Electro-Active Polymers (EAPs): A Promising Route to Design Bio-Organic/Bioinspired Platforms with on Demand Functionalities
Abstract
:1. Introduction
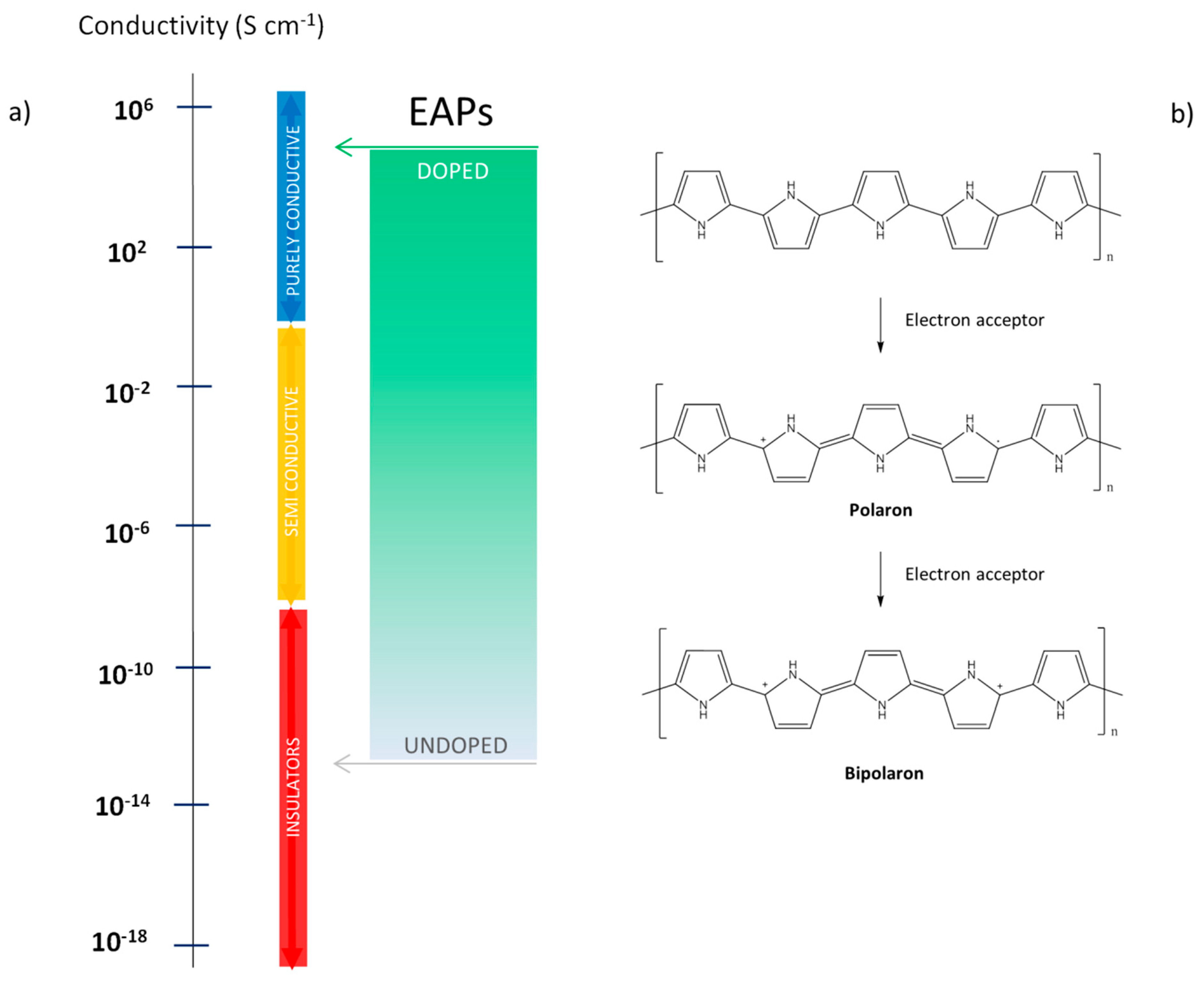
1.1. Mechanism of EAPs Polymers: Conductivity Source and Doping
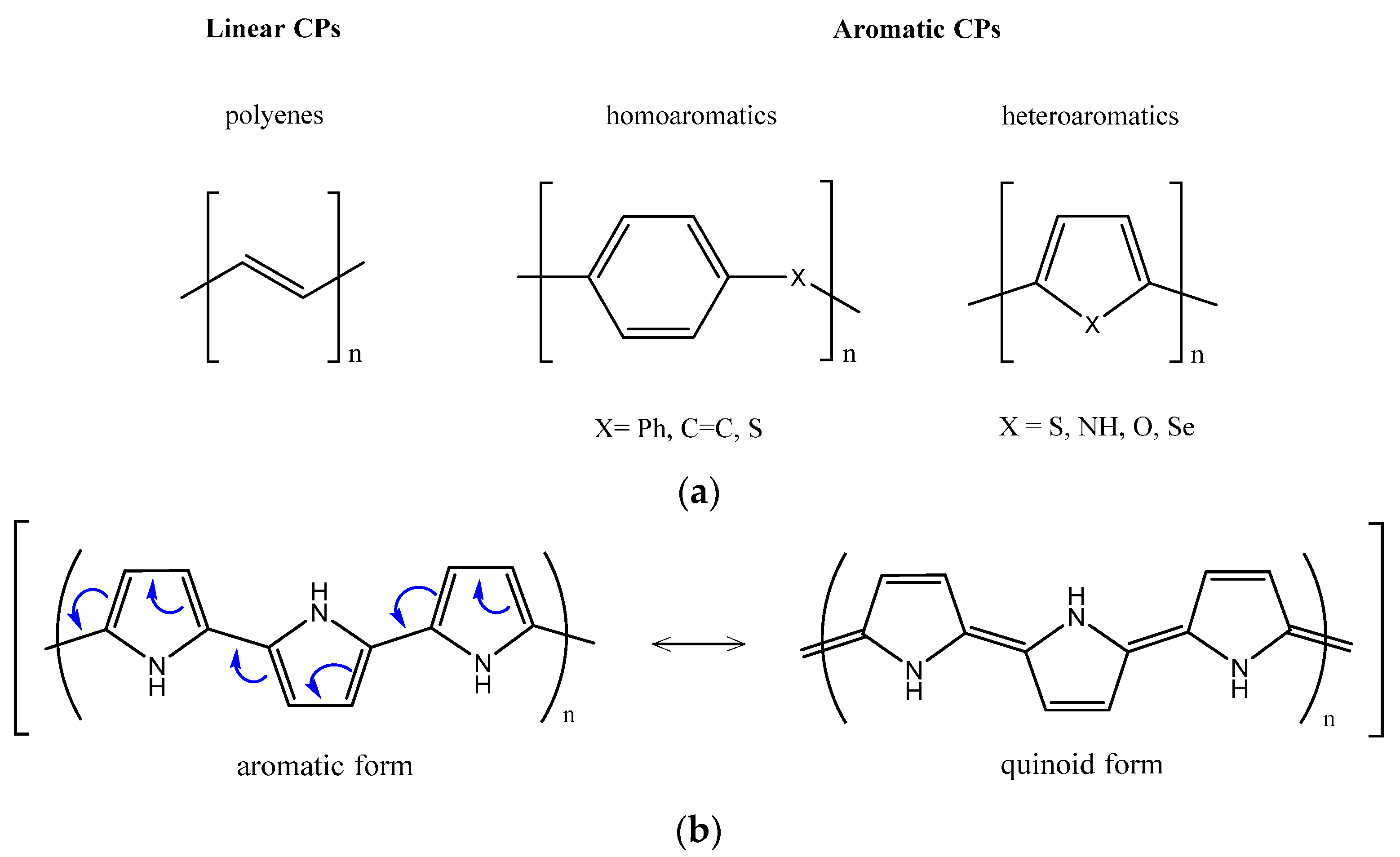
1.2. EAP Classification
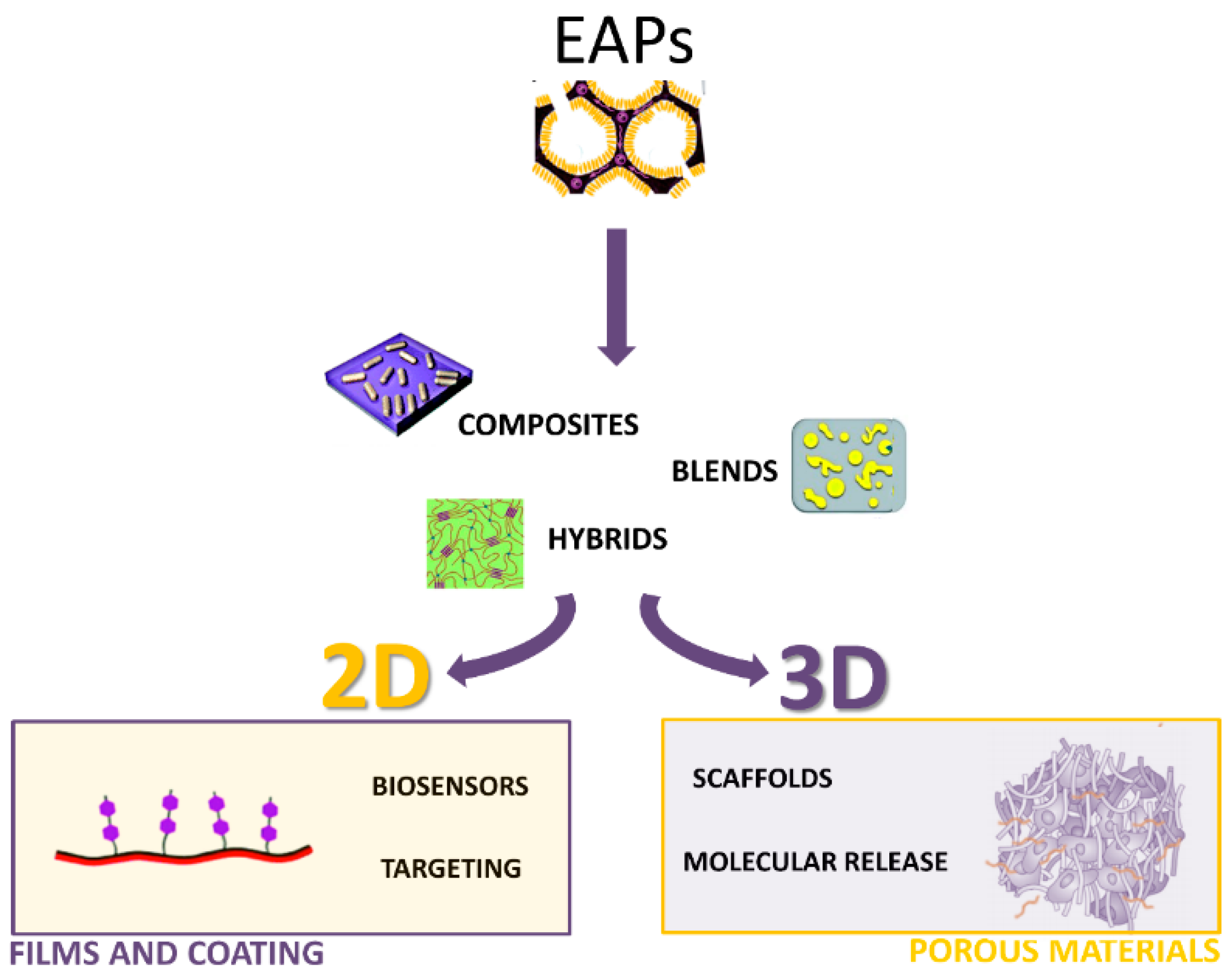
2. EAPs Processing
2.1. Two-Dimensional (2D) Processes for Coatings
2.2. Three-Dimensional (3D) Processing
2.2.1. Blends
2.2.2. Composites
2.2.3. Porous Materials
3. Applications in Biomedical Field
3.1. EAPs to Guide Cell Functions: Tissue Engineering
3.2. EAPs to Target Drugs and Biological Molecules: Drug Delivery
3.3. EAPs to Monitor Cell Activity: Biosensors
4. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Guarino, V.; Alvarez-Perez, M.A.; Borriello, A.; Napolitano, T.; Ambrosio, L. Conductive PANi/PEGDA macroporous hydrogels for nerve regeneration. Adv. Healthc. Mater. 2013, 2, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, B.D.; Chaubey, A.; Singh, S.P. Prospects of conducting polymers in biosensors. Anal. Chim. Acta 2006, 578, 59–74. [Google Scholar] [CrossRef] [PubMed]
- Wallace, G.G.; Spinks, G.M.; Kane-Maguire, L.A.P.; Teasdate, P.R. Conductive Electroactive Polimers—Intelligent Polymer System, 3rd ed.; CRC Press LLC.: Boca Raton, FL, USA, 2003. [Google Scholar]
- Balint, R.; Cassidy, N.J.; Cartmell, S.H. Conductive polymers: Towards a smart biomaterial for tissue engineering. Acta Biomat. 2014, 10, 2341–2353. [Google Scholar] [CrossRef] [PubMed]
- Ivory, D.M.; Miller, G.G.; Sowa, J.M.; Shacklette, L.W.; Chance, R.R.; Baughman, R.H. Highly conducting charge-transfer complexes of poly(p-phenylene). J. Chem. Phys. 1979, 71, 1506. [Google Scholar] [CrossRef]
- Heeger, A.J.; MacDiarmid, A.G.; Shirakawa, H. The Nobel Prize in Chemistry, 2000: Conductive Polymers; The Royal Swedish Academy of Sciences: Stockholm, Sweden, 2000. [Google Scholar]
- Guimard, N.K.; Gomez, N.; Schmidt, C.E. Conducting polymers in biomedical engineering. Prog. Polym. Sci. 2007, 32, 876–921. [Google Scholar] [CrossRef]
- Kaur, G.; Adhikari, R.; Cass, P.; Bown, P.; Gunatillake, M. Electrically conductive polymers and composites for biomedical applications. RSC Adv. 2015, 5, 37553–37567. [Google Scholar] [CrossRef]
- Chiang, C.K.; Fincher, C.R.; Park, Y.W., Jr.; Heeger, A.J.; Shirakawa, H.; Louis, E.J.; Gau, S.C.; MacDiarmid, A.G. Electrical conductivity in doped polyacetylene. Phys. Rev. Lett. 1978, 40, 1472. [Google Scholar] [CrossRef]
- Harrison, W.A. Elementary theory of heteroconjunctions. J. Vac. Sci. Technol. 1977, 14, 11016. [Google Scholar] [CrossRef]
- Roncali, J. Synthetic principles for bandgap control in linear π-conjugated systems. Chem. Rev. 1997, 97, 173–205. [Google Scholar] [CrossRef] [PubMed]
- Brédas, J.L.; Street, G.B. Polarons, bipolarons and solitons in conducting polymers. Acc. Chem. Res. 1985, 18, 309–315. [Google Scholar] [CrossRef]
- Smela, E. Conjugated polymer actuators for biomedical applications. Adv. Mater. 2003, 15, 481–494. [Google Scholar] [CrossRef]
- Iseki, M.; Saito, K.; Kuhara, K.; Mizukami, A. Electrochemical exchange process of dopant anions in polypyrrole. Synth. Met. 1991, 40, 117–126. [Google Scholar] [CrossRef]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.H.; Baharvand, H.; Kiani, S.; Al-Deyab, S.S.; Ramakrishna, S. Application of conductive polymers, scaffolds and electrical stimulation for nerve tissue engineering. J. Tissue Eng. Regen. Med. 2011, 15, e17–e35. [Google Scholar] [CrossRef] [PubMed]
- Tourillon, G. Polythiophene and its derivatives. In Handbook of Conducting Polymers, 2nd ed.; Skotheim, T.A., Ed.; Marcel Dekker, INC.: New York, NY, USA, 1986; Volume 1, pp. 293–350. [Google Scholar]
- Holland, E.R.; Pomfret, S.J.; Adams, P.N.; Abell, L.; Monkman, A.P. Doping dependent transport properties of polyaniline-CSA films. Synt. Met. 1997, 84, 777–778. [Google Scholar] [CrossRef]
- Sinha, S.; Bhadra, S.; Khastgir, D. Effect of dopant type on the properties of polyaniline. J. Appl. Polym. Sci. 2009, 112, 3135–3140. [Google Scholar] [CrossRef]
- Heeger, A.J.; Kivelson, S.; Schrieffer, J.R.; Su, W.-P. Solitons in conducting polymers. Rev. Mod. Phys. 1988, 60, 781. [Google Scholar] [CrossRef]
- Garnier, F.; Hajlaoui, R.; Yassar, A.; Srivastava, P. All-polymer field-effect transistor realized by printing techniques. Science 1994, 265, 1684–1686. [Google Scholar] [CrossRef] [PubMed]
- Schön, J.H.; Dodabalapur, A.; Bao, Z.; Kloc, C.; Schenker, O.; Batlogg, B. Gate-induced superconductivity in a solution-processed organic polymer film. Nature 2001, 410, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Lu, J.; Matthews, B.; Mau, A.W.H. Doping of conducting polymers by sulfonated fullerene derivatives and dendrimers. J. Phys. Chem. B 1998, 102, 4049–4053. [Google Scholar] [CrossRef]
- Cao, Y.; Andreatta, A.; Heeger, A.J.; Smith, P. Influence of chemical polymerization conditions on the properties of polyaniline. Polymer 1989, 30, 2305–2311. [Google Scholar] [CrossRef]
- Pron, A.; Genoud, F.; Menardo, C.; Nechtschein, M. The effect of the oxidation conditions on the chemical polymerization of polyaniline. Synth. Met. 1988, 24, 193–201. [Google Scholar] [CrossRef]
- Wallace, G.G.; Smyth, M.; Zhao, H. Conducting electroactive polymer-based biosensors. Trends Anal. Chem. 1999, 18, 245–251. [Google Scholar] [CrossRef]
- Shi, G.; Rouabhia, M.; Wang, Z.; Dao, L.H.; Zhang, Z. A novel electrically conductive and biodegradable composite made of polypyrrole nanoparticles and polylactide. Biomaterials 2004, 25, 2477–2488. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.Y.; Langer, R.; Ingber, D.E. Electrically conducting polymers can noninvasively control the shape and growth of mammalian cells. Proc. Natl. Acad. Sci. USA 1994, 91, 3201–3204. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, M.R.; Murray, P.; Spinks, G.M.; Wallace, G.G. Mechanism of electromechanical actuation in polypyrrole. Synt. Met. 1995, 73, 247–256. [Google Scholar] [CrossRef]
- Wallace, G.; Spinks, G. Conducting polymers—Bridging the bionic interface. Soft Matter 2007, 3, 665–671. [Google Scholar] [CrossRef]
- Ding, C.; Qian, X.; Yu, G.; An, X. Dopant effect and characterization of polypyrrole-cellulose composites prepared by in situ polymerization process. Cellulose 2010, 17, 1067–1077. [Google Scholar] [CrossRef]
- Boyle, A.; Genies, E.; Fouletier, M. Electrochemical behaviour of polypyrrole films doped with ATP anions. J. Electroanal. Chem. Interf. Electrochem. 1990, 279, 179–186. [Google Scholar] [CrossRef]
- Shin, M.-C.; Kim, H.-S. Electrochemical characterization of polypyrrole/glucose oxidase biosensor: Part I. Influence of enzyme concentration on the growth and properties of the film. Biosens. Bioelectron. 1996, 11, 161–169. [Google Scholar] [CrossRef]
- Li, H.C.; Khor, E. A collagen-poiypyrrole hybrid: Influence of 3-butanesulfonate substitution. Macromol. Chem. Phys. 1995, 196, 1801–1812. [Google Scholar]
- Guiseppi-Elie, A. Electroconductive hydrogels: Synthesis, characterization and biomedical applications. Biomaterials 2010, 31, 2701–2716. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zhuang, X.; Hu, J.; Lang, L.; Zhang, P.; Wang, Y.; Chen, X.; Wei, Y.; Jing, X. Synthesis of biodegradable and electroactive multiblock polylactide and aniline pentamer copolymer for tissue engineering applications. Biomacromolecules 2008, 9, 850–858. [Google Scholar] [CrossRef] [PubMed]
- Ateh, D.D.; Navsaria, H.A.; Vadgama, P. Polypyrrole-based conducting polymers and interactions with biological tissues. J. R. Soc. Interface 2006, 3, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Mintmire, J.W.; White, C.T.; Elert, M.L. Heteroatom effects in heterocyclic ring chain polymers. Synth. Met. 1986, 16, 235–243. [Google Scholar]
- Ouyang, J.; Chu, C.-W.; Chen, F.-C.; Xu, Q.; Yang, Y. High-conductivity poly(3,4-ethylenedioxythiophene): Poly(styrene sulfonate) film and its application in polymer optoelectronic devices. Adv. Funct. Mater. 2005, 2, 203–208. [Google Scholar] [CrossRef]
- Ouyang, J.; Xu, Q.; Chu, C.W.; Yang, Y.; Li, G.; Shinar, J. On the mechanism of conductivity enhancement in poly(3,4-ethylenedioxythiophene): Poly(styrene sulfonate) film through solvent treatment. Polymer 2004, 45, 8443–8450. [Google Scholar] [CrossRef]
- Rozlosnik, N. New directions in medical biosensors employing poly(3,4-ethylenedioxy thiophene) derivative-based electrodes. Anal. Bioanal. Chem. 2009, 395, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Sharmaeur, R.C. Advances in conductive polymers. Polym. J. 1998, 34, 1053–1060. [Google Scholar] [CrossRef]
- Green, R.A.; Lovell, N.H.; Wallace, G.G.; Poole-Warren, L.A. Conducting polymers for neural interfaces: Challenges in developing an effective long-term implant. Biomaterials 2008, 29, 3393–3399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yan, Y.; Li, S.; Feng, T. The synthesis and characterization of a novel biodegradable and electroactive polyphosphazene for nerve regeneration. Mat. Sci. Eng. C 2010, 30, 160–166. [Google Scholar] [CrossRef]
- Shastri, V.R. Evaluation of Polypyrrole thin Films as Substratum for Mammalian Cell Culture; Rensselaer Polytechnic Institute: Troy, NY, USA, 1995. [Google Scholar]
- Garner, B.; Georgevich, A.; Hodgson, A.J.; Liu, L.; Wallace, G.G. Polypyrrole-heparine composites as stimulus-responsive substrates for endothelial cell growth. J. Biomed. Mater. Res. 1999, 44, 121–129. [Google Scholar] [CrossRef]
- Lee, J.Y.; Bashur, C.A.; Goldstein, A.S.; Schmidt, C.E. Polypyrrole-coated electrospun PLGA nanofibers for neural tissue applications. Biomaterials 2009, 30, 4325–4335. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.E.; Shastri, V.R.; Vacanti, J.P.; Langer, R. Stimulation of neurite outgrowth using an electrically conducting polymer. Proc. Natl. Acad. Sci. USA 1997, 94, 8948–8953. [Google Scholar] [CrossRef] [PubMed]
- Gomez, N.; Lee, J.Y.; Nickels, J.D.; Schmidt, C.E. Micropatterned polypyrrole: A combination of electrical and topographical characteristics for the stimulation of cells. Adv. Funct. Mater. 2007, 17, 1645–1653. [Google Scholar] [CrossRef] [PubMed]
- Brahim, S.; Narinesingh, D.; Guiseppi-Elie, A. Amperometric determination of cholesterol in serum using a cholesterol oxidase biosensor with a polypyrrole/ hydrogel membrane. Anal. Chim. Acta 2001, 448, 27–36. [Google Scholar] [CrossRef]
- Wang, C.H.; Dong, Y.Q.; Sengothi, K.; Tan, K.L.; Kang, E.T. In-vivo tissue response to polyaniline. Synth. Met. 1999, 102, 1313–1314. [Google Scholar] [CrossRef]
- Kamalesh, S.; Tan, P.; Wang, J.; Lee, T.; Kang, E.-T.; Wang, C.-H. Biocompatibility of electroactive polymers in tissues. J. Biomed. Mater. Res. 2000, 52, 467–478. [Google Scholar] [CrossRef]
- Bidez, P.R.; Li, S.; MacDiarmid, A.G.; Venancio, E.C.; Wei, Y.; Lelkes, P.I. Polyaniline, an electroactive polymer, supports adhesion and proliferation of cardiac myoblasts. J. Biomat. Sci. Polym. E. 2006, 17, 199–212. [Google Scholar] [CrossRef]
- Huang, W.-S.; Humphrey, B.D.; MacDiarmid, A.G. Polyaniline, a novel conducting polymer. Morphology and chemistry of its oxidation and reduction in aqueous electrolytes. J. Chem. Soc. Faraday Trans. 1986, 82, 2385–2400. [Google Scholar] [CrossRef]
- Anand, J.; Palaniappan, S.; Sathyanarayana, D.N. Conducting polyaniline blends and composites. Prog. Polym. Sci. 1998, 23, 993–1018. [Google Scholar] [CrossRef]
- Bhadraa, S.; Khastgir, D.; N. Singha, K.; Lee, J.H. Progress in preparation, processing and applications of polyaniline. Prog. Polym. Sci. 2009, 34, 783–810. [Google Scholar] [CrossRef]
- Kim, D.-H.; Abidian, M.; Martin, D.C. Conducting polymers grown in hydrogel scaffolds coated on neural prosthetic devices. J. Biomed. Mater. Res. A 2004, 71, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Widge, A.S.; Jeffries-El, M.; Cui, X.; Lagenaur, C.F.; Matsuoka, Y. Self-assembled monolayers of polythiophene conductive polymers improve biocompatibility and electrical impedance of neural electrodes. Biosens. Bioelectron. 2007, 22, 1723–1732. [Google Scholar] [CrossRef] [PubMed]
- Peramo, A.; Urbanchek, M.G.; Spanninga, S.A.; Povlich, L.K.; Cederna, P.; Martin, D.C. In situ polymerization of a conductive polymer in acellular muscle tissue constructs. Tissue Eng. Part A 2008, 14, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Richardson-Burns, S.M.; Hendricks, J.L.; Martin, D.C. Electrochemical polymerization of conducting polymers in living neural tissue. J. Neural. Eng. 2007, 4, L6–L13. [Google Scholar] [CrossRef] [PubMed]
- Qazi, T.H.; Rai, R.; Dippold, D.; Roether, J.E.; Schubert, D.W.; Rosellini, E.; Barbani, N.; Boccaccini, A.R. Development and characterization of novel electrically conductive PANI-PGS composites for cardiac tissue engineering applications. Acta Biomater. 2014, 10, 2434–2445. [Google Scholar] [CrossRef] [PubMed]
- Zilberman, M.; Siegmann, A.; Narkis, M. Conductivity and structure of melt-processed polyaniline binary and ternary blends. Polym. Adv. Technol. 2000, 11, 20–26. [Google Scholar] [CrossRef]
- Zilberman, M.; Siegmann, A.; Narkis, M. Melt-processed electrically conductive polymer/polyaniline blends. J. Macromol. Sci. B 1998, 37, 301–318. [Google Scholar] [CrossRef]
- Macinnes, D.; Funt, B.L. Poly-o-methoxyaniline: A new soluble conducting polymer. Synth. Met. 1988, 25, 23542. [Google Scholar] [CrossRef]
- Pud, A.A.; Shapoval, G.S.; Vinogradnyi, P.G. Electropolymerization of pyrrole in polymer matrices. Russ. J. Electrochem. 2000, 36, 447. [Google Scholar] [CrossRef]
- Wan, Y.; Wen, D.J. Preparation and characterization of porous conducting poly(d,l-lactide) composite membranes. J. Membr. Sci. 2005, 246, 193–194. [Google Scholar] [CrossRef]
- Borriello, A.; Guarino, V.; Schiavo, L.; Alvarez-Perez, M.A.; Ambrosio, L. Optimizing PANi doped electroactive substrates as patches for the regeneration of cardiac muscle. J. Mater. Sci. Mat. Med. 2011, 22, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, U.M.; Kumar, S.V.; Nagiah, N.; Sivagnanam, S.T. Fabrication of polyvinyl alcohol-polyvinylpyrrolidone blend scaffolds via electrospinning for tissue engineering applications. Int. J. Polym. Mat. Polym. Biomater. 2014, 63, 476–485. [Google Scholar] [CrossRef]
- Li, L.; Ge, L.; Ma, P.X.; Guo, B. Injectable conducting interpenetrating polymer network hydrogels from gelatin-graft-polyaniline and oxidized dextran with enhanced mechanical properties. RSC Adv. 2015, 5, 92490–92498. [Google Scholar] [CrossRef]
- Li, W.J.; Cooper, J.A.; Mauck, R.L.; Tuan, R.S. Fabrication and characterization of six electrospun poly(α-hydroxy ester) based fibrous scaffolds for tissue engineering application. Acta Biomater. 2006, 2, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.L.; Finne-Wistrand, A.; Albertsson, A.C. Molecular architecture of electroactive and biodegradable copolymers composed of polylactide and carboxyl-capped aniline trimer. Biomacromolecules 2010, 11, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Pomfret, S.J.; Adams, P.N.; Comfort, N.P. Electrical and mechanical properties of polyaniline fibres produced by a one-step wet spinning. Polymer 2000, 41, 2265–2269. [Google Scholar] [CrossRef]
- McKeon, K.D.; Lewis, A.; Freeman, J.W. Electrospun poly(d,l-lactide) and polyaniline scaffold characterization. J. Appl. Polym. Sci. 2010, 115, 1566–1572. [Google Scholar] [CrossRef]
- Yu, Q.Z.; Shi, M.M.; Deng, M.; Wang, M. Morphology and conductivity of polyaniline sub-micron fibers prepared by electrospinning. Mater. Sci. Eng. B 2008, 150, 70–76. [Google Scholar] [CrossRef]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.H.; Ramakrishna, S. Electrical stimulation of nerve cells using conductive nanofibrous scaffolds for nerve tissue engineering. Tissue Eng. A 2009, 15, 3605–3619. [Google Scholar] [CrossRef] [PubMed]
- Struèmpler, R.; Glatz-Reichenbach, J. Conducting polymer composites. J. Electroceramics 1999, 3, 329–346. [Google Scholar] [CrossRef]
- Takano, T.; Mikazuki, A.; Kobayashi, T. Conductive polypyrrole composite films prepared using wet cast technique with a pyrrole–cellulose acetate solution. Polym. Eng. Sci. 2014, 54, 78–84. [Google Scholar]
- Broda, C.R.; Lee, J.Y.; Sirivisoot, S.; Schmidt, C.E.; Harrison, B.S. A chemically polymerized electrically conducting composite of polypyrrole nanoparticles and polyurethane for tissue engineering. J. Biomed. Mater. Res. Part A 2011, 98, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Sarvari, R.; Massoumi, B.; Jaymand, M.; Beygi-Khosrowshahic, Y.; Abdollahid, M. Novel three-dimensional, conducting, biocompatible, porous, and elastic polyanilinebased scaffolds for regenerative therapies. RSC Adv. 2016, 6, 19437. [Google Scholar] [CrossRef]
- Hatchett, D.; Josowicz, M.; Janata, J. Acid doping of polyaniline: Spectroscopic and electrochemical studies. J. Phys. Chem. B 1999, 103, 10992–10998. [Google Scholar] [CrossRef]
- Severs, N.J. The cardiac muscle cell. Bioassays 2000, 22, 188–199. [Google Scholar] [CrossRef]
- Dvir, T.; Timko, B.P.; Brigham, M.D.; Naik, S.R.; Karajanagi, S.S.; Levy, O.; Jin, H.; Parker, K.K.; Langer, R.; Kohane, D.S. Nanowired three-dimensional cardiac patches. Nat. Nanotechnol. 2011, 6, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Baheiraei, N.; Yeganeh, H.; Ai, J.; Gharibi, R.; Ebrahimi-Barough, S.; Azami, M.; Vahdat, S.; Baharvand, H. Preparation of a porous conductive scaffold from aniline pentamer-modified polyurethane/PCL blend for cardiac tissue engineering. Biomed. Mater. Res. Part A 2015, 103A, 3179–3187. [Google Scholar] [CrossRef] [PubMed]
- Baniasadi, H.; Ramazani, A.; Mashayekhan, S.; Farani, M.R.; Ghaderinezhad, F.; Dabaghi, M. Design, Fabrication, and characterization of novel porous conductive scaffolds for nerve tissue engineering. Int. J. Polym. Mat. Polym. Biomater. 2015, 64, 969–977. [Google Scholar] [CrossRef]
- Wan, A.M.D.; Inal, S.; Williams, T.; Wang, K.; Leleux, P.; Estevez, L.; Giannelis, E.P.; Fischbach, C.; Malliaras, G.G.; Gourdon, D. 3D conducting polymer platforms for electrical control of protein conformation and cellular functions. J. Mater. Chem. B 2015, 3, 5040–5048. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Cooper, A.I. Aligned porous structures by directional freezing. Adv. Mater. 2007, 19, 1529–1533. [Google Scholar] [CrossRef]
- Estevez, L.; Dua, R.; Bhandari, N.; Ramanujapuram, A.; Wang, P.; Giannelis, E.P. A facile approach for the synthesis of monolithic hierarchical porous carbons–high performance materials for amine based CO2 capture and supercapacitor electrode. Energy Environ. Sci. 2013, 6, 1785–1790. [Google Scholar] [CrossRef]
- Schwartz, M.A.; Chen, C.S. Deconstructing dimensionality. Science 2013, 339, 402–404. [Google Scholar] [CrossRef] [PubMed]
- Cukierman, E.; Pankov, R.; Stevens, D.R.; Yamada, K.M. Taking cell-matrix adhesions to the third dimension. Science 2001, 294, 1708–1712. [Google Scholar] [CrossRef] [PubMed]
- Khodagholy, D.; Doublet, T.; Quilichini, P.; Gurfinkel, M.; Leleux, P.; Ghestem, A.; Ismailova, E.; Herve, T.; Sanaur, S.; Bernard, C.; et al. In vivo recordings of brain activity using organic transistors. Nat. Commun. 2013, 4, 1575. [Google Scholar]
- Moczko, E.; Istamboulie, G.; Calas-Blanchard, C.; Rouillon, R.; Noguer, T. Biosensor employing screen-printed PEDOT:PSS for sensitive detection of phenolic compounds in water. J. Polym. Sci. Polym. Chem. A 2012, 50, 2085–2292. [Google Scholar] [CrossRef]
- Snyder-Mackler, L. Electrical Stimulation: Its Role in Growth, Repair and Remolding of the Musculoskeletal System; Jonathan Black/Praaeger: New York, NY, USA, 1997. [Google Scholar]
- Kitchen, S. Electrotherapy: Evidencebased Practice; Churchill Livingstone: Edinburgh, UK, 2002. [Google Scholar]
- Paul, D.H. The Physiology of Nerve Cells; Blackwell Scientific: Philadelphia, PA, USA, 1975. [Google Scholar]
- Mattioli-Belmonte, M.; Giavaresi, G.; Biagini, G.; Virgili, L.; Giacomini, M.; Fini, M.; Giantomassi, F.; Natali, D.; Torricelli, P.; Giardino, R. Tailoring biomaterial compatibility: In vivo tissue response versus in vitro cell behavior. Int. J. Artif. Organs 2003, 26, 1077–1085. [Google Scholar] [PubMed]
- Guarino, V.; Causa, F.; Ambrosio, L. Bioactive scaffolds for bone and ligament tissue. Exp. Rev. Med. Dev. 2007, 4, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Lin, J.; Roy, K. Effect of 3D scaffold and dynamic culture condition on the global gene expression profile of mouse embryonic stem cells. Biomaterials 2006, 27, 5978–5989. [Google Scholar] [CrossRef] [PubMed]
- Mata, A.; Kim, E.J.; Boehm, C.A.; Fleischman, A.J.; Muschler, G.F.; Roy, S. A three-dimensional scaffold with precise micro-architecture and surface micro-textures. Biomaterials 2009, 30, 4610–4617. [Google Scholar] [CrossRef] [PubMed]
- Stout, D.A.; Yoo, J.; Santiago-Miranda, A.N.; Webster, T.J. Mechanisms of greater cardiomyocyte functions on conductive nanoengineered composites for cardiovascular application. Int. J. Nanomed. 2012, 7, 5653–5669. [Google Scholar]
- Peckham, P.H.; Knutson, J.S. Functional electrical stimulation for neuromuscular applications. Annu. Rev. Biomed. Eng. 2005, 7, 327–360. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; He, W.; Huang, J.; Liu, S.; Wu, G.; Teng, W.; Wang, Q.; Dong, Y. Synthesis of water soluble, biodegradable, and electroactive polysaccharide crosslinker with aldehyde and carboxylic groups for biomedical applications. Macromol. Biosci. 2011, 11, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Sirivisoot, S.; Pareta, R.; Harrison, B.S. Protocol and cell responses in three-dimensional conductive collagen gel scaffolds with conductive polymer nanofibres for tissue regeneration. Interf. Focus 2014, 6, 20130050. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, C.W.; Bai, M.Y.; Chang, Y.; Chung, M.F.; Lee, T.Y.; Wu, C.T.; Maiti, B.; Liao, Z.-X.; Li, R.-K.; Sung, H.-W. Electrical coupling of isolated cardiomyocyte clusters grown on aligned conductive nanofibrous meshes for their synchronized beating. Biomaterials 2013, 34, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Holzwarth, J.M.; Yan, Y.; Zheng, H.; Yin, Y.; Li, S.; Ma, P.X. Conductive PPY/PDLLA conduit for peripheral nerve regeneration. Biomaterials 2014, 35, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Guarino, V.; Cirillo, V.; Ambrosio, L. Bicomponent electrospun scaffolds to design extracellular matrix tissue analogs. Exp. Rev. Med. Dev. 2016, 13, 83–102. [Google Scholar] [CrossRef] [PubMed]
- Cen, L.; Neoh, K.G.; Li, Y.; Kang, E.T. Assessment of in vitro bioactivity of hyaluronic acid and sulfated hyaluronic acid functionalized electroactive polymer. Biomacromolecules 2004, 5, 2238–2246. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yue, Z.; Higgins, M.J.; Wallace, G.G. Conducting polymers with immobilised fibrillar collagen for enhanced neural interfacing. Biomaterials 2011, 32, 7309–7317. [Google Scholar] [CrossRef] [PubMed]
- Stauffer, W.R.; Cui, X.T. Polypyrrole doped with 2 peptide sequences from laminin. Biomaterials 2006, 27, 2405–2413. [Google Scholar] [CrossRef] [PubMed]
- Ateh, D.D.; Vadgama, P.; Navsaria, H.A. Culture of human keratinocytes on polypyrrole-based conducting polymers. Tissue Eng. 2006, 12, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Collier, J.H.; Camp, J.P.; Hudson, T.W.; Schmidt, C.E. Synthesis and characterization of polypyrrole hyaluronic acid composite biomaterials for tissue engineering applications. J. Biomed. Mater. Res. 2000, 50, 574–584. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, J.W.; Schmidt, C.E. Neuroactive conducting scaffolds: Nerve growth factor conjugation on active ester-functionalized polypyrrole. J. Royal. Soc. Interface 2008, 6, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Gomez, N.; Schmidt, C.E. Nerve growth factor-immobilized polypyrrole: Bioactive electrically conducting polymer for enhanced neurite extension. J. Biomed. Mater. Res. A 2007, 81, 135–149. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.L.; Glavas, L.; Albertsson, A.C. Biodegradable and electrically conducting polymers for biomedical applications. Prog. Polym. Sci. 2013, 38, 1263–1286. [Google Scholar] [CrossRef]
- Liu, Y.D.; Hu, J.; Zhuang, X.L.; Zhang, P.B.; Wei, Y.; Wang, X.H.; Chen, X.S. Synthesis and characterization of novel biodegradable and electroactive hydrogel based on aniline oligomer and gelatin. Macromol. Biosci. 2011, 12, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Li, L.C.; Ge, J.; Guo, B.L.; Ma, P.X. In situ forming biodegradable electroactive hydrogels. Polym. Chem. 2014, 5, 2880–2890. [Google Scholar] [CrossRef]
- Gil, S.; Mano, J.F. Magnetic composite biomaterials for tissue engineering. Biomater. Sci. 2014, 2, 812–818. [Google Scholar] [CrossRef]
- Fernández-Oliveras, A.; Rubiño, M.; Pérez, M.M. Scattering and absorption properties of biomaterials for dental restorative applications. J. Eur. Opt. Soc. 2013, 8, 13056. [Google Scholar] [CrossRef]
- Ribeiro, C.; Sencadas, V.; Correia, D.M.; Lanceros-Méndez, S. Piezoelectric polymers as biomaterials for tissue engineering applications. Coll. Surf. B 2015, 136, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Iannotti, V.; Ausanio, G.; Lanotte, L.; Lanotte, L. Magneto-piezoresistivity in iron particle-filled silicone: An alternative outlook for reading magnetic field intensity and direction. eXPRESS Polym. Lett. 2016, 10, 65–71. [Google Scholar] [CrossRef]
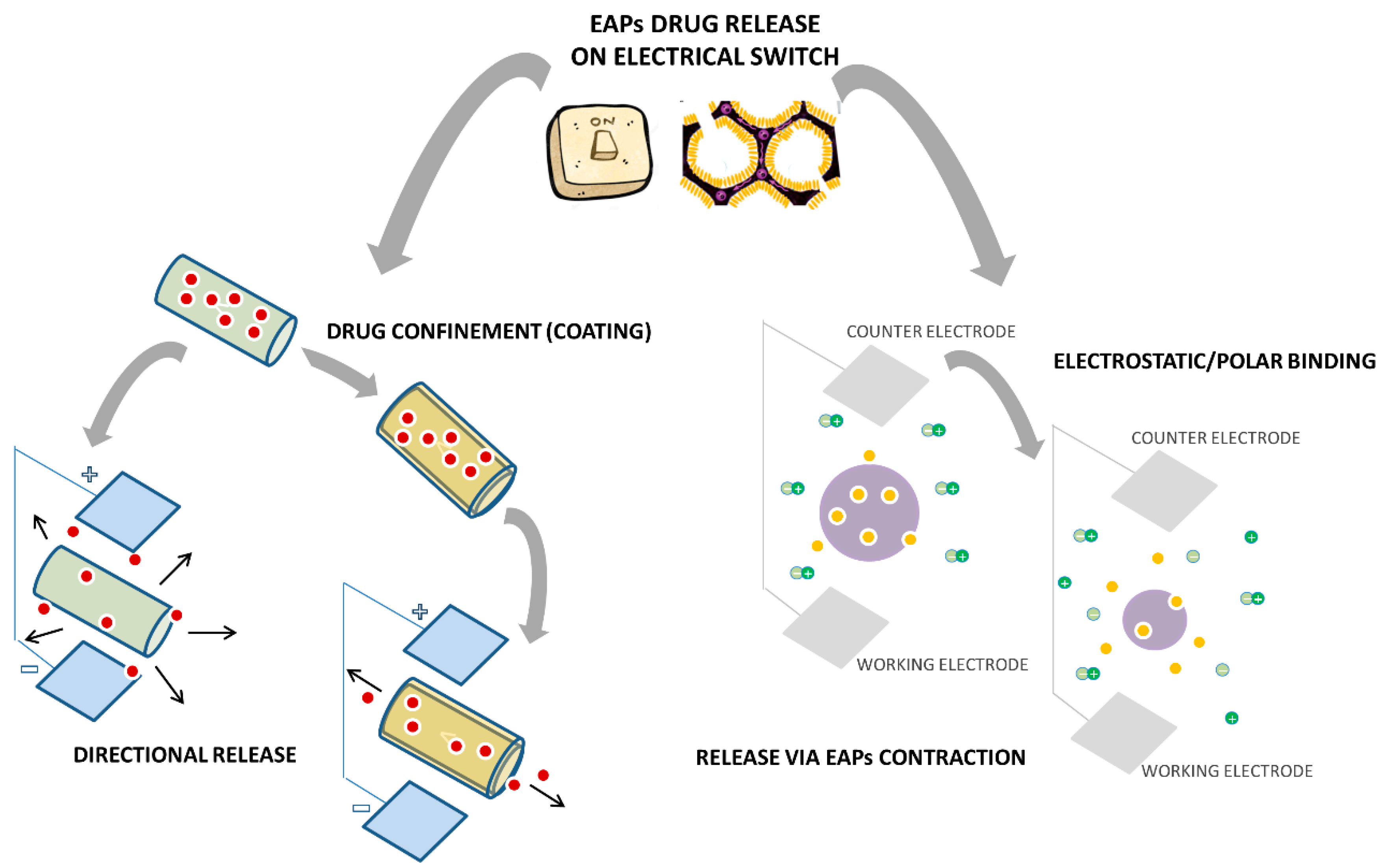
- Stassen, I.; Sloboda, T.; Hambitzer, G. Membrane with controllable permeability for drugs. Synth. Met. 1995, 71, 2243–2244. [Google Scholar] [CrossRef]
- Bartlett, P.N.; Whitaker, R.G. Modified electrode surface in amperometric biosensors. Med. Biol. Eng. Comput. 1988, 28, 10–17. [Google Scholar] [CrossRef]
- Pernaut, J.M.; Reynolds, J.R. Use of conducting electroactive polymers for drug delivery and sensing of bioactive molecules. A redox chemistry approach. J. Phys. Chem. B 2000, 104, 4080–4090. [Google Scholar] [CrossRef]
- Wadhwa, R.; Lagenaur, C.F.; Cui, X.T. Electrochemically controlled release of dexamethasone from conducting polymer polypyrrole coated electrode. J. Control Release 2006, 110, 531–541. [Google Scholar] [CrossRef] [PubMed]
- George, P.M.; Lyckman, A.W.; LaVan, D.A.; Hegde, A.; Leung, Y.; Avasare, R.; Testa, C.; Alexander, P.M.; Langer, R.; Sur, M. Fabrication and biocompatibility of polypyrrole implants suitable for neural prosthetics. Biomaterials 2005, 26, 3511–3519. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Cui, X.T. Electrochemically controlled release based on nanoporous conducting polymers. Electrochem. Commun. 2009, 11, 402–404. [Google Scholar]
- Hodgson, A.J.; John, M.J.; Campbell, T.; Georgevich, A.; Woodhouse, S.; Aoki, T. Integration of biocomponents with synthetic structures: Use of conducting polymer polyelectrolyte composites. Proc. SPIE Int. Soc. Opt. Eng. 1996, 2716, 164–176. [Google Scholar]
- Li, Y.; Neoh, K.G.; Cen, L.; Kang, E.T. Controlled release of heparin from polypyrrole-poly(vinyl alcohol) assembly by electrical stimulation. J. Biomed. Mater. Res. A 2005, 73A, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.Y.; Hetke, J.F.; Wiler, J.A.; Anderson, D.J.; Martin, D.C. Electrochemical deposition and characterization of poly(3,4-ethylenedioxythiophene) on neural microelectrode arrays. Sens. Actuators A 2001, 89, 92–102. [Google Scholar] [CrossRef]
- Kim, D.-H.; Richardson-Burns, S.M.; Hendricks, J.L.; Sequera, C.; Martin, D.C. Effect of immobilized nerve growth factor on conductive polymers: Electrical properties and cellular response. Adv. Funct. Mater. 2007, 17, 79–86. [Google Scholar] [CrossRef]
- Abidian, M.R.; Kim, D.-H.; Martin, D.C. Conducting-polymer nanotubes for controlled drug release. Adv. Mater. 2006, 17, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.H.; Lira, L.M.; Córdoba de Torresi, S.I.J. Zero-order release profiles from a multistimuli responsive electro-conductive hydrogel. Biomater. Nanobiotech. 2012, 3, 262–268. [Google Scholar] [CrossRef]
- Guo, B.L.; Finne-Wistrand, A.; Albertsson, A.C. Degradable and electroactive hydrogels with tunable electrical conductivity and swelling behavior. Chem. Mater. 2011, 23, 1254–1262. [Google Scholar] [CrossRef]
- Justin, G.A.; Zhu, S.; Nicholson, T.R.; Maskrod, J.; Mbugua, J.; Chase, M.; Jung, J.-H.; Mercado, R.M.L. On-demand controlled release of anti-inflammatory and analgesic drugs from conducting polymer films to aid in wound healing. In Proceedings of the IEEE International Conference on Engineering Medicine and Biology Society, San Diego, CA, USA, 28 September 2012; pp. 1206–1209.
- Kamaly, N.; Yameen, B.; Wu, J.; Farokhzad, O.C. Degradable controlled-release polymers and polymeric nanoparticles: Mechanisms of controlling drug release. Chem. Rev. 2016, 116, 2602–2663. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, T.; Mir, I.A.; Kumar, D. Biomolecular immobilization on conducting polymers for biosensing applications. Biosens. Bioelectron. 2007, 28, 791–805. [Google Scholar] [CrossRef] [PubMed]
- Gerard, M.; Chaubey, A.; Malhotra, B.D. Application of conducting polymers to biosensors. Biosens. Bioelectron. 2002, 17, 345–359. [Google Scholar] [CrossRef]
- Jha, S.; Kanungo, M.; D’souze, S.F. Entrapment of live microbial cells in electropolymerized polyaniline and their use as urea biosensor. Biosen. Bioelectron. 2009, 24, 2637–2642. [Google Scholar] [CrossRef] [PubMed]
- Vidal, J.-C.; Garcia-Ruiz, E.; Castillo, J.-R. Recent advances in electropolymerized conducting polymers in amperometric biosensors. Microchim. Acta 2003, 143, 93–111. [Google Scholar] [CrossRef]
- Ramanavičiusa, A.; Ramanavičienėa, A.; Malinauskasc, A. Electrochemical sensors based on conducting polymer-polypyrrole. Electrochim. Acta 2006, 51, 6025–6037. [Google Scholar] [CrossRef]
- Richardson-Burns, S.M.; Hendricks, J.L.; Foster, B.; Povlich, L.K.; Kim, D.-H.; Martin, D.C. Polymerization of the conducting polymer poly(3,4-ethylenedioxythiophene) (PEDOT) around living neural cells. Biomaterials 2007, 28, 1539–1552. [Google Scholar] [CrossRef] [PubMed]
- Centonze, D.; Malitesta, C.; Palmisano, F.; Zambonin, P.G. Permeation of solutes through an electropolymerized ultrathin poly-O-phenylenediamine film used as an enzyme-entrapping membrane. Electroanalysis 1994, 6, 423–429. [Google Scholar] [CrossRef]
- Cosnier, S. Biosensors based on electropolymerized films: New trends. Anal. Bioanal. Chem. 2003, 377, 507–520. [Google Scholar] [CrossRef] [PubMed]
- Cosnier, S. Biomolecule immobilization on electrode surfaces by entrapment or attachment to electrochemically polymerized films. A review. Biosens. Bioelectron. 1999, 14, 443–456. [Google Scholar] [CrossRef]
- Palmisano, F.; Centonze, D.; Malitesta, C.; Zambonin, P.G. Correlation between permselectivity and chemical structure of overoxidized polypyrrole membranes used in electroproduced enzyme biosensors. Anal. Chem. 1995, 67, 2207–2211. [Google Scholar] [CrossRef]
- Cosnier, S.; Galland, B.; Gondran, C.; Le Pellec, A. Electrogeneration of biotinylated functionalized polypyrroles for the simple immobilization of enzymes. Electroanalysis 1998, 10, 808–813. [Google Scholar] [CrossRef]
- Park, J.; Kim, H.K.; Son, Y. Glucose biosensor constructed from capped conducting microtubules of PEDOT. Sens. Actuators B 2008, 133, 244–250. [Google Scholar] [CrossRef]
- Santhosh, P.; Manesh, K.M.; Uthayakumar, S.; Komathi, S.; Gopalan, A.I.; Lee, K.-P. Fabrication of enzymatic glucose biosensor based on palladium nanoparticles dispersed onto poly(3,4-ethylenedioxythiophene) nanofibers. Bioelectrochemistry 2009, 75, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Njagi, J.; Andreescu, S. Stable enzyme biosensors based on chemically synthesized Au–polypyrrole nanocomposites. Biosens. Bioelectron. 2007, 23, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Wisniewski, N.; Reichert, M. Methods for reducing biosensor membrane biofouling. Coll. Surface B 2000, 18, 18197–18219. [Google Scholar] [CrossRef]
- Quinn, C.A.P.; Connor, R.E.; Heller, A. Biocompatible, glucose-permeable hydrogel for in situ coating of implantable biosensors. Biomaterials 1997, 18, 1665–1670. [Google Scholar] [CrossRef]
- Guiseppi-Elie, A.; Lei, C.; Baughman, R.H. Direct electron transfer of glucose oxidase on carbon nanotubes. Nanotechnology 2002, 13, 559–564. [Google Scholar] [CrossRef]
- Paleček, E.; Jelen, F. Electrochemistry of nucleic acids and development of DNA sensors development of DNA sensors. Crit. Rev. Anal. Chem. 2002, 32, 261–270. [Google Scholar] [CrossRef]
- Lee, T.-Y.; Shim, Y.-B. Direct DNA hybridization detection based on the oligonucleotide-functionalized conductive polymer. Anal. Chem. 2001, 73, 5629–5632. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Soeller, C.; Vigar, N.; Kilmartin, P.A.; Cannell, M.B.; Bowmaker, G.A.; Cooney, R.P.; Travas-Sejdic, J. Label-free electrochemical DNA sensor based on functionalised conducting copolymer. Biosens. Bioelectron. 2005, 20, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Soeller, C.; Travas-Sejdic, J. Novel conducting polymers for DNA sensing. Macromolecules 2007, 40, 909–914. [Google Scholar] [CrossRef]
- Weng, C.-H.; Yeh, W.-M.; Ho, K.-C.; Lee, G.-B. A microfluidic system utilizing molecularly imprinted polymer films for amperometric detection of morphine. Sens. Actuators B 2007, 121, 576–582. [Google Scholar] [CrossRef]
- Sirivisoot, S.; Pareta, R.A.; Webster, T.J. A conductive nanostructured polymer electrodeposited on titanium as a controllable, local drug delivery platform. J. Biomed. Mater. Res. Part A 2011, 99, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Löffler, S.; Richter-Dahlfors, A. Phase angle spectroscopy on transparent conducting polymer electrodes for real-time measurement of epithelial barrier integrity. J. Mater. Chem. B 2015, 3, 4997–5000. [Google Scholar] [CrossRef]

| Conducting polymers | Maximum conductivity range | References | |
|---|---|---|---|
| Poly(acetylene) (PAc) |  | 102–103 S∙cm−1 | [6,11,15,39] |
| Poly(p-vinylene) (PPV) | 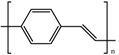 | ||
| Poly(p-phenylene) (PPP) | 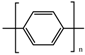 | 1–102 S∙cm−1 | [6,11,12,15,39] |
| Poly(p-phenylene sulphide) (PPS) |  | ||
| Polypyrrole (PPy) | 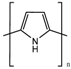 | ||
| Polyaniline (PANI) |  | ||
| Polythiophene (PTh) | 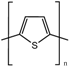 | ||
| Poly(3,4-ethylenedioxythiophene) (PEDOT) | 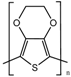 | 10−1–10 S∙cm−1 | [12,39,40] |
| Poly(isothianaphtene) (PITN) | 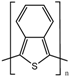 | ||
| EAPs | Key properties | Limitations | Main biomedical applications |
|---|---|---|---|
| PPy | High conductivity | Insolubility in organic | Neural probes |
| High stability in air | solvents | Drug delivery | |
| Electroactivity pH [4,5,6,7,8,9,10,11] | Poor processability | Coatings | |
| Biocompatibility | No biodegradability | Tissue engineering | |
| Low solubility in water | Biosensing | ||
| Bio-actuators | |||
| PANI | High conductivity | Low solubility in organic solvents | Drug delivery |
| Enviromental stability | Electroactivity pH < 4 Poor processability | Coatings | |
| Suitable redox properties | No biodegradability | Tissue engineering | |
| Biocompatibility | Bio-actuators | ||
| PTh | High conductivity | Instability in air | Tissue engineering |
| Biocompatibility | Low solubility in organic solvents | Biosensing | |
| High doping levels | Poor processability | Coatings | |
| No biodegradability | |||
| PEDOT | High transparency | Low solubility in organic solvents | Biosensing field |
| High stability in its oxidation state vs. biological reducing agent | Poor processability | Neural probes | |
| Low oxidation potential | No biodegradability | Drug delivery | |
| High compatibility with aqueous electrolytes |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarino, V.; Zuppolini, S.; Borriello, A.; Ambrosio, L. Electro-Active Polymers (EAPs): A Promising Route to Design Bio-Organic/Bioinspired Platforms with on Demand Functionalities. Polymers 2016, 8, 185. https://doi.org/10.3390/polym8050185
Guarino V, Zuppolini S, Borriello A, Ambrosio L. Electro-Active Polymers (EAPs): A Promising Route to Design Bio-Organic/Bioinspired Platforms with on Demand Functionalities. Polymers. 2016; 8(5):185. https://doi.org/10.3390/polym8050185
Chicago/Turabian StyleGuarino, Vincenzo, Simona Zuppolini, Anna Borriello, and Luigi Ambrosio. 2016. "Electro-Active Polymers (EAPs): A Promising Route to Design Bio-Organic/Bioinspired Platforms with on Demand Functionalities" Polymers 8, no. 5: 185. https://doi.org/10.3390/polym8050185
APA StyleGuarino, V., Zuppolini, S., Borriello, A., & Ambrosio, L. (2016). Electro-Active Polymers (EAPs): A Promising Route to Design Bio-Organic/Bioinspired Platforms with on Demand Functionalities. Polymers, 8(5), 185. https://doi.org/10.3390/polym8050185