Chitosan-Based Active Packaging Films Incorporating Terminalia catappa Leaf Extract and Zinc Oxide Precursors for Sustainable Food Packaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.1.1. Chitosan
2.1.2. Other Chemicals and Reagents
2.1.3. Determination of Chitosan Molecular Weight
2.2. Microwave-Assisted Extraction and Drying of Terminalia catappa Leaf Extract
2.2.1. Preparation of Terminalia catappa Leaf Powder
2.2.2. Extraction
2.2.3. Comparative Drying Methods
2.2.4. UV–Visible Spectroscopy
2.2.5. FTIR Analysis of Terminalia catappa Leaf Extract
2.2.6. Total Phenolic Content (TPC)
2.2.7. Antioxidant Assays of Terminalia catappa Leaf Extract
DPPH Analysis
ABTS Analysis
2.3. Synthesis of ZnO by the Hydrothermal Method
2.3.1. Preparation of Hydrothermally Synthesised ZnO Powder
2.3.2. XRD Analysis of Synthesised ZnO Powder
2.3.3. Scanning Electron Microscopy (SEM)
2.3.4. FT-IR Analysis
2.3.5. Thermogravimetric Analysis (TGA)
2.4. Chitosan-Based Active Packaging Films Incorporating Terminalia catappa Leaf Extract and Zinc Oxide
2.4.1. Preparation of the Films
2.4.2. Colour Properties
2.4.3. Haze and Transmittance Properties
2.4.4. Scanning Electron Microscopy (SEM) and Energy-Dispersive X-Ray Spectroscopy (EDX)
2.4.5. XRD Analysis of Composite Films
2.4.6. Thickness and Mechanical Properties
2.4.7. ATR-FTIR Analysis of Composite Films
2.4.8. Moisture Content
2.4.9. Water Vapour Permeability (WVP) and Water Vapour Transmission Rate (WVTR)
2.4.10. Contact Angle (CA)
2.4.11. Thermal Analysis of Composite Films (DSC and TGA)
2.4.12. Disc-Diffusion Antimicrobial Assay of Composite Films
2.4.13. Antioxidant Activity of Film Extracts
2.5. Preliminary Visual Quality Assessment of Packaged Bananas
2.6. Statistical Analysis
3. Results and Discussion
3.1. Microwave-Assisted Extraction of T. catappa Leaf Extract
3.1.1. Temperature Profile, Extraction Yield, Total Phenolic Content, and Antioxidant Activities
3.1.2. UV–Vis Spectroscopy
3.1.3. FTIR Analysis
3.1.4. Selection of Preferred Extraction Conditions
3.2. Synthesis and Characterization of ZnO Nanostructures
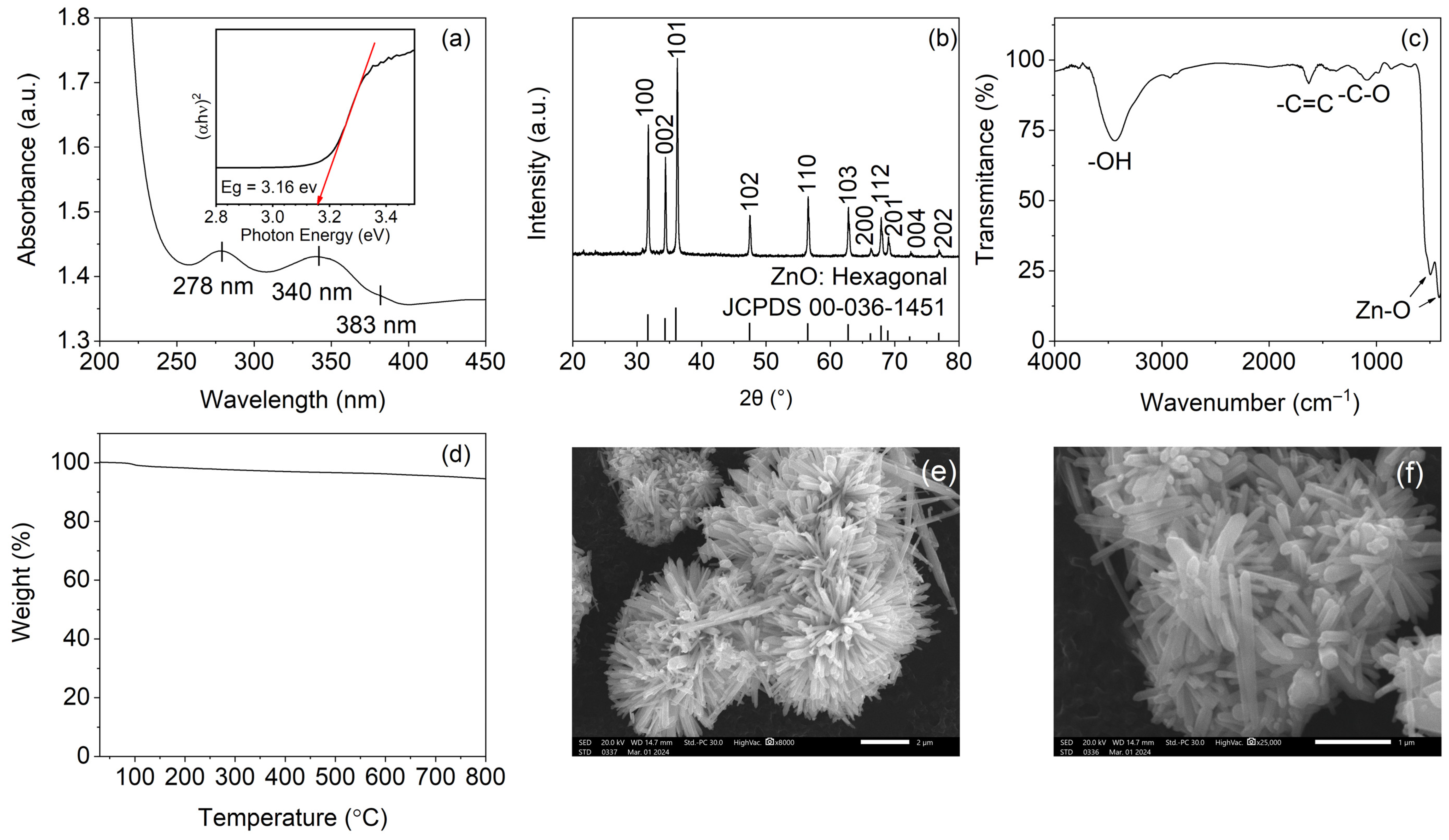
3.2.1. UV–Vis Spectroscopy and Optical Band Gap
3.2.2. X-Ray Diffraction
3.2.3. FTIR Spectroscopy
3.2.4. Thermogravimetric Analysis
3.2.5. Scanning Electron Microscopy
3.2.6. Summary of ZnO Characterization
3.3. Characterization of Chitosan-Based Active Packaging Films
3.3.1. Structural and Morphological Characterization of Films: XRD, FTIR, SEM, and EDX
X-Ray Diffraction (XRD) Analysis

Fourier-Transform Infrared (FTIR) Spectroscopy
Scanning Electron Microscopy (SEM) and Energy-Dispersive X-Ray Spectroscopy (EDX)
3.3.2. Visual Appearance and Optical Properties
3.3.3. Mechanical Properties
3.3.4. Thermal Properties of Composite Films (DSC and TGA)
3.3.5. Moisture Content, Water Vapour Barrier (WVTR, WVP), and Contact Angle
3.3.6. Antioxidant Activity of Composite Films
3.3.7. Antimicrobial Activity of Composite Films
3.3.8. Preliminary Application: Visual Assessment of Packaged Bananas
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS | 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| ANOVA | Analysis of variance |
| ATR | Attenuated total reflectance |
| CA | Contact angle |
| CFU | Colony-forming units |
| DLS | Dynamic light scattering |
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| DSC | Differential scanning calorimetry |
| EAB | Elongation at break |
| EDX | Energy-dispersive X-ray spectroscopy |
| Eg | Optical band gap |
| FC | Folin–Ciocalteu |
| FD | Freeze drying |
| FTIR | Fourier-transform infrared spectroscopy |
| FWHM | Full width at half maximum |
| GAE | Gallic acid equivalents |
| HAT | Hydrogen atom transfer |
| HSD | Honestly significant difference |
| JCPDS | Joint Committee on Powder Diffraction Standards |
| KBr | Potassium bromide |
| KOH | Potassium hydroxide |
| MAE | Microwave-assisted extraction |
| MC | Moisture content |
| MIC | Minimum inhibitory concentration |
| MW | Molecular weight |
| NA | Nutrient agar |
| NPs | Nanoparticles |
| PTFE | Polytetrafluoroethylene |
| ROS | Reactive oxygen species |
| SD | Standard deviation |
| SEM | Scanning electron microscopy |
| SET | Single electron transfer |
| TE | Terminalia catappa leaf extract |
| TEAC | Trolox equivalent antioxidant capacity |
| Tg | Glass transition temperature |
| TGA | Thermogravimetric analysis |
| Tm | Melting temperature |
| TPC | Total phenolic content |
| TS | Tensile strength |
| UV–Vis | Ultraviolet–visible spectroscopy |
| VD | Vacuum drying |
| WVP | Water vapour permeability |
| WVTR | Water vapour transmission rate |
| XRD | X-ray diffraction |
| ZnO | Zinc oxide |
References
- Mondi. Why One-Third of Food Is Wasted and How Packaging Can Help. Available online: https://www.mondigroup.com/news-and-insight/2025/why-does-one-third-of-the-worlds-food-never-make-it-to-the-table-and-how-can-packaging-help/ (accessed on 22 September 2025).
- Cheng, J.; Gao, R.; Zhu, Y.; Lin, Q. Applications of biodegradable materials in food packaging: A review. Alex. Eng. J. 2024, 91, 70–83. [Google Scholar] [CrossRef]
- Donkor, L.; Kontoh, G.; Yaya, A.; Bediako, J.K.; Apalangya, V. Bio-based and sustainable food packaging systems: Relevance, challenges, and prospects. Appl. Food Res. 2023, 3, 100356. [Google Scholar] [CrossRef]
- Dutta, D.; Sit, N. Application of natural extracts as active ingredient in biopolymer-based packaging systems. J. Food Sci. Technol. 2023, 60, 1888–1902. [Google Scholar] [CrossRef] [PubMed]
- Versino, F.; Ortega, F.; Monroy, Y.; Rivero, S.; López, O.V.; García, M.A. Sustainable and bio-based food packaging: A review on past and current design innovations. Foods 2023, 12, 1057. [Google Scholar] [CrossRef]
- Jogaiah, S.; Mujtaba, A.G.; Mujtaba, M.; Archana; De Britto, S.; Geetha, N.; Belorkar, S.A.; Shetty, H.S. Chitosan-metal and metal oxide nanocomposites for active and intelligent food packaging: A comprehensive review of emerging trends and associated challenges. Carbohydr. Polym. 2025, 357, 123459. [Google Scholar] [CrossRef]
- Liu, T.; Li, J.; Tang, Q.; Qiu, P.; Gou, D.; Zhao, J. Chitosan-based materials: An overview of potential applications in food packaging. Foods 2022, 11, 1490. [Google Scholar] [CrossRef]
- Haghighi, H.; Licciardello, F.; Fava, P.; Siesler, H.W.; Pulvirenti, A. Recent advances on chitosan-based films for sustainable food packaging applications. Food Packag. Shelf Life 2020, 26, 100551. [Google Scholar] [CrossRef]
- Kumar, S.; Mukherjee, A.; Dutta, J. Chitosan-based nanocomposite films and coatings: Emerging antimicrobial food packaging alternatives. Trends Food Sci. Technol. 2020, 97, 196–209. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Varma, A.J.; Deshpande, S.V.; Kennedy, J.F. Metal complexation by chitosan and its derivatives: A review. Carbohydr. Polym. 2004, 55, 77–93. [Google Scholar] [CrossRef]
- Kola, V.; Carvalho, I.S. Plant extracts as additives in biodegradable films and coatings in active food packaging. Food Biosci. 2023, 54, 102860. [Google Scholar] [CrossRef]
- Cazón, P.; Mateus, A.R.; Silva, A.S. Advances in active packaging using natural biopolymers and fruit by-products for enhanced food preservation. Food Res. Int. 2025, 213, 116439. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.; Lall, A.; Kumar, S.; Patil, T.D.; Gaikwad, K.K. Plant-based edible films and coatings for food-packaging applications: Recent advances, applications, and trends. Sustain. Food Technol. 2024, 2, 1428–1455. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Yong, H.; Qin, Y.; Liu, J.; Liu, J. Development of multifunctional food packaging films based on chitosan, TiO2 nanoparticles and anthocyanin-rich black plum peel extract. Food Hydrocoll. 2019, 94, 80–92. [Google Scholar] [CrossRef]
- Takó, M.; Kerekes, E.B.; Zambrano, C.; Kotogán, A.; Papp, T.; Krisch, J.; Vágvölgyi, C. Plant phenolics and phenolic-enriched extracts as antimicrobial agents against food-contaminating microorganisms. Antioxidants 2020, 9, 165. [Google Scholar] [CrossRef]
- Chung, K.-T.; Wong, T.Y.; Wei, C.-I.; Huang, Y.-W.; Lin, Y. Tannins and human health: A review. Crit. Rev. Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef]
- Ozogul, Y.; Ucar, Y.; Tadesse, E.E.; Rathod, N.; Kulawik, P.; Trif, M.; Esatbeyoglu, T.; Ozogul, F. Tannins for food preservation and human health: A review of current knowledge. Appl. Food Res. 2025, 5, 100738. [Google Scholar] [CrossRef]
- Cosme, F.; Aires, A.; Pinto, T.; Oliveira, I.; Vilela, A.; Gonçalves, B. A comprehensive review of bioactive tannins in foods and beverages: Functional properties, health benefits, and sensory qualities. Molecules 2025, 30, 800. [Google Scholar] [CrossRef]
- Nguena-Dongue, B.-N.; Amobonye, A.; Waleguele, C.C.; Asong, S.T.; Tchuenguia, C.V.; Menkem, E.Z.; Pillai, S. In vitro antimicrobial activities and phytochemical profiling of Cananga odorata, Terminalia catappa, and Terminalia mantaly. J. Ethnopharmacol. 2025, 352, 120144. [Google Scholar] [CrossRef]
- Chanda, S.; Rakholiya, K.; Nair, R. Antimicrobial activity of Terminalia catappa L. leaf extracts against some clinically important pathogenic microbial strains. Chin. Med. 2011, 2, 171–177. [Google Scholar] [CrossRef]
- Allyn, O.Q.; Kusumawati, E.; Nugroho, R.A. Antimicrobial activity of Terminalia catappa brown leaf extracts against Staphylococcus aureus ATCC 25923 and Pseudomonas aeruginosa ATCC 27853. F1000Research 2018, 7, 1406. [Google Scholar] [CrossRef] [PubMed]
- Sani, M.A.; Azizi-Lalabadi, M.; Tavassoli, M.; Mohammadi, K.; McClements, D.J. Recent advances in the development of smart and active biodegradable packaging materials. Nanomaterials 2021, 11, 1331. [Google Scholar] [CrossRef] [PubMed]
- Kołodziejczak-Radzimska, A.; Jesionowski, T. Zinc oxide—From synthesis to application: A review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef] [PubMed]
- Gökmen, G.G.; Mirsafi, F.S.; Leißner, T.; Akan, T.; Mishra, Y.K.; Kışla, D. Zinc oxide nanomaterials: Safeguarding food quality and sustainability. Compr. Rev. Food Sci. Food Saf. 2024, 23, e70051. [Google Scholar] [CrossRef]
- Lebaka, V.R.; Ravi, P.; Reddy, M.C.; Thummala, C.; Mandal, T.K. Zinc oxide nanoparticles in modern science and technology: Multifunctional roles in healthcare, environmental remediation, and industry. Nanomaterials 2025, 15, 754. [Google Scholar] [CrossRef]
- Hwang, C.; Choi, M.-H.; Kim, H.-E.; Jeong, S.-H.; Park, J.-U. Reactive oxygen species-generating hydrogel platform for enhanced antibacterial therapy. NPG Asia Mater. 2022, 14, 72. [Google Scholar] [CrossRef]
- David, C.A.; Galceran, J.; Rey-Castro, C.; Puy, J.; Companys, E.; Salvador, J.; Marcigüe, J.; Pérez, S. Dissolution kinetics and solubility of ZnO nanoparticles followed by AGNES. J. Phys. Chem. C 2012, 116, 11758–11767. [Google Scholar] [CrossRef]
- Ejima, H.; Richardson, J.J.; Liang, K.; Best, J.P.; van Koeverden, M.P.; Such, G.K.; Cui, J.; Caruso, F. One-step assembly of coordination complexes for versatile film and particle engineering. Science 2013, 341, 154–157. [Google Scholar] [CrossRef]
- Kasaai, M.R. Calculation of Mark–Houwink–Sakurada (MHS) equation viscometric constants for chitosan in any solvent–temperature system using experimental reported viscometric constants data. Carbohydr. Polym. 2007, 68, 477–488. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Baruah, S.; Dutta, J. Hydrothermal growth of ZnO nanostructures. Sci. Technol. Adv. Mater. 2009, 10, 013001. [Google Scholar] [CrossRef] [PubMed]
- Bulcha, B.; Tesfaye, J.L.; Anatol, D.; Shanmugam, R.; Dwarampudi, L.P.; Nagaprasad, N.; Ramasamy, K.T.; Krishnaraj, R. Synthesis of zinc oxide nanoparticles by hydrothermal methods and spectroscopic investigation of ultraviolet radiation protective properties. J. Nanomater. 2021, 2021, 8617290. [Google Scholar] [CrossRef]
- Janotti, A.; Van de Walle, C.G. Fundamentals of zinc oxide as a semiconductor. Rep. Prog. Phys. 2009, 72, 126501. [Google Scholar] [CrossRef]
- Siripatrawan, U.; Harte, B.R. Physical properties and antioxidant activity of an active film from chitosan incorporated with green tea extract. Food Hydrocoll. 2010, 24, 770–775. [Google Scholar] [CrossRef]
- Kanatt, S.R.; Rao, M.S.; Chawla, S.P.; Sharma, A. Active chitosan–polyvinyl alcohol films with natural extracts. Food Hydrocoll. 2012, 29, 290–297. [Google Scholar] [CrossRef]
- Leceta, I.; Guerrero, P.; de la Caba, K. Functional properties of chitosan-based films. Carbohydr. Polym. 2013, 93, 339–346. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Negi, Y.S. Effect of varying filler concentration on zinc oxide nanoparticle loaded chitosan films. J. Polym. Environ. 2017, 25, 1087–1098. [Google Scholar] [CrossRef]
- Rhim, J.-W.; Park, H.-M.; Ha, C.-S. Bio-nanocomposites for food packaging applications. Prog. Polym. Sci. 2013, 38, 1629–1652. [Google Scholar] [CrossRef]
- Noshirvani, N.; Ghanbarzadeh, B.; Mokarram, R.R.; Hashemi, M. Novel active packaging based on carboxymethyl cellulose–chitosan–ZnO NPs nanocomposite for food preservation. Food Packag. Shelf Life 2017, 11, 106–114. [Google Scholar] [CrossRef]
- Goy, R.C.; de Britto, D.; Assis, O.B.G. A review of the antimicrobial activity of chitosan. Polímeros 2009, 19, 241–247. [Google Scholar] [CrossRef]
- Jiang, Y.; Joyce, D.C.; Macnish, A.J. Extension of the shelf life of banana fruit by 1-methylcyclopropene in combination with polyethylene bags. Postharvest Biol. Technol. 1999, 16, 187–193. [Google Scholar] [CrossRef]
- Devlieghere, F.; Vermeulen, A.; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as a coating on fruit and vegetables. Food Microbiol. 2004, 21, 703–714. [Google Scholar] [CrossRef]
- Dahmoune, F.; Nayak, B.; Moussi, K.; Remini, H.; Madani, K. Optimization of microwave-assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem. 2015, 166, 585–595. [Google Scholar] [CrossRef]
- López-Salazar, H.E.; Corrales-García, H.; Aguilar-Jiménez, C.A.; Valdivia-López, M.Á.; Núñez-Gastélum, J.A.; Anaya-Esparza, L.M.; Montalvo-González, E.; Hernández-Fuentes, A.D. Microwave-assisted extraction of functional compounds from plants: A review. BioResources 2023, 18, 6614–6654. [Google Scholar] [CrossRef]
- Ratti, C. Hot air and freeze-drying of high-value foods: A review. J. Food Eng. 2001, 49, 311–319. [Google Scholar] [CrossRef]
- Shofian, N.M.; Hamid, A.A.; Osman, A.; Saari, N.; Anwar, F.; Dek, M.S.P.; Hairuddin, M.R. Effect of freeze-drying on the antioxidant compounds and antioxidant activity of selected tropical fruits. Int. J. Mol. Sci. 2011, 12, 4678–4692. [Google Scholar] [CrossRef]
- Floegel, A.; Kim, D.-O.; Chung, S.-J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J. Food Compos. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Chyau, C.-C.; Ko, P.-T.; Mau, J.-L. Antioxidant properties of aqueous extracts from Terminalia catappa leaves. LWT Food Sci. Technol. 2006, 39, 1099–1108. [Google Scholar] [CrossRef]
- Kinoshita, S.; Inoue, Y.; Nakama, S.; Ichiba, T.; Aniya, Y. Antioxidant and hepatoprotective actions of medicinal herb, Terminalia catappa L. from Okinawa Island and its tannin corilagin. Phytomedicine 2007, 14, 755–762. [Google Scholar] [CrossRef]
- Terças, A.G.; Monteiro, A.S.; Moffa, E.B.; dos Santos, J.R.A.; de Sousa, E.M.; Pinto, A.R.B.; da Silva Costa, P.C.; Borges, A.C.R.; Torres, L.M.B.; Barros, A.K.D.; et al. Phytochemical characterization of Terminalia catappa Linn. extracts and their antifungal activities against Candida spp. Front. Microbiol. 2017, 8, 595. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Webster, F.X.; Kiemle, D.J. Spectrometric Identification of Organic Compounds, 7th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Djurišić, A.B.; Leung, Y.H. Optical properties of ZnO nanostructures. Small 2006, 2, 944–961. [Google Scholar] [CrossRef]
- Wang, Z.L. Zinc oxide nanostructures: Growth, properties and applications. J. Phys. Condens. Matter 2004, 16, R829–R858. [Google Scholar] [CrossRef]
- Polsongkram, D.; Chamninok, P.; Pukird, S.; Chow, L.; Lupan, O.; Chai, G.; Khallaf, H.; Park, S.; Schulte, A. Effect of synthesis conditions on the growth of ZnO nanorods via hydrothermal method. Phys. B Condens. Matter 2008, 403, 3713–3717. [Google Scholar] [CrossRef]
- Hong, R.; Pan, T.; Qian, J.; Li, H. Synthesis and surface modification of ZnO nanoparticles. Chem. Eng. J. 2006, 119, 71–81. [Google Scholar] [CrossRef]
- Borah, P.; Baishya, H.; Mukherjee, A.; Mitra, A.; Dutta, J.; Kumar, S. Plant-mediated synthesis of zinc oxide nanoparticles and their applications in biopolymer-based sustainable food packaging: A state-of-the-art review. Trends Food Sci. Technol. 2025, 162, 105101. [Google Scholar] [CrossRef]
- Karuppan Perumal, M.K.; Rajasekaran, M.B.S.; Rajan Renuka, R.; Samrot, A.V.; Nagarajan, M. Zinc oxide nanoparticles and their nanocomposites as an imperative coating for smart food packaging. Appl. Food Res. 2025, 5, 100849. [Google Scholar] [CrossRef]
- Moezzi, A.; McDonagh, A.M.; Cortie, M.B. Zinc oxide particles: Synthesis, properties and applications. Chem. Eng. J. 2012, 185–186, 1–22. [Google Scholar] [CrossRef]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on zinc oxide nanoparticles: Antibacterial activity and toxicity mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef]
- Zare, M.; Namratha, K.; Ilyas, S.; Sultana, A.; Hezam, A.; Sunil, L.; Surmeneva, M.A.; Surmenev, R.A.; Nayan, M.B.; Ramakrishna, S.; et al. Emerging trends for ZnO nanoparticles and their applications in food packaging. ACS Food Sci. Technol. 2022, 2, 763–781. [Google Scholar] [CrossRef]
- Jiang, A.; Patel, R.; Padhan, B.; Palimkar, S.; Galgali, P.; Adhikari, A.; Varga, I.; Patel, M. Chitosan-based biodegradable composite for antibacterial food packaging application. Polymers 2023, 15, 2235. [Google Scholar] [CrossRef] [PubMed]
- Wahab, R.; Ansari, S.G.; Kim, Y.S.; Seo, H.K.; Kim, G.S.; Khang, G.; Shin, H.-S. Low temperature solution synthesis and characterization of ZnO nano-flowers. Mater. Res. Bull. 2007, 42, 1640–1648. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dobretsov, S.; Dutta, J. Chitosan–zinc oxide nanoparticle composite coating for active food packaging applications. Innov. Food Sci. Emerg. Technol. 2016, 38, 231–237. [Google Scholar] [CrossRef]
- Yong, H.; Liu, J. Active packaging films and edible coatings based on polyphenol-rich propolis extract: A review. Compr. Rev. Food Sci. Food Saf. 2021, 20, 2106–2145. [Google Scholar] [CrossRef]
- Siripatrawan, U.; Vitchayakitti, W. Improving functional properties of chitosan films as active food packaging by incorporating with propolis. Food Hydrocoll. 2016, 61, 695–702. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Alves, M.M.; Santos, C.F.; Ribeiro, I.A.C.; Rodrigues, C.; Coelhoso, I.; Fernando, A.L. Biodegradable Chitosan Films with ZnO Nanoparticles Synthesized Using Food Industry By-Products—Production and Characterization. Coatings 2021, 11, 646. [Google Scholar] [CrossRef]
- McDonald, M.; Mila, I.; Scalbert, A. Precipitation of metal ions by plant polyphenols: Optimal conditions and origin of precipitation. J. Agric. Food Chem. 1996, 44, 599–606. [Google Scholar] [CrossRef]
- Wang, H.; Qian, J.; Ding, F. Emerging chitosan-based films for food packaging applications. J. Agric. Food Chem. 2018, 66, 395–413. [Google Scholar] [CrossRef]
- Lawrie, G.; Keen, I.; Drew, B.; Chandler-Temple, A.; Rintoul, L.; Fredericks, P.; Grøndahl, L. Interactions between alginate and chitosan biopolymers characterized using FTIR and XPS. Biomacromolecules 2007, 8, 2533–2541. [Google Scholar] [CrossRef]
- Li, L.-H.; Deng, J.-C.; Deng, H.-R.; Liu, Z.-L.; Xin, L. Synthesis and characterization of chitosan/ZnO nanoparticle composite membranes. Carbohydr. Res. 2010, 345, 994–998. [Google Scholar] [CrossRef]
- Ab Rahman, M.A.A.; Sy Mohamad, S.F.; Mohamad, S. Development and characterization of bio-composite films made from bacterial cellulose derived from oil palm frond juice fermentation, chitosan, and glycerol. Trends Sci. 2023, 20, 4919. [Google Scholar] [CrossRef]
- Yong, H.; Wang, X.; Bai, R.; Miao, Z.; Zhang, X.; Liu, J. Development of antioxidant and intelligent pH-sensing packaging films by incorporating purple-fleshed sweet potato extract into chitosan matrix. Food Hydrocoll. 2019, 90, 216–224. [Google Scholar] [CrossRef]
- Liu, J.; Huang, J.; Hu, Z.; Li, G.; Hu, L.; Chen, X.; Hu, Y. Chitosan-based films with antioxidant of bamboo leaves and ZnO nanoparticles for application in active food packaging. Int. J. Biol. Macromol. 2021, 189, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Rahman, P.M.; Mujeeb, V.M.A.; Muraleedharan, K. Flexible chitosan-nano ZnO antimicrobial pouches as a new material for extending the shelf life of raw meat. Int. J. Biol. Macromol. 2017, 97, 382–391. [Google Scholar] [CrossRef]
- Shankar, S.; Teng, X.; Li, G.; Rhim, J.-W. Preparation, characterization, and antimicrobial activity of gelatin/ZnO nanocomposite films. Food Hydrocoll. 2015, 45, 264–271. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Soares, N.F.F.; Coimbra, J.S.R.; de Andrade, N.J.; Cruz, R.S.; Medeiros, E.A.A. Zinc oxide nanoparticles: Synthesis, antimicrobial activity and food packaging applications. Food Bioprocess Technol. 2012, 5, 1447–1464. [Google Scholar] [CrossRef]
- Padmavathy, N.; Vijayaraghavan, R. Enhanced bioactivity of ZnO nanoparticles—An antimicrobial study. Sci. Technol. Adv. Mater. 2008, 9, 035004. [Google Scholar] [CrossRef]
- Li, X.; Li, W.; Jiang, Y.; Ding, Y.; Yun, J.; Tang, Y.; Zhang, P. Effect of nano-ZnO-coated active packaging on quality of fresh-cut “Fuji” apple. Int. J. Food Sci. Technol. 2011, 46, 1947–1955. [Google Scholar] [CrossRef]
- Raghupathi, K.R.; Koodali, R.T.; Manna, A.C. Size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir 2011, 27, 4020–4028. [Google Scholar] [CrossRef]
- Sanuja, S.; Agalya, A.; Umapathy, M.J. Synthesis and characterization of zinc oxide–neem oil–chitosan bionanocomposite for food packaging application. Int. J. Biol. Macromol. 2015, 74, 76–84. [Google Scholar] [CrossRef]
- Yan, D.; Li, Y.; Liu, Y.; Li, N.; Zhang, X.; Yan, C. Antimicrobial properties of chitosan and chitosan derivatives in the treatment of enteric infections. Molecules 2021, 26, 7136. [Google Scholar] [CrossRef]
- EFSA Panel on Food Contact Materials, Enzymes, Flavourings and Processing Aids (CEF). Safety Assessment of the Substance Zinc Oxide, Nanoparticles, for Use in Food Contact Materials. EFSA J. 2016, 14, 4408. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EU) No 10/2011 of 14 January 2011 on plastic materials and articles intended to come into contact with food. Off. J. Eur. Union 2011, 12, 1–89. [Google Scholar]






| Sample ID | Zn | Zn | Sample ID | Zn | Zn | Sample ID | Zn | Zn | Sample ID | Zn | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|
| (wt%) | (at%) | (wt%) | (at%) | (wt%) | (at%) | (wt%) | (at%) | ||||
| T0Z0 | n.d. | n.d. | T1Z0 | n.d. | n.d. | T2Z0 | n.d. | n.d. | T3Z0 | n.d. | n.d. |
| T0Z1 | 6.37 | 1.42 | T1Z1 | 5.32 | 1.17 | T2Z1 | 4.28 | 0.94 | T3Z1 | 5.16 | 1.15 |
| T0Z2 | 11.13 | 2.58 | T1Z2 | 9.91 | 2.28 | T2Z2 | 11.44 | 2.67 | T3Z2 | 9.52 | 2.20 |
| T0Z3 | 15.08 | 3.62 | T1Z3 | 16.26 | 3.95 | T2Z3 | 14.74 | 3.56 | T3Z3 | 14.79 | 3.57 |
| Sample ID | L* | a* | b* | ΔE* | Haze (%) | Transmittance (%) | Thickness (mm) | Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|---|---|---|---|---|---|---|
| T0Z0 | 83.3 ± 0.6 b | −0.30 ± 0.01 e | 26 ± 2 c | Standard | 54.7 ± 1.8 f | 86.9 ± 0.8 a | 0.072 ± 0.013 i | 6.0 ± 0.5 f | 21.4 ± 0.9 cdef |
| T1Z0 | 58 ± 2 c | 16 ± 2 c | 46 ± 2 a | 35.7 ± 1.7 e | 62.2 ± 1.2 d | 58 ± 4 b | 0.080 ± 0.012 hi | 5.4 ± 1.6 f | 29.1 ± 0.8 bc |
| T2Z0 | 48 ± 2 d | 19.9 ± 0.6 a | 35 ± 2 b | 41.1 ± 1.5 d | 71.1 ± 1.6 c | 43 ± 4 de | 0.088 ± 0.010 fgh | 8.7 ± 1.4 f | 29 ± 3 bc |
| T3Z0 | 43 ± 2 fg | 19.4 ± 0.3 a | 26 ± 4 c | 45 ± 2 bc | 78.6 ± 0.3 b | 34 ± 2 g | 0.100 ± 0.014 def | 5.8 ± 0.8 f | 22 ± 4 cde |
| T0Z1 | 86.8 ± 0.1 a | −1.0 ± 0.1 e | 12.0 ± 0.4 d | 16.2 ± 1.8 g | 42.0 ± 0.6 g | 88.9 ± 0.4 a | 0.082 ± 0.010 hi | 15.7 ± 1.9 e | 28.5 ± 1.8 bc |
| T0Z2 | 87.5 ± 0.2 a | −0.8 ± 0.1 e | 7.9 ± 0.8 e | 19.3 ± 0.8 f | 41.5 ± 1.8 g | 89.7 ± 0.1 a | 0.087 ± 0.015 gh | 27 ± 3 cd | 33 ± 5 bcd |
| T0Z3 | 85.3 ± 0.2 ab | 0.3 ± 0.1 e | 14.3 ± 0.3 d | 12.7 ± 0.3 h | 29 ± 2 h | 88.9 ± 0.4 a | 0.10 ± 0.02 efg | 36 ± 4 a | 24 ± 4 cde |
| T1Z1 | 58.2 ± 1.1 c | 15.5 ± 0.7 cd | 47.8 ± 0.8 a | 36.3 ± 0.5 e | 58.1 ± 0.6 e | 57.4 ± 1.4 b | 0.086 ± 0.010 gh | 18 ± 3 e | 37 ± 4 a |
| T1Z2 | 58.3 ± 1.0 c | 14.3 ± 0.6 d | 45.7 ± 0.3 a | 34.5 ± 1.0 e | 58.8 ± 0.9 e | 53 ± 2 c | 0.090 ± 0.011 fgh | 29.4 ± 1.2 bc | 32 ± 3 ab |
| T1Z3 | 56.8 ± 0.9 c | 14.3 ± 0.6 d | 44.3 ± 0.1 a | 34.9 ± 0.9 e | 59.5 ± 1.1 e | 52.9 ± 0.7 c | 0.092 ± 0.016 fgh | 33 ± 4 ab | 16 ± 2 efg |
| T2Z1 | 46.1 ± 1.4 de | 19.5 ± 0.8 a | 32 ± 4 b | 42 ± 2 cd | 72.4 ± 0.9 c | 44.3 ± 1.2 d | 0.106 ± 0.015 cde | 14.8 ± 1.7 e | 28 ± 4 bcd |
| T2Z2 | 44 ± 3 ef | 19.4 ± 0.1 a | 31.2 ± 1.5 b | 42.5 ± 0.8 cd | 71.9 ± 0.7 c | 40 ± 2 ef | 0.107 ± 0.012 cde | 23.9 ± 1.1 d | 20 ± 3 def |
| T2Z3 | 42.8 ± 1.4 efg | 17.6 ± 0.8 b | 26 ± 2 c | 44.2 ± 1.0 bc | 71.5 ± 0.4 c | 38.4 ± 1.4 f | 0.114 ± 0.010 bc | 30 ± 3 bc | 14 ± 4 fgh |
| T3Z1 | 40.2 ± 1.4 g | 17.8 ± 0.1 b | 23 ± 2 c | 46.8 ± 1.5 b | 78.3 ± 0.3 b | 32.9 ± 1.5 g | 0.136 ± 0.011 a | 14 ± 2 e | 23 ± 4 cde |
| T3Z2 | 34.4 ± 0.3 h | 16.8 ± 0.2 bc | 13.5 ± 0.5 d | 53.3 ± 0.3 a | 79.6 ± 0.8 ab | 31 ± 3 g | 0.111 ± 0.015 cd | 27 ± 2 cd | 10 ± 5 gh |
| T3Z3 | 36.2 ± 1.1 h | 16.0 ± 0.1 c | 16.2 ± 1.4 d | 50.8 ± 1.3 a | 81.5 ± 0.7 a | 21.5 ± 1.5 h | 0.123 ± 0.015 b | 27 ± 4 cd | 7 ± 2 h |
| Sample ID | DSC | TGA | ||
|---|---|---|---|---|
| Tg (°C) | Tm (°C) | Weight Loss (%) | Residue (%) | |
| T0Z0 | 74.5 | 146.6 | 82.3 | 17.7 |
| T1Z0 | 75.5 | 166.2 | 84.3 | 15.7 |
| T2Z0 | 78.0 | 163.3 | 85.0 | 15.0 |
| T3Z0 | 78.2 | 164.6 | 78.5 | 21.5 |
| T0Z1 | 72.4 | 155.7 | 76.9 | 23.1 |
| T0Z2 | 83.0 | 153.1 | 73.6 | 26.4 |
| T0Z3 | 103.3 | 156.9 | 73.0 | 27.0 |
| T1Z1 | 75.9 | 153.1 | 77.8 | 22.2 |
| T1Z2 | 100.0 | 159.1 | 72.6 | 27.4 |
| T1Z3 | 110.7 | 153.8 | 71.0 | 29.0 |
| T2Z1 | 105.6 | 156.4 | 77.3 | 22.7 |
| T2Z2 | 111.1 | 159.3 | 70.5 | 29.5 |
| T2Z3 | 117.9 | 151.3 | 68.3 | 31.8 |
| T3Z1 | 91.0 | 159.9 | 73.0 | 27.0 |
| T3Z2 | 111.7 | 165.8 | 70.4 | 29.6 |
| T3Z3 | 122.9 | 166.4 | 68.9 | 31.2 |
| Sample ID | Moisture Content (%) | WVTR (g/(m2·h)) | WVP (×10−8 g·cm/(cm2·day·Pa)) | Contact Angle (°) |
|---|---|---|---|---|
| T0Z0 | 19.8 ± 0.6 e | 2.16 ± 0.07 a | 1.41 ± 0.03 ab | 92 ± 9 a |
| T1Z0 | 18.6 ± 0.5 de | 2.08 ± 0.09 ab | 1.48 ± 0.07 ab | 88 ± 5 a |
| T2Z0 | 18.6 ± 1.2 de | 2.09 ± 0.08 ab | 1.45 ± 0.09 ab | 80 ± 4 a |
| T3Z0 | 17.7 ± 0.7 d | 2.08 ± 0.09 ab | 1.70 ± 0.08 a | 71 ± 8 a |
| T0Z1 | 15.54 ± 0.14 c | 2.05 ± 0.13 ab | 1.12 ± 0.07 cde | 79 ± 9 a |
| T0Z2 | 13.3 ± 0.3 ab | 2.04 ± 0.03 ab | 1.218 ± 0.004 bcde | 89 ± 9 a |
| T0Z3 | 12.7 ± 0.3 ab | 1.81 ± 0.18 ab | 1.13 ± 0.12 cde | 85 ± 3 a |
| T1Z1 | 14.6 ± 0.9 bc | 2.04 ± 0.14 ab | 1.20 ± 0.15 bcde | 87 ± 3 a |
| T1Z2 | 13.4 ± 0.4 ab | 2.0 ± 0.2 ab | 1.01 ± 0.12 e | 86.0 ± 1.7 a |
| T1Z3 | 13.1 ± 0.2 ab | 2.02 ± 0.17 ab | 1.32 ± 0.06 bcd | 94 ± 3 a |
| T2Z1 | 14.2 ± 1.2 bc | 2.00 ± 0.17 ab | 1.11 ± 0.15 de | 70 ± 9 a |
| T2Z2 | 13.4 ± 0.5 ab | 1.98 ± 0.18 ab | 1.4 ± 0.2 ab | 89 ± 4 a |
| T2Z3 | 12.2 ± 0.3 a | 1.8 ± 0.3 ab | 1.4 ± 0.2 bc | 79 ± 2 a |
| T3Z1 | 15.33 ± 0.17 c | 2.0 ± 0.2 ab | 1.44 ± 0.11 ab | 70 ± 9 a |
| T3Z2 | 13.3 ± 0.2 ab | 1.97 ± 0.14 ab | 1.5 ± 0.1 ab | 78 ± 10 a |
| T3Z3 | 12.0 ± 0.7 a | 1.7 ± 0.3 b | 1.48 ± 0.09 ab | 88 ± 7 a |
| Sample ID | Inhibition Zone (mm) | |
|---|---|---|
| E. coli (Gram-Negative) | S. aureus (Gram-Positive) | |
| T0Z0 | 0 e | 0 d |
| T1Z0 | 0 e | 0 d |
| T2Z0 | 0 e | 0 d |
| T3Z0 | 0 e | 0 d |
| T0Z1 | 11.3 ± 1.6 d | 0 d |
| T0Z2 | 19.3 ± 1.8 ab | 16.8 ± 1.0 b |
| T0Z3 | 22 ± 3 a | 21 ± 2 a |
| T1Z1 | 14.6 ± 1.2 cd | 0 d |
| T1Z2 | 20 ± 2 ab | 17.3 ± 0.9 b |
| T1Z3 | 23 ± 2 a | 20.6 ± 0.1 a |
| T2Z1 | 14.2 ± 0.8 cd | 0 d |
| T2Z2 | 18 ± 3 bc | 15.5 ± 1.0 bc |
| T2Z3 | 20 ± 2 ab | 19.9 ± 1.4 a |
| T3Z1 | 0 e | 0 d |
| T3Z2 | 17 ± 3 bc | 14 ± 2 c |
| T3Z3 | 20.8 ± 1.7 ab | 19.5 ± 0.4 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Thongchai, P.; Wannapasit, P.; Teerasirida, K. Chitosan-Based Active Packaging Films Incorporating Terminalia catappa Leaf Extract and Zinc Oxide Precursors for Sustainable Food Packaging. Polymers 2026, 18, 928. https://doi.org/10.3390/polym18080928
Thongchai P, Wannapasit P, Teerasirida K. Chitosan-Based Active Packaging Films Incorporating Terminalia catappa Leaf Extract and Zinc Oxide Precursors for Sustainable Food Packaging. Polymers. 2026; 18(8):928. https://doi.org/10.3390/polym18080928
Chicago/Turabian StyleThongchai, Prem, Paitoon Wannapasit, and Kulyada Teerasirida. 2026. "Chitosan-Based Active Packaging Films Incorporating Terminalia catappa Leaf Extract and Zinc Oxide Precursors for Sustainable Food Packaging" Polymers 18, no. 8: 928. https://doi.org/10.3390/polym18080928
APA StyleThongchai, P., Wannapasit, P., & Teerasirida, K. (2026). Chitosan-Based Active Packaging Films Incorporating Terminalia catappa Leaf Extract and Zinc Oxide Precursors for Sustainable Food Packaging. Polymers, 18(8), 928. https://doi.org/10.3390/polym18080928







