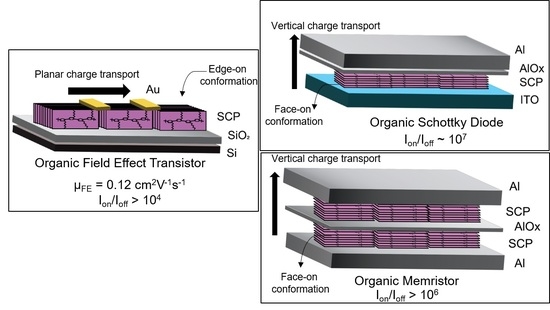
The Influence of a Microstructural Conformation of Oriented Floating Films of Semiconducting Polymers on Organic Device Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Thin Film Characterization
2.3. OFET Fabrication
2.4. OSD Fabrication
2.5. Organic Bistable Memristor Fabrication
3. Results and Discussion
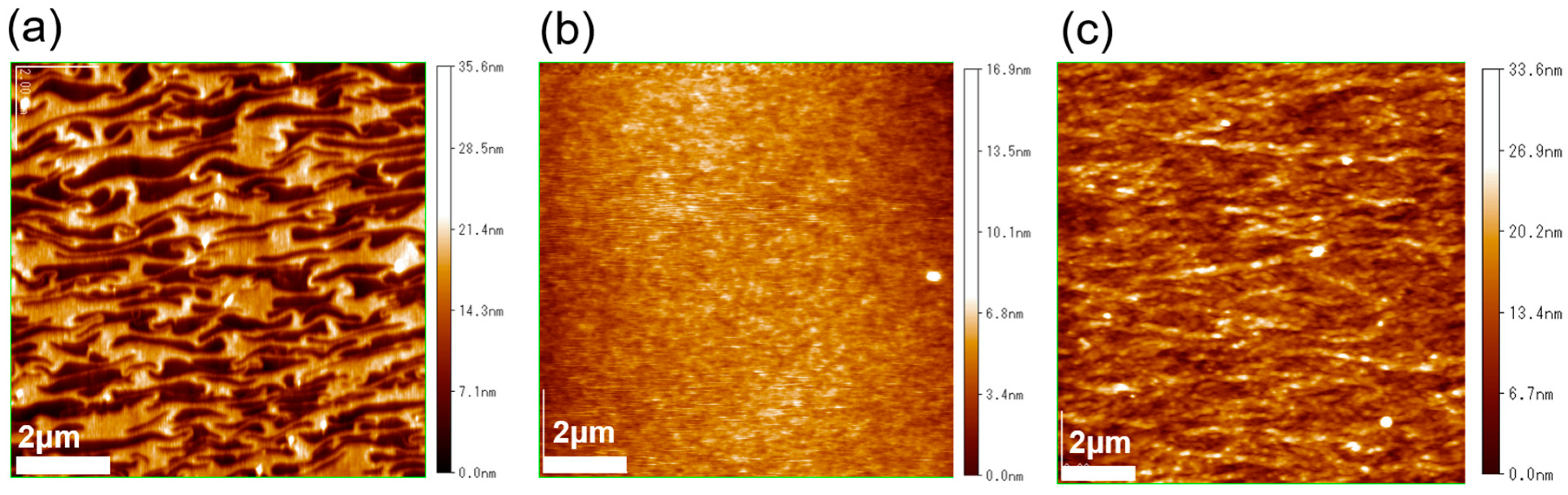
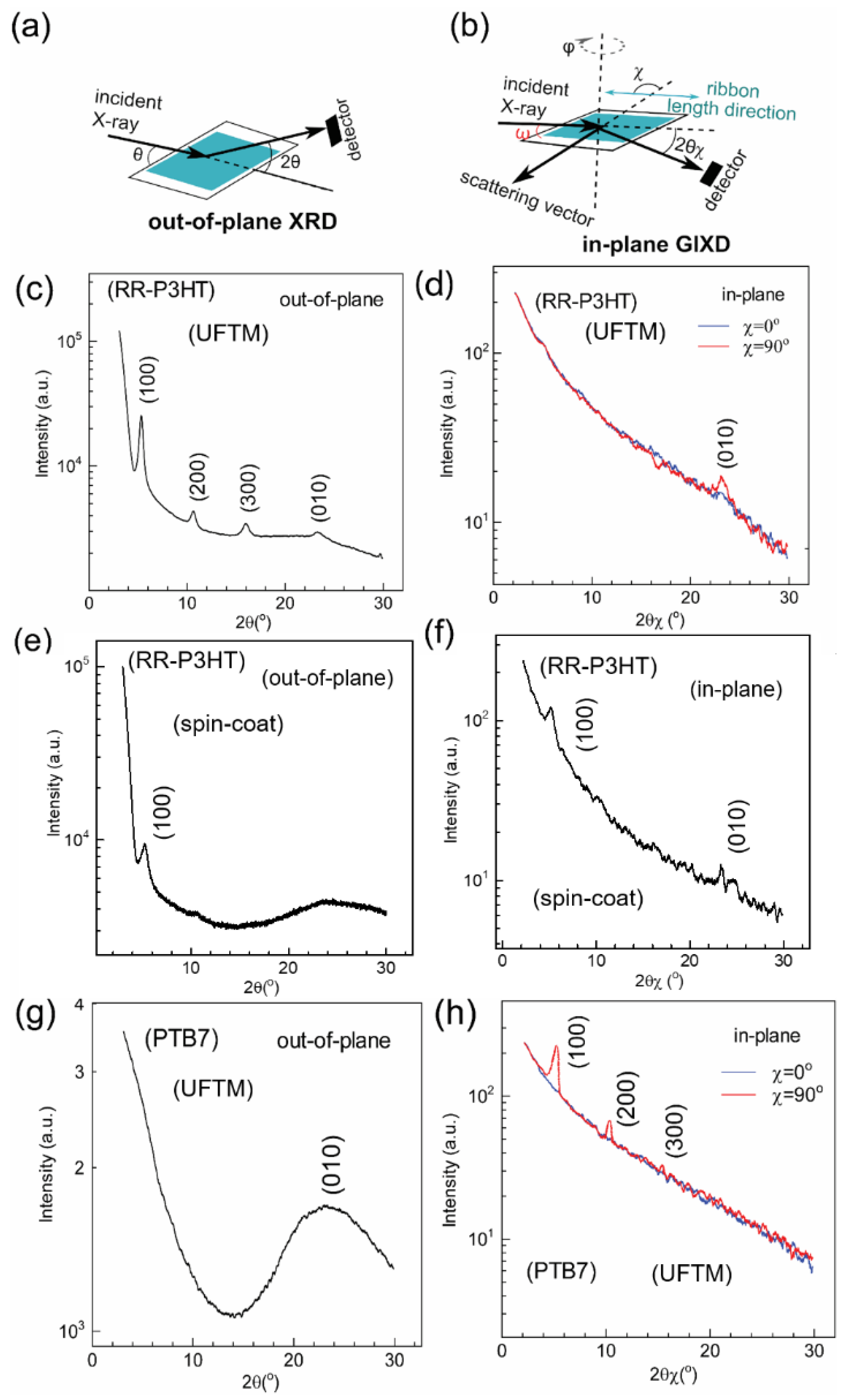
3.1. Thin-Film Microstructure
3.2. Device Characteristics
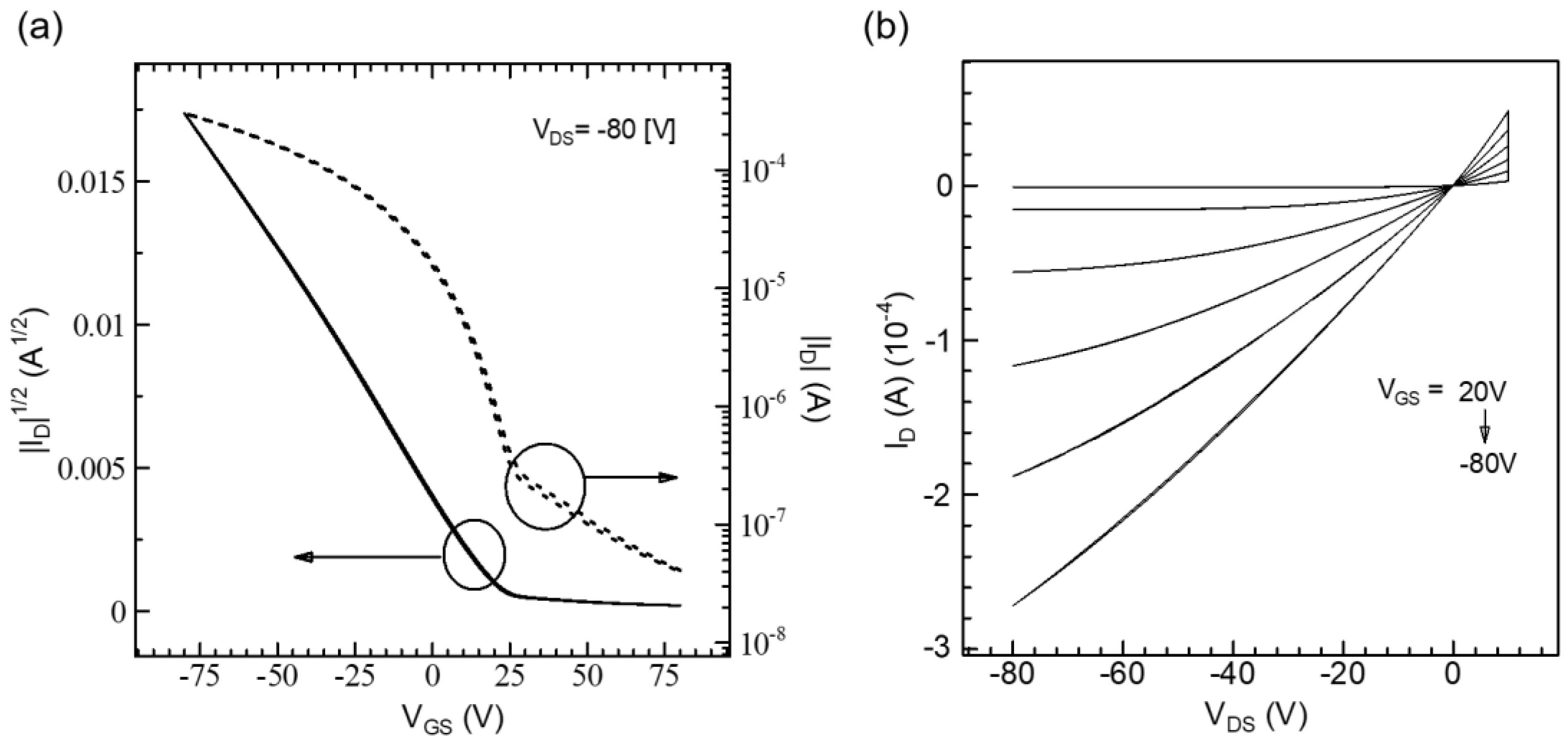
3.2.1. OFET Performance
3.2.2. OSD Performance
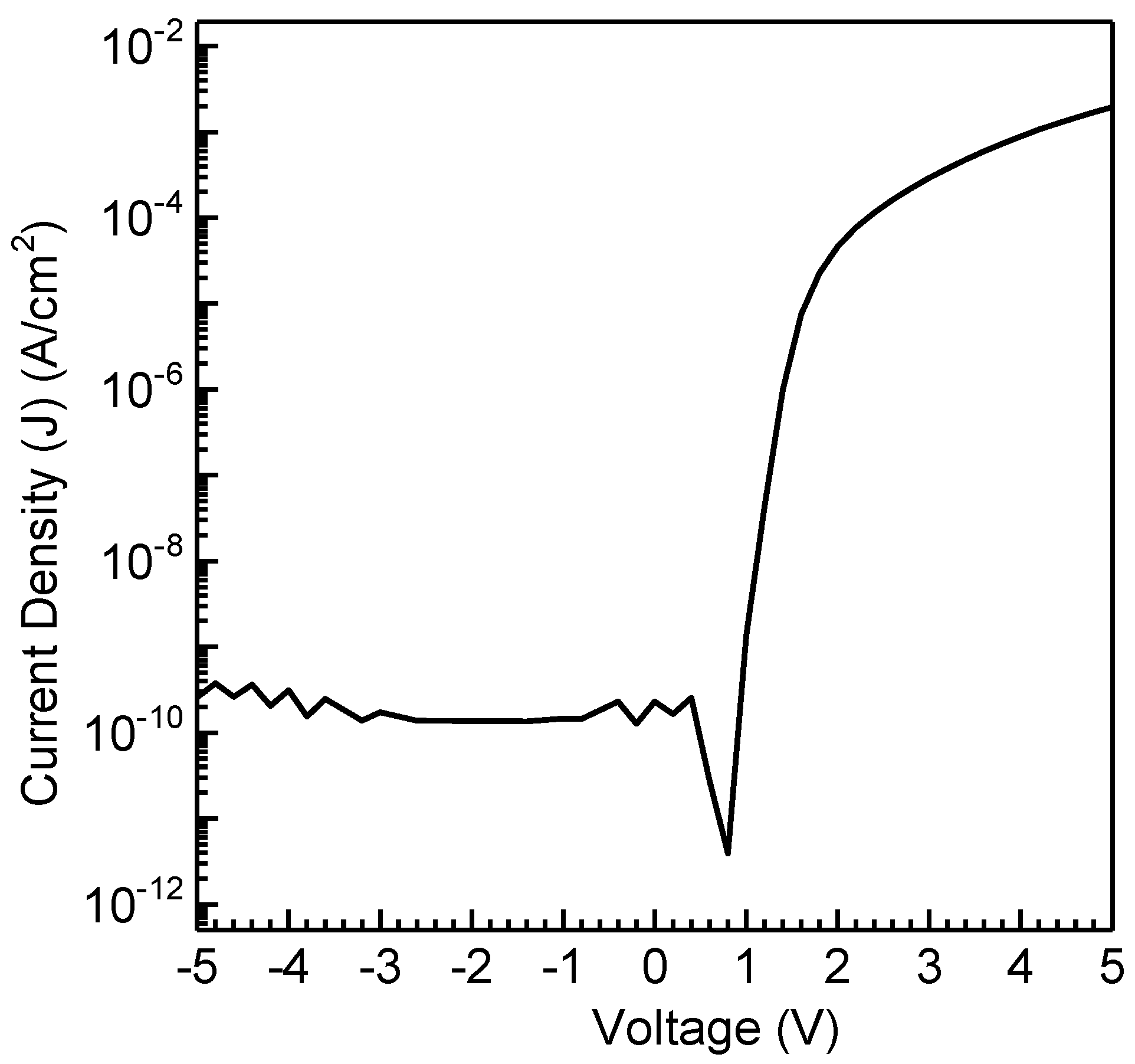
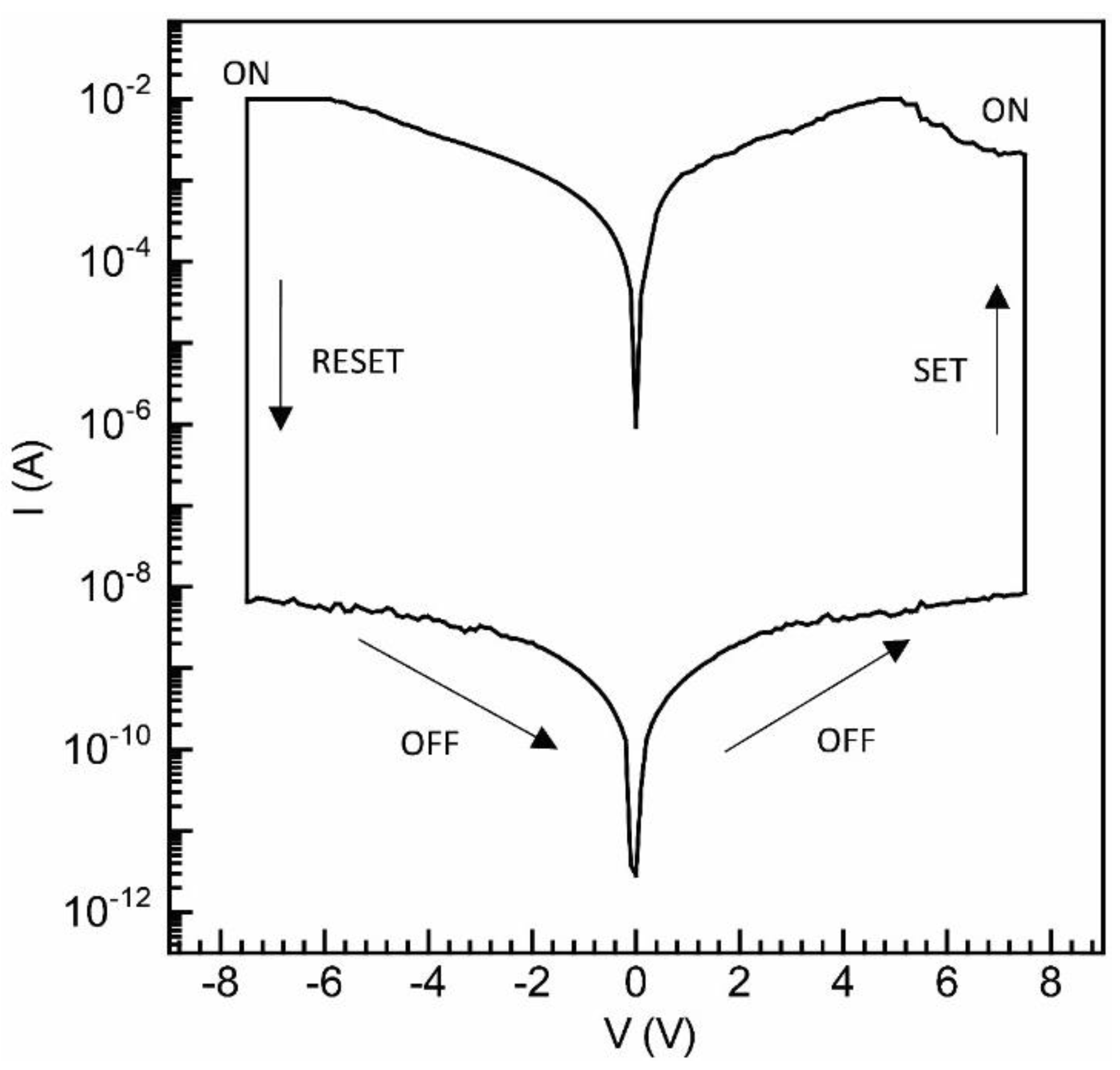
3.2.3. Organic Bistable Memristor
| (a) OFETs: | |||
| Technique | µ (cm2V−1s−1) | On/Off Ratio | Reference |
| UFTM | 0.12 | 104 | This work |
| Drop casting | 8.4 × 10−2 | 104 | [44] |
| Solvent-drop casting | 0.16 | 104 | [45] |
| Spin coating | 3.8 × 10−2 | 2.4 × 105 | [46] |
| Spin coating | 1.4 × 10−2 | 1 × 104 | [47] |
| Dip coating | 8.5 × 10−2 | 1.5 × 104 | [48] |
| (b) OSDs: | |||
| Technique | On/Off Ratio | Reference | |
| Spin coating | 8.6 × 106 | This work | |
| Nanofiber solution casting | 1.2 × 103 | [49] | |
| Wet deposition | 106 | [50] | |
| Spin coating | 1.11 × 106 | [30] | |
| Spin coating | 103 | [30] | |
| Spin coating | 8.8 × 102 | [38] | |
| (c) Organic memristors: | |||
| Polymer | Technique | On/Off Ratio | Reference |
| PTB-7 | UFTM | 2.1 × 106 | This work |
| P3HT:LiClO4 | Spin coating | 103 | [51] |
| PVA | Spin coating | 104 | [52] |
| Cu-TCNQ | Vapor deposition | 4 × 102 | [53] |
| PVA-PEDOT: PSS | Spin coating | 102 | [54] |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sirringhaus, H. Device Physics of Solution-Processed Organic Field-Effect Transistors. Adv. Mater. 2005, 17, 2411–2425. [Google Scholar] [CrossRef]
- Sirringhaus, H. 25th Anniversary Article: Organic Field-Effect Transistors: The Path Beyond Amorphous Silicon. Adv. Mater. 2014, 26, 1319–1335. [Google Scholar] [CrossRef]
- Oh, J.Y.; Rondeau-Gagné, S.; Chiu, Y.-C.; Chortos, A.; Lissel, F.; Wang, G.-J.N.; Schroeder, B.C.; Kurosawa, T.; Lopez, J.; Katsumata, T.; et al. Intrinsically Stretchable and Healable Semiconducting Polymer for Organic Transistors. Nature 2016, 539, 411–415. [Google Scholar] [CrossRef]
- Xu, J.; Wu, H.-C.; Zhu, C.; Ehrlich, A.; Shaw, L.; Nikolka, M.; Wang, S.; Molina-Lopez, F.; Gu, X.; Luo, S.; et al. Multi-Scale Ordering in Highly Stretchable Polymer Semiconducting Films. Nat. Mater. 2019, 18, 594–601. [Google Scholar] [CrossRef]
- Abdullah, M.; Al-Nashy, B.O.; Korotcenkov, G.; Al-Khursan, A.H. QDs of Wide Band Gap II–VI Semiconductors Luminescent Properties and Photodetector Applications. In Handbook of II-VI Semiconductor-Based Sensors and Radiation Detectors; Springer International Publishing: Cham, Switzerland, 2023; pp. 399–425. [Google Scholar]
- Sirringhaus, H.; Brown, P.J.; Friend, R.H.; Nielsen, M.M.; Bechgaard, K.; Langeveld-Voss, B.M.W.; Spiering, A.J.H.; Janssen, R.A.J.; Meijer, E.W.; Herwig, P.; et al. Two-Dimensional Charge Transport in Self-Organized, High-Mobility Conjugated Polymers. Nature 1999, 401, 685–688. [Google Scholar] [CrossRef]
- Gurau, M.C.; Delongchamp, D.M.; Vogel, B.M.; Lin, E.K.; Fischer, D.A.; Sambasivan, S.; Richter, L.J. Measuring Molecular Order in Poly(3-Alkylthiophene) Thin Films with Polarizing Spectroscopies. Langmuir 2007, 23, 834–842. [Google Scholar] [CrossRef]
- O’Connor, B.T.; Reid, O.G.; Zhang, X.; Kline, R.J.; Richter, L.J.; Gundlach, D.J.; DeLongchamp, D.M.; Toney, M.F.; Kopidakis, N.; Rumbles, G. Morphological Origin of Charge Transport Anisotropy in Aligned Polythiophene Thin Films. Adv. Funct. Mater. 2014, 24, 3422–3431. [Google Scholar] [CrossRef]
- Ning, G.-H.; Chen, Z.; Gao, Q.; Tang, W.; Chen, Z.; Liu, C.; Tian, B.; Li, X.; Loh, K.P. Salicylideneanilines-Based Covalent Organic Frameworks as Chemoselective Molecular Sieves. J. Am. Chem. Soc. 2017, 139, 8897–8904. [Google Scholar] [CrossRef] [PubMed]
- Bhimanapati, G.R.; Lin, Z.; Meunier, V.; Jung, Y.; Cha, J.; Das, S.; Xiao, D.; Son, Y.; Strano, M.S.; Cooper, V.R.; et al. Recent Advances in Two-Dimensional Materials beyond Graphene. ACS Nano 2015, 9, 11509–11539. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.-F.; Sun, B.; Breiby, D.W.; Nielsen, M.M.; Sölling, T.I.; Giles, M.; McCulloch, I.; Sirringhaus, H. Enhanced Mobility of Poly(3-Hexylthiophene) Transistors by Spin-Coating from High-Boiling-Point Solvents. Chem. Mater. 2004, 16, 4772–4776. [Google Scholar] [CrossRef]
- Yang, H.; LeFevre, S.W.; Ryu, C.Y.; Bao, Z. Solubility-Driven Thin Film Structures of Regioregular Poly(3-Hexyl Thiophene) Using Volatile Solvents. Appl. Phys. Lett. 2007, 90, 172116. [Google Scholar] [CrossRef]
- Soeda, J.; Matsui, H.; Okamoto, T.; Osaka, I.; Takimiya, K.; Takeya, J. Highly Oriented Polymer Semiconductor Films Compressed at the Surface of Ionic Liquids for High-Performance Polymeric Organic Field-Effect Transistors. Adv. Mater. 2014, 26, 6430–6435. [Google Scholar] [CrossRef]
- Kajiya, D.; Ozawa, S.; Koganezawa, T.; Saitow, K. Enhancement of Out-of-Plane Mobility in P3HT Film by Rubbing: Aggregation and Planarity Enhanced with Low Regioregularity. J. Phys. Chem. C 2015, 119, 7987–7995. [Google Scholar] [CrossRef]
- Osaka, I.; Takimiya, K. Backbone Orientation in Semiconducting Polymers. Polymer 2015, 59, A1–A15. [Google Scholar] [CrossRef]
- Zhao, F.; Wang, C.; Zhan, X. Morphology Control in Organic Solar Cells. Adv. Energy Mater. 2018, 8, 1703147. [Google Scholar] [CrossRef]
- Pandey, M.; Kumari, N.; Nagamatsu, S.; Pandey, S.S. Recent Advances in the Orientation of Conjugated Polymers for Organic Field-Effect Transistors. J. Mater. Chem. C 2019, 7, 13323–13351. [Google Scholar] [CrossRef]
- Ma, J.; Hashimoto, K.; Koganezawa, T.; Tajima, K. Enhanced Vertical Carrier Mobility in Poly(3-Alkylthiophene) Thin Films Sandwiched between Self-Assembled Monolayers and Surface-Segregated Layers. Chem. Commun. 2014, 50, 3627–3630. [Google Scholar] [CrossRef]
- Skrypnychuk, V.; Wetzelaer, G.-J.A.H.; Gordiichuk, P.I.; Mannsfeld, S.C.B.; Herrmann, A.; Toney, M.F.; Barbero, D.R. Ultrahigh Mobility in an Organic Semiconductor by Vertical Chain Alignment. Adv. Mater. 2016, 28, 2359–2366. [Google Scholar] [CrossRef]
- Skrypnychuk, V.; Boulanger, N.; Yu, V.; Hilke, M.; Mannsfeld, S.C.B.; Toney, M.F.; Barbero, D.R. Enhanced Vertical Charge Transport in a Semiconducting P3HT Thin Film on Single Layer Graphene. Adv. Funct. Mater. 2015, 25, 664–670. [Google Scholar] [CrossRef]
- Wang, F.; Hashimoto, K.; Segawa, H.; Tajima, K. Effects of Chain Orientation in Self-Organized Buffer Layers Based on Poly(3-Alkylthiophene)s for Organic Photovoltaics. ACS Appl. Mater. Interfaces 2018, 10, 8901–8908. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Hashimoto, K.; Koganezawa, T.; Tajima, K. End-On Orientation of Semiconducting Polymers in Thin Films Induced by Surface Segregation of Fluoroalkyl Chains. J. Am. Chem. Soc. 2013, 135, 9644–9647. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-C.; Chen, S.-Y.; Yang, Y.-W.; Hsu, C.-S.; Tajima, K. Face-on Reorientation of π-Conjugated Polymers in Thin Films by Surface-Segregated Monolayers. J. Mater. Chem. A 2020, 8, 6268–6275. [Google Scholar] [CrossRef]
- Don Park, Y.; Lim, J.A.; Lee, H.S.; Cho, K. Interface Engineering in Organic Transistors. Mater. Today 2007, 10, 46–54. [Google Scholar] [CrossRef]
- Sharma, S.; Vats, A.K.; Tang, L.; Kaishan, F.; Toyoda, J.; Nagamatsu, S.; Ando, Y.; Tamagawa, M.; Tanaka, H.; Pandey, M.; et al. High Field-Effect Mobility in Oriented Thin Films of D-A Type Semiconducting Polymers by Engineering Stable Interfacial System. Chem. Eng. J. 2023, 469, 143932. [Google Scholar] [CrossRef]
- Syafutra, H.; Toyoda, J.; Pandey, M.; Kumari, N.; Benten, H.; Nakamura, M. Perfectness of the Main-Chain Alignment in the Conjugated Polymer Films Prepared by the Floating Film Transfer Method. Appl. Phys. Lett. 2022, 120, 203301. [Google Scholar] [CrossRef]
- DeLongchamp, D.M.; Vogel, B.M.; Jung, Y.; Gurau, M.C.; Richter, C.A.; Kirillov, O.A.; Obrzut, J.; Fischer, D.A.; Sambasivan, S.; Richter, L.J.; et al. Variations in Semiconducting Polymer Microstructure and Hole Mobility with Spin-Coating Speed. Chem. Mater. 2005, 17, 5610–5612. [Google Scholar] [CrossRef]
- Rivnay, J.; Steyrleuthner, R.; Jimison, L.H.; Casadei, A.; Chen, Z.; Toney, M.F.; Facchetti, A.; Neher, D.; Salleo, A. Drastic Control of Texture in a High Performance N-Type Polymeric Semiconductor and Implications for Charge Transport. Macromolecules 2011, 44, 5246–5255. [Google Scholar] [CrossRef]
- Biniek, L.; Leclerc, N.; Heiser, T.; Bechara, R.; Brinkmann, M. Large Scale Alignment and Charge Transport Anisotropy of PBTTT Films Oriented by High Temperature Rubbing. Macromolecules 2013, 46, 4014–4023. [Google Scholar] [CrossRef]
- Kumari, N.; Pandey, M.; Hamada, K.; Hirotani, D.; Nagamatsu, S.; Hayase, S.; Pandey, S.S. Role of Device Architecture and AlOX Interlayer in Organic Schottky Diodes and Their Interpretation by Analytical Modeling. J. Appl. Phys. 2019, 126, 125501. [Google Scholar] [CrossRef]
- Sharma, S.; Nagamatsu, S.; Singh, V.; Pandey, S.S. Facile Fabrication and Characterization of Oriented and Multilayer Thin Films of Solution Processable Conjugated Polymer. Phys. Status Solidi 2023, 220, 2300194. [Google Scholar] [CrossRef]
- Ma, L.; Pyo, S.; Ouyang, J.; Xu, Q.; Yang, Y. Nonvolatile Electrical Bistability of Organic/Metal-Nanocluster/Organic System. Appl. Phys. Lett. 2003, 82, 1419–1421. [Google Scholar] [CrossRef]
- Nagamatsu, S.; Misaki, M.; Chikamatsu, M.; Kimura, T.; Yoshida, Y.; Azumi, R.; Tanigaki, N.; Yase, K. Crystal Structure of Friction-Transferred Poly(2,5-Dioctyloxy-1,4-Phenylenevinylene). J. Phys. Chem. B 2007, 111, 4349–4354. [Google Scholar] [CrossRef]
- Sharma, S.; Vats, A.K.; Pandey, M.; Nagamatsu, S.; Chen, J.-C.; Pandey, S.S. Unraveling the Implications of Macromolecular Orientation on the Planar and Vertical Charge Transport in Organic Electronic Devices. ACS Appl. Polym. Mater. 2022, 4, 8315–8323. [Google Scholar] [CrossRef]
- Sharma, S.; Kumari, N.; Nagamatsu, S.; Nakamura, M.; Pandey, S.S. Bistable Resistive Memory Switches Fabricated by Floating Thin Films of Conjugated Polymers. Mater. Today Electron. 2023, 4, 100043. [Google Scholar] [CrossRef]
- Liu, C.; Li, G.; Di Pietro, R.; Huang, J.; Noh, Y.-Y.; Liu, X.; Minari, T. Device Physics of Contact Issues for the Overestimation and Underestimation of Carrier Mobility in Field-Effect Transistors. Phys. Rev. Appl. 2017, 8, 034020. [Google Scholar] [CrossRef]
- Choi, H.H.; Cho, K.; Frisbie, C.D.; Sirringhaus, H.; Podzorov, V. Critical Assessment of Charge Mobility Extraction in FETs. Nat. Mater. 2018, 17, 2–7. [Google Scholar] [CrossRef]
- Gunduz, B.; Yahia, I.S.; Yakuphanoglu, F. Electrical and Photoconductivity Properties of P-Si/P3HT/Al and p-Si/P3HT:MEH-PPV/Al Organic Devices: Comparison Study. Microelectron. Eng. 2012, 98, 41–57. [Google Scholar] [CrossRef]
- Güllü, Ö.; Aydoğan, Ş.; Türüt, A. High Barrier Schottky Diode with Organic Interlayer. Solid State Commun. 2012, 152, 381–385. [Google Scholar] [CrossRef]
- Kaneto, K.; Takashima, W. Fabrication and Characteristics of Schottky Diodes Based on Regioregular Poly(3-Hexylthiophene)/Al Junction. Curr. Appl. Phys. 2001, 1, 355–361. [Google Scholar] [CrossRef]
- Sze, S.M.; Ng, K.K. Physics of Semiconductor Devices; Wiley: Hoboken, NJ, USA, 2006; ISBN 9780471143239. [Google Scholar]
- Ma, L.P.; Liu, J.; Yang, Y. Organic Electrical Bistable Devices and Rewritable Memory Cells. Appl. Phys. Lett. 2002, 80, 2997–2999. [Google Scholar] [CrossRef]
- Bozano, L.D.; Kean, B.W.; Beinhoff, M.; Carter, K.R.; Rice, P.M.; Scott, J.C. Organic Materials and Thin-Film Structures for Cross-Point Memory Cells Based on Trapping in Metallic Nanoparticles. Adv. Funct. Mater. 2005, 15, 1933–1939. [Google Scholar] [CrossRef]
- Sethuraman, K.; Ochiai, S.; Kojima, K.; Mizutani, T. Performance of Poly(3-Hexylthiophene) Organic Field-Effect Transistors on Cross-Linked Poly(4-Vinyl Phenol) Dielectric Layer and Solvent Effects. Appl. Phys. Lett. 2008, 92, 183302. [Google Scholar] [CrossRef]
- Singh, K.A.; Sauvé, G.; Zhang, R.; Kowalewski, T.; McCullough, R.D.; Porter, L.M. Dependence of Field-Effect Mobility and Contact Resistance on Nanostructure in Regioregular Poly(3-Hexylthiophene) Thin Film Transistors. Appl. Phys. Lett. 2008, 92, 263303. [Google Scholar] [CrossRef]
- Han, T.; Sun, L.; Guo, Y.; Ding, S.; Jin, G.; Jiang, C.; Huang, X.; Zhang, X.; Chang, F. New Strategy for Enhancing Performance of P3HT-Based Organic Field-Effect Transistor via Regulating the Precipitated Speed of Solute in Volatile Solvents. Polym. Test. 2020, 90, 106788. [Google Scholar] [CrossRef]
- Na, J.Y.; Kang, B.; Sin, D.H.; Cho, K.; Park, Y.D. Understanding Solidification of Polythiophene Thin Films during Spin-Coating: Effects of Spin-Coating Time and Processing Additives. Sci. Rep. 2015, 5, 13288. [Google Scholar] [CrossRef]
- Podzorov, V.; Sysoev, S.E.; Loginova, E.; Pudalov, V.M.; Gershenson, M.E. Single-Crystal Organic Field Effect Transistors with the Hole Mobility ~8 cm2/Vs. Appl. Phys. Lett. 2003, 83, 3504–3506. [Google Scholar] [CrossRef]
- Chaudhary, V.; Pandey, R.K.; Prakash, R.; Singh, A.K. Self-Assembled H-Aggregation Induced High Performance Poly (3-Hexylthiophene) Schottky Diode. J. Appl. Phys. 2017, 122, 225501. [Google Scholar] [CrossRef]
- Ferchichi, K.; Pecqueur, S.; Guerin, D.; Bourguiga, R.; Lmimouni, K. High Rectification Ratio in Polymer Diode Rectifier through Interface Engineering with Self-Assembled Monolayer. Electron. Mater. 2021, 2, 445–453. [Google Scholar] [CrossRef]
- Feng, Y.; Gao, X.; Zhong, Y.-N.; Wu, J.-L.; Xu, J.-L.; Wang, S.-D. Solution-Processed Polymer Thin-Film Memristors with an Electrochromic Feature and Frequency-Dependent Synaptic Plasticity. Adv. Intell. Syst. 2019, 1, 1900022. [Google Scholar] [CrossRef]
- Oh, S.; Kim, H.; Kim, S.E.; Kim, M.-H.; Park, H.-L.; Lee, S.-H. Biodegradable and Flexible Polymer-Based Memristor Possessing Optimized Synaptic Plasticity for Eco-Friendly Wearable Neural Networks with High Energy Efficiency. Adv. Intell. Syst. 2023, 5, 2200272. [Google Scholar] [CrossRef]
- Yang, F.; Sun, L.; Duan, Q.; Dong, H.; Jing, Z.; Yang, Y.; Li, R.; Zhang, X.; Hu, W.; Chua, L. Vertical-organic-nanocrystal-arrays for Crossbar Memristors with Tuning Switching Dynamics toward Neuromorphic Computing. SmartMat 2021, 2, 99–108. [Google Scholar] [CrossRef]
- Nguyen, V.C.; Lee, P.S. Coexistence of Write Once Read Many Memory and Memristor in Blend of Poly(3,4-Ethylenedioxythiophene): Polystyrene Sulfonate and Polyvinyl Alcohol. Sci. Rep. 2016, 6, 38816. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, S.; Gaurav, K.V.; Nagamatsu, S.; Pandey, S.S. The Influence of a Microstructural Conformation of Oriented Floating Films of Semiconducting Polymers on Organic Device Performance. Polymers 2024, 16, 710. https://doi.org/10.3390/polym16050710
Sharma S, Gaurav KV, Nagamatsu S, Pandey SS. The Influence of a Microstructural Conformation of Oriented Floating Films of Semiconducting Polymers on Organic Device Performance. Polymers. 2024; 16(5):710. https://doi.org/10.3390/polym16050710
Chicago/Turabian StyleSharma, Shubham, Kumar Vivek Gaurav, Shuichi Nagamatsu, and Shyam S. Pandey. 2024. "The Influence of a Microstructural Conformation of Oriented Floating Films of Semiconducting Polymers on Organic Device Performance" Polymers 16, no. 5: 710. https://doi.org/10.3390/polym16050710
APA StyleSharma, S., Gaurav, K. V., Nagamatsu, S., & Pandey, S. S. (2024). The Influence of a Microstructural Conformation of Oriented Floating Films of Semiconducting Polymers on Organic Device Performance. Polymers, 16(5), 710. https://doi.org/10.3390/polym16050710