SANS from Salt-Free Aqueous Solutions of Hydrophilic and Highly Charged Star-Branched Polyelectrolytes
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. SANS Measurements
3. Results and Discussion
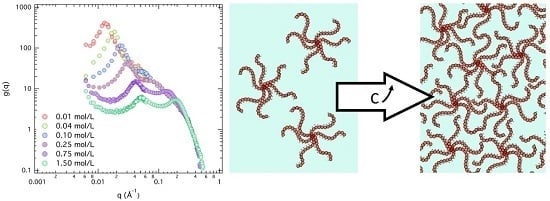
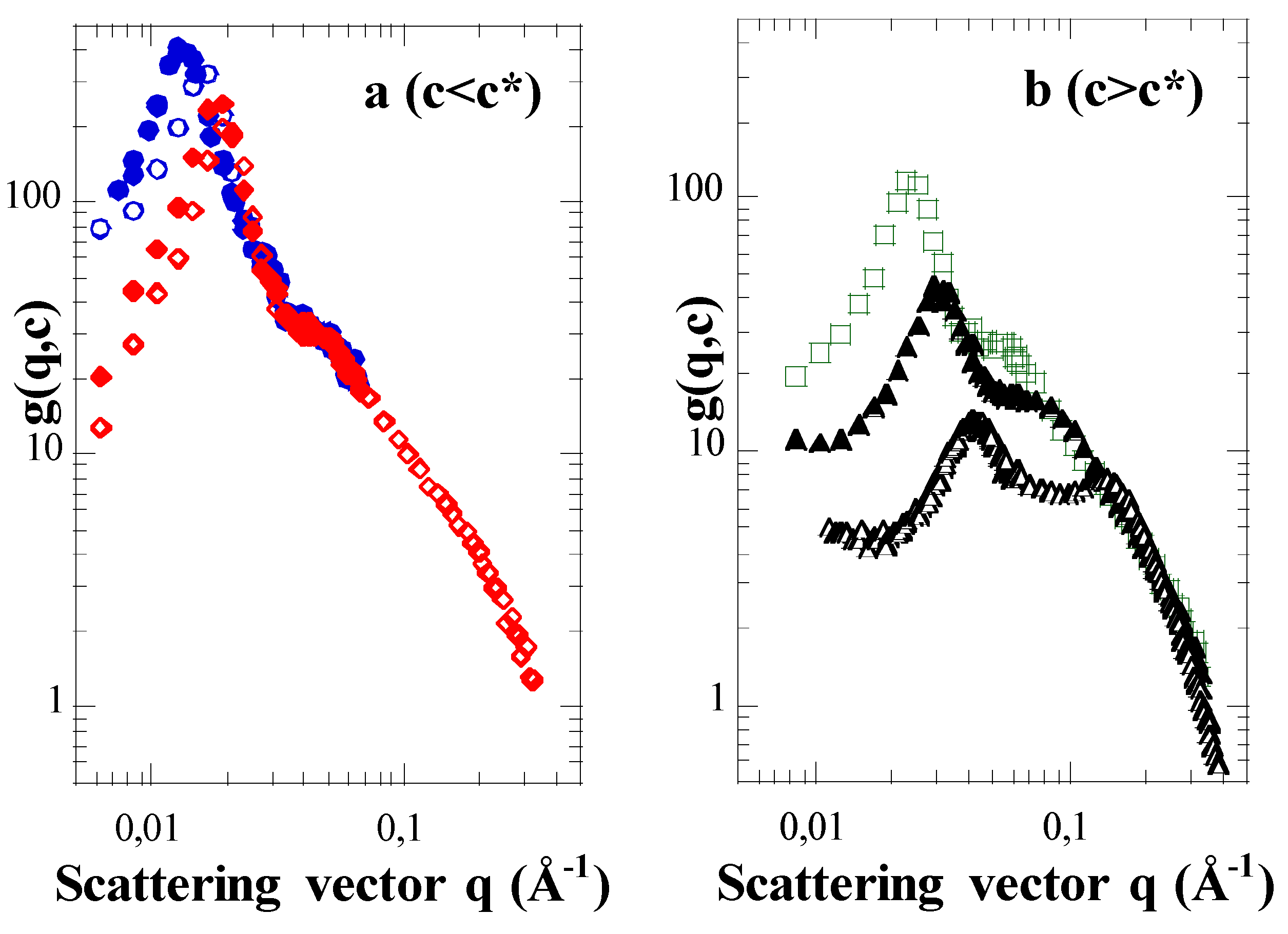
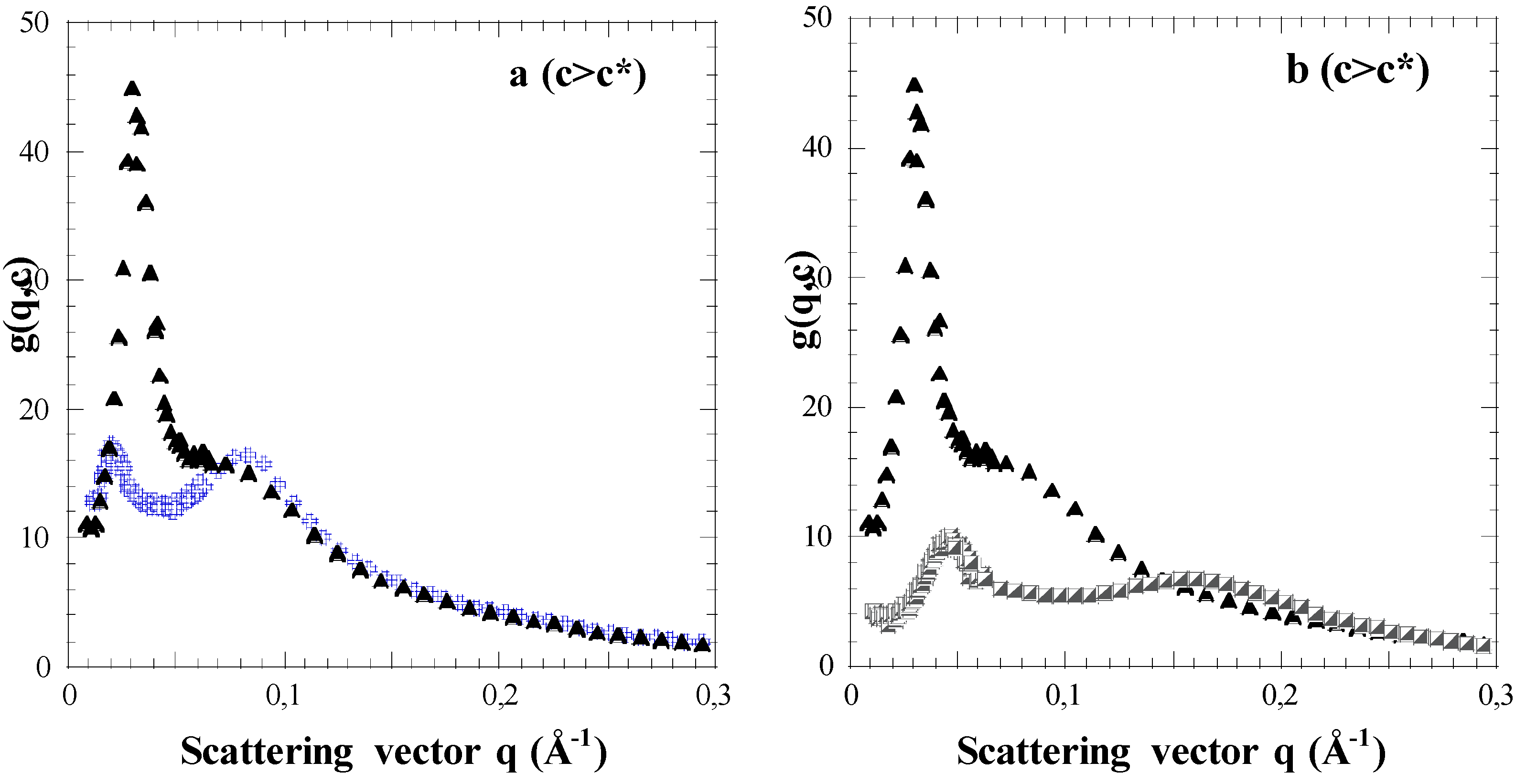
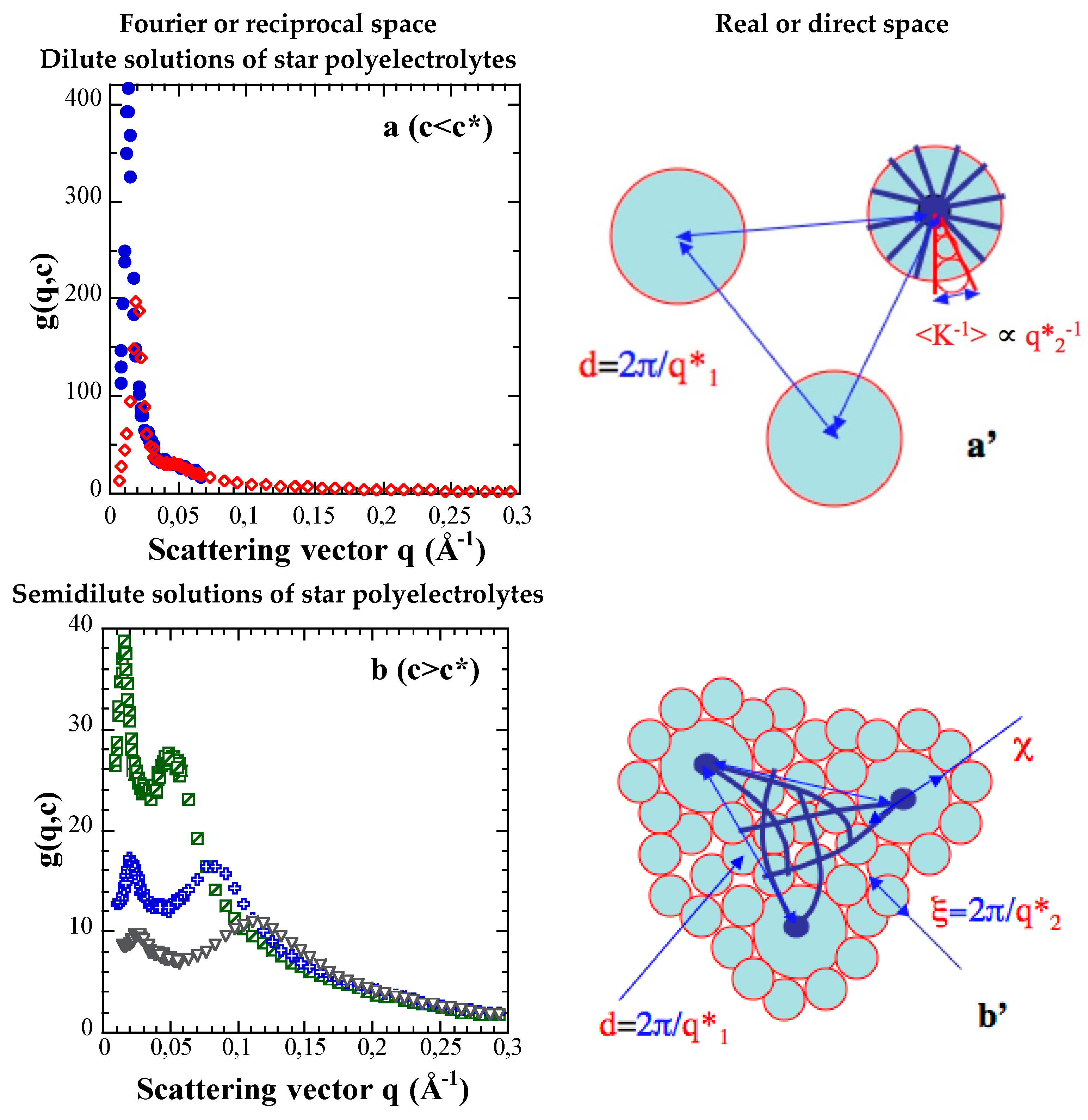
3.1. Results: Effects of Concentration c and Arm Degree of Polymerization Na
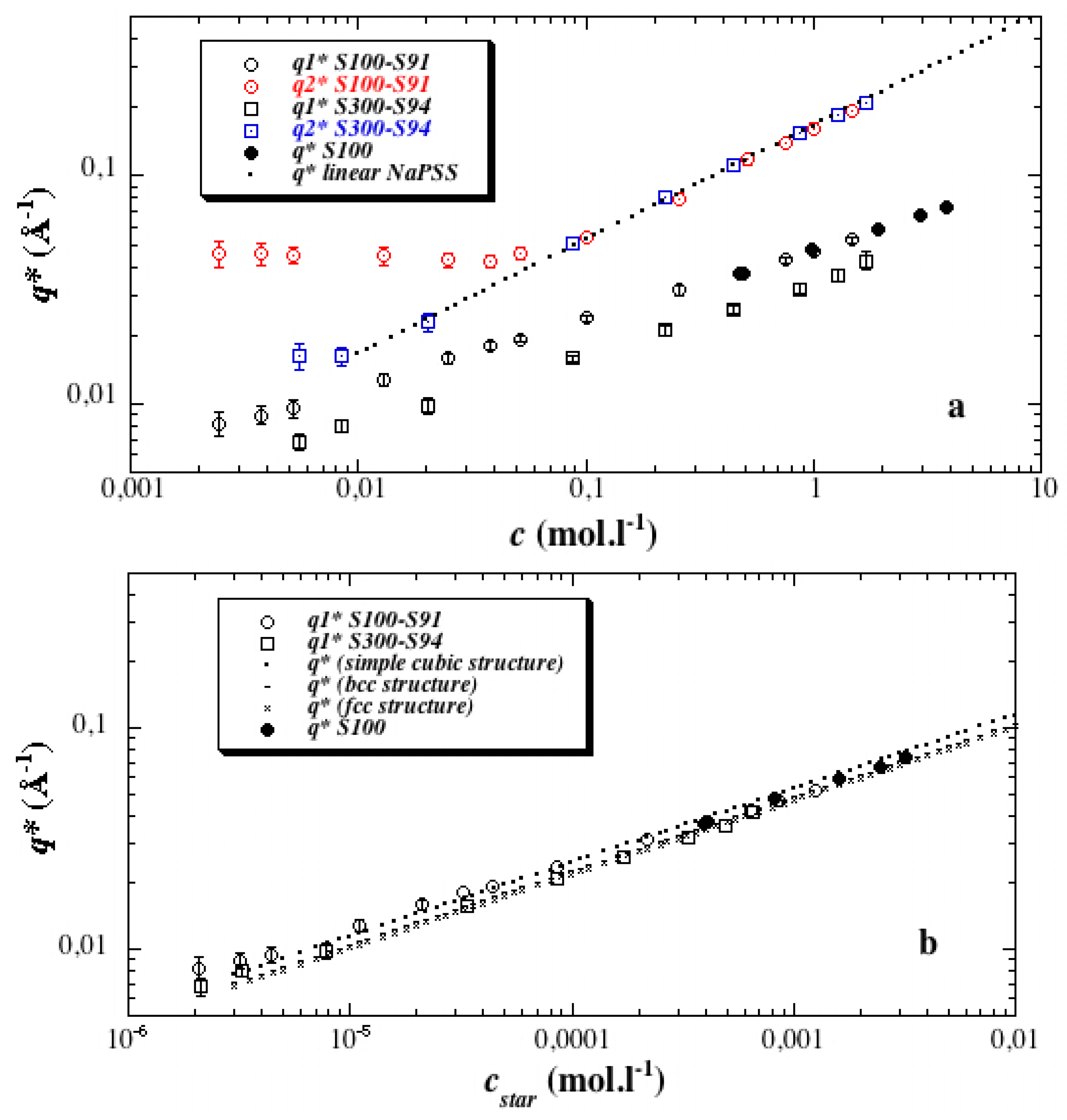
3.2. First Discussions: Using the Variations of the Maxima Positions q1* and q2* with Concentration c to Attribute Them to Defined Distances (“Indexation”)
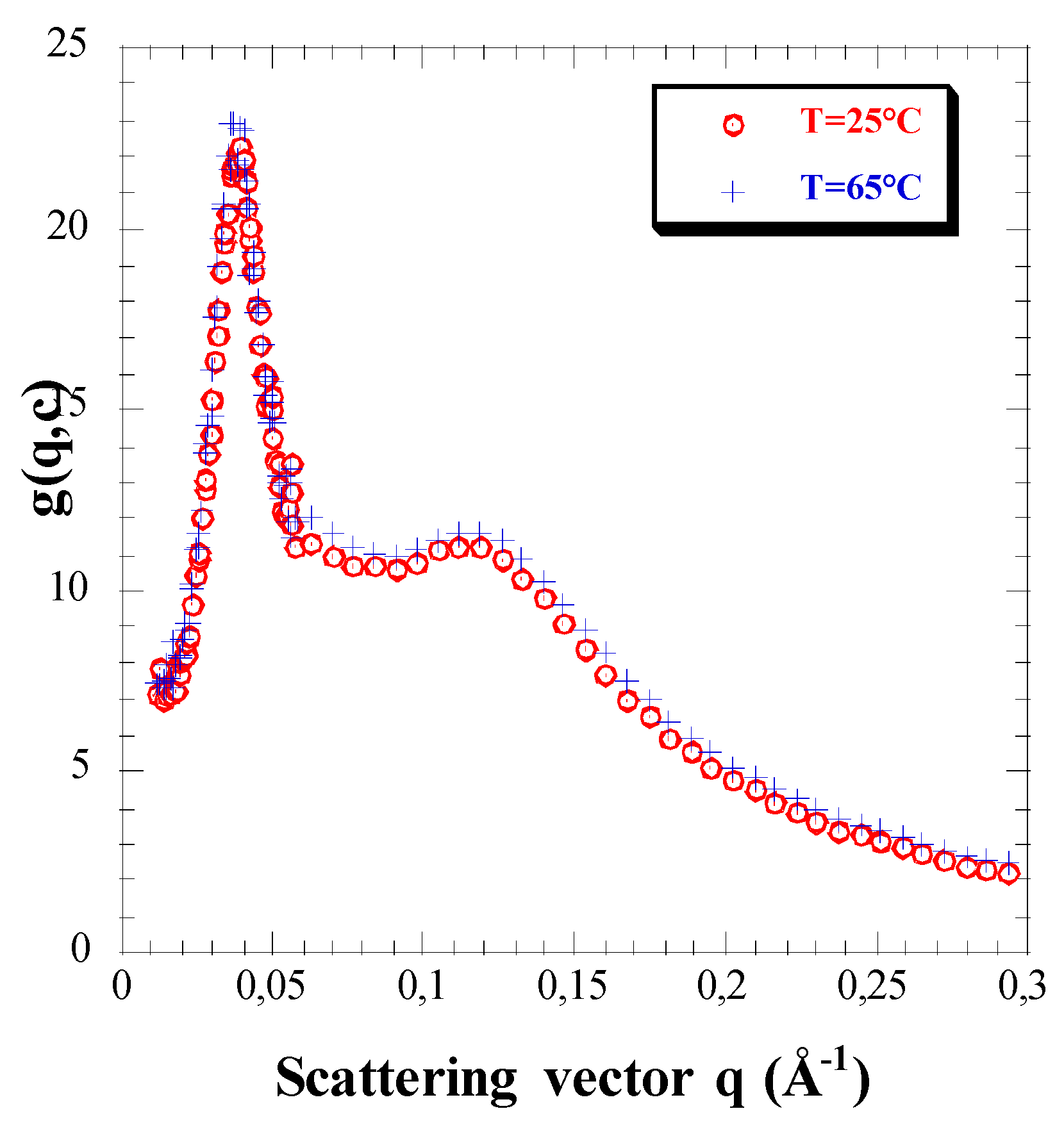
3.3. Effect of Temperature
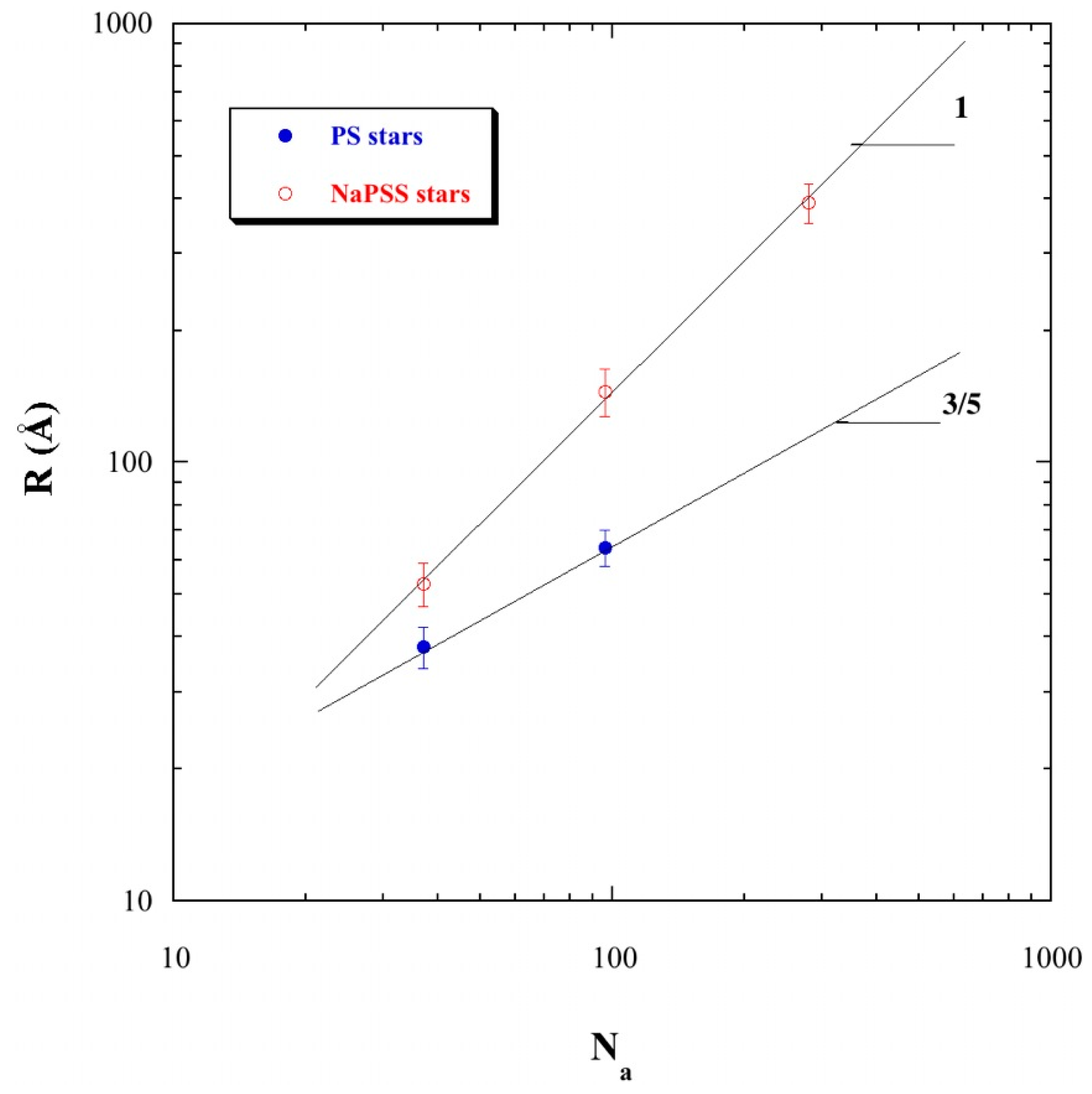
3.4. Direct Estimate of the Star Size from the c Dependence of the First Maximum and Comparison with Theories
3.4.1. Estimate of the Size R from the Star Overlap Concentration
3.4.2. Comparing with Scaling Theory
3.4.3. Comparison with the Wormlike Chain Model
3.5. Counterions Condensed and/or Trapped Inside the Stars: Information from the Second Maximum
3.5.1. Estimating the Inner Counterions Concentration from q2*
3.5.2. A Heterogeneous Radial Distribution? Present State of the Art
3.5.3. A Heterogeneous Radial Distribution? Return from Our SANS Experiments
3.6. Final Picture in Real Space
4. Summary and Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PE | Polyelectrolyte |
| SANS | Small Angle Neutron Scattering |
| SAXS | Small Angle X Rays Scattering |
| DVB | DiVinylBenzene |
| PS | Polystyrene |
| NaPSS | Sodium DiVinylBenzene Polystyrene Sulfonate |
| SEC | Size Exclusion Chromatography |
References
- Schmidt, M. Polyelectrolytes with Defined Molecular Architecture I & II; Springer-Verlag: Berlin/Heidelberg, Germany, 2004; Volume 165 & 166. [Google Scholar]
- Grest, G.S.; Fetters, L.J.; Huang, J.S.; Richter, D. Advances in Chemical Physics; Prigogine, I., Rice, S.A., Eds.; John Wiley & Sons: San Francisco, CA, USA, 1996; Volume XCIV, p. 67. [Google Scholar]
- Witten, T.A.; Pincus, P.A.; Cates, M.E. Macrocrystal ordering in star polymer solutions. Europhys. Lett. 1986, 2, 137–140. [Google Scholar] [CrossRef]
- Daoud, M.; Cotton, J.-P. Star shaped polymers: A model for the conformation and its concentration dependence. J. Phys. 1982, 43, 531–538. [Google Scholar] [CrossRef]
- Marques, C.M.; Charitat, T.; Izzo, D.; Mendes, E. Scattering from solutions of star polymers. Eur. Phys. J. B 1998, 3, 353–358. [Google Scholar] [CrossRef]
- Heinrich, M.; Rawiso, M.; Zilliox, J.-G.; Lesieur, P.; Simon, J.-P. Small-angle X-ray scattering from salt-free solutions of star-branched polyelectrolytes. Eur. Phys. J. E 2001, 4, 131–142. [Google Scholar] [CrossRef]
- Heinrich, M. Structure de solutions aqueuses de polyélectrolytes en étoile. Ph.D. Thesis, Université Louis Pasteur de Strasbourg, Strasbourg, France, 1998. [Google Scholar]
- Guenoun, P.; Muller, F.; Delsanti, M.M.; Auvray, L.; Chen, Y.J.; Mays, J.W.; Tirrell, M. Rodlike behavior of polyelectrolyte brushes. Phys. Rev. Lett. 1998, 81, 3872–3875. [Google Scholar] [CrossRef]
- Muller, F.; Delsanti, M.; Auvray, L.; Yang, J.; Chen, Y.J.; Mays, J.W.; Demé, B.; Tirrell, M.; Guenoun, P. Ordering of urchin-like charged copolymer micelles: Electrostatic, packing and polyelectrolyte correlations. Eur. Phys. J. E 2000, 3, 45–53. [Google Scholar] [CrossRef]
- Roger, M.; Guenoun, P.; Muller, F.; Belloni, L.; Delsanti, M. Monte Carlo simulations of star-branched polyelectrolyte micelles. Eur. Phys. J. E 2002, 9, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Jusufi, A.; Likos, C.N.; Löwen, H. Conformations and Interactions of Star-Branched Polyelectrolytes. Phys. Rev. Lett. 2002, 88, 18301–18304. [Google Scholar] [CrossRef] [PubMed]
- Jusufi, A.; Likos, C.N.; Löwen, H. Counterion-induced entropic interactions in solutions of strongly stretched, osmotic polyelectrolyte stars. J. Chem. Phys. 2002, 116, 11011–11027. [Google Scholar] [CrossRef]
- Jusufi, A.; Likos, C.N. Colloqium: Star-branched polyelectrolytes: The physics of their conformations and interactions. Rev. Mod. Phys. 2009, 81, 1753–1772. [Google Scholar] [CrossRef]
- Shusharina, N.P.; Rubinstein, M. Concentration regimes in solutions of polyelectrolyte stars. Macromolecules 2008, 41, 203–217. [Google Scholar] [CrossRef]
- Groenewegen, W.; Egelhaaf, S.U.; Lapp, A.; van der Maarel, J.R.C. Concentration regimes in solutions of polyelectrolyte stars. Macromolecules 2000, 33, 3283–3293. [Google Scholar] [CrossRef]
- Moinard, D.; Taton, D.; Gnanou, Y.; Rochas, C.; Borsali, R. SAXS from four-arm polyelectrolyte stars in semi-dilute solutions. Macromol. Chem. Phys. 2003, 204, 89–97. [Google Scholar] [CrossRef]
- Cotton, J.-P.; Moan, M. Inter-polyionic orientation in polyelectrolyte solution, observed by small angle neutron scattering. J. Phys. Lett. 1976, 37, 75–76. [Google Scholar] [CrossRef]
- Nierlich, M.; Williams, C.E.; Boué, F.; Cotton, J.-P.; Daoud, M.; Farnoux, B.; Jannink, G.; Picot, C.; Moan, M.; Wolf, C.; et al. Small angle neutron scattering by semi-dilute solutions of polyelectrolyte. J. Phys. (Paris) 1979, 40, 701–704. [Google Scholar] [CrossRef]
- Nierlich, M.; Boué, F.; Lapp, A.; Oberthür, R.C. Characteristic lengths of the structure of salt free polyelectrolyte solutions. A neutron scattering study. Colloid Polym. Sci. 1985, 263, 955–964. [Google Scholar] [CrossRef]
- Kaji, K.; Urakawa, H.; Kanaya, T.; Kitamaru, R. Phase diagram of polyelectrolyte solutions. J. Phys. 1988, 49, 993–1000. [Google Scholar] [CrossRef]
- Ise, N.; Okubo, T.; Kunigi, S.; Matsuoka, H.; Yamamoto, K.I.; Ishii, Y. “Ordered” structure in dilute solutions of sodium polystyrenesulfonates as studied by small-angle x-ray scattering. J. Chem. Phys. 1984, 81, 3294–3306. [Google Scholar] [CrossRef]
- De Gennes, P.G.; Pincus, P.; Velasco, R.M.; Brochard, F. Remarks on polyelectrolyte conformation. J. Phys. 1976, 37, 1461–1473. [Google Scholar] [CrossRef]
- Hayter, J.B.; Jannink, G.; Brochard-Wyart, F.; de Gennes, P.-G. Correlations and dynamics of polyelectrolyte solutions. J. Phys. Lett. 1980, 41, 451–454. [Google Scholar] [CrossRef]
- Dobrynin, A.V.; Colby, R.H.; Rubinstein, M. Scaling theory of polyelectrolyte solutions. Macromolecules 1995, 28, 1859–1871. [Google Scholar] [CrossRef]
- Essafi, W.; Lafuma, F.; Williams, C.E. Effect of solvent quality on the behaviour of highly charged polyelectrolytes. J. Phys. II 1995, 5, 1269–1275. [Google Scholar] [CrossRef]
- Essafi, W. Structure des polyélectrolytes fortement chargés. Etude DXPA d’un polyélectrolyte hydrophobe, le PSSNa partiellement sulfoné. Ph.D. Thesis, Université Paris 11, Orsay, France, 1996. [Google Scholar]
- Essafi, W.; Spiteri, M.N.; Williams, C.; Boué, F. Hydrophobic polyelectrolytes in better polar solvent. Structure and chain conformation as seen by SAXS and SANS. Macromolecules 2009, 42, 9568–9580. [Google Scholar]
- Spiteri, M.-N. Conformation et arrangement des polyélectrolytes en solutions semi-diluée. Etude par diffusion de neutrons aux petits angles. Ph.D. Thesis, Université Paris 11, Orsay, France, 1997. [Google Scholar]
- Dobrynin, A.V.; Rubinstein, M.; Obukhov, S. Cascade of transitions of polyelectrolytes in poor solvents. Macromolecules 1996, 29, 2974–2979. [Google Scholar] [CrossRef]
- Dobrynin, A.V.; Rubinstein, M. Hydrophobic polyelectrolytes. Macromolecules 1999, 32, 915–922. [Google Scholar] [CrossRef]
- Mann, B.A.F.; Kremer, K.; Lenz, O.; Holm, C. Hydrogels in poor solvents: a molecular dynamics study. Macromol. Theory Simul. 2011, 20, 721–734. [Google Scholar] [CrossRef]
- Worsfold, D.J.; Zilliox, J.G.; Rempp, P. Préparation et caractérisation de polymères-modèle à structure en étoile, par copolymérisation séquencée anionique. Can. J. Chem. 1969, 47, 3379–3385. [Google Scholar] [CrossRef]
- Rawiso, M. De l'intensité à la structure en physico-chimie des polymères. J. Phys. IV 1999, 9, Pr1–Pr147. [Google Scholar]
- Makowski, H.S.; Lundberg, R.D.; Singhal, G.S. Flexible polymeric compositions comprising a normally plastic polymer sulfonated to about 0.2 to about 10 mole % sulfonate. U.S. Patent 3870841, 1975. [Google Scholar]
- Essafi, W.; Lafuma, F.; Williams, C.E. Macroion Characterization from Dilute Solutions to Complex Fluids; Schmitz, K.S., Ed.; ACS Symposium Series 548; American Chemical Society: Washington, DC, USA, 1994; Chapter 21. [Google Scholar]
- Tondre, C.; Zana, R. Apparent molal volumes of polyelectrolytes in aqueous solutions. J. Phys. Chem. 1972, 76, 3451–3459. [Google Scholar] [CrossRef]
- Millero, F.J. The partial molal volumes of electrolytes in aqueous solutions. In Water and Aqueous Solutions; Horne, R.A., Ed.; Wiley Interscience: New York, NY, USA, 1972; Chapter 13. [Google Scholar]
- Cotton, J.-P. Introduction to scattering experiment. In Neutron X-ray and Light Scattering: Introduction to an Investigate Tool for Colloidal and Polymeric Systems; Lindner, P., Zemb, Th., Eds.; North-Holland Delta Series: Amsterdam, The Netherlands, 1991; Chapter 2; pp. 19–31. [Google Scholar]
- Lindner, P. Scattering experiments: experimental aspects, initial data reduction and absolute calibration. In Neutrons, X-rays and Light: Scattering Methods Applied to Soft Condensed Matter; Lindner, P., Zemb, Th., Eds.; North-Holland Delta Series: Amsterdam, The Netherlands, 2002; Chapter 2; pp. 23–48. [Google Scholar]
- Brûlet, A.; Lairez, D.; Lapp, A.; Cotton, J.-P. Improvement of data treatment in small-angle neutron scattering. J. Appl. Cryst. 2007, 40, 165–177. [Google Scholar] [CrossRef]
- Sears, G.D. Neutron scattering lengths and cross sections. Neutron News 1992, 3, 26–37. [Google Scholar] [CrossRef]
- Van Krevelen, D.W.; te Nijenhuis, K. Properties of Polymers, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Combet, J.; Isel, F.; Rawiso, M.; Boué, F. Scattering functions of flexible polyelectrolytes in the presence of mixed valence counterions: condensation and scaling. Macromolecules 2005, 38, 7456–7469. [Google Scholar] [CrossRef]
- Boué, F.; Cotton, J.-P.; Lapp, A.; Jannink, G. A direct measurement of the polyion conformation in aqueous solutions at different temperatures. Small angle neutron scattering of PSSNa using zero average and full contrast. J. Chem. Phys. 1994, 101, 2562–2568. [Google Scholar] [CrossRef]
- Essafi, W.; Haboubi, N.; Williams, C.; Boué, F. Weak temperature dependence of structure in hydrophobic polyelectrolyte aqueous solution (PSSNa): correlation between scattering and viscosity. J. Phys. Chem. B 2011, 115, 8951–8960. [Google Scholar] [CrossRef] [PubMed]
- Borisov, O.V. Conformations of star-branched polyelectrolytes. J. Phys. II 1996, 6, 1–19. [Google Scholar] [CrossRef]
- Borisov, O.V.; Zhulina, E.B. Effects of ionic strength and charge annealing in star-branched polyelectrolytes. Eur. Phys. J. B 1998, 4, 205–217. [Google Scholar] [CrossRef]
- Kassapidou, K.; Jesse, W.; Kuil, M.E.; Lapp, A.; Egelhaaf, S.; van der Maarel, J.R.C. Structure and charge distribution in DNA and poly(styrenesulfonate) aqueous solutions. Macromolecules 1997, 30, 2671–2684. [Google Scholar] [CrossRef]
- Dubois, E.; Boué, F. Conformation of polystyrene sulfonate polyions in the presence of multivalent ions: Small angle neutron scattering experiments. Macromolecules 2001, 34, 3684–3697. [Google Scholar] [CrossRef]
- Combet, J.; Lorchat, P.; Rawiso, M. Salt-free aqueous solutions of polyelectrolytes: Small angle X-ray and neutron scattering characterization. Eur. Phys. J. Spec. Top. 2012, 213, 243–265. [Google Scholar] [CrossRef]
- Boris, D.C.; Colby, R.H. Rheology of sulfonated polystyrene solutions. Macromolecules 1998, 31, 5746–5755. [Google Scholar] [CrossRef]
- Zhulina, E.B. Phase diagram for semi-rigid macromolecules grafted to a solid sphere. Polym. Sci. USSR 1984, 26, 885–891. [Google Scholar] [CrossRef]
- Birshtein, T.M.; Zhulina, E.B. Conformations of star-branched macromolecules. Polymer 1984, 25, 1453–1461. [Google Scholar] [CrossRef]
- Birshtein, T.M.; Zhulina, E.B.; Borisov, O.V. Temperature-concentration diagram for a solution of star-branched macromolecules. Polymer 1986, 27, 1078–1086. [Google Scholar] [CrossRef]
| Sample | XL % | <Ma>N g·mol−1 | Ia | <Na>N | MN g·mol−1 | I | τDVB % | <f> | σ/<f> | τS % | <m> g·mol−1 | <v> cm3·mol−1 | τW % | c* mol·L−1 | Φ* % |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S40 | 3 | 3.8 k | 1.05 | 36 | 40 k | 1.13 | 3.70 | 10.0 | 0.35 | 0 | 105.1 | 96.68 | 0 | 1.924 | 18.60 |
| S40-S85 | 3 | – | 1.05 | 36 | – | 1.13 | 3.70 | 10.0 | 0.35 | 85 ± 2 | 188.6 | 109.90 | 10.7 | 0.515 | 5.66 |
| S100 | <2 | 10 k | 1.09 | 96 | 126 k | 1.06 | 1.86 | 10.8 | 0.32 | 0 | 104.6 | 96.54 | 0 | 0.993 | 9.59 |
| S100-S91 | <2 | – | 1.09 | 96 | – | 1.06 | 1.86 | 10.8 | 0.32 | 91 ± 5 | 195.8 | 111.22 | 10 | 0.075 | 0.83 |
| S300 | <2 | 29 k | 1.10 | 280 | 364 k | 1.07 | 0.64 | 12.4 | 0.25 | 0 | 104.3 | 96.45 | 0 | – | – |
| S300-S94 | <2 | – | 1.10 | 280 | – | 1.07 | 0.64 | 12.4 | 0.25 | 94 ± 5 | 199.6 | 111.93 | 11 | 0.01 | 0.11 |
| Sample | <Na>N | R (Å) | q2* (Å−1) | q2*<Na>N (Å−1) | q2*R |
|---|---|---|---|---|---|
| S40-S85 | 36 | 53 | 0.116 | 4.2 | 6.1 |
| S100-S91 | 96 | 145 | 0.045 | 4.3 | 6.5 |
| S300-S94 | 280 | 391 | 0.0162 | 4.5 | 6.3 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boué, F.; Combet, J.; Demé, B.; Heinrich, M.; Zilliox, J.-G.; Rawiso, M. SANS from Salt-Free Aqueous Solutions of Hydrophilic and Highly Charged Star-Branched Polyelectrolytes. Polymers 2016, 8, 228. https://doi.org/10.3390/polym8060228
Boué F, Combet J, Demé B, Heinrich M, Zilliox J-G, Rawiso M. SANS from Salt-Free Aqueous Solutions of Hydrophilic and Highly Charged Star-Branched Polyelectrolytes. Polymers. 2016; 8(6):228. https://doi.org/10.3390/polym8060228
Chicago/Turabian StyleBoué, François, Jérôme Combet, Bruno Demé, Martine Heinrich, Jean-Georges Zilliox, and Michel Rawiso. 2016. "SANS from Salt-Free Aqueous Solutions of Hydrophilic and Highly Charged Star-Branched Polyelectrolytes" Polymers 8, no. 6: 228. https://doi.org/10.3390/polym8060228
APA StyleBoué, F., Combet, J., Demé, B., Heinrich, M., Zilliox, J.-G., & Rawiso, M. (2016). SANS from Salt-Free Aqueous Solutions of Hydrophilic and Highly Charged Star-Branched Polyelectrolytes. Polymers, 8(6), 228. https://doi.org/10.3390/polym8060228