Taming Waste Heterogeneity for Plastics Circularity with Optimized Sample Preparation Protocols for Quality Assessment
Abstract
1. Introduction
2. Materials and Methods
2.1. Polymeric Materials and Model Waste Composition
2.2. Theoretical Approach to Evaluate Sample Preparation Techniques
2.3. Preparation of the Model Waste Samples
2.4. Characterization
2.4.1. Attenuated Total Reflectance (ATR) Fourier Transform Infrared (FTIR) Spectroscopy
2.4.2. High-Performance Liquid Chromatography Coupled to Mass Spectrometry (HPLC-MS)
2.4.3. X-Ray Fluorescence Spectrometry (XRF)
2.4.4. Gel Permeation Chromatography (GPC)
2.4.5. Melt Flow Rate (MFR)
2.4.6. Thermal Properties
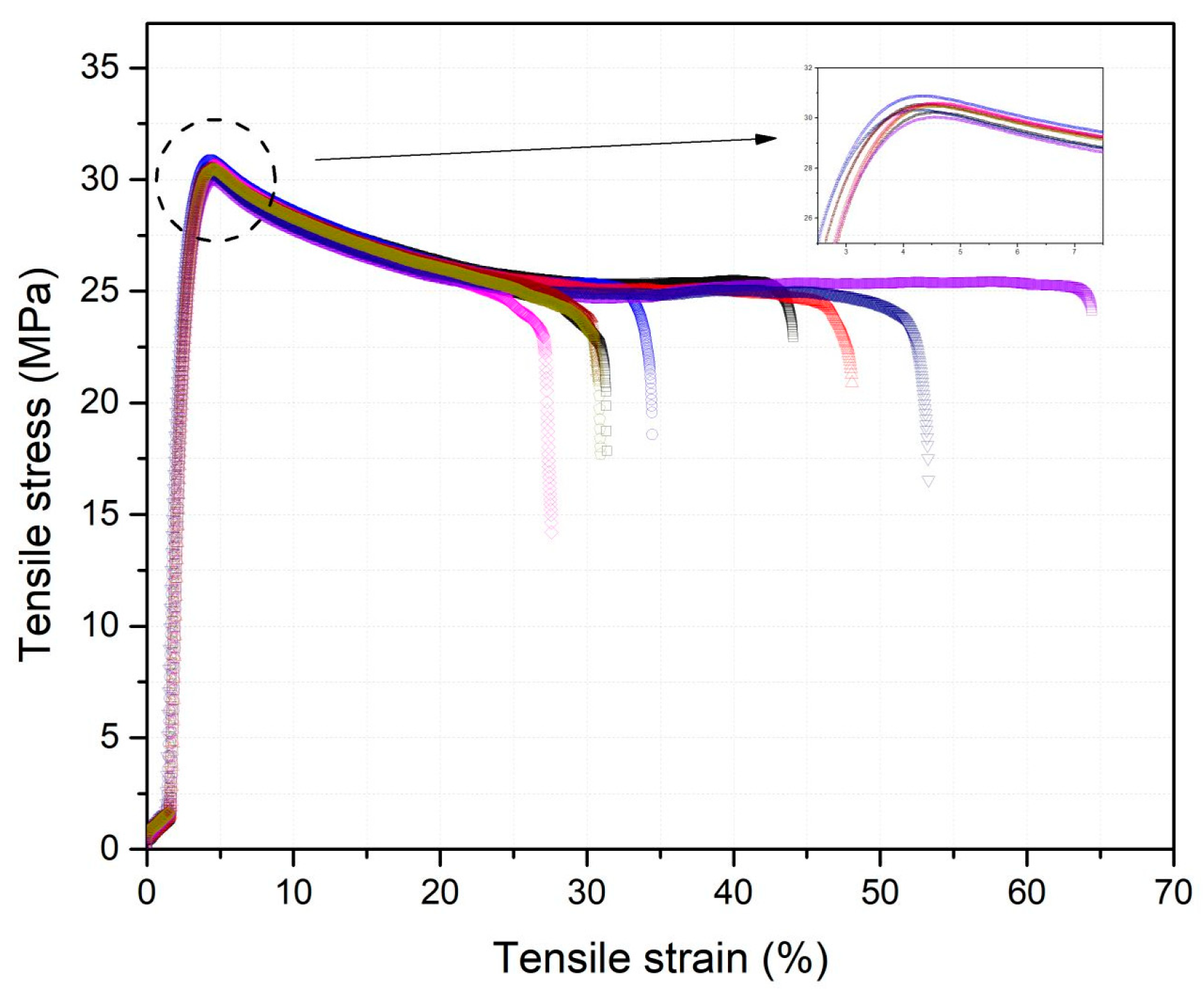
2.4.7. Mechanical Properties—Uniaxial Tensile Testing
2.4.8. X-Ray Micro-Computed Tomography (mCT)
3. Results and Discussion
3.1. Challenges of Sampling and Sample Preparation in Plastics Recycling—PP Rich Waste
3.2. Theoretical Approach to Evaluate Sample Preparation Techniques—ABS Rich Waste
3.3. Experimental Approach to Evaluate Sample Preparation Techniques—ABS Rich Waste
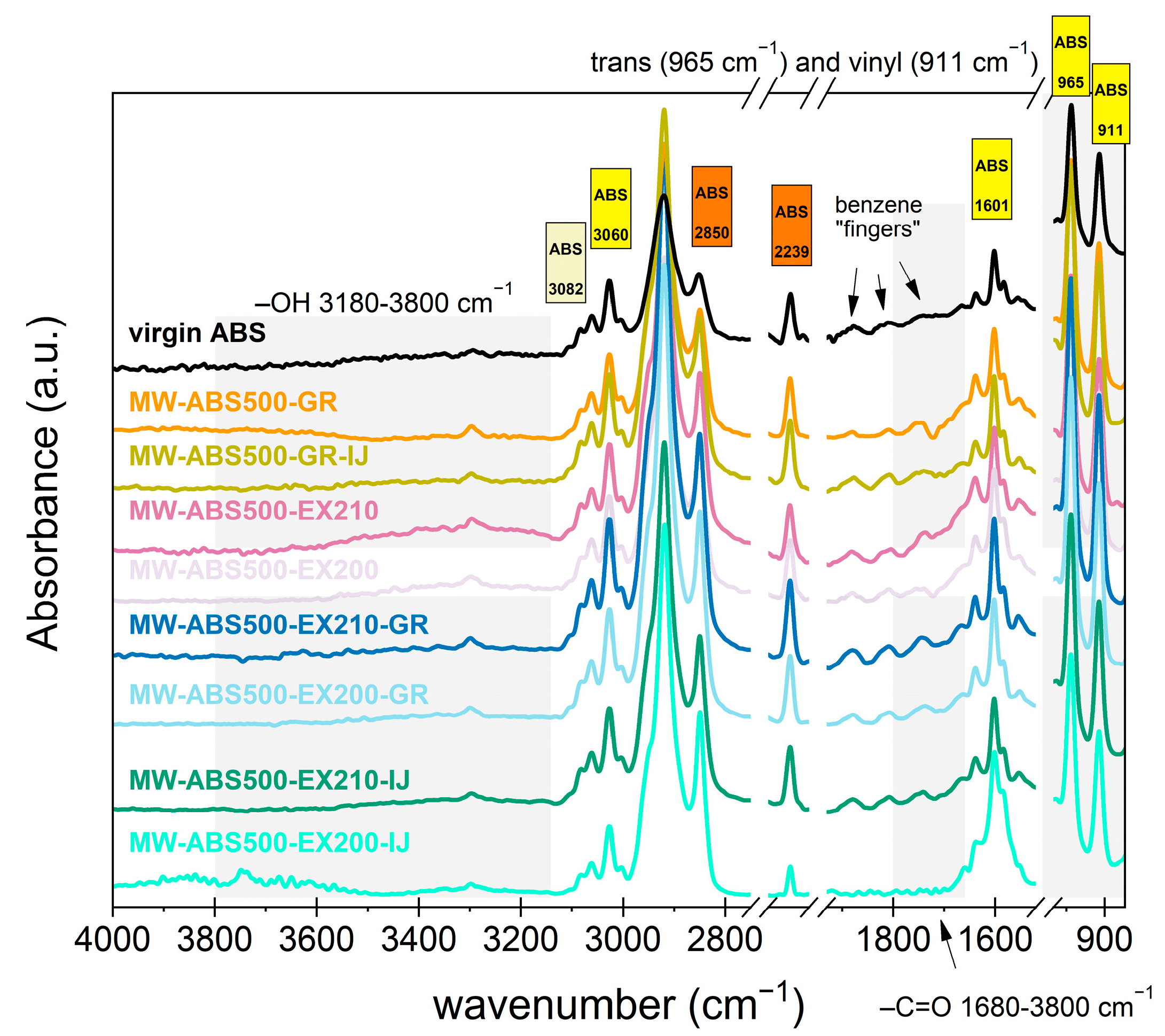
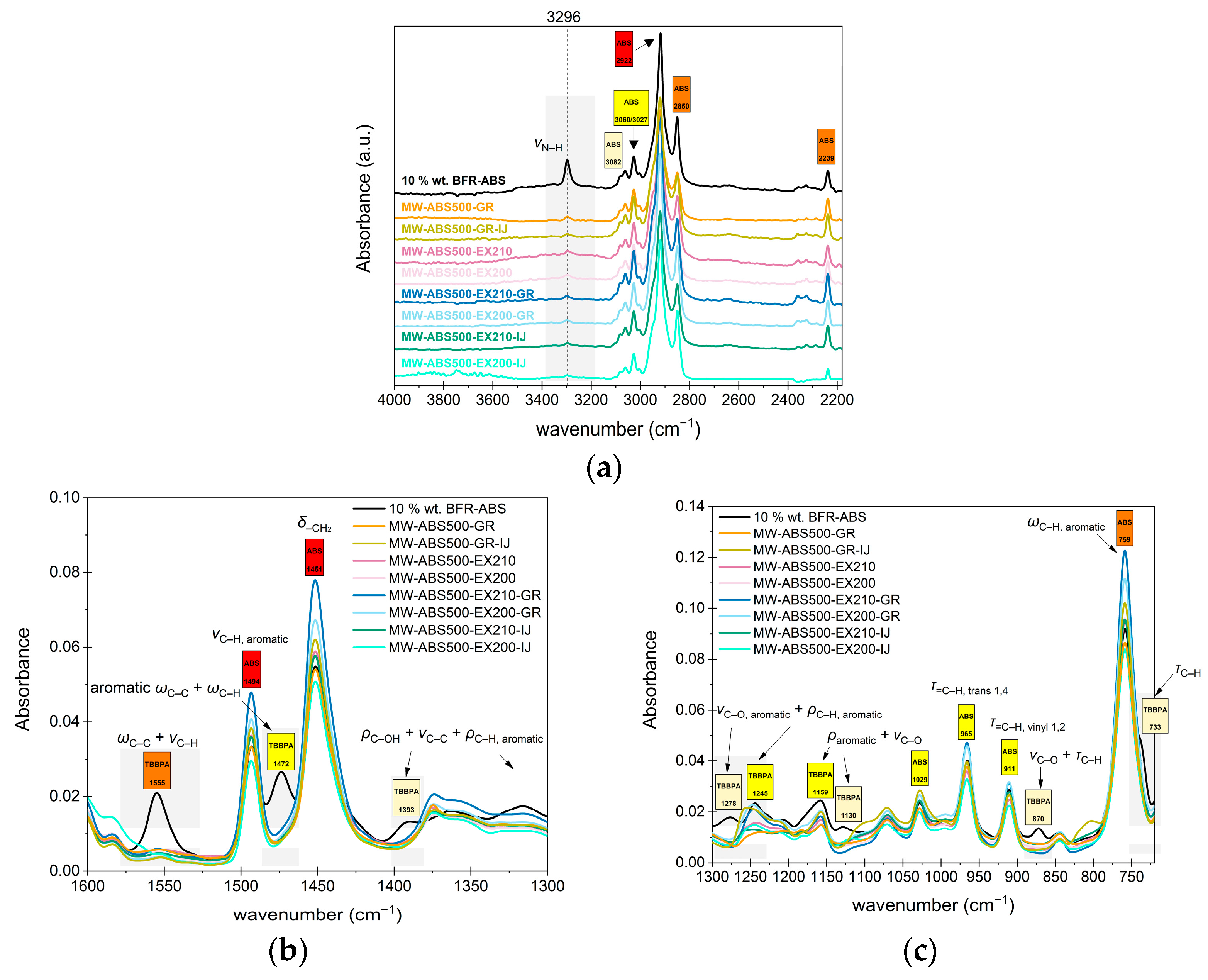
3.3.1. Evaluate Possible Thermomechanical Degradation of Samples
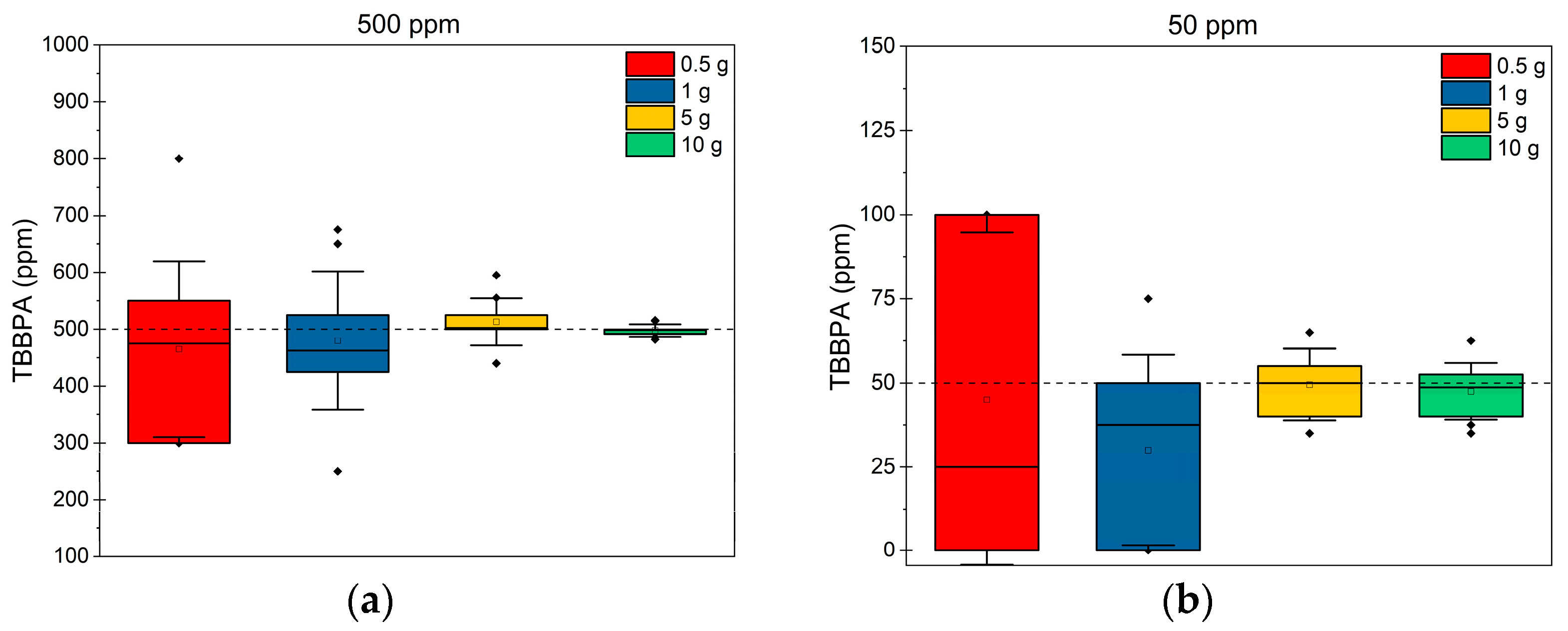
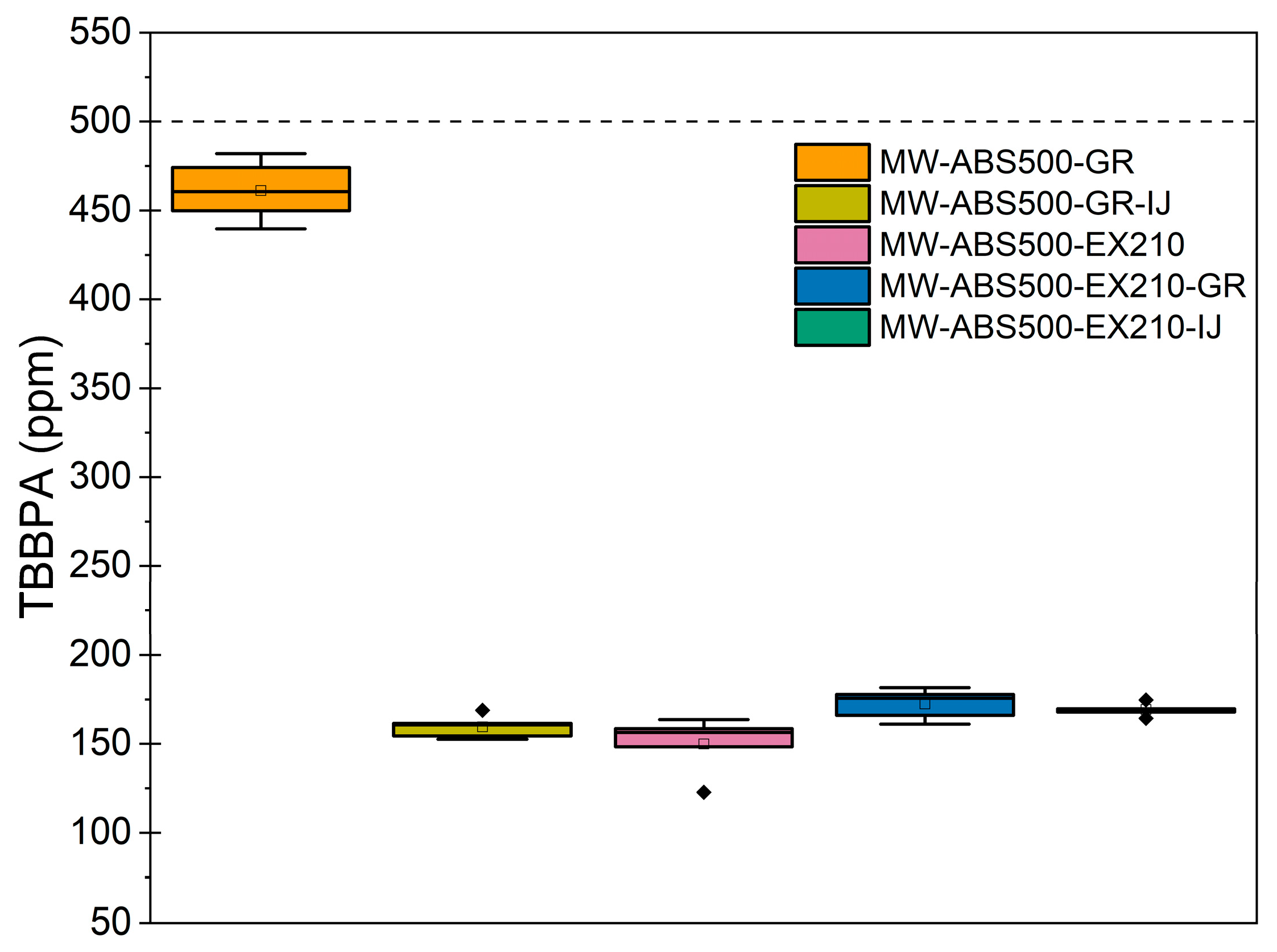
3.3.2. Evaluate the Efficiency to Define Content of Undesirable Additives (Non-Polymeric Contaminant)—TBBPA Content
3.3.3. Evaluate the Efficiency to Define Content of Polymeric Contaminants—HIPS, PP, HDPE Contents
3.3.4. Evaluate the Efficiency to Define Melt Processability
3.4. Establishing Proper Homogenization Procedures for Each Analytical Technique—Proof of Concept for the PP-Rich Waste
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Pottinger, A.S.; Geyer, R.; Biyani, N.; Martinez, C.C.; Nathan, N.; Morse, M.R.; Liu, C.; Hu, S.; De Bruyn, M.; Boettiger, C.; et al. Pathways to Reduce Global Plastic Waste Mismanagement and Greenhouse Gas Emissions by 2050. Science 2024, 386, 1168–1173. [Google Scholar] [CrossRef] [PubMed]
- Mangold, H. The Frontier of Plastics Recycling: Rethinking Waste as a Resource for High-Value Applications. Macromol. Chem. Phys. 2022, 223, 2100488. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, Use, and Fate of All Plastics Ever Made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Plastics—The Fast Facts 2024. Plastics Europe. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-fast-facts-2024/ (accessed on 20 March 2025).
- Golkaram, M.; Demets, R.; Vogels, J.T.W.E.; Urbanus, J.H.; Christoula, A.; Elbing, R.; Meester, S.D.; Ragaert, K. RecyQMeter: Application-Specific Quality of Recycled Plastics. Waste Manag. 2025, 202, 114846. [Google Scholar] [CrossRef]
- Pietrasik, M.; Wilbik, A.; Damoiseaux, Y.; Derks, T.; Karambiri, E.; de Koster, S.; van de Velde, D.; Ragaert, K.; Teng, S.Y. Capturing Variability in Material Property Predictions for Plastics Recycling via Machine Learning. Digit. Chem. Eng. 2025, 15, 100239. [Google Scholar] [CrossRef]
- Roosen, M.; Tonini, D.; Albizzati, P.F.; Caro, D.; Cristóbal, J.; Lase, I.S.; Ragaert, K.; Dumoulin, A.; De Meester, S. Operational Framework to Quantify “Quality of Recycling” across Different Material Types. Environ. Sci. Technol. 2023, 57, 13669–13680. [Google Scholar] [CrossRef]
- CEN/TR 15310-1:2006; Characterization of Waste—Sampling of Waste Materials—Part 1: Guidance on Selection and Application of Criteria for Sampling under Various Conditions. European Committee for Standardization: Brussels, Belgium, 2006.
- CEN/TR 15310-2:2006; Characterization of Waste—Sampling of Waste Materials—Part 2: Guidance on Sampling Techniques. European Committee for Standardization: Brussels, Belgium, 2006.
- CEN/TR 15310-3:2006; Characterization of Waste—Sampling of Waste Materials—Part 3: Guidance on Procedures for Sub-Sampling in the Field. European Committee for Standardization: Brussels, Belgium, 2006.
- CEN/TR 15310-4:2006; Characterization of Waste—Sampling of Waste Materials—Part 4: Guidance on Procedures for Sample Packaging, Storage, Preservation, Transport and Delivery. European Committee for Standardization: Brussels, Belgium, 2006.
- CEN/TR 15310-5:2006; Characterization of Waste—Sampling of Waste Materials—Part 5: Guidance on the Process of Defining the Sampling Plan. European Committee for Standardization: Brussels, Belgium, 2006.
- CEN/TS 16010:2020; Plastics—Recycled Plastics—Sampling Procedures for Testing Plastics Waste and Recyclates. European Committee for Standardization: Brussels, Belgium, 2020.
- CEN/TS 16011:2013; Plastics—Recycled Plastics—Sample Preparation. European Committee for Standardization: Brussels, Belgium, 2013.
- EN 14899:2005; Characterization of Waste—Sampling of Waste Materials—Framework for the Preparation and Application of a Sampling Plan. European Committee for Standardization: Brussels, Belgium, 2006.
- ASTM D4687-14; Standard Guide for General Planning of Waste Sampling. ASTM International: West Conshohocken, PA, USA, 2022.
- ASTM D7204-23; Standard Practice for Sampling Waste Streams on Conveyors. ASTM International: West Conshohocken, PA, USA, 2023.
- Directive 2009/48/EC of the European Parliament and of the Council of 18 June 2009 on the Safety of Toys (OJ L 170); The European Parliament and the Council of the European Union: Strasbourg, France, 2022.
- Directive 2011/65/EU of the European Parliament and of the Council of 8 June 2011 on the Restriction of the Use of Certain Hazardous Substances in Electrical and Electronic Equipment (OJ L 174); The European Parliament and the Council of the European Union: Strasbourg, France, 2014.
- OEKO-TEX® Standard 100 (Edition 01.2024); OEKO-TEX®: Zürich, Switzerland, 2024.
- Regulation (EC) No 1935/2004 of the European Parliament and of the Council of 27 October 2004 on Materials and Articles Intended to Come into Contact with Food (OJ L 338); The European Parliament and the Council of the European Union: Strasbourg, France, 2021.
- Berwald, A.; Dimitrova, G.; Feenstra, T.; Onnekink, J.; Peters, H.; Vyncke, G.; Ragaert, K. Design for Circularity Guidelines for the EEE Sector. Sustainability 2021, 13, 3923. [Google Scholar] [CrossRef]
- Scaffaro, R.; Botta, L.; Di Benedetto, G. Physical Properties of Virgin-Recycled ABS Blends: Effect of Post-Consumer Content and of Reprocessing Cycles. Eur. Polym. J. 2012, 48, 637–648. [Google Scholar] [CrossRef]
- Freymond, C.; Guinault, A.; Charbuillet, C.; Fayolle, B. Reprocessing of Polymer Blends from WEEE: A Methodology for Predicting Embrittlement. Polym. Test. 2022, 106, 107458. [Google Scholar] [CrossRef]
- Karahaliou, E.-K.; Tarantili, P.A. Stability of ABS Compounds Subjected to Repeated Cycles of Extrusion Processing. Polym. Eng. Sci. 2009, 49, 2269–2275. [Google Scholar] [CrossRef]
- Lase, I.S.; Ragaert, K.; Dewulf, J.; De Meester, S. Multivariate Input-Output and Material Flow Analysis of Current and Future Plastic Recycling Rates from Waste Electrical and Electronic Equipment:The Case of Small Household Appliances. Resour. Conserv. Recycl. 2021, 174, 105772. [Google Scholar] [CrossRef]
- ISO 527-2:2025; Plastics—Determination of Tensile Properties—Part 2: Test Conditions for Moulding and Extrusion Plastics. International Organization for Standardization: Geneva, Switzerland, 2025.
- ISO 1133-1:2011; Plastics—Determination of the Melt Mass-Flow Rate (MFR) and Melt Volume-Flow Rate (MVR) of Thermoplastics—Part 1: Standard Method. International Organization for Standardization: Geneva, Switzerland, 2011.
- ISO 1133-2:2011; Plastics—Determination of the Melt Mass-Flow Rate (MFR) and Melt Volume-Flow Rate (MVR) of Thermoplastics—Part 2: Method for Materials Sensitive to Time-Temperature History and/or Moisture. International Organization for Standardization: Geneva, Switzerland, 2011.
- ISO 19062-2:2019; Plastics—Acrylonitrile-Butadiene-Styrene (ABS) Moulding and Extrusion Materials—Part 2: Preparation of Test Specimens and Determination of Properties. International Organization for Standardization: Geneva, Switzerland, 2019.
- Kouranou, D.; Galanopoulou, C.; Korres, D.M.; Tzani, A.; Detsi, A.; Vouyiouka, S. Restabilization of Post-Consumer Recycled Polypropylene with Natural Antioxidants from Spent Coffee Residue. J. Appl. Polym. Sci. 2024, 142, e56494. [Google Scholar] [CrossRef]
- Krejčová, A.; Pouzar, M.; Černohorský, T.; Pešková, K. The Cryogenic Grinding as the Important Homogenization Step in Analysis of Inconsistent Food Samples. Food Chem. 2008, 109, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Shimada, J.; Kabuki, K. The Mechanism of Oxidative Degradation of ABS Resin. Part I. The Mechanism of Thermooxidative Degradation. J. Appl. Polym. Sci. 1968, 12, 655–669. [Google Scholar] [CrossRef]
- Shimada, J.; Kabuki, K. The Mechanism of Oxidative Degradation of ABS Resin. Part II. The Mechanism of Photooxidative Degradation. J. Appl. Polym. Sci. 1968, 12, 671–682. [Google Scholar] [CrossRef]
- Signoret, C.; Caro-Bretelle, A.-S.; Lopez-Cuesta, J.-M.; Ienny, P.; Perrin, D. Alterations of Plastics Spectra in MIR and the Potential Impacts on Identification towards Recycling. Resour. Conserv. Recycl. 2020, 161, 104980. [Google Scholar] [CrossRef]
- Desrousseaux, C.; Cueff, R.; Aumeran, C.; Garrait, G.; Mailhot-Jensen, B.; Traoré, O.; Sautou, V. Fabrication of Acrylonitrile-Butadiene-Styrene Nanostructures with Anodic Alumina Oxide Templates, Characterization and Biofilm Development Test for Staphylococcus Epidermidis. PLoS ONE 2015, 10, e0135632. [Google Scholar] [CrossRef]
- Roos, Y.H.; Drusch, S. Physical State and Molecular Mobility. In Phase Transitions in Foods; Elsevier: Amsterdam, The Netherlands, 2016; pp. 19–47. [Google Scholar]
- Hammer, C.F. Polymeric Plasticizers. In Polymer Blends; Elsevier: Amsterdam, The Netherlands, 1978; pp. 219–241. [Google Scholar]
- Langer, E.; Bortel, K.; Waskiewicz, S.; Lenartowicz-Klik, M. Essential Quality Parameters of Plasticizers. In Plasticizers Derived from Post-Consumer PET; Elsevier: Amsterdam, The Netherlands, 2020; pp. 45–100. [Google Scholar]
- Wypych, G. Effect of Plasticizers on Properties of Plasticized Materials. In Handbook of Plasticizers; Elsevier: Amsterdam, The Netherlands, 2023; pp. 229–311. [Google Scholar]
- Romanova, N.; Shafigullin, L.; Gabdrakhmanov, A.; Buyatova, S. Thermal Properties of Products Based on ABS/PC. MATEC Web Conf. 2019, 298, 00016. [Google Scholar] [CrossRef]
- Oral, M.A.; Ersoy, O.G.; Serhatli, E.İ. Effect of Acrylonitrile–Butadiene–Styrene/Polyethylene Terephthalate Blends on Dimensional Stability, Morphological, Physical and Mechanical Properties and after Aging at Elevated Temperature. J. Plast. Film. Sheeting 2018, 34, 394–417. [Google Scholar] [CrossRef]
- Signoret, C.; Caro-Bretelle, A.-S.; Lopez-Cuesta, J.-M.; Ienny, P.; Perrin, D. MIR Spectral Characterization of Plastic to Enable Discrimination in an Industrial Recycling Context: I. Specific Case of Styrenic Polymers. Waste Manag. 2019, 95, 513–525. [Google Scholar] [CrossRef]
- Boldizar, A.; Möller, K. Degradation of ABS during Repeated Processing and Accelerated Ageing. Polym. Degrad. Stab. 2003, 81, 359–366. [Google Scholar] [CrossRef]
- Smith, P.; McLauchlin, A.; Franklin, T.; Yan, P.; Cunliffe, E.; Hasell, T.; Kurlin, V.; Kerr, C.; Attwood, J.; Shaver, M.P.; et al. A Data-Driven Analysis of HDPE Post-Consumer Recyclate for Sustainable Bottle Packaging. Resour. Conserv. Recycl. 2024, 205, 107538. [Google Scholar] [CrossRef]









| Sample Preparation Technique | Sample | TBBPA (ppm) | Tmelt (°C) |
|---|---|---|---|
| Masterbatch—Extrusion | 10% wt. BFR-ABS | 100,000 | — |
| Cryogenic Grinding (GR) | MW-ABS50(0)-GR | 50(0) | — |
| Cryogenic Grinding (GR) + Injection (IJ) | MW-ABS500-GR-IJ | 500 | — |
| Extrusion (EX) | MW-ABS50(0)-EX210 | 50(0) | 210 |
| MW-ABS500-EX200 | 500 | 200 | |
| Extrusion (EX) + Cryogenic Grinding (GR) | MW-ABS50(0)-EX210-GR | 50(0) | 210 |
| MW-ABS500-EX200-GR | 500 | 200 | |
| Extrusion (EX) + Injection (IJ) | MW-ABS500-EX210-IJ | 500 | 210 |
| MW-ABS500-EX200-IJ | 500 | 200 |
| Element | P | Si | Ca | Ti | Br | Sb | Fe | Zn | Pb | Cd | Cr |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Concentration (ppm) | 92 | 40,814 | 33,656 | 8478 | 416 | 225 | 806 | 197 | 16 | <5 | 16 |
| SD (ppm) | 11 | 6561 | 7879 | 852 | 54 | 48 | 54 | 27 | 5 | 2 | 11 |
| RSD (%) | 11.9 | 16.1 | 23.4 | 10.0 | 12.9 | 21.3 | 6.7 | 13.7 | 31.3 | 40 | 68.8 |
| Sample (xi) | MFR (230 °C, 2.16 kg) g/10 min | ||||
|---|---|---|---|---|---|
| Replicate 1 | Replicate 2 | Replicate 3 | Replicate 4 | Replicate 5 | |
| x1 | 7.7 | 9.1 | 8.7 | 9.1 | 9.9 |
| x2 | 8.8 | 7.2 | 8.1 | 10.5 | 9.4 |
| x3 | 8.3 | 8.0 | 8.7 | 9.4 | 8.7 |
| x4 | 8.7 | 7.9 | 7.5 | 9.6 | 9.3 |
| x5 | 7.8 | 8.8 | 8.4 | 10.0 | 9.7 |
| x6 | 9.3 | 7.6 | 7.9 | 9.9 | 8.6 |
| x7 | 7.9 | 5.9 | 7.8 | 9.2 | 10.1 |
| x8 | 7.7 | 6.8 | 8.3 | 9.9 | 9.6 |
| x9 | 7.6 | 6.5 | 8.0 | 11.0 | 10.4 |
| Average (g/10 min) | 8.2 | 7.5 | 8.2 | 9.8 | 9.5 |
| SD (g/10 min) | 0.6 | 1.1 | 0.4 | 0.6 | 0.6 |
| RSD (%) | 7.3 | 14.0 | 5.0 | 6.3 | 6.2 |
| Max−Min (g/10 min) | 1.7 | 3.2 | 1.2 | 1.9 | 1.7 |
| 15% Average (g/10 min) | 1.2 | 1.1 | 1.2 | 1.5 | 1.4 |
| Specimen (xi) | E (MPa) | σy (MPa) | εy (%) |
|---|---|---|---|
| x1 | 1501 | 26.5 | 6.1 |
| x2 | 1340 | 24.6 | 5.7 |
| x3 | 1524 | 26.3 | 5.9 |
| x4 | 1524 | 27.0 | 6.1 |
| x5 | 1629 | 27.6 | 6.2 |
| x6 | 1589 | 27.4 | 6.4 |
| x7 | 1716 | 28.2 | 6.0 |
| x8 | 1674 | 28.4 | 6.1 |
| x9 | 1411 | 26.6 | 5.9 |
| x10 | 1576 | 26.6 | 5.9 |
| Average | 1548 | 26.9 | 6.0 |
| SD | 115 | 1.1 | 0.2 |
| RSD (%) | 7.4 | 4.1 | 3.5 |
| Sample | (g/mol) | (g/mol) | Đ |
|---|---|---|---|
| ABS (virgin polymer) | 25,800 | 123,000 | 4.8 |
| MW-ABS500-GR | 24,700 | 118,000 | 4.8 |
| MW-ABS500-GR-IJ | 22,600 | 123,000 | 5.4 |
| MW-ABS500-EX210 | 24,200 | 126,000 | 5.2 |
| MW-ABS500-EX200 | 25,100 | 126,000 | 5.0 |
| MW-ABS500-EX210-GR | 24,000 | 126,000 | 5.3 |
| MW-ABS500-EX200-GR | 23,700 | 126,000 | 5.3 |
| MW-ABS500-EX210-IJ | 24,300 | 122,000 | 5.0 |
| Sample | MFR (220 °C, 10 kg) g/10 min | ||||
|---|---|---|---|---|---|
| Average | SD | RSD (%) | Max−Min | 15%∙Average a | |
| MW-ABS500-GR | 67.2 | 3.0 | 4.5 | 5 replicates < 15%∙Average | 4.2–9.4 |
| MW-ABS500-GR-IJ | 53.1 | 2.5 | 4.7 | 5 replicates < 15%∙Average | 1.0–4.2 |
| MW-ABS500-EX210 | 51.9 | 2.0 | 3.9 | 3 replicates < 15%∙Average | 3.3–47.7 |
| MW-ABS500-EX200 | 53.7 | 1.0 | 1.9 | 5 replicates < 15%∙Average | 1.7–7.2 |
| MW-ABS500-EX210-GR | 50.0 | 1.3 | 2.5 | 5 replicates < 15%∙Average | 1.4–3.7 |
| MW-ABS500-EX200-GR | 49.3 | 1.0 | 1.9 | 5 replicates < 15%∙Average | 1.2–2.2 |
| MW-ABS500-EX210-IJ | 55.7 | 4.7 | 8.5 | 4 replicates < 15%∙Average | 1.8–21.6 |
| MW-ABS500-EX200-IJ | 54.5 | 2.2 | 4.0 | 5 replicates < 15%∙Average | 0.9–3.6 |
| Sample Preparation Technique | Analytical Technique | Waste Characteristic Analyzed |
|---|---|---|
| Cryogenic grinding | HPLC-MS | Quantified non-polymeric contaminants |
| Extrusion + Cryogenic grinding | XRF | Elemental composition |
| Extrusion + Injection | MFR DSC Tensile testing | Melt flow behavior and processability Melting and crystallization Mechanical performance |
| All | ATR-FTIR TGA GPC | Detected polymeric contaminants Thermal stability Degradation extent via molecular weight determination |
| Sample (xi) | MFR (230 °C, 2.16 kg) g/10 min | ||||
|---|---|---|---|---|---|
| Replicate 1 | Replicate 2 | Replicate 3 | Replicate 4 | Replicate 5 | |
| x1 | 6.1 | 6.2 | 6.0 | 6.0 | 5.9 |
| x2 | 6.0 | 6.1 | 6.0 | 6.1 | 5.9 |
| x3 | 6.0 | 6.1 | 6.0 | 6.0 | 5.9 |
| x4 | 6.1 | 6.0 | 6.0 | 6.0 | 5.9 |
| x5 | 5.9 | 5.9 | 6.1 | 6.0 | 5.8 |
| x6 | 5.9 | 5.9 | 5.7 | 6.0 | 5.6 |
| x7 | 6.0 | 6.0 | 5.6 | 6.0 | 5.7 |
| x8 | 5.9 | 5.9 | 5.6 | 6.1 | 5.6 |
| x9 | 5.8 | 5.8 | 5.5 | 5.8 | 5.6 |
| Average (g/10 min) | 5.8 | 5.9 | 5.7 | 5.9 | 5.7 |
| SD (g/10 min) | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| RSD (%) | 4.2 | 3.4 | 4.3 | 3.1 | 3.1 |
| Max−Min (g/10 min) | 0.8 | 0.6 | 0.6 | 0.6 | 0.5 |
| 15% Average (g/10 min) | 0.9 | 0.9 | 0.9 | 0.9 | 0.9 |
| Specimen (xi) | σy (MPa) | εy (%) | E (MPa) |
|---|---|---|---|
| x1 | 30.2 | 4.5 | 2270 |
| x2 | 30.0 | 4.2 | 2265 |
| x3 | 30.6 | 4.4 | 2295 |
| x4 | 30.9 | 4.2 | 2270 |
| x5 | 30.6 | 4.5 | 2249 |
| x6 | 30.6 | 4.6 | 2209 |
| x7 | 30.0 | 4.5 | 2161 |
| x8 | 30.3 | 4.2 | 2265 |
| x9 | 30.5 | 4.4 | 2253 |
| x10 | 30.4 | 4.4 | 2249 |
| Average | 30.4 | 4.4 | 2249 |
| SD | 0.3 | 0.2 | 38 |
| RSD (%) | 0.9% | 0.0% | 1.7% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Panagiotopoulos, C.; Podara, C.; Gkartzou, E.; Karamitrou, M.; Kosanovic-Milickovic, T.; Silber, M.; Meyer, L.; von Vacano, B.; Neiva, A.R.C.; Knoop, J.-H.; et al. Taming Waste Heterogeneity for Plastics Circularity with Optimized Sample Preparation Protocols for Quality Assessment. Polymers 2026, 18, 409. https://doi.org/10.3390/polym18030409
Panagiotopoulos C, Podara C, Gkartzou E, Karamitrou M, Kosanovic-Milickovic T, Silber M, Meyer L, von Vacano B, Neiva ARC, Knoop J-H, et al. Taming Waste Heterogeneity for Plastics Circularity with Optimized Sample Preparation Protocols for Quality Assessment. Polymers. 2026; 18(3):409. https://doi.org/10.3390/polym18030409
Chicago/Turabian StylePanagiotopoulos, Christos, Christina Podara, Eleni Gkartzou, Melpo Karamitrou, Tatjana Kosanovic-Milickovic, Mara Silber, Lars Meyer, Bernhard von Vacano, Ana Rita Carvalho Neiva, Jan-Hendrik Knoop, and et al. 2026. "Taming Waste Heterogeneity for Plastics Circularity with Optimized Sample Preparation Protocols for Quality Assessment" Polymers 18, no. 3: 409. https://doi.org/10.3390/polym18030409
APA StylePanagiotopoulos, C., Podara, C., Gkartzou, E., Karamitrou, M., Kosanovic-Milickovic, T., Silber, M., Meyer, L., von Vacano, B., Neiva, A. R. C., Knoop, J.-H., Martínez-García, A., Ibáñez-García, A., Pavlidou, S., Poudeh, L., Charitidis, C. A., & Vouyiouka, S. N. (2026). Taming Waste Heterogeneity for Plastics Circularity with Optimized Sample Preparation Protocols for Quality Assessment. Polymers, 18(3), 409. https://doi.org/10.3390/polym18030409









