Temperature Induced Solubility Transitions of Various Poly(2-oxazoline)s in Ethanol-Water Solvent Mixtures
Abstract
:1. Introduction
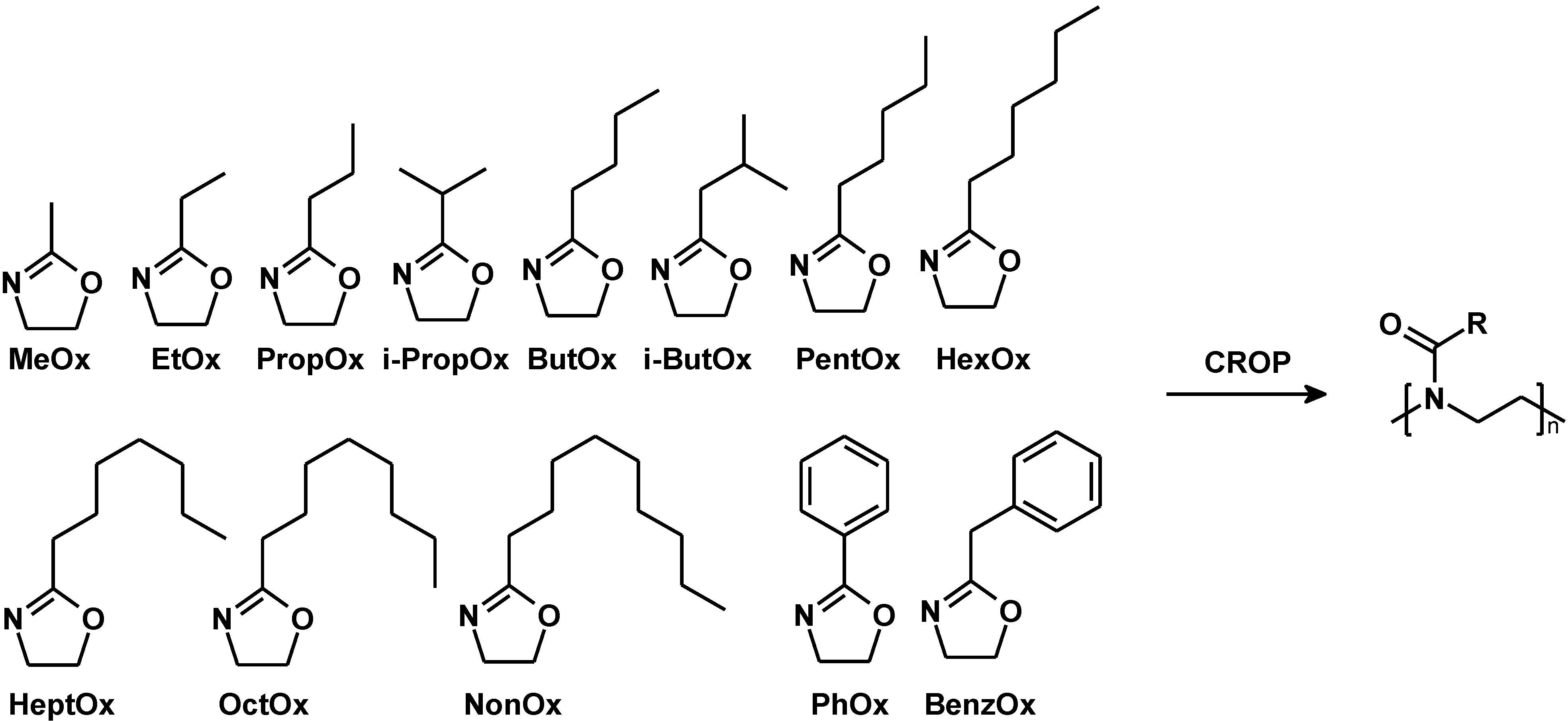
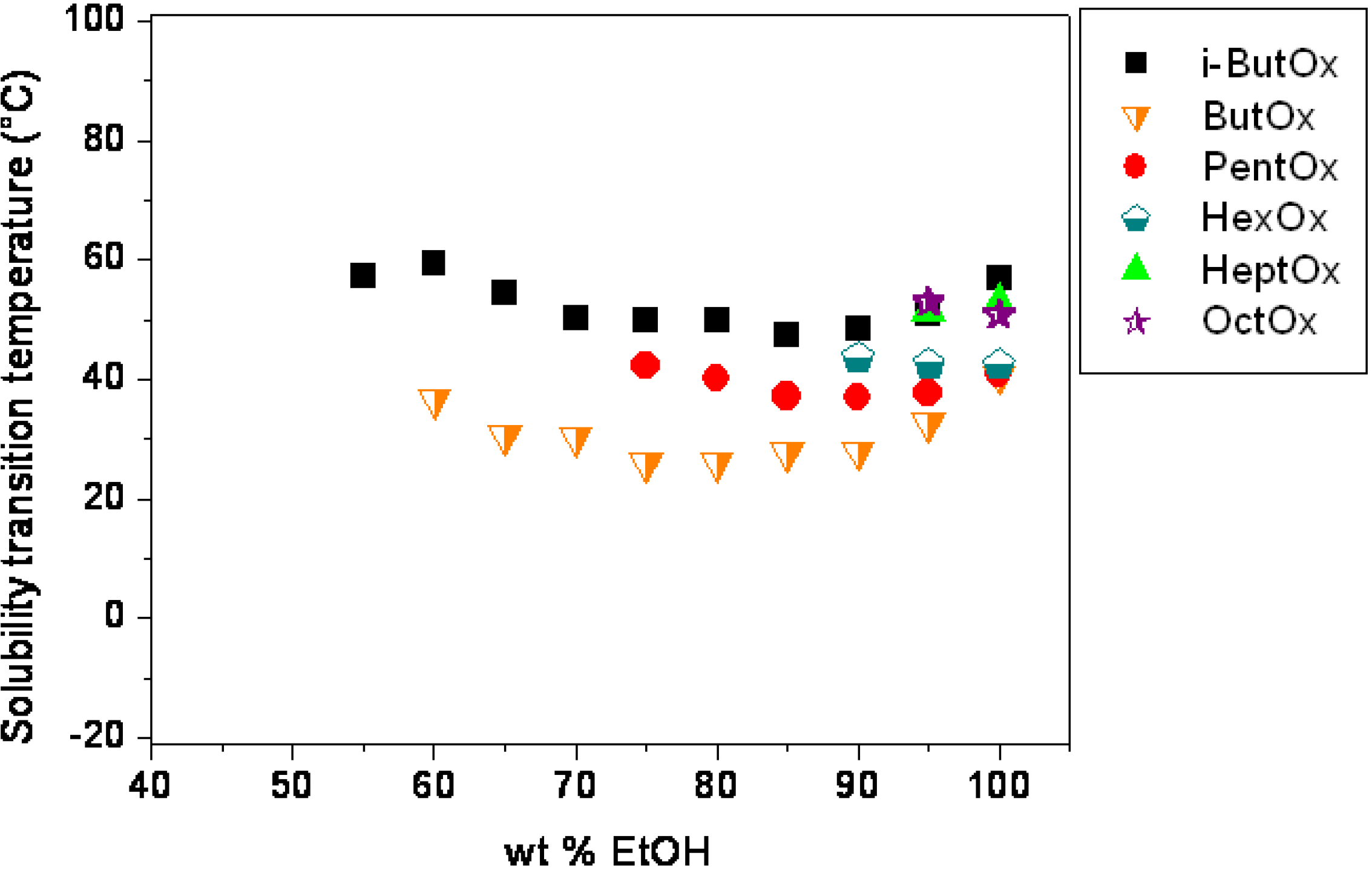
2. Results and Discussion
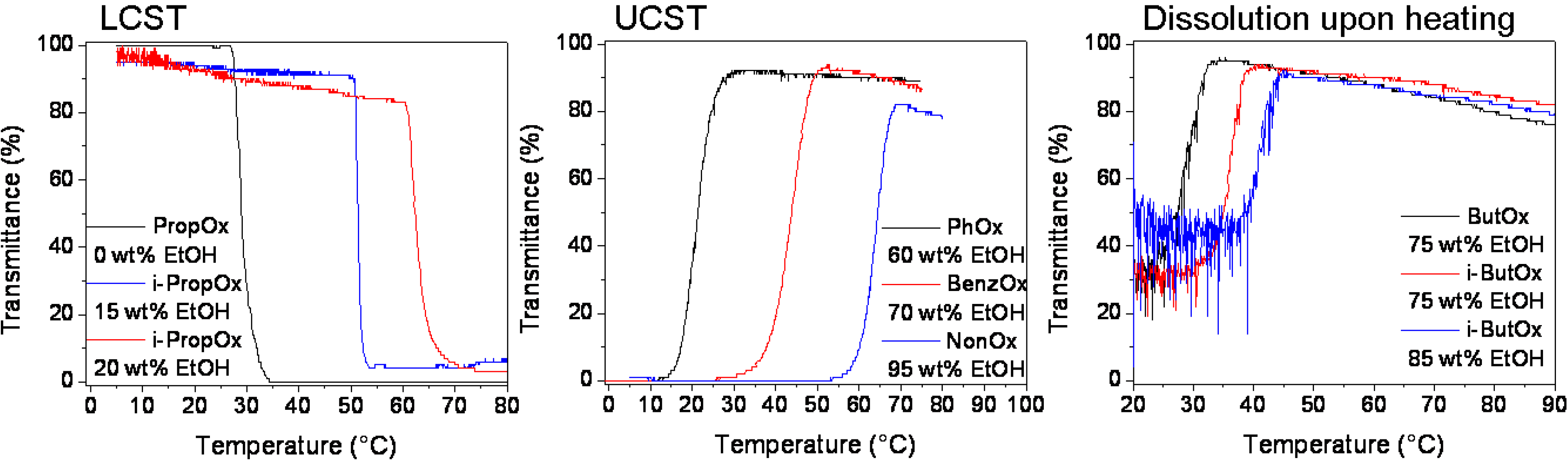

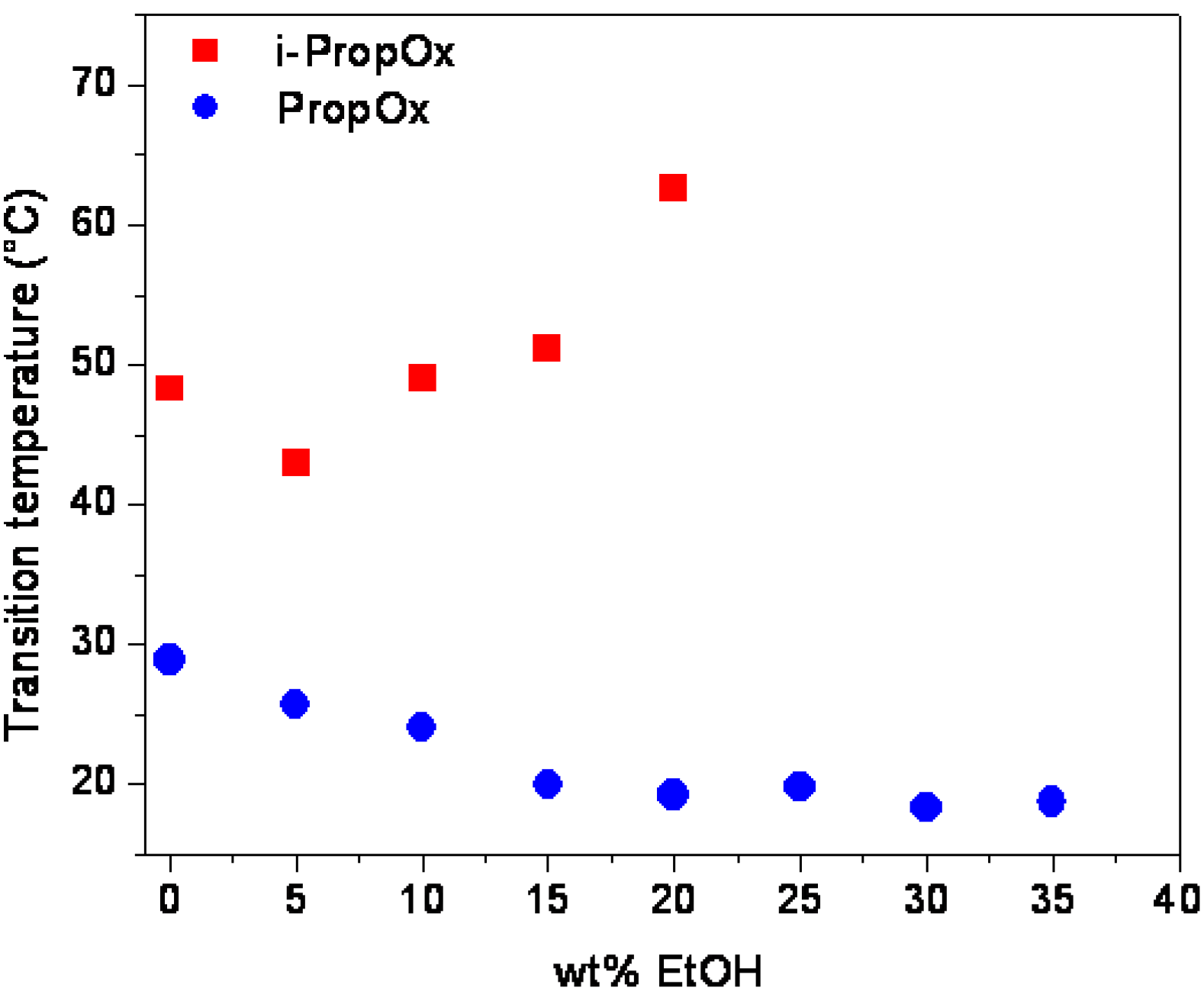
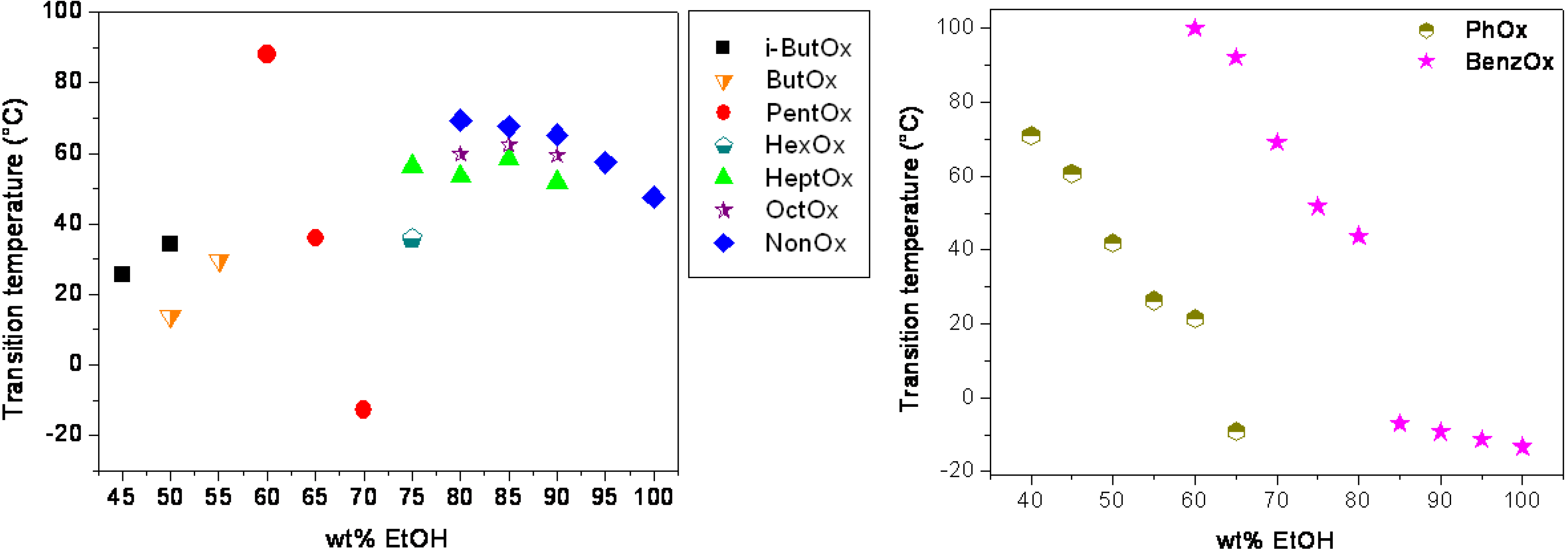
3. Experimental Section
4. Conclusions
Acknowledgements
References and Notes
- Tomalia, D.A.; Sheetz, D.P. Homopolymerization of 2-alkyl- and 2-aryl-2-oxazolines. J. Polym. Sci. A Polym. Chem. 1966, 4, 2253–2265. [Google Scholar] [CrossRef]
- Seeliger, W.; Aufderhaar, E.; Diepers, W.; Feinauer, R.; Nehring, R.; Thier, W.; Hellmann, H. Recent syntheses and reactions of cyclic imidic esters. Angew. Chem. 1966, 20, 913–927. [Google Scholar] [CrossRef]
- Kagiya, T.; Narisawa, S.; Maeda, T.; Fukui, K. Ring-opening polymerization of 2-substituted 2-oxazolines. Polym. Lett. 1966, 4, 441–445. [Google Scholar] [CrossRef]
- Bassiri, T.G.; Levy, A.; Litt, M. Polymerization of cyclic imino ethers. I. Oxazolines. Polym. Lett. 1967, 5, 871–879. [Google Scholar] [CrossRef]
- Hoogenboom, R. Poly(2-oxazoline)s: A polymer class with numerous potential applications. Angew. Chem. Int. Ed. 2009, 48, 7978–7994. [Google Scholar] [CrossRef]
- Adams, N.; Schubert, U.S. Poly(2-oxazolines) in biological and biomedical application contexts. Adv. Drug Delivery Rev. 2007, 59, 1504–1520. [Google Scholar] [CrossRef]
- Knop, K.; Hoogenboom, R.; Fischer, D.; Schubert, U.S. Poly(ethylene glycol) in drug delivery: Pros and cons as well as potential alternatives. Angew. Chem. Int. Ed. 2010. [Google Scholar] [CrossRef]
- Schlaad, H.; Diehl, C.; Gress, A.; Meyer, M.; Demirel, A.L.; Nur, Y.; Bertin, A. Poly(2-oxazoline)s as smart bioinspired polymers. Macromol. Rapid Commun. 2010, 31, 511–525. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.T.; Cornelissen, J.J.L.M.; Nolte, R.J.M.; van Hest, J.C.M. Polymeric monosaccharide receptors responsive at neutral pH. J. Am. Chem. Soc. 2009, 131, 13908–13909. [Google Scholar] [CrossRef] [PubMed]
- Qing, G.; Wang, X.; Fuchs, H.; Sun, T. Nucleotide-responsive wettability on a smart polymer surface. J. Am. Chem. Soc. 2009, 131, 8370–8371. [Google Scholar] [CrossRef] [PubMed]
- Amir, R.J.; Zhong, S.; Pochan, D.J.; Hawker, C.J. Enzymatically triggered self-assembly of block copolymers. J. Am. Chem. Soc. 2009, 131, 13949–13951. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.A.; Hamilton, A.; Yang, J.; Cremar, L.D.; van Gough, D.; Potisek, S.L.; Ong, M.T.; Braun, P.V.; Martínez, T.J.; White, S.R.; Moore, J. S.; Sottos, N.R. Force-induced activation of covalent bonds in mechanoresponsive polymeric materials. Nature 2009, 459, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Piermattei, A.; Karthikeyan, S.; Sijbesma, R.P. Activating catalysts with mechanical force. Nat. Chem. 2009, 1, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Sershen, S.R.; Westcott, S.L.; Halas, N.J.; West, J.L. Temperature-sensitive polymer-nanoshell composites for photothermally modulated drug delivery. J. Biomed. Mater. Res. 2000, 51, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Keurentjes, J.T.F.; Kemmere, M.F.; Bruinewoud, H.; Vertommen, M.A.M.E.; Rovers, S.A.; Hoogenboom, R.; Stemkens, L.F.S.; Peters, F.L.A.M.A.; Tielen, N.J.C.; van Asseldonk, D.T.A.; Gabriel, A.F.; Joosten, E.A.; Marcus, M.A.E. Externally triggered glass transition switch for localized on-demand drug delivery. Angew. Chem. Int. Ed. 2009, 48, 9867–9870. [Google Scholar] [CrossRef]
- Wood, K.C.; Zacharia, N.S.; Schmidt, D.J.; Wrightman, S.N.; Andaya, B.J.; Hammond, P.T. Electroactive controlled release thin films. Proc. Natl. Acad. Sci. USA 2008, 105, 2280–2285. [Google Scholar] [CrossRef] [PubMed]
- Hergt, R.; Hiergeist, R.; Hilger, I.; Kaiser, W.A.; Lapatnikov, Y.; Margel, S.; Richter, U. Maghemite nanoparticles with very high AC-losses for application in RF-magnetic hyperthermia. J. Magnetism Magn. Mater. 2004, 270, 345–357. [Google Scholar] [CrossRef]
- Schild, H.G. Poly(N-isopropylacrylamide): Experiment, theory and application. Prog. Polym. Sci. 1992, 17, 163–249. [Google Scholar] [CrossRef]
- Russell, T.P. Surface responsive materials. Science 2002, 297, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Oosten, C.L.; Bastiaansen, C.W.M.; Broer, D.J. Printed artificial cilia from liquid-crystal network actuators modularly driven by light. Nat. Mater. 2009, 8, 677–682. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, C.; Ritter, H. Color change of N-isopropylacrylamide copolymer bearing Reichardts dye as optical sensor for lower critical solution temperature and for host-guest interaction with β-cyclodextrin. J. Am. Chem. Soc. 2007, 129, 3502–3503. [Google Scholar] [CrossRef] [PubMed]
- Gota, C.; Okabe, K.; Funatsu, T.; Harada, Y.; Uchiyama, S. Hydrophilic fluorescent nanogel thermometer for intracellular thermometry. J. Am. Chem. Soc. 2009, 131, 2766–2767. [Google Scholar] [CrossRef] [PubMed]
- Pietsch, C.; Hoogenboom, R.; Schubert, U.S. Soluble polymeric dual sensor for temperature and pH value. Angew. Chem. Int. Ed. 2009, 48, 5653–5656. [Google Scholar] [CrossRef]
- Kudaibergenov, S.; Jaeger, W.; Laschewsky, A. Polymeric betaines: Synthesis, characterization and application. Adv. Polym. Sci. 2006, 201, 157–224. [Google Scholar]
- Dimitrov, I.; Trzebicka, B.; Mueller, A.H.E.; Dworak, A.; Tsvetanov, C.B. Thermosensitive water-soluble copolymers with doubly responsive reversibly interacting entities. Prog. Polym. Sci. 2007, 32, 1275–1343. [Google Scholar] [CrossRef]
- Costa, R.O.R.; Freitas, R.F.S. Phase behavior of poly(N-isopropylacrylamide) in binary aqueous solutions. Polymer 2002, 43, 5879–5885. [Google Scholar] [CrossRef]
- Lin, P.; Clash, C.; Pearce, E.M.; Kwei, T.K.; Aponte, M.A. Solubility and miscibility of poly(ethyl oxazoline). J. Polym. Sci. B Polym. Phys. 1988, 26, 603–619. [Google Scholar] [CrossRef]
- Uyama, H.; Kobayashi, S. A novel thermo-sensitive polymer. Poly(2-iso-propyl-2-oxazoline). Chem. Lett. 1992, 21, 1643–1646. [Google Scholar] [CrossRef]
- Park, J.-S.; Kataoka, K. Comprehensive and accurate control of thermosensitivity of poly(2-alkyl-2-oxazoline)s via well-defined gradient or random copolymerization. Macromolecules 2007, 40, 3599–3609. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Thijs, H.M.L.; Jochems, M.J.H.C.; van Lankvelt, B.M.; Fijten, M.W.M.; Schubert, U.S. Tuning the LCST of poly(2-oxazoline)s by varying composition and molecular weight: alternatives to poly(N-isopropylacrylamide)? Chem. Commun. 2008, 44, 5758–5760. [Google Scholar] [CrossRef]
- Afroze, F.; Nies, E.; Berghmans, H. Phase transitions in the system poly(N-isopropylacrylamide)/water and swelling behaviour of the corresponding networks. J. Mol. Struct. 2000, 554, 55–68. [Google Scholar] [CrossRef]
- Christova, D.; Velichkova, R.; Loos, W.; Goethals, E.J.; Du Prez, F. New thermo-responsive polymer materials based on poly(2-ethyl-2-oxazoline) segments. Polymer 2003, 44, 2255–2261. [Google Scholar] [CrossRef]
- Meyer, M.; Antonietti, M.; Schlaad, H. Unexpected thermal characteristics of aqueous solutions of poly(2-isopropyl-2-oxazoline). Soft Matter 2007, 3, 430–431. [Google Scholar] [CrossRef]
- Demirel, A.L.; Meyer, M.; Schlaad, H. Formation of polyamide nanofibers by directional crystallization in aqueous solution. Angew. Chem. Int. Ed. 2007, 46, 8622–8624. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Thijs, H.M.L.; Wouters, D.; Hoeppener, S.; Schubert, U.S. Tuning solution polymer properties by binary water-ethanol solvent mixtures. Soft Matter 2008, 4, 103–107. [Google Scholar] [CrossRef]
- Lambermont-Thijs, H.M.L.; Hoogenboom, R.; Fustin, C.-A.; Bomal-D’Haese, C.; Gohy, J.-F.; Schubert, U.S. Solubility behavior of amphiphilic block and random copolymers based on 2-ethyl-2-oxazoline and 2-nonyl-2-oxazoline in binary water-ethanol mixtures. J. Polym. Sci. A Polym. Chem. 2009, 47, 515–522. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Lambermont-Thijs, H.M.L.; Jochems, M.J.H.C.; Hoeppener, S.; Guerlain, C.; Fustin, C.-A.; Gohy, J.-F.; Schubert, U.S. A schizophrenic gradient copolymer: switching and reversing poly(2-oxazoline) micelles based on UCST and subtle solvent changes. Soft Matter 2009, 5, 3590–3592. [Google Scholar] [CrossRef]
- Shultz, A.R.; Flory, P.J. Phase equilibria in polymer-solvent systems. III. Three-component systems. J. Am. Chem. Soc. 1953, 75, 5681–5685. [Google Scholar] [CrossRef]
- Wolf, B.A.; Blaum, G. Measured and calculated solubility of polymers in mixed solvents: Monotony and cosolvency. J. Polym. Sci. B Polym. Phys. 1975, 13, 1115–1132. [Google Scholar] [CrossRef]
- Frank, H.S.; Evans, M.W. Free volume and entropy in condensed systems iii. Entropy in binary liquid mixtures; partial molal entropy in dilute solutions; structure and thermodynamics in aqueous electrolytes. J. Chem. Phys. 1945, 13, 507–532. [Google Scholar] [CrossRef]
- Franks, F.; Ives, D.J.G. The structural properties of alcohol-water mixtures. Q. Rev. Chem. Soc. 1966, 20, 1–44. [Google Scholar] [CrossRef]
- Noskov, S.Y.; Lamoureux, G.; Roux, B. Molecular dynamics study of hydration in ethanol-water mixtures using a polarizable force field. J. Phys. Chem. B 2005, 109, 6705–6713. [Google Scholar] [CrossRef] [PubMed]
- Escalera, J.B.; Bustamante, P.; Martin, A. Predicting the solubility of drugs in solvent mixtures: Multiple solubility maxima and the chameleonic effect. J. Pharm. Pharmacol. 1994, 46, 172–175. [Google Scholar] [CrossRef] [PubMed]
- Bustamante, P.; Navarro, J.; Romero, S.; Escalera, B. Thermodynamic origin of the solubility profile of drugs showing one or two maxima against the polarity of aqueous and nonaqueous mixtures: niflumic acid and caffeine. J. Pharm. Sci. 2002, 91, 874–883. [Google Scholar] [CrossRef] [PubMed]
- Piccarolo, S.; Titomanlio, G. Synergism in the swelling and solubility of poly(methyl methacrylate) in presence of ethanol/water mixtures. Makromol. Chem. Rapid Commun. 1982, 3, 383–387. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Rogers, S.; Can, A.; Becer, C.R.; Guerrero-Sanchez, C.; Wouters, D.; Hoeppener, S.; Schubert, U.S. Self-assembly of double hydrophobic block copolymers in water-ethanol mixtures: From micelles to thermo-responsive micellar gels. Chem. Commun. 2009, 37, 5582–5584. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Fijten, M.W.M.; Thijs, H.M.L.; van Lankvelt, B.M.; Schubert, U.S. Microwave-assisted synthesis and properties of a series of poly(2 alkyl-2-oxazoline)s. Des. Monomers Polym. 2005, 8, 659–671. [Google Scholar] [CrossRef]
- Kempe, K.; Lobert, M.; Hoogenboom, R.; Schubert, U.S. Screening the synthesis of 2-substituted-2-oxazolines. J. Comb. Chem. 2009, 11, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Beom Lee, S.; Cong, S.-C.; Jin, J.-I.; Sohn, Y.S. Solvent effect on the lower critical solution temperature of biodegradable thermosensitive poly(organophosphazenes). Polym. Bull. 2000, 45, 389–396. [Google Scholar]
- Bharatiya, B.; Guo, C.; Ma, J.H.; Hassan, P.A.; Bahadur, P. Aggregation and clouding behavior of aqueous solution of EO–PO block copolymer in presence of n-alkanols. Eur. Polym. J. 2007, 43, 1883–1891. [Google Scholar] [CrossRef]
- Hoogenboom, R.; Becer, C.R.; Guerrero-Sanchez, C.; Hoeppener, S.; Schubert, U.S. Solubility and thermoresponsiveness of PMMA in alcohol-water solvent mixtures. Aust. J. Chem. 2010, 63. [Google Scholar] [CrossRef]
- Kempe, K.; Lobert, M.; Hoogenboom, R.; Schubert, U.S. Synthesis and characterization of a series of diverse poly(2-oxazoline)s. J. Polym. Sci. A Polym. Chem. 2009, 47, 3829–3838. [Google Scholar] [CrossRef]
- Birch, M.; Fussel, S.J.; Higginson, P.D.; McDowall, N.; Marziano, I. Towards a PAT-based strategy for crystallization development. Org. Process. Res. Dev. 2005, 9, 360–364. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lambermont-Thijs, H.M.L.; Kuringen, H.P.C.v.; Put, J.P.W.v.d.; Schubert, U.S.; Hoogenboom, R. Temperature Induced Solubility Transitions of Various Poly(2-oxazoline)s in Ethanol-Water Solvent Mixtures. Polymers 2010, 2, 188-199. https://doi.org/10.3390/polym2030188
Lambermont-Thijs HML, Kuringen HPCv, Put JPWvd, Schubert US, Hoogenboom R. Temperature Induced Solubility Transitions of Various Poly(2-oxazoline)s in Ethanol-Water Solvent Mixtures. Polymers. 2010; 2(3):188-199. https://doi.org/10.3390/polym2030188
Chicago/Turabian StyleLambermont-Thijs, Hanneke M. L., Huub P. C. van Kuringen, Jeroen P. W. van der Put, Ulrich S. Schubert, and Richard Hoogenboom. 2010. "Temperature Induced Solubility Transitions of Various Poly(2-oxazoline)s in Ethanol-Water Solvent Mixtures" Polymers 2, no. 3: 188-199. https://doi.org/10.3390/polym2030188
APA StyleLambermont-Thijs, H. M. L., Kuringen, H. P. C. v., Put, J. P. W. v. d., Schubert, U. S., & Hoogenboom, R. (2010). Temperature Induced Solubility Transitions of Various Poly(2-oxazoline)s in Ethanol-Water Solvent Mixtures. Polymers, 2(3), 188-199. https://doi.org/10.3390/polym2030188





