Multimeric, Multifunctional Derivatives of Poly(ethylene glycol)
Abstract
: This article reviews the use of multifunctional polymers founded on high-molecular weight poly(ethylene glycol) (PEG). The design of new PEG derivatives assembled in a dendrimer-like multimeric fashion or bearing different functionalities on the same molecule is described. Their use as new drug delivery systems based on the conjugation of multiple copies or diversely active drugs on the same biocompatible support is illustrated.1. Introduction
For the application of biopharmaceuticals in human therapy, the covalent coupling of poly(ethylene glycol) (PEG) chains to drugs, or PEGylation, has been an outstanding innovation. Important pioneering work in this field was performed by Davis and Abuchowski, laying the cornerstone for the commercial success of this technology [1]. Even though many attempts have been undertaken to develop new polymers with improved properties, none of these new substances have been able to compete with poly(ethylene glycol) for this application. This can be explained by the biocompatibility of PEG and the good experience with PEG as a low-cost additive for the pharmaceutical and cosmetic industry over the last decades. An ideal PEG reagent fulfills at least the following criteria: (a) a polydispersity index close to 1.00, in order to assure a reproducible high quality; (b) availability of terminal reactive groups for the coupling reaction; (c) non-toxic and non-immunogenic, biochemically stable linker; (d) options for site-specific coupling.
Many drugs require further modifications to decrease adverse side effects, limit nonspecific activity, increase circulation time, control release time-profile, modify biodistribution, etc. In most cases, such modifications involve the so-called ‘prodrug approach’. A prodrug is a biologically inactive derivative of a parent drug molecule that usually requires an enzymatic transformation within the body in order to release the active drug, and has improved delivery properties over the parent molecule [2]. On this basis, the conjugation of PEG to many bioactive molecules is an upcoming methodology for drug development and allows for a substantial improvement of their pharmacokinetic and pharmacodynamic properties. Protein, peptides and small bioactive molecules hold great promise as therapeutic agents but they also possess several shortcomings that limit their usefulness. These disadvantages include their susceptibility to destruction by proteolytic enzymes, short circulating half-life, low solubility, rapid kidney clearance and their propensity to generate neutralizing antibodies [3]. Hence, PEGylation is an alternative method that overcomes these deficiencies for both proteins and low molecular weight agents. Some advantages are (i) increased water solubility (important for very low soluble molecules like, for example, many anticancer drugs such as taxol or camptothecin); (ii) enhanced bioavailability and prolonged plasma half-life due to the increased hydrodynamic volume that reduces the kidney clearance (kidneys filter substances on the basis of size, thus larger molecules clear more slowly); (iii) protection towards degrading enzymes; (iv) prevention or reduction of aggregation, immunogenicity and antigenicity; and (v) specific accumulation in organs, tissues or cells, by actively targeted polymers or exploiting the known ‘enhanced permeability and retention (EPR) effect’ [4]. The U.S. Food and Drug Administration has approved PEG for use as a vehicle or additive or excipients in foods, cosmetics and pharmaceuticals. It shows little toxicity, and is eliminated from the body intact by either the kidneys (for PEGs < 30 kDa) or in the faeces (for PEGs < 20 kDa) [5,6].
PEGylation processes may be classified into two categories: ‘first-generation’ and ‘second-generation’ processes. First-generation processes are associated with different pitfalls such as the presence of mixtures of isomers, diol contamination, unstable bonds, and changes in the bioactivity of some biomolecules. Nevertheless, despite these limitations, some first-generation PEGylated drugs are still in use today, for example, pegademase (Adagen®), a PEGylated form of the enzyme adenosine deaminase for the treatment of severe combined immunodeficiency disease. Instead, the second-generation PEGylation led to the (i) synthesis of larger PEG polymers in order to improve the pharmacokinetic and pharmacodynamic effects achieved by the conjugates prepared with lower molecular mass PEGs; (ii) synthesis and use of heterobifunctional PEGs containing dissimilar terminal groups, which are useful for applications in immunoassays, biosensors and probes to link macromolecules to surfaces, as well as for the targeting of drugs, liposomes or viruses to specific tissues [7]; (iii) the preparation of branched PEGs of greatly increased molecular masses (>40 KDa) which shields the protein surface better than a linear PEG of the same size and are more effective in protecting the conjugated protein from proteolytic enzymes and antibodies [8].
PEG, a polyether backbone polymer, is synthesized by ring opening polymerization of ethylene oxide using in common cases or usually methanol or water as an initiator. The reaction gives products with one or two end chain hydroxyl groups termed monomethoxy-PEG (mPEG-OH) or diol-PEG (HO-PEG-OH), respectively. Usually, the polymerization process produces a family of PEG molecules with wide Gaussian's distribution of molecular weights. In this form, PEG is not suitable for use in drug conjugation, although it is largely employed in pharmaceutical technology as excipient. The lone hydroxyl group in the case of the methoxy form, or the two in the case of PEG diol, can be modified to be reactive towards different chemical groups by several activation strategies. Nowadays, a number of activated PEGs are commercially available. These derivatives are commonly monofunctional, either in their linear or branched structure. Thus, in contrast with other water soluble biocompatible polymers, such as N-(2-hydroxypropyl)-methacryl amide (HPMA) copolymer [9] and polyglutamic acid (PGA) [10], the functionality of PEG is limited to its two chain termini regardless of the molecular weight. Usually, an increased loading value is needed in therapeutic agents with low biological activity, which would otherwise require the administration of a high dose of conjugate with consequent high viscosity of the solution. To this purpose, PEG diols, PEG dendrons [11,12], or dendrimers [13,14], have been proposed for small drug delivery thanks to the possibility of reaching higher drug to polymer ratios and further approaches have been made to synthesize new multifunctional PEGs [15-18]. Additionally, PEG-dendrimer hybrids where constructed where the PEG components are attached to the apex [19] or to the peripheral groups [20] of dendrimers. More recently, various core functionalities have been introduced on those biodegradable PEGylated dendrimers [21].
2. Multimeric PEG-Based Supports
As previously mentioned, commercial PEG derivatives show good physico-chemical properties, terminal reactive functional groups, as well as advantageous solubilising properties. However, a reduced size homogeneity and loading capacity could hamper their pharmacological application, and these adverse characteristics increase with higher molecular weights. With a view to improving their overall features, many modifications of linear bifunctional PEGs have been proposed [22-24], but their synthesis can be complex and time-consuming and they are scarcely hydrolyzed under physiological conditions [25,26]. Additionally, there are some difficulties in preparing multimeric derivatives using high molecular weight PEG owing to the reduced reactivity of the starting units [27]. Moreover, analogous high-capacity soluble polymeric supports, consisting of aliphatic dendritic polyethers and of hyperbranched polyethers-polyols, require laborious, multi-step preparation [28-31]. A different approach has been recently proposed where a simple “click” copolymerization procedure provides linear PEGs bearing multiple pendant hydroxyl groups [32] along the final chain. Some further example of linear activated PEGs, multi-armed activated PEG derivatives and branched activated PEGs in a wide variety of molecular weights are now commercially available. In Figure 1, the structural schemes of some of those derivatives are described.
Recently, Bonora and coll. proposed a synthetic strategy aimed to the production of new branched, high-molecular weight multimeric PEG-based systems (MultiPEGs) starting from inexpensive commercial PEG moieties assembled through a divergent dendrimeric fashion. These new derivatives were obtained from smaller commercial bifunctional PEGs (M.W. = 2,000, 3,000 and 6,000 Da) which were selectively and temporarily protected at only one of their two starting reactive ends. Activation of the residual hydroxylic groups unable the assembling of PEG units with appropriate linkers through successive condensation reactions following different synthetic routes [33]. The resulting multifunctional poly(ethylene glycol) derivatives (MultiPEGs), were purified by molecular exclusion chromatography or extensive dialysis. The final polymeric derivatives, ranging from 10,000 to 24,000 Da, are characterized by a higher number of terminal reacting groups (up to 6 times, as in the PentaPEG(2000)-(OH)12) than that found in linear, commercial PEGs of the same molecular dimensions. The number of synthetic steps compared with the nature of the final product (in terms of dimension and loading value) and the purification passages played a critical role in the final yield of these processes.
The reported synthetic processes of these multifunctional polymers have been performed on a gram scale, but a scaling up could be reasonably afforded for commercial purposes. When used as drug carrier, these MultiPEGs will allow the administration of a correspondingly lower amount of polymer, to achieve the same level of drug concentration within the body, in comparison with commercial bifunctional PEGs of the same size. Moreover, the introduction of an urethane bond within the final polyether backbone will likely allow for more advantageous biodegradability properties in comparison with other similar branched derivatives. In fact, some hydrolysis at the level of the urethane bond [34] is expected to take place in physiological media. Also, it should be emphasized that the use of low-dispersed polymers as starting building units will also guarantee the low level of polydispersity for the larger, final MultiPEG. Further development of this synthetic strategy allows the preparation of bifunctional derivatives containing reactive groups with different chemical properties as the (OH)8TetraPEG(6000)-NH2, that permit the possible conjugation of two unlike molecules [35]. The schematic structures of some relevant MultiPEGs are described in Figure 2.
3. Multifunctional PEG Derivatives with High Drug Loading
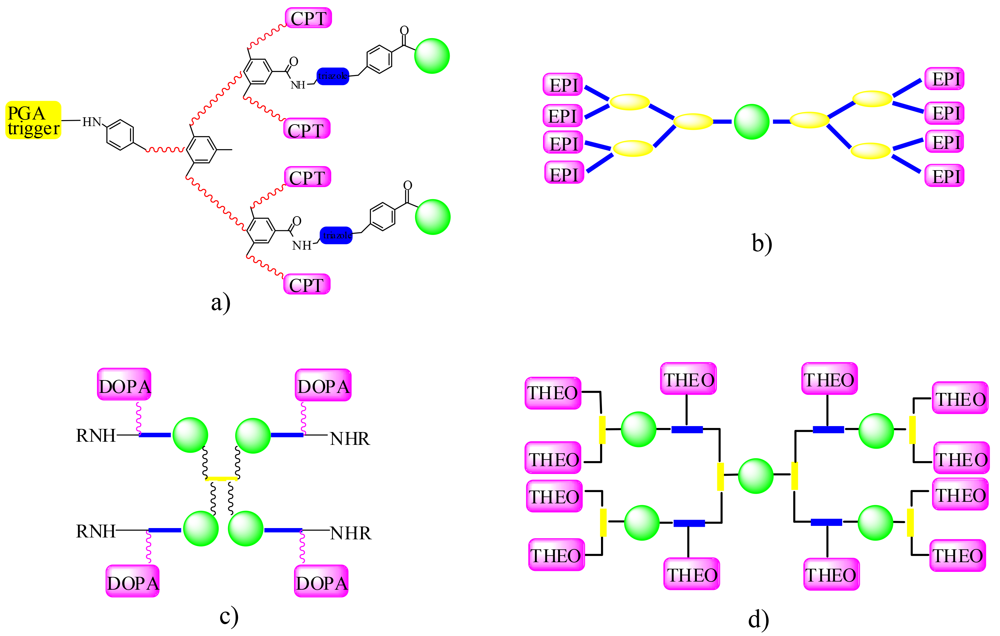
As mentioned, since PEG has a maximum of two active sites, it is only possible to conjugate two drug molecules on a single polymeric support, limiting the loading capacity of such a carrier. Then, in order to increase the load of the drug delivery system, branched polymers and high-molecular weight multimeric PEG-based systems (MultiPEGs) can have been realised. An increased number of functional groups, compared to linear PEGs of the same molecular size, and chemical bonds of different stability within the main polymeric backbone represent the main features of these derivatives. Their recent application as drug delivery of some pharmacologically active molecules is described in the following. In Figure 3 some schematic structure of the examples reported here is illustrated.
In the field of anticancer drug therapy, Camptosar® [CPT-11, Irinotecan] is a prodrug that is approved for the treatment of advanced colorectal cancer. The compound selectively inhibits topoisomerase I by trapping the enzyme during the cleavage of DNA, thereby prohibiting faithful cell division and protein production. The active metabolite of CPT-11, SN38 (7-ethyl-10-hydroxy-camptothecin), has 100- to 1,000-fold more potent cytotoxic activity in tissue cell culture compared to CPT-11 [36]. Besides, it has poor solubility in any pharmaceutically acceptable excipient and cannot be used for systemic applications [37]. Using multiarm PEG linkers, Zhao [38] developed several novel PEG-SN38 conjugates that have high drug loading and high water solubility. Starting from the four-arm-PEG, in order to increase the loading of SN38, four PEG-SN38 conjugates with different aminoacid spacers were obtained. The chemistry for the conjugation of SN38 ensures that the anticancer molecule is locked into its active closed lactone form in the body until intact SN38 is released from the PEG conjugate. In addition, Camptothecin (CPT) has been also covalently linked to dendrimers synthesized by the copper-catalyzed azide-alkyne cycloaddition (CuAAC) [39], where a 5 kDa PEG molecule has been incorporated into the G2 dendrimer, a self-immolative dendrimer synthesized characterized by degradation upon exposure to the enzyme penicillin G-amidase (PGA) to yield two triazole-PEG molecules and four molecules of the anticancer drug camptothecin.
Epirubicin (EPI) is an anthracycline drug, favoured over doxorubicin in some chemotherapy regimens as it appears to cause fewer side-effects such as cardiotoxicity. Pasut [12] synthesized PEG-dendrimers with high loading capacity using amino adipic acid or β-glutamic acid as branching molecules, built on a PEG diol of Mw 10,000 Da. The use of specific amino bicarboxylic acids (amino adipic acid or β-glutamic acid) facilitated the linking of the hindered molecule epirubicin to the multibranched PEG. Epirubicin binding was achieved through EDC/HOBT activation of the PEG-dendrimer carboxylic group in anhydrous DMF leading to a series of derivatives. The dendrimers obtained with β-glutamic acid possess COOH groups at the same distance from the core while dendrimers from amino adipic acid present the COOH groups at different levels from the core, due to the different lengths between the two arms of the bicarboxylic acid. The synthesized conjugates showed better stability than free epirubicin in different pH buffers and in plasma or the preliminary pharmacokinetic studies conducted in mice demonstrated increased blood residence time compared to free epirubicin. This is due to the reduced kidney clearance as a consequence of the increased size.
An additional example of multifunctional PEGylating derivatives was given by the synthesis of PEG-DOPA polymers with high coupling efficiencies. In this study, PEG-DOPA molecules of varying molecular weight and architecture were synthesized using standard peptide coupling chemistry and investigated for their ability to form cross-linked hydrogels under oxidative conditions [40].
A recent finding demonstrated that PEG has a beneficial effect in early and long-term cold ischemia-reperfusion (I/R) injury, in renal medulla injury, and in the isolated perfused rat kidney. Nitrogen oxide (NO), one of the primary antithrombotic factors produced by endothelial cells, inhibits platelet and leukocyte adhesion and aggregation and interaction of platelets with damaged vessels. Therefore, the hypothesis that PEG could repair endothelial cell damage during postischemic reperfusion has led to the development of a novel macromolecular donor of NO in which 1, 8, and 16 NO-releasing molecules were covalently bound to PEG [41].
Ara-C (Cytosine arabinose, 1-(β-D-arabinofuranosyl)cytosine) is one of the older chemotherapy drugs and it is most commonly used in the treatment of acute and chronic humans leukemias. Its rapid clearance is due to the enzymatic conversion to the inactive and more soluble metabolite 1-(β-D-arabinofuranosyl)uracil (Ara-U) by the catabolic action of cytosine nucleoside deaminases widely distributed in both normal and cancerous tissue. The PEG-conjugation involves the N4 amino group of Ara-C pyrimidine ring and the carboxylic group of an amino acid spacer, but the presence of only one or two functionalisable hydroxyl groups is a severe limitation [42]. This drawback was circumvented by the conjugation of tetrafunctional or octafunctional PEG to drug. Tetrafunctional PEG was synthesized through conjugation of a bicarboxylic amino acid (L-2-aminoadipic) to the two hydroxyl groups of PEG while, by a second conjugation step between L-2-aminoadipic and tetrafunctional PEG, an octafunctional PEG was obtained. Studies toward Ara-C deamination by cytidine deaminase demonstrated that the conjugation protect toward the N4 deamination of Ara-C to Ara-U. In vitro activity of PEG-Ara-C derivatives demonstrated a much lower cytotoxicity of Ara-C conjugates than free Ara-C. The drug release rate from the conjugates is pH dependent, but it is also influenced by the polymer structure and molecular weight. The polymer moiety has an influence on the pharmacokinetic profile, thus PEG-(AD)2-(AD)4-(Ara-C)8 (AD = aminoadipic acid), PEG-(AD)2-(Ara-C)4 and mPEG20000-Lys-Ara-C present a prolonged blood residence time, which is due to the mass and shape of PEG for mPEG20000-Lys-Ara-C and to the mass of PEG and branching of the polymer moiety in the case of PEG-(AD)2-(AD)4-(Ara-C)8 and PEG-(AD)2-(Ara-C)4. These last two compounds possess the advantage of higher loading, which is a critical aspect in macromolecular prodrugs.
A new high-loading branched MultiPEGs [43] characterized by the presence of several functional groups on the same high-molecular weight derivative, as well as by adequate physicochemical properties, were evaluated for a drug delivery system with a well-studied sample drug like theophylline [44]. This molecule is used as a bronchodilator, but, in spite of its good oral bioavailability, the rather short half-life and its narrow therapeutic range (10–20 µg/mL) in plasma make it necessary to administer the drug relatively often, while the peak levels achieved shortly after administration are associated with the observed side effects. Active theophylline molecules were covalently linked to two new multifunctional high-molecular weight poly(ethylene glycol) derivatives (MultiPEG) yielding PentaPEG2000-[7-(hydroxymethyl)theophylline]12 and TetraPEG6000-[7-(hydroxymethyl)theophylline]10 [34] derivatives. The drug loading was increased up to six times in comparison with commercial PEG of the same molecular weight. A clear increase of the time of persistence within the body and a concomitant improvement of the overall pharmacokinetic properties of the synthesized prodrugs were also observed. These studies sustain the use of these new PEG-based polymeric supports as a valuable alternative for an effective drug delivery system.
Recently, [45] the attention was focused on the pharmacokinetic study of THEO-MultiPEG conjugates after oral administration in rabbit. Pharmacokinetic behavior was studied according to a two-compartment modified approach accounting for drug permeation and elimination in the blood. The PK analysis led on the basis of an ad hoc developed mathematical model revealed that, essentially, the effect of PEG is to reduce both the elimination and the absorption constants. The evaluation of the MultiPEG-drug permeability revealed that the presence of PEG causes a reduction of approximately one order of magnitude with respect to the value relating to the parent drug. The branched high-molecular weight MultiPEG proved to be a reliable drug delivery system able to prolong theophylline staying in the blood after oral administration of a THEO-MultiPEG solution. In addition, the modified compartment approach allowed, contrarily to usual compartment models, to estimate gastro-intestinal apparent permeability (P) of THEO conjugated derivative resorting to PK experimental data. This analysis showed that P and the absorption constant abruptly decreased in the case of THEO-MultiPEG systems regardless of molecular weight of polymeric moiety. In contrast, the elimination constant showed a reduction approximately proportional to molecular weight, while the distribution volume showed a small tendency to increase with the size of the polymeric chain.
New multi-arm PEGs have also been exploited for the preparation of PEG-irinotecan (NKTR-102) and PEG-docetaxel (NKTR-105) by Nektar Therapeutics. The drug has been covalently bound to a four-arm PEG. In preclinical studies, NKTR-102 showed prolonged pharmacokinetic profiles with a half-life of 15 days compared to 4 h with free irinotecan. NKTR-105 has shown good preclinical activity in colon and lung cancer xenograft models [46]. These products have just entered phase 2 clinical studies.
4. PEG-Conjugated Systems for Targeted Delivery or Combination Therapy
A different methodology, focused on the use of PEG-derivatives characterized by two orthogonal functionalities, is aimed at the production of polymeric conjugates where dissimilar but coactive molecules are conjugated to the same support. These polymeric derivatives can be adopted to modulate the systemic delivery of drugs; their use as targeted delivery systems to cell surface receptors is an interesting approach in order to treat diseased but spare healthy cells.
An interesting approach utilizes folic acid as a ligand for the folic acid receptor. Folic acid presents low molecular weight, stability, non-immunogenicity, low cost, significant differences in expression level of folate receptor (FR) on normal and cancer cell surfaces and comparatively simpler conjugation chemistry. Anthracycline antibiotics, particularly doxorubicin (DOX), rank among the most used cancerostatics in current oncological chemotherapy. However, it sometimes causes strong side effects which could lead to congestive heart failure and death. Yoo [47] conjugated separately folate (FOL) and DOX at α- and ω-terminal end of a PEG chain to produce FOL-PEG-DOX. The FOL-PEG-DOX conjugate has a targeting moiety at one end and an anti-cancer drug moiety at the other end in a single flexible PEG chain structure. It was hypothesized that FOL-PEG-DOX could sterically stabilize deprotonated and hydrophobic DOX nano-aggregates in an aqueous solution by anchoring the conjugated DOX moiety to DOX aggregates while exposing the more hydrophilic FOL moiety outside. The doxorubicin nano-aggregates showed a greater extent of intracellular uptake against folate-receptor-positive cancer cells than folate-receptor-negative cells, indicating that the cellular uptake occurred via a folate-receptor-mediated endocytosis mechanism. They also exhibited more potent cytotoxic effect on KB cells than free doxorubicin.
Newly, [48] two series of targeted and non-targeted PEG-epirubicin conjugates were synthesised, using folic acid as targeting moiety. The heterobifunctional HO-PEG-COOH was used as a polymeric carrier. A dendron structure was synthesised at one end of the PEG chain with the aim of increasing the number of folic acid molecules while L-2-aminoadipic acid was used as branching unit. The targeted conjugates showed a cytotoxicity profile dependent on both the number of folic acid units per PEG chain and the level of folic acid receptor expression in the tested cell lines. Differently, the non-targeted conjugates, although being more active, had almost the same cytotoxicity in the cell lines considered.
Gemcitabine, 2′,2′-difluoro-2′-deoxycytidine (dFdC), is one of the most effective cytotoxic agents for lung cancer non-small cell carcinoma (NSCLC), but it is also commonly used to treat pancreatic, bladder and breast cancer. Unfortunately, gemcitabine possesses a rapid body clearance that limits its efficacy; thus, a frequent administration schedule at high drug doses is required and this leads to significant side effects. To overcome these limitations, recently two folic acid targeted PEG-gemcitabine conjugates, differing in the drug loading, were prepared [49] and in order to evaluate their active targeting superiority three non-targeted conjugates were also synthesized. Anyway, for all types of conjugates, the drug linkage involved the N4-amino group of the pyrimidine ring of gemcitabine and the COOH of PEG, while folic acid was linked to the PEG amino group mainly through its γ carboxylic group. It was chosen as targeting agent because its receptor is often over-expressed in many tumors. Polymer conjugation of gemcitabine increased the drug plasma half-life, by reducing its kidney clearance. The targeted conjugates showed a higher antiproliferative activity and a higher selectivity than non-targeted ones when studied against KB-3-1 cell line that over-expressed the folic acid receptor. The decreased cytotoxicity of these targeted conjugates in cell lines that do not over-express the folic acid receptor can indicate that these conjugates need a receptor-mediated endocytosis mechanism for cell penetration.
Recently, nanoparticles based on gold chemistry have attracted significant research and practical attention. They are versatile agents with a variety of biomedical applications as well as for drug and gene delivery [50]. For systemic applications, long-circulating nanoparticles are desidered for passive targeting to tumors and inflammatory sites. Surface modification of gold nanoparticles through the PEG spacer would allow the modified nanoparticles to remain in the systemic circulation for a prolonged period and provide flexibility to the attached ligand for efficient interaction with its target. For the development of surface-functionalized gold nanoparticles as cellular probes and delivery agents, Shenoy synthesized hetero-bifunctional poly(ethylene glycol) (MW 1,500) having a thiol group on one terminus and a reactive functional group on the other for use as a flexible spacer [51]. Coumarin, a model fluorescent dye, was conjugated to one end of the PEG spacer and gold nanoparticles were modified with coumarin-PEG-thiol. Using the PEG spacer, the gold nano-platform can be conjugated with a variety of biologically relevant ligands such as fluorescent dyes, antibodies, etc. in order to target, probe, and induce a stimulus at the target site. Another important objective was the demonstration of the attachment to antibodies, which has led to the synthesis of PEG derivatized biomolecules including labeled antibody conjugates [52] with excellent fluorescence characteristics, permitting cellular affinity studies.
In order to investigate the capabilities of an actively targeting superparamagnetic nanoparticle to specifically deliver drug and MRI contrast agents to cancer cells, iron oxide nanoparticles were also synthesized and conjugated to both a chemotherapeutic agent, methotrexate, and a targeting ligand, chlorotoxin, through a poly(ethylene glycol) linker [53]. The targeting nanoparticle demonstrated preferential accumulation and increased cytotoxicity in tumor cells. Furthermore, prolonged retention of these nanoparticles was observed within tumors in vivo.
Another non-invasive modality to treat solid tumors includes the photodynamic therapy (PDT) which involves the delivery of photosensitizers to tumor tissues followed by irradiation with a laser of appropriate wavelength. Currently available clinical photosensitizers are low molecular weight compounds with poor tumor specificity. In order to improve therapeutic efficacy of the photosensitizers, a modified poly-(L-glutamic acid)-(mesochlorin e6)-(Gd-DO3A) conjugate with poly(ethylene glycol) was synthesized and its efficacy in contrast-enhanced MRI-guided photodynamic therapy was evaluated [54]. The PEGylated conjugate had longer blood circulation, lower liver uptake and higher tumor accumulation than the non-pegylated conjugate.
In addition, bifunctional PEG molecules provide a novel approach not only in multidrug PEG-conjugated systems but also to retargeting viral vectors without the need to genetically modify the vector. The coupling of the protein FGF2 to the surface of an adenovirus (Ad) confers a novel route of entry upon the virus. Moreover, polymer modification of adenovirus vectors results in reduced localization of adenovirus to nontarget tissues [55]. This kind of approach may lead to the development of a gene therapy vector capable of targeting a therapeutic gene to diseased cells, while reducing toxicity in other tissues. A new process has been recently set up in this field that permits the modification of inexpensive commercial PEGs by the introduction of two different functional groups, orthogonally protected, at their extremities. The final introduction of any couple of different molecules at the ends of the same polymeric chain is consequently easily allowed [56].
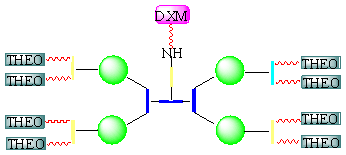
An additional example of mixed multidrug PEG-conjugated systems was given by the synthesis of a new multimeric, multifunctional PEG-based construct (MultiPEG) conjugated to two different drugs, theophylline (THEO) and dexamethasone (DXM), considered as model drugs [43]. The investigation of these drugs, characterized by low solubility in water and poor bioavailability, included PEGylation both on linear [44] as well as on new multimeric PEGs [34] and found that polymer conjugation could offer new advantages for the delivery and release of drugs such as an increased drug loading capability and a long lasting release. In this new approach, the two drugs have been conjugated on the same, new multifunctional MultiPEG, in a ratio which matches the usual therapeutic amounts when separately dosed. The effect of the bifunctional polymeric carrier on the in vitro stability and the permeation across the intestinal membrane of these drugs, once conjugated on the same support, have been evaluated (Figure 4).
5. Conclusions
As observed, PEGylation can be a very helpful and promising technique to improve the pharmacological properties of many biopharmaceuticals due to the considerable importance of achieving new innovative and specialized drug delivery systems. Thus, intelligent drug delivery systems such as PEGylation will determine the commercial success of the pharmaceutical industry in the future. With its broad applicability, high efficiency and comparably low costs, PEGylation will certainly maintain a leading position. An area that still needs further development deals with the problem of obtaining monodisperse or at least very low polydisperse PEGs, mainly with high molecular weight polymer species. This polydispersity comes to the fore as a quality problem for modified drugs; as a consequence, long and linear poly(ethylene glycol) chains used for conjugation, available today as a mixture of polymeric chains with different chain lengths, should be produced in a monodisperse fashion, as recently proposed [57]. As described, a new area of PEG-based developments is related to the presence of a higher number of functional groups on this material, since the better biological performances are given by high-molecular weight PEGs, negatively counterbalanced by their reduced loading capacity per unit of weight. Moreover, further activities are needed to improve the production of PEG derivatives carrying functional groups that are selectively and reversibly protected. These derivatives will allow a step-by-step assembling of new conjugates in which different, but complementary, pharmacologically active units are carried on the same polymeric moiety.



References
- Davis, F.F. The origin of PEGnology. Adv. Drug Deliv. Rev. 2007, 54, 457–458. [Google Scholar]
- Maeda, H.; Seymour, L.W.; Miyamoto, Y. Conjugates of anticancer agents and polymers: Advantages of macromolecular therapeutics in vivo. Bioconjug. Chem. 2007, 3, 351–362. [Google Scholar]
- Harris, J.M.; Chess, R.B. Effect of pegylation on pharmaceuticals. Nat. Rev. 2003, 2, 214–221. [Google Scholar]
- Maeda, H.; Wu, J.; Sawa, T.; Matsumura, Y.; Hori, K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: A review. J. Control. Release 2000, 65, 271–284. [Google Scholar]
- Yamaoka, T.; Tabata, Y.; Ikada, Y. Distribution and tissue uptake of poly(ethylene glycol) with different molecular weights after intravenous administration to mice. J. Pharm. Sci. 1994, 83, 601–606. [Google Scholar]
- Yamaoka, T.; Tabata, Y.; Ikada, Y. Fate of water-soluble polymers administered via different routes. J. Pharm. Sci. 1995, 84, 349–354. [Google Scholar]
- Bentley, M.D.; Harris, J.M.; Kozlowski, A. Heterobifunctional Polyethylene Glycol Derivatives and Methods for Their Preparation. U.S. Patent 2,010,063,328, 11 March 2010. [Google Scholar]
- Pasut, G.; Veronese, F.M. Polymer-drug conjugation, recent achievements and general strategies. Prog. Polym. Sci. 2007, 32, 933–961. [Google Scholar]
- Kopecek, J.; Kopeckova, P.; Minko, T.; Lu, Z. HPMA copolymer-anticancer drug conjugates: Design, activity, and mechanism of action. Eur. J. Pharm. Biopharm. 2000, 50, 61–81. [Google Scholar]
- Li, C. Poly(L-glutamic acid)-anticancer drug conjugates poly(L-glutamic acid)-anticancer drug conjugates. Adv. Drug Delivery Rev. 2002, 54, 695–713. [Google Scholar]
- Choe, Y.H.; Conover, C.D.; Wu, D.; Royzen, M.; Gervacio, Y; Borowski, V. Anticancer drug delivery systems: Multiloaded N4-acyl poly(ethylene glycol) prodrugs of ara-C: II. Efficacy in ascites and solid tumors. J. Control Release 2002, 79, 55–70. [Google Scholar]
- Pasut, G.; Scaramuzza, S.; Schiavon, O.; Mendichi, R.; Veronese, F.M. PEG-epirubicin conjugates with high loading. J. Bioac. Comp. Pol. 2005, 20, 13–30. [Google Scholar]
- Berna, M.; Dalzoppo, D.; Pasut, G.; Manunta, M.; Izzo, L.; Jones, A.T. Novel monodisperse PEG-dendrons as new tools for targeted drug delivery: Synthesis, characterization and cellular uptake. Biomacromolecules 2006, 7, 146–153. [Google Scholar]
- Gajbhiye, V.; Kumar, P.V.; Tekade, R.K.; Jain, N.K. Pharmaceutical and biomedical potential of PEGylated dendrimers. Curr. Pharm. Des. 2004, 13, 415–429. [Google Scholar]
- Nathan, A.; Zalipsky, S.; Kohn, J. Strategies for covalent attachment of doxorubicin to PEG. J. Bioact. Compat. Polym. 1994, 9, 239–251. [Google Scholar]
- Pechar, M.; Ulbrich, K.; Subr, V.; Seymour, L.W.; Schacht, E.H. Poly(ethylene glycol) multiblock copolymer as a carrier of anti-cancer drug doxorubicin. Bioconjugate Chem. 2000, 11, 131–139. [Google Scholar]
- Cheng, J.; Khin, K.T.; Jensen, G.S.; Liu, A.; Davis, M.E. Synthesis of linear, β-Cyclodextrin-based polymers and their camptothecin conjugates. Bioconjugate Chem. 2003, 14, 1007–1017. [Google Scholar]
- Kumar, R.; Chen, M.H.; Parmar, V.S.; Samuelson, L.A.; Kumar, J.; Nicolosi, R.; Yoganathan, S.; Watterson, A.C. Supramolecular assemblies based on copolymers of PEG600 and functionalized aromatic diesters for drug delivery applications. J. Am. Chem. Soc. 2004, 126, 10640–10644. [Google Scholar]
- De Jesus, O.L.P.; Ihre, H.R.; Gagne, L.; Fréchet, J.M.L.; Szoka, F.C. Olyester dendritic system for drug delivery applications: In vitro and in vivo evaluations. Bioconjugate Chem. 2002, 13, 453–461. [Google Scholar]
- Gillies, E.R.; Fréchet, J.M.L. Designing macromolecules for therapeutic applications: Polyester dendrimer-poly(ethylene oxide) “bow-tie” hybrids with tunable molecular weight and architecture. J. Am. Chem. Soc. 2002, 124, 14137–14146. [Google Scholar]
- Giullaudeu, S.J.; Fox, E.M.; Haidar, Y.M.; Dy, E.; Szoka, F.C.; Fréchet, J.M.L. PEGylated dendrimers with core functionality for biological applications. Bioconjugate Chem. 2008, 19, 461–469. [Google Scholar]
- Benaglia, M.; Annunziata, R.; Cinquini, M.; Cozzi, F.; Ressel, S. Synthesis of new poly(ethyleneglycol)s with a high loading capacity. J. Org. Chem. 1998, 63, 8628–8629. [Google Scholar]
- Reed, N.N.; Janda, K.D. Steath star polymers: A new high-loading scaffold for liquid-phase organic synthesis. Org. Lett. 2000, 2, 1311–1313. [Google Scholar]
- Guiotto, A.; Pozzobon, M.; Sanavio, C.; Schiavon, O.; Orsolini, P.; Veronese, F.M. An improved procedure for the synthesis of branched polyethylene glycols (PEGs) with the reporter dipeptide Met-beta Ala for protein conjugation. Bioorg. Med. Chem. Lett. 2002, 12, 177–180. [Google Scholar]
- Monfardini, C.; Schiavon, O.; Caliceti, P.; Morpurgo, M.; Harris, M.J.; Veronese, F.M. A branched monomethoxypoly(ethylene glycol) for protein modification. Bioconjugate Chem. 1995, 6, 62–69. [Google Scholar]
- Harris, J.M.; Veronese, F.M.; Caliceti, P.; Schiavon, O. Multiarmed, Monofunctional Polymer for Coupling to Molecules and Surfaces. U.S. Patent 5,932,462, 3 August 1999. [Google Scholar]
- Fishman, M.; Farrah, E.; Zhong, J.H.; Paramanantham, S.; Carrera, C.; Lee-Ruff, E. Synthesis and investigation of novel branched PEG-based soluble polymer supports. J. Org. Chem. 2003, 68, 9843–9846. [Google Scholar]
- Gillies, E.R.; Fréchet, J.M. Dendrimers and dendritic polymers in drug delivery. Drug Discov. Today 2005, 10, 35–43. [Google Scholar]
- Haag, R.; Sunder, A.; Stumbé, J.F. An approach to glycerol dendrimers and pseudo-dendritic polyglycerols. J. Am. Chem. Soc. 2000, 122, 2954–2955. [Google Scholar]
- Ihe, H.; De Jesús, O.L.P.; Fréchet, J.M. Fast and convenient divergent synthesis of aliphatic ester dendrimers by anhydride coupling. J. Am. Chem. Soc. 2001, 123, 5908–5917. [Google Scholar]
- Sunder, A.; Mülhaupt, R.; Haag, R.; Frey, H. Chiral hyperbranched dendron-analogs. Macromolecules 2000, 33, 253–254. [Google Scholar]
- Liu, X.M.; Thakur, A.; Wang, D. Efficient synthesis of linear multifunctional poly(ethylene glycol) by copper(I)-catalyzed Huisgen 1,3-dipolar cycloaddition. Biomacromolecules 2007, 8, 2653–2658. [Google Scholar]
- Ballico, M.; Drioli, S.; Bonora, G.M. MultiPEG: High molecular weight multifunctional poly(ethylen glycol)s assembled by a dendrimer-like approach. Eur. J. Org. Chem. 2005, 2005, 2064–2073. [Google Scholar]
- Zacchigna, M.; Di Luca, G.; Cateni, F.; Maurich, V.; Ballico, M.; Bonora, G.M.; Drioli, S. New MultiPEG-conjugated theophylline derivatives: Synthesis and pharmacological evaluations. Eur. J. Pharm. Sci. 2007, 30, 343–350. [Google Scholar]
- Drioli, S.; Bonora, G.M.; Ballico, M. Synthesis and characterization of new multifunctional high-molecular weight PEG derivatives (MultiPEG)s. Open Org. Chem. J. 2008, 2, 17–25. [Google Scholar]
- Chabot, G.G. Clinical pharmacokinetics of irinotecan. Clin. Pharmacokinet. 1997, 33, 245–259. [Google Scholar]
- Zhang, J.A.; Xuan, T.; Parmar, M.; Ma, L.; Ugwu, S.; Ali, S.; Ahmad, I. Development and characterization of a novel liposome-based formulation of SN-38. Int. J. Pharm. 2004, 270, 93–107. [Google Scholar]
- Zhao, H.; Rubio, B.; Sapra, P.; Wu, D.; Reddy, P.; Sai, P.; Martinez, A.; Gao, Y.; Lozanguiez, Y.; Longley, C.; et al. Novel prodrugs of SN38 using multiarm poly(ethylene glycol) linkers. Bioconjugate Chem. 2008, 19, 849–859. [Google Scholar]
- Johnson, J.A.; Finn, M.G.; Koberstein, J.T.; Turro, N.J. Construction of linear polymers, dendrimers, networks, and other polymeric architectures by copper-catalyzed azide-alkyne cycloaddition “click” chemistry. Macromol. Rapid Commun. 2008, 29, 1052–1072. [Google Scholar]
- Lee, B.P.; Dalsin, J.L.; Messersmith, P.B. Enzymatic and non-enzymatic pathways to formation of Dopa-modified PEG hydrogels. Polym. Prepr. 2001, 42, 151–152. [Google Scholar]
- Bertuglia, S.; Veronese, F.M.; Pasut, G. Polyethylene glycol and a novel developed polyethylene glycol-nitric oxide normalize arteriolar response and oxidative stress in ischemia-reperfusion. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, 1536–1544. [Google Scholar]
- Schiavon, O.; Pasut, G.; Moro, S.; Orsolini, P.; Guiotto, A.; Veronese, F.M. PEG-Ara-C conjugates for controlled release. Eur. J. Med. Chem. 2004, 39, 123–133. [Google Scholar]
- Bonora, G.M.; Drioli, S.; Ballico, M. Multifunctional Polyethylene Glycol Derivatives: Preparation and Use. WO 03/093346 A1. 2003. [Google Scholar]
- Zacchigna, M.; Di Luca, G.; Cateni, F.; Zorzet, S.; Maurich, V. Improvement of physicochemical and biopharmaceutical properties of theophylline by poly(ethylene glycol) conjugates. Farmaco 2003, 58, 1307–1312. [Google Scholar]
- Grassi, M.; Bonora, G.M.; Drioli, S.; Cateni, F.; Zacchigna, M. Pharmacokinetic analysis of multi PEG-theophylline conjugates. Biopharm. Drug Dispos. 2011. submitted. [Google Scholar]
- Pasut, G.; Veronese, F.M. PEG conjugates in clinical development or use as anticancer agents: An overview. Adv. Drug Delivery Rev. 2009, 61, 1177–1188. [Google Scholar]
- Yoo, H.S.; Park, T.G. Folate-receptor-targeted delivery of doxorubicin nano-aggregates stabilized by doxorubicin-PEG-folate conjugate. J. Control. Release 2004, 100, 247–256. [Google Scholar]
- Canal, F.; Vicent, M.J.; Pasut, G.; Schiavon, O. Relevance of folic acid/polymer ratio in targeted PEG-epirubicin conjugates. J. Control. Release 2010, 146, 388–399. [Google Scholar]
- Pasut, G.; Canal, F.; Dalla Via, L.; Arpicco, S.; Veronese, F.M.; Schiavon, O. Antitumoral activity of PEG-gemcitabine prodrugs targeted by folic acid. J. Control. Release 2008, 127, 239–248. [Google Scholar]
- Thomas, M.; Klibanov, A. Conjugation to gold nanoparticles enhances polyethylenimine's transfer of plasmid DNA into mammalian cells. Proc. Natl. Acad. Sci. USA 2003, 100, 9138–9143. [Google Scholar]
- Shenoy, D.; Fu, W.; Li, J.; Crasto, C.; Jones, G.; DiMarzio, C.; Sridhar, S.; Amiji, M. Surface functionalization of gold nanoparticles using hetero-bifunctional poly(ethylene glycol) spacer for intracellular tracking and delivery. Int. J. Nanomed. 2006, 1, 51–57. [Google Scholar]
- Li, J.; Crasto, C.F.; Weinberg, J.S.; Amiji, M.; Shenoy, D.; Sridhar, S.; Bubley, G.J.; Jones, G.B. An approach to heterobifunctional poly(ethyleneglycol) bioconjugates. Bioorg. Med. Chem. Lett. 2005, 15, 5558–5561. [Google Scholar]
- Sun, C.; Fang, C.; Stephen, Z.; Veiseh, O.; Hansen, S.; Lee, D.; Ellenbogen, R.G.; Olson, J.; Zhang, M. Tumor-targeted drug delivery and MRI contrast enhancement by chlorotoxin-conjugated iron oxide nanoparticles. Nanomedicine 2008, 3, 495–505. [Google Scholar]
- Vaidya, A.; Sun, Y.; Feng, Y.; Emerson, L.; Jeong, E.; Lu, Z. Contrast-enhanced MRI-guided photodynamic cancer therapy with a PEGylated bifunctional polymer conjugate. Pharm. Res. 2008, 25, 2002–2011. [Google Scholar]
- Lanciotti, J.; Song, A.; Doukas, J.; Sosnowski, B.; Pierce, G.; Gregory, R.; Wadsworth, S.; Riordan, C. Targeting adenoviral vectors using heterofunctional polyethylene Glicol FGF2 Conjugates. Mol. Ther. 2003, 8, 99–107. [Google Scholar]
- Campaner, P.; Drioli, S.; Bonora, G.M. Synthesis of selectively end-modified high-molecular weight polyethylenglycol. Lett. Org. Chem. 2006, 10, 773–779. [Google Scholar]
- Niculescu-Duvaz, D.; Getaz, J.; Springer, C.J. Long functionalized poly(ethylene glycol)s of defined molecula weight: Synthesis and application in solid-phase synthesis of conjugates. Bioconjugate Chem. 2008, 19, 973–981. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zacchigna, M.; Cateni, F.; Drioli, S.; Bonora, G.M. Multimeric, Multifunctional Derivatives of Poly(ethylene glycol). Polymers 2011, 3, 1076-1090. https://doi.org/10.3390/polym3031076
Zacchigna M, Cateni F, Drioli S, Bonora GM. Multimeric, Multifunctional Derivatives of Poly(ethylene glycol). Polymers. 2011; 3(3):1076-1090. https://doi.org/10.3390/polym3031076
Chicago/Turabian StyleZacchigna, Marina, Francesca Cateni, Sara Drioli, and Gian Maria Bonora. 2011. "Multimeric, Multifunctional Derivatives of Poly(ethylene glycol)" Polymers 3, no. 3: 1076-1090. https://doi.org/10.3390/polym3031076
APA StyleZacchigna, M., Cateni, F., Drioli, S., & Bonora, G. M. (2011). Multimeric, Multifunctional Derivatives of Poly(ethylene glycol). Polymers, 3(3), 1076-1090. https://doi.org/10.3390/polym3031076