Recent Applications of Chitosan and Its Derivatives in Antibacterial, Anticancer, Wound Healing, and Tissue Engineering Fields
Abstract
1. Introduction
1.1. Why Chitosan–Highly Versatile and Widely Acceptable
1.2. History of Chitosan
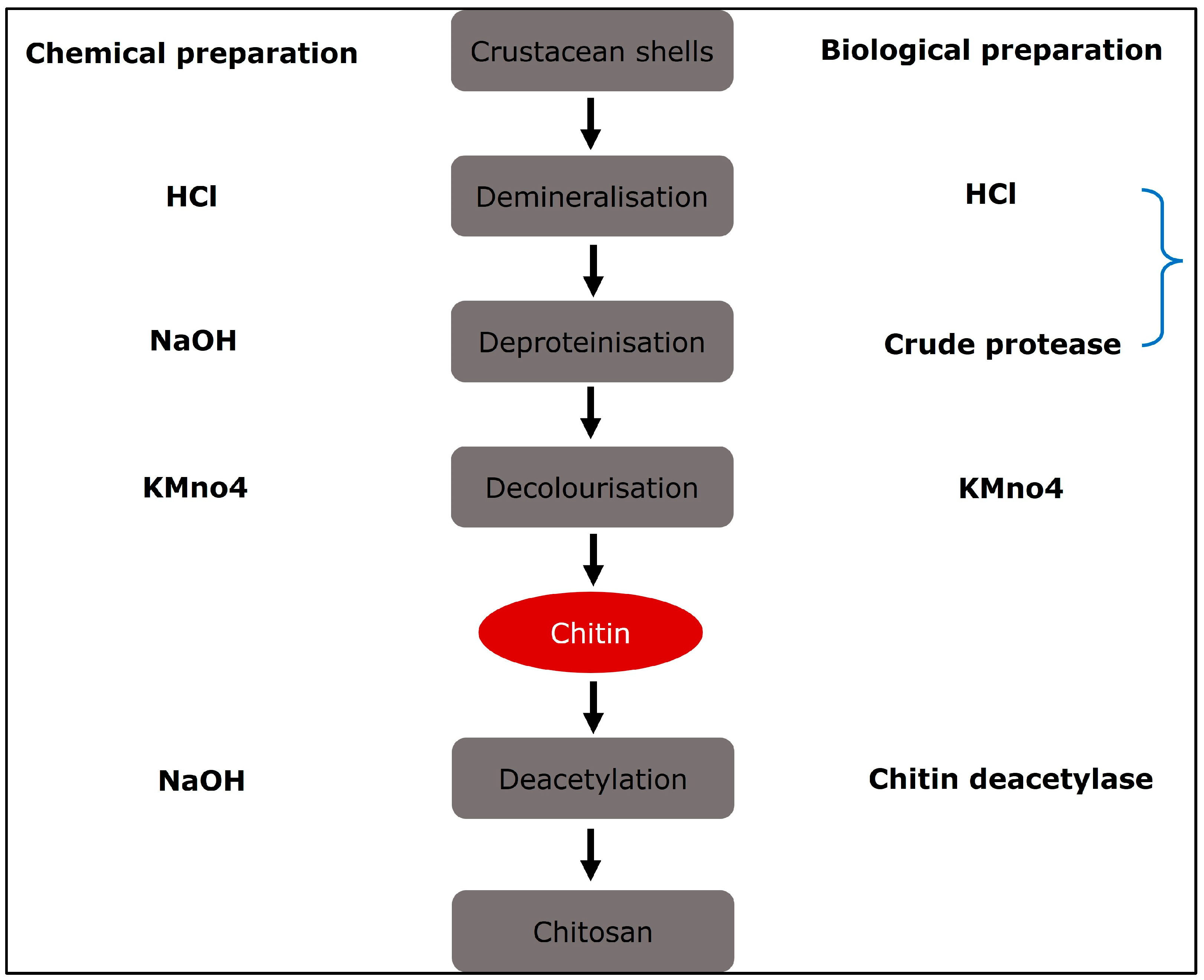
2. Production of Chitosan
Chitosan Derivatives
3. Advantages and Limitations
4. Bioactivities of Chitosan
4.1. Anti-Inflammatory and Antimicrobial Activity of Chitosan
4.2. Latest Developments in Chitosan’s Antimicrobial Activity
| S. No. | Type of Formulation | Size of NP/Fibres | Microbes | Special Comments | References |
|---|---|---|---|---|---|
| 1. | Bletilla striata (BSP) film | 0.097 ± 0.004 to 0.136 ± 0.003 mm thickness | Escherichia coli staphylococcus aureus and Pseudomonas aeruginosa | In this study, the author reported that the prepared BSP/CS films may be used to create biomaterials for new wound dressings. | [81] |
| 2. | Eucalyptus-oil-loaded chitosan nanofibres | 48.26 nm | Staphylococcus aureus | The developed nano-chitosan/Eucalyptus oil/cellulose acetate nanofibre has excellent antimicrobial properties and shows promise as a wound healing dressing. | [82] |
| 3. | CSNPs | 141.20 nm | S. aureus and P. aeruginosa | The dose-dependent antibacterial activity was observed. | [83] |
| 4. | Lecithin-coated CSNPs | 235 ± 20 nm | S. aureus | A 2-fold decrease in the survival rate of S. aureus was observed. | [84] |
| 5. | CS-AgNPs | 10–30 nm | Methicillin-resistant streptococcus aureus (MRSA) | MRSA was not detected in all treated groups. | [85] |
| 6. | CSNPs | 408.30 ± 53.17 nm | S. aureus | More than 90% inhibition rate was observed in CSNP-treated groups. | [86] |
| 7. | AgNPs | - | S. aureus, P. aeruginosa, and E. coli | The prepared sponges loaded with AgNPs show excellent antibacterial activity against S. aureus, P. aeruginosa, and E. coli. | [87] |
| 8. | CS-AgNPs | 10–50 nm | MRSA and P. aeruginosa | The CS-AgNPs show good antibacterial activity against MRSA and P. aeruginosa. | [88] |
| 9. | CS-AgNPs | 22.80 nm | E. coli | The CS film loaded with CS-AgNPs showed higher inhibition against E. coli compared to CS-AgNP solution. | [89] |
| 10. | CS fibres | - | Escherichia coli, Staphylococcus aureus, and Candida albicans | The prepared fibres showed excellent antibacterial activity. | [90] |
| 11. | CS nanofibres | 140–170 | E. coli and S. aureus | The CS nanofibres loaded with cinnamon extract enhance the antibacterial activity. | [91] |
| 12. | CSNPs | 208.40 ± 15.70 nm | E. coli and S. aureus | The CSNPs show dose-dependent antibacterial activity and have greater effect against S. aureus than E. coli. | [92] |
| 13. | CSNPs | 51.67 ± 12.55 nm | S. aureus, P. aeruginosa, E. coli, B. subtilis, and C. albicans | The CSNP loaded with SSD show greater effect against gram +ve bacteria than gram −ve bacteria. | [93] |
| 14. | AgNPs | 5–10 nm | Staphylococcus aureus and Pseudomonas aeruginosa | The 3D scaffold loaded with AgNPs showed excellent antibacterial activity. | [94] |
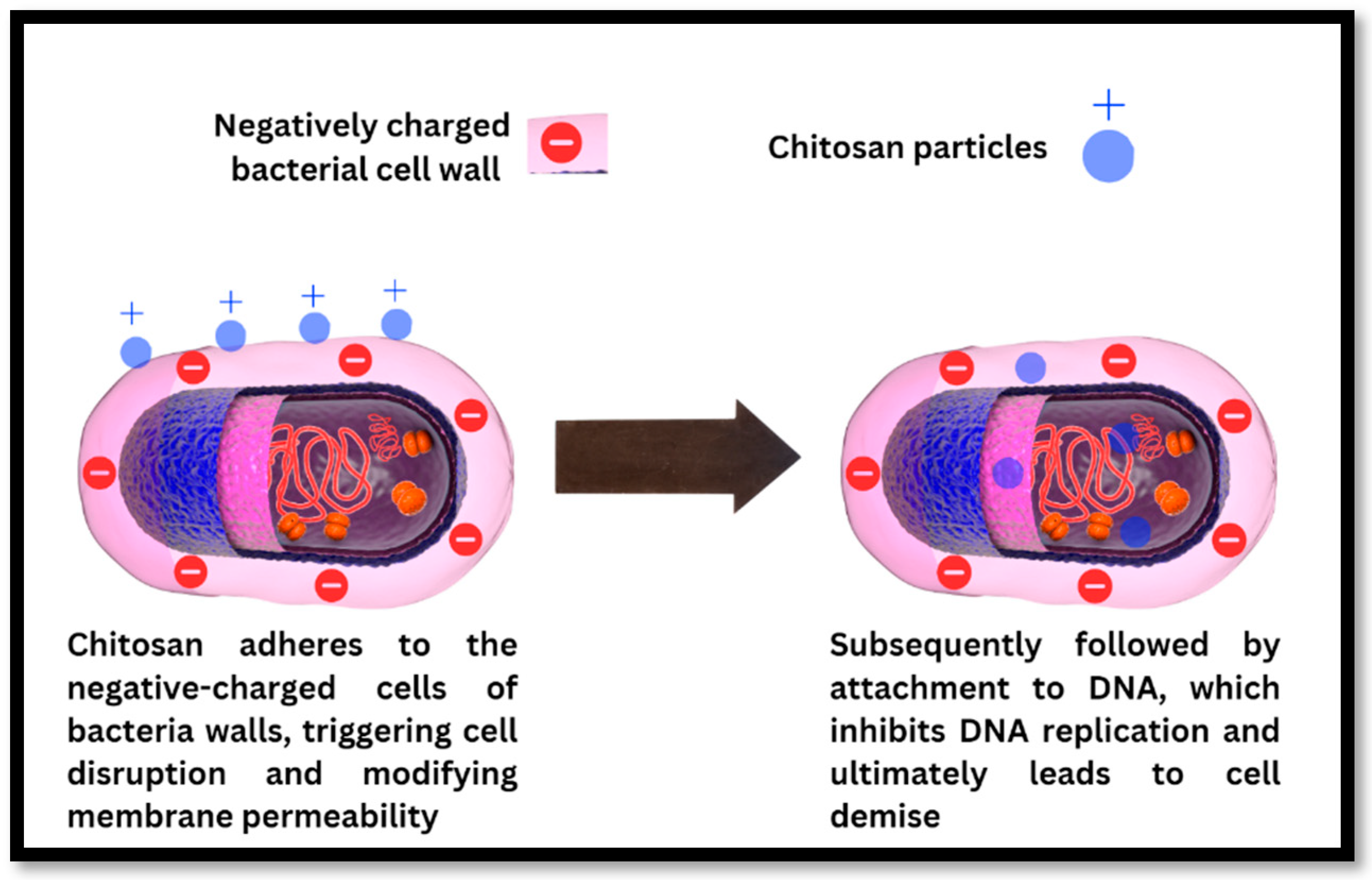
Antibacterial Mechanism of Action of Chitosan
4.3. Wound Healing Activity of Chitosan
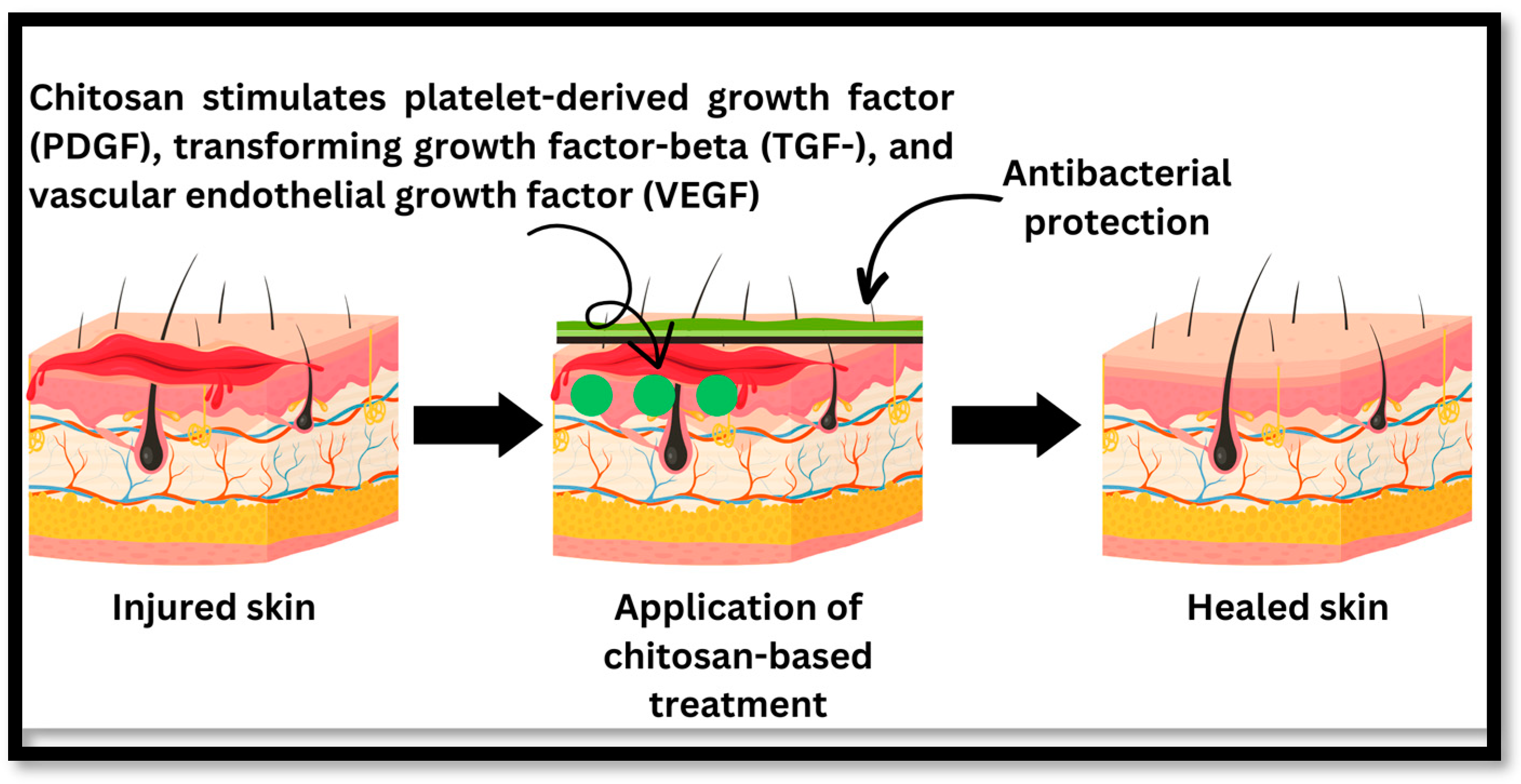
Mechanism of Wound Healing of Chitosan
| Type of Formulation | Active Compound | Size | Type of Wounds | Zeta Potential | Special Comments | References |
|---|---|---|---|---|---|---|
| Quercetin alginate/chitosan gel | Quercetin | - | Excision wound | - | In comparison to free quercetin, the developed gel was topically efficacious and showed synergistic wound healing potential in Wistar albino rats. | [112] |
| Chitosan nanofibres | Bromelain and silver nanoparticle | 145 ± 73 nm | Second-degree burn | - | In this study, the authors revealed that the prepared bromelain-and-silver-nanoparticle-based CS nanofibres are a promising solution for wound healing. | [113] |
| CSNPs | Dunaliella salina | 425.19 ± 4.21 to 496.89 ± 7.62 nm | Full-thickness excision lesions | 22.51 ± 0.50 to 29.21 ± 0.33 mV | This is the first study to show that D. salina-loaded nanoparticles effectively cure wounds. | [5] |
| Chitosan nanofibres | Cissus quadrangularis (CQ) | 77.9–97.4 nm | Cell line study | - | Bilayer sponges containing natural CQ extract exhibited encouraging outcomes as a prospective biomaterial for wound healing purposes. | [114] |
| CSNPs | Epidermal growth factor (EGF) | 63.5–127 nm | Excision wounds | +35 to +40 mV | The developed chitosan nanoparticles are a viable medium for safe delivery of EGF for wound healing applications. | [115] |
| Chitosan hydrogels | Melanin nanoparticles | 216 ± 30 nm | - | +30 ± 14 mV | This work proposes a unique three-dimensional therapy based on natural macromolecules that accelerates wound healing through dual routes while minimising the wound stress response, which is important for the development of therapeutic phototherapy techniques. | [6] |
| CSNPs | Gallic acid | 252.90 ± 3.09 nm | Excision wound | +33.50 ± 0.30 mV | The hexosamine and collagen content were highest in the CSNP-treated group. | [116] |
| Hydrogel | Lupeol-loaded chitosan–Ag+ nanoparticle | 291.9 ± 23.1 to 508.1 ± 26.9 nm | Infected full-thickness wounds | - | The developed hydrogel exhibits significant promise as a versatile therapeutic platform with the capacity to expedite wound healing and proficiently combat bacterial infections in clinical environments. | [117] |
| CSNPs | Curcumin | 359 ± 65 nm | Full-thickness wound | −10.70 ± 0.10 mV | The curcumin-loaded-CSNP-treated groups show the highest collagen content. | [118] |
| Patches | Doxycycline | 50–100 nm | Excision wound | – 24.4 mV | The authors reported that the chitosan-based skin patch incorporating doxycycline holds promise as a viable dressing for the management and enhancement of skin wound healing. | [119] |
| AgNPs | Ag | 190–200 nm | Full-thickness wound | - | CS-AgNPs generated hydroxyproline content of 27.53 0.47 mg/g, 1.6 times greater than the control group and nearly identical to the level seen in the original tissue. | [120] |
| CS-AgNPs | Ag | 200 nm | Excisional wound | - | The result from this study reveals that the prepared CS-AgNPs enhance the antibacterial and wound healing activity. | [121] |
| CSNPs | Melatonin | 160–165 nm | Diabetic full-thickness wound | +25 Mv | No significant difference in collagen content was observed. | [122] |
| CSNPs | Curcumin | 257–260 nm | Full-thickness wound | +30 ± 14 mV | The curcumin-loaded CSNPs hasten the wound healing due to synergistic action shown by chitosan and curcumin. | [123] |
| CSNPs | Insulin | 294–300 nm | Full-thickness wound | +17.89 ± 0.74 mV | The prepared scaffold helps in wound healing; there was a 45% reduction in wound size. | [124] |
| CSNP-loaded nanofibres | Curcumin | 32.17 ± 0.39 nm | Excisional wound | - | The prepared electrospun nanofibres show superior antibacterial and antioxidant properties. The nanofibres loaded with curcumin nanoparticles also help in wound healing. | [125] |
4.4. The Recent Updates on the Wound Healing Activity of Chitosan
4.5. Chitosan in Tissue Engineering
4.6. The Recent Updates on Chitosan in Tissue Engineering
4.7. Chitosan in Delivery Systems for Anticancer Drugs
4.8. Recent Updates on Chitosan in Cancer Research
5. Research Gap and Future Direction
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iber, B.T.; Kasan, N.A.; Torsabo, D.; Omuwa, J.W. A Review of Various Sources of Chitin and Chitosan in Nature. J. Renew. Mater. 2022, 10, 1097. [Google Scholar] [CrossRef]
- Kumar, M.; Mahmood, S.; Chopra, S.; Bhatia, A. Biopolymer Based Nanoparticles and Their Therapeutic Potential in Wound Healing—A Review. Int. J. Biol. Macromol. 2024, 267, 131335. [Google Scholar] [CrossRef] [PubMed]
- Morin-Crini, N.; Lichtfouse, E.; Torri, G.; Crini, G. Applications of Chitosan in Food, Pharmaceuticals, Medicine, Cosmetics, Agriculture, Textiles, Pulp and Paper, Biotechnology, and Environmental Chemistry. Environ. Chem. Lett. 2019, 17, 1667–1692. [Google Scholar] [CrossRef]
- Ahmad, S.I.; Ahmad, R.; Khan, M.S.; Kant, R.; Shahid, S.; Gautam, L.; Hasan, G.M.; Hassan, M.I. Chitin and Its Derivatives: Structural Properties and Biomedical Applications. Int. J. Biol. Macromol. 2020, 164, 526–539. [Google Scholar] [CrossRef] [PubMed]
- El-Baz, F.K.; Salama, A.; Ali, S.I.; El-Hashemy, H.A. Dunaliella Salina Chitosan Nanoparticles as a Promising Wound Healing Vehicles: In-Vitro and in-Vivo Study. OpenNano 2023, 12, 100165. [Google Scholar] [CrossRef]
- Kong, X.; Chen, H.; Li, F.; Zhang, F.; Jiang, Y.; Song, J.; Sun, Y.; Zhao, B.; Shi, J. Three-Dimension Chitosan Hydrogel Loading Melanin Composite Nanoparticles for Wound Healing by Anti-Bacteria, Immune Activation and Macrophage Autophagy Promotion. Int. J. Biol. Macromol. 2023, 237, 124176. [Google Scholar] [CrossRef]
- Xu, Y.X.; Kim, K.M.; Hanna, M.A.; Nag, D. Chitosan–Starch Composite Film: Preparation and Characterization. Ind. Crops Prod. 2005, 21, 185–192. [Google Scholar] [CrossRef]
- Denkbaş, E.B.; Kilicay, E.; Birlikseven, C.; Öztürk, E. Magnetic Chitosan Microspheres: Preparation and Characterization. React. Funct. Polym. 2002, 50, 225–232. [Google Scholar] [CrossRef]
- Machałowski, T.; Rusak, A.; Wiatrak, B.; Haczkiewicz-Leśniak, K.; Popiel, A.; Jaroszewicz, J.; Żak, A.; Podhorska-Okołów, M.; Jesionowski, T. Naturally Formed Chitinous Skeleton Isolated from the Marine Demosponge Aplysina Fistularis as a 3D Scaffold for Tissue Engineering. Materials 2021, 14, 2992. [Google Scholar] [CrossRef] [PubMed]
- Garg, Y.; Kumar, M.; Sharma, G.; Katare, O.P.; Chopra, S.; Bhatia, A. Systematic Designing and Optimization of Polymeric Nanoparticles Using Central Composite Design: A Novel Approach for Nose-to-Brain Delivery of Donepezil Hydrochloride. J. Clust. Sci. 2023, 35, 1007–1019. [Google Scholar] [CrossRef]
- Kumar, M.; Hilles, A.R.; Almurisi, S.H.A.; Bhatia, A.; Mahmood, S. Micro and Nano-Carriers-Based Pulmonary Drug Delivery System: Their Current Updates, Challenges, and Limitations—A Review. JCIS Open 2023, 12, 100095. [Google Scholar] [CrossRef]
- Kumar, M.; Kumar, D.; Garg, Y.; Mahmood, S.; Chopra, S.; Bhatia, A. Marine-Derived Polysaccharides and Their Therapeutic Potential in Wound Healing Application-A Review. Int. J. Biol. Macromol. 2023, 253, 127331. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, K.; Yui, T.; Okuyama, K. Three D Structures of Chitosan. Int. J. Biol. Macromol. 2004, 34, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.N.V.R.; Muzzarelli, R.; Muzzarelli, C.; Sashiwa, H.; Domb, A.J. Chitosan Chemistry and Pharmaceutical Perspectives. Chem. Rev. 2004, 104, 6017–6084. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Meng, Q.; Li, Q.; Liu, J.; Zhou, M.; Jin, Z.; Zhao, K. Chitosan Derivatives and Their Application in Biomedicine. Int. J. Mol. Sci. 2020, 21, 487. [Google Scholar] [CrossRef] [PubMed]
- Mohan, K.; Ganesan, A.R.; Ezhilarasi, P.N.; Kondamareddy, K.K.; Rajan, D.K.; Sathishkumar, P.; Rajarajeswaran, J.; Conterno, L. Green and Eco-Friendly Approaches for the Extraction of Chitin and Chitosan: A Review. Carbohydr. Polym. 2022, 287, 119349. [Google Scholar] [CrossRef] [PubMed]
- Crini, G. Historical Review on Chitin and Chitosan Biopolymers. Environ. Chem. Lett. 2019, 17, 1623–1643. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef]
- Percot, A.; Viton, C.; Domard, A. Optimization of Chitin Extraction from Shrimp Shells. Biomacromolecules 2003, 4, 12–18. [Google Scholar] [CrossRef]
- Jung, W.J.; Jo, G.H.; Kuk, J.H.; Kim, K.Y.; Park, R.D. Extraction of Chitin from Red Crab Shell Waste by Cofermentation with Lactobacillus Paracasei Subsp. Tolerans KCTC-3074 and Serratia Marcescens FS-3. Appl. Microbiol. Biotechnol. 2006, 71, 234–237. [Google Scholar] [CrossRef]
- Kafetzopoulos, D.; Martinou, A.; Bouriotis, V. Bioconversion of Chitin to Chitosan: Purification and Characterization of Chitin Deacetylase from Mucor Rouxii. Proc. Natl. Acad. Sci. USA 1993, 90, 2564–2568. [Google Scholar] [CrossRef] [PubMed]
- No, H.K.; Meyers, S.P. Preparation and Characterization of Chitin and Chitosan—A Review. J. Aquat. Food Prod. Technol. 1995, 4, 27–52. [Google Scholar] [CrossRef]
- Mourya, V.K.; Inamdar, N.N. Chitosan-Modifications and Applications: Opportunities Galore. React. Funct. Polym. 2008, 68, 1013–1051. [Google Scholar] [CrossRef]
- Takbirgou, H.; Emam-Djomeh, Z.; Salami, M.; Askari, G. Chitin and Chitosan-Based Polymer Blends, Interpenetrating Polymer Networks, and Gels. In Handbook of Natural Polymers, Volume 1; Elsevier: Amsterdam, The Netherlands, 2023; pp. 153–175. [Google Scholar]
- Aguirre-Pranzoni, C.; García, M.G.; Ochoa, N.A. Structural and Conformational Changes on Chitosan after Green Heterogeneous Synthesis of Phenyl Derivatives. Carbohydr. Polym. 2023, 312, 120843. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Ghosal, K.; Kumar, M.; Mahmood, S.; Thomas, S. A Detailed Discussion on Interpenetrating Polymer Network (IPN) Based Drug Delivery System for the Advancement of Health Care System. J. Drug Deliv. Sci. Technol. 2022, 79, 104095. [Google Scholar] [CrossRef]
- Khalil, T.E.; Abdel-Salam, A.H.; Mohamed, L.A.; El-Meligy, E.; El-Dissouky, A. Crosslinked Modified Chitosan Biopolymer for Enhanced Removal of Toxic Cr (VI) from Aqueous Solution. Int. J. Biol. Macromol. 2023, 234, 123719. [Google Scholar] [CrossRef] [PubMed]
- Harugade, A.; Sherje, A.P.; Pethe, A. Chitosan: A Review on Properties, Biological Activities and Recent Progress in Biomedical Applications. React. Funct. Polym. 2023, 191, 105634. [Google Scholar] [CrossRef]
- Yu, J.; Hu, N.; Hou, L.; Hang, F.; Li, K.; Xie, C. Effect of Deacetylation of Chitosan on Physicochemical Properties, Antioxidant and Antibacterial Activities of Chitosan-mannose Derivatives. J. Sci. Food Agric. 2023, 103, 6394–6405. [Google Scholar] [CrossRef] [PubMed]
- Mohite, P.; Shah, S.R.; Singh, S.; Rajput, T.; Munde, S.; Ade, N.; Prajapati, B.G.; Paliwal, H.; Mori, D.D.; Dudhrejiya, A.V. Chitosan and Chito-Oligosaccharide: A Versatile Biopolymer with Endless Grafting Possibilities for Multifarious Applications. Front. Bioeng. Biotechnol. 2023, 11, 1190879. [Google Scholar] [CrossRef] [PubMed]
- Thanou, M.; Verhoef, J.C.; Junginger, H.E. Oral Drug Absorption Enhancement by Chitosan and Its Derivatives. Adv. Drug Deliv. Rev. 2001, 52, 117–126. [Google Scholar] [CrossRef]
- Pathak, K.; Misra, S.K.; Sehgal, A.; Singh, S.; Bungau, S.; Najda, A.; Gruszecki, R.; Behl, T. Biomedical Applications of Quaternized Chitosan. Polymers 2021, 13, 2514. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.G.; Zhang, X.; Sun, S.J.; Sun, G.J.; Yao, K.D.; Liang, D.C.; Guo, G.; Zhang, J.Y. N-Alkylated Chitosan as a Potential Nonviral Vector for Gene Transfection. Bioconjugate Chem. 2003, 14, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Wang, Z. Advances in Alkylated Chitosan and Its Applications for Hemostasis. Macromol 2022, 2, 346–360. [Google Scholar] [CrossRef]
- Je, J.-Y.; Kim, S.-K. Reactive Oxygen Species Scavenging Activity of Aminoderivatized Chitosan with Different Degree of Deacetylation. Bioorganic Med. Chem. 2006, 14, 5989–5994. [Google Scholar] [CrossRef] [PubMed]
- Negm, N.A.; Hefni, H.H.H.; Abd-Elaal, A.A.A.; Badr, E.A.; Abou Kana, M.T.H. Advancement on Modification of Chitosan Biopolymer and Its Potential Applications. Int. J. Biol. Macromol. 2020, 152, 681–702. [Google Scholar] [CrossRef] [PubMed]
- Tiwary, A.K.; Sapra, B.; Kaur, G.; Rana, V. Chitosan: Modifications and Applications in Dosage Form Design. In Chitosan: Manufacture, Properties and Usage; Nova Science Publishers: New York, NY, USA, 2011; pp. 71–132. [Google Scholar]
- Pokhrel, S.; Yadav, P.N. Functionalization of Chitosan Polymer and Their Applications. J. Macromol. Sci. Part A 2019, 56, 450–475. [Google Scholar] [CrossRef]
- Ardean, C.; Davidescu, C.M.; Nemeş, N.S.; Negrea, A.; Ciopec, M.; Duteanu, N.; Negrea, P.; Duda-Seiman, D.; Musta, V. Factors Influencing the Antibacterial Activity of Chitosan and Chitosan Modified by Functionalization. Int. J. Mol. Sci. 2021, 22, 7449. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.F.; Guan, Y.L.; Yang, D.Z.; Li, Z.; De Yao, K. Antibacterial Action of Chitosan and Carboxymethylated Chitosan. J. Appl. Polym. Sci. 2001, 79, 1324–1335. [Google Scholar]
- Kim, C.H.; Choi, K.S. Synthesis and Properties of Carboxyalkyl Chitosan Derivatives. J. Ind. Eng. Chem. 1998, 4, 19–25. [Google Scholar]
- Khamidullina, L.A.; Tobysheva, P.D.; Cherepanova, O.E.; Puzyrev, I.S.; Pestov, A.V. Carboxyalkyl Derivatives of Chitosan As Promising Growth and Development Regulators of Medical Plants. Her. Russ. Acad. Sci. 2023, 93, 180–186. [Google Scholar] [CrossRef]
- Shigemasa, Y.; Usui, H.; Morimoto, M.; Saimoto, H.; Okamoto, Y.; Minami, S.; Sashiwa, H. Chemical Modification of Chitin and Chitosan 1: Preparation of Partially Deacetylated Chitin Derivatives via a Ring-Opening Reaction with Cyclic Acid Anhydrides in Lithium Chloride/N, N-Dimethylacetamide. Carbohydr. Polym. 1999, 39, 237–243. [Google Scholar] [CrossRef]
- Reis, B.; Gerlach, N.; Steinbach, C.; Haro Carrasco, K.; Oelmann, M.; Schwarz, S.; Müller, M.; Schwarz, D. A Complementary and Revised View on the N-Acylation of Chitosan with Hexanoyl Chloride. Mar. Drugs 2021, 19, 385. [Google Scholar] [CrossRef] [PubMed]
- Sashiwa, H. Chemical Aspects of Chitin and Chitosan Derivatives. In Chitin and Chitosan Derivatives: Advances in Drug Discovery and Developments; Kim, S.-K., Ed.; Taylor & Francis: Abingdon, UK, 2014; pp. 93–111. [Google Scholar]
- Farinha, I.; Freitas, F. Chemically Modified Chitin, Chitosan, and Chitinous Polymers as Biomaterials. In Handbook of Chitin and Chitosan; Elsevier: Amsterdam, The Netherlands, 2020; pp. 43–69. [Google Scholar]
- Bernkop-Schnürch, A.; Hopf, T.E. Synthesis and in Vitro Evaluation of Chitosan-Thioglycolic Acid Conjugates. Sci. Pharm. 2001, 69, 109–118. [Google Scholar] [CrossRef]
- Kast, C.E.; Bernkop-Schnürch, A. Thiolated Polymers—Thiomers: Development and in Vitro Evaluation of Chitosan–Thioglycolic Acid Conjugates. Biomaterials 2001, 22, 2345–2352. [Google Scholar] [CrossRef]
- Chen, Q.; Qi, Y.; Jiang, Y.; Quan, W.; Luo, H.; Wu, K.; Li, S.; Ouyang, Q. Progress in Research of Chitosan Chemical Modification Technologies and Their Applications. Mar. Drugs 2022, 20, 536. [Google Scholar] [CrossRef]
- Ishihara, M. Photocrosslinkable Chitosan Hydrogel as a Wound Dressing and a Biological Adhesive. Trends Glycosci. Glycotechnol. 2002, 14, 331–341. [Google Scholar] [CrossRef]
- Ono, K.; Saito, Y.; Yura, H.; Ishikawa, K.; Kurita, A.; Akaike, T.; Ishihara, M. Photocrosslinkable Chitosan as a Biological Adhesive. J. Biomed. Mater. Res. An Off. J. Soc. Biomater. Jpn. Soc. Biomater. 2000, 49, 289–295. [Google Scholar] [CrossRef]
- Heras, A.; Rodriguez, N.M.; Ramos, V.M.; Agullo, E. N-Methylene Phosphonic Chitosan: A Novel Soluble Derivative. Carbohydr. Polym. 2001, 44, 1–8. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Shafi, M.E.; Zabermawi, N.M.; Arif, M.; Batiha, G.E.; Khafaga, A.F.; Abd El-Hakim, Y.M.; Al-Sagheer, A.A. Antimicrobial and Antioxidant Properties of Chitosan and Its Derivatives and Their Applications: A Review. Int. J. Biol. Macromol. 2020, 164, 2726–2744. [Google Scholar] [CrossRef]
- Wang, W.; Xue, C.; Mao, X. Chitosan: Structural Modification, Biological Activity and Application. Int. J. Biol. Macromol. 2020, 164, 4532–4546. [Google Scholar] [CrossRef]
- Freitas, E.D.; Moura, C.F., Jr.; Kerwald, J.; Beppu, M.M. An Overview of Current Knowledge on the Properties, Synthesis and Applications of Quaternary Chitosan Derivatives. Polymers 2020, 12, 2878. [Google Scholar] [CrossRef]
- Riaz Rajoka, M.S.; Mehwish, H.M.; Wu, Y.; Zhao, L.; Arfat, Y.; Majeed, K.; Anwaar, S. Chitin/Chitosan Derivatives and Their Interactions with Microorganisms: A Comprehensive Review and Future Perspectives. Crit. Rev. Biotechnol. 2020, 40, 365–379. [Google Scholar] [CrossRef] [PubMed]
- Mukhtar, M.; Fényes, E.; Bartos, C.; Zeeshan, M.; Ambrus, R. Chitosan Biopolymer, Its Derivatives and Potential Applications in Nano-Therapeutics: A Comprehensive Review. Eur. Polym. J. 2021, 160, 110767. [Google Scholar] [CrossRef]
- Pellis, A.; Guebitz, G.M.; Nyanhongo, G.S. Chitosan: Sources, Processing and Modification Techniques. Gels 2022, 8, 393. [Google Scholar] [CrossRef]
- Nayak, A.K.; Ahmad, S.A.; Hasnain, M.S. Chitosan-Based Nanoparticles in Drug Delivery. In Chitosan in Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2022; pp. 55–82. [Google Scholar]
- Alavi, M.; Rai, M. Antibacterial and Wound Healing Activities of Micro/Nanocarriers Based on Carboxymethyl and Quaternized Chitosan Derivatives. In Biopolymer-Based Nano Films; Elsevier: Amsterdam, The Netherlands, 2021; pp. 191–201. [Google Scholar]
- Parhi, R. Drug Delivery Applications of Chitin and Chitosan: A Review. Environ. Chem. Lett. 2020, 18, 577–594. [Google Scholar] [CrossRef]
- Gull, N.; Khan, S.M.; Butt, O.M.; Islam, A.; Shah, A.; Jabeen, S.; Khan, S.U.; Khan, A.; Khan, R.U.; Butt, M.T.Z. Inflammation Targeted Chitosan-Based Hydrogel for Controlled Release of Diclofenac Sodium. Int. J. Biol. Macromol. 2020, 162, 175–187. [Google Scholar] [CrossRef]
- Sharma, D.; Arora, S.; Banerjee, A.; Singh, J. Improved Insulin Sensitivity in Obese-Diabetic Mice via Chitosan Nanomicelles Mediated Silencing of pro-Inflammatory Adipocytokines. Nanomed. Nanotechnol. Biol. Med. 2021, 33, 102357. [Google Scholar] [CrossRef]
- Safarzadeh, M.; Sadeghi, S.; Azizi, M.; Rastegari-Pouyani, M.; Pouriran, R.; Hoseini, M.H.M. Chitin and Chitosan as Tools to Combat COVID-19: A Triple Approach. Int. J. Biol. Macromol. 2021, 183, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Le Thi, M.Y.P. Chitosan Oligosaccharides Suppress Oxidized LDL-Induced Foam Cell Formation and Inflammatory Response in RAW264. 7 Macrophages. Master’s Thesis, Pukyong National University, Busan, Republic of Korea, 2022. [Google Scholar]
- Li, T.; Yang, J.; Weng, C.; Liu, P.; Huang, Y.; Meng, S.; Li, R.; Yang, L.; Chen, C.; Gong, X. Intra-Articular Injection of Anti-Inflammatory Peptide-Loaded Glycol Chitosan/Fucoidan Nanogels to Inhibit Inflammation and Attenuate Osteoarthritis Progression. Int. J. Biol. Macromol. 2021, 170, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Sukul, M.; Sahariah, P.; Lauzon, H.L.; Borges, J.; Másson, M.; Mano, J.F.; Haugen, H.J.; Reseland, J.E. In Vitro Biological Response of Human Osteoblasts in 3D Chitosan Sponges with Controlled Degree of Deacetylation and Molecular Weight. Carbohydr. Polym. 2021, 254, 117434. [Google Scholar] [CrossRef]
- Iswanti, F.C.; Nurulita, I.; Djauzi, S.; Sadikin, M.; Witarto, A.B.; Yamazaki, T. Preparation, Characterization, and Evaluation of Chitosan-Based Nanoparticles as CpG ODN Carriers. Biotechnol. Biotechnol. Equip. 2019, 33, 390–396. [Google Scholar] [CrossRef]
- Eskandani, R.; Kazempour, M.; Farahzadi, R.; Sanaat, Z.; Eskandani, M.; Adibkia, K.; Vandghanooni, S.; Mokhtarzadeh, A. Engineered Nanoparticles as Emerging Gene/Drug Delivery Systems Targeting the Nuclear Factor-ΚB Protein and Related Signaling Pathways in Cancer. Biomed. Pharmacother. 2022, 156, 113932. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, R.; Tran, H.D.N.; Kurniawan, N.D.; Moonshi, S.S.; Whittaker, A.K.; Ta, H.T. Chitosan Nanococktails Containing Both Ceria and Superparamagnetic Iron Oxide Nanoparticles for Reactive Oxygen Species-Related Theranostics. ACS Appl. Nano Mater. 2021, 4, 3604–3618. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, L.; Tan, W.; Li, Q.; Dong, F.; Guo, Z. Preparation of Chitosan-Rosmarinic Acid Derivatives with Enhanced Antioxidant and Anti-Inflammatory Activities. Carbohydr. Polym. 2022, 296, 119943. [Google Scholar] [CrossRef] [PubMed]
- Confederat, L.G.; Tuchilus, C.G.; Dragan, M.; Sha’at, M.; Dragostin, O.M. Preparation and Antimicrobial Activity of Chitosan and Its Derivatives: A Concise Review. Molecules 2021, 26, 3694. [Google Scholar] [CrossRef]
- Yilmaz Atay, H. Antibacterial Activity of Chitosan-Based Systems. Funct. Chitosan Drug Deliv. Biomed. Appl. 2019, 457–489. [Google Scholar]
- Al-Zahrani, S.S.; Bora, R.S.; Al-Garni, S.M. Antimicrobial Activity of Chitosan Nanoparticles. Biotechnol. Biotechnol. Equip. 2021, 35, 1874–1880. [Google Scholar] [CrossRef]
- Li, J.; Zhuang, S. Antibacterial Activity of Chitosan and Its Derivatives and Their Interaction Mechanism with Bacteria: Current State and Perspectives. Eur. Polym. J. 2020, 138, 109984. [Google Scholar] [CrossRef]
- Jøraholmen, M.W.; Bhargava, A.; Julin, K.; Johannessen, M.; Škalko-Basnet, N. The Antimicrobial Properties of Chitosan Can Be Tailored by Formulation. Mar. Drugs 2020, 18, 96. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Kim, K.D.; Chun, S.C. Antibacterial Activity of Chitosan Nanoparticles: A Review. Processes 2020, 8, 1173. [Google Scholar] [CrossRef]
- Sheikh, M.; Mehnaz, S.; Sadiq, M.B. Prevalence of Fungi in Fresh Tomatoes and Their Control by Chitosan and Sweet Orange (Citrus Sinensis) Peel Essential Oil Coating. J. Sci. Food Agric. 2021, 101, 6248–6257. [Google Scholar] [CrossRef] [PubMed]
- Elmehbad, N.Y.; Mohamed, N.A.; Abd El-Ghany, N.A. Evaluation of the Antimicrobial and Anti-Biofilm Activity of Novel Salicylhydrazido Chitosan Derivatives Impregnated with Titanium Dioxide Nanoparticles. Int. J. Biol. Macromol. 2022, 205, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Omer, A.M.; Eltaweil, A.S.; El-Fakharany, E.M.; Abd El-Monaem, E.M.; Ismail, M.M.F.; Mohy-Eldin, M.S.; Ayoup, M.S. Novel Cytocompatible Chitosan Schiff Base Derivative as a Potent Antibacterial, Antidiabetic, and Anticancer Agent. Arab. J. Sci. Eng. 2023, 48, 7587–7601. [Google Scholar] [CrossRef]
- Li, Y.; Ma, Z.; Yang, X.; Gao, Y.; Ren, Y.; Li, Q.; Qu, Y.; Chen, G.; Zeng, R. Investigation into the Physical Properties, Antioxidant and Antibacterial Activity of Bletilla Striata Polysaccharide/Chitosan Membranes. Int. J. Biol. Macromol. 2021, 182, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Elbhnsawi, N.A.; Elwakil, B.H.; Hassanin, A.H.; Shehata, N.; Elshewemi, S.S.; Hagar, M.; Olama, Z.A. Nano-Chitosan/Eucalyptus Oil/Cellulose Acetate Nanofibers: Manufacturing, Antibacterial and Wound Healing Activities. Membranes 2023, 13, 604. [Google Scholar] [CrossRef] [PubMed]
- Jafari, A.; Hassanajili, S.; Karimi, M.B.; Emami, A.; Ghaffari, F.; Azarpira, N. Effect of Organic/Inorganic Nanoparticles on Performance of Polyurethane Nanocomposites for Potential Wound Dressing Applications. J. Mech. Behav. Biomed. Mater. 2018, 88, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Dhanalakshmi, V.; Nimal, T.R.; Sabitha, M.; Biswas, R.; Jayakumar, R. Skin and Muscle Permeating Antibacterial Nanoparticles for Treating Staphylococcus Aureus Infected Wounds. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Song, C.; Yang, C.; Guo, Q.; Yao, M. Low Molecular Weight Chitosan-Coated Silver Nanoparticles Are Effective for the Treatment of MRSA-Infected Wounds. Int. J. Nanomed. 2017, 12, 295. [Google Scholar] [CrossRef]
- Basha, M.; AbouSamra, M.M.; Awad, G.A.; Mansy, S.S. A Potential Antibacterial Wound Dressing of Cefadroxil Chitosan Nanoparticles in Situ Gel: Fabrication, in Vitro Optimization and in Vivo Evaluation. Int. J. Pharm. 2018, 544, 129–140. [Google Scholar] [CrossRef]
- Wu, Z.; Zhou, W.; Deng, W.; Xu, C.; Cai, Y.; Wang, X. Antibacterial and Hemostatic Thiol-Modified Chitosan-Immobilized AgNPs Composite Sponges. ACS Appl. Mater. Interfaces 2020, 12, 20307–20320. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Malaikozhundan, B.; Parthasarathy, A.; Saravanakumar, K.; Wang, M.-H.; Vaseeharan, B. Nano Biomedical Potential of Biopolymer Chitosan-Capped Silver Nanoparticles with Special Reference to Antibacterial, Antibiofilm, Anticoagulant and Wound Dressing Material. J. Clust. Sci. 2020, 31, 355–366. [Google Scholar] [CrossRef]
- Pansara, C.; Mishra, R.; Mehta, T.; Parikh, A.; Garg, S. Formulation of Chitosan Stabilized Silver Nanoparticle-Containing Wound Healing Film: In Vitro and in Vivo Characterization. J. Pharm. Sci. 2020, 109, 2196–2205. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Fu, J.; Tian, X.; Hua, T.; Poon, T.; Koo, M.; Chan, W. Characteristics of Chitosan Fiber and Their Effects towards Improvement of Antibacterial Activity. Carbohydr. Polym. 2022, 280, 119031. [Google Scholar] [CrossRef]
- Ahmadi, S.; Hivechi, A.; Bahrami, S.H.; Milan, P.B.; Ashraf, S.S. Cinnamon Extract Loaded Electrospun Chitosan/Gelatin Membrane with Antibacterial Activity. Int. J. Biol. Macromol. 2021, 173, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zheng, Y.; Shen, Y.; Shi, Y.; Li, F.; Su, C.; Zhao, L. Chitosan Nanoparticles Loaded Hydrogels Promote Skin Wound Healing through the Modulation of Reactive Oxygen Species. Artif. Cells Nanomed. Biotechnol. 2018, 46, 138–149. [Google Scholar] [CrossRef] [PubMed]
- El-Feky, G.S.; Sharaf, S.S.; El Shafei, A.; Hegazy, A.A. Using Chitosan Nanoparticles as Drug Carriers for the Development of a Silver Sulfadiazine Wound Dressing. Carbohydr. Polym. 2017, 158, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Aranaz, I.; Navarro-García, F.; Morri, M.; Acosta, N.; Casettari, L.; Heras, A. Evaluation of Chitosan Salt Properties in the Production of AgNPs Materials with Antibacterial Activity. Int. J. Biol. Macromol. 2023, 235, 123849. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, A.; Nanditha, C.K.; Kumar, G.S.V. ECM-Mimicking Nanofibrous Scaffold Enriched with Dual Growth Factor Carrying Nanoparticles for Diabetic Wound Healing. Nanoscale Adv. 2021, 3, 3085–3092. [Google Scholar] [CrossRef] [PubMed]
- Şalva, E.; Alan, S.; Karakoyun, B.; Çakalağaoğlu, F.; Özbaş, S.; Akbuğa, J. Investigation of Therapeutic Effects in the Wound Healing of Chitosan/PGM-CSF Complexes. Braz. J. Pharm. Sci. 2022, 58, e19668. [Google Scholar]
- Gritsch, L.; Liverani, L.; Lovell, C.; Boccaccini, A.R. Polycaprolactone Electrospun Fiber Mats Prepared Using Benign Solvents: Blending with Copper (II)-Chitosan Increases the Secretion of Vascular Endothelial Growth Factor in a Bone Marrow Stromal Cell Line. Macromol. Biosci. 2020, 20, 1900355. [Google Scholar] [CrossRef]
- Lin, I.-C.; Wang, T.-J.; Wu, C.-L.; Lu, D.-H.; Chen, Y.-R.; Yang, K.-C. Chitosan-Cartilage Extracellular Matrix Hybrid Scaffold Induces Chondrogenic Differentiation to Adipose-Derived Stem Cells. Regen. Ther. 2020, 14, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Moeini, A.; Pedram, P.; Makvandi, P.; Malinconico, M.; d’Ayala, G.G. Wound Healing and Antimicrobial Effect of Active Secondary Metabolites in Chitosan-Based Wound Dressings: A Review. Carbohydr. Polym. 2020, 233, 115839. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.; Hasan, N.; Cao, J.; Lee, J.; Hlaing, S.P.; Yoo, J.-W. Chitosan-Based Nitric Oxide-Releasing Dressing for Anti-Biofilm and in Vivo Healing Activities in MRSA Biofilm-Infected Wounds. Int. J. Biol. Macromol. 2020, 142, 680–692. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Kumar, D.; Mahmood, S.; Singh, V.; Chopra, S.; Hilles, A.R.; Bhatia, A. Nanotechnology-Driven Wound Healing Potential of Asiaticoside: A Comprehensive Review. RSC Pharm. 2024, 1, 9–36. [Google Scholar] [CrossRef]
- Shamloo, A.; Aghababaie, Z.; Afjoul, H.; Jami, M.; Bidgoli, M.R.; Vossoughi, M.; Ramazani, A.; Kamyabhesari, K. Fabrication and Evaluation of Chitosan/Gelatin/PVA Hydrogel Incorporating Honey for Wound Healing Applications: An in Vitro, in Vivo Study. Int. J. Pharm. 2021, 592, 120068. [Google Scholar] [CrossRef] [PubMed]
- Sathiyaseelan, A.; Saravanakumar, K.; Mariadoss, A.V.A.; Wang, M.-H. Antimicrobial and Wound Healing Properties of FeO Fabricated Chitosan/PVA Nanocomposite Sponge. Antibiotics 2021, 10, 524. [Google Scholar] [CrossRef] [PubMed]
- Askari, M.; Afshar, M.; Khorashadizadeh, M.; Zardast, M.; Naghizadeh, A. Wound Healing Effects of Chitosan Nanosheets/Honey Compounds in Male BALB/c Mice. Int. J. Low. Extrem. Wounds 2022, 15347346221074584. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, B.; Sha, D.; Liu, Y.; Xu, J.; Shi, K.; Yu, C.; Ji, X. Injectable and Antibacterial ε-Poly (l-Lysine)-Modified Poly (Vinyl Alcohol)/Chitosan/AgNPs Hydrogels as Wound Healing Dressings. Polymer 2021, 212, 123155. [Google Scholar] [CrossRef]
- Umar, A.K.; Sriwidodo, S.; Maksum, I.P.; Wathoni, N. Film-Forming Spray of Water-Soluble Chitosan Containing Liposome-Coated Human Epidermal Growth Factor for Wound Healing. Molecules 2021, 26, 5326. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Keshwania, P.; Chopra, S.; Mahmood, S.; Bhatia, A. Therapeutic Potential of Nanocarrier-Mediated Delivery of Phytoconstituents for Wound Healing: Their Current Status and Future Perspective. AAPS PharmSciTech 2023, 24, 155. [Google Scholar] [CrossRef]
- Kumar, M.; Ge, Y.; Hilles, A.R.; Bhatia, A.; Mahmood, S. A Review on Polysaccharides Mediated Electrospun Nanofibers for Diabetic Wound Healing: Their Current Status with Regulatory Perspective. Int. J. Biol. Macromol. 2023, 234, 123696. [Google Scholar] [CrossRef]
- Kumar, M.; Thakur, A.; Mandal, U.K.; Thakur, A.; Bhatia, A. Foam-Based Drug Delivery: A Newer Approach for Pharmaceutical Dosage Form. AAPS PharmSciTech 2022, 23, 244. [Google Scholar] [CrossRef]
- Kumar, M.; Mahmood, S.; Mandal, U.K. An Updated Account On Formulations And Strategies for The Treatment Of Burn Infection—A Review. Curr. Pharm. Des. 2022, 28, 1480–1492. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Mori, T.; Fujinaga, T. Topical Formulations and Wound Healing Applications of Chitosan. Adv. Drug Deliv. Rev. 2001, 52, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Nalini, T.; Khaleel Basha, S.; Mohamed Sadiq, A.; Sugantha Kumari, V. Fabrication and Evaluation of Nanoencapsulated Quercetin for Wound Healing Application. Polym. Bull. 2023, 80, 515–540. [Google Scholar] [CrossRef]
- Saghafi, Y.; Baharifar, H.; Najmoddin, N.; Asefnejad, A.; Maleki, H.; Sajjadi-Jazi, S.M.; Bonkdar, A.; Shams, F.; Khoshnevisan, K. Bromelain-and Silver Nanoparticle-Loaded Polycaprolactone/Chitosan Nanofibrous Dressings for Skin Wound Healing. Gels 2023, 9, 672. [Google Scholar] [CrossRef]
- Aker, S.D.; Tamburaci, S.; Tihminlioglu, F. Development of Cissus Quadrangularis-Loaded POSS-Reinforced Chitosan-Based Bilayer Sponges for Wound Healing Applications: Drug Release and In Vitro Bioactivity. ACS Omega 2023, 8, 19674–19691. [Google Scholar] [CrossRef] [PubMed]
- Montazeri, S.; Rastegari, A.; Mohammadi, Z.; Nazari, M.; Yousefi, M.; Samadi, F.Y.; Najafzadeh, S.; Aghsami, M. Chitosan Nanoparticle Loaded by Epidermal Growth Factor as a Potential Protein Carrier for Wound Healing: In Vitro and in Vivo Studies. IET Nanobiotechnol. 2023, 17, 204–211. [Google Scholar] [CrossRef]
- Kaparekar, P.S.; Pathmanapan, S.; Anandasadagopan, S.K. Polymeric Scaffold of Gallic Acid Loaded Chitosan Nanoparticles Infused with Collagen-Fibrin for Wound Dressing Application. Int. J. Biol. Macromol. 2020, 165, 930–947. [Google Scholar] [CrossRef]
- Chu, W.; Wang, P.; Ma, Z.; Peng, L.; Guo, C.; Fu, Y.; Ding, L. Lupeol-Loaded Chitosan-Ag+ Nanoparticle/Sericin Hydrogel Accelerates Wound Healing and Effectively Inhibits Bacterial Infection. Int. J. Biol. Macromol. 2023, 243, 125310. [Google Scholar] [CrossRef]
- Zahiri, M.; Khanmohammadi, M.; Goodarzi, A.; Ababzadeh, S.; Farahani, M.S.; Mohandesnezhad, S.; Bahrami, N.; Nabipour, I.; Ai, J. Encapsulation of Curcumin Loaded Chitosan Nanoparticle within Poly (ε-Caprolactone) and Gelatin Fiber Mat for Wound Healing and Layered Dermal Reconstitution. Int. J. Biol. Macromol. 2020, 153, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Khosravian, P.; Javdani, M.; Noorbakhnia, R.; Moghtadaei-Khorasgani, E.; Barzegar, A. Preparation and Evaluation of Chitosan Skin Patches Containing Mesoporous Silica Nanoparticles Loaded by Doxycycline on Skin Wound Healing. Arch. Dermatol. Res. 2023, 315, 1333–1345. [Google Scholar] [CrossRef] [PubMed]
- Hajji, S.; Khedir, S.B.; Hamza-Mnif, I.; Hamdi, M.; Jedidi, I.; Kallel, R.; Boufi, S.; Nasri, M. Biomedical Potential of Chitosan-Silver Nanoparticles with Special Reference to Antioxidant, Antibacterial, Hemolytic and in Vivo Cutaneous Wound Healing Effects. Biochim. Biophys. Acta BBA Gen. Subj. 2019, 1863, 241–254. [Google Scholar] [CrossRef]
- Shao, J.; Wang, B.; Li, J.; Jansen, J.A.; Walboomers, X.F.; Yang, F. Antibacterial Effect and Wound Healing Ability of Silver Nanoparticles Incorporation into Chitosan-Based Nanofibrous Membranes. Mater. Sci. Eng. C 2019, 98, 1053–1063. [Google Scholar] [CrossRef] [PubMed]
- Correa, V.L.R.; Martins, J.A.; de Souza, T.R.; Rincon, G.d.C.N.; Miguel, M.P.; de Menezes, L.B.; Amaral, A.C. Melatonin Loaded Lecithin-Chitosan Nanoparticles Improved the Wound Healing in Diabetic Rats. Int. J. Biol. Macromol. 2020, 162, 1465–1475. [Google Scholar] [CrossRef]
- Shende, P.; Gupta, H. Formulation and Comparative Characterization of Nanoparticles of Curcumin Using Natural, Synthetic and Semi-Synthetic Polymers for Wound Healing. Life Sci. 2020, 253, 117588. [Google Scholar] [CrossRef]
- Ehterami, A.; Salehi, M.; Farzamfar, S.; Vaez, A.; Samadian, H.; Sahrapeyma, H.; Mirzaii, M.; Ghorbani, S.; Goodarzi, A. In Vitro and in Vivo Study of PCL/COLL Wound Dressing Loaded with Insulin-Chitosan Nanoparticles on Cutaneous Wound Healing in Rats Model. Int. J. Biol. Macromol. 2018, 117, 601–609. [Google Scholar] [CrossRef]
- Fahimirad, S.; Abtahi, H.; Satei, P.; Ghaznavi-Rad, E.; Moslehi, M.; Ganji, A. Wound Healing Performance of PCL/Chitosan Based Electrospun Nanofiber Electrosprayed with Curcumin Loaded Chitosan Nanoparticles. Carbohydr. Polym. 2021, 259, 117640. [Google Scholar] [CrossRef]
- Mourya, V.K.; Inamdar, N.N. Trimethyl Chitosan and Its Applications in Drug Delivery. J. Mater. Sci. Mater. Med. 2009, 20, 1057–1079. [Google Scholar] [CrossRef]
- Abu Elella, M.H.; Abdallah, H.M.; Gamal, H.; Moustafa, E.B.; Goda, E.S. Rational Design of Biocompatible IPNs Hydrogels Containing Carboxymethyl Starch and Trimethyl Chitosan Chloride with High Antibacterial Activity. Cellulose 2022, 29, 7317–7330. [Google Scholar] [CrossRef]
- Geng, Y.; Xue, H.; Zhang, Z.; Panayi, A.C.; Knoedler, S.; Zhou, W.; Mi, B.; Liu, G. Recent Advances in Carboxymethyl Chitosan-Based Materials for Biomedical Applications. Carbohydr. Polym. 2023, 305, 120555. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Yang, M.; Woo, M.W.; Li, Y.; Han, W.; Dang, X. High-Mechanical Strength Carboxymethyl Chitosan-Based Hydrogel Film for Antibacterial Wound Dressing. Carbohydr. Polym. 2021, 256, 117590. [Google Scholar] [CrossRef]
- Wibel, R.; Braun, D.E.; Hämmerle, L.; Jörgensen, A.M.; Knoll, P.; Salvenmoser, W.; Steinbring, C.; Bernkop-Schnürch, A. In Vitro Investigation of Thiolated Chitosan Derivatives as Mucoadhesive Coating Materials for Solid Lipid Nanoparticles. Biomacromolecules 2021, 22, 3980–3991. [Google Scholar] [CrossRef] [PubMed]
- Samprasit, W.; Opanasopit, P.; Chamsai, B. Mucoadhesive Chitosan and Thiolated Chitosan Nanoparticles Containing Alpha Mangostin for Possible Colon-Targeted Delivery. Pharm. Dev. Technol. 2021, 26, 362–372. [Google Scholar] [CrossRef]
- Sun, X.; Zhang, J.; Mi, Y.; Chen, Y.; Tan, W.; Li, Q.; Dong, F.; Guo, Z. Synthesis, Characterization, and the Antioxidant Activity of the Acetylated Chitosan Derivatives Containing Sulfonium Salts. Int. J. Biol. Macromol. 2020, 152, 349–358. [Google Scholar] [CrossRef]
- Mio, L.; Sacco, P.; Donati, I. Influence of Temperature and Polymer Concentration on the Nonlinear Response of Highly Acetylated Chitosan–Genipin Hydrogels. Gels 2022, 8, 194. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Wu, L.; Yan, H.; Jiang, Z.; Li, S.; Li, W.; Bai, Y.; Wang, H.; Cheng, Z.; Kong, D. Microchannelled Alkylated Chitosan Sponge to Treat Noncompressible Hemorrhages and Facilitate Wound Healing. Nat. Commun. 2021, 12, 4733. [Google Scholar] [CrossRef]
- Chen, X.; Yan, G.; Chen, M.; Yang, P.; Xu, B. Alkylated Chitosan-Attapulgite Composite Sponge for Rapid Hemostasis. Biomater. Adv. 2023, 153, 213569. [Google Scholar] [CrossRef] [PubMed]
- Suflet, D.M.; Popescu, I.; Pelin, I.M.; Ichim, D.L.; Daraba, O.M.; Constantin, M.; Fundueanu, G. Dual Cross-Linked Chitosan/PVA Hydrogels Containing Silver Nanoparticles with Antimicrobial Properties. Pharmaceutics 2021, 13, 1461. [Google Scholar] [CrossRef]
- Guo, S.; Zhang, X.; Ma, R.; Ge, X.; Shen, H.; Liang, W.; Zhang, G.; Li, W. Preparation and Characterization of Polyvinyl Alcohol/Glutaraldehyde Cross-Linked Chitosan/ε-Polylysine Degradable Composite Film and Its Antibacterial Effect. J. Food Eng. 2023, 359, 111698. [Google Scholar] [CrossRef]
- Chang, H.K.; Yang, D.H.; Ha, M.Y.; Kim, H.J.; Kim, C.H.; Kim, S.H.; Choi, J.W.; Chun, H.J. 3D Printing of Cell-Laden Visible Light Curable Glycol Chitosan Bioink for Bone Tissue Engineering. Carbohydr. Polym. 2022, 287, 119328. [Google Scholar] [CrossRef] [PubMed]
- Inbaraj, B.S.; Chen, B.-Y.; Liao, C.-W.; Chen, B.-H. Green Synthesis, Characterization and Evaluation of Catalytic and Antibacterial Activities of Chitosan, Glycol Chitosan and Poly (γ-Glutamic Acid) Capped Gold Nanoparticles. Int. J. Biol. Macromol. 2020, 161, 1484–1495. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, L.; Koschella, A.; Tied, A.; Pfeifer, A.; Heinze, T. Sulfoethylation of Polysaccharides—A Comparative Study. Carbohydr. Polym. 2020, 246, 116533. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, L.; Heinze, T. Structure Design of Polysaccharides–Chemoselective Sulfoethylation of Chitosan. Eur. Polym. J. 2020, 140, 109978. [Google Scholar] [CrossRef]
- Bolshakov, I.N.; Gornostaev, L.M.; Fominykh, O.I.; Svetlakov, A.V. Synthesis, Chemical and Biomedical Aspects of the Use of Sulfated Chitosan. Polymers 2022, 14, 3431. [Google Scholar] [CrossRef] [PubMed]
- Karami, F.; Saber-Samandari, S. Synthesis and Characterization of a Novel Hydrogel Based on Carboxymethyl Chitosan/Sodium Alginate with the Ability to Release Simvastatin for Chronic Wound Healing. Biomed. Mater. 2023, 18, 025001. [Google Scholar] [CrossRef] [PubMed]
- Nie, L.; Wei, Q.; Sun, M.; Ding, P.; Wang, L.; Sun, Y.; Ding, X.; Okoro, O.V.; Jiang, G.; Shavandi, A. Injectable, Self-Healing, Transparent, and Antibacterial Hydrogels Based on Chitosan and Dextran for Wound Dressings. Int. J. Biol. Macromol. 2023, 233, 123494. [Google Scholar] [CrossRef] [PubMed]
- Bakhsheshi-Rad, H.R.; Hadisi, Z.; Ismail, A.F.; Aziz, M.; Akbari, M.; Berto, F.; Chen, X.B. In Vitro and in Vivo Evaluation of Chitosan-Alginate/Gentamicin Wound Dressing Nanofibrous with High Antibacterial Performance. Polym. Test. 2020, 82, 106298. [Google Scholar] [CrossRef]
- Xie, Y.; Liao, X.; Zhang, J.; Yang, F.; Fan, Z. Novel Chitosan Hydrogels Reinforced by Silver Nanoparticles with Ultrahigh Mechanical and High Antibacterial Properties for Accelerating Wound Healing. Int. J. Biol. Macromol. 2018, 119, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Archana, D.; Dutta, J.; Dutta, P.K. Evaluation of Chitosan Nano Dressing for Wound Healing: Characterization, in Vitro and in Vivo Studies. Int. J. Biol. Macromol. 2013, 57, 193–203. [Google Scholar] [CrossRef]
- Fan, Y.; Lu, Q.; Liang, W.; Wang, Y.; Zhou, Y.; Lang, M. Preparation and Characterization of Antibacterial Polyvinyl Alcohol/Chitosan Sponge and Potential Applied for Wound Dressing. Eur. Polym. J. 2021, 157, 110619. [Google Scholar] [CrossRef]
- Kim, J.O.; Noh, J.-K.; Thapa, R.K.; Hasan, N.; Choi, M.; Kim, J.H.; Lee, J.-H.; Ku, S.K.; Yoo, J.-W. Nitric Oxide-Releasing Chitosan Film for Enhanced Antibacterial and in Vivo Wound-Healing Efficacy. Int. J. Biol. Macromol. 2015, 79, 217–225. [Google Scholar] [CrossRef]
- Dong, F.; Li, S. Wound Dressings Based on Chitosan-Dialdehyde Cellulose Nanocrystals-Silver Nanoparticles: Mechanical Strength, Antibacterial Activity and Cytotoxicity. Polymers 2018, 10, 673. [Google Scholar] [CrossRef]
- Rahmani, H.; Najafi, S.H.M.; Ashori, A.; Fashapoyeh, M.A.; Mohseni, F.A.; Torkaman, S. Preparation of Chitosan-Based Composites with Urethane Cross Linkage and Evaluation of Their Properties for Using as Wound Healing Dressing. Carbohydr. Polym. 2020, 230, 115606. [Google Scholar] [CrossRef]
- Sudheesh Kumar, P.T.; Lakshmanan, V.-K.; Anilkumar, T.V.; Ramya, C.; Reshmi, P.; Unnikrishnan, A.G.; Nair, S.V.; Jayakumar, R. Flexible and Microporous Chitosan Hydrogel/Nano ZnO Composite Bandages for Wound Dressing: In Vitro and in Vivo Evaluation. ACS Appl. Mater. Interfaces 2012, 4, 2618–2629. [Google Scholar] [CrossRef]
- Ushmarov, D.; Gumenyuk, S.; Gumenyuk, A.; Gaivoronskaya, T.; Grigoriev, T.; Lukanina, K.; Patsaev, T.; Domenyuk, D. Preclinical Trials for Advanced Chitosan-Based Coatings in Treating Purulent Wounds. Arch. EuroMedica 2021, 11, 63–71. [Google Scholar] [CrossRef]
- Intini, C.; Elviri, L.; Cabral, J.; Mros, S.; Bergonzi, C.; Bianchera, A.; Flammini, L.; Govoni, P.; Barocelli, E.; Bettini, R. 3D-Printed Chitosan-Based Scaffolds: An in Vitro Study of Human Skin Cell Growth and an in-Vivo Wound Healing Evaluation in Experimental Diabetes in Rats. Carbohydr. Polym. 2018, 199, 593–602. [Google Scholar] [CrossRef]
- Aramwit, P.; Yamdech, R.; Ampawong, S. Controlled Release of Chitosan and Sericin from the Microspheres-Embedded Wound Dressing for the Prolonged Anti-Microbial and Wound Healing Efficacy. AAPS J. 2016, 18, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Madni, A.; Kousar, R.; Naeem, N.; Wahid, F. Recent Advancements in Applications of Chitosan-Based Biomaterials for Skin Tissue Engineering. J. Bioresour. Bioprod. 2021, 6, 11–25. [Google Scholar] [CrossRef]
- Mohebbi, S.; Nezhad, M.N.; Zarrintaj, P.; Jafari, S.H.; Gholizadeh, S.S.; Saeb, M.R.; Mozafari, M. Chitosan in Biomedical Engineering: A Critical Review. Curr. Stem Cell Res. Ther. 2019, 14, 93–116. [Google Scholar] [CrossRef]
- Xu, J.; Fang, H.; Zheng, S.; Li, L.; Jiao, Z.; Wang, H.; Nie, Y.; Liu, T.; Song, K. A Biological Functional Hybrid Scaffold Based on Decellularized Extracellular Matrix/Gelatin/Chitosan with High Biocompatibility and Antibacterial Activity for Skin Tissue Engineering. Int. J. Biol. Macromol. 2021, 187, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Ressler, A. Chitosan-Based Biomaterials for Bone Tissue Engineering Applications: A Short Review. Polymers 2022, 14, 3430. [Google Scholar] [CrossRef] [PubMed]
- Kołodziejska, M.; Jankowska, K.; Klak, M.; Wszoła, M. Chitosan as an Underrated Polymer in Modern Tissue Engineering. Nanomaterials 2021, 11, 3019. [Google Scholar] [CrossRef] [PubMed]
- Kazimierczak, P.; Benko, A.; Nocun, M.; Przekora, A. Novel Chitosan/Agarose/Hydroxyapatite Nanocomposite Scaffold for Bone Tissue Engineering Applications: Comprehensive Evaluation of Biocompatibility and Osteoinductivity with the Use of Osteoblasts and Mesenchymal Stem Cells. Int. J. Nanomed. 2019, 14, 6615–6630. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, F.S.; Sharifianjazi, F.; Esmaeilkhanian, A.; Salehi, E. Chitosan Films and Scaffolds for Regenerative Medicine Applications: A Review. Carbohydr. Polym. 2021, 273, 118631. [Google Scholar] [CrossRef]
- Pahlevanzadeh, F.; Emadi, R.; Valiani, A.; Kharaziha, M.; Poursamar, S.A.; Bakhsheshi-Rad, H.R.; Ismail, A.F.; RamaKrishna, S.; Berto, F. Three-Dimensional Printing Constructs Based on the Chitosan for Tissue Regeneration: State of the Art, Developing Directions and Prospect Trends. Materials 2020, 13, 2663. [Google Scholar] [CrossRef] [PubMed]
- Yousefiasl, S.; Manoochehri, H.; Makvandi, P.; Afshar, S.; Salahinejad, E.; Khosraviyan, P.; Saidijam, M.; Soleimani Asl, S.; Sharifi, E. Chitosan/Alginate Bionanocomposites Adorned with Mesoporous Silica Nanoparticles for Bone Tissue Engineering. J. Nanostructure Chem. 2023, 13, 389–403. [Google Scholar] [CrossRef]
- Suo, H.; Zhang, J.; Xu, M.; Wang, L. Low-Temperature 3D Printing of Collagen and Chitosan Composite for Tissue Engineering. Mater. Sci. Eng. C 2021, 123, 111963. [Google Scholar] [CrossRef] [PubMed]
- Bhuiyan, M.H.; Clarkson, A.N.; Ali, M.A. Optimization of Thermoresponsive Chitosan/β-Glycerophosphate Hydrogels for Injectable Neural Tissue Engineering Application. Colloids Surf. B Biointerfaces 2023, 224, 113193. [Google Scholar] [CrossRef]
- Ali, A.; Hasan, A.; Negi, Y.S. Effect of Carbon Based Fillers on Xylan/Chitosan/Nano-HAp Composite Matrix for Bone Tissue Engineering Application. Int. J. Biol. Macromol. 2022, 197, 1–11. [Google Scholar] [CrossRef]
- Wasupalli, G.K.; Verma, D. Thermosensitive Injectable Hydrogel Based on Chitosan-Polygalacturonic Acid Polyelectrolyte Complexes for Bone Tissue Engineering. Carbohydr. Polym. 2022, 294, 119769. [Google Scholar] [CrossRef] [PubMed]
- Nazar, V.; Kashi, M.; Haghbin Nazarpak, M.; Shahryari, E.; Mehrjoo, M. Gelatin Hydrogel Reinforced by Graphene Oxide Grafted Chitosan for Cartilage Tissue Engineering Application. Int. J. Polym. Mater. Polym. Biomater. 2022, 72, 1120–1131. [Google Scholar] [CrossRef]
- Cheng, R.; Cao, Y.; Yan, Y.; Shen, Z.; Zhao, Y.; Zhang, Y.; Sang, S.; Han, Y. Fabrication and Characterization of Chitosan-Based Composite Scaffolds for Neural Tissue Engineering. Int. J. Polym. Mater. Polym. Biomater. 2022, 71, 831–841. [Google Scholar] [CrossRef]
- Gunes, O.C.; Kara, A.; Baysan, G.; Bugra Husemoglu, R.; Akokay, P.; Ziylan Albayrak, A.; Ergur, B.U.; Havitcioglu, H. Fabrication of 3D Printed Poly (Lactic Acid) Strut and Wet-Electrospun Cellulose Nano Fiber Reinforced Chitosan-Collagen Hydrogel Composite Scaffolds for Meniscus Tissue Engineering. J. Biomater. Appl. 2022, 37, 683–697. [Google Scholar] [CrossRef] [PubMed]
- Nosrati, H.; Banitalebi-Dehkordi, M.; Khodaei, M.; Sharifi, E.; Asadpour, S.; Mansouri, K.; Soleimannejad, M. Preparation and in Vitro Characterization of Electrospun Scaffolds Composed of Chitosan, Gelatin and 58S Bioactive Glass Nanoparticles for Skin Tissue Engineering. J. Shahrekord Univ. Med. Sci. 2022, 24, 1–6. [Google Scholar] [CrossRef]
- Sadeghianmaryan, A.; Naghieh, S.; Yazdanpanah, Z.; Sardroud, H.A.; Sharma, N.K.; Wilson, L.D.; Chen, X. Fabrication of Chitosan/Alginate/Hydroxyapatite Hybrid Scaffolds Using 3D Printing and Impregnating Techniques for Potential Cartilage Regeneration. Int. J. Biol. Macromol. 2022, 204, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Bei, Z.; Wei, J.; Yan, X.; Wen, H.; Cao, Y.; Li, H. Mussel-Inspired Injectable Chitosan Hydrogel Modified with Catechol for Cell Adhesion and Cartilage Defect Repair. J. Mater. Chem. B 2022, 10, 1019–1030. [Google Scholar] [CrossRef]
- Singh, A.K.; Pramanik, K. Fabrication and Investigation of Physicochemical and Biological Properties of 3D Printed Sodium Alginate-chitosan Blend Polyelectrolyte Complex Scaffold for Bone Tissue Engineering Application. J. Appl. Polym. Sci. 2023, 140, e53642. [Google Scholar] [CrossRef]
- Wasupalli, G.K.; Verma, D. Development of Chitosan-polygalacturonic Acid Polyelectrolyte Complex Fibrous Scaffolds Using the Hydrothermal Treatment for Bone Tissue Engineering. J. Biomed. Mater. Res. Part A 2023, 111, 354–366. [Google Scholar] [CrossRef]
- Giordano-Kelhoffer, B.; Rodríguez-Gonzalez, R.; Perpiñan-Blasco, M.; Buitrago, J.O.; Bosch, B.M.; Perez, R.A. A Novel Chitosan Composite Biomaterial with Drug Eluting Capacity for Maxillary Bone Regeneration. Materials 2023, 16, 685. [Google Scholar] [CrossRef]
- Samie, M.; Khan, A.F.; Rahman, S.U.; Iqbal, H.; Yameen, M.A.; Chaudhry, A.A.; Galeb, H.A.; Halcovitch, N.R.; Hardy, J.G. Drug/Bioactive Eluting Chitosan Composite Foams for Osteochondral Tissue Engineering. Int. J. Biol. Macromol. 2023, 229, 561–574. [Google Scholar] [CrossRef] [PubMed]
- Peifen, M.; Mengyun, L.; Jinglong, H.; Danqian, L.; Yan, T.; Liwei, X.; Han, Z.; Jianlong, D.; Lingyan, L.; Guanghui, Z. New Skin Tissue Engineering Scaffold with Sulfated Silk Fibroin/Chitosan/Hydroxyapatite and Its Application. Biochem. Biophys. Res. Commun. 2023, 640, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Gao, S.; Shui, S.; Liu, S.; Qu, H.; Liu, C.; Zheng, L. Small Interfering RNA-Loaded Chitosan Hydrochloride/Carboxymethyl Chitosan Nanoparticles for Ultrasound-Triggered Release to Hamper Colorectal Cancer Growth in Vitro. Int. J. Biol. Macromol. 2020, 162, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Lara-Velazquez, M.; Alkharboosh, R.; Norton, E.S.; Ramirez-Loera, C.; Freeman, W.D.; Guerrero-Cazares, H.; Forte, A.J.; Quiñones-Hinojosa, A.; Sarabia-Estrada, R. Chitosan-Based Non-Viral Gene and Drug Delivery Systems for Brain Cancer. Front. Neurol. 2020, 11, 740. [Google Scholar] [CrossRef] [PubMed]
- Moramkar, N.; Bhatt, P. Insight into Chitosan Derived Nanotherapeutics for Anticancer Drug Delivery and Imaging. Eur. Polym. J. 2021, 154, 110540. [Google Scholar] [CrossRef]
- Gaikwad, G.; Rohra, N.; Kumar, C.; Jadhav, S.; Sarma, H.D.; Borade, L.; Chakraborty, S.; Bhagwat, S.; Dandekar, P.; Jain, R. A Facile Strategy for Synthesis of a Broad Palette of Intrinsically Radiolabeled Chitosan Nanoparticles for Potential Use in Cancer Theranostics. J. Drug Deliv. Sci. Technol. 2021, 63, 102485. [Google Scholar] [CrossRef]
- Nikkhoo, A.; Rostami, N.; Farhadi, S.; Esmaily, M.; Ardebili, S.M.; Atyabi, F.; Baghaei, M.; Haghnavaz, N.; Yousefi, M.; Aliparasti, M.R. Codelivery of STAT3 SiRNA and BV6 by Carboxymethyl Dextran Trimethyl Chitosan Nanoparticles Suppresses Cancer Cell Progression. Int. J. Pharm. 2020, 581, 119236. [Google Scholar] [CrossRef]
- Bastaki, S.; Aravindhan, S.; Saheb, N.A.; Kashani, M.A.; Dorofeev, A.E.; Kiani, F.K.; Jahandideh, H.; Dargani, F.B.; Aksoun, M.; Nikkhoo, A. Codelivery of STAT3 and PD-L1 SiRNA by Hyaluronate-TAT Trimethyl/Thiolated Chitosan Nanoparticles Suppresses Cancer Progression in Tumor-Bearing Mice. Life Sci. 2021, 266, 118847. [Google Scholar] [CrossRef] [PubMed]
- Zaharoff, D.A.; Hoffman, B.S.; Hooper, H.B.; Benjamin, C.J., Jr.; Khurana, K.K.; Hance, K.W.; Rogers, C.J.; Pinto, P.A.; Schlom, J.; Greiner, J.W. Intravesical Immunotherapy of Superficial Bladder Cancer with Chitosan/Interleukin-12. Cancer Res. 2009, 69, 6192–6199. [Google Scholar] [CrossRef]
- Al-Nemrawi, N.K.; Altawabeyeh, R.M.; Darweesh, R.S. Preparation and Characterization of Docetaxel-PLGA Nanoparticles Coated with Folic Acid-Chitosan Conjugate for Cancer Treatment. J. Pharm. Sci. 2022, 111, 485–494. [Google Scholar] [CrossRef]
- Manimaran, D.; Elangovan, N.; Mani, P.; Subramanian, K.; Ali, D.; Alarifi, S.; Palanisamy, C.P.; Zhang, H.; Rangasamy, K.; Palanisamy, V. Isolongifolene-Loaded Chitosan Nanoparticles Synthesis and Characterization for Cancer Treatment. Sci. Rep. 2022, 12, 19250. [Google Scholar] [CrossRef]
- Shakeran, Z.; Keyhanfar, M.; Varshosaz, J.; Sutherland, D.S. Biodegradable Nanocarriers Based on Chitosan-Modified Mesoporous Silica Nanoparticles for Delivery of Methotrexate for Application in Breast Cancer Treatment. Mater. Sci. Eng. C 2021, 118, 111526. [Google Scholar] [CrossRef] [PubMed]
- Amini, Z.; Rudsary, S.S.; Shahraeini, S.S.; Dizaji, B.F.; Goleij, P.; Bakhtiari, A.; Irani, M.; Sharifianjazi, F. Magnetic Bioactive Glasses/Cisplatin Loaded-Chitosan (CS)-Grafted-Poly (ε-Caprolactone) Nanofibers against Bone Cancer Treatment. Carbohydr. Polym. 2021, 258, 117680. [Google Scholar] [CrossRef]
- Zhu, X.; Yu, Z.; Feng, L.; Deng, L.; Fang, Z.; Liu, Z.; Li, Y.; Wu, X.; Qin, L.; Guo, R. Chitosan-Based Nanoparticle Co-Delivery of Docetaxel and Curcumin Ameliorates Anti-Tumor Chemoimmunotherapy in Lung Cancer. Carbohydr. Polym. 2021, 268, 118237. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yu, H.; Wang, S.; Gai, C.; Cui, X.; Xu, Z.; Li, W.; Zhang, W. Targeted Delivery of Quercetin by Nanoparticles Based on Chitosan Sensitizing Paclitaxel-Resistant Lung Cancer Cells to Paclitaxel. Mater. Sci. Eng. C 2021, 119, 111442. [Google Scholar] [CrossRef]
- Naruphontjirakul, P.; Viravaidya-Pasuwat, K. Development of Anti-HER2-Targeted Doxorubicin–Core-Shell Chitosan Nanoparticles for the Treatment of Human Breast Cancer. Int. J. Nanomed. 2019, 14, 4105–4121. [Google Scholar] [CrossRef] [PubMed]
- Samadi, A.; Haseli, S.; Pourmadadi, M.; Rashedi, H.; Yazdian, F.; Navaei-Nigjeh, M. Curcumin-Loaded Chitosan-Agarose-Montmorillonite Hydrogel Nanocomposite for the Treatment of Breast Cancer. In Proceedings of the 2020 27th National and 5th International Iranian Conference on Biomedical Engineering (ICBME), Tehran, Iran, 26–27 November 2020; IEEE: Piscataway, NJ, USA, 2020; pp. 148–153. [Google Scholar]
- Aslzad, S.; Heydari, P.; Abdolahinia, E.D.; Amiryaghoubi, N.; Safary, A.; Fathi, M.; Erfan-Niya, H. Chitosan/Gelatin Hybrid Nanogel Containing Doxorubicin as Enzyme-Responsive Drug Delivery System for Breast Cancer Treatment. Colloid Polym. Sci. 2023, 301, 273–281. [Google Scholar] [CrossRef]
- Wang, Y.; Qian, J.; Yang, M.; Xu, W.; Wang, J.; Hou, G.; Ji, L.; Suo, A. Doxorubicin/Cisplatin Co-Loaded Hyaluronic Acid/Chitosan-Based Nanoparticles for in Vitro Synergistic Combination Chemotherapy of Breast Cancer. Carbohydr. Polym. 2019, 225, 115206. [Google Scholar] [CrossRef] [PubMed]
- Bayat, F.; Pourmadadi, M.; Eshaghi, M.M.; Yazdian, F.; Rashedi, H. Improving Release Profile and Anticancer Activity of 5-Fluorouracil for Breast Cancer Therapy Using a Double Drug Delivery System: Chitosan/Agarose/γ-Alumina Nanocomposite@ Double Emulsion. J. Clust. Sci. 2023, 34, 2565–2577. [Google Scholar] [CrossRef]
- Wan, S.; Fan, Q.; Wu, Y.; Zhang, J.; Qiao, G.; Jiang, N.; Yang, J.; Liu, Y.; Li, J.; Chiampanichayakul, S. Curcumin-Loaded Platelet Membrane Bioinspired Chitosan-Modified Liposome for Effective Cancer Therapy. Pharmaceutics 2023, 15, 631. [Google Scholar] [CrossRef]
- Zarepour, A.; Egil, A.C.; Cokol Cakmak, M.; Esmaeili Rad, M.; Cetin, Y.; Aydinlik, S.; Ozaydin Ince, G.; Zarrabi, A. Fabrication of a Dual-Drug-Loaded Smart Niosome-g-Chitosan Polymeric Platform for Lung Cancer Treatment. Polymers 2023, 15, 298. [Google Scholar] [CrossRef] [PubMed]
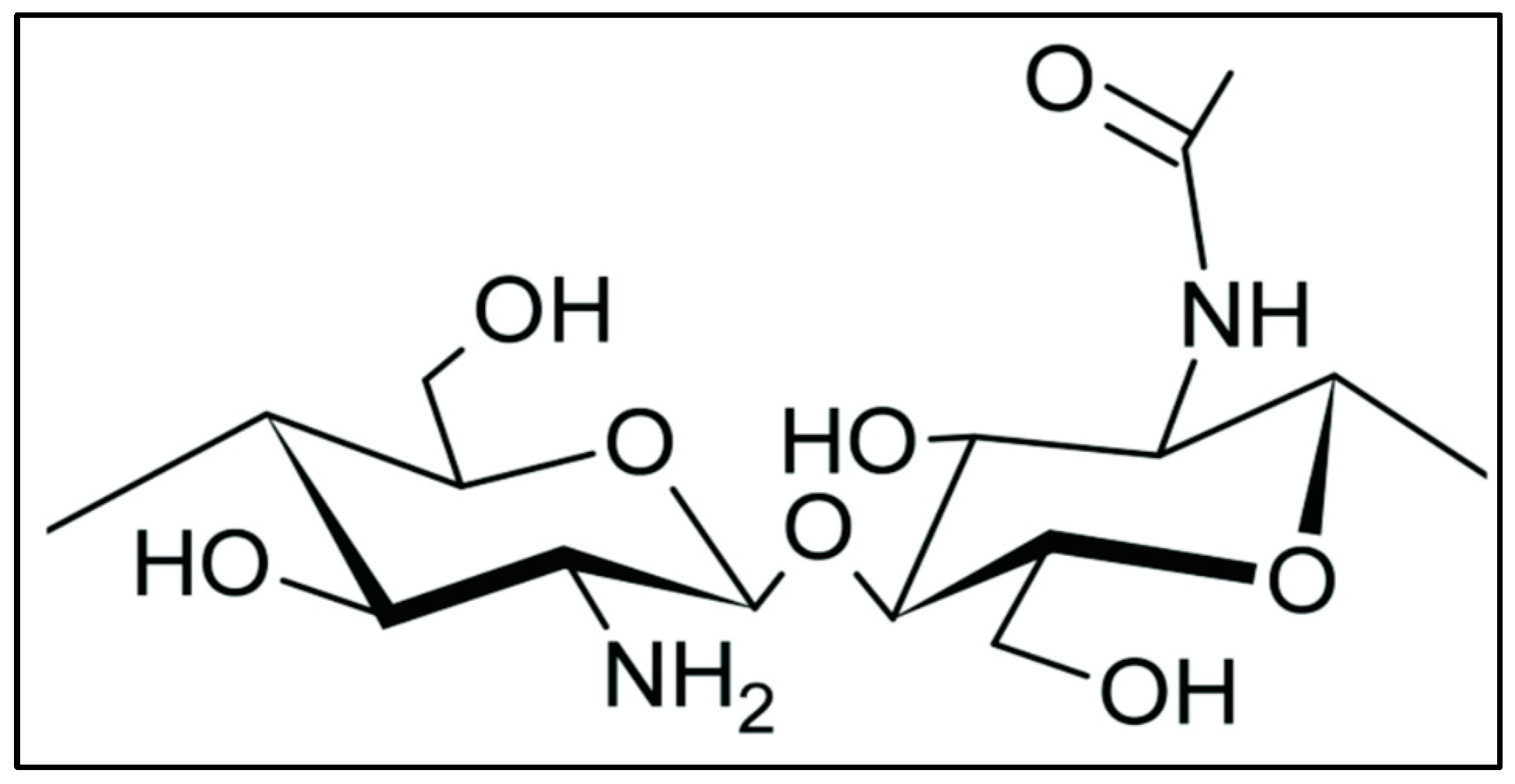

| Derivatives | Importance | Modification | Characteristics | Application | References |
|---|---|---|---|---|---|
| Quaternised chitosan | The mucoadhesive characteristics increase proportionally with the degree of quaternisation. This is similar to the enhanced cationic nature of chitosan, which influences its interaction with negatively charged mucin, resulting in mucoadhesion. | 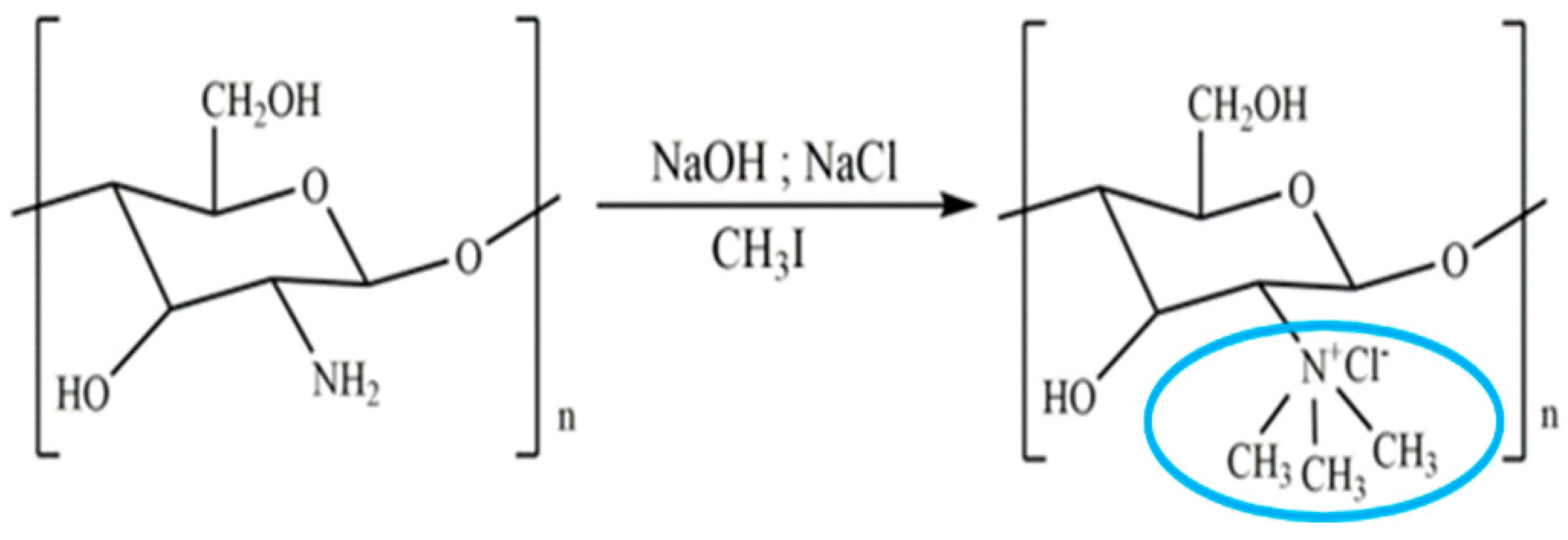 | It helps various drugs to pass mucosal barrier and improves the water solubility and antioxidant activity. | Nasal vaccine adjuvant and antimicrobial agent. | [23,31,32] |
| Alkyl chitosan | It is proposed that the increased transfection efficiency of alkylated chitosan is due to increased cell entry facilitated by hydrophobic interactions, as well as easier unpacking of DNA from alkylated chitosan carriers due to the weakening of electrostatic attractions between DNA and alkylated chitosan. |  | Improves transfection efficiency and forms clots with blood to improve coagulation efficiency. | Gene delivery and haemostatic dressing. | [23,33,34] |
| Highly cationic derivatives | Highly cationic derivatives of chitosan have been prepared, and their cationic nature is essential to many of their applications, including bio-adhesion, absorption enhancement, transfection efficiency, and biological activities like antitumour, antimicrobial, anti-inflammatory, and anti-hypercholesterolemic effects. | 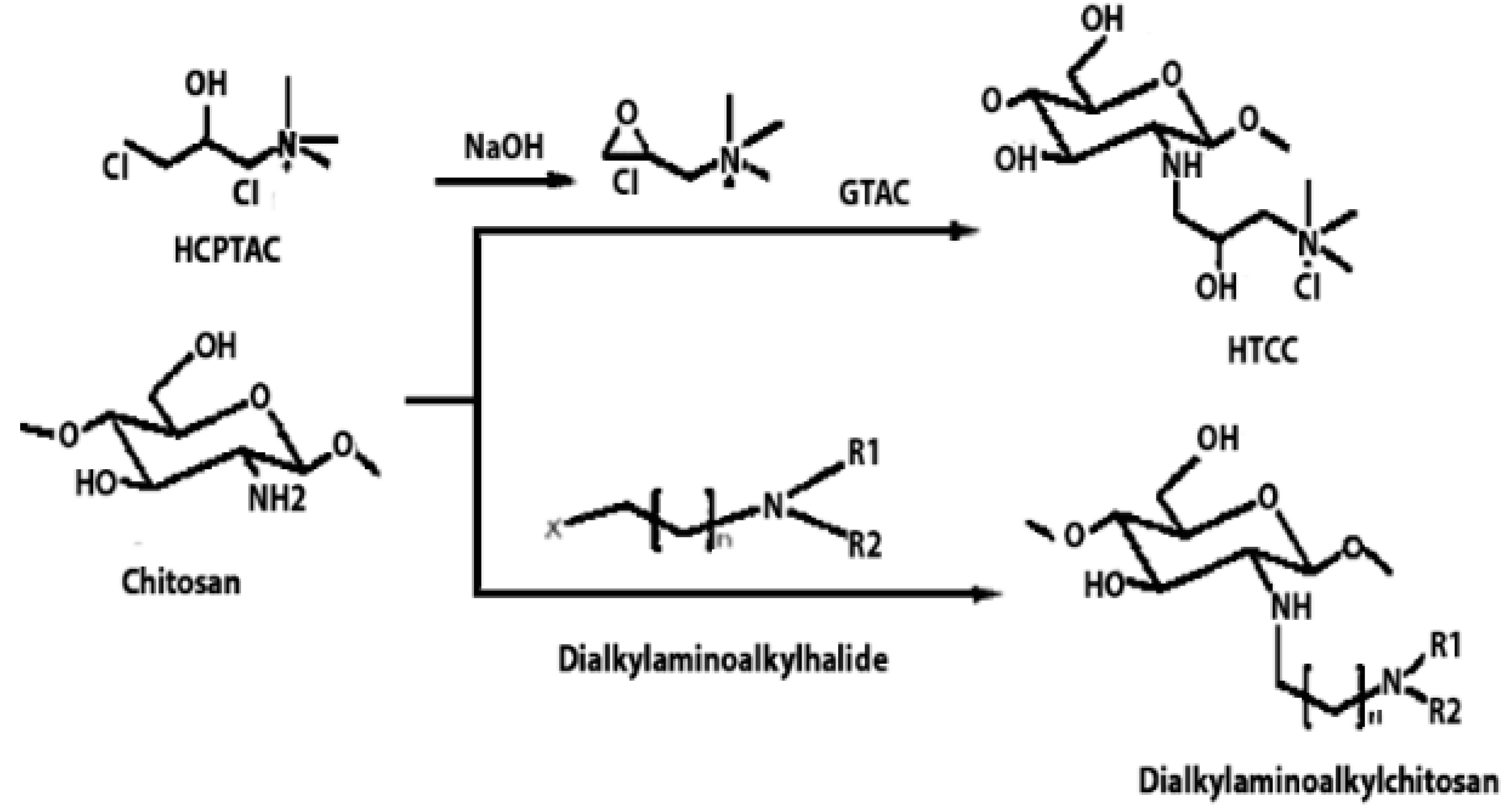 | Enhanced water solubility, mucoadhesive properties, and improved interaction with negatively charged cell membranes. | Tissue regeneration and wound healing. | [35,36] |
| Hydroxyalkyl chitosan | Hydroxyethyl chitosan has great biocompatibility and biodegradability and is suitable for medicinal applications. It also has good bacteriostatic and hygroscopic moisturising properties and may be used as a natural textile softening and finishing agent. |  | Improved mechanical strength and enhanced antibacterial activity and bioavailability. | Drug delivery carrier. | [37,38,39] |
| Carboxyalkyl chitosan | Carboxymethyl chitosan is a water-soluble chitosan derivative with antibacterial, anticancer, antitumour, antifungal, and antioxidant characteristics that is employed in both drug administration and enzyme delivery. | 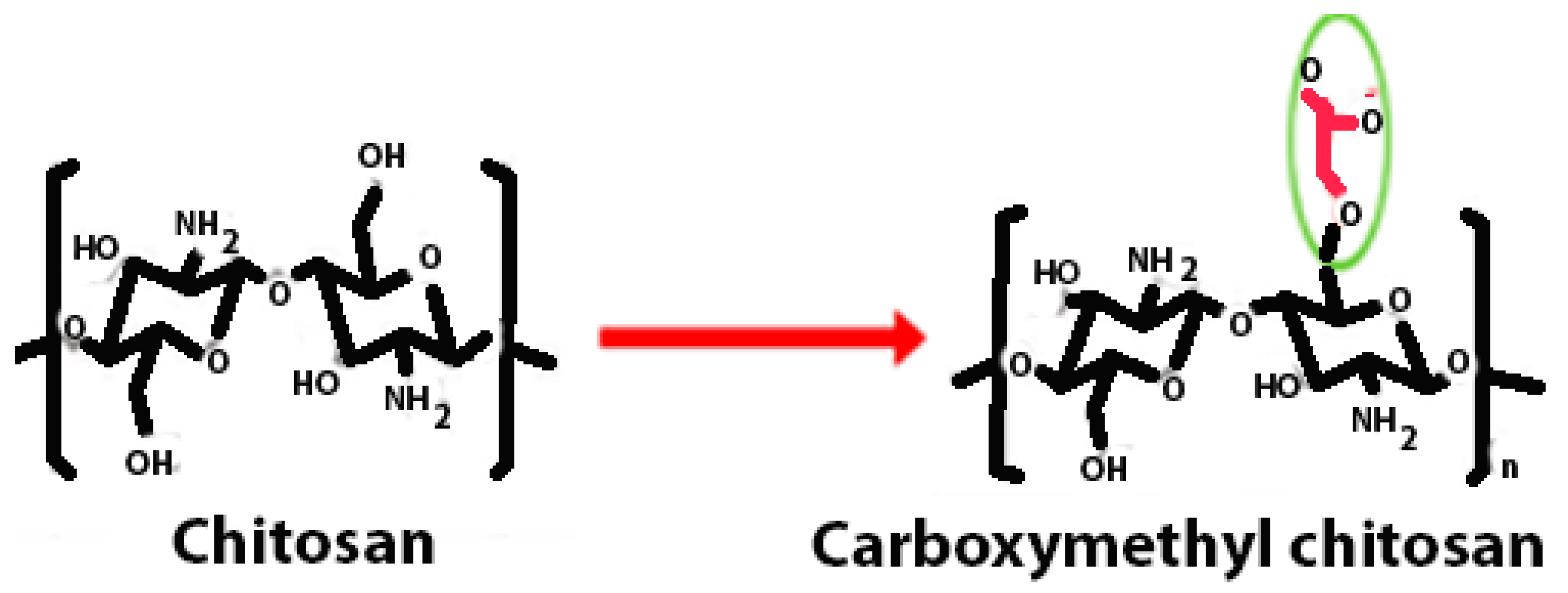 | Improved water solubility and DNA binding capacity. | DNA delivery and site-specific protein delivery. | [40,41,42] |
| N-Acyl chitosan | N-acylated chitosan improves water solubility. | 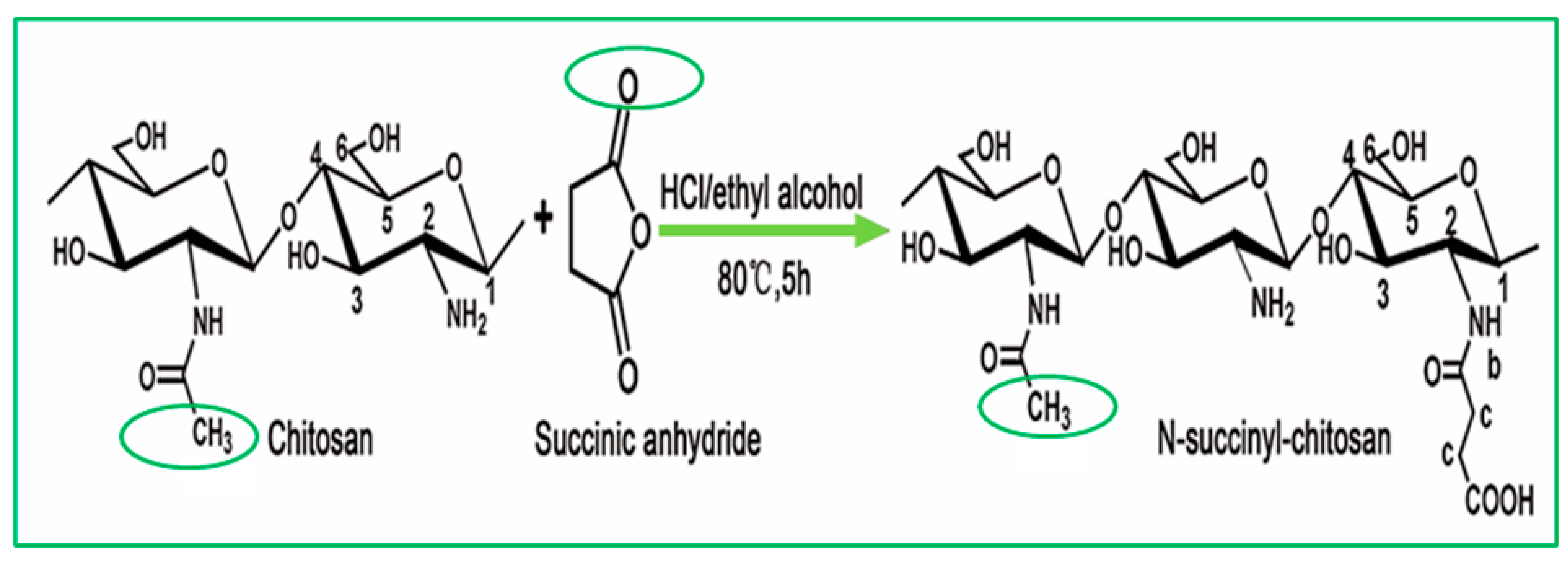 | Improved encapsulation, stability, and adhesion. | Drug carrier. | [43,44] |
| O-Acyl chitosan | It improves its fat solubility and hydrophobicity. |  | Improved adhesion and antibacterial activity. | Antimicrobial agent. | [45,46] |
| Thiolated chitosan | Thiolated chitosan offers various advantages over native chitosan, including increased solubility at low degrees of substitution and mucoadhesive and cellular penetration capabilities. | 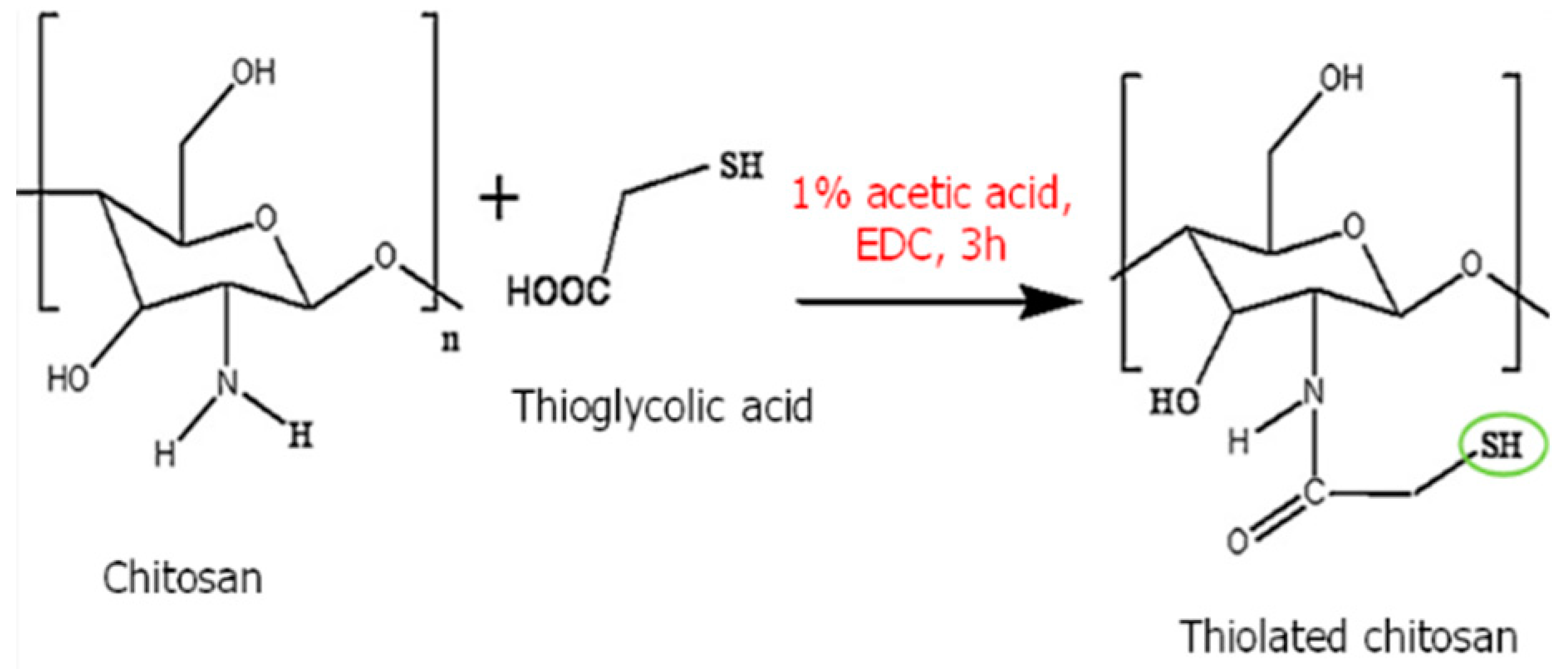 | Increases the solubility and extends the release of drugs. | Drug carrier. | [47,48,49] |
| Azidated chitosan | Enhancement of mechanical stability. | 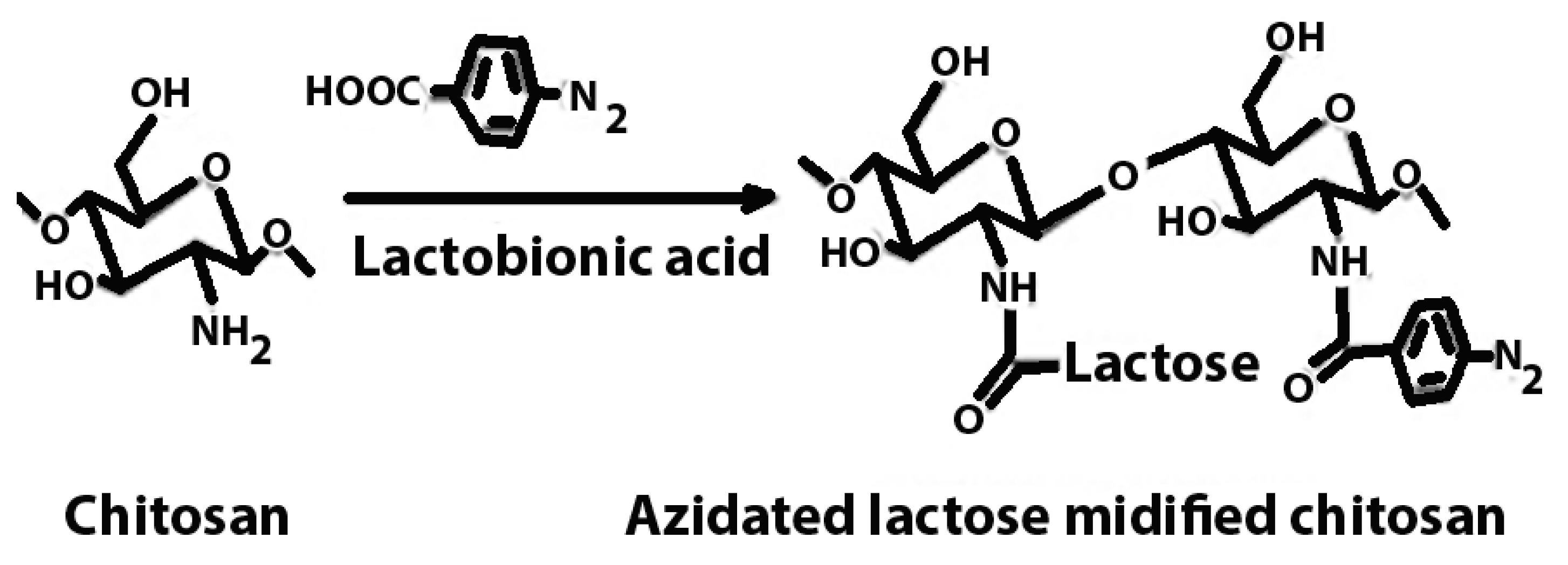 | Enhanced mechanical strength, prolonged drug release kinetics, and targeted delivery. | Tissue engineering scaffolds in regenerative medicine. | [50,51] |
| Phosphorylated chitosan | Phosphorylated chitosan (PC), a water-soluble chitosan derivative, has various beneficial wound healing characteristics, including haemostatic capabilities, metal chelating capacity, antioxidant, anti-inflammatory, antibacterial, and angiogenic activity. |  | Promoting proliferation and osteogenic differentiation of MC3T3-E1s. | Bone tissue engineering. | [49,52] |
| Chitosan Derivatives | Method of Preparation | Mechanical Properties | Special Comments | References |
|---|---|---|---|---|
| Trimethyl chitosan | Trimethylation | Mucoadhesion, tensile strength flexibility, and biodegradability. | Trimethyl chitosan tends to possess enhanced mucoadhesive properties compared to its unmodified counterpart, facilitating prolonged contact with tissue surfaces and providing a stable platform for drug delivery or tissue regeneration. Moreover, its increased water solubility and cationic charge may contribute to improved interaction with biological matrices, potentially enhancing cell adhesion, migration, and proliferation. | [126,127] |
| Carboxymethyl chitosan | Reductive or direct alkylation | Carboxymethyl chitosan combines the inherent biocompatibility and biodegradability of chitosan with the improved solubility and versatility conferred by carboxymethylation. | Carboxymethyl chitosan’s mechanical properties play a crucial role in facilitating the healing process. Its flexibility allows it to conform to the irregular contours of wounds, ensuring close contact with the wound bed and promoting a conducive environment for tissue regeneration. Chitosan’s adhesive properties enable it to adhere firmly to the wound site, preventing the displacement of dressings and promoting cell attachment and migration, which are essential steps in tissue repair. | [128,129] |
| Thiolated chitosan | Thiolation | Improved mechanical strength and elasticity compared to unmodified chitosan. | The modified form of chitosan, which incorporates thiol (sulfhydryl) groups, exhibits enhanced mucoadhesive properties, allowing for prolonged residence at the wound site or on mucosal surfaces. | [130,131] |
| Acylated chitosan | Acylation | Enhanced tensile strength, flexibility, and adhesion properties. | The mechanical stability of acylated chitosan materials ensures prolonged support to the healing tissue and promotes cell migration, proliferation, and tissue regeneration. | [34,132,133] |
| Alkylated chitosan | Alkylation | Enhance material’s flexibility, tensile strength, and elasticity. | The alkylated chitosan’s improved mechanical properties enable it to conform to wound contours, withstand mechanical stresses, and provide structural support during tissue regeneration processes. | [134,135] |
| Crosslinked chitosan | Condensation | Improved tensile strength, elasticity, and durability. | These enhanced mechanical properties enable crosslinked chitosan to withstand the dynamic mechanical stresses encountered in wound environments and tissue engineering constructs, providing stable support for tissue regeneration processes. | [136,137] |
| Glycol chitosan | Conjugation | Enhanced flexibility, biocompatibility, and biodegradability. | Glycol chitosan’s mechanical properties play a critical role in providing structural support to wound dressings and tissue scaffolds, facilitating cell adhesion, migration, and proliferation in the wound site or tissue defect. | [138,139] |
| Sulfoethyl chitosan | Alkylation | Enhanced adhesion, biocompatibility, biodegradability, and flexibility. | This derivative possesses enhanced mucoadhesive properties compared to unmodified chitosan, allowing for prolonged residence at mucosal surfaces, such as those found in wounds or damaged tissues. Its mechanical strength, flexibility, and adhesion properties enable it to conform to irregular wound contours, providing a protective barrier and promoting cell attachment, proliferation, and tissue regeneration. | [140,141,142] |
| Ref. | Key Materials | Methodology | Preclinical Findings | Clinical Relevance | Bacterial Strains Tested | Antibacterial Mechanism | Safety and Biocompatibility | Future Research Directions |
|---|---|---|---|---|---|---|---|---|
| [145] | Chitosan-alginate and gentamicin | Electrospinning and in vitro and in vivo evaluations | Improved antibacterial activity, enhanced skin regeneration in Balb/C mice, and promotion of collagen deposition and formation of new blood vessels and hair follicles in treated wounds. | Potential use in preventing infections in wounds, specifically in skin regeneration and scaffolding for tissue engineering applications. | Staphylococcus aureus and Escherichia coli | Gentamicin inhibits protein formation, disrupts nucleic acid reproduction, and causes bacterial cell membrane rupture. | Demonstrated minimal cytotoxicity in L929 cell lines, supporting safe topical application. | Recommended studies on long-term effects, comparison with other antibacterial agents, and scaling up for clinical trials. |
| [146] | Chitosan, Ag nanoparticles, and LiOH/urea | Synthesis using LiOH/urea, characterisation (FTIR, XRD, SEM, TEM), mechanical testing, swelling studies, antibacterial activity studies, and wound healing studies in Sprague Dawley rats | Hydrogels showed ultrahigh mechanical properties with significant enhancement in compressive strength (15.95 ± 1.95 MPa). Demonstrated excellent antibacterial performance and accelerated wound healing with increased re-epithelialisation and collagen deposition compared to controls. | Could be used as an advanced wound dressing for skin regeneration, especially in scenarios requiring robust mechanical properties and high antibacterial activity. | Staphylococcus aureus and Escherichia coli | Silver nanoparticles disrupt bacterial membranes and prevent DNA replication, enhancing the antibacterial efficacy of the hydrogel. | Minimal cytotoxicity indicated in preclinical tests, suggesting good biocompatibility for topical application. | Further studies suggested for long-term effects and potential scaling up for clinical trials. |
| [147] | Chitosan, pectin, and TiO2 nanoparticles | Synthesis of nano-dressing, characterisation (FTIR, TGA, DSC, SEM, TEM), mechanical testing, antibacterial activity, and in vivo wound healing in albino rats | Nano-dressing enhanced wound healing evidenced by increased re-epithelialisation and collagen deposition. Showed good antibacterial properties and mechanical strength. | Potential for use as an advanced wound dressing material in medical settings due to its enhanced biocompatibility and effective wound healing capabilities. | Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, Bacillus subtilis, and Aspergillus niger | TiO2 nanoparticles disrupt microbial cell membranes, enhancing antibacterial activity. | Demonstrated minimal cytotoxicity and favourable blood compatibility, indicating safety for topical application. | Further studies recommended on long-term effects and evaluation in clinical settings. |
| [148] | PVA, chitosan, DMAEMA, and 1-bromobutane | Freeze-drying, crosslinking with glutaraldehyde vapor, FTIR, SEM, mechanical testing, antibacterial activity studies, and cytotoxicity and haemolysis tests | The prepared sponges showed improved water absorption (up to 2300%) and flexibility, significant antibacterial activity of nearly 100% against E. coli and S. aureus, excellent haemocompatibility, and cytocompatibility. | Ideal for advanced wound dressing applications due to its high porosity, enhanced mechanical properties, and effective antibacterial capabilities. | Escherichia coli and Staphylococcus aureus | Quaternary ammonium compounds in the sponge disrupt bacterial cell membranes, leading to high antibacterial efficacy. | Showed minimal haemolysis and cytotoxicity, indicating good compatibility for clinical use. | Further research could explore long-term clinical applications and effectiveness in diverse microbial environments. |
| [149] | Chitosan (CS) and S-nitrosoglutathione (GSNO) | Preparation of NO-releasing chitosan films (CS/NO), characterisation (FTIR, SEM, mechanical testing), antibacterial activity studies, and in vivo wound healing in rats | CS/NO films showed enhanced wound healing compared to CS films alone, with increased re-epithelialisation and improved histopathological outcomes. Strong antibacterial activity against Pseudomonas aeruginosa and Staphylococcus aureus. | Potential use as an advanced wound dressing for treating full-thickness wounds with enhanced antibacterial properties to prevent infections and promote wound healing. | Pseudomonas aeruginosa and Staphylococcus aureus | NO release from GSNO disrupts bacterial cell functions and structures, leading to antibacterial effects. | No adverse effects reported in in vivo studies, indicating good biocompatibility for topical application. | Suggest further studies on the long-term effects of CS/NO films and their clinical applications in human subjects. |
| [150] | Chitosan (CS), Dialdehyde Cellulose Nanocrystals (DCNCs), and silver nanoparticles (AgNPs) | Synthesis of AgNPs using periodate oxidation of CNC, forming of wound dressings via solution casting method, mechanical and antibacterial testing, and cytotoxicity assays | Enhanced mechanical properties and antibacterial activity of wound dressings; good cytocompatibility indicated by low cytotoxicity to NIH3T3 cells. | Potential for clinical application as advanced wound dressing materials due to enhanced durability, efficacy against infections, and safety. | Gram-positive and gram-negative bacteria and fungi including Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, Enterobacter cloacae, Streptococcus pneumoniae, Pseudomonas aeruginosa, Candida albicans, Candida glabrata, and Candida krusei | AgNPs provide high antibacterial activity through interactions with microbial membranes, leading to cell damage. | Demonstrated minimal cytotoxicity and favourable biocompatibility for clinical use, supporting safe topical application. | Further research to explore long-term clinical effectiveness and potential for commercial scaling is suggested. |
| [151] | Chitosan (CS), Polyvinyl Alcohol (PVA), Polyvinylpyrrolidone (PVP), and Hexamethylene 1,6-di(aminocarboxysulfonate) (HMDACS) | Preparation of composite films using solvent casting method, characterisation (FTIR, SEM), mechanical testing, and antibacterial activity studies | Composite films showed improved mechanical properties, significant antibacterial activity, and optimal hydrophilicity for wound contact. | Potential for use as advanced wound dressings, particularly in settings requiring robust mechanical properties and effective antibacterial protection. | Escherichia coli and Staphylococcus aureus | Antibacterial activity attributed to the structured network of chitosan and its interaction with bacterial membranes, enhanced by PVA and PVP. | Demonstrated safety and minimal cytotoxicity, indicating suitability for clinical use in wound care. | Further research on long-term clinical performance and scalability of the production process. |
| [152] | Chitosan and nano-ZnO (nZnO) | Preparation of chitosan hydrogel/nZnO composite bandages, characterisation (FT-IR, XRD, SEM), and in vitro and in vivo evaluation | Composite bandages enhanced wound healing, showing faster re-epithelialisation and collagen deposition in vivo in Sprague Dawley rats. Demonstrated good swelling, cytocompatibility, and cell infiltration properties. | Promising for burn wounds, chronic wounds, and diabetic foot ulcers due to enhanced mechanical strength, flexibility, and significant antibacterial properties. | Staphylococcus aureus and Escherichia coli | Nano-ZnO provides antibacterial activity by damaging microbial cell walls and preventing microbial colonisation. | Showed minimal cytotoxicity and good biocompatibility, supporting its safety for clinical applications. | Explore long-term clinical effectiveness and potential customisation for specific wound care needs. |
| [153] | Chitosan and various additives for density and porosity control | Development and testing of chitosan-based wound coatings on male Wistar–Kyoto rats and rabbits, with evaluations including mechanical properties, biodegradation, and in vivo wound healing effectiveness | Coatings showed effective framework function, proper drainage, and supported rapid healing processes in purulent wounds. Specific coatings recommended based on wound type (purulent vs. granulating). | Promising for use in clinical settings for treating various types of wounds, particularly purulent wounds requiring robust and effective antibacterial barriers. | Staphylococcus aureus | Chitosan’s natural antibacterial properties enhanced by structural modifications to optimise contact with pathogens. | Demonstrated good biocompatibility with no adverse effects reported in animal tests, supporting potential for safe clinical use. | Further clinical trials to validate efficacy in human subjects and explore broader applications in wound care. |
| [154] | Chitosan and 3D printing technology | Preparation of 3D-printed chitosan scaffolds, in vitro cell growth studies, and in vivo wound healing tests on diabetic rats | Enhanced wound healing in diabetic rats; improved cellular adhesion and proliferation in vitro. | Potential application in diabetic wound management; showcases the effectiveness of 3D-printed scaffolds in regenerative medicine. | Not specified in the manuscript | N/A | Demonstrated biocompatibility with human skin cells, supporting safe application in medical treatments. | Suggest further studies to assess long-term effects and potential clinical applications in human diabetic wound care. |
| [155] | Chitosan, hyaluronic acid, and zinc oxide nanoparticles (ZnO NPs) | Fabrication of chitosan-based patches using solvent casting, characterisation (SEM, FTIR), antibacterial testing, haemostatic evaluation, and in vivo wound healing in rats | Patches showed excellent haemostatic properties and enhanced antibacterial activity and promoted faster wound healing in rat models. | Suitable for advanced wound care, particularly in managing bleeding and preventing infection in wounds. | Staphylococcus aureus and Escherichia coli | Zinc oxide nanoparticles disrupt bacterial membranes, enhancing the antibacterial properties of the patch. | Demonstrated biocompatibility and non-toxicity in animal studies, indicating safety for potential clinical use. | Further development for clinical trials and exploration of its efficacy across various wound types and patient demographics. |
| S. No. | Carrier System | Tissue Type | In Vitro/In Vivo Tests | References |
|---|---|---|---|---|
| 1. | Hydrogels | Neural tissue | PC 12 cell in vitro | [166] |
| 2. | Nanocomposite matrix | Bone tissue | MG-63 cell line | [167] |
| 3. | Injectable hydrogels | Bone tissue | MG-63 cell line | [168] |
| 4. | Hydrogels | Cartilage tissue | - | [169] |
| 5. | Scaffolds | Neural tissue | Rat pheochromocytoma (PC12) | [170] |
| 6 | Hydrogel composite scaffolds | Meniscus tissue | Rabbit mesenchymal stem cells | [171] |
| 7. | Nanocomposite scaffolds | Skin tissue | Attenuated total reflectance–Fourier-transform infrared spectroscopy (ATR-FTIR) | [172] |
| 8. | Three-dimensional (3D)-printed hydrogel scaffolds | Cartilage tissue | ATCD5 cells | [173] |
| 9. | Injectable chitosan hydrogel | Cartilage tissue | The chondrogenic effect of bone mesenchymal stem cells (BMSCs) within the chitosan hydrogel was also assessed in vitro | [174] |
| 10. | 3D-printed scaffold | Bone tissue | MG-63 cell line | [175] |
| 11. | Fibrous scaffolds | Bone tissue | The in vitro study showed that scaffolds fabricated at 90 °C promoted better MG63 cell attachment, proliferation, and differentiation | [176] |
| S. No. | Carrier System | Active Compound | Size | Entrapment Efficiency | Key Findings | References |
|---|---|---|---|---|---|---|
| 1. | Nanoparticles | Docetaxel | 250.3 ± 1.7 nm | 85% | The NP components were non-toxic and safe to human cells. The prepared nanoparticles may be used as effective carriers for chemotherapeutic agents targeting carcinogenetic tissues. | [187] |
| 2. | Nanoparticles | Isolongifolene | 200–250 nm | 79.05 ± 4.60% | Isolongifolene-loaded chitosan nanoparticles were shown to be plasma-compatible and to have a consistent release pattern. As a result, chitosan-loaded nanoparticles might be used as an effective adjuvant in cancer therapy to address multi-drug resistance in solid tumours. | [188] |
| 3. | Nanoparticles | Methotrexate | 73.2 ± 4.9 nm | - | Methotrexate-loaded nanoparticles inhibited the viability of breast cancer cells when exposed to low doses. The tiny size of this efficient cytotoxic nanoparticle delivery method makes it a promising technique for use in breast cancer. | [189] |
| 4. | Nanofibre | Cisplatin | 200–350 nm | - | The prepared nanofibres are biocompatible and effective in cancer treatment. | [190] |
| 5. | Nanoparticles | Docetaxel and curcumin | 100–150 nm | 7.82% and 6.48 | The prepared nanoparticle loaded with curcumin and docetaxel can ameliorate the immunosuppressive microenvironment to promote the inhibition of tumour growth. | [191] |
| 6. | Nanoparticles | Quercetin | 291.1 ± 9.1 | 11.17 ± 1.12% | The prepared nanoparticles decrease tumour growth and provide a prospective strategy for the treatment of paclitaxel-resistant lung cancer. | [192] |
| 7. | Nanoparticles | Doxorubicin | 27.31 ± 0.78 nm | 74.40 ± 1.60% | At pH 5.0, the drug was promptly and totally liberated from the nanoparticles. The anti-HER2 conjugated OCP copolymer nanoparticles had the lowest IC50 in vitro, indicating a boost in the therapeutic effectiveness of DOX to treat human breast cancer. | [193] |
| 8. | Hydrogel nanocomposite | Curcumin | 30 nm | 62% | The proposed nanostructure is a promising vehicle with the ability to improve curcumin loading and enable prolonged curcumin release while causing considerable cytotoxicity in MCF-7 cells. | [194] |
| 9. | Hybrid nanogel containing gold nanoparticles | Doxorubicin | 119.3 nm | 56% | The present research demonstrated that the designed enzyme-responsive nanogel could potentially target various solid tumours both actively and passively. | [195] |
| 10. | Nanoparticles | Doxorubicin and Cisplatin | 160 nm | 21.4% for DOX and 81.71 for CP | In human breast cancer MCF-7 cells, DOX plus cisplatin had a synergistic cell-killing impact. The findings clearly show that the innovative nanoplatform holds great promise for synergistic combination treatment of breast cancer. | [196] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mawazi, S.M.; Kumar, M.; Ahmad, N.; Ge, Y.; Mahmood, S. Recent Applications of Chitosan and Its Derivatives in Antibacterial, Anticancer, Wound Healing, and Tissue Engineering Fields. Polymers 2024, 16, 1351. https://doi.org/10.3390/polym16101351
Mawazi SM, Kumar M, Ahmad N, Ge Y, Mahmood S. Recent Applications of Chitosan and Its Derivatives in Antibacterial, Anticancer, Wound Healing, and Tissue Engineering Fields. Polymers. 2024; 16(10):1351. https://doi.org/10.3390/polym16101351
Chicago/Turabian StyleMawazi, Saeid Mezail, Mohit Kumar, Noraini Ahmad, Yi Ge, and Syed Mahmood. 2024. "Recent Applications of Chitosan and Its Derivatives in Antibacterial, Anticancer, Wound Healing, and Tissue Engineering Fields" Polymers 16, no. 10: 1351. https://doi.org/10.3390/polym16101351
APA StyleMawazi, S. M., Kumar, M., Ahmad, N., Ge, Y., & Mahmood, S. (2024). Recent Applications of Chitosan and Its Derivatives in Antibacterial, Anticancer, Wound Healing, and Tissue Engineering Fields. Polymers, 16(10), 1351. https://doi.org/10.3390/polym16101351







