Hydrogels—Advanced Polymer Platforms for Drug Delivery
Abstract
1. Introduction
2. Methodology
3. Synthesis, Essential and Specific Properties of Hydrogels
3.1. Synthesis, Architecture, and Compositional Aspects
3.2. Swelling Behavior
3.3. Diffusion in Hydrogels
3.4. Biocompatibility
3.5. Biodegradability
4. Polymeric Materials Used in Hydrogel Synthesis
5. Stimulus-Responsive Smart Hydrogels
5.1. pH-Sensitive Hydrogels
5.2. Temperature-Sensitive Hydrogels
5.3. Glucose-Responsive Hydrogels
5.4. Enzyme-Sensitive Hydrogels
5.5. ROS-Sensitive Hydrogels
5.6. Multiresponsive Hydrogels
6. Advanced Hydrogel Platforms
6.1. Hydrogels with Integrated Biosensors for Early Diagnosis of the Pathological Microenvironment
6.2. Advanced Hydrogel Platforms for Adaptive Drug Release
6.3. Hydrogels Integrated with Wearable Devices and Smart Microtechnologies
7. The Main Administration Routes of Hydrogels
7.1. Oral Administration
7.2. Injectable Route
7.3. Transmucosal Administration
7.4. Cutaneous and Transdermal Route
8. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| PLGA | Poly(lactic-co-glycolic acid) |
| PEGDA | Poly(ethylene glycol) diacrylate |
| PEG | Poly(ethylene glycol) |
| CS | Chitosan |
| DN | Double Network |
| UDP | Uridine Diphosphate |
| HA | Hyaluronic Acid |
| PEGDMA | Poly(ethylene glycol) dimethacrylate |
| PCL | Polycaprolactone |
| PLA | Polylactic Acid |
| PVA | Poly(vinyl alcohol) |
| PNIPAm | Poly(N-isopropylacrylamide) |
| PDEAm | Poly(N,N-diethylacrylamide) |
| PU | Polyurethane |
| CMCS | Carboxymethyl Chitosan |
| MAA | Methacrylic Acid |
| PVP | Polyvinylpyrrolidone |
| GO | Graphene Oxide |
| TNT | Titanium Nanotubes |
| AgNPs | Silver Nanoparticles |
| IPN | Interpenetrating Polymer Network |
| MMP2 | Matrix Metalloproteinase-2 |
| MMP9 | Matrix Metalloproteinase-9 |
| siRNA | Small Interfering RNA |
| mRNA | Messenger RNA |
| PGE2 | Prostaglandin E2 |
| TNFα | Tumor Necrosis Factor-alpha |
| GoX | Glucose Oxidase |
| PBA | Phenylboronic Acid |
| ConA | Concanavalin A |
| GC | Glycated Chitosan |
| LCST | Lower Critical Solution Temperature |
| UCST | Upper Critical Solution Temperature |
| VEGF | Vascular Endothelial Growth Factor |
| shRNA | Short Hairpin RNA |
| GMS | Glyceryl Monostearate |
| MPO | Myeloperoxidase |
| SOD | Superoxide Dismutase |
| GSH | Glutathione |
| ADA | Dialdehyde Alginate |
| OCMC | O-Carboxymethyl Chitosan |
| IFNγ | Interferon-gamma |
| PLM | Persistent Luminescent Material |
| AR | Rheumatoid Arthritis |
| DMARD | Disease-Modifying Antirheumatic Drug |
| NSAIDs | Nonsteroidal Anti-Inflammatory Drugs |
| COX-2 | Cyclooxygenase-2 |
| RA GLP-1 | Glucagon-Like Peptide-1 Receptor Agonist |
| RHAMM | Receptor for Hyaluronic Acid-Mediated Motility |
| LYVE-1 | Lymphatic Vessel Endothelial Hyaluronic Acid Receptor 1 |
| TLR | Toll-Like Receptor |
| ROS | Reactive Oxygen Species |
References
- Zhang, Y.; Wu, B.M. Current Advances in Stimuli-Responsive Hydrogels as Smart Drug Delivery Carriers. Gels 2023, 9, 838. [Google Scholar] [CrossRef]
- Thang, N.H.; Chien, T.B.; Cuong, D.X. Polymer-Based Hydrogels Applied in Drug Delivery: An Overview. Gels 2023, 9, 523. [Google Scholar] [CrossRef] [PubMed]
- Protsak, I.S.; Morozov, Y.M. Fundamentals and Advances in Stimuli-Responsive Hydrogels and Their Applications: A Review. Gels 2025, 11, 30. [Google Scholar] [CrossRef]
- Sánchez-Cid, P.; Jiménez-Rosado, M.; Romero, A.; Pérez-Puyana, V. Novel Trends in Hydrogel Development for Biomedical Applications: A Review. Polymers 2022, 14, 3023. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Hou, L.; Ching, Y.C.; Ching, K.Y.; Hai, N.D.; Chuah, C.H. A Review of Recent Advances of Cellulose-Based Intelligent-Responsive Hydrogels as Vehicles for Controllable Drug Delivery System. Int. J. Biol. Macromol. 2024, 264, 130525. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Li, F.; Wang, K.; Wang, Q.; Liu, H.; Sun, X.; Xie, D. Synthesis, Characterizations, and Release Mechanisms of Carboxymethyl Chitosan-Graphene Oxide-Gelatin Composite Hydrogel for Controlled Delivery of Drug. Inorg. Chem. Commun. 2023, 155, 110965. [Google Scholar] [CrossRef]
- Lin, S.-H.; Hsu, S. Smart Hydrogels for in Situ Tissue Drug Delivery. J. Biomed. Sci. 2025, 32, 70. [Google Scholar] [CrossRef]
- Solanki, R.; Bhatia, D. Stimulus-Responsive Hydrogels for Targeted Cancer Therapy. Gels 2024, 10, 440. [Google Scholar] [CrossRef]
- Buttacavoli, M.; Di Cara, G.; Roz, E.; Pucci-Minafra, I.; Feo, S.; Cancemi, P. Integrated Multi-Omics Investigations of Metalloproteinases in Colon Cancer: Focus on MMP2 and MMP9. Int. J. Mol. Sci. 2021, 22, 12389. [Google Scholar] [CrossRef]
- Oliveira, T.P.; Da Silva, J.G.V.; Costa, P.M.; De Oliveira, M.C.D.; Coelho, R.C.; Santos Nascimento, A.M.S.; Silva-Filho, E.C.; Dos Santos, F.E.P.; Muniz, E.C. Study of Doxorubicin Release Carried out by pH-Responsive Chitosan-Alginate Hydrogels. J. Mol. Liq. 2025, 429, 127523. [Google Scholar] [CrossRef]
- Yang, K.; Lu, R.; Mei, J.; Cao, K.; Zeng, T.; Hua, Y.; Huang, X.; Li, W.; Yin, Y. The War between the Immune System and the Tumor—Using Immune Biomarkers as Tracers. Biomark. Res. 2024, 12, 51. [Google Scholar] [CrossRef]
- Nishida, A.; Andoh, A. The Role of Inflammation in Cancer: Mechanisms of Tumor Initiation, Progression, and Metastasis. Cells 2025, 14, 488. [Google Scholar] [CrossRef]
- Pathan, I.; Raza, M.A.; Roy, A.; Badwaik, H.; Sakure, K.; Uddin, A. Recent Advances in Thermo-Responsive Hydrogels for Ocular Drug Delivery: Materials, Mechanisms, and Clinical Potential. J. Drug Deliv. Sci. Technol. 2025, 114, 107537. [Google Scholar] [CrossRef]
- Wu, C.; Bi, C.; Kim, G.; Yang, Z.; Li, S.; Dai, T.; Wu, X.; Tan, J.; He, N.; Li, S. Oral Colon-Targeted Responsive Chitosan/Pectin-Based Nanoparticles Propels the Application of Tofacitinib in Colitis Therapy. Sci. Rep. 2025, 15, 1569. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Gu, Z.; Li, W.; Guo, L.; Wang, L.; Guo, L.; Ma, S.; Han, B.; Chang, J. pH-Sensitive O-Carboxymethyl Chitosan/Sodium Alginate Nanohydrogel for Enhanced Oral Delivery of Insulin. Int. J. Biol. Macromol. 2022, 223, 433–445. [Google Scholar] [CrossRef]
- Lu, X.; Lu, X.; Yang, P.; Zhang, Z.; Lv, H. Honokiol Nanosuspensions Loaded Thermosensitive Hydrogels as the Local Delivery System in Combination with Systemic Paclitaxel for Synergistic Therapy of Breast Cancer. Eur. J. Pharm. Sci. 2022, 175, 106212. [Google Scholar] [CrossRef]
- Abdellatif, A.A.H.; Mohammed, A.M.; Saleem, I.; Alsharidah, M.; Al Rugaie, O.; Ahmed, F.; Osman, S.K. Smart Injectable Chitosan Hydrogels Loaded with 5-Fluorouracil for the Treatment of Breast Cancer. Pharmaceutics 2022, 14, 661. [Google Scholar] [CrossRef] [PubMed]
- Dubashynskaya, N.V.; Bokatyi, A.N.; Sall, T.S.; Egorova, T.S.; Demyanova, E.V.; Dubrovskii, Y.A.; Murashko, E.A.; Anufrikov, Y.A.; Shasherina, A.Y.; Vlasova, E.N.; et al. Hyaluronan/B12-Chitosan Polyelectrolyte Complex for Oral Colistin Administration. Int. J. Biol. Macromol. 2024, 263, 130177. [Google Scholar] [CrossRef] [PubMed]
- Dubashynskaya, N.V.; Petrova, V.A.; Skorik, Y.A. Biopolymer Drug Delivery Systems for Oromucosal Application: Recent Trends in Pharmaceutical R&D. Int. J. Mol. Sci. 2024, 25, 5359. [Google Scholar] [CrossRef]
- Li, M.; Zhou, S.; Zhang, S.; Xie, X.; Nie, J.; Wang, Q.; Ma, L.; Cheng, Y.; Luo, J. Transdermal Delivery of CRISPR/Cas9-Mediated Melanoma Gene Therapy via Polyamines-Modified Thermosensitive Hydrogels. J. Nanobiotechnol. 2025, 23, 441. [Google Scholar] [CrossRef]
- Melo, B.L.; Lima-Sousa, R.; Alves, C.G.; Moreira, A.F.; Correia, I.J.; De Melo-Diogo, D. Chitosan-Based Injectable in Situ Forming Hydrogels Containing Dopamine-Reduced Graphene Oxide and Resveratrol for Breast Cancer Chemo-Photothermal Therapy. Biochem. Eng. J. 2022, 185, 108529. [Google Scholar] [CrossRef]
- Tavakoli, J.; Tang, Y. Hydrogel Based Sensors for Biomedical Applications: An Updated Review. Polymers 2017, 9, 364. [Google Scholar] [CrossRef]
- Wang, M.L.; Yeon, P.; Mofidfar, M.; Chamberlayne, C.; Xu, H.; Annes, J.P.; Zare, R.N.; Arbabian, A. A Wireless Implantable Closed-Loop Electrochemical Drug Delivery System. IEEE Trans. Biomed. Circuits Syst. 2025, 19, 777–790. [Google Scholar] [CrossRef]
- Yue, W.; Guo, Y.; Wu, J.-K.; Ganbold, E.; Kaushik, N.K.; Jaiswal, A.; Yu, N.; Wang, Y.; Lei, Y.-F.; Oh, B.; et al. A Wireless, Battery-Free Microneedle Patch with Light-Cured Swellable Hydrogel for Minimally-Invasive Glucose Detection. Nano Energy 2024, 131, 110194. [Google Scholar] [CrossRef]
- Wei, X.; Zhao, X.; Sui, D.; Chen, X.; Yang, W. Peptide Hydrogel Based Electrochemical Biosensor for Simultaneous Monitoring of H2O2 and NO Released from Three-Dimensional Cultured Breast Cancer Cells. Microchim. Acta 2024, 191, 523. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, H.; Hu, H.; Hu, S.; Ma, L. An Enzyme-Responsive Hydrogel of Ferrocene-Grafted Carboxymethyl Chitosan as a Soft Electrochemical Sensor for MMP-9 Detection. Int. J. Biol. Macromol. 2024, 268, 131582. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Liu, S.; Yu, W.; Wang, L.; Lv, F.; Yang, L.; Yu, H.; Shi, H.; Huang, Y. Hydrogel Based Flexible Wearable Sweat Sensor for SERS-AI Monitoring Treatment Effect of Lung Cancer. Sens. Actuators B Chem. 2025, 427, 137155. [Google Scholar] [CrossRef]
- Rykowska, I.; Nowak, I.; Nowak, R.; Michałkiewicz, O. Biodegradable Contact Lenses for Targeted Ocular Drug Delivery: Recent Advances, Clinical Applications, and Translational Perspectives. Molecules 2025, 30, 2542. [Google Scholar] [CrossRef]
- Yadav, P.; Singh, S.; Jaiswal, S.; Kumar, R. Synthetic and Natural Polymer Hydrogels: A Review of 3D Spheroids and Drug Delivery. Int. J. Biol. Macromol. 2024, 280, 136126. [Google Scholar] [CrossRef]
- Ho, T.-C.; Chang, C.-C.; Chan, H.-P.; Chung, T.-W.; Shu, C.-W.; Chuang, K.-P.; Duh, T.-H.; Yang, M.-H.; Tyan, Y.-C. Hydrogels: Properties and Applications in Biomedicine. Molecules 2022, 27, 2902. [Google Scholar] [CrossRef]
- Karoyo, A.H.; Wilson, L.D. A Review on the Design and Hydration Properties of Natural Polymer-Based Hydrogels. Materials 2021, 14, 1095. [Google Scholar] [CrossRef]
- Hoque, M.; Alam, M.; Wang, S.; Zaman, J.U.; Rahman, M.d.S.; Johir, M.; Tian, L.; Choi, J.-G.; Ahmed, M.B.; Yoon, M.-H. Interaction Chemistry of Functional Groups for Natural Biopolymer-Based Hydrogel Design. Mater. Sci. Eng. R Rep. 2023, 156, 100758. [Google Scholar] [CrossRef]
- Zhao, L.; Zhou, Y.; Zhang, J.; Liang, H.; Chen, X.; Tan, H. Natural Polymer-Based Hydrogels: From Polymer to Biomedical Applications. Pharmaceutics 2023, 15, 2514. [Google Scholar] [CrossRef]
- Santhamoorthy, M.; Kim, S.-C. A Review of the Development of Biopolymer Hydrogel-Based Scaffold Materials for Drug Delivery and Tissue Engineering Applications. Gels 2025, 11, 178. [Google Scholar] [CrossRef]
- Berradi, A.; Aziz, F.; Achaby, M.E.; Ouazzani, N.; Mandi, L. A Comprehensive Review of Polysaccharide-Based Hydrogels as Promising Biomaterials. Polymers 2023, 15, 2908. [Google Scholar] [CrossRef]
- Varaprasad, K.; Raghavendra, G.M.; Jayaramudu, T.; Yallapu, M.M.; Sadiku, R. A Mini Review on Hydrogels Classification and Recent Developments in Miscellaneous Applications. Mater. Sci. Eng. C 2017, 79, 958–971. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; Sharma, M.; Devi, M. Hydrogels: An Overview of Its Classifications, Properties, and Applications. J. Mech. Behav. Biomed. Mater. 2023, 147, 106145. [Google Scholar] [CrossRef] [PubMed]
- Friday, C.; Edgar, K. Recent Developments in All-Polysaccharide Hydrogels: Synthesis, Properties, and Biomedical Applications. Carbohydr. Polym. 2026, 376, 124806. [Google Scholar] [CrossRef]
- Ren, G.; Zhuge, C.; Zhu, Y.; Hu, R.; Jiang, Y.; Song, X.; Wu, W.; Wang, M.; Zhou, J.; Huang, M.; et al. Entirely Physical Crosslinking Strategy: pH-Tuned Double-Network Hydrogel Composed of Ferulic Acid-ε-Polylysine and Polyvinyl Alcohol. Food Chem. 2026, 505, 148019. [Google Scholar] [CrossRef] [PubMed]
- Kharkar, P.M.; Kiick, K.L.; Kloxin, A.M. Designing Degradable Hydrogels for Orthogonal Control of Cell Microenvironments. Chem. Soc. Rev. 2013, 42, 7335–7372. [Google Scholar] [CrossRef]
- Radulescu, D.-M.; Neacsu, I.A.; Grumezescu, A.-M.; Andronescu, E. New Insights of Scaffolds Based on Hydrogels in Tissue Engineering. Polymers 2022, 14, 799. [Google Scholar] [CrossRef]
- Malektaj, H.; Drozdov, A.D.; deClaville Christiansen, J. Mechanical Properties of Alginate Hydrogels Cross-Linked with Multivalent Cations. Polymers 2023, 15, 3012. [Google Scholar] [CrossRef] [PubMed]
- Massana Roquero, D.; Othman, A.; Melman, A.; Katz, E. Iron(III)-Cross-Linked Alginate Hydrogels: A Critical Review. Mater. Adv. 2022, 3, 1849–1873. [Google Scholar] [CrossRef]
- Ručigaj, A.; Golobič, J.; Kopač, T. The Role of Multivalent Cations in Determining the Cross-Linking Affinity of Alginate Hydrogels: A Combined Experimental and Modeling Study. Chem. Eng. J. Adv. 2024, 20, 100678. [Google Scholar] [CrossRef]
- Hu, W.; Wang, Z.; Xiao, Y.; Zhang, S.; Wang, J. Advances in Crosslinking Strategies of Biomedical Hydrogels. Biomater. Sci. 2019, 7, 843–855. [Google Scholar] [CrossRef]
- Zhuo, S.; Liang, Y.; Wu, Z.; Zhao, X.; Han, Y.; Guo, B. Supramolecular Hydrogels for Wound Repair and Hemostasis. Mater. Horiz. 2024, 11, 37–101. [Google Scholar] [CrossRef]
- Choi, Y.; Koh, H.Y.; Han, J.Y.; Seo, S. Synthesis of Hydrogel-Based Microgels and Nanogels Toward Therapeutic and Biomedical Applications. Appl. Sci. 2025, 15, 1368. [Google Scholar] [CrossRef]
- Zhang, S.; Ren, D.; Zhao, Q.; Peng, M.; Wang, X.; Zhang, Z.; Liu, W.; Huang, F. Observation of Topological Hydrogen-Bonding Domains in Physical Hydrogel for Excellent Self-Healing and Elasticity. Nat. Commun. 2025, 16, 2371. [Google Scholar] [CrossRef]
- Xie, Y.; Yu, W.; Xia, T.; O’Reilly, R.K.; Dove, A.P. Stereocomplex-Driven Morphological Transition of Coil–Rod–Coil Poly(Lactic Acid)-Based Cylindrical Nanoparticles. Macromolecules 2023, 56, 7689–7697. [Google Scholar] [CrossRef]
- Xia, T.; Li, X.; Wu, Y.; Lu, X. Synthesis and Thermally-Induced Gelation of Interpenetrating Nanogels. J. Colloid. Interface Sci. 2024, 669, 754–765. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.; Reddy, R.; Jiang, Q. Crosslinking Biopolymers for Biomedical Applications. Trends Biotechnol. 2015, 33, 362–369. [Google Scholar] [CrossRef]
- Nosrati-Siahmazgi, V.; Abbaszadeh, S.; Musaie, K.; Eskandari, M.R.; Rezaei, S.; Xiao, B.; Ghorbani-Bidkorpeh, F.; Shahbazi, M.-A. NIR-Responsive Injectable Hydrogel Cross-Linked by Homobifunctional PEG for Photo-Hyperthermia of Melanoma, Antibacterial Wound Healing, and Preventing Post-Operative Adhesion. Mater. Today Bio 2024, 26, 101062. [Google Scholar] [CrossRef]
- Karami, P.; Rana, V.K.; Zhang, Q.; Boniface, A.; Guo, Y.; Moser, C.; Pioletti, D.P. NIR Light-Mediated Photocuring of Adhesive Hydrogels for Noninvasive Tissue Repair via Upconversion Optogenesis. Biomacromolecules 2022, 23, 5007–5017. [Google Scholar] [CrossRef] [PubMed]
- Raza, M.A. Ionizing Radiation Crosslinked Chitosan-Based Hydrogels for Environmental Remediation. Gels 2025, 11, 492. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Hussain, C.M. 3D-Printed Hydrogel for Diverse Applications: A Review. Gels 2023, 9, 960. [Google Scholar] [CrossRef]
- Dell, A.C.; Wagner, G.; Own, J.; Geibel, J.P. 3D Bioprinting Using Hydrogels: Cell Inks and Tissue Engineering Applications. Pharmaceutics 2022, 14, 2596. [Google Scholar] [CrossRef]
- Gnatowski, T.; Kwiecińska-Piróg, J.; Bogiel, T. Development of 3D-Printed Hydrogel Disks as Standardized Platform for Evaluating Excipient Impact on Metronidazole’s Antimicrobial Activity. Pharmaceutics 2025, 17, 749. [Google Scholar] [CrossRef] [PubMed]
- Uysal, B.; Madduma-Bandarage, U.S.K.; Jayasinghe, H.G.; Madihally, S. 3D-Printed Hydrogels from Natural Polymers for Biomedical Applications: Conventional Fabrication Methods, Current Developments, Advantages, and Challenges. Gels 2025, 11, 192. [Google Scholar] [CrossRef]
- Yoon, J.; Han, H.; Jang, J. Nanomaterials-Incorporated Hydrogels for 3D Bioprinting Technology. Nano Converg. 2023, 10, 52. [Google Scholar] [CrossRef]
- Yu, J.; Huang, X.; Wu, F.; Feng, S.; Cheng, R.; Xu, J.; Cui, T.; Li, J. 3D-Printed Hydrogel Scaffolds Loaded with Flavanone@ZIF-8 Nanoparticles for Promoting Bacteria-Infected Wound Healing. Gels 2024, 10, 835. [Google Scholar] [CrossRef]
- Baishya, G.; Parasar, B.; Limboo, M.; Kumar, R.; Dutta, A.; Hussain, A.; Phukan, M.M.; Saikia, D. Advancements in Nanocomposite Hydrogels: A Comprehensive Review of Biomedical Applications. Discov. Mater. 2024, 4, 40. [Google Scholar] [CrossRef]
- Chelu, M.; Calderon Moreno, J.M.; Musuc, A.M.; Popa, M. Natural Regenerative Hydrogels for Wound Healing. Gels 2024, 10, 547. [Google Scholar] [CrossRef]
- Paoletti, L.; Ferrigno, G.; Zoratto, N.; Secci, D.; Di Meo, C.; Matricardi, P. Reinforcement of Dextran Methacrylate-Based Hydrogel, Semi-IPN, and IPN with Multivalent Crosslinkers. Gels 2024, 10, 773. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Mundy, D.C.; Le, P.; Seo, Y.A.; Logan, C.M.; Fernandes-Cunha, G.M.; Basco, C.A.; Myung, D. In Situ-Forming Collagen-Hyaluronate Semi-Interpenetrating Network Hydrogel Enhances Corneal Defect Repair. Trans. Vis. Sci. Technol. 2022, 11, 22. [Google Scholar] [CrossRef]
- Lara-Rico, R.; López-Badillo, C.M.; Claudio-Rizo, J.A.; Cabrera-Munguía, D.A.; Becerra-Rodríguez, J.J.; Espinosa-Neira, R.; Cruz-Ortiz, B.R. Smart Hydrogels Based on Semi-Interpenetrating Polymeric Networks of Collagen-Polyurethane-Alginate for Soft/Hard Tissue Healing, Drug Delivery Devices, and Anticancer Therapies. Biopolymers 2023, 114, e23538. [Google Scholar] [CrossRef]
- Dhand, A.P.; Galarraga, J.H.; Burdick, J.A. Enhancing Biopolymer Hydrogel Functionality through Interpenetrating Networks. Trends Biotechnol. 2021, 39, 519–538. [Google Scholar] [CrossRef]
- Wu, J.; Xue, W.; Yun, Z.; Liu, Q.; Sun, X. Biomedical Applications of Stimuli-Responsive “Smart” Interpenetrating Polymer Network Hydrogels. Mater. Today Bio 2024, 25, 100998. [Google Scholar] [CrossRef]
- Bernhard, S.; Tibbitt, M.W. Supramolecular Engineering of Hydrogels for Drug Delivery. Adv. Drug Deliv. Rev. 2021, 171, 240–256. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhu, X.; Zhao, J.; Ling, G.; Zhang, P. Biomedical Applications of Supramolecular Hydrogels with Enhanced Mechanical Properties. Adv. Colloid Interface Sci. 2023, 321, 103000. [Google Scholar] [CrossRef]
- Fang, K.; Wang, R.; Zhang, H.; Zhou, L.J.; Xu, T.; Xiao, Y.; Zhou, Y.; Gao, G.R.; Chen, J.; Liu, D.L.; et al. Mechano-Responsive, Tough, and Antibacterial Zwitterionic Hydrogels with Controllable Drug Release for Wound Healing Applications. ACS Appl. Mater. Interfaces 2020, 12, 52307. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, W.; Jiang, Y.; Wang, H.; Chen, G.; Guo, M. Physicochemical, Structural, and Biological Properties of Polysaccharides from Dandelion. Molecules 2019, 24, 1485. [Google Scholar] [CrossRef]
- Racovita, S.; Lungan, M.A.; Bunia, I.; Popa, M.; Vasiliu, S. Adsorption and Release Studies of Cefuroxime Sodium from Acrylic Ion Exchange Resin Microparticles Coated with Gellan. React. Funct. Polym. 2016, 105, 103–113. [Google Scholar] [CrossRef]
- Madhusudhan, A.; Suhagia, T.A.; Sharma, C.; Jaganathan, S.K.; Purohit, S.D. Carbon Based Polymeric Nanocomposite Hydrogel Bioink: A Review. Polymers 2024, 16, 3318. [Google Scholar] [CrossRef]
- Salem, M.A.S.; Bhat, A.S.; Mehandi, R.; Abugu, H.O.; Onwujiogu, V.C.; Orjiocha, S.I.; Chinonso, E.F. Advances in Sensing Technologies Using Functional Carbon Nanotube-Hydrogel Nanocomposites. Inorg. Chem. Commun. 2025, 175, 114139. [Google Scholar] [CrossRef]
- Makhado, E.; Seleka, W.M. Innovative Self-Assembled Silver Nanoparticles on Reduced Graphene Oxide Hydrogel Nanocomposite for Improved Electrochemical Hydrogen Generation and Sensing. Sci. Rep. 2025, 15, 28595. [Google Scholar] [CrossRef] [PubMed]
- Chronopoulou, L.; Binaymotlagh, R.; Cerra, S.; Haghighi, F.H.; Di Domenico, E.G.; Sivori, F.; Fratoddi, I.; Mignardi, S.; Palocci, C. Preparation of Hydrogel Composites Using a Sustainable Approach for In Situ Silver Nanoparticles Formation. Materials 2023, 16, 2134. [Google Scholar] [CrossRef] [PubMed]
- Rumon, M.M.H. Advances in Cellulose-Based Hydrogels: Tunable Swelling Dynamics and Their Versatile Real-Time Applications. RSC Adv. 2025, 15, 11688–11729. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Han, Q.; Feng, L.; Zhang, L. A Novel Solution-Diffusion-Flory-Rehner Model to Predict Flux Behavior during Forward Osmosis with Thermo-Responsive Hydrogel as Draw Agent. J. Membr. Sci. 2023, 675, 121553. [Google Scholar] [CrossRef]
- Revathi, D.; Panda, S.; Deshmukh, K.; Khotele, N.; Murthy, V.R.K.; Pasha, S.K.K. Smart Hydrogels for Sensing and Biosensing—Preparation, Smart Behaviours, and Emerging Applications—A Comprehensive Review. Polym. Test. 2025, 150, 108912. [Google Scholar] [CrossRef]
- De Piano, R.; Caccavo, D.; Barba, A.A.; Lamberti, G. Polyelectrolyte Hydrogels in Biological Systems: Modeling of Swelling and Deswelling Behavior. Chem. Eng. Sci. 2023, 279, 118959. [Google Scholar] [CrossRef]
- Zhu, C.; Zhang, X.; Gan, J.; Geng, D.; Bian, X.; Cheng, Y.; Tang, N. A pH-Sensitive Hydrogel Based on Carboxymethylated Konjac Glucomannan Crosslinked by Sodium Trimetaphosphate: Synthesis, Characterization, Swelling Behavior and Controlled Drug Release. Int. J. Biol. Macromol. 2023, 232, 123392. [Google Scholar] [CrossRef]
- Sabbagh, F.; Deshmukh, A.R.; Choi, Y.; Kim, B.S. Effect of Microsphere Concentration on Catechin Release from Microneedle Arrays. ACS Appl. Mater. Interfaces 2024, 16, 28276–28289. [Google Scholar] [CrossRef]
- Sabbagh, F. pH-Responsive Transdermal Release from Poly(Vinyl Alcohol)-Coated Liposomes and Transethosomes: Investigating the Role of Coating in Delayed Drug Delivery. ACS Appl. Bio Mater. 2025, 8, 4093–4103. [Google Scholar] [CrossRef]
- Suhail, M.; Wahab, A.; Wang, G.; An, S.; Kiran, B.; Jabeen, N.; Iqbal, M.Z.; Kong, X.; Wu, P.-C. Preparation of Natural Polymer-Based Hydrogels for Oral Delivery of Anti-Malarial Drug: In Vitro and Cytotoxicity Evaluation. RSC Adv. 2025, 15, 30378–30386. [Google Scholar] [CrossRef]
- Gao, K.; Xu, K. Advancements and Prospects of pH-Responsive Hydrogels in Biomedicine. Gels 2025, 11, 293. [Google Scholar] [CrossRef]
- Lavrentev, F.V.; Shilovskikh, V.V.; Alabusheva, V.S.; Yurova, V.Y.; Nikitina, A.A.; Ulasevich, S.A.; Skorb, E.V. Diffusion-Limited Processes in Hydrogels with Chosen Applications from Drug Delivery to Electronic Components. Molecules 2023, 28, 5931. [Google Scholar] [CrossRef]
- Mohsin, M.E.A.; Siddiqa, A.J.; Mousa, S.; Shrivastava, N.K. Design, Characterization, and Release Kinetics of a Hybrid Hydrogel Drug Delivery System for Sustained Hormone Therapy. Polymers 2025, 17, 999. [Google Scholar] [CrossRef] [PubMed]
- Amsden, B. Solute Diffusion within Hydrogels. Mechanisms and Models. Macromolecules 1998, 31, 8382–8395. [Google Scholar] [CrossRef]
- Nguyen, D.-V.; Yuan, Y.; Kukumberg, M.; Wang, L.; Lim, S.H.; Hassanbhai, A.M.; Chong, M.; Kofidis, T.; Tan, E.C.K.; Seliktar, D.; et al. Controlled Release of Vancomycin from PEGylated Fibrinogen Polyethylene Glycol Diacrylate Hydrogel. Biomater. Adv. 2024, 161, 213896. [Google Scholar] [CrossRef] [PubMed]
- Amirthalingam, S.; Rajendran, A.K.; Moon, Y.G.; Hwang, N.S. Stimuli-Responsive Dynamic Hydrogels: Design, Properties and Tissue Engineering Applications. Mater. Horiz. 2023, 10, 3325–3350. [Google Scholar] [CrossRef]
- Toews, P.M.; Velraj, A.; Bates, J.S. Stimuli-Responsive Hydrogels, Their Mass Transfer, Intermolecular Interactions, and Applications in Biomedical Devices. J. Mater. Sci. Mater. Eng. 2025, 20, 66. [Google Scholar] [CrossRef]
- Onaciu, A.; Munteanu, R.A.; Moldovan, A.I.; Moldovan, C.S.; Berindan-Neagoe, I. Hydrogels Based Drug Delivery Synthesis, Characterization and Administration. Pharmaceutics 2019, 11, 432. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, M.; Roopmani, P.; Rajendran, J.; Narayan, K.P.; Giri, J. Injectable, in-Situ Forming, Tunable, Biocompatible Gelatin Hydrogels for Biomedical Applications. Int. J. Biol. Macromol. 2025, 285, 138200. [Google Scholar] [CrossRef]
- Zhou, G.; Cao, Y.; Yan, Y.; Xu, H.; Zhang, X.; Yan, T.; Wan, H. Injectable Hydrogels Based on Hyaluronic Acid and Gelatin Combined with Salvianolic Acid B and Vascular Endothelial Growth Factor for Treatment of Traumatic Brain Injury in Mice. Molecules 2024, 29, 1705. [Google Scholar] [CrossRef]
- Patil, V.A.; Masters, K.S. Engineered Collagen Matrices. Bioengineering 2020, 7, 163. [Google Scholar] [CrossRef]
- Parvin, N.; Joo, S.W.; Mandal, T.K. Injectable Biopolymer-Based Hydrogels: A Next-Generation Platform for Minimally Invasive Therapeutics. Gels 2025, 11, 383. [Google Scholar] [CrossRef]
- Garcia-Garcia, A.; Muñana-González, S.; Lanceros-Mendez, S.; Ruiz-Rubio, L.; Alvarez, L.P.; Vilas-Vilela, J.L. Biodegradable Natural Hydrogels for Tissue Engineering, Controlled Release, and Soil Remediation. Polymers 2024, 16, 2599. [Google Scholar] [CrossRef]
- Nanda, D.; Behera, D.; Pattnaik, S.S.; Behera, A.K. Advances in Natural Polymer-Based Hydrogels: Synthesis, Applications, and Future Directions in Biomedical and Environmental Fields. Discov. Polym. 2025, 2, 6. [Google Scholar] [CrossRef]
- Segneanu, A.-E.; Bejenaru, L.E.; Bejenaru, C.; Blendea, A.; Mogoşanu, G.D.; Biţă, A.; Boia, E.R. Advancements in Hydrogels: A Comprehensive Review of Natural and Synthetic Innovations for Biomedical Applications. Polymers 2025, 17, 2026. [Google Scholar] [CrossRef]
- Liu, J.; Du, C.; Huang, W.; Lei, Y. Injectable Smart Stimuli-Responsive Hydrogels: Pioneering Advancements in Biomedical Applications. Biomater. Sci. 2024, 12, 8–56. [Google Scholar] [CrossRef] [PubMed]
- Piotrowska, U.; Szatko, J.; Nowakowska, A.; Klimaszewska, E.; Ogorzałek, M.; Sobczak, M. Chitosan-Based Drug Delivery Systems for Targeted Chemotherapy in Colorectal Cancer: A Scoping Review. Mar. Drugs 2025, 23, 467. [Google Scholar] [CrossRef]
- Cao, J.; Wu, B.; Yuan, P.; Liu, Y.; Hu, C. Research Progress of Sodium Alginate-Based Hydrogels in Biomedical Engineering. Gels 2025, 11, 758. [Google Scholar] [CrossRef]
- Román-Guerrero, A.; Cortés-Camargo, S.; Alpizar-Reyes, E.; Fabela-Morón, M.F.; Cruz-Olivares, J.; Velázquez-Gutiérrez, S.K.; Pérez-Alonso, C. Chemically Modified Alginate-Based Hydrogel-Matrices in Drug Delivery. Macromol 2025, 5, 36. [Google Scholar] [CrossRef]
- Bahwal, H.O.; Akhtar, K.; Bawazir, W.A.; Alharthi, S.H.; Khan, S.B. Preparation of Crosslinked Alginate Hydrogels for the Adsorption and Sustainable Release of Doxorubicin Hydrochloride. Polymers 2025, 17, 3294. [Google Scholar] [CrossRef]
- Rubio-Martin Del Campo, K.N.; Rivas-Gastelum, M.F.; Garcia-Amezquita, L.E.; Sepulveda-Villegas, M.; López-Mena, E.R.; Mejía-Méndez, J.L.; Sánchez-López, A.L. From Nature to Science: A Review of the Applications of Pectin-Based Hydrogels. Macromol 2025, 5, 58. [Google Scholar] [CrossRef]
- Grabowski, M.; Gmyrek, D.; Żurawska, M.; Trusek, A. Hyaluronic Acid: Production Strategies, Gel-Forming Properties, and Advances in Drug Delivery Systems. Gels 2025, 11, 424. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Wang, J.; Kong, L.; Wang, X.; Li, Q.; Zhang, L.; Shi, J.; Duan, J.; Mu, H. ROS-Responsive Hyaluronic Acid Hydrogel for Targeted Delivery of Probiotics to Relieve Colitis. Int. J. Biol. Macromol. 2022, 222, 1476–1486. [Google Scholar] [CrossRef]
- Ata, O.; Kokini, J.L.; Tavman, S.; Yazar, G. Advances in Collagen-/Gelatin-Based Hydrogels: Rheological Properties and Applications. Macromol 2025, 5, 55. [Google Scholar] [CrossRef]
- Moreno, J.M.C.; Chelu, M.; Popa, M. Biocompatible Stimuli-Sensitive Natural Hydrogels: Recent Advances in Biomedical Applications. Gels 2025, 11, 993. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, R.; Yang, S.; Li, S.; Gao, Z. Design and Application of Stimuli-Responsive DNA Hydrogels: A Review. Mater. Today Bio 2022, 16, 100430. [Google Scholar] [CrossRef] [PubMed]
- Mo, F.; Jiang, K.; Zhao, D.; Wang, Y.; Song, J.; Tan, W. DNA Hydrogel-Based Gene Editing and Drug Delivery Systems. Adv. Drug Deliv. Rev. 2021, 168, 79–98. [Google Scholar] [CrossRef]
- Zhong, R.; Talebian, S.; Mendes, B.B.; Wallace, G.; Langer, R.; Conde, J.; Shi, J. Hydrogels for RNA Delivery. Nat. Mater. 2023, 22, 818–831. [Google Scholar] [CrossRef]
- Vasile, C.; Pamfil, D.; Stoleru, E.; Baican, M. New Developments in Medical Applications of Hybrid Hydrogels Containing Natural Polymers. Molecules 2020, 25, 1539. [Google Scholar] [CrossRef] [PubMed]
- Getya, D.; Gitsov, I. Synthesis and Applications of Hybrid Polymer Networks Based on Renewable Natural Macromolecules. Molecules 2023, 28, 6030. [Google Scholar] [CrossRef]
- Zhao, Z.; Qin, Z.; Zhao, T.; Li, Y.; Hou, Z.; Hu, H.; Su, X.; Gao, Y. Crosslinked Biodegradable Hybrid Hydrogels Based on Poly(Ethylene Glycol) and Gelatin for Drug Controlled Release. Molecules 2024, 29, 4952. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Cao, S.; Yang, D.; Shang, L.; Hang, Y.; Wang, P.; Zhang, S.; Li, C.; Zhang, M.; Gao, X. Proteomics Profiling Reveals Pharmaceutical Excipient PEG400 Induces Nuclear-Receptor-Activation-Affected Lipid Metabolism and Metabolic Enzyme Expression. Int. J. Mol. Sci. 2025, 26, 1732. [Google Scholar] [CrossRef]
- Ali, A.; Nouseen, S.; Saroj, S.; Shegane, M.; Majumder, P.; Puri, A.; Rakshit, T.; Manna, D.; Pal, S. Repurposing Pinacol Esters of Boronic Acids for Tuning Viscoelastic Properties of Glucose-Responsive Polymer Hydrogels: Effects on Insulin Release Kinetics. J. Mater. Chem. B 2022, 10, 7591–7599. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Zhong, H.-J.; Ding, H.; Yu, B.; Ma, X.; Liu, X.; Chong, C.-M.; He, J. Polyvinyl Alcohol (PVA)-Based Hydrogels: Recent Progress in Fabrication, Properties, and Multifunctional Applications. Polymers 2024, 16, 2755. [Google Scholar] [CrossRef]
- Su, J.; Li, J.; Liang, J.; Zhang, K.; Li, J. Hydrogel Preparation Methods and Biomaterials for Wound Dressing. Life 2021, 11, 1016. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, C.; Yang, D.; Ruan, J.; Luo, Z.; Quan, P.; Fang, L. Mechanism Insight on Drug Skin Delivery from Polyurethane Hydrogels: Roles of Molecular Mobility and Intermolecular Interaction. Eur. J. Pharm. Sci. 2021, 161, 105783. [Google Scholar] [CrossRef]
- Plugariu, I.-A.; Gradinaru, L.M.; Avadanei, M.; Rosca, I.; Nita, L.E.; Maxim, C.; Bercea, M. Thermosensitive Polyurethane-Based Hydrogels as Potential Vehicles for Meloxicam Delivery. Pharmaceuticals 2023, 16, 1510. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.J.; Rajendran, R.R.; Mohanto, S.; Agarwal, U.; Panda, K.; Dhotre, K.; Manne, R.; Deepak, A.; Zafar, A.; Yasir, M.; et al. Poly(N-Isopropylacrylamide)-Based Hydrogels for Biomedical Applications: A Review of the State-of-the-Art. Gels 2022, 8, 454. [Google Scholar] [CrossRef] [PubMed]
- Thirupathi, K.; Phan, T.T.V.; Santhamoorthy, M.; Ramkumar, V.; Kim, S.-C. pH and Thermoresponsive PNIPAm-Co-Polyacrylamide Hydrogel for Dual Stimuli-Responsive Controlled Drug Delivery. Polymers 2022, 15, 167. [Google Scholar] [CrossRef]
- Rana, M.M.; De La Hoz Siegler, H. Evolution of Hybrid Hydrogels: Next-Generation Biomaterials for Drug Delivery and Tissue Engineering. Gels 2024, 10, 216. [Google Scholar] [CrossRef]
- Rizwan, M.; Yahya, R.; Hassan, A.; Yar, M.; Azzahari, A.; Selvanathan, V.; Sonsudin, F.; Abouloula, C. pH Sensitive Hydrogels in Drug Delivery: Brief History, Properties, Swelling, and Release Mechanism, Material Selection and Applications. Polymers 2017, 9, 137. [Google Scholar] [CrossRef]
- Karchoubi, F.; Afshar Ghotli, R.; Pahlevani, H.; Baghban Salehi, M. New Insights into Nanocomposite Hydrogels; a Review on Recent Advances in Characteristics and Applications. Adv. Ind. Eng. Polym. Res. 2024, 7, 54–78. [Google Scholar] [CrossRef]
- Baddi, S.; Dang-i, A.Y.; Gao, F.; Qiu, X.; Feng, C. Physical Strategies to Engineer Supramolecular Composite Hydrogels for Advanced Biomedical Applications. Prog. Mater. Sci. 2025, 151, 101428. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, L.; Wang, L.; Li, Z.; Yu, Y.; Zheng, Y.; Lian, D.; Li, X.; Chen, H.; Mei, L.; et al. An Injectable Hydrogel Based on Bi2Se3 Nanosheets and Hyaluronic Acid for Chemo-Photothermal Synergistic Therapy. Int. J. Biol. Macromol. 2023, 244, 125064. [Google Scholar] [CrossRef]
- Choi, S.; Jo, J.; Park, J.; Kim, S.; Jeong, S.; Jeong, S.Y.; Jung, S.H.; Choi, E.; Kim, H. Stimuli-Responsive, Methyl Cellulose-Based, Interpenetrating Network Hydrogels: Non-Covalent Design, Injectability, and Controlled Release. Carbohydr. Polym. 2025, 347, 122689. [Google Scholar] [CrossRef]
- Chaudhary, H.K.; Singh, P.; Niveria, K.; Yadav, M.; Malik, A.; Verma, A.K. Microcrystalline Cellulose and Itaconic Acid pH Sensitive Semi-Interpenetrating Network Hydrogel for Oral Insulin Delivery. Int. J. Biol. Macromol. 2024, 282, 136804. [Google Scholar] [CrossRef] [PubMed]
- Hua, J.; Huang, R.; Yu, M.; You, R.; Wang, L.; Yan, S.; Huang, Y.; Zhang, Q. High-Performance Silk Fibroin/Hyaluronic Acid Interpenetrating Network Hydrogel Microneedles for Diabetes Management. Int. J. Biol. Macromol. 2025, 298, 140357. [Google Scholar] [CrossRef]
- Li, C.; Li, H.; Guo, J.; Li, L.; Xi, X.; Yu, Y. Biocompatible Supramolecular Pseudorotaxane Hydrogels for Controllable Release of Doxorubicin in Ovarian Cancer SKOV-3 Cells. RSC Adv. 2020, 10, 689–697. [Google Scholar] [CrossRef]
- Bera, S.; Datta, H.K.; Dastidar, P. An Injectable Supramolecular Hydrogel as a Self-Drug-Delivery System for Local Chemoimmunotherapy against Melanoma. Biomater. Sci. 2023, 11, 5618–5633. [Google Scholar] [CrossRef]
- Deniaud-Bouët, E.; Hardouin, K.; Potin, P.; Kloareg, B.; Hervé, C. A Review about Brown Algal Cell Walls and Fucose-Containing Sulfated Polysaccharides: Cell Wall Context, Biomedical Properties and Key Research Challenges. Carbohydr. Polym. 2017, 175, 395–408. [Google Scholar] [CrossRef]
- Hamman, J.H. Chitosan Based Polyelectrolyte Complexes as Potential Carrier Materials in Drug Delivery Systems. Mar. Drugs 2010, 8, 1305–1322. [Google Scholar] [CrossRef]
- Madziva, H.; Kailasapathy, K.; Phillips, M. Alginate–Pectin Microcapsules as a Potential for Folic Acid Delivery in Foods. J. Microencapsul. 2005, 22, 343–351. [Google Scholar] [CrossRef]
- Egle, R.; Milek, M.; Mlinarič-Raščan, I.; Fahr, A.; Kristl, J. A Novel Gene Delivery System for Stable Transfection of Thiopurine-S-Methyltransferase Gene in Versatile Cell Types. Eur. J. Pharm. Biopharm. 2008, 69, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Fishman, M.L.; Hicks, K.B. Pectin in Controlled Drug Delivery—A Review. Cellulose 2006, 14, 15–24. [Google Scholar] [CrossRef]
- Anal, A.K.; Stevens, W.F.; Remuñán-López, C. Ionotropic Cross-Linked Chitosan Microspheres for Controlled Release of Ampicillin. Int. J. Pharm. 2006, 312, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Jha, A.K.; Harrington, D.A.; Farach-Carson, M.C.; Jia, X. Hyaluronic Acid-Based Hydrogels: From a Natural Polysaccharide to Complex Networks. Soft Matter 2012, 8, 3280. [Google Scholar] [CrossRef] [PubMed]
- Prestwich, G.D. Hyaluronic Acid-Based Clinical Biomaterials Derived for Cell and Molecule Delivery in Regenerative Medicine. J. Control. Release 2011, 155, 193–199. [Google Scholar] [CrossRef]
- Zhu, J.; Marchant, R.E. Design Properties of Hydrogel Tissue-Engineering Scaffolds. Expert. Rev. Med. Devices 2011, 8, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials 2010, 3, 1863–1887. [Google Scholar] [CrossRef]
- Klouda, L.; Mikos, A.G. Thermoresponsive Hydrogels in Biomedical Applications. Eur. J. Pharm. Biopharm. 2008, 68, 34–45. [Google Scholar] [CrossRef]
- Schmaljohann, D. Thermo- and pH-Responsive Polymers in Drug Delivery☆. Adv. Drug Deliv. Rev. 2006, 58, 1655–1670. [Google Scholar] [CrossRef]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in Pharmaceutical Formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Jayakumar, R.; Prabaharan, M.; Nair, S.V.; Tamura, H. Novel Chitin and Chitosan Nanofibers in Biomedical Applications. Biotechnol. Adv. 2010, 28, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Guelcher, S.A. Biodegradable Polyurethanes: Synthesis and Applications in Regenerative Medicine. Tissue Eng. Part B Rev. 2008, 14, 3–17. [Google Scholar] [CrossRef]
- Zhang, X.-Z.; Yang, Y.-Y.; Chung, T.-S.; Ma, K.-X. Preparation and Characterization of Fast Response Macroporous Poly(N-Isopropylacrylamide) Hydrogels. Langmuir 2001, 17, 6094–6099. [Google Scholar] [CrossRef]
- Bhattarai, N.; Gunn, J.; Zhang, M. Chitosan-Based Hydrogels for Controlled, Localized Drug Delivery. Adv. Drug Deliv. Rev. 2010, 62, 83–99. [Google Scholar] [CrossRef]
- Correa, S.; Grosskopf, A.K.; Lopez Hernandez, H.; Chan, D.; Yu, A.C.; Stapleton, L.M.; Appel, E.A. Translational Applications of Hydrogels. Chem. Rev. 2021, 121, 11385–11457. [Google Scholar] [CrossRef] [PubMed]
- Steinle, H.; Ionescu, T.-M.; Schenk, S.; Golombek, S.; Kunnakattu, S.-J.; Tutku Özbek, M.; Schlensak, C.; Wendel, H.P.; Avci-Adali, M. Incorporation of Synthetic mRNA in Injectable Chitosan-Alginate Hybrid Hydrogels for Local and Sustained Expression of Exogenous Proteins in Cells. Int. J. Mol. Sci. 2018, 19, 1313. [Google Scholar] [CrossRef]
- Wang, B.; Qiu, D.; Gu, Y.; Shan, Z.; Shi, R.; Luo, J.; Qi, S.; Wang, Y.; Jiang, B.; Jin, Y. A Lignin-Based Controlled/Sustained Release Hydrogel by Integrating Mechanical Strengthening and Bioactivities of Lignin. J. Bioresour. Bioprod. 2025, 10, 62–76. [Google Scholar] [CrossRef]
- Wu, L.; Huang, S.; Zheng, J.; Qiu, Z.; Lin, X.; Qin, Y. Synthesis and Characterization of Biomass Lignin-Based PVA Super-Absorbent Hydrogel. Int. J. Biol. Macromol. 2019, 140, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Damm, C.; Goepferich, A. Dual Cross-Linked Hydrogels as Promising Materials for Drug Delivery. Int. J. Pharm. 2025, 683, 125929. [Google Scholar] [CrossRef]
- Bordbar-Khiabani, A.; Gasik, M. Smart Hydrogels for Advanced Drug Delivery Systems. Int. J. Mol. Sci. 2022, 23, 3665. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, Y.; Li, T.; Zhang, J.; Tian, H. Stimuli-responsive Hydrogels: Fabrication and Biomedical Applications. VIEW 2022, 3, 20200112. [Google Scholar] [CrossRef]
- Xue, C.; Xu, X.; Zhang, L.; Liu, Y.; Liu, S.; Liu, Z.; Wu, M.; Shuai, Q. Self-Healing/pH-Responsive/Inherently Antibacterial Polysaccharide-Based Hydrogel for a Photothermal Strengthened Wound Dressing. Colloids Surf. B Biointerfaces 2022, 218, 112738. [Google Scholar] [CrossRef]
- Vegad, U.; Patel, M.; Khunt, D.; Zupančič, O.; Chauhan, S.; Paudel, A. pH Stimuli-Responsive Hydrogels from Non-Cellulosic Biopolymers for Drug Delivery. Front. Bioeng. Biotechnol. 2023, 11, 1270364. [Google Scholar] [CrossRef]
- Long, J.; Zhou, G.; Yu, X.; Xu, J.; Hu, L.; Pranovich, A.; Yong, Q.; Xie, Z.-H.; Xu, C. Harnessing Chemical Functionality of Xylan Hemicellulose towards Carbohydrate Polymer-Based pH/Magnetic Dual-Responsive Nanocomposite Hydrogel for Drug Delivery. Carbohydr. Polym. 2024, 343, 122461. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yao, H.; Ye, J.; Wang, Z.; Cong, H.; Yu, B. pH-Responsive Polymers: Classification, Response Mode, Properties and Biomedical Applications. Chem. Eng. J. 2025, 523, 168589. [Google Scholar] [CrossRef]
- Schoeller, J.; Itel, F.; Wuertz-Kozak, K.; Gaiser, S.; Luisier, N.; Hegemann, D.; Ferguson, S.J.; Fortunato, G.; Rossi, R.M. pH-Responsive Chitosan/Alginate Polyelectrolyte Complexes on Electrospun PLGA Nanofibers for Controlled Drug Release. Nanomaterials 2021, 11, 1850. [Google Scholar] [CrossRef]
- Hosonuma, M.; Yoshimura, K. Association between pH Regulation of the Tumor Microenvironment and Immunological State. Front. Oncol. 2023, 13, 1175563. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, G.; Wang, C.; Li, C.; Yao, P. Shear-Responsive Injectable Supramolecular Hydrogel Releasing Doxorubicin Loaded Micelles with pH-Sensitivity for Local Tumor Chemotherapy. Int. J. Pharm. 2017, 530, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Yuan, M.; Liu, L.; Zhang, K.; Zhao, B.; He, B.; Liang, Y.; Li, F. pH-Responsive Wound Dressings: Advances and Prospects. Nanoscale Horiz. 2023, 8, 422–440. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Lang, Y.; Li, C.; Liu, S.; Chang, M.-W. Biomimetic 3D Composite Scaffold with pH-Responsive Micropatterns for Wound Healing. Chem. Eng. J. 2024, 485, 149646. [Google Scholar] [CrossRef]
- Patroklou, G.; Triantafyllopoulou, E.; Goula, P.-E.; Karali, V.; Chountoulesi, M.; Valsami, G.; Pispas, S.; Pippa, N. pH-Responsive Hydrogels: Recent Advances in Pharmaceutical Applications. Polymers 2025, 17, 1451. [Google Scholar] [CrossRef]
- Tang, Q.; Zhao, D.; Yang, H.; Wang, L.; Zhang, X. A pH-Responsive Self-Healing Hydrogel Based on Multivalent Coordination of Ni2+ with Polyhistidine-Terminated PEG and IDA-Modified Oligochitosan. J. Mater. Chem. B 2019, 7, 30–42. [Google Scholar] [CrossRef]
- Yadav, J.; Chahal, S.; Kumar, P.; Kumar, C. Thermo-Responsive Smart Hydrogels: Molecular Engineering, Dynamic Cross-Linking Strategies, and Therapeutics Applications. Gels 2025, 12, 12. [Google Scholar] [CrossRef]
- Chatterjee, S.; Hui, P.C.; Kan, C. Thermoresponsive Hydrogels and Their Biomedical Applications: Special Insight into Their Applications in Textile Based Transdermal Therapy. Polymers 2018, 10, 480. [Google Scholar] [CrossRef]
- Moon, S.H.; Park, S.J.; Lee, Y.W.; Yang, Y.J. LCST/UCST Behavior of Polysaccharides for Hydrogel Fabrication. RSC Adv. 2024, 14, 35754–35768. [Google Scholar] [CrossRef]
- Tanga, S.; Aucamp, M.; Ramburrun, P. Injectable Thermoresponsive Hydrogels for Cancer Therapy: Challenges and Prospects. Gels 2023, 9, 418. [Google Scholar] [CrossRef]
- Li, B.; Shemkai, J.C.; Zhang, Q.; Qiao, F.; Yu, N.; Zhao, J.; Zuo, W.; Yang, J. Fabrication of an Alginate-Poloxamer 407 Thermosensitive Carboxymethyl Chitosan Stabilized Nanosuspension Hydrogel for Enhanced Ocular Delivery of Clobetasol Propionate in Uveitis Management. Int. J. Biol. Macromol. 2025, 319, 145356. [Google Scholar] [CrossRef]
- Andrgie, A.T.; Darge, H.F.; Mekonnen, T.W.; Birhan, Y.S.; Hanurry, E.Y.; Chou, H.-Y.; Wang, C.-F.; Tsai, H.-C.; Yang, J.M.; Chang, Y.-H. Ibuprofen-Loaded Heparin Modified Thermosensitive Hydrogel for Inhibiting Excessive Inflammation and Promoting Wound Healing. Polymers 2020, 12, 2619. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; She, K.; He, R.; Xu, Q. Application and Progress of Temperature-Sensitive Hydrogels in Cartilage Injury Repair. Front. Bioeng. Biotechnol. 2025, 13, 1602303. [Google Scholar] [CrossRef] [PubMed]
- Pal, S.; Rakshit, T.; Saha, S.; Jinagal, D. Glucose-Responsive Materials for Smart Insulin Delivery: From Protein-Based to Protein-Free Design. ACS Mater. Au 2025, 5, 239–252. [Google Scholar] [CrossRef]
- Bercea, M.; Lupu, A. Recent Insights into Glucose-Responsive Concanavalin A-Based Smart Hydrogels for Controlled Insulin Delivery. Gels 2024, 10, 260. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Navarrete, M.; Pérez-López, A.; Guillot, A.J.; Cordeiro, A.S.; Melero, A.; Aparicio-Blanco, J. Latest Advances in Glucose-Responsive Microneedle-Based Systems for Transdermal Insulin Delivery. Int. J. Biol. Macromol. 2024, 263, 130301. [Google Scholar] [CrossRef]
- Yao, Y.; Ji, K.; Wang, Y.; Gu, Z.; Wang, J. Materials and Carriers Development for Glucose-Responsive Insulin. Acc. Mater. Res. 2022, 3, 960–970. [Google Scholar] [CrossRef]
- Li, X.; Fu, M.; Wu, J.; Zhang, C.; Deng, X.; Dhinakar, A.; Huang, W.; Qian, H.; Ge, L. pH-Sensitive Peptide Hydrogel for Glucose-Responsive Insulin Delivery. Acta Biomater. 2017, 51, 294–303. [Google Scholar] [CrossRef]
- Yang, J.; Cao, Z. Glucose-Responsive Insulin Release: Analysis of Mechanisms, Formulations, and Evaluation Criteria. J. Control. Release 2017, 263, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Luck, L.A.; Punihaole, D.; Madura, J.D.; Asher, S.A. Photonic Crystal Protein Hydrogel Sensor Materials Enabled by Conformationally Induced Volume Phase Transition. Chem. Sci. 2016, 7, 4557–4562. [Google Scholar] [CrossRef]
- Taylor, M.J.; Gregory, R.; Tomlins, P.; Jacob, D.; Hubble, J.; Sahota, T.S. Closed-Loop Glycaemic Control Using an Implantable Artificial Pancreas in Diabetic Domestic Pig (Sus scrofa domesticus). Int. J. Pharm. 2016, 500, 371–378. [Google Scholar] [CrossRef]
- Lin, K.; Yi, J.; Mao, X.; Wu, H.; Zhang, L.-M.; Yang, L. Glucose-Sensitive Hydrogels from Covalently Modified Carboxylated Pullulan and Concanavalin A for Smart Controlled Release of Insulin. React. Funct. Polym. 2019, 139, 112–119. [Google Scholar] [CrossRef]
- Huang, Q.; Wang, L.; Yu, H.; Ur-Rahman, K. Advances in Phenylboronic Acid-Based Closed-Loop Smart Drug Delivery System for Diabetic Therapy. J. Control. Release 2019, 305, 50–64. [Google Scholar] [CrossRef]
- Kurnia, K.A.; Setyaningsih, W.; Darmawan, N.; Yuliarto, B. A Comprehensive Study on the Impact of the Substituent on pKa of Phenylboronic Acid in Aqueous and Non-Aqueous Solutions: A Computational Approach. J. Mol. Liq. 2021, 326, 115321. [Google Scholar] [CrossRef]
- Marco-Dufort, B.; Tibbitt, M.W. Design of Moldable Hydrogels for Biomedical Applications Using Dynamic Covalent Boronic Esters. Mater. Today Chem. 2019, 12, 16–33. [Google Scholar] [CrossRef]
- VandenBerg, M.A.; Webber, M.J. Biologically Inspired and Chemically Derived Methods for Glucose-Responsive Insulin Therapy. Adv. Healthc. Mater. 2019, 8, 1801466. [Google Scholar] [CrossRef]
- Lu, Y.; Yu, H.; Wang, L.; Shen, D.; Liu, J. Preparation of Phenylboronic Acid-based Glucose-Responsive Hydrogels and Microneedles for Regulated Delivery of Insulin. Eur. Polym. J. 2023, 192, 112061. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, J.; Wang, C.; Yu, F.; Yu, W.; Zhao, Z. A Glucose Responsive Multifunctional Hydrogel with Antibacterial Properties and Real-Time Monitoring for Diabetic Wound Treatment. Biomater. Sci. 2025, 13, 275–286. [Google Scholar] [CrossRef]
- Morariu, S. Advances in the Design of Phenylboronic Acid-Based Glucose-Sensitive Hydrogels. Polymers 2023, 15, 582. [Google Scholar] [CrossRef]
- Yu, J.; Wang, J.; Zhang, Y.; Chen, G.; Mao, W.; Ye, Y.; Kahkoska, A.R.; Buse, J.B.; Langer, R.; Gu, Z. Glucose-Responsive Insulin Patch for the Regulation of Blood Glucose in Mice and Minipigs. Nat. Biomed. Eng. 2020, 4, 499–506. [Google Scholar] [CrossRef]
- Kumi, M.; Ejeromedoghene, O.; Sudane, W.D.; Zhang, Z. Unlocking the Biological Response of Smart Stimuli-Responsive Hydrogels and Their Application in Biological Systems. Eur. Polym. J. 2024, 209, 112906. [Google Scholar] [CrossRef]
- Sobczak, M. Enzyme-Responsive Hydrogels as Potential Drug Delivery Systems—State of Knowledge and Future Prospects. Int. J. Mol. Sci. 2022, 23, 4421. [Google Scholar] [CrossRef] [PubMed]
- Chandrawati, R. Enzyme-Responsive Polymer Hydrogels for Therapeutic Delivery. Exp. Biol. Med. 2016, 241, 972–979. [Google Scholar] [CrossRef]
- Xie, X.; Wang, Y.; Deng, B.; Blatchley, M.R.; Lan, D.; Xie, Y.; Lei, M.; Liu, N.; Xu, F.; Wei, Z. Matrix Metalloproteinase-Responsive Hydrogels with Tunable Retention for on-Demand Therapy of Inflammatory Bowel Disease. Acta Biomater. 2024, 186, 354–368. [Google Scholar] [CrossRef]
- Noddeland, H.K.; Lind, M.; Jensen, L.B.; Petersson, K.; Skak-Nielsen, T.; Larsen, F.H.; Malmsten, M.; Heinz, A. Design and Characterization of Matrix Metalloproteinase-Responsive Hydrogels for the Treatment of Inflammatory Skin Diseases. Acta Biomater. 2023, 157, 149–161. [Google Scholar] [CrossRef]
- Delgado-Pujol, E.J.; Martínez, G.; Casado-Jurado, D.; Vázquez, J.; León-Barberena, J.; Rodríguez-Lucena, D.; Torres, Y.; Alcudia, A.; Begines, B. Hydrogels and Nanogels: Pioneering the Future of Advanced Drug Delivery Systems. Pharmaceutics 2025, 17, 215. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, Y.; Miao, H.; Yan, M.; Liu, C.; Wang, Y.; Zhang, J.; Fu, Z. Application of MMP-2-Responsive in Situ Forming Injectable Hydrogel in Preventing the Recurrence of Oral Squamous Cell Carcinoma. RSC Adv. 2025, 15, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Hu, Y.; Lin, Z.; Du, L.; Hu, Y.; Ouyang, L.; Xie, X.; Cheng, P.; Liao, J.; Lu, L.; et al. MMP-9 Responsive Hydrogel Promotes Diabetic Wound Healing by Suppressing Ferroptosis of Endothelial Cells. Bioact. Mater. 2025, 43, 240–254. [Google Scholar] [CrossRef]
- Vildanova, R.R.; Petrova, S.F.; Kolesov, S.V.; Khutoryanskiy, V.V. Biodegradable Hydrogels Based on Chitosan and Pectin for Cisplatin Delivery. Gels 2023, 9, 342. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kanika; Kumar, V.; Ahmad, A.; Mishra, R.K.; Nadeem, A.; Siddiqui, N.; Ansari, M.M.; Raza, S.S.; Kondepudi, K.K.; et al. Colon-Adhering Delivery System with Inflammation Responsiveness for Localized Therapy of Experimental Colitis. ACS Biomater. Sci. Eng. 2023, 9, 4781–4793. [Google Scholar] [CrossRef]
- Nam, M.; Lee, J.W.; Cha, G.D. Biomedical Application of Enzymatically Crosslinked Injectable Hydrogels. Gels 2024, 10, 640. [Google Scholar] [CrossRef]
- Liu, J.; Jia, B.; Li, Z.; Li, W. Reactive Oxygen Species-Responsive Polymer Drug Delivery Systems. Front. Bioeng. Biotechnol. 2023, 11, 1115603. [Google Scholar] [CrossRef]
- Lu, P.; Ruan, D.; Huang, M.; Tian, M.; Zhu, K.; Gan, Z.; Xiao, Z. Harnessing the Potential of Hydrogels for Advanced Therapeutic Applications: Current Achievements and Future Directions. Sig. Transduct. Target. Ther. 2024, 9, 166. [Google Scholar] [CrossRef]
- Kennedy, L.; Sandhu, J.K.; Harper, M.-E.; Cuperlovic-Culf, M. Role of Glutathione in Cancer: From Mechanisms to Therapies. Biomolecules 2020, 10, 1429. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Cui, J.; Lin, K.; Xie, J.; Wang, X. Recent Advances in Smart Stimuli-Responsive Biomaterials for Bone Therapeutics and Regeneration. Bone Res. 2022, 10, 17. [Google Scholar] [CrossRef]
- Lee, H.J.; Jeong, B. ROS-Sensitive Degradable PEG–PCL–PEG Micellar Thermogel. Small 2020, 16, 1903045. [Google Scholar] [CrossRef]
- Mollazadeh, S.; Mackiewicz, M.; Yazdimamaghani, M. Recent Advances in the Redox-Responsive Drug Delivery Nanoplatforms: A Chemical Structure and Physical Property Perspective. Mater. Sci. Eng. C 2021, 118, 111536. [Google Scholar] [CrossRef]
- Ye, H.; Zhou, Y.; Liu, X.; Chen, Y.; Duan, S.; Zhu, R.; Liu, Y.; Yin, L. Recent Advances on Reactive Oxygen Species-Responsive Delivery and Diagnosis System. Biomacromolecules 2019, 20, 2441–2463. [Google Scholar] [CrossRef] [PubMed]
- De La Torre, C.; Coll, C.; Ultimo, A.; Sancenón, F.; Martínez-Máñez, R.; Ruiz-Hernández, E. In Situ-Forming Gels Loaded with Stimuli-Responsive Gated Mesoporous Silica Nanoparticles for Local Sustained Drug Delivery. Pharmaceutics 2023, 15, 1071. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Ding, J.; Li, Z.; Rong, Y.; He, C.; Chen, X. ROS-Responsive Thermosensitive Polypeptide Hydrogels for Localized Drug Delivery and Improved Tumor Chemoimmunotherapy. Biomater. Sci. 2024, 12, 3100–3111. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Zang, M.; Zhang, Y.; Chen, Y.; Du, J.; Yan, A.; Gu, J.; Li, Y.; Wei, S.; Xu, J.; et al. A Bioresponsive Diselenide-Functionalized Hydrogel with Cascade Catalytic Activities for Enhanced Local Starvation- and Hypoxia-Activated Melanoma Therapy. Acta Biomater. 2023, 167, 182–194. [Google Scholar] [CrossRef]
- Le Thi, P.; Tran, D.L.; Hoang Thi, T.T.; Lee, Y.; Park, K.D. Injectable Reactive Oxygen and Nitrogen Species-Controlling Hydrogels for Tissue Regeneration: Current Status and Future Perspectives. Regen. Biomater. 2022, 9, rbac069. [Google Scholar] [CrossRef]
- Ma, X.; Cao, B.; Liu, X. Microenvironment-Responsive Peptide Hydrogels: Molecular Mechanisms, Design and Frontiers. Front. Bioeng. Biotechnol. 2025, 13, 1692319. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Heydarpour, R.; Tehrani, Z.M. Multi-Stimuli-Responsive Hydrogels and Their Medical Applications. New J. Chem. 2021, 45, 15705–15717. [Google Scholar] [CrossRef]
- Garshasbi, H.R.; Soleymani, S.; Naghib, S.M.; Mozafari, M.R. Multi-Stimuli-Responsive Hydrogels for Therapeutic Systems: An Overview on Emerging Materials, Devices, and Drugs. Curr. Pharm. Des. 2024, 30, 2027–2046. [Google Scholar] [CrossRef] [PubMed]
- Jo, Y.-J.; Gulfam, M.; Jo, S.-H.; Gal, Y.-S.; Oh, C.-W.; Park, S.-H.; Lim, K.T. Multi-Stimuli Responsive Hydrogels Derived from Hyaluronic Acid for Cancer Therapy Application. Carbohydr. Polym. 2022, 286, 119303. [Google Scholar] [CrossRef]
- Zhou, Y.; Dai, F.; Zhao, S.; Li, Z.; Liang, H.; Wang, X.; Zhao, L.; Tan, H. pH and Glucose Dual-Responsive Hydrogels Promoted Diabetic Wound Healing by Remodeling the Wound Microenvironment. Adv. Healthc. Mater. 2025, 14, 2500810. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Y.; Long, L.; Hu, C.; Kong, Q.; Wang, Y. A Spatiotemporal Release Platform Based on pH/ROS Stimuli-Responsive Hydrogel in Wound Repairing. J. Control. Release 2022, 341, 147–165. [Google Scholar] [CrossRef]
- Haidari, H.; Vasilev, K.; Cowin, A.J.; Kopecki, Z. Bacteria-Activated Dual pH- and Temperature-Responsive Hydrogel for Targeted Elimination of Infection and Improved Wound Healing. ACS Appl. Mater. Interfaces 2022, 14, 51744–51762. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yang, W.; Li, Y.; Ma, X.; Xie, Y.; Zhou, G.; Liu, S. Dual-Temperature/pH-Sensitive Hydrogels with Excellent Strength and Toughness Crosslinked Using Three Crosslinking Methods. Gels 2024, 10, 480. [Google Scholar] [CrossRef]
- Nicolae, A.-M.; Badea, M.; Bucurica, S.; Rasaliu, F.; Constantinescu, E.M. Intelligent Biosensors Based on Hyaluronic Acid Hydrogels for Monitoring Chronic Wound Healing with the Involvement of Artificial Intelligence. Biosensors 2025, 15, 773. [Google Scholar] [CrossRef] [PubMed]
- Man, T.; Yu, G.; Zhu, F.; Huang, Y.; Wang, Y.; Su, Y.; Deng, S.; Pei, H.; Li, L.; Ye, H.; et al. Antidiabetic Close Loop Based on Wearable DNA–Hydrogel Glucometer and Implantable Optogenetic Cells. JACS Au 2024, 4, 1500–1508. [Google Scholar] [CrossRef]
- Cicha, I.; Priefer, R.; Severino, P.; Souto, E.B.; Jain, S. Biosensor-Integrated Drug Delivery Systems as New Materials for Biomedical Applications. Biomolecules 2022, 12, 1198. [Google Scholar] [CrossRef]
- Li, Y.; Luo, S.; Gui, Y.; Wang, X.; Tian, Z.; Yu, H. Difunctional Hydrogel Optical Fiber Fluorescence Sensor for Continuous and Simultaneous Monitoring of Glucose and pH. Biosensors 2023, 13, 287. [Google Scholar] [CrossRef]
- Zhang, H.; Hu, H.; Li, Y.; Wang, J.; Ma, L. A Ferrocene-Based Hydrogel as Flexible Electrochemical Biosensor for Oxidative Stress Detection and Antioxidation Treatment. Biosens. Bioelectron. 2024, 248, 115997. [Google Scholar] [CrossRef]
- Zhou, Y.; Liang, X.; Shen, Z.; Zhang, R.; Zhang, G.; Yu, B.; Li, Y.; Xu, F.-J. Glucose-Responsive Hydrogel with Adaptive Insulin Release to Modulate Hyperglycemic Microenvironment and Promote Wound Healing. Biomaterials 2026, 326, 123641. [Google Scholar] [CrossRef]
- Yuan, Y.; Yang, Y.; Ji, Z.; Feng, J.; Shu, L.; Xiao, S.; Huang, Z. Wound Microenvironment Sensing and Self-Adjusting Hydrogel with Glucose, ROS, and MMP-9 Responsiveness for Improving Microcirculation of Diabetes Foot Ulcers. Chem. Eng. J. 2025, 505, 159537. [Google Scholar] [CrossRef]
- Tai, Q.-D.; Tang, Y.; Xie, S.-T.; Ye, Y.-Y.; Tang, X.; Lyu, Q.; Fan, Z.-J.; Liao, Y.-H. Glucose-Responsive Nanozyme Hydrogel for Glycemic Control and Catalytic Anti-Infective Therapy in Diabetic Wound Healing. Mater. Today Bio 2025, 35, 102405. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Tao, C.; Wang, J.; Le, Y.; Zhang, J. MMP-Responsive in Situ Forming Hydrogel Loaded with Doxorubicin-Encapsulated Biodegradable Micelles for Local Chemotherapy of Oral Squamous Cell Carcinoma. RSC Adv. 2019, 9, 31264–31273. [Google Scholar] [CrossRef]
- Li, B.; Li, M.; Wang, Y. Smart Hydrogels in Wearable Electronics for Wound Treatments. Small 2025, 21, e07368. [Google Scholar] [CrossRef]
- Du, N.; Fan, Y.; Zhang, Y.; Huang, H.; Lyu, Y.; Cai, R.; Zhang, Y.; Zhang, T.; Guan, Y.; Nan, K. Wireless, Programmable, and Refillable Hydrogel Bioelectronics for Enhanced Diabetic Wound Healing. Adv. Sci. 2024, 11, 2407820. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Xiang, H.; Xin, M.; Li, R.; Zhou, Y.; Pang, D.; Jia, X.; Yuan, H.; Chao, D. A Wearable Iontophoresis Enables Dual-Responsive Transdermal Delivery for Atopic Dermatitis Treatment. J. Colloid. Interface Sci. 2025, 678, 908–919. [Google Scholar] [CrossRef]
- Deng, P.; Shi, Z.; Fang, F.; Xu, Y.; Zhou, L.; Liu, Y.; Jin, M.; Chen, T.; Wang, Y.; Cao, Y.; et al. Wireless Matrix Metalloproteinase-9 Sensing by Smart Wound Dressing with Controlled Antibacterial Nanoparticles Release toward Chronic Wound Management. Biosens. Bioelectron. 2025, 268, 116860. [Google Scholar] [CrossRef] [PubMed]
- Richards, D.A.; Thomas, M.R.; Szijj, P.A.; Foote, J.; Chen, Y.; Nogueira, J.C.F.; Chudasama, V.; Stevens, M.M. Employing Defined Bioconjugates to Generate Chemically Functionalised Gold Nanoparticles for in Vitro Diagnostic Applications. Nanoscale 2021, 13, 11921–11931. [Google Scholar] [CrossRef]
- Nunziata, G.; Pollonio, D.; Lacroce, E.; Rossi, F. Smart pH-Responsive Polymers in Biomedical Applications: Nanoparticles, Hydrogels, and Emerging Hybrid Platforms. Mater. Today Chem. 2025, 49, 103063. [Google Scholar] [CrossRef]
- Kuntoji, G.; Kousar, N.; Gaddimath, S.; Koodlur Sannegowda, L. Macromolecule–Nanoparticle-Based Hybrid Materials for Biosensor Applications. Biosensors 2024, 14, 277. [Google Scholar] [CrossRef]
- Niu, Y.; Zhao, Z.; Yang, L.; Lv, D.; Sun, R.; Zhang, T.; Li, Y.; Bao, Q.; Zhang, M.; Wang, L.; et al. Towards Intelligent Wound Care: Hydrogel-Based Wearable Monitoring and Therapeutic Platforms. Polymers 2025, 17, 1881. [Google Scholar] [CrossRef]
- Khadeeja Thanha, K.P.; Ayisha Sana, P.; Ajesh, J.S.; Naseef, P.P.; Tharayil, H.; Lubaib, P.; Pramod, K. Advanced Smart Bioelectronics for Wound Healing: Biosensing, Drug Delivery, and Artificial Intelligence. Int. J. Pharm. 2025, 684, 126098. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, H.; Liu, F.; Su, Y.; Han, K.; Liu, Y.; Guan, F.; Liu, H.; Ma, S. Artificial Intelligence-Enabled Hydrogels: Innovations and Applications. J. Mater. Chem. B 2025, 13, 14967–14981. [Google Scholar] [CrossRef]
- Saeidi, M.; Chenani, H.; Orouji, M.; Adel Rastkhiz, M.; Bolghanabadi, N.; Vakili, S.; Mohamadnia, Z.; Hatamie, A.; Simchi, A. (Arash) Electrochemical Wearable Biosensors and Bioelectronic Devices Based on Hydrogels: Mechanical Properties and Electrochemical Behavior. Biosensors 2023, 13, 823. [Google Scholar] [CrossRef] [PubMed]
- Chenani, H.; Saeidi, M.; Rastkhiz, M.A.; Bolghanabadi, N.; Aghaii, A.H.; Orouji, M.; Hatamie, A.; Simchi, A. Challenges and Advances of Hydrogel-Based Wearable Electrochemical Biosensors for Real-Time Monitoring of Biofluids: From Lab to Market. A Review. Anal. Chem. 2024, 96, 8160–8183. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Kim, D.Y.; Lim, S.I.; Lim, H.-R. Hydrogel-Based Biointerfaces: Recent Advances, Challenges, and Future Directions in Human–Machine Integration. Gels 2025, 11, 232. [Google Scholar] [CrossRef]
- Taleb, H.; Nasser, A.; Andrieux, G.; Charara, N.; Motta Cruz, E. Wireless Technologies, Medical Applications and Future Challenges in WBAN: A Survey. Wirel. Netw. 2021, 27, 5271–5295. [Google Scholar] [CrossRef]
- Zhong, L.; He, S.; Lin, J.; Wu, J.; Li, X.; Pang, Y.; Li, Z. Technological Requirements and Challenges in Wireless Body Area Networks for Health Monitoring: A Comprehensive Survey. Sensors 2022, 22, 3539. [Google Scholar] [CrossRef]
- Uyanga, K.A.; Onyeukwu, E.J.; Han, J. Advancing Wearable Technologies with Hydrogels: Innovations and Future Perspectives. Gels 2025, 11, 988. [Google Scholar] [CrossRef]
- Chen, H.; Islam, W.; El Halabi, J.; Li, L.; Selaru, F.M. Innovative Gastrointestinal Drug Delivery Systems: Nanoparticles, Hydrogels, and Microgrippers. Front. Biosci. (Landmark Ed.) 2025, 30, 25281. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Lu, Y.; Quan, H.; Wang, Y.; Song, S.; Guo, H. Advanced Oral Drug Delivery Systems for Gastrointestinal Targeted Delivery: The Design Principles and Foundations. J. Nanobiotechnol. 2025, 23, 400. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.; Jamil, Q.A.; Noreen, S.; Ashraf, M.A.; Madni, A.; Mahmood, H.; Shoukat, H.; Raza, M.R. Preparation and Evaluation of pH-Sensitive Chitosan/Alginate Nanohybrid Mucoadhesive Hydrogel Beads: An Effective Approach to a Gastro-Retentive Drug Delivery System. Pharmaceutics 2024, 16, 1451. [Google Scholar] [CrossRef]
- Grosso, R.; Benito, E.; Carbajo-Gordillo, A.I.; García-Martín, M.G.; Perez-Puyana, V.; Sánchez-Cid, P.; de-Paz, M.-V. Biodegradable Guar-Gum-Based Super-Porous Matrices for Gastroretentive Controlled Drug Release in the Treatment of Helicobacter pylori: A Proof of Concept. Int. J. Mol. Sci. 2023, 24, 2281. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Škalko-Basnet, N.; Obuobi, S. Alginate/Polyethylene Glycol Diacrylate Shape Memory Hydrogel Films for Gastric Retention and Antibiotic Delivery in H. pylori Infection. Carbohydr. Polym. Technol. Appl. 2025, 12, 100967. [Google Scholar] [CrossRef]
- Zainab, F.; Mir, S.; Khan, S.W.; Awwad, N.S.; Ibrahium, H.A. Silane-Crosslinked Graphene Oxide Reinforced Chitosan/Sodium Alginate Hydrogel for Controlled Release of Insulin. RSC Adv. 2025, 15, 19983–20005. [Google Scholar] [CrossRef]
- Wu, H.; Nan, J.; Yang, L.; Park, H.J.; Li, J. Insulin-Loaded Liposomes Packaged in Alginate Hydrogels Promote the Oral Bioavailability of Insulin. J. Control. Release 2023, 353, 51–62. [Google Scholar] [CrossRef]
- Wang, D.; Wang, W.; Wang, P.; Wang, C.; Niu, J.; Liu, Y.; Chen, Y. Research Progress of Colon-Targeted Oral Hydrogel System Based on Natural Polysaccharides. Int. J. Pharm. 2023, 643, 123222. [Google Scholar] [CrossRef]
- Rabeh, M.E.; Vora, L.K.; Moore, J.V.; Bayan, M.F.; McCoy, C.P.; Wylie, M.P. Dual Stimuli-Responsive Delivery System for Self-Regulated Colon-Targeted Delivery of Poorly Water-Soluble Drugs. Biomater. Adv. 2024, 157, 213735. [Google Scholar] [CrossRef]
- Sun, W.; Fan, B.; Qin, X.; Zhang, X.; Zhang, P.; Zhang, Y. Synergistic ROS/Enzyme Dual-Responsive Oral Drug Delivery System: A Novel Multi-Mechanistic Platform for Spatiotemporal Control and Overcoming Drug Resistance in Colorectal Cancer Therapy. Mater. Today Bio 2025, 33, 101920. [Google Scholar] [CrossRef]
- Sorolla, M.A.; Hidalgo, I.; Sorolla, A.; Montal, R.; Pallisé, O.; Salud, A.; Parisi, E. Microenvironmental Reactive Oxygen Species in Colorectal Cancer: Involved Processes and Therapeutic Opportunities. Cancers 2021, 13, 5037. [Google Scholar] [CrossRef]
- Sampaio Moura, N.; Schledwitz, A.; Alizadeh, M.; Patil, S.A.; Raufman, J.-P. Matrix Metalloproteinases as Biomarkers and Therapeutic Targets in Colitis-Associated Cancer. Front. Oncol. 2024, 13, 1325095. [Google Scholar] [CrossRef]
- Catalano, T.; Selvaggi, F.; Cotellese, R.; Aceto, G.M. The Role of Reactive Oxygen Species in Colorectal Cancer Initiation and Progression: Perspectives on Theranostic Approaches. Cancers 2025, 17, 752. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; He, S.; Liao, B.; Wang, M.; Lin, H.; Hu, B.; Lan, X.; Shu, Z.; Zhang, C.; Yu, M.; et al. Orally Administrated Hydrogel Harnessing Intratumoral Microbiome and Microbiota-Related Immune Responses for Potentiated Colorectal Cancer Treatment. Research 2024, 7, 0364. [Google Scholar] [CrossRef] [PubMed]
- Omidian, H.; Chowdhury, S.D. Advancements and Applications of Injectable Hydrogel Composites in Biomedical Research and Therapy. Gels 2023, 9, 533. [Google Scholar] [CrossRef]
- Yang, W.; Chen, J.; Zhao, Z.; Wu, M.; Gong, L.; Sun, Y.; Huang, C.; Yan, B.; Zeng, H. Recent Advances in Fabricating Injectable Hydrogels via Tunable Molecular Interactions for Bio-Applications. J. Mater. Chem. B 2024, 12, 332–349. [Google Scholar] [CrossRef] [PubMed]
- Garg, A. In-Situ Gel: A Smart Carrier for Drug Delivery. Int. J. Pharm. 2024, 652, 123819. [Google Scholar] [CrossRef] [PubMed]
- Naser, Y.A.; Tekko, I.A.; Vora, L.K.; Peng, K.; Anjani, Q.K.; Greer, B.; Elliott, C.; McCarthy, H.O.; Donnelly, R.F. Hydrogel-Forming Microarray Patches with Solid Dispersion Reservoirs for Transdermal Long-Acting Microdepot Delivery of a Hydrophobic Drug. J. Control. Release 2023, 356, 416–433. [Google Scholar] [CrossRef]
- Maxwell, C.J.; Soltisz, A.M.; Rich, W.W.; Choi, A.; Reilly, M.A.; Swindle-Reilly, K.E. Tunable Alginate Hydrogels as Injectable Drug Delivery Vehicles for Optic Neuropathy. J. Biomed. Mater. Res. 2022, 110, 1621–1635. [Google Scholar] [CrossRef]
- Gil-Cabrerizo, P.; Saludas, L.; Prósper, F.; Abizanda, G.; Echanove-González De Anleo, M.; Ruiz-Villalba, A.; Garbayo, E.; Blanco-Prieto, M.J. Development of an Injectable Alginate-Collagen Hydrogel for Cardiac Delivery of Extracellular Vesicles. Int. J. Pharm. 2022, 629, 122356. [Google Scholar] [CrossRef]
- Rusu, A.G.; Nita, L.E.; Simionescu, N.; Ghilan, A.; Chiriac, A.P.; Mititelu-Tartau, L. Enzymatically-Crosslinked Gelatin Hydrogels with Nanostructured Architecture and Self-Healing Performance for Potential Use as Wound Dressings. Polymers 2023, 15, 780. [Google Scholar] [CrossRef]
- Li, Y.; Meng, H.; Liu, Y.; Lee, B.P. Fibrin Gel as an Injectable Biodegradable Scaffold and Cell Carrier for Tissue Engineering. Sci. World J. 2015, 2015, 685690. [Google Scholar] [CrossRef]
- Xu, M.; Fu, T.; Zhang, C.; An, Z.; Yan, J.; Lu, Z.; Wu, H.; Liu, J.; Qiu, L.; Shi, L.; et al. Prolonged, Staged, and Self-Regulated Methotrexate Release Coupled with ROS Scavenging in an Injectable Hydrogel for Rheumatoid Arthritis Therapy. J. Control. Release 2024, 375, 60–73. [Google Scholar] [CrossRef]
- d’Aquino, A.I.; Maikawa, C.L.; Nguyen, L.T.; Lu, K.; Hall, I.A.; Jons, C.K.; Kasse, C.M.; Yan, J.; Prossnitz, A.N.; Chang, E.; et al. Use of a Biomimetic Hydrogel Depot Technology for Sustained Delivery of GLP-1 Receptor Agonists Reduces Burden of Diabetes Management. Cell Rep. Med. 2023, 4, 101292. [Google Scholar] [CrossRef]
- Di Franco, S.; Alfieri, A.; Sansone, P.; Pota, V.; Coppolino, F.; Frangiosa, A.; Maffei, V.; Pace, M.C.; Passavanti, M.B.; Fiore, M. Hydrogel-Based Formulations to Deliver Analgesic Drugs: A Scoping Review of Applications and Efficacy. Biomedicines 2025, 13, 2465. [Google Scholar] [CrossRef]
- Guo, L.; Fu, Z.; Li, H.; Wei, R.; Guo, J.; Wang, H.; Qi, J. Smart Hydrogel: A New Platform for Cancer Therapy. Adv. Colloid. Interface Sci. 2025, 340, 103470. [Google Scholar] [CrossRef]
- Cheah, K.E.; Yeoh, P.S.Q.; Lai, K.W.; Wu, S.; Muhamad, F. Smart and Injectable Hydrogels for Cancer Treatment and Other Biomedical Applications. Biomater. Adv. 2026, 180, 214624. [Google Scholar] [CrossRef]
- Farajollahi, A. Injectable Hydrogel: A Promising Frontier in Cancer Therapy. Prog. Biophys. Mol. Biol. 2025, 199, 46–68. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, L.; Zhang, H.; Liao, B.; Li, Y. Progress of Research in In Situ Smart Hydrogels for Local Antitumor Therapy: A Review. Pharmaceutics 2022, 14, 2028. [Google Scholar] [CrossRef]
- Mohammadzadeh, V.; Atapour-Mashhad, H.; Shahvali, S.; Salehi, B.; Shaban, M.; Shirzad, M.; Salahvarzi, A.; Mohammadi, M. Hydrogels as Advanced Drug Delivery Platforms for Cancer Immunotherapy: Promising Innovations and Future Outlook. J. Nanobiotechnol 2025, 23, 545. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Song, H.; Zhou, X.; Chen, Y.; Liu, Q.; Gao, X.; Zhu, X.; Chen, D. Novel Facile Thermosensitive Hydrogel as Sustained and Controllable Gene Release Vehicle for Breast Cancer Treatment. Eur. J. Pharm. Sci. 2019, 134, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Fathi, A.; Gholami, M.; Motasadizadeh, H.; Malek-Khatabi, A.; Sedghi, R.; Dinarvand, R. Thermoresponsive in Situ Forming and Self-Healing Double-Network Hydrogels as Injectable Dressings for Silymarin/Levofloxacin Delivery for Treatment of Third-Degree Burn Wounds. Carbohydr. Polym. 2024, 331, 121856. [Google Scholar] [CrossRef] [PubMed]
- Lo, Y.-W.; Sheu, M.-T.; Chiang, W.-H.; Chiu, Y.-L.; Tu, C.-M.; Wang, W.-Y.; Wu, M.-H.; Wang, Y.-C.; Lu, M.; Ho, H.-O. In Situ Chemically Crosslinked Injectable Hydrogels for the Subcutaneous Delivery of Trastuzumab to Treat Breast Cancer. Acta Biomater. 2019, 86, 280–290. [Google Scholar] [CrossRef]
- Khanal, M.; Gohil, S.V.; Kuyinu, E.; Kan, H.-M.; Knight, B.E.; Baumbauer, K.M.; Lo, K.W.-H.; Walker, J.; Laurencin, C.T.; Nair, L.S. Injectable Nanocomposite Analgesic Delivery System for Musculoskeletal Pain Management. Acta Biomater. 2018, 74, 280–290. [Google Scholar] [CrossRef]
- Li, Y.; Chen, Y.; Xue, Y.; Jin, J.; Xu, Y.; Zeng, W.; Liu, J.; Xie, J. Injectable Hydrogel Delivery System with High Drug Loading for Prolonging Local Anesthesia. Adv. Sci. 2024, 11, 2309482. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, P.; Jiang, Y.; Li, L.; Ju, F.; Cheng, Q.; Zhou, Y.L.; Zhou, Y. Sustained-Release Esketamine Based Nanoparticle-Hydrogel Delivery System for Neuropathic Pain Management. Int. J. Nanomed. 2023, 18, 1131–1143. [Google Scholar] [CrossRef]
- Brown, P.; Pratt, A.G.; Hyrich, K.L. Therapeutic Advances in Rheumatoid Arthritis. BMJ 2024, 384, e070856. [Google Scholar] [CrossRef]
- Oliveira, I.M.; Fernandes, D.C.; Cengiz, I.F.; Reis, R.L.; Oliveira, J.M. Hydrogels in the Treatment of Rheumatoid Arthritis: Drug Delivery Systems and Artificial Matrices for Dynamic in Vitro Models. J. Mater. Sci. Mater. Med. 2021, 32, 74. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.K.; Ghosh, S.; More, N.; Choppadandi, M.; Sinha, M.; Srivalliputtur, S.B.; Velayutham, R.; Kapusetti, G. ECM-Mimetic, NSAIDs Loaded Thermo-Responsive, Immunomodulatory Hydrogel for Rheumatoid Arthritis Treatment. BMC Biotechnol. 2024, 24, 26. [Google Scholar] [CrossRef]
- Meng, M.; Chen, L.; Zhao, J.; Wang, C.; Hao, R.; Cai, Y.; Li, L.; Sun, H.; Liu, M. Prodrug-Loaded Micelles Targeting Articular Cavity Reactive Oxygen Species for Rheumatoid Arthritis Treatment. Mater. Today Bio 2025, 35, 102552. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Seo, J.; Kim, Y.H.; Ju, H.J.; Kim, S.; Ji, Y.B.; Lee, H.B.; Kim, H.S.; Choi, S.; Kim, M.S. Enhanced Intra-Articular Therapy for Rheumatoid Arthritis Using Click-Crosslinked Hyaluronic Acid Hydrogels Loaded with Toll-like Receptor Antagonizing Peptides. Acta Biomater. 2023, 172, 188–205. [Google Scholar] [CrossRef] [PubMed]
- Coghi, P.; Chen, Z.; Leong, K.F.; Yu, X.; Hosmane, N.S.; Zhu, Y. Metal Coordination-Based Nanomaterials: Novel Drug Delivery System for Treatment of Rheumatoid Arthritis. Coord. Chem. Rev. 2026, 546, 217049. [Google Scholar] [CrossRef]
- Ke, Z.; Ma, Q.; Ye, X.; Wang, Y.; Jin, Y.; Zhao, X.; Su, Z. Peptide GLP-1 Receptor Agonists: From Injection to Oral Delivery Strategies. Biochem. Pharmacol. 2024, 229, 116471. [Google Scholar] [CrossRef]
- Wang, H.; Chen, Z.; Gu, S.; Wang, Y.; Wang, Y.; Gao, C.; Shi, J.; Ding, J.; Wang, Q.; Yu, L. Long-Acting Management of Diabetes and Associated Complications Using an Injectable Thermosensitive Hydrogel Incorporating IgG-Conjugated GLP-1RA. Theranostics 2026, 16, 1833–1854. [Google Scholar] [CrossRef]
- Dutta, K.; Das, R.; Ling, J.; Monibas, R.M.; Carballo-Jane, E.; Kekec, A.; Feng, D.D.; Lin, S.; Mu, J.; Saklatvala, R.; et al. In Situ Forming Injectable Thermoresponsive Hydrogels for Controlled Delivery of Biomacromolecules. ACS Omega 2020, 5, 17531–17542. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, P.; Koskinen, M.; Mikkola, J.; Sarkhel, S.; Leino, L.; Seth, A.; Madalli, S.; Will, S.; Howard, V.G.; Brant, H.; et al. Injectable Biodegradable Silica Depot: Two Months of Sustained Release of the Blood Glucose Lowering Peptide, Pramlintide. Pharmaceutics 2022, 14, 553. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Leng, P.; Liu, Y. Oral Drug Delivery with Nanoparticles into the Gastrointestinal Mucosa. Fundam. Clin. Pharmacol. 2021, 35, 86–96. [Google Scholar] [CrossRef]
- Palmeira-de-Oliveira, R.; Oliveira, A.S.; Rolo, J.; Tomás, M.; Palmeira-de-Oliveira, A.; Simões, S.; Martinez-de-Oliveira, J. Women’s Preferences and Acceptance for Different Drug Delivery Routes and Products. Adv. Drug Deliv. Rev. 2022, 182, 114133. [Google Scholar] [CrossRef]
- Raju, N.A.; Popat, N.R. Overall Review: On Mucosal Drug Delivery System. Int. J. Creat. Res. Thoughts 2024, 12, 38–51. [Google Scholar]
- Zhao, Y.; Ran, B.; Xie, X.; Gu, W.; Ye, X.; Liao, J. Developments on the Smart Hydrogel-Based Drug Delivery System for Oral Tumor Therapy. Gels 2022, 8, 741. [Google Scholar] [CrossRef]
- Zaheer, M.; Waqas, M.K.; Arshad, S.; Ijaz, M.; Asim, M.H.; Rabbani, I.; Hussain, T.; Murtaza, G.; Batool, N.; Mahmood, A.; et al. Mucoadhesive Hydrogel Film Based on Cyclodextrin Cross-Linked with Thiolated Hydroxyethyl Cellulose for Mucosal Delivery of Cisplatin. J. Mol. Liq. 2025, 433, 127940. [Google Scholar] [CrossRef]
- Wu, H.; Li, C.; Yuan, H.; Zhao, J.; Li, S. Brain Delivery Strategies for Biomacromolecular Drugs: Intranasal Administration. Int. J. Nanomed. 2025, 20, 6463–6487. [Google Scholar] [CrossRef]
- Yeruva, T.; Yang, S.; Doski, S.; Duncan, G.A. Hydrogels for Mucosal Drug Delivery. ACS Appl. Bio Mater. 2023, 6, 1684–1700. [Google Scholar] [CrossRef]
- Su, Y.; Sun, B.; Gao, X.; Liu, S.; Hao, R.; Han, B. Chitosan Hydrogel Doped with PEG-PLA Nanoparticles for the Local Delivery of miRNA-146a to Treat Allergic Rhinitis. Pharmaceutics 2020, 12, 907. [Google Scholar] [CrossRef]
- Pina Costa, C.; Nižić Nodilo, L.; Silva, R.; Martins, E.; Zadravec, D.; Kalogjera, L.; Nuno Moreira, J.; Manuel Sousa Lobo, J.; Hafner, A.; Catarina Silva, A. In Situ Hydrogel Containing Diazepam-Loaded Nanostructured Lipid Carriers (DZP-NLC) for Nose-to-Brain Delivery: Development, Characterization and Deposition Studies in a 3D-Printed Human Nasal Cavity Model. Int. J. Pharm. 2023, 644, 123345. [Google Scholar] [CrossRef]
- Lynch, C.R.; Kondiah, P.P.D.; Choonara, Y.E.; Du Toit, L.C.; Ally, N.; Pillay, V. Hydrogel Biomaterials for Application in Ocular Drug Delivery. Front. Bioeng. Biotechnol. 2020, 8, 228. [Google Scholar] [CrossRef]
- Li, Q.; Cao, Y.; Wang, P. Recent Advances in Hydrogels for the Diagnosis and Treatment of Dry Eye Disease. Gels 2022, 8, 816. [Google Scholar] [CrossRef]
- Tyson, S.L.; Bafna, S.; Gira, J.P.; Goldberg, D.F.; Jones, J.J.; Jones, M.P.; Kim, J.K.; Martel, J.M.; Nordlund, M.L.; Piovanetti-Perez, I.K.; et al. Multicenter Randomized Phase 3 Study of a Sustained-Release Intracanalicular Dexamethasone Insert for Treatment of Ocular Inflammation and Pain after Cataract Surgery. J. Cataract. Refract. Surg. 2019, 45, 204–212. [Google Scholar] [CrossRef]
- Yan, K.; Zhang, Q.; Liu, Q.; Han, Y.; Liu, Z. Advances in Adhesive Hydrogels Applied for Ophthalmology: An Overview Focused on the Treatment. Theranostics 2025, 15, 915–942. [Google Scholar] [CrossRef]
- Holgado, M.A.; Anguiano-Domínguez, A.; Martín-Banderas, L. Contact Lenses as Drug-Delivery Systems: A Promising Therapeutic Tool. Arch. Soc. Española Oftalmol. (Engl. Ed.) 2020, 95, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Franco, P.; De Marco, I. Contact Lenses as Ophthalmic Drug Delivery Systems: A Review. Polymers 2021, 13, 1102. [Google Scholar] [CrossRef]
- Desai, D.T.; Maulvi, F.A.; Desai, A.R.; Shukla, M.R.; Desai, B.V.; Khadela, A.D.; Shetty, K.H.; Shah, D.O.; Willcox, M.D.P. In Vitro and in Vivo Evaluation of Cyclosporine-Graphene Oxide Laden Hydrogel Contact Lenses. Int. J. Pharm. 2022, 613, 121414. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-J.; Huh, J.; Hahn, S.K. Smart Theranostic Contact Lenses. J. Control. Release 2025, 379, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Li, S.; Li, J.; Falcone, N.; Cui, Q.; Shah, S.; Hartel, M.C.; Yu, N.; Young, P.; De Barros, N.R.; et al. Lab-on-a-Contact Lens: Recent Advances and Future Opportunities in Diagnostics and Therapeutics. Adv. Mater. 2022, 34, 2108389. [Google Scholar] [CrossRef]
- Kim, T.Y.; Lee, G.-H.; Mun, J.; Cheong, S.; Choi, I.; Kim, H.; Hahn, S.K. Smart Contact Lens Systems for Ocular Drug Delivery and Therapy. Adv. Drug Deliv. Rev. 2023, 196, 114817. [Google Scholar] [CrossRef] [PubMed]
- Ioniță, M.; Vlăsceanu, G.M.; Toader, A.G.; Manole, M. Advances in Therapeutic Contact Lenses for the Management of Different Ocular Conditions. J. Pers. Med. 2023, 13, 1571. [Google Scholar] [CrossRef]
- Yang, Z.; Wu, X.; Wang, H.; Zhou, J.; Lin, X.; Yang, P. Vagina, a Promising Route for Drug Delivery. J. Drug Deliv. Sci. Technol. 2024, 93, 105397. [Google Scholar] [CrossRef]
- AlAnsari, R.; Hasan, B.; Deen, G.R.; Torsten, U. Hydrogel- and Nanocomposite-Based Drug-Delivery Strategies in the Treatment of Vaginal Infections. Polymers 2024, 16, 775. [Google Scholar] [CrossRef]
- Dedeloudi, A.; Siamidi, A.; Pavlou, P.; Vlachou, M. Recent Advances in the Excipients Used in Modified Release Vaginal Formulations. Materials 2022, 15, 327. [Google Scholar] [CrossRef]
- Dos Santos, A.M.; Carvalho, S.G.; Araujo, V.H.S.; Carvalho, G.C.; Gremião, M.P.D.; Chorilli, M. Recent Advances in Hydrogels as Strategy for Drug Delivery Intended to Vaginal Infections. Int. J. Pharm. 2020, 590, 119867. [Google Scholar] [CrossRef] [PubMed]
- Mork, S.; Eilertsen, C.; Škalko-Basnet, N.; Jøraholmen, M.W. Formulation Matters: Assessment of the Correlation between Mucoadhesiveness and Type of Chitosan Formulation for Vaginal Application. J. Drug Deliv. Sci. Technol. 2025, 114, 107564. [Google Scholar] [CrossRef]
- Enggi, C.K.; Isa, H.T.; Sulistiawati, S.; Ardika, K.A.R.; Wijaya, S.; Asri, R.M.; Mardikasari, S.A.; Donnelly, R.F.; Permana, A.D. Development of Thermosensitive and Mucoadhesive Gels of Cabotegravir for Enhanced Permeation and Retention Profiles in Vaginal Tissue: A Proof of Concept Study. Int. J. Pharm. 2021, 609, 121182. [Google Scholar] [CrossRef]
- Almomen, A.; Cho, S.; Yang, C.-H.; Li, Z.; Jarboe, E.A.; Peterson, C.M.; Huh, K.M.; Janát-Amsbury, M.M. Thermosensitive Progesterone Hydrogel: A Safe and Effective New Formulation for Vaginal Application. Pharm. Res. 2015, 32, 2266–2279. [Google Scholar] [CrossRef]
- Permana, A.D.; Asri, R.M.; Amir, M.N.; Himawan, A.; Arjuna, A.; Juniarti, N.; Utami, R.N.; Mardikasari, S.A. Development of Thermoresponsive Hydrogels with Mucoadhesion Properties Loaded with Metronidazole Gel-Flakes for Improved Bacterial Vaginosis Treatment. Pharmaceutics 2023, 15, 1529. [Google Scholar] [CrossRef]
- Afloarea, O.-T.; Nacu, I.; Vereștiuc, L.; Yilmaz, C.N.; Panainte, A.D.; Peptu, C.A.; Ostafe, I.-G.; Bibire, N. In Vitro and Ex Vivo Evaluation of Novel Methacrylated Chitosan-PNIPAAm-Hyaluronic Acid Hydrogels Loaded with Progesterone for Applications in Vaginal Delivery. Polymers 2024, 16, 2160. [Google Scholar] [CrossRef]
- Taurin, S.; Almomen, A.A.; Pollak, T.; Kim, S.J.; Maxwell, J.; Peterson, C.M.; Owen, S.C.; Janát-Amsbury, M.M. Thermosensitive Hydrogels a Versatile Concept Adapted to Vaginal Drug Delivery. J. Drug Target. 2018, 26, 533–550. [Google Scholar] [CrossRef]
- Tahir, D.; Ardiansyah, A.; Heryanto, H.; Noor, E.E.M.; Mohamed, M.A. Chitosan-Based Hydrogels: A Comprehensive Review of Transdermal Drug Delivery. Int. J. Biol. Macromol. 2025, 298, 140010. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, S.; Chen, J.; Yang, S.; Wu, C.; Zhao, Q.; Zhang, H.; Wei, K.; Zhang, J.; Zhang, R.; et al. Transdermal Drugs and Delivery Strategies: History, Advances and Future Prospects. Chin. Chem. Lett. 2025, 112170. in press. [Google Scholar] [CrossRef]
- Rathore, P.N.; Pal, A.; Pandey, S.; Maji, S.; Ram, A. A Review on Exploring the Emerging Role of Novel Transdermal Gel Drug Delivery Systems for Skin Diseases. Next Res. 2025, 2, 100373. [Google Scholar] [CrossRef]
- Asad, M.I.; Khan, D.; Rehman, A.U.; Elaissari, A.; Ahmed, N. Development and In Vitro/In Vivo Evaluation of pH-Sensitive Polymeric Nanoparticles Loaded Hydrogel for the Management of Psoriasis. Nanomaterials 2021, 11, 3433. [Google Scholar] [CrossRef] [PubMed]
- Wan, L.; Wang, M.; Song, Y.; Sun, G.; Zhang, R.; Chen, Z.; Zhou, Y.; Ma, K.; Zheng, R.; Gluchman, M.; et al. Multifunctional Hydrogel for the Treatment of Atopic Dermatitis: Current Advances and Translational Challenges. Eur. J. Pharm. Sci. 2025, 214, 107288. [Google Scholar] [CrossRef] [PubMed]
- Zagórska-Dziok, M.; Sobczak, M. Hydrogel-Based Active Substance Release Systems for Cosmetology and Dermatology Application: A Review. Pharmaceutics 2020, 12, 396. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Dong, S.; Xu, W.; Tu, S.; Yan, L.; Zhao, C.; Ding, J.; Chen, X. Antibacterial Hydrogels. Adv. Sci. 2018, 5, 1700527. [Google Scholar] [CrossRef]
- Casadidio, C.; Censi, R. Prospects of Hydrogels in Wound Healing: Toward the Next Generation of Smart Biomaterials. Pharmaceutics 2025, 17, 1486. [Google Scholar] [CrossRef] [PubMed]
- Mota, F.A.R.; Pereira, S.A.P.; Araújo, A.R.T.S.; Passos, M.L.C.; Saraiva, M.L.M.F.S. Biomarkers in the Diagnosis of Wounds Infection: An Analytical Perspective. TrAC Trends Anal. Chem. 2021, 143, 116405. [Google Scholar] [CrossRef]
- Psarrou, M.; Mitraki, A.; Vamvakaki, M.; Kokotidou, C. Stimuli-Responsive Polysaccharide Hydrogels and Their Composites for Wound Healing Applications. Polymers 2023, 15, 986. [Google Scholar] [CrossRef]
- Huang, Y.; Mu, L.; Zhao, X.; Han, Y.; Guo, B. Bacterial Growth-Induced Tobramycin Smart Release Self-Healing Hydrogel for Pseudomonas aeruginosa-Infected Burn Wound Healing. ACS Nano 2022, 16, 13022–13036. [Google Scholar] [CrossRef]
- Shah, S.A.; Sohail, M.; Khan, S.; Minhas, M.U.; De Matas, M.; Sikstone, V.; Hussain, Z.; Abbasi, M.; Kousar, M. Biopolymer-Based Biomaterials for Accelerated Diabetic Wound Healing: A Critical Review. Int. J. Biol. Macromol. 2019, 139, 975–993. [Google Scholar] [CrossRef] [PubMed]
- Saghazadeh, S.; Rinoldi, C.; Schot, M.; Kashaf, S.S.; Sharifi, F.; Jalilian, E.; Nuutila, K.; Giatsidis, G.; Mostafalu, P.; Derakhshandeh, H.; et al. Drug Delivery Systems and Materials for Wound Healing Applications. Adv. Drug Deliv. Rev. 2018, 127, 138–166. [Google Scholar] [CrossRef]
- Jia, B.; Li, G.; Cao, E.; Luo, J.; Zhao, X.; Huang, H. Recent Progress of Antibacterial Hydrogels in Wound Dressings. Mater. Today Bio 2023, 19, 100582. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Wang, P.; Xie, J.; Tang, Z.; Fu, J.; Ning, Y.; Zhong, Q.; Wang, D.; Lei, M.; Mai, H.; et al. A Multifunctional Injectable, Self-Healing, and Adhesive Hydrogel-Based Wound Dressing Stimulated Diabetic Wound Healing with Combined Reactive Oxygen Species Scavenging, Hyperglycemia Reducing, and Bacteria-Killing Abilities. J. Nanobiotechnol. 2024, 22, 444. [Google Scholar] [CrossRef]
- Phatale, V.; Vaiphei, K.K.; Jha, S.; Patil, D.; Agrawal, M.; Alexander, A. Overcoming Skin Barriers through Advanced Transdermal Drug Delivery Approaches. J. Control. Release 2022, 351, 361–380. [Google Scholar] [CrossRef]
- Bashyal, S.; Shin, C.Y.; Hyun, S.M.; Jang, S.W.; Lee, S. Preparation, Characterization, and In Vivo Pharmacokinetic Evaluation of Polyvinyl Alcohol and Polyvinyl Pyrrolidone Blended Hydrogels for Transdermal Delivery of Donepezil HCl. Pharmaceutics 2020, 12, 270. [Google Scholar] [CrossRef]
- Alex, M.; Alsawaftah, N.M.; Husseini, G.A. State-of-All-the-Art and Prospective Hydrogel-Based Transdermal Drug Delivery Systems. Appl. Sci. 2024, 14, 2926. [Google Scholar] [CrossRef]
- Hou, X.; Li, J.; Hong, Y.; Ruan, H.; Long, M.; Feng, N.; Zhang, Y. Advances and Prospects for Hydrogel-Forming Microneedles in Transdermal Drug Delivery. Biomedicines 2023, 11, 2119. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Matsumoto, H.; Moro-oka, Y.; Tanaka, M.; Miyahara, Y.; Suganami, T.; Matsumoto, A. Smart Microneedle Fabricated with Silk Fibroin Combined Semi-Interpenetrating Network Hydrogel for Glucose-Responsive Insulin Delivery. ACS Biomater. Sci. Eng. 2019, 5, 5781–5789. [Google Scholar] [CrossRef]
- Zhu, M.; Liu, Y.; Jiang, F.; Cao, J.; Kundu, S.C.; Lu, S. Combined Silk Fibroin Microneedles for Insulin Delivery. ACS Biomater. Sci. Eng. 2020, 6, 3422–3429. [Google Scholar] [CrossRef]
- Smith, F.; Sabri, A.H.; Heppel, M.; Fonseca, I.; Chowdhury, F.; Cheung, K.; Willmor, S.; Rawson, F.; Marlow, M. The Clinical and Translational Prospects of Microneedle Devices, with a Focus on Insulin Therapy for Diabetes Mellitus as a Case Study. Int. J. Pharm. 2022, 628, 122234. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, L.; Cui, Y. A Wearable, Rapidly Manufacturable, Stability-Enhancing Microneedle Patch for Closed-Loop Diabetes Management. Microsyst. Nanoeng. 2024, 10, 112. [Google Scholar] [CrossRef]
- Karthikeyan, L.; Kang, H.W. Recent Progress in Multifunctional Theranostic Hydrogels: The Cornerstone of next-Generation Wound Care Technologies. Biomater. Sci. 2025, 13, 4358–4389. [Google Scholar] [CrossRef] [PubMed]

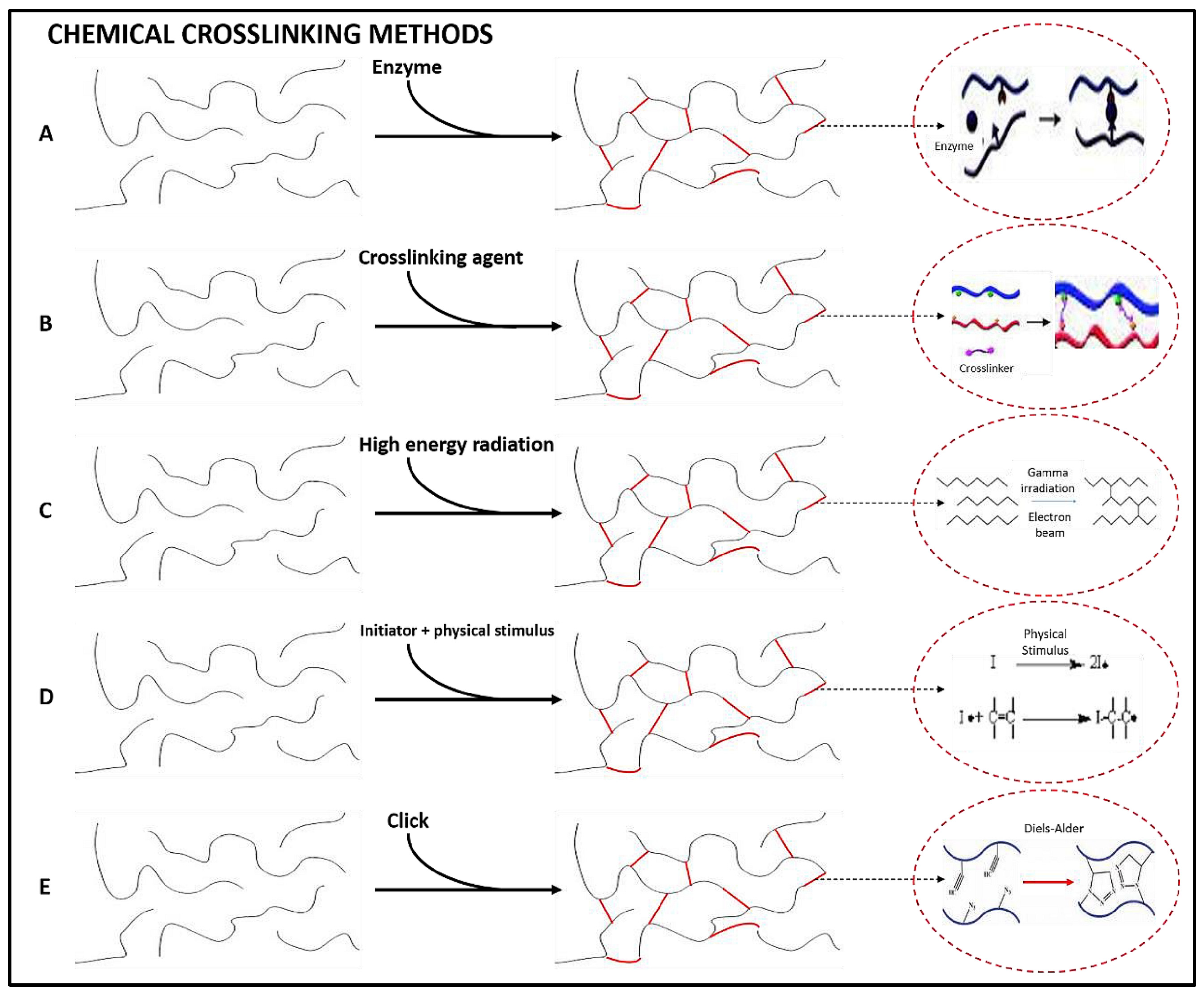


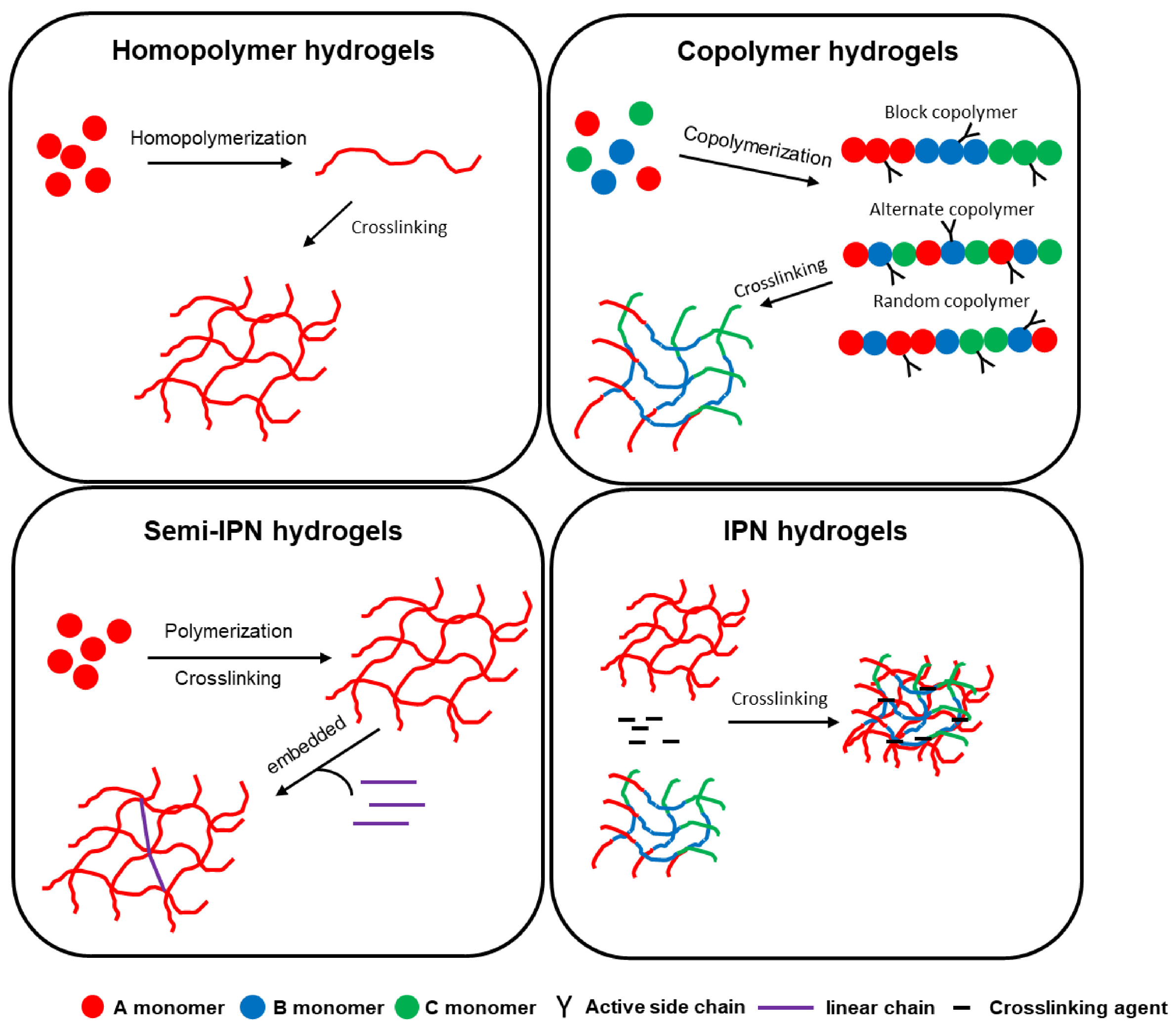




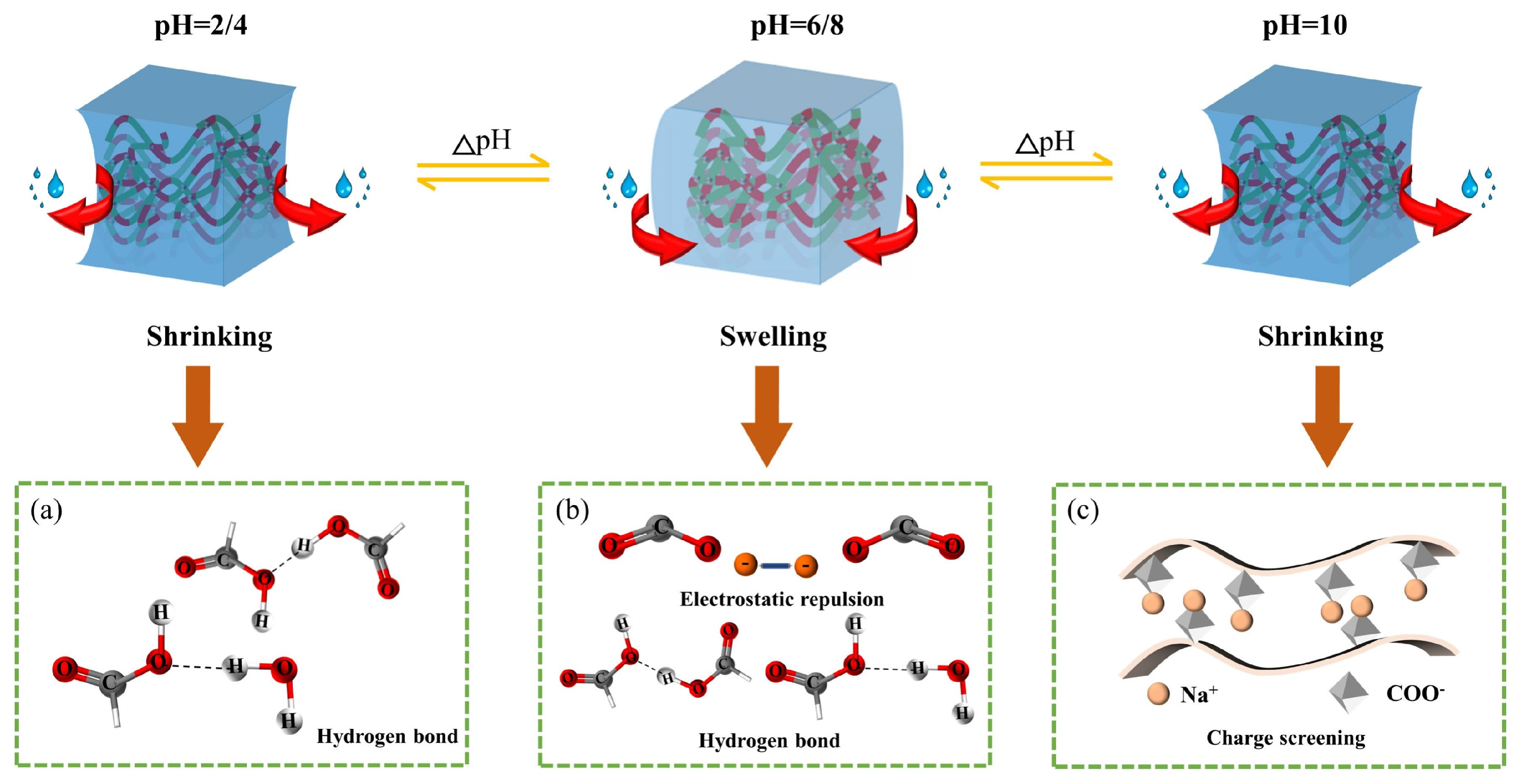



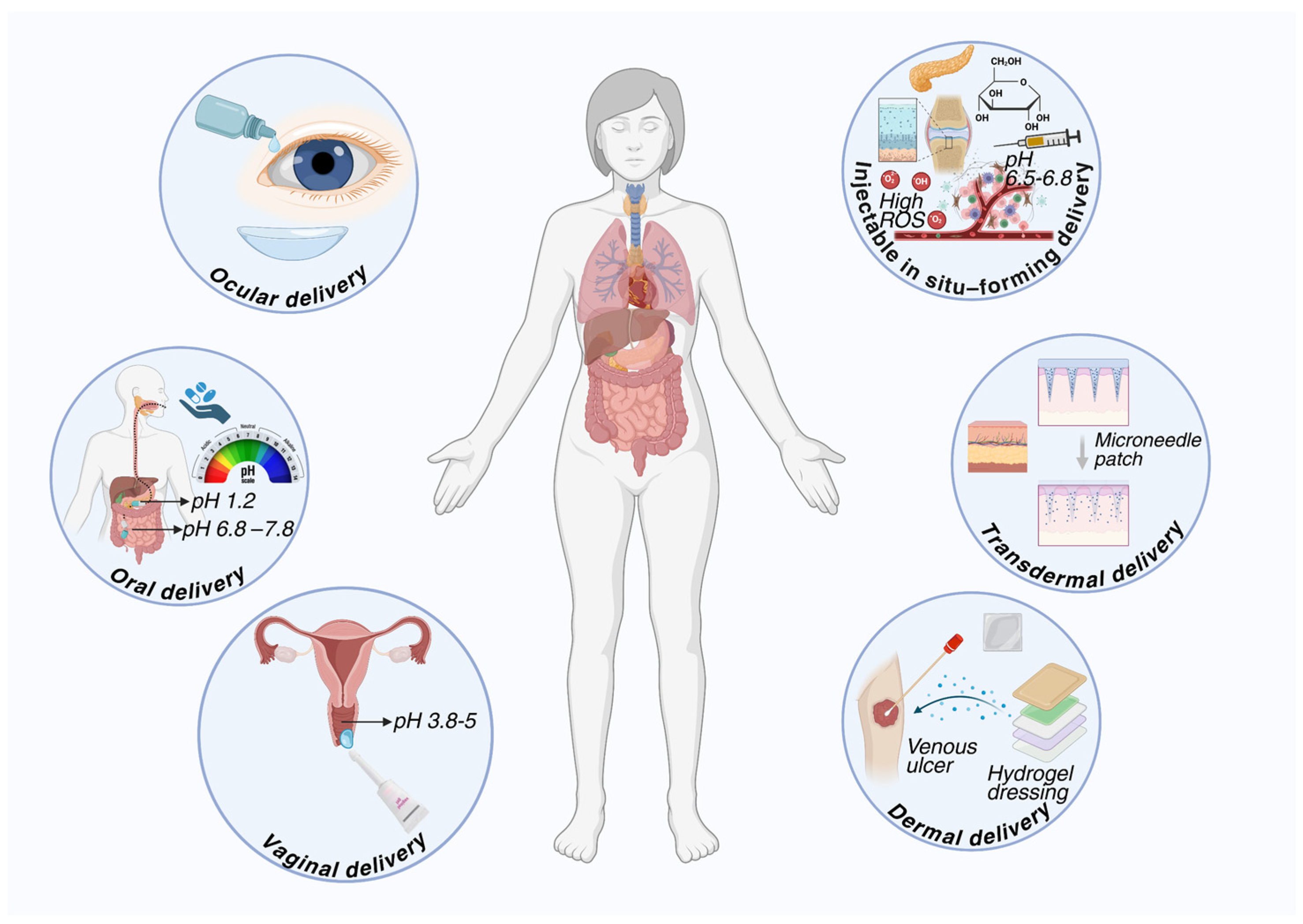


| Material | Class | Relevant Properties for Drug Delivery Systems (DDSs) | Type of Response/Functionality | Typical Applications in Drug Delivery | Main Limitations | Ref. |
|---|---|---|---|---|---|---|
| Chitosan | Natural Polysaccharide | Cationic, Mucoadhesive Biodegradable Antibacterial activity, Biocompatible | pH-responsive, Thermo-responsive, Paracellular permeability | Controlled oral administration (proteins, peptides, insulin, anticancer drugs), Injectable, Wound healing | Low solubility at neutral pH, limited mechanical strength, Low thermal stability, high batch-to-batch variability | [101] |
| Alginate | Natural Polysaccharide | Anionic, ionic gelation (“egg-box”), Biocompatible | pH-dependent swelling, Sustained release, Ionic strength | Oral administration, Colon-targeted DDS, In situ injectable | Non-enzymatically biodegradable, low mechanical stability, low mechanical strength | [102,103,104] |
| Pectin | Natural Polysaccharide | Anionic, degree-of-methoxylation-dependent gelation, Biocompatible, Biodegradable Stable in acidic environment | pH- and enzyme-responsive (colonic microbiota), Mucoadhesive, High water absorption | Oral administration, Colon-targeted DDS, Platform for antibacterial and anticancer formulations | Modest mechanical strength, structural variability, Limited control over release kinetics | [105] |
| Hyaluronic acid | Natural Polysaccharide | Anionic, bioactive, high water retention, CD44 affinity, Biocompatible, Biodegradable hydrophilic, and viscoelastic | Enzyme (hyaluronidase), ROS- and pH-responsive | Targeted delivery (anticancer), Oral, in situ injectable, transdermal | Accelerated enzymatic degradation in vivo, low mechanical stability | [106,107] |
| Collagen/gelatin | Natural Structural Proteins | Biocompatibility, biodegradability, high porosity, thermo-/chemically gelable, RGD-mediated cell adhesion | Enzyme- and ROS-responsive, in situ gelation | Oral, ocular, in situ injectable administration, DDS for oncological therapy | Structural variability, rapid enzymatic degradation, modest mechanical strength | [108,109] |
| DNA | Nucleic acid (natural biopolymer) | Biocompatibility, biodegradability, complementary sequence programmability, porosity, tunable multifunctionality | pH-responsive, thermo-responsive, sol–gel transitions, programmed volume changes | DDS, biosensing, protein- or nucleic-acid-based therapeutic systems | Uncertain biological stability, uncontrolled drug release, undesired immune response, insufficient in vivo monitoring, reduced predictability, poor reproducibility | [110,111] |
| RNA | Nucleic acid (natural biopolymer) | Biocompatible, biodegradable | pH-responsive, enzyme-responsive, thermo-responsive | Drug release (DDS), gene therapy, vaccine delivery, antitumor therapy | Enzymatic instability, rapid degradation, limited release control, need for vectors, complex formulation, low stability | [112] |
| Lignin | Natural | Biocompatibility, bioregenerability, amorphous, anionic, porous structure | Hydrophilicity, antioxidant, antimicrobial | DDS, tissue scaffold, wound dressings | Requires chemical modification for functionalization, uneven release, structural variability, difficult standardization | [113,114] |
| Poly(ethyleneglycol) (PEG) | Synthetic Polymer | Biocompatible, water-soluble, non-toxic, adjustable molecular weights, easily derivatizable, biologically inert, precise crosslinking control | Controlled diffusion; stimulus-responsive platform | Injectable, Nanocarriers, Protein delivery | Lack of intrinsic bioactivity, potential systemic effects | [115,116] |
| Poly(vinylalcohol)(PVA) | Synthetic Polymer | Hydrophilic, structural stability, hydrogen bonding | Swelling-controlled release; glucose-responsive platform | Oral administration, Smart insulin delivery systems | Low biodegradability | [117,118] |
| Polyurethane | Synthetic Polymer | Biocompatible tunable mechanical properties, block-segmented polymer structure | Thermosensitivity; pH-responsive in specific derivatives | Controlled transdermal release, Active dressings | Potential cytotoxicity of diisocyanates Top of Form Bottom of Form | [119,120,121] |
| Poly(N-isopropylacrylamide) (PNIPAm) | Synthetic Polymer | Critical temperature (LCST) approx. 32 °C | Thermo-responsive, dual response (pH and temperature) | Injectable systems, On-demand DDS | Limited biodegradability, modest loading capacity | [122,123] |
| Material/Form | Swelling Ratio (%) | Degradation Rate | Mechanical Properties (kPa) | Biocompatibility | Release Efficiency (%) | Ref. |
|---|---|---|---|---|---|---|
| Chitosan | 200–400 | Slow–Moderate (enzymatic) | 1–10 | ++++ | 60–80 | [101,134,135] |
| Chitosan/Alginate Hybrid | 300–600 | Moderate (tunable) | 5–30 | ++++ | 70–90 | |
| Alginate | 300–800 | Slow (non-enzymatic) | 0.5–5 | ++++ | 55–75 | [102,103,104,136,137] |
| Alginate/Pectin Hybrid | 400–900 | Moderate (enzyme + pH) | 2–15 | ++++ | 65–85 | |
| Pectin | 400–1000 | Moderate (colonic enzymes) | 0.5–8 | ++++ | 60–80 | [105,138,139] |
| Pectin/Chitosan Hybrid | 350–800 | Moderate–Slow (tunable) | 5–20 | ++++ | 70–88 | |
| Hyaluronic acid (HA) | 500–2000 | Fast (hyaluronidase) | 0.1–2 | ++++ | 50–70 | [106,107,140,141] |
| HA/Collagen Hybrid | 600–1800 | Moderate (tunable crosslinking) | 2–20 | ++++ | 65–85 | |
| Collagen/ gelatin | 300–700 | Fast (collagenase/MMP) | 0.5–5 | ++++ | 55–75 | [108,109,142,143] |
| Collagen/PEG Hybrid | 400–900 | Moderate (crosslink- dependent) | 5–50 | ++++ | 70–90 | |
| Poly (ethylene glycol) (PEG) | 100–500 | Very slow (hydrolytic) | 1–100 (tunable) | ++++ | 60–85 | [115,116,144,145] |
| PEG/PNIPAm Hybrid | 200–600 | Slow–Moderate | 5–80 | +++ | 75–95 | |
| Poly(vinylalcohol)(PVA) | 100–300 | Very slow (non-biodegradable) | 10–200 | +++ | 50–70 | [117,118,146,147] |
| PVA/ Chitosan Hybrid | 200–500 | Slow–Moderate (partial) | 15–150 | ++++ | 65–85 | |
| Polyurethane | 50–200 | Slow (hydrolytic/oxidative) | 100–10,000 | ++ | 40–65 | [119,120,121,148] |
| Polyurethane/PEG Hybrid | 100–350 | Moderate (tunable) | 50–5000 | +++ | 55–80 | |
| PNIPAm | 100–400 | Slow (non-biodegradable) | 1–20 | ++ | 50–70 | [122,123,149,150] |
| PNIPAm/Alginate Hybrid | 200–600 | Moderate (ionic + thermal) | 5–40 | +++ | 70–90 | |
| DNA | 500–1000 | Tunable via cross-linking | 800–1200 | ++++ | 50–90 | [110,111,112,151,152] |
| Chitosan/Alginate mRNA Hybrid | 300–650 | Slow | 50–200 | ++++ | 42 | |
| Lignin/PVA/Chitosan | 540–900 | Slow | 900 | +++ | 40–95 | [153,154] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ene, R.; Iacob, A.-T.; Fulga, I.; Di Gioia, M.L.; Dragostin, I.; Fulga, A.; Samal, S.K.; Dragostin, O.-M. Hydrogels—Advanced Polymer Platforms for Drug Delivery. Polymers 2026, 18, 709. https://doi.org/10.3390/polym18060709
Ene R, Iacob A-T, Fulga I, Di Gioia ML, Dragostin I, Fulga A, Samal SK, Dragostin O-M. Hydrogels—Advanced Polymer Platforms for Drug Delivery. Polymers. 2026; 18(6):709. https://doi.org/10.3390/polym18060709
Chicago/Turabian StyleEne (Vatcu), Rodica, Andreea-Teodora Iacob, Iuliu Fulga, Maria Luisa Di Gioia, Ionut Dragostin, Ana Fulga, Sangram Keshari Samal, and Oana-Maria Dragostin. 2026. "Hydrogels—Advanced Polymer Platforms for Drug Delivery" Polymers 18, no. 6: 709. https://doi.org/10.3390/polym18060709
APA StyleEne, R., Iacob, A.-T., Fulga, I., Di Gioia, M. L., Dragostin, I., Fulga, A., Samal, S. K., & Dragostin, O.-M. (2026). Hydrogels—Advanced Polymer Platforms for Drug Delivery. Polymers, 18(6), 709. https://doi.org/10.3390/polym18060709










