Investigation of Morphological, Chemical, and Thermal Properties of Biodegradable Food Packaging Films Synthesised by Direct Utilisation of Cassava (Monihot esculanta) Bagasse
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Film Preparation
2.2.1. Preparation of Film-Forming Mixture
2.2.2. Synthesis of Cassava Bagasse-Based Biodegradable Food Packaging Film
2.3. Film Characterisation
2.3.1. Macroscopic Characteristics
2.3.2. SEM Analysis
2.3.3. Fourier Transform Infrared (FTIR) Analysis
2.3.4. Thermogravimetric Analysis
3. Results and Discussion
3.1. Macroscopic View
3.2. Surface Morphology of the Films
3.3. Availability of Functional Groups
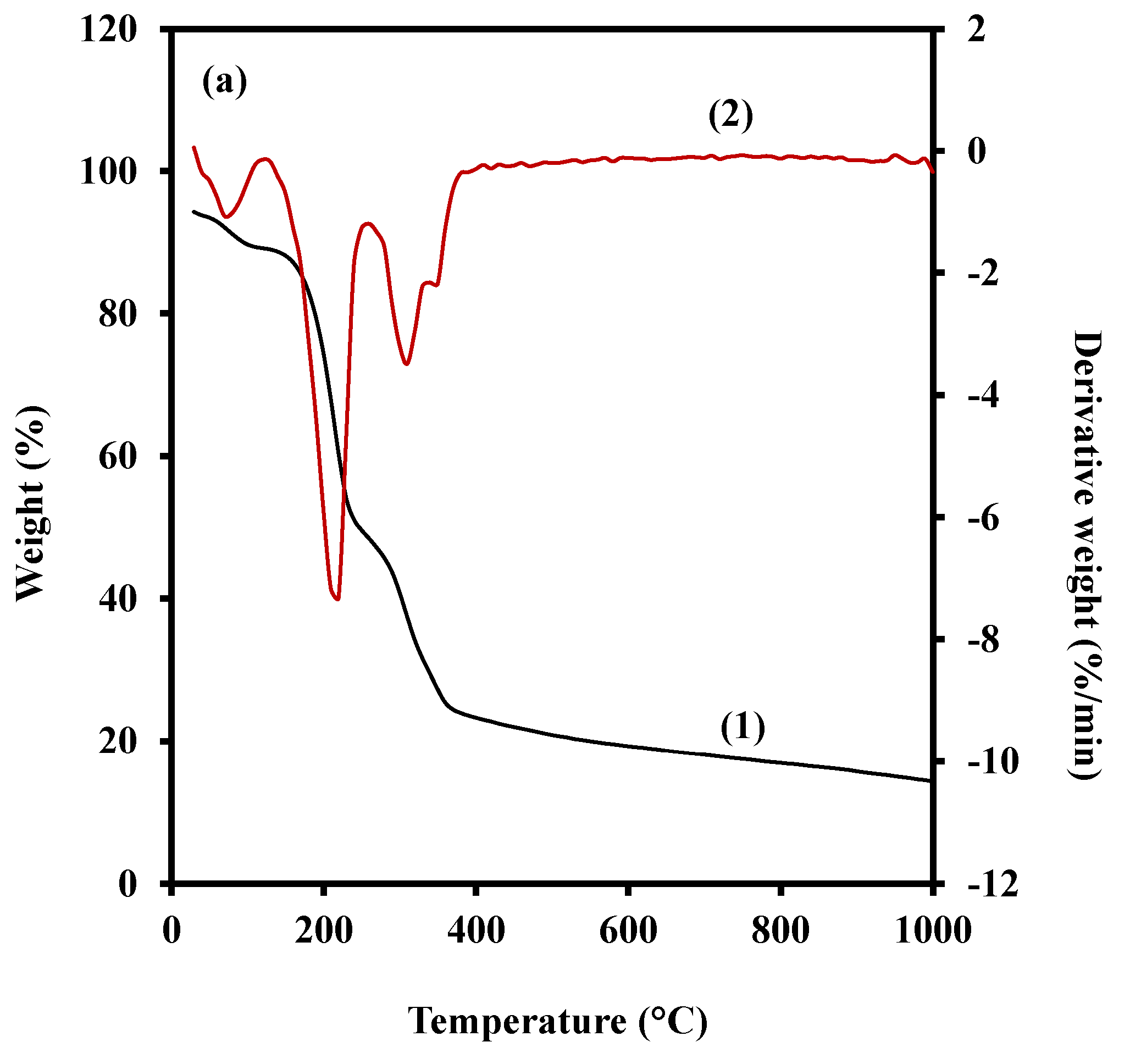
3.4. Thermal Stability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gopinathar, P.; Prabha, G.; Ravichandra, D.K. The Role of Packaging in Manufacturing—A Brief Understanding. IOSR J. Bus. Manag. 2016, 18, 1–7. [Google Scholar]
- Guillard, V.; Gaucel, S.; Fornaciari, C.; Angellier-Coussy, H.; Buche, P.; Gontard, N. The Next Generation of Sustainable Food Packaging to Preserve Our Environment in a Circular Economy Context. Front. Nutr. 2018, 5, 121. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, S.A.; Zannou, O.; Bahmid, N.A.; Fidan, H.; Alamou, A.-F.; Nagdalian, A.A.; Hassoun, A.; Fernando, I.; Ibrahim, S.A.; Arsyad, M. Consumer behavior towards nanopackaging—A new trend in the food industry. Future Foods 2022, 6, 100191. [Google Scholar] [CrossRef]
- Salgado, P.R.; Di Giorgio, L.; Musso, Y.S.; Mauri, A.N. Recent Developments in Smart Food Packaging Focused on Biobased and Biodegradable Polymers. Front. Sustain. Food Syst. 2021, 5, 630393. [Google Scholar]
- Asgher, M.; Qamar, S.A.; Bilal, M.; Iqbal, H.M.N. Bio-based active food packaging materials: Sustainable alternative to conventional petrochemical-based packaging materials. Food Res. Int. 2020, 137, 109625. [Google Scholar] [PubMed]
- Oliveira, C.F.d.P.; Fidalgo, N.N.; Valera, T.S. Thermoplastic Starch: Preparation Method. In ANTEC 2014, Proceedings of the Technical Conference & Exhibition, Las Vegas, NV, USA, 28–30 April 2014; Society of Plastics Engineers: Danbury, CT, USA, 2014; pp. 431–434. [Google Scholar]
- DeBruyn, J.; Bandopadhyay, S.; Hayes, D.; Inglis, D.; Miles, C. Performance and Adoptability Biodegradable Mulch. 2015. Available online: https://ag.tennessee.edu/biodegradablemulch/Documents/biodegradation_factsheet.pdf (accessed on 20 October 2021).
- Kumar Tiwari, A.; Gautam, M.; Maurya, H.K. Recent Development of Biodegradation Techniques of Polymer. Int. J. Res. Granthaalayah 2018, 6, 414–452. [Google Scholar] [CrossRef]
- Tang, X.; Alavi, S. Structure and physical properties of starch/poly vinyl alcohol/laponite RD nanocomposite films. J. Agric. Food Chem. 2012, 60, 1954–1962. [Google Scholar] [CrossRef]
- Shahabi-Ghahfarrokhi, I.; Goudarzi, V.; Babaei-Ghazvini, A. Production of starch-based biopolymer by green photochemical reaction at different UV region as a food packaging material: Physicochemical characterization. Int. J. Biol. Macromol. 2019, 122, 201–209. [Google Scholar] [CrossRef]
- Sudheesh, C.; Sunooj, K.V.; Sasidharan, A.; Sabu, S.; Basheer, A.; Navaf, M.; Raghavender, C.; Sinha, S.K.; George, J. Energetic neutral N2 atoms treatment on the kithul (Caryota urens) starch biodegradable film: Physico-chemical characterization. Food Hydrocoll. 2020, 103, 105650. [Google Scholar] [CrossRef]
- Torrenegra, M.; Solano, R.; Herrera, A.; León, G. Preparation of biodegradable films based on modified Colombian starches from Ipomoea batatas, Manihot esculenta, Dioscorea rotundata and Zea mays. Mater. Technol. 2019, 34, 157–166. [Google Scholar] [CrossRef]
- Xu, A.; Guo, K.; Liu, T.; Bian, X.; Zhang, L.; Wei, C. Effects of Different Isolation Media on Structural and Functional Properties of Starches from Root Tubers of Purple, Yellow and White Sweet Potatoes. Molecules 2018, 23, 2135. [Google Scholar] [CrossRef]
- Swapna Joseph, C.; Harish Prashanth, K.V.; Rastogi, N.K.; Indiramma, A.R.; Yella Reddy, S.; Raghavarao, K.S.M.S. Optimum Blend of Chitosan and Poly-(ε-caprolactone) for Fabrication of Films for Food Packaging Applications. Food Bioprocess Technol. 2009, 4, 1179–1185. [Google Scholar] [CrossRef]
- Tripathi, S.; Mehrotra, G.K.; Dutta, P.K. Physicochemical and bioactivity of cross-linked chitosan-PVA film for food packaging applications. Int. J. Biol. Macromol. 2009, 45, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Zehra, A.; Wani, S.M.; Jan, N.; Bhat, T.A.; Rather, S.A.; Malik, A.R.; Hussain, S.Z. Development of chitosan-based biodegradable films enriched with thyme essential oil and additives for potential applications in packaging of fresh collard greens. Sci. Rep. 2022, 12, 16923. [Google Scholar] [CrossRef]
- Taktak, W.; Kchaou, H.; Hamdi, M.; Li, S.; Nasri, M.; Karra-Chaâbouni, M.; Nasri, R. Design of Bioinspired Emulsified Composite European Eel Gelatin and Protein Isolate-Based Food Packaging Film: Thermal, Microstructural, Mechanical, and Biological Features. Coatings 2020, 10, 26. [Google Scholar] [CrossRef]
- Xiao, Y.; Liu, Y.; Kang, S.; Wang, K.; Xu, H. Development and evaluation of soy protein isolate-based antibacterial nanocomposite films containing cellulose nanocrystals and zinc oxide nanoparticles. Food Hydrocoll. 2020, 106, 105898. [Google Scholar] [CrossRef]
- Gvozdenko, A.A.; Siddiqui, S.A.; Blinov, A.V.; Golik, A.B.; Nagdalian, A.A.; Maglakelidze, D.G.; Statsenko, E.N.; Pirogov, M.A.; Blinova, A.A.; Sizonenko, M.N.; et al. Synthesis of CuO nanoparticles stabilized with gelatin for potential use in food packaging applications. Sci Rep. 2022, 12, 12843. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Niu, X.; Yang, J.; Fan, R.; Shi, J.; Ullah, N.; Feng, X.; Chen, L. Active biodegradable films based on the whole potato peel incorporated with bacterial cellulose and curcumin. Int. J. Biol. Macromol. 2020, 150, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Zahan, K.A.; Azizul, N.M.; Mustapha, M.; Tong, W.Y.; Abdul Rahman, M.S.; Sahuri, I.S. Application of bacterial cellulose film as a biodegradable and antimicrobial packaging material. Mater. Today Proc. 2020, 31, 83–88. [Google Scholar] [CrossRef]
- Amin, M.R.; Chowdhury, M.A.; Kowser, M.A. Characterization and performance analysis of composite bioplastics synthesized using titanium dioxide nanoparticles with corn starch. Heliyon 2019, 5, e02009. [Google Scholar] [CrossRef]
- Rahman, R.; Sood, M.; Gupta, N.; Bandral, J.D.; Hameed, F.; Ashraf, S. Bioplastics for Food Packaging: A Review. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 2311–2321. [Google Scholar] [CrossRef]
- Weligama Thuppahige, V.T.; Karim, M.A. A comprehensive review on the properties and functionalities of biodegradable and semi biodegradable food packaging materials. Compr. Rev. Food Sci. Food Saf. 2022, 21, 689–718. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.C.; Elias, T.M.; Ribeiro, K.d.O.; Soares, M.S., Jr.; Caliari, M. Microbiological and physicochemical profiles of the sour cassava starch and bagasse obtained from cassava agroindustry. Food Sci. Technol. 2019, 39, 803–809. [Google Scholar] [CrossRef]
- da Silva, É.C.; Cereda, M.P.; Denck Colman, T.A.; Mottin Demiate, I.; Schnitzler, E. Characterisation of cassava bagasse in different granulometries from two starch processing plants. J. Microbiol. Biotechnol. Food Sci. 2015, 05, 99–102. [Google Scholar] [CrossRef]
- Silviana, S.; Brandon, P.B.; Silawanda, B.A. Preparation of Cassava Bagasse Starch-Based Biodegradable Film Reinforced with Chicken Feet Gelatin, Citric Acid as Crosslinker, and Glycerol as Plasticizer. Indones. J. Chem. 2018, 18, 688–695. [Google Scholar] [CrossRef]
- Silviana, S.; Dzulkarom, M.C. Synthesis of Cassava Bagasse Starch-Based Biocomposite Reinforced Woven Bamboo Fibre with Lime Juice as Crosslinker and Epoxidized Waste Cooking Oil (EWCO) as Bioplasticizer. J. Phys. Conf. Ser. 2019, 1295, 012076. [Google Scholar] [CrossRef]
- de Morais Teixeira, E.; Da Róz, A.L.; de Carvalho, A.J.F.; da Silva Curvelo, A.A. Preparation and Characterisation of Thermoplastic Starches from Cassava Starch, Cassava Root and Cassava Bagasse. Macromol. Symp. 2005, 229, 266–275. [Google Scholar] [CrossRef]
- Farias, F.O.; Jasko, A.C.; Colman, T.A.D.; Pinheiro, L.A.; Schnitzler, E.; Barana, A.C.; Demiate, I. Characterisation of Cassava Bagasse and Composites Prepared by Blending with Low-Density Polyethylene. Braz. Arch. Biol. Technol. 2014, 57, 821–830. [Google Scholar] [CrossRef]
- Leite, A.; Zanon, C.D.; Menegalli, F.C. Isolation and characterization of cellulose nanofibers from cassava root bagasse and peelings. Carbohydr. Polym 2017, 157, 962–970. [Google Scholar] [CrossRef]
- Versino, F.; García, M.A. Cassava (Manihot esculenta) starch films reinforced with natural fibrous filler. Ind. Crops Prod. 2014, 58, 305–314. [Google Scholar] [CrossRef]
- Lai, S.M.; Don, T.M.; Huang, Y.C. Preparation, and properties of biodegradable thermoplastic starch/poly (hydroxy butyrate) blends. J. Appl. Polym. Sci. 2006, 100, 2371–2379. [Google Scholar] [CrossRef]
- Gul, W.; Khan, A.; Shakoor, A. Impact of Hot Pressing Pressure on Medium Density Fiberboard (MDF) Performance. AAPPS Bull. 2018, Hal Archives 1, Hal-01612472. [Google Scholar]
- Kachel-Jakubowska, M.; Matwijczuk, A.; Gagoś, M. Analysis of the physicochemical properties of post-manufacturing waste derived from production of methyl esters from rapeseed oil. Int. Agrophys. 2017, 31, 175–182. [Google Scholar] [CrossRef]
- Cheng, H.; Chen, L.; McClements, D.J.; Yang, T.; Zhang, Z.; Ren, F.; Miao, M.; Tian, Y.; Jin, Z. Starch-based biodegradable packaging materials: A review of their preparation, characterization and diverse applications in the food industry. Trends Food Sci. Technol. 2021, 114, 70–82. [Google Scholar]
- Basiak, E.; Lenart, A.; Debeaufort, F. How Glycerol and Water Contents Affect the Structural and Functional Properties of Starch-Based Edible Films. Polymers 2018, 10, 412. [Google Scholar] [CrossRef]
- Hancock, B.C.; Zografi, G. The relationship between the glass transition temperature and the water content of amorphous pharmaceutical solids. Pharm. Res. 1994, 11, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Taghizadeh, A.; Favis, B.D. Effect of high molecular weight plasticizers on the gelatinization of starch under static and shear conditions. Carbohydr. Polym. 2013, 92, 1799–1808. [Google Scholar] [CrossRef]
- Ertugrul, U.; Tas, O.; Namli, S.; Oztop, M.H. A preliminary investigation of caramelisation and isomerisation of allulose at medium temperatures and alkaline pHs: A comparison study with other monosaccharides. Int. J. Food Sci. Technol. 2021, 56, 5334–5339. [Google Scholar] [CrossRef]
- Majzoobi, M.; Pesaran, Y.; Mesbahi, G.; Golmakani, M.T.; Farahnaky, A. Physical properties of biodegradable films from heat-moisture-treated rice flour and rice starch. Starch-Stärke 2015, 67, 1053–1060. [Google Scholar] [CrossRef]
- Ketiku, A.O.; Oyenuga, V.A. Changes in the Carbohydrate Constituents of Cassava Root-tuber (Manihot utilissima Pohl) During Growth. J. Sci. Food Agric. 1972, 23, 1451–1456. [Google Scholar] [CrossRef]
- Reis, K.C.; Pereira, J.; Smith, A.C.; Carvalho, C.W.P.; Wellner, N.; Yakimets, I. Characterization of polyhydroxybutyrate-hydroxyvalerate (PHB-HV)/maize starch blend films. J. Food Eng. 2008, 89, 361–369. [Google Scholar] [CrossRef]
- Jost, V.; Langowski, H.-C. Effect of different plasticisers on the mechanical and barrier properties of extruded cast PHBV films. Eur. Polym. J. 2015, 68, 302–312. [Google Scholar] [CrossRef]
- Righetti, M.C.; Cinelli, P.; Mallegni, N.; Stabler, A.; Lazzeri, A. Thermal and Mechanical Properties of Biocomposites Made of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and Potato Pulp Powder. Polymers 2019, 11, 308. [Google Scholar] [CrossRef]
- Versino, F.; López, O.V.; García, M.A. Sustainable use of cassava (Manihot esculenta) roots as raw material for biocomposites development. Ind. Crops Prod. 2015, 65, 79–89. [Google Scholar] [CrossRef]
- Versino, F.; Lopez, O.V.; Garcia, M.A.; Zaritzky, N.E. Starch-based films and food coatings: An overview. Starch-Stärke 2016, 68, 1026–1037. [Google Scholar] [CrossRef]
- Heydari, A.; Alemzadeh, I.; Vossoughi, M. Functional properties of biodegradable corn starch nanocomposites for food packaging applications. Mater. Des. 2013, 50, 954–961. [Google Scholar] [CrossRef]
- Choy, S.Y.; Prasad, K.M.; Wu, T.Y.; Raghunandan, M.E.; Yang, B.; Phang, S.M.; Ramanan, R.N. Isolation, characterization, and the potential use of starch from jackfruit seed wastes as a coagulant aid for treatment of turbid water. Environ. Sci. Pollut. Res. 2017, 24, 2876–2889. [Google Scholar] [CrossRef]
- Ferreira, D.C.M.; Molina, G.; Pelissari, F.M. Biodegradable trays based on cassava starch blended with agro-industrial residues. Compos. Part B Eng. 2020, 183, 107682. [Google Scholar] [CrossRef]
- Bodirlau, R.; Teaca, C.-A.; Spiridon, I. Influence of natural fillers on the properties of starch-based biocomposite films. Compos. Part B Eng. 2013, 44, 575–583. [Google Scholar] [CrossRef]
- Danish, M.; Mumtaz, M.W.; Fakhar, M.; Rashid, U. Response Surface Methodology: An Imperative Tool for the Optimized Purification of the Residual Glycerol from Biodiesel Production Process. Chiang Mai J. Sci. 2016, 43, 1–13. [Google Scholar]
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Ismarrubie Zahari, N. Preparation and Characterization of Cassava Starch/Peel Composite Film. Polym. Compos. 2018, 39, 1704–1715. [Google Scholar] [CrossRef]
- Luchese, C.L.; Benelli, P.; Spada, J.C.; Tessaro, I.C. Impact of the starch source on the physicochemical properties and biodegradability of different starch-based films. J. Appl. Polym. Sci. 2018, 135, 46564. [Google Scholar] [CrossRef]
- Guimarães, M.; Botaro, V.R.; Novack, K.M.; Teixeira, F.G.; Tonoli, G.H.D. High moisture strength of cassava starch/polyvinyl alcohol-compatible blends for the packaging and agricultural sectors. J. Polym. Res. 2015, 22, 192. [Google Scholar] [CrossRef]
- Aggarwal, P.; Dollimore, D.; Heon, K. Comparative thermal analysis of two biopolymers, starch and cellulose. J. Therm. Anal. 1997, 50, 7–17. [Google Scholar] [CrossRef]
- Maqsood, M.; Seide, G. Investigation of the Flammability and Thermal Stability of Halogen-Free Intumescent System in Biopolymer Composites Containing Biobased Carbonization Agent and Mechanism of Their Char Formation. Polymers 2018, 11, 48. [Google Scholar] [CrossRef] [PubMed]
- Ramgobin, A.; Fontaine, G.; Penverne, C.; Bourbigot, S. Thermal atability and fine properties of salen and mtallosalens as fire retardants in thermoplastic polyurethane (TPU). Material. 2017, 10, 665. [Google Scholar] [CrossRef] [PubMed]



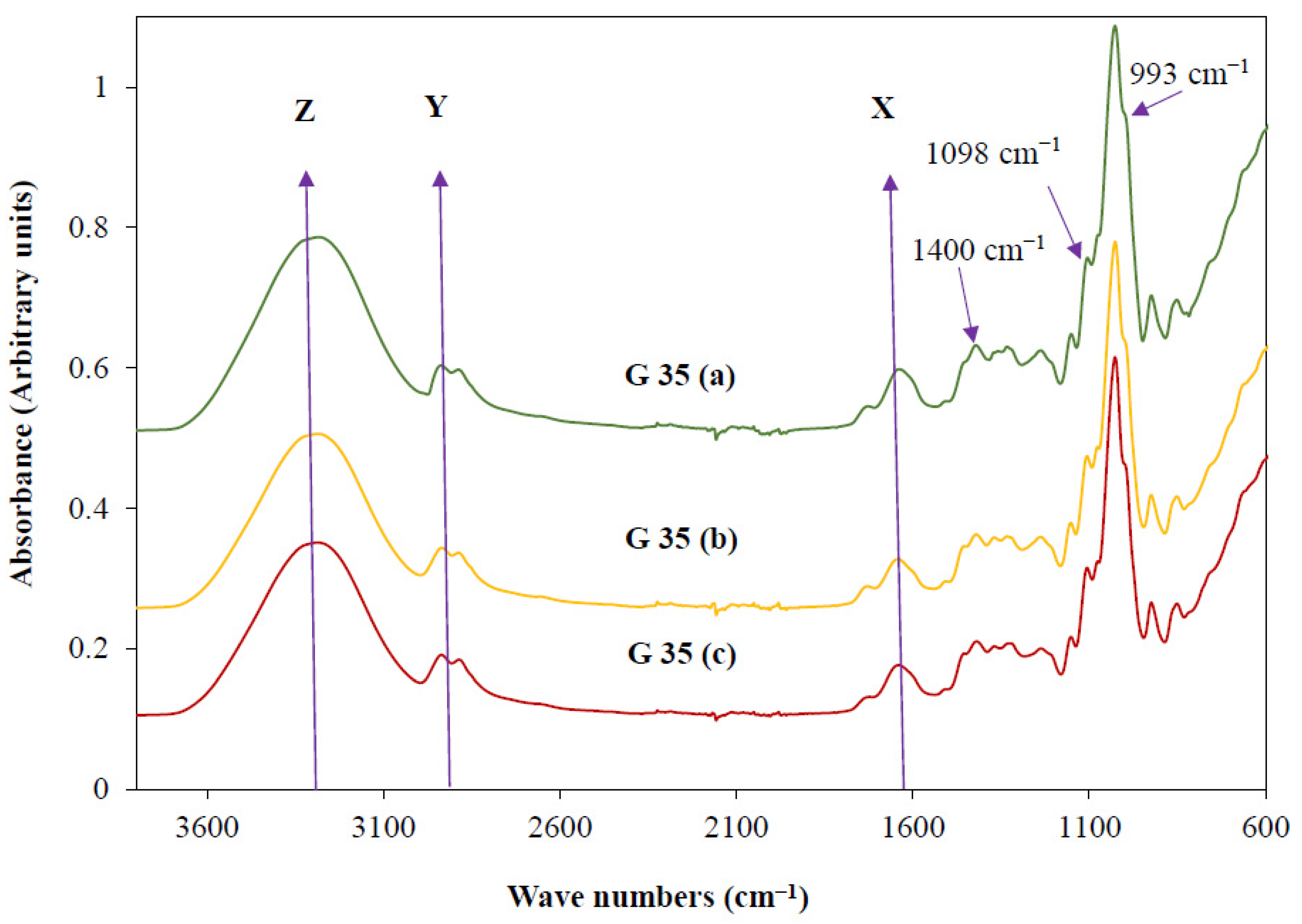

| Film | Powdered Cassava Bagasse 1 | Glycerol 1 | Water 1 | Glycerol + Water |
|---|---|---|---|---|
| G 30 | 47 | 30 | 23 | 53 |
| G 35 | 37 | 35 | 28 | 63 |
| Film | Stage 1 | Stage 2 | Stage 3 | Stage 4 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mass Loss | Residual Mass | Peak Temper-Ature | Mass Loss | Residual Mass | Peak Temper-Ature | Mass Loss | Residual Mass | Peak Temper-Ature | Mass Loss | Residual Mass | Peak Temper-Ature | |
| G 35 A | 5.04 | 89.10 | 97.6 | 40.50 | 48.60 | 178.6 | 25.11 | 23.48 | 355.2 | 9.23 | 14.24 | 567.1 |
| G 35 B | 4.00 | 90.09 | 91.9 | 37.73 | 52.36 | 179.1 | 24.90 | 27.46 | 348.3 | 12.02 | 15.44 | 993.6 |
| G 35 C | 4.09 | 89.73 | 30.6 | 40.63 | 49.10 | 175.9 | 26.08 | 23.03 | 352.8 | 12.37 | 10.65 | 995.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weligama Thuppahige, V.T.; Moghaddam, L.; Welsh, Z.G.; Karim, A. Investigation of Morphological, Chemical, and Thermal Properties of Biodegradable Food Packaging Films Synthesised by Direct Utilisation of Cassava (Monihot esculanta) Bagasse. Polymers 2023, 15, 767. https://doi.org/10.3390/polym15030767
Weligama Thuppahige VT, Moghaddam L, Welsh ZG, Karim A. Investigation of Morphological, Chemical, and Thermal Properties of Biodegradable Food Packaging Films Synthesised by Direct Utilisation of Cassava (Monihot esculanta) Bagasse. Polymers. 2023; 15(3):767. https://doi.org/10.3390/polym15030767
Chicago/Turabian StyleWeligama Thuppahige, Vindya Thathsaranee, Lalehvash Moghaddam, Zachary G. Welsh, and Azharul Karim. 2023. "Investigation of Morphological, Chemical, and Thermal Properties of Biodegradable Food Packaging Films Synthesised by Direct Utilisation of Cassava (Monihot esculanta) Bagasse" Polymers 15, no. 3: 767. https://doi.org/10.3390/polym15030767
APA StyleWeligama Thuppahige, V. T., Moghaddam, L., Welsh, Z. G., & Karim, A. (2023). Investigation of Morphological, Chemical, and Thermal Properties of Biodegradable Food Packaging Films Synthesised by Direct Utilisation of Cassava (Monihot esculanta) Bagasse. Polymers, 15(3), 767. https://doi.org/10.3390/polym15030767






