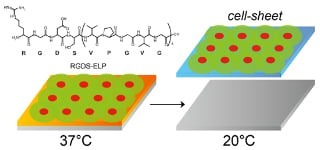
Fabrication of Thermo-Responsive Molecular Layers from Self-Assembling Elastin-Like Oligopeptides Containing Cell-Binding Domain for Tissue Engineering
Abstract
:1. Introduction
2. Experimental Section
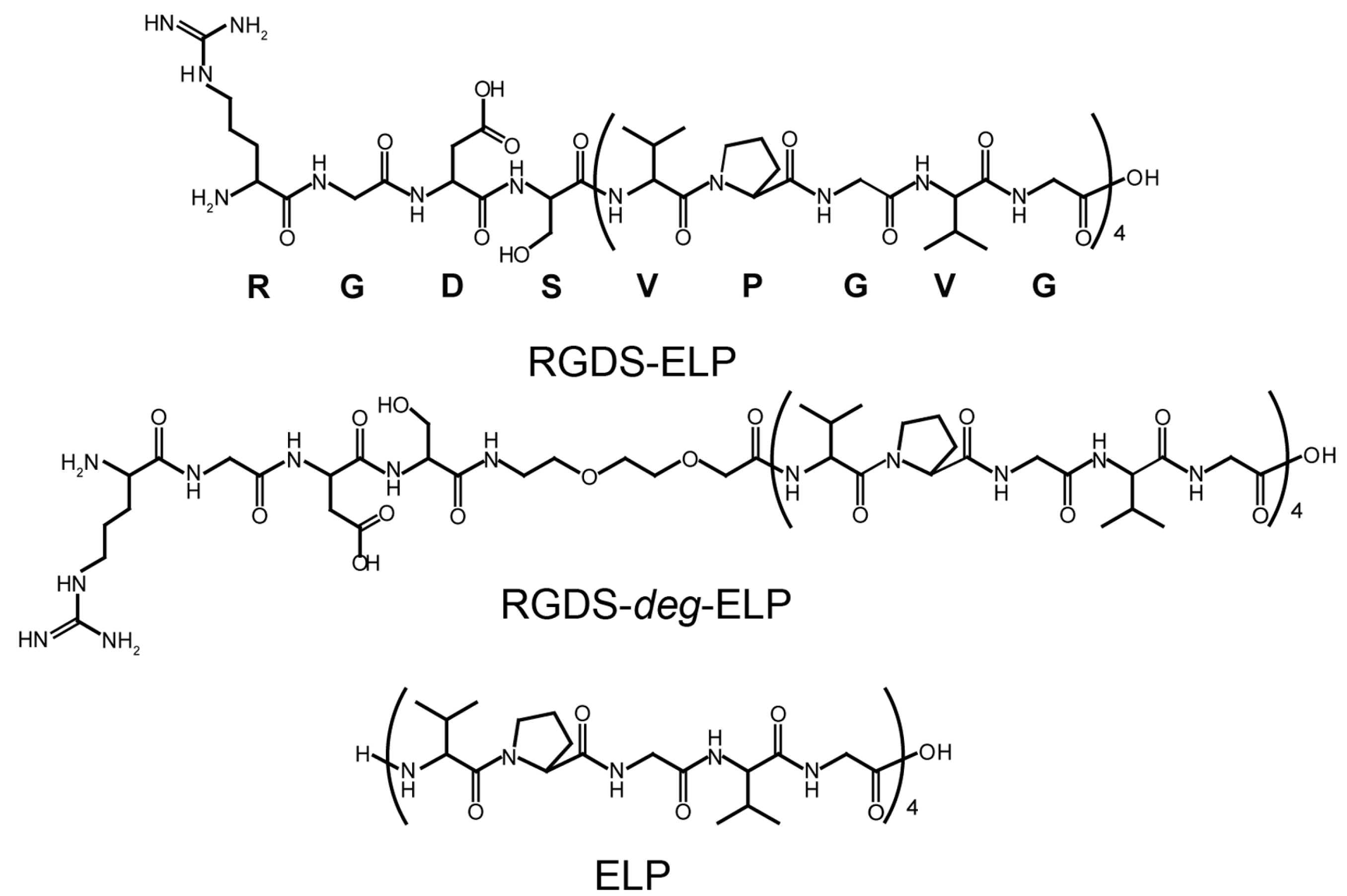
2.1. Peptide Preparation
2.2. Circular Dichroism (CD) Spectroscopy and FTIR Spectroscopy Measurements
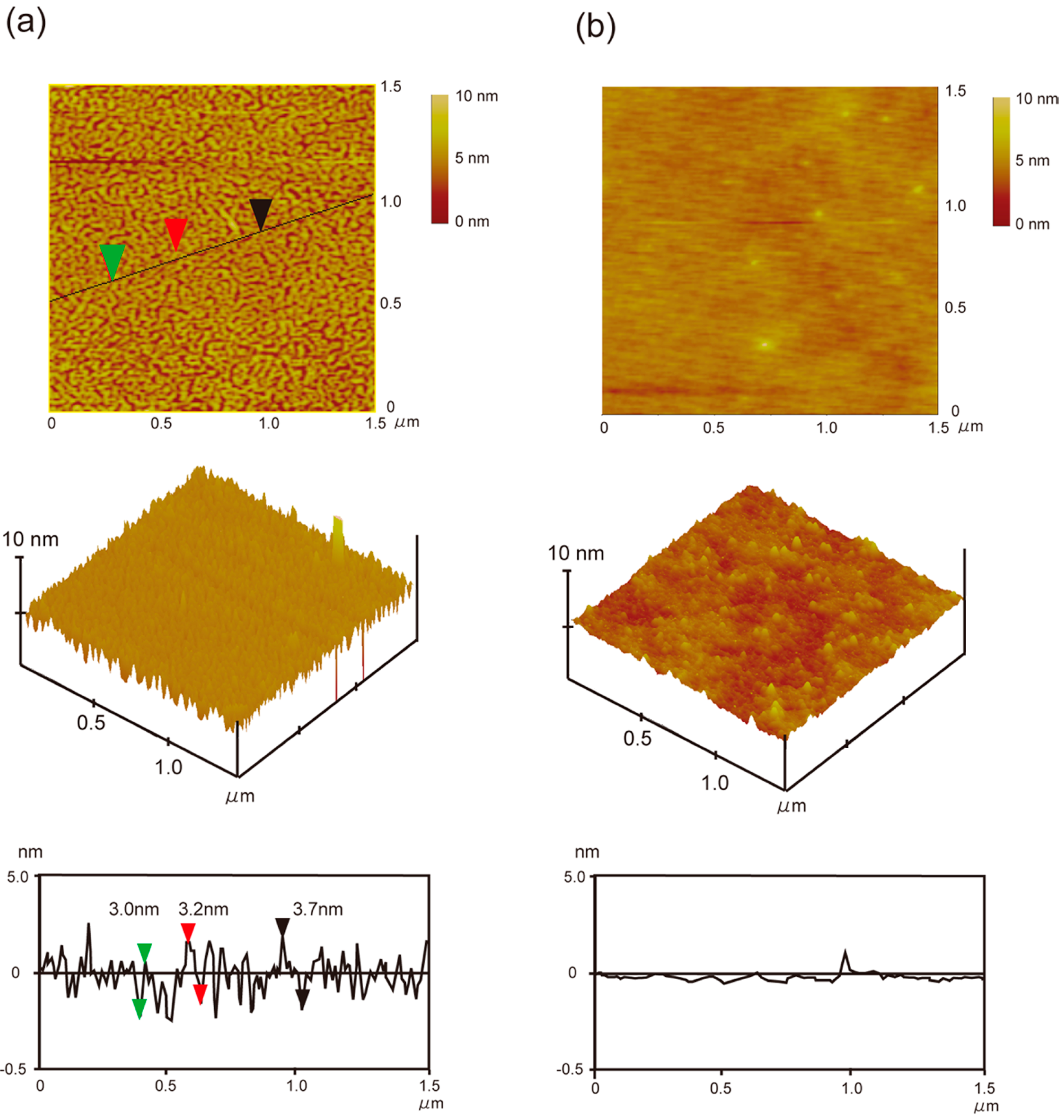
2.3. Atomic Force Microscopy (AFM) and Contact Angle Measurements
2.4. Cell Attachment
3. Results and Discussion
3.1. Preparation of Thermo-Responsive Oligopeptides Containing Cell-Binding Domain and Their Conformational Property in Aqueous Solutions
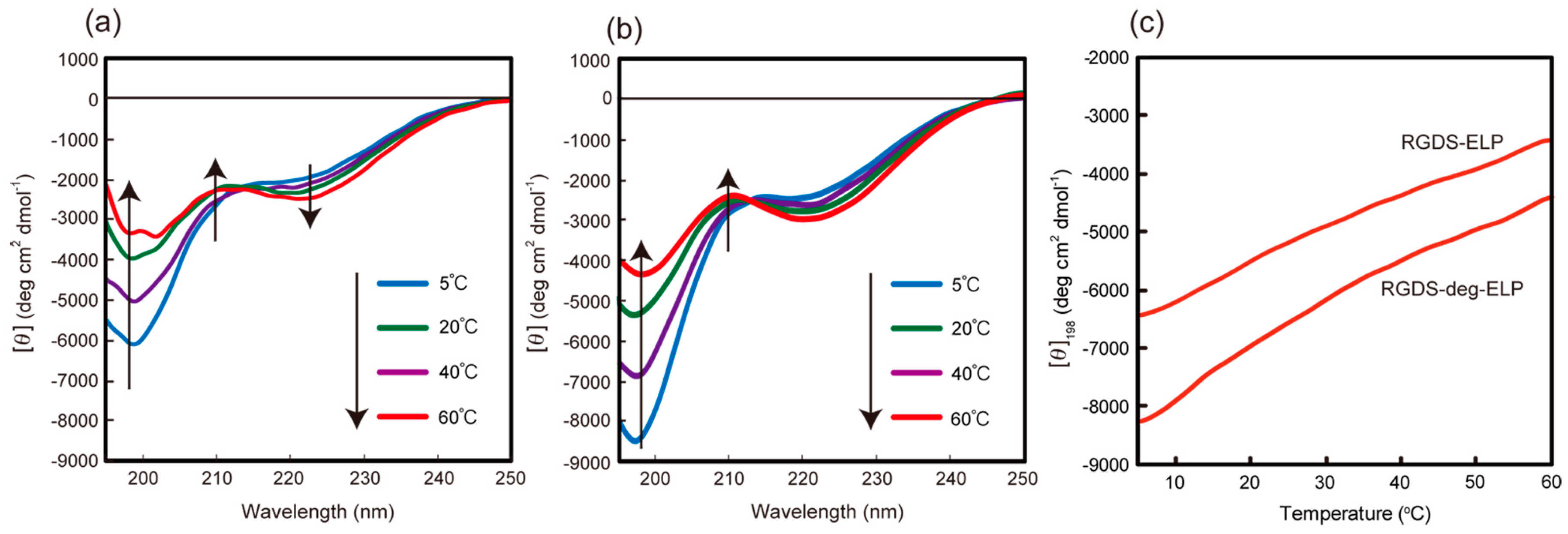
3.2. Thermo-Induced Self-Assembly of Oligo(ELP)s Containing Cell-Binding Domain onto Hydrophobic Surfaces
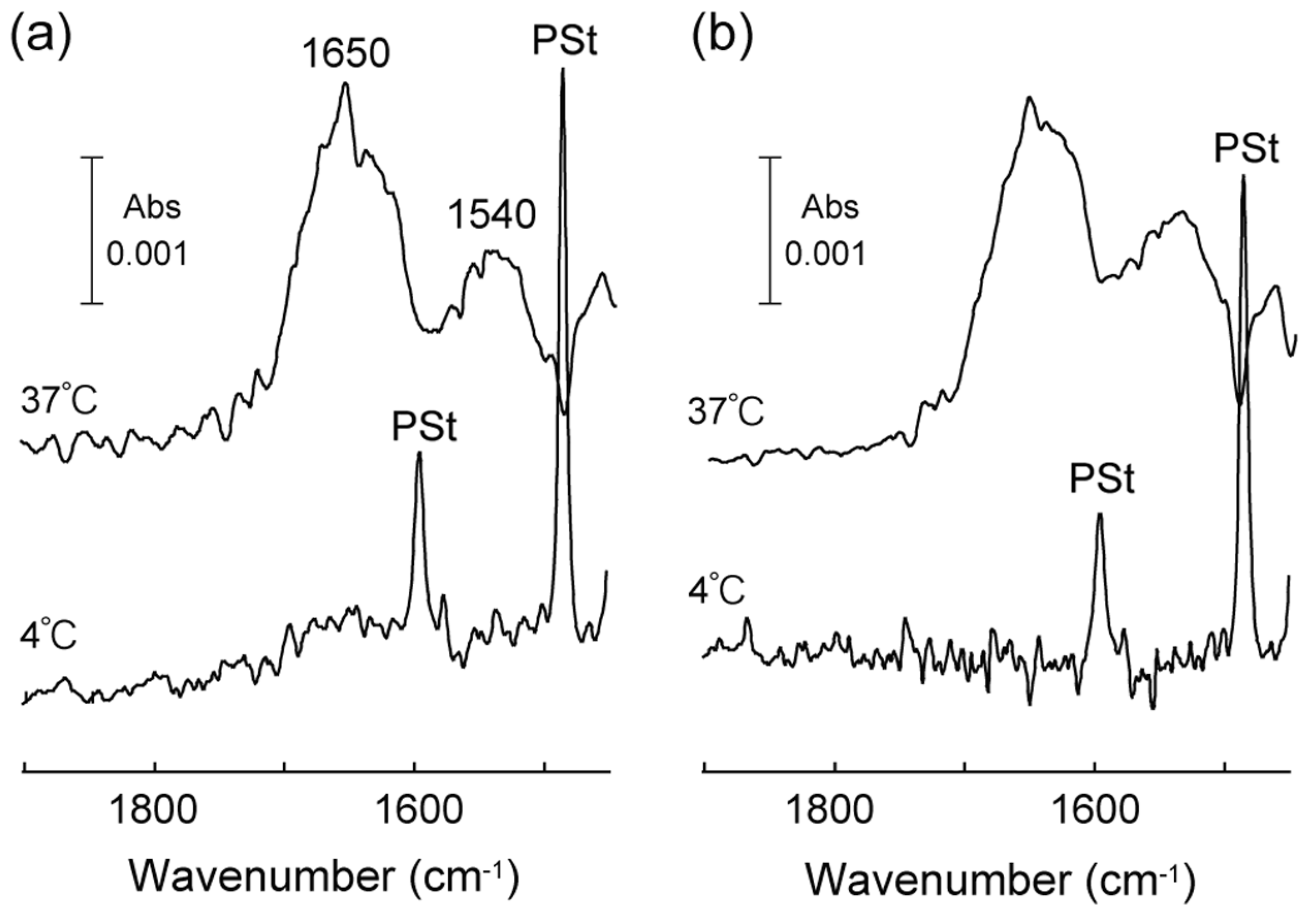
3.3. Cell Adhesive Properties of Self-Assembled Oligo(ELP) Layers and Cell Recovery
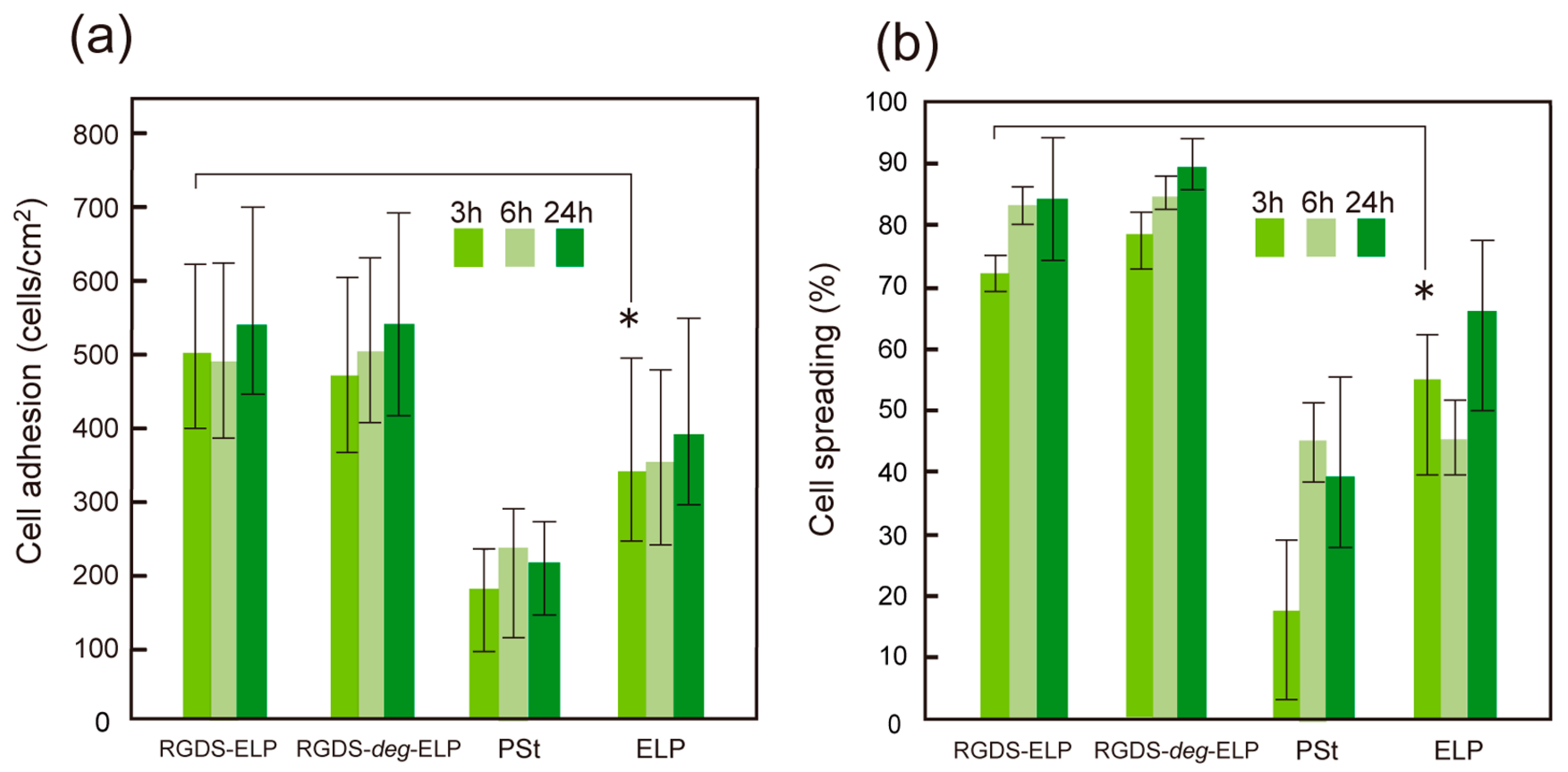

4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stevens, M.M.; George, J.H. Exploring and engineering the cell surface interface. Science 2005, 310, 1135–1138. [Google Scholar] [CrossRef]
- Lutolf, M.P.; Hubbell, J.A. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat. Biotechnol. 2005, 23, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Ruoslahti, E. Fibronectin and its receptors. Annu. Rev. Biochem. 1988, 57, 375–413. [Google Scholar] [CrossRef] [PubMed]
- Pierschbacher, M.D.; Ruoslahti, E. Cell attachment activity of fibronectin can be duplicated by small synthetic fragments of the molecule. Nature 1984, 309, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Ruoslahti, E.M.; Pierschbacher, D. New perspectives in cell adhesion: RGD and integrins. Science 1987, 238, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Beers, K.L.; MichelleByrd, H.C.; VanderHart, D.L.; Washburn, N.R. Solid-phase ATRP synthesis of peptide−polymer hybrids. J. Am. Chem. Soc. 2004, 126, 3472–3476. [Google Scholar] [CrossRef] [PubMed]
- Tugulu, S.; Silacci, P.; Stergiopulos, N.; Klok, H.-A. RGD-functionalized polymer brushes as substrates for the integrin specific adhesion of human umbilical vein endothelial cells. Biomaterials 2007, 28, 2536–2546. [Google Scholar] [CrossRef] [PubMed]
- Petersen, S.; Loschonsky, S.; Prucker, O.; Ruhen, J.; Biesalski, M. Cell micro-arrays from surface attached peptide–polymer monolayers. Phys. Status Solid A 2009, 206, 468–473. [Google Scholar] [CrossRef]
- Koga, T.; Teraguchi, Y.; Higashi, N. Preparation of PHEMA copolymers containing cell-binding peptides as graft chains and their cell adhesive properties. Trans. Mater. Res. Soc. Jpn. 2012, 37, 533–536. [Google Scholar] [CrossRef]
- Okano, T.; Yamada, N.; Sakai, H.; Sakurai, Y. A novel recovery system for cultured cells using plasma-treated polystyrene dishes grafted with poly(N-isopropylacrylamide). J. Biomed. Mater. Res. 1993, 27, 1243–1251. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yamato, M.; Nishida, K.; Ohki, T.; Kanzaki, M.; Sekine, H.; Shimizu, T.; Okano, T. Cell delivery in regenerative medicine: The cell sheet engineering approach. J. Controll. Release 2006, 116, 193–203. [Google Scholar] [CrossRef]
- Ebara, M.; Yamato, M.; Aoyagi, T.; Kikuchi, A.; Sakai, K.; Okano, T. Temperature-responsive cell culture surfaces enable “on−off” affinity control between cell integrins and RGDS ligands. Biomacromolecules 2004, 5, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Akiyama, Y.; Yamato, M.; Okano, T. Comb-type grafted poly(N-isopropylacrylamide) gel modified surfaces for rapid detachment of cell sheet. Biomaterials 2010, 31, 7435–7443. [Google Scholar] [CrossRef] [PubMed]
- Urry, D.W. Molecular machines: How motion and other functions of living organisms can result from reversible chemical changes. Angew. Chem. Int. Ed. 1993, 32, 819–841. [Google Scholar] [CrossRef]
- Urry, D.W. Free energy transduction in polypeptides and proteins based on inverse temperature transitions. Prog. Biophys. Mol. Biol. 1992, 57, 23–57. [Google Scholar] [CrossRef] [PubMed]
- Meyer, D.E.; Chilkoti, A. Quantification of the effects of chain length and concentration on the thermal behavior of elastin-like polypeptides. Biomacromolecules 2004, 5, 846–851. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.R.; Conticello, V.P. Self-assembly of block copolymers derived from elastin-mimetic polypeptide sequences. Adv. Drug Deliv. Rev. 2002, 54, 1057–1073. [Google Scholar] [CrossRef] [PubMed]
- Chilkoti, A.; Dreher, M.R.; Meyer, D.E. Design of thermally responsive, recombinant polypeptide carriers for targeted drug delivery. Adv. Drug Deliv. Rev. 2002, 54, 1093–1111. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Yang, D.S.C.; Trabbic-Carlson, K.; Kim, B.; Chilkoti, A.; Filipe, C.D.M. Self-cleavable stimulus responsive tags for protein purification without chromatography. J. Am. Chem. Soc. 2005, 127, 11228–11229. [Google Scholar] [CrossRef] [PubMed]
- Herrero-Vanrell, R.; Rincón, A.C.; Alonso, M.; Reboto, V.; Molina-Martinez, I.T.; Rodríguez-Cabello, J.C. Self-assembled particles of an elastin-like polymer as vehicles for controlled drug release. J. Controll. Release 2005, 102, 113–122. [Google Scholar] [CrossRef]
- Heilshorn, S.C.; Liu, J.C.; Tirrell, D.A. Cell-binding domain context affects cell behavior on engineered proteins. Biomacromolecules 2005, 6, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Mie, M.; Mizushima, Y.; Kobatake, E. Novel extracellular matrix for cell sheet recovery using genetically engineered elastin-like protein. J. Biomed. Mater. Res. B 2008, 86B, 283–290. [Google Scholar] [CrossRef]
- Na, K.; Jung, J.; Kim, O.; Lee, J.; Lee, T.G.; Park, Y.H.; Hyun, J. “Smart” biopolymer for a reversible stimuli-responsive platform in cell-based biochips. Langmuir 2008, 24, 4917–4923. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.; Custodio, C.A.; Testera, A.M.; Arias, F.J.; Rodríguez-Cabello, J.C.; Alves, N.M.; Mano, J.F. Stimuli-responsive thin coatings using elastin-like polymers for biomedical applications. Adv. Funct. Mater. 2009, 19, 3210–3218. [Google Scholar] [CrossRef]
- Haghpanah, J.S.; Yuvienco, C.; Civay, D.E.; Babara, H.; Baker, P.J.; Khapli, S.; Voloshchuk, N.; Gunasekar, S.K.; Muthukumer, M.; Montclare, J.K. Artificial protein block copolymers blocks comprising two distinct self-assembling domains. Chembiochem 2009, 10, 2733–2735. [Google Scholar] [CrossRef] [PubMed]
- Pierna, M.; Santos, M.; Arias, F.J.; Alonso, M.; Rodríguez-Cabello, J.C. Efficient cell and cell-sheet harvesting based on smart surfaces coated with a multifunctional and self-organizing elastin-like recombinamer. Biomacromolecules 2013, 14, 1893–1903. [Google Scholar] [CrossRef] [PubMed]
- Koga, T.; Nishiuma, T.; Higashi, N. Thermo-responsive polymer micelle triggered by conformational switch of elastin-like peptides. Kobunshi Ronbunshu 2010, 67, 679–685. [Google Scholar] [CrossRef] [Green Version]
- Higashi, N.; Ochiai, T.; Kanazawa, C.; Koga, T. Site-specific adsorption of gold nanoparticles coated with thermo-responsive peptides. Polym. J. 2013, 45, 523–528. [Google Scholar] [CrossRef] [Green Version]
- Koga, T.; Iimura, M.; Higashi, N. Novel peptide-shelled dendrimer with dramatically changeable thermo-responsive character. Macromol. Biosci. 2012, 12, 1043–1047. [Google Scholar] [CrossRef] [PubMed]
- Higashi, N.; Yasufuku, K.; Matsuo, Y.; Matsumoto, T.; Koga, T. Thermo-responsive multilayer films from ionic polymers with elastin-like peptide as graft chains. Colloid Interface Sci. Commun. 2014, 1, 50–53. [Google Scholar] [CrossRef]
- Reiersen, H.; Clarke, A.R.; Rees, A.R. Short elastin-like peptides exhibit the same temperature-induced structural transitions as elastin polymers: Implications for protein engineering. J. Mol. Biol. 1998, 283, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Nuhn, H.; Klok, H.-A. Secondary structure formation and LCST behavior of short elastin-like peptides. Biomacromolecules 2008, 9, 2755–2763. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koga, T.; Nakamoto, K.; Odawara, K.; Matsuoka, T.; Higashi, N. Fabrication of Thermo-Responsive Molecular Layers from Self-Assembling Elastin-Like Oligopeptides Containing Cell-Binding Domain for Tissue Engineering. Polymers 2015, 7, 134-146. https://doi.org/10.3390/polym7010134
Koga T, Nakamoto K, Odawara K, Matsuoka T, Higashi N. Fabrication of Thermo-Responsive Molecular Layers from Self-Assembling Elastin-Like Oligopeptides Containing Cell-Binding Domain for Tissue Engineering. Polymers. 2015; 7(1):134-146. https://doi.org/10.3390/polym7010134
Chicago/Turabian StyleKoga, Tomoyuki, Kazuhiro Nakamoto, Koji Odawara, Tomoo Matsuoka, and Nobuyuki Higashi. 2015. "Fabrication of Thermo-Responsive Molecular Layers from Self-Assembling Elastin-Like Oligopeptides Containing Cell-Binding Domain for Tissue Engineering" Polymers 7, no. 1: 134-146. https://doi.org/10.3390/polym7010134
APA StyleKoga, T., Nakamoto, K., Odawara, K., Matsuoka, T., & Higashi, N. (2015). Fabrication of Thermo-Responsive Molecular Layers from Self-Assembling Elastin-Like Oligopeptides Containing Cell-Binding Domain for Tissue Engineering. Polymers, 7(1), 134-146. https://doi.org/10.3390/polym7010134