Effects of Poly-γ-Glutamic Acid Molecular Weight on Lettuce Growth, Soil Properties, and Bacterial Community Structure
Abstract
1. Introduction
2. Materials and Methods
2.1. γ-PGA, Soil and Chemical Materials
2.2. Infiltration Soil Column Experiment
2.3. Design of Pot Experiment
2.4. Assays of Soil Chemical Characteristics and Lettuce Growth
2.5. Analysis of Soil Bacterial Community
2.6. Statistical Analysis
3. Results
3.1. Effect of γ-PGA on Red Soil Infiltration
3.2. Lettuce Traits Analysis
3.3. Effect of γ-PGA on Soil NH4+-N, NO3−-N, AP, and AK Contents
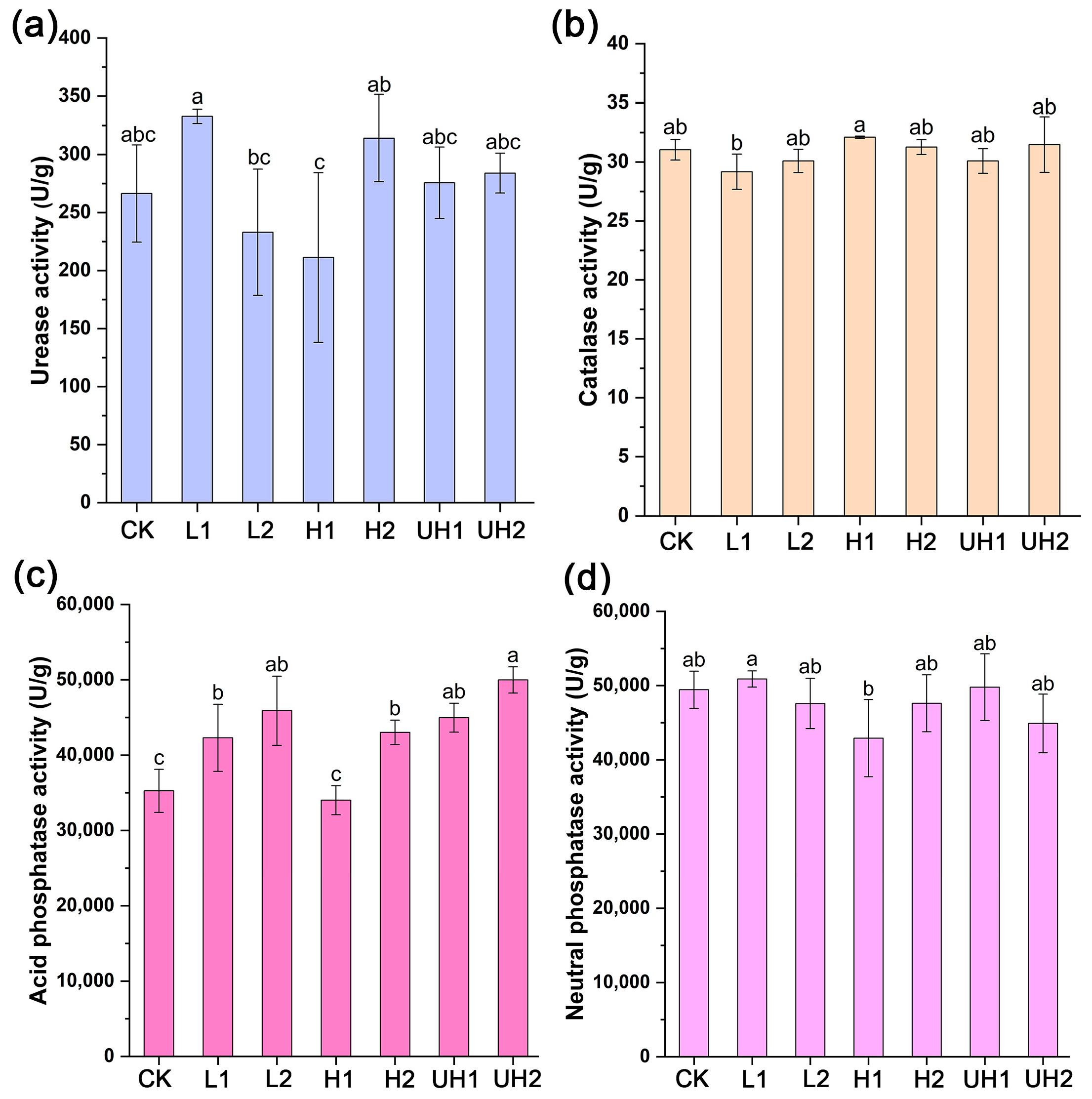
3.4. Soil Enzyme Activities
3.5. Soil Bacterial Diversity
3.6. Similarity Analysis of Soil Bacterial Communities
3.7. Soil Bacterial Community Composition
3.8. Relationship Between the Bacterial Community and Soil Biochemical Factors
4. Discussion
4.1. The Decrease in Soil Infiltration Rates by γ-PGA Application
4.2. Influence of γ-PGAs with Different Mws on Lettuce Growth
4.3. Correlations Between Soil Bacterial Community and Biochemical Properties
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| γ-PGA | Poly-γ-Glutamic Acid |
| Mw | Molecular Weight |
| N | Nitrogen |
| P | Phosphorus |
| K | Potassium |
| Ammonium N | NH4+-N |
| Nitrate N | NO3−-N |
| AP | Available P |
| AN | Available N |
| AK | Available K |
| OTUs | Operational Taxonomic Units |
| PCoA | Principal Co-Ordinates Analysis |
| LDA | Linear Discriminant Analysis |
| LEfSe | LDA Effect Size |
| dbRDA | Distance-Based Redundancy Analysis |
References
- Ashiuchi, M.; Misono, H. Biochemistry and molecular genetics of poly-γ-glutamate synthesis. Appl. Microbiol. Biotechnol. 2002, 59, 9–14. [Google Scholar] [CrossRef]
- Liang, J.; Shi, W. Poly-γ-glutamic acid improves water-stable aggregates, nitrogen and phosphorus uptake efficiency, water-fertilizer productivity, and economic benefit in barren desertified soils of Northwest China. Agric. Water Manag. 2021, 245, 106551. [Google Scholar] [CrossRef]
- Yin, A.; Jia, Y.; Qiu, T.; Gao, M.; Cheng, S.; Wang, X.; Sun, Y. Poly-γ-glutamic acid improves the drought resistance of maize seedlings by adjusting the soil moisture and microbial community structure. Appl. Soil Ecol. 2018, 129, 128–135. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, X.; Gao, D.; Wang, L.; Li, J.; Wei, Z.; Shi, Y. Effects of poly-γ-glutamic acid (γ-PGA) on plant growth and its distribution in a controlled plant-soil system. Sci. Rep. 2017, 7, 6090. [Google Scholar] [CrossRef]
- Bai, N.; Zhang, H.; Li, S.; Zheng, X.; Zhang, J.; Sun, L.; Lv, W. Effects of application rates of poly-γ-glutamic acid on vegetable growth and soil bacterial community structure. Appl. Soil Ecol. 2020, 147, 103405. [Google Scholar] [CrossRef]
- Zhang, L.; Gao, J.; Wang, L.; Sun, Y.; Dong, X.; Pei, J.; Wei, Z.; Zhang, L.; Shi, Y. Poly-γ-glutamic acid differentially alters the abundances and communities of N functional genes involved in urea hydrolysis, nitrification and denitrification when applied with different nitrogen fertilizers. Appl. Soil Ecol. 2023, 190, 105015. [Google Scholar] [CrossRef]
- Bai, N.; Zhang, H.; He, Y.; Zhang, J.; Zheng, X.; Zhang, H.; Zhang, Y.; Lv, W.; Li, S. Effects of Bacillus subtilis A-5 and its fermented γ-polyglutamic acid on the rhizosphere bacterial community of Chinese cabbage. Front. Microbiol. 2022, 13, 954489. [Google Scholar] [CrossRef]
- Hong, L.; Wei, L.; Fanglan, G.; Jiao, L.; Shiheng, T.; Hong, Y.; Yao, R.; Xinyue, G.; Can, Y. Unveiling the regulatory mechanism of poly-γ-glutamic acid on soil characteristics under drought stress through integrated metagenomics and metabolomics analysis. Front. Microbiol. 2024, 15, 1387223. [Google Scholar] [CrossRef]
- Xu, Z.; Wan, C.; Xu, X.; Feng, X.; Xu, H. Effect of poly (γ-glutamic acid) on wheat productivity, nitrogen use efficiency and soil microbes. J. Soil Sci. Plant Nut. 2013, 13, 744–755. [Google Scholar] [CrossRef]
- Jing, B.; Shi, W.; Liu, L.; Wang, Y. Poly-γ-glutamic acid improved biological nitrogen fixation, water-nitrogen productivity, and nitrate residue in cotton/soybean intercropping. J. Sci. Food Agric. 2023, 103, 7284–7292. [Google Scholar] [CrossRef]
- Zhai, Z.; Shi, W.; Liu, L.; Jing, B. Poly-γ-glutamic acid enhances corn nitrogen use efficiency and yield by decreasing gaseous nitrogen loss and increasing mineral nitrogen accumulation. Soil Tillage Res. 2025, 249, 106480. [Google Scholar] [CrossRef]
- Zhao, C.; Zhang, Y.; Wei, X.; Hu, Z.; Zhu, F.; Xu, L.; Luo, M.; Liu, H. Production of ultra-high molecular weight poly-γ-glutamic acid with Bacillus licheniformis P-104 and characterization of its flocculation properties. Appl. Biochem. Biotechnol. 2013, 170, 562–572. [Google Scholar] [CrossRef] [PubMed]
- Sha, Y.; Huang, Y.; Zhu, Y.; Sun, T.; Luo, Z.; Qiu, Y.; Zhan, Y.; Lei, P.; Li, S.; Xu, H. Efficient biosynthesis of low-molecular-weight poly-γ-glutamic acid based on stereochemistry regulation in Bacillus amyloliquefaciens. ACS Synth. Biol. 2020, 9, 1395–1405. [Google Scholar] [CrossRef]
- Shu, S.; Sha, X.M.; Hu, Z.Z.; Ma, Q.L.; Qiao, J.J.; Fang, T.; Jiang, W.L.; Tu, Z.C. Improving gelling properties of fish gelatin by γ-polyglutamic acid with four different molecular weights. Int. J. Food Sci. Technol. 2023, 58, 6588–6597. [Google Scholar] [CrossRef]
- Qiao, M.; Zhang, T.; Miao, M. Minced beef meat paste characteristics: Gel properties, water distribution, and microstructures regulated by medium molecular mass of γ-poly-glutamic acid. Foods 2024, 13, 510. [Google Scholar] [CrossRef]
- Zhang, L.; Wei, Z.; Wang, L.; Sun, Y.; Pei, J.; Wang, J.; Gao, J.; Zhang, L.; Shi, Y. Fate of urea and ammonium sulfate in the plant and soil system as affected by poly-γ-glutamic acid. J. Soil Sci. Plant Nut. 2022, 22, 2457–2468. [Google Scholar] [CrossRef]
- Shi, W.; Yue, X.; Liang, J.; Wen, L.; Li, J.; Li, N.; Lu, Y. Poly-γ-glutamic acid enhances the wheat yield, water use efficiency and soil physicochemical properties of the arid area in the Northwest China. Arch. Agron. Soil Sci. 2023, 69, 2663–2676. [Google Scholar] [CrossRef]
- Guo, J.; Shi, W.; Wen, L.; Shi, X.; Li, J. Effects of a super-absorbent polymer derived from poly-γ-glutamic acid on water infiltration, field water capacity, soil evaporation, and soil water-stable aggregates. Arch. Agron. Soil Sci. 2019, 66, 1627–1638. [Google Scholar] [CrossRef]
- Guo, J.; Shi, W.; Li, J.; Zhai, Z. Effects of poly-γ-glutamic acid and poly-γ-glutamic acid super absorbent polymer on the sandy loam soil hydro-physical properties. PLoS ONE 2021, 16, e0245365. [Google Scholar] [CrossRef]
- Regestein née Meissner, L.; Arndt, J.; Palmen, T.G.; Jestel, T.; Mitsunaga, H.; Fukusaki, E.; Büchs, J. Investigation of poly(γ-glutamic acid) production via online determination of viscosity and oxygen transfer rate in shake flasks. J. Biol. Eng. 2017, 11, 23. [Google Scholar] [CrossRef]
- Siebielec, S.; Siebielec, G.; Klimkowicz-Pawlas, A.; Gałązka, A.; Grządziel, J.; Stuczyński, T. Impact of water stress on microbial community and activity in sandy and loamy soils. Agronomy 2020, 10, 1429. [Google Scholar] [CrossRef]
- Zeng, W.; Liu, Y.; Shu, L.; Guo, Y.; Wang, L.; Liang, Z. Production of ultra-high-molecular-weight poly-γ-glutamic acid by a newly isolated Bacillus subtilis strain and genomic and transcriptomic analyses. Biotechnol. J. 2024, 19, 2300614. [Google Scholar] [CrossRef]
- Kandeler, E.; Gerber, H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol. Fertil. Soils 1988, 6, 68–72. [Google Scholar] [CrossRef]
- Orta-Zavalza, E.; Briones-Martin-del-Campo, M.; Castano, I.; Penas, A.D.L. Catalase activity assay in Candida glabrata. Bio Protoc. 2014, 4, e1072. [Google Scholar] [CrossRef]
- He, Z.; Zhang, M.; Wilson, M.J. Distribution and Classification of Red Soils in China. In The Red Soils of China: Their Nature, Management and Utilization; Wilson, M.J., He, Z., Yang, X., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 29–33. [Google Scholar]
- Baligar, V.C.; Fageria, N.K.; Eswaran, H.; Wilson, M.J.; He, Z. Nature and Properties of Red Soils of the World. In The Red Soils of China: Their Nature, Management and Utilization; Wilson, M.J., He, Z., Yang, X., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 7–27. [Google Scholar]
- Dong, W.; Zhang, X.; Wang, H.; Dai, X.; Sun, X.; Qiu, W.; Yang, F. Effect of different fertilizer application on the soil fertility of paddy soils in red soil region of Southern China. PLoS ONE 2012, 7, e44504. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Zhou, Y.; Shen, Q.; Zhang, F. Effect of ammonium and nitrate nutrition on some physiological processes in higher plants-growth, photosynthesis, photorespiration, and water relations. Plant Biol. 2008, 9, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Lei, P.; Feng, X.; Xu, X.; Xu, H.; Yang, H.; Tang, W. Effect of poly(γ-glutamic acid) on microbial community and nitrogen pools of soil. Acta Agric. Scand. Sect. B Soil Plant Sci. 2013, 63, 657–668. [Google Scholar] [CrossRef]
- Ho, G.H.; Ho, T.I.; Hsieh, K.H.; Su, Y.C.; Lin, P.Y.; Yang, J.; Yang, K.H.; Yang, S.C. γ-Polyglutamic acid produced by Bacillus subtilis (Natto): Structural characteristics, chemical properties and biological functionalities. J. Chin. Chem. Soc. 2006, 53, 1363–1384. [Google Scholar] [CrossRef]
- Wang, H.; Yan, Z.; Chen, Z.; Song, X.; Zhang, J.; Li, S.-L.; Müller, C.; Ju, X.; Zhu-Barker, X. Microbial ammonium immobilization promoted soil nitrogen retention under high moisture conditions in intensively managed fluvo-aquic soils. Biol. Fertil. Soils 2024, 61, 641–651. [Google Scholar] [CrossRef]
- Yan, Z.; Bond-Lamberty, B.; Todd-Brown, K.E.; Bailey, V.L.; Li, S.; Liu, C.; Liu, C. A moisture function of soil heterotrophic respiration that incorporates microscale processes. Nat. Commun. 2018, 9, 2562. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, J.; Zhang, K.; Li, S.; Zhang, Y. Effect of γ-PGA and γ-PGA SAP on soil microenvironment and the yield of winter wheat. PLoS ONE 2023, 18, e0288299. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, K.; Halmschlag, B.; Briel, S.; Sieben, M.; Putri, S.; Fukusaki, E.; Blank, L.M.; Büchs, J. Online measurement of the viscosity in shake flasks enables monitoring of γ-PGA production in depolymerase knockout mutants of Bacillus subtilis with the phosphat-starvation inducible promoter Ppst. Biotechnol. Prog. 2022, 39, e3293. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, Z.; Wang, Y.; Cheng, W.; Mou, H. Production of a water-soluble fertilizer containing amino acids by solid-state fermentation of soybean meal and evaluation of its efficacy on the rapeseed growth. J. Biotechnol. 2014, 187, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Liu, G.; Xue, S.; Wang, G. Soil bacterial community dynamics reflect changes in plant community and soil properties during the secondary succession of abandoned farmland in the Loess Plateau. Soil Biol. Biochem. 2016, 97, 40–49. [Google Scholar] [CrossRef]
- You, T.; Liu, D.; Chen, J.; Yang, Z.; Dou, R.; Gao, X.; Wang, L. Effects of metal oxide nanoparticles on soil enzyme activities and bacterial communities in two different soil types. J. Soils Sediments 2017, 18, 211–221. [Google Scholar] [CrossRef]
- Janssen Peter, H. Identifying the dominant soil bacterial taxa in libraries of 16S rRNA and 16S rRNA genes. Appl. Environ. Microbiol. 2006, 72, 1719–1728. [Google Scholar] [CrossRef]
- Kersters, K.; De Vos, P.; Gillis, M.; Swings, J.; Vandamme, P.; Stackebrandt, E. Introduction to the Proteobacteria. In The Prokaryotes: Volume 5: Proteobacteria: Alpha and Beta Subclasses; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 3–37. [Google Scholar]
- Hu, X.; Liu, X.; Qiao, L.; Zhang, S.; Su, K.; Qiu, Z.; Li, X.; Zhao, Q.; Yu, C. Study on the spatial distribution of ureolytic microorganisms in farmland soil around tailings with different heavy metal pollution. Sci. Total Environ. 2021, 775, 144946. [Google Scholar] [CrossRef]
- Krzmarzick, M.J.; Crary, B.B.; Harding, J.J.; Oyerinde, O.O.; Leri, A.C.; Myneni, S.C.B.; Novak, P.J. Natural niche for organohalide-respiring Chloroflexi. Appl. Environ. Microbiol. 2012, 78, 393–401. [Google Scholar] [CrossRef]
- Hug, L.A.; Castelle, C.J.; Wrighton, K.C.; Thomas, B.C.; Sharon, I.; Frischkorn, K.R.; Williams, K.H.; Tringe, S.G.; Banfield, J.F. Community genomic analyses constrain the distribution of metabolic traits across the Chloroflexi phylum and indicate roles in sediment carbon cycling. Microbiome 2013, 1, 22. [Google Scholar] [CrossRef]





| Treatments | CK | L1 | L2 | H1 | H2 | UH1 | UH2 |
|---|---|---|---|---|---|---|---|
| γ-PGA rate (w:w) | 0 | 0.1% | 0.2% | 0.1% | 0.2% | 0.1% | 0.2% |
| Treatments | Shannon | Simpson | ACE | Chao1 | Coverage |
|---|---|---|---|---|---|
| CK | 5.289 a | 0.012 c | 1557 a | 1505 a | 0.985 |
| L1 | 4.394 b | 0.034 a | 995 b | 978 b | 0.99 |
| L2 | 5.213 a | 0.014 c | 1552 a | 1430 a | 0.986 |
| H1 | 4.45 b | 0.033 ab | 1207 b | 1115 b | 0.989 |
| H2 | 4.517 b | 0.029 b | 1111 b | 1085 b | 0.989 |
| UH1 | 5.289 a | 0.013 c | 1523 a | 1489 a | 0.985 |
| UH2 | 5.191 a | 0.015 c | 1529 a | 1458 a | 0.985 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lin, Y.; Wang, L.; Shu, L.; Chen, H.; Liang, Z.; Zeng, W. Effects of Poly-γ-Glutamic Acid Molecular Weight on Lettuce Growth, Soil Properties, and Bacterial Community Structure. Polymers 2026, 18, 640. https://doi.org/10.3390/polym18050640
Lin Y, Wang L, Shu L, Chen H, Liang Z, Zeng W. Effects of Poly-γ-Glutamic Acid Molecular Weight on Lettuce Growth, Soil Properties, and Bacterial Community Structure. Polymers. 2026; 18(5):640. https://doi.org/10.3390/polym18050640
Chicago/Turabian StyleLin, Yu, Linye Wang, Lin Shu, Huizhen Chen, Zhiqun Liang, and Wei Zeng. 2026. "Effects of Poly-γ-Glutamic Acid Molecular Weight on Lettuce Growth, Soil Properties, and Bacterial Community Structure" Polymers 18, no. 5: 640. https://doi.org/10.3390/polym18050640
APA StyleLin, Y., Wang, L., Shu, L., Chen, H., Liang, Z., & Zeng, W. (2026). Effects of Poly-γ-Glutamic Acid Molecular Weight on Lettuce Growth, Soil Properties, and Bacterial Community Structure. Polymers, 18(5), 640. https://doi.org/10.3390/polym18050640





