Novel Polyarylene Ether Nitrile/BaTiO3-Decorated WS2 Nanohybrid Crosslinked Nanocomposites for Thermal-Stable Dielectrics
Abstract
1. Introduction
2. Experimental Methods
2.1. Materials
2.2. Preparation of WS2@BT Nanoparticles
2.3. Preparation of PEN/WS2@BT Nanocomposite Films
2.4. Characterization
3. Results and Discussion
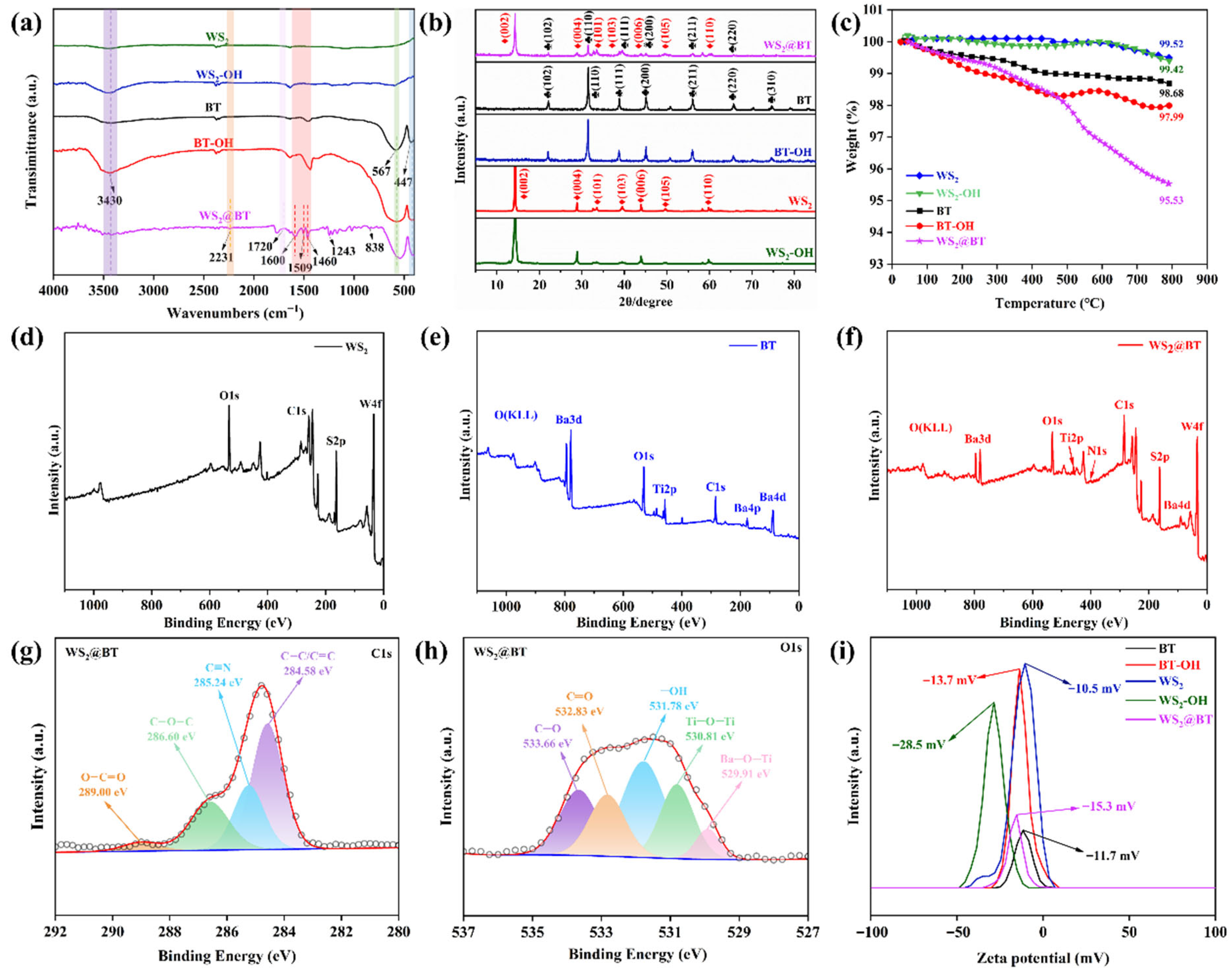
3.1. Characterization of WS2@BT Nanofilles
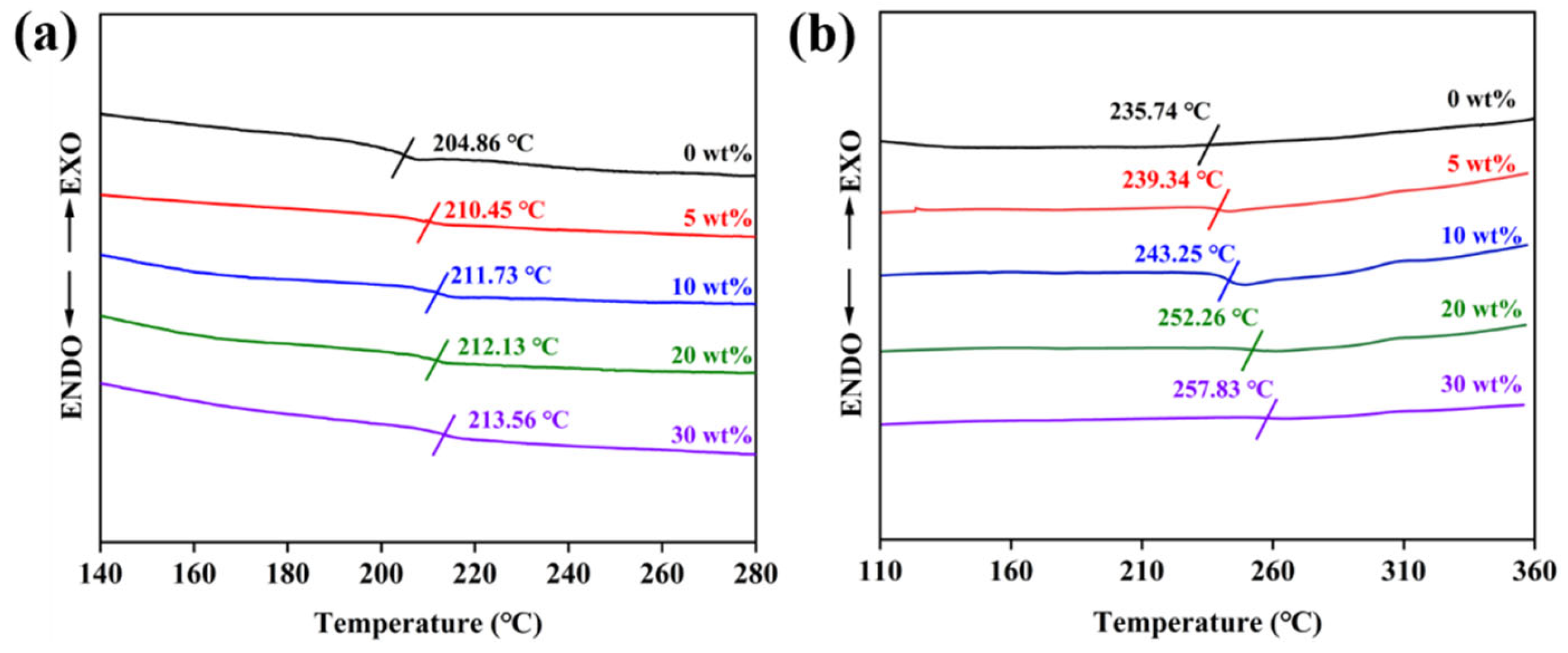
3.2. The Properties of Nanocomposites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Yan, J.H.; Liu, X.Y.; Ma, C.R.; Huang, Y.C.; Yang, G.W. All-dielectric materials and related nanophotonic applications. Mater. Sci. Eng. R Rep. 2020, 141, 100563. [Google Scholar] [CrossRef]
- Wang, G.; Lu, Z.L.; Li, Y.; Li, L.H.; Ji, H.F.; Feteira, A.; Zhou, D.; Wang, D.W.; Zhang, S.J.; Reaney, I.M. Electroceramics for high-energy density capacitors: Current status and future perspectives. Chem. Rev. 2021, 121, 6124–6172. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.C.; Zhou, T.T.; Zhao, Z.Y.; Wu, X.F.; Wei, P.; Yu, G.; Wang, H.; Pang, L.F. Significantly enhanced dielectric polarization and breakdown strength of polyvinylidene fluoride via grafting with sulfobetaine methacrylate. Macromolecules 2025, 58, 13022–13032. [Google Scholar] [CrossRef]
- Yuan, Q.B.; Wang, Y.; Zhang, Z.; Duan, M.Y.; Li, X.; Yang, H.; Yao, B.; Wang, Y.F. Polymer-based nanocomposites with one-dimensional nanofillers for dielectric energy storage at high temperatures. ACS Appl. Polym. Mater. 2024, 6, 3678–3689. [Google Scholar] [CrossRef]
- Yang, M.Z.; Guo, M.F.; Xu, E.X.; Ren, W.B.; Wang, D.; Li, S.; Zhang, S.J.; Nan, C.W.; Shen, Y. Polymer nanocomposite dielectrics for capacitive energy storage. Nat. Nanotechnol. 2024, 19, 45–53. [Google Scholar] [CrossRef]
- Guo, R.; Luo, H.; Yan, M.; Zhou, X.; Zhou, K.; Zhang, D. Significantly enhanced breakdown strength and energy density in sandwich-structured nanocomposites with low-level BaTiO3 nanowires. Nano Energy 2020, 75, 105412. [Google Scholar] [CrossRef]
- Hu, X.; Zhang, H.; Wu, D.; Yin, D.; Zhu, N.; Guo, K.; Lu, C. PVDF-based matrix with covalent bonded BaTiO3 nanowires enabled ultrahigh energy density and dielectric properties. Chem. Eng. J. 2023, 451, 138391. [Google Scholar] [CrossRef]
- Tsyganov, A.; Vikulova, M.; Zotov, I.; Burmistrov, I.; Gorokhovsky, A.; Gorshkov, N. Synergistic effect of CaCu3Ti4O12 ceramic and Ti3C2Tx MXene nanoflakes on the dielectric properties of poly(vinylidene fluoride) composites. Mater. Chem. Phys. 2023, 303, 128714. [Google Scholar] [CrossRef]
- Singh, M.; Apata, I.E.; Samant, S.; Wu, W.; Tawade, B.V.; Pradhan, N.; Raghavan, D.; Karim, A. Nanoscale strategies to enhance the energy storage capacity of polymeric dielectric capacitors: Review of recent advances. Polym. Rev. 2021, 61, 295–334. [Google Scholar] [CrossRef]
- Chen, S.Y.; Yang, S.; Zuo, F.; Wang, P.; You, Y. Novel three-dimensional wintersweet-like BaTiO3-decorated halloysite nanotube nanohybrids/polyarylene ether nitrile nanocomposites for flexible thermal-resistant dielectrics. Appl. Surf. Sci. 2024, 656, 159690. [Google Scholar] [CrossRef]
- Singh, M.; Tiwary, S.K.; Karim, A. Sub-nano fillers for high-temperature storage. Nat. Energy 2024, 9, 157–165. [Google Scholar] [CrossRef]
- Chen, S.Y.; Yang, S.; Chen, S.S.; Zuo, F.; You, Y. Polyarylene ether nitrile dielectric films modified by HNTs@PDA hybrids for high-temperature resistant organic electronics field. Nanotechnol. Rev. 2023, 12, 20230117. [Google Scholar] [CrossRef]
- Sang, X.; Li, X.; Zhang, D.; Zhang, X.; Wang, H.; Li, S. Improved dielectric properties and energy-storage densities of BaTiO3-Doped PVDF composites by heat treatment and surface modification of BaTiO3. J. Phys. D Appl. Phys. 2022, 55, 215501. [Google Scholar] [CrossRef]
- Qiao, J.; Liu, Z.; Mu, H.; Liu, C. Effect of high-entropy spinel ferrite (Mn0.2Zr0.2Cu0.2Ca0.2Ni0.2)Fe2O4 doping concentration on the ferroelectric properties of PVDF-based polymers. Polymers 2023, 15, 2688. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Xue, W.; Yang, Y.; Li, Y.; Wang, S.; Zhan, Y.; Li, W.; Hao, J.; Zha, J.-W.; Liu, C. PMMA brush-modified graphene for flexible energy storage PVDF dielectric films. Compos. Commun. 2023, 37, 101411. [Google Scholar] [CrossRef]
- Zhang, T.D.; Sun, H.; Yin, C.; Jung, Y.H.; Min, S.; Zhang, Y.; Zhang, C.; Chen, Q.; Lee, K.J.; Chi, Q. Recent progress in polymer dielectric energy storage: From film fabrication and modification to capacitor performance and application. Prog. Mater. Sci. 2023, 146, 101207. [Google Scholar] [CrossRef]
- Likhi, F.H.; Singh, M.; Potdukhe, H.R.; Ajayan, P.M.; Rahman, M.M.; Karim, A. Tuning dielectric properties with nanofiller dimensionality in polymer nanocomposites. ACS Appl. Mater. Interfaces 2024, 16, 34567–34577. [Google Scholar] [CrossRef]
- Zhou, T.; Zha, J.W.; Cui, R.Y.; Fan, B.H.; Yuan, J.K.; Dang, Z.M. Improving dielectric properties of BaTiO3/ferroelectric polymer composites by employing surface hydroxylated BaTiO3 nanoparticles. ACS Appl. Mater. Interfaces 2011, 3, 2184–2188. [Google Scholar] [CrossRef]
- Luo, H.; Zhou, X.; Ellingford, C.; Zhang, Y.; Chen, S.; Zhou, K.; Zhang, D.; Bowen, C.R.; Wan, C. Interface design for high energy density polymer nanocomposites. Chem. Soc. Rev. 2019, 48, 4424–4465. [Google Scholar] [CrossRef]
- Liu, X.D.; Li, L.; Wang, M.; Yang, Y.; You, Y.; Yang, X.; Wang, P. In-situ polymerization of PANI nanocone array on PEN nanofibrous membranes for solar-driven interfacial evaporation. Sep. Purif. Technol. 2024, 344, 127109. [Google Scholar] [CrossRef]
- You, Y.; Liu, S.N.; Tu, L.; Wang, Y.J.; Zhan, C.H.; Du, X.Y.; Wei, R.B.; Liu, X.B. Controllable fabrication of poly(arylene ether nitrile) dielectrics for thermal-resistant film capacitors. Macromolecules 2019, 52, 5850–5859. [Google Scholar] [CrossRef]
- Chen, S.S.; Chen, S.Y.; Yang, S.; Wang, P.; You, Y. Core-shell structure polydopamine and polyethylenimine co-assisted MoS2-BaTiO3/polyarylene ether nitrile nanocomposites for high-temperature resistant organic dielectrics. J. Alloys Compd. 2024, 977, 173336. [Google Scholar] [CrossRef]
- Liu, S.N.; Chen, Y.N.; Wu, J.Q.; Zhang, T.; Tong, L.F.; Liu, X.B. Covalent interface and crosslinked network engineering enable polyarylene ether nitrile nanocomposites with enhanced thermal conductivity and high-temperature energy storage. Chem. Eng. J. 2025, 518, 164719. [Google Scholar] [CrossRef]
- Xiao, Q.; Han, W.H.; Yang, R.Q.; You, Y.; Wei, R.B.; Liu, X.B. Mechanical, dielectric, and thermal properties of polyarylene ether nitrile and boron nitride nanosheets composites. Polym. Compos. 2018, 39, e1598–e1605. [Google Scholar] [CrossRef]
- Pant, H.; Patra, M.; Verma, A.; Vadera, S.; Kumar, N. Study of the dielectric properties of barium titanate–polymer composites. Acta Mater. 2006, 54, 3163–3169. [Google Scholar] [CrossRef]
- Bertolazzi, S.; Brivio, J.; Kis, A. Stretching and breaking of ultrathin MoS2. ACS Nano 2011, 5, 9703–9709. [Google Scholar] [CrossRef]
- Haba, D.; Brunner, A.J.; Barbezat, M.; Spetter, D.; Tremel, W.; Pinter, G. Correlation of epoxy material properties with the toughening effect of fullerene-like WS2 nanoparticles. Eur. Polym. J. 2016, 84, 125–136. [Google Scholar] [CrossRef]
- Haba, D.; Brunner, A.J.; Pinter, G. Dispersion of fullerene-like WS2 nanoparticles within epoxy and the resulting fracture mechanics. Compos. Sci. Technol. 2015, 119, 55–61. [Google Scholar] [CrossRef]
- Stefanescu, E.A.; Tan, X.L.; Lin, Z.Q.; Bowler, N.; Kessler, M.R. Multifunctional fiberglass-reinforced PMMA-BaTiO3 structural/dielectric composites. Polymer 2011, 52, 2016–2024. [Google Scholar] [CrossRef]
- Pu, Z.J.; Zheng, X.Y.; Tian, Y.H.; Hu, L.Q.; Zhong, J.C. Flexible ultrahigh-temperature polymer-based dielectrics with high permittivity for film capacitor applications. Polymers 2017, 9, 596. [Google Scholar] [CrossRef]
- He, L.; Lin, G.; Liu, X.B.; Tong, L.F. Polyarylene ether nitrile composites film with self-reinforcing effect by cross-linking and crystallization synergy. Polymer 2022, 262, 125457. [Google Scholar] [CrossRef]
- Liu, Y.; Higaki, Y.J.; Mukai, M.; Takahara, A. Molecular aggregation structure and water repellency of Poly(perfluorohexyl acrylate) with a carbamate linkage. Polymer 2019, 182, 121846. [Google Scholar] [CrossRef]
- Kassis, C.M.; Steehler, J.K.; Betts, D.E.; Guan, Z.B.; Romack, T.J.; DeSimone, J.M.; Linton, R.W. XPS studies of fluorinated acrylate polymers and block copolymers with polystyrene. Macromolecules 1996, 29, 3247–3254. [Google Scholar] [CrossRef]
- Zafeiropoulos, N.E.; Vickers, P.E.; Baillie, C.A.; Watts, J.F. An experimental investigation of modified and unmodified flax fibres with XPS, ToF-SIMS and ATR-FTIR. J. Mater. Sci. 2003, 38, 3903–3914. [Google Scholar] [CrossRef]
- Wang, G.B.; Labow, R.S.; Santerre, J.P. Probing the surface chemistry of a hydrated segmented polyurethane and a comparison with its dry surface chemical structure. Macromolecules 2000, 33, 7321–7327. [Google Scholar] [CrossRef]
- Briggs, D.; Beamson, G. Primary and secondary oxygen-induced C1s binding energy shifts in x-ray photoelectron spectroscopy of polymers. Anal. Chem. 1992, 64, 1729–1736. [Google Scholar] [CrossRef]
- Nayak, S.; Sahoo, B.; Chaki, T.K.; Khastgir, D. Facile preparation of uniform barium titanate (BaTiO3) multipods with high permittivity: Impedance and temperature dependent dielectric behavior. RSC Adv. 2014, 4, 1212–1224. [Google Scholar] [CrossRef]
- Peng, J.; Cao, T.; You, Y.; Liu, X.B.; Huang, Y.M. Fabrication of low dielectric constant fluorinated poly(arylene ether nitrile) composites by cross-linking with metal-organic frameworks. Polymer 2023, 283, 126229. [Google Scholar] [CrossRef]
- Meeporn, K.; Thongbai, P. Flexible La1.5Sr0.5NiO4/poly(vinylidene fluoride) composites with an ultra high dielectric constant: A comparative study. Compos. Part B Eng. 2020, 182, 107738. [Google Scholar] [CrossRef]
- Yu, S.Y.; Mahmood, J.; Noh, H.J.; Seo, J.M.; Jung, S.M.; Shin, S.H.; Im, Y.K.; Jeon, I.Y.; Baek, J.B. Direct synthesis of a covalent triazine-based framework from aromatic amides. Angew. Chem. Int. Ed. 2018, 57, 6518–6522. [Google Scholar] [CrossRef]
- Pu, Z.J.; Tang, H.L.; Huang, X.; Yang, J.; Zhan, Y.Q.; Zhao, R.; Liu, X.B. Effect of surface functionalization of SiO2 particles on the interfacial and mechanical properties of PEN composite films. Colloids Surf. A 2012, 415, 125–133. [Google Scholar] [CrossRef]
- Feng, M.N.; Chen, M.; Qiu, J.; He, M.; Huang, Y.M.; Lin, J. Improving dielectric properties of poly(arylene ether nitrile) composites by employing core-shell structured BaTiO3@polydopamine and MoS2@polydopamine interlinked with poly(ethylene imine) for high-temperature applications. J. Alloys Compd. 2021, 856, 158213. [Google Scholar] [CrossRef]
- Sun, W.; Lu, X.J.; Jiang, J.Y.; Zhang, X.; Hu, P.H.; Li, M.; Lin, Y.H.; Nan, C.W.; Shen, Y. Dielectric and energy storage performances of polyimide/BaTiO3 nanocomposites at elevated temperatures. J. Appl. Phys. 2017, 121, 244101. [Google Scholar] [CrossRef]
- Zuo, P.Y.; Jiang, J.H.; Chen, D.L.; Lin, J.Y.; Zhao, Z.P.; Sun, B.W.; Zhuang, Q.X. Enhanced interfacial and dielectric performance for polyetherimide nanocomposites through tailoring shell polarities. ACS Appl. Mater. Interfaces 2023, 15, 23792–23803. [Google Scholar] [CrossRef]
- Li, J.P.; Jiang, J.H.; Chen, Y.; Liu, X.Y.; Zuo, P.Y.; Cheng, Q.L.; Zhuang, Q.X. Enhanced dielectric performance with high-temperature stability by interface-modulation of the core–shell structured imide-polymer@BT nanohybrids in PEI-based nanocomposites. J. Mater. Chem. C 2023, 11, 8420–8429. [Google Scholar] [CrossRef]
- Tang, H.L.; Wang, P.; Zheng, P.L.; Liu, X.B. Core-shell structured BaTiO3@polymer hybrid nanofiller for poly(arylene ether nitrile) nanocomposites with enhanced dielectric properties and high thermal stability. Compos. Sci. Technol. 2016, 123, 79–86. [Google Scholar] [CrossRef]
- Feng, M.N.; Li, C.T.; He, M.; Huang, Y.M.; Luo, J.H. Poly(arylene ether nitrile) ternary dielectric composites modulated via polydopamine-assisted BaTiO3 decorating MoS2 sheets. Ceram. Int. 2020, 46, 22085–22094. [Google Scholar] [CrossRef]
- Ma, J.C.; Azhar, U.; Zong, C.Y.; Zhang, Y.B.; Xu, A.H.; Zhai, C.C.; Zhang, L.; Zhang, S.X. Core-shell structured PVDF@BT nanoparticles for dielectric materials: A novel composite to prove the dependence of dielectric properties on ferroelectric shell. Mater. Des. 2019, 168, 107556. [Google Scholar] [CrossRef]
- Xu, W.H.; Yang, G.; Jin, L.; Liu, J.; Zhang, Y.H.; Zhang, Z.C.; Jiang, Z.H. High-k polymer nanocomposites filled with hyperbranched phthalocyanine-coated BaTiO3 for high-temperature and elevated field applications. ACS Appl. Mater. Interfaces 2018, 10, 11384–11394. [Google Scholar] [CrossRef]
- Yu, K.; Niu, Y.; Bai, Y.; Zhou, Y.C.; Wang, H. Poly(vinylidene fluoride) polymer based nanocomposites with significantly reduced energy loss by filling with core-shell structured BaTiO3/SiO2 nanoparticles. Appl. Phys. Lett. 2013, 102, 103904. [Google Scholar] [CrossRef]











Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mao, X.; Zhou, J.; Wu, J.; Chen, S.; Wang, P.; You, Y. Novel Polyarylene Ether Nitrile/BaTiO3-Decorated WS2 Nanohybrid Crosslinked Nanocomposites for Thermal-Stable Dielectrics. Polymers 2026, 18, 747. https://doi.org/10.3390/polym18060747
Mao X, Zhou J, Wu J, Chen S, Wang P, You Y. Novel Polyarylene Ether Nitrile/BaTiO3-Decorated WS2 Nanohybrid Crosslinked Nanocomposites for Thermal-Stable Dielectrics. Polymers. 2026; 18(6):747. https://doi.org/10.3390/polym18060747
Chicago/Turabian StyleMao, Xiaohua, Jingtao Zhou, Junda Wu, Siyi Chen, Pan Wang, and Yong You. 2026. "Novel Polyarylene Ether Nitrile/BaTiO3-Decorated WS2 Nanohybrid Crosslinked Nanocomposites for Thermal-Stable Dielectrics" Polymers 18, no. 6: 747. https://doi.org/10.3390/polym18060747
APA StyleMao, X., Zhou, J., Wu, J., Chen, S., Wang, P., & You, Y. (2026). Novel Polyarylene Ether Nitrile/BaTiO3-Decorated WS2 Nanohybrid Crosslinked Nanocomposites for Thermal-Stable Dielectrics. Polymers, 18(6), 747. https://doi.org/10.3390/polym18060747








