Strong Screening Effect of Polyhedral Oligomeric Silsesquioxanes (POSS) Nanoparticles on Hydrogen Bonded Polymer Blends
Abstract
:1. Introduction
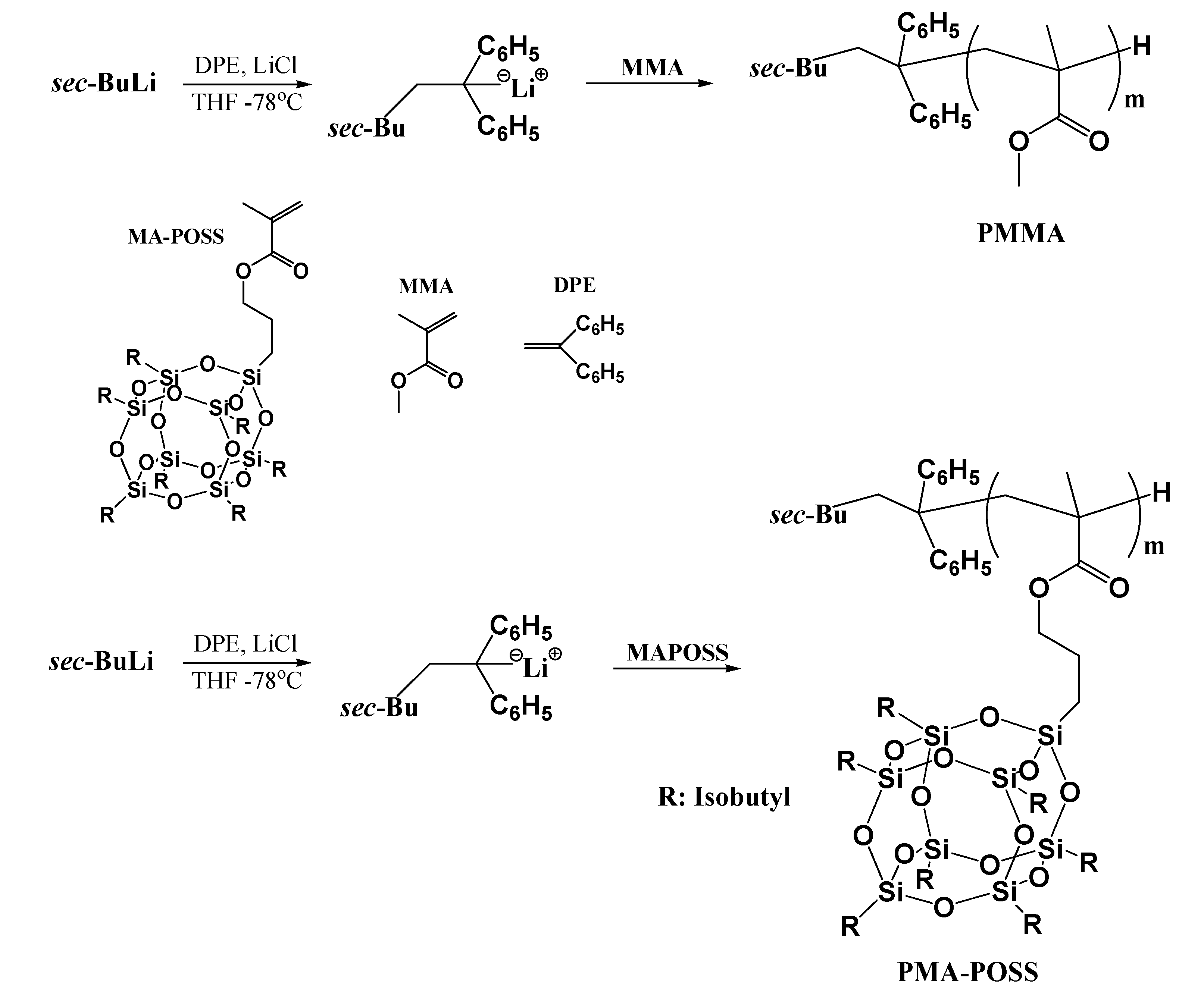
2. Experimental Section
2.1. Materials
2.2. PMMA Homopolymer through Living Anionic Polymerization
| Polymer | V | Mw | DP | PDI | Equilibrium Constant | |||
|---|---|---|---|---|---|---|---|---|
| K2 | KB | KA b | KC c | |||||
| PVPh a | 100 | 120 | 66 | 1.73 | 21.6 | 66.8 | 10 | 0 |
| Phenolic | 84 | 105 | 6 | 2.40 | 23.3 | 52.3 | 20 | 0 |
| BPA | 176 | 228 | – | – | 21.0 | 66.8 | 20 | 0 |
| PMMA d | 84.9 | 100 | 403 | 1.04 | – | – | – | – |
| PMA-POSS d | 826 | 926 | 41 | 1.07 | – | – | – | – |
2.3. PMA-POSS Homopolymer through Living Anionic Polymerization
2.4. Blend Preparations
2.5. Characterization
3. Results and Discussion
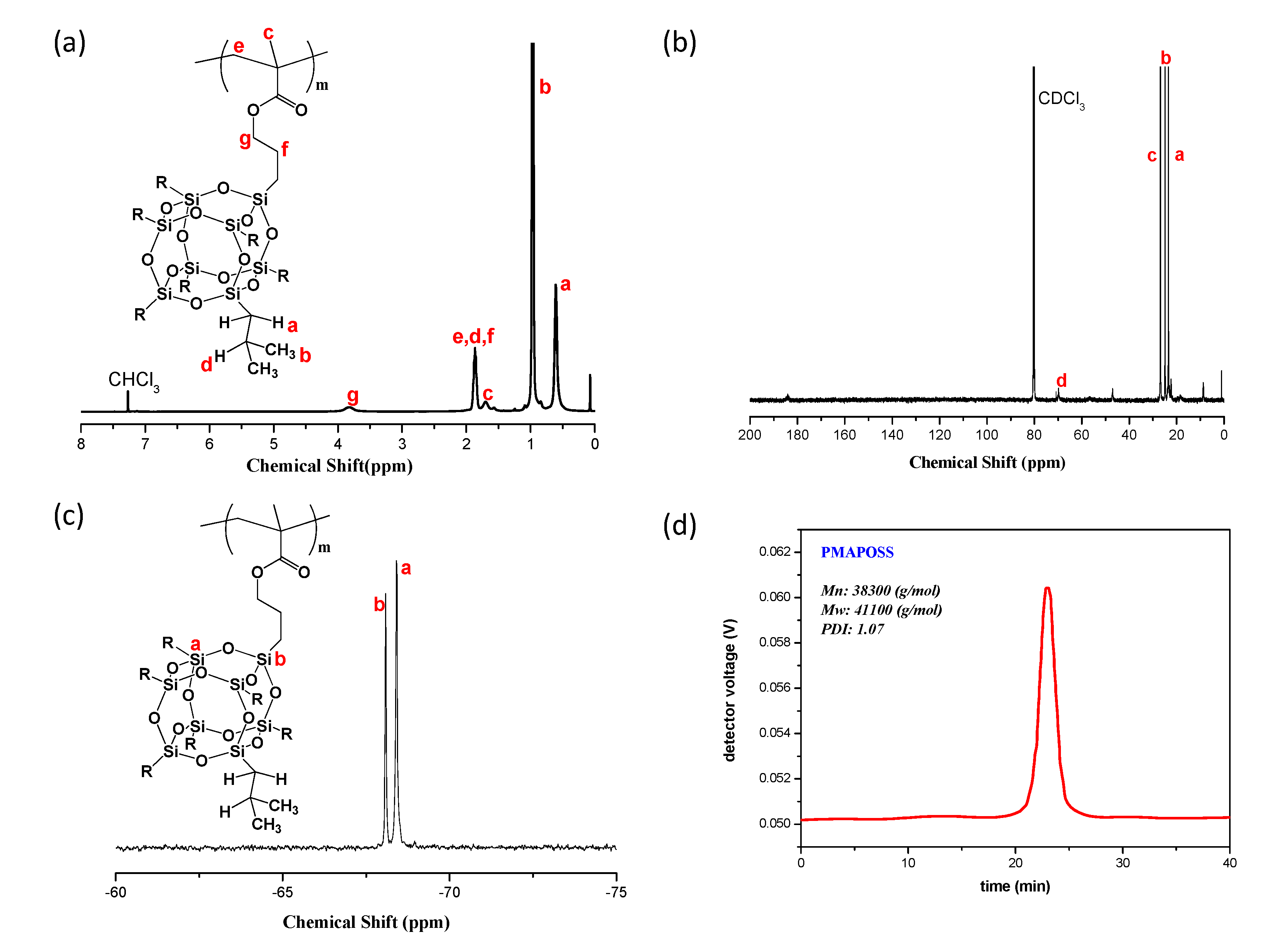
3.1. Synthesis of PMA-POSS
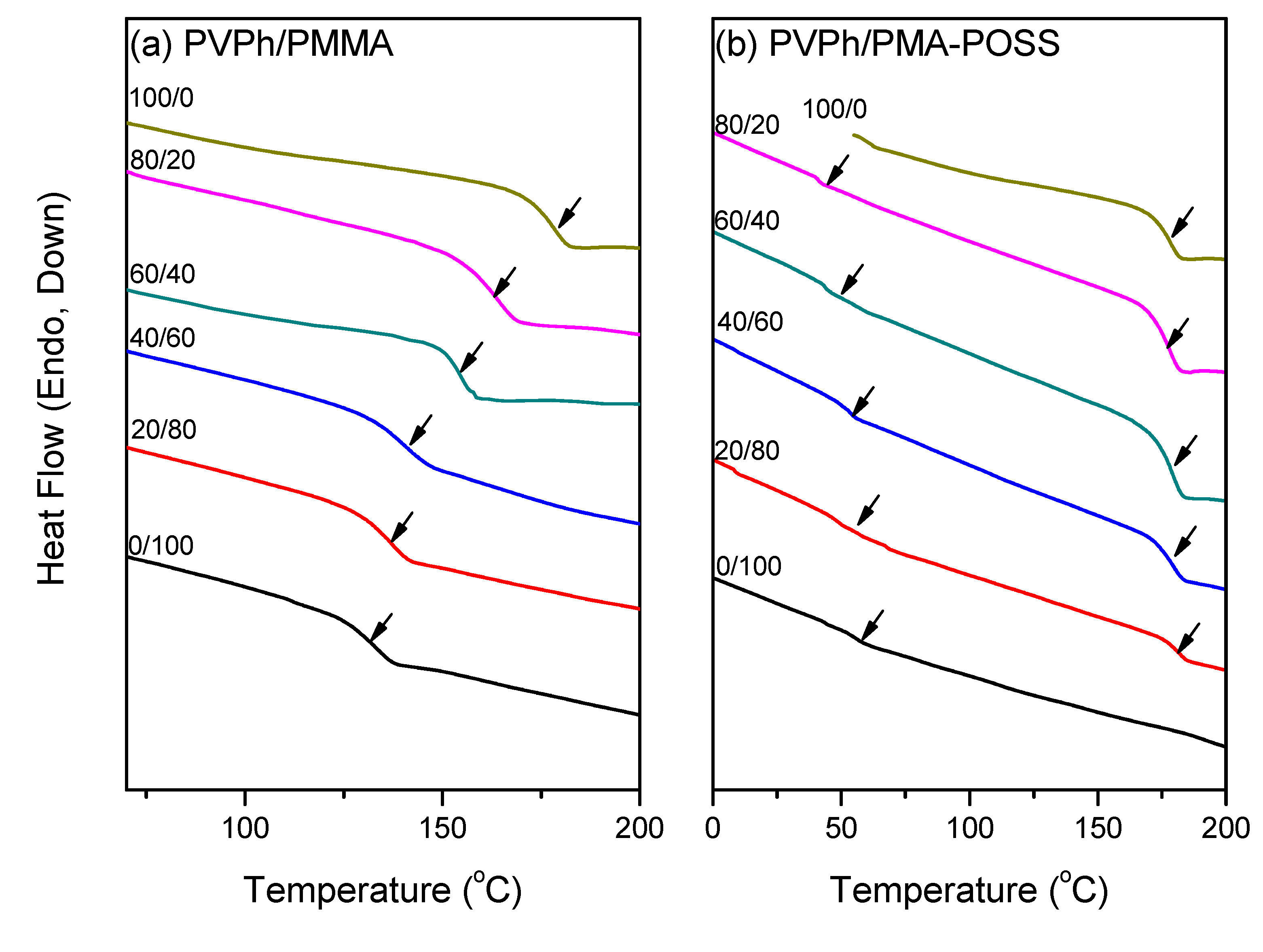
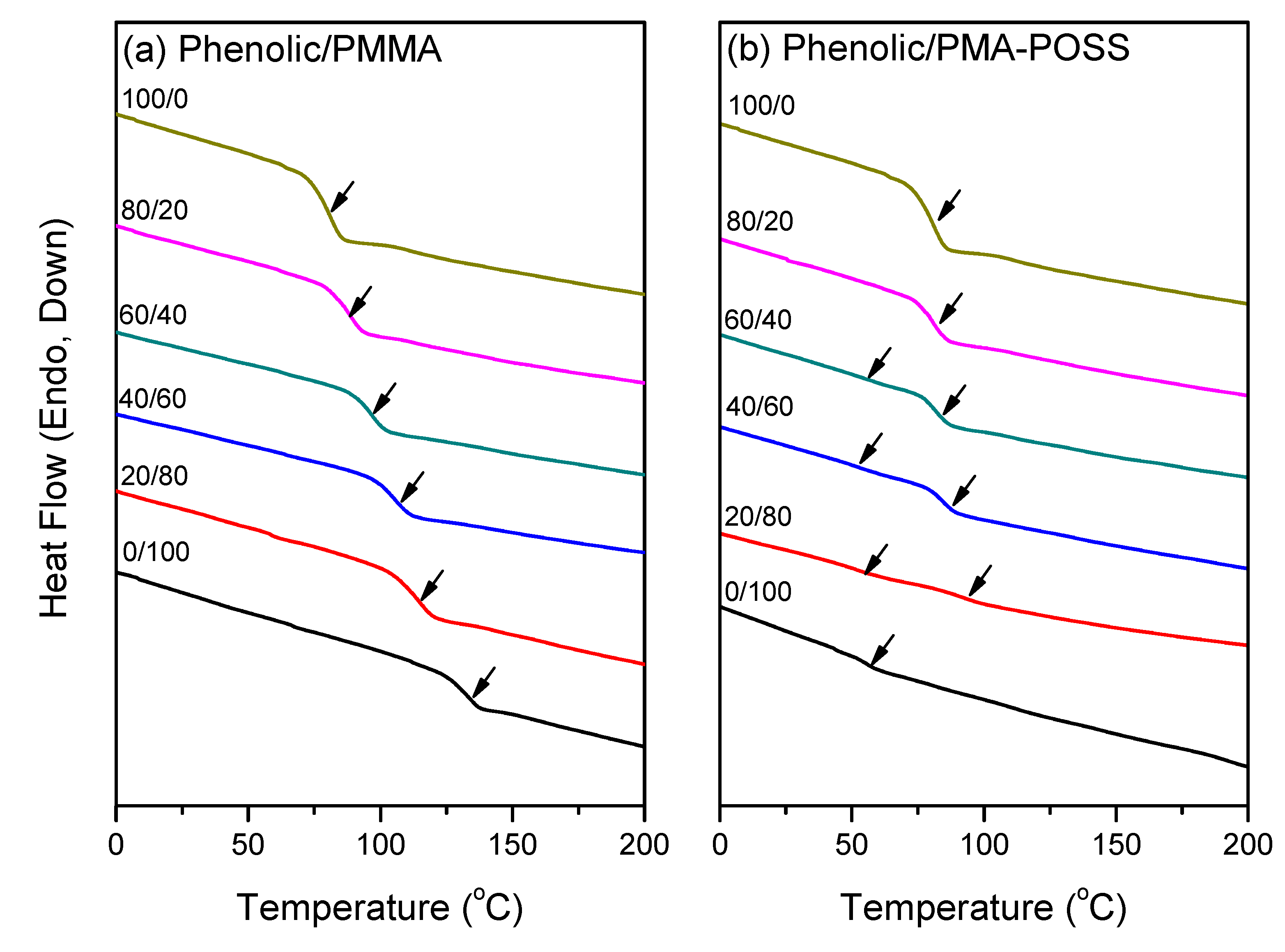
3.2. DSC Analyses
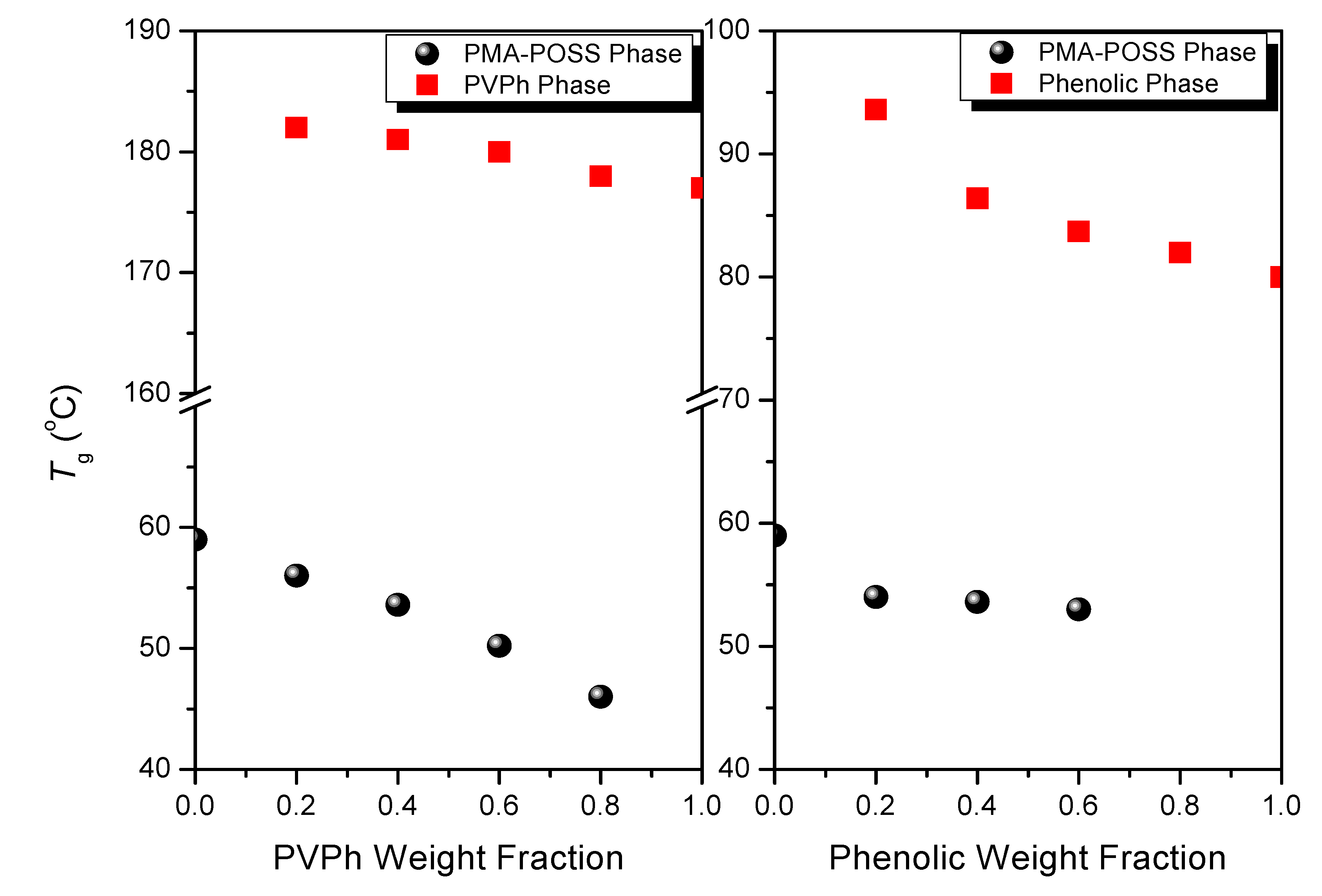
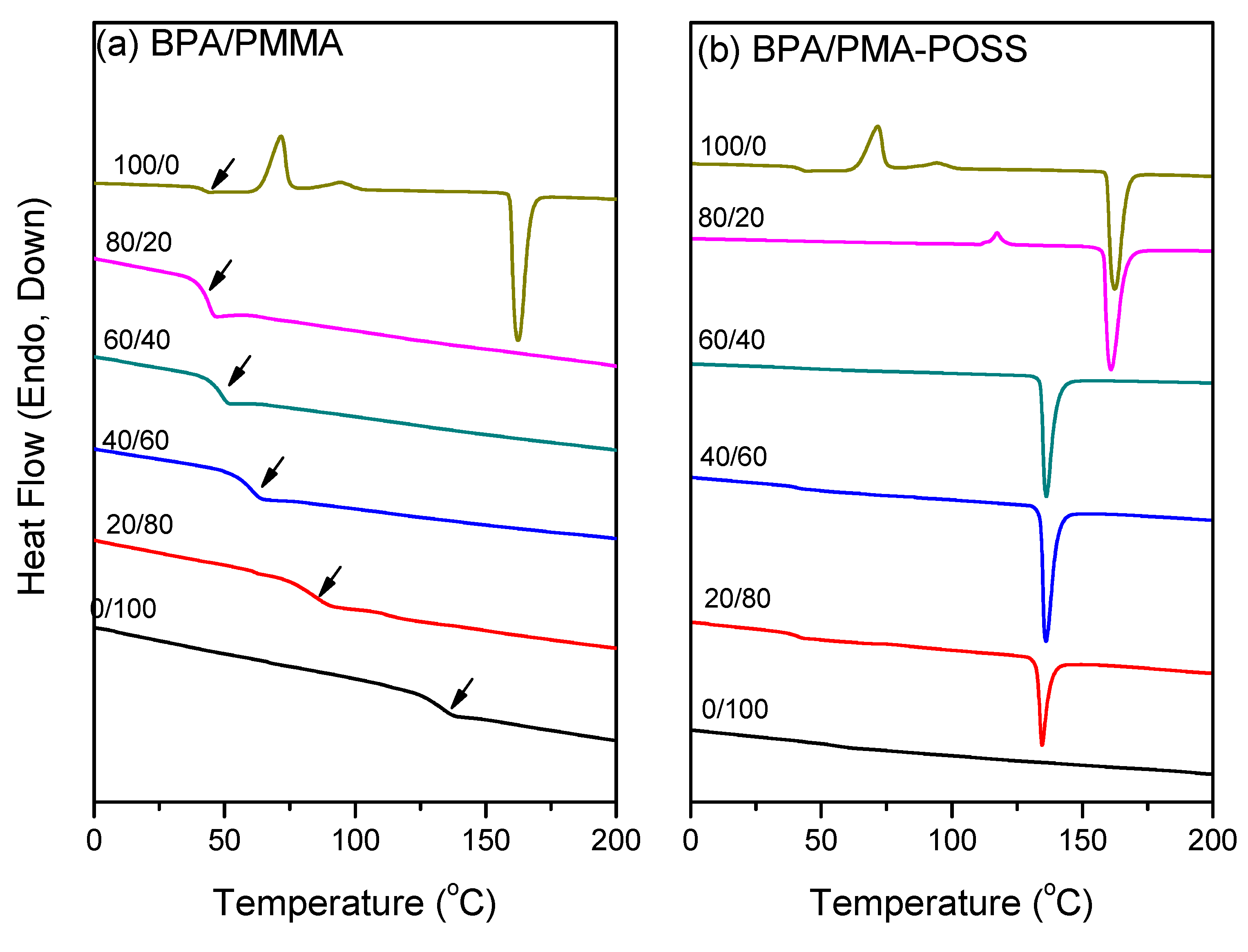
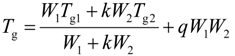
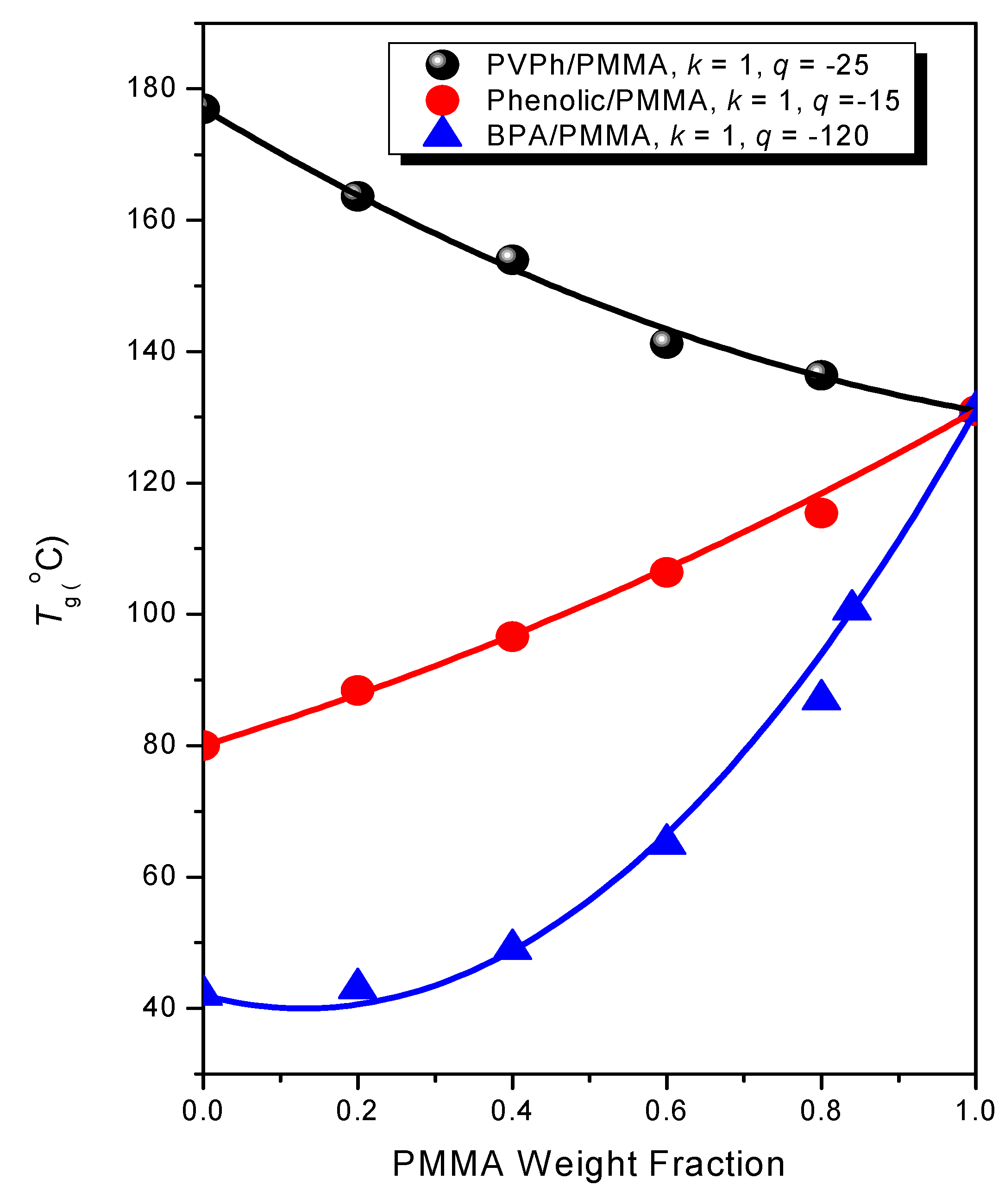
3.3. FTIR Spectra of PMMA Homopolymer Blend Systems
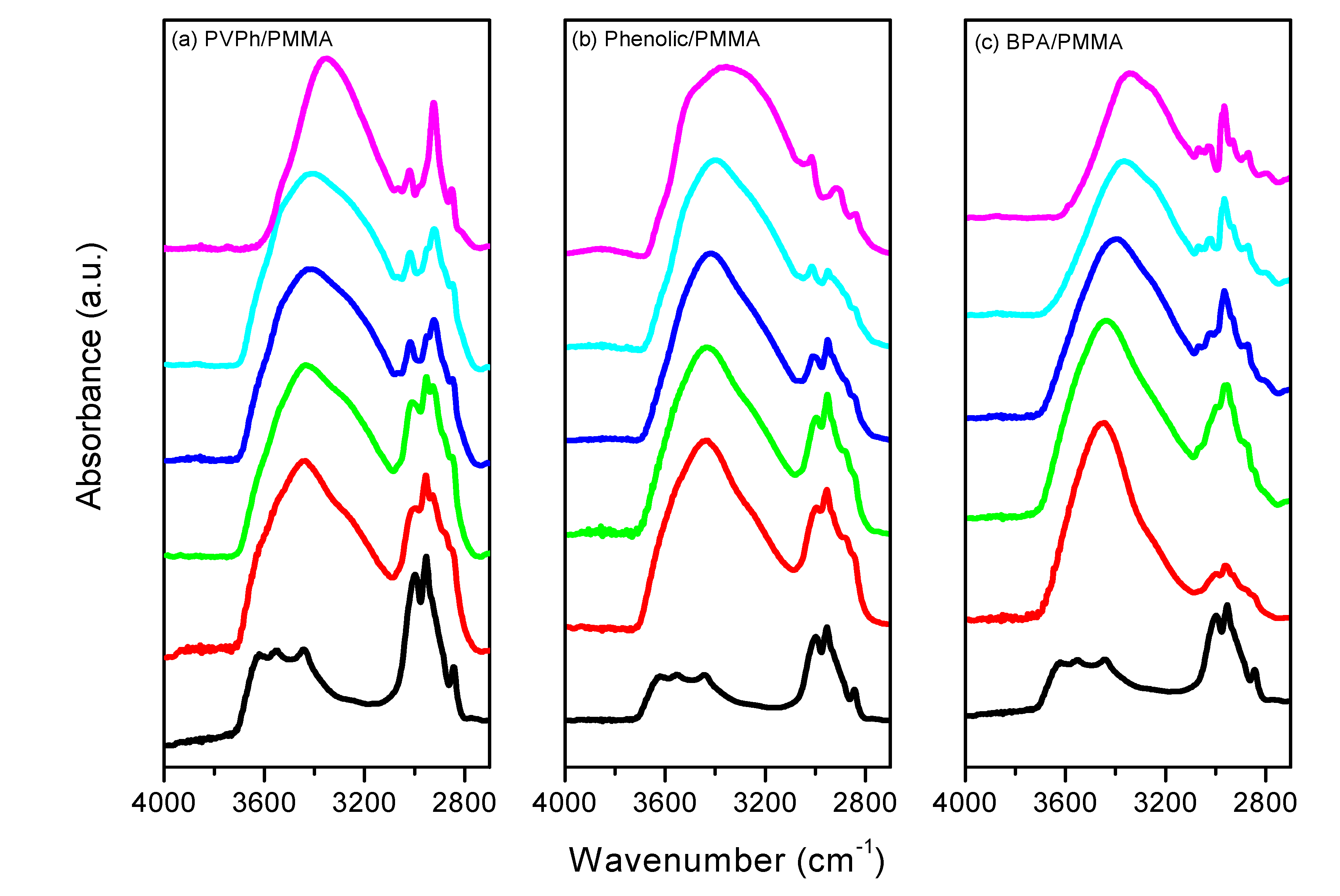
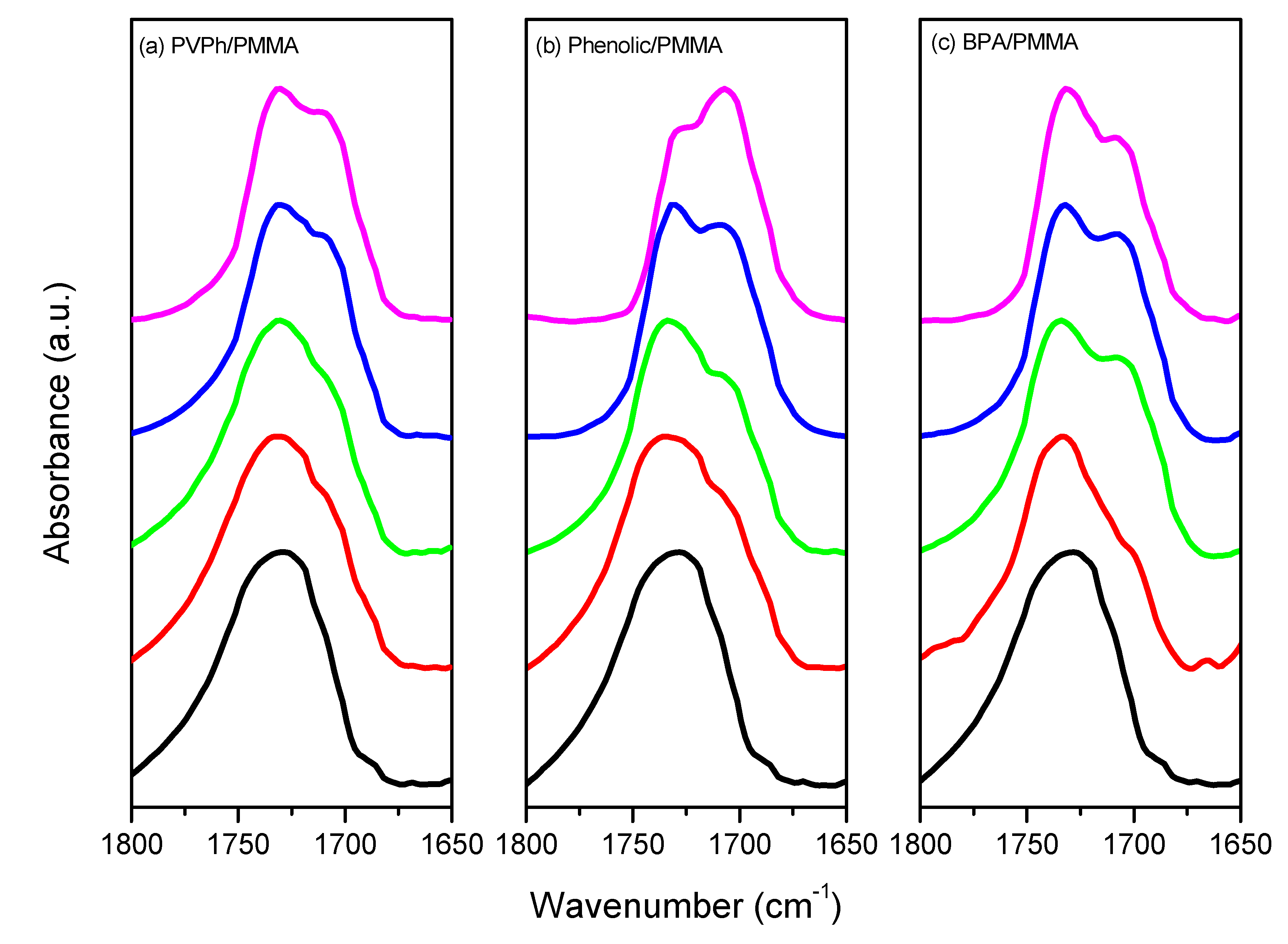
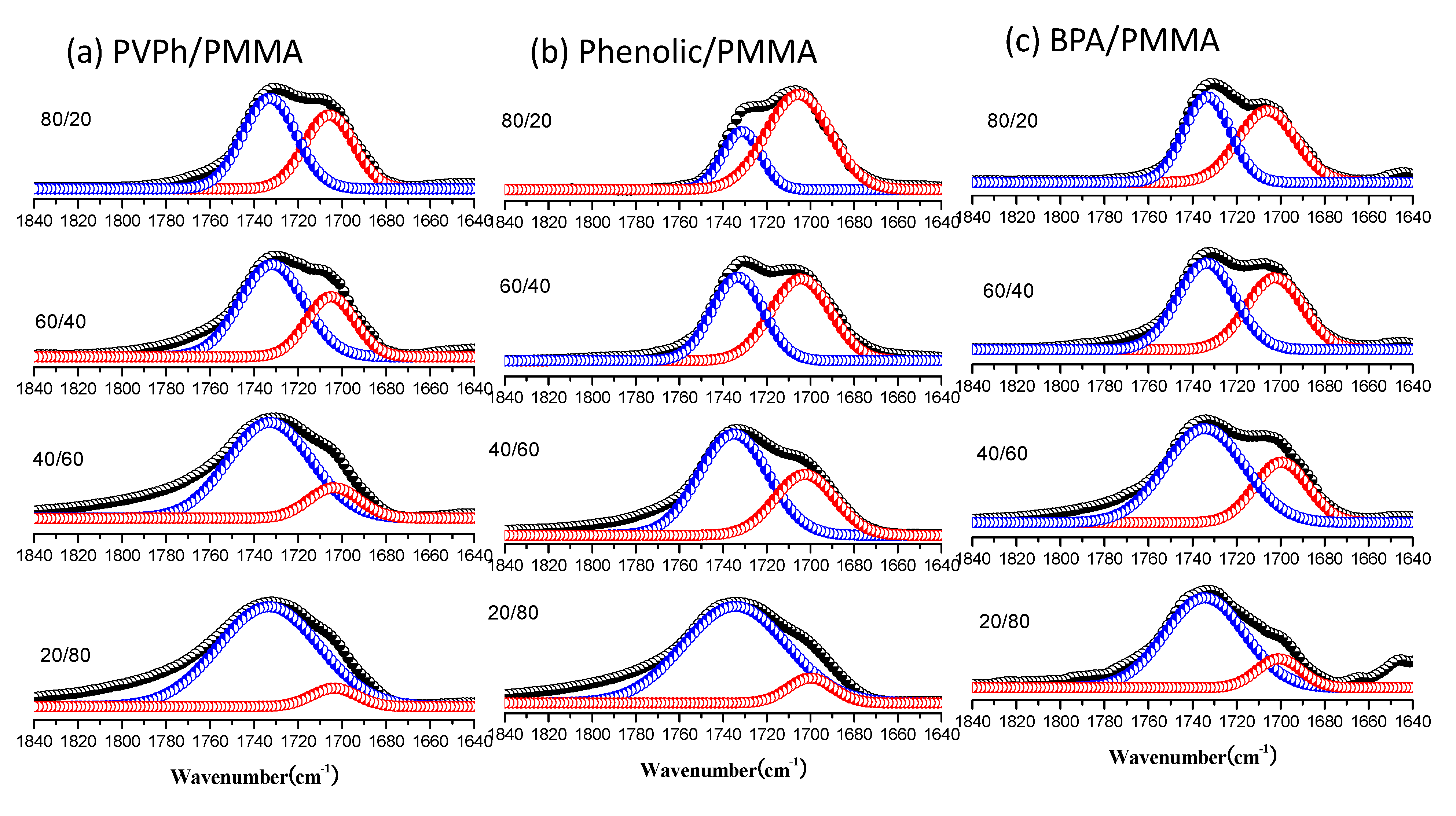
| Sample (wt%) | Free C=O | Hydrogen-bonded C=O | fb (%) | |||
|---|---|---|---|---|---|---|
| vf (cm−1) | Af (%) | vb (cm−1) | Ab (%) | |||
| PVPh/PMMA | ||||||
| 0/100 | 1734 | 100 | – | – | – | |
| 20/80 | 1733 | 92.4 | 1704 | 7.6 | 5.1 | |
| 40/60 | 1733 | 83.9 | 1704 | 16.1 | 11.3 | |
| 60/40 | 1732 | 67.2 | 1705 | 32.8 | 24.5 | |
| 80/20 | 1733 | 57.0 | 1705 | 43.0 | 33.4 | |
| Phenolic/PMMA | ||||||
| 20/80 | 1734 | 84.9 | 1703 | 15.1 | 10.6 | |
| 40/60 | 1735 | 65.9 | 1703 | 34.1 | 25.6 | |
| 60/40 | 1733 | 46.8 | 1704 | 53.2 | 43.1 | |
| 80/20 | 1732 | 26.8 | 1705 | 73.2 | 64.5 | |
| BPA/PMMA | ||||||
| 20/80 | 1734 | 89.9 | 1705 | 10.1 | 6.9 | |
| 40/60 | 1734 | 69.8 | 1705 | 30.2 | 22.3 | |
| 60/40 | 1734 | 55.2 | 1705 | 44.8 | 35.1 | |
| 80/20 | 1733 | 49.2 | 1705 | 50.8 | 40.7 | |

3.4. FTIR Spectra of PMMA-POSS Homopolymer Blend Systems
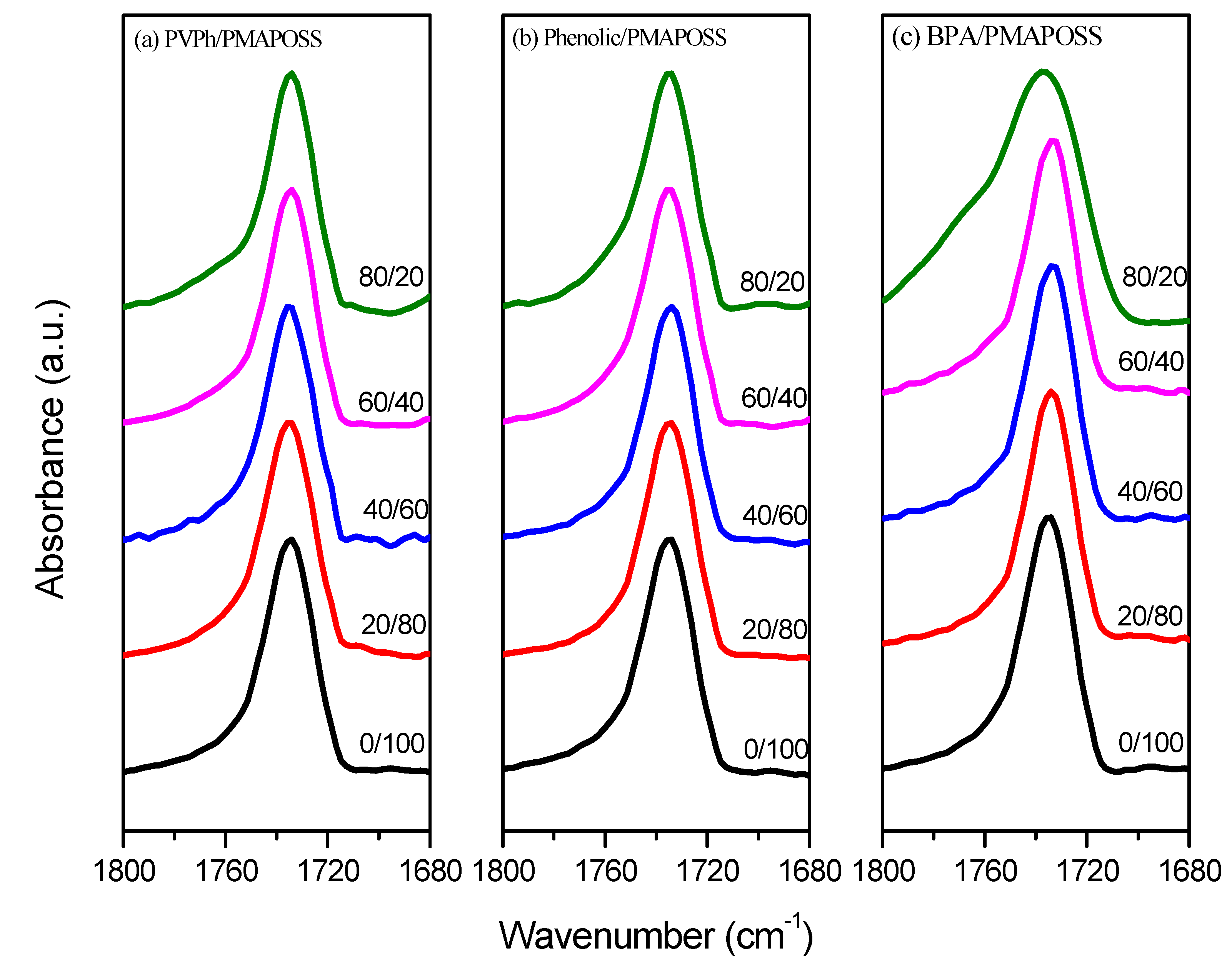
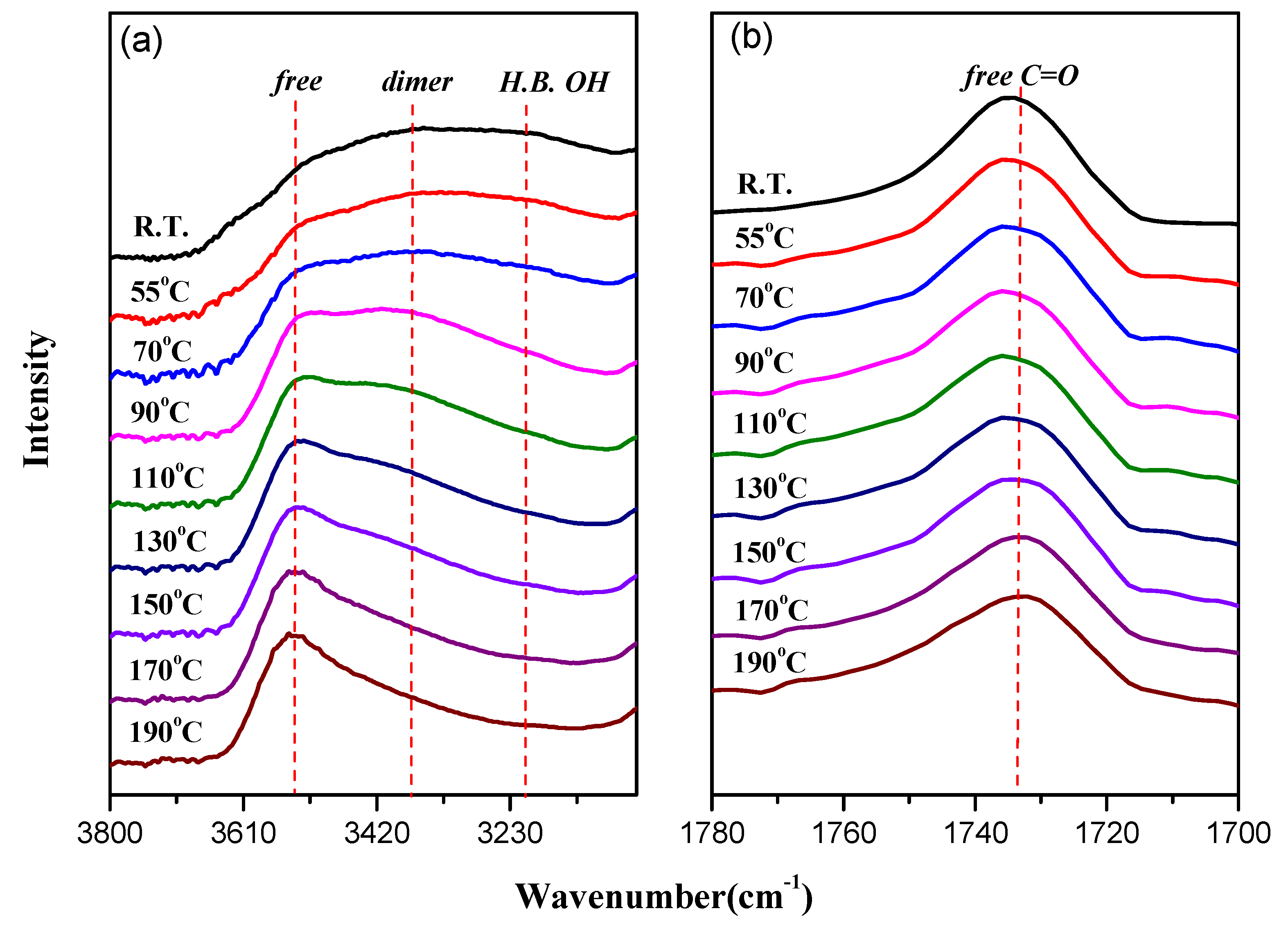
3.5. WAXD Analyses of PMMA-POSS Homopolymer Blend Systems
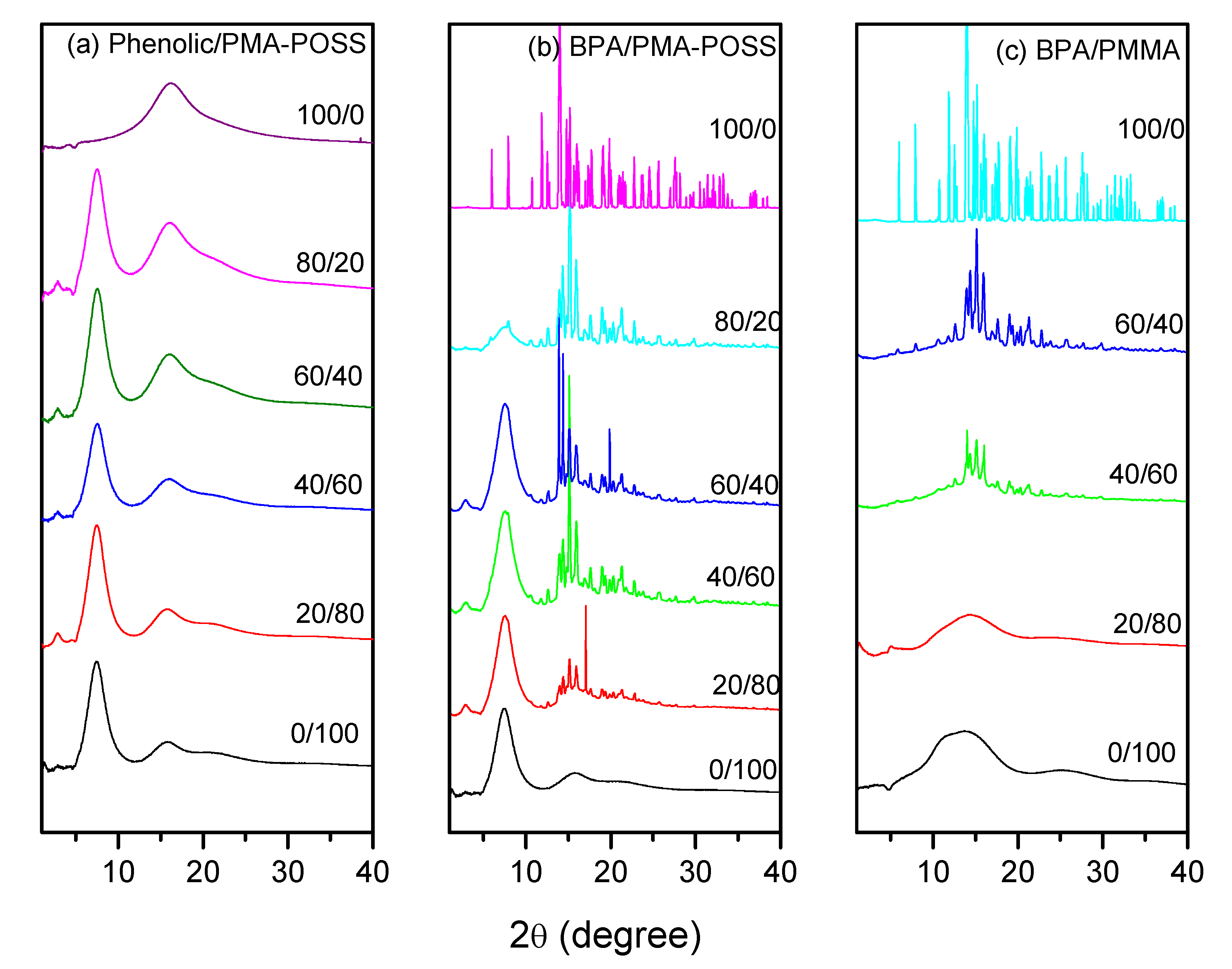
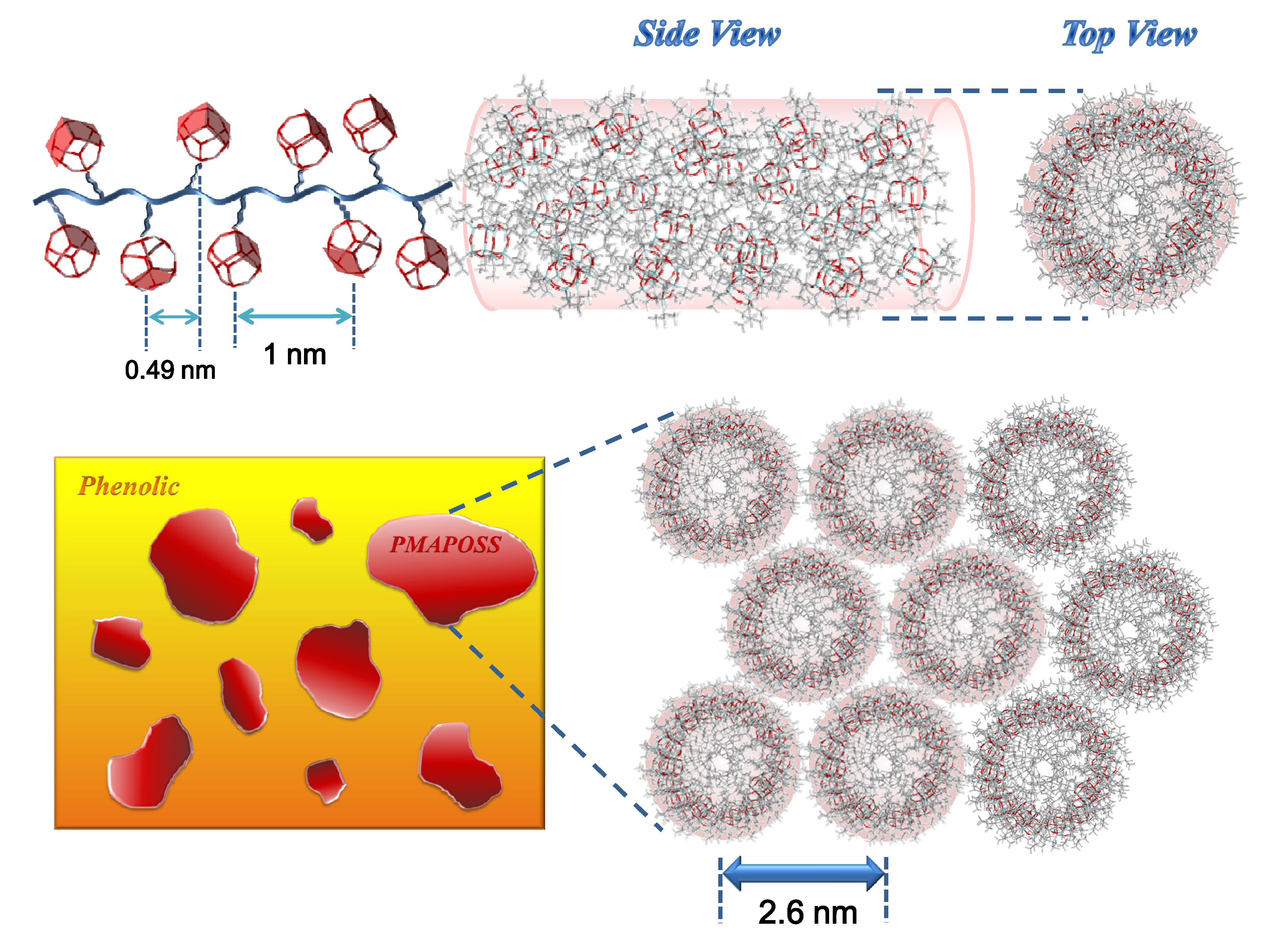
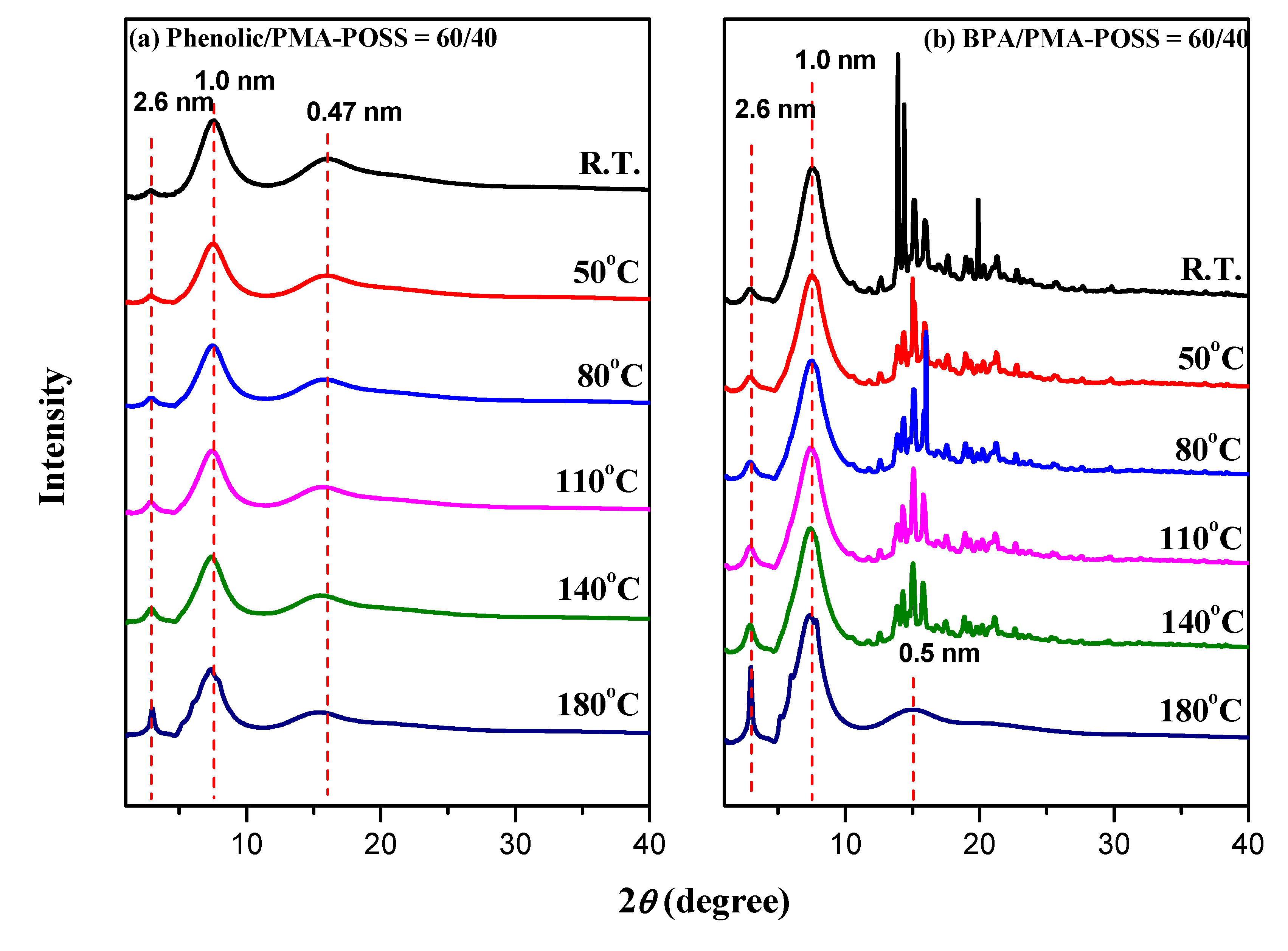
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Coleman, M.M.; Painter, P.C. Hydrogen bonded polymer blends. Prog. Polym. Sci. 1995, 20, 1–59. [Google Scholar] [CrossRef]
- He, Y.; Zhu, B.; Inoue, Y. Hydrogen bonds in polymer blends. Prog. Polym. Sci. 2004, 29, 1021–1051. [Google Scholar] [CrossRef]
- Kuo, S.W. Hydrogen-bonding in polymer blends. J. Polym. Res. 2008, 15, 459–486. [Google Scholar] [CrossRef]
- Kuo, S.W.; Huang, C.F.; Tung, P.H.; Huang, W.J.; Huang, J.M.; Chang, F.C. Synthesis, thermal properties, and specific interactions of high Tg increase in poly(2,6-dimethyl-1,4-phenylene oxide)-block-polystyrene copolymers. Polymer 2005, 46, 9348–9361. [Google Scholar] [CrossRef]
- Kuo, S.W.; Huang, W.J.; Huang, C.F.; Chan, S.C.; Chang, F.C. Miscibility, Specific Interactions, and Spherulite Growth Rates of Binary Poly(acetoxystyrene)/Poly(ethylene oxide) Blends. Macromolecules 2004, 37, 4164–4173. [Google Scholar] [CrossRef]
- Zhang, X.; Takegoshi, K.; Hikichi, K. Poly(vinylphenol)/poly(methyl acrylate) and poly(vinylphenol)/poly(methyl methacrylate) blends: Hydrogen bonding, miscibility, and blending effects on molecular motions as studied by carbon-13 CP/MAS NMR. Macromolecules 1991, 24, 5756–5762. [Google Scholar] [CrossRef]
- Li, D.; Brisson, J. DMTA and FTIR Investigation of the Phase Behavior of Poly(methyl methacrylate)–Poly(4-vinylphenol) Blends. Macromolecules 1996, 29, 868–874. [Google Scholar] [CrossRef]
- Li, D.; Brisson, J. Hydrogen bonds in poly(methyl methacrylate)-poly(4-vinyl phenol) blends: 1. Quantitative analysis using FTi.r. spectroscopy. Polymer 1998, 39, 793–800. [Google Scholar] [CrossRef]
- Dong, J.; Ozaki, Y. FTIR and FT-Raman Studies of Partially Miscible Poly(methyl methacrylate)/Poly(4-vinylphenol) Blends in Solid States. Macromolecules 1997, 30, 286–292. [Google Scholar] [CrossRef]
- Goh, S.H.; Siow, K.S. Miscibility of poly(p-vinyl phenol) with polymethacrylates. Polym. Bull. 1987, 17, 453–457. [Google Scholar]
- Serman, S.J.; Painter, P.C.; Coleman, M.M. Studies of the phase behaviour of poly(vinyl phenol)-poly(n-alkyl methacrylate) blends. Polymer 1991, 32, 1049–1058. [Google Scholar] [CrossRef]
- Lin, C.L.; Chen, W.C.; Liao, C.S.; Su, Y.C.; Huang, C.F.; Kuo, S.W.; Chang, F.C. Sequence Distribution and Polydispersity Index Affect the Hydrogen-Bonding Strength of Poly(vinylphenol-co-methyl methacrylate) Copolymers. Macromolecules 2005, 38, 6435–6444. [Google Scholar] [CrossRef]
- Kuo, S.W.; Chang, F.C. Miscibility and Hydrogen Bonding in Blends of Poly(vinylphenol-co-methyl methacrylate) with Poly(ethylene oxide). Macromolecules 2001, 34, 4089–4097. [Google Scholar] [CrossRef]
- Chen, W.C.; Kuo, S.W.; Jeng, U.S.; Chang, F.C. Self-Assembly through Competitive Interactions of Miscible Diblock Copolymer/Homopolymer Blends: Poly(vinylphenol-b-methyl methacrylate)/Poly(vinylpyrrolidone) Blend. Macromolecules 2008, 41, 1401–1410. [Google Scholar] [CrossRef]
- Kuo, S.W. Hydrogen bond-mediated self-assembly and supramolecular structures of diblock copolymer mixtures. Polym. Int. 2009, 58, 455–464. [Google Scholar] [CrossRef]
- Ni, Y.P.; Becquart, F.; Chen, J.D.; Taha, M. Polyurea–Urethane Supramolecular Thermo-Reversible Networks. Macromolecules 2013, 46, 1066–1074. [Google Scholar] [CrossRef]
- Wang, S.J.; Xu, Y.S.; Yang, S.; Chen, E.Q. Phase Behavior of a Hydrogen-Bonded Polymer with Lamella-to-Cylinder Transition: Complex of Poly(4-vinylpyridine) and Small Dendritic Benzoic Acid Derivative. Macromolecules 2012, 45, 8760–8769. [Google Scholar] [CrossRef]
- Kuo, S.W.; Chen, C.J. Functional Polystyrene Derivatives Influence the Miscibility and Helical Peptide Secondary Structures of Poly(γ-benzyl l-glutamate). Macromolecules 2012, 45, 2442–2452. [Google Scholar] [CrossRef]
- Painter, P.C.; Veytsman, B.; Kumar, S.; Shenoy, S.; Graf, J.F.; Xu, Y.; Coleman, M.M. Intramolecular Screening Effects in Polymer Mixtures. 1. Hydrogen-Bonded Polymer Blends. Macromolecules 1997, 30, 932–942. [Google Scholar] [CrossRef]
- Pehlert, G.J.; Painter, P.C.; Veytsman, B.; Coleman, M.M. Functional Group Accessibility in Hydrogen-Bonded Polymer Blends. 2. Miscibility Map of 2,3-Dimethylbutadiene- stat-vinylphenol Blends with Ethylene-stat-vinyl acetate. Macromolecules 1997, 30, 3671–3677. [Google Scholar] [CrossRef]
- Pehlert, G.J.; Painter, P.C.; Coleman, M.M. Functional Group Accessibility in Hydrogen-Bonded Polymer Blends. 3. Steric Shielding Effects. Macromolecules 1998, 31, 8423–8424. [Google Scholar] [CrossRef]
- Coleman, M.M.; Xu, Y.; Painter, P.C. Compositional heterogeneities in hydrogen-bonded polymer blends: infrared spectroscopic results. Macromolecules 1994, 27, 127–134. [Google Scholar] [CrossRef]
- Pruthtikul, R.; Coleman, M.; Painter, P.C.; Tan, N.B. Screening Effects in Solutions of a Hyperbranched, Dendrimer-Like Polyester. Macromolecules 2001, 34, 4145–4150. [Google Scholar] [CrossRef]
- Huang, C.F.; Kuo, S.W.; Lin, H.C.; Chen, J.K.; Chen, Y.K.; Xu, H.Y.; Chang, F.C. Thermal properties, miscibility and specific interactions in comparison of linear and star poly(methyl methacrylate) blend with phenolic. Polymer 2004, 45, 5913–5921. [Google Scholar] [CrossRef]
- Huang, C.F.; Kuo, S.W.; Lin, F.L.; Huang, W.J.; Wang, C.F.; Chen, W.Y.; Chang, F.C. Influence of PMMA-Chain-End Tethered Polyhedral Oligomeric Silsesquioxanes on the Miscibility and Specific Interaction with Phenolic Blends. Macromolecules 2006, 39, 300–308. [Google Scholar] [CrossRef]
- Kuo, S.W.; Chang, F.C. POSS related polymer nanocomposites. Prog. Polym. Sci. 2011, 36, 1649–1696. [Google Scholar] [CrossRef]
- Zhang, W.A.; Muller, A.H.E. Architecture, self-assembly and properties of well-defined hybrid polymers based on polyhedral oligomeric silsequioxane (POSS). Prog. Polym. Sci. 2013, 38, 1121–1162. [Google Scholar] [CrossRef]
- Tanaka, K.; Chujo, Y. Unique properties of amphiphilic POSS and their applications. Polym. J. 2013, 45, 247–254. [Google Scholar] [CrossRef]
- Xu, H.; Kuo, S.W.; Lee, J.S.; Chang, F.C. Glass transition temperatures of poly(hydroxystyrene-co-vinylpyrrolidone-co-isobutylstyryl polyhedral oligosilsesquioxanes). Polymer 2002, 43, 5117–5124. [Google Scholar] [CrossRef]
- Lee, Y.J.; Huang, J.M.; Kuo, S.W.; Lu, J.S.; Chang, F.C. Polyimide and polyhedral oligomeric silsesquioxane nanocomposites for low-dielectric applications. Polymer 2005, 46, 173–181. [Google Scholar] [CrossRef]
- Lin, H.C.; Kuo, S.W.; Huang, C.F.; Chang, F.C. Thermal and Surface Properties of Phenolic Nanocomposites Containing Octaphenol Polyhedral Oligomeric Silsesquioxane. Macromol. Rapid Commun. 2006, 27, 537–541. [Google Scholar] [CrossRef]
- Zhang, W.C.; Li, X.M.; Yang, R.J. Blowing-out effect and temperature profile in condensed phase in flame retarding epoxy resins by phosphorus-containing oligomeric silsesquioxane. Polym. Adv. Tech. 2013, 24, 951–961. [Google Scholar] [CrossRef]
- Li, Y.W.; Wang, Z.; Zheng, J.K.; Su, H.; Lin, F.; Guo, K.; Feng, X.; Wesdemiotis, C.; Becker, M.L.; Cheng, S.Z.D.; Zhang, W.B. Cascading One-Pot Synthesis of Single-Tailed and Asymmetric Multitailed Giant Surfactants. ACS Macro Lett. 2013, 2, 1026–1032. [Google Scholar] [CrossRef]
- Yue, K.; Liu, C.; Guo, K.; Yu, X.F.; Huang, M.J.; Li, Y.W.; Wesdemiotis, C.; Cheng, S.Z.D.; Zhang, W.B. Sequential “Click” Approach to Polyhedral Oligomeric Silsesquioxane-Based Shape Amphiphiles. Macromolecules 2012, 45, 8126–8134. [Google Scholar] [CrossRef]
- Hu, W.H.; Huang, K.W.; Chiou, C.W.; Kuo, S.W. Complementary Multiple Hydrogen Bonding Interactions Induce the Self-Assembly of Supramolecular Structures from Heteronucleobase-Functionalized Benzoxazine and Polyhedral Oligomeric Silsesquioxane Nanoparticles. Macromolecules 2012, 45, 9020–9028. [Google Scholar] [CrossRef]
- Kuo, S.W.; Chen, C.J. Using Hydrogen-Bonding Interactions To Control the Peptide Secondary Structures and Miscibility Behavior of Poly(L-glutamate)s with Phenolic Resin. Macromolecules 2011, 44, 7315–7326. [Google Scholar] [CrossRef]
- Li, J.G.; Lin, Y.D.; Kuo, S.W. From Microphase Separation to Self-Organized Mesoporous Phenolic Resin through Competitive Hydrogen Bonding with Double-Crystalline Diblock Copolymers of Poly(ethylene oxide-b-ε-caprolactone). Macromolecules 2011, 44, 9295–9309. [Google Scholar] [CrossRef]
- Li, J.G.; Chu, W.C.; Jeng, U.S.; Kuo, S.W. In Situ Monitoring of the Reaction-Induced Self-Assembly of Phenolic Resin Templated by Diblock Copolymers. Macromol. Chem. Phys. 2013, 214, 2115–2123. [Google Scholar] [CrossRef]
- Hirai, T.; Leolukman, M.; Jin, S.; Goseki, R.; Ishida, Y.; Kakimoto, M.; Hayakawa, T.; Ree, M.; Gopalan, P. Hierarchical Self-Assembled Structures from POSS-Containing Block Copolymers Synthesized by Living Anionic Polymerization. Macromolecules 2009, 42, 8835–8843. [Google Scholar] [CrossRef]
- Ishida, Y.; Hira, T.; Goseki, R.; Tokita, M.; Kakimoto, A.; Hayakawa, T. Synthesis and self-assembly of thermotropic block copolymer with long alkyl tethered cage silsesquioxane in the side chain. J. Polym. Sci. A Polym. Chem. 2011, 49, 2653–2664. [Google Scholar] [CrossRef]
- Wu, Y.C.; Kuo, S.W. Synthesis and characterization of polyhedral oligomeric silsesquioxane (POSS) with multifunctional benzoxazine groups through click chemistry. Polymer 2010, 51, 3948–3955. [Google Scholar] [CrossRef]
- Huang, K.W.; Kuo, S.W. High-Performance Polybenzoxazine Nanocomposites Containing Multifunctional POSS Cores Presenting Vinyl-Terminated Benzoxazine Groups. Macromol. Chem. Phys. 2010, 211, 2301–2311. [Google Scholar] [CrossRef]
- Kuo, S.W.; Chan, S.C.; Chang, F.C. Miscibility enhancement on the immiscible binary blend of poly(vinyl acetate) and poly(vinyl pyrrolidone) with bisphenol A. Polymer 2002, 43, 3653–3660. [Google Scholar] [CrossRef]
- Kwei, T.K. The effect of hydrogen bonding on the glass transition temperatures of polymer mixtures. J. Polym. Sci. Polym. Lett. Ed. 1984, 22, 307–313. [Google Scholar] [CrossRef]
- Kuo, S.W.; Chan, S.C.; Chang, F.C. The effect of hydrogen bonding on the glass transition temperatures of polymer mixtures. Macromolecules 2003, 36, 6653–6661. [Google Scholar] [CrossRef]
- Kuo, S.W.; Chang, F.C. Miscibility Behavior and Specific Interaction of Phenolic Resin with Poly(acetoxystyrene) Blends. Macromol. Chem. Phys. 2002, 203, 868–878. [Google Scholar] [CrossRef]
- Lee, Y.J.; Kuo, S.W.; Huang, W.J.; Lee, H.Y.; Chang, F.C. Miscibility, specific interactions, and self-assembly behavior of phenolic/polyhedral oligomeric silsesquioxane hybrids. J. Polym. Sci. Polym. Phys. 2004, 42, 1127–1136. [Google Scholar] [CrossRef]
- Kuo, S.W.; Lin, H.C.; Huang, W.J.; Huang, C.F.; Chang, F.C. Hydrogen bonding interactions and miscibility between phenolic resin and octa(acetoxystyryl) polyhedral oligomeric silsesquioxane (AS-POSS) nanocomposites. J. Polym. Sci. Polym. Phys. 2006, 44, 673–686. [Google Scholar] [CrossRef]
- Jin, S.; Hira, T.; Ahn, B.; Rho, Y.; Kim, K.W.; Kakimoto, M.A.; Gopalan, P.; Hayakawa, T.; Ree, M. Synchrotron Grazing Incidence X-ray Scattering Study of the Morphological Structures in Thin Films of a Polymethacrylate Diblock Copolymer Bearing POSS Moieties. J. Phys. Chem. B 2010, 114, 8033–8042. [Google Scholar]
- Chen, Y.; Kang, E.T. New approach to nanocomposites of polyimides containing polyhedral oligomeric silsesquioxane for dielectric applications. Mater. Lett. 2004, 58, 3716–3719. [Google Scholar] [CrossRef]
- Li, X.D.; Goh, S.H.; Zheng, J.W. Specific interactions and phase behavior of poly(2-vinylpridine)/bisphenol blends. J. Appl. Polym. Sci. 2003, 87, 1137–1143. [Google Scholar] [CrossRef]
- Chen, W.C.; Kuo, S.W.; Lu, C.H.; Chang, F.C. Self-Assembly Structures through Competitive Interactions of Crystalline–Amorphous Diblock Copolymer/Homopolymer Blends: Poly(ε-caprolactone-b-4-vinyl pyridine)/Poly(vinyl phenol). Macromolecules 2009, 42, 3580–3590. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chiou, C.-W.; Lin, Y.-C.; Wang, L.; Hirano, C.; Suzuki, Y.; Hayakawa, T.; Kuo, S.-W. Strong Screening Effect of Polyhedral Oligomeric Silsesquioxanes (POSS) Nanoparticles on Hydrogen Bonded Polymer Blends. Polymers 2014, 6, 926-948. https://doi.org/10.3390/polym6030926
Chiou C-W, Lin Y-C, Wang L, Hirano C, Suzuki Y, Hayakawa T, Kuo S-W. Strong Screening Effect of Polyhedral Oligomeric Silsesquioxanes (POSS) Nanoparticles on Hydrogen Bonded Polymer Blends. Polymers. 2014; 6(3):926-948. https://doi.org/10.3390/polym6030926
Chicago/Turabian StyleChiou, Chin-Wei, Yung-Chih Lin, Lei Wang, Chiharu Hirano, Yoshinori Suzuki, Teruaki Hayakawa, and Shiao-Wei Kuo. 2014. "Strong Screening Effect of Polyhedral Oligomeric Silsesquioxanes (POSS) Nanoparticles on Hydrogen Bonded Polymer Blends" Polymers 6, no. 3: 926-948. https://doi.org/10.3390/polym6030926
APA StyleChiou, C.-W., Lin, Y.-C., Wang, L., Hirano, C., Suzuki, Y., Hayakawa, T., & Kuo, S.-W. (2014). Strong Screening Effect of Polyhedral Oligomeric Silsesquioxanes (POSS) Nanoparticles on Hydrogen Bonded Polymer Blends. Polymers, 6(3), 926-948. https://doi.org/10.3390/polym6030926





