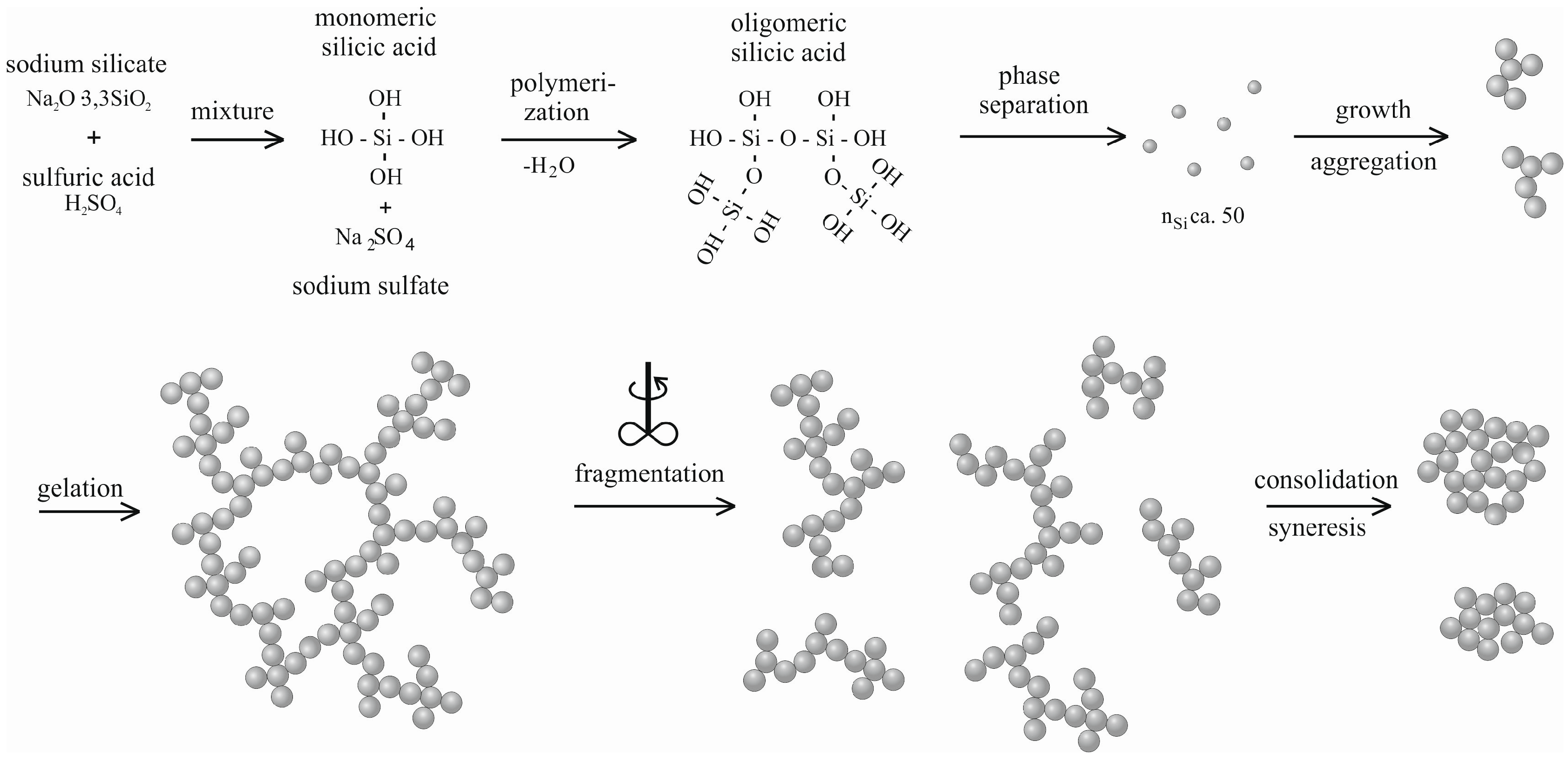
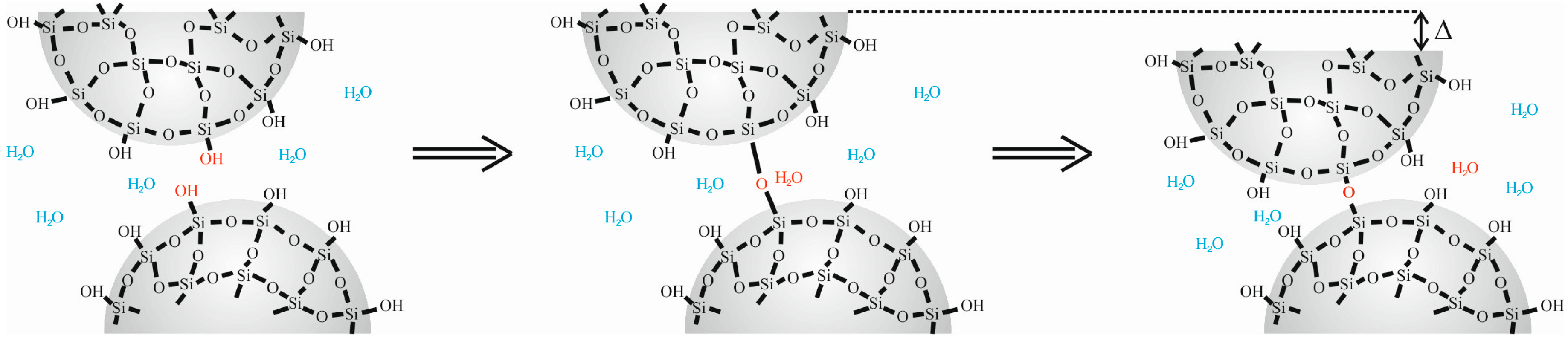
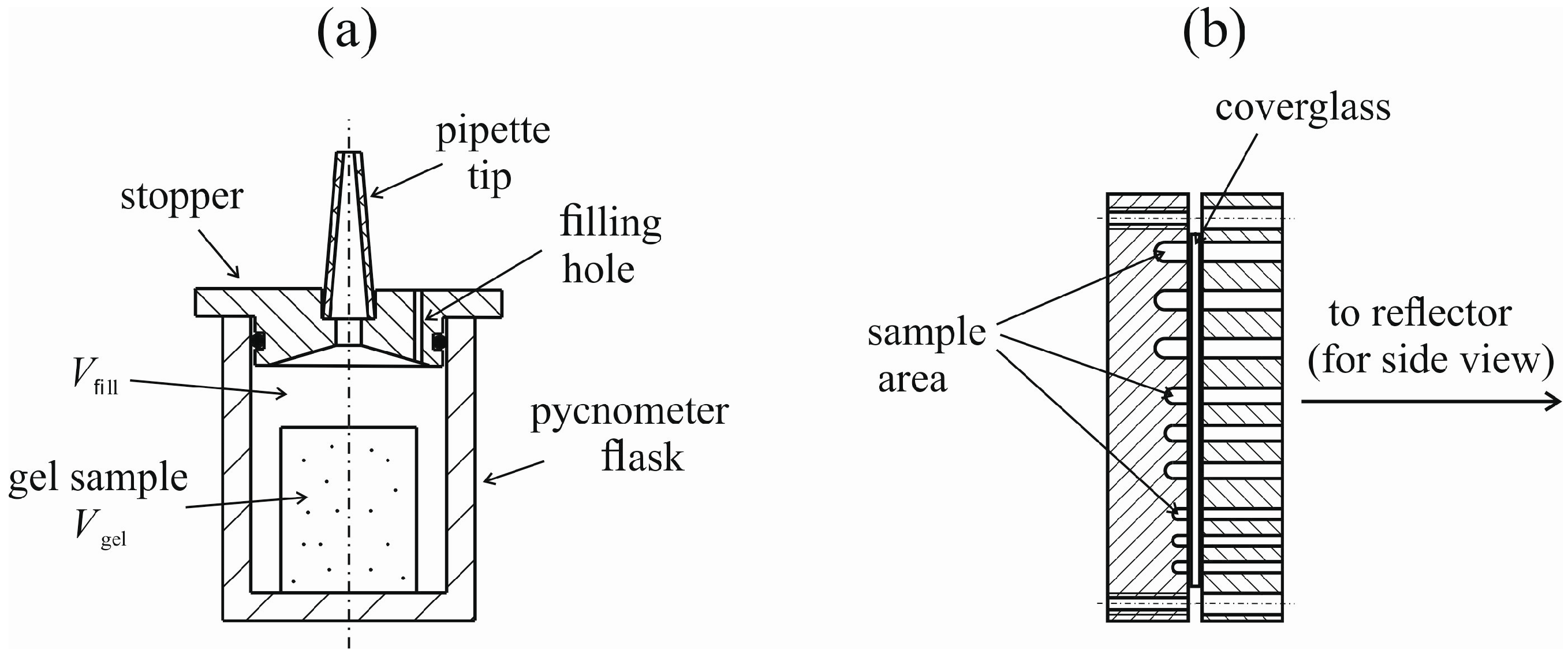
4.1. Natural Syneresis
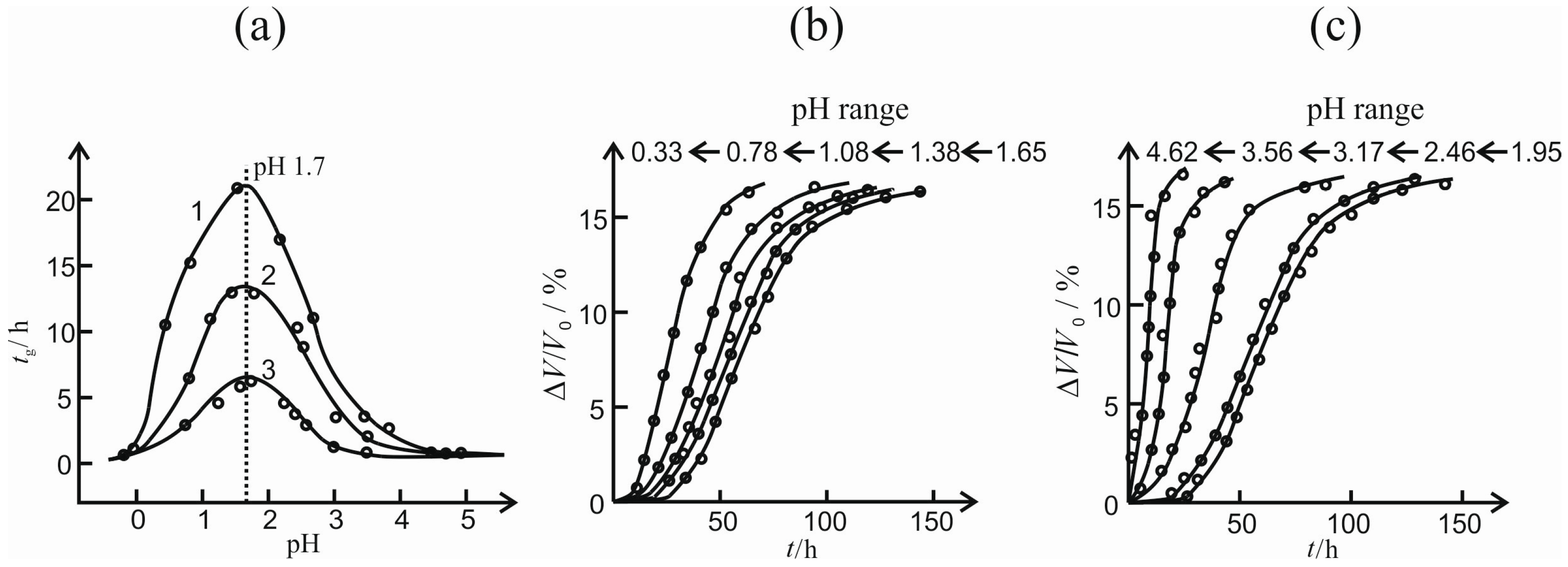
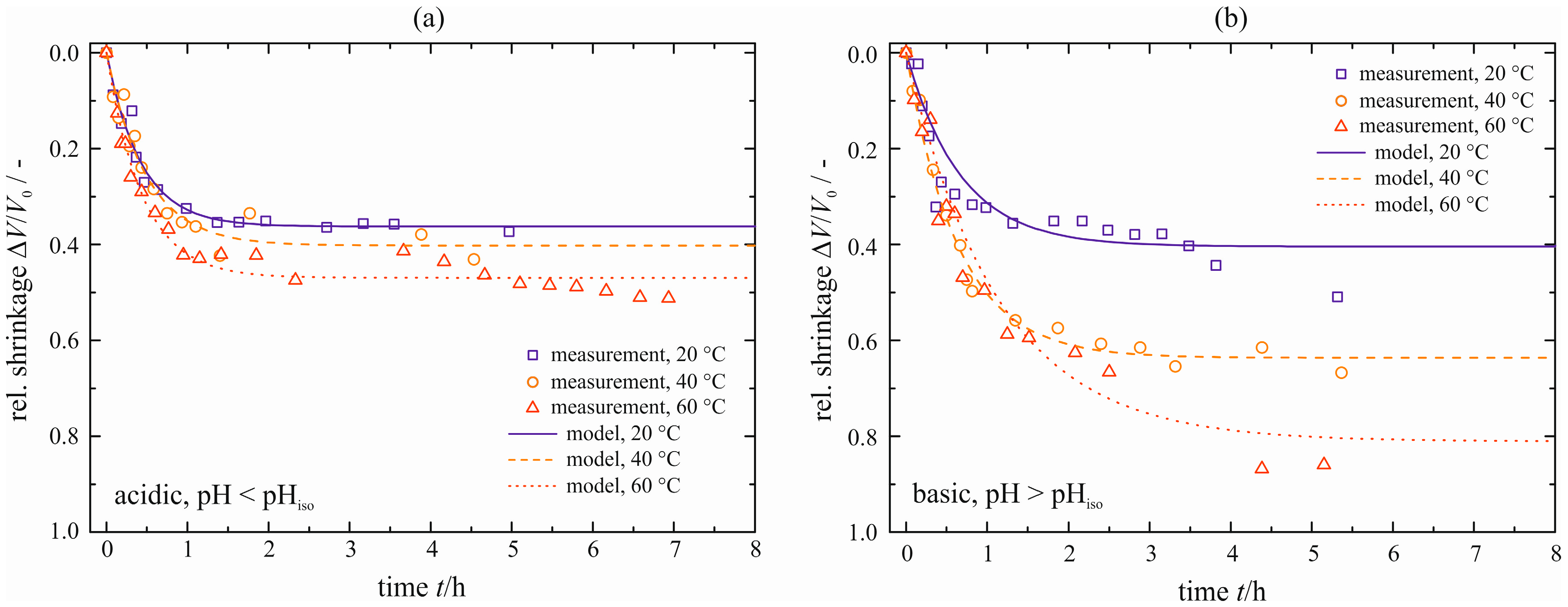
The temporal courses of the relative shrinkage Δ
V/
V0 of the cylindrical samples (
dCyl = 12 mm and
VGel ≈ 3 mL) are determined experimentally for three different temperatures and are depicted in
Figure 8 for an acid- and base-catalyzed gel. For the acid-catalyzed gel, a maximum shrinkage of Δ
V/
V0|
max = 0.20 is reached independently of the temperature. The phenomenon of a slightly lower value for the maximum shrinkage with increasing temperature stated by Ponomerova [
15] cannot be proved or disproved when the error bars are regarded. However, the temperature affects the kinetics of syneresis reflected by the different slopes for
t < 50 h. With increasing temperature, syneresis is accelerated. A simple model based upon a kinetic rate equation is proposed for a quantitative description of the courses, see Equation (3). A physically based model is still to be made.
Equation (3) comprises two parameters that represent the maximum shrinkage Δ
V/
V0|
max (the value for
t→∞) and the characteristic time constant τ (taking into consideration the kinetics at the beginning). These two parameters are characteristic for the measurements and are used for further comparison of the influence of different process conditions. Their values are listed in
Table 1.
Figure 8.
Relative shrinkage Δ
V/
V0 of the cylindrical samples, natural syneresis for different temperatures ϑ, acid-catalyzed [
5] (
a), and base-catalyzed (
b).
Figure 8.
Relative shrinkage Δ
V/
V0 of the cylindrical samples, natural syneresis for different temperatures ϑ, acid-catalyzed [
5] (
a), and base-catalyzed (
b).
Table 1.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ for the cylindrical samples (dCyl = 12 mm and VGel ≈ 3 mL), natural syneresis, model parameters.
Table 1.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ for the cylindrical samples (dCyl = 12 mm and VGel ≈ 3 mL), natural syneresis, model parameters.
| Temperature ϑ/°C | Maximum Shrinkage ΔV/V0|max/- | Time Constant τ/h |
|---|
| Acid-catalyzed pH < pHiso | Base-catalyzed pH > pHiso | Acid-catalyzed pH < pHiso | Base-catalyzed pH > pHiso |
|---|
| 20 | 0.20 | 0.30 | 21.3 | 50.1 |
| 40 | 0.20 | 0.39 | 11.7 | 32.0 |
| 60 | 0.20 | 0.51 | 5.0 | 27.5 |
A different behavior is observed for the base-catalyzed gel, see
Figure 8b. Both the maximum shrinkage and the rate increase with temperature. In direct comparison to the acid-catalyzed gel, the base-catalyzed gel exhibits larger values for the maximum shrinkage. They reach from Δ
V/
V0|
max = 0.30 to 0.51. This can be explained by the different structure of the solid skeleton. The acid-catalyzed gel is built up of compact primary particles that are connected loosely, resulting in a translucent gel tending to brittle fracture. However, the base-catalyzed gel behaves in an opposite way. It is composed of loosely connected primary particles that form a softer network than the acid-catalyzed gel. As a result of these different structures, the base-catalyzed gel exhibits a larger maximum shrinkage [
1]. The shrinkages measured are fitted with the same model, see Equation (3). The base-catalyzed gel consolidates at a much slower velocity, reflected by larger values for the characteristic time constant τ (see
Table 1). One explanation might be the difference in the solid structure of the gels. In the case of the acid-catalyzed gel, the pore liquid can flow more easily through the gel network due to the compact, but loosely connected primary particles. For the base-catalyzed gel, a denser network forms, resulting in a slower drainage of the pore liquid. Scattering experiments of these differently catalyzed gels emphasize these differences in their solid structures and optical appearances [
8]. Regarding the maximum shrinkage, there are small deviations between the measured and modeled data. The measured data seem to decrease further even after
t > 250 h, especially for higher temperatures. These deviations are accepted in order to facilitate a comparison to the acid-catalyzed gel.
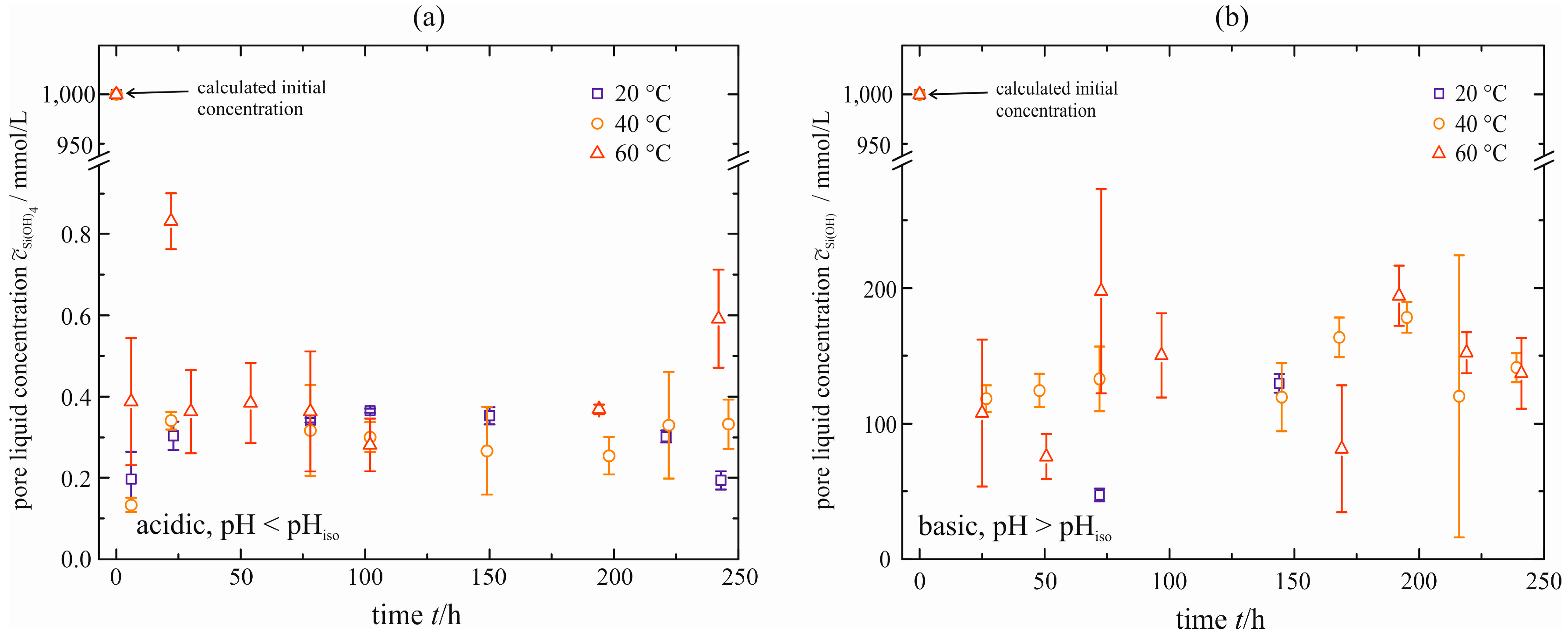
The concentration of monomeric silicic acid in the pore liquid squeezed out is determined to monitor a possible residual supersaturation
S. Therefore, the pore liquid is analyzed spectroscopically, as described by Alexander [
17]. The courses are shown in
Figure 9. The initial concentration
of monomeric silicic acid is determined and calculated by the weight of sodium silicate solution used. A very strong reduction to concentrations in order of
= 0.2–0.4 mmol/L is obtained for the acid-catalyzed gel (
Figure 9a). They are within the solubility limit of ≤2 mmol/L given by Iler [
2]. There is no distinct change with time
t and temperature. The latter can be explained by the solubility of monomeric silicic acid that is nearly independent of temperature in this range of pH [
3]. In the case of the base-catalyzed gel (
Figure 9b), this reduction is much less distinct (
= 100–200 mmol/L). This is due to the higher pH, resulting in the increase of solubility
. As already stated for the acid-catalyzed gel, there is no distinct dependency on temperature although the solubility of monomeric silicic acid increases strongly with temperature for the base-catalyzed polymerization. This might be due to the fact that the spectroscopic analysis could not be performed at elevated temperature, but at room temperature (~20 °C). Thus, monomeric silicic acid can polymerize and lower the concentration in the pore liquid squeezed out.
Figure 9.
Concentration of monomeric silicic acid in the pore liquid of cylindrical samples, acid-catalyzed (a) and base-catalyzed (b), natural syneresis.
Figure 9.
Concentration of monomeric silicic acid in the pore liquid of cylindrical samples, acid-catalyzed (a) and base-catalyzed (b), natural syneresis.
For reasons still not explained, the concentration measured is higher than the solubility calculated with Equation (1). This fact is confirmed by the pore liquid that becomes turbid if stored for several days at constant temperature. It seems as if new solid is formed when the pore liquid is separated from the solid. However, an unambiguous increase of the concentration with temperature, as presented previously in the state-of-the-art, cannot be stated. If any, there is only a small tendency for both gels.
The sample size affects the syneresis behavior strongly. Therefore, small gel droplets (
dDrop ≈ 3 mm) instead of the cylindrical samples are prepared and examined for their syneresis behavior depending on pH and temperature. The courses of shrinkage of the gel droplets are depicted in
Figure 10.
Compared to
Figure 8, the different scaling of the
x-axis should be noted. Smaller samples consolidate much faster, clearly showing the dependency between the syneresis rate and sample size. Furthermore, the maximum shrinkage Δ
V/
V0|
max increases for both types of catalysts and, in the case of the acid-catalyzed gel, even fans out with increasing temperature. This behavior cannot be observed for the cylindrical samples and, thus, must be attributed to the smaller sample size. There is no great difference between the courses for both types of catalyst during the first 30 min. This is due to the constant temperature of 20 °C during this period. Thereafter, the temperature is set to the values given in
Figure 10. Thus, each droplet must be heated firstly to this temperature. This might be the reason for the almost similar course in the case of 40 and 60 °C for
t ≤ 2 h, especially for the base-catalyzed gel droplets. All model parameters for the gel droplets are summarized in
Table 2.
Figure 10.
Relative shrinkage ΔV/V0 of the gel droplets, natural syneresis for different temperatures ϑ, acid-catalyzed (a) and base-catalyzed (b).
Figure 10.
Relative shrinkage ΔV/V0 of the gel droplets, natural syneresis for different temperatures ϑ, acid-catalyzed (a) and base-catalyzed (b).
Table 2.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ for the gel droplets (dDrop ≈ 3 mm), natural syneresis, model parameters.
Table 2.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ for the gel droplets (dDrop ≈ 3 mm), natural syneresis, model parameters.
| Temperature ϑ/°C | Maximum Shrinkage ΔV/V0|max/- | Time Constant τ/h |
|---|
| Acid-catalyzed pH < pHiso | Base-catalyzed pH > pHiso | Acid-catalyzed pH < pHiso | Base-catalyzed pH > pHiso |
|---|
| 20 | 0.37 | 0.30 | 0.40 | 0.67 |
| 40 | 0.41 | 0.39 | 0.64 | 0.57 |
| 60 | 0.48 | 0.51 | 0.77 | 0.95 |
An analysis of the pore liquid as performed for the cylindrical gel samples is not possible due to the very small amounts of pore liquid squeezed out of the gel droplets.
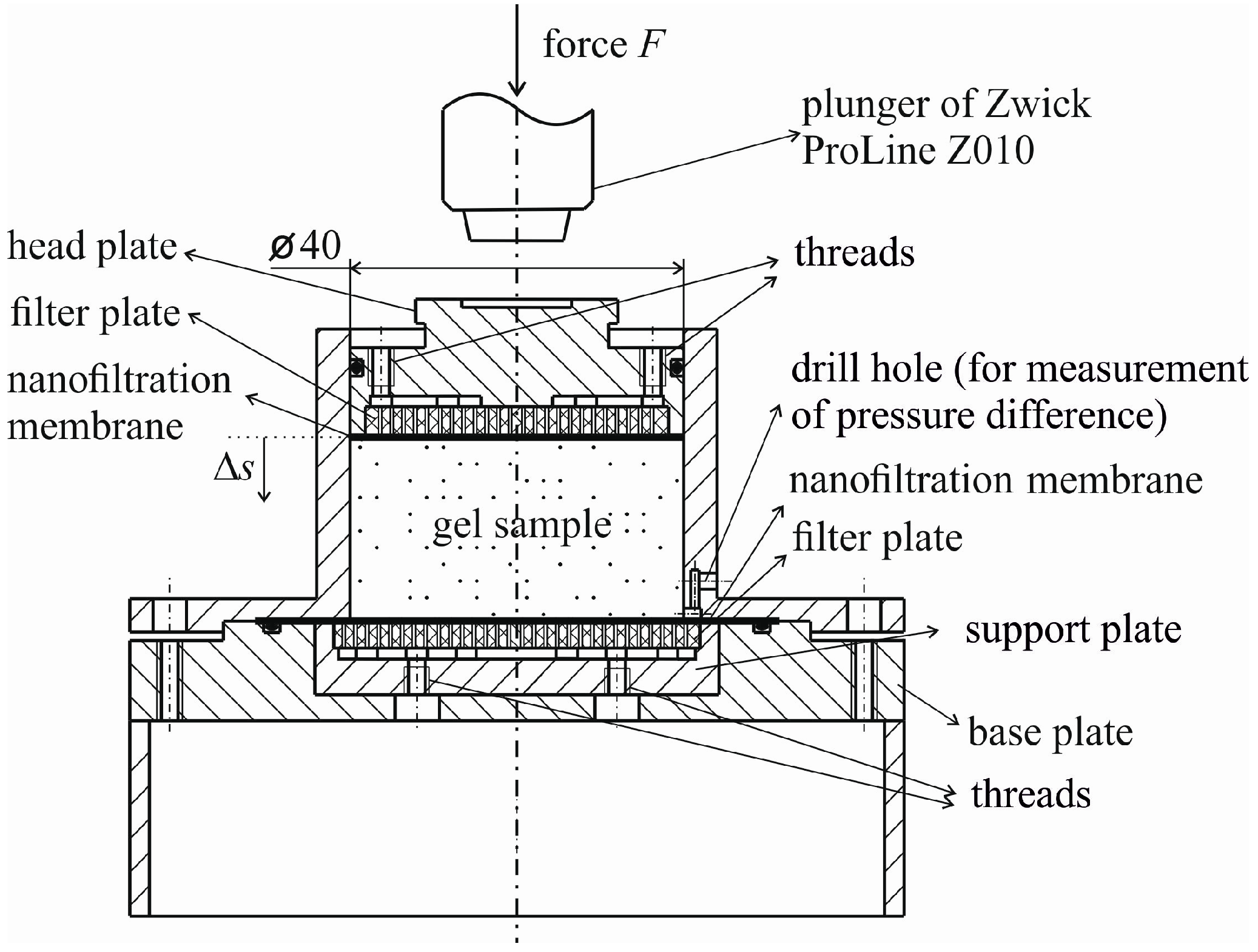
4.2. Enforced Syneresis
An additional process parameter, that is the pressure difference Δ
p induced by the external force
F, is introduced for enforced syneresis. Generally, a membrane is necessary to retard the solid formed. Two different membranes are investigated with regard to their suitability for enforced syneresis. A simple and easy to perform experiment comprising a pressure nutsch and the specific membrane is set up. The pressure nutsch is filled with Si standard solution (Certipur
®, Merck Millipore) already used for the calibration of UV/Vis measurements. The nutsch is operated with pressurized air at Δ
pair = 4 bar and the concentration in the permeate is determined with the same method described in the previous section.
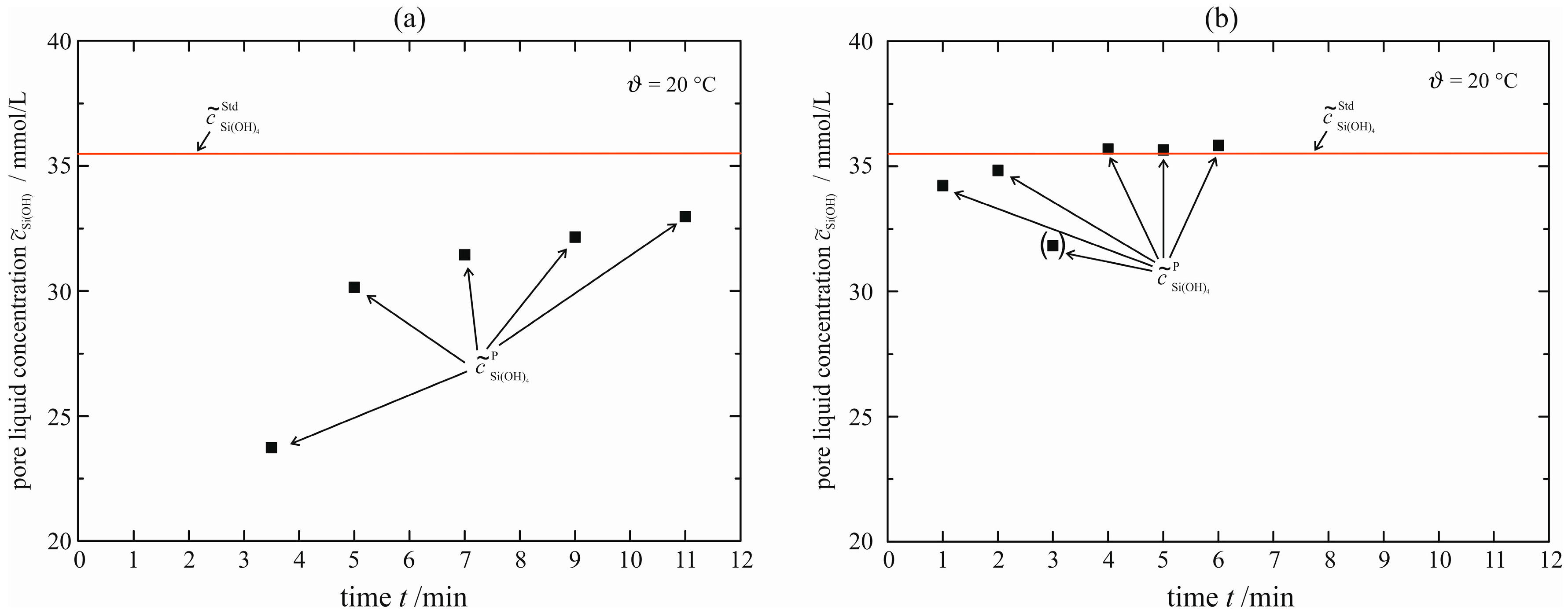
Figure 11 shows the courses of permeate concentration for the two different membranes. The Si standard solution has a molar concentration of
= 35.7 mmol/L and is depicted as a horizontal line. The permeate concentrations
(a permeate volume of
VP = 1 mL is chosen) for the membrane Dow Filmtec NF270 (
Figure 11a) is always lower than the standard solution that is filled into the pressure nutsch. Thus, that membrane retards the monomeric silicic acid and, when applied to enforced syneresis, an influence on the syneresis process cannot be excluded. Using the membrane Microdyn-Nadir NP010, the permeate concentrations reach the concentration of the standard solution almost immediately. This means that much less monomeric silicic acid is stored inside or held back by the membrane. For this reason, the latter membrane is used to retard the solid in the case of enforced syneresis. In the previous publication [
5], experiments of the acid-catalyzed gel were performed with the membrane Dow Filmtec NF270. All these experiments are repeated with the membrane Microdyn-Nadir NP010. There is no significant difference in the results between the two membranes. However, this is not valid for the base-catalyzed gel.
Figure 11.
Concentration of monomeric silicic acid in the permeate for membrane Dow Filmtec NF270 (a) and Microdyn-Nadir NP010 (b) upon filtration of Si standard solution (Certipur®, Merck Millipore).
Figure 11.
Concentration of monomeric silicic acid in the permeate for membrane Dow Filmtec NF270 (a) and Microdyn-Nadir NP010 (b) upon filtration of Si standard solution (Certipur®, Merck Millipore).
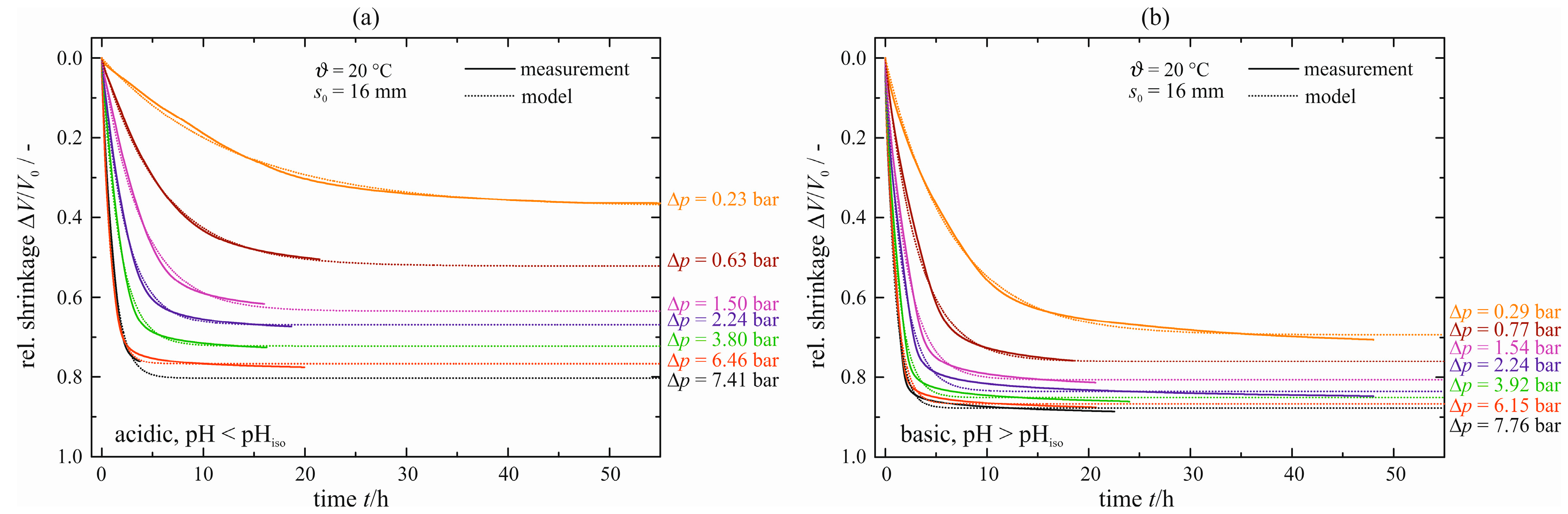
The courses of enforced syneresis for ϑ = 20 °C with an initial gel sample height
s0 = 16 mm are shown in
Figure 12. Both measured (solid lines) and modeled data (dashed lines), see Equation (3), are given. As might be expected, all curves are shifted to larger maximum shrinkages and shorter times with increasing pressure difference Δ
p, independently of the catalyst. When comparing the acid- with the base-catalyzed gel, the latter exhibits a less distinctive spread although the same range of pressure differences Δ
p is covered. A reason might be the softer solid structure, leading to a lower resistance against the pressure difference. This is supported by a larger maximum shrinkage for equal pressure differences Δ
p (e.g., Δ
V/
V0|
max = 0.8 and 0.63 at Δ
p = 1.54 and 1.50 bar, respectively).
Figure 12.
Relative shrinkage ΔV/V0 of enforced syneresis for s0 = 16 mm and ϑ = 20 °C, acid-catalyzed (a) and base-catalyzed (b).
Figure 12.
Relative shrinkage ΔV/V0 of enforced syneresis for s0 = 16 mm and ϑ = 20 °C, acid-catalyzed (a) and base-catalyzed (b).
All values for the maximum shrinkage Δ
V/
V0|
max and the characteristic time constant τ are summarized in
Table 3. As can be seen, the maximum shrinkage almost halved from maximum to minimum pressure difference for the acid-catalyst gel, whereas the base-catalyst gel shows a reduction of merely one fifth. Exactly the inverse applies for the characteristic time constant τ. A 13-fold increase stands against a 6–7-fold increase.
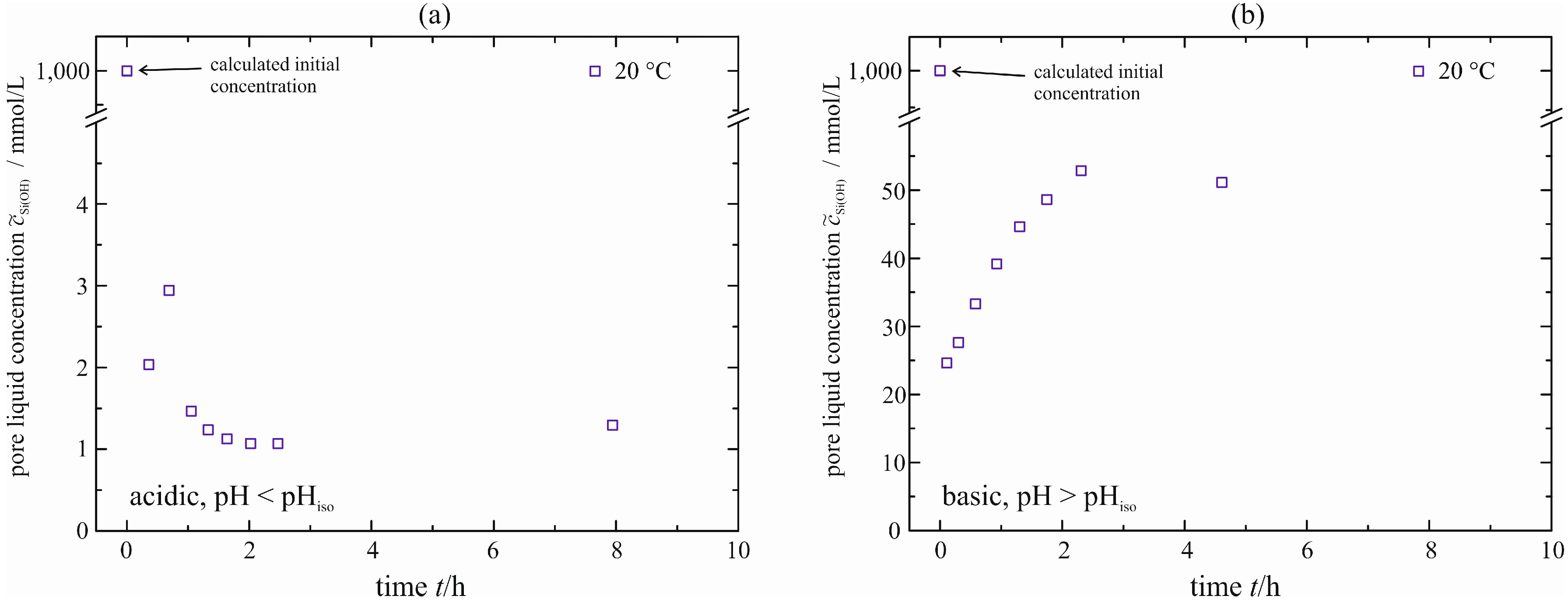
Until now, the concentration of monomeric silicic acid in the permeate is given merely for Si standard solution filled in. Adopting the same procedure, the concentration in the permeate is measured for the acid- and base-catalyzed gel in order to compare them with the concentrations of natural syneresis. This is done exemplarily for Δ
p = 3.80–3.92 bar and ϑ = 20 °C. The courses are given in
Figure 13. In the case of the acid-catalyzed gel, there is a strong reduction in the concentration from
= 1000 mmol/L to
≈ 1.0–1.5 mmol/L. A decrease in concentration (from 1000 to 25 mmol/L) and a subsequent increase to approximately 55 mmol/L is observed for the base-catalyzed gel. Neither are determined for natural syneresis (see
Figure 9). We assume an additional solid formation directly on the membrane due to the rising turbidity of the pore liquid for natural syneresis described already in
Section 4.1. To prove this assumption, the pore liquid of natural syneresis (base-catalyzed gel) is filled into the pressure nutsch. Therefore, the influence of the solid is eliminated and only the behavior of pore liquid and membrane is investigated. The analysis of the pore liquid with respect to monomeric silicic acid yields a very similar diagram to
Figure 13b.
Table 3.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ, enforced syneresis, s0 = 16 mm and ϑ = 20 °C.
Table 3.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ, enforced syneresis, s0 = 16 mm and ϑ = 20 °C.
| Pressure Difference Δp/bar | Maximum Shrinkage ΔV/V0|max/- | Time Constant τ/h |
|---|
| Acid-catalyzed pH < pHiso | Base-catalyzed pH > pHiso | Acid-catalyzed pH < pHiso | Base-catalyzed pH > pHiso |
|---|
| 0.23–0.29 | 0.38 | 0.69 | 13.1 | 6.5 |
| 0.63–0.77 | 0.52 | 0.76 | 5.9 | 3.2 |
| 1.50–1.54 | 0.63 | 0.81 | 3.8 | 2.1 |
| 2.24–2.24 | 0.67 | 0.83 | 2.3 | 1.7 |
| 3.80–3.92 | 0.72 | 0.85 | 1.7 | 1.1 |
| 6.15–6.46 | 0.77 | 0.87 | 1.1 | 1.0 |
| 7.41–7.76 | 0.80 | 0.88 | 1.0 | 0.9 |
Figure 13.
Concentration of monomeric silicic acid in the pore liquid, acid-catalyzed (a) and base-catalyzed (b), enforced syneresis for Δp = 3.80–3.92 bar.
Figure 13.
Concentration of monomeric silicic acid in the pore liquid, acid-catalyzed (a) and base-catalyzed (b), enforced syneresis for Δp = 3.80–3.92 bar.
A decrease and a subsequent increase of permeate concentration is observed. In addition, the time which elapsed between two samples to collect a permeate volume of
VP = 1 mL increased from a few minutes directly at the beginning to several hours for the last sample although the driving pressure was constant (Δ
p = 4 bar) and there was still enough retentate volume present in the cell (
VR ≈ ¾
VR (
t = 0)). This is an indication of a buildup of an additional flow resistance. Indeed, there is a thin layer on the membrane after the termination of the experiment. However, this layer is found only for pore liquid of the base-catalyzed gel. We conclude that the layer is built up during enforced syneresis, but has no significant influence on the maximum shrinkage due to its negligibly small thickness. By contrast, there is an influence on the kinetics of syneresis that has not been able to be eliminated up to now. The courses and model parameters of enforced syneresis for ϑ = 40 and 60 °C are given in the
Figures S3 and S4 and in the
Tables S1 and S2 in the
Supplementary Materials. Due to the increased temperature, the rate of syneresis is accelerated (smaller values for the characteristic time constant τ) for both types of catalyst. However, the maximum shrinkage Δ
V/
V0|
max is almost unaffected for the acid-catalyzed gel, whereas larger values are obtained in the case of the base-catalyzed gel. Thus, this is a first indication of an analogous behavior between natural and enforced syneresis.
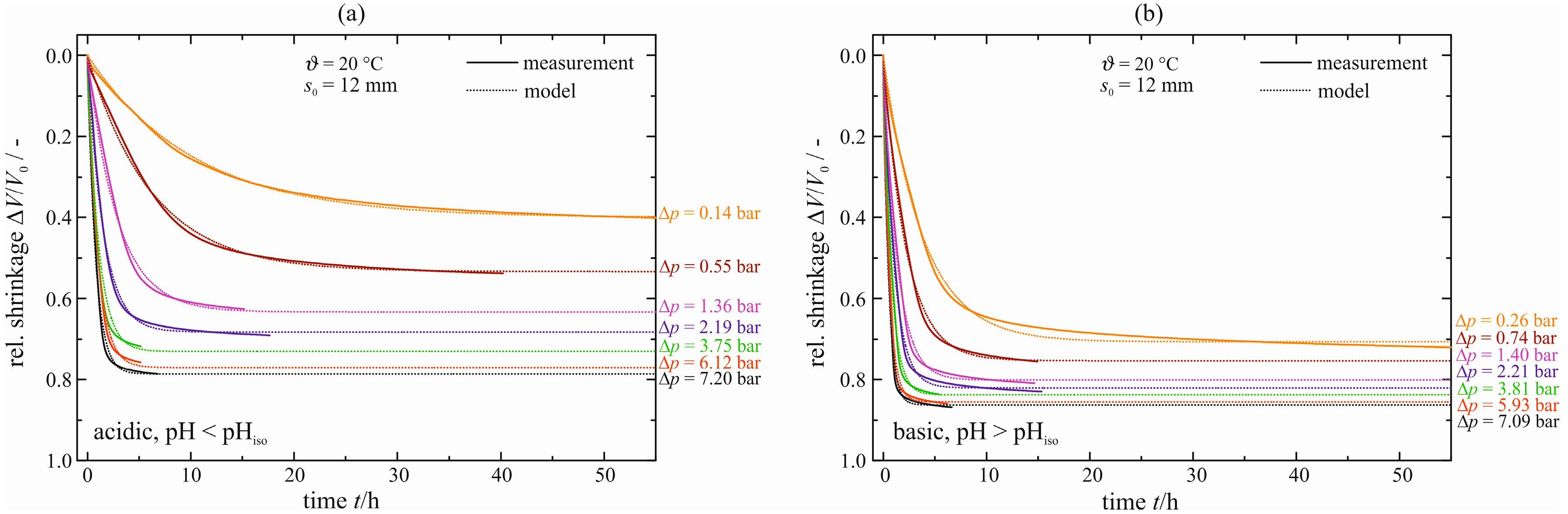
The distances that are flowed through by the pore liquid have to be equal for a quantitative comparison between natural and enforced syneresis. Assuming a radial flow within the cylindrical gel samples, this distance corresponds to the radius, that is
= 6 mm. For the enforced syneresis presented above, this distance matches half of the initial gel sample height,
= 8 mm. These differences, albeit small, affect at least the kinetics and, potentially, the maximum shrinkage. Therefore, the initial sample height is reduced to
s0 = 12 mm. The temporal courses of shrinkage are shown in
Figure 14.
Figure 14.
Relative shrinkage ΔV/V0 of enforced syneresis for s0 = 12 mm and ϑ = 20 °C, acid-catalyzed (a) and base-catalyzed (b).
Figure 14.
Relative shrinkage ΔV/V0 of enforced syneresis for s0 = 12 mm and ϑ = 20 °C, acid-catalyzed (a) and base-catalyzed (b).
As might be expected from the results of natural syneresis, the kinetics of syneresis is accelerated, compared with
Figure 12. This is expressed by smaller values for the characteristic time constant τ, e.g., from τ = 2.3 to 1.5 h for the acid-catalyzed gel with Δ
p = 2.24 and 2.19 bar, respectively. Thus, enforced syneresis behaves analogously to natural syneresis with respect to initial sample size. However, the maximum shrinkage Δ
V/
V0|
max is virtually unaffected by the reduction of the initial gel sample height, contrary to the results of the cylindrical gel samples and droplets. All values can be found in
Table S3 in the
Supplementary Materials. A possible explanation is the very different order of size reduction between the cylindrical gel samples and droplets for natural syneresis,
i.e., the reduction of gel sample height by 4 mm for enforced syneresis. The courses of shrinkage for elevated temperature of 40 and 60 °C and their model parameters can be found in
Figures S5 and S6 and
Tables S4 and S5 in the
Supplementary Materials. The maximum shrinkage Δ
V/
V0|
max is nearly independent of the temperature for the acid-catalyzed gel, whereas it increases with temperature in the case of the base-catalyzed gel. In direct comparison to the larger initial gel sample height, see
Figure 12, the maximum shrinkage is equal with a small tendency to larger values. However, the rate of syneresis is accelerated due to the reduced initial gel sample height, represented by smaller values for the characteristic time constant τ. In summary, enforced syneresis behaves analogously to natural syneresis with respect to changes in the initial sample dimensions.
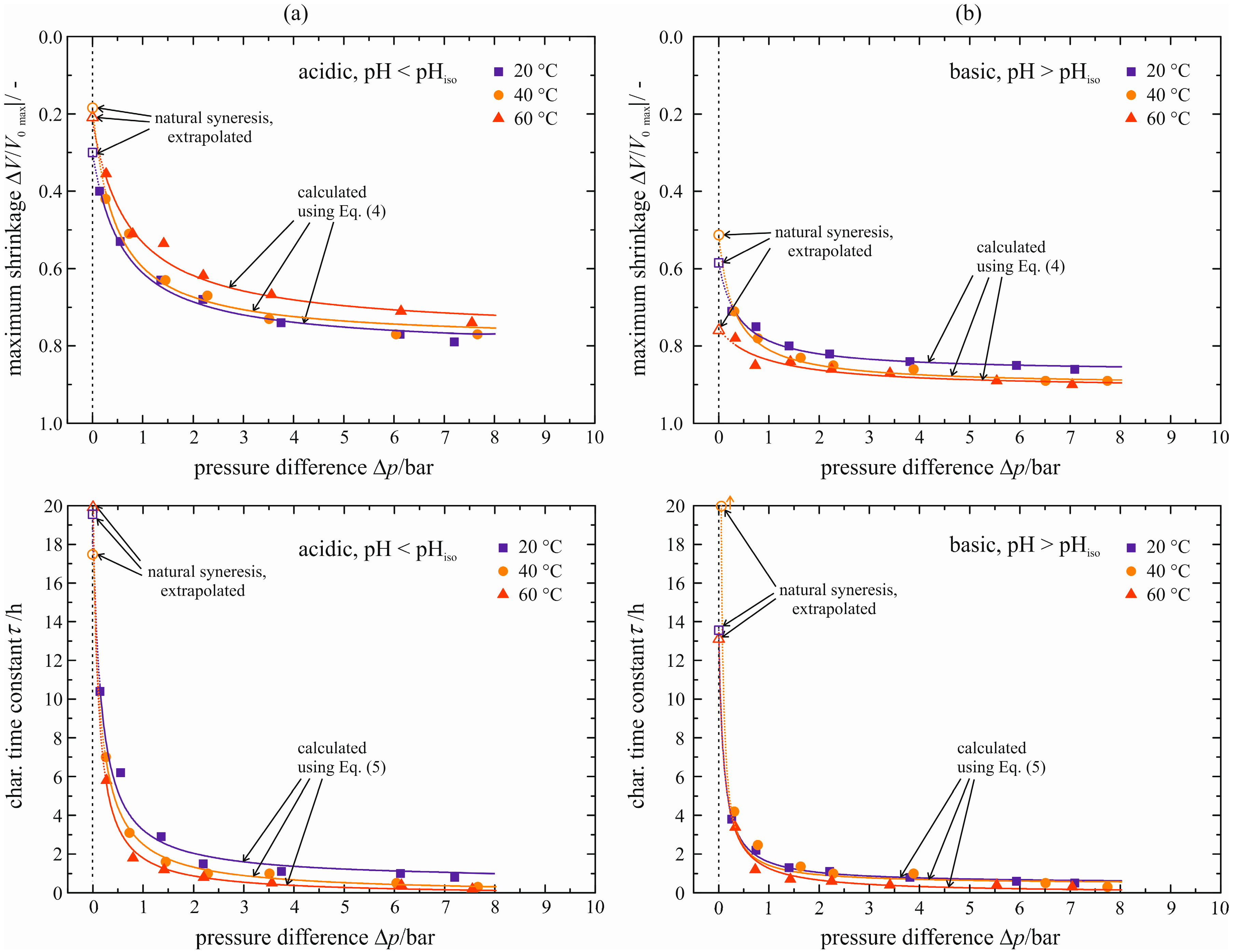
4.3. Correlative Model
The model correlates the maximum shrinkage Δ
V/
V0|
max and the characteristic time constant τ to the applied pressure difference Δ
p. They are represented by the filled symbols in
Figure 15. Their dependence on the pressure difference Δ
p may be fitted separately for each temperature with the following equations:
Figure 15.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ for acid-catalyzed (a) and base-catalyzed (b) gel as a function of pressure difference Δp.
Figure 15.
Maximum shrinkage ΔV/V0|max and characteristic time constant τ for acid-catalyzed (a) and base-catalyzed (b) gel as a function of pressure difference Δp.
These equations are chosen with respect to the course of the values and to the possibility of a mathematical extrapolation towards Δ
p = 0 bar. All values for the coefficients
A–
F can be found in
Tables S6 and S7 in the
Supplementary Materials. The courses of these equations are split in two regions. The first region comprises the pressure differences that are covered experimentally and is depicted as solid lines in
Figure 15. Equations (4) and (5) reflect the progression of enforced syneresis sufficiently. This region is used to compare the temperature-dependent behavior of enforced with natural syneresis. The second region of the courses deals with the extrapolation towards Δ
p = 0 bar, shown as dotted lines in
Figure 15. The values obtained for Δ
p = 0 bar are shown by the blank symbols.
A slightly smaller value for the maximum shrinkage Δ
V/
V0|
max can be detected with increasing temperature for the acid-catalyzed gel (
Figure 15a). This is not in accordance with the temperature behavior for natural syneresis (see
Figure 8). The extrapolation towards Δ
p = 0 bar yields maximum shrinkages for natural syneresis of Δ
V/
V0|
max = 0.19 to 0.30 that are in the range of the values measured for natural syneresis. In the case of the base-catalyzed gel (
Figure 15b), larger values for the maximum shrinkage are obtained with increasing temperature as already measured for natural syneresis (
Figure 8). The extrapolated values for Δ
p = 0 bar are Δ
V/
V0|
max = 0.52 to 0.75 depending on the temperature and, thus, almost twice as large as those measured for natural syneresis. Here, the correlative model overestimates natural syneresis. Nevertheless, larger shrinkages for the base-catalyzed gel are predicted in comparison to the acid-catalyzed gel that is accordance with the measurements. Independent of the catalyst, the syneresis velocity increases with temperature, reflected by smaller values for the characteristic time constant (
Figure 15). This behavior is analogous to natural syneresis. However, the acceleration is less distinctive and less unambiguous for the base-catalyzed gel. The values predicted through extrapolation towards Δ
p = 0 bar vary between 17.4 h ≤ τ (Δ
p = 0 bar) ≤ 21.2 h for the acid-catalyzed gel. They match partially or are at least in the range of the values determined for natural syneresis, see
Table 1. In the case of the base-catalyzed gel, the characteristic time constant takes values of 13.2 h ≤ τ (Δ
p = 0 bar) ≤ 50.6 h. Again, these values are related to those of natural syneresis, but the correct assignment to temperature is not given. Here, the largest value for τ is assigned to ϑ = 40 °C and not to the lowest temperature of ϑ = 20 °C. This is due to the sensitivity of the extrapolation especially for small pressure differences. Thus, special attention must be paid to the correlative model to predict natural syneresis.