The Key to Solving Plastic Packaging Wastes: Design for Recycling and Recycling Technology
Abstract
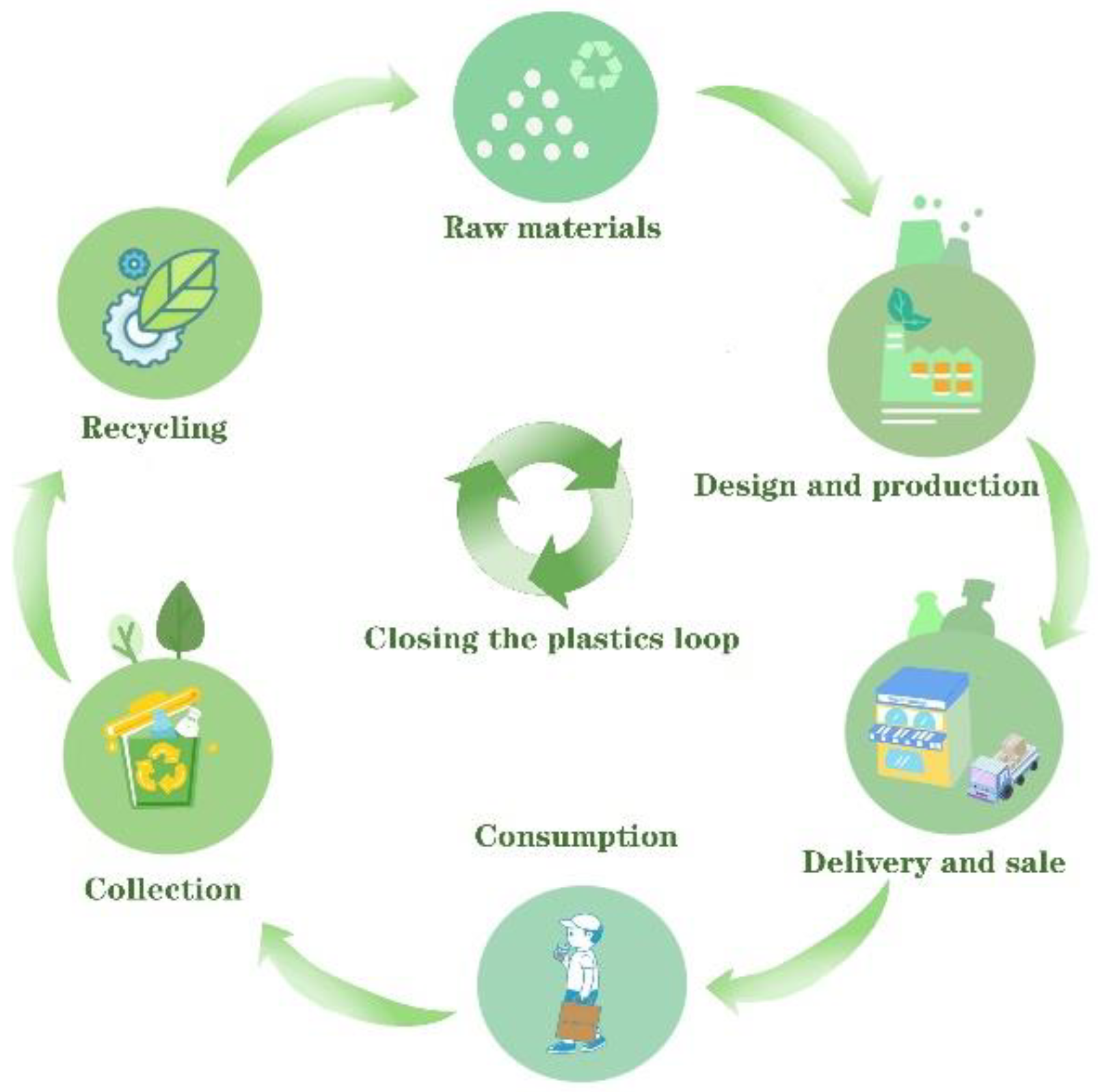
1. Introduction
2. Design for Recycling of Plastic Packaging
2.1. Design for Recycling
- (1)
- Recyclable materials and marks
- (2)
- Recovery techniques and methods
- (3)
- Recycling economy
- (4)
- Structure and design of recyclable parts
2.2. Recyclability and Influencing Factors
- (1)
- Base resin
- (2)
- Barrier layer/coating/additives
- (3)
- Labels/inks/adhesive
- (4)
- Closures and pumps
- (5)
- Color and dimensions
- (6)
- Attachments
2.3. Application Cases and Strategies
- (1)
- Material design
- (2)
- Structural design
- (3)
- Decoration design
3. Recycling Technology of Plastic Packaging Wastes
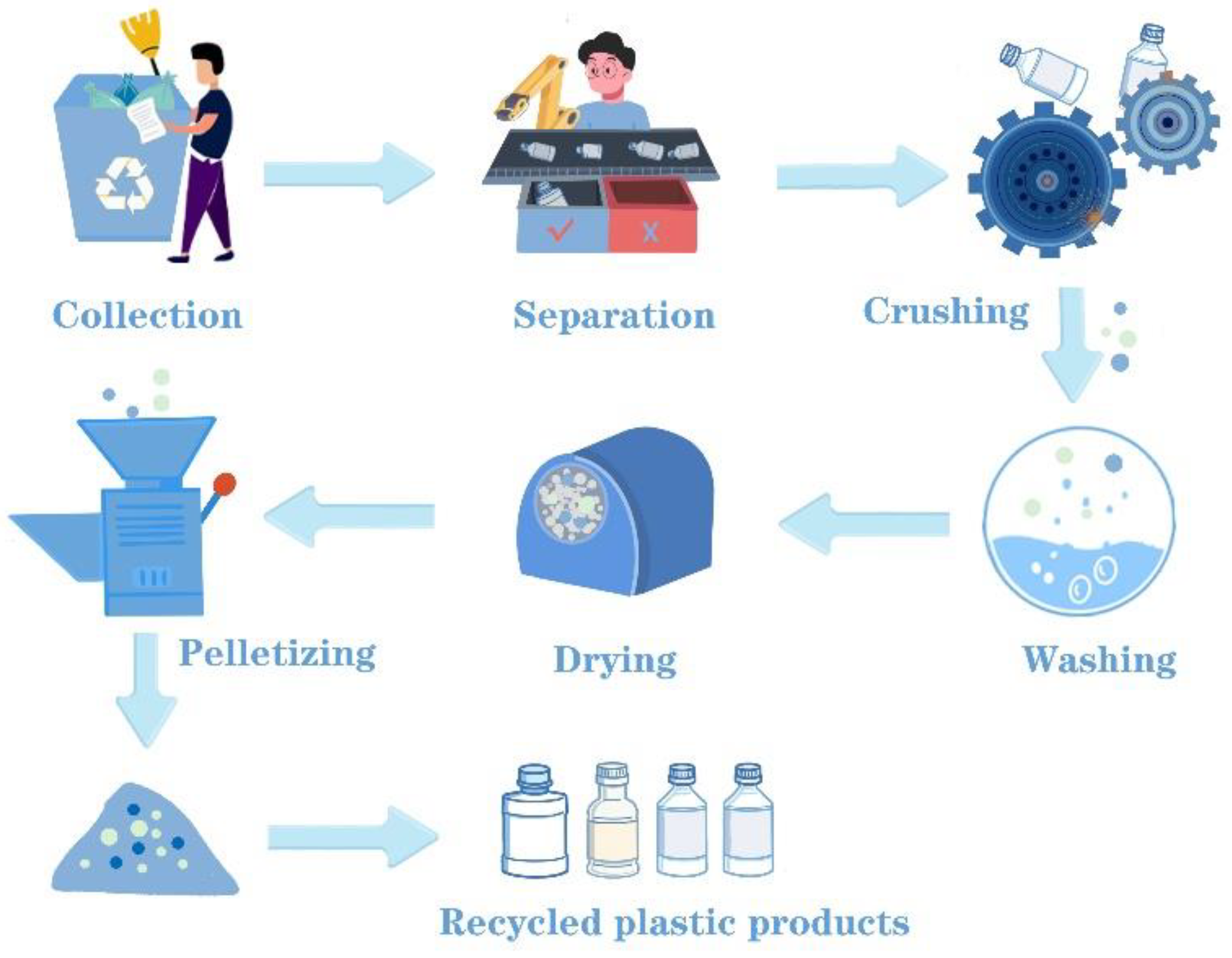
3.1. Mechanical Recycling
3.1.1. Automatic Sorting Methods
- (1)
- Air sorting
- (2)
- Flotation and froth flotation
- (3)
- Melt filtration
- (4)
- NIR and X-rays
3.1.2. Individual Plastics
3.1.3. Mixed Plastics Waste
3.2. Chemical Recycling
3.2.1. Thermoplastic Waste
3.2.2. Thermosetting Plastics Waste
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, N.; Sun, Z.X.; Feng, L.S.; Zheng, M.Z.; Chi, D.C.; Meng, W.Z.; Hou, Z.Y.; Bai, W.; Li, K.Y. Plastic film mulching for water-efficient agricultural applications and degradable films materials development research. Mater. Manuf. Process. 2015, 30, 143–154. [Google Scholar] [CrossRef]
- Raheem, D. Application of plastics and paper as food packaging materials-an overview. Emir. J. Food Agric. 2012, 25, 177–188. [Google Scholar] [CrossRef]
- Reichmanis, E.; Katz, H.; Kloc, C.; Maliakal, A. Plastic electronic devices: From materials design to device applications. Bell Labs Tech. J. 2005, 10, 87–105. [Google Scholar] [CrossRef]
- Jahnke, A. A discussion of single-use plastics in medical settings. Reinf. Plast. 2020, 64, 190–192. [Google Scholar] [CrossRef]
- Chauhan, V.; Kärki, T.; Varis, J. Review of natural fiber-reinforced engineering plastic composites, their applications in the transportation sector and processing techniques. J. Thermoplast. Compos. 2022, 35, 1169–1209. [Google Scholar] [CrossRef]
- Trzepieciński, T.; Najm, S.M.; Sbayti, M.; Belhadjsalah, H.; Szpunar, M.; Lemu, H.G. New advances and future possibilities in forming technology of hybrid metal-polymer composites used in aerospace applications. J. Compos. Sci. 2021, 5, 217. [Google Scholar] [CrossRef]
- Keya, K.N.; Kona, N.A.; Koly, F.A.; Maraz, K.M.; Islam, M.N.; Khan, R.A. Natural fiber reinforced polymer composites: History, types, advantages and applications. Mater. Eng. Res. 2019, 1, 69–85. [Google Scholar] [CrossRef]
- Porta, R.; Sabbah, M.; Pierro, P.D. Biopolymers as food packaging materials. Int. J. Mol. Sci. 2020, 21, 4942. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Awasthi, A.K.; Wei, F.; Tan, Q.; Li, J. Single-use plastics: Production, usage, disposal, and adverse impacts. Sci. Total Environ. 2021, 752, 141772. [Google Scholar] [CrossRef] [PubMed]
- Chamas, A.; Moon, H.; Zheng, J.; Qiu, Y.; Tabassum, T.; Jang, J.H.; Abu-Omar, M.; Scott, S.L.; Suh, S. Degradation rates of plastics in the environment. ACS Sustain. Chem. Eng. 2020, 8, 3494–3511. [Google Scholar] [CrossRef]
- Ali, S.S.; Elsamahy, T.; Koutra, E.; Kornaros, M.; El-Sheekh, M.; Abdelkarim, E.A.; Zhu, D.; Sun, J. Degradation of conventional plastic wastes in the environment: A review on current status of knowledge and future perspectives of disposal. Sci. Total Environ. 2021, 771, 144719. [Google Scholar] [CrossRef]
- Global Plastics Outlook: Economic Drivers, Environmental Impacts and Policy Options. Available online: https://www.oecd-ilibrary.org/environment/global-plastics-outlook_de747aef-en (accessed on 1 March 2023).
- Khoo, K.S.; Ho, L.Y.; Lim, H.R.; Leong, H.Y.; Chew, K.W. Plastic waste associated with the COVID-19 pandemic: Crisis or Opportunity? J. Hazard. Mater. 2021, 417, 126108. [Google Scholar] [CrossRef] [PubMed]
- Ebner, N.; Iacovidou, E. The challenges of Covid-19 pandemic on improving plastic waste recycling rates. Sustain. Prod. Consump. 2021, 28, 726–735. [Google Scholar] [CrossRef] [PubMed]
- Adyel, T.M. Accumulation of plastic waste during COVID-19. Science 2020, 369, 1314–1315. [Google Scholar] [CrossRef]
- Dris, R.; Agarwal, S.; Laforsch, C. Plastics: From a success story to an environmental problem and a global challenge. Glob. Chall. 2020, 4, 2000026. [Google Scholar] [CrossRef]
- Wang, Z.; Huo, J.; Duan, Y. The impact of government incentives and penalties on willingness to recycle plastic waste: An evolutionary game theory perspective. Front. Environ. Sci. Eng. 2020, 14, 29. [Google Scholar] [CrossRef]
- Shin, S.K.; Um, N.; Kim, Y.J.; Cho, N.H.; Jeon, T.W. New policy framework with plastic waste control plan for effective plastic waste management. Sustainability 2020, 12, 6049. [Google Scholar] [CrossRef]
- Antonopoulos, I.; Faraca, G.; Tonini, D. Recycling of post-consumer plastic packaging waste in the EU: Recovery rates, material flows, and barriers. Waste Manag. 2021, 126, 694–705. [Google Scholar] [CrossRef]
- Liu, J.; Yang, Y.; An, L.; Liu, Q.; Ding, J. The Value of China’s Legislation on Plastic Pollution Prevention in 2020. B. Environ. Contam. Tox. 2022, 108, 601–608. [Google Scholar] [CrossRef]
- Ferdous, W.; Manalo, A.; Siddique, R.; Mendis, P.; Zhuge, Y.; Wong, H.S.; Lokuge, W.; Aravinthan, T.; Schubel, P. Recycling of landfill wastes (tyres, plastics and glass) in construction—A review on global waste generation, performance, application and future opportunities. Resour. Conserv. Recycl. 2021, 173, 105745. [Google Scholar] [CrossRef]
- Chen, H.L.; Nath, T.K.; Chong, S.; Foo, V.; Gibbins, C.; Lechner, A.M. The plastic waste problem in Malaysia: Management, recycling and disposal of local and global plastic waste. SN Appl. Sci. 2021, 3, 437. [Google Scholar] [CrossRef]
- Millican, J.M.; Agarwal, S. Plastic Pollution: A Material Problem? Macromolecules 2021, 54, 4455–4469. [Google Scholar] [CrossRef]
- Godfrey, L. Waste plastic, the challenge facing developing countries—Ban it, change it, collect it? Recycling 2019, 4, 3. [Google Scholar] [CrossRef]
- Silva Filho, C.R.; Velis, C.A. United Nations’ plastic pollution treaty pathway puts waste and resources management sector at the centre of massive change. Waste Manag. Res. 2022, 40, 487–489. [Google Scholar] [CrossRef]
- Aryan, Y.; Yadav, P.; Samadder, S.P. Life Cycle Assessment of the existing and proposed plastic waste management options in India: A case study. J. Clean. Prod. 2019, 211, 1268–1283. [Google Scholar] [CrossRef]
- Faraca, G.; Martinez-Sanchez, V.; Astrup, T.F. Environmental life cycle cost assessment: Recycling of hard plastic waste collected at Danish recycling centres. Resour. Conserv. Recycl. 2019, 143, 299–309. [Google Scholar] [CrossRef]
- Antelava, A.; Damilos, S.; Hafeez, S.; Manos, G.; Al-Salem, S.M.; Sharma, B.K.; Kohli, K.; Constantinou, A. Plastic solid waste (PSW) in the context of life cycle assessment (LCA) and sustainable management. Environ. Manag. 2019, 64, 230–244. [Google Scholar] [CrossRef] [PubMed]
- Alhazmi, H.; Almansour, F.H.; Aldhafeeri, Z. Plastic Waste Management: A Review of Existing Life Cycle Assessment Studies. Sustainability 2021, 13, 5340. [Google Scholar] [CrossRef]
- Liu, M.; Wen, J.; Feng, Y.; Zhang, L.; Wu, J.; Wang, J.; Yang, X. A benefit evaluation for recycling medical plastic waste in China based on material flow analysis and life cycle assessment. J. Clean. Prod. 2022, 368, 133033. [Google Scholar] [CrossRef]
- Fagnani, D.E.; Tami, J.L.; Copley, G.; Clemons, M.N.; Getzler, Y.D.Y.L.; McNeil, A.J. 100th Anniversary of Macromolecular Science Viewpoint: Redefining Sustainable Polymers. ACS Macro Lett. 2021, 10, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Vogt, B.D.; Stokes, K.K.; Kumar, S.K. Why Is Recycling of Postconsumer Plastics So Challenging? ACS Appl. Polym. Mater. 2021, 3, 4325–4346. [Google Scholar] [CrossRef]
- Nkwachukwu, O.I.; Chima, C.H.; Ikenna, A.O.; Albert, L. Focus on potential environmental issues on plastic world towards a sustainable plastic recycling in developing countries. Int. J. Ind. Chem. 2013, 4, 34. [Google Scholar] [CrossRef]
- Jang, Y.; Lee, G.; Kwon, Y.; Lim, J.; Jeong, J. Recycling and management practices of plastic packaging waste towards a circular economy in South Korea. Resour. Conserv. Recycl. 2020, 158, 104798. [Google Scholar] [CrossRef]
- Horodytska, O.; Valdés, F.J.; Fullana, A. Plastic flexible films waste management—A state of art review. Waste Manag. 2018, 77, 413–425. [Google Scholar] [CrossRef]
- Chen, S. British dairy farmers’ management attitudes towards agricultural plastic waste: Reduce, reuse, recycle. Polym. Int. 2022, 71, 1418–1424. [Google Scholar] [CrossRef]
- Gill, Y.Q.; Khurshid, M.; Abid, U.; Ijaz, M.W. Review of hospital plastic waste management strategies for Pakistan. Environ. Sci. Pollut. R. 2022, 29, 9408–9421. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Z.; Zhang, J.; Fan, G.; Wu, D. A quantitative study on the benefit of various waste classifications. Adv. Civ. Eng. 2021, 2021, 6660927. [Google Scholar] [CrossRef]
- Meng, X.; Wen, Z.; Qian, Y. Multi-agent based simulation for household solid waste recycling behavior. Resour. Conserv. Recycl. 2018, 128, 535–545. [Google Scholar] [CrossRef]
- Ishii, K.; Eubanks, C.F.; Marco, P.D. Design for product retirement and material life-cycle. Mater. Design 1994, 15, 225–233. [Google Scholar] [CrossRef]
- Alting, L. Life cycle engineering and design. CIRP Ann. 1995, 44, 569–580. [Google Scholar] [CrossRef]
- Boothroyd, G.; Alting, L. Design for assembly and disassembly. CIRP Ann. 1992, 41, 625–636. [Google Scholar] [CrossRef]
- Kriwet, A.; Zussman, E.; Seliger, G. Systematic integration of design-for-recycling into product design. Int. J. Prod. Econ. 1995, 38, 15–22. [Google Scholar] [CrossRef]
- Hagelüken, C.; Corti, C.W. Recycling of gold from electronics: Cost-effective use through ‘Design for Recycling’. Gold Bull. 2010, 43, 209–220. [Google Scholar] [CrossRef]
- Shu, L.H.; Flowers, W.C. Application of a design-for-remanufacture framework to the selection of product life-cycle fastening and joining methods. Robot. Com.-Int. Manuf. 1999, 15, 179–190. [Google Scholar] [CrossRef]
- Grenchus, E.; Keene, R.; Nobs, C. Demanufacturing of information technology equipment. In Proceedings of the IEEE Symposium on Electronics and the Environment, San Francisco, CA, USA, 5–7 May 1997. [Google Scholar]
- Zussman, E.; Kriwet, A.; Seliger, G. Disassembly-oriented assessment methodology to support design for recycling. CIRP Ann. 1994, 43, 9–14. [Google Scholar] [CrossRef]
- Sodhi, M.; Knight, W.A. Product design for disassembly and bulk recycling. CIRP Ann. 1998, 47, 115–118. [Google Scholar] [CrossRef]
- Ferrão, P.; Amaral, J. Design for recycling in the automobile industry: New approaches and new tools. J. Eng. Design 2006, 17, 447–462. [Google Scholar] [CrossRef]
- Soh, S.L.; Ong, S.K.; Nee, A.Y.C. Design for assembly and disassembly for remanufacturing. Assem. Autom. 2016, 36, 12–24. [Google Scholar] [CrossRef]
- Favi, C.; Marconi, M.; Germani, M.; Mandolini, M. A design for disassembly tool oriented to mechatronic product demanufacturing and recycling. Adv. Eng. Inform. 2019, 39, 62–79. [Google Scholar] [CrossRef]
- Masanet, E.; Horvath, A. Assessing the benefits of design for recycling for plastics in electronics: A case study of computer enclosures. Mater. Design 2007, 28, 1801–1811. [Google Scholar] [CrossRef]
- Gaustad, G.; Olivetti, E.; Kirchain, R. Design for recycling: Evaluation and efficient alloy modificatio. J. Ind. Ecol. 2010, 14, 286–308. [Google Scholar] [CrossRef]
- Gallagher, J.; Basu, B.; Browne, M.; Kenna, A.; McCormack, S.; Pilla, F.; Styles, D. Adapting stand-alone renewable energy technologies for the circular economy through eco-design and recycling. J. Ind. Ecol. 2019, 23, 133–140. [Google Scholar] [CrossRef]
- Selke, S.E.; Lai, C.C. Recyclability aspects of packaging design. Resour. Recycl. 1988, 6, 36–37, 66–70. [Google Scholar]
- Navin-Chandra, D. The recovery problem in product design. J. Eng. Design 1994, 5, 65–86. [Google Scholar] [CrossRef]
- Knight, W.A.; Sodhi, M. Design for bulk recycling: Analysis of materials separation. CIRP Ann. 2000, 49, 83–86. [Google Scholar] [CrossRef]
- Cândido, L.; Kindlein, W.; Demori, R.; Carli, L.; Mauler, R.; Oliveira, R. The recycling cycle of materials as a design project tool. J. Clean. Prod. 2011, 19, 1438–1445. [Google Scholar] [CrossRef]
- Corabieru, P.; Corabieru, A.; Vasilescu, D.D. New approaches in the design of plastic products for easy recycling. Environ. Eng. Manag. J. 2014, 13, 1997–2004. [Google Scholar] [CrossRef]
- Leal, J.M.; Pompidou, S.; Charbuillet, C.; Perry, N. Design for and from recycling: A circular ecodesign approach to improve the circular economy. Sustainability 2020, 12, 9861. [Google Scholar] [CrossRef]
- Köhler, A.R. Challenges for eco-design of emerging technologies: The case of electronic textiles. Mater. Design 2013, 51, 51–60. [Google Scholar] [CrossRef]
- Houe, R.; Grabot, B. Assessing the compliance of a product with an eco-label: From standards to constraints. Int. J. Prod. Econ. 2009, 121, 21–38. [Google Scholar] [CrossRef]
- Dodbiba, G.; Fujita, T. Progress in separating plastic materials for recycling. Phys. Sep. Sci. Eng. 2004, 13, 165–182. [Google Scholar] [CrossRef]
- Ishii, K.; Eubanks, C.F.; Marks, M. Evaluation methodology for post-manufacturing issues in life-cycle design. Concurr. Eng.-Res. A 1993, 1, 61–68. [Google Scholar] [CrossRef]
- de Aguiar, J.; de Oliveira, L.; da Silva, J.O.; Bond, D.; Scalice, R.K.; Becker, D. A design tool to diagnose product recyclability during product design phase. J. Clean. Prod. 2017, 141, 219–229. [Google Scholar] [CrossRef]
- Tan, J.; Wang, Q.; Chen, S.; Li, Z.; Sun, J.; Liu, W.; Yang, W.; Xiang, X.; Sun, X.; Duan, X. Recycling-oriented cathode materials design for lithium-ion batteries: Elegant structures versus complicated compositions. Energy Storage Mater. 2021, 41, 380–394. [Google Scholar] [CrossRef]
- Plastics Recyclers. International Plastics Recycling Groups Announce Global Definition of “Plastic Recyclability”. Available online: https://www.plasticsrecyclers.eu/ (accessed on 15 January 2023).
- The Association of Plastic Recyclers. APR Design® Guide for Plastics Recyclability. Available online: https://plasticsrecycling.org/apr-design-guide (accessed on 15 January 2023).
- RecyClass. Design for Recycling Guidelines. Available online: https://recyclass.eu/recyclass/definition/ (accessed on 15 January 2023).
- Zhang, R.; Ma, X.; Shen, X.; Zhai, Y.; Zhang, T.; Ji, C.; Hong, J. PET bottles recycling in China: An LCA coupled with LCC case study of blanket production made of waste PET bottles. J. Environ. Manag. 2020, 260, 110062. [Google Scholar] [CrossRef]
- Benyathiar, P.; Kumar, P.; Carpenter, G.; Brace, J.; Mishra, D.K. Polyethylene terephthalate (PET) bottle-to-bottle recycling for the beverage industry: A review. Polymers 2022, 14, 2366. [Google Scholar] [CrossRef] [PubMed]
- Katsura, T.; Sasaki, H. On-going solutions to environmental issues in plastic packaging. Packag. Technol. Sci. 2001, 14, 87–95. [Google Scholar] [CrossRef]
- Recoup. Recyclability by Design 2022. Available online: https://www.recoup.org/p/173/recoup-reports (accessed on 15 January 2023).
- Aliaga, C.; Ferreira, B.; Hortal, M.; Pancorbo, M.Á.; López, J.M.; Navas, F.J. Influence of RFID tags on recyclability of plastic packaging. Waste Manag. 2011, 31, 1133–1138. [Google Scholar] [CrossRef]
- Rhein, S.; Sträter, K.F. Corporate self-commitments to mitigate the global plastic crisis: Recycling rather than reduction and reuse. J. Clean. Prod. 2021, 296, 126571. [Google Scholar] [CrossRef]
- Kahlert, S.; Bening, C.R. Why pledges alone will not get plastics recycled: Comparing recyclate production and anticipated demand. Resour. Conserv. Recycl. 2022, 181, 106279. [Google Scholar] [CrossRef]
- Perry, N.; Bernard, A.; Laroche, F.; Pompidou, S. Improving design for recycling—Application to composites. CIRP Ann. 2012, 61, 151–154. [Google Scholar] [CrossRef]
- Kaiser, K.; Schmid, M.; Schlummer, M. Recycling of polymer-based multilayer packaging: A review. Recycling 2018, 3, 1. [Google Scholar] [CrossRef]
- Colgate. Faq Toothpaste|Colgate®. Available online: https://www.colgate.com/en-us/power-of-optimism/faq (accessed on 15 January 2023).
- Guerritore, M.; Olivieri, F.; Castaldo, R.; Avolio, R.; Cocca, M.; Errico, M.E.; Galdi, M.R.; Carfagna, C.; Gentile, G. Recyclable-by-design mono-material flexible packaging with high barrier properties realized through graphene hybrid coatings. Resour. Conserv. Recycl. 2022, 179, 106126. [Google Scholar] [CrossRef]
- Meereboer, K.W.; Misra, M.; Mohanty, A.K. Review of recent advances in the biodegradability of polyhydroxyalkanoate (PHA) bioplastics and their composites. Green Chem. 2020, 22, 5519–5558. [Google Scholar] [CrossRef]
- Brillian, M.; Kristina, H.J.; Silalahi, R.V. Label size evaluation of ready-to-drink beverages based on brand knowledge assessment with brand awareness as a parameter. ASEAN Mark. J. 2019, 10, 31–35. [Google Scholar]
- Fernandez, C.M.; Alves, J.; Gaspar, P.D.; Lima, T.M.; Silva, P.D. Innovative processes in smart packaging. A systematic review. J. Sci. Food Agric. 2023, 103, 986–1003. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Liew, M.S. Tertiary recycling of plastics waste: An analysis of feedstock, chemical and biological degradation methods. J. Mater. Cycles Waste. Manag. 2021, 23, 32–43. [Google Scholar] [CrossRef]
- Datta, J.; Kopczyńska, P. From polymer waste to potential main industrial products: Actual state of recycling and recovering. Crit. Rev. Environ. Sci. Technol. 2016, 46, 905–946. [Google Scholar] [CrossRef]
- Schyns, Z.O.G.; Shaver, M.P. Mechanical recycling of packaging plastics: A review. Macromol. Rapid Commun. 2021, 42, 2000415. [Google Scholar] [CrossRef]
- Briassoulis, D.; Hiskakis, M.; Babou, E. Technical specifications for mechanical recycling of agricultural plastic waste. Waste Manag. 2013, 33, 1516–1530. [Google Scholar] [CrossRef]
- Damayanti, D.; Saputri, D.R.; Marpaung, D.S.S.; Yusupandi, F.; Sanjaya, A.; Simbolon, Y.M.; Asmarani, W.; Ulfa, M.; Wu, H.S. Current Prospects for Plastic Waste Treatment. Polymers 2022, 14, 3133. [Google Scholar] [CrossRef]
- Davidson, M.G.; Furlong, R.A.; McManus, M.C. Developments in the life cycle assessment of chemical recycling of plastic waste—A review. J. Clean. Prod. 2021, 293, 126163. [Google Scholar] [CrossRef]
- Eriksson, O.; Finnveden, G. Plastic waste as a fuel-CO2-neutral or not? Energy Environ. Sci. 2009, 2, 907–914. [Google Scholar] [CrossRef]
- Khoo, H.H. LCA of plastic waste recovery into recycled materials, energy and fuels in Singapore. Resour. Conserv. Recycl. 2019, 145, 67–77. [Google Scholar] [CrossRef]
- Cui, J.; Forssberg, E. Mechanical recycling of waste electric and electronic equipment: A review. J. Hazard. Mater. 2003, 99, 243–263. [Google Scholar] [CrossRef] [PubMed]
- Al-Salem, S.M.; Lettieri, P.; Baeyens, J. Recycling and recovery routes of plastic solid waste (PSW): A review. Waste Manag. 2009, 29, 2625–2643. [Google Scholar] [CrossRef]
- Janajreh, I.; Alshrah, M.; Zamzam, S. Mechanical recycling of PVC plastic waste streams from cable industry: A case study. Sustain. Cities Soc. 2015, 18, 13–20. [Google Scholar] [CrossRef]
- Lawrence, K.; Cooper, V.; Kissoon, P. Sustaining voluntary recycling programmes in a country transitioning to an integrated solid waste management system. J. Environ. Manag. 2020, 257, 109966. [Google Scholar] [CrossRef] [PubMed]
- Thoden van Velzen, E.U.; Chu, S.; Alvarado Chacon, F.; Brouwer, M.T.; Molenveld, K. The impact of impurities on the mechanical properties of recycled polyethylene. Packag. Technol. Sci. 2021, 34, 219–228. [Google Scholar] [CrossRef]
- Ruj, B.; Pandey, V.; Jash, P.; Srivastava, V.K. Sorting of plastic waste for effective recycling. J. Appl. Sci. Eng. Res. 2015, 4, 564–571. [Google Scholar]
- Lange, J.P. Managing plastic waste ─ sorting, recycling, disposal, and product redesign. ACS Sustain. Chem. Eng. 2021, 9, 15722–15738. [Google Scholar] [CrossRef]
- Wang, C.; Wang, H.; Fu, J.; Liu, Y. Flotation separation of waste plastics for recycling—A review. Waste Manag. 2015, 41, 28–38. [Google Scholar] [CrossRef]
- Saisinchai, S. Separation of PVC from PET/PVC mixtures using flotation by calcium lignosulfonate depressant. Eng. J. 2014, 18, 46–53. [Google Scholar] [CrossRef]
- Ragaert, K.; Delva, L.; Geem, K.V. Mechanical and chemical recycling of solid plastic waste. Waste Manag. 2017, 69, 24–58. [Google Scholar] [CrossRef] [PubMed]
- Luijsterburg, B.J.; Jobse, P.S.; Spoelstra, A.B.; Goossens, J.G.P. Solid-state drawing of post-consumer isotactic poly(propylene): Effect of melt filtration and carbon black on structural and mechanical properties. Waste Manag. 2016, 54, 53–61. [Google Scholar] [CrossRef]
- Wu, X.; Li, J.; Yao, L.; Xu, Z. Auto-sorting commonly recovered plastics from waste household appliances and electronics using near-infrared spectroscopy. J. Clean. Prod. 2020, 246, 118732. [Google Scholar] [CrossRef]
- Rani, M.; Marchesi, C.; Federici, S.; Rovelli, G.; Alessandri, I.; Vassalini, I.; Ducoli, S.; Borgese, L.; Zacco, A.; Bilo, F.; et al. Miniaturized Near-Infrared (Micro NIR) Spectrometer in Plastic Waste Sorting. Materials 2019, 12, 2740. [Google Scholar] [CrossRef]
- Singh, B.; Sharma, N. Mechanistic implications of plastic degradation. Polym. Degrad. Stab. 2008, 93, 561–584. [Google Scholar] [CrossRef]
- Thomas, N.L.; Clarke, J.; McLauchlin, A.R.; Patrick, S.G. Oxo-degradable plastics: Degradation, environmental impact and recycling. Waste Resour. Manag. 2012, 165, 133–140. [Google Scholar]
- Folarin, O.M.; Sadiku, E.R. Thermal stabilizers for poly (vinyl chloride): A review. Int. J. Phys. Sci. 2011, 6, 4323–4330. [Google Scholar]
- Maris, J.; Bourdon, S.; Brossard, J.M.; Cauret, L.; Fontaine, L.; Montembault, V. Mechanical recycling: Compatibilization of mixed thermoplastic wastes. Polym. Degrad. Stab. 2018, 147, 245–266. [Google Scholar] [CrossRef]
- Manila, F.P.L. Effect of fillers on the properties of recycled polymers. Macromol. Symp. 2003, 194, 101–110. [Google Scholar] [CrossRef]
- Lewandowski, K.; Skórczewska, K. A brief review of poly(vinyl chloride) (PVC) recycling. Polymers 2022, 14, 3035. [Google Scholar] [CrossRef]
- Asawakosinchai, A.; Jubsilp, C.; Mora, P.; Rimdusit, S. Organic heat stabilizers for polyvinyl chloride (PVC): A synergistic behavior of eugenol and uracil derivative. J. Mater. Eng. Perform. 2017, 26, 4781–4788. [Google Scholar] [CrossRef]
- Sadat-Shojai, M.; Bakhshandeh, G.R. Recycling of PVC wastes. Polym. Degrad. Stab. 2011, 96, 404–415. [Google Scholar] [CrossRef]
- Singh, A.K.; Bedi, R.; Kaith, B.S. Composite materials based on recycled polyethylene terephthalate and their properties–A comprehensive review. Compos. Part B-Eng. 2021, 219, 108928. [Google Scholar] [CrossRef]
- Nasir, A.; Yasin, T.; Islam, A. Thermo-oxidative degradation behavior of recycled polypropylene. J. Appl. Polym. Sci. 2011, 119, 3315–3320. [Google Scholar] [CrossRef]
- Das, P.; Tiwari, P. Thermal degradation study of waste polyethylene terephthalate (PET) under inert and oxidative environments. Thermochim. Acta 2019, 679, 178340. [Google Scholar] [CrossRef]
- Colucci, G.; Simon, H.; Roncato, D.; Martorana, B.; Badini, C. Effect of recycling on polypropylene composites reinforced with glass fibres. J. Thermoplast. Compos. 2017, 30, 707–723. [Google Scholar] [CrossRef]
- Thumsorn, S.; Negoro, T.; Thodsaratpreeyakul, W.; Inoya, H.; Okoshi, M.; Hamada, H. Effect of ammonium polyphosphate and fillers on flame retardant and mechanical properties of recycled PET injection molded. Polym. Adv. Technol. 2017, 28, 979–985. [Google Scholar] [CrossRef]
- Chowreddy, R.R.; Nord-Varhaug, K.; Rapp, F. Recycled polyethylene terephthalate/carbon nanotube composites with improved processability and performance. J. Mater. Sci. 2018, 53, 7017–7029. [Google Scholar] [CrossRef]
- La Mantia, F.P. Recycling of heterogeneous plastics wastes: Ι—Blends with low-density polyethylene. Polym. Degrad. Stab. 1992, 37, 145–148. [Google Scholar] [CrossRef]
- La Mantia, F.P. Recycling of heterogeneous plastics wastes: II—The role of modifier agents. Polym. Degrad. Stab. 1993, 42, 213–218. [Google Scholar] [CrossRef]
- Mamoor, G.M.; Shahid, W.; Mushtaq, A.; Amjad, U.; Mehmood, U. Recycling of mixed plastics waste containing polyethylene, polyvinylchloride and polyethylene terephthalate. Chem. Eng. Res. Bull. 2013, 16, 25–32. [Google Scholar] [CrossRef]
- Paul, D.R.; Barlow, J.W. Polymer blends. J. Macromol. Sci. Rev. Macromol. Chem. 1980, 18, 109–168. [Google Scholar] [CrossRef]
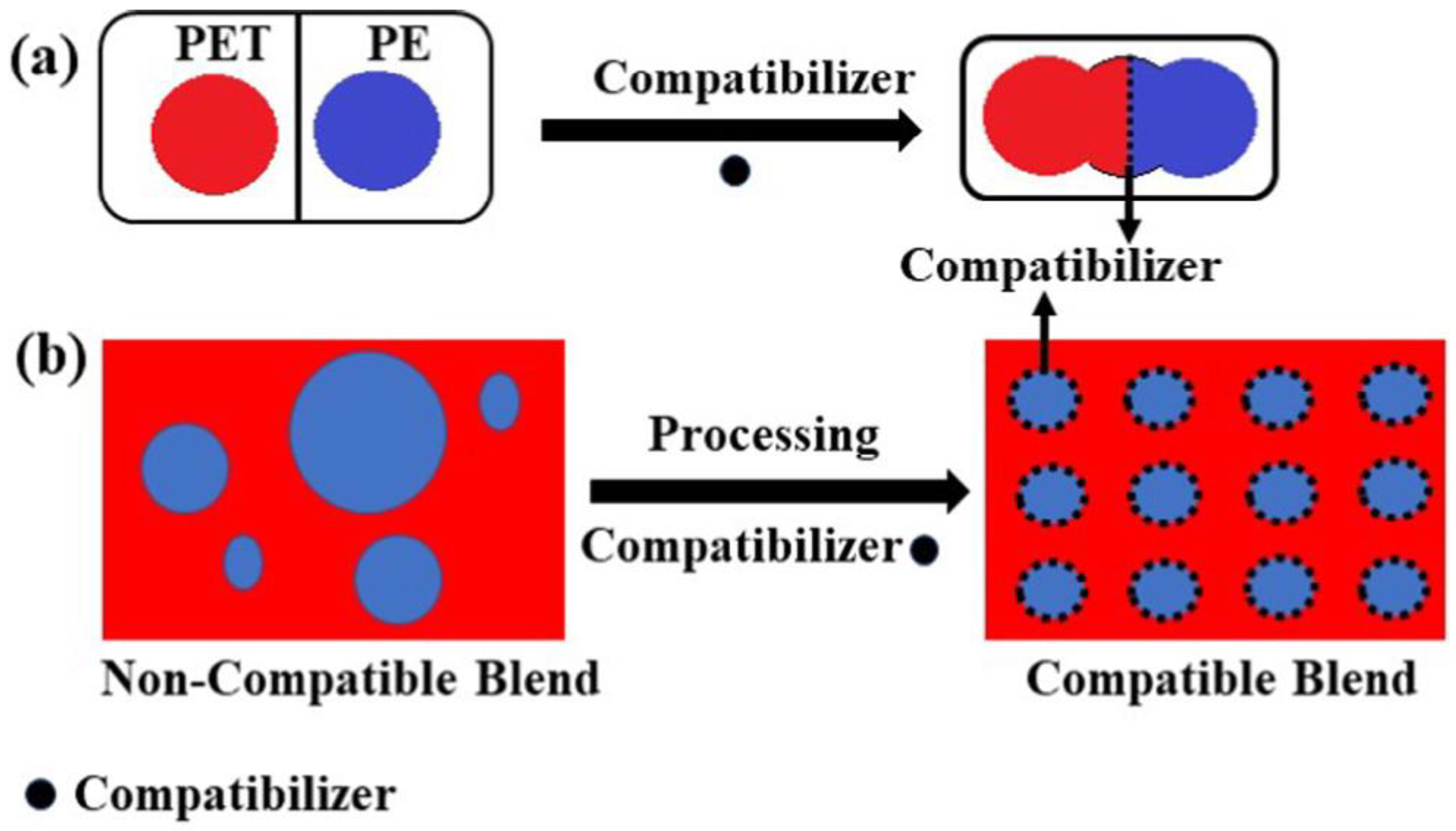
- Koning, C.; Van Duin, M.; Pagnoulle, C.; Jerome, R. Strategies for compatibilization of polymer blends. Prog. Polym. Sci. 1998, 23, 707–757. [Google Scholar] [CrossRef]
- Utracki, L.A. Compatibilization of polymer blends. Can. J. Chem. Eng. 2002, 80, 1008–1016. [Google Scholar] [CrossRef]
- Graziano, A.; Jaffer, S.; Sain, M. Review on modification strategies of polyethylene/polypropylene immiscible thermoplastic polymer blends for enhancing their mechanical behavior. J. Elastom. Plast. 2019, 51, 291–336. [Google Scholar] [CrossRef]
- Techawinyutham, L.; Tengsuthiwat, J.; Srisuk, R.; Techawinyutham, W.; Rangappa, S.M.; Siengchin, S. Recycled LDPE/PETG blends and HDPE/PETG blends: Mechanical, thermal, and rheological properties. J. Mater. Res. Technol. 2021, 15, 2445–2458. [Google Scholar] [CrossRef]
- Vazquez, Y.V.; Barbosa, S.E. Recycling of mixed plastic waste from electrical and electronic equipment. Added value by compatibilization. Waste Manag. 2016, 53, 196–203. [Google Scholar] [CrossRef]
- Macosko, C.W.; Jeon, H.K.; Hoye, T.R. Reactions at polymer–polymer interfaces for blend compatibilization. Prog. Polym. Sci. 2005, 30, 939–947. [Google Scholar] [CrossRef]
- Dorigato, A. Recycling of polymer blends. Adv. Ind. Eng. Polym. Res. 2021, 4, 53–69. [Google Scholar] [CrossRef]
- Rajasekaran, D.; Maji, P.K. Recycling of plastic wastes with poly (ethylene-co-methacrylic acid) copolymer as compatibilizer and their conversion into high-end product. Waste Manag. 2018, 74, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Pracella, M.; Rolla, L.; Chionna, D.; Galeski, A. Compatibilization and properties of poly(ethylene terephthalate)/polyethylene blends based on recycled materials. Macromol. Chem. Phys. 2002, 203, 1473–1485. [Google Scholar] [CrossRef]
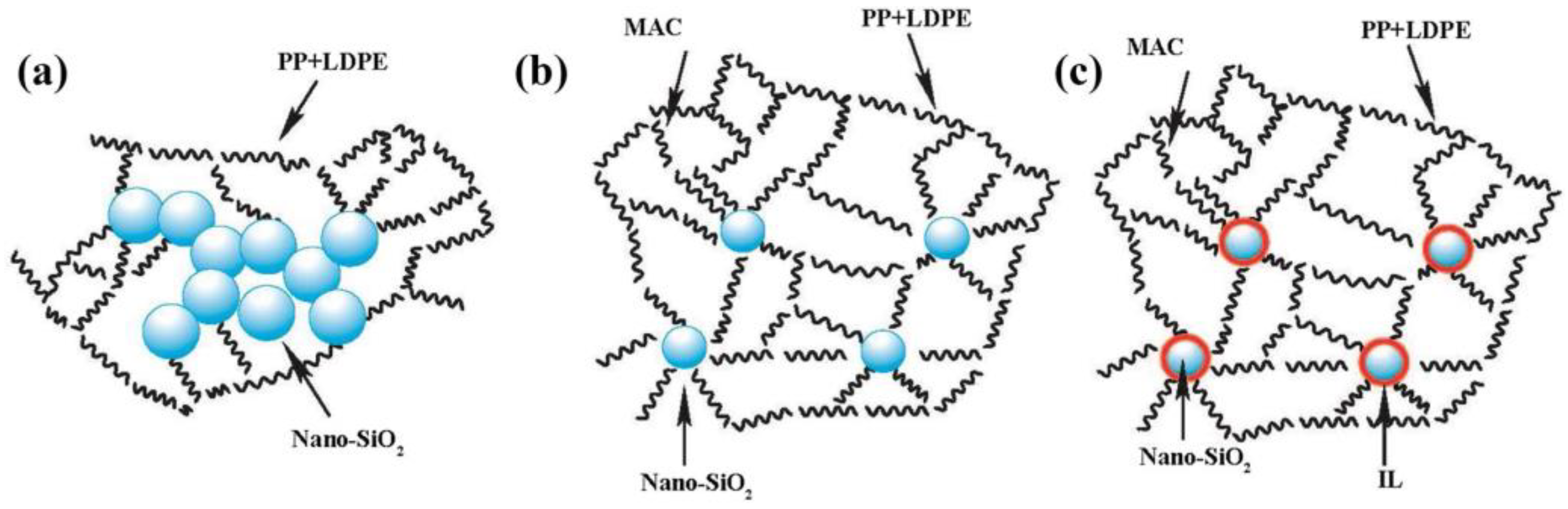
- Garofalo, E.; Di Maio, L.; Scarfato, P.; Di Gregorio, F.; Incarnato, L. Reactive compatibilization and melt compounding with nanosilicates of post-consumer flexible plastic packagings. Polym. Degrad. Stab. 2018, 152, 52–63. [Google Scholar] [CrossRef]
- Ahmadlouydarab, M.; Chamkouri, M.; Chamkouri, H. Compatibilization of immiscible polymer blends (R-PET/PP) by adding PP-g-MA as compatibilizer: Analysis of phase morphology and mechanical properties. Polym. Bull. 2020, 77, 5753–5766. [Google Scholar] [CrossRef]
- Touati, Z.; Boulahia, H.; Belhaneche-Bensemra, N.; Massardier, V. Modification of diss fibers for biocomposites based on recycled low-density polyethylene and polypropylene blends. Waste Biomass Valor. 2019, 10, 2365–2378. [Google Scholar] [CrossRef]
- Martikka, O.; Kärki, T. Promoting recycling of mixed waste polymers in wood-polymer composites using compatibilizers. Recycling 2019, 4, 6. [Google Scholar] [CrossRef]
- Fredi, G.; Dorigato, A. Recycling of bioplastic waste: A review. Adv. Ind. Eng. Polym. Res. 2021, 4, 159–177. [Google Scholar] [CrossRef]
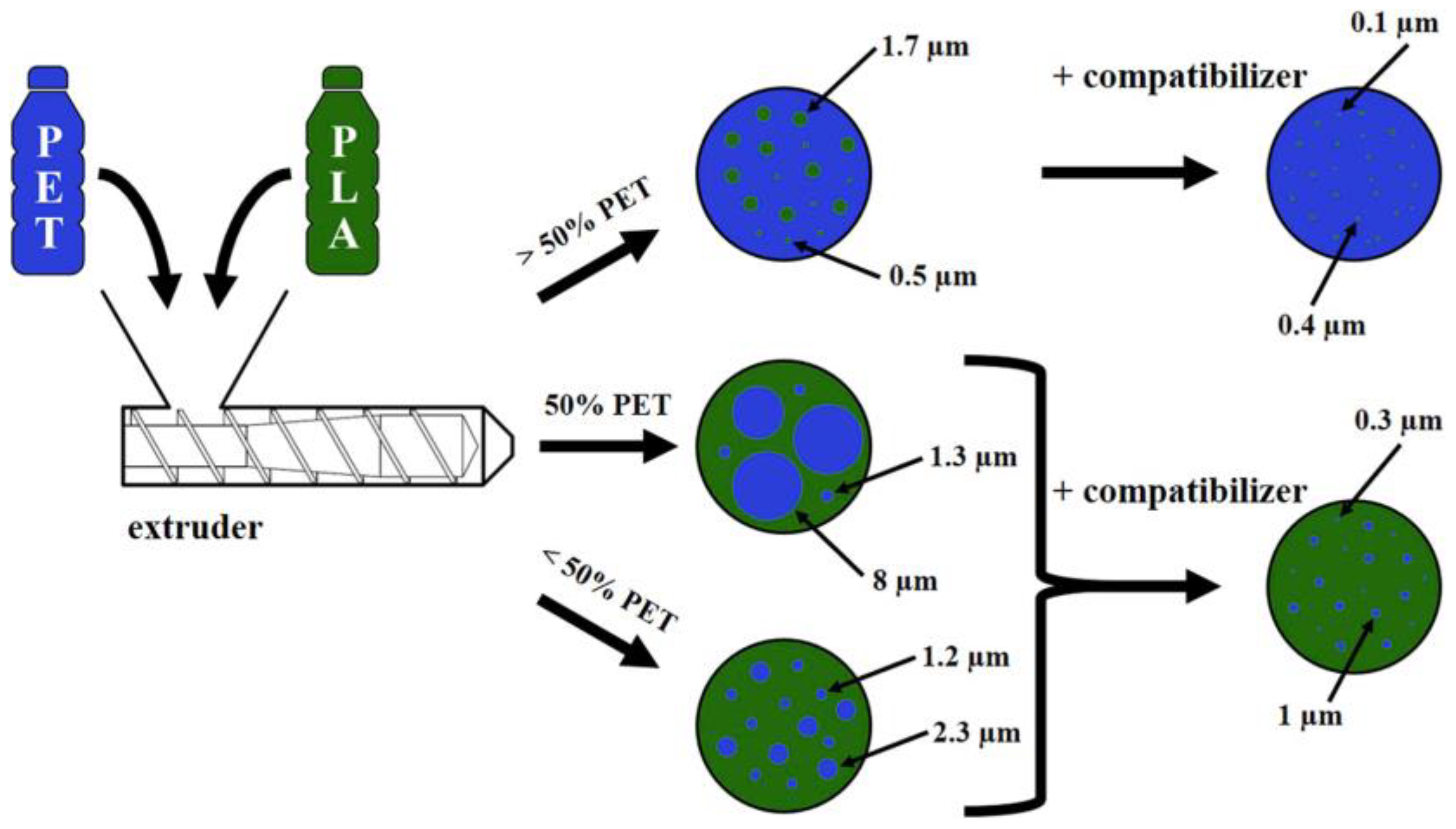
- Sánchez, A.C.; Collinson, S.R. The selective recycling of mixed plastic waste of polylactic acid and polyethylene terephthalate by control of process conditions. Eur. Polym. J. 2011, 47, 1970–1976. [Google Scholar] [CrossRef]
- Beltrán, F.R.; Gaspar, G.; Chomachayi, M.D.; Jalali-Arani, A.; Lozano-Pérez, A.A.; Cenis, J.L.; Orden, M.U.; Pérez, E.; Urreaga, J.M.M. Influence of addition of organic fillers on the properties of mechanically recycled PLA. Environ. Sci. Pollut. Res. 2021, 28, 24291–24304. [Google Scholar] [CrossRef]
- Alaerts, L.; Augustinus, M.; Van Acker, K. Impact of bio-based plastics on current recycling of plastics. Sustainability 2018, 10, 1487. [Google Scholar] [CrossRef]
- Gere, D.; Czigany, T. Future trends of plastic bottle recycling: Compatibilization of PET and PLA. Polym. Test. 2020, 81, 106160. [Google Scholar] [CrossRef]
- Kaminsky, W. Chemical recycling of mixed plastics of pyrolysis. Adv. Polym. Technol. 1995, 14, 337–344. [Google Scholar] [CrossRef]
- Lee, J.; Kwon, E.E.; Lam, S.S.; Chen, W.H.; Rinklebe, J.; Park, Y.K. Chemical recycling of plastic waste via thermocatalytic routes. J. Clean. Prod. 2021, 321, 128989. [Google Scholar] [CrossRef]
- Liu, X.H.; Xu, S.M.; Zhang, F.; Wang, X.L.; Wang, Y.Z. Chemical upcycling of polymeric materials. Acta Polym. Sin. 2022, 53, 1005–1022. [Google Scholar]
- Awoyera, P.O.; Adesina, A. Plastic wastes to construction products: Status, limitations and future perspective. Case Stud. Constr. Mater. 2020, 12, e00330. [Google Scholar] [CrossRef]
- Dogu, O.; Pelucchi, M.; Van de Vijver, R.; Van Steenberge, P.H.M.; D’hooge, D.R.; Cuoci, A.; Mehl, M.; Frassoldati, A.; Faravelli, T.; Van Geem, K.M. The chemistry of chemical recycling of solid plastic waste via pyrolysis and gasification: State-of-the-art, challenges, and future directions. Prog. Energy Combust. Sci. 2021, 84, 100901. [Google Scholar] [CrossRef]
- Thiounn, T.; Smith, R.C. Advances and approaches for chemical recycling of plastic waste. J. Polym. Sci. 2020, 58, 1347–1364. [Google Scholar] [CrossRef]
- Jiang, J.; Shi, K.; Zhang, X.; Yu, K.; Zhang, H.; He, J.; Ju, Y.; Liu, J. From plastic waste to wealth using chemical recycling: A review. J. Environ. Chem. Eng. 2022, 10, 106867. [Google Scholar] [CrossRef]
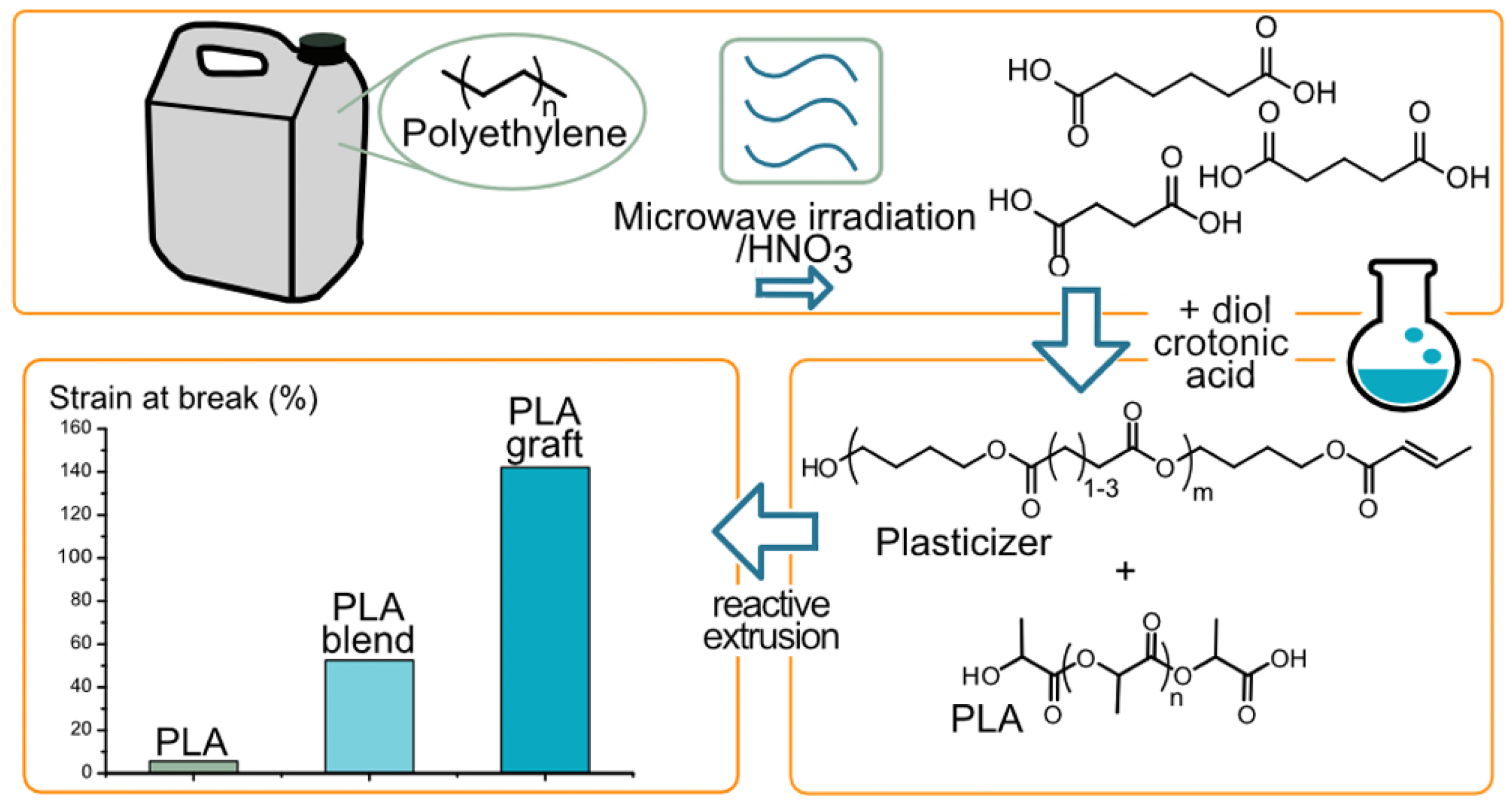
- Bäckström, E.; Odelius, K.; Hakkarainen, M. Trash to treasure: Microwave-assisted conversion of polyethylene to functional chemicals. Ind. Eng. Chem. Res. 2017, 56, 14814–14821. [Google Scholar] [CrossRef]
- Bäckström, E.; Odelius, K.; Hakkarainen, M. Designed from recycled: Turning polyethylene waste to covalently attached polylactide plasticizers. ACS Sustain. Chem. Eng. 2019, 7, 11004–11013. [Google Scholar] [CrossRef]
- Kots, P.A.; Liu, S.; Vance, B.C.; Wang, C.; Sheehan, J.D.; Vlachos, D.G. Polypropylene plastic waste conversion to lubricants over Ru/TiO2 catalysts. ACS Catal. 2021, 11, 8104–8115. [Google Scholar] [CrossRef]
- Sharma, P.; Lochab, B.; Kumar, D.; Roy, P.K. Sustainable bis-benzoxazines from cardanol and PET-derived terephthalamides. ACS Sustain. Chem. Eng. 2016, 4, 1085–1093. [Google Scholar] [CrossRef]
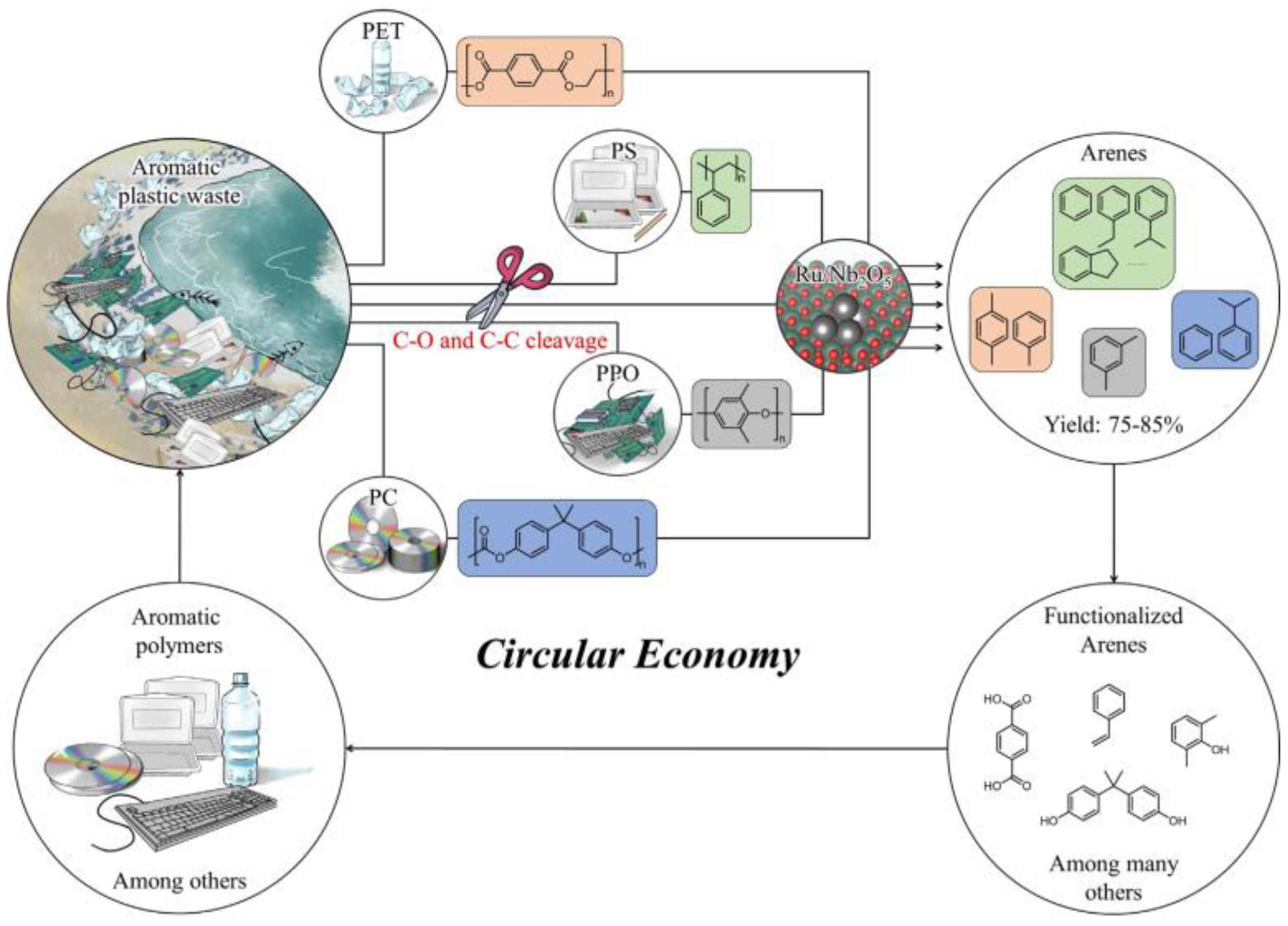
- Jing, Y.; Wang, Y.; Furukawa, S.; Xia, J.; Sun, C.; Hülsey, M.J.; Wang, H.; Guo, Y.; Liu, X.; Yan, N. Towards the circular economy: Converting aromatic plastic waste back to arenes over a Ru/Nb2O5 catalyst. Angew. Chem. Int. Ed. 2021, 60, 5527–5535. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Strachan, A. Molecular simulations of crosslinking process of thermosetting polymers. Polymer 2010, 51, 6058–6070. [Google Scholar] [CrossRef]
- Quirino, R.L.; Monroe, K.; Fleischer III, C.H.; Biswas, E.; Kessler, M.R. Thermosetting polymers from renewable sources. Polym. Int. 2021, 7, 167–180. [Google Scholar] [CrossRef]
- Liu, T.; Zhao, B.; Zhang, J. Recent development of repairable, malleable and recyclable thermosetting polymers through dynamic transesterification. Polymer 2020, 194, 122392. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, Z.; Wang, B.; Li, P.; Zhu, J.; Ma, S. Closed-loop chemical recycling of thermosetting polymers and their applications: A review. Green Chem. 2022, 24, 5691–5708. [Google Scholar] [CrossRef]
- Kim, Y.N.; Kim, Y.O.; Kim, S.Y.; Park, M.; Yang, B.; Kim, J.; Jung, Y.C. Application of supercritical water for green recycling of epoxy-based carbon fiber reinforced plastic. Compos. Sci. Technol. 2019, 173, 66–72. [Google Scholar] [CrossRef]
- Chen, X.; Chen, S.; Xu, Z.; Zhang, J.; Miao, M.; Zhang, D. Degradable and recyclable bio-based thermoset epoxy resins. Green Chem. 2020, 22, 4187–4198. [Google Scholar] [CrossRef]
- Hanaoka, T.; Arao, Y.; Kayaki, Y.; Kuwata, S.; Kubouchi, M. New approach to recycling of epoxy resins using nitric acid: Regeneration of decomposed products through hydrogenation. ACS Sustain. Chem. Eng. 2021, 9, 12520–12529. [Google Scholar] [CrossRef]
- Memon, H.; Wei, Y.; Zhu, C. Recyclable and reformable epoxy resins based on dynamic covalent bonds–Present, past, and future. Polym. Test. 2022, 105, 107420. [Google Scholar] [CrossRef]
- Das, M.; Chacko, R.; Varughese, S. An efficient method of recycling of CFRP waste using peracetic acid. ACS Sustain. Chem. Eng. 2018, 6, 1564–1571. [Google Scholar] [CrossRef]
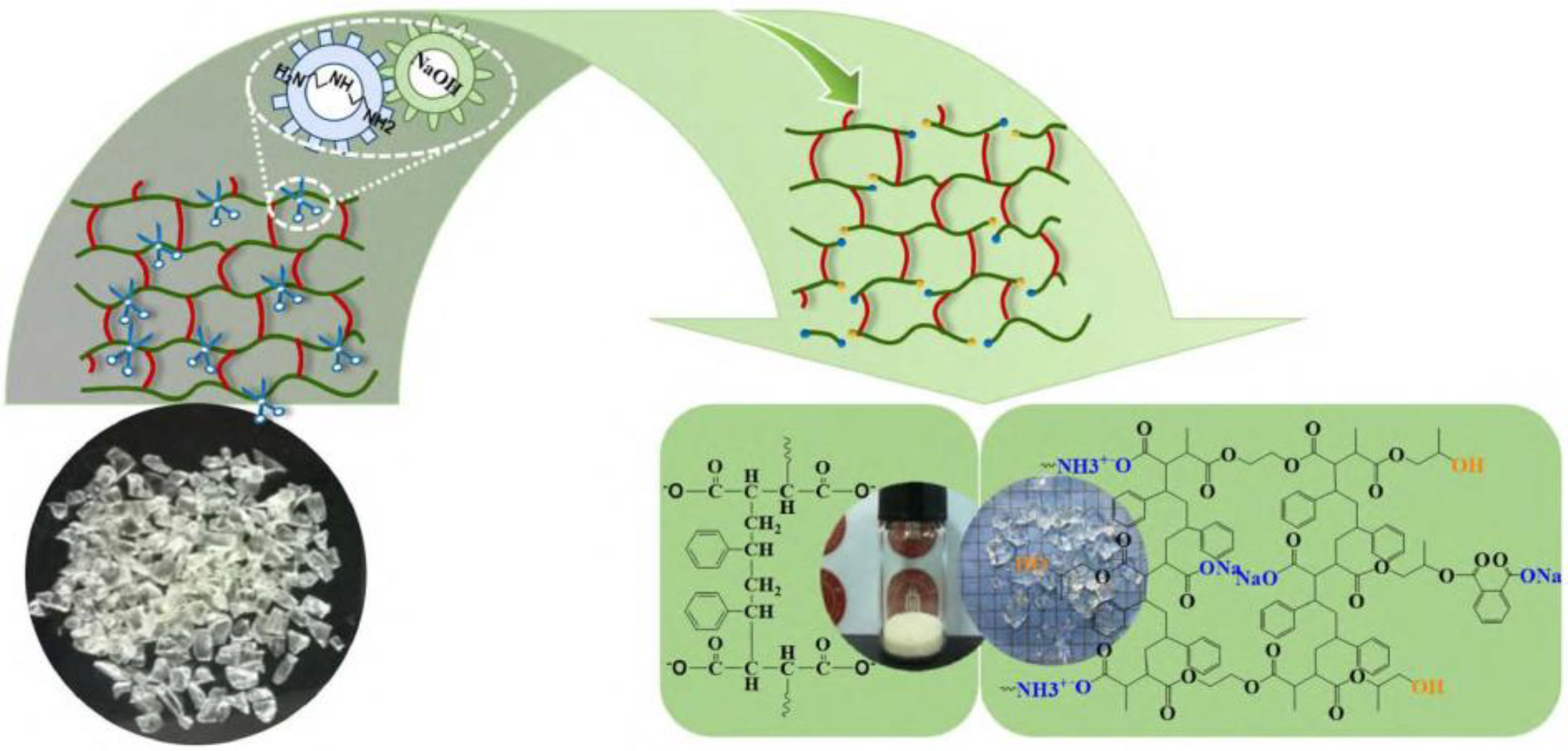
- Wang, Y.; Cui, X.; Yang, Q.; Deng, T.; Wang, Y.; Yang, Y.; Jia, S.; Qin, Z.; Hou, X. Chemical recycling of unsaturated polyester resin and its composites via selective cleavage of the ester bond. Green Chem. 2015, 17, 4527–4532. [Google Scholar] [CrossRef]
- An, W.; Wang, X.L.; Yang, Y.; Xu, H.; Xu, S.; Wang, Y.Z. Synergistic catalysis of binary alkalis for the recycling of unsaturated polyester under mild conditions. Green Chem. 2019, 21, 3006–3012. [Google Scholar] [CrossRef]
- An, W.; Wang, X.L.; Liu, X.; Wu, G.; Xu, S.; Wang, Y.Z. Chemical recovery of thermosetting unsaturated polyester resins. Green Chem. 2022, 24, 701–712. [Google Scholar] [CrossRef]


| Component | Original Design | Improved Design | Packaging Form | Applicable Products | Brand Name |
|---|---|---|---|---|---|
| Barrier layer | aluminized PET multilayer composites | Mono-polyolefin HDPE * PET PP | soft tube blister pack skin packaging packaging film | tooth paste cosmetics cheese fruit paste vegetable paste milk powder | Nestle Colgate Essel Gerber |
| Adhesive | solvent based/ pressure-sensitive/melt adhesive | washable adhesive self-adhesive non-glue structure | bottle bucket can flexible package express package | daily chemical/beauty products drinks/milk machine oil commodity | UPM Raflatac Avery Dennison China Post Amazon |
| Label | PVC shrink label paper label multi-layer label | PET shrink label PETG * shrink label wood-based PE Peelable label No label label reduction Laser printing and embossing Electronic tag | bottle container flexible pack | Mouth wash coffee/tea/juice carbonated/ isotonic drink gum | ORION Darlie Master Kong Eastroc Beverage Coca Cola Pepsi Pulpy Orange Evian |
| Pump | metal spring metal/glass bead PP/PE | All-plastic pump 100% PP 100% PE PP/r-PP | bottle container | daily chemical/ cleaning products medicine skin care | Tianzhou Aptar Group Berry Global Rieke Packaging |
| Color | green/blue white/black opaque | transparent unpigmented | bottle | coffee/juice carbonated drink milk/tea daily chemical skin care | Spite Fido Amcor Unilever Cnnice |
| Attachment | separated cap aluminum foil | attached cap EVA * TPE * | bottle soft bag | drinks sauce | Alpla Sidel Coca Cola |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, Q.; Zhu, H. The Key to Solving Plastic Packaging Wastes: Design for Recycling and Recycling Technology. Polymers 2023, 15, 1485. https://doi.org/10.3390/polym15061485
Ding Q, Zhu H. The Key to Solving Plastic Packaging Wastes: Design for Recycling and Recycling Technology. Polymers. 2023; 15(6):1485. https://doi.org/10.3390/polym15061485
Chicago/Turabian StyleDing, Qian, and Heping Zhu. 2023. "The Key to Solving Plastic Packaging Wastes: Design for Recycling and Recycling Technology" Polymers 15, no. 6: 1485. https://doi.org/10.3390/polym15061485
APA StyleDing, Q., & Zhu, H. (2023). The Key to Solving Plastic Packaging Wastes: Design for Recycling and Recycling Technology. Polymers, 15(6), 1485. https://doi.org/10.3390/polym15061485





