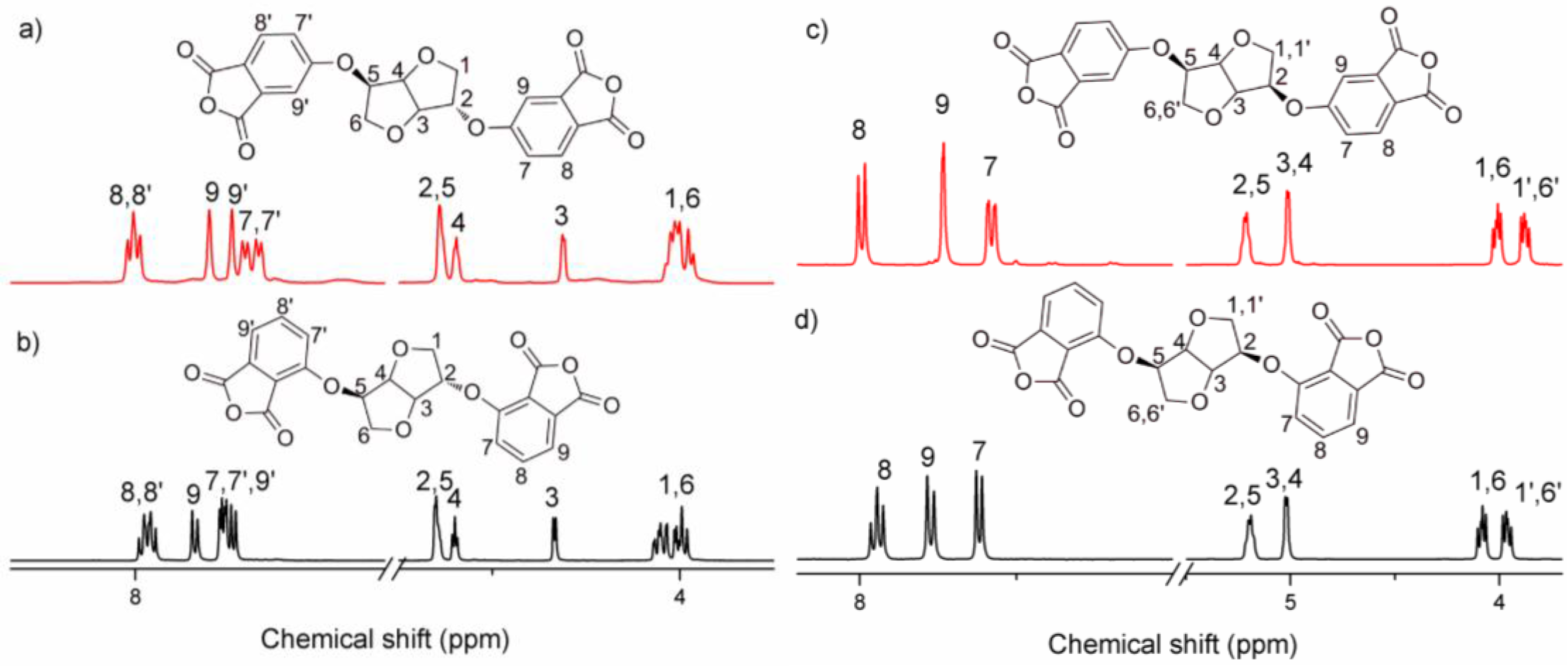
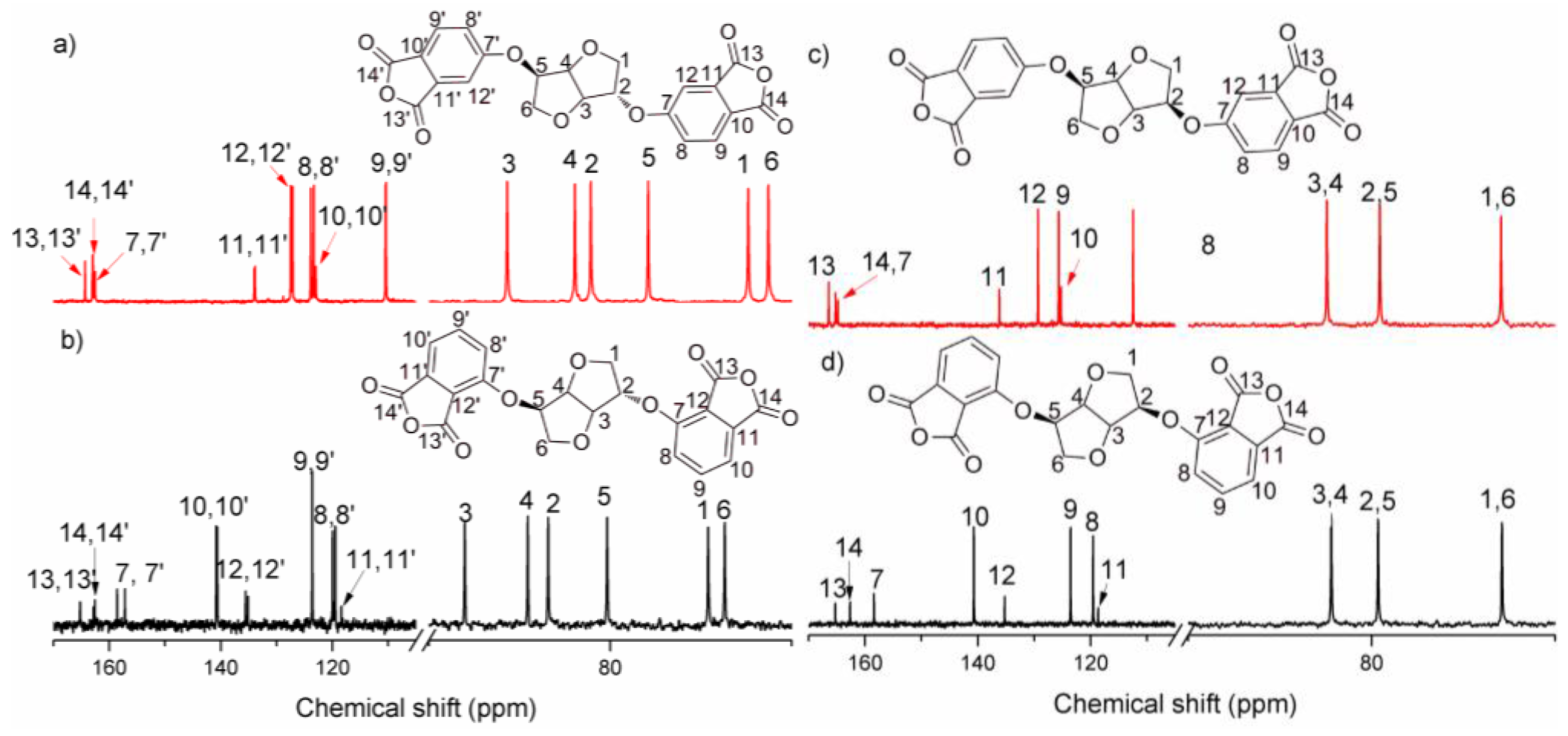
2.2.2. 1,4:3,6-Dianhydro-2,5-di-O-(2,3-dicarboxyphenyl)-d-sorbitol Dianhydride (3,3′-ISDPA)
2.2.3. 1,4:3,6-Dianhydro-2,5-di-O-(2,3-dicarboxyphenyl)-d-mannitol Dianhydride (3,3′-IMDPA)
2.2.4. 1,4:3,6-Dianhydro-2,5-di-O-(3,4-dicarboxyphenyl)-d-sorbitol Dianhydride (4,4′-ISDPA)
2.2.5. 1,4:3,6-Dianhydro-2,5-di-O-(3,4-dicarboxyphenyl)-d-mannitol Dianhydride (4,4′-IMDPA)
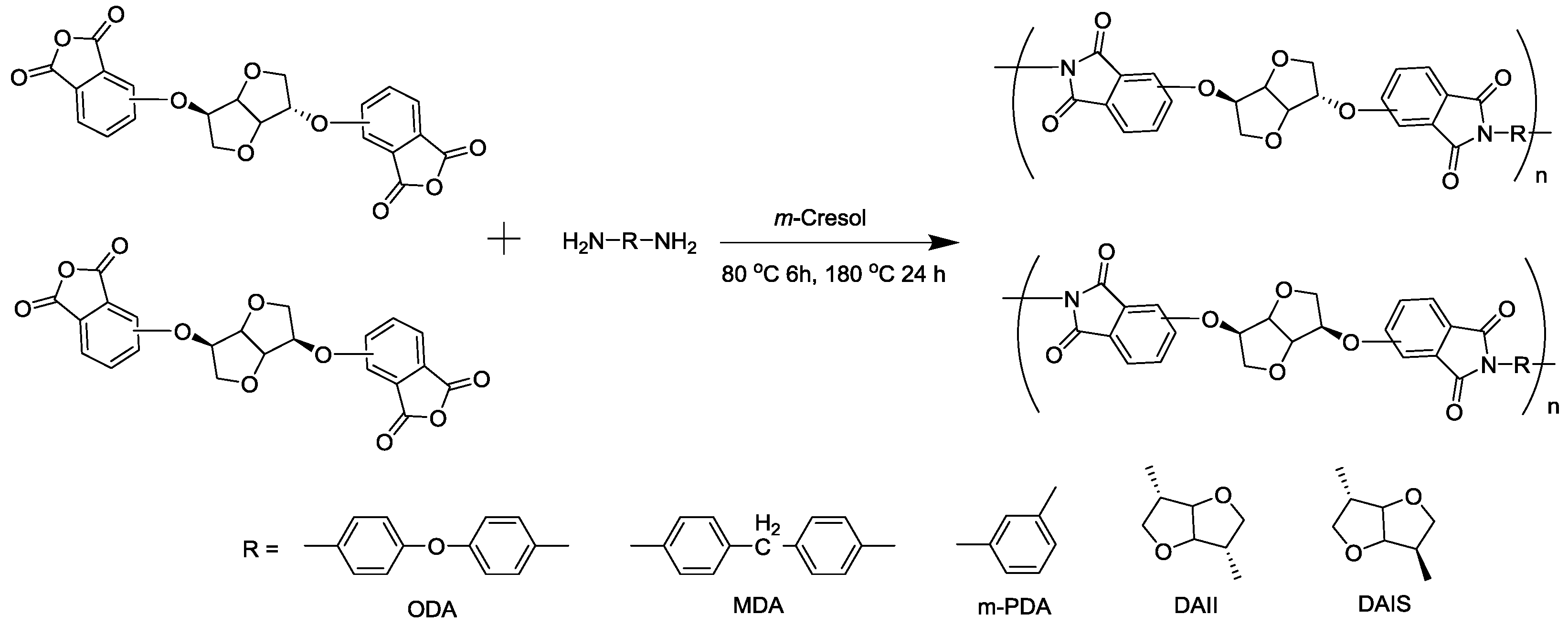
2.3. Synthesis of Polymers
2.4. Film Formation and Characterization
3. Results and Discussion
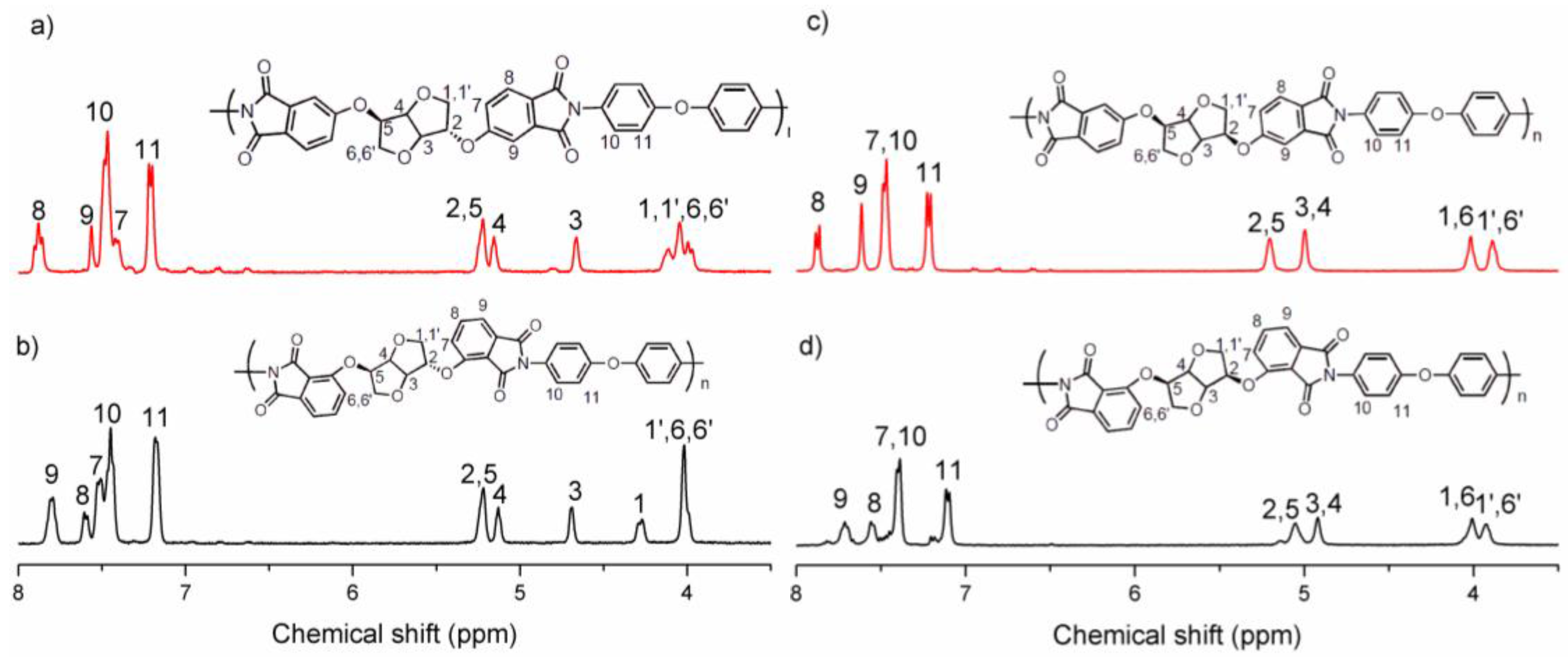
3.1. Synthesis of Monomers
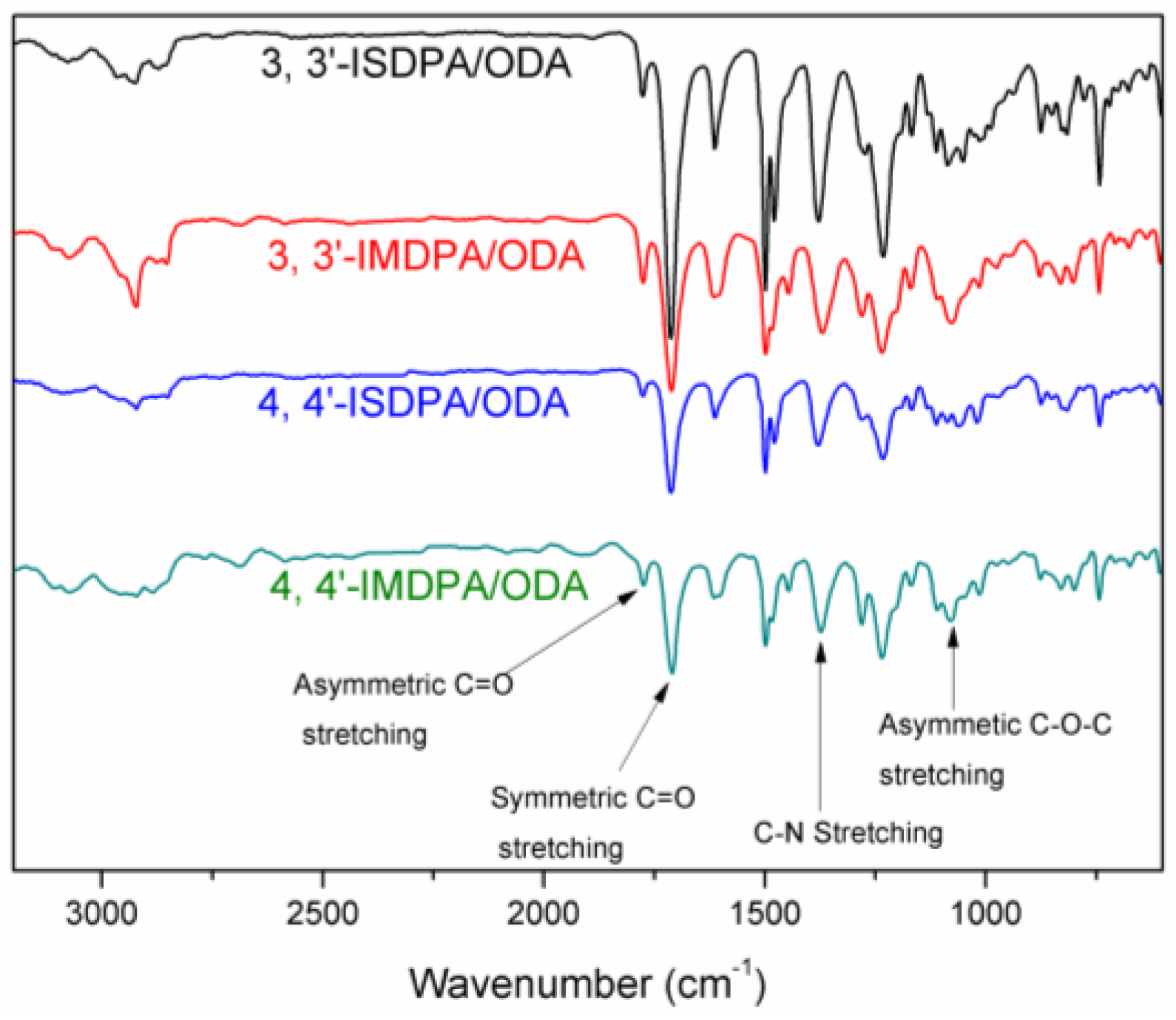
3.2. Synthesis of Polymers
3.3. Properties of Bio-Based Poly(Ether Imide)s
3.3.1. Bio-Based Contents and Solubility
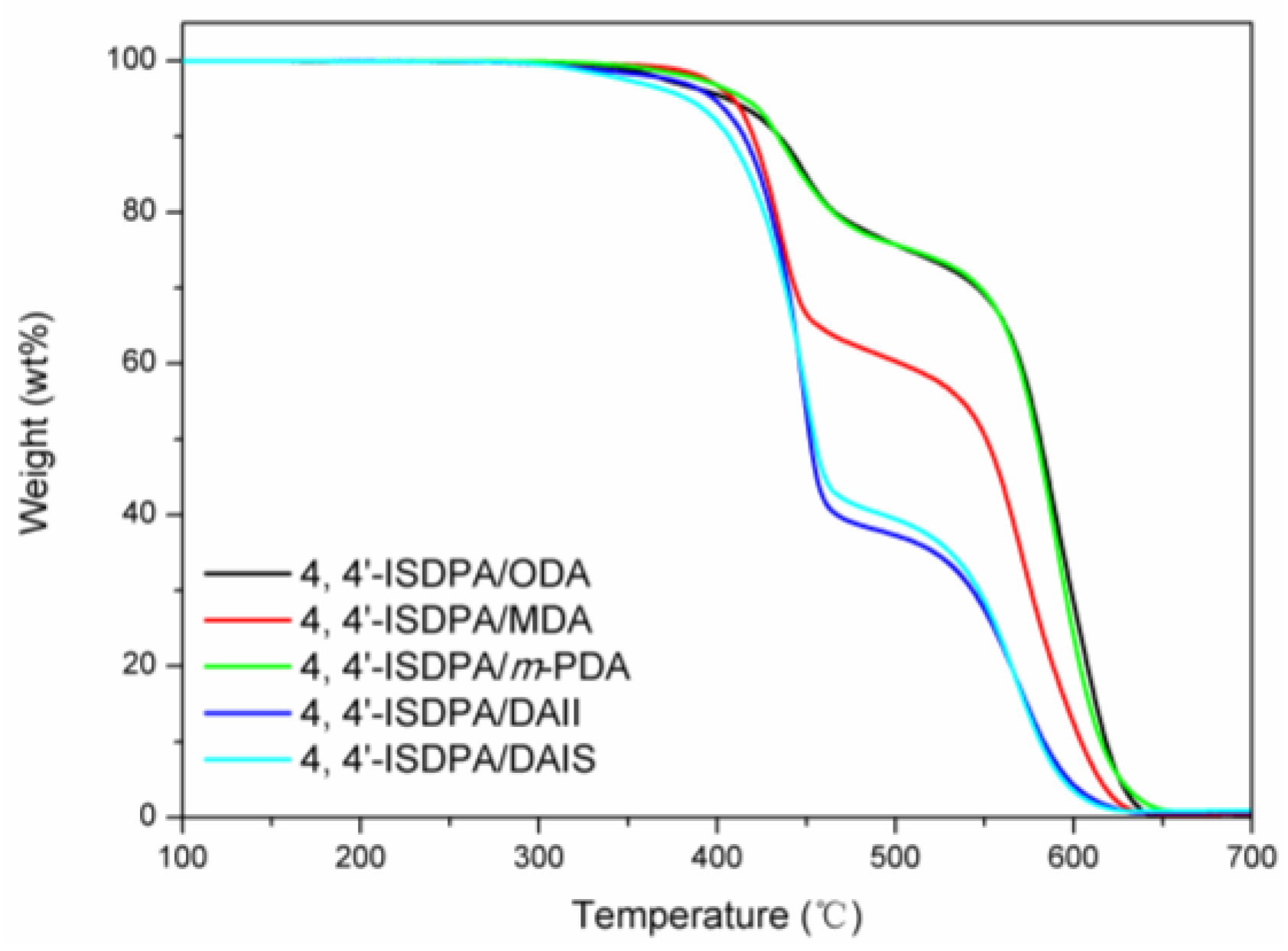
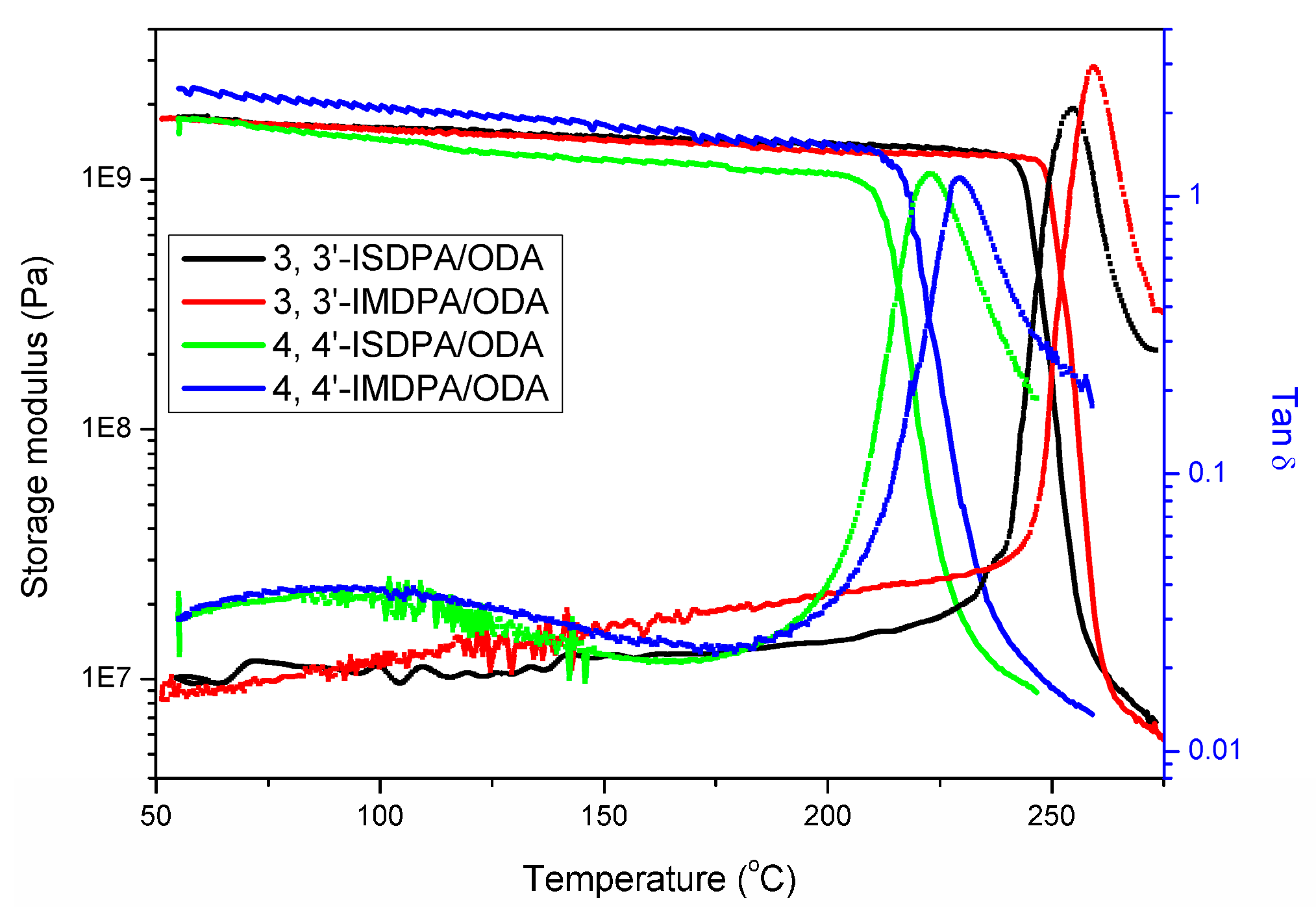
3.3.2. Thermal Properties
3.3.3. Mechanical Properties
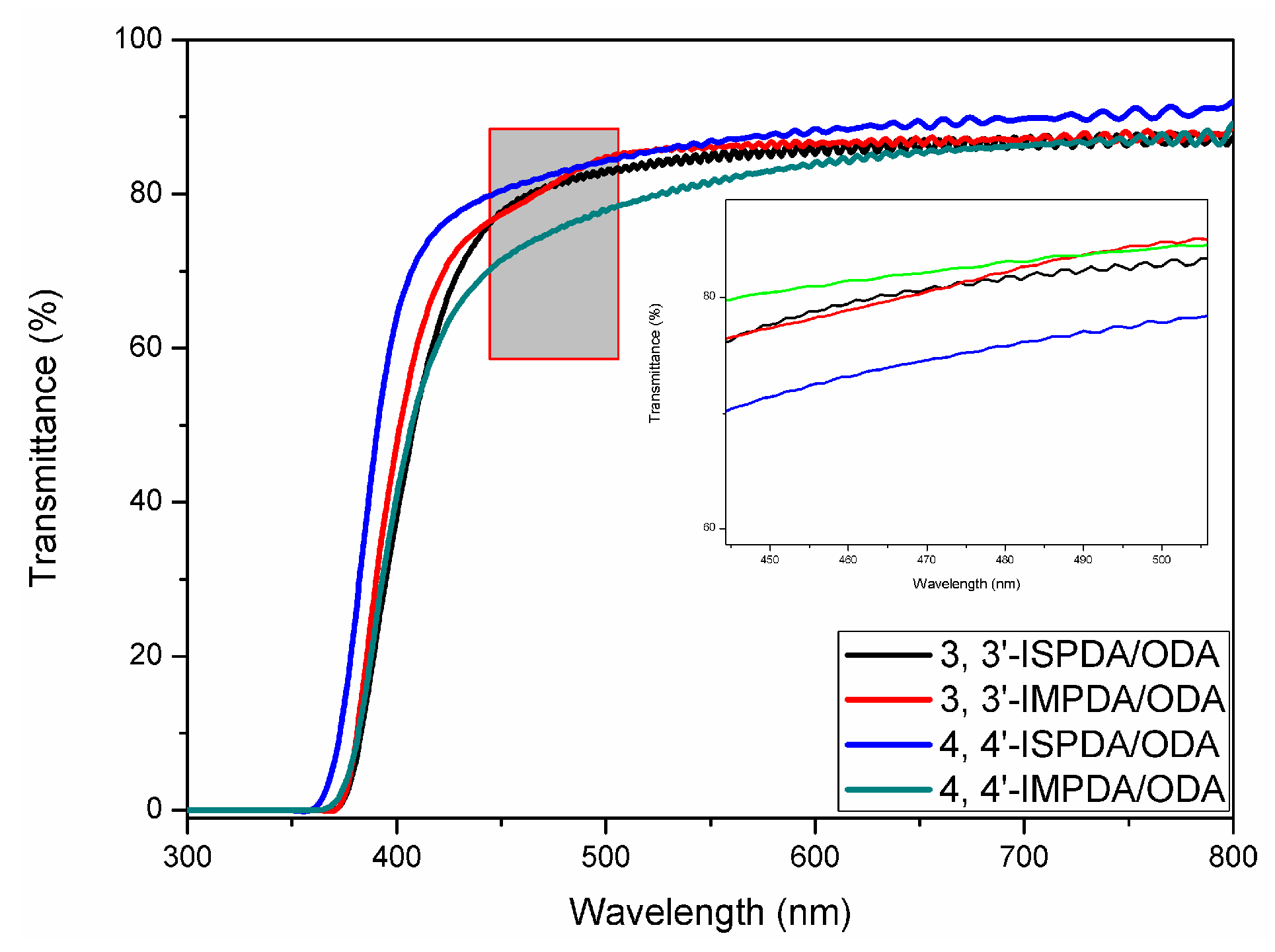
3.3.4. Optical Properties
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Delidovich, I.; Hausoul, P.J.C.; Deng, L.; Pfützenreuter, R.; Rose, M.; Regina Palkovits, R. Alternative Monomers Based on Lignocellulose and Their Use for Polymer Production. Chem. Rev. 2016, 116, 1540–1599. [Google Scholar] [CrossRef] [PubMed]
- Galbis, J.A.; García-Martín, M.G.; de Paz, M.V.; Galbis, E. Synthetic Polymers from Sugar-Based Monomers. Chem. Rev. 2016, 116, 1600–1636. [Google Scholar] [CrossRef] [PubMed]
- Gandini, A.; Lacerda, T.M.; Carvalho, A.J.F.; Trovatti, E. Progress of Polymers from Renewable Resources: Furans, Vegetable Oils, and Polysaccharides. Chem. Rev. 2016, 116, 1637–1669. [Google Scholar] [CrossRef] [PubMed]
- Braun, D.; Bergmann, M. 1,4:3,6-Dianhydrohexite als Bausteine für Polymere. Adv. Synth. Catal. 1992, 334, 298–310. [Google Scholar]
- Rose, M.; Palkovits, R. Isosorbide as a Renewable Platform Chemical for Versatile Applications-Quo Vadis? ChemSusChem 2012, 5, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Fenouillot, F.; Rousseau, A.; Colomines, G.; Saint-Loup, R.; Pascault, J.-P. Polymers from Renewable 1,4:3,6-Dianhydrohexitols (isosorbide, isomannide and isoidide): A Review. Prog. Polym. Sci. 2010, 35, 578–622. [Google Scholar] [CrossRef]
- Scott, A. Roquette embraces biobased materials. C&EN 2014, 90, 16–17. [Google Scholar]
- Yoon, W.J.; Oh, K.S.; Koo, J.M.; Kim, J.R.; Lee, K.J.; Im, S.S. Advanced Polymerization and Properties of Biobased High Tg polyester of Isosorbide and 1,4-Cyclohexanedicarboxylic Acid through in situ Acetylation. Macromolecules 2013, 46, 2930–2940. [Google Scholar] [CrossRef]
- Kricheldorf, H.R.; Weidner, S.M. Copolyesters of Lactide, Isosorbide, and Terephthalic Acid-Biobased, Biodegradable, High-Tg Engineering Plastics. Macromol. Chem. Phys. 2013, 214, 726–733. [Google Scholar] [CrossRef]
- Li, Q.; Zhu, W.; Li, C.; Guan, G.; Zhang, D.; Xiao, Y.; Zheng, L. A non-phosgene process to homopolycarbonate and copolycarbonates of isosorbide using dimethyl carbonate: Synthesis, characterization, and properties. J. Polym. Sci. Polym. Chem. 2013, 51, 1387–1397. [Google Scholar] [CrossRef]
- Lee, C.H.; Takagi, H.; Okamoto, H.; Kato, M. Improving the mechanical properties of isosorbide copolycarbonates by varying the ratio of comonomers. J. Appl. Polym. Sci. 2013, 127, 530–534. [Google Scholar] [CrossRef]
- Philip, B.; Sreekumar, K. Optically active poly(ester-amide)s: Synthesis and characterization. Polym. Int. 2001, 50, 1318–1323. [Google Scholar] [CrossRef]
- Mitsubishi Chemical. World-First-DURABIO, Bio-Based Engineering Plastic from Mitsubishi Chemical, Used on the Front Panel of Sharp’s New AQUOS CRYSTAL 2 Smartphone. Available online: http://www.m-kagaku.co.jp/english/newsreleases/00258.html (accessed on 22 September 2017).
- Mitsubishi Chemical. New Biobased Engineering Plastic “DURABIO”. Available online: http://www.m-kagaku.co.jp/english/products/business/polymer/sustainable/details/1194667_3255.html (accessed on 22 September 2017).
- Belgacem, C.; Medimagh, R.; Kricheldorf, H.; Romdhane, H.B.; Chatti, S. Copolyethersulfones of 1,4:3,6-dianhydrohexitols and bisphenol A. Des. Monomers Polym. 2016, 19, 248–255. [Google Scholar] [CrossRef]
- Park, S.; Choi, J.; Ju, S.; Jegal, J.; Lee, K.M.; Hwang, S.Y.; Oh, D.X.; Park, J. Copolycarbonates of bio-based rigid isosorbide and flexible 1,4-cyclohexanedimethanol: Merits over bisphenol-A based polycarbonate. Polymer 2017, 116, 153–159. [Google Scholar] [CrossRef]
- Montgomery, R.; Wiggins, L.F. The Anhydrides of Polyhydric Alcohols. Part V. 2:5-Diamino 1:4–3:6-Dianhydro Mannitol and Sorbitol and Their Sulphanilamide Derivatives. J. Chem. Soc. 1946, 393–396. [Google Scholar] [CrossRef]
- Thiyagarajan, S.; Gootjes, L.; Vogelzang, W.; van Haveren, J.; Lutz, M.; van Es, D.S. Renewable Rigid Diamines: Efficient, Stereospecific Synthesis of High Purity Isohexide Diamines. ChemSusChem 2011, 4, 1823–1829. [Google Scholar] [CrossRef] [PubMed]
- Thiyagarajan, S.; Gootjes, L.; Vogelzang, W.; van Haveren, J.; Lutz, M.; van Es, D.S. Chiral Building Blocks from Biomass: 2,5-Diamino-2,5-dideoxy-1,4:3,6-dianhydroiditol. Tetrahedron 2011, 67, 383–389. [Google Scholar] [CrossRef]
- Hayes, R.; Brandenburg, C. New Bis(2-hydroxyethyl)isosorbide Used as Monomer for Polymer Materials, e.g., Polyesters, Polyurethanes and Polycarbonates. US Patent US6608167-B1, 19 August 2003. [Google Scholar]
- Wu, J.; Eduard, P.; Thiyagarajan, S.; Van Haveren, J.; Van Es, D.S.; Koning, C.E.; Luts, M.; Guerra, C.F. Isohexide Derivatives from Renewable Resources as Chiral Building Blocks. ChemSusChem 2011, 4, 599–603. [Google Scholar] [CrossRef] [PubMed]
- Caouthar, A.A.; Roger, P.; Tessier, M.; Chatti, S.; Blais, J.C.; Bortolussi, M. Synthesis and Characterization of New Polyamides Derived from di(4-cyanophenyl)isosorbide. Eur. Polym. J. 2007, 43, 220–230. [Google Scholar] [CrossRef]
- Bachmann, F.; Reimer, J.; Ruppenstein, M.; Thiem, J. Synthesis of a novel starch-derived AB-type polyurethane. Macromol. Rapid Commun. 1998, 19, 21–26. [Google Scholar] [CrossRef]
- Zenner, M.D.; Xia, Y.; Chen, J.S.; Kessler, M.R. Polyurethanes from Isosorbide-Based Diisocyanates. ChemSusChem 2013, 6, 1182–1185. [Google Scholar] [CrossRef] [PubMed]
- Thiem, J.; Luders, H. Synthesis and Properties of Polyurethanes Derived from Diaminodianhydroalditols. Makromol. Chem. Phys. 1986, 187, 2775–2785. [Google Scholar] [CrossRef]
- Besset, C.; Pascault, J.; Fleury, E.; Drockenmuller, E.; Bernard, J. Structure-properties Relationship of Biosourced Stereocontrolled Polytriazoles from Click Chemistry Step Growth Polymerization of Diazide and Dialkyne Dianhydrohexitols. Biomacromolecules 2010, 11, 2797–2803. [Google Scholar] [CrossRef] [PubMed]
- Thiem, J.; Bachmann, F. Synthesis and Properties of polyamides Derived from Anhydroalditols and Dianhydroalditols. Macromol. Chem. Phys. 1991, 192, 2163–2182. [Google Scholar] [CrossRef]
- Caouthar, A.; Loupy, A.; Bortolussi, M.; Blais, J.C.; Dubreucq, L.; Meddour, A. Synthesis and Characterization of New Polyamides Based on Diphenylaminoisosorbide. J. Polym. Sci. Polym. Chem. 2005, 43, 6480–6491. [Google Scholar] [CrossRef]
- Bachmann, F.; Reimer, J.; Ruppenstein, M.; Thiem, J. Synthesis of Novel Polyurethanes and Polyureas by Polyaddition Reactions of Dianhydrohexitol Configurated Diisocyanates. Marcomol. Chem. Phys. 2001, 202, 3410–3419. [Google Scholar] [CrossRef]
- Wu, J.; Jasinska, L.; Dudenko, D.; Rozanski, A.; Hansen, M.R.; van Es, D.; Koning, C.E. An Investigation of Polyamides Based on Isoidide-2,5-dimethyleneamine as a Green Rigid Building Block with Enhanced Reactivity. Macromolecules 2012, 45, 9333–9346. [Google Scholar] [CrossRef]
- Jasinska, L.; Villani, M.; Dudenko, D.; van Asselen, O.; Klop, E.; Rastogi, S.; Hansen, M.R.; Koning, C.E. Novel, Fully Biobased Semicrystalline Polyamides. Macromolecules 2011, 44, 3458–3466. [Google Scholar] [CrossRef]
- Jasinska, L.; Villani, M.; Dudenko, D.; van Asselen, O.; Klop, E.; Rastogi, S.; Hansen, M.R.; Koning, C.E. Local Conformation and Cocrystallization Phenomena in Renewable Diaminoisoidide-based Polyamides Studied by FT-IR, Solid State NMR, and WAXD. Macromolecules 2012, 45, 2796–2808. [Google Scholar] [CrossRef]
- Jasinska, L.; Dudenko, D.; Rozanski, A.; Thiyagarajan, S.; Sowinski, P.; van Es, D.; Shu, J.; Hansen, M.R.; Koning, C.E. Structure and Molecular Dynamics in Renewable Polyamides from Dideoxy-diamino Isohexide. Macromolecules 2012, 45, 5653–5666. [Google Scholar] [CrossRef]
- Gallagher, J.J.; Hillmyer, M.A.; Reineke, T.M. Acrylic Triblock Copolymers Incorporating Isosorbide for Pressure Sensitive Adhesives. ACS Sustain. Chem. Eng. 2016, 4, 3379–3387. [Google Scholar] [CrossRef]
- Sroog, C.E. Polyimides. Prog. Polym. Sci. 1991, 16, 561–694. [Google Scholar] [CrossRef]
- Wilson, D.; Stenzewberger, H.D.; Hergenrother, P.M. Polyimides; Springer: Dordrecht, The Netherlands, 1990; pp. 58–77. [Google Scholar]
- Abajo, J.H.; Campa, J.G. Processable Aromatic Polyimides. Adv. Polym. Sci. 1999, 140, 23–59. [Google Scholar]
- Ding, M. Isomeric Polyimides. Prog. Polym. Sci. 2007, 32, 623–668. [Google Scholar] [CrossRef]
- Martin, E.R.; Timothy, E.L. Synthetic Methods in Step-Growth Polymers; John Wiley & Son: Hoboken, NJ, USA, 2003; pp. 265–326. [Google Scholar]
- Pohanish, R.P. Sitting’s Handbook of Toxic and Hazardous Chemicals and Carcinogens, 6th ed.; Elsevier Science Publisher: Amsterdam, The Netherlands, 2012; pp. 873–875. [Google Scholar]
- Suvannasara, P.; Tateyama, S.; Miyasato, A.; Matsumura, K.; Shimoda, T.; Ito, T.; Yamagata, Y.; Fujita, T.; Takaya, N. Biobased Polyimides from 4-Aminocinnamic Acid Photodimer. Macromolecules 2014, 47, 1586–1593. [Google Scholar] [CrossRef]
- Kumar, A.; Tateyama, S.; Yasaki, K.; Ali, M.A.; Takaya, N.; Singh, R.; Kaneko, T. Ultrahigh performance bio-based polyimides from 4,4′-diaminostilbene. Polymer 2016, 83, 182–189. [Google Scholar] [CrossRef]
- Hu, J.; Wang, Z.; Lu, Z.; Chen, C.; Shi, M.; Wang, J.; Zhao, E.; Zeng, K.; Yang, G. Bio-based adenine-containing high performance polyimide. Polymer 2017, 119, 59–65. [Google Scholar] [CrossRef]
- Shingte, R.D.; Tawade, B.V.; Wadgaonkar, P.P. Partially Bio-based Processable Polyimides Based on Aromatic Diamine Derived from Cardanol. Green Mater. 2017, 5, 1–11. [Google Scholar]
- Ji, X.; Wang, Z.K.; Yan, J.; Wang, Z. Partially Bio-based Polyimides from Isohexide-derived Diamines. Polymer 2015, 74, 38–45. [Google Scholar] [CrossRef]
- Ji, X.; Yan, J.; Liu, X.; Wang, Z.K.; Wang, Z. Synthesis and Properties of Polyimides Derived from Bis(4-aminophenyl)isohexides. High Perform. Polym. 2017, 29, 197–204. [Google Scholar] [CrossRef]
- Mi, Z.; Liu, Z.; Tian, C.; Zhao, X.; Zhou, H.; Wang, D.; Chen, C. Soluble polyimides containing 1,4:3,6-dianhydro-d-glucidol and fluorinated units: Preparation, characterization, optical, and dielectric properties. J. Polym. Sci. Polym. Chem. 2017, 55, 3253–3265. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, R.; Huang, H.; Liu, L.; Wang, L.; Chen, Y. Synthesis of Novel Biobased Polyimides Derived from Isomannide with Good Optical Transparecy, Solubility and Thermal Stability. RSC Adv. 2015, 5, 67574–67582. [Google Scholar] [CrossRef]
- Rozhanskii, I.; Okuyama, K.; Goto, K. Synthesis and properties of polyimides derived from isomeric biphenyltetracarboxylic dianhydrides. Polymer 2000, 41, 7057–7065. [Google Scholar] [CrossRef]
- Norton, G.A.; Devlin, S.L. Determining the Modern Carbon Content of Biobased Products Using Radiocarbon Analysis. Bioresour. Technol. 2006, 97, 2084–2090. [Google Scholar] [CrossRef] [PubMed]
- Takekoshi, T.; Kochanowski, J.E.; Manello, J.S.; Webber, M.J. Polyetherimides: Synthesis and characterization. Polym. Prepr. 1983, 24, 312–313. [Google Scholar]
- Wu, Z.; Han, B.; Zhang, C.; Zhu, D.; Gao, L.; Ding, M.; Yang, Z. Novel soluble and optically active polyimides containing axially asymmetric 9,9′-spirobifluorene units: Synthesis, thermal, optical and chiral properties. Polymer 2012, 53, 5706–5716. [Google Scholar] [CrossRef]
- Zhi, J.; Guan, Y.; Cui, J.; Liu, A.; Zhu, Z.; Wan, X.; Zhou, Q. Synthesis and characterization of optically active helical vinyl polymers via free radical polymerization. J. Polym. Sci. Polym. Chem. 2009, 47, 2408–2421. [Google Scholar] [CrossRef]
- Abe, M.; Ohmori, K.; Suzuki, K.; Yamamoto, T. Synthesis and chiroptical properties of π-conjugated polymer consisting of dihydropentahelicene units with axial chirality. J. Polym. Sci. Polym. Chem. 2010, 48, 1844–1848. [Google Scholar] [CrossRef]
- Kieber, R.J., III; Silver, S.A.; Kennemur, J.G. Stereochemical effects on the mechanical and viscoelastic properties of renewable polyurethanes derived from isohexides and hydroxymethylfurfural. Polym. Chem. 2017, 8, 4822–4829. [Google Scholar] [CrossRef]
- Mi, Q.; Gao, L.; Ding, M. Optically active aromatic polyimides having axially dissymmetric 1,1′-binaphthalene-2,2′-diyl units. Macromolecules 1996, 29, 5758–5759. [Google Scholar] [CrossRef]
- Mi, Q.; Gao, L.; Li, L.; Ma, Y.; Zhang, X.; Ding, M. Synthesis and properties of optically active aromatic polyimides derived from optically pure 1,1′-bi-2-naphthol. J. Polym. Sci. Polym. Chem. 1997, 35, 3287–3297. [Google Scholar] [CrossRef]
- Mi, Q.; Ma, Y.; Gao, L.; Ding, M. Synthesis and characterization of optically active aromatic polyimides derived from 2,2-bis(2-trifluoro-4-aminophenoxy)-1,1-binaphthyl and aromatic tetracarboxylic dianhydrides. J. Polym. Sci. Polym. Chem. 1999, 37, 4536–4540. [Google Scholar] [CrossRef]
- Song, N.; Gao, L.; Ding, M. Optically active poly(amide-imide)s containing axially dissymmetric 1,1-binaphthyl moieties. J. Polym. Sci. Polym. Chem. 1999, 37, 3147–3154. [Google Scholar] [CrossRef]
- Song, N.; Qi, W.; Qiu, X.; Gao, L.; Ding, M. Synthesis and chirooptical properties of optically active poly(ester imide)s based on axially asymmetric 1,1′-binaphthyls. J. Polym. Sci. Polym. Chem. 2004, 42, 4318–4326. [Google Scholar] [CrossRef]
- Kudo, K.; Nonokawa, D.; Li, J.; Shiraishi, S. Synthesis of optically active alicyclic polyimides from a chiral, nonracemic dianhydride. J. Polym. Sci. Polym. Chem. 2002, 40, 4038–4044. [Google Scholar] [CrossRef]

| Polyimides | ηinh (dL·g−1) a | Bio-Based content (%) b | Solubility c | |||||
|---|---|---|---|---|---|---|---|---|
| m-Cresol | DMAc | CHCl3 | NMP | DMSO | 1,4-Dioxane | |||
| 3,3′-ISDPA/ODA | 0.69 | 17.6 | ++ | ++ | - | ++ | + | +- |
| 3,3′-ISDPA/MDA | 0.56 | 17.1 | ++ | ++ | +- | ++ | +- | +- |
| 3,3′-ISDPA/m-PDA | 0.54 | 21.4 | ++ | + | - | ++ | + | - |
| 3,3′-ISDPA/DAII | 0.51 | 44.4 | ++ | +- | - | ++ | + | +- |
| 3,3′-ISDPA/DAIS | 0.75 | 44.4 | ++ | +- | +- | ++ | + | - |
| 3,3′-IMDPA/ODA | 1.50 | 17.6 | ++ | ++ | - | ++ | ++ | - |
| 3,3′-IMDPA/MDA | 1.30 | 17.1 | ++ | + | - | ++ | ++ | - |
| 3,3′-IMDPA/m-PDA | - d | 21.4 | +- | + | - | ++ | ++ | - |
| 3,3′-IMDPA/DAII | 2.40 | 44.4 | ++ | - | - | - | - | - |
| 3,3′-IMDPA/DAIS | 0.72 | 44.4 | ++ | +- | - | +- | - | - |
| 4,4′-ISDPA/ODA | 0.69 | 17.6 | ++ | + | +- | + | +- | - |
| 4,4′-ISDPA/MDA | 0.59 | 17.1 | ++ | +- | +- | ++ | +- | - |
| 4,4′-ISDPA/m-PDA | 0.46 | 21.4 | ++ | +- | - | + | + | - |
| 4,4′-ISDPA/DAII | 0.41 | 44.4 | ++ | +- | +- | ++ | +- | - |
| 4,4′-ISDPA/DAIS | 0.98 | 44.4 | ++ | - | - | +- | +- | - |
| 4,4′-IMDPA/ODA | 0.72 | 17.6 | ++ | - | - | +- | +- | - |
| 4,4′-IMDPA/MDA | 1.61 | 17.1 | ++ | + | - | + | + | - |
| 4,4′-IMDPA/m-PDA | 0.46 | 21.4 | ++ | + | - | ++ | + | - |
| 4,4′-IMDPA/DAII | 0.44 | 44.4 | ++ | - | +- | ++ | + | - |
| 4,4′-IMDPA/DAIS | 0.76 | 44.4 | ++ | - | +- | + | + | - |
| Polyimides | Tg a (°C) | Tg b (°C) | T5% c (°C) | Tensile strength (MPa) | Modulus (GPa) | Elongation at break (%) |
|---|---|---|---|---|---|---|
| 3,3′-ISDPA/ODA | 255 | 250 | 413 | 114 | 2.7 | 7.6 |
| 3,3′-ISDPA/MDA | 247 | 247 | 404 | 101 | 2.5 | 6.8 |
| 3,3′-ISDPA/m-PDA | 260 | 260 | 399 | 80 | 3.1 | 2.9 |
| 3,3′-ISDPA/DAII | 255 | 249 | 384 | 80 | 3.6 | 2.6 |
| 3,3′-ISDPA/DAIS | 253 | 255 | 388 | 106 | 3.2 | 6.3 |
| 3,3′-IMDPA/ODA | 259 | 261 | 411 | 74 | 2.2 | 3.8 |
| 3,3′-IMDPA/MDA | 252 | 254 | 406 | 85 | 2.2 | 4.3 |
| 3,3′-IMDPA/m-PDA | - d | 262 | 410 | - d | - d | - d |
| 3,3′-IMDPA/DAII | - d | 262 | 399 | - d | - d | - d |
| 3,3′-IMDPA/DAIS | - d | 259 | 393 | - d | - d | - d |
| 4,4′-ISDPA/ODA | 222 | 226 | 405 | 98 | 2.9 | 14.7 |
| 4,4′-ISDPA/MDA | 230 | 227 | 409 | 111 | 2.7 | 13.4 |
| 4,4′-ISDPA/m-PDA | 239 | 239 | 415 | 132 | 3.3 | 8.4 |
| 4,4′-ISDPA/DAII | 243 | 249 | 398 | 117 | 3.2 | 8.3 |
| 4,4′-ISDPA/DAIS | 239 | 239 | 384 | 129 | 3.5 | 12.6 |
| 4,4′-IMDPA/ODA | 229 | 225 | 413 | 116 | 2.7 | 15.9 |
| 4,4′-IMDPA/MDA | 246 | 251 | 402 | 115 | 3.0 | 24.5 |
| 4,4′-IMDPA/m-PDA | 246 | 246 | 408 | 134 | 3.6 | 10.4 |
| 4,4′-IMDPA/DAII | 245 | 244 | 393 | 116 | 3.4 | 11.1 |
| 4,4′-IMDPA/DAIS | 242 | 239 | 386 | 119 | 3.5 | 9.3 |
| Polyimides a | Specific rotation (°) b | λ0 (nm) c | T450 (%) d |
|---|---|---|---|
| 3,3′-ISDPA/ODA | (−) 100 | 373 | 78 |
| 3,3′-ISDPA/MDA | (−) 95 | 378 | 64 |
| 3,3′-ISDPA/m-PDA | (−) 91 | 377 | 61 |
| 3,3′-ISDPA/DAII | (+) 86 | 370 | 76 |
| 3,3′-ISDPA/DAIS | (+) 124 | 368 | 65 |
| 3,3′-IMDPA/ODA | (+) 230 | 373 | 77 |
| 3,3′-IMDPA/MDA | (+) 212 | 373 | 83 |
| 3,3′-IMDPA/m-PDA | (+) 258 | 365 | - |
| 4,4′-ISDPA/ODA | (+) 123 | 364 | 80 |
| 4,4′-ISDPA/MDA | (+) 123 | 373 | 63 |
| 4,4′-ISDPA/m-PDA | (+) 140 | 377 | 67 |
| 4,4′-ISDPA/DAII | (+) 203 | 369 | 78 |
| 4,4′-ISDPA/DAIS | (+) 248 | 365 | 72 |
| 4,4′-IMDPA/ODA | (+) 235 | 370 | 71 |
| 4,4′-IMDPA/MDA | (+) 233 | 363 | 80 |
| 4,4′-IMDPA/m-PDA | (+) 285 | 372 | 71 |
| 4,4′-IMDPA/DAII | (+) 344 | 369 | 66 |
| 4,4′-IMDPA/DAII | (+) 370 | 365 | 70 |
| 3,3′-ISDPA/Ph | (−) 26 | - | - |
| 3,3′-IMDPA/Ph | (+) 189 | - | - |
| 4,4′-ISDPA/Ph | (+) 121 | - | - |
| 4,4′-IMDPA/Ph | (+) 293 | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, X.; Wang, Z.; Wang, Z.; Yan, J. Bio-Based Poly(Ether Imide)s from Isohexide-Derived Isomeric Dianhydrides. Polymers 2017, 9, 569. https://doi.org/10.3390/polym9110569
Ji X, Wang Z, Wang Z, Yan J. Bio-Based Poly(Ether Imide)s from Isohexide-Derived Isomeric Dianhydrides. Polymers. 2017; 9(11):569. https://doi.org/10.3390/polym9110569
Chicago/Turabian StyleJi, Xiaodong, Zikun Wang, Zhen Wang, and Jingling Yan. 2017. "Bio-Based Poly(Ether Imide)s from Isohexide-Derived Isomeric Dianhydrides" Polymers 9, no. 11: 569. https://doi.org/10.3390/polym9110569
APA StyleJi, X., Wang, Z., Wang, Z., & Yan, J. (2017). Bio-Based Poly(Ether Imide)s from Isohexide-Derived Isomeric Dianhydrides. Polymers, 9(11), 569. https://doi.org/10.3390/polym9110569




