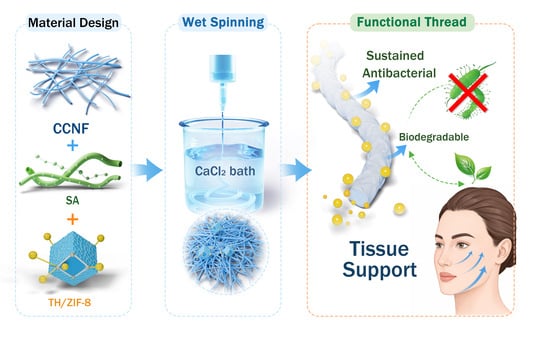
MOF-Enabled Nanocellulose Composite Threads for Sustained Antibacterial Drug Delivery and Minimally Invasive Soft-Tissue Lifting
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Preparation of AB-CCNF/SA Threads
2.3. Characterization
2.4. In Vitro Experiments
2.5. In Vivo Experiments
2.6. Statistical Analysis
3. Results and Discussion
3.1. Fabrication of AB-CCNF/SA Threads
3.2. Drug Loading of ZIF-8
3.3. Characterization and Drug Release of the Threads
3.4. In Vitro Application Performances
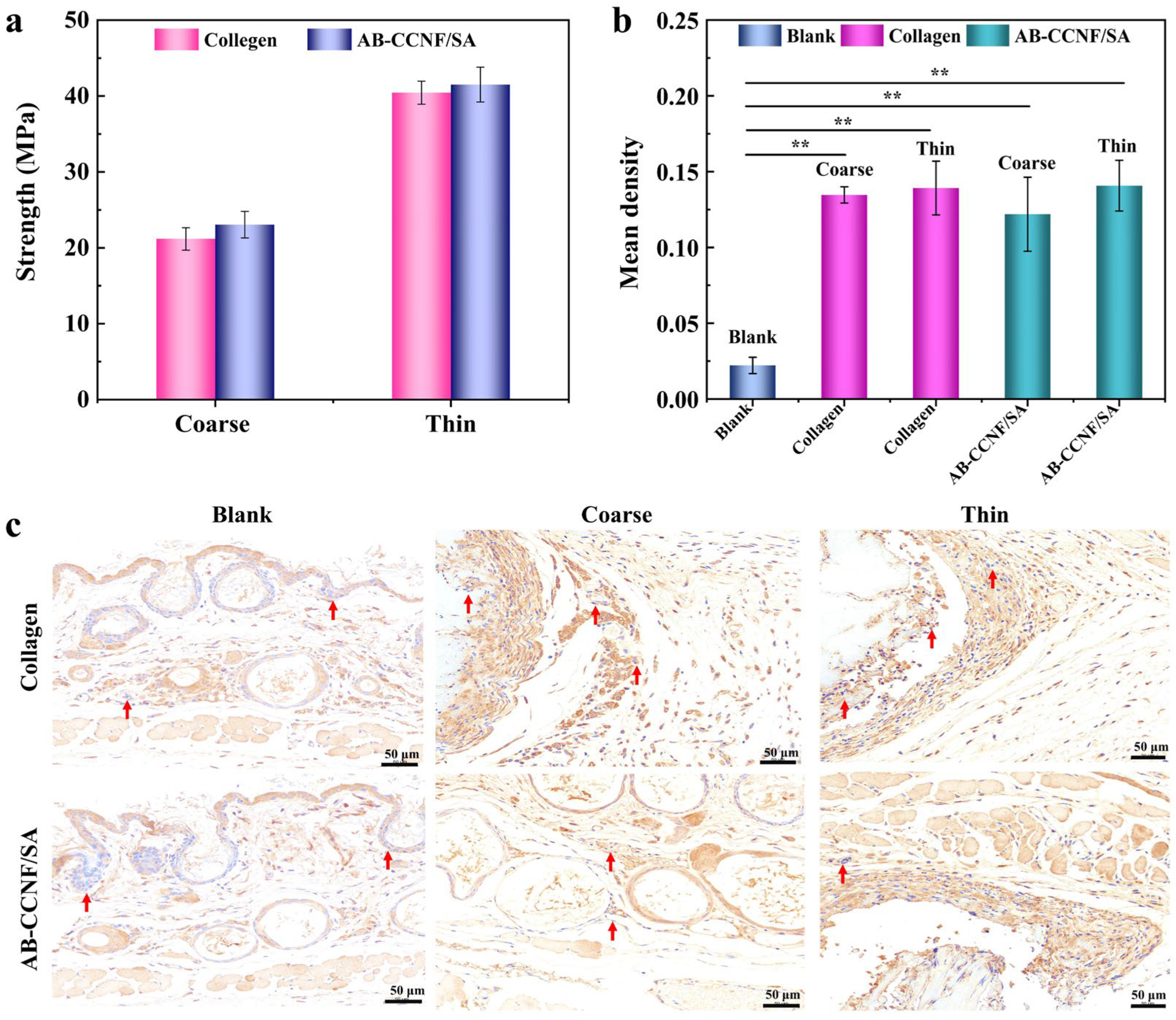
3.5. Model In Vivo Application Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CCNF | carboxymethylated cellulose nanofibrils |
| SA | sodium alginate |
| ZIF-8 | zeolitic Imidazolate Framework-8 |
| TH | tetracycline Hydrochloride |
| S. aureus | Staphylococcus aureus |
| E. coli | Escherichia coli |
References
- Siquier-Dameto, G.; Boadas-Vaello, P.; Verdú, E. Intradermal treatment with a hyaluronic acid complex supplemented with amino acids and antioxidant vitamins improves cutaneous hydration and viscoelasticity in healthy subjects. Antioxidants 2024, 13, 770. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, M.; Khan, A.; Gupta, M. Skin Ageing: Pathophysiology and current market treatment approaches. Curr. Aging Sci. 2020, 13, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Csekes, E.; Račková, L. Skin aging, cellular senescence and natural polyphenols. Int. J. Mol. Sci. 2021, 22, 12641. [Google Scholar] [CrossRef]
- Bonté, F.; Girard, D.; Archambault, J.-C.; Desmoulière, A. Skin changes during ageing. In Biochemistry and Cell Biology of Ageing: Part II Clinical Science; Subcellular Biochemistry; Springer: Singapore, 2019; Volume 91, pp. 249–280. [Google Scholar] [CrossRef]
- Hau, K.C.; Jain, S. Malar reshaping technique using bidirectional barb thread suspension procedure for 3-dimensional aging in Asian faces. Int. J. Women’s Dermatol. 2021, 7, 747–755. [Google Scholar] [CrossRef]
- Kim, M.S.; An, J.; Lee, J.H.; Lee, S.H.; Min, S.; Kim, Y.B.; Song, M.; Park, S.H.; Nam, K.Y.; Park, H.J.; et al. Clinical validation of face-fit surface-lighting micro light-emitting diode mask for skin anti-aging treatment. Adv. Mater. 2024, 36, 2411651. [Google Scholar] [CrossRef]
- Gülbitti, H.A.; Colebunders, B.; Pirayesh, A.; Bertossi, D.; van der Lei, B. Thread-lift sutures: Still in the lift? A systematic review of the literature. Plast. Reconstr. Surg. 2018, 141, 341e–347e. [Google Scholar] [CrossRef]
- Niu, Z.; Han, Y.; Jin, R.; Li, Y.; Liu, J.; Li, N.; Li, W.; Li, D.; Chen, Y.; Han, Y. Complications following facial thread-lifting. Chin. J. Plast. Reconstr. Surg. 2020, 2, 204–211. [Google Scholar] [CrossRef]
- Bertossi, D.; Botti, G.; Gualdi, A.; Fundarò, P.; Nocini, R.; Pirayesh, A.; van der Lei, B. Effectiveness, longevity, and complications of facelift by barbed suture insertion. Aesthet. Surg. J. 2019, 39, 241–247. [Google Scholar] [CrossRef]
- Wanitphakdeedecha, R.; Yan, C.; Ng, J.N.C.; Fundarò, S. Absorbable barbed threads for lower facial soft-tissue repositioning in Asians. Dermatol. Ther. 2021, 11, 1395–1408. [Google Scholar] [CrossRef]
- Ma, H.; Zhao, J.; Liu, Y.; Liu, L.; Yu, J.; Fan, Y. Controlled delivery of aspirin from nanocellulose-sodium alginate interpenetrating network hydrogels. Ind. Crops Prod. 2023, 192, 116081. [Google Scholar] [CrossRef]
- Wang, X.; Zhong, C.; Zhong, Y.; Fan, Z.; Liu, Z.; Xu, P.; Deng, X.; Guo, J.; Sawant, T.R.; Zhou, M. Impressive merits of nanocellulose driving sustainable beauty. Carbohydr. Polym. 2025, 353, 123270. [Google Scholar] [CrossRef]
- Xue, H.; Zhang, Y.; Zhao, Z.; Gao, H.; Bao, W.; Li, J.; Zhang, Z.; Wang, Q.; He, Q. A Review: Sources, preparation and application of nanocellulose. J. Polym. Mater. 2025, 42, 379–409. [Google Scholar] [CrossRef]
- Geng, R.; Wang, J.; Zhang, Z.; Dong, Q.; Wu, F.; Chen, S.; Su, T.; Qi, X. Adsorption of antibiotics by polydopamine-modified salecan hydrogel: Performance, kinetics and mechanism studies. Chem. Eng. J. 2023, 454, 140446. [Google Scholar] [CrossRef]
- Wang, M.; Wang, Y.; Liu, J.; Yu, H.; Liu, P.; Yang, Y.; Sun, D.; Kang, H.; Wang, Y.; Tang, J.; et al. Integration of advanced biotechnology for green carbon. Green Carbon 2024, 2, 164–175. [Google Scholar] [CrossRef]
- Kalita, H.; Hazarika, A.; Kandimalla, R.; Kalita, S.; Devi, R. Development of banana (Musa balbisiana) pseudo stem fiber as a surgical bio-tool to avert post-operative wound infections. RSC Adv. 2018, 8, 36791–36801. [Google Scholar] [CrossRef] [PubMed]
- Samulin Erdem, J.; Alswady-Hoff, M.; Ervik, T.K.; Skare, Ø.; Ellingsen, D.G.; Zienolddiny, S. Cellulose nanocrystals modulate alveolar macrophage phenotype and phagocytic function. Biomaterials 2019, 203, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, R.A.; Zuhri, M.Y.M.; Aisyah, H.A.; Asyraf, M.R.M.; Hassan, S.A.; Zainudin, E.S.; Sapuan, S.M.; Sharma, S.; Bangar, S.P.; Jumaidin, R.; et al. Natural fiber-reinforced polylactic acid, polylactic acid blends and their composites for advanced applications. Polymers 2022, 14, 202. [Google Scholar] [CrossRef] [PubMed]
- Lamm, M.E.; Li, K.; Qian, J.; Wang, L.; Lavoine, N.; Newman, R.; Gardner, D.J.; Li, T.; Hu, L.; Ragauskas, A.J.; et al. Recent advances in functional materials through cellulose nanofiber templating. Adv. Mater. 2021, 33, 2005538. [Google Scholar] [CrossRef]
- Lei, W.; Li, Z.; Fang, C.; Gao, Y.; Zhou, X.; Pu, M.; Li, L.; Yang, M. High-efficiency synergistic modification of polylactic acid by micrometer-sized cellulose fibers and cellulose nanocrystals. Int. J. Biol. Macromol. 2025, 307, 142258. [Google Scholar] [CrossRef]
- Wu, M.; Liu, Y.; Liu, C.; Cui, Q.; Zheng, X.; Fatehi, P.; Li, B. Core–shell filament with excellent wound healing property made of cellulose nanofibrils and guar gum via interfacial polyelectrolyte complexation spinning. Small 2023, 19, 2205867. [Google Scholar] [CrossRef]
- Wan, A.C.A.; Cutiongco, M.F.A.; Tai, B.C.U.; Leong, M.F.; Lu, H.F.; Yim, E.K.F. Fibers by interfacial polyelectrolyte complexation–processes, materials and applications. Mater. Today 2016, 19, 437–450. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, L.; Wang, W.; Xiang, Y.; Liu, J.; An, Y.; Shi, J.; Qi, H.; Huang, Z. Sodium alginate/sodium lignosulfonate hydrogel based on inert Ca2+ activation for water conservation and growth promotion. Environ. Res. 2024, 246, 118144. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, R.; Ci, M.; Sui, S.; Zhu, P. Sodium alginate/cellulose nanocrystal fibers with enhanced mechanical strength prepared by wet spinning. J. Eng. Fibers Fabr. 2019, 14, 1558925019847553. [Google Scholar] [CrossRef]
- Soomro, N.A.; Wu, Q.; Amur, S.A.; Liang, H.; Ur Rahman, A.; Yuan, Q.; Wei, Y. Natural drug physcion encapsulated zeolitic imidazolate framework, and their application as antimicrobial agent. Colloids Surf. B Biointerfaces 2019, 182, 110364. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Huo, Y.; Fan, Q.; Li, M.; Liu, H.; Li, B.; Li, Y. Cellulose nanofibrils composite hydrogel with polydopamine@zeolitic imidazolate framework-8 encapsulated in used as efficient vehicles for controlled drug release. J. Ind. Eng. Chem. 2021, 102, 343–350. [Google Scholar] [CrossRef]
- Rahman, M.; Kabir, M.; Islam, T.; Wang, Y.; Meng, Q.; Liu, H.; Chen, S.; Wu, S. Curcumin-loaded ZIF-8 nanomaterials: Exploring drug loading efficiency and biomedical performance. ACS Omega 2025, 10, 3067–3079. [Google Scholar] [CrossRef]
- Pandit, A.H.; Nisar, S.; Imtiyaz, K.; Nadeem, M.; Mazumdar, N.; Rizvi, M.M.A.; Ahmad, S. Injectable, self-healing, and biocompatible N,O-carboxymethyl chitosan/multialdehyde guar gum hydrogels for sustained anticancer drug delivery. Biomacromolecules 2021, 22, 3731–3745. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Wang, S.; Xie, Z.; Yu, S.; Li, L.; Xiao, H.; Song, Y. Hyaluronic acid coating on the surface of curcumin-loaded zif-8 nanoparticles for improved breast cancer therapy: An In Vitro and In Vivo study. Colloids Surf. B Biointerfaces 2021, 203, 111759. [Google Scholar] [CrossRef]
- Mihai, M.; Racovita, S.; Vasiliu, A.-L.; Doroftei, F.; Barbu-Mic, C.; Schwarz, S.; Steinbach, C.; Simon, F. Autotemplate microcapsules of CaCO3/pectin and nonstoichiometric complexes as sustained tetracycline hydrochloride delivery carriers. ACS Appl. Mater. Interfaces 2017, 9, 37264–37278. [Google Scholar] [CrossRef]
- Long, X.; Lu, Y.-L.; Guo, H.; Tang, Y.-P. Recent advances in solid residues resource utilization in traditional Chinese medicine. ChemistrySelect 2023, 8, e202300383. [Google Scholar] [CrossRef]
- Zhao, S.; Fang, Z.; Liu, Y.; Li, G.; Lin, X.; Chen, K.; Qiu, X. Enhancing aging resistance of transparent paper: Structural modification of wood fibers via carboxymethylation. ACS Sustain. Chem. Eng. 2024, 12, 11244–11252. [Google Scholar] [CrossRef]
- Fraschini, C.; Chauve, G.; Bouchard, J. TEMPO-mediated surface oxidation of cellulose nanocrystals (CNCs). Cellulose 2017, 24, 2775–2790. [Google Scholar] [CrossRef]
- Zhang, S.; Yi, J.; Yuan, X.; Zhang, Z.; Shan, Z.; Wang, H. Fabrication and characterization of carrageenan-based multifunctional films integrated with gallic acid@ZIF-8 for beef preservation. Int. J. Biol. Macromol. 2024, 274, 133319. [Google Scholar] [CrossRef]
- Wang, S.; Wang, S.; Yang, L.; Wang, P.; Song, H.; Liu, H. pH-responsive aminated mesoporous silica microspheres modified with soybean hull polysaccharides for curcumin encapsulation and controlled release. Food Chem. 2024, 454, 139832. [Google Scholar] [CrossRef]
- Abbasnezhad, N.; Zirak, N.; Shirinbayan, M.; Kouidri, S.; Salahinejad, E.; Tcharkhtchi, A.; Bakir, F. Controlled Release from polyurethane films: Drug release mechanisms. J. Appl. Polym. Sci. 2020, 138, 50083. [Google Scholar] [CrossRef]
- Wu, M.; Ding, L.; Bai, X.; Cao, Y.; Rahmaninia, M.; Li, B.; Li, B. Cellulose-based suture: State of art, challenge, and future outlook. J. Bioresour. Bioprod. 2025, 10, 295–309. [Google Scholar] [CrossRef]
- You, J.; Cao, J.; Zhao, Y.; Zhang, L.; Zhou, J.; Chen, Y. Improved mechanical properties and sustained release behavior of cationic cellulose nanocrystals reinforeced cationic cellulose injectable hydrogels. Biomacromolecules 2016, 17, 2839–2848. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, S.; So, J.-H.; Kim, K.; Koo, H.-J. Cytotoxicity of gallium–indium liquid metal in an aqueous environment. ACS Appl. Mater. Interfaces 2018, 10, 17448–17454. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, Q.; Huo, Y.; Li, M.; Liu, H.; Li, B. Construction of nanocellulose-based composite hydrogel with a double packing structure as an intelligent drug carrier. Cellulose 2021, 28, 6953–6966. [Google Scholar] [CrossRef]
- Mertaniemi, H.; Escobedo-Lucea, C.; Sanz-Garcia, A.; Gandía, C.; Mäkitie, A.; Partanen, J.; Ikkala, O.; Yliperttula, M. Human stem cell decorated nanocellulose threads for biomedical applications. Biomaterials 2016, 82, 208–220. [Google Scholar] [CrossRef]
- Hojabri, M.; Tayebi, T.; Kasravi, M.; Aghdaee, A.; Ahmadi, A.; Mazloomnejad, R.; Tarasi, R.; Shaabani, A.; Bahrami, S.; Niknejad, H. Wet-spinnability and crosslinked fiber properties of alginate/hydroxyethyl cellulose with varied proportion for potential use in tendon tissue engineering. Int. J. Biol. Macromol. 2023, 240, 124492. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cheng, S.; Qin, K.; Yang, X.; Wang, H.; Man, C.; Zhao, Q.; Jiang, Y. Apigenin@ZIF-8 with pH-responsive sustained release function added to propolis-gelatin films achieved an outstanding antibacterial effect. Food Packag. Shelf Life 2023, 40, 101191. [Google Scholar] [CrossRef]
- Peng, J.; Zhang, Z.; Hu, C.; Wang, Z.; Kang, Y.; Chen, W.; Ao, T. Hierarchically porous ZIF-8 for tetracycline hydrochloride elimination. J. Sol-Gel Sci. Technol. 2021, 99, 339–353. [Google Scholar] [CrossRef]
- Wang, Z.; Fu, Q.; Xie, D.; Wang, F.; Zhang, G.; Shan, H. Facile fabrication of zeolitic imidazolate framework-8@regenerated cellulose nanofibrous membranes for effective adsorption of tetracycline hydrochloride. Molecules 2024, 29, 4146. [Google Scholar] [CrossRef] [PubMed]
- Manivel, C.M.; Zheng, A.L.T.; Kannan, R.; Seenivasagam, S.; Yun Hin, T.Y.; Boonyuen, S.; Lease, J.; Andou, Y.; Tan, K.B. ZIF-8/graphene composite for photocatalytic degradation under low intensity UV irradiation and antibacterial applications. Sci. Rep. 2025, 15, 33875. [Google Scholar] [CrossRef]
- Pandey, S.K.; Sharmah, B.; Manna, P.; Chawngthu, Z.; Kumar, S.; Trivedi, A.K.; Saha, S.K.; Das, J. Fabrication of hydrophobic drug-loaded zeolitic imidazolate framework-8 (ZIF-8) for enhanced anti-bacterial activity. J. Mol. Struct. 2024, 1312, 138452. [Google Scholar] [CrossRef]
- Lei, W.; Fang, C.; Zhou, X.; Yin, Q.; Pan, S.; Yang, R.; Liu, D.; Ouyang, Y. Cellulose nanocrystals obtained from office waste paper and their potential application in PET packing materials. Carbohydr. Polym. 2018, 181, 376–385. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, L.; Huang, L.; Zhang, W.; Wang, R.; Yue, T.; Sun, J.; Li, G.; Wang, J. The highly efficient elimination of intracellular bacteria via a metal organic framework (MOF)-based three-in-one delivery system. Nanoscale 2019, 11, 9468–9477. [Google Scholar] [CrossRef]
- Nam, H.S.; An, J.; Chung, D.J.; Kim, J.-H.; Chung, C.-P. Controlled release behavior of bioactive molecules from photo-reactive hyaluronic acid-alginate scaffolds. Macromol. Res. 2006, 14, 530–538. [Google Scholar] [CrossRef]
- Plascencia Martinez, D.F.; Quiroz Castillo, J.M.; Ospina Orejarena, A.; Pérez Gallardo, A.; Méndez Merino, E.; Trimmer López, G.A.; López Peña, I.Y.; Hernández Martínez, D.; López Gastelum, K.A.; Leyva Verduzco, A.A.; et al. Comparative study of single and coaxial electrospun antimicrobial cross-linked scaffolds enriched with aloe vera: Characterization, antimicrobial activity, drug delivery, cytotoxicity, and cell proliferation on adipose stem cells and human skin fibroblast. ACS Omega 2024, 9, 41157–41170. [Google Scholar] [CrossRef] [PubMed]
- Rethinam, S.; Thotapalli Parvathaleswara, S.; Nandhagobal, G.; Alagumuthu, T.; Robert, B. Preparation of absorbable surgical suture: Novel approach in biomedical application. J. Drug Deliv. Sci. Technol. 2018, 47, 454–460. [Google Scholar] [CrossRef]
- Naser, M.A.; Sayed, A.M.; Abdelmoez, W.; El-Wakad, M.T.; Abdo, M.S. Biodegradable suture development-based albumin composites for tissue engineering applications. Sci. Rep. 2024, 14, 7912. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Xu, W.; Wu, D.; Liu, Z.; Song, Y.; Zhou, A.; Xiong, R.; Huang, C. Bio-based pH-responsive nanofibrous membranes for controlled release of tetracycline hydrochloride. J. Appl. Polym. Sci. 2024, 141, e55384. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, D.; Mensaha, A.; Wang, Q.; Cai, Y.; Wei, Q. Ultrafast gelation of multifunctional hydrogel/composite based on self-catalytic Fe3+/tannic acid-cellulose nanofibers. J. Colloid Interface Sci. 2022, 606, 1457–1468. [Google Scholar] [CrossRef]
- Hong, G.-W.; Park, S.Y.; Yi, K.-H. Revolutionizing Thread lifting: Evolution and techniques in facial rejuvenation. J. Cosmet. Dermatol. 2024, 23, 2537–2542. [Google Scholar] [CrossRef] [PubMed]
- Wong, V. Hanging by a thread: Choosing the right thread for the right patient. J. Dermatol. Cosmetol. 2017, 1, 00021. [Google Scholar] [CrossRef]
- Stein, P.; Vitavska, O.; Kind, P.; Hoppe, W.; Wieczorek, H.; Schürer, N.Y. The biological basis for poly-L-lactic acid-induced augmentation. J. Dermatol. Sci. 2015, 78, 26–33. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sun, M.; Wu, M.; Wang, P.; Li, B.; Yu, G.; Du, H.; Lou, T.; Li, B. MOF-Enabled Nanocellulose Composite Threads for Sustained Antibacterial Drug Delivery and Minimally Invasive Soft-Tissue Lifting. Polymers 2026, 18, 1186. https://doi.org/10.3390/polym18101186
Sun M, Wu M, Wang P, Li B, Yu G, Du H, Lou T, Li B. MOF-Enabled Nanocellulose Composite Threads for Sustained Antibacterial Drug Delivery and Minimally Invasive Soft-Tissue Lifting. Polymers. 2026; 18(10):1186. https://doi.org/10.3390/polym18101186
Chicago/Turabian StyleSun, Meng, Meiyan Wu, Ping Wang, Bing Li, Guang Yu, Haishun Du, Tao Lou, and Bin Li. 2026. "MOF-Enabled Nanocellulose Composite Threads for Sustained Antibacterial Drug Delivery and Minimally Invasive Soft-Tissue Lifting" Polymers 18, no. 10: 1186. https://doi.org/10.3390/polym18101186
APA StyleSun, M., Wu, M., Wang, P., Li, B., Yu, G., Du, H., Lou, T., & Li, B. (2026). MOF-Enabled Nanocellulose Composite Threads for Sustained Antibacterial Drug Delivery and Minimally Invasive Soft-Tissue Lifting. Polymers, 18(10), 1186. https://doi.org/10.3390/polym18101186