NMR and GPC Analysis of Alkyd Resins: Influence of Synthesis Method, Vegetable Oil and Polyol Content
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. NMR Spectroscopy
2.3. Gel Permeation Chromatography
3. Results and Discussion
3.1. 1D 1H-NMR Spectra
3.2. 1D 13C-NMR Spectra
3.3. 2D NMR Spectra
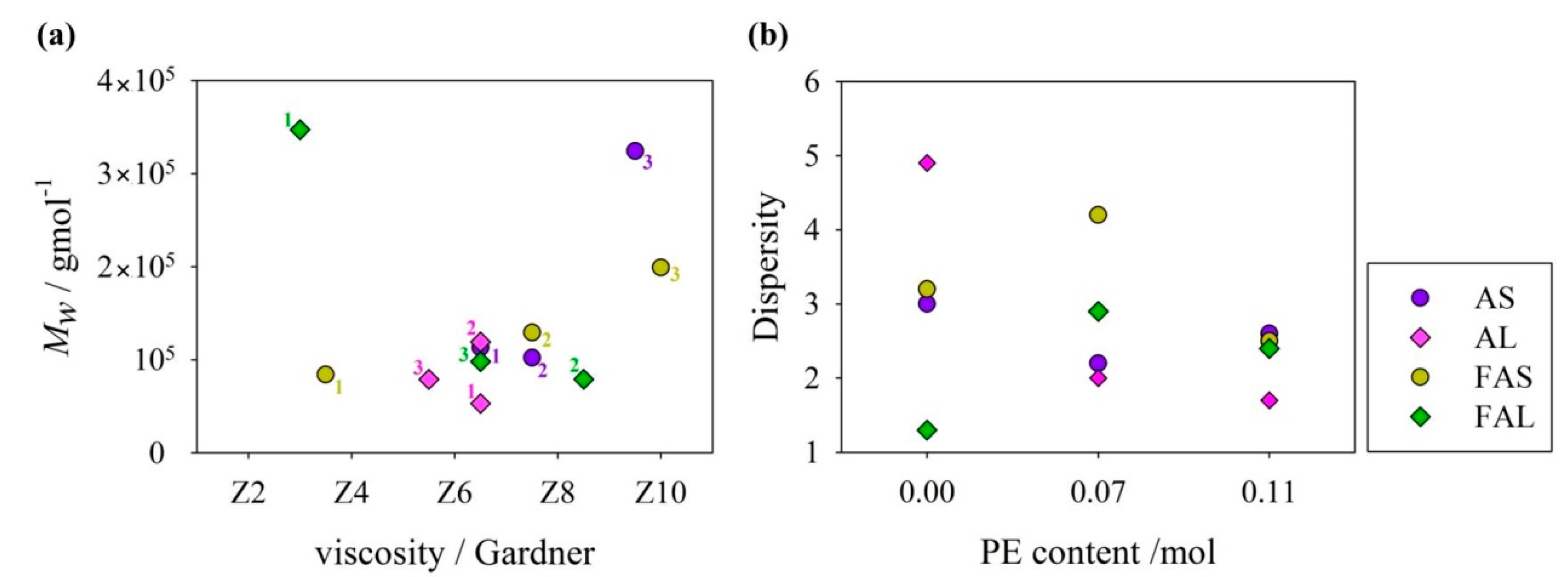
3.4. GPC Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bouwman, E.; Van Gorkum, R. A study of new manganese complexes as potential driers for alkyd paints. J. Coat. Technol. Res. 2007, 4, 491–503. [Google Scholar] [CrossRef]
- Simpson, N.; Maaijen, K.; Roelofsen, Y.; Hage, R. The Evolution of Catalysis for Alkyd Coatings: Responding to Impending Cobalt Reclassification with Very Active Iron and Manganese Catalysts, Using Polydentate Nitrogen Donor Ligands. Catalysts 2019, 9, 825. [Google Scholar] [CrossRef]
- Dizman, C.; Ozman, E. Preparation of rapid (chain-stopped) alkyds by incorporation of gum rosin and investigation of coating properties. Turk. J. Chem. 2020, 44, 932–940. [Google Scholar] [CrossRef] [PubMed]
- Al-Zahrani, F.A.M.; Lin, L.; El-Wahab, H.A. Preparation and evaluation of high–performance modified alkyd resins based on and study of their anticorrosive properties for surface coating applications. E-Polymers 2022, 22, 781–792. [Google Scholar] [CrossRef]
- Ploeger, R.; Scalarone, D.; Chiantore, O. The characterization of commercial artists’ alkyd paints. J. Cult. Herit. 2008, 9, 412–419. [Google Scholar] [CrossRef]
- Bellatin Arciniega, L.; Meza Yapu, R.; Obregón Valencia, D.; Hadzich, A.; Costa, M.A.; Ispas, A.; Bund, A.; Flores, S. Alkyds with artistic applications based on drying oils, multifunctional polyalcohols and different polybasic acids. J. Appl. Polym. Sci. 2023, 140, e53746. [Google Scholar] [CrossRef]
- Khorasani, S.N.; Ataei, S.; Neisiany, R.E. Microencapsulation of a coconut oil-based alkyd resin into poly (melamine-urea-formaldehyde) as shell for self-healing purposes. Prog. Org. Coat. 2017, 111, 99–106. [Google Scholar] [CrossRef]
- Çömlekçi, G.K.; Ulutan, S. Encapsulation of linseed oil and linseed oil based alkyd resin by urea formaldehyde shell for self-healing systems. Prog. Org. Coat. 2018, 121, 190–200. [Google Scholar] [CrossRef]
- Taylor, S.R. Coatings for corrosion protection: An overview. In Encyclopedia of Materials: Science and Technology, 2nd ed.; Buschow, K.H.J., Cahn, R.W., Flemings, M.C., Ilschner, B., Kramer, E.J., Mahajan, S., Veyssière, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; pp. 1263–1269. [Google Scholar] [CrossRef]
- Van Haveren, J.; Oostveen, E.A.; Miccichè, F.; Noordover, B.A.J.; Koning, C.E.; van Benthem, R.A.T.M.; Frissen, A.E.; Weijnen, J.G.J. Resins and additives for powder coatings and alkyd paints, based on renewable resources. J. Coat. Technol. Res. 2007, 4, 177–186. [Google Scholar] [CrossRef]
- Hofland, A. Alkyd resins: From down and out to alive and kicking. Prog. Org. Coat. 2012, 73, 274–282. [Google Scholar] [CrossRef]
- Spasojević, P.M.; Panić, V.V.; Džunuzović, J.V.; Marinković, A.D.; Woortman, A.J.J.; Loos, K.; Popović, I.G. High performance alkyd resins synthesized from postconsumer PET bottles. RSC Adv. 2015, 5, 62273–62283. [Google Scholar] [CrossRef]
- Tuck, N. Raw Materials Used for Alkyd Preparation. In Waterborne and Solvent Based Alkyds and Their End User Applications; Sita Technology Limited: Edinburgh, UK, 2000; Volume 6, pp. 19–47. [Google Scholar]
- Vallejo, P.P.; López, B.L.; Murillo, E.A. Hyperbranched phenolic-alkyd resins with high solid content. Prog. Org. Coat. 2015, 87, 213–221. [Google Scholar] [CrossRef]
- Prashantha, M.A.B.; Premachandra, B.A.J.K.; Amarasinghe, A.D.U.S. Synthesis of fast drying long oil alkyd resins using seed oil of Karawila (Momordica charantia). Indian J. Chem. Technol. 2017, 24, 47–54. [Google Scholar]
- Hadzich, A.; Gross, G.A.; Leimbach, M.; Ispas, A.; Bund, A.; Flores, S. Characterization of Plukenetia volubilis L. fatty acid-based alkyd resins. Pol. Test. 2020, 82, 106296. [Google Scholar] [CrossRef]
- Hadzich, A.; Gross, G.A.; Leimbach, M.; Ispas, A.; Bund, A.; Flores, S. Effect of polyalcohols on the anticorrosive behaviour of alkyd coatings prepared with drying oils. Prog. Org. Coat. 2020, 145, 105671. [Google Scholar] [CrossRef]
- Nanvaee, A.A.; Yahya, R.; Gan, S.N. Alkyd resins are still of major important binders in organic coatings. In Proceedings of the Malaysia Polymer International Conference (MPIC 2009), Kuala Lumpur, Malaysia, 21–22 October 2009; pp. 65–69. [Google Scholar]
- Onukwli, O.D.; Igbokwe, P.K. Production and Characterization of Castor Oil-Modified Alkyd Resins. J. Eng. Appl. Sci. 2008, 3, 161–165. [Google Scholar]
- Alam, M.; Alandis, N.M. Development of Ambient Cured Polyesteramide Coatings from Linseed Oil: A Sustainable Resource. J. Polym. Environ. 2011, 19, 391–397. [Google Scholar] [CrossRef]
- Boruah, M.; Gogoi, P.; Adhikari, B.; Dolui, S.K. Preparation and characterization of Jatropha Curcas oil based alkyd resin suitable for surface coating. Prog. Org. Coat. 2012, 74, 596–602. [Google Scholar] [CrossRef]
- Maurer, N.E.; Hatta-Sakoda, B.; Pascual-Chagman, G.; Rodriguez-Saona, L.E. Characterization and authentication of a novel vegetable source of omega-3 fatty acids, sacha inchi (Plukenetia volubilis L.) oil. Food Chem. 2012, 134, 1173–1180. [Google Scholar] [CrossRef]
- Popa, V.-M.; Gruia, A.; Raba, D.-N.; Dumbrava, D.; Moldovan, C.; Bordean, D.; Mateescu, C. Fatty acids composition and oil characteristics of linseed (Linum Usitatissimum L.) from Romania. J. Agroaliment. Process. Technol. 2012, 18, 136–140. [Google Scholar]
- Flores, S.; Flores, A.; Calderón, C.; Obregón, D. Synthesis and characterization of sacha inchi (Plukenetia volubilis L.) oil-based alkyd resin. Prog. Org. Coat. 2019, 136, 105289. [Google Scholar] [CrossRef]
- Obregón, D.; Toledo, C.; Hadzich, A.; Flores, S. Low viscosity alkyd resins based on trimethylolpropane and Peruvian oil. J. Polym. Res. 2021, 28, 203. [Google Scholar] [CrossRef]
- Obregón, D.; Hadzich, A.; Bellatin, L.; Flores, S. Microwave-assisted synthesis of alkyd resins using response surface methodology. Chem. Eng. Process. Process Intensif. 2023, 183, 109221. [Google Scholar] [CrossRef]
- Hadzich, A.; Flores, S. Physicochemical Characterization of Medium Alkyd Resins Prepared with a Mixture of Linum usitatissimum L. and Plukenetia volubilis L. Oils. IJCME 2019, 13, 319–322. [Google Scholar]
- Gómez, C.; Inciarte, H.; Orozco, L.M.; Cardona, S.; Villada, Y.; Rios, L. Interesterification and blending with Sacha Inchi oil as strategies to improve the drying properties of Castor Oil. Prog. Org. Coat. 2022, 162, 106572. [Google Scholar] [CrossRef]
- Spyros, A. Characterization of Unsaturated Polyester and Alkyd Resins Using One- and Two-Dimensional NMR Spectroscopy. J. Appl. Polym. Sci. 2003, 88, 1881–1888. [Google Scholar] [CrossRef]
- Assanvo, E.F.; Gogoi, P.; Dolui, S.K.; Baruah, S.D. Synthesis, characterization, and performance characteristics of alkyd resins based on Ricinodendron heudelotii oil and their blending with epoxy resins. Ind. Crops Prod. 2015, 65, 293–302. [Google Scholar] [CrossRef]
- Chiplunkar, P.P.; Pratap, A.P. Utilization of sunflower acid oil for synthesis of alkyd resin. Prog. Org. Coat. 2016, 93, 61–67. [Google Scholar] [CrossRef]
- Ibrahim, K.A.; Abu-sbeih, K.A.; Al-Trawneh, I.; Bourghli, L. Preparation and Characterization of Alkyd Resins of Jordan Valley Tomato Oil. J. Polym. Environ. 2014, 22, 553–558. [Google Scholar] [CrossRef]
- Rämänen, P.; Maunu, S.L. Structure of tall oil fatty acid-based alkyd resins and alkyd-acrylic copolymers studied by NMR spectroscopy. Prog. Org. Coat. 2014, 77, 361–368. [Google Scholar] [CrossRef]
- Glenn, A.; Jensen, A.T.; Machado, F. Salvia hispanica L. (Chia) Oil as a Potential Renewable Raw Material for the Production of Air-Dry Alkyd Resins. ACS Appl. Polym. Mater. 2021, 3, 6186–6197. [Google Scholar] [CrossRef]
- Aigbodion, A.I.; Pillai, C.K.S. Synthesis and molecular weight characterization of rubber seed oil-modified alkyd resins. J. Appl. Polym. Sci. 2001, 79, 2431–2438. [Google Scholar] [CrossRef]
- Mustafa, S.M.; Gan, S.N.; Yahya, R. Synthesis and Characterization of Novel Alkyds Derived From Palm Oil Based Polyester Resin. Asian J. Chem. 2013, 25, 8737–8740. [Google Scholar] [CrossRef]
- Yin, X.; Duan, H.; Wang, X.; Sun, L.; Sun, W.; Qi, H.; Ma, L. An investigation on synthesis of alkyd resin with sorbitol. Prog. Org. Coat. 2013, 77, 674–678. [Google Scholar] [CrossRef]
- Ifijen, I.H.; Maliki, M.; Odiachi, I.J.; Aghedo, O.N.; Ohiocheoya, E.B. Review on Solvents Based Alkyd Resins and Water Borne Alkyd Resins: Impacts of Modification on Their Coating Properties. Chem. Afr. 2022, 5, 211–225. [Google Scholar] [CrossRef]
- Odetoye, T.E.; Ogunniyi, D.S.; Olatunji, G.A. Studies on the preparation of Parinari polyandra benth seed oil alkyd resins. J. Appl. Polym. Sci. 2012, 127, 4610–4616. [Google Scholar] [CrossRef]
- Islam, M.R.; Beg, M.D.H.; Jamari, S.S. Development of vegetable-oil-based polymers. J. Appl. Polym. Sci. 2014, 131, 9016–9028. [Google Scholar] [CrossRef]
- Kadam, A.; Pawar, M.; Yemul, O.; Thamke, V.; Kodam, K. Biodegradable biobased epoxy resin from karanja oil. Polymer 2015, 72, 82–92. [Google Scholar] [CrossRef]
- Mańczyk, K.; Szewczyk, P. Highly branched high solids alkyd resins. Prog. Org. Coat. 2002, 44, 99–109. [Google Scholar] [CrossRef]
- Nosal, H.; Nowicki, J.; Warzała, M.; Semeniuk, I.; Sabura, E. Synthesis and characterization of alkyd resins based on Camelina sativa oil, glycerol and selected epoxidized vegetable oils as functional modifiers. Prog. Org. Coat. 2016, 101, 553–568. [Google Scholar] [CrossRef]
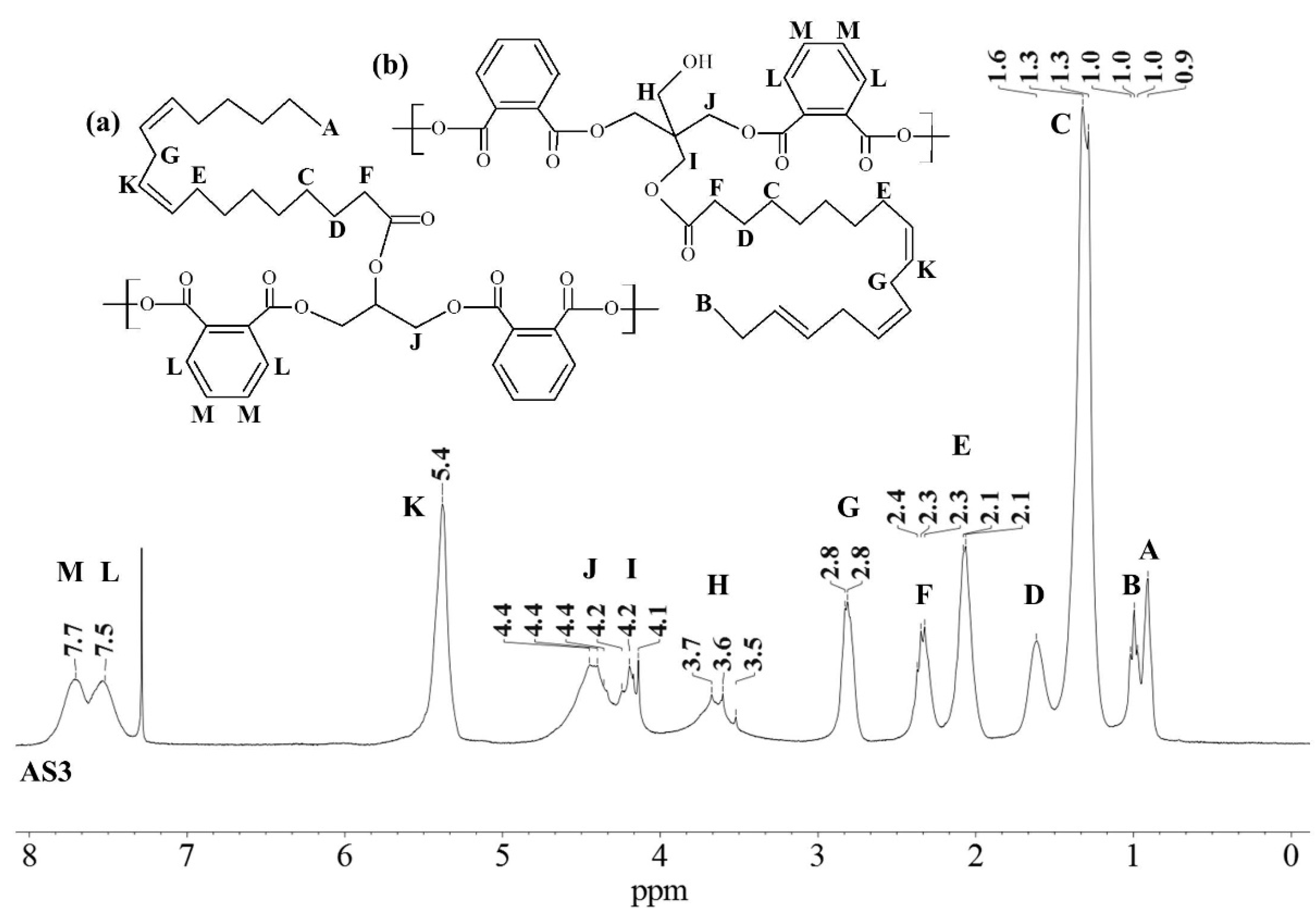






| Sample Code | Source | Polyol Ratio (GC:PE) | Fatty Acid Monoglyceride (FA) | Triglyceride, Oil (A) |
|---|---|---|---|---|
| AS1 | sacha inchi | 1:0 | x | |
| AS2 | sacha inchi | 0.5:0.5 | x | |
| AS3 | sacha inchi | 0.2:0.8 | x | |
| AL1 | linseed | 1:0 | x | |
| AL2 | linseed | 0.5:0.5 | x | |
| AL3 | linseed | 0.2:0.8 | x | |
| FAS1 | sacha inchi | 1:0 | x | |
| FAS2 | sacha inchi | 0.5:0.5 | x | |
| FAS3 | sacha inchi | 0.2:0.8 | x | |
| FAL1 | linseed | 1:0 | x | |
| FAL2 | linseed | 0.5:0.5 | x | |
| FAL3 | linseed | 0.2:0.8 | x |
| Peak | 1H Chemical Shift Range (ppm) | Group | ||||
|---|---|---|---|---|---|---|
| FAS3 | AS3 | AL3 | AS1 | AL1 | ||
| M | 7.91–7.63 | 7.88–7.63 | 7.97–7.64 | 7.96–7.65 | 7.99–7.65 | 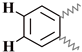 |
| L | 7.62–7.37 | 7.61–7.37 | 7.61–7.36 | 7.63–7.38 | 7.65–7.38 |  |
| K | 5.73–5.21 | 5.59–5.23 | 5.80–5.18 | 5.75–5.18 | 5.86–5.21 | 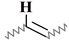 |
| J | 4.85–4.28 | 4.70–4.31 | 4.74–4.28 | 4.81–4.28 | 4.77–4.28 | 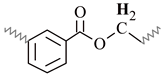 |
| I | 4.28–4.04 | 4.29–4.08 | 4.27–4.04 | 4.24–4.07 | 4.26–4.07 | 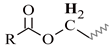 |
| H | 3.88–3.39 | 3.83–3.47 | 3.86–3.34 | 3.99–3.46 | 3.93–3.51 | 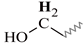 |
| G | 2.94–2.63 | 2.96–2.70 | 2.94–2.68 | 2.92–2.66 | 2.89–2.65 | 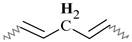 |
| F | 2.41–2.21 | 2.43–2.23 | 2.47–2.22 | 2.50–2.23 | 2.43–2.23 |  |
| E | 2.21–1.87 | 2.19–1.96 | 2.19–1.91 | 2.23–1.89 | 2.18–1.88 | 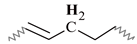 |
| D | 1.70–1.50 | 1.72–1.51 | 1.74–1.52 | 1.73–1.52 | 1.74–1.50 | 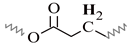 |
| C | 1.50–1.12 | 1.49–1.17 | 1.49–1.17 | 1.48–1.18 | 1.47–1.16 | 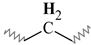 |
| B | 1.08–0.94 | 1.07–0.95 | 1.06–0.95 | 1.05–0.95 | 1.06–0.95 | 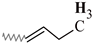 |
| A | 0.96–0.80 | 0.95–0.84 | 0.94–0.83 | 0.94–0.82 | 0.94–0.85 |  |
| Sample | (104 g/mol) | (104 g/mol) | Dispersity (Đ) |
|---|---|---|---|
| AS1 | 3.4 | 10.2 | 3.0 |
| AS2 | 5.1 | 11.3 | 2.2 |
| AS3 | 12.5 | 32.4 | 2.6 |
| AL1 | 2.5 | 11.9 | 4.9 |
| AL2 | 3.9 | 7.9 | 2.0 |
| AL3 | 3.1 | 5.3 | 1.7 |
| FAS1 | 2.6 | 8.4 | 3.2 |
| FAS2 | 3.1 | 12.9 | 4.2 |
| FAS3 | 8 | 19.9 | 2.5 |
| FAL1 | 26.9 | 34.7 | 1.3 |
| FAL2 | 2.7 | 7.9 | 2.9 |
| FAL3 | 4.1 | 9.8 | 2.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hadzich, A.; Flores, S.; Masucci, A.E.; Gomez, E.D.; Groß, G.A. NMR and GPC Analysis of Alkyd Resins: Influence of Synthesis Method, Vegetable Oil and Polyol Content. Polymers 2023, 15, 1993. https://doi.org/10.3390/polym15091993
Hadzich A, Flores S, Masucci AE, Gomez ED, Groß GA. NMR and GPC Analysis of Alkyd Resins: Influence of Synthesis Method, Vegetable Oil and Polyol Content. Polymers. 2023; 15(9):1993. https://doi.org/10.3390/polym15091993
Chicago/Turabian StyleHadzich, Antonella, Santiago Flores, Ashley E. Masucci, Enrique D. Gomez, and G. Alexander Groß. 2023. "NMR and GPC Analysis of Alkyd Resins: Influence of Synthesis Method, Vegetable Oil and Polyol Content" Polymers 15, no. 9: 1993. https://doi.org/10.3390/polym15091993
APA StyleHadzich, A., Flores, S., Masucci, A. E., Gomez, E. D., & Groß, G. A. (2023). NMR and GPC Analysis of Alkyd Resins: Influence of Synthesis Method, Vegetable Oil and Polyol Content. Polymers, 15(9), 1993. https://doi.org/10.3390/polym15091993






