Preparation and Application of Cellulose-Based Thermosensitive Polymer in Water-Based Drilling Fluid
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of HAAN
2.3. Characterization
2.3.1. Fourier Transform Infrared (FT-IR)
2.3.2. 1H NMR Spectroscopic Characterization
2.3.3. Thermogravimetric Analysis
2.3.4. Temperature-Sensitive Behavior Test
2.3.5. High Temperature Rheological Property Test
2.4. Conventional Performance Evaluation of Drilling Fluid
2.4.1. API Filtration Performance Evaluation
2.4.2. Rheological Property Evaluation
2.5. Mud Cake Morphology Analysis
3. Results and Discussion
3.1. Optimization of Synthetic Conditions of HAAN
3.2. Characterization of HAAN
3.2.1. FTIR Analysis of HAAN
3.2.2. 1H NMR Analysis of HAAN
3.2.3. Thermal Analysis of HAAN
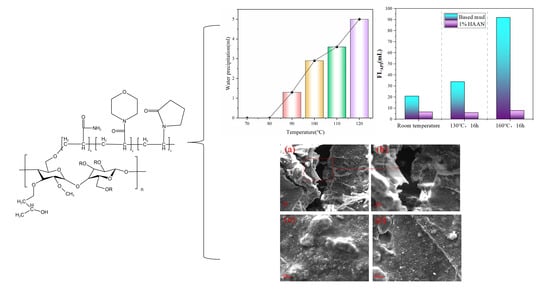
3.2.4. Temperature-Sensitive Behavior
3.2.5. High-Temperature Rheological Analysis
3.2.6. Temperature Tolerance Test
3.3. Seal Performance of HAAN Plugging Agent
3.3.1. Rheological Property Analysis and Filtration Property Analysis
3.3.2. Salt Tolerance Test
3.3.3. Morphology Analysis of Mud Cake
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, T.; Liang, Y.; Wang, Z.; Ji, Q. Dynamic Prediction of Shale Gas Drilling Costs Based on Machine Learning. Appl. Sci. 2024, 14, 10984. [Google Scholar] [CrossRef]
- Wang, Z.C.; Shi, Y.Z.; Wen, L.; Jiang, H.; Jiang, Q.C.; Huang, S.P.; Xie, W.R.; Li, R.; Jin, H.; Zhang, Z.J.; et al. Exploring the potential of oil and gas resources in Sichuan Basin with Super Basin Thinking. Pet. Explor. Dev. 2022, 49, 977–990. [Google Scholar] [CrossRef]
- Feng, G.; Wang, G.F.; Xie, H.Q.; Hu, Y.Q.; Meng, T.; Li, G. A Review of Exploration and Development Technologies for Coal, Oil, and Natural Gas. Energies 2024, 17, 3600. [Google Scholar] [CrossRef]
- Liu, Y.X.; Zhang, Y.J.; Xu, Q.J.; Sun, M.J. The Thinking of Shale Gas Development in China According to the US Shale Gas Development Experiments. Adv. Mater. Res. 2013, 616–618, 939–943. [Google Scholar] [CrossRef]
- Li, L.; Wu, F.; Cao, Y.Y.; Cheng, F.; Wang, D.L.; Li, H.Z.; Yu, Z.Q.; You, J. Sustainable development index of shale gas exploitation in China, the UK, and the US. Environ. Sci. Ecotechnol. 2022, 12, 100202. [Google Scholar] [CrossRef]
- Xu, X.F.; Chen, C.L.; Zhou, Y.; Pan, J.Y.; Song, W.; Zhu, K.L.; Wang, C.H.; Li, S.B. Study of the Wellbore Instability Mechanism of Shale in the Jidong Oilfield under the Action of Fluid. Energies 2023, 16, 2989. [Google Scholar] [CrossRef]
- Guo, B.Y.; Li, G.; Song, J.Z.; Li, J. A feasibility study of gas-lift drilling in unconventional tight oil and gas reservoirs. J. Nat. Gas Sci. Eng. 2017, 37, 551–559. [Google Scholar] [CrossRef]
- He, Y.B.; Jiang, G.C.; Dong, T.F.; Wang, G.S.; He, J.; Dou, H.J.; Du, M.L. Research progress and development tendency of polymer drilling fluid technology for unconventional gas drilling. Front. Energy Res. 2023, 10, 1058412. [Google Scholar] [CrossRef]
- Lai, H.; Lv, S.Z.; Lai, Z.Y.; Liu, L.B.; Lu, Z.Y. Utilization of oil-based mud drilling cuttings wastes from shale gas extraction for cement clinker production. Environ. Sci. Pollut. Res. 2020, 27, 33075–33084. [Google Scholar] [CrossRef]
- Al Moajil, A.M.; Nasr-El-Din, H.A. Formation Damage Caused by Improper Mn3O4-Based Filter-Cake-Cleanup Treatments. J. Can. Pet. Technol. 2013, 52, 64–74. [Google Scholar] [CrossRef]
- Yang, J.; Sun, J.S.; Wang, R.; Qu, Y.Z. Treatment of drilling fluid waste during oil and gas drilling: A review. Environ. Sci. Pollut. Res. 2023, 30, 19662–19682. [Google Scholar] [CrossRef]
- Zhou, S.S.; Song, J.J.; Xu, P.; He, M.; Xu, M.B.; You, F.C. A review on tribology, characterization and lubricants for water-based drilling fluids. Geoenergy Sci. Eng. 2023, 229, 212074. [Google Scholar] [CrossRef]
- Anderson, R.L.; Ratcliffe, I.; Greenwell, H.C.; Williams, P.A.; Cliffe, S.; Coveney, P.V. Clay swelling—A challenge in the oilfield. Earth-Sci. Rev. 2010, 98, 201–216. [Google Scholar] [CrossRef]
- Sadeghalvaad, M.; Sabbaghi, S. The effect of the TiO2/polyacrylamide nanocomposite on water-based drilling fluid properties. Powder Technol. 2015, 272, 113–119. [Google Scholar] [CrossRef]
- Ahmed, H.M.; Kamal, M.S.; Al-Harthi, M. Polymeric and low molecular weight shale inhibitors: A review. Fuel 2019, 251, 187–217. [Google Scholar] [CrossRef]
- Risal, A.R.; Manan, M.A.; Yekeen, N.; Azli, N.B.; Samin, A.M.; Tan, X.K. Experimental investigation of enhancement of carbon dioxide foam stability, pore plugging, and oil recovery in the presence of silica nanoparticles. Pet. Sci. 2019, 16, 344–356. [Google Scholar] [CrossRef]
- Abdullah, A.H.; Ridha, S.; Mohshim, D.F.; Maoinser, M.A. An experimental investigation into the rheological behavior and filtration loss properties of water-based drilling fluid enhanced with a polyethyleneimine-grafted graphene oxide nanocomposite. RSC Adv. 2024, 14, 10431–10444. [Google Scholar] [CrossRef]
- Xiao, Y.R.; Liu, X.J.; Ding, Y.; Liang, L.X.; Xiong, J. Study on the Dispersion Stability and Plugging Performance of Dopamine-Modified Multiwalled Carbon Nanotubes. Energy Fuel 2023, 37, 19590–19601. [Google Scholar] [CrossRef]
- Barry, M.M.; Jung, Y.; Lee, J.K.; Phuoc, T.X.; Chyu, M.K. Fluid filtration and rheological properties of nanoparticle additive and intercalated clay hybrid bentonite drilling fluids. J. Pet. Sci. Eng. 2015, 127, 338–346. [Google Scholar] [CrossRef]
- Ospanov, Y.K.; Kudaikulova, G.A.; Moldabekov, M.S.; Zhaksylykova, M.Z. Improving Shale Stability through the Utilization of Graphene Nanopowder and Modified Polymer-Based Silica Nanocomposite in Water-Based Drilling Fluids. Processes 2024, 12, 1616. [Google Scholar] [CrossRef]
- Liu, F.; Zheng, Z.; Wang, X.Y.; Li, X.Q.; Zhang, Z.X.; Wang, X.W.; Dai, X.D.; Xin, Y.P.; Liu, Q.X.; Yao, H.L.; et al. Novel modified nano-silica/polymer composite in water-based drilling fluids to plug shale pores. Energy Sources Part A 2022, 44, 8662–8678. [Google Scholar] [CrossRef]
- Kotsuchibashi, Y. Recent advances in multi-temperature-responsive polymeric materials. Polym. J. 2020, 52, 681–689. [Google Scholar] [CrossRef]
- Koetting, M.C.; Peters, J.T.; Steichen, S.D.; Peppas, N.A. Stimulus-responsive hydrogels: Theory, modern advances, and applications. Mater. Sci. Eng. R 2015, 93, 1–49. [Google Scholar] [CrossRef] [PubMed]
- Doberenz, F.; Zeng, K.; Willems, C.; Zhang, K.; Groth, T. Thermo-responsive polymers and their biomedical application in tissue engineering—A review. J. Mater. Chem. B 2020, 8, 607–628. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Shi, W.; Tang, L.; Chen, S.Q.; Zhou, X.F.; Huang, D.C.; Lai, N.J. Temperature-Responsive Nano-SiO2 Composite for Improving Shale Stability in Oil-Based Drilling Fluids. Energy Fuel 2024, 38, 7733–7745. [Google Scholar] [CrossRef]
- Zhu, W.X.; Luo, Y.H.; Li, H.D.; Gong, Y.F. Application of a Novel Temperature-Sensitive Polymer PNA-SiO2 as a Rheological Agent in Geothermal Drilling. ACS Omega 2025, 10, 13011–13026. [Google Scholar] [CrossRef]
- Wang, W.J.; Qiu, Z.S.; Zhong, H.Y.; Huang, W.A.; Dai, W.H. Thermo-sensitive polymer nanospheres as a smart plugging agent for shale gas drilling operations. Pet. Sci. 2017, 14, 116–125. [Google Scholar] [CrossRef]
- Pan, Y.; Zhang, X.Y.; Zhan, Q.R.; Yang, S.C.; Wang, Y.C.; Guan, J.; Yang, G.; Yang, P.; Qayum, Z.U.A. Temperature-Sensitive Modified Bentonite Based on NIPAM for Drilling Fluid: Experimental and Molecular Simulation Studies. Molecules 2023, 28, 3839. [Google Scholar] [CrossRef]
- Li, S.P.; Yang, S.C.; Zhang, X.Y.; Feng, Q.; Guo, M.Z.; Pan, Y. Preparation of temperature-sensitive CMC-BENTONITE-NIPAM applied to deepwater drilling fluids. Matéria 2025, 30, 20250416. [Google Scholar] [CrossRef]
- Xie, B.Q.; Zhang, X.B.; Li, Y.G.; Liu, W.; Luo, M.W. Application a novel thermo-sensitive copolymer as a potential rheological modifier for deepwater water-based drilling fluids. Colloids Surf. A 2019, 581, 123848. [Google Scholar] [CrossRef]
- Zhou, S.-S.; Song, J.-J.; Xu, M.-B.; Xu, P.; You, F.-C.; Pu, L. Rheological and filtration properties of water-based drilling fluids as influenced by cellulose nanomaterials: Different aspect ratios and modified groups. Cellulose 2023, 30, 3667–3683. [Google Scholar] [CrossRef]
- Zhang, J.; Youngblood, J.P. Cellulose nanofibril-based hybrid coatings with enhanced moisture barrier properties. Mater. Adv. 2025, 6, 2833–2844. [Google Scholar] [CrossRef]
- Lei, M.; Sun, J.S.; Zhu, H.Y.; Huang, W.A.; Cai, Z.X. Synthesis and characterization of natural rosin-modified silica nanocomposite and its green multifunctional applications for drilling fluid. Colloids Surf. A 2024, 702, 134994. [Google Scholar] [CrossRef]
- Iqbal, M.A.; Hassan, S.U.; Mahmood, A.; Al-Masry, W.; Lee, J.; Lee, H.J.; Akhter, T.; Park, C.H. Dual responsive phase behavior of nonionic cellulose-based polymer brushes by visible-light-driven organocatalyzed atom transfer radical polymerization. Int. J. Biol. Macromol. 2025, 305, 140856. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, M.L.; Liberman, L.; Ertem, S.P.; Edmund, J.; Bates, F.S.; Lodge, T.P. Methyl cellulose solutions and gels: Fibril formation and gelation properties. Prog. Polym. Sci. 2021, 112, 101324. [Google Scholar] [CrossRef]
- Lei, M.; Huang, W.A.; Sun, J.S.; Shao, Z.X.; Zhao, L.; Zheng, K.X.; Fang, Y.W. Synthesis and characterization of thermo-responsive polymer based on carboxymethyl chitosan and its potential application in water-based drilling fluid. Colloids Surf. A 2021, 629, 127478. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Wang, J.C.; Chen, D.F.; Ge, S.; Fu, J.F.; Wu, X.Y.; Li, H.; Wang, R.R.; Han, R.L.; Tian, X.; et al. A Smart Plugging System of Temperature-Sensitive Gels Based on Cellulose Ether. ACS Omega 2025, 10, 23359–23373. [Google Scholar] [CrossRef]
- Bai, X.; Li, K.; Hu, H.; Yan, Y.; Luo, Y. Synthesis and properties of poly(acrylamide-co-N-vinylpyrrolidone-co-sodium p-styrene sulfonate) as an anionic fluid loss additive. J. Polym. Res. 2023, 30, 180. [Google Scholar] [CrossRef]
- Bonetti, L.; De Nardo, L.; Variola, F.; Fare, S. Evaluation of the subtle trade-off between physical stability and thermo-responsiveness in crosslinked methylcellulose hydrogels. Soft Matter 2020, 16, 5577–5587. [Google Scholar] [CrossRef]
- Bonetti, L.; De Nardo, L.; Variola, F.; Farè, S. In-situ Raman spectroscopy: An effective technique for the quantification of LCST transition of methylcellulose hydrogels. Mater. Lett. 2020, 274, 128011. [Google Scholar] [CrossRef]
- Wagner, M.; Pietsch, C.; Kerth, A.; Traeger, A.; Schubert, U.S. Physicochemical Characterization of the Thermo-Induced Self-Assembly of Thermo-Responsive PDMAEMA-b-PDEGMA Copolymers. J. Polym. Sci. Part A Polym. Chem. 2015, 53, 924–935. [Google Scholar] [CrossRef]
- Beg, M.; Kumar, P.; Choudhary, P.; Sharma, S. Effect of high temperature ageing on TiO2 nanoparticles enhanced drilling fluids: A rheological and filtration study. Upstream Oil Gas Technol. 2020, 5, 100019. [Google Scholar] [CrossRef]
- Kamali, F.; Saboori, R.; Sabbaghi, S. Fe3O4-CMC nanocomposite performance evaluation as rheology modifier and fluid loss control characteristic additives in water-based drilling fluid. J. Pet. Sci. Eng. 2021, 205, 108912. [Google Scholar] [CrossRef]










| Factor | |||||
|---|---|---|---|---|---|
| Level | A | B | C | D | E |
| 1 | A1 (1%) | B1 (5:3:2) | C1 (1:0.5) | D1 (2 h) | E1 (0.5%) |
| 2 | A2 (3%) | B2 (6:2:2) | C2 (1:1) | D2 (3 h) | E2 (1%) |
| 3 | A3 (5%) | B3 (4:3:3) | C3 (1:1.5) | D3 (4 h) | E3 (1.5%) |
| 4 | A4 (7%) | B4 (4:4:2) | C4 (1:2) | D4 (5 h) | E4 (2%) |
| Test No. | A | B | C | D | E | FLHTHP (130 °C) |
|---|---|---|---|---|---|---|
| 1 | 1 | 1 | 1 | 1 | 1 | 33 |
| 2 | 1 | 2 | 2 | 2 | 2 | 43 |
| 3 | 1 | 3 | 3 | 3 | 3 | 30 |
| 4 | 1 | 4 | 4 | 4 | 4 | 69 |
| 5 | 2 | 1 | 2 | 3 | 4 | 7 |
| 6 | 2 | 2 | 1 | 4 | 3 | 20.5 |
| 7 | 2 | 3 | 4 | 1 | 2 | 17 |
| 8 | 2 | 4 | 3 | 2 | 1 | 21.5 |
| 9 | 3 | 1 | 3 | 4 | 2 | 18.5 |
| 10 | 3 | 2 | 4 | 3 | 1 | 59 |
| 11 | 3 | 3 | 1 | 2 | 4 | 27 |
| 12 | 3 | 4 | 2 | 1 | 3 | 25 |
| 13 | 4 | 1 | 4 | 2 | 3 | 11 |
| 14 | 4 | 2 | 3 | 1 | 4 | 28 |
| 15 | 4 | 3 | 2 | 4 | 1 | 15.5 |
| 16 | 4 | 4 | 1 | 3 | 2 | 18.5 |
| K1 | 43.75 | 17.375 | 24.75 | 32.25 | 25.75 | |
| K2 | 16.5 | 37.625 | 22.625 | 24.25 | 25.625 | |
| K3 | 32.375 | 22.375 | 24.5 | 21.625 | 28.625 | |
| K4 | 18.25 | 33.5 | 39 | 32.75 | 30.875 | |
| R | 27.25 | 20.25 | 16.375 | 11.125 | 5.25 |
| Concentration of HPMC | Monomer Ratio | Total Mass Fraction of Grafting Monomers | Reaction Time | Initiator Concentration | FLHTHP (130 °C) |
|---|---|---|---|---|---|
| 3% | 5:3:2 | 1:1 | 4 h | 1% | 7 |
| Drilling Fluid System | Condition | AV (mPa·s) | PV (mPa·s) | YP (Pa) | FLAPI (mL) |
|---|---|---|---|---|---|
| Based mud | Before aging | 9 | 6 | 3 | 21 |
| 160 °C, 16 h | 2.5 | 2 | 0.24 | 92 | |
| Based mud + 1%HAAN | Before aging | 21 | 15.5 | 3 | 6.6 |
| 160 °C, 16 h | 16 | 14 | 1 | 8 | |
| 1%HAAN + 2.5%NaCl | Before aging | 26 | 19 | 7 | 6 |
| 160 °C, 16 h | 18 | 14 | 4 | 6.3 | |
| 1%HAAN + 5%NaCl | Before aging | 29 | 19 | 10 | 5 |
| 160 °C, 16 h | 21.5 | 17 | 9 | 7 | |
| 1%HAAN + 7.5%NaCl | Before aging | 26 | 13 | 13 | 6.1 |
| 160 °C, 16 h | 25 | 24 | 1 | 9.8 | |
| 1%HAAN + 10%NaCl | Before aging | 43.5 | 29 | 14 | 6 |
| 160 °C, 16 h | 13 | 13 | 0 | 15 | |
| 1%HAAN + 0.5%CaCl2 | Before aging | 25.5 | 18 | 7 | 6.3 |
| 160 °C, 16 h | 30 | 22 | 7.5 | 6.8 | |
| 1%HAAN + 1%KCl | Before aging | 35.5 | 25 | 10 | 7.2 |
| 160 °C, 16 h | 29.5 | 23 | 6 | 10 | |
| 1%HAAN + 2%KCl | Before aging | 35 | 21 | 13 | 7.6 |
| 160 °C, 16 h | 22.5 | 18 | 4 | 13.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Bai, X.; Xue, Z.; Wang, M.; Song, M.; Yang, M.; You, J.; Luo, Y. Preparation and Application of Cellulose-Based Thermosensitive Polymer in Water-Based Drilling Fluid. Polymers 2026, 18, 1187. https://doi.org/10.3390/polym18101187
Bai X, Xue Z, Wang M, Song M, Yang M, You J, Luo Y. Preparation and Application of Cellulose-Based Thermosensitive Polymer in Water-Based Drilling Fluid. Polymers. 2026; 18(10):1187. https://doi.org/10.3390/polym18101187
Chicago/Turabian StyleBai, Xiaodong, Zeyu Xue, Moubo Wang, Molin Song, Mengqian Yang, Jianpeng You, and Yumei Luo. 2026. "Preparation and Application of Cellulose-Based Thermosensitive Polymer in Water-Based Drilling Fluid" Polymers 18, no. 10: 1187. https://doi.org/10.3390/polym18101187
APA StyleBai, X., Xue, Z., Wang, M., Song, M., Yang, M., You, J., & Luo, Y. (2026). Preparation and Application of Cellulose-Based Thermosensitive Polymer in Water-Based Drilling Fluid. Polymers, 18(10), 1187. https://doi.org/10.3390/polym18101187