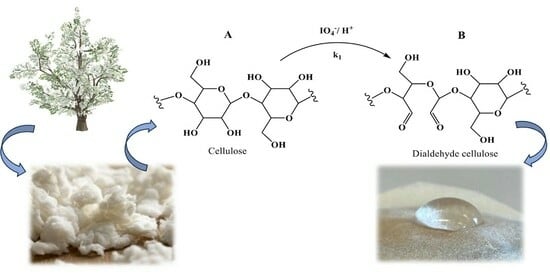
Kinetics of Periodate-Mediated Oxidation of Cellulose
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of DAC
2.3. Determination of Aldehyde Content
2.4. Fourier-Transform Infrared Spectroscopy, FTIR
2.5. Field Emission Scanning Electron Microscopy, FE-SEM
2.6. Contact Angle Measurements
2.7. Design of Oxidation Experiments for Kinetic Modeling
3. Results and Discussion
3.1. Characterization of the Synthesized DAC
3.2. Morphology of DAC and Cellulose Fibers
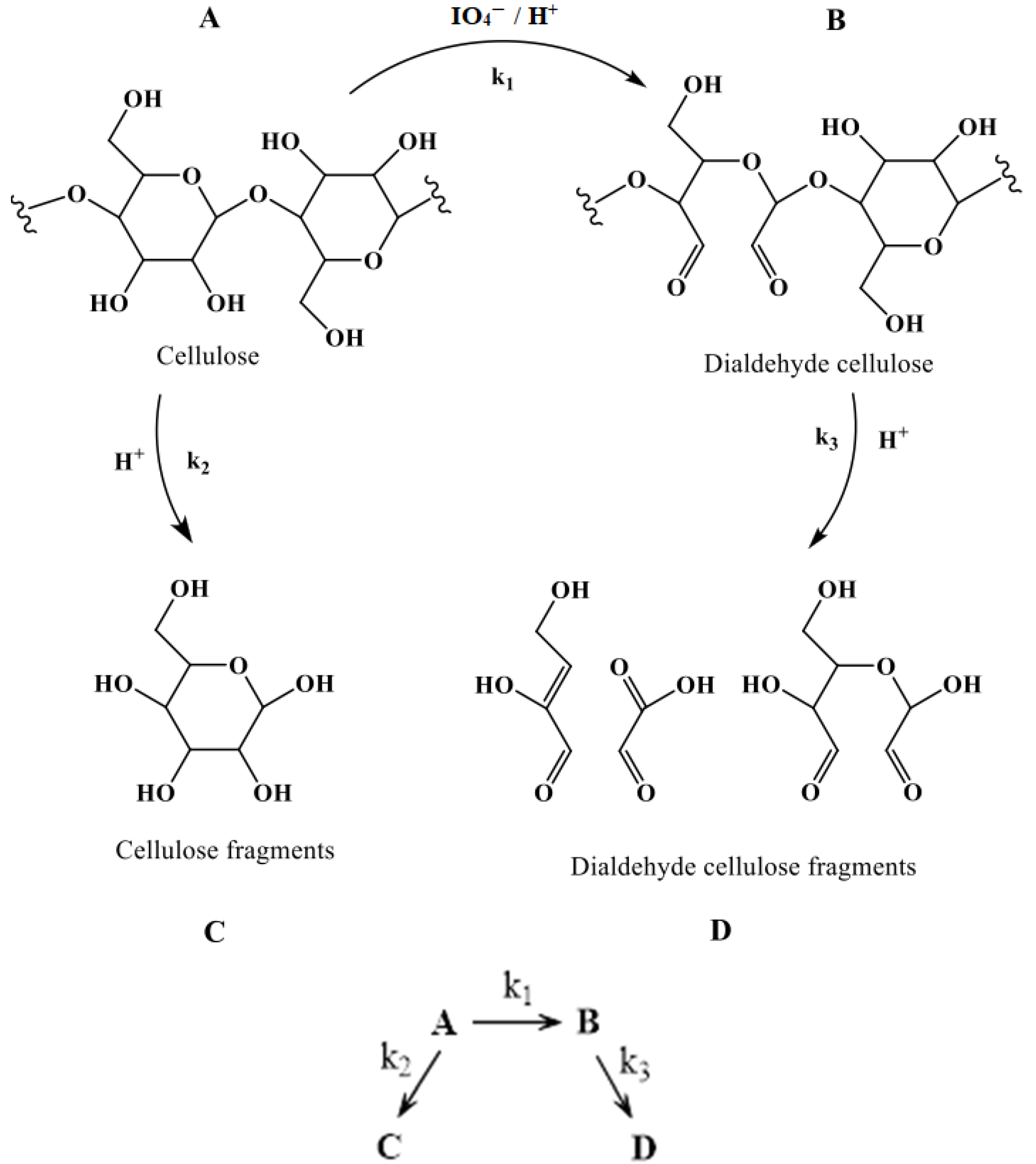
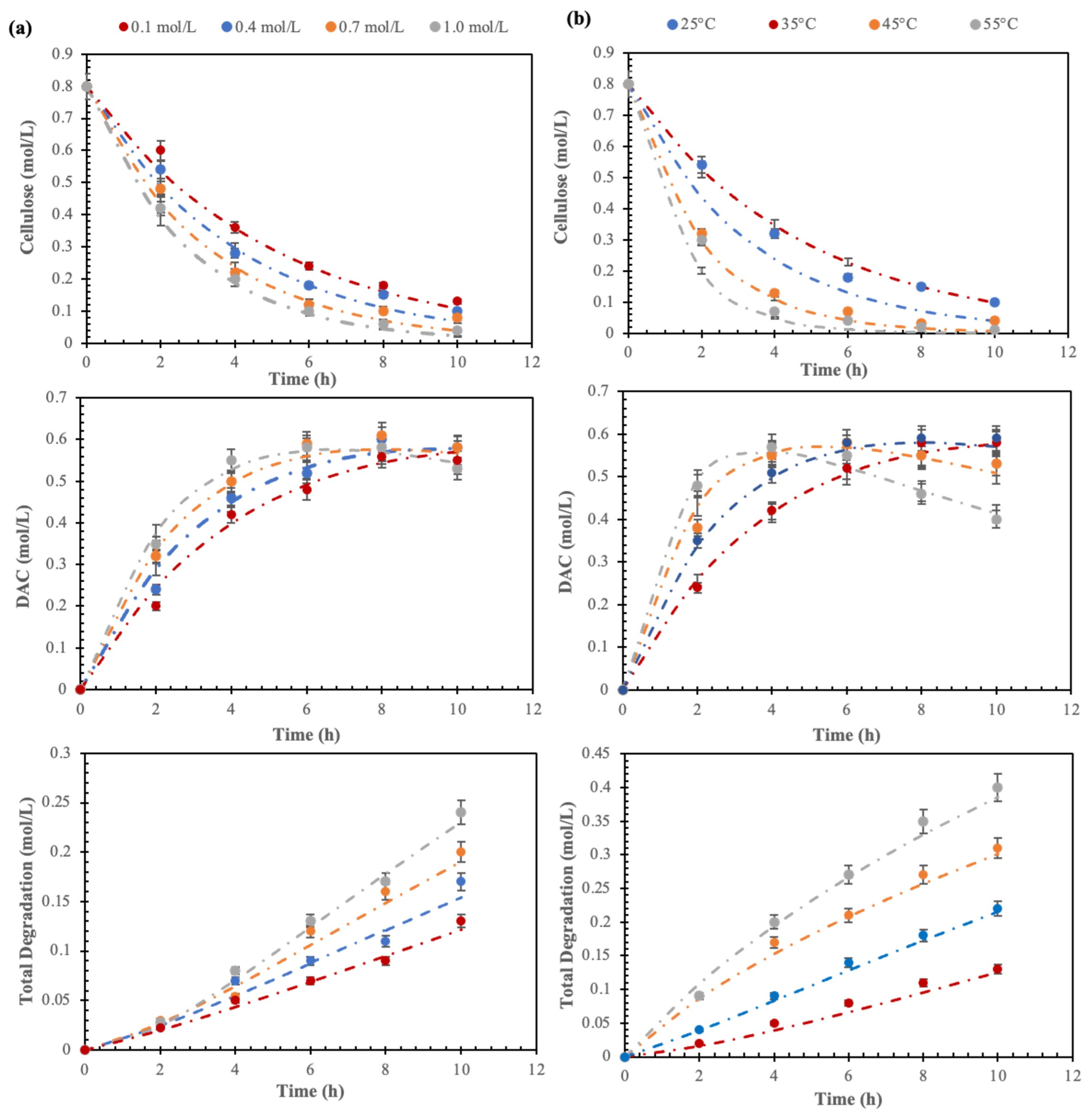
3.3. Cellulose Oxidation Kinetics
3.4. Synthesis Parameter Impact on Cellulose Oxidation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heinze, T.; El Seoud, O.A.; Koschella, A.; Heinze, T.; El Seoud, O.A.; Koschella, A. Production and characteristics of cellulose from different sources. In Cellulose Derivatives: Synthesis, Structure, and Properties, 1st ed.; Springer: Cham, Switzerland, 2018; pp. 1–38. [Google Scholar]
- Kim, U.-J.; Kuga, S.; Wada, M.; Okano, T.; Kondo, T. Periodate oxidation of crystalline cellulose. Biomacromolecules 2000, 1, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Chavan, V.; Sarwade, B.; Varma, A. Morphology of cellulose and oxidised cellulose in powder form. Carbohydr. Polym. 2002, 50, 41–45. [Google Scholar] [CrossRef]
- Kim, U.-J.; Kuga, S. Ion-exchange chromatography by dicarboxyl cellulose gel. J. Chromatogr. A 2001, 919, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Potthast, A.; Kostic, M.; Schiehser, S.; Kosma, P.; Rosenau, T. Studies on oxidative modifications of cellulose in the periodate system: Molecular weight distribution and carbonyl group profiles. Holzforschung 2007, 61, 662–666. [Google Scholar] [CrossRef]
- Vicini, S.; Princi, E.; Luciano, G.; Franceschi, E.; Pedemonte, E.; Oldak, D.; Kaczmarek, H.; Sionkowska, A. Thermal analysis and characterisation of cellulose oxidised with sodium methaperiodate. Thermochim. Acta 2004, 418, 123–130. [Google Scholar] [CrossRef]
- Koshani, R.; Tavakolian, M.; van de Ven, T.G. Natural emulgel from dialdehyde cellulose for lipophilic drug delivery. ACS Sust. Chem. Eng. 2021, 9, 4487–4497. [Google Scholar] [CrossRef]
- Wu, M.; Kuga, S. Cationization of cellulose fabrics by polyallylamine binding. J. Appl. Polym. Sci. 2006, 100, 1668–1672. [Google Scholar] [CrossRef]
- Nichols, B.L.; Chen, J.; Mischnick, P.; Edgar, K.J. selective oxidation of 2-hydroxypropyl ethers of cellulose and dextran: Simple and efficient introduction of versatile ketone groups to polysaccharides. Biomacromolecules 2020, 21, 4835–4849. [Google Scholar] [CrossRef]
- Nypelö, T.; Berke, B.; Spirk, S.; Sirviö, J.A. Periodate oxidation of wood polysaccharides—Modulation of hierarchies. Carbohydr. Polym. 2021, 252, 117105. [Google Scholar] [CrossRef]
- Isobe, N.; Lee, D.-S.; Kwon, Y.-J.; Kimura, S.; Kuga, S.; Wada, M.; Kim, U.-J. Immobilization of protein on cellulose hydrogel. Cellulose 2011, 18, 1251–1256. [Google Scholar] [CrossRef]
- Huang, X.; Hadi, P.; Joshi, R.; Alhamzani, A.G.; Hsiao, B.S. A comparative study of mechanism and performance of anionic and cationic dialdehyde nanocelluloses for dye adsorption and separation. ACS Omega 2023, 8, 8634–8649. [Google Scholar] [CrossRef]
- Dalei, G.; Das, S.; Pradhan, M. Dialdehyde cellulose as a niche material for versatile applications: An overview. Cellulose 2022, 29, 5429–5461. [Google Scholar] [CrossRef]
- Maekawa, E.; Koshijima, T. Properties of 2, 3-dicarboxy cellulose combined with various metallic ions. J. Appl. Polym. Sci. 1984, 29, 2289–2297. [Google Scholar] [CrossRef]
- Varma, A.J.; Kulkarni, M.P. Oxidation of cellulose under controlled conditions. Polym. Degr. Stab. 2002, 77, 25–27. [Google Scholar] [CrossRef]
- Sirvio, J.; Hyvakko, U.; Liimatainen, H.; Niinimaki, J.; Hormi, O. Periodate oxidation of cellulose at elevated temperatures using metal salts as cellulose activators. Carbohydr. Polym. 2011, 83, 1293–1297. [Google Scholar] [CrossRef]
- Goldfinger, G.; Mark, H.; Siggia, S. Kinetics of oxidation of cellulose with periodic acid. Ind. Eng. Chem. 1943, 35, 1083–1086. [Google Scholar] [CrossRef]
- Xu, Y.H.; Huang, C. Effect of sodium periodate selective oxidation on crystallinity of cotton cellulose. AMR 2011, 197–198, 1201–1204. [Google Scholar] [CrossRef]
- Lucia, A.; van Herwijnen, H.W.; Oberlerchner, J.T.; Rosenau, T.; Beaumont, M. Resource-Saving Production of Dialdehyde Cellulose: Optimization of the Process at High Pulp Consistency. ChemSusChem 2019, 12, 4679–4684. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, L.; Song, X.; Song, H.; Zhao, J.R.; Wang, S. A kinetic model for oxidative degradation of bagasse pulp fiber by sodium periodate. Carbohydr. Polym. 2012, 90, 218–223. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, L.P.; Hudebine, D.; Guillaume, D.; Verstraete, J.J. A Review of Kinetic Modeling Methodologies for Complex Processes. Oil Gas Sci. Technol. 2016, 71, 45–49. [Google Scholar] [CrossRef]
- Taylor, C.J.; Seki, H.; Dannheim, F.M.; Willis, M.J.; Clemens, G.; Taylor, B.A.; Chamberlain, T.W.; Bourne, R.A. An automated computational approach to kinetic model discrimination and parameter estimation. React. Chem. Eng. 2021, 6, 1404–1411. [Google Scholar] [CrossRef] [PubMed]
- Larsson, P.A.; Berglund, L.A.; Wågberg, L. Ductile all-cellulose nanocomposite films fabricated from core–shell structured cellulose nanofibrils. Biomacromolecules 2014, 15, 2218–2223. [Google Scholar] [CrossRef] [PubMed]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical review of rate constants for reactions of hydrated electrons, hydrogen atoms and hydroxyl radicals (⋅OH/⋅O− in aqueous solution. J. Phys. Chem. Ref. Data 1988, 17, 513–886. [Google Scholar] [CrossRef]
- Painter, T.J. Control of depolymerisation during the preparation of reduced dialdehyde cellulose. Carbohydr. Res. 1988, 179, 259–268. [Google Scholar] [CrossRef]
- Symons, M. Evidence for formation of free-radical intermediates in some reactions involving periodate. J. Chem. Soc. 1955, 2794–2796. [Google Scholar] [CrossRef]
- Zhao, H.; Heindel, N.D. Determination of degree of substitution of formyl groups in polyaldehyde dextran by the hydroxylamine hydrochloride method. Pharm. Res. 1991, 8, 400–402. [Google Scholar] [CrossRef] [PubMed]
- Larsson, P.A.; Gimåker, M.; Wågberg, L. The influence of periodate oxidation on the moisture sorptivity and dimensional stability of paper. Cellulose 2008, 15, 837–847. [Google Scholar] [CrossRef]
- Schwanninger, M.; Rodrigues, J.; Pereira, H.; Hinterstoisser, B. Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vib. Spec. 2004, 36, 23–40. [Google Scholar] [CrossRef]
- Simon, J.; Tsetsgee, O.; Iqbal, N.A.; Sapkota, J.; Ristolainen, M.; Rosenau, T.; Potthast, A. A fast method to measure the degree of oxidation of dialdehyde celluloses using multivariate calibration and infrared spectroscopy. Carbohydr. Polym. 2022, 278, 118887. [Google Scholar] [CrossRef]
- Plappert, S.F.; Quraishi, S.; Pircher, N.; Mikkonen, K.S.; Veigel, S.; Klinger, K.M.; Potthast, A.; Rosenau, T.; Liebner, F.W. Transparent, flexible, and strong 2, 3-dialdehyde cellulose films with high oxygen barrier properties. Biomacromolecules 2018, 19, 2969–2978. [Google Scholar] [CrossRef]
- Sulaeva, I.; Klinger, K.M.; Amer, H.; Henniges, U.; Rosenau, T.; Potthast, A. Determination of molar mass distributions of highly oxidized dialdehyde cellulose by size exclusion chromatography and asymmetric flow field-flow fractionation. Cellulose 2015, 22, 3569–3581. [Google Scholar] [CrossRef]
- Maguire, R.J. Kinetics of the hydrolysis of cellulose by β-1, 4-glucan cellobiohydrolase of Trichoderma viride. Can. J. Biochem. 1977, 55, 644–650. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Ray, A.R.; Vasudevan, P. Biodegradation studies on periodate oxidized cellulose. Biomaterials 1982, 3, 16–20. [Google Scholar] [CrossRef]
- Potthast, A.; Rosenau, T.; Kosma, P. Analysis of oxidized functionalities in cellulose. Adv. Polym. Sci. 2006, 205, 1–48. [Google Scholar]
- Li, H.; Wu, B.; Mu, C.; Lin, W. Concomitant degradation in periodate oxidation of carboxymethyl cellulose. Carbohydr. Polym. 2011, 84, 881–886. [Google Scholar] [CrossRef]
- Simon, J.; Schlapp-Hackl, I.; Sapkota, J.; Ristolainen, M.; Rosenau, T.; Potthast, A. Towards Tailored Dialdehyde Cellulose Derivatives: A Strategy for Tuning the Glass Transition Temperature. ChemSusChem 2023, e20230079136. [Google Scholar] [CrossRef]
- Mostafa, A.G.; Gaith, E.A.; Akl, M.A. Aminothiol supported dialdehyde cellulose for efficient and selective removal of Hg(II) from aquatic solutions. Sci. Rep. 2023, 13, 19507. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, E.S.A.; Dacrory, S.; Essawy, H.A.; Ibrahim, H.S.; Ammar, N.S.; Kamel, S. Sustainable grafted chitosan-dialdehyde cellulose with high adsorption capacity of heavy metal. BMC Chem. 2023, 17, 117. [Google Scholar] [CrossRef]
- Liu, P.; Pang, B.; Dechert, S.; Zhang, X.C.; Andreas, L.B.; Fischer, S.; Meyer, F.; Zhang, K. Structure selectivity of alkaline periodate oxidation on lignocellulose for facile isolation of cellulose nanocrystals. Angew. Chem. 2020, 59, 3218–3225. [Google Scholar] [CrossRef]
- Yang, C.W.; Choi, H.M. Preparation of water-soluble dialdehyde nanocelluloses by periodate oxidation under microwave irradiation. Cellulose Chem. Technol. 2020, 54, 247–258. [Google Scholar] [CrossRef]




| Experiment Number | Temperature (°C) | Concentration of IO4− (mol/L) |
|---|---|---|
| 1 | 0.01 | |
| 2 | 25 | 0.04 |
| 3 | 0.07 | |
| 4 | 1.0 | |
| 5 | 25 | |
| 6 | 35 | 0.5 |
| 7 | 45 | |
| 8 | 55 |
| Temperature (°C) | Concentration (mol/L) | k1 (h−1) | k2 (h−1) | k3 (h−1) | |
|---|---|---|---|---|---|
| IO4− concentration variation | 0.01 | 0.1896 | 0.0110 | 0.0213 | |
| 0.04 | 0.2357 | 0.0128 | 0.0270 | ||
| 25 | 0.07 | 0.2893 | 0.0135 | 0.0352 | |
| 1.0 | 0.3475 | 0.0165 | 0.0420 | ||
| Temperature variation | 25 | 0.2363 | 0.0108 | 0.0281 | |
| 35 | 0.5 | 0.2910 | 0.0117 | 0.0353 | |
| 45 | 0.4441 | 0.0519 | 0.0421 | ||
| 55 | 0.6121 | 0.0809 | 0.0630 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sultana, N.; Edlund, U.; Guria, C.; Westman, G. Kinetics of Periodate-Mediated Oxidation of Cellulose. Polymers 2024, 16, 381. https://doi.org/10.3390/polym16030381
Sultana N, Edlund U, Guria C, Westman G. Kinetics of Periodate-Mediated Oxidation of Cellulose. Polymers. 2024; 16(3):381. https://doi.org/10.3390/polym16030381
Chicago/Turabian StyleSultana, Nazmun, Ulrica Edlund, Chandan Guria, and Gunnar Westman. 2024. "Kinetics of Periodate-Mediated Oxidation of Cellulose" Polymers 16, no. 3: 381. https://doi.org/10.3390/polym16030381
APA StyleSultana, N., Edlund, U., Guria, C., & Westman, G. (2024). Kinetics of Periodate-Mediated Oxidation of Cellulose. Polymers, 16(3), 381. https://doi.org/10.3390/polym16030381