Abstract
A novel gel undergoes the Belousov-Zhabotinsky (BZ) reaction in strong-acid-free conditions. Under such conditions, the gel can switch the BZ reaction on or off in conventional self-oscillating gels that undergo self-oscillation only in aqueous solutions with strong acids, such as HNO3 or H2SO4. The self-oscillation of the polymer chain can be controlled by varying the temperature, owing to its thermoresponsive property. Moreover, the polymer chain undergoes viscosity self-oscillations in strong-acid-free conditions. In this review, the direct observation of self-oscillations in polymer chains attached to glass or gold surfaces, by using scanning probe microscopy and quartz crystal microbalances with dissipation monitoring, is discussed.
1. Introduction
Electroactive polymers (EAPs) are materials that change their shape when an external electric input is switched on or off. EAPs can be easily cut and manipulated because of their soft physical properties and are the subject of ever increasing attention because of their applications in soft robots, soft actuators, artificial muscles, and sensors [1,2,3,4,5,6,7,8,9,10,11,12,13]. Two types of EAPs exist: electronic EAPs, driven by Coulombic interactions including ferroelectric forces, dielectric forces, and electrostatics; and ionic EAPs, which change their shape by moving ions between two electrodes because the electric stimuli cause different electric potentials between the two electrodes. To generate strong forces and large deformations, electric EAPs require the application of high voltages. In contrast, ionic EAPs are structurally altered by low voltages (<5 V); however, the force applied to ionic EAPs is lower than that in electronic EAPs. In the area of robotics, EAPs have been envisaged as functional soft actuators in the realization of soft robots, which are flexible, lightweight, and consume small amounts of electrical energy. However, to drive soft robots and actuators composed of EAPs, an external device to provide the appropriate electric stimulus is indispensable. The total system including external input devices, power cords, and an energy source is significantly larger than the actuator itself.
In contrast, all living organisms involve the isothermal conversion of chemical energy into mechanical work; biological systems are significantly more efficient compared to soft robots constructed from EAPs. To construct autonomous soft robots comparable to living organisms, self-oscillating polymer systems have been developed [14,15,16]. The energy source for self-oscillating polymer systems is the Belousov-Zhabotinsky (BZ) reaction. The BZ reaction is well known for exhibiting temporal and spatiotemporal oscillating phenomena under unstirred and constant temperature conditions [17,18,19,20,21,22,23]. The BZ reaction has been intensively studied as a simple model for the formation of spatiotemporal structures. The overall BZ reaction process consists of the oxidation of an organic substrate by an oxidizing agent in the presence of a metal catalyst under strongly acidic conditions. In the BZ reaction, the metal catalyst undergoes periodic redox self-oscillation, with a concurrent change in the solubility of the metal catalyst. The self-oscillating polymer system covalently bonded to the metal catalyst undergoes autonomous volume changes induced by the BZ reaction, owing to periodic changes in the solubility of the metal catalyst moiety. Self-oscillating polymer systems are of two types: self-oscillating polymer chains [15] and self-oscillating polymer gels [16]. These polymer systems are composed of N-isopropylacrylamide (NIPAAm) and [ruthenium(4-vinyl-4’-methyl-2, 2-bipyridine)bis(2,2’-bipyridine)bis(hexafluorophosphate)] (Ru(bpy)3). The self-oscillating polymer gel undergoes a swelling-deswelling self-oscillation, and the self-oscillating polymer chains cause aggregation-disaggregation self-oscillations at constant temperatures. The operation of the self-oscillating polymer system requires three BZ substrates: malonic acid (MA), sodium bromate (NaBrO3), and nitric or sulfuric acid. The operating conditions of the self-oscillating polymer system are, therefore, limited to strongly acidic conditions, with the attendant safety risks arising through the use of strong acids. To reduce the safety risks in such processes, Hara et al. attempted to devise an autonomous polymer system that underwent self-oscillation under strong-acid-free conditions. In their report, as the first step towards achieving a self-oscillating polymer system, acrylamide-2-methylpropane sulfonic acid (AMPS) was incorporated into a conventional self-oscillating polymer chain (poly(NIPAAm-co-Ru(bpy)3)) as a pH control site [24,25]. The AMPS-containing self-oscillating polymer chain underwent the soluble-insoluble self-oscillation under strong-acid-free conditions. Subsequently, methacrylamide propyl trimethyl ammonium chloride (MAPTAC) with a positively charged group was incorporated into poly(NIPAAm-co-Ru(bpy)3) as the release site of an anionic oxidizing agent (bromate ion) [26]; the bromate ion was introduced into the MAPTAC-containing polymer via an ion-exchange process. The MAPTAC-containing self-oscillating polymer system underwent self-oscillation under oxidant-free conditions. The AMPS and MAPTAC moieties were then simultaneously incorporated in the poly(NIPAAm-co-Ru(bpy)3) as the pH-control and oxidant supply sites, respectively [27]. The novel self-oscillating polymer chain facilitated the aggregation-disaggregation self-oscillation when only the MA solution was added, however a self-oscillating polymer gel that undergoes the BZ reaction under strong-acid-free conditions did not develop; the development of a new strong acid-free gel is therefore necessary.
This review discusses a novel gel that undergoes the BZ reaction under strong-acid-free conditions [28]. Under strong-acid-free conditions, the novel gel can control the on/off switching of the BZ reaction in conventional self-oscillating (poly(NIPAAm-co-Ru(bpy)3-co-AMPS) gels that require a strong acid. Autonomous polymer systems containing pH control sites with a high potential for controlling self-oscillating behavior [29,30,31,32,33] are introduced. Information about the detailed motion of the self-oscillating polymer chain is of great significance for the design of autonomous molecular robot systems; techniques for the direct observation of self-oscillating polymer chains, such as scanning probe microscopy (SPM) and a quartz crystal microbalance with dissipation (QCM-D) [34,35] are introduced.
2. Strong-Acid-Free Gel
In this section, the self-oscillating behavior of the strong-acid-free gel is discussed [28]. The strong-acid-free gel possesses a semi-interpenetrating polymer network (IPN) structure; the first network consists of a poly(AMPS) gel, while the second network comprises a self-oscillating polymer chain (poly(NIPAAm-co-Ru(bpy)3-co-AMPS)). The red-green-blue (RGB) analysis of the strong-acid-free gel is shown in Figure 1A. The green and red self-oscillations occur distinctly in strong-acid-free conditions. This result indicated that the BZ reaction occurred in the novel gel without the direct addition of strong acid. The period of the self-oscillation as a function of the BZ substrate (MA) concentration demonstrated a linear relationship, as shown in Figure 1B. The logarithmic plot was fitted to a linear equation and expressed as a[substrate]b, where a and b are experimental constants, and the square brackets indicate the initial substrate concentration. This result demonstrated that the self-oscillation period was similar to normal self-oscillating polymer systems containing high H+ concentrations [36,37,38].
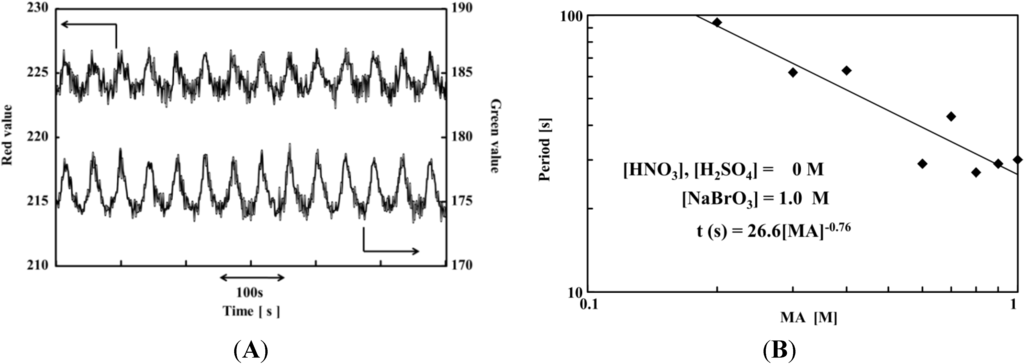
Figure 1.
(A) Red-green-blue (RGB) analysis of the strong-acid-free gel ([NaBrO3] = 1.2 M, [MA] = 0.6 M); (B) Logarithmic plot of period (t in s) vs. initial malonic acid (MA) concentration at [NaBrO3] = 1.0 M. (Reproduced with permission from [28], Copyright 2013, American Chemical Society).
Figure 2A shows a schematic illustration of the connected/disconnected experimental setup under strong-acid-free conditions; in the case of the connected experiment, the two gels were pressed together by silicone rubber. The RGB analysis of the strong-acid-free gel and the normal self-oscillating gel shows that the chemical wave in the normal gel was only observed when the two gels were connected. In contrast, when the novel gel was detached from the normal gel, the BZ reaction stopped in the normal gel, as the normal gel could not produce the immediate substrates for the BZ reaction owing to the high pH. Utilizing this phenomenon, we can control the on/off switching of the self-oscillation in the normal gel by manipulating the adhesion state of the two types of gel in strong-acid-free conditions. This phenomenon is useful for the construction of autonomous soft robots and actuators in safe conditions. The normal gel that was not attached to the novel gel was placed in the same solution as shown in Figure 2A as a control experiment. There was no observation of self-oscillating behavior in the control normal gel in the connected and disconnected state; the on/off switching of the BZ reaction in the gel was caused by the adhesion of the strong-acid-free gel to the normal gel in strong-acid-free conditions [28].
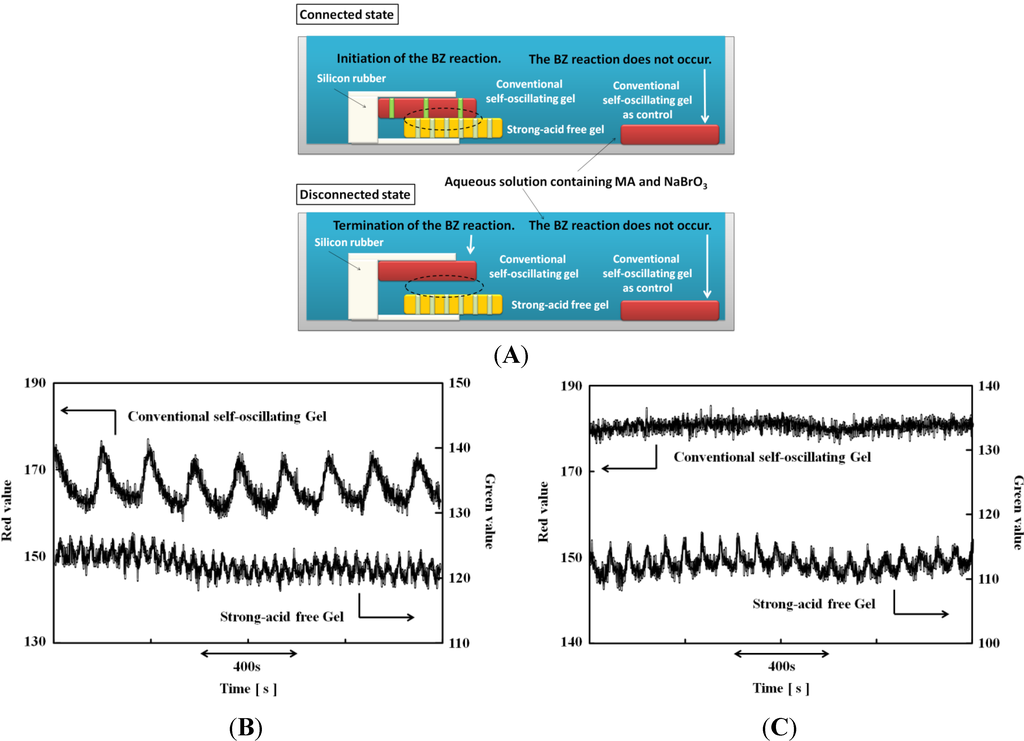
Figure 2.
(A) Schematic illustration of the experimental setup for the Belousov-Zhabotinsky (BZ) reaction in strong-acid-free conditions, regulated via connection/disconnection of the gels; (B) RGB analysis of the connected gels; (C) RGB analysis of the disconnected gels ([NaBrO3] = 1.0 M, [MA] = 0.5 M). (Reprinted with permission from [28], Copyright 2013, American Chemical Society).
3. On/Off Switching of the AMPS-Containing Self-Oscillating Polymer Chain
In this section, the on/off switching of the self-oscillation in the polymer chain (AMPS-containing self-oscillating polymer chain) by changing the external temperature is introduced [29]. The AMPS-containing polymer chain was composed of NIPAAm, Ru(bpy)3, and AMPS (See Figure 3). The lower critical solution temperature (LCST) of the AMPS-containing polymer chain with 50 wt% AMPS moiety in the reduced state was 14 °C [29]. In contrast, there was no observation of the LCST of the polymer chain in the oxidized state. These measurements demonstrated that the solubility of the AMPS-containing polymer chain in the reduced and oxidized states was significantly different because the LCST is determined by the solubility of the polymer chain. This different solubility of the AMPS-containing polymer chain in the reduced and oxidized states was the driving force for the soluble-insoluble self-oscillation induced by the BZ reaction.
Figure 4 presents a phase diagram of the temperature (12–36 °C) and the concentration of sodium bromate (NaBrO3) for the self-oscillation of AMPS-containing solutions at a fixed MA concentration (0.1 M). As shown in Figure 4, the self-oscillation period of the AMPS-containing solution was controlled by changing the temperature and the sodium bromate concentration.
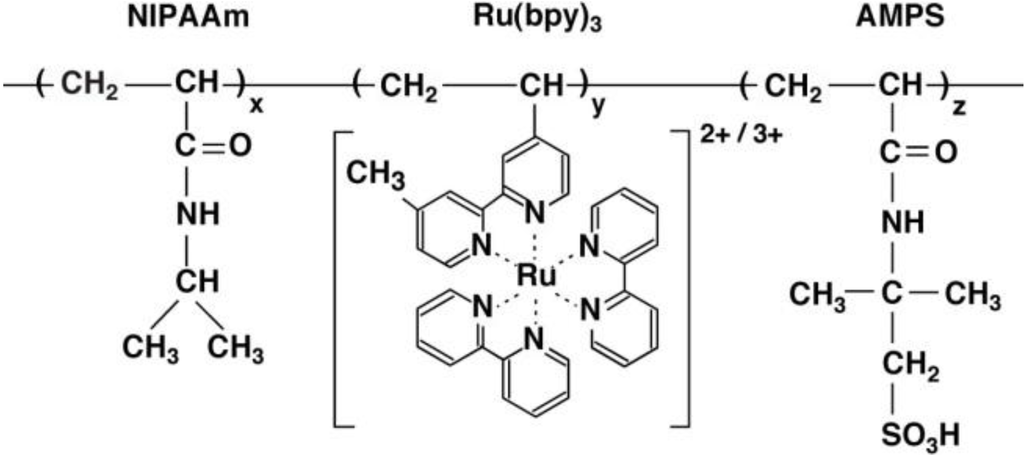
Figure 3.
Chemical structure of poly(NIPAA-co-Ru(bpy)3-co-AMPS).
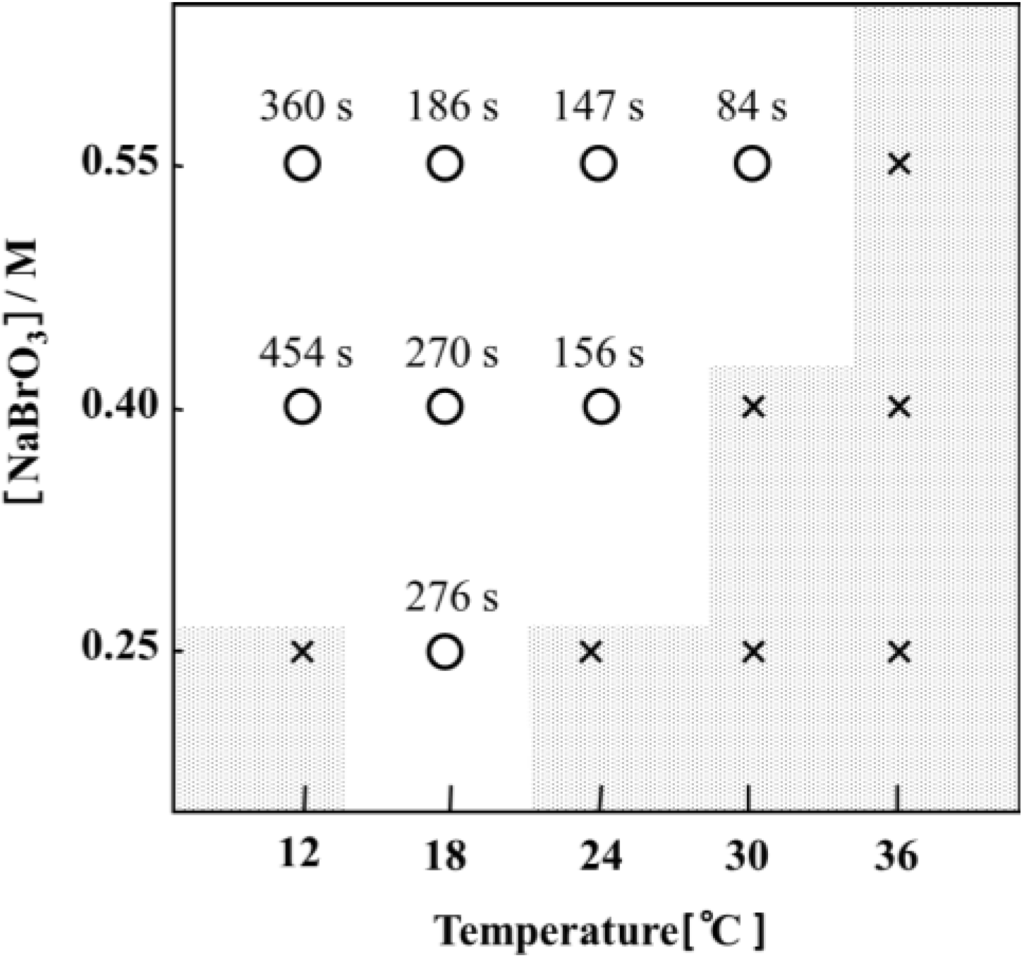
Figure 4.
Phase diagram of the oscillating region given by the initial NaBrO3 concentration and temperature at a fixed MA concentration (0.1 M). The number above the plot denotes the oscillation period. A cross indicates that no oscillation occurred. Polymer solutions were prepared by dissolving the polymer (2.0 wt%) into an aqueous solution containing the two BZ substrates (MA and NaBrO3). The transmittance at 570 nm as a function of time was monitored by a spectrophotometer. (Reprinted with permission from [29], Copyright 2005, American Chemical Society).
The soluble-insoluble self-oscillation of the AMPS-containing polymer chain can be switched by controlling the external temperatures. As shown in Figure 5, the self-oscillation stopped at 36 °C because the LCST of the AMPS-containing polymer chain in the reduced state is 14 °C; the polymer chain aggregated above the LCST. In the BZ reaction, the time in the reduced state is much longer than that in the oxidized state; the self-oscillating behavior of the polymer chain is dominated by the solubility of the polymer chain in the reduced state. As shown in Figure 5A, the polymer chain does not oscillate when the temperature is decreased to 12 °C. At this specific set of BZ substrate conditions ([MA] = 0.1 M and [NaBrO3] = 0.25 M), the self-oscillation only occurs at 18 °C. In contrast, when [NaBrO3] was 0.40 and 0.55 M at fixed MA concentration (0.1 M), the self-oscillation began when the temperature was decreased to 12 °C. As shown in Figure 5, transmittance dropped before 36 °C owing to the temperature changing process. Furthermore, the transmittance value (the baseline) of the self-oscillation at 12 °C was different in Figure 5B,C; the transmittance value of the self-oscillating polymer chain was greatly affected by the ionic strength of the solution [38]. In high ionic strength conditions, the solubility of the polymer chain decreased, with a concomitant decrease in transmittance. Therefore, the baseline of the transmittance of the self-oscillation at [NaBrO3] = 0.40 M was higher than that at [NaBrO3] = 0.55 M. In addition, the amplitude of the self-oscillation at [NaBrO3] = 0.55 M was higher than that at [NaBrO3] = 0.40 M; this observation is explained by the Field-Koros-Noyes (FKN) mechanism [39,40]. The FKN mechanism divides the BZ reaction into three main processes: the consumption of Br− ions (process A), the autocatalytic formation of HBrO2 (process B), and the formation of Br− ions (process C).
A: BrO3− + 2 Br− + 3 H+ →3 HOBr
B: BrO3− + HBrO2 + 2 Mred + 3 H+ →2 HBrO2 + 2 Mox + H2O
C: 2 Mox + MA + BrMA→f Br− + 2 Mred + other products
Mox and Mred represent the metal catalyst for the BZ reaction in the oxidized and reduced states, respectively. In process B, the Ru(bpy)3 is oxidized; in contrast, the oxidized metal catalyst moiety is reduced in process C. The high concentration of NaBrO3 increases the mole fraction of the Ru(bpy)33+ moiety, which is rationalized by process B. The amplitude of the self-oscillation is determined by the mole fraction of the Ru(bpy)33+ moiety because the solubility of the polymer is determined by the concentration of the oxidized Ru moiety [35]; the amplitude of the self-oscillation is higher when the concentration of sodium bromate is increased.
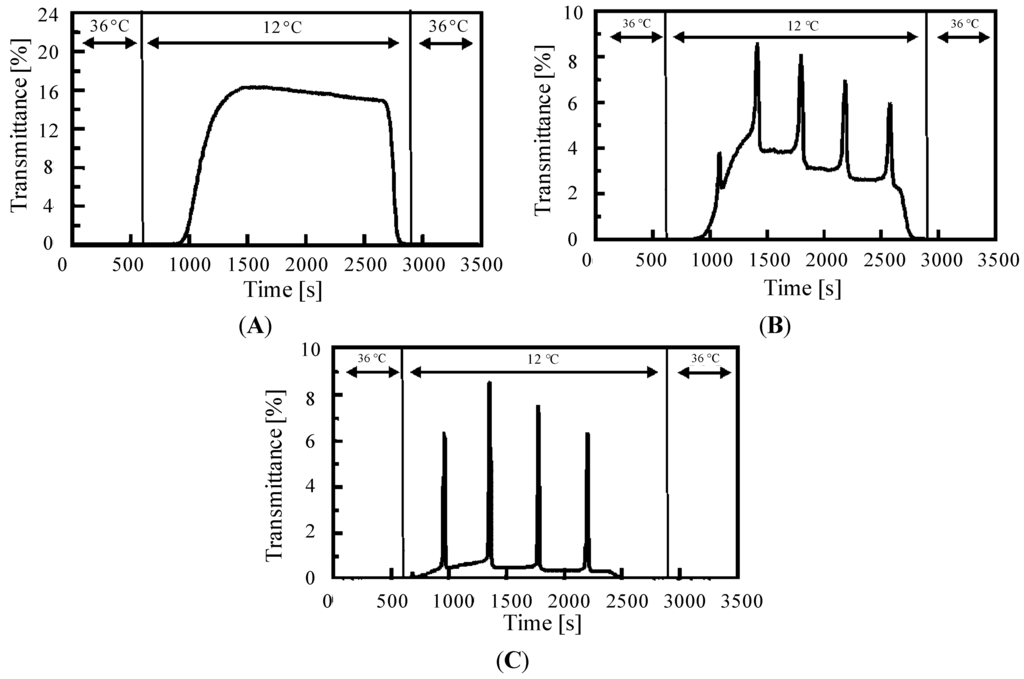
Figure 5.
Oscillating profiles of the optical transmittance for AMPS-containing solutions (2.0 wt%) in response to stepwise temperature changes between 12 and 36 °C for several substrate concentrations; (A) [MA] = 0.1 M and [NaBrO3] = 0.25 M; (B) [MA] = 0.1 M and [NaBrO3] = 0.40 M; and (C) [MA] = 0.1 M and [NaBrO3] = 0.55 M. (Reprinted with permission from [29], Copyright 2005, American Chemical Society).
4. Viscosity Self-Oscillation of the AMPS-Containing Polymer Solution
In this section, the viscosity self-oscillating behavior of the AMPS-containing polymer solution is discussed [30]. Figure 6A shows the viscosity self-oscillation of the 12 wt% AMPS-containing polymer solution at constant temperature (12 °C). The viscosity increased rapidly due to the carbon dioxide (CO2) generated by the BZ reaction in the polymer solution; a large amount of CO2 bubbles were observed in the AMPS-containing polymer solution. When the BZ reaction stopped, the viscosity decreased. As the AMPS-containing polymer solution generated CO2, the hydrophobic Ru moieties in the polymer chain gathered at the liquid-gas interface [30]. The polymer chain in contact with the liquid-gas interface become more rigid, and the viscosity increased. In the case of the 12 wt% polymer solution, the amplitude of the viscosity self-oscillation was about 4 mPa·s. When the temperature was increased (15, 18, 21, 24, and 27 °C), viscosity self-oscillation for the 12 wt% polymer solution was not observed. In contrast, the 6 wt% polymer solution underwent viscosity self-oscillation in the temperature range 12–24 °C, as shown in Figure 6B. Above 27 °C, viscosity self-oscillation was not observed. Figure 6C shows the period of the viscosity self-oscillation for the 6 wt% polymer solution; the period decreased with increasing temperature because the self-oscillation of the polymer solution was in accordance with the Arrhenius equation [36,37,38].
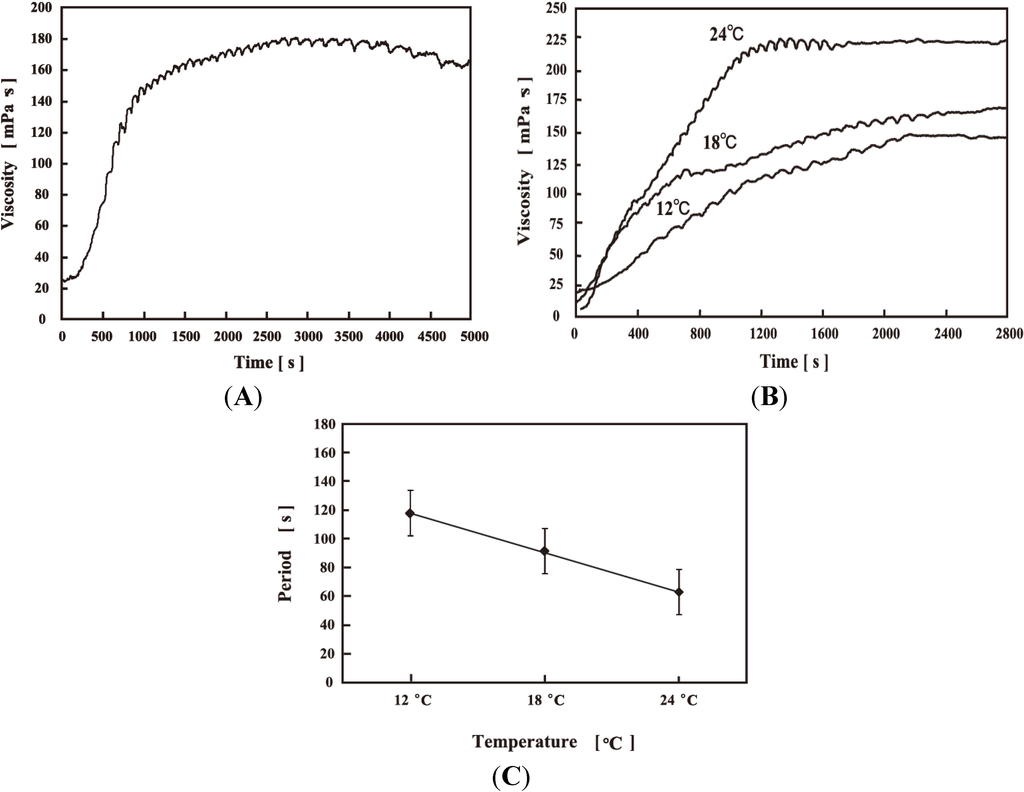
Figure 6.
(A) Viscosity self-oscillation of 12 wt% poly(NIPAAm-co-Ru(bpy)3-co-AMPS) solution at 12 °C; (B) Viscosity self-oscillating behavior of 6 wt% polymer solution; (C) Viscosity self-oscillation period of 6 wt% polymer solutions at different temperatures. (Reprinted with permission from [30], Copyright 2008, American Institute of Physics).
5. Direct Observation of the Autonomous Oscillation of the Single Polymer Chain
The direct observation of polymer chain self-oscillation was recently achieved by using SPM and QCM-D [34,35]. To measure self-oscillating behavior via SPM and QCM-D, polymer chains containing an anchoring unit to solid surfaces (glass and gold surfaces) were synthesized. The self-oscillating polymer chain was composed of NIPAAm, Ru(bpy)3, and an N-succimidyl group to tether the polymer to the selected surface, as shown in Figure 7.
The polymer chain incorporating the anchoring site was immobilized on an aminosilane-coupled glass plate (see Figure 7). The attached polymer chain underwent self-oscillation only in aqueous solution including the three BZ substrates (see Figure 8A,B); self-oscillation in the control solution without three BZ substrates was not observed, as the BZ reaction did not occur. As shown in Figure 8A, the amplitude and period of the self-oscillation of the polymer chain chemically bonded to the glass plate was unstable because the solution was not stirred. In addition, the self-oscillation of the polymer chain does not cause damping; while the polymer chain was covalently bonded to the glass surface, the polymer chain does not aggregate, and there is no observation of damping behavior in the SPM measurements. Dynamic light scattering (DLS) experiments for the anchored polymer chain (in both reduced and oxidized states) were conducted in an aqueous solution including 0.3 M HNO3 solutions containing 1 mM Ce2(SO4)3 or 1 mM Ce(SO4)2. The cumulant diameter of the polymer chain in the reduced and oxidized states was 23.9 and 59.6 nm, respectively, demonstrating that the conformation of the polymer chain was greatly affected by the oxidation state of the Ru moiety. The SPM measurements therefore detected the self-oscillation on the glass surface in an aqueous solution including the three BZ substrates.
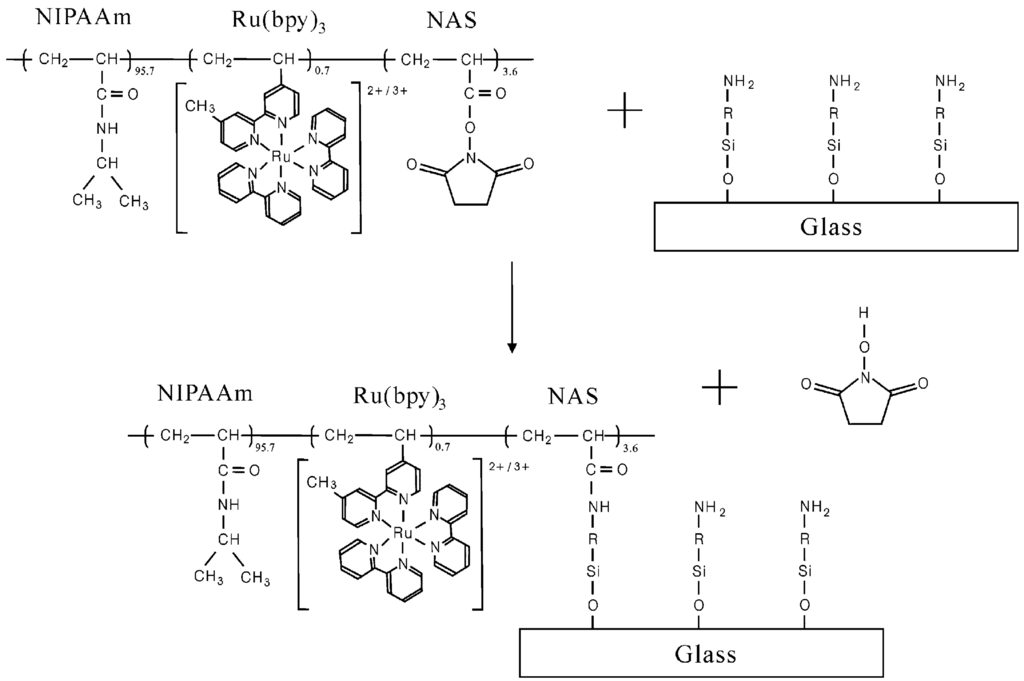
Figure 7.
Preparation of self-oscillating polymers on glass surfaces. (Reprinted with permission from [34], Copyright 2006, American Chemical Society).
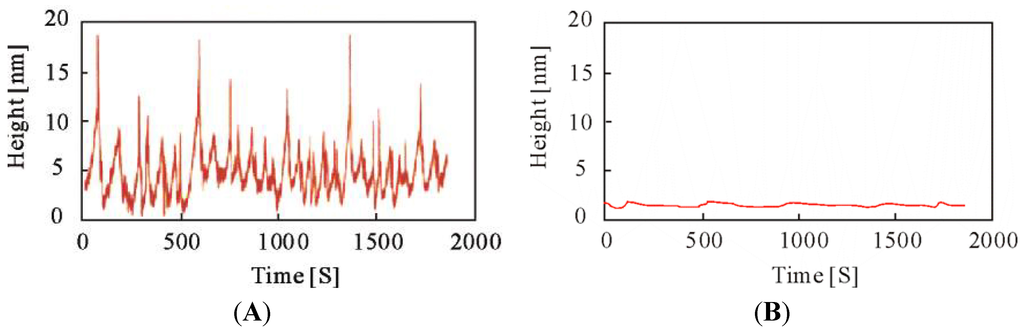
Figure 8.
(A) Self-oscillating behavior of a polymer chain on a glass surface detected by SPM in aqueous solution including MA (0.1 M), NaBrO3 (0.3 M), and HNO3 (0.3 M); (B) Behavior of the polymer chain on the glass surface in aqueous solution without three BZ substrates. (Reprinted with permission from [34], Copyright 2006, American Chemical Society).
The time dependence of the change in the resonance frequency (Δf) of the self-oscillating polymer chain was measured using QCM-D at constant temperature (20 °C) [35]. The self-oscillation of Δf originates in the adsorption-desorption self-oscillation of water molecules on the polymer chain. In the BZ reaction, the formation of the polymer chain undergoes the self-oscillation as shown by SPM measurements (See Figure 8). The water molecules around the polymer chain attached or detached synchronously with the change in the polymer chain’s conformation, because of the concurrent solubility changes of the polymer. The period of Δf was stable and approximately 400 s (see Figure 9B).
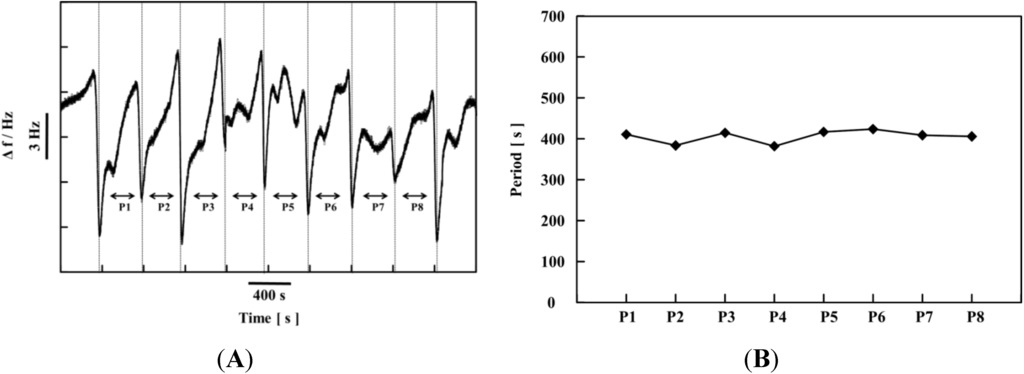
Figure 9.
(A) Self-oscillation at the resonance frequency (Δf) for fixed initial concentrations of the BZ substrates ([MA] = 0.1 M, [HNO3] = 0.3 M, and [NaBrO3] = 0.5 M) at 20 °C; (B) Period of self-oscillation of the resonance frequency (Δf). (Reprinted with permission from [35], Copyright 2013, American Chemical Society).
Figure 10A shows the time-dependent changes in Δf and dissipation (ΔD) that reflect the changes in the conformation of the polymers attached to gold surfaces. The D factor is defined as follows:
D = Edissipated/2πEstored
Edissipated is the energy dissipated during one oscillation, and Estored is the energy stored in the oscillating system [41]. As shown in Figure 10A, ΔD undergoes the self-oscillation in synchronization with the Δf self-oscillation. When Δf decreases due to the adsorption of water molecules onto the polymer chain, ΔD increases because the flexibility of the polymer chain increases [35]. Conversely, as Δf increases, because of the detachment of the water molecules from the polymer chain attached to the gold surface, ΔD decreases due to the increased rigidity of the polymer chains. Figure 10B shows the relationship between Δf and ΔD. The shape indicates that the frequencies of the Δf and ΔD self-oscillations are identical, but differ in phase and amplitude. The size of the ellipse is different in self-oscillations 1, 2, and 3 because the rates of the oxidation and reduction of the Ru moiety in the BZ reaction are different in each cycle.
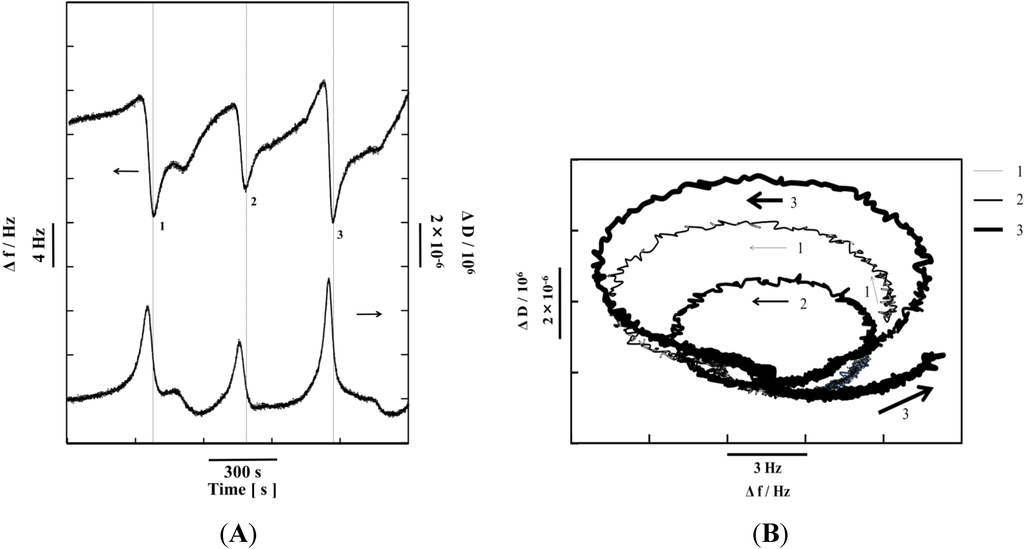
Figure 10.
(A) Time dependence of the resonance frequency (Δf) and energy dissipation (ΔD); (B) Plots of ΔD against Δf for the self-oscillating polymer chain during the BZ reaction attached to gold surfaces. The notations 1, 2, and 3 correspond to part (A). (Reprinted with permission from [35], Copyright 2013, American Chemical Society).
6. Conclusions
This review demonstrates that novel gels can control the on/off switching of the BZ reaction in the conventional self-oscillating gel in strong-acid-free conditions. In addition, the self-oscillation of the AMPS-containing polymer chain can be controlled by changing the external temperature. These on/off switching functions are of significance in the control of autonomous actuators and molecular robots under safe conditions. Moreover, viscosity self-oscillations of the polymer chain in strong-acid-free conditions were observed, which were significantly affected by the temperature and the concentration of the polymer chain. The direct observation of self-oscillations in single polymer chains attached to glass surfaces was achieved via SPM measurements, clarifying that polymer chains on such surfaces undergo self-oscillations induced by the BZ reaction. Furthermore, by using QCM-D, it was demonstrated that the water adsorption-desorption self-oscillation on the polymer chain occurs in synchronization with conformational changes in the polymer chain.
Acknowledgments
This work was carried out under the auspices of the New Energy and Industrial Technology Development Organization (NEDO) of Japan under the Industrial Technology Research Grant Program in 2011. We were also supported by Grants-in-Aid (KAKENHI) for Challenging Exploratory Research (24656178).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Oguro, K.; Kawami, Y.; Takenaka, H. An Actuator Element of Polyelectrolyte Gel Membrane-Electrode Composite. Bull. Gov. Ind. Res. Inst. Osaka 1992, 43, 21–24. [Google Scholar]
- Baughman, R.H.; Cui, C.; Zakhidov, A.A.; Iqbal, Z.; Barisci, J.N.; Spinks, G.M.; Wallace, G.G.; Mazzoldi, A.; de Rossi, D.; Rinzler, A.G.; et al. Carbon Nanotube Actuators. Science 1999, 284, 1340–1344. [Google Scholar] [CrossRef]
- Pelrine, R.; Kornbluh, R.; Pei, Q.; Joseph, J. High-Speed Electrically Actuated Elastomers with Strain Greater Than 100%. Science 2000, 287, 836–839. [Google Scholar] [CrossRef]
- Bar-Cohen, Y. EAP Actuators as Artificial Muscles—Reality, Potential, and Challenges; International Society for Optical Engineering (SPIE): Bellingham, WA, USA, 2001; pp. 4–44. [Google Scholar]
- Fukushima, T.; Asaka, K.; Kosaka, A.; Aida, T. Fully Plastic Actuator through Layer-by-Layer Casting with Ionic-Liquid-Based Bucky Gel. Angew. Chem. 2005, 117, 2462–2465. [Google Scholar] [CrossRef]
- Mukai, K.; Asaka, K.; Sugino, T.; Kiyohara, K.; Takeuchi, I.; Terasawa, N.; Futaba, D.N.; Hata, K.; Fukushima, T.; Aida, T. Highly Conductive Sheets from Millimeter-Long Single-Walled Carbon Nanotubes and Ionic Liquids: Application to Fast-Moving, Low-Voltage Electromechanical Actuators Operable in Air. Adv. Mater. 2009, 21, 1582–1585. [Google Scholar] [CrossRef]
- Zhang, Q.M.; Li, H.; Poh, M.; Xia, F.; Cheng, Z.-Y.; Xu, H.; Huang, C. An All-Organic Composite Actuator Material with a High Dielectric Constant. Nature 2002, 419, 284–287. [Google Scholar] [CrossRef]
- Wissler, M.; Mazza, E. Modeling of a Pre-strained Circular Actuator Made of Dielectric Elastomer Actuators. Sens. Actuators A 2005, 120, 184–192. [Google Scholar] [CrossRef]
- Shankar, R.; Ghosh, T.K.; Spontak, R.J. Dielectric Elastomers as Next-Generation Polymeric Actuators. Soft Matter 2007, 3, 1116–1129. [Google Scholar] [CrossRef]
- Pons, J.L. Emerging Actuator Technologies: A Micromechatronic Approach; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Smela, E.; Inganäs, O.; Lundström, I. Controlled Folding of Micrometer-Size Structures. Science 1995, 268, 1735–1738. [Google Scholar]
- Baughman, R.H. Conducting Polymer Artificial Muscles. Synth. Met. 1996, 78, 339–353. [Google Scholar] [CrossRef]
- Doi, M.; Matsumoto, M.; Hirose, Y. Deformation of Ionic Polymer Gels by Electric Fields. Macromolecules 1992, 25, 5504–5511. [Google Scholar] [CrossRef]
- Ishiwatari, T.; Kawaguchi, M.; Mitsuishi, M. Oscillatory Reactions in Polymer Systems. J. Polym. Sci. Part A Polym. Chem. 1984, 22, 2699–2704. [Google Scholar] [CrossRef]
- Yoshida, R.; Sakai, T.; Ito, S.; Yamaguchi, T. Self-Oscillation of Polymer Chains with Rhythmical Soluble-Insoluble Changes. J. Am. Chem. Soc. 2002, 124, 8095–8098. [Google Scholar] [CrossRef]
- Yoshida, R.; Takahashi, T.; Yamaguchi, T.; Ichijo, H. Self-Oscillating Gel. J. Am. Chem. Soc. 1996, 118, 5134–5135. [Google Scholar] [CrossRef]
- Zaikin, A.N.; Zhabotinsky, A.M. Concentration Wave Propagation in Two-Dimensional Liquid-Phase Self-Oscillating System. Nature 1970, 225, 535–537. [Google Scholar] [CrossRef]
- Reusser, E.J.; Field, R.J. The Transition from Phase Waves to Trigger Waves in a Model of the Zhabotinsky Reaction. J. Am. Chem. Soc. 1979, 101, 1063–1094. [Google Scholar] [CrossRef]
- Gyorgyi, L.; Turanyi, T.; Field, R.J. Mechanistic Details of the Oscillatory Belousov-Zhabotinsky Reaction. J. Phys. Chem. 1990, 94, 7162–7170. [Google Scholar] [CrossRef]
- Scott, S.K. Chemical Chaos, 1st ed.; Oxford University Press: Oxford, UK, 1991. [Google Scholar]
- Field, R.J.; Burger, M. Oscillations and Traveling Waves in Chemical Systems; John Wiley & Sons: New York, NY, USA, 1985. [Google Scholar]
- Nicolis, G.; Prigogine, I. Self-Organization in Nonequilibrium. Systems; John Wiley & Sons: New York, NY, USA, 1977. [Google Scholar]
- Murray, J.D. Mathematical Biology; Springer-Verlag: Berlin, Germany, 1990. [Google Scholar]
- Hara, Y.; Yoshida, R. Self-Oscillation of Polymer Chains Induced by the Belousov-Zhabotinsky Reaction under Acid-Free Conditions. J. Phys. Chem. B 2005, 109, 9451–9454. [Google Scholar] [CrossRef]
- Hara, Y.; Yoshida, R. Damping Behavior of Aggregation-Disaggregation Self-Oscillation for a Polymer Chain. Macromol. Rapid Commun. 2009, 30, 1656–1662. [Google Scholar] [CrossRef]
- Hara, Y.; Sakai, T.; Maeda, S.; Hashimoto, S.; Yoshida, R. Self-Oscillating Soluble−Insoluble Changes of a Polymer Chain Including an Oxidizing Agent Induced by the Belousov−Zhabotinsky Reaction. J. Phys. Chem. B 2005, 109, 23316–23319. [Google Scholar] [CrossRef]
- Hara, Y.; Yoshida, R. Self-Oscillating Polymer Fueled by Organic Acid. J. Phys. Chem. B 2008, 112, 8427–8429. [Google Scholar] [CrossRef]
- Hara, Y.; Yamaguchi, Y.; Mayama, H. Switching the BZ Reaction with a Strong-Acid-Free Gel. J. Phys. Chem. B 2014, 118, 634–638. [Google Scholar] [CrossRef]
- Hara, Y.; Yoshida, R. Control of Oscillating Behavior for the Self-Oscillating Polymer with pH-Control Site. Langmuir 2005, 21, 9773–9776. [Google Scholar] [CrossRef]
- Hara, Y.; Yoshida, R. A Viscosity Self-Oscillation of Polymer Solution Induced by the BZ Reaction under Acid-Free Conditions. J. Chem. Phys. 2008, 128, 224904. [Google Scholar] [CrossRef]
- Maeda, S.; Hara, Y.; Yoshida, R.; Hashimoto, S. Control of the Dynamic Motion of a Gel Actuator Driven by the Belousov–Zhabotinsky Reaction. Macromol. Rapid Commun. 2008, 29, 401–405. [Google Scholar] [CrossRef]
- Maeda, S.; Hara, Y.; Sakai, T.; Yoshida, R.; Hashimoto, S. Self-Walking Gel. Adv. Mater. 2007, 19, 3480–3484. [Google Scholar] [CrossRef]
- Maeda, S.; Hara, Y.; Yoshida, R.; Hashimoto, S. Peristaltic Motion of Polymer Gels. Angew. Chem. Int. Ed. 2008, 120, 6792–6795. [Google Scholar] [CrossRef]
- Ito, Y.; Hara, T.; Uetsuka, H.; Hasuda, H.; Onishi, H.; Arakawa, H.; Ikai, A.; Yoshida, R. AFM Observation of Immobilized Self-Oscillating Polymer. J. Phys. Chem. B 2006, 110, 5170–5173. [Google Scholar] [CrossRef]
- Hara, Y.; Mayama, H.; Yamaguchi, Y.; Takenaka, Y.; Fukuda, F. Direct Observation of Periodic Swelling and Collapse of Polymer Chains Induced by the Belousov–Zhabotinsky Reaction. J. Phys. Chem. B 2013, 117, 14351–14357. [Google Scholar]
- Hara, Y.; Rumana, J.A. Activation Energy of Aggregation-Disaggregation Self-Oscillation of Polymer Chains. Int. J. Mol. Sci. 2012, 13, 16281–16290. [Google Scholar] [CrossRef]
- Miyakawa, K.; Sakamoto, F.; Yoshida, R.; Yamaguchi, T.; Kokufuta, E. Chemical Waves in Self-Oscillating Gels. Phys. Rev. E 2000, 62, 793–798. [Google Scholar] [CrossRef]
- Hara, Y.; Takenaka, Y. Autonomous Oscillation of Polymer Chains Induced by the Belousov–Zhabotinsky Reaction. Sensors 2014, 14, 1497–1510. [Google Scholar] [CrossRef]
- Field, R.J.; Koros, E.; Noyes, R.M. Oscillations in Chemical Systems. II. Thorough Analysis of Temporal Oscillation in the Bromate-Cerium-Malonic Acid System. J. Am. Chem. Soc. 1972, 94, 8649–8664. [Google Scholar] [CrossRef]
- Field, R.J.; Noyes, R.M. Oscillations in Chemical Systems. IV. Limit Cycle Behavior in a Model of a Real Chemical Reaction. J. Chem. Phys. 1974, 60, 1877–1884. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, G. Periodic Swelling and Collapse of Polyelectrolyte Brushes Driven by Chemical Oscillation. J. Phys. Chem. B 2008, 112, 10137–10141. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).