Evaluation of the Biological Effects of Externally Tunable, Hydrogel Encapsulated Quantum Dot Nanospheres in Escherichia coli
Abstract
:1. Introduction
2. Experimental Section
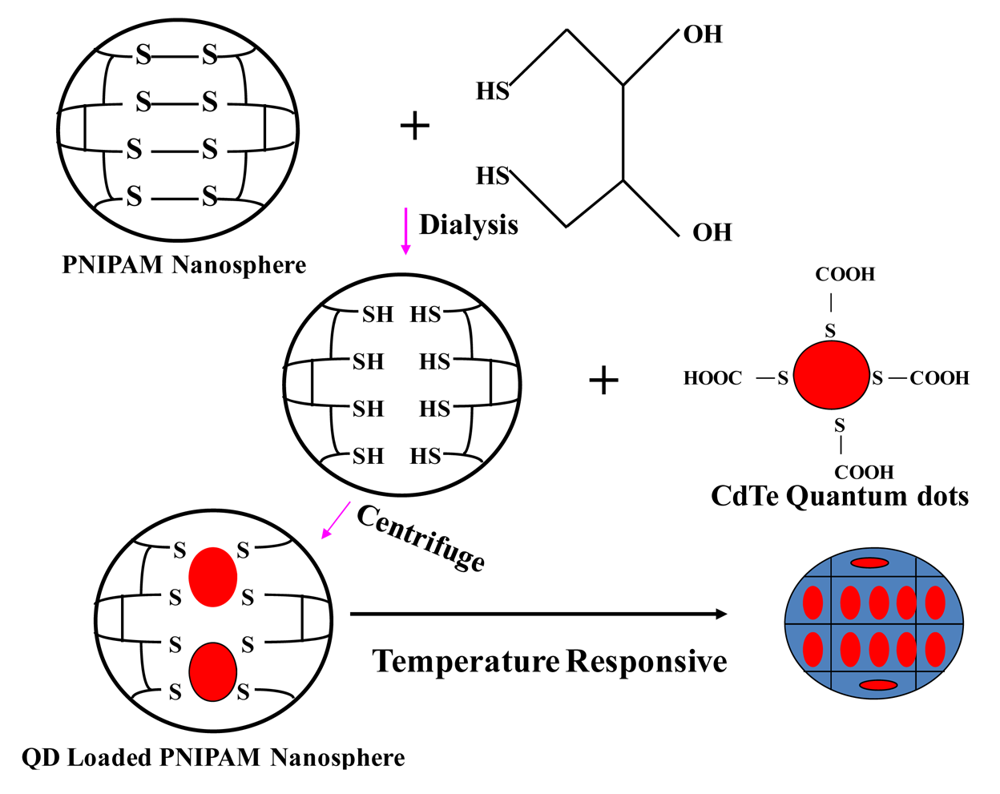
2.1. Synthesis of CdTe-PNIPAM Nanospheres
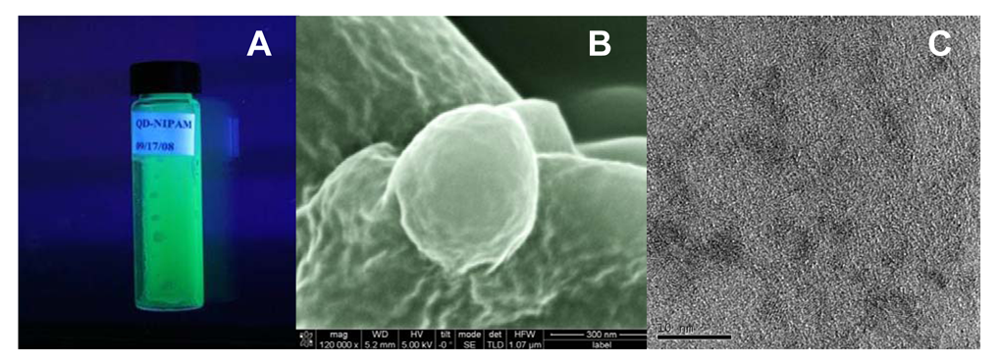
2.2. Scanning and Transmission Electron Microscopy (SEM and TEM)
2.3. Bacterial Cell Culture
2.4. Transformation of E. coli with pGLO
3. Results and Discussion
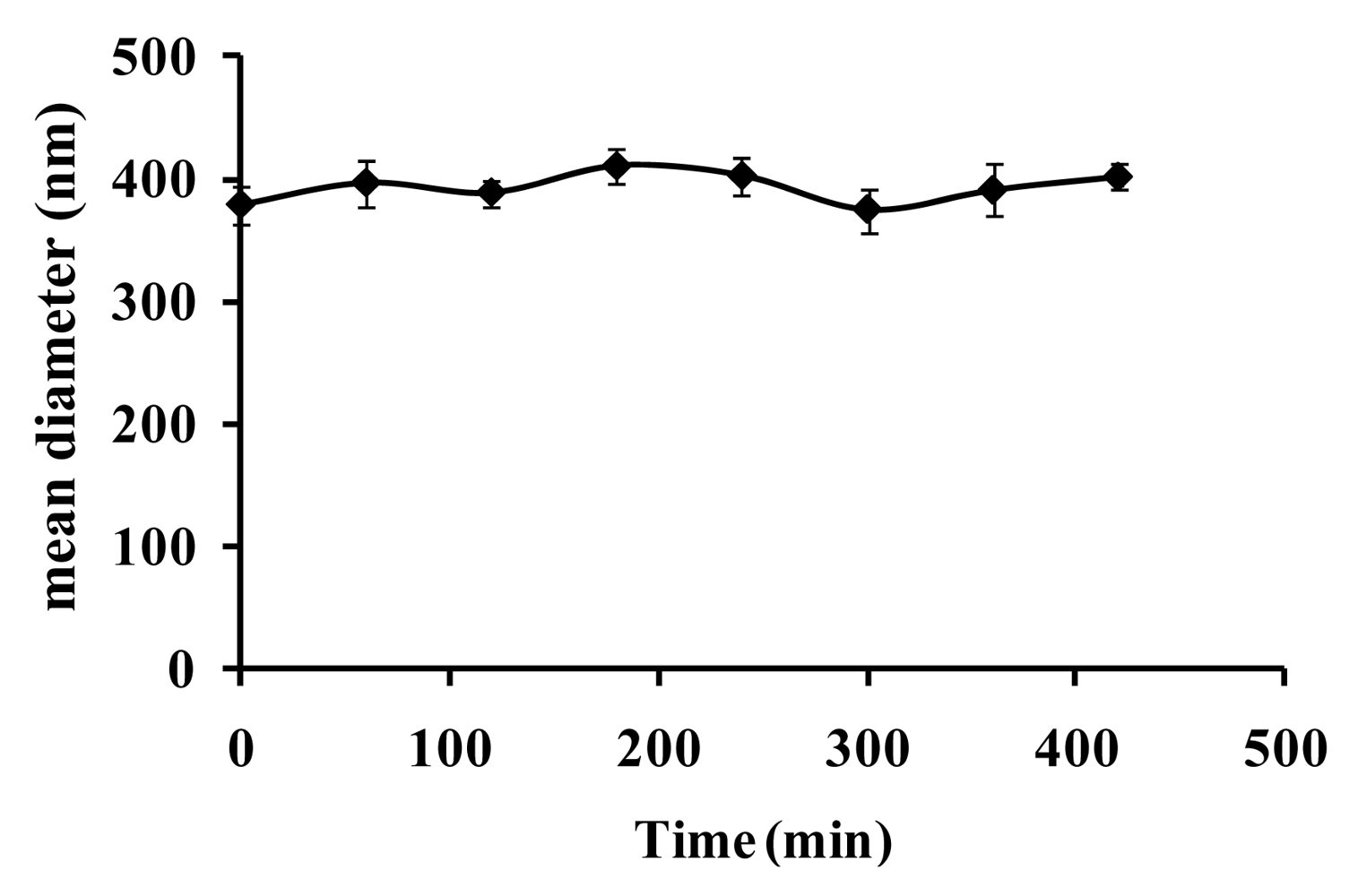
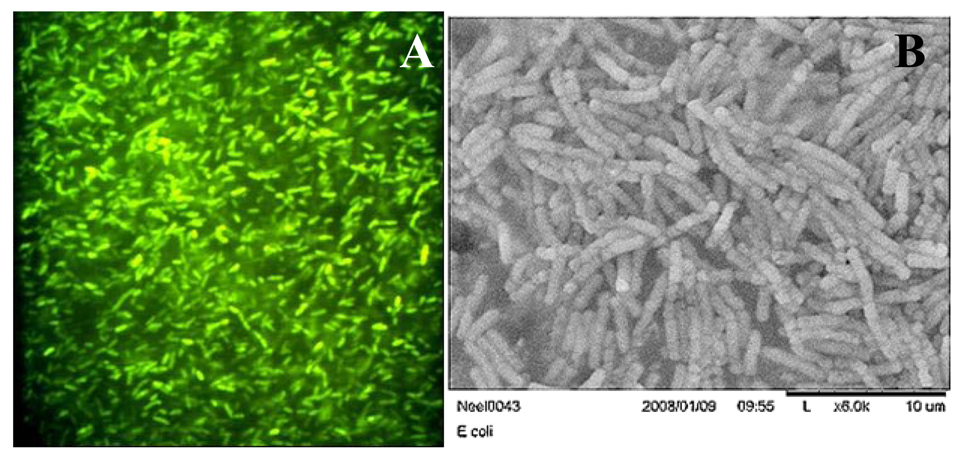
3.1. Physicochemical characterization of CdTe-PNIPAM Nanospheres
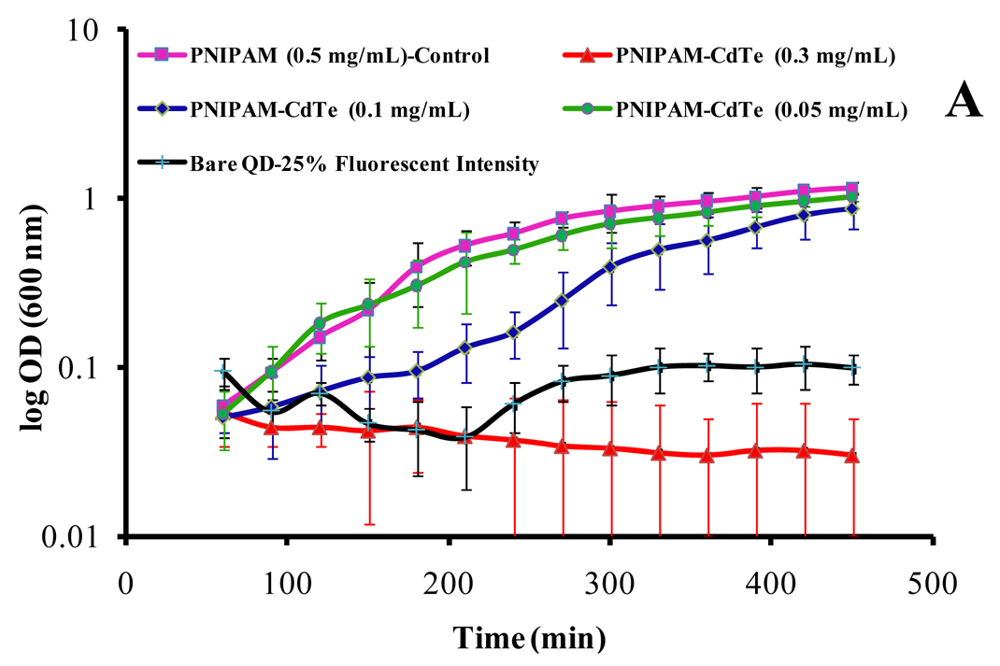
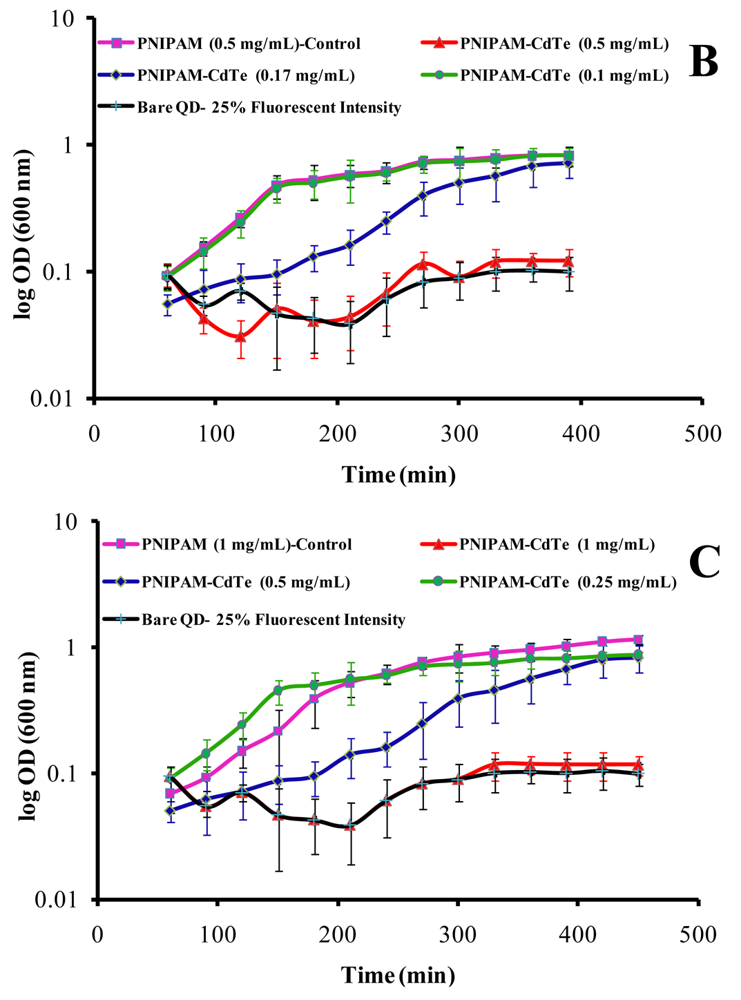
3.2. Microbial Growth Experiments and GFP Expression
4. Conclusions
Acknowledgments
References
- Alivisatos, P. The use of nanocrystals in biological detection. Nat. Biotechnol. 2004, 22, 47–52. [Google Scholar]
- Niemeyer, C.M. Nanoparticles, proteins, and nucleic acids: Biotechnology meets materials science. Angew. Chem. Int. Ed. 2001, 40, 4128–4158. [Google Scholar]
- Bagalkot, V.; Zhang, L.; Levy-Nissenbaum, E.; Jon, S.; Kantoff, P.W.; Langer, R.; Farokhzad, O.C. Quantum dot-aptamer conjugates for synchronous cancer imaging, therapy, and sensing of drug delivery based on bi-fluorescence resonance energy transfer. Nano Let. 2007, 7, 3065–3070. [Google Scholar]
- Bakalova, R.; Ohba, H.; Zhelev, Z.; Ishikawa, M.; Baba, Y. Quantum dots as photosensitizers? Nat. a-z Index 2004, 22, 1360–1361. [Google Scholar]
- Ballou, B.; Ernst, L.A.; Andreko, S.; Harper, T.; Fitzpatrick, J.A.J.; Waggoner, A.S.; Bruchez, M.P. Sentinel lymph node imaging using quantum dots in mouse tumor models. Bioconjugate Chem. 2007, 18, 389–396. [Google Scholar]
- Bruchez, M., Jr.; Moronne, M.; Gin, P.; Weiss, S.; Alivisatos, A.P. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016. [Google Scholar]
- GhoshMitra, S.; Diercks, D.R.; Mills, N.C.; Hynds, D.A.L.; Ghosh, S. Excellent biocompatibility of semiconductor quantum dots encased in multifunctional poly(N-isopropylacrylamide) nanoreservoirs and nuclear specific labeling of growing neurons. Appl. Phys. Lett. 2011, 98, 103702. [Google Scholar]
- Smith, A.M.; Ruan, G.; Rhyner, M.N.; Nie, S. Engineering luminescent quantum dots for in vivo molecular and cellular imaging. Ann. Biomed. Eng. 2006, 34, 3–14. [Google Scholar]
- Hu, Z.; Chen, Y.; Wang, C.; Zheng, Y.; Li, Y. Polymer gels with engineered environmentally responsive surface patterns. Nature 1998, 393, 149–152. [Google Scholar]
- Ghosh, S.; GhoshMitra, S.; Cai, T.; Diercks, D.R.; Mills, N.C.; Hynds, D.A.L. Alternating magnetic field controlled, multifunctional nano-reservoirs: Intracellular uptake and improved biocompatibility. Nanoscale Res. Lett. 2010, 5, 195–204. [Google Scholar]
- Garner, B.W.; Cai, T.; Ghosh, S.; Hu, Z.; Neogi, A. Refractive index change due to volume-phase transition in polyacrylamide gel nanospheres for optoelectronics and bio-photonics. Appl. Phys. Express 2009, 2, 057001. [Google Scholar]
- Agrawal, M.; Rubio-Retama, J.; Zafeiropoulos, N.; Gaponik, N.; Gupta, S.; Cimrova, V.; Lesnyak, V.; Lopez-Cabarcos, E.; Tzavalas, S.; Rojas-Reyna, R. Switchable photoluminescence of CdTe nanocrystals by temperature-responsive microgels. Langmuir 2008, 24, 9820–9824. [Google Scholar]
- Ghosh, S.; Yang, C.; Cai, T.; Hu, Z.; Neogi, A. Oscillating magnetic field-actuated microvalves for micro- and nanofluidics. J. Phys. D 2009, 42, 135501. [Google Scholar]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 2004, 4, 11–18. [Google Scholar]
- Zhang, T.; Stilwell, J.L.; Gerion, D.; Ding, L.; Elboudwarej, O.; Cooke, P.A.; Gray, J.W.; Alivisatos, A.P.; Chen, F.F. Cellular effect of high doses of silica-coated quantum dot profiled with high throughput gene expression analysis and high content cellomics measurements. Nano Lett. 2006, 6, 800–808. [Google Scholar]
- Cho, S.J.; Maysinger, D.; Jain, M.; Röder, B.; Hackbarth, S.; Winnik, F.M. Long-term exposure to CdTe quantum dots causes functional impairments in live cells. Langmuir 2007, 23, 1974–1980. [Google Scholar]
- Hardman, R. A toxicologic review of quantum dots: Toxicity depends on physicochemical and environmental factors. Environ. Health Perspect. 2006, 114, 165–172. [Google Scholar]
- Hoshino, A.; Manabe, N.; Fujioka, K.; Suzuki, K.; Yasuhara, M.; Yamamoto, K. Use of fluorescent quantum dot bioconjugates for cellular imaging of immune cells, cell organelle labeling, and nanomedicine: Surface modification regulates biological function, including cytotoxicity. J. Artif. Organs 2007, 10, 149–157. [Google Scholar]
- Kim, J.; Park, Y.; Yoon, T.H.; Yoon, C.S.; Choi, K. Phototoxicity of CdSe/ZnSe quantum dots with surface coatings of 3-mercaptopropionic acid or tri-n-octylphosphine oxide/gum arabic in Daphnia magna under environmentally relevant UV-B light. Aquat. Toxicol. 2010, 97, 116–124. [Google Scholar]
- Lu, Z.; Li, C.M.; Bao, H.; Qiao, Y.; Toh, Y.; Yang, X. Mechanism of antimicrobial activity of CdTe quantum dots. Langmuir 2008, 24, 5445–5452. [Google Scholar]
- Su, Y.; He, Y.; Lu, H.; Sai, L.; Li, Q.; Li, W.; Wang, L.; Shen, P.; Huang, Q.; Fan, C. The cytotoxicity of cadmium based, aqueous phase-synthesized, quantum dots and its modulation by surface coating. Biomaterials 2009, 30, 19–25. [Google Scholar]
- Su, Y.; Hu, M.; Fan, C.; He, Y.; Li, Q.; Li, W.; Wang, L.; Shen, P.; Huang, Q. The cytotoxicity of CdTe quantum dots and the relative contributions from released cadmium ions and nanoparticle properties. Biomaterials 2010, 31, 4829–4834. [Google Scholar]
- Hezinger, A.; Teβmar, J.; Göpferich, A. Polymer coating of quantum dots—A powerful tool toward diagnostics and sensorics. Eur. J. Pharm. Biopharm. 2008, 68, 138–152. [Google Scholar]
- Williams, D.; Ehrman, S.; Pulliam Holoman, T. Evaluation of the microbial growth response to the inorganic nanoparticles. J. Nanobiotechnol. 2006. [Google Scholar] [CrossRef]
- Jiang, G.; Shen, Z.; Niu, J.; Bao, Y.; Chen, J.; He, T. Toxicological assessment of TiO2 nanoparticles by recombinant Escherichia coli bacteria. J. Environ. Monit. 2011, 13, 42–48. [Google Scholar]
- Neogi, A.; Ghosh, S.; Li, J.; Cai, T.; Hu, Z. Enhanced luminescence efficiency from hydrogel microbead encapsulated quantum dots. Mater. Res. Soc. Symp. Proc. 2007, 959, 202–207. [Google Scholar]
- Häfeli, U.; Riffle, J.; Harris-Shekhawat, L.; Carmichael-Baranauskas, A.; Mark, F.; Dailey, J.; Bardenstein, D. Cell uptake and in vitro toxicity of magnetic nanoparticles suitable for drug delivery. Mol. Pharm. 2009, 6, 1417–1428. [Google Scholar]
- Frimpong, R.; Hilt, J. Poly(n-isopropylacrylamide)-based hydrogel coatings on magnetite nanoparticles via atom transfer radical polymerization. Nanotechnology 2008, 19, 175101. [Google Scholar]
- Helbig, K.; Grosse, C.; Nies, D. Cadmium toxicity in glutathione nutants of Escherichia coli. J. Bacteriol. 2008, 190, 5439–5454. [Google Scholar]
- Lovrić, J.; Bazzi, H.; Cuie, Y.; Fortin, G.; Winnik, F.; Maysinger, D. Differences in subcellular distribution and toxicity of green and red emitting CdTe quantum dots. J. Mol. Med. 2005, 83, 377–385. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
GhoshMitra, S.; Cai, T.; Diercks, D.; Hu, Z.; Roberts, J.; Dahiya, J.; Mills, N.; Hynds, D.; Ghosh, S. Evaluation of the Biological Effects of Externally Tunable, Hydrogel Encapsulated Quantum Dot Nanospheres in Escherichia coli. Polymers 2011, 3, 1243-1254. https://doi.org/10.3390/polym3031243
GhoshMitra S, Cai T, Diercks D, Hu Z, Roberts J, Dahiya J, Mills N, Hynds D, Ghosh S. Evaluation of the Biological Effects of Externally Tunable, Hydrogel Encapsulated Quantum Dot Nanospheres in Escherichia coli. Polymers. 2011; 3(3):1243-1254. https://doi.org/10.3390/polym3031243
Chicago/Turabian StyleGhoshMitra, Somesree, Tong Cai, David Diercks, Zhibing Hu, James Roberts, Jai Dahiya, Nathaniel Mills, DiAnna Hynds, and Santaneel Ghosh. 2011. "Evaluation of the Biological Effects of Externally Tunable, Hydrogel Encapsulated Quantum Dot Nanospheres in Escherichia coli" Polymers 3, no. 3: 1243-1254. https://doi.org/10.3390/polym3031243
APA StyleGhoshMitra, S., Cai, T., Diercks, D., Hu, Z., Roberts, J., Dahiya, J., Mills, N., Hynds, D., & Ghosh, S. (2011). Evaluation of the Biological Effects of Externally Tunable, Hydrogel Encapsulated Quantum Dot Nanospheres in Escherichia coli. Polymers, 3(3), 1243-1254. https://doi.org/10.3390/polym3031243




