Stability and Composting Behaviour of PLA–Starch Laminates Containing Active Extracts and Cellulose Fibres from Rice Straw
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Obtaining Cellulosic and Active Fractions from RS
2.3. Film Preparation
2.3.1. TPS Monolayers
2.3.2. PLA Monolayers
2.3.3. Bilayer Preparation
2.4. Characterisation of the Films
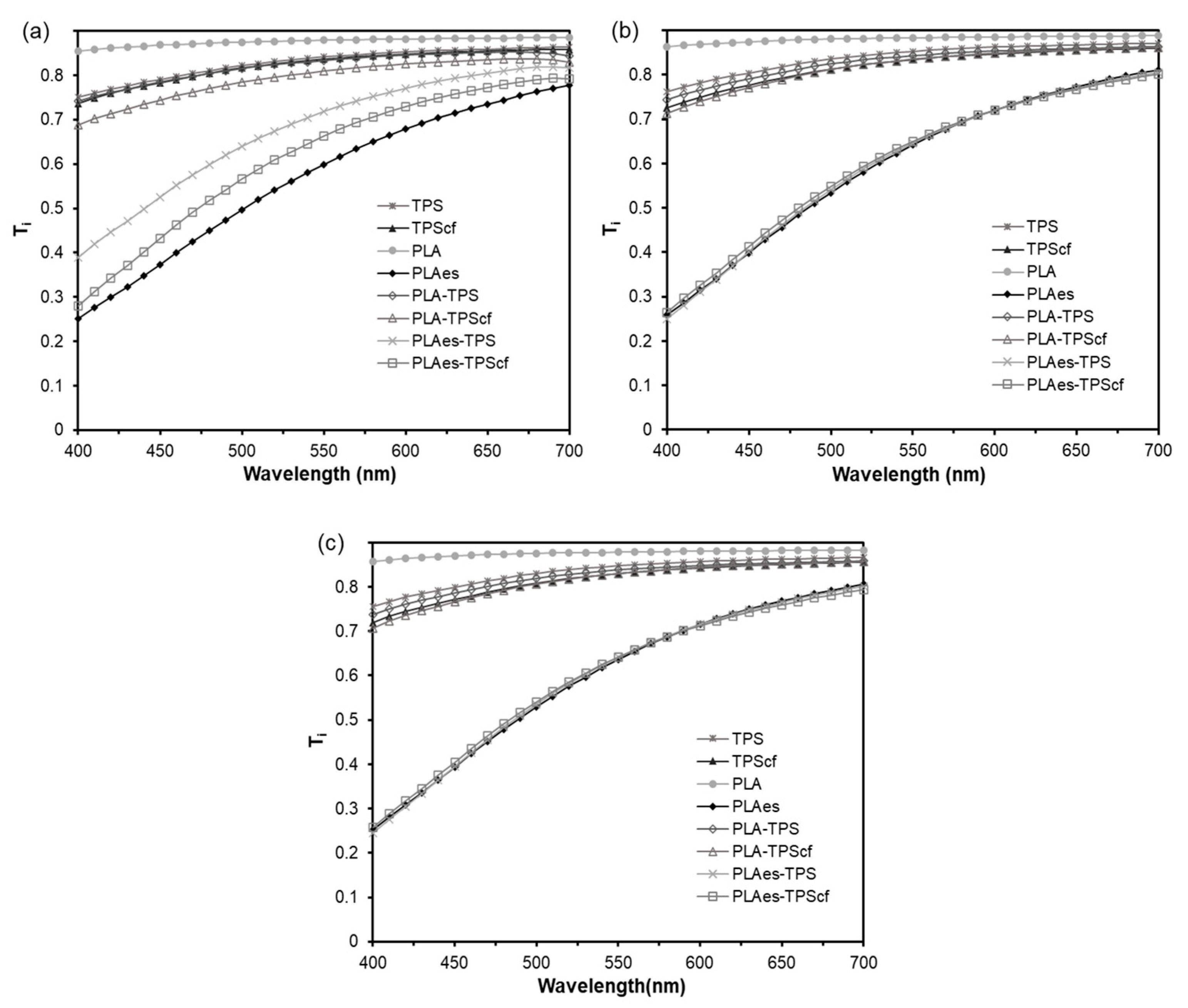
2.4.1. Optical Properties
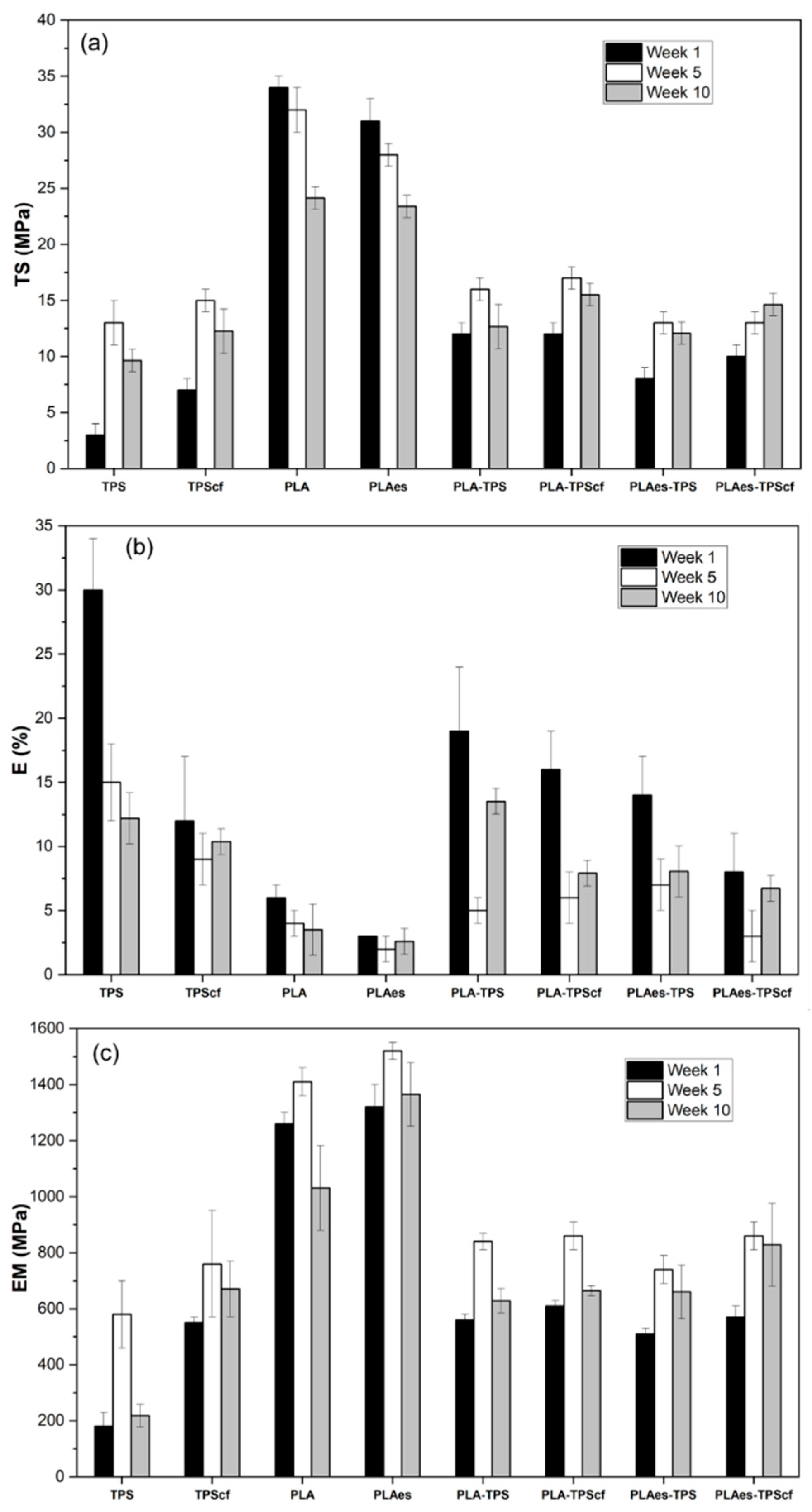
2.4.2. Tensile Properties
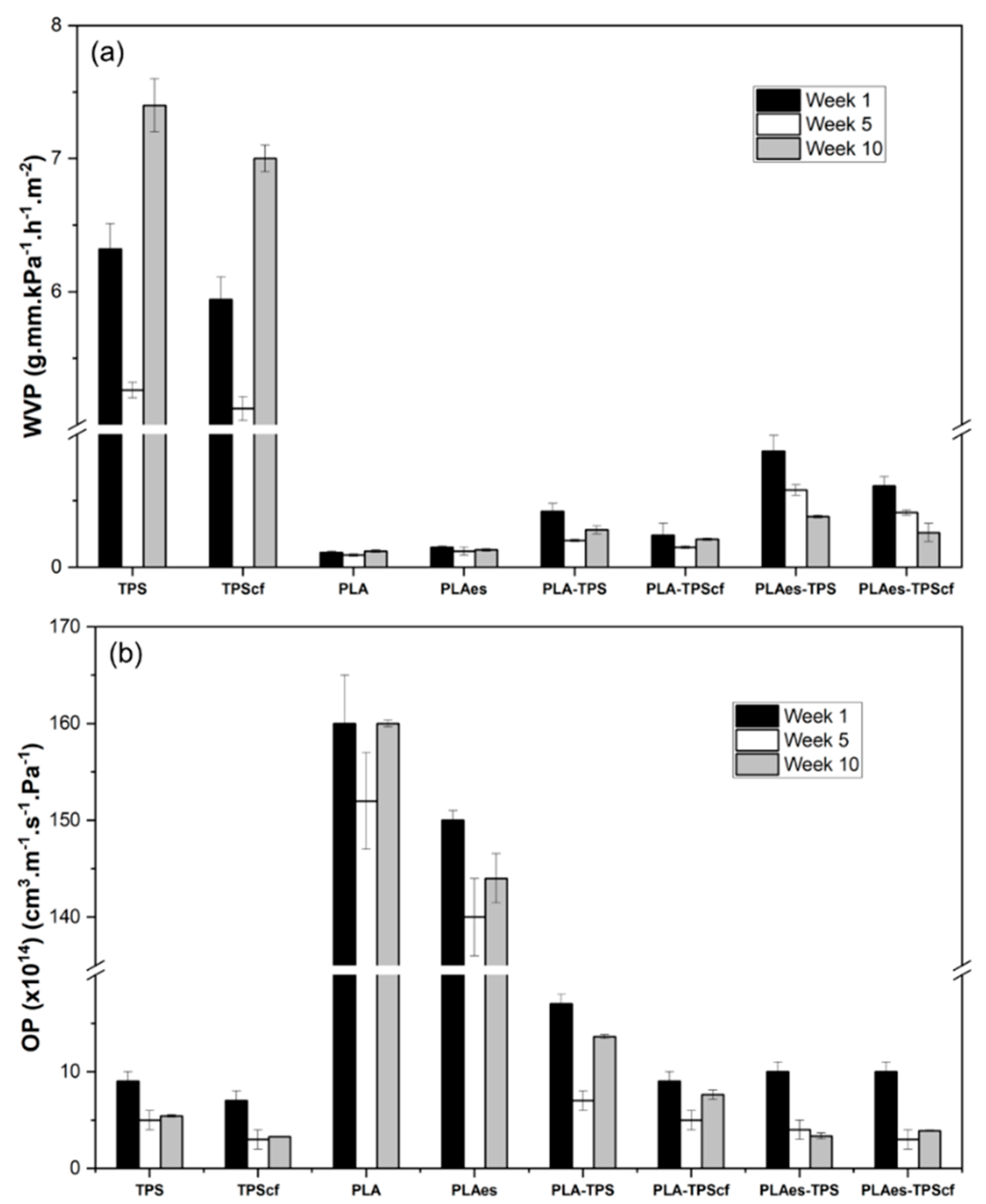
2.4.3. Barrier Properties
2.4.4. Bioactive Properties
2.5. Composting Properties of the Films
2.5.1. Compost Conditioning and Preparation of Synthetic Solid Residue
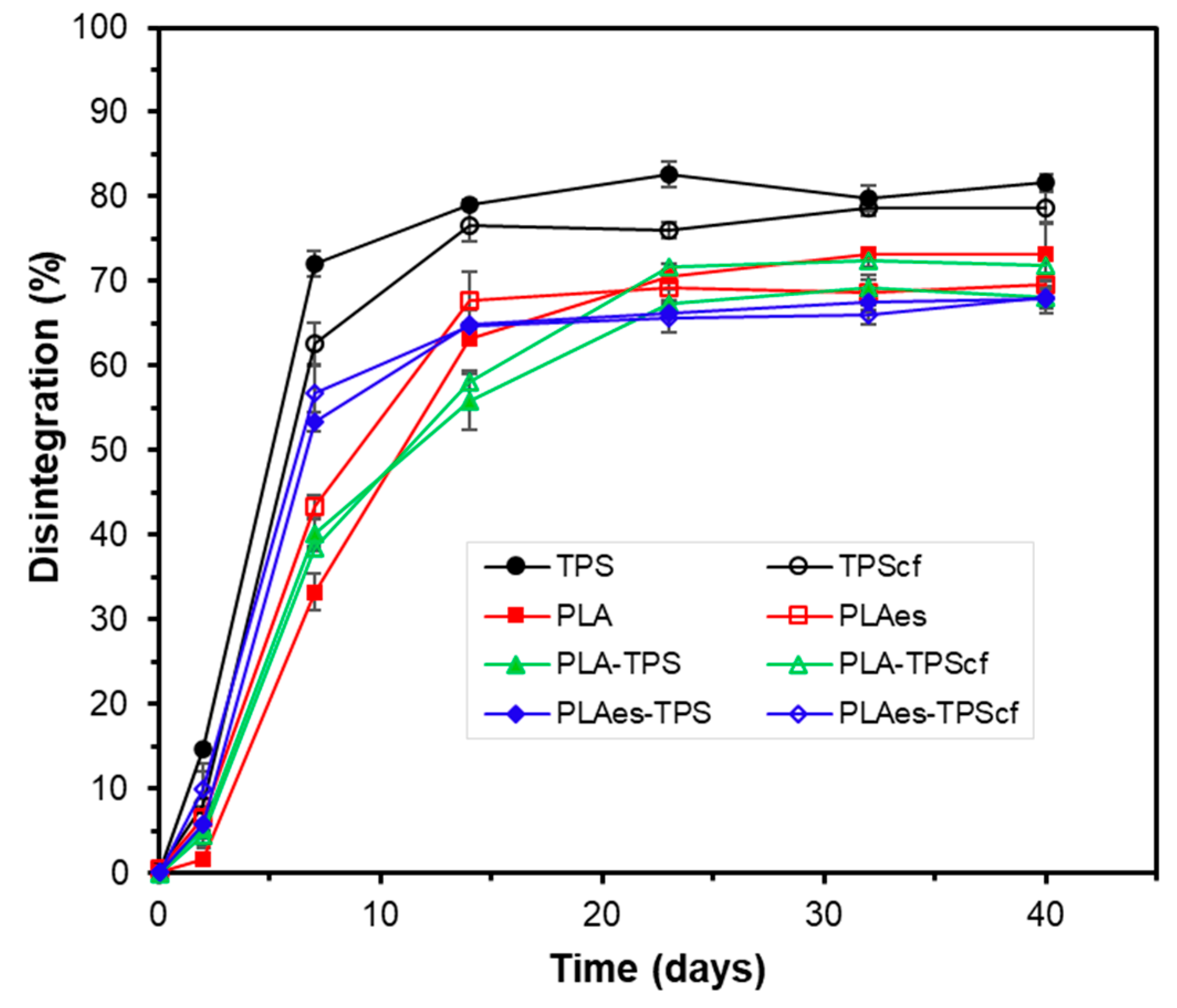
2.5.2. Disintegration Test
2.5.3. Biodegradation Test
2.6. Statistical Analysis
3. Results and Discussion
3.1. Changes in Physical Properties of the Films during Storage
3.1.1. Barrier Properties
3.1.2. Tensile Properties
3.1.3. Optical Properties
3.1.4. Bioactive Properties
3.2. Biodegradability of the Mono and Bilayer Films
3.2.1. Moisture Content, Thicknesses, and Elemental Carbon of the Films
3.2.2. Compost Characteristics
3.2.3. Disintegration Test
3.2.4. Biodegradation Test
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Shlush, E.; Davidovich-Pinhas, M. Bioplastics for Food Packaging. Trends Food Sci. Technol. 2022, 125, 66–80. [Google Scholar] [CrossRef]
- Thakur, S.; Chaudhary, J.; Sharma, B.; Verma, A.; Tamulevicius, S.; Thakur, V.K. Sustainability of Bioplastics: Opportunities and Challenges. Curr. Opin. Green Sustain. Chem. 2018, 13, 68–75. [Google Scholar] [CrossRef]
- Barnes, D.K.A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and Fragmentation of Plastic Debris in Global Environments. Phil. Trans. R. Soc. B 2009, 364, 1985–1998. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Peng, Z.; Zhu, Z.-R.; Fu, W.; Dai, X.; Ni, B.-J. The Atmospheric Microplastics Deposition Contributes to Microplastic Pollution in Urban Waters. Water Res. 2022, 225, 119116. [Google Scholar] [CrossRef] [PubMed]
- Ivar Do Sul, J.A.; Costa, M.F. The Present and Future of Microplastic Pollution in the Marine Environment. Environ. Pollut. 2014, 185, 352–364. [Google Scholar] [CrossRef]
- Andrady, A.L.; Neal, M.A. Applications and Societal Benefits of Plastics. Phil. Trans. R. Soc. B 2009, 364, 1977–1984. [Google Scholar] [CrossRef]
- Peanparkdee, M.; Iwamoto, S. Bioactive Compounds from By-Products of Rice Cultivation and Rice Processing: Extraction and Application in the Food and Pharmaceutical Industries. Trends Food Sci. Technol. 2019, 86, 109–117. [Google Scholar] [CrossRef]
- Singh, L.; Brar, B.S. A Review on Rice Straw Management Strategies. Nat. Environ. Pollut. Technol. 2021, 20, 1485–1493. [Google Scholar] [CrossRef]
- Sadh, P.K.; Chawla, P.; Bhandari, L.; Duhan, J.S. Bio-Enrichment of Functional Properties of Peanut Oil Cakes by Solid State Fermentation Using Aspergillus Oryzae. J. Food Meas. Charact. 2018, 12, 622–633. [Google Scholar] [CrossRef]
- Freitas, P.A.V.; González-Martínez, C.; Chiralt, A. Application of Ultrasound Pre-Treatment for Enhancing Extraction of Bioactive Compounds from Rice Straw. Foods 2020, 9, 1657. [Google Scholar] [CrossRef]
- Dilamian, M.; Noroozi, B. Rice straw agri-waste for water pollutant adsorption: Relevant mesoporous super hydrophobic cellulose aerogel. Carbohydr. Polym. 2021, 251, 117016. [Google Scholar] [CrossRef] [PubMed]
- Freitas, P.A.V.; González-Martínez, C.; Chiralt, A. Applying Ultrasound-Assisted Processing to Obtain Cellulose Fibres from Rice Straw to Be Used as Reinforcing Agents. Innov. Food Sci. Emerg. Technol. 2022, 76, 102932. [Google Scholar] [CrossRef]
- Freitas, P.A.V.; González-Martínez, C.; Chiralt, A. Using Rice Straw Fractions to Develop Reinforced, Active PLA-Starch Bilayers for Meat Preservation. Food Chem. 2023, 405, 134990. [Google Scholar] [CrossRef]
- Freitas, P.A.V.; González-Martínez, C.; Chiralt, A. Antioxidant Starch Composite Films Containing Rice Straw Extract and Cellulose Fibres. Food Chem. 2023, 400, 134073. [Google Scholar] [CrossRef]
- Baghi, F.; Gharsallaoui, A.; Dumas, E.; Ghnimi, S. Advancements in Biodegradable Active Films for Food Packaging: Effects of Nano/Microcapsule Incorporation. Foods 2022, 11, 760. [Google Scholar] [CrossRef]
- Kaiser, K.; Schmid, M.; Schlummer, M. Recycling of Polymer-Based Multilayer Packaging: A Review. Recycling 2017, 3, 1. [Google Scholar] [CrossRef]
- Anukiruthika, T.; Sethupathy, P.; Wilson, A.; Kashampur, K.; Moses, J.A.; Anandharamakrishnan, C. Multilayer Packaging: Advances in Preparation Techniques and Emerging Food Applications. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1156–1186. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, W.; Zhu, W.; McClements, D.J.; Liu, X.; Liu, F. A Review of Multilayer and Composite Films and Coatings for Active Biodegradable Packaging. npj Sci. Food 2022, 6, 18. [Google Scholar] [CrossRef] [PubMed]
- Galdeano, M.C.; Mali, S.; Grossmann, M.V.E.; Yamashita, F.; García, M.A. Effects of Plasticizers on the Properties of Oat Starch Films. Mater. Sci. Eng. C 2009, 29, 532–538. [Google Scholar] [CrossRef]
- Avérous, L.; Fringant, C.; Moro, L. Plasticized Starch–Cellulose Interactions in Polysaccharide Composites. Polymer 2001, 42, 6565–6572. [Google Scholar] [CrossRef]
- Freitas, P.A.V.; Bas Gil, N.J.; González-Martínez, C.; Chiralt, A. Antioxidant Poly (Lactic Acid) Films with Rice Straw Extract for Food Packaging Applications. Food Packag. Shelf Life 2022, 34, 101003. [Google Scholar] [CrossRef]
- Muller, J.; González-Martínez, C.; Chiralt, A. Poly(Lactic) Acid (PLA) and Starch Bilayer Films, Containing Cinnamaldehyde, Obtained by Compression Moulding. Eur. Polym. J. 2017, 95, 56–70. [Google Scholar] [CrossRef]
- Freitas, P.A. Valorisation of Rice Straw by Obtaining Active Compounds and Cellulosic Materials for the Development of Biodegradable Food Packaging Systems. Doctoral Thesis, Universitat Politècnica de València, Valencia, Spain, 2022. [Google Scholar]
- Freitas, P.A.V.; Arias, C.I.L.F.; Torres-Giner, S.; González-Martínez, C.; Chiralt, A. Valorization of Rice Straw into Cellulose Microfibers for the Reinforcement of Thermoplastic Corn Starch Films. Appl. Sci. 2021, 11, 8433. [Google Scholar] [CrossRef]
- ASTM D882–12; Standard Test Method for Tensile Properties of Thin Plastic Sheeting. American Society for Testing and Materials (ASTM): West Conshohocken, PA, USA, 2012.
- ASTM E96/E96M; Standard Test Methods for Water Vapor Transmission of Materials. American Society for Testing and Materials (ASTM): West Conshohocken, PA, USA, 2005.
- ASTM D3985-025; Standard Test Method for Oxygen Gas Transmission Rate through Plastic Film and Sheeting Using s Coulometric Sensor. ASTM: West Conshohocken, PA, USA, 2010; pp. 1–7.
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- ISO 20200; Plastics—Determination of the Degree of Disintegration of Plastic Materials under Composting Conditions in a Laboratory-Scale TES. European Committee fot Standarization: Bruxelles, Belgium, 2023.
- UNE-EN ISO 14885-1; Determinación de la Biodegradabilidad Aeróbica Final de Materiales Plásticos en Condiciones de Compostaje Controladas. Meétodo Según el Análisis de Dióxido de Carbono Generado. European Committee fot Standarization: Bruxelles, Belgium, 2013.
- Hosseini, S.F.; Javidi, Z.; Rezaei, M. Efficient Gas Barrier Properties of Multi-Layer Films Based on Poly(Lactic Acid) and Fish Gelatin. Int. J. Biol. Macromol. 2016, 92, 1205–1214. [Google Scholar] [CrossRef] [PubMed]
- Siracusa, V. Food Packaging Permeability Behaviour: A Report. Int. J. Polym. Sci. 2012, 2012, 302029. [Google Scholar] [CrossRef]
- Wang, S.; Li, C.; Copeland, L.; Niu, Q.; Wang, S. Starch Retrogradation: A Comprehensive Review. Comp. Rev. Food Sci. Food Safe 2015, 14, 568–585. [Google Scholar] [CrossRef]
- Ke, T.; Sun, X. Melting Behavior and Crystallization Kinetics of Starch and Poly(Lactic Acid) Composites. J. Appl. Polym. Sci. 2003, 89, 1203–1210. [Google Scholar] [CrossRef]
- Acioli-Moura, R.; Sun, X.S. Thermal Degradation and Physical Aging of Poly(Lactic Acid) and Its Blends with Starch. Polym. Eng. Sci. 2008, 48, 829–836. [Google Scholar] [CrossRef]
- Vilarinho, F.; Stanzione, M.; Buonocore, G.G.; Barbosa-Pereira, L.; Sendón, R.; Vaz, M.F.; Sanches Silva, A. Green Tea Extract and Nanocellulose Embedded into Polylactic Acid Film: Properties and Efficiency on Retarding the Lipid Oxidation of a Model Fatty Food. Food Packag. Shelf Life 2021, 27, 100609. [Google Scholar] [CrossRef]
- Fourati, Y.; Magnin, A.; Putaux, J.-L.; Boufi, S. One-Step Processing of Plasticized Starch/Cellulose Nanofibrils Nanocomposites via Twin-Screw Extrusion of Starch and Cellulose Fibers. Carbohydr. Polym. 2020, 229, 115554. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.-T.; Auras, R.; Rubino, M. Processing Technologies for Poly(Lactic Acid). Prog. Polym. Sci. 2008, 33, 820–852. [Google Scholar] [CrossRef]
- Benito-González, I.; López-Rubio, A.; Martínez-Sanz, M. High-Performance Starch Biocomposites with Celullose from Waste Biomass: Film Properties and Retrogradation Behaviour. Carbohydr. Polym. 2019, 216, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Bi, X.; Huang, W.; Wu, J.; Hu, X.; Liao, X. Changes of Quality of High Hydrostatic Pressure Processed Cloudy and Clear Strawberry Juices during Storage. Innov. Food Sci. Emerg. Technol. 2012, 16, 181–190. [Google Scholar] [CrossRef]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Anthocyanin Stability and Antioxidant Activity of Spray-Dried Açai (Euterpe Oleracea Mart.) Juice Produced with Different Carrier Agents. Food Res. Int. 2010, 43, 907–914. [Google Scholar] [CrossRef]
- Balaguer, M.P.; Villanova, J.; Cesar, G.; Gavara, R.; Hernandez-Munoz, P. Compostable Properties of Antimicrobial Bioplastics Based on Cinnamaldehyde Cross-Linked Gliadins. Chem. Eng. J. 2015, 262, 447–455. [Google Scholar] [CrossRef]
- Silva, A.P.M.; Oliveira, A.V.; Pontes, S.M.A.; Pereira, A.L.S.; Souza Filho, M.D.S.M.; Rosa, M.F.; Azeredo, H.M.C. Mango Kernel Starch Films as Affected by Starch Nanocrystals and Cellulose Nanocrystals. Carbohydr. Polym. 2019, 211, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Pavon, C.; Aldas, M.; López-Martínez, J.; Hernández-Fernández, J.; Arrieta, M.P. Films Based on Thermoplastic Starch Blended with Pine Resin Derivatives for Food Packaging. Foods 2021, 10, 1171. [Google Scholar] [CrossRef]
- Kale, G.; Auras, R.; Singh, S.P. Comparison of the Degradability of Poly(Lactide) Packages in Composting and Ambient Exposure Conditions. Packag. Technol. Sci. 2007, 20, 49–70. [Google Scholar] [CrossRef]
- Chinaglia, S.; Tosin, M.; Degli-Innocenti, F. Biodegradation Rate of Biodegradable Plastics at Molecular Level. Polym. Degrad. Stab. 2018, 147, 237–244. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Fortunati, E.; Dominici, F.; Rayón, E.; López, J.; Kenny, J.M. PLA-PHB/Cellulose Based Films: Mechanical, Barrier and Disintegration Properties. Polym. Degrad. Stab. 2014, 107, 139–149. [Google Scholar] [CrossRef]
- Donlan, R.M. Biofilms: Microbial Life on Surfaces. Emerg. Infect. Dis. 2002, 8, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Sheel, A.; Pant, D. Microbial Depolymerization. In Waste Bioremediation. Energy, Environment, and Sustainability; Springer: Singapore, 2018; pp. 61–103. [Google Scholar]
- Castro-Aguirre, E.; Auras, R.; Selke, S.; Rubino, M.; Marsh, T. Enhancing the biodegradation rate of poly (lactic acid) films and PLA bio-nanocomposites in simulated composting through bioaugmentation. Polym. Degrad. Stab. 2018, 154, 46–54. [Google Scholar] [CrossRef]
- Shen, J.; Bartha, R. Priming Effect of Glucose Polymers in Soil-Based Biodegradation Tests. Soil Biol. Biochem. 1997, 29, 1195–1198. [Google Scholar] [CrossRef]



| Film | L* | Cab* | hab* | ||||||
|---|---|---|---|---|---|---|---|---|---|
| S1 | S5 | S10 | S1 | S5 | S10 | S1 | S5 | S10 | |
| PLA | 90.7 ± 0.2 a,1 | 90.8 ± 0.1 a,1 | 90.1 ± 0.3 a,1 | 2.5 ± 0.13 e,1 | 2.42 ± 0.12 e,1 | 2.33 ± 0.17 e,1 | 99.6 ± 0.8 a,1 | 100.1 ± 0.21 a,1 | 100.7 ± 0.5 a,1 |
| PLAes | 67.6 ± 0.7 f,1 | 68.3 ± 1.7 f,1 | 66.2 ± 1.2 f,1 | 34.57 ± 0.7 a,1 | 32.85 ± 3.0 a,1 | 33.72 ± 1.20 a,1 | 77.1 ± 0.3 f,1 | 79.0 ± 1.0 f,1 | 78.5 ± 0.5 f,1 |
| TPS | 88.5 ± 0.1 b,1 | 89.0 ± 0.1 b,1 | 87.8 ± 0.9 b,1 | 7.57 ± 0.10 d,1 | 7.3 ± 0.88 d,1 | 7.12 ± 0.33 d,1 | 92.5 ± 0.1 b,1 | 94.7 ± 0.7 b,1 | 95.4 ± 0.8 b,1 |
| TPScf | 88.1 ± 0.2 b,1 | 87.8 ± 0.3 b,1 | 88.3 ± 0.7 b,1 | 8.25 ± 0.35 cd,1 | 8.7 ± 0.48 cd,1 | 8.2 ± 0.28 cd,1 | 92.6 ± 0.2 b,1 | 92.4 ± 0.4 b,1 | 92.1 ± 0.6 b,1 |
| PLA-TPS | 87.3 ± 0.3 b,1 | 88.4 ± 0.5 b,1 | 87.9 ± 0.7 b,1 | 7.58 ± 0.11 d,1 | 7.3 ± 1.22 d,1 | 7.1 ± 1.31 d,1 | 92.2 ± 0.3 b,1 | 95.1 ± 1.2 b,1 | 93.1 ± 2.1 b,1 |
| PLA-TPScf | 86.0 ± 0.5 c,1 | 87.2 ± 0.3 c,1 | 87.8 ± 0.4 c,1 | 9.50 ± 0.31 c,1 | 9.79 ± 0.36 c,1 | 9.34 ± 0.22 c,1 | 90.9 ± 0.4 c,1 | 92.7 ± 0.5 c,1 | 91.4 ± 0.5 c,1 |
| PLAes-TPS | 76.9 ± 1.2 d,1 | 74.5 ± 3.0 d,1 | 75.4 ± 1.6 d,1 | 27.92 ± 2.06 b,1 | 25.83 ± 2.98 b,1 | 25.14 ± 1.28 b,1 | 83.4 ± 0.7 d,1 | 80.4 ± 1.6 d,1 | 80.7 ± 0.7 d,1 |
| PLAes-TPScf | 72.2 ± 0.9 e,1 | 72.0 ± 2.0 e,1 | 73.1 ± 1.1 e,1 | 34.41 ± 1.22 a,1 | 34.32 ± 2.68 a,1 | 34.24 ± 2.47 a,1 | 81.0 ± 0.6 e,1 | 81.0 ± 1.4 e,1 | 80.5 ± 0.2 e,1 |
| Sample | Moisture Content (%) | Thickness (µm) | C (%) |
|---|---|---|---|
| TPS | 8.3 ± 0.2 a | 0.171 ± 0.026 bc | 40.3 ± 0.1 |
| TPScf | 8.0 ± 0.6 a | 0.185 ± 0.025 b | 40.2 ± 0.2 |
| PLA | 0.7 ± 0.1 c | 0.146 ± 0.008 c | 48.7 ± 0.1 |
| PLAes | 0.9 ± 0.1 c | 0.144 ± 0.007 c | 49.0 ± 0.5 |
| PLA-TPS | 6.9 ± 0.3 b | 0.278 ± 0.033 a | 45.0 ± 0.4 |
| PLA-TPScf | 6.7 ± 0.4 b | 0.273 ± 0.012 a | 43.8 ± 0.1 |
| PLAes-TPS | 6.7 ± 0.3 b | 0.269 ± 0.013 a | 44.7 ± 0.1 |
| PLAes-TPScf | 6.6 ± 0.1 b | 0.262 ± 0.007 a | 44.8 ± 0.2 |
| MCC | - | - | 42.2 ± 0.1 |
| Sample | VS (g·100 g−1 DS) | R (%) | Desintegration D73 (%) | |
|---|---|---|---|---|
| Pre-Composting | Post-Composting | |||
| SSR | 90.6 ± 1.8 | 88.3 ± 2.1 | 43.2 | - |
| TPS | - | 85.1 ± 1.2 | 48.1 | 84 ± 1 |
| TPScf | - | 83.1 ± 3.4 | 45.1 | 83 ± 1 |
| PLA | - | 82.4 ± 1.5 | 51.3 | 75 ± 1 |
| PLAes | - | 87.3 ± 2.7 | 49.2 | 80 ± 2 |
| PLA-TPS | - | 85.4 ± 1.5 | 48.2 | 75 ± 2 |
| PLA-TPScf | - | 83.4 ± 2.7 | 53.2 | 74 ± 3 |
| PLAes-TPS | - | 80.4 ± 2.4 | 47.6 | 70 ± 2 |
| PLAes-TPScf | - | 83.4 ± 1.2 | 49.2 | 71 ± 3 |
| Sample | n | k (Days) | Bmax (%) | R2 |
|---|---|---|---|---|
| MCC | 1.2 | 23.6 | 185 | 0.995 |
| TPS | 1.0 | 20.1 | 198 | 0.994 |
| TPScf | 0.9 | 22.3 | 196 | 0.996 |
| PLA | 5.6 | 58.5 | 155 | 0.992 |
| PLAes | 4.2 | 84.0 | 125 | 0.992 |
| PLA-TPS | 0.4 | 35.2 | 163 | 0.998 |
| PLA-TPScf | 0.9 | 38.4 | 172 | 0.985 |
| PLAes-TPS | 0.7 | 45.8 | 147 | 0.996 |
| PLAes-TPScf | 1.0 | 39.4 | 129 | 0.990 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freitas, P.A.V.; González-Martínez, C.; Chiralt, A. Stability and Composting Behaviour of PLA–Starch Laminates Containing Active Extracts and Cellulose Fibres from Rice Straw. Polymers 2024, 16, 1474. https://doi.org/10.3390/polym16111474
Freitas PAV, González-Martínez C, Chiralt A. Stability and Composting Behaviour of PLA–Starch Laminates Containing Active Extracts and Cellulose Fibres from Rice Straw. Polymers. 2024; 16(11):1474. https://doi.org/10.3390/polym16111474
Chicago/Turabian StyleFreitas, Pedro A. V., Chelo González-Martínez, and Amparo Chiralt. 2024. "Stability and Composting Behaviour of PLA–Starch Laminates Containing Active Extracts and Cellulose Fibres from Rice Straw" Polymers 16, no. 11: 1474. https://doi.org/10.3390/polym16111474
APA StyleFreitas, P. A. V., González-Martínez, C., & Chiralt, A. (2024). Stability and Composting Behaviour of PLA–Starch Laminates Containing Active Extracts and Cellulose Fibres from Rice Straw. Polymers, 16(11), 1474. https://doi.org/10.3390/polym16111474








