Microscopic Dynamics and Topology of Polymer Rings Immersed in a Host Matrix of Longer Linear Polymers: Results from a Detailed Molecular Dynamics Simulation Study and Comparison with Experimental Data
Abstract
:1. Introduction
2. Molecular Model and Simulated Systems
3. Results and Discussion
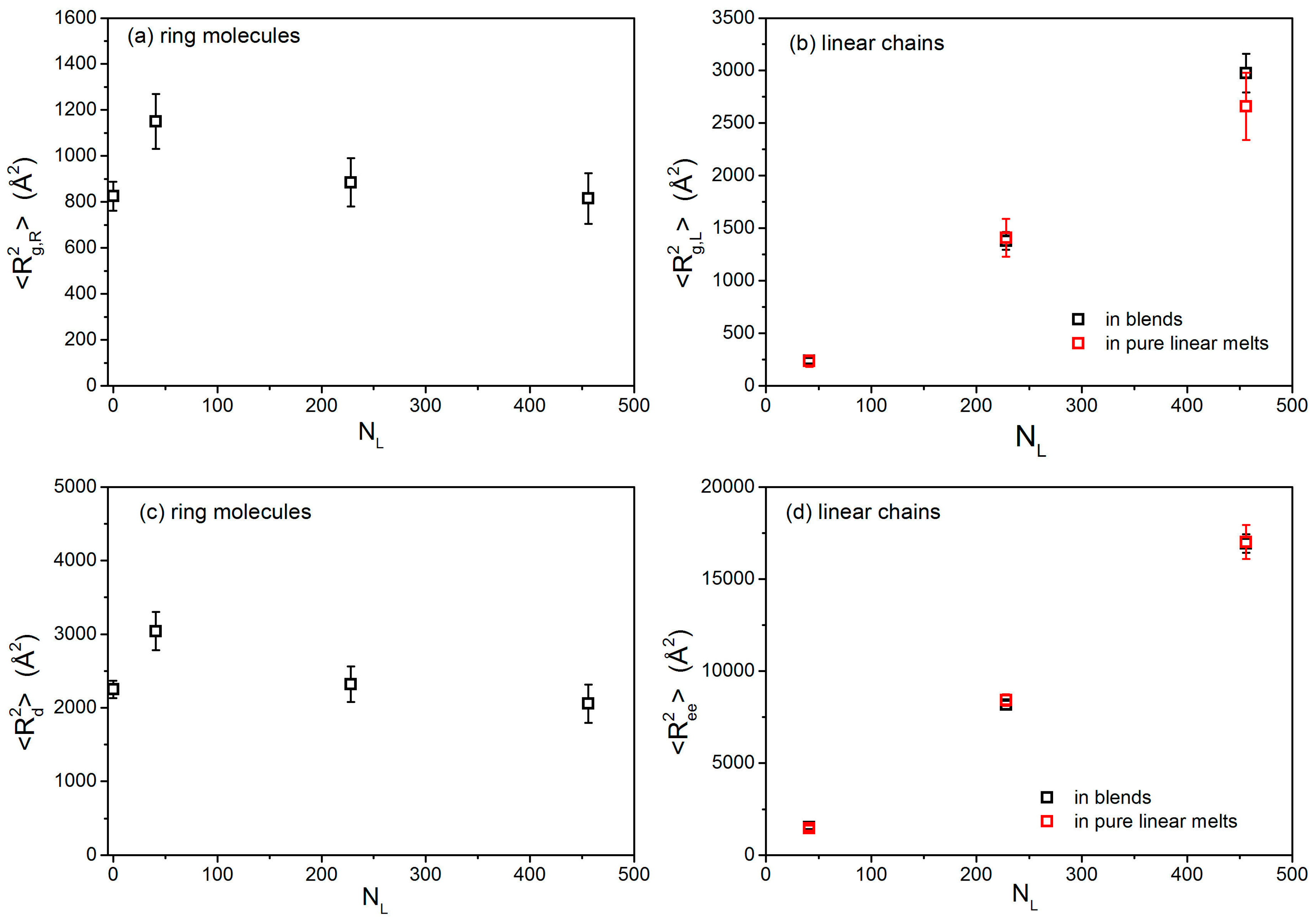
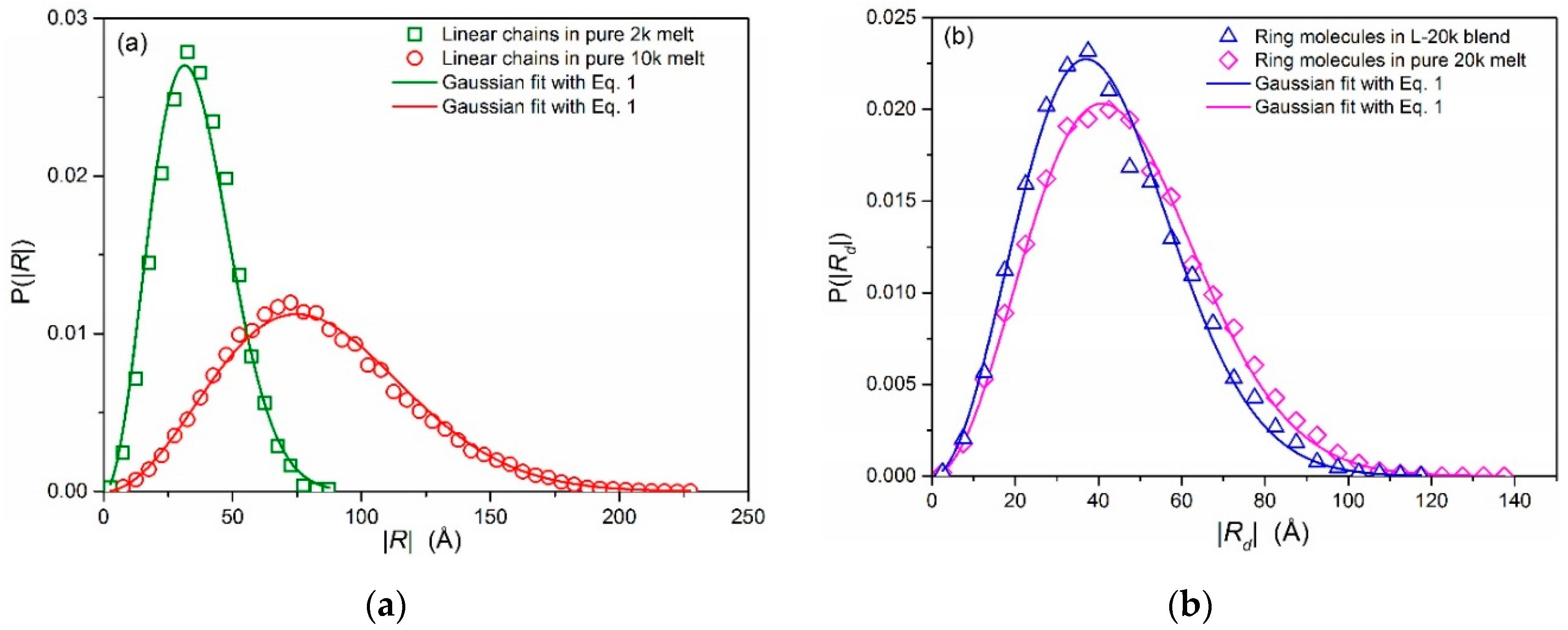
3.1. Conformational Properties
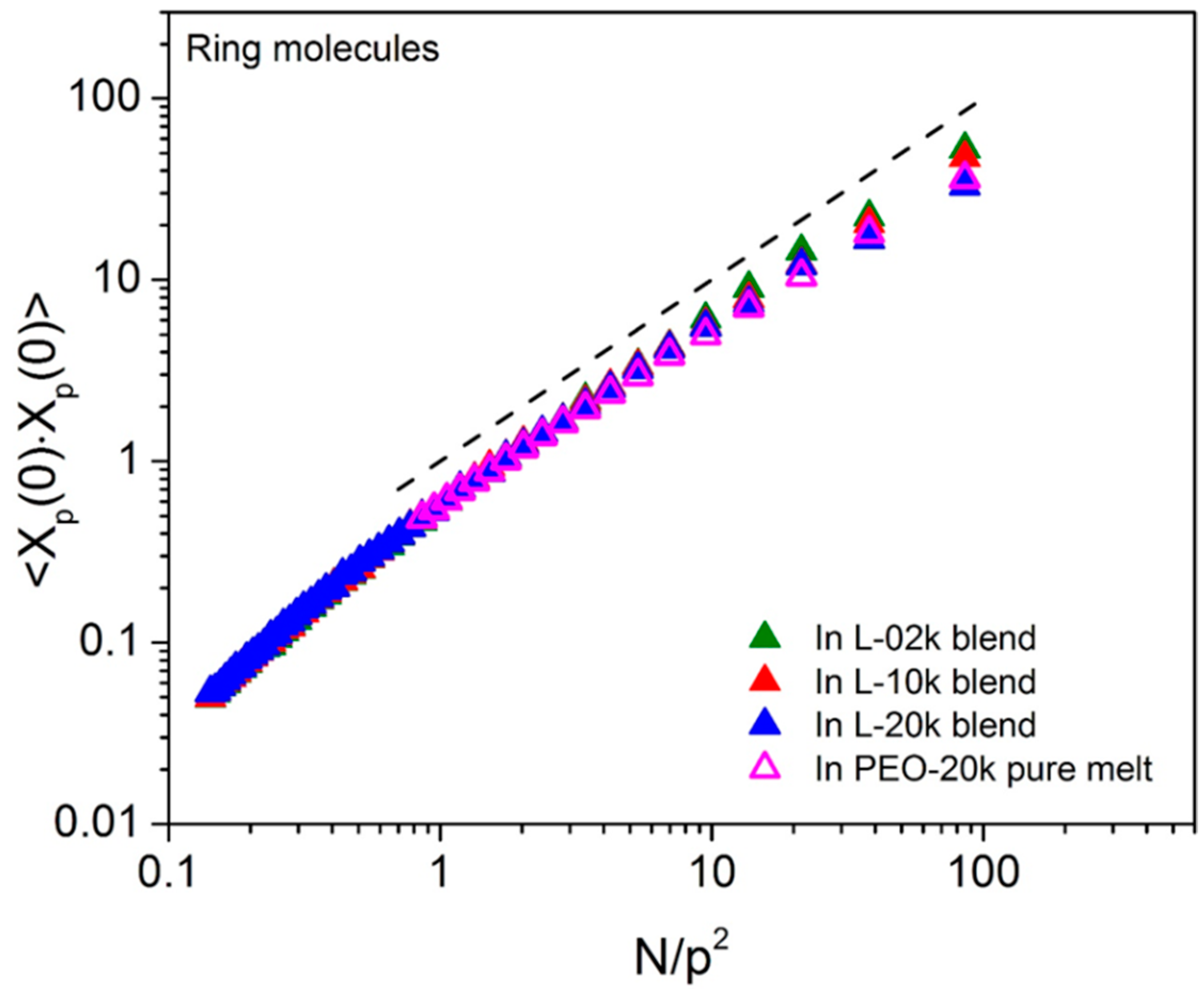
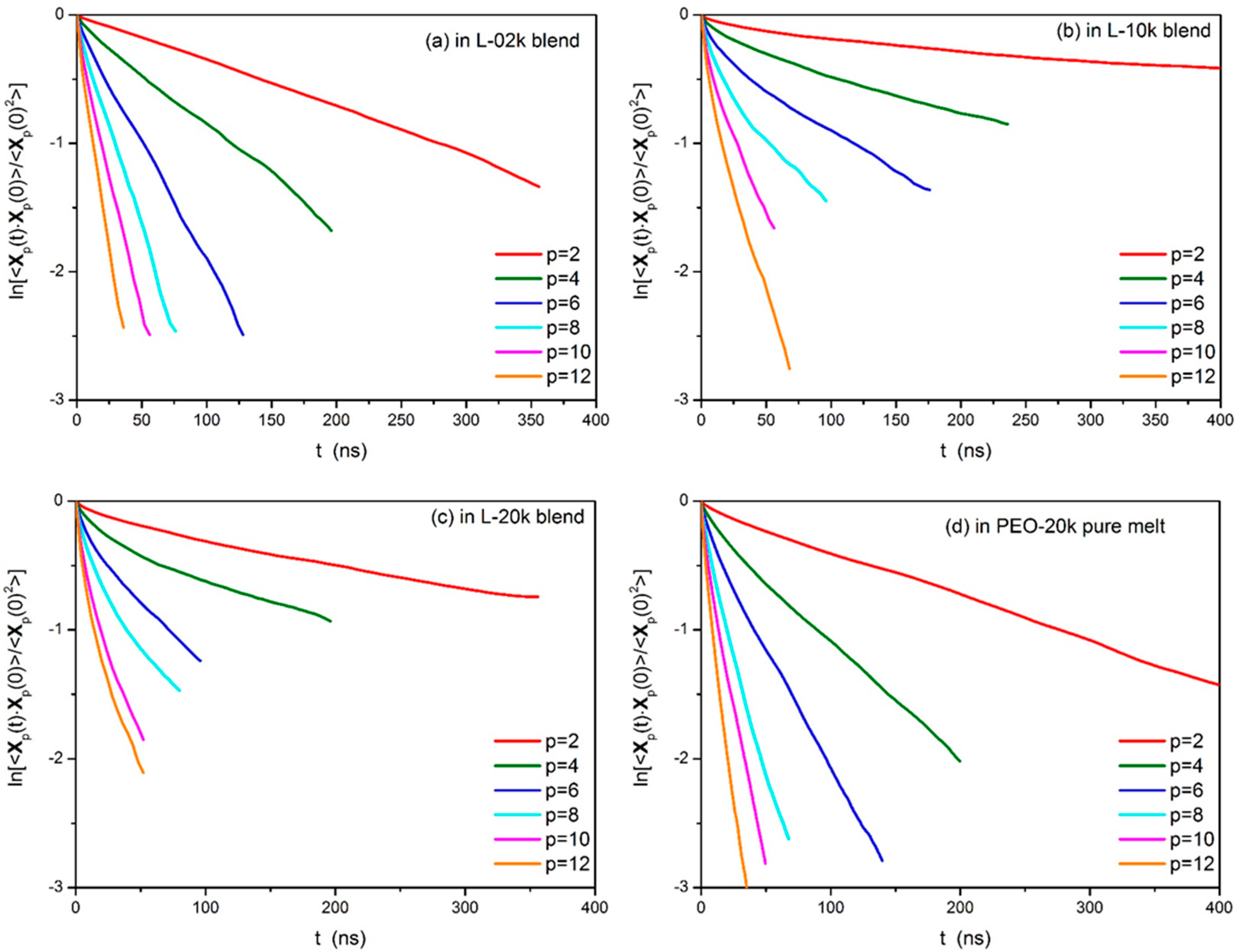
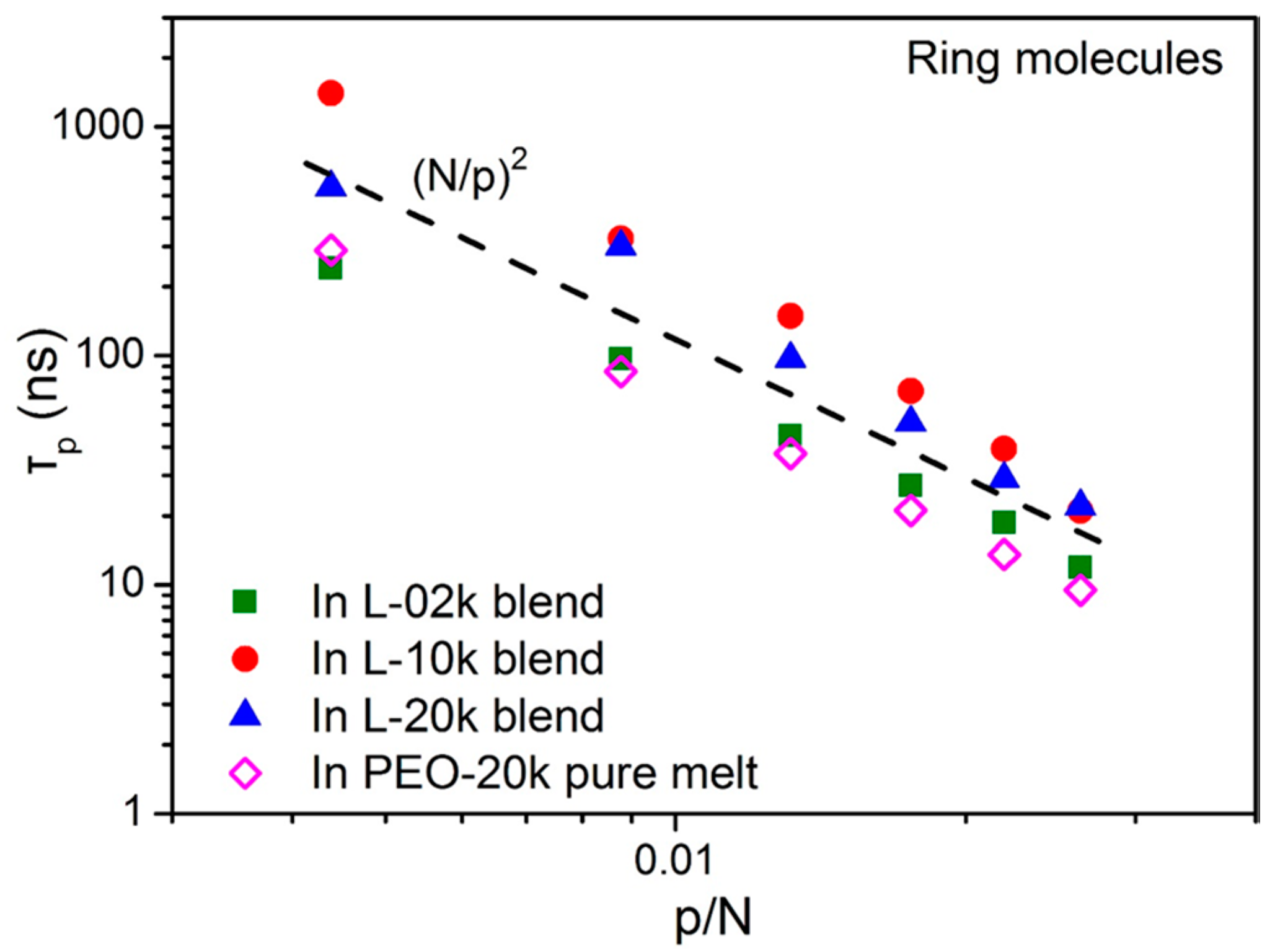
3.2. Normal Mode Analysis
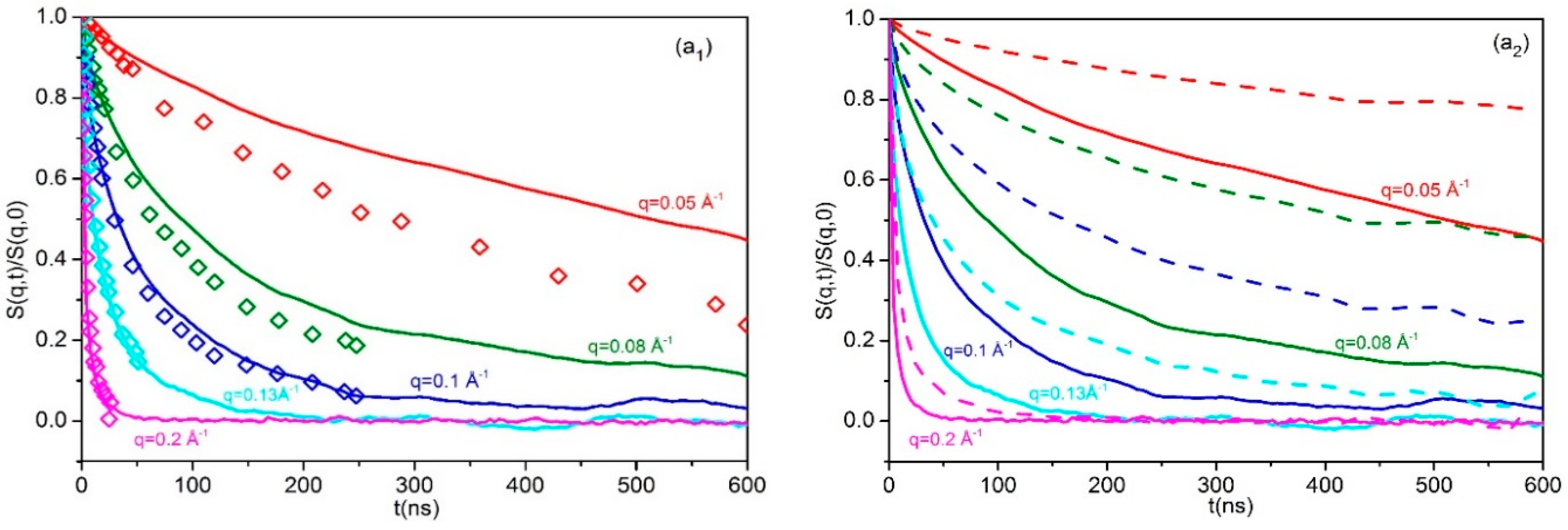
3.3. Dynamic Structure Factor
- (a)
- In the short L-02k blend (Figure 6a1), the computed curves for all q’s decay monotonically and smoothly over short and long time scales (that extend up to 600ns). The rate of decay is steeper at short times and deceases as the time increases, but in a very smooth way. As a result, only quantitative differences are observed from the corresponding spectra computed for the neat ring 20k PEO melt (Figure 6a2).
- (b)
- In the L-10k blend, a totally different picture emerges. Compared to the pure 20k ring or the behavior of rings in the unentangled L-02k blend, the simulation results here indicate an initial rapid decay at short times but then a rather asymptotic and time-independent behavior which leads to plateau values for that depend on the momentum transfer q. The time scale of the fast initial decay depends on the wavenumber q but overall is seen to be between 30 and 100 ns, i.e., on the order of the entanglement time τe for entangled linear PEO melts at 413K, see Section 3.4 and Section 3.6 below. A closer inspection reveals that the initial fast decay is even steeper than the one recorded in the corresponding pure ring PEO-20k melt, implying more freedom for motion. According to Goossen et al. [21], at these short times, rings in the L-10k blend enjoy free 3-d Rouse motion in the tubes formed by the surrounding (moderately entangled, number of entanglements Z ≅ 5) L-10k linear chains. At later times, strong topological interactions (hindrance effects [21]) set in, which cause to cross over to time-independent plateau values exhibiting no sign of any further decay.
- (c)
- In the longer L-20k blend, the dynamic structure factor exhibits the same qualitative behavior as in the L-10k blend. Again, we observe the rapid initial decay at very short times (on the order of 30 to 100 ns depending on the wavenumber q), followed by the time-independent behavior towards plateau values that are practically the same with those observed in the L-10k blend.
- (d)
- The qualitative agreement between predicted and experimentally measured spectra in the L-02k and L-20k blends is excellent. The quantitative agreement, on the other hand, is fairly good but improves for larger q values (larger than approximately 0.10 Å−1). For lower q values the simulation results systematically overpredict the measured NSE data. We believe that any quantitative differences between simulated and experimentally measured spectra should be attributed to the united-atom nature of the force-field employed in the MD simulations.
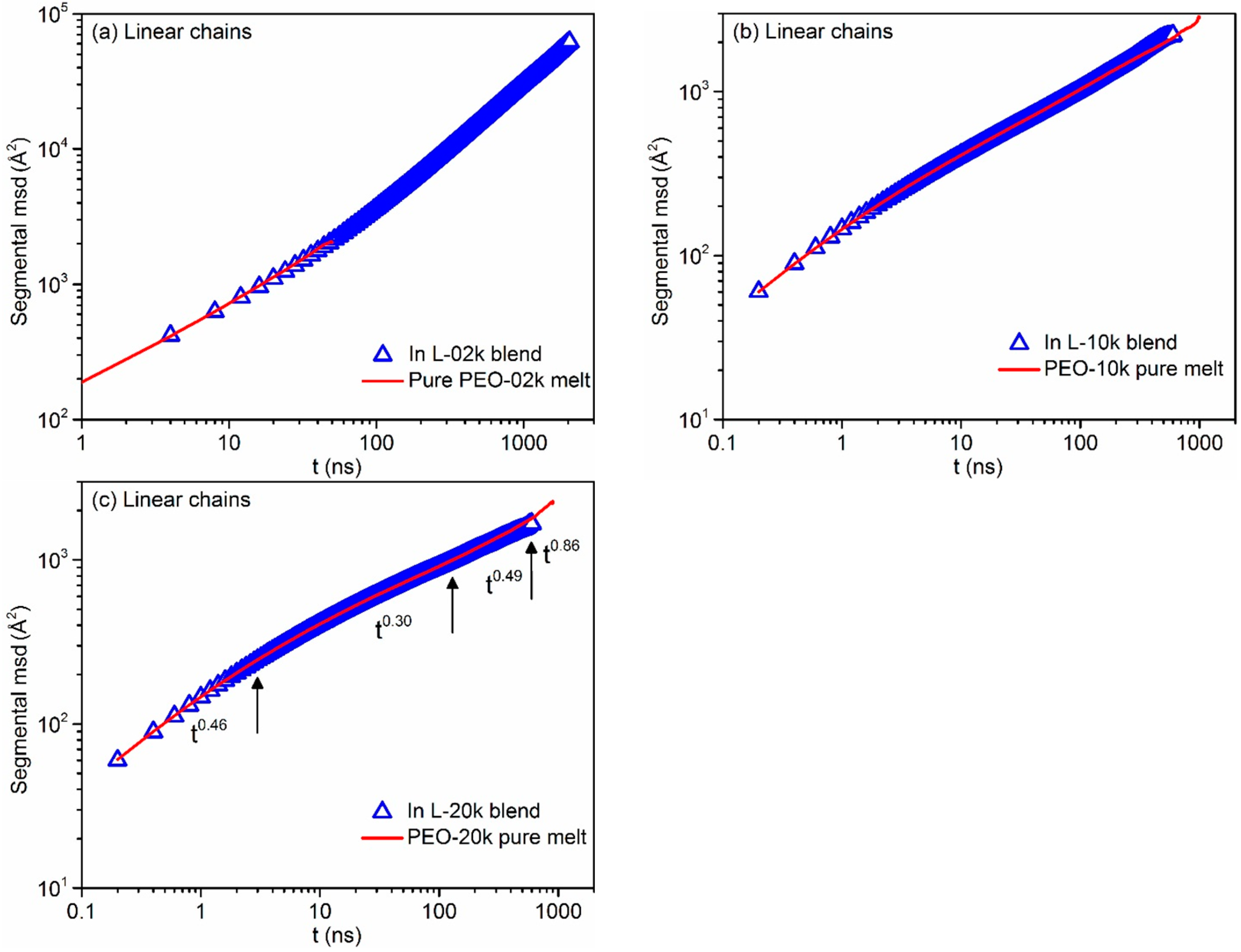
3.4. Mean Square Displacement of Atomistic Segments
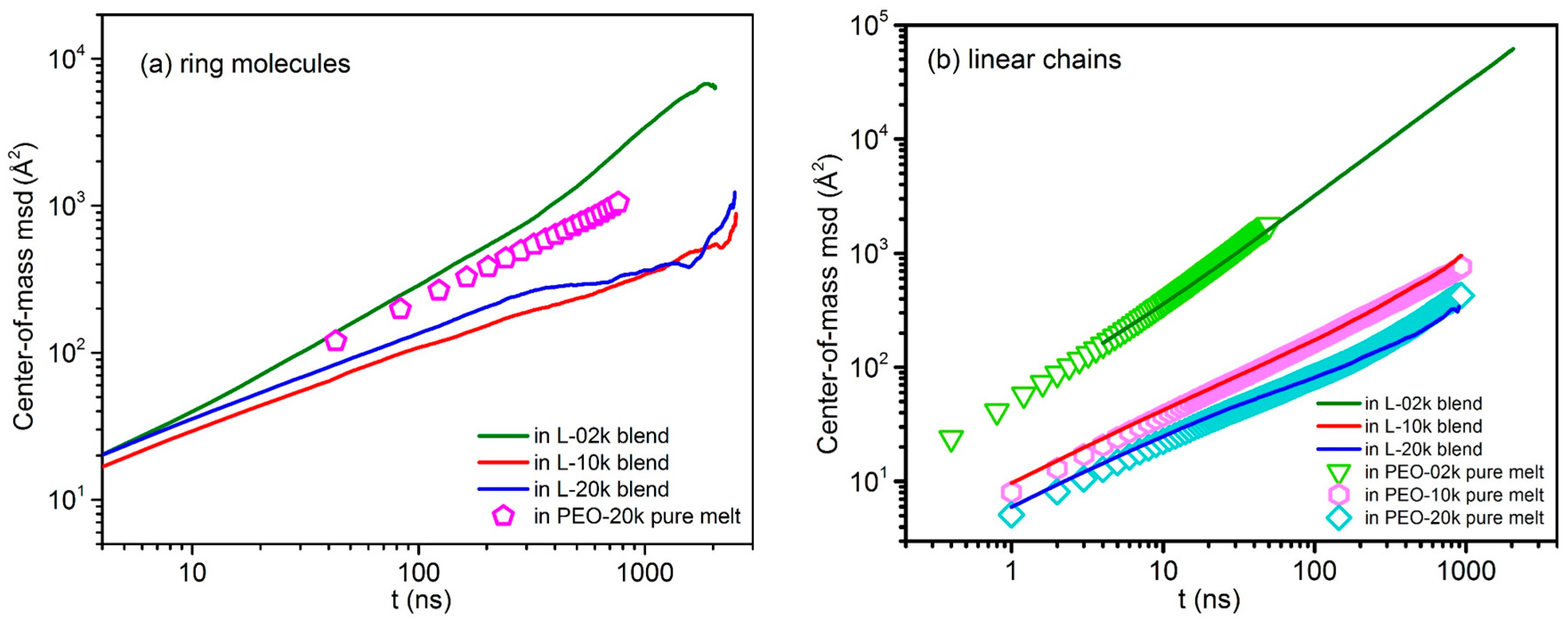
3.5. Mean-Square-Displacement of Chains Centers-of-Mass
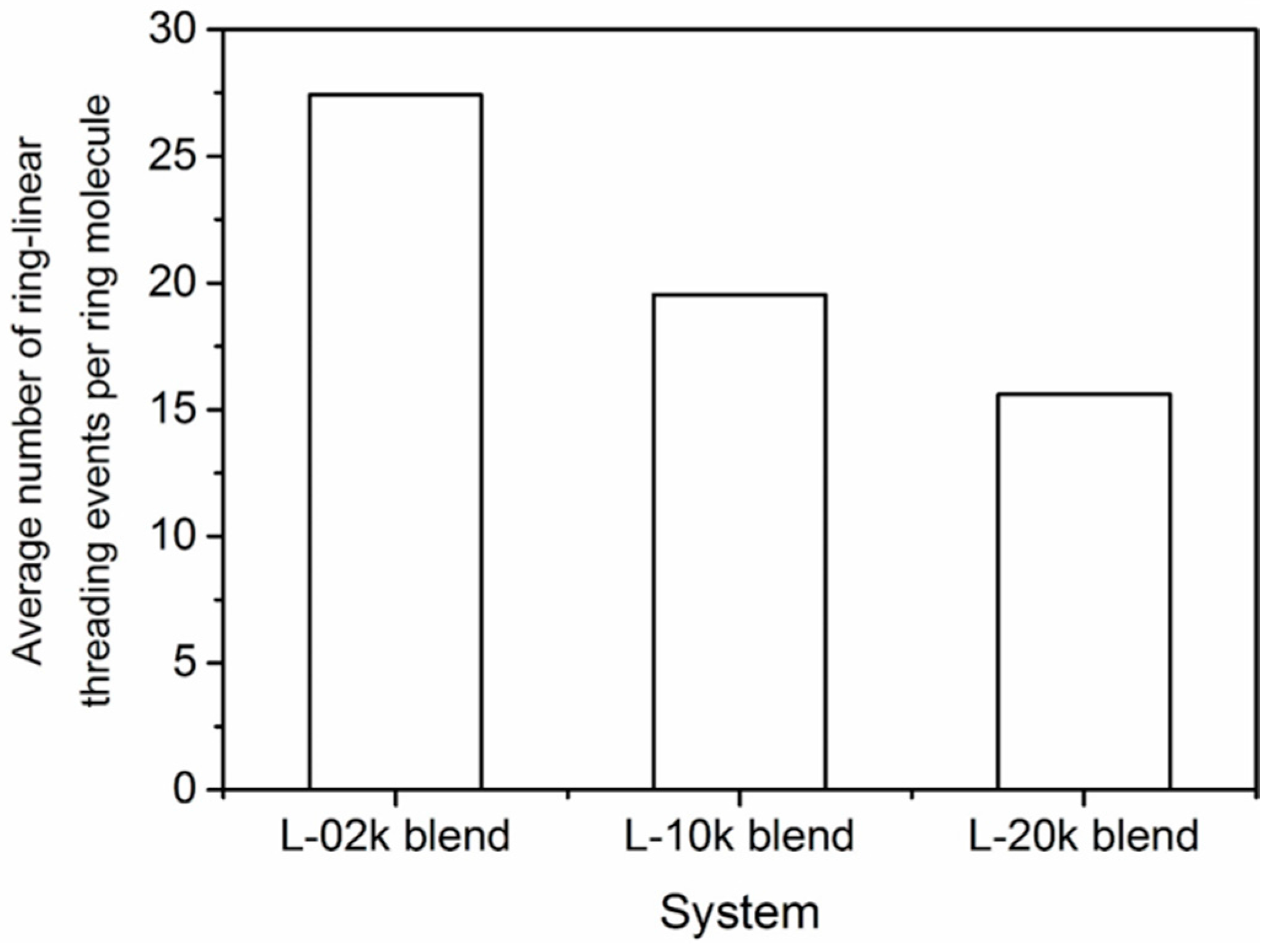
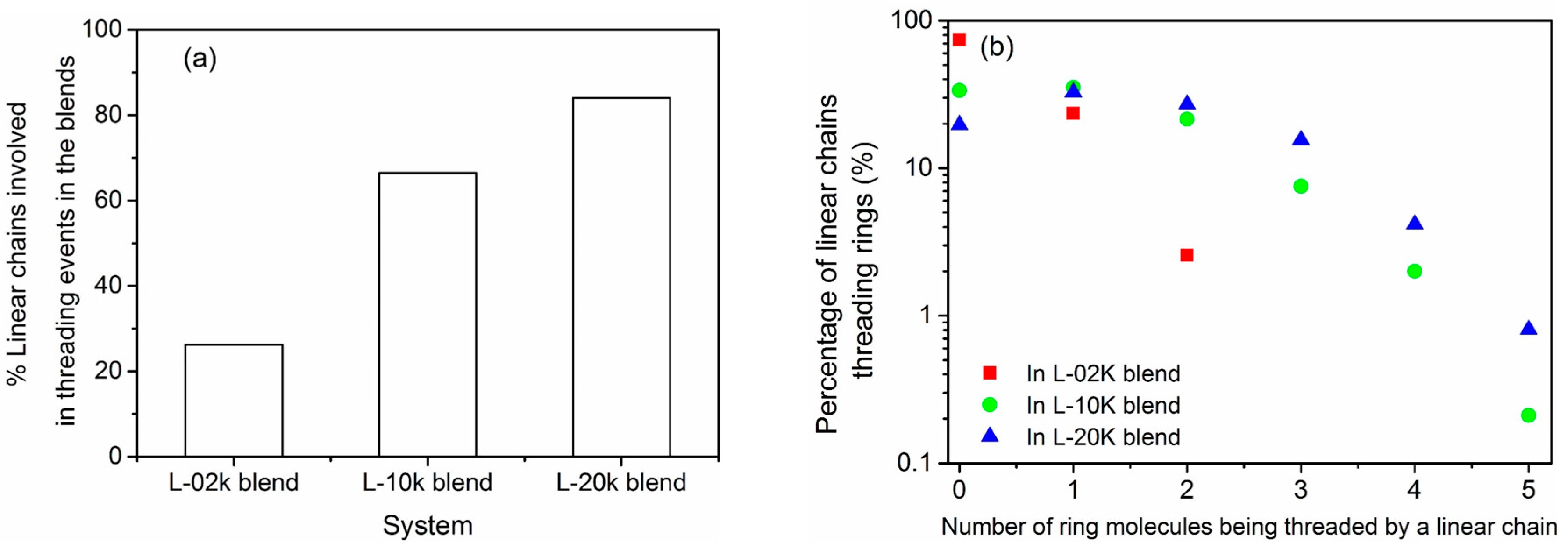
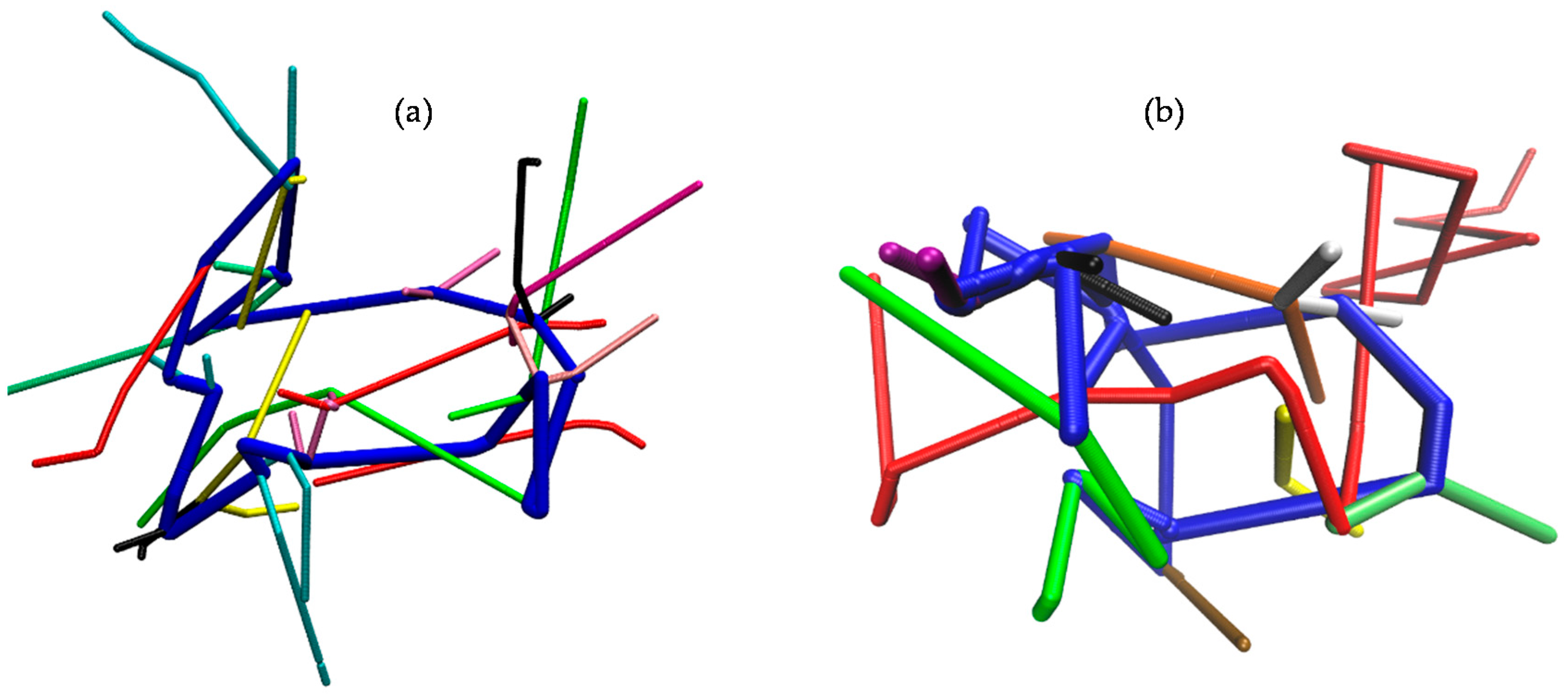
3.6. Topological Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Roovers, J. The melt properties of ring polystyrenes. Macromolecules 1985, 18, 1359–1361. [Google Scholar] [CrossRef]
- Lutz, P.; McKenna, G.B.; Rempp, P.; Strazielle, C. Solution properties of ring-shaped polystyrenes. Macromol. Chem. Rapid Commun. 1986, 7, 599–605. [Google Scholar] [CrossRef]
- Grosberg, A.Y.; Nechaev, S.K.; Shakhnovich, E.I. The role of topological constraints in the kinetics of collapse of macromolecules. J. Phys. 1988, 49, 2095–2100. [Google Scholar] [CrossRef]
- Roovers, J.; Toporowski, P.M. Synthesis and characterization of ring polybutadienes. J. Polym. Sci. B 1988, 26, 1251–1259. [Google Scholar] [CrossRef]
- McKenna, G.B.; Hostetler, B.J.; Hadjichristidis, N.; Fetters, L.J.; Plazek, D.J. A study of the linear viscoelastic properties of cyclic polystyrenes using creep and recovery measurements. Macromolecules 1989, 22, 1834–1852. [Google Scholar] [CrossRef]
- Roovers, J. Synthesis and properties of ring polybutadienes. Rubber Chem. Technol. 1989, 62, 33–41. [Google Scholar] [CrossRef]
- Zhang, R.; Mattice, W.L. Molecular dynamics study of the conformational properties of cyclohexadecane. J. Chem. Phys. 1993, 98, 9888–9894. [Google Scholar] [CrossRef]
- Obukhov, S.P.; Rubinstein, M.; Duke, T. Dynamics of a ring polymer in a gel. Phys. Rev. Lett. 1994, 73, 1263–1266. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Wittmer, J.P.; Cates, M.E. Topological effects in ring polymers: A computer simulation study. Phys. Rev. E 1996, 53, 5063–5074. [Google Scholar] [CrossRef]
- Semlyen, J.A. Cyclic Polymers, 2nd ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2000. [Google Scholar]
- Lee, H.C.; Lee, H.; Lee, W.; Chang, T.; Roovers, J. Fractionation of cyclic polystyrene from linear precursor by HPLC at the chromatographic critical condition. Macromolecules 2000, 33, 8119–8121. [Google Scholar] [CrossRef]
- Von Meerwall, E.; Ozisik, R.; Mattice, W.L.; Pfister, P.M. Self-diffusion of linear and cyclic alkanes, measured with pulsed-gradient spin-echo nuclear magnetic resonance. J. Chem. Phys. 2003, 118, 3867–3873. [Google Scholar] [CrossRef]
- Arrighi, V.; Gagliardi, S.; Dagger, A.C.; Semlyen, J.A.; Higgins, J.S.; Shenton, M.J. Conformation of cyclics and linear chain polymers in bulk by SANS. Macromolecules 2004, 37, 8057–8065. [Google Scholar] [CrossRef]
- Watanabe, H.; Inoue, T.; Matsumiya, Y. Transient conformational change of bead-spring ring chain during creep process. Macromolecules 2006, 39, 5419–5426. [Google Scholar] [CrossRef]
- Rosa, A.; Everaers, R. Structure and Dynamics of Interphase Chromosomes. PLoS Comput. Biol. 2008, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kapnistos, M.; Lang, M.; Vlassopoulos, D.; Pyckhout-Hintzen, W.; Richter, D.; Cho, D.; Chang, T.; Rubinstein, M. Unexpected power-law stress relaxation of entangled ring polymers. Nat. Mater. 2008, 7, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Brás, A.R.; Pasquino, R.; Koukoulas, T.; Tsolou, G.; Holderer, O.; Radulescu, A.; Allgaier, J.; Mavrantzas, V.G.; Pyckhout-Hintzen, W.; Wischnewski, A.; Vlassopoulos, D.; Richter, D. Structure and dynamics of polymer rings by neutron scattering: Breakdown of the rouse model. Soft Matter 2011, 7, 11169–11176. [Google Scholar] [CrossRef]
- Pasquino, R.; Vasilakopoulos, T.C.; Jeong, Y.C.; Lee, H.; Rogers, S.; Sakellariou, G.; Allgaier, J.; Takano, A.; Brás, A.R.; Chang, T.; et al. Viscosity of ring polymer melts. ACS Macro Lett. 2013, 2, 874–878. [Google Scholar] [CrossRef] [PubMed]
- Brás, R.; Gooßen, S.; Krutyeva, M.; Radulescu, A.; Farago, B.; Allgaier, J.; Pyckhout-Hintzen, W.; Wischnewski, A.; Richter, D. Compact structure and non-Gaussian dynamics of ring polymer melts. Soft Matter 2014, 10, 3649–3655. [Google Scholar] [CrossRef] [PubMed]
- Gooßen, S.; Brás, A.R.; Krutyeva, M.; Sharp, M.; Falus, P.; Feoktystov, A.; Gasser, U.; Pyckhout-Hintzen, W.; Wischnewski, A.; Richter, D. Molecular scale dynamics of large ring polymers. Phys. Rev. Lett. 2014, 113, 168302. [Google Scholar] [CrossRef] [PubMed]
- Gooßen, S.; Krutyeva, M.; Sharp, M.; Feoktystov, A.; Allgaier, J.; Pyckhout-Hintzen, W.; Wischnewski, A.; Richter, D. Sensing polymer chain dynamics through ring topology: A neutron spin echo study. Phys. Rev. Lett. 2015, 115, 148302. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Matsubara, K.; Ohta, Y.; Nakano, T.; Kawaguchi, D.; Takahashi, Y.; Takano, A.; Matsushita, Y. Melt rheology of ring polystyrenes with ultrahigh purity. Macromolecules 2015, 48, 3140–3147. [Google Scholar] [CrossRef]
- Yan, Z.-C.; Costanzo, S.; Jeong, Y.; Chang, T.; Vlassopoulos, D. Linear and nonlinear shear rheology of a marginally entangled ring polymer. Macromolecules 2016, 49, 1444–1453. [Google Scholar] [CrossRef]
- Ge, T.; Panyukov, S.; Rubinstein, M. Self-similar conformations and dynamics in entangled melts and solutions of nonconcatenated ring polymers. Macromolecules 2016, 49, 708–722. [Google Scholar] [CrossRef] [PubMed]
- Hur, K.; Winkler, R.G.; Yoon, D.Y. Comparison of ring and linear Polyethylene from molecular dynamics simulations. Macromolecules 2006, 39, 3975–3977. [Google Scholar] [CrossRef]
- Iyer, B.V.S.; Lele, A.K.; Shanbhag, S. What is the size of a ring polymer in a ring–linear blend? Macromolecules 2007, 40, 5995–6000. [Google Scholar] [CrossRef]
- Tsolou, G.; Stratikis, N.; Baig, C.; Stephanou, P.S.; Mavrantzas, V.G. Melt structure and dynamics of unentangled polyethylene rings: Rouse theory, atomistic molecular dynamics simulation, and comparison with the linear analogues. Macromolecules 2010, 43, 10692–10713. [Google Scholar] [CrossRef]
- Milner, S.T.; Newhall, J.D. Stress relaxation in entangled melts of unlinked ring polymers. Phys. Rev. Lett. 2010, 105, 208302. [Google Scholar] [CrossRef] [PubMed]
- Hur, K.; Jeong, C.; Winkler, R.G.; Lacevic, N.; Gee, R.H.; Yoon, D.Y. Chain dynamics of ring and linear polyethylene melts from molecular dynamics simulations. Macromolecules 2011, 44, 2311–2315. [Google Scholar] [CrossRef]
- Halverson, J.D.; Lee, W.B.; Grest, G.S.; Grosberg, A.Y.; Kremer, K. Molecular dynamics simulation study of nonconcatenated ring polymers in a melt. I. Statics. J. Chem. Phys. 2011, 134, 204904. [Google Scholar] [CrossRef] [PubMed]
- Halverson, J.D.; Lee, W.B.; Grest, G.S.; Grosberg, A.Y.; Kremer, K. Molecular dynamics simulation study of nonconcatenated ring polymers in a melt. II. Dynamics. J. Chem. Phys. 2011, 134, 204905. [Google Scholar] [CrossRef] [PubMed]
- Halverson, J.D.; Grest, G.S.; Grosberg, A.Y.; Kremer, K. Rheology of ring polymer melts: From linear contaminants to ring/linear blends. Phys. Rev. Lett. 2012, 108, 038301. [Google Scholar] [CrossRef] [PubMed]
- Tsalikis, D.G.; Koukoulas, T.; Mavrantzas, V.G. Dynamic, conformational and topological properties of ring–linear poly(ethylene oxide) blends from molecular dynamics simulations. React. Funct. Polym. 2014, 80, 61–70. [Google Scholar] [CrossRef]
- Michieletto, D.; Marenduzzo, D.; Orlandini, E.; Alexander, G.P.; Turner, M.S. Threading dynamics of ring polymers in a gel. ACS Macro Lett. 2014, 3, 255–259. [Google Scholar] [CrossRef]
- Tsalikis, G.D.; Mavrantzas, V.G. Threading of ring poly(ethylene oxide) molecules by linear chains in the melt. ACS Macro Lett. 2014, 3, 763–766. [Google Scholar] [CrossRef]
- Lee, E.; Kim, S.; Jung, Y. Slowing down of ring polymer diffusion caused by inter-ring threading. Macromol. Rapid Commun. 2015, 36, 1115–1121. [Google Scholar] [CrossRef] [PubMed]
- Tsalikis, D.G.; Mavrantzas, V.G.; Vlassopoulos, D. Analysis of slow modes in ring polymers: Threading of rings controls slow relaxation. ACS Macro Lett. 2016, 5, 755–760. [Google Scholar] [CrossRef]
- Edwards, S.F. Theory of rubber elasticity. Br. Polym. J. 1977, 9, 140–143. [Google Scholar] [CrossRef]
- Doi, M.; Edwards, S.F. The Theory of Polymer Dynamics; Oxford University Press: Oxford, UK, 1986. [Google Scholar]
- DeGennes, P.G. Scaling Concepts in Polymer Physics; Cornell University Press: Ithaca, NY, USA, 1979. [Google Scholar]
- Rubinstein, M.; Colby, R.H. Polymer Physics; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Fischer, J.; Paschek, D.; Geiger, A.; Sadowski, G. Modeling of aqueous poly(oxyethylene) solutions: 1. atomistic simulations. J. Phys. Chem. B 2008, 112, 2388–2398. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.; Paschek, D.; Geiger, A.; Sadowski, G. Modeling of aqueous poly(oxyethylene) solutions: 1. atomistic simulations. J. Phys. Chem. B 2008, 112, 8849–8850. [Google Scholar] [CrossRef]
- Hong, B.; Chremos, A.; Panagiotopoulos, A.Z. Dynamics in coarse-grained models for oligomer-grafted silica nanoparticles. J. Chem. Phys. 2012, 136, 204904. [Google Scholar] [CrossRef] [PubMed]
- Borodin, O.; Douglas, R.; Smith, G.D.; Trouw, F.; Petrucci, S. MD simulations and experimental study of structure, dynamics, and thermodynamics of poly(ethylene oxide) and its oligomers. J. Phys. Chem. B 2003, 107, 6813–6823. [Google Scholar] [CrossRef]
- Zheng, P.; Meng, X.; Wu, J.; Liu, L. Density and viscosity measurements of dimethoxymethane and 1,2-dimethoxyethane from 243 K to 373 K up to 20 MPa. Int. J. Thermophys. 2008, 29, 1244–1256. [Google Scholar] [CrossRef]
- Das, B.; Roy, M.N.; Hazra, D.K. Densities and viscosities of the binary aqueous mixture of tetrahydrofuran and 1, 2 dimethoxyethane at 298, 308 and 318 K. Indian J. Chem. Technol. 1994, 1, 93–97. [Google Scholar]
- Kusano, K. Densities, refractive indexes, and normal boiling points of 1,2-disubstituted ethylene glycol derivatives. J. Chem. Eng. Data 1978, 23, 141–143. [Google Scholar] [CrossRef]
- Pal, A.; Sharma, S. Excess Molar Volumes and Viscosities of 1-Propanol + Ethylene Glycol, + Ethylene Glycol Monomethyl, + Ethylene Glycol Dimethyl, + Diethylene Glycol Dimethyl, + Triethylene Glycol Dimethyl, + Diethylene Glycol Diethyl, and + Diethylene Glycol Dibutyl Ethers at 298.15 K. J. Chem. Eng. Data 1998, 43, 532–536. [Google Scholar]
- Conesa, A.; Shen, S.; Coronas, A. Liquid densities, kinematic viscosities, and heat capacities of some ethylene glycol dimethyl ethers at temperatures from 283.15 to 423.15 K. Int. J. Thermophys. 1998, 19, 1343–1358. [Google Scholar] [CrossRef]
- Wick, C.D.; Theodorou, D.N. Connectivity-altering Monte Carlo simulations of the end group effects on volumetric properties for poly(ethylene oxide). Macromolecules 2004, 37, 7026–7033. [Google Scholar] [CrossRef]
- Dee, G.T.; Ougizawa, T.; Walsh, D.J. The pressure-volume-temperature properties of polyethylene, poly(dimethyl siloxane), poly(ethylene glycol) and poly(propylene glycol) as a function of molecular weight. Polymer 1992, 33, 3462–3469. [Google Scholar] [CrossRef]
- Annis, B.K.; Borodin, O.; Smith, G.D.; Benmore, C.J.; Soper, A.K.; Londono, J.D. The structure of a poly(ethylene oxide) melt from neutron scattering and molecular dynamics simulations. J. Chem. Phys. 2001, 115, 10998–11003. [Google Scholar] [CrossRef]
- Van Der Spoel, E.L.D.; Hess, B.; Groenhof, G.; Mark, A.E.; Berendsen, H.J.C. GROMACS: Fast, flexible, and free. J. Comp. Chem. 2005, 26, 1701–1718. [Google Scholar] [CrossRef] [PubMed]
- Nosé, S. Constant temperature molecular dynamics methods. Prog. Theor. Phys. Suppl. 1991, 103, 1–46. [Google Scholar] [CrossRef]
- Hoover, G.W. Canonical dynamics: Equilibrium phase-space distributions. Phys. Rev. A 1985, 31, 1695–1697. [Google Scholar] [CrossRef]
- Parrinello, M.; Rahman, A. Polymorphic transitions in single crystals: A new molecular dynamics method. J. Appl. Phys. 1981, 52, 7182–7190. [Google Scholar] [CrossRef]
- Scienomics, MAPS Platform, Version 3.4.2, France. 2015. Available online: http://www.scienomics.com/ (accessed on 28 July 2016).
- Tzoumanekas, C.; Theodorou, D.N. Topological analysis of linear polymer melts: A statistical approach. Macromolecules 2006, 39, 4592–4604. [Google Scholar] [CrossRef]
- Harmandaris, V.A.; Mavrantzas, V.G.; Theodorou, D.N.; Kröger, M.; Ramírez, J.; Öttinger, H.C.; Vlassopoulos, D. Crossover from Rouse to entangled polymer melt regime: Signals from long, detailed atomistic molecular dynamics simulations, supported by rheological experiments. Macromolecules 2003, 36, 1376–1387. [Google Scholar] [CrossRef]
- Tsolou, G.; Mavrantzas, V.G.; Theodorou, D.N. Detailed atomistic molecular dynamics simulation of cis-1,4-poly(butadiene). Macromolecules 2005, 38, 1478–1492. [Google Scholar] [CrossRef]
- Karayiannis, N.C.; Mavrantzas, V.G. Hierarchical modelling of the dynamics of polymers with a non-linear molecular architecture: Calculation of branch point friction and chain reptation time of H-shaped polyethylene melts from long molecular dynamics simulations. Macromolecules 2005, 38, 8583–8596. [Google Scholar] [CrossRef]
- Stephanou, P.S.; Baig, C.; Tsolou, G.; Mavrantzas, V.G.; Kröger, M. Quantifying chain reptation in entangled polymer melts: Topological and dynamical mapping of atomistic simulation results onto the tube model. J. Chem. Phys. 2010, 132, 124904. [Google Scholar] [CrossRef] [PubMed]
- Foteinopoulou, K.; Karayiannis, N.C.; Mavrantzas, V.G.; Kröger, M. Primitive path identification and entanglement statistics in polymer melts: Results from direct topological analysis on atomistic polyethylene models. Macromolecules 2006, 39, 4207–4216. [Google Scholar] [CrossRef]
- Cremer, T.; Cremer, M.; Dietzel, S.; Müller, S.; Solovei, I.; Fakan, S. Chromosome territories-A functional nuclear landscape. Curr. Opin. Cell Biol. 2006, 18, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Jun, S.; Mulder, B. Entropy-driven spatial organization of highly confined polymers: Lessons for the bacterial chromosome. Proc. Nat. Acad. Sci. USA 2006, 103, 12388–12393. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, M.H.; Gallucci, J.C.; Yin, H. Cyclic esters and cyclodepsipeptides derived from lactide and 2,5-morpholinediones. Proc. Nat. Acad. Sci. USA 2006, 103, 15315–15320. [Google Scholar] [CrossRef] [PubMed]
- Meaburn, K.J.; Misteli, T. Cell biology: Chromosome territories. Nature 2007, 445, 379–381. [Google Scholar] [CrossRef] [PubMed]
- Marenduzzo, D.; Orlandini, E.; Stasiak, A.; Sumners, D.W.; Tubiana, L.; Micheletti, C. DNA–DNA interactions in bacteriophage capsids are responsible for the observed DNA knotting. Proc. Natl. Acad. Sci. USA 2009, 106, 22269–22274. [Google Scholar] [CrossRef] [PubMed]



| System | Host matrix | Number of ring PEO-20k molecules | Number of linear PEO chains | Volume fraction of ring molecules |
|---|---|---|---|---|
| 1 | L-02k | 8 | 720 | 0.1 |
| 2 | L-10k | 8 | 144 | 0.1 |
| 3 | L-20k | 8 | 72 | 0.1 |
| System | Ring molecules | Linear chains | |||
|---|---|---|---|---|---|
| (Å2) | (Å2) | (Å2) | (Å2) | ||
| 02k pure linear melt | - | - | 235 ± 55 | 1475 ± 140 | 6.3 ± 0.7 |
| 10k pure linear melt | - | - | 1410 ± 180 | 8420 ± 310 | 6.0 ± 0.8 |
| 20k pure linear melt | - | - | 2660 ± 320 | 17,015 ± 930 | 6.4 ± 1.0 |
| L-02k blend | 1150 ± 120 | 3040 ± 260 | 240 ± 25 | 1525 ± 105 | 6.3 ± 1.4 |
| L-10k blend | 885 ± 105 | 2320 ± 240 | 1380 ± 85 | 8160 ± 220 | 5.9 ± 08 |
| L-20k blend | 815 ± 110 | 2055 ± 260 | 2975 ± 185 | 16,925 ± 500 | 5.7 ± 0.8 |
| 20k pure ring melt | 825 ± 63 | 2250 ± 120 | - | - | - |
| System | of ring molecules (Å) | of ring molecules (Å) | of linear chains (Å) | of linear chains (Å) |
|---|---|---|---|---|
| 02k pure linear melt | 12 ± 3 | 6.7 ± 0.4 | ||
| 10k pure linear melt | 12 ± 3 | 6.7 ± 0.4 | ||
| 20k pure linear melt | 11 ± 3 | 6.7 ± 0.4 | ||
| 20k pure ring melt | 35 ± 3 | 6.1 ± 0.4 | ||
| L-02k blend | 28 ± 3 | 6.1 ± 0.4 | 13 ± 3 | 6.7 ± 0.4 |
| L-10k blend | 31 ± 3 | 6.1 ± 0.4 | 12 ± 3 | 6.7 ± 0.4 |
| L-20k blend | 38 ± 4 | 6.1 ± 0.4 | 12 ± 3 | 6.7 ± 0.4 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papadopoulos, G.D.; Tsalikis, D.G.; Mavrantzas, V.G. Microscopic Dynamics and Topology of Polymer Rings Immersed in a Host Matrix of Longer Linear Polymers: Results from a Detailed Molecular Dynamics Simulation Study and Comparison with Experimental Data. Polymers 2016, 8, 283. https://doi.org/10.3390/polym8080283
Papadopoulos GD, Tsalikis DG, Mavrantzas VG. Microscopic Dynamics and Topology of Polymer Rings Immersed in a Host Matrix of Longer Linear Polymers: Results from a Detailed Molecular Dynamics Simulation Study and Comparison with Experimental Data. Polymers. 2016; 8(8):283. https://doi.org/10.3390/polym8080283
Chicago/Turabian StylePapadopoulos, George D., Dimitrios G. Tsalikis, and Vlasis G. Mavrantzas. 2016. "Microscopic Dynamics and Topology of Polymer Rings Immersed in a Host Matrix of Longer Linear Polymers: Results from a Detailed Molecular Dynamics Simulation Study and Comparison with Experimental Data" Polymers 8, no. 8: 283. https://doi.org/10.3390/polym8080283
APA StylePapadopoulos, G. D., Tsalikis, D. G., & Mavrantzas, V. G. (2016). Microscopic Dynamics and Topology of Polymer Rings Immersed in a Host Matrix of Longer Linear Polymers: Results from a Detailed Molecular Dynamics Simulation Study and Comparison with Experimental Data. Polymers, 8(8), 283. https://doi.org/10.3390/polym8080283