Enzyme Initiated Radical Polymerizations
Abstract
:1. Introduction
2. Mechanism(s) of Enzyme Initiated Radical Polymerizations
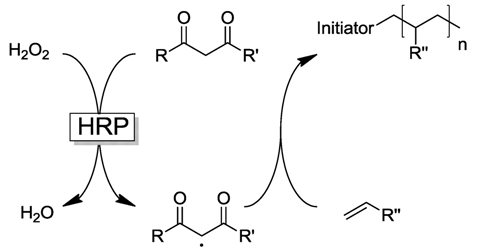
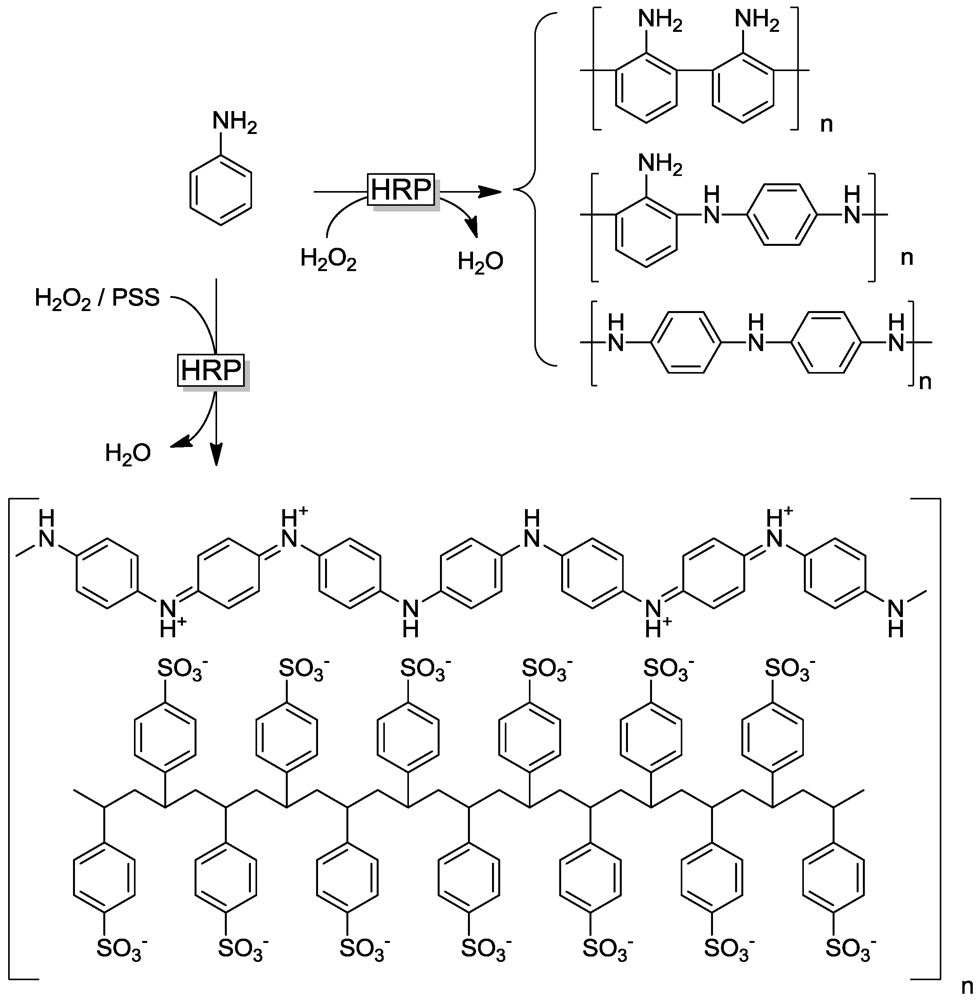
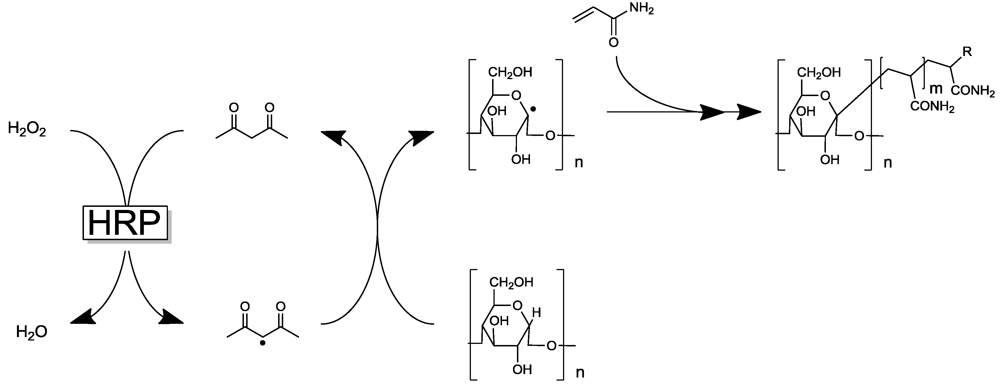
2.1. Peroxidase-Initiated Polymerizations
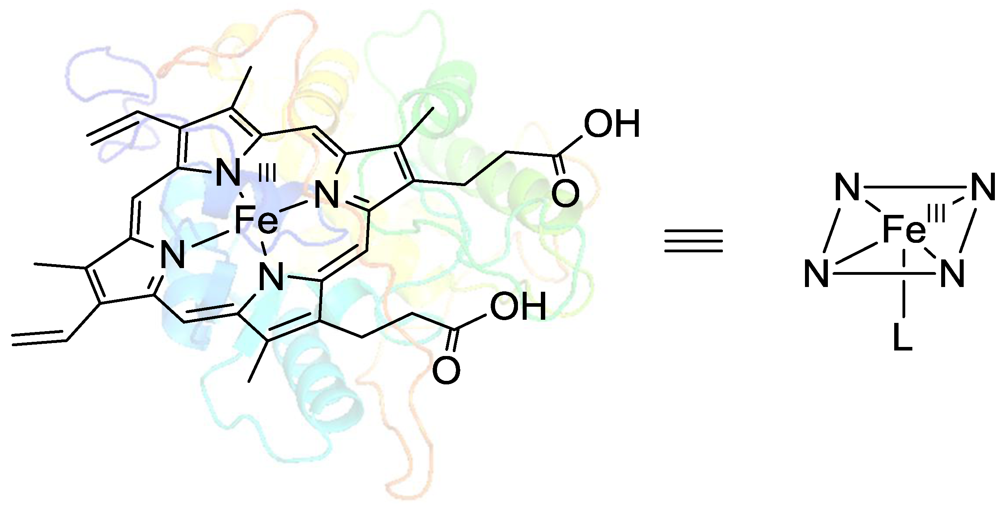
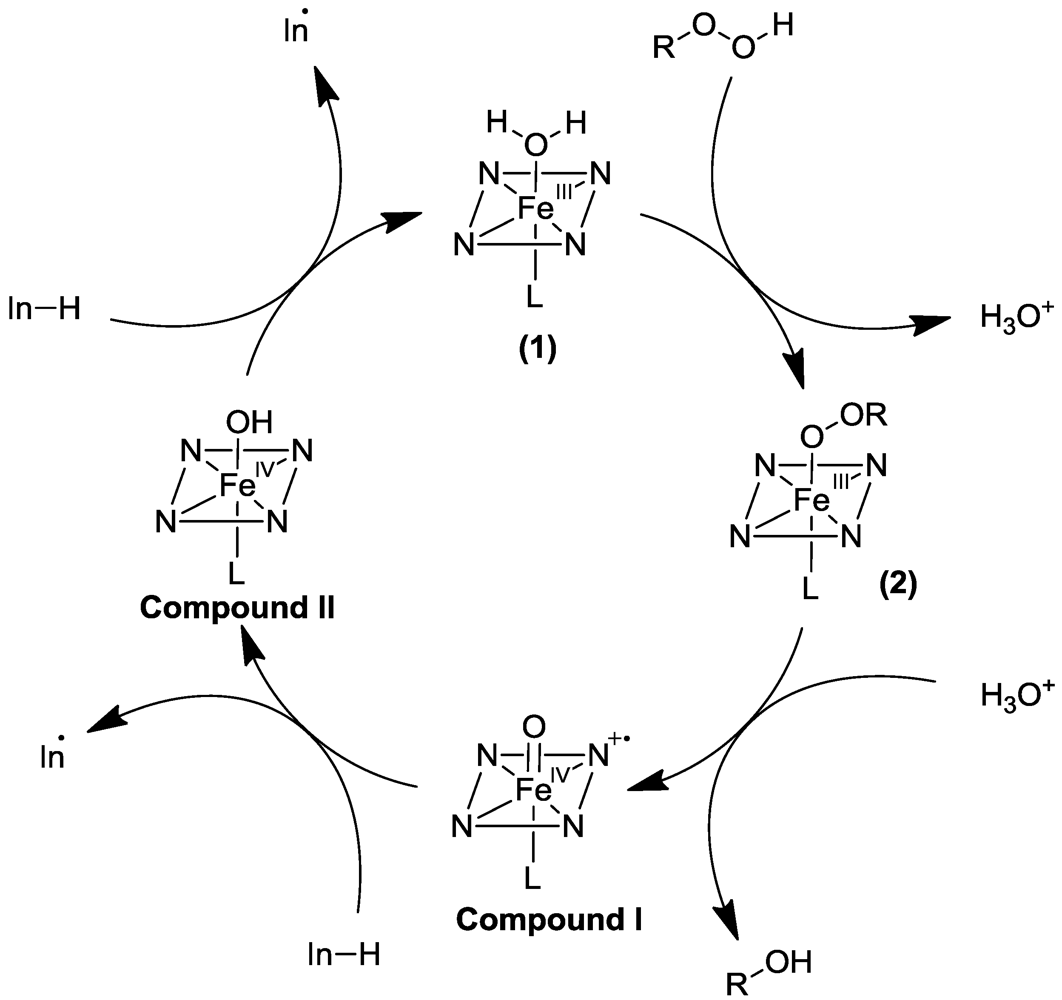
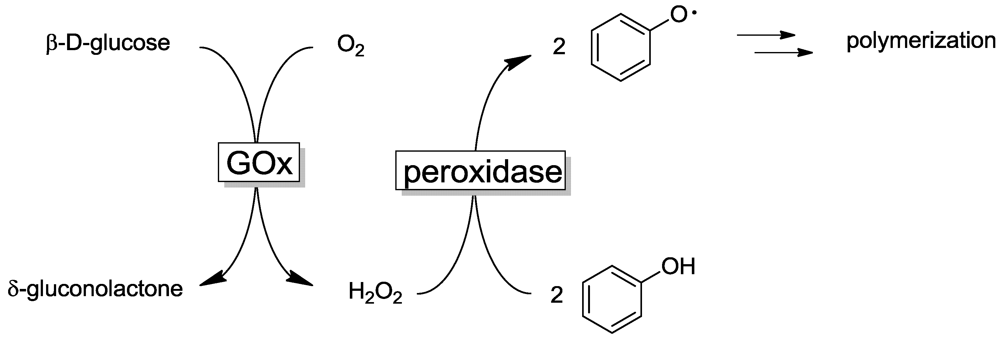
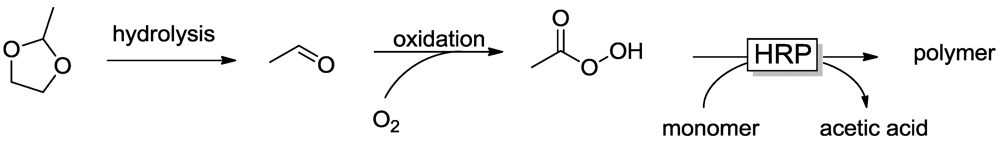
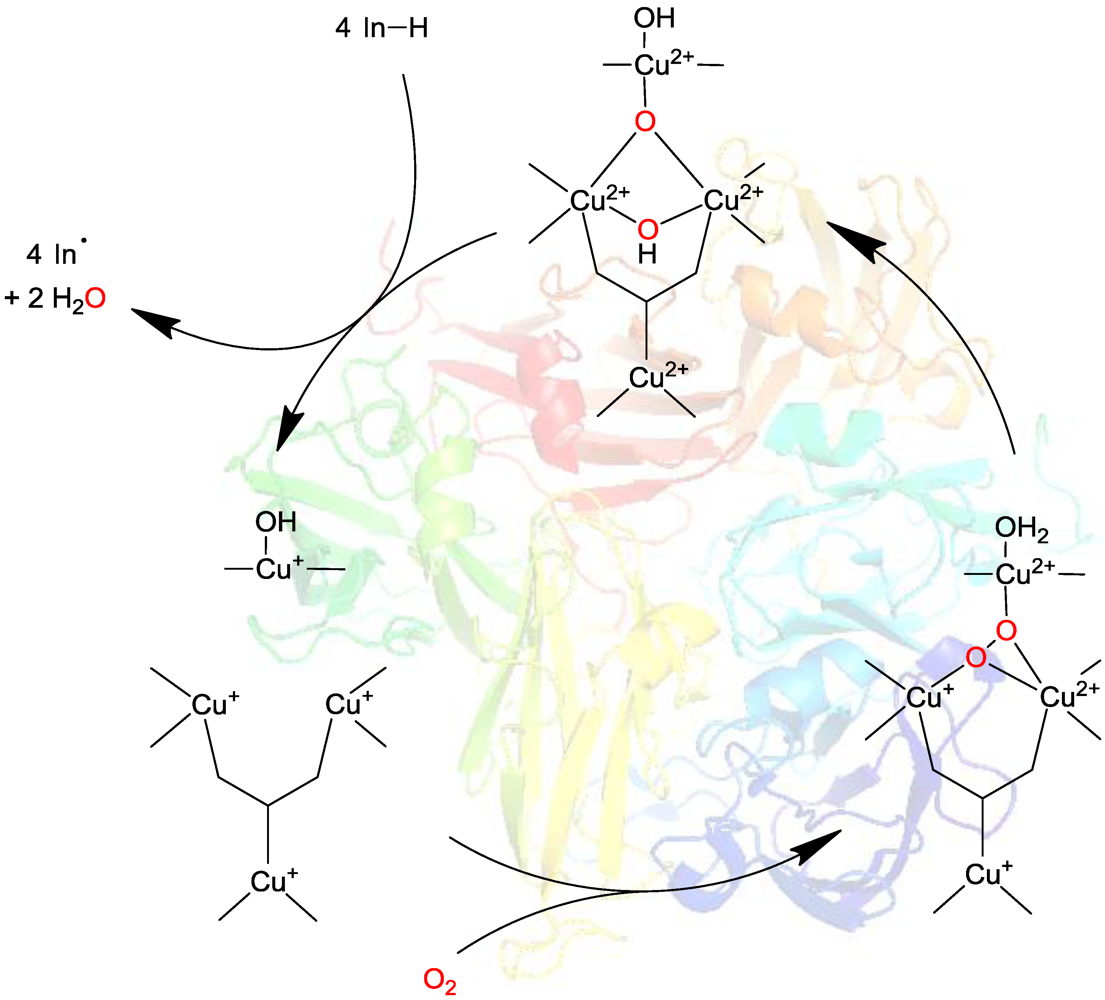
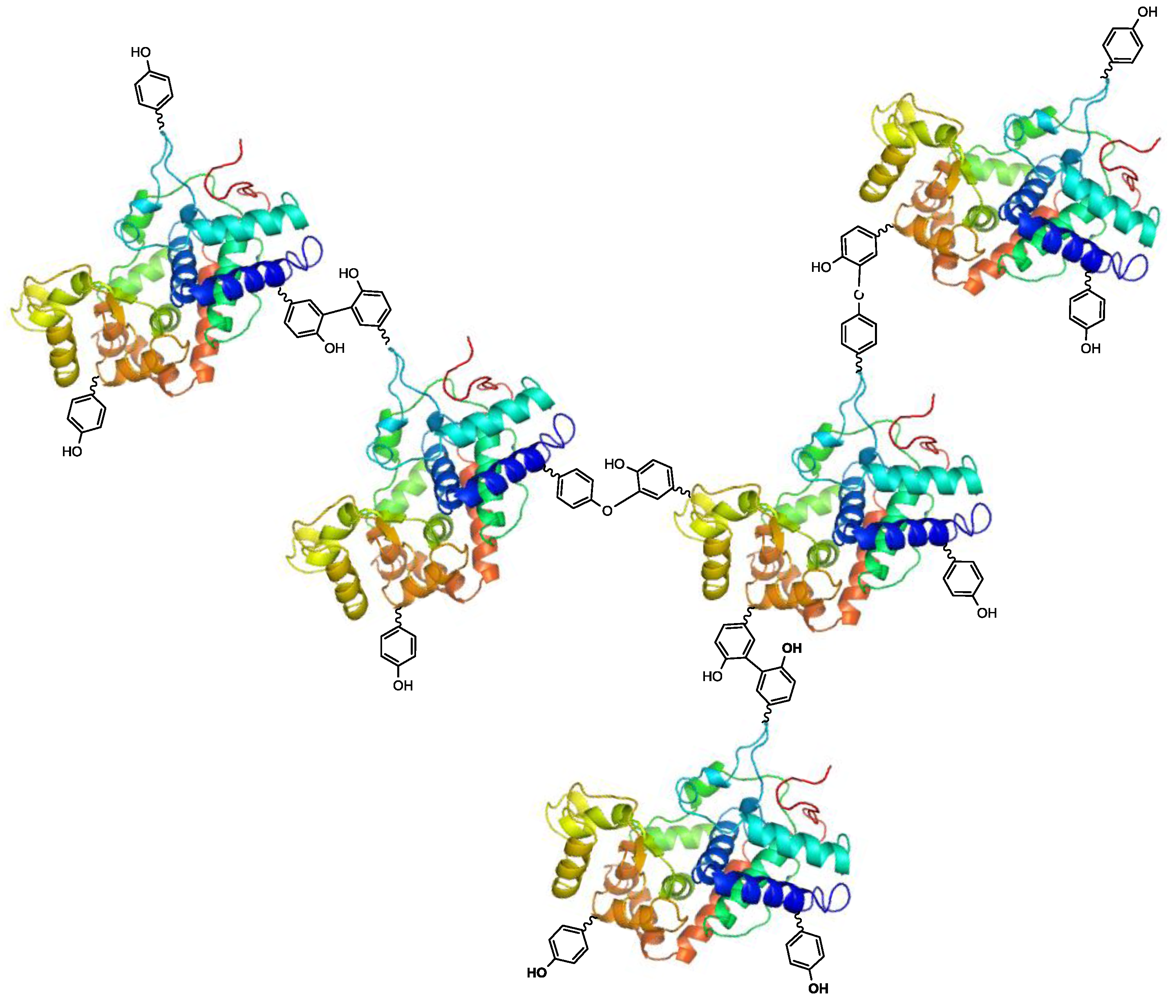
2.2. Laccase-Initiated Polymerization
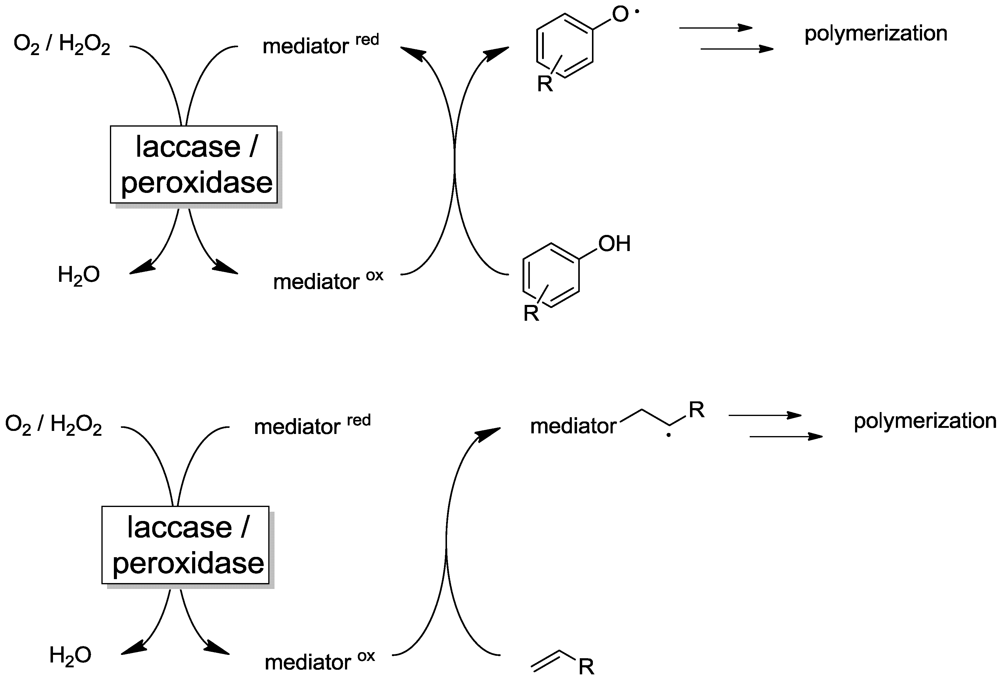
2.3. Laccase-/Peroxidase-Mediator-Systems (LMS/PMS)
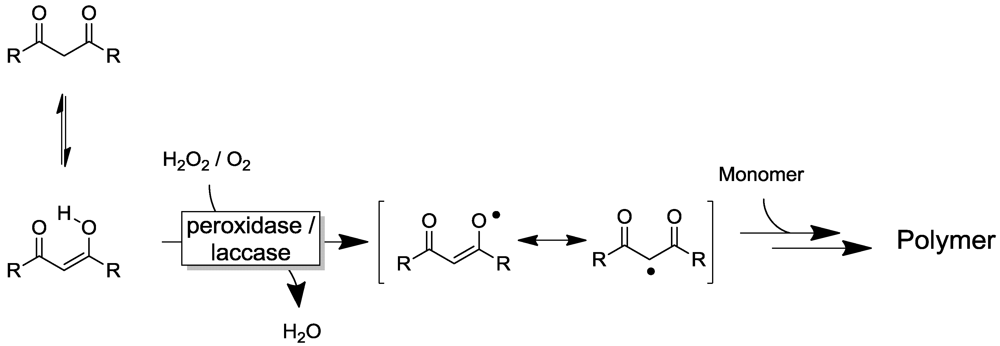
2.4. Other Enzyme Systems/Miscellaneous
3. Controlling the Structure of the Polymers
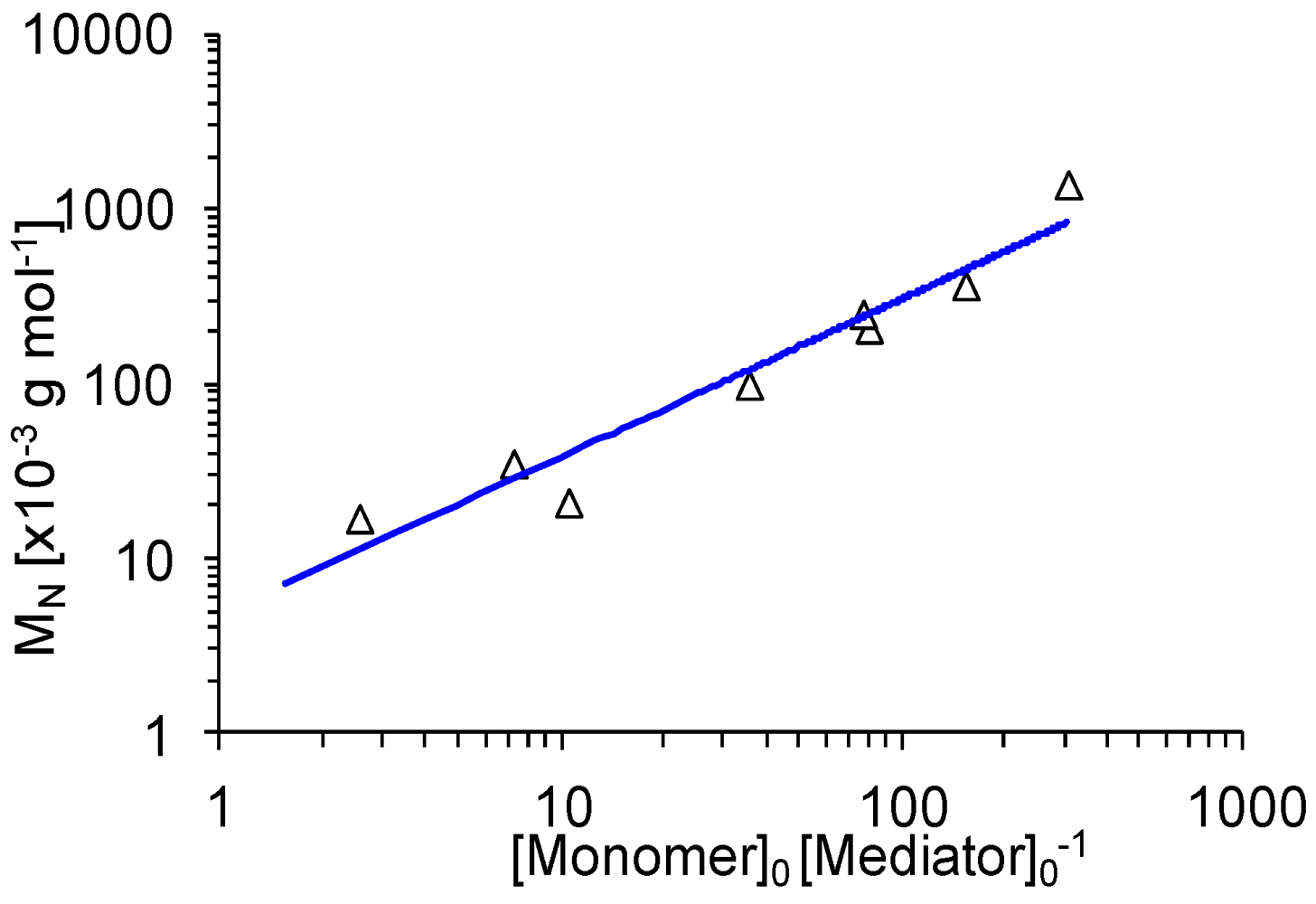
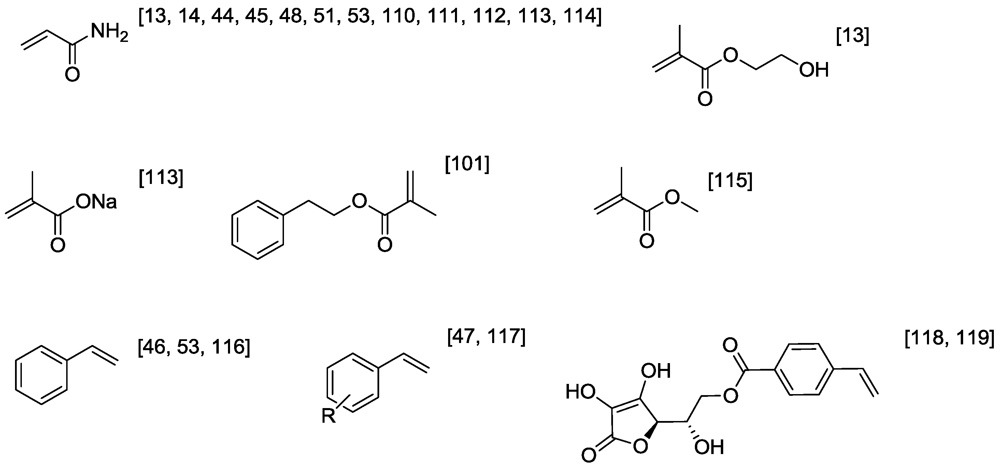
3.1. Controlling Vinyl Polymerization
| Initiator | Yield [%] | MW [×10−3 g·mol−1] | PD |
|---|---|---|---|
| Styrene polymerization [47] | |||
 | 17 | 27 | 2.1 |
 | 59 | 68 | 2.0 |
 | 14 | 80 | 2.0 |
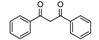 | 14 | 97 | 2.2 |
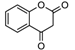 | 14 | 57 | 1.6 |
| Acrylamide polymerization [48] | |||
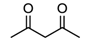 | 93 | 124 | 2.5 |
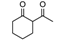 | 84 | 56 | 2.9 |
 | 76 | 5 | 4.4 |
 | 72 | 27 | 3.3 |
 | 78 | 85 | 2.7 |
 | 38 | 10.5 | 3.9 |
3.2. Controlling Polymerization of Aromatics
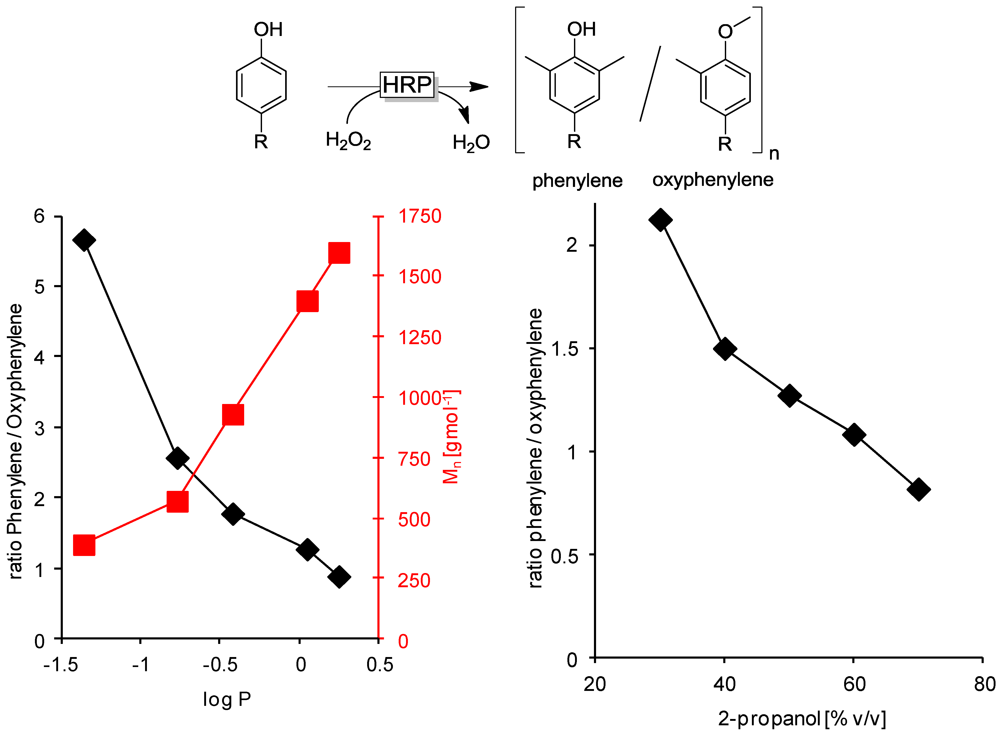
3.2.1. Cosolvents
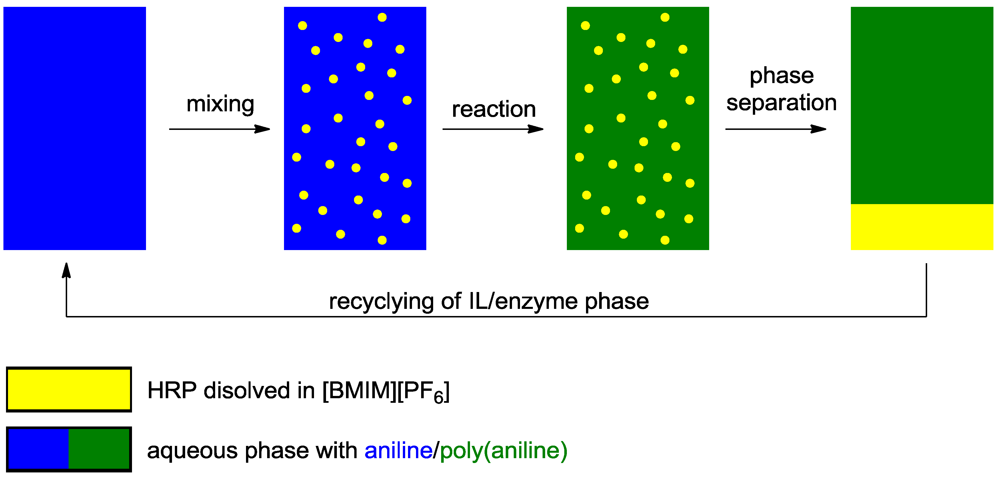
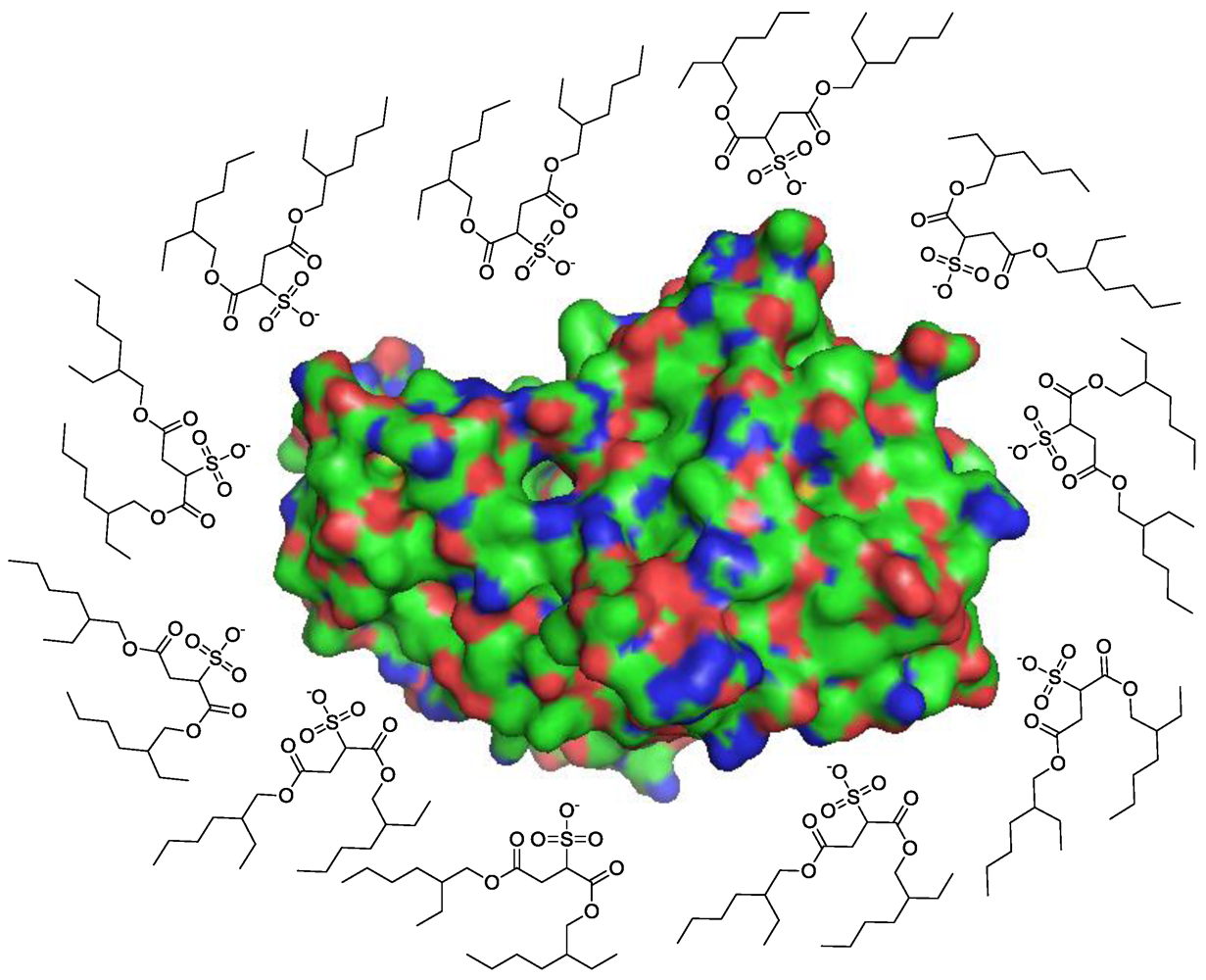
3.2.2. Templates
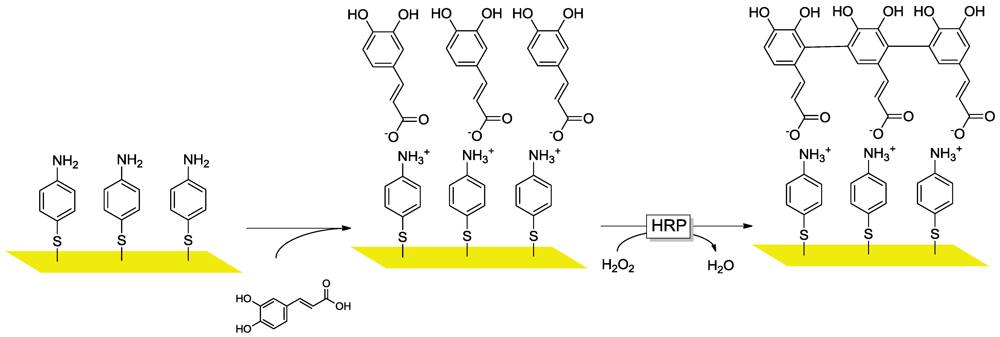
3.2.3. Substrate Engineering
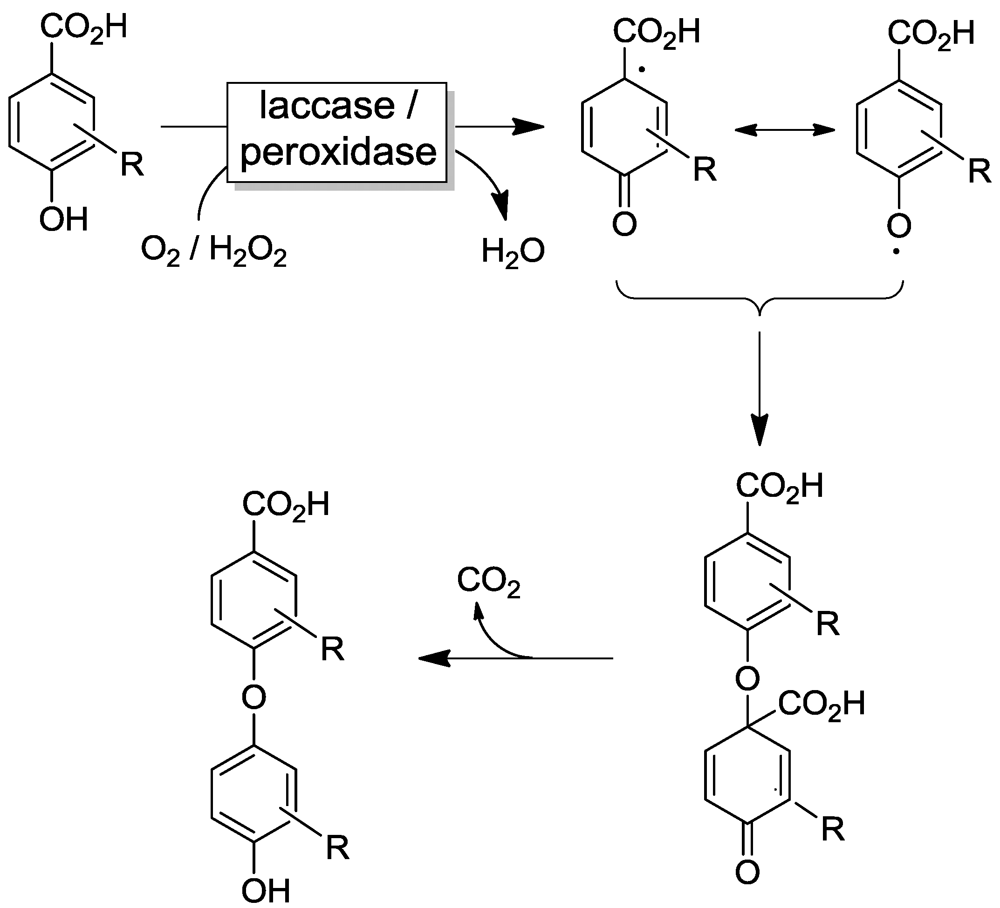
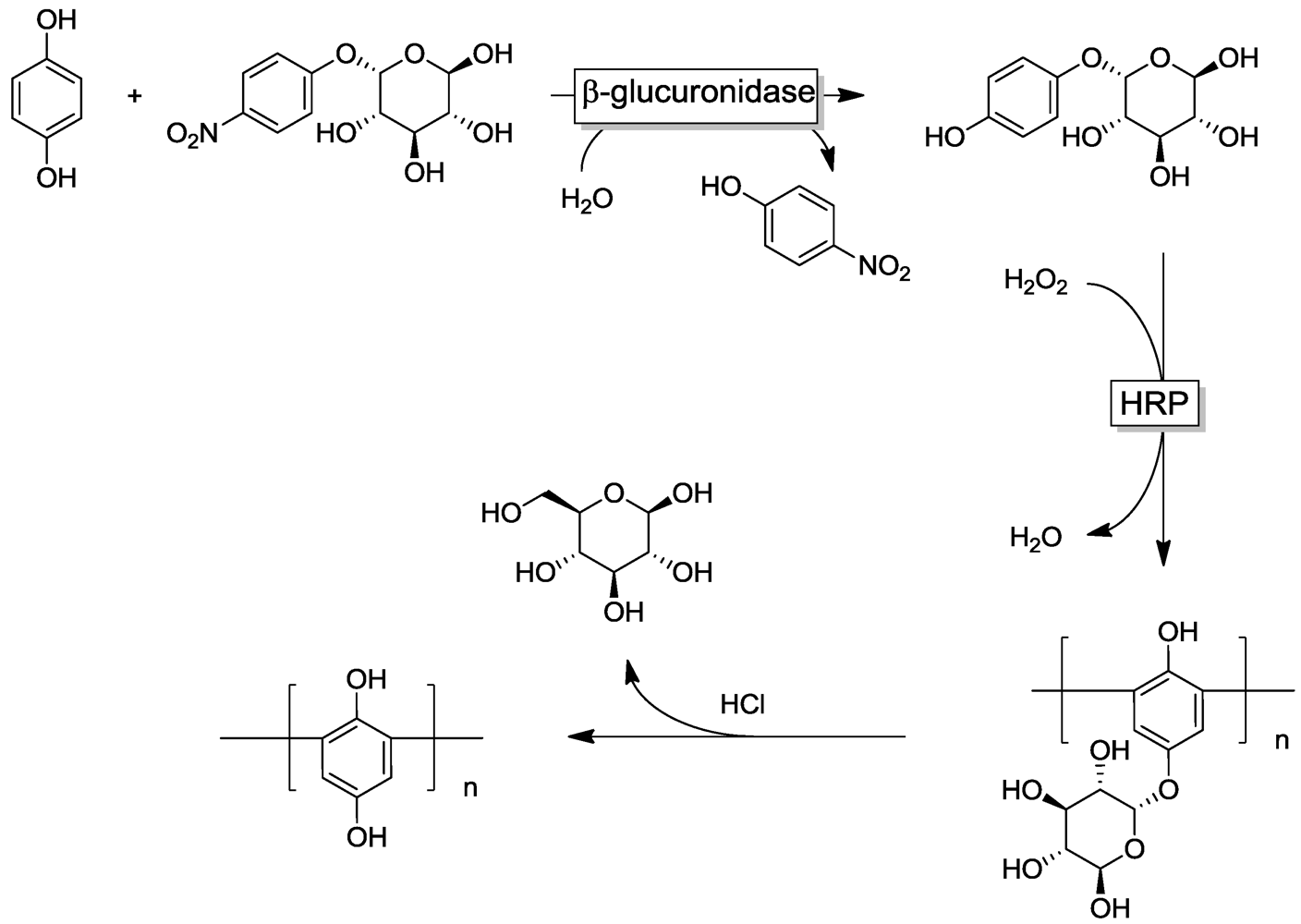
4. Selected Examples
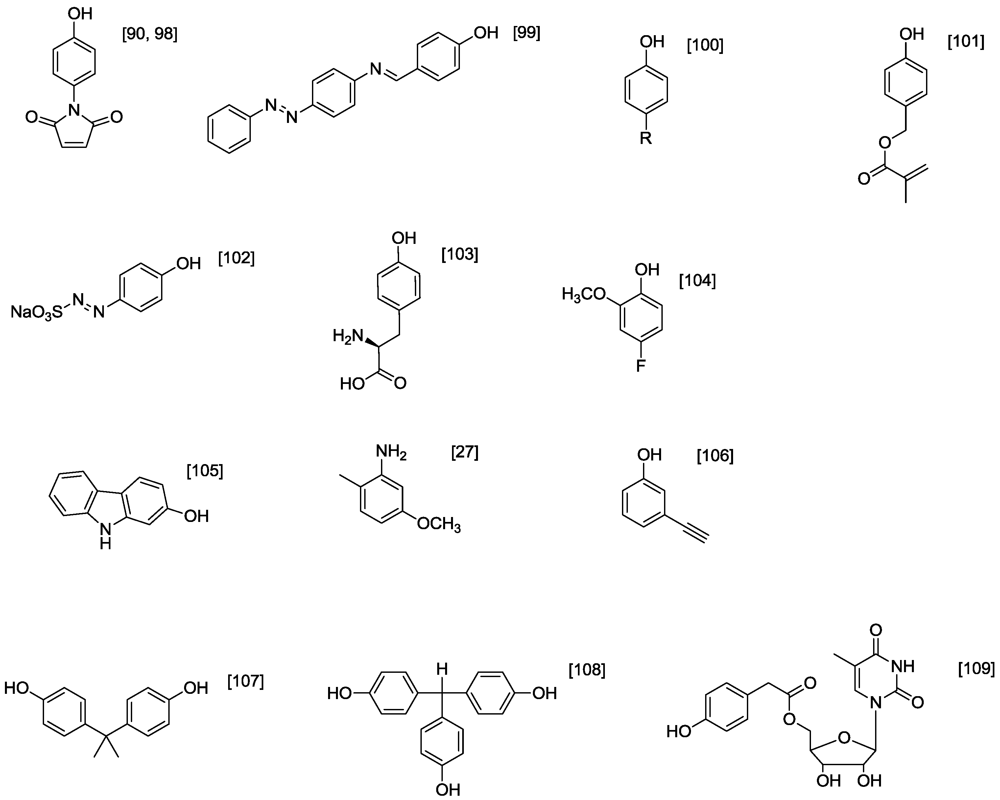
4.1. Polyphenols
| monomer | catalyst | reference |
|---|---|---|
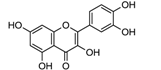 | HRP | [120] |
| laccase | [121] | |
| Quercitin | ||
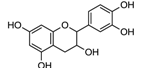 | laccase | [122,123,124] |
| Catechin | ||
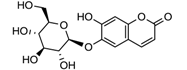 | laccase | [125,126] |
| Esculin | ||
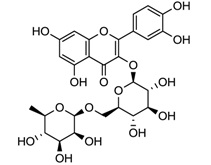 | laccase | [127,128] |
| Rutin | ||
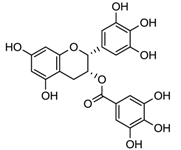 | laccase | [129] |
| Epigallocatechin |
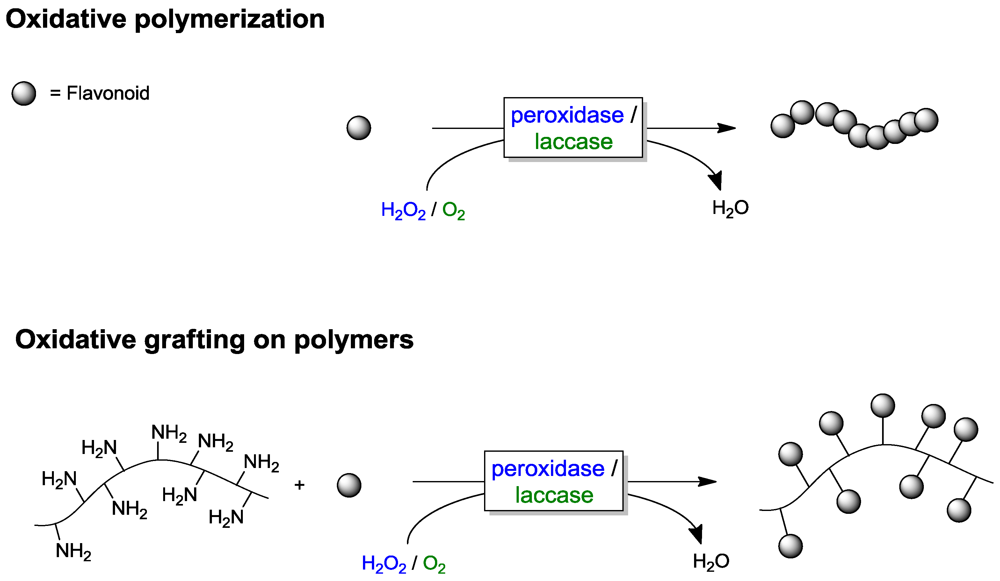
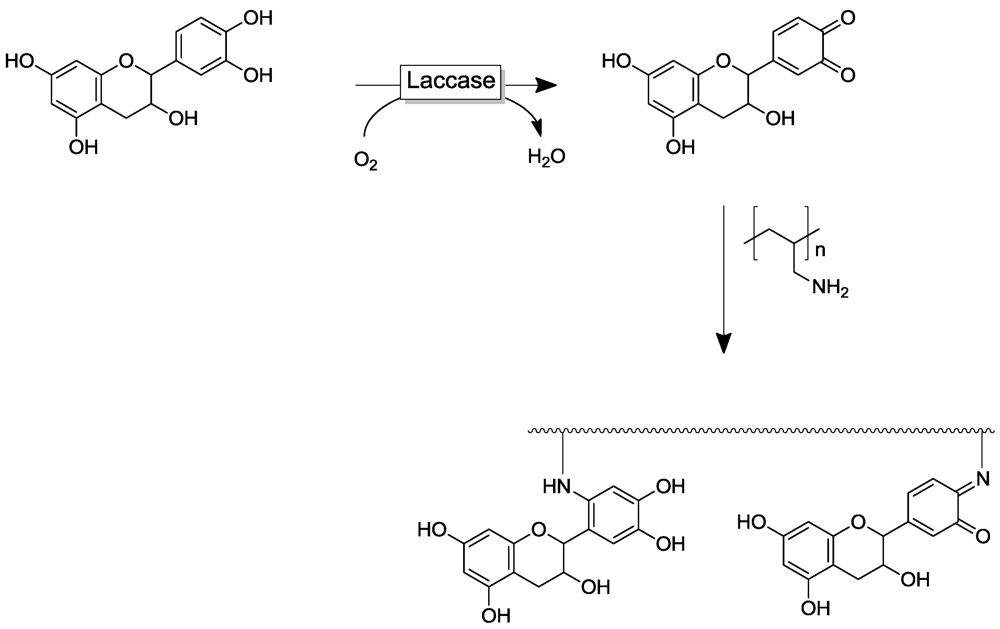
4.2. Polymer Modification
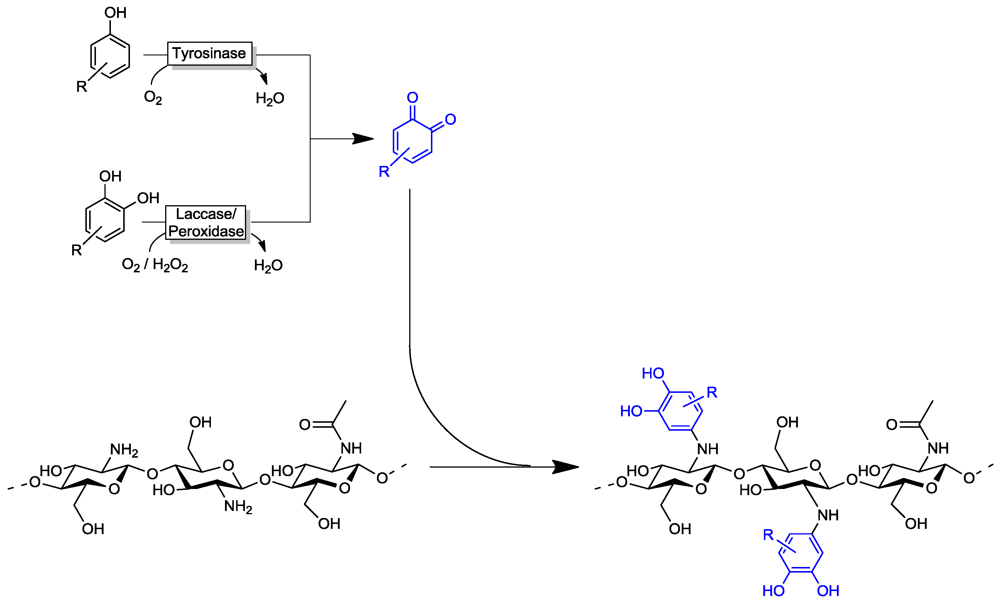
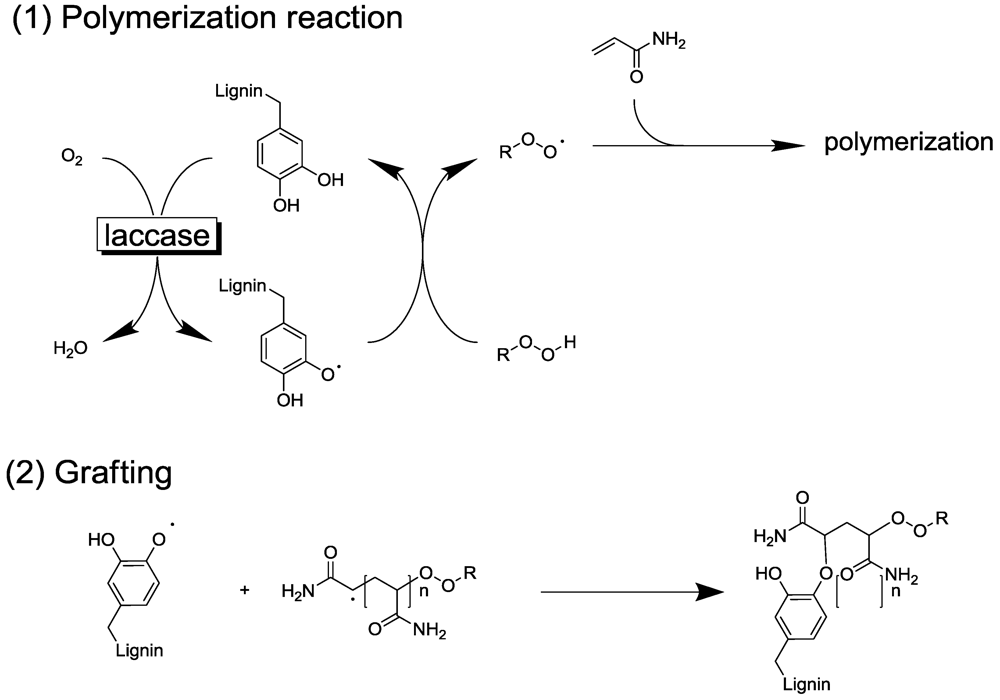
4.3. Artificial Urushi
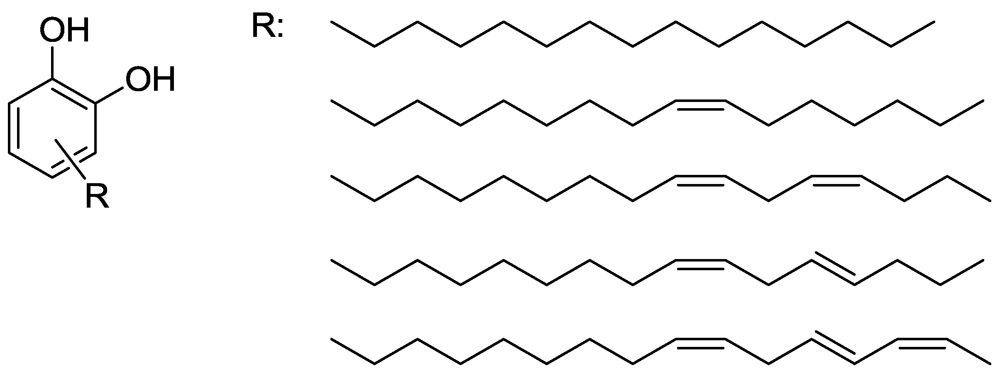
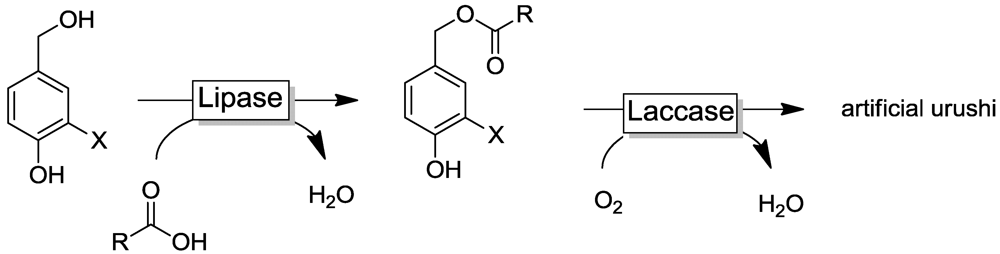
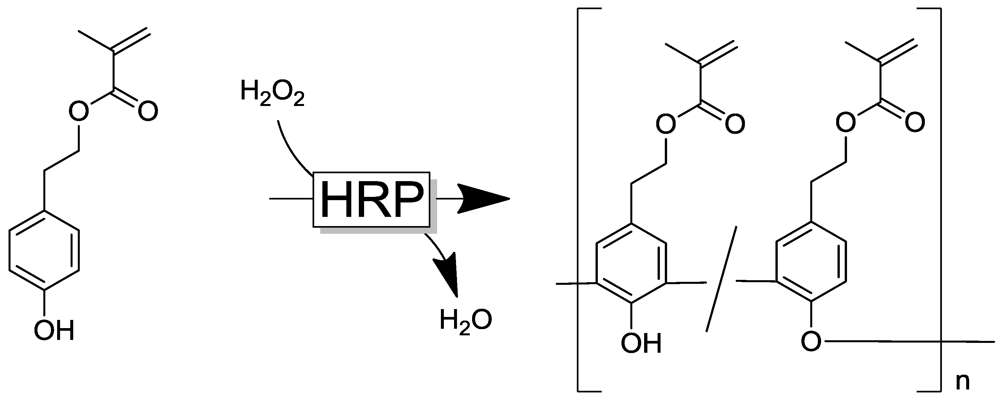
4.4. Selected Examples for Chemoselective Polymerization
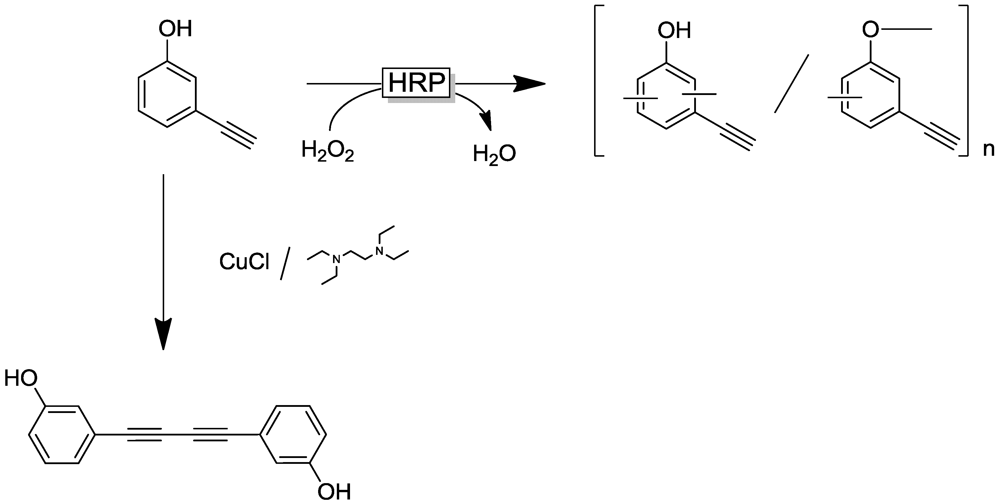
4.5. Phenol Detoxification
5. Conclusions
Acknowledgments
References
- Wenda, S.; Illner, S.; Mell, A.; Kragl, U. Industrial biotechnology—The future of green chemistry? Green Chem. 2011, 13, 3007–3047. [Google Scholar] [CrossRef]
- Hollmann, F.; Arends, I.W.C.E.; Buehler, K.; Schallmey, A.; Buhler, B. Enzyme-mediated oxidations for the chemist. Green Chem. 2011, 13, 226–265. [Google Scholar] [CrossRef]
- Hollmann, F.; Arends, I.W.C.E.; Holtmann, D. Enzymatic reductions for the chemist. Green Chem. 2011, 13, 2285–2313. [Google Scholar] [CrossRef]
- Kuhn, D.; Kholiq, M.A.; Heinzle, E.; Bühler, B.; Schmid, A. Intensification and economic and ecological assessment of a biocatalytic oxyfunctionalization process. Green Chem. 2010, 12, 815–827. [Google Scholar] [CrossRef]
- Thum, O.; Oxenbøll, K.M. Biocatalysis: A sustainable process for production of cosmetic ingredients. In Proceedings of the International Federation of Societies of Cosmetic Chemists, IFSCC Congress 2006, Osaka, Japan, 16–19 October 2006.
- Henderson, R.K.; Jiminez-Gonzalez, C.; Preston, C.; Constable, D.J.C.; Woodley, J.M. EHS & LCA assessment for 7-ACA synthesis A case study for comparing biocatalytic & chemical synthesis. Ind. Biotechnol. 2008, 4, 180–192. [Google Scholar] [CrossRef]
- Sheldon, R.A. E factors, green chemistry and catalysis: An odyssey. Chem. Commun. 2008, 3352–3365. [Google Scholar] [CrossRef]
- Kobayashi, S.; Makino, A. Enzymatic polymer synthesis: An opportunity for green polymer chemistry. Chem. Rev. 2009, 109, 5288–5353. [Google Scholar] [CrossRef]
- Uyama, H.; Kobayashi, S.; Ritter, H.; Kaplan, D. Enzymatic synthesis and properties of polymers from polyphenols. In Enzyme-Catalyzed Synthesis of Polymers; Springer: Berlin, Germany and Heidelberg, Germany, 2006; Volume 194, pp. 51–67. [Google Scholar]
- Singh, A.; Kaplan, D.L. Enzyme-based vinyl polymerization. J. Polym. Environ. 2002, 10, 85–91. [Google Scholar] [CrossRef]
- Uyama, H. Artificial polymeric flavonoids: Synthesis and applications. Macromol. Biosci. 2007, 7, 410–422. [Google Scholar] [CrossRef]
- Reihmann, M.; Ritter, H. Synthesis of phenol polymers using peroxidases. In Enzyme-Catalyzed Synthesis of Polymers; Kobayashi, S., Ritter, H., Kaplan, D., Eds.; Springer-Verlag: Berlin, Germany, 2006; Volume 194, pp. 1–49. [Google Scholar]
- Derango, R.; Chiang, L.-C.; Dowbenko, R.; Lasch, J. Enzyme-mediated polymerization of acrylic monomers. Biotechnol. Technol. 1992, 6, 523–526. [Google Scholar]
- Tsujimoto, T.; Uyama, H.; Kobayashi, S. Polymerization of vinyl monomers using oxidase catalysts. Macromol. Biosci. 2001, 1, 228–232. [Google Scholar] [CrossRef]
- Kausaite, A.; Ramanaviciene, A.; Ramanavicius, A. Polyaniline synthesis catalysed by glucose oxidase. Polymer 2009, 50, 1846–1851. [Google Scholar] [CrossRef]
- Hofrichter, M.; Ullrich, R.; Pecyna, M.J.; Liers, C.; Lundell, T. New and classic families of secreted fungal heme peroxidases. Appl. Microbiol. Biotechnol. 2010, 87, 871–897. [Google Scholar] [CrossRef]
- Van Rantwijk, F.; Sheldon, R.A. Selective oxygen transfer catalysed by heme peroxidases: Synthetic and mechanistic aspects. Curr. Opin. Biotechnol. 2000, 11, 554–564. [Google Scholar] [CrossRef]
- Hanson, R.L.; Howell, J.M.; LaPorte, T.L.; Donovan, M.J.; Cazzulino, D.L.; Zannella, V.; Montana, M.A.; Nanduri, V.B.; Schwarz, S.R.; Eiring, R.F.; et al. Synthesis of allysine ethylene acetal using phenylalanine dehydrogenase from Thermoactinomyces intermedius. Enzym. Microb. Technol. 2000, 26, 348–358. [Google Scholar] [CrossRef]
- Valderrama, B.; Ayala, M.; Vazquez-Duhalt, R. Suicide inactivation of peroxidases and the challenge of engineering more robust enzymes. Chem. Biol. 2002, 9, 555–565. [Google Scholar] [CrossRef]
- Hiner, A.N.P.; Hernández-Ruiz, J.; Williams, G.A.; Arnao, M.B.; García-Cánovas, F.; Acosta, M. Catalase-like oxygen production by horseradish peroxidase must predominantly be an enzyme-catalyzed reaction. Arch. Biochem. Biophys. 2001, 392, 295–302. [Google Scholar] [CrossRef]
- Collinson, E.; Dainton, F.S.; McNaughton, G.S. The polymerization of acrylamide in aqueous solution. Part 1.The X- and g-ray initiated reaction. Trans. Faraday Soc. 1957, 53, 476–488. [Google Scholar] [CrossRef]
- Hollmann, F.; Arends, I.W.C.E.; Buehler, K. Biocatalytic redox reactions for organic synthesis: Nonconventional regeneration methods. ChemCatChem 2010, 2, 762–782. [Google Scholar] [CrossRef]
- Churakova, E.; Kluge, M.; Ullrich, R.; Arends, I.; Hofrichter, M.; Hollmann, F. Specific photobiocatalytic oxyfunctionalization reactions. Angew. Chem. Int. Ed. 2011, 50, 10716–10719. [Google Scholar]
- Perez, D.I.; Mifsud Grau, M.; Arends, I.W.C.E.; Hollmann, F. Visible light-driven and chloroperoxidase-catalyzed oxygenation reactions. Chem. Commun. 2009, 6848–6850. [Google Scholar]
- Uyama, H.; Kurioka, H.; Kobayashi, S. Novel bienzymatic catalysis system for oxidative polymerization of phenols. Polym. J. 1997, 29, 190–192. [Google Scholar] [CrossRef]
- Taboada-Puig, R.; Junghanns, C.; Demarche, P.; Moreira, M.T.; Feijoo, G.; Lema, J.M.; Agathos, S.N. Combined cross-linked enzyme aggregates from versatile peroxidase and glucose oxidase: Production, partial characterization and application for the elimination of endocrine disruptors. Bioresour. Technol. 2011, 102, 6593–6599. [Google Scholar]
- Kim, S.C.; Huh, P.; Kumar, J.; Kim, B.; Lee, J.O.; Bruno, F.F.; Samuelson, L.A. Synthesis of polyaniline derivatives via biocatalysis. Green Chem. 2007, 9, 44–48. [Google Scholar] [CrossRef]
- Kunamneni, A.; Camarero, S.; Garcia-Burgos, C.; Plou, F.; Ballesteros, A.; Alcalde, M. Engineering and applications of fungal laccases for organic synthesis. Microb. Cell Factories 2008, 7. [Google Scholar] [CrossRef]
- Witayakran, S.; Ragauskas, A.J. Synthetic applications of laccase in green chemistry. Adv. Synth. Catal. 2009, 351, 1187–1209. [Google Scholar] [CrossRef]
- Riva, S. Laccases: Blue enzymes for green chemistry. Trends Biotechnol. 2006, 24, 219–226. [Google Scholar] [CrossRef]
- Cañas, A.I.; Camarero, S. Laccases and their natural mediators: Biotechnological tools for sustainable eco-friendly processes. Biotechnol. Adv. 2010, 28, 694–705. [Google Scholar] [CrossRef]
- Won, K.; Kim, Y.H.; An, E.S.; Lee, Y.S.; Song, B.K. Horseradish peroxidase-catalyzed polymerization of cardanol in the presence of redox mediators. Biomacromolecules 2004, 5, 1–4. [Google Scholar] [CrossRef]
- Chelikani, R.; Kim, Y.H.; Yoon, D.Y.; Kim, D.S. Enzymatic polymerization of natural anacardic acid and antibiofouling effects of polyanacardic acid coatings. Appl. Biochem. Biotechnol. 2009, 157, 263–277. [Google Scholar] [CrossRef]
- Song, H.K.; Palmore, G.T.R. Conductive polypyrrole via enzyme catalysis. J. Phys. Chem. B 2005, 109, 19278–19287. [Google Scholar]
- Cruz-Silva, R.; Amaro, E.; Escamilla, A.; Nicho, M.E.; Sepulveda-Guzman, S.; Arizmendi, L.; Romero-Garcia, J.; Castillon-Barraza, F.F.; Farias, M.H. Biocatalytic synthesis of polypyrrole powder, colloids, and films using horseradish peroxidase. J. Colloid Interface Sci. 2008, 328, 263–269. [Google Scholar] [CrossRef]
- Eibes, G.; Debernardi, G.; Feijoo, G.; Moreira, M.T.; Lema, J.M. Oxidation of pharmaceutically active compounds by a ligninolytic fungal peroxidase. Biodegradation 2011, 22, 539–550. [Google Scholar] [CrossRef]
- Kim, S.; Silva, C.; Evtuguin, D.V.; Gamelas, J.A.F.; Cavaco-Paulo, A. Polyoxometalate/laccase-mediated oxidative polymerization of catechol for textile dyeing. Appl. Microbiol. Biotechnol. 2011, 89, 981–987. [Google Scholar] [CrossRef]
- Nagarajan, S.; Kumar, J.; Bruno, F.F.; Samuelson, L.A.; Nagarajan, R. Biocatalytically synthesized poly(3,4-ethylenedioxythiophene). Macromolecules 2008, 41, 3049–3052. [Google Scholar]
- Sigg, S.J.; Seidi, F.; Renggli, K.; Silva, T.B.; Kali, G.; Bruns, N. Horseradish peroxidase as a catalyst for atom transfer radical polymerization. Macromol. Rapid Commun. 2011, 32, 1710–1715. [Google Scholar] [CrossRef]
- Ng, Y.H.; di Lena, F.; Chai, C.L.L. PolyPEGA with predetermined molecular weights from enzyme-mediated radical polymerization in water. Chem. Commun. 2011, 47, 6464–6466. [Google Scholar]
- Ng, Y.H.; di Lena, F.; Chai, C.L.L. Metalloenzymatic radical polymerization using alkyl halides as initiators. Polym. Chem. 2011, 2, 589–594. [Google Scholar] [CrossRef]
- Tsarevsky, N.V.; Matyjaszewski, K. “Green” atom transfer radical polymerization: From process design to preparation of well-defined environmentally friendly polymeric materials. Chem. Rev. 2007, 107, 2270–2299. [Google Scholar] [CrossRef]
- Rodrigues, A.P.; da Fonseca, L.M.; de Faria Oliveira, O.M.; Brunetti, I.L.; Ximenes, V.F. Oxidation of acetylacetone catalyzed by horseradish peroxidase in the absence of hydrogen peroxide. Biochim. Biophys. Acta 2006, 1760, 1755–1761. [Google Scholar]
- Durand, A.; Lalot, T.; Brigodiot, M.; Maréchal, E. Enzyme-mediated radical initiation of acrylamide polymerization: Main characteristics of molecular weight control. Polymer 2001, 42, 5515–5521. [Google Scholar] [CrossRef]
- Hollmann, F.; Gumulya, Y.; Toelle, C.; Liese, A.; Thum, O. Evaluation of the laccase from myceliophthora thermophila as industrial biocatalyst for polymerization reactions. Macromolecules 2008, 41, 8520–8524. [Google Scholar] [CrossRef]
- Qi, G.G.; Jones, C.W.; Schork, F.J. Enzyme-initiated miniemulsion polymerization. Biomacromolecules 2006, 7, 2927–2930. [Google Scholar] [CrossRef]
- Singh, A.; Ma, D.; Kaplan, D.L. Enzyme-mediated free radical polymerization of styrene. Biomacromolecules 2000, 1, 592–596. [Google Scholar] [CrossRef]
- Teixeira, D.; Lalot, T.; Brigodiot, M.; Marechal, E. β-diketones as key compounds in free-radical polymerization by enzyme-mediated initiation. Macromolecules 1999, 32, 70–72. [Google Scholar] [CrossRef]
- Baader, W.J.; Bohne, C.; Cilento, G.; Dunford, H.B. Peroxidase-catalyzed formation of triplet acetone and chemiluminescence from isobutyraldehyde and molecular oxygen. J. Biol. Chem. 1985, 260, 10217–10225. [Google Scholar]
- Parravano, G. Chain reactions induced by enzymic systems. J. Am. Chem. Soc. 1951, 73, 183–184. [Google Scholar] [CrossRef]
- Emery, O.; Lalot, T.; Brigodiot, M.; Maréchal, E. Free-Radical polymerization of acrylamide by horseradish peroxidase-mediated initiation. J. Polym. Sci. A 1997, 35, 3331–3333. [Google Scholar] [CrossRef]
- Ikeda, R.; Tanaka, H.; Uyama, H.; Kobayashi, S. Laccase-catalyzed polymerization of acrylamide. Macromol. Rapid Commun. 1998, 19, 423–425. [Google Scholar] [CrossRef]
- Villarroya, S.; Thurecht, K.J.; Howdle, S.M. HRP-mediated inverse emulsion polymerisation of acrylamide in supercritical carbon dioxide. Green Chem. 2008, 10, 863–867. [Google Scholar] [CrossRef]
- Nieto, M.; Nardecchia, S.; Peinado, C.; Catalina, F.; Abrusci, C.; Gutierrez, M.C.; Ferrer, M.L.; del Monte, F. Enzyme-induced graft polymerization for preparation of hydrogels: Synergetic effect of laccase-immobilized-cryogels for pollutants adsorption. Soft Matter 2010, 6, 3533–3540. [Google Scholar]
- Ayyagari, M.S.R.; Kaplan, D.L.; Chatterjee, S.; Walker, J.E.; Akkara, J.A. Solvent effects in horseradish peroxidase-catalyzed polyphenol synthesis. Enzym. Microb. Technol. 2002, 30, 3–9. [Google Scholar] [CrossRef]
- Akkara, J.A.; Ayyagari, M.S.R.; Bruno, F.F. Enzymatic synthesis and modification of polymers in nonaqueous solvents. Trends Biotechnol. 1999, 17, 67–73. [Google Scholar] [CrossRef]
- Dordick, J.S.; Marletta, M.A.; Klibanov, A.M. Polymerization of phenols catalyzed by peroxidase in nonaqueous media. Biotechnol. Bioeng. 1987, 30, 31–36. [Google Scholar] [CrossRef]
- Oguchi, T.; Tawaki, S.; Uyama, H.; Kobayashi, S. Soluble polyphenol. Macromol. Rapid Commun. 1999, 20, 401–403. [Google Scholar] [CrossRef]
- Mita, N.; Maruichi, N.; Tonami, H.; Nagahata, R.; Tawaki, S.; Uyama, H.; Kobayashi, S. Enzymatic oxidative polymerization of p-t-butylphenol and characterization of the product polymer. Bull. Chem. Soc. Jpn. 2003, 76, 375–379. [Google Scholar]
- Mita, N.; Tawaki, S.; Uyama, H.; Kobayashi, S. Structural control in enzymatic oxidative polymerization of phenols with varying the solvent and substituent nature. Chem. Lett. 2002, 402–403. [Google Scholar]
- Mita, N.; Tawaki, S.; Uyama, H.; Kobayashi, S. Precise structure control of enzymatically synthesized polyphenols. Bull. Chem. Soc. Jpn. 2004, 77, 1523–1527. [Google Scholar] [CrossRef]
- Mita, N.; Tawaki, S.-I.; Uyama, H.; Kobayashi, S. Laccase-catalyzed oxidative polymerization of phenols. Macromol. Biosci. 2003, 3, 253–257. [Google Scholar] [CrossRef]
- Sgalla, S.; Fabrizi, G.; Cacchi, S.; Macone, A.; Bonamore, A.; Boffi, A. Horseradish peroxidase in ionic liquids: Reactions with water insoluble phenolic substrates. J. Mol. Catal. B 2007, 44, 144–148. [Google Scholar] [CrossRef]
- Eker, B.; Zagorevski, D.; Zhu, G.Y.; Linhardt, R.J.; Dordick, J.S. Enzymatic polymerization of phenols in room-temperature ionic liquids. J. Mol. Catal. B 2009, 59, 177–184. [Google Scholar] [CrossRef]
- Zaragoza-Gasca, P.; Villamizar-Gálvez, O.J.; García-Arrazola, R.; Gimeno, M.; Bárzana, E. Use of ionic liquid for the enzyme-catalyzed polymerization of phenols. Polym. Adv. Technol. 2010, 21, 454–456. [Google Scholar]
- Rumbau, V.; Marcilla, R.; Ochoteco, E.; Pomposo, J.A.; Mecerreyes, D. Ionic liquid immobilized enzyme for biocatalytic synthesis of conducting polyaniline. Macromolecules 2006, 39, 8547–8549. [Google Scholar] [CrossRef]
- Nakamura, R.; Matsushita, Y.; Umemoto, K.; Usuki, A.; Fukushima, K. Enzymatic polymerization of coniferyl alcohol in the presence of cyclodextrins. Biomacromolecules 2006, 7, 1929–1934. [Google Scholar] [CrossRef]
- Reihmann, M.H.; Ritter, H. Oxidative oligomerization of cyclodextrin-complexed bifunctional phenols catalyzed by horseradish peroxidase in water. Macromol. Chem. Phys. 2000, 201, 798–804. [Google Scholar] [CrossRef]
- Mita, N.; Tawaki, S.; Uyama, H.; Kobayashi, S. Enzymatic oxidative polymerization of phenol in an aqueous solution in the presence of a catalytic amount of cyclodextrin. Macromol. Biosci. 2002, 2, 127–130. [Google Scholar] [CrossRef]
- Dordick, J.S.; Marletta, M.A.; Klibanov, A.M. Peroxidase depolymerize lignin in organic media but not in water. Proc. Natl. Acad. Sci. USA 1986, 83, 6255–6257. [Google Scholar]
- Angerer, P.S.; Studer, A.; Witholt, B.; Li, Z. Oxidative polymerization of a substituted phenol with ion-paired horseradish peroxidase in an organic solvent. Macromolecules 2005, 38, 6248–6250. [Google Scholar]
- Oshima, T.; Sato, M.; Shikaze, Y.; Ohto, K.; Inoue, K.; Baba, Y. Enzymatic polymerization of o-phenylendiamine with cytochrome c activated by a calixarene derivative in organic media. Biochem. Eng. J. 2007, 35, 66–70. [Google Scholar] [CrossRef]
- Walde, P.; Guo, Z.W. Enzyme-catalyzed chemical structure-controlling template polymerization. Soft Matter 2011, 7, 316–331. [Google Scholar] [CrossRef]
- Samuelson, L.A.; Anagnostopoulos, A.; Alva, K.S.; Kumar, J.; Tripathy, S.K. Biologically derived conducting and water soluble polyaniline. Macromolecules 1998, 31, 4376–4378. [Google Scholar]
- Nabid, M.R.; Taheri, S.S.; Sedghi, R.; Rezaei, S.J.T. Synthesis and characterization of chemiluminescent conducting polyluminol via biocatalysis. Macromol. Res. 2011, 19, 280–285. [Google Scholar] [CrossRef]
- Nabid, M.R.; Sedghi, R.; Entezami, A.A. Enzymatic oxidation of alkoxyanilines for preparation of conducting polymers. J. Appl. Polym. Sci. 2007, 103, 3724–3729. [Google Scholar] [CrossRef]
- Huh, P.; Kim, S.C.; Kim, Y.; Wang, Y.; Singh, J.; Kumar, J.; Samuelson, L.A.; Kim, B.S.; Jo, N.J.; Lee, J.O. Optical and electrochemical detection of saccharides with poly(aniline-co-3-am nobenzeneboronic acid) prepared from enzymatic polymerization. Biomacromolecules 2007, 8, 3602–3607. [Google Scholar] [CrossRef]
- Karamyshev, A.V.; Shleev, S.V.; Koroleva, O.V.; Yaropolov, A.I.; Sakharov, I.Y. Laccase-catalyzed synthesis of conducting polyaniline. Enzym. Microb. Technol. 2003, 33, 556–564. [Google Scholar]
- Tewari, A.; Kokil, A.; Ravichandran, S.; Nagarajan, S.; Bouldin, R.; Samuelson, L.A.; Nagarajan, R.; Kumar, J. Soybean peroxidase catalyzed enzymatic synthesis of pyrrole/EDOT copolymers. Macromol. Chem. Phys. 2010, 211, 1610–1617. [Google Scholar]
- Vasil’eva, I.; Morozova, O.; Shumakovich, G.; Yaropolov, A. Synthesis of electroconductive polyaniline using immobilized laccase. Appl. Biochem. Microbiol. 2009, 45, 27–30. [Google Scholar] [CrossRef]
- Nagarajan, R.; Tripathy, S.; Kumar, J.; Bruno, F.F.; Samuelson, L. An enzymatically synthesized conducting molecular complex of polyaniline and poly(vinylphosphonic acid). Macromolecules 2000, 33, 9542–9547. [Google Scholar]
- Thiyagarajan, M.; Samuelson, L.A.; Kumar, J.; Cholli, A.L. Helical conformational specificity of enzymatically synthesized water-soluble conducting polyaniline nanocomposites. J. Am. Chem. Soc. 2003, 125, 11502–11503. [Google Scholar]
- Nagarajan, R.; Liu, W.; Kumar, J.; Tripathy, S.K.; Bruno, F.F.; Samuelson, L.A. Manipulating DNA conformation using intertwined conducting polymer chains. Macromolecules 2001, 34, 3921–3927. [Google Scholar]
- Ma, Y.; Zhang, J.; Zhang, G.; He, H. Polyaniline nanowires on si surfaces fabricated with DNA templates. J. Am. Chem. Soc. 2004, 126, 7097–7101. [Google Scholar]
- Streltsov, A.V.; Morozova, O.V.; Arkharova, N.A.; Klechkovskaya, V.V.; Staroverova, I.N.; Shumakovich, G.P.; Yaropolov, A.I. Synthesis and characterization of conducting polyaniline prepared by laccase-catalyzed method in sodium dodecylbenzenesulfonate micellar solutions. J. Appl. Polym. Sci. 2009, 114, 928–934. [Google Scholar]
- Streltsov, A.V.; Shumakovich, G.P.; Morozova, O.V.; Gorbacheva, M.A.; Yaropolov, A.I. Micellar laccase-catalyzed synthesis of electroconductive polyaniline. Appl. Biochem. Microbiol. 2008, 44, 264–270. [Google Scholar] [CrossRef]
- Bouldin, R.; Ravichandran, S.; Kokil, A.; Garhwal, R.; Nagarajan, S.; Kumar, J.; Bruno, F.F.; Samuelson, L.A.; Nagarajan, R. Synthesis of polypyrrole with fewer structural defects using enzyme catalysis. Synth. Met. 2011, 161, 1611–1617. [Google Scholar]
- Kim, Y.J.; Shibata, K.; Uyama, H.; Kobayashi, S. Synthesis of ultrahigh molecular weight phenolic polymers by enzymatic polymerization in the presence of amphiphilic triblock copolymer in water. Polymer 2008, 49, 4791–4795. [Google Scholar] [CrossRef]
- Kim, Y.J.; Uyama, H.; Kobayashi, S. Enzymatic template polymerization of phenol in the presence of water-soluble polymers in an aqueous medium. Polym. J. 2004, 36, 992–998. [Google Scholar] [CrossRef]
- Kim, Y.J.; Uyama, H.; Kobayashi, S. Peroxidase-catalyzed oxidative polymerization of phenol with a nonionic polymer surfactant template in water. Macromol. Biosci. 2004, 4, 497–502. [Google Scholar] [CrossRef]
- Kim, Y.J.; Uyama, H.; Kobayashi, S. Regioselective synthesis of poly(phenylene) as a complex with poly(ethylene glycol) by template polymerization of phenol in water. Macromolecules 2003, 36, 5058–5060. [Google Scholar] [CrossRef]
- Bruno, F.F.; Nagarajan, R.; Stenhouse, P.; Yang, K.; Kumar, J.; Tripathy, S.K.; Samuelson, L.A. Polymerization of water-soluble conductive polyphenol using horseradish peroxidase. J. Macromol. Sci. Pure Appl. Chem. 2001, 38, 1417–1426. [Google Scholar] [CrossRef]
- Peng, Y.; Liu, H.W.; Zhang, X.Y.; Li, Y.S.; Liu, S.Y. CNT templated regioselective enzymatic polymerization of phenol in water and modification of surface of MWNT thereby. J. Polym. Sci. Pol. Chem. 2009, 47, 1627–1635. [Google Scholar] [CrossRef]
- Xu, P.; Uyama, H.; Whitten, J.E.; Kobayashi, S.; Kaplan, D.L. Peroxidase-catalyzed in situ polymerization of surface orientated caffeic acid. J. Am. Chem. Soc. 2005, 127, 11745–11753. [Google Scholar]
- Ikeda, R.; Sugihara, J.; Uyama, H.; Kobayashi, S. Enzymatic oxidative polymerization of 4-hydroxybenzoic acid derivatives to poly(phenylene oxide)s. Polym. Int. 1998, 47, 295–301. [Google Scholar] [CrossRef]
- Wang, P.; Martin, B.D.; Parida, S.; Rethwisch, D.G.; Dordick, J.S. Multienzymic synthesis of poly(hydroquinone) for use as a redox polymer. J. Am. Chem. Soc. 1995, 117, 12885–12886. [Google Scholar]
- Reihmann, M.H.; Ritter, H. Regioselective HRP-catalyzed polymerization of 4-amino-phenol. J. Macromol. Sci. Pure Appl. Chem. 2002, A39, 1369–1382. [Google Scholar]
- Reihmann, M.H.; Ritter, H. Oxidative copolymerization of para-functionalized phenols catalyzed by horseradish peroxidase and thermocrosslinking via Diels-Alder and (1+3) cycloaddition. Macromol. Biosci. 2001, 1, 170–176. [Google Scholar]
- Turac, E.; Sahmetlioglu, E. Oxidative polymerization of 4-[(4-phenylazo-phenyimino)-methyl]-phenol catalyzed by horseradish peroxidase. Synth. Met. 2010, 160, 169–172. [Google Scholar] [CrossRef]
- Uyama, H.; Kurioka, H.; Sugihara, J.; Komatsu, I.; Kobayashi, S. Oxidative polymerization of p-alkylphenols catalyzed by horseradish peroxidase. J. Polym. Sci. Pol. Chem. 1997, 35, 1453–1459. [Google Scholar] [CrossRef]
- Uyama, H.; Lohavisavapanich, C.; Ikeda, R.; Kobayashi, S. Chemoselective polymerization of a phenol derivative having a methacryl group by peroxidase catalyst. Macromolecules 1998, 31, 554–556. [Google Scholar]
- Robert, J.P.; Uyama, H.; Kobayashi, S.; Jordan, R.; Nuyken, O. First diazosulfonate homopolymer by enzymatic polymerization. Macromol. Rapid Commun. 2003, 24, 185–189. [Google Scholar] [CrossRef]
- Fukuoka, T.; Tachibana, Y.; Tonami, H.; Uyama, H.; Kobayashi, S. Enzymatic polymerization of tyrosine derivatives. Peroxidase- and protease-catalyzed synthesis of poly(tyrosine)s with different structures. Biomacromolecules 2002, 3, 768–774. [Google Scholar] [CrossRef]
- Zaragoza-Gasca, P.; Gimeno, M.; Hernandez, J.M.; Barzana, E. Novel photoconductive polyfluorophenol synthesized by an enzyme. J. Mol. Catal. B 2011, 72, 25–27. [Google Scholar] [CrossRef]
- Bilici, A.; Kaya, Ä.; Yildirim, M.; Dogan, F. Enzymatic polymerization of hydroxy-functionalized carbazole monomer. J. Mol. Catal. B 2010, 64, 89–95. [Google Scholar] [CrossRef]
- Tonami, H.; Uyama, H.; Kobayashi, S.; Fujita, T.; Taguchi, Y.; Osada, K. Chemoselective oxidative polymerization of m-ethynylphenol by peroxidase catalyst to a new reactive polyphenol. Biomacromolecules 2000, 1, 149–151. [Google Scholar] [CrossRef]
- Kim, Y.H.; An, E.S.; Park, S.Y.; Lee, J.O.; Kim, J.H.; Song, B.K. Polymerization of bisphenol a using Coprinus cinereus peroxidase (CiP) and its application as a photoresist resin. J. Mol. Catal. B 2007, 44, 149–154. [Google Scholar] [CrossRef]
- Kadota, J.; Fukuoka, T.; Uyama, H.; Hasegawa, K.; Kobayashi, S. New positive-type photoresists based on enzymatically synthesized polyphenols. Macromol. Rapid Commun. 2004, 25, 441–444. [Google Scholar] [CrossRef]
- Wang, P.; Dordick, J.S. Enzymatic synthesis of unique thymidine-containing polyphenols. Macromolecules 1998, 31, 941–943. [Google Scholar] [CrossRef]
- Durand, A.; Lalot, T.; Brigodiot, M.; Maréchal, E. Enzyme-mediated initiation of acrylamide polymerization: Reaction mechanism. Polymer 2000, 41, 8183–8192. [Google Scholar]
- Lalot, T.; Brigodiot, M.; Maréchal, E. A kinetic approach to acrylamide radical polymerization by horse radish peroxidase-mediated initiation. Polym. Int. 1999, 48, 288–292. [Google Scholar] [CrossRef]
- Zhao, Q.; Sun, J.Z.; Ren, H.; Zhou, Q.Y.; Lin, Q.C. Horseradish peroxidase immobilized in macroporous hydrogel for acrylamide polymerization. J. Polym. Sci. Pol. Chem. 2008, 46, 2222–2232. [Google Scholar] [CrossRef]
- Kalra, B.; Gross, R.A. HRP-mediated polymerizations of acrylamide and sodium acrylate. Green Chem. 2002, 4, 174–178. [Google Scholar] [CrossRef]
- Shogren, R.L.; Willett, J.L.; Biswas, A. HRP-mediated synthesis of starch-polyacrylamide graft copolymers. Carbohyd. Polym. 2009, 75, 189–191. [Google Scholar] [CrossRef]
- Kalra, B.; Gross, R.A. Horseradish peroxidase mediated free radical polymerization of methyl methacrylate. Biomacromolecules 2000, 1, 501–505. [Google Scholar] [CrossRef]
- Shan, J.; Kitamura, Y.; Yoshizawa, H. Emulsion polymerization of styrene by horseradish peroxidase-mediated initiation. Colloid Polym. Sci. 2005, 284, 108–111. [Google Scholar] [CrossRef]
- Singh, A.; Roy, S.; Samuelson, L.; Bruno, F.; Nagarajan, R.; Kumar, J.; John, V.; Kaplan, D.L. Peroxidase, hematin, and pegylated-hematin catalyzed vinyl polymerizations in water. J. Macromol. Sci. A 2001, 38, 1219–1230. [Google Scholar] [CrossRef]
- Singh, A.; Kaplan, D.L. Vitamin C functionalized poly(methyl methacrylate) for free radical scavenging. J. Macromol. Sci. Part A 2004, 41, 1377–1386. [Google Scholar] [CrossRef]
- Singh, A.; Kaplan, D.L. Biocatalytic route to ascorbic acid-modified polymers for free-radical scavenging. Adv. Mat. 2003, 15, 1291–1294. [Google Scholar] [CrossRef]
- Bruno, F.F.; Trotta, A.; Fossey, S.; Nagarajan, S.; Nagarajan, R.; Samuelson, L.A.; Kumar, J. Enzymatic synthesis and characterization of poly quercetin. J. Macromol. Sci. Part A 2010, 47, 1191–1196. [Google Scholar] [CrossRef]
- Desentis-Mendoza, R.M.; Hernandez-Sanchez, H.; Moreno, A.; Emilio, R.D.C.; Chel-Guerrero, L.; Tamariz, J.; Jaramillo-Flores, M.E. Enzymatic polymerization of phenolic compounds using laccase and tyrosinase from Ustilago maydis. Biomacromolecules 2006, 7, 1845–1854. [Google Scholar]
- Ma, H.-L.; Kermasha, S.; Gao, J.-M.; Borges, R.M.; Yu, X.-Z. Laccase-catalyzed oxidation of phenolic compounds in organic media. J. Mol. Catal. B 2009, 57, 89–95. [Google Scholar] [CrossRef]
- Kurisawa, M.; Chung, J.E.; Uyama, H.; Kobayashi, S. Laccase-catalyzed synthesis and antioxidant property of poly(catechin). Macromol. Biosci. 2003, 3, 758–764. [Google Scholar] [CrossRef]
- Bruno, F.F.; Nagarajan, S.; Nagarajan, R.; Kumar, J.; Samuelson, L.A. Biocatalytic synthesis of water-soluble oligo(catechins). J. Macromol. Sci. 2005, A42, 1547–1554. [Google Scholar]
- Anthoni, J.; Chebil, L.; Lionneton, F.; Magdalou, J.; Humeau, C.; Ghoul, M. Automated analysis of synthesized oligorutin and oligoesculin by laccase. Can. J. Chem. 2011, 89, 964–970. [Google Scholar] [CrossRef]
- Anthoni, J.; Humeau, C.; Maia, E.R.; Chebil, L.; Engasser, J.M.; Ghoul, M. Enzymatic synthesis of oligoesculin: Structure and biological activities characterizations. Eur. Food Res. Technol. 2010, 231, 571–579. [Google Scholar] [CrossRef]
- Kurisawa, M.; Chung, J.E.; Uyama, H.; Kobayashi, S. Enzymatic synthesis and antioxidant properties of poly(rutin). Biomacromolecules 2003, 4, 1394–1399. [Google Scholar] [CrossRef]
- Uzan, E.; Portet, B.; Lubrano, C.; Milesi, S.; Favel, A.; Lesage-Meessen, L.; Lomascolo, A. Pycnoporus laccase-mediated bioconversion of rutin to oligomers suitable for biotechnology applications. Appl. Microbiol. Biotechnol. 2011, 90, 97–105. [Google Scholar] [CrossRef]
- Kurisawa, M.; Chung, J.E.; Uyama, H.; Kobayashi, S. Oxidative coupling of epigallocatechin gallate amplifies antioxidant activity and inhibits xanthine oxidase activity. Chem. Commun. 2004, 294–295. [Google Scholar]
- Kudanga, T.; Nyanhongo, G.S.; Guebitz, G.M.; Burton, S. Potential applications of laccase-mediated coupling and grafting reactions: A review. Enzym. Microb. Technol. 2011, 48, 195–208. [Google Scholar] [CrossRef]
- Chung, J.E.; Kurisawa, M.; Tachibana, Y.; Uyama, H.; Kobayashi, S. Enzymatic synthesis and antioxidant property of poly(allylamine)-catechin conjugate. Chem. Lett. 2003, 32, 620–621. [Google Scholar] [CrossRef]
- Gogoi, P.; Hazarika, S.; Dutta, N.N.; Rao, P.G. Kinetics and mechanism on laccase catalyzed synthesis of poly(allylamine)-catechin conjugate. Chem. Eng. J. 2010, 163, 86–92. [Google Scholar] [CrossRef]
- Gogoi, P.; Hazarika, S.; Dutta, N.N.; Rao, P.G. Laccase catalysed conjugation of catechin with poly(allylamine): Solvent effect. Chem. Eng. J. 2009, 155, 810–815. [Google Scholar] [CrossRef]
- Chung, J.E.; Kurisawa, M.; Uyama, H.; Kobayashi, S. Enzymatic synthesis and antioxidant property of gelatin-catechin conjugates. Biotechnol. Lett. 2003, 25, 1993–1997. [Google Scholar] [CrossRef]
- Gaffar Hossain, K.M.; Díaz González, M.; Monmany, J.M.D.; Tzanov, T. Effects of alkyl chain lengths of gallates upon enzymatic wool functionalisation. J. Mol. Catal. B 2010, 67, 231–235. [Google Scholar] [CrossRef]
- Vachoud, L.; Chen, T.; Payne, G.F.; Vazquez-Duhalt, R. Peroxidase catalyzed grafting of gallate esters onto the polysaccharide chitosan. Enzym. Microb. Technol. 2001, 29, 380–385. [Google Scholar] [CrossRef]
- Cho, Y.S.; Kim, S.K.; Ahn, C.B.; Je, J.Y. Preparation, characterization, and antioxidant properties of gallic acid-grafted-chitosans. Carbohydr. Polym. 2011, 83, 1617–1622. [Google Scholar] [CrossRef]
- Vartiainen, J.; Rättö, M.; Lantto, R.; Nättinen, K.; Hurme, E. Tyrosinase-catalysed grafting of food-grade gallates to chitosan: Surface properties of novel functional coatings. Packag. Technol. Sci. 2008, 21, 317–328. [Google Scholar] [CrossRef]
- Pasanphan, W.; Buettner, G.R.; Chirachanchai, S. Chitosan gallate as a novel potential polysaccharide antioxidant: An EPR study. Carbohydr. Res. 2010, 345, 132–140. [Google Scholar] [CrossRef]
- Aljawish, A.; Chevalot, I.; Piffaut, B.; Rondeau-Mouro, C.; Girardin, M.; Jasniewski, J.; Scher, J.l.; Muniglia, L. Functionalization of chitosan by laccase-catalyzed oxidation of ferulic acid and ethyl ferulate under heterogeneous reaction conditions. Carbohydr. Polym. 2012, 87, 537–544. [Google Scholar]
- Sousa, F.; Guebitz, G.M.; Kokol, V. Antimicrobial and antioxidant properties of chitosan enzymatically functionalized with flavonoids. Proc. Biochem. 2009, 44, 749–756. [Google Scholar] [CrossRef]
- Ihara, N.; Kurisawa, M.; Chung, J.E.; Uyama, H.; Kobayashi, S. Enzymatic synthesis of a catechin conjugate of polyhedral oligomeric silsesquioxane and evaluation of its antioxidant activity. Appl. Microbiol. Biotechnol. 2005, 66, 430–433. [Google Scholar] [CrossRef]
- Kim, S.; Cavaco-Paulo, A. Laccase-catalysed protein-flavonoid conjugates for flax fibre modification. Appl. Microbiol. Biotechnol. 2012, 93, 585–600. [Google Scholar] [CrossRef]
- Silva, C.; Matama, T.; Kim, S.; Padrao, J.; Nugroho Prasetyo, E.; Kudanga, T.; Nyanhongo, G.S.; Guebitz, G.M.; Casal, M.; Cavaco-Paulo, A. Antimicrobial and antioxidant linen via laccase-assisted grafting. React. Funct. Polym. 2011, 71, 713–720. [Google Scholar] [CrossRef]
- Chen, T.; Kumar, G.; Harris, M.T.; Smith, P.J.; Payne, G.F. Enzymatic grafting of hexyloxyphenol onto chitosan to alter surface and rheological properties. Biotechnol. Bioeng. 2000, 70, 564–573. [Google Scholar] [CrossRef]
- Brzonova, I.; Steiner, W.; Zankel, A.; Nyanhongo, G.S.; Guebitz, G.M. Enzymatic synthesis of catechol and hydroxyl-carboxic acid functionalized chitosan microspheres for iron overload therapy. Eur. J. Pharm. Biopharm. 2011, 79, 294–303. [Google Scholar] [CrossRef]
- Matheis, G.; Whitaker, J.R. Peroxidase-catalyzed cross linking of proteins. J. Protein Chem. 1984, 3, 35–48. [Google Scholar] [CrossRef]
- Oudgenoeg, G.; Hilhorst, R.; Piersma, S.R.; Boeriu, C.G.; Gruppen, H.; Hessing, M.; Voragen, A.G.J.; Laane, C. Peroxidase-mediated cross-linking of a tyrosine-containing peptide with ferulic acid. J. Agric. Food Chem. 2001, 49, 2503–2510. [Google Scholar]
- Mattinen, M.L.; Hellman, M.; Permi, P.; Autio, K.; Kalkkinen, N.; Buchert, J. Effect of protein structure on laccase-catalyzed protein oligomerization. J. Agric. Food Chem. 2006, 54, 8883–8890. [Google Scholar]
- Minamihata, K.; Goto, M.; Kamiya, N. Site-specific protein cross-linking by peroxidase-catalyzed activation of a tyrosine-containing peptide tag. Bioconjugate Chem. 2011, 22, 74–81. [Google Scholar] [CrossRef]
- Kuuva, T.; Lantto, R.; Reinikainen, T.; Buchert, J.; Autio, K. Rheological properties of laccase-induced sugar beet pectin gels. Food Hydrocoll. 2003, 17, 679–684. [Google Scholar] [CrossRef]
- Micard, V.; Thibault, J.F. Oxidative gelation of sugar-beet pectins: Use of laccases and hydration properties of the cross-linked pectins. Carbohydr. Polym. 1999, 39, 265–273. [Google Scholar]
- Chen, B.; McClements, D.J.; Gray, D.A.; Decker, E.A. Stabilization of soybean oil bodies by enzyme (laccase) cross-linking of adsorbed beet pectin coatings. J. Agric. Food Chem. 2010, 58, 9259–9265. [Google Scholar]
- Ogushi, Y.; Sakai, S.; Kawakami, K. Synthesis of enzymatically-gellable carboxymethylcellulose for biomedical applications. J. Biosci. Bioeng. 2007, 104, 30–33. [Google Scholar] [CrossRef]
- Sakai, S.; Hashimoto, I.; Ogushi, Y.; Kawakami, K. Peroxidase-catalyzed cell encapsulation in subsieve-size capsules of alginate with phenol moieties in water-immiscible fluid dissolving H2O2. Biomacromolecules 2007, 8, 2622–2626. [Google Scholar] [CrossRef]
- Lee, F.; Chung, J.E.; Kurisawa, M. An injectable enzymatically crosslinked hyaluronic acid-tyramine hydrogel system with independent tuning of mechanical strength and gelation rate. Soft Matter 2008, 4, 880–887. [Google Scholar] [CrossRef]
- Jin, R.; Hiemstra, C.; Zhong, Z.Y.; Feijen, J. Enzyme-mediated fast in situ formation of hydrogels from dextran-tyramine conjugates. Biomaterials 2007, 28, 2791–2800. [Google Scholar] [CrossRef]
- Park, K.M.; Shin, Y.M.; Joung, Y.K.; Shin, H.; Park, K.D. In situ forming hydrogels based on tyramine conjugated 4-Arm-PPO-PEO via enzymatic oxidative reaction. Biomacromolecules 2010, 11, 706–712. [Google Scholar] [CrossRef]
- Sakai, S.; Yamada, Y.; Zenke, T.; Kawakami, K. Novel chitosan derivative soluble at neutral pH and in situ gellable via peroxidase-catalyzed enzymatic reaction. J. Mater. Chem. 2009, 19, 230–235. [Google Scholar]
- Jayakumar, R.; Prabaharan, M.; Reis, R.L.; Mano, J.F. Graft copolymerized chitosan—Present status and applications. Carbohydr. Polym. 2005, 62, 142–158. [Google Scholar] [CrossRef]
- Kumar, G.; Smith, P.J.; Payne, G.F. Enzymatic grafting of a natural product onto chitosan to confer water solubility under basic conditions. Biotechnol. Bioeng. 1999, 63, 154–165. [Google Scholar] [CrossRef]
- Mai, C.; Milstein, O.; Hüttermann, A. Fungal laccase grafts acrylamide onto lignin in presence of peroxides. Appl. Microbiol. Biotechnol. 1999, 51, 527–531. [Google Scholar] [CrossRef]
- Mai, C.; Milstein, O.; Hüttermann, A. Chemoenzymatical grafting of acrylamide onto lignin. J. Biotechnol. 2000, 79, 173–183. [Google Scholar]
- Tsujimoto, T.; Ando, N.; Oyabu, H.; Uyama, H.; Kobayashi, S. Laccase-catalyzed curing of natural phenolic lipids and product properties. J. Macromol. Sci. A 2007, 44, 1055–1060. [Google Scholar] [CrossRef]
- Tsujimoto, T.; Ikeda, R.; Uyama, H.; Kobayashi, S. Crosslinkable polyphenols from urushiol analogues. Macromol. Chem. Phys. 2001, 202, 3420–3425. [Google Scholar] [CrossRef]
- Kobayashi, S.; Uyama, H.; Ikeda, R. Artificial urushi. Chem. Eur. J. 2001, 7, 4755–4760. [Google Scholar]
- Ikeda, R.; Tanaka, H.; Oyabu, H.; Uyama, H.; Kobayashi, S. Preparation of artificial urushi via an environmentally benign process. Bull. Chem. Soc. Japan 2001, 74, 1067–1073. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, Y.H.; Won, K.; Song, B.K. Enzymatic synthesis and curing of polycardol from renewable resources. J. Mol. Catal. B. 2009, 57, 312–316. [Google Scholar] [CrossRef]
- Kim, Y.H.; Won, K.; Kwon, J.M.; Jeong, H.S.; Park, S.Y.; An, E.S.; Song, B.K. Synthesis of polycardanol from a renewable resource using a fungal peroxidase from Coprinus cinereus. J. Mol. Catal. B 2005, 34, 33–38. [Google Scholar] [CrossRef]
- Kim, Y.H.; An, E.S.; Song, B.K.; Kim, D.S.; Chelikani, R. Polymerization of cardanol using soybean peroxidase and its potential application as anti-biofilm coating material. Biotechnol. Lett. 2003, 25, 1521–1524. [Google Scholar] [CrossRef]
- Reihmann, M.H.; Ritter, H. Enzymatically catalyzed synthesis of photocrosslinkable oligophenols. Macromol. Chem. Phys. 2000, 201, 1593–1597. [Google Scholar] [CrossRef]
- Bilici, A.; Kaya, I.; Yildirim, M. Peroxidase-catalyzed synthesis of polyphenols bearing aldehyde units. Des. Monomers Polym. 2011, 14, 353–366. [Google Scholar] [CrossRef]
- Majeau, J.A.; Brar, S.K.; Tyagi, R.D. Laccases for removal of recalcitrant and emerging pollutants. Biores. Technol. 2010, 101, 2331–2350. [Google Scholar]
- Husain, Q. Peroxidase mediated decolorization and remediation of wastewater containing industrial dyes: A review. Rev. Environ. Sci. Bio Technol. 2010, 9, 117–140. [Google Scholar] [CrossRef]
- Minussi, R.C.; Pastore, G.M.; Duran, N. Potential applications of laccase in the food industry. Trends Food Sci. Technol. 2002, 13, 205–216. [Google Scholar] [CrossRef]
- Lopez, C.; Moreira, M.T.; Feijoo, G.; Lema, J.M. Economic comparison of enzymatic reactors and advanced oxidation processes applied to the degradation of phenol as a model compound. Biocatal. Biotransf. 2011, 29, 344–353. [Google Scholar]
- Kadhim, H.; Graham, C.; Barratt, P.; Evans, C.S.; Rastall, R.A. Removal of phenolic compounds in water using Coriolus versicolor grown on wheat bran. Enz. Microb. Technol. 1999, 24, 303–307. [Google Scholar] [CrossRef]
- Uhnakova, B.; Ludwig, R.; Peknicova, J.; Homolka, L.; Lisa, L.; Sulc, M.; Petrickova, A.; Elzeinova, F.; Pelantova, H.; Monti, D.; et al. Biodegradation of tetrabromobisphenol A by oxidases in basidiomycetous fungi and estrogenic activity of the biotransformation products. Bioresour. Technol. 2011, 102, 9409–9415. [Google Scholar]
- Gaitan, I.J.; Medina, S.C.; Gonzalez, J.C.; Rodriguez, A.; Espejo, A.J.; Osma, J.F.; Sarria, V.; Almeciga-Diaz, C.J.; Sanchez, O.F. Evaluation of toxicity and degradation of a chlorophenol mixture by the laccase produced by Trametes pubescens. Bioresour. Technol. 2011, 102, 3632–3635. [Google Scholar]
- Lisov, A.V.; Pozhidaeva, Z.A.; Stepanova, E.V.; Koroleva, O.V.; Leontievsky, A.A. Conversion of polychloronhenols by laccases with 1-hydroxybenzotriazole as a mediator. Appl. Biochem. Microbiol. 2007, 43, 616–619. [Google Scholar] [CrossRef]
- Leontievsky, A.A.; Myasoedova, N.M.; Baskunov, B.P.; Evans, C.S.; Golovleva, L.A. Transformation of 2,4,6-trichlorophenol by the white rot fungi Panus tigrinus and Coriolus versicolor. Biodegradation 2000, 11, 331–340. [Google Scholar] [CrossRef]
- Schultz, A.; Jonas, U.; Hammer, E.; Schauer, F. Dehalogenation of chlorinated hydroxybiphenyls by fungal laccase. Appl. Environ. Microbiol. 2001, 67, 4377–4381. [Google Scholar] [CrossRef]
- Watanabe, C.; Kashiwada, A.; Matsuda, K.; Yamada, K. Soybean peroxidase-catalyzed treatment and removal of BPA and bisphenol derivatives from aqueous solutions. Environ. Prog. Sustain. Energy 2011, 30, 81–91. [Google Scholar] [CrossRef]
- Fukuda, T.; Uchida, H.; Suzuki, M.; Miyamoto, H.; Morinaga, H.; Nawata, H.; Uwajima, T. Transformation products of bisphenol A by a recombinant Trametes vilosa laccase and their estrogenic activity. J. Chem. Technol. Biotechnol. 2004, 79, 1212–1218. [Google Scholar] [CrossRef]
- Tsutsumi, Y.; Haneda, T.; Nishida, T. Removal of estrogenic activities of bisphenol A and nonylphenol by oxidative enzymes from lignin-degrading basidiomycetes. Chemosphere 2001, 42, 271–276. [Google Scholar] [CrossRef]
- Lloret, L.; Hollmann, F.; Eibes, G.; Feijoo, G.; Moreira, M.; Lema, J. Immobilisation of laccase on Eupergit supports and its application for the removal of endocrine disrupting chemicals in a packed-bed reactor. Biodegradation 2012, 1–14. [Google Scholar]
- Lloret, L.; Eibes, G.; Feijoo, G.; Moreira, M.T.; Lema, J.M.; Hollmann, F. Immobilization of laccase by encapsulation in a sol-gel matrix and its characterization and use for the removal of estrogens. Biotechnol. Prog. 2011, 27, 1570–1579. [Google Scholar] [CrossRef]
- Lloret, L.; Eibes, G.; Lu-Chau, T.A.; Moreira, M.T.; Feijoo, G.; Lema, J.M. Laccase-catalyzed degradation of anti-inflammatories and estrogens. Biochem. Eng. J. 2010, 51, 124–131. [Google Scholar] [CrossRef]
- Wang, C.J.; Thiele, S.; Bollag, J.M. Interaction of 2,4,6-trinitrotoluene (TNT) and 4-amino-2,6-dinitrotoluene with humic monomers in the presence of oxidative enzymes. Arch. Environ. Contam. Toxicol. 2002, 42, 1–8. [Google Scholar] [CrossRef]
- Thiele, S.; Fernandes, E.; Bollag, J.M. Enzymatic transformation and binding of labeled 2,4,6-trinitrotoluene to humic substances during an anaerobic/aerobic incubation. J. Environ. Qual. 2002, 31, 437–444. [Google Scholar] [CrossRef]
- Collins, P.J.; Kotterman, M.J.J.; Field, J.A.; Dobson, A.D.W. Oxidation of anthracene and benzo[a]pyrene by laccases from Trametes versicolor. Appl. Environ. Microbiol. 1996, 62, 4563–4567. [Google Scholar]
- Zeng, J.; Lin, X.G.; Zhang, J.; Li, X.Z.; Wong, M.H. Oxidation of polycyclic aromatic hydrocarbons by the bacterial laccase CueO from E. coli. Appl. Microbiol. Biotechnol. 2011, 89, 1841–1849. [Google Scholar] [CrossRef]
- Wang, J.Q.; Ogata, M.; Hirai, H.; Kawagishi, H. Detoxification of aflatoxin B(1) by manganese peroxidase from the white-rot fungus Phanerochaete sordida YK-624. FEMS Microbiol. Lett. 2011, 314, 164–169. [Google Scholar] [CrossRef]
- Eibes, G.; Cajthaml, T.; Moreira, M.T.; Feijoo, G.; Lema, J.M. Enzymatic degradation of anthracene, dibenzothiophene and pyrene by manganese peroxidase in media containing acetone. Chemosphere 2006, 64, 408–414. [Google Scholar] [CrossRef]
- Eibes, G.; Lu-Chau, T.; Feijoo, G.; Moreira, M.T.; Lema, J.M. Complete degradation of anthracene by Manganese Peroxidase in organic solvent mixtures. Enzym. Microb. Technol. 2005, 37, 365–372. [Google Scholar] [CrossRef]
- Mielgo, I.; Lopez, C.; Moreira, M.T.; Feijoo, G.; Lema, J.M. Oxidative degradation of azo dyes by manganese peroxidase under optimized conditions. Biotechnol. Prog. 2003, 19, 325–331. [Google Scholar] [CrossRef]
- Minussi, R.C.; Miranda, M.A.; Silva, J.A.; Ferreira, C.V.; Aoyama, H.; Marangoni, S.; Rotilio, D.; Pastore, G.M.; Duran, N. Purification, characterization and application of laccase from Trametes versicolor for colour and phenolic removal of olive mill wastewater in the presence of 1-hydroxybenzotriazole. Afr. J. Biotechnol. 2007, 6, 1248–1254. [Google Scholar]
- Suda, T.; Hata, T.; Kawai, S.; Okamura, H.; Nishida, T. Treatment of tetracycline antibiotics by laccase in the presence of 1-hydroxybenzotriazole. Bioresour. Technol. 2012, 103, 498–501. [Google Scholar]
- Gross, R.A.; Kumar, A.; Kalra, B. Polymer synthesis by in vitro enzyme catalysis. Chem. Rev. 2001, 101, 2097–2124. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hollmann, F.; Arends, I.W.C.E. Enzyme Initiated Radical Polymerizations. Polymers 2012, 4, 759-793. https://doi.org/10.3390/polym4010759
Hollmann F, Arends IWCE. Enzyme Initiated Radical Polymerizations. Polymers. 2012; 4(1):759-793. https://doi.org/10.3390/polym4010759
Chicago/Turabian StyleHollmann, Frank, and Isabel W. C. E. Arends. 2012. "Enzyme Initiated Radical Polymerizations" Polymers 4, no. 1: 759-793. https://doi.org/10.3390/polym4010759
APA StyleHollmann, F., & Arends, I. W. C. E. (2012). Enzyme Initiated Radical Polymerizations. Polymers, 4(1), 759-793. https://doi.org/10.3390/polym4010759