Effects of Recycled Polyethylene on Natural Rubber Composite Blends Filled with Aluminum Trihydroxide and Polyurethane Waste: Mechanical and Dynamic Mechanical Properties, Flammability
Abstract
1. Introduction
2. Experimental and Characterization
2.1. Materials
2.2. Preparation of NR/PE Composites Filled with PU Waste, SiO2, and ATH
3. Characterization
3.1. Mechanical Properties
3.2. Impact Strength
3.3. Dynamic Mechanical Properties
3.4. Thermal Properties
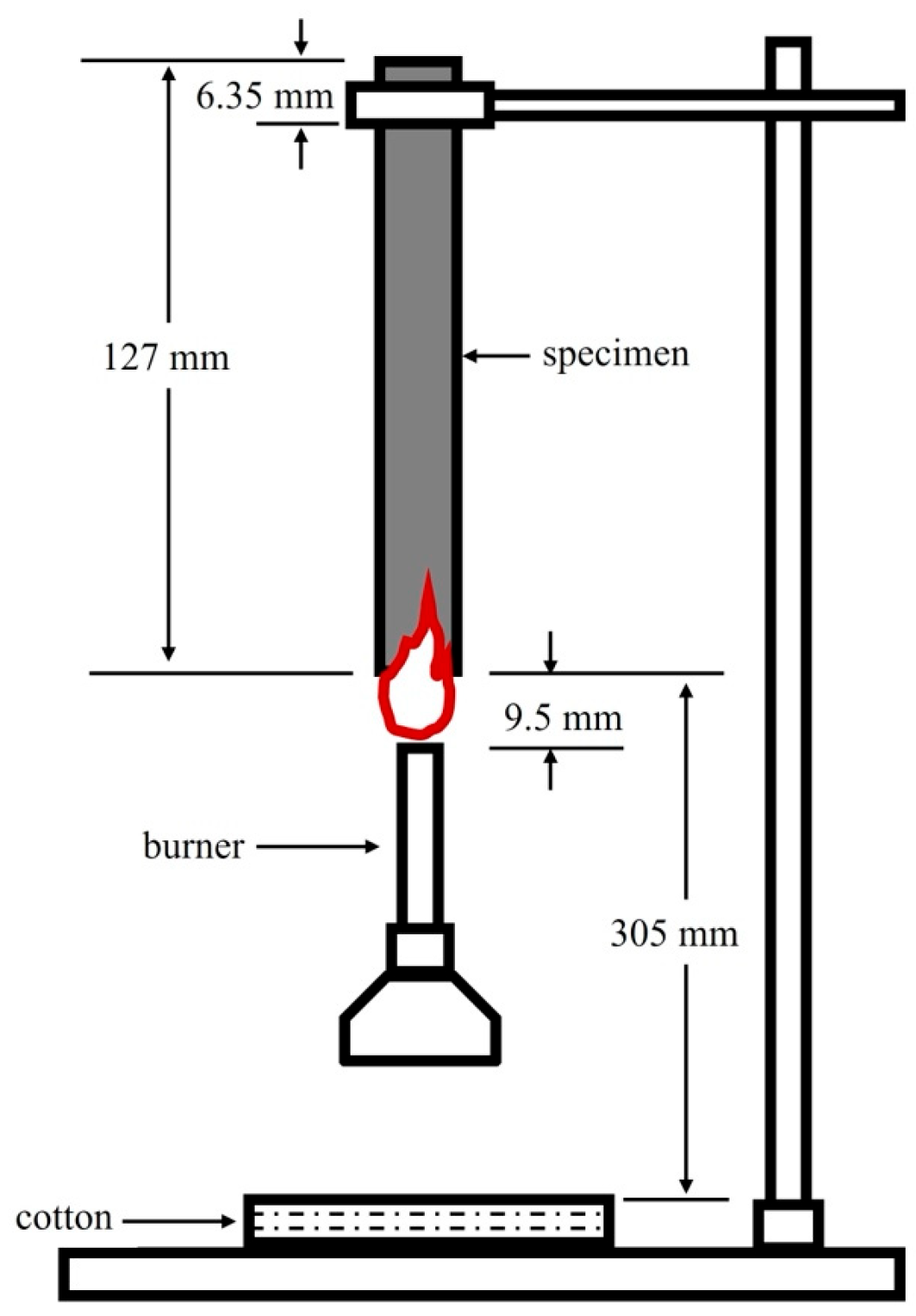
3.5. UL-94 Flammability Standard Test
4. Results and Discussion
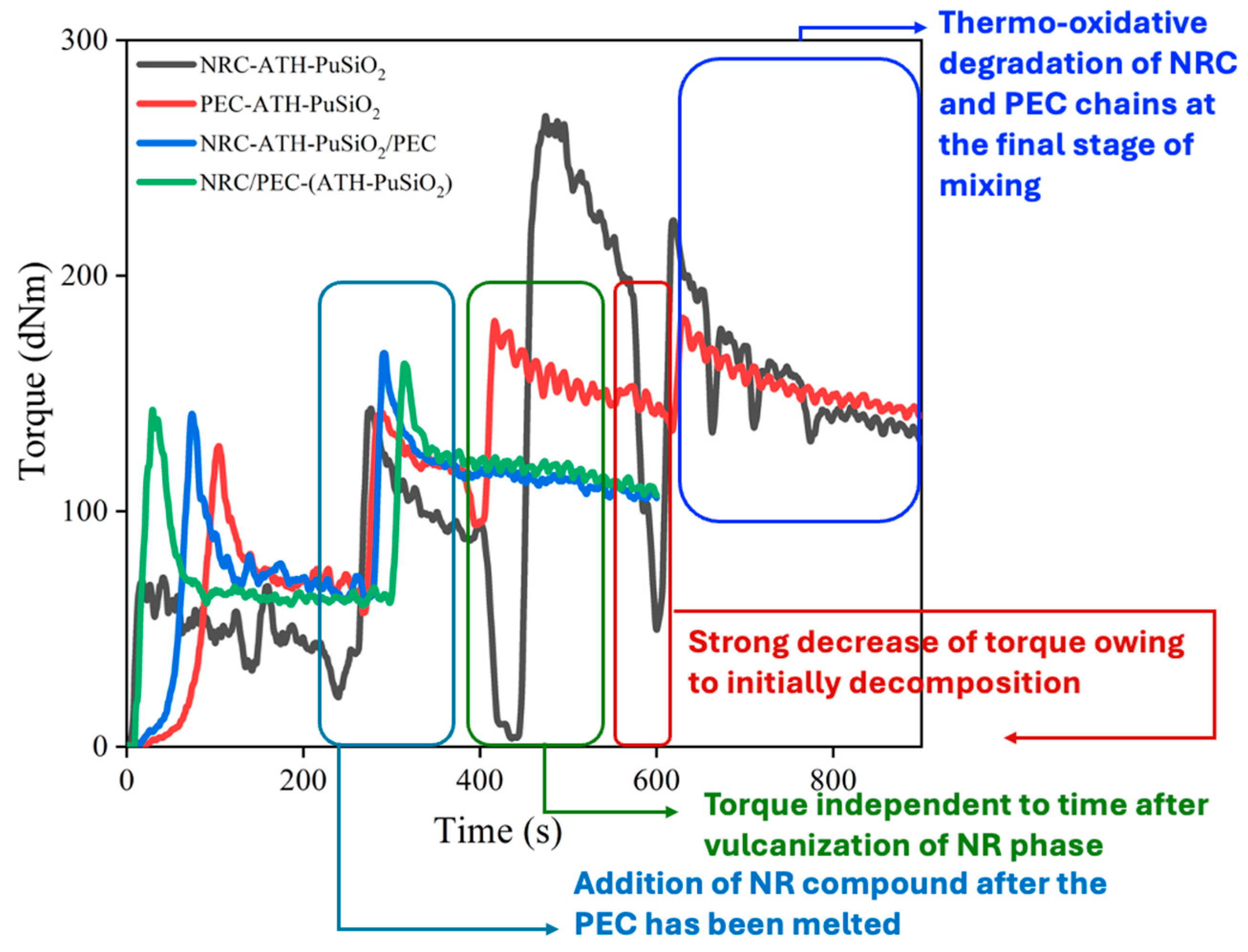
4.1. Mixing Torque
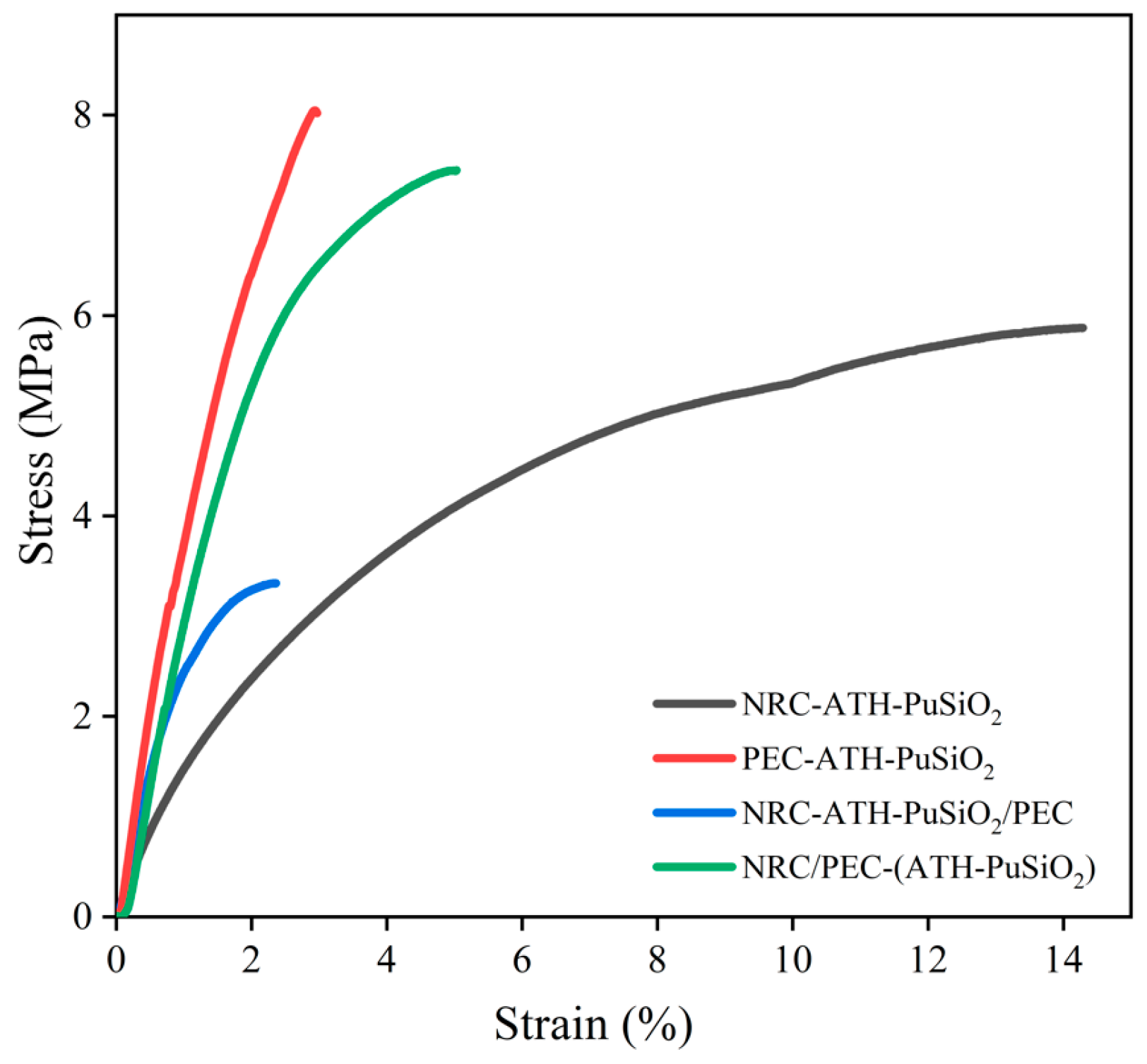
4.2. Mechanical Properties
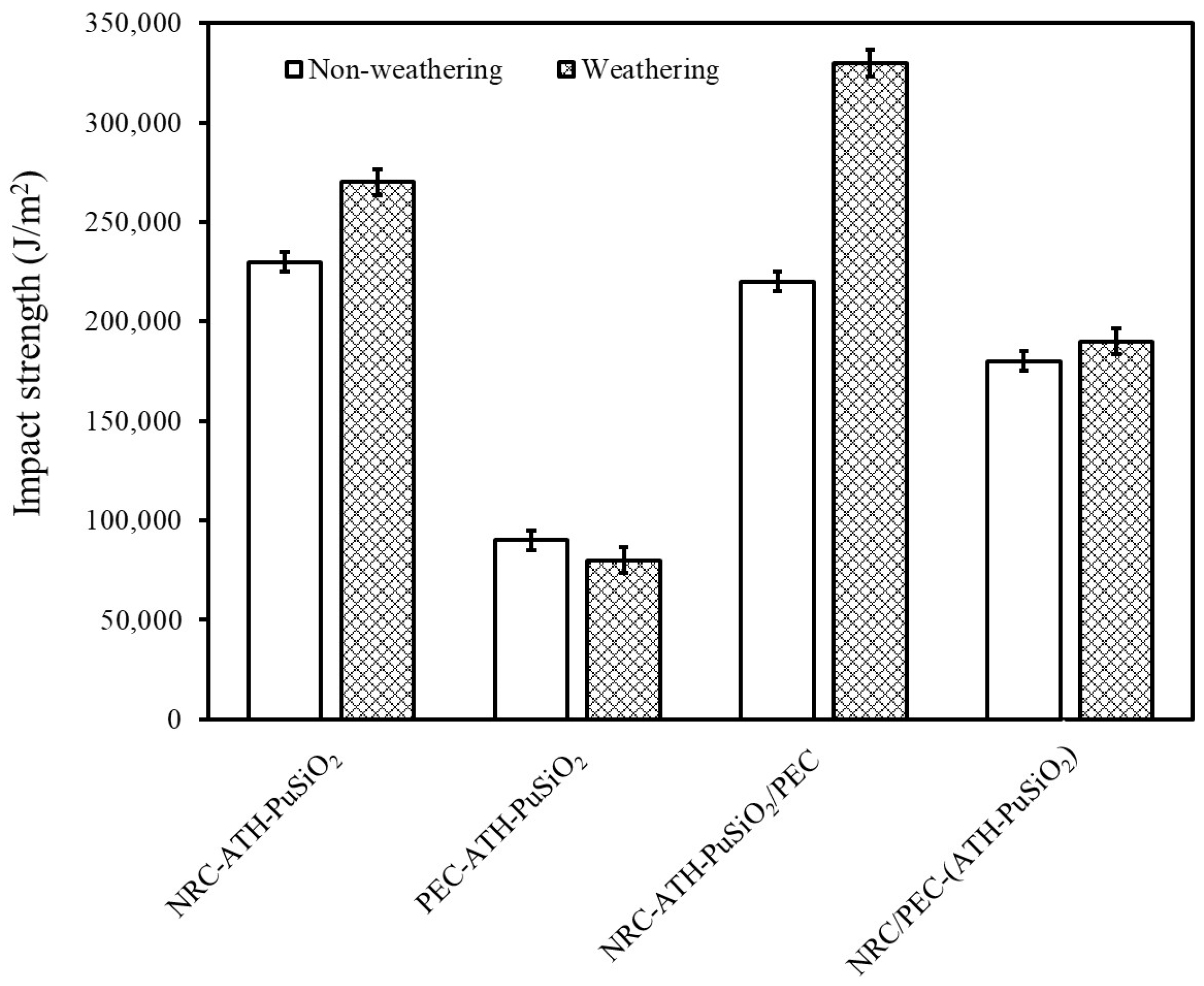
4.3. Impact Strength
4.4. Dynamic Mechanical Properties
4.5. Thermal Properties
4.6. UL-94 Flammability
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Yao, Z.; Seong, H.J.; Jang, Y.S. Environmental toxicity and decomposition of polyethylene. Ecotoxicol. Environ. Saf. 2022, 242, 113933. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Sánchez, F.; Olayo, R.; Manzur, A. Effect of NR and EPDM on the rheology of HDPE/PP blends. Polym. Bull. 1999, 42, 481–488. [Google Scholar] [CrossRef]
- Maziad, N.A.; El-Nashar, D.E.; Sadek, E.M. The effects of a silane coupling agent on properties of rice husk-filled maleic acid anhydride compatibilized natural rubber/low-density polyethylene blend. J. Mater. Sci. 2009, 44, 2665–2673. [Google Scholar] [CrossRef]
- Srilathakutty, R.; Joseph, R.; George, K.E. Studies on microcellular soles based on natural rubber/polyethylene blends. J. Mater. Sci. 1999, 34, 1493–1495. [Google Scholar] [CrossRef]
- Wickramaarachchi, W.V.W.H.; Walpalage, S.; Egodage, S.M. Identification of the polyethylene grade most suitable for natural rubber-polyethylene blends used for roofing application. Eng. J. Inst. Eng. 2016, 49, 9–14. [Google Scholar] [CrossRef]
- Zoran, S. Polyurethanes. In Handbook of Polymer Synthesis; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Hepburn, C. Polyurethane Elastomers; Applied Science Publishers: London, UK, 1982. [Google Scholar]
- Bhowmick, A.K.; Stephens, H.L. Handbook of Elastomers; Marcel Dekker, Inc.: New York, NY, USA, 2001. [Google Scholar]
- Chen, T.K.; Tien, Y.I.; Wei, K.H. Synthesis and characterization of novel segmented polyurethane/clay nanocomposites. Polymer 2001, 41, 1345–1353. [Google Scholar] [CrossRef]
- Tien, Y.I.; Wei, K.H. High-tensile-property layered silicates/ polyurethane nanocomposites by using reactive silicates as pseudo chain extenders. Macromolecules 2001, 34, 9045–9052. [Google Scholar] [CrossRef]
- Xiong, J.; Liu, Y.; Yang, X.; Wang, X. Thermal and mechanical properties of polyurethane/montmorillonite nanocomposites based on a novel reactive modifier. Polym. Degrad. Stab. 2001, 86, 549–555. [Google Scholar] [CrossRef]
- Berta, M.; Lindsay, C.; Pans, G.; Camino, G. Effect of chemical structure on combustion and thermal behavior of polyurethane elastomer layered silicate nanocomposites. Polym. Degrad. Stab. 2006, 91, 1179–1191. [Google Scholar] [CrossRef]
- Meng, X.; Du, X.; Wang, Z.; Bi, W.; Tang, T. The investigation of exfoliation process of organic modified montmorillonite in thermoplastic polyurethane with different molecular weights. Compos. Sci. Technol. 2008, 68, 1815–1821. [Google Scholar] [CrossRef]
- Chattopadhyay, D.K.; Webster, D.C. Thermal stability and flame retardancy of polyurethanes. Prog. Polym. Sci. 2009, 34, 1068–1133. [Google Scholar] [CrossRef]
- Liang, J.; Xu, Y.; Huang, Y.; Zhang, L.; Wang, Y.; Ma, Y.; Li, F.; Guo, T.; Chen, Y. Infrared-triggered actuators from graphene-based nanocomposites. J. Phys. Chem. C 2009, 113, 9921–9927. [Google Scholar] [CrossRef]
- Kim, H.; Miura, Y.; Macosko, C.W. Graphene/polyurethane nanocomposites for improved gas barrier and electrical conductivity. Chem. Mater. 2010, 22, 3441–3450. [Google Scholar] [CrossRef]
- Chang, C.C.; Chang, C.H. Preparation and characterization of polyurethane-gold nanocomposites prepared using encapsulated gold nanoparticles. Polym. Int. 2010, 59, 910–916. [Google Scholar] [CrossRef]
- Liu, H.L.; Dai, S.A.; Fu, K.Y.; Hsu, S. Antibacterial properties of silver nanoparticles in three different sizes and their nanocomposites with a new waterborne polyurethane. Int. J. Nanomed. 2010, 5, 1017–1028. [Google Scholar] [CrossRef] [PubMed]
- Chwang, C.P.; Liu, C.D.; Huang, S.W.; Chao, D.Y.; Lee, S.N. Synthesis and characterization of high dielectric constant polyaniline/polyurethane blends. Synth. Met. 2004, 142, 275–281. [Google Scholar] [CrossRef]
- Vicentini, D.S.; Barra, G.M.O.; Bertolino, J.R.; Pires, A.T.N. Polyaniline/thermoplastic polyurethane blends: Preparation and evaluation of electrical conductivity. Eur. Polym. J. 2007, 43, 4565–4572. [Google Scholar] [CrossRef]
- Gan, L.M.; Ni, H.Y.; Zhou, Y.J.; Chen, J. Study on vulcanization and thermal decomposition behaviors of methyl vinyl silicone rubber/polyurethane rubber blends. J. Macromol. Sci. Phys. 2011, 50, 1491–1499. [Google Scholar] [CrossRef]
- Shun, Z.; Hongdian, L.; Lei, S.; Zhengzhou, W.; Yuan, H.; Jianxiong, N.; Weiyi, X. Microencapsulated ammonium polyphosphate with polyurethane shell: Application to flame retarded polypropylene/ethylene-propylene diene terpolymer blends. J. Macromol. Sci. Pure Appl. Chem. 2009, 46, 136–144. [Google Scholar] [CrossRef]
- Emi, G.B.; Andela, P.; Ivan, S.; Mirela, L. Blends of thermoplastic polyurethane and polypropylene. II. Thermal and morphological behavior. J. Appl. Polym. Sci. 2010, 117, 1378–1384. [Google Scholar] [CrossRef]
- Wang, N.; Mi, L.; Wu, Y.; Zhang, J.; Fang, Q. Double-layered co-microencapsulated ammonium polyphosphate and mesoporous MCM-41 in intumescent flame-retardant natural rubber composites. J. Therm. Anal. Calorim. 2014, 115, 1173–1181. [Google Scholar] [CrossRef]
- Khanlari, S.; Kokabi, M. Thermal stability, aging properties, and flame resistance of NR-based nanocomposite. J. Appl. Polym. Sci. 2011, 119, 855–862. [Google Scholar] [CrossRef]
- Radhakrishnan, N.M.N.; Gopinathan, N.M.R. Thermogravimetric analysis of PVC/NR-b-PU blend. J. Therm. Anal. Calorim. 2011, 103, 863–872. [Google Scholar] [CrossRef]
- Rajalingam, V.; Radhakrishnan, G.; Francis, J.D. Thermoset polyurethanes from hydroxyl-terminated natural rubber. J. Appl. Polym. Sci. 1991, 43, 1385–1386. [Google Scholar] [CrossRef]
- Sung, C.S.P.; Schneider, N.S. Temperature dependence of hydrogen bonding in toluene diisocyanate based polyurethanes. Macromolecules 1977, 10, 452–458. [Google Scholar] [CrossRef]
- Teo, L.S.; Chen, C.Y.; Kuo, J.F. Fourier transform infrared spectroscopy study on effects of temperature on hydrogen bonding in amine-containing polyurethanes and poly(urethane–urea)s. Macromolecules 1997, 6, 1793–1799. [Google Scholar] [CrossRef]
- Seymour, R.W.; Cooper, S.L. Thermal analysis of polyurethane block polymers. Macromolecules 1973, 6, 48–53. [Google Scholar] [CrossRef]
- Lim, S.K.; Bee, S.T.; Sin, L.T.; Tee, T.T.; Ratnam, C.T.; Hui, D.; Rahmat, A.R. A review of application of ammonium polyphosphate as intumescent flame retardant in thermoplastic composites. Compos. B Eng. 2016, 84, 155–174. [Google Scholar] [CrossRef]
- Lewin, M.; Weil, E.D. Mechanisms and modes of action in flame retardancy of polymers. Fire Ret. Mater. 2001, 1, 31–68. [Google Scholar] [CrossRef]
- Mouritz, A.P.; Gibson, A.G. Fire Properties of Polymer Composite Materials; Solid Mechanics and Its Applications; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007; Volume 143. [Google Scholar]
- ISO37:2011; Standard Test Method for Rubber, Vulcanized or Thermoplastic—Mechanical Properties. International Standard: Determination of Tensile Stress-Strain Properties; International Organization for Standardization: Geneva, Switzerland, 2011.
- ASTM D2240-05; Standard Test Method for Rubber Property—Durometer Hardness. ASTM International: West Conshohocken, PA, USA, 2010.
- ASTM D256-10; Standard Test Methods for Determining the Izod Pendulum Impact Resistance of Plastics. ASTM International: West Conshohocken, PA, USA, 2018.
- IEC 60695-11-10; Fire hazard testing—Part 11-10: Test Flames—50 W Horizontal and Vertical Flame Test Methods. International Electrotechnical Commission: Geneva, Switzerland, 2013.
- Wan, L.; Deng, C.; Zhao, Z.Y.; Chen, H.; Wang, Y.Z. Flame retardation of natural rubber: Strategy and recent progress. Polymers 2020, 12, 429. [Google Scholar] [CrossRef]
- Laoutid, F.; Bonnaud, L.; Alexandre, M.; Lopez-Cuesta, J.M.; Dubois, P. New prospects in flame retardant polymer materials: From fundamentals to nanocomposites. Mater. Sci. Eng. 2009, 63, 100–125. [Google Scholar] [CrossRef]
- Namita, R.C.; Chaki, T.K.; Bhowmick, A.K. Thermal characterization of thermoplastic elastomeric natural rubber-polypropylene blends. Thermochim. Acta. 1991, 176, 149–161. [Google Scholar] [CrossRef]
- Bendjaouahdou, C.; Salima Bensaad, S. Aging studies of a polypropylene and natural rubber blend. Int. J. Ind. Chem. 2018, 9, 345–352. [Google Scholar] [CrossRef]
- Wissamitanan, T.; Dechwayukul, C.; Kalkornsurapranee, E.; Thongruang, W. Proper blends of biodegradable polycaprolactone and natural rubber for 3D printing. Polymers 2020, 12, 2416. [Google Scholar] [CrossRef] [PubMed]
- Burgoa, A.; Hernandez, R.; Vilas, J.L. Toward superior applications of thermoplastic elastomer blends: Double Tg increase and improved ductility. Polym. Int. 2019, 68, 1130–1139. [Google Scholar] [CrossRef]
- Katančić, Z.; Krehula, L.K.; Siročić, A.P.; Grozdanić, V.; Hrnjak-Murgić, Z. Effect of modified nanofillers on fire retarded high-density polyethylene/wood composites. J. Compos. Mater. 2013, 48, 3771–3783. [Google Scholar] [CrossRef]



| Chemicals | NRC-ATH-PuSiO2 | PEC-ATH-PuSiO2 | NRC-ATH-PuSiO2/PEC | NRC/PEC-(ATH-PuSiO2) |
|---|---|---|---|---|
| Pure PE existing | 0 phr | 100 phr | 50 phr | 10 phr |
| Part 1 (phr) | ||||
| Pure NR | 100 | - | - | - |
| Pure PE | - | 100 | - | - |
| PU waste | 200 | 200 | - | - |
| SiO2 | 100 | 100 | - | - |
| ATH | 140 | 140 | - | - |
| Steric acid | 1 | - | - | - |
| ZnO | 5 | - | - | - |
| TMQ | 1 | - | - | - |
| MBTs | 1 | - | - | - |
| Sulfur | 2 | - | - | - |
| Part 2 (phr) | ||||
| Pure PE | - | - | 50 | - |
| PEC-ATH-SiO2 | - | - | - | 50 |
| NRC-ATH-SiO2 | - | - | 50 | 50 |
| Samples | Young’s Modulus (MPa) | Tensile Strength (MPa) | Elongation at Break (%) | Hardness (Shore A) |
|---|---|---|---|---|
| NRC-ATH-PuSiO2 | 1.54 ± 0.15 | 5.87 ± 0.28 | 14.00 ± 1.01 | 94.5 ± 1.04 |
| PEC-ATH-PuSiO2 | 3.84 ± 0.09 | 8.02 ± 0.42 | 2.96 ± 0.22 | 97.0 ± 2.07 |
| NRC-ATH-PuSiO2/PEC | 2.59 ± 0.17 | 3.30 ± 0.73 | 2.89 ± 0.68 | 84.0 ± 0.50 |
| NRC/PEC-(ATH-PuSiO2) | 2.96 ± 0.16 | 7.45 ± 0.46 | 5.22 ± 0.56 | 96.0 ± 0.35 |
| Samples | Initial E’ (MPa) | Tg1 (°C) | Tg2 (°C) | Tan δ at 0 °C |
|---|---|---|---|---|
| NRC-ATH-PuSiO2 | 1787 | - | −52 | 0.09 |
| PEC-ATH-PuSiO2 | 2071 | −129 | - | 0.06 |
| NRC-ATH-PuSiO2/PEC | 1514 | −127 | −54 | 0.09 |
| NRC/PEC-(ATH-PuSiO2) | 2024 | −126 | −56 | 0.07 |
| Samples | Decomposition (%) | Temperature (°C) | |
|---|---|---|---|
| First Step | Second Step | t2 | |
| NRC-ATH-PuSiO2 | 50 | 13 | 238 |
| PEC-ATH-PuSiO2 | 63 | 8 | 254 |
| NRC-ATH-PuSiO2/PEC | 76 | 6 | 268 |
| NRC/PEC-(ATH-PuSiO2) | 58 | 10 | 245 |
| Samples | Fire Classification |
|---|---|
| NRC-ATH-PuSiO2 | V-0 |
| PEC-ATH-PuSiO2 | V-2 |
| NRC-ATH-PuSiO2/PEC | V-2 |
| NRC/PEC-(ATH-PuSiO2) | V-0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tilokavichai, V.; Pannarungsee, O.; Luangchuang, P.; Nakaramontri, Y. Effects of Recycled Polyethylene on Natural Rubber Composite Blends Filled with Aluminum Trihydroxide and Polyurethane Waste: Mechanical and Dynamic Mechanical Properties, Flammability. Polymers 2024, 16, 1657. https://doi.org/10.3390/polym16121657
Tilokavichai V, Pannarungsee O, Luangchuang P, Nakaramontri Y. Effects of Recycled Polyethylene on Natural Rubber Composite Blends Filled with Aluminum Trihydroxide and Polyurethane Waste: Mechanical and Dynamic Mechanical Properties, Flammability. Polymers. 2024; 16(12):1657. https://doi.org/10.3390/polym16121657
Chicago/Turabian StyleTilokavichai, Varanya, Onanong Pannarungsee, Piyawadee Luangchuang, and Yeampon Nakaramontri. 2024. "Effects of Recycled Polyethylene on Natural Rubber Composite Blends Filled with Aluminum Trihydroxide and Polyurethane Waste: Mechanical and Dynamic Mechanical Properties, Flammability" Polymers 16, no. 12: 1657. https://doi.org/10.3390/polym16121657
APA StyleTilokavichai, V., Pannarungsee, O., Luangchuang, P., & Nakaramontri, Y. (2024). Effects of Recycled Polyethylene on Natural Rubber Composite Blends Filled with Aluminum Trihydroxide and Polyurethane Waste: Mechanical and Dynamic Mechanical Properties, Flammability. Polymers, 16(12), 1657. https://doi.org/10.3390/polym16121657






