Aromatic Copolyamides with Anthrazoline Units in the Backbone: Synthesis, Characterization, Pervaporation Application
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
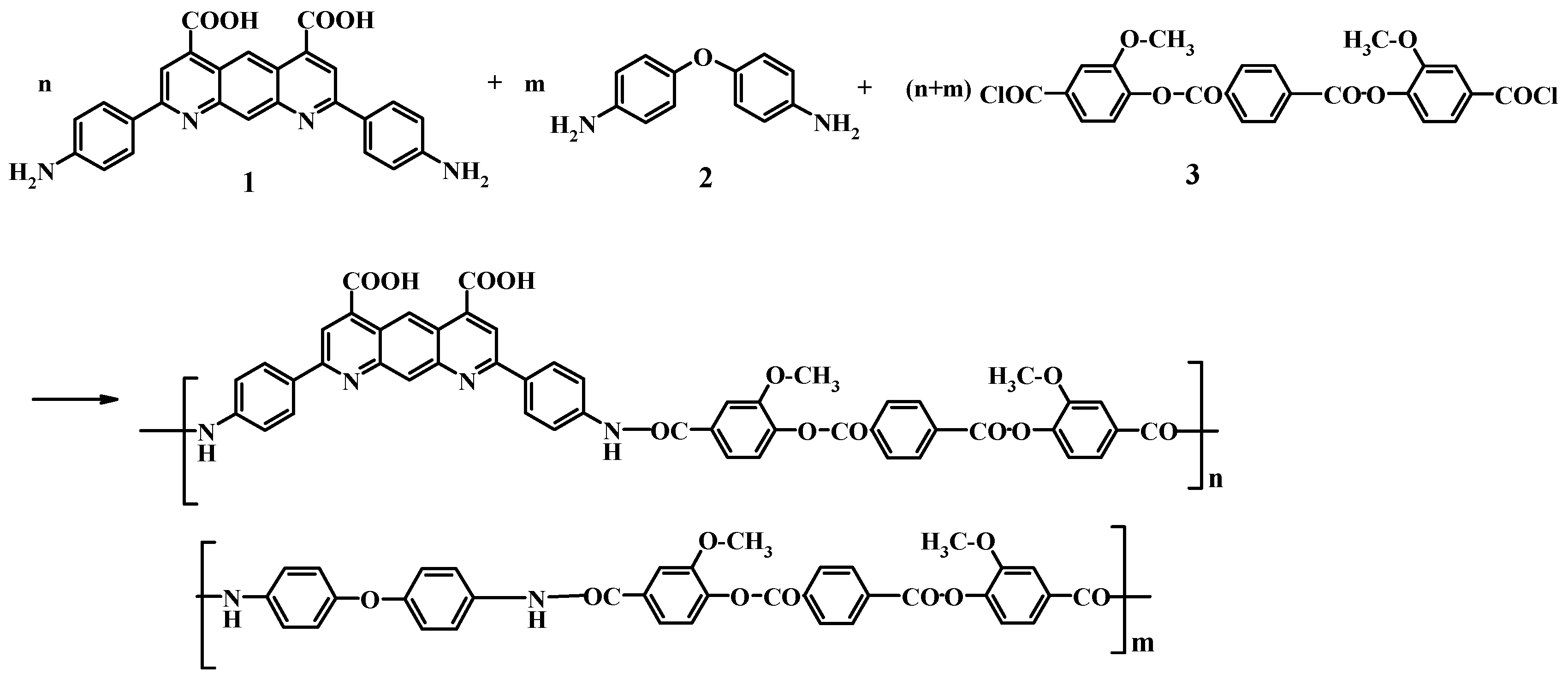
2.2. Polymer Synthesis
2.3. Film Preparation
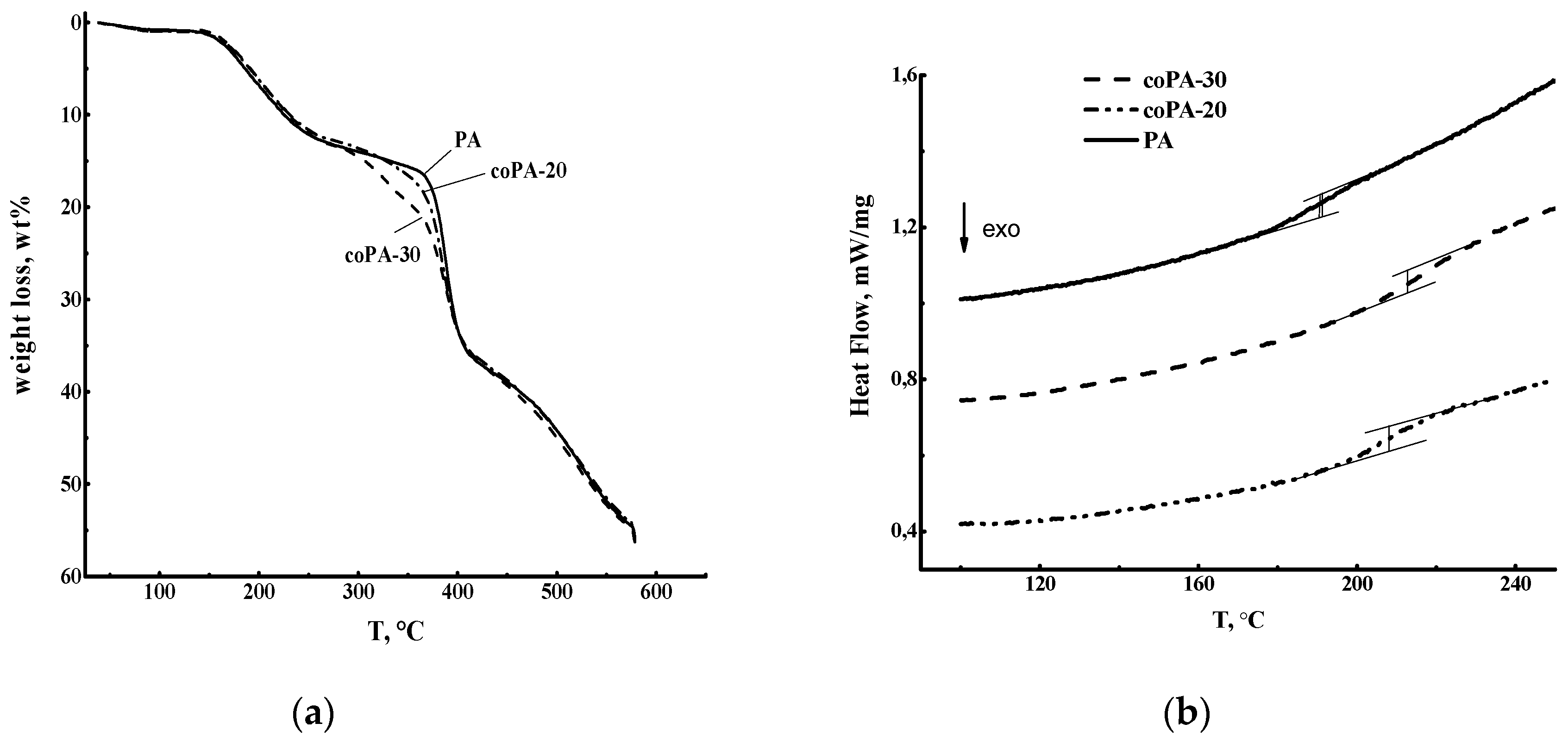
2.4. Thermogravimetric Analysis (TGA)
2.5. Scanning Electron Microscopy
2.6. Physical Properties
2.7. Sorption Experiment
2.8. Pervaporation
3. Results and Discussion
3.1. Physical and Thermal Properties
3.2. Films Structure
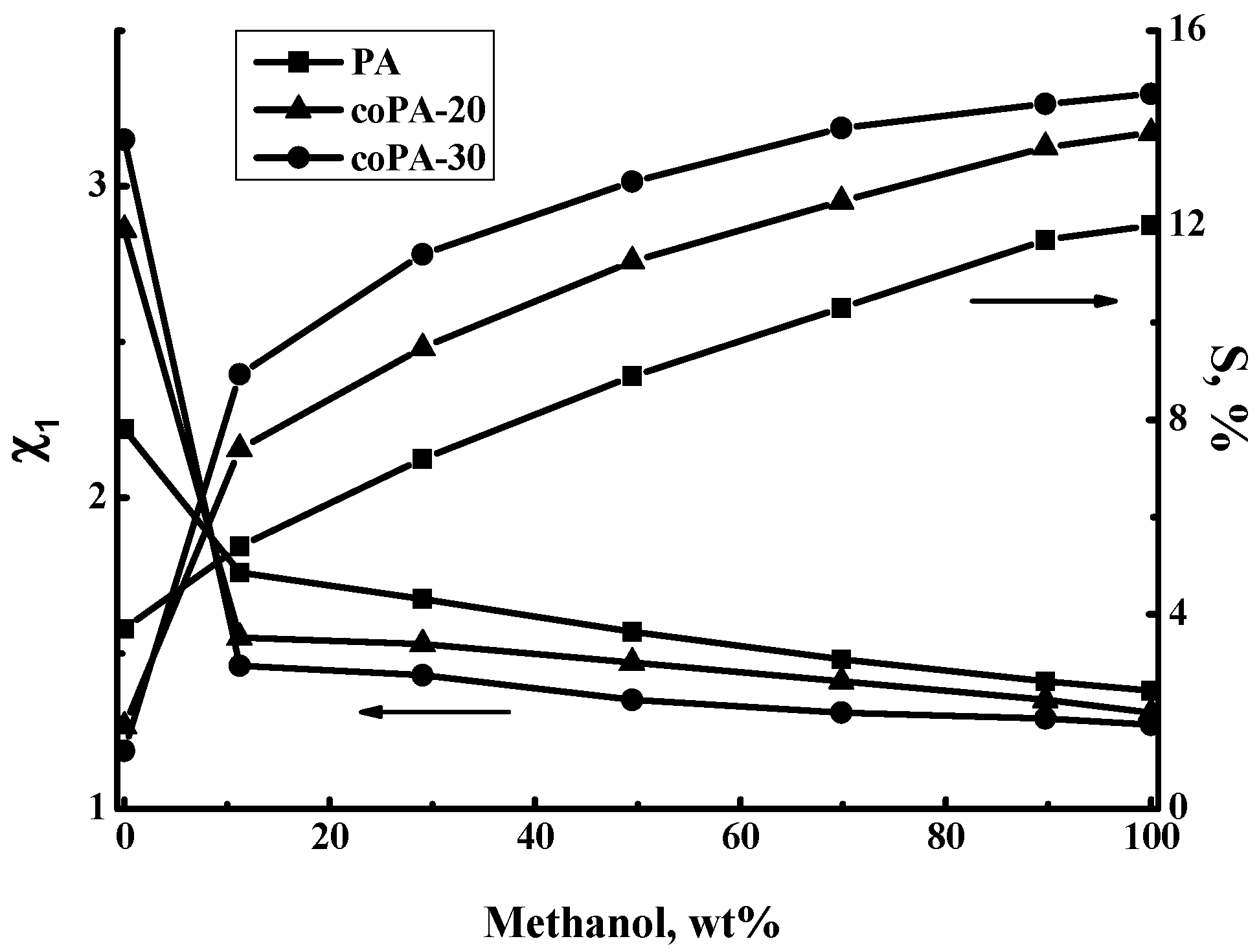
3.3. Sorption Study
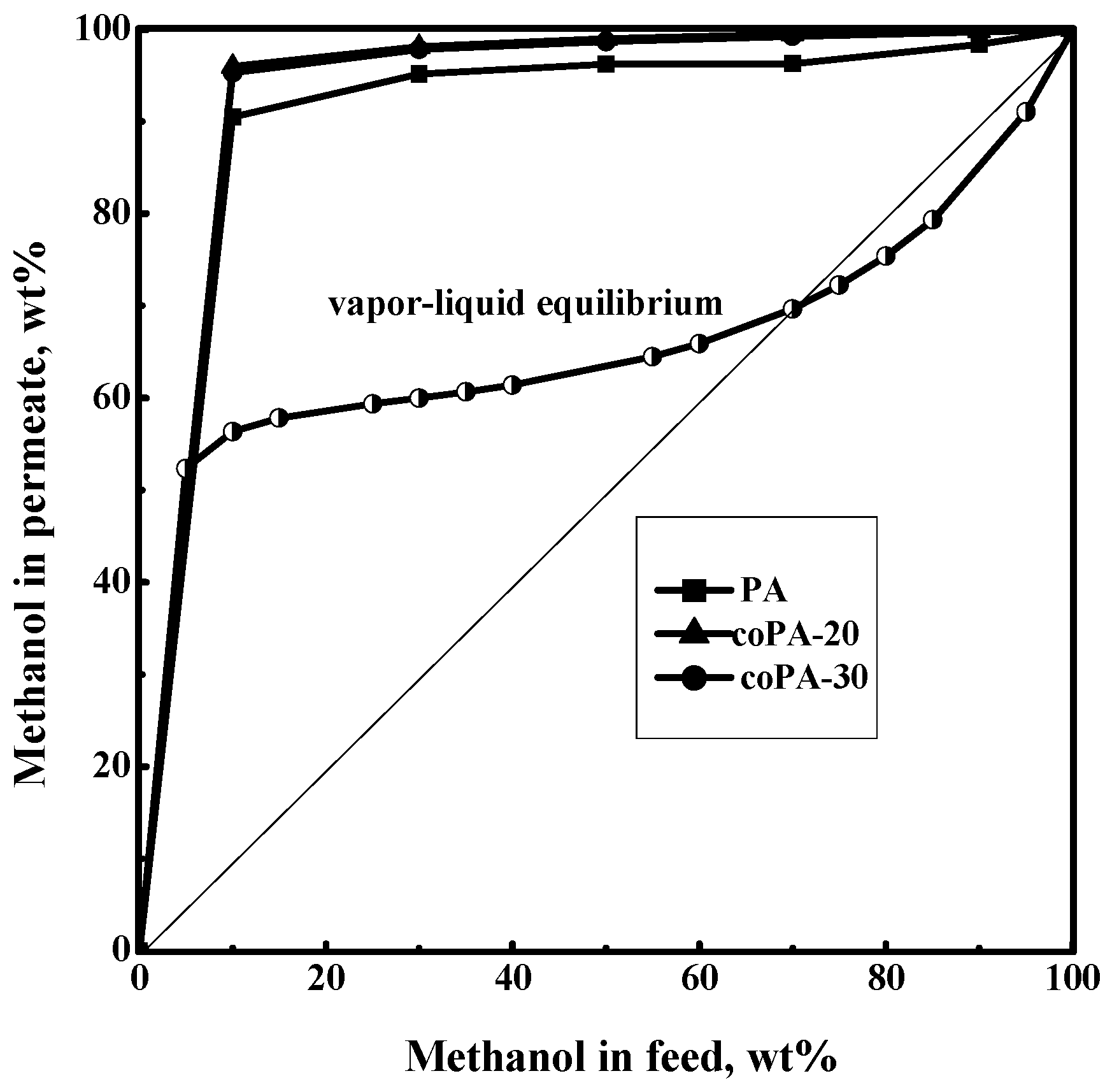
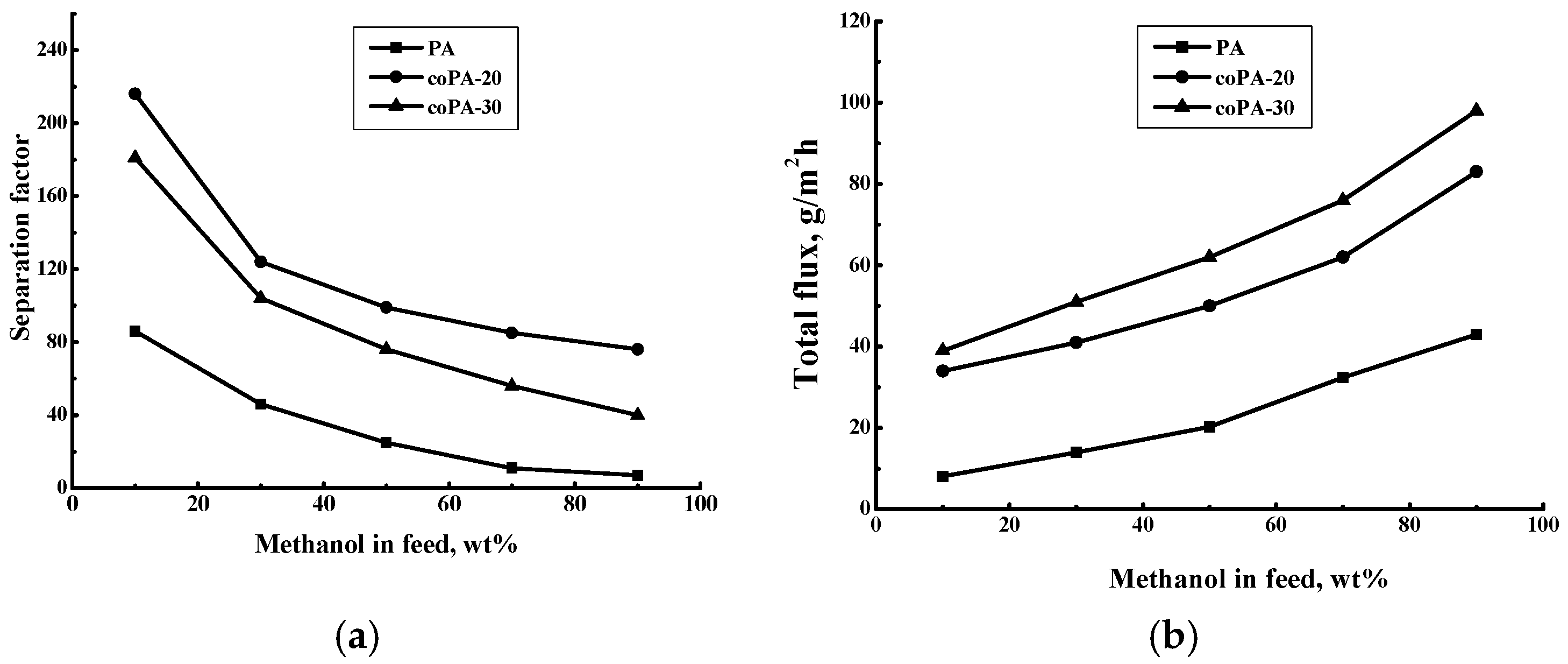
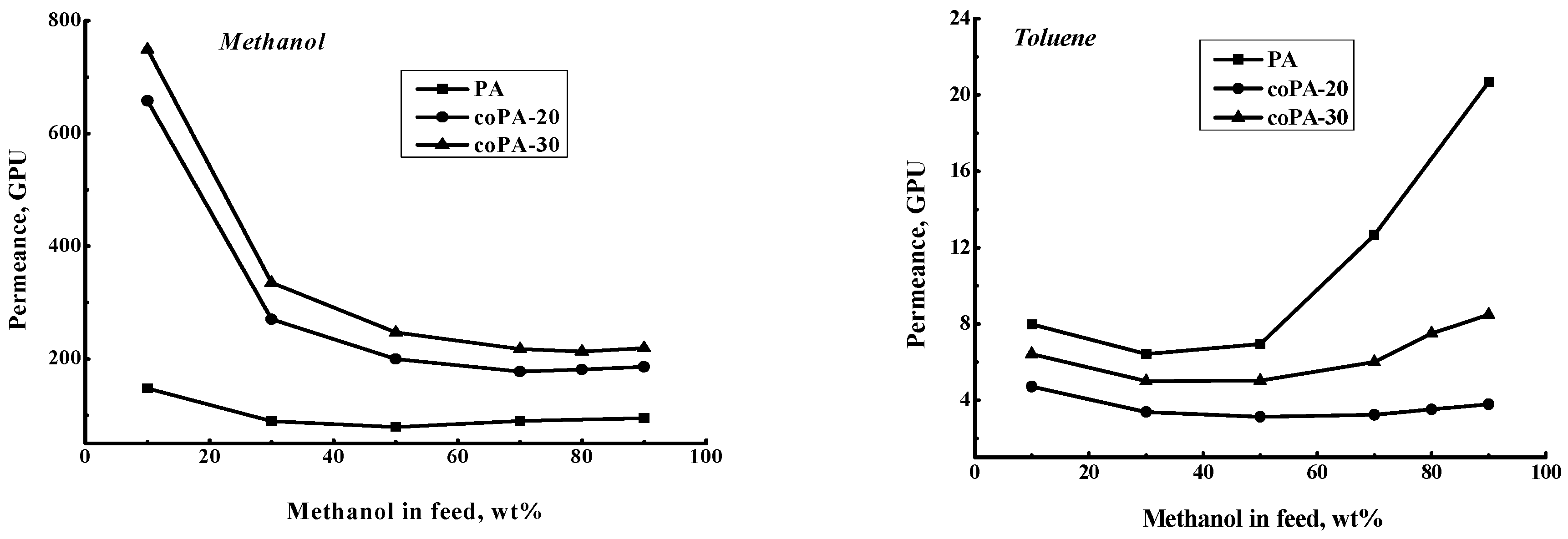
3.4. Pervaporation of Methanol–Toluene Mixture
3.5. Comparison of Pervaporation Properties with Literature Data
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Liaw, D.-J.; Wang, K.-L.; Huang, Y.-C.; Lee, K.-R.; Lai, J.-Y.; Ha, C.-S. Advanced polyimide materials: Syntheses, physical properties and applications. Prog. Polym. Sci. 2012, 37, 907–974. [Google Scholar] [CrossRef]
- Bessonov, M.I.; Koton, M.M.; Kudryavtsev, V.V.; Laius, L.A. Polyimides—Thermally Stable Polymers; Plenum: New York, NY, USA, 1987; p. 318. [Google Scholar]
- Ohya, H.; Kudryavtsev, V.V.; Semenova, S.I. Polyimide Membranes. Applications, Fabrications and Properties; Kodansha Ltd.: Tokyo, Japan, 1996; p. 328. [Google Scholar]
- Pulyalina, A.Y.; Polotskaya, G.A.; Veremeychik, K.Y.; Goikhman, M.Y.; Podeshvo, I.V.; Toikka, A.M. Ethanol purification from methanol via pervaporation using polybenzoxazinoneimide membrane. Fuel Process. Technol. 2015, 139, 178–185. [Google Scholar] [CrossRef]
- Miki, M.; Horiuchi, H.; Yamada, Y. Synthesis and gas transport properties of hyperbranched polyimide–silica hybrid/composite membranes. Polymers 2013, 5, 1362–1379. [Google Scholar] [CrossRef]
- Yampolskii, Y.; Pinnau, I.; Freeman, B. Materials Science of Membranes for Gas and Vapor Separation; Wiley: Hoboken, NJ, USA, 2006; p. 466. [Google Scholar]
- Wang, Y.; Shang, Y.; Li, X.; Tian, T.; Gao, L.; Jiang, L. Fabrication of CO2 facilitated transport channels in block copolymer through supramolecular assembly. Polymers 2014, 6, 1403–1413. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Kim, J.-H.; Lee, S.-B.; Rhee, H.-W. Polysiloxaneimide membranes for removal of VOCs from water by pervaporation. J. Appl. Polym. Sci. 2000, 77, 2691–2702. [Google Scholar] [CrossRef]
- Song, Z.W.; Zhu, J.M.; Jiang, L.Y. Novel polysiloxaneimide/polyetherimide/non-woven fabric composite membranes for organophilic pervaporation. J. Membr. Sci. 2014, 472, 77–90. [Google Scholar] [CrossRef]
- Konietzny, R.; Barth, C.; Harms, S.; Raetzke, K.; Koelsch, P.; Staudt, C. Structural investigations and swelling behavior of 6FDA copolyimide thin films. Polym. Int. 2011, 60, 1670–1678. [Google Scholar] [CrossRef]
- Chang, B.-J.; Chang, Y.-H.; Kim, D.-K.; Kim, J.-H.; Lee, S.-B. New copolyimide membranes for the pervaporation of trichloroethylene from water. J. Membr. Sci. 2005, 248, 99–107. [Google Scholar] [CrossRef]
- Pulyalina, A.; Polotskaya, G.; Goikhman, M.; Podeshvo, I.; Kalyuzhnaya, L.; Chislov, M.; Toikka, A. Study on polybenzoxazinone membrane in pervaporation processes. J. Appl. Polym. Sci. 2013, 130, 4024–4031. [Google Scholar] [CrossRef]
- Le, N.L.; Wang, Y.; Chung, T.S. Synthesis, cross-linking modifications of 6FDA-NDA/DABA polyimide membranes for ethanol dehydration via pervaporation. J. Membr. Sci. 2012, 415–416, 109–121. [Google Scholar] [CrossRef]
- Pientka, Z.; Brozova, L.; Pulyalina, A.Y.; Goikhman, M.Y.; Podeshvo, I.V.; Gofman, I.V.; Saprykina, N.N.; Polotskaya, G.A. Synthesis and characterization of polybenzoxazinone and its prepolymer using gas separation. Macromol. Chem. Phys. 2013, 214, 2867–2874. [Google Scholar] [CrossRef]
- Pulyalina, A.Y.; Polotskaya, G.A.; Kalyuzhnaya, L.M.; Sushchenko, I.G.; Meleshko, T.K.; Yakimanskii, A.V.; Chislov, M.V.; Toikka, A.M. Sorption and transport of aqueous isopropanol solutions in polyimide-poly(aniline-co-anthranilic acid) composites. Russ. J. Appl. Chem. 2011, 84, 840–846. [Google Scholar] [CrossRef]
- Babalou, A.A.; Rafia, N.; Ghasemzadeh, K. Fundamentals of membrane distillation. In Pervaporation, Vapour Permeation and Membrane Distillation; Elsevier Ltd.: Amsterdam, The Netherlands, 2015; pp. 277–316. [Google Scholar]
- Baker, R.W. Pervaporation. In Membrane Technology and Applications; Wiley: Hoboken, NJ, USA, 2012; pp. 379–416. [Google Scholar]
- Adoor, S.G.; Manjeshwar, L.S.; Naidu, B.V.K.; Sairam, M.; Aminabhavi, T.M. Poly(vinyl alcohol)/poly(methyl methacrylate) blend membranes for pervaporation separation of water + isopropanol and water + 1,4-dioxane mixtures. J. Membr. Sci. 2006, 280, 594–602. [Google Scholar] [CrossRef]
- Smitha, B.; Suhanya, D.; Sridhar, S.; Ramakrishna, M. Separation of organic-organic mixtures by pervaporation–A review. J. Membr. Sci. 2004, 241, 1–21. [Google Scholar] [CrossRef]
- Bracke, W. Polymers with anthrazoline units in the main chain. Macromolecules 1969, 3, 286–289. [Google Scholar] [CrossRef]
- Higgins, J.; Janović, Z. Polyanthrazolines. J. Polym. Sci. Part B Polym. Lett. 1972, 10, 357–360. [Google Scholar] [CrossRef]
- Imai, Y.; Johnson, E.F.; Katto, T.; Kurihara, M.; Stille, J.K. Synthesis of aromatic polymers containing anthrazoline and isoanthrazoline units. J. Polym. Sci. Polym. Chem. Ed. 1975, 13, 2233–2249. [Google Scholar] [CrossRef]
- Agrawal, A.; Jenekhe, S. Electrochemical Properties and Electronic Structures of Conjugated Polyquinolines and Polyanthrazolines. Chem. Mater. 1996, 8, 579–589. [Google Scholar] [CrossRef]
- Morrison, R.T.; Boyd, R.N. Alcohols. Production and Physical Properties. In Organic Chemistry; Prentice Hall: Bergen County, NJ, USA, 1992; pp. 476–500. [Google Scholar]
- Renge, V.C.; Meshram, A.K.; Badukale, P.K. Separation of methanol & toluene from spent toluene and methanol mixture azeotropically. IJCPS J. 2013, 2, 19–25. [Google Scholar]
- Avagimova, N.; Polotskaya, G.; Saprykina, N.; Toikka, A.; Pientka, Z. Mixed matrix membranes based on polyamide/montmorillonite for pervaporation of methanol–toluene mixture. Sep. Sci. Technol. 2013, 48, 2513–2523. [Google Scholar] [CrossRef]
- Polotskaya, G.A.; Meleshko, T.K.; Sushchenko, I.G.; Yakimansky, A.V.; Pulyalina, A.Y.; Toikka, A.M.; Pientka, Z. Membranes based on polyimide-polyaniline nanocomposites for pervaporation of organic mixtures. J. Appl. Polym. Sci. 2010, 117, 2175–2182. [Google Scholar] [CrossRef]
- Khayet, M.; Villaluenga, J.P.G.; Godino, M.P.; Mengual, J.I.; Seoane, B.; Khulbe, K.C.; Matsuura, T. Preparation and application of dense poly(phenylene oxide) membranes in pervaporation. J. Colloid Interface Sci. 2004, 278, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.A.; Pangarkar, V.G. Methanol-selective membranes for the pervaporative separation of methanol-toluene mixtures. J. Membr. Sci. 2000, 167, 187–201. [Google Scholar] [CrossRef]
- Mandal, S.; Pangarkar, V.G. Separation of methanol-benzene and methanol-toluene mixtures by pervaporation: Effects of thermodynamics and structural phenomenon. J. Membr. Sci. 2002, 201, 175–190. [Google Scholar] [CrossRef]
- Park, H.C.; Meertens, R.M.; Muldef, M.H.V. Pervaporation of alcohol-toluene mixtures through polymer blend membranes of poly(acrylic acid) and poly(vinyl alcohol). J. Membr. Sci. 1994, 90, 265–274. [Google Scholar] [CrossRef]
- Singha, N.R.; Kuila, S.B.; Das, P.; Ray, S.K. Separation of toluene-methanol mixtures by pervaporation using crosslink IPN membranes. Chem. Eng. Process. Process Intensif. 2009, 48, 1560–1565. [Google Scholar] [CrossRef]
- Huang, R.Y.M.; Moon, G.Y.; Pal, R. N-acetylated chitosan membranes for the pervaporation separation of alcohol/toluene mixtures. J. Membr. Sci. 2000, 176, 223–231. [Google Scholar] [CrossRef]
- Tang, J.; Sirkar, K.K.; Majumdar, S. Permeation and sorption of organic solvents and separation of their mixtures through an amorphous perfluoropolymer membrane in pervaporation. J. Membr. Sci. 2013, 447, 345–354. [Google Scholar] [CrossRef]
- Garg, P.; Singh, R.P.; Choudhary, V. Selective polydimethylsiloxane/polyimide blended IPN pervaporation membrane for methanol/toluene azeotrope separation. Sep. Purif. Technol. 2011, 76, 407–418. [Google Scholar] [CrossRef]
- Goikhman, M.; Podeshvo, I.; Mikhailov, G.; Baklagina, Y.; Kudryavtsev, V.; Lukasov, S.; Lebedeva, M.; Bobrova, N.; Sazanov, Y.; Fedorova, G.; et al. A new bifunctional monomer, terephthaloylbis(3-methoxy-4-oxybenzoyl) dichloride, and polyamides prepared from it. Russ. J. Appl. Chem. 1997, 70, 1793–1797. [Google Scholar]
- Imaji, M.; Takeda, H.; Kawada, M.; Nokami, J.; Tsuji, J. Facile transformation of substituted allyl malonates to monocarboxylic acids and esters by the reaction with ammonium formate catalyzed by palladium complexes. Org. Chem. 1989, 54, 5395–5397. [Google Scholar]
- Bondi, A. Catalog of Molecular propertiies. In Physical Properties of Molecular Crystals, Liquids and Glasses; Wiley: New York, NY, USA, 1968; p. 485. [Google Scholar]
- Askadskii, A.A. Transfer in polymers and solubility. In Chemical Structure and Physical Properties of Polymers; Galitskaya, L.I., Ed.; Chemistry: Moscow, Russia, 1983; pp. 215–243. (In Russian) [Google Scholar]
- Barton, A.F.M. CRC Handbook of Solubility Parameters and Other Cohesion Parameters; CRC Press: Boka Raton, FL, USA, 1991; p. 768. [Google Scholar]
- Wolińska–Grabczyk, A. Transport of liquid hydrocarbons in the polyurethane–based membranes. J. Membr. Sci. 2007, 302, 59–69. [Google Scholar] [CrossRef]
- Crank, J.; Park, G.S. Diffusion in Polymers; Academic Press: New York, NY, USA, 1968; p. 452. [Google Scholar]
- Tager, A.A. Theory of polymer solutions. In Physical Chemistry of Polymers, 4th ed.; Rogailina, L.A., Ed.; Nauchnyi Mir: Moscow, Russia, 2007; pp. 379–406. (In Russian) [Google Scholar]
- Polotskaya, G.A.; Pulyalina, A.Y.; Rostovtseva, V.A.; Toikka, A.M.; Saprykina, N.N.; Vinogradova, L.V. Effect of polystyrene stars with fullerene C60 cores on pervaporation properties of poly(phenylene oxide) membrane. Polym. Int. 2016, 65, 407–414. [Google Scholar] [CrossRef]
- Mulder, M. Introduction. In Basic Principles of Membrane Technology; Springer: Dordrecht, The Netherlands, 1996; pp. 1–21. [Google Scholar]
- Koter, S.; Kujawska, A.; Kujawski, W. Modeling of transport and separation in a thermopervaporation process. J. Membr. Sci. 2015, 480, 129–138. [Google Scholar] [CrossRef]
- Baker, R.W.; Wijmans, J.G.; Huang, Y. Permeability, permeance and selectivity: A preferred way of reporting pervaporation performance data. J. Membr. Sci. 2010, 348, 346–352. [Google Scholar] [CrossRef]
- Naidu, B.V.K.; Aminabhavi, T.M. Pervaporation separation of water/2-propanol mixtures by use of the blend membranes of sodium alginate and (hydroxyethyl)cellulose: Roles of permeate−membrane interactions, zeolite filling, and membrane swelling. Ind. Eng. Chem. Res. 2005, 44, 7481–7489. [Google Scholar] [CrossRef]
- Chovau, S.; Van der Bruggen, B.; Luis, P. Application of the mass-based UNIQUAC model to membrane systems: A critical revision. J. Chem. Thermodyn. 2012, 48, 260–266. [Google Scholar] [CrossRef]
- Adoor, S.G.; Prathab, B.; Manjeshwar, L.S.; Aminabhavi, T.M. Mixed matrix membranes of sodium alginate and poly(vinyl alcohol) for pervaporation dehydration of isopropanol at different temperatures. Polymer 2007, 48, 5417–5430. [Google Scholar] [CrossRef]
- Burke, D.E.; Williams, G.C.; Plank, C.A. Vapor-liquid equilibria for the methanol-toluene system. J. Chem. Eng. Data 1964, 9, 212–214. [Google Scholar] [CrossRef]



| Film | Glass transition temperature, °C | Density, g/сm3 | Solubility parameter δ, (J/сm3)0.5 | Fractional free volume |
|---|---|---|---|---|
| PA | 184 | 1.307 | 24.84 | 0.342 |
| coPA-20 | 204 | 1.313 | 24.92 | 0.355 |
| coPA-30 | 211 | 1.319 | 24.96 | 0.361 |
| Polymer | T, °C | Total flux, g/m2h | Methanol in permeate, wt % | Permeance (P/l)Methanol, GPU | Selectivity (βMethanol-Toluene) | References |
|---|---|---|---|---|---|---|
| coPA-20 | 50 | 107.0 | 97.5 | 301 | 11 | Present work |
| coPA-30 | 50 | 126.0 | 96.3 | 316 | 7.0 | |
| Polyamide/MMT (3%) | 50 | 112 | 99.2 | 78 | 11 | [26] |
| Polyimide | 50 | 16.0 | 92.1 | 9.9 | 1.0 | [27] |
| Polyimide/polyanyline | 50 | 12.5 | 94.9 | 8.0 | 1.6 | [27] |
| Poly(phenyleneoxide) | 30 | 300 | 98.6 | 502 | 19 | [28] |
| Cellulose acetate | 30 | 1400 | 92.1 | 2188 | 3.2 | [29,30] |
| Cellulose triacetate | 30 | 370 | 92.1 | 578 | 3.2 | [29,30] |
| Cellulose acetate/Cellulose butyrate | 30 | 500 | 92.1 | 781 | 3.2 | [29] |
| Polyphthalamide/poly(vinylalcohol) | 30 | 250 | 99.6 | 422 | 68 | [31] |
| Poly(vinylalcohol)/(Hydroxy(ethyl) methacrylate + aceticanhydride) | 30 | 1100 | 98.6 | 1840 | 19 | [32] |
| Chitosan | 35 | 1000 | 99.4 | 1318 | 45 | [33] |
| Chitosan/aceticanhydride (4%) | 35 | 1000 | 99.3 | 1932 | 57 | |
| Copolymer of perfluoro-2,2-dimethyl-1,1,3-dioxole and tetrafluoroethylene (25% toluene) | 50 | 1.7 | 95.2 | 96 | 15 | [34] |
| Interpenetrating network of vinylterminated poly(dimethylsiloxane) and aromatic polyimide (90:10) | 30 | 52 | 92.3 | 2839 | 9.3 | [35] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polotskaya, G.A.; Pulyalinа, A.Y.; Goikhman, M.Y.; Podeshvo, I.V.; Valieva, I.A.; Toikka, A.M. Aromatic Copolyamides with Anthrazoline Units in the Backbone: Synthesis, Characterization, Pervaporation Application. Polymers 2016, 8, 362. https://doi.org/10.3390/polym8100362
Polotskaya GA, Pulyalinа AY, Goikhman MY, Podeshvo IV, Valieva IA, Toikka AM. Aromatic Copolyamides with Anthrazoline Units in the Backbone: Synthesis, Characterization, Pervaporation Application. Polymers. 2016; 8(10):362. https://doi.org/10.3390/polym8100362
Chicago/Turabian StylePolotskaya, Galina A., Alexandra Yu. Pulyalinа, Mikhail Ya. Goikhman, Irina V. Podeshvo, Irina A. Valieva, and Alexander M. Toikka. 2016. "Aromatic Copolyamides with Anthrazoline Units in the Backbone: Synthesis, Characterization, Pervaporation Application" Polymers 8, no. 10: 362. https://doi.org/10.3390/polym8100362
APA StylePolotskaya, G. A., Pulyalinа, A. Y., Goikhman, M. Y., Podeshvo, I. V., Valieva, I. A., & Toikka, A. M. (2016). Aromatic Copolyamides with Anthrazoline Units in the Backbone: Synthesis, Characterization, Pervaporation Application. Polymers, 8(10), 362. https://doi.org/10.3390/polym8100362










