Influence of Process Parameters on the Characteristics of Additively Manufactured Parts Made from Advanced Biopolymers
Abstract
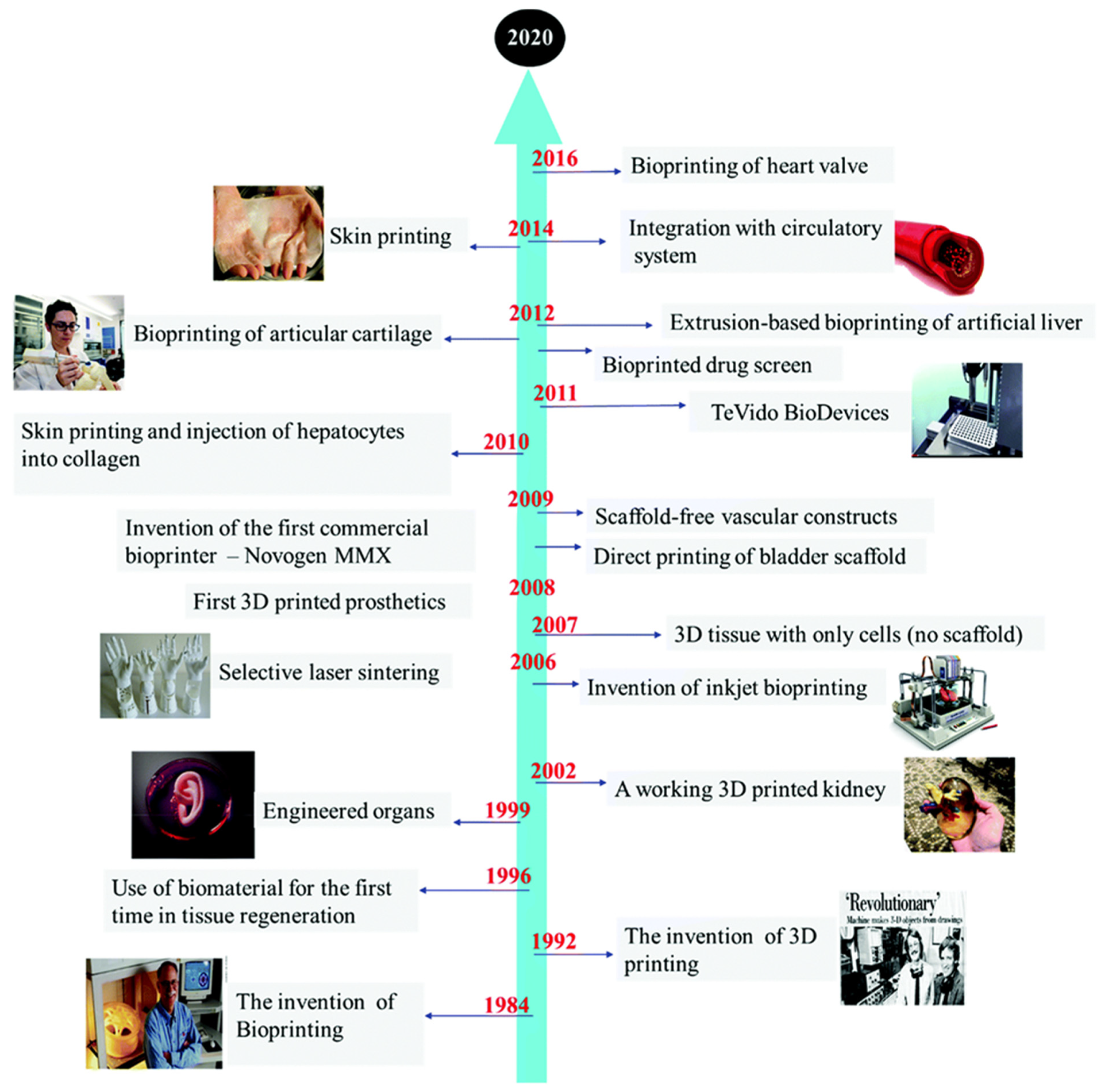
1. Introduction

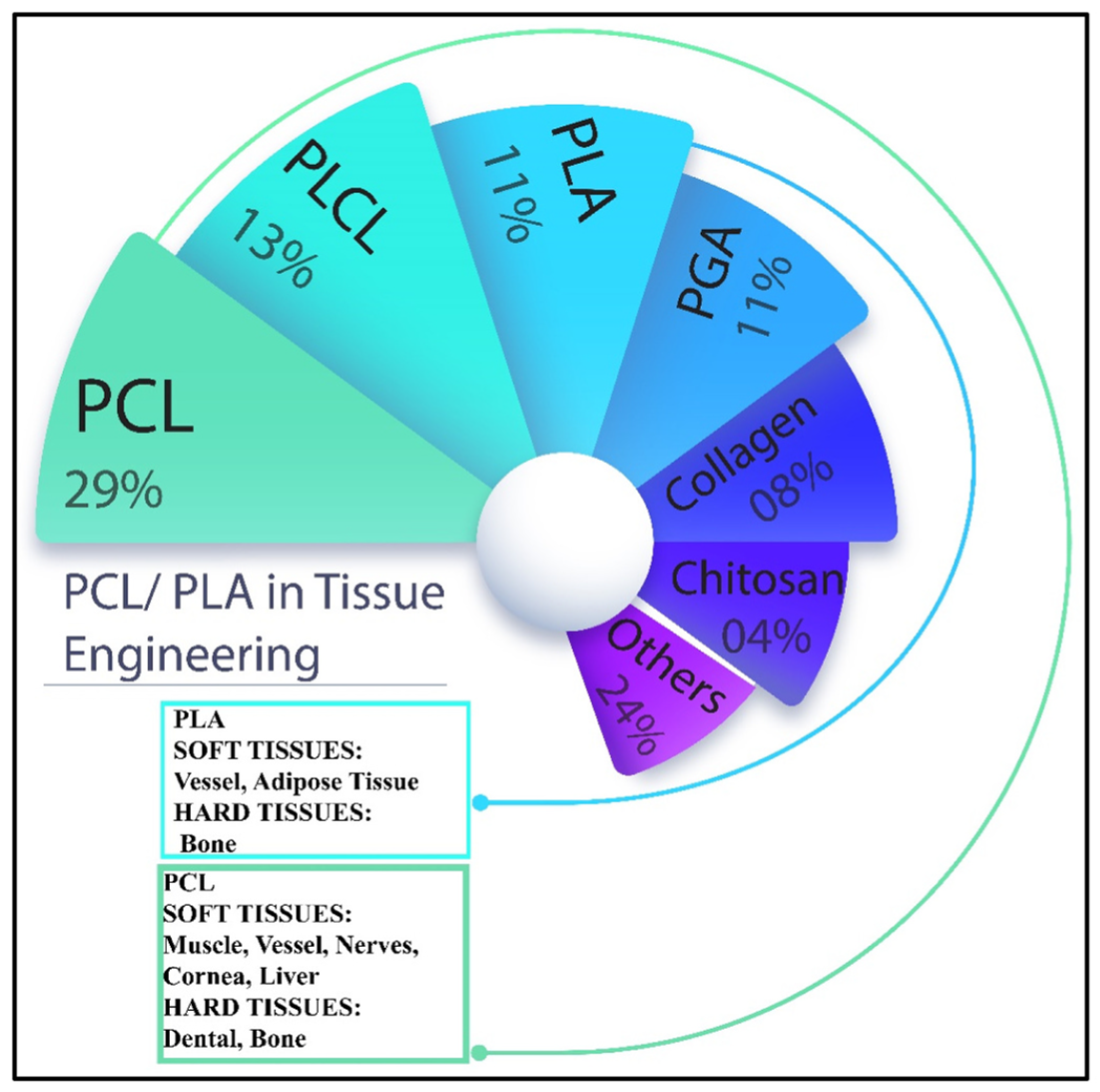
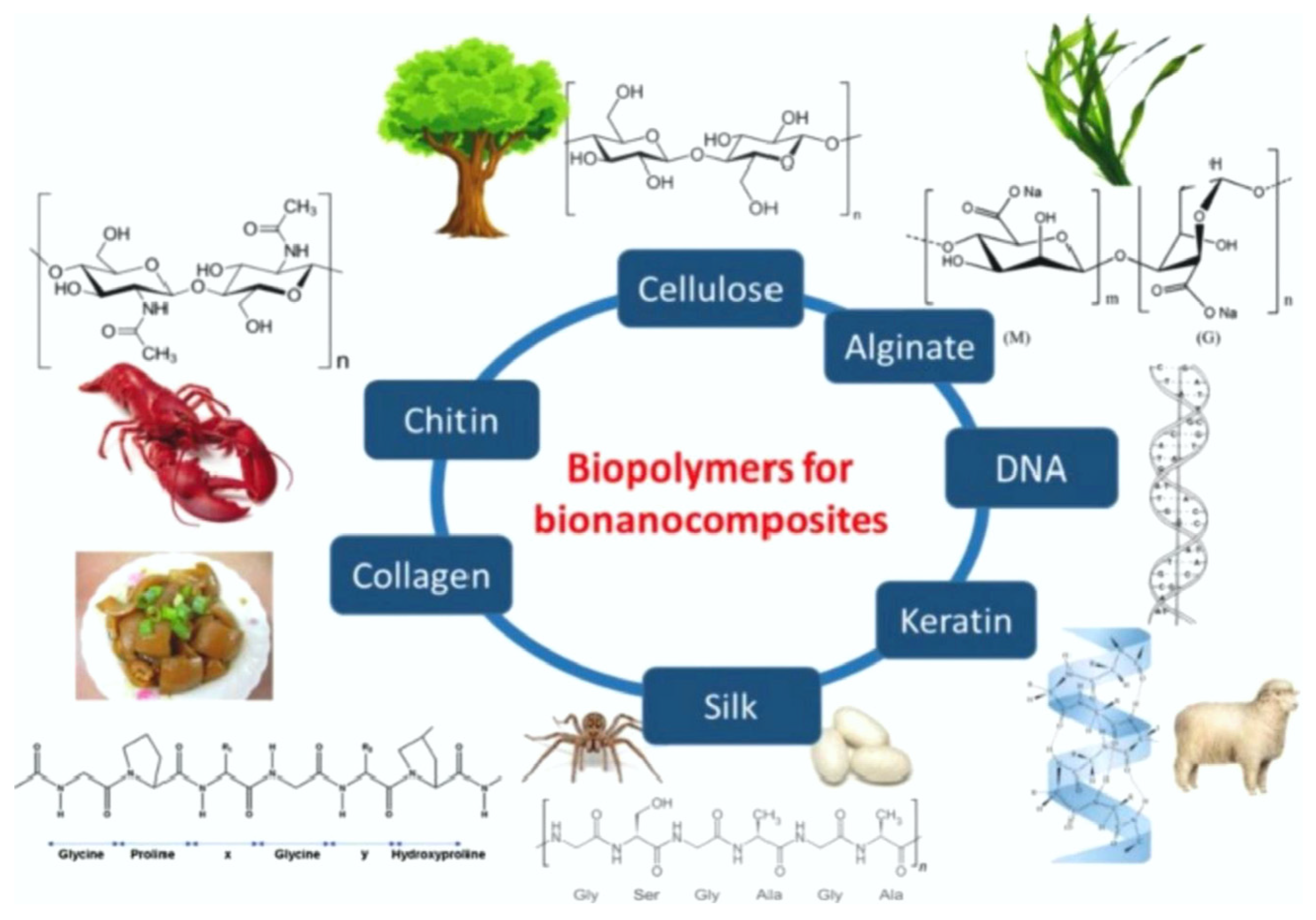
2. Biopolymer Materials
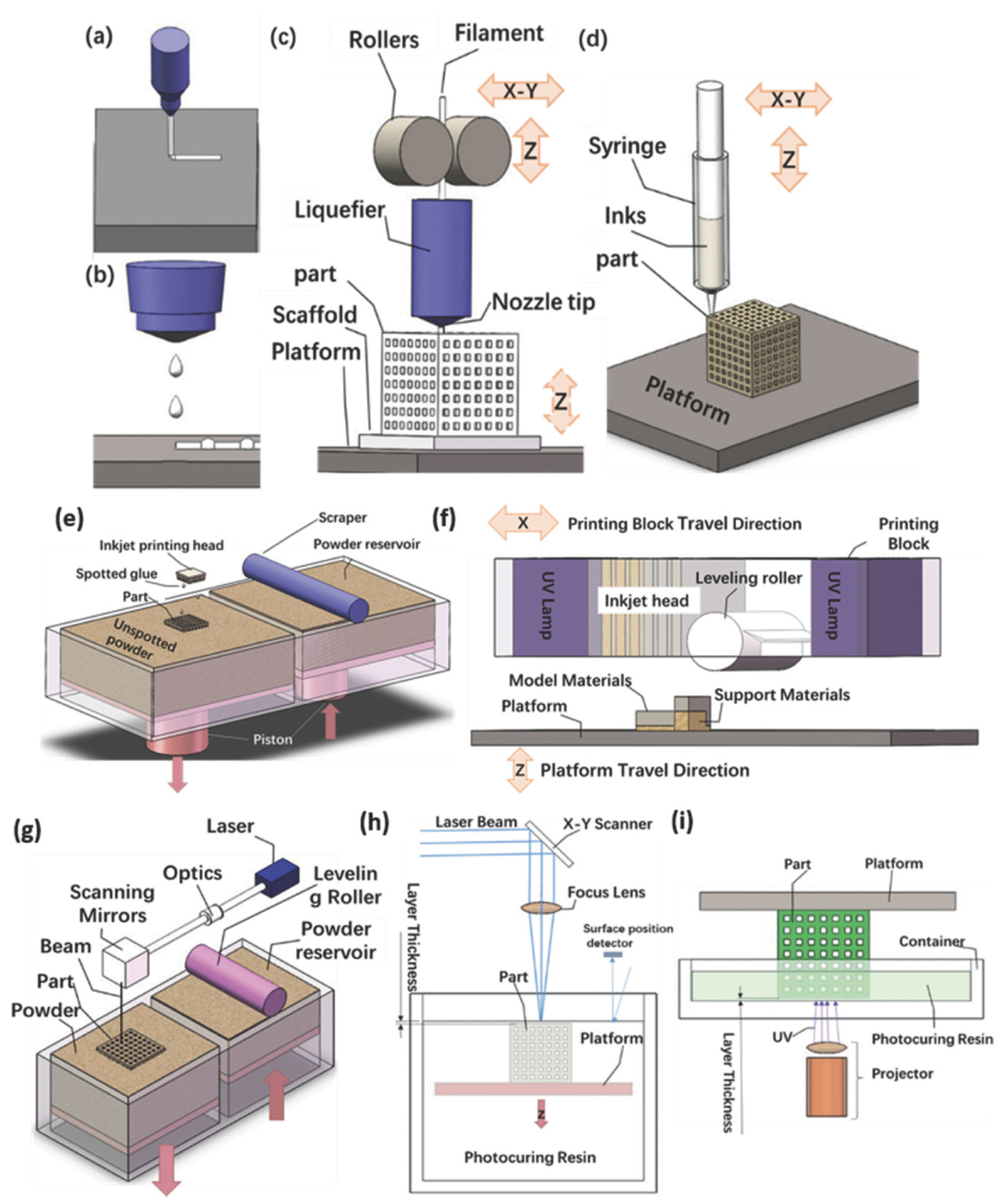
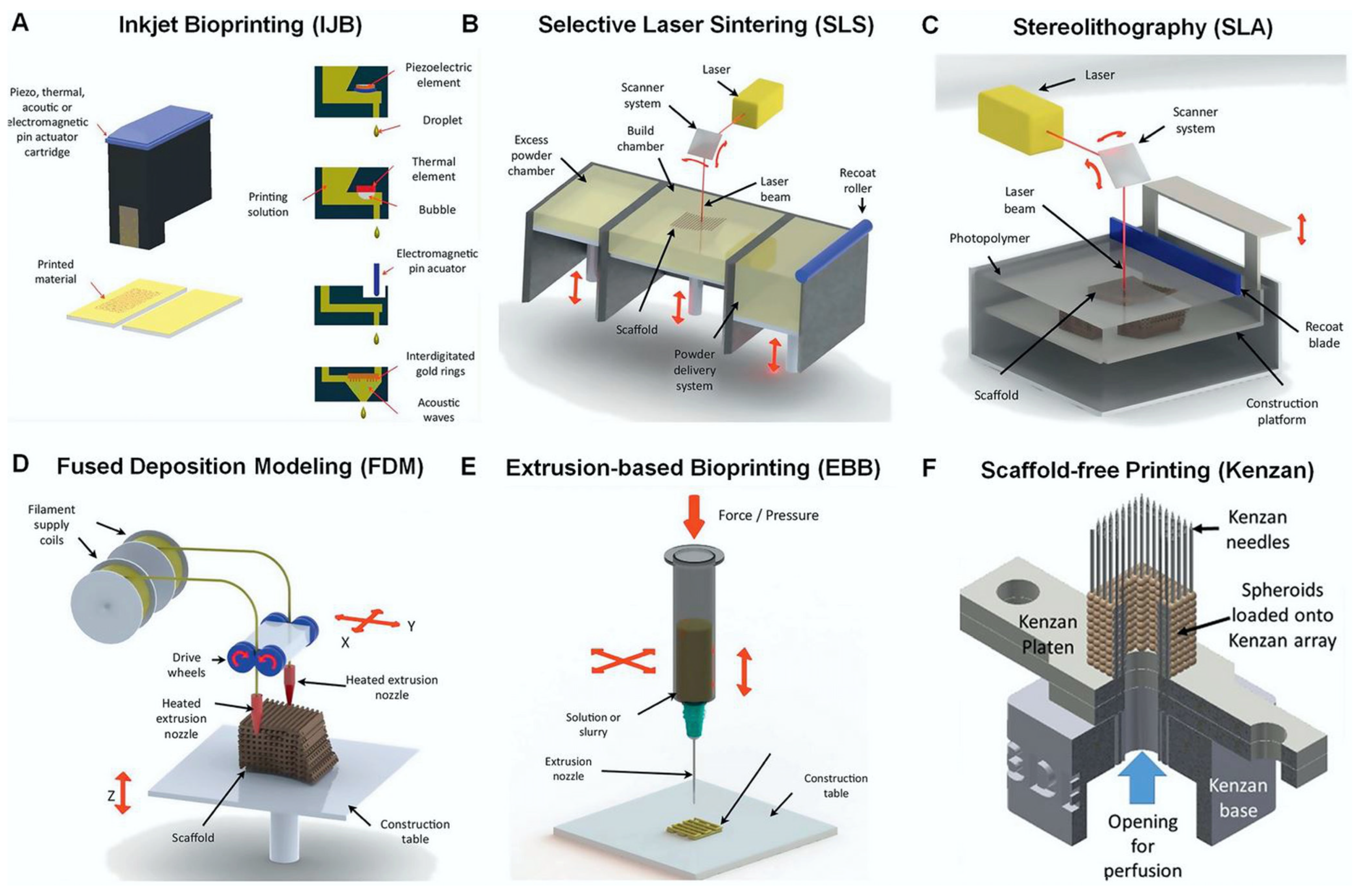
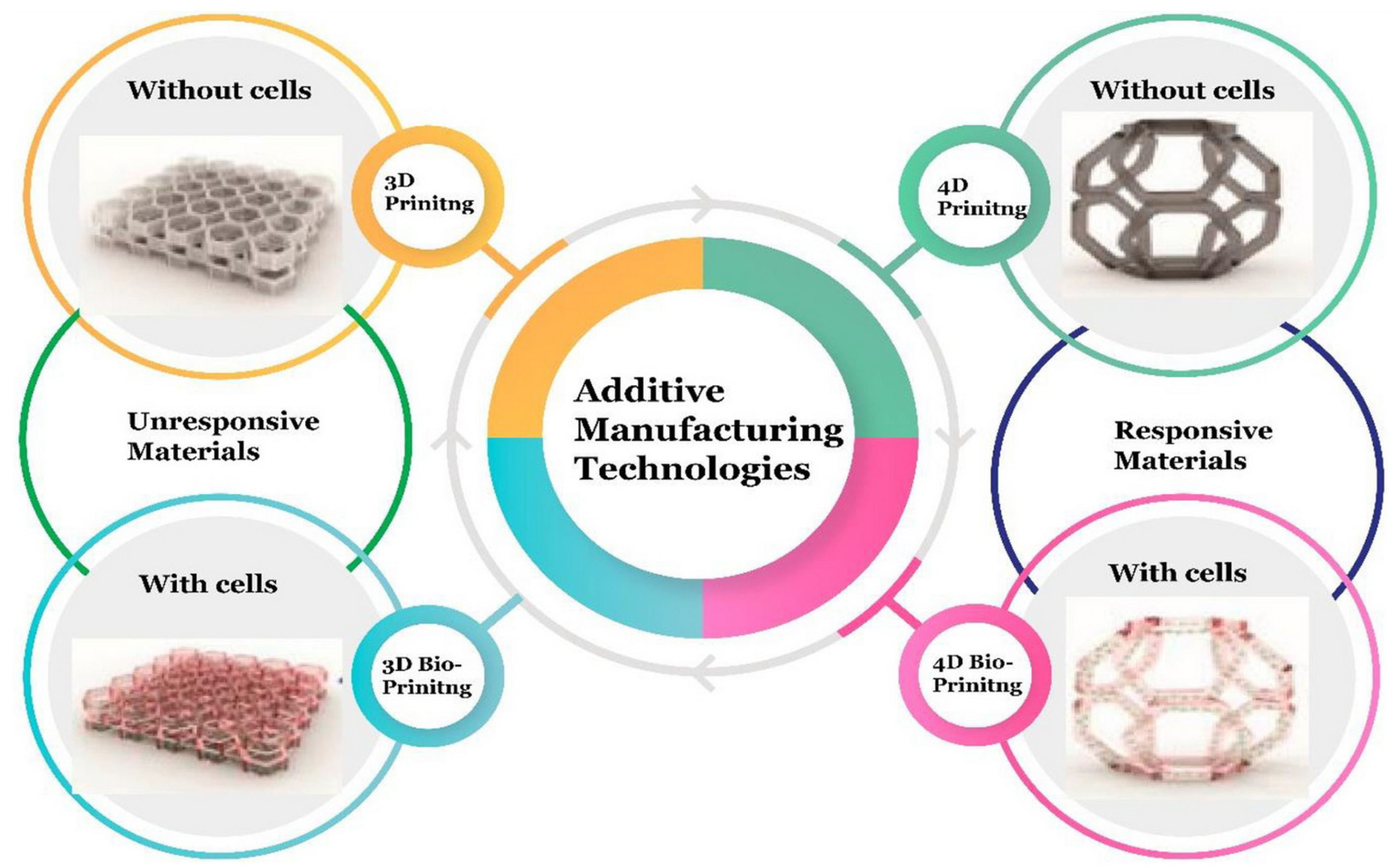
3. Additive Manufacturing Technologies
4. Manufacturing Parameters for AM of Advanced Biopolymers
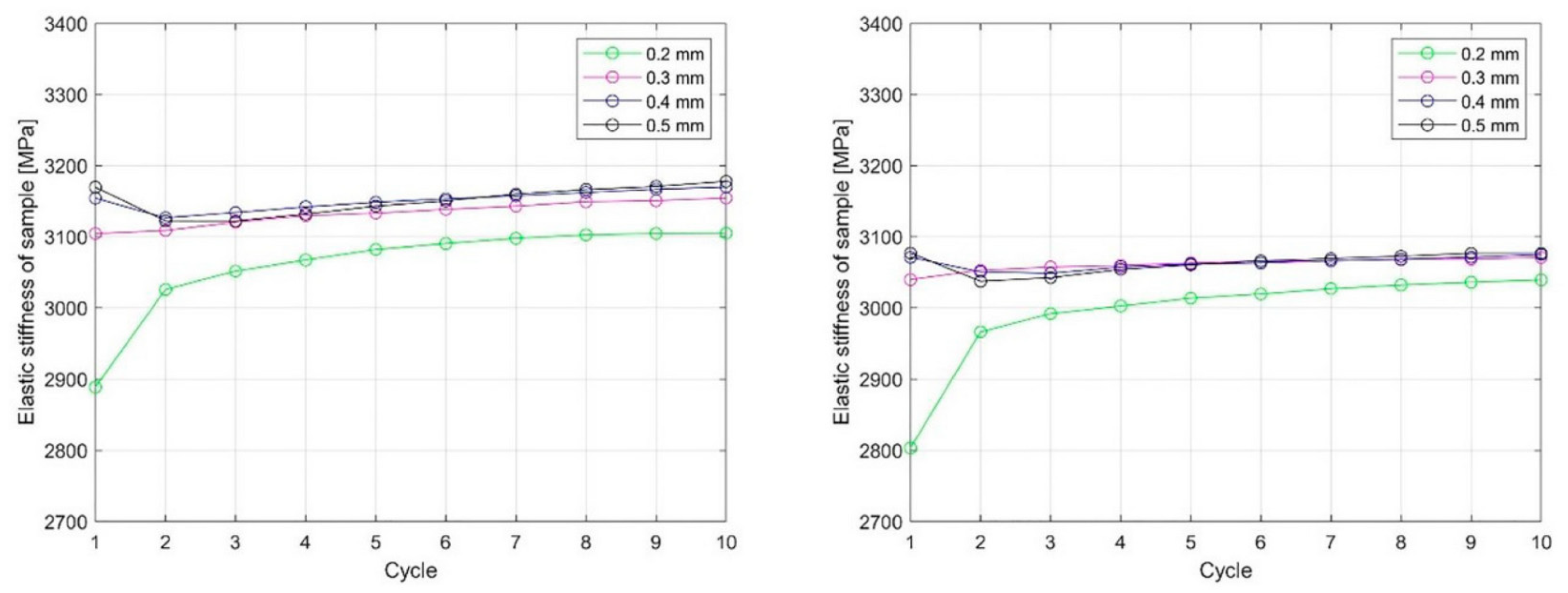
4.1. Vat-Based Polymerization Processes
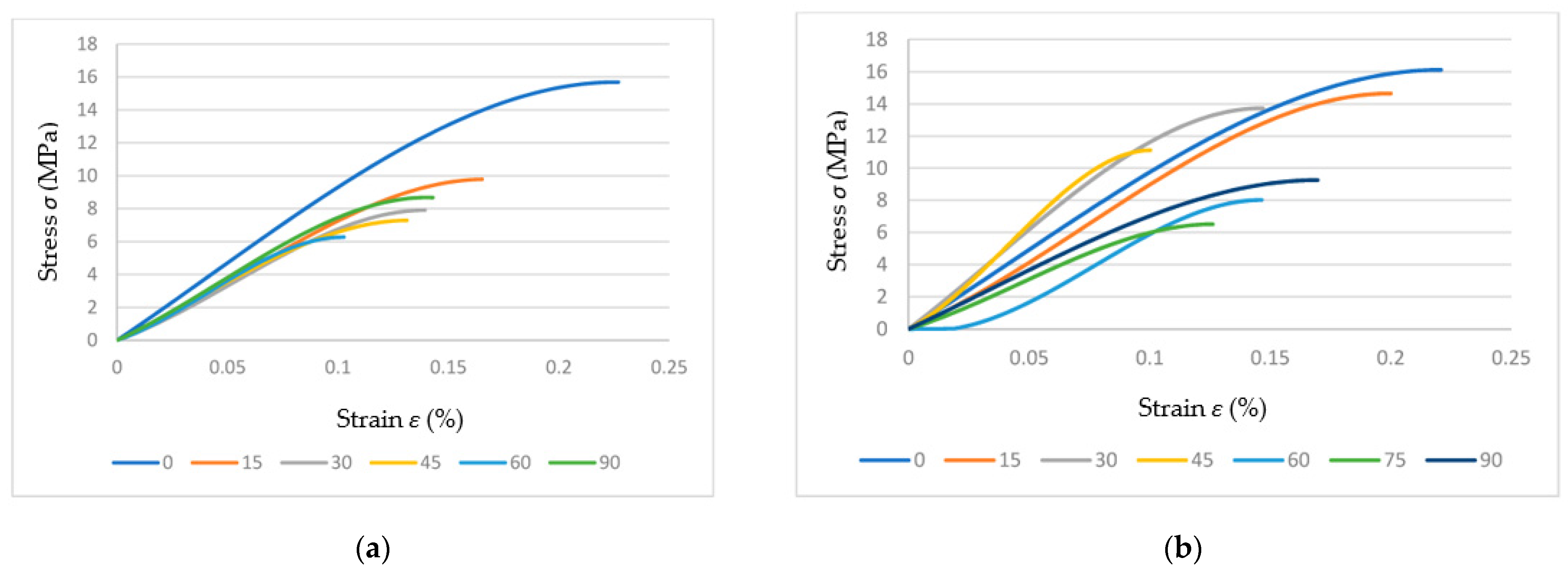
4.2. Fused Deposition Modeling (FDM)
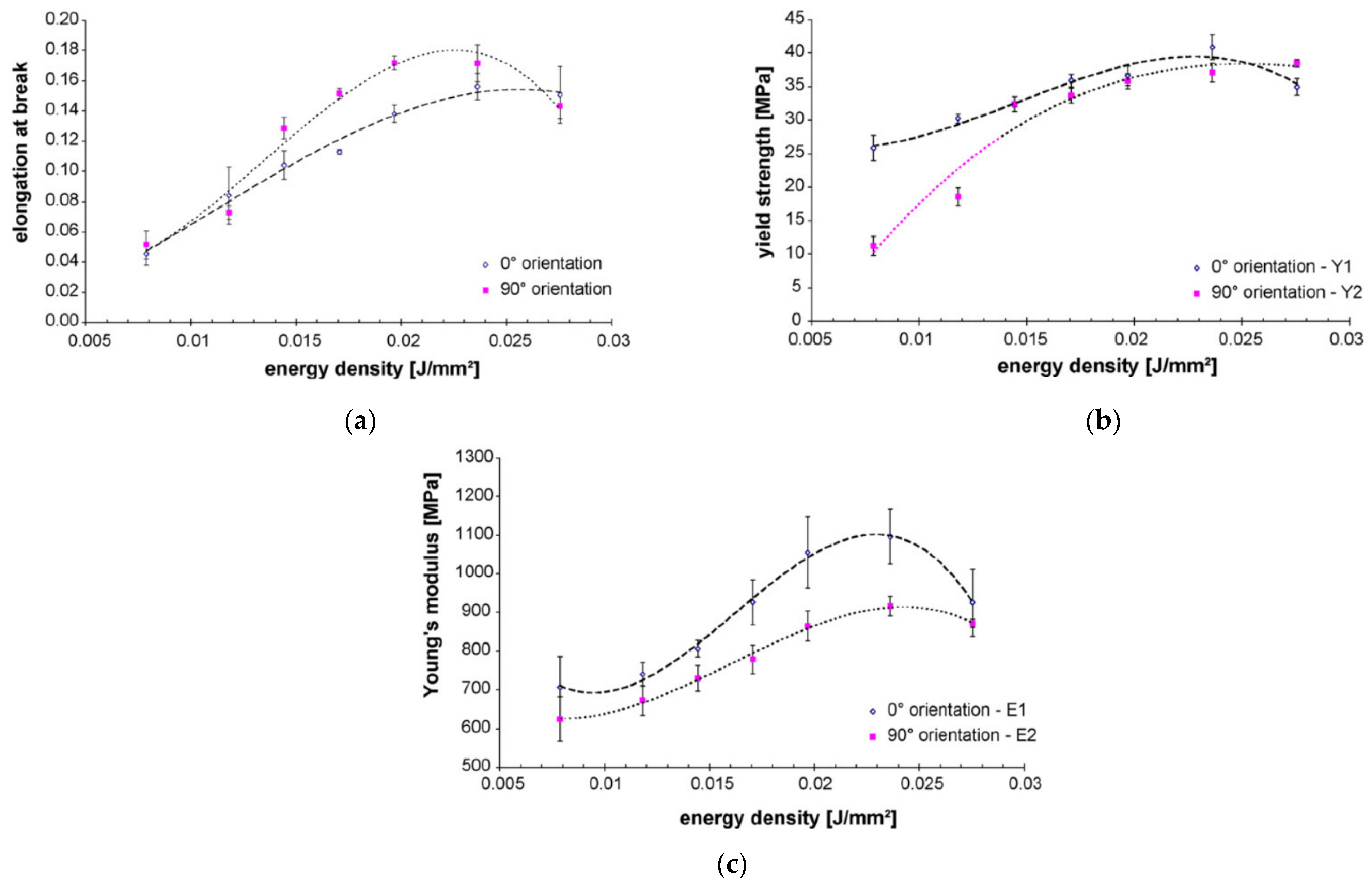
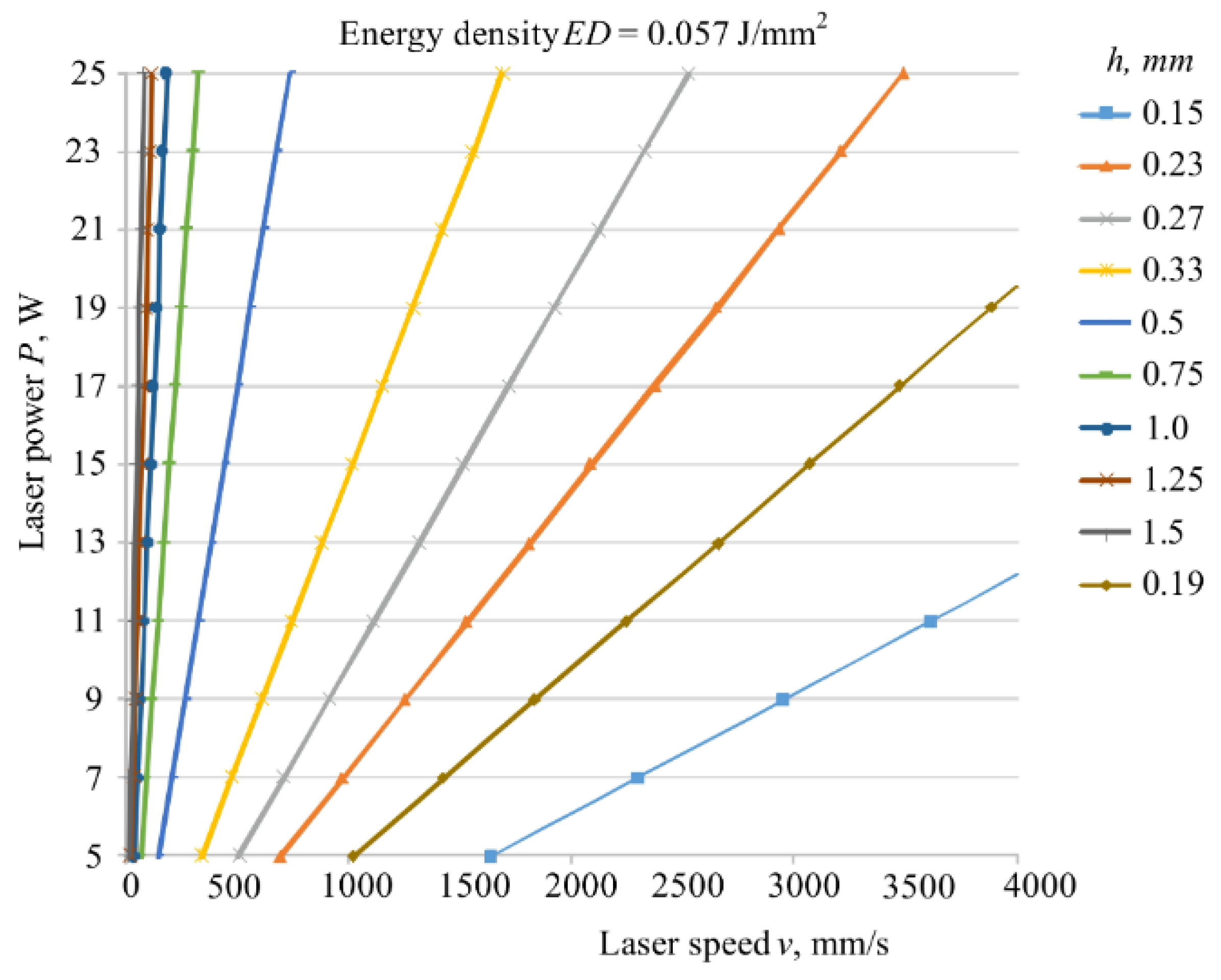
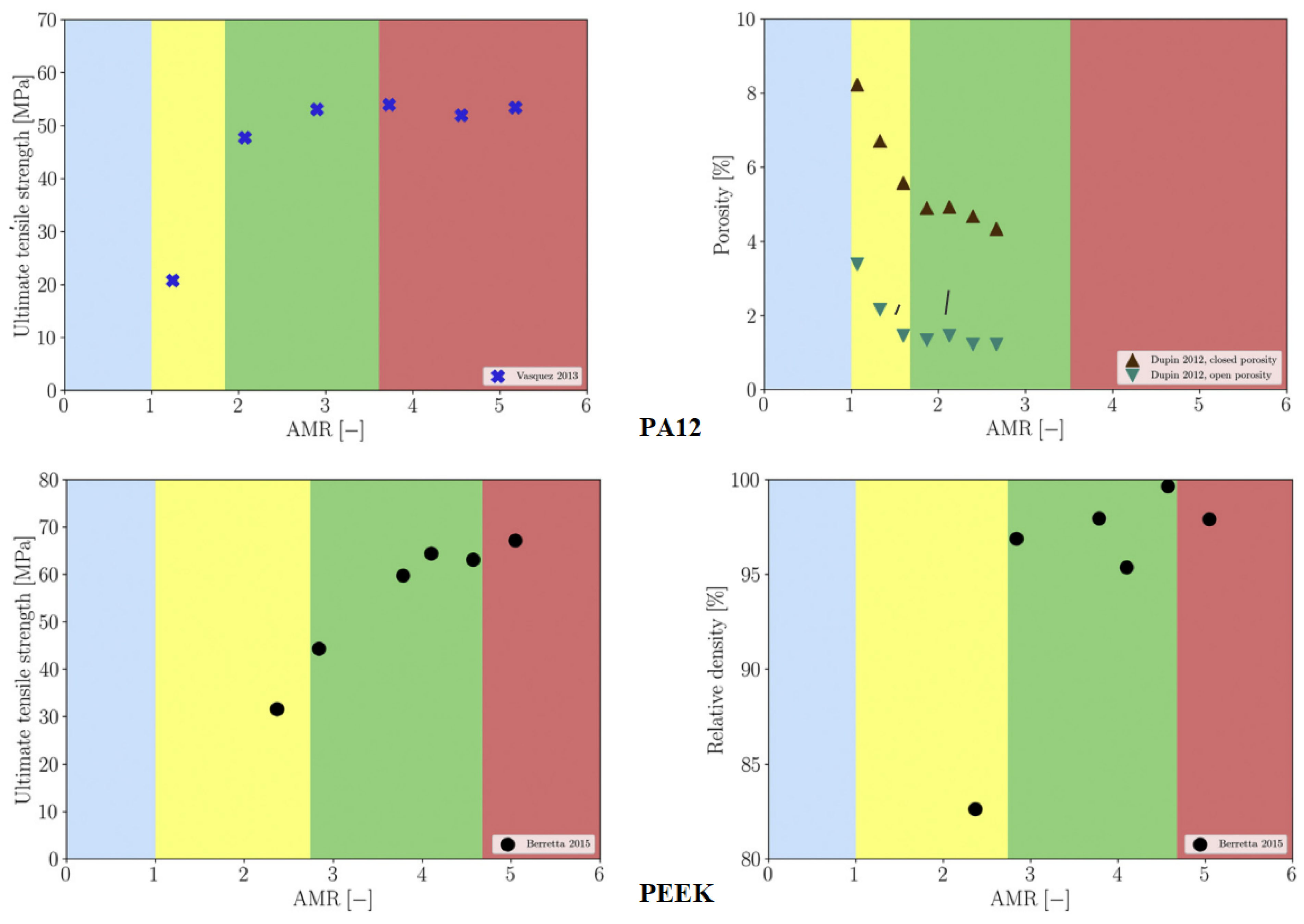
4.3. Selective Laser Sintering
5. Influence of the Structure and/or Inner Structure on Part Properties
6. Complexity of Additive Manufactured Parts
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 2PP | two-photon polymerization |
| 3D | 3-dimensional |
| 4D | 4-dimensional |
| ABS | acrylonitrile butadiene styrene |
| AM | additive manufacturing |
| AMR | attenuation melt ratio |
| ANOVA | analysis of variance |
| ASTM | American Society for Testing and Materials |
| BioPE | bio-based polyethylene |
| CAD | computer-aided design |
| CF | carbon fiber |
| Cu | copper |
| DLP | digital light processing |
| DMLS | direct metal laser sintering |
| DOE | design of experiments |
| EBB | extrusion-based bioprinting |
| EBM | electron-beam melting |
| FDM | fused deposition modeling |
| Fe | ferrum |
| FEA | finite element analysis |
| FFF | fused filament fabrication |
| HA | hydroxyapatite |
| HDPE | high-density polyethylene |
| LAP | lithium phenyl-2,4,6-trimethylbenzoylphosphinate |
| LENS | laser-engineered net shaping |
| LOM | laminated object manufacturing |
| LVI | low-velocity impact |
| Mg | magnesium |
| PA12 | polyamide 12 |
| PA6.10 | polyamide 6.10 |
| PA66 | polyamide 66 |
| PA2200 | polyamide 2200 |
| PCL | polycaprolactone |
| PDA | biopolymer polydopamine |
| PE | polyethylene |
| PEG | polyethylene glycol |
| PEGDA | polyethylene glycol diacrylate |
| PEEK | polyether ether ketone |
| PETG | polyethylene terephthalate glycol |
| PGA | polyglycolic acid |
| PHA | polyhydroxyalkanoate |
| PLA | polylactic acid |
| PLCL | poly(L-lactide-co-e-caprolactone) |
| PLGA | poly(lactide-co-glycolide) |
| PLLA | poly(l-lactide) |
| PMMA | poly(methyl methacrylate) |
| POM | polyoxymethylene |
| PP | polypropylene |
| PVA | polyvinyl alcohol |
| SEM | scanning electron microscope |
| SHS | selective heat sintering |
| SLA | stereolithography |
| SLM | selective laser melting |
| SLS | selective laser sintering |
| SMM | shape-memory material |
| SMP | shape-memory polymer |
| TCP | tricalcium phosphate |
| TE | tissue engineering |
| Ti | titanium |
| US FDA | United States Food and Drug Administration |
| UV | ultraviolet |
| WF | wood flour |
| WOS | Web of Science |
References
- Saleh Alghamdi, S.; John, S.; Roy Choudhury, N.; Dutta, N.K. Additive Manufacturing of Polymer Materials: Progress, Promise and Challenges. Polymers 2021, 13, 753. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Zhang, Y.; Ramanujan, D.; Ramani, K.; Chen, Y.; Christopher, B.; Williams, C.; Wang, C.L.; Yung, C.; Song Zhang, S.; et al. The status, challenges, and future of additive manufacturing in engineering. Comput. Aided Des. 2015, 69, 65–89. [Google Scholar] [CrossRef]
- ISO/ASTM 52900:2021; Additive Manufacturing—General Principles—Fundamentals and Vocabulary. ISO: Geneva, Switzerland, 2021.
- Vishnumaya Varma, M.; Kandasubramanian, B.; Ibrahim, S.M. 3D printed scaffolds for biomedical applications. Mater. Chem. Phys. 2020, 255, 123642. [Google Scholar] [CrossRef]
- Available online: http://www.dpm.ftn.uns.ac.rs/sr/vesti/722-humanost-na-delu-kroz-zajednicki-rad-idealab-ova-u-regionu (accessed on 7 December 2022).
- Zhakeyev, A.; Wang, P.; Zhang, L.; Shu, W.; Wang, H.X. Additive Manufacturing: Unlocking the Evolution of Energy Materials. J. Adv. Sci. 2017, 4, 1700187. [Google Scholar] [CrossRef] [PubMed]
- Dickens, P.M. Research Developments in Rapid Prototyping. Proc. Inst. Mech. Eng. Part B J. Eng. Manuf. 1995, 209, 261–266. [Google Scholar] [CrossRef]
- Jacobs, P.F. Rapid prototyping & manufacturing: Fundamentals of stereolithography. In Society of Manufacturing Engineers; SME: Southfield, MI, USA, 1992; pp. 196–221. Available online: https://sci-hub.ru/10.1016/0278-6125(93)90311-g (accessed on 7 December 2022).
- Hauber, D. Automatic production of P/M parts directly from a computer aided design model. Int. J. Powder Metal 1986, 24, 337–342. [Google Scholar]
- Hull, C. StereoLithography: Plastic prototypes from CAD data without tooling. Mod. Cast. 1988, 78, 38. [Google Scholar]
- Swainson, W.K. Method, Medium and Apparatus for Producing Three-Dimensional Figure Product. U.S. Patent 4041476, 23 July 1971. [Google Scholar]
- Swainson, W.K.; Kramer, D. Three-Dimensional Pattern Making. U.S. Patent 4333165, 1 December 1977. [Google Scholar]
- Hull, C.W. Apparatus for Production of Three-Dimensional Objects by Stereolithography. U.S. Patent 4575330, 8 August 1984. [Google Scholar]
- Fudim, E.V. Method and Apparatus for Production of Three-Dimensional Objects by Photosolidification. U.S. Patent 4801477, 29 September 1987. [Google Scholar]
- Zivanović, S.; Tabaković, S.; Randjelović, S. Rapid prototyping of art sculptural shapes according to the sample. J. Adv. Technol. Mat. 2019, 44, 27–32. [Google Scholar] [CrossRef]
- Bonaldi, R. Functional finishes for high-performance apparel. In High-Perform. Apparel; Woodhead Publishing Series in Textiles; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; pp. 129–156. [Google Scholar] [CrossRef]
- Lee, J. A 3D Food Printing Process for the New Normal Era: A Review. Processes 2021, 9, 1495. [Google Scholar] [CrossRef]
- Agunbiade, A.O.; Song, L.; Agunbiade, O.J.; Ofoedu, C.E.; Chacha, J.S.; Duguma, H.T.; Hossaini, S.M.; Rasaq, W.A.; Shorstkii, I.; Osuji, C.M.; et al. Potentials of 3D extrusion-based printing in resolving food processing challenges: A perspective review. J. Food Process Eng. 2022, 45, e13996. [Google Scholar] [CrossRef]
- Liu, H.; Liu, Z.; Hao, S. Design of a Throat-extended FDM Extruder for Multi-axis 3D Printing. Stroj. Vest. J. Mechan. Eng. 2021, 67, 180–190. [Google Scholar] [CrossRef]
- Valentinčič, J.; Sevšek, L.; Prijatelj, M.; Sabotin, I.; Jerman, M.; Lebar, A. Towards Production of Microfeatures on a Custom-Made Stereolitographic DLP Printer. Proc. Manuf. Syst. 2018, 13, 51–55. [Google Scholar]
- Valentinčič, J.; Prijatelj, M.; Jerman, M.; Lebar, A.; Sabotin, I. Characterization of a custom-made digital light processing stereolithographic printer based on a slanted groove micromixer geometry. J. Micro Nano Manuf. 2020, 8, 010911-1–010911-6. [Google Scholar] [CrossRef]
- Taghizadeh, M.; Taghizadeh, A.; Yazdi, M.; Zarrintaj, P.; Stadler, F.; Ramsey, J.; Habibzadeh, S.; Rad, S.H.; Naderi, G.; Saeb, M.R.; et al. Chitosan-based inks for 3D printing and bioprinting. Green Chem. 2022, 24, 62–101. [Google Scholar] [CrossRef]
- Qasim, M.; Chae, D.S.; Lee, N.Y. Advancements and frontiers in nano-based 3D and 4D scaffolds for bone and cartilage tissue engineering. Int. J. Nanomed. 2019, 14, 4333–4351. [Google Scholar] [CrossRef] [PubMed]
- Puppi, D.; Chiellini, F. Biodegradable Polymers for Biomedical Additive Manufacturing. Appl. Mater. Today 2020, 20, 100700. [Google Scholar] [CrossRef]
- Mohan, D.; Teong, Z.K.; Bakir, A.N.; Sajab, M.S.; Kaco, H. Extending Cellulose-Based Polymers Application in Additive Manufacturing Technology: A Review of Recent Approaches. Polymers 2020, 12, 1876. [Google Scholar] [CrossRef]
- Cometa, S.; Bonifacio, M.A.; Tranquillo, E.; Gloria, A.; Domingos, M.; De Giglio, E. A 3D Printed Composite Scaffold Loaded with Clodronate to Regenerate Osteoporotic Bone: In Vitro Characterization. Polymers 2021, 13, 150. [Google Scholar] [CrossRef]
- Balart, R.; Montanes, N.; Dominici, F.; Boronat, T.; Torres-Giner, S. Environmentally Friendly Polymers and Polymer Composites. Materials 2020, 13, 4892. [Google Scholar] [CrossRef]
- Dobrzyńska-Mizera, M.; Knitter, M.; Woźniak-Braszak, A.; Baranowski, M.; Sterzyński, T.; Di Lorenzo, M.L. Poly(l-Lactic Acid)/Pine Wood Bio-Based Composites. Materials 2020, 13, 3776. [Google Scholar] [CrossRef]
- Petković, D.; Madić, M.; Radenković, G. Knee Prosthesis Biomaterial Selection by Using MCDM Solver. J. Adv. Technol. Mat. 2021, 46, 37–41. [Google Scholar] [CrossRef]
- Calignano, F.; Manfredi, D.; Ambmbrosio, E.P.; Biamino, S.; Lombmbardi, M.; Atzeni, E.; Salmi, A.; Minetola, P.; Iuliano, L.; Fino, P. Overview on Additive Manufacturing Technologies. Proc. IEEE 2017, 105, 593–612. [Google Scholar] [CrossRef]
- Chicos, L.-A.; Pop, M.A.; Zaharia, S.-M.; Lancea, C.; Buican, G.R.; Pascariu, I.S.; Stamate, V.-M. Fused Filament Fabrication of Short Glass Fiber-Reinforced Polylactic Acid Composites: Infill Density Influence on Mechanical and Thermal Properties. Polymers 2022, 14, 4988. [Google Scholar] [CrossRef] [PubMed]
- Kozior, T.; Mamun, A.; Trabelsi, M.; Sabantina, L.; Ehrmann, A. Quality of the Surface Texture and Mechanical Properties of FDM Printed Samples after Thermal and Chemical Treatment. Stroj. Vest. J. Mech. Eng. 2020, 66, 105–113. [Google Scholar] [CrossRef]
- Beaman, J.J.; Barlow, J.W.; Bourell, D.L.; Crawford, R.H.; Marcus, H.L.; McAlea, K.P. Solid Freeform Fabrication: A New Direction in Manufacturing; Kluwer Academic Publishers: Norwell, MA, USA, 1997; Volume 2061, pp. 25–49. [Google Scholar]
- Pepelnjak, T.; Karimi, A.; Maček, A.; Mole, N. Altering the Elastic Properties of 3D Printed Poly-Lactic Acid (PLA) Parts by Compressive Cyclic Loading. Materials 2020, 13, 4456. [Google Scholar] [CrossRef]
- Dogan, O. Short-term Creep Behaviour of Different Polymers Used in Additive Manufacturing under Different Thermal and Loading Conditions. Stroj. Vest. J. Mech. Eng. 2022, 68, 451–460. [Google Scholar] [CrossRef]
- Karimi, A.; Mole, N.; Pepelnjak, T. Numerical Investigation of the Cycling Loading Behavior of 3D-Printed Poly-Lactic Acid (PLA) Cylindrical Lightweight Samples during Compression Testing. Appl. Sci. 2022, 12, 8018. [Google Scholar] [CrossRef]
- Fu, Z.; Ouyang, L.; Xu, R.; Yang, Y.; Sun, W. Responsive biomaterials for 3D bioprinting: A review. Mater. Today 2022, 52, 112–132. [Google Scholar] [CrossRef]
- Altıparmak, A.C.; Yardley, V.A.; Shi, Z.; Lin, J. Extrusion-based additive manufacturing technologies: State of the art and future perspectives. J. Manuf. Process. 2022, 83, 607–636. [Google Scholar] [CrossRef]
- Karki, S.; Gohain, S.M.B.; Yadav, D.; Ingole, P.G. Nanocomposite and bio-nanocomposite polymeric materials/membranes development in energy and medical sector: A review. Int. J. Biol. Macromol. 2021, 193-B, 2121–2139. [Google Scholar] [CrossRef]
- Zorko, D.; Demšar, I.; Tavčar, J. An investigation on the potential of bio-based polymers for use in polymer gear transmissions. Polym. Test. 2021, 93, 106994. [Google Scholar] [CrossRef]
- Reichert, C.L.; Bugnicourt, E.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, A.; Canesi, I.; Braca, F.; Martínez, B.M.; Alonso, R.; Agostinis, L.; et al. Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability. Polymers 2020, 12, 1558. [Google Scholar] [CrossRef]
- Wu, H.; Fahy, W.P.; Kim, S.; Kim, H.; Zhao, N.; Pilato, L.; Kafi, A.; Bateman, S.; Koo, J.H. Recent developments in polymers/polymer nanocomposites for additive manufacturing. Prog. Mater. Sci. 2022, 111, 100638. [Google Scholar] [CrossRef]
- Singamneni, S.; Behera, M.P.; Truong, D.; Le Guen, M.J.; Macrae, E.; Pickering, K. Direct extrusion 3D printing for a softer PLA-based bio-polymer composite in pellet form. J. Mater. Res. Technol. 2021, 15, 936–949. [Google Scholar] [CrossRef]
- Huang, S.; Fu, X. Naturally derived materials-based cell and drug delivery systems in skin regeneration. J. Control Release 2010, 142, 149–159. [Google Scholar] [CrossRef]
- Singh, N.; Singh, G. Advances in polymers for bio-additive manufacturing: A state of art review. J. Manuf. Process. 2021, 72, 439–457. [Google Scholar] [CrossRef]
- Peng, X.; Dong, K.; Wu, Z.; Wang, J.; Wang, L.W. A review on emerging biodegradable polymers for environmentally benign transient electronic skins. J. Mater. Sci. 2021, 56, 16765–16789. [Google Scholar] [CrossRef]
- Arif, Z.U.; Khalid, M.Y.; Noroozi, R.; Sadeghianmaryan, A.; Jalalvand, M.; Hossain, M. Recent advances in 3D-printed polylactide and polycaprolactone-based biomaterials for tissue engineering applications. Int. J. Biol. Macromol. 2022, 218, 930–968. [Google Scholar] [CrossRef]
- Ryan, K.R.; Down, M.P.; Hurst, N.J.; Edmund, M.; Keefe, E.M.; Banks, C.E. Additive manufacturing (3D printing) of electrically conductive polymers and polymer nanocomposites and their applications. eScience 2022, 2–4, 365–381. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, H.-J.; Kim, S.-Y.; Seok, J.M.; Lee, J.H.; Kim, W.D.; Kwon, I.K.; Shin-Young Park, S.-Y.; Park, S.A. In situ gold nanoparticle growth on polydopamine-coated 3D-printed scaffolds improves osteogenic differentiation for bone tissue engineering applications: In vitro and in vivo studies. Nanoscale 2018, 10, 15447–15453. [Google Scholar] [CrossRef]
- Miao, S.; Zhu, W.; Castro, N.; Nowicki, M.; Zhou, X.; Cui, H.; Fisher, J.P.; Zhang, L.G. 4D printing smart biomedical scaffolds with novel soybean oil epoxidized acrylate. Sci. Rep. 2016, 6, 27226. [Google Scholar] [CrossRef] [PubMed]
- González-Henríquez, C.M.; Sarabia-Vallejos, M.A.; Rodriguez-Hernandez, J. Polymers for additive manufacturing and 4D-printing: Materials, methodologies, and biomedical applications. Prog. Polym. Sci. 2019, 94, 57–116. [Google Scholar] [CrossRef]
- Lendlein, A.; Langer, R. Biodegradable, elastic shape-memory polymers for potential biomedical applications. Science 2002, 296, 1673–1676. [Google Scholar] [CrossRef] [PubMed]
- Kelch, S.; Steuer, S.; Schmidt, A.M.; Lendlein, A. Shape-Memory Polymer Networks from Oligo[(ε-hydroxycaproate)-co-glycolate]dimethacrylates and Butyl Acrylate with Adjustable Hydrolytic Degradation Rate. Biomacromolecules 2007, 8, 1018–1027. [Google Scholar] [CrossRef]
- Sokolowski, W.; Metcalfe, A.; Hayashi, S.; Yahia, L.; Raymond, J. Medical applications of shape memory polymers. Biomed. Mater. 2007, 2, 23–27. [Google Scholar] [CrossRef]
- Zhang, Y.-F.; Li, Z.; Li, H.; Li, H.; Xiong, Y.; Zhu, X.; Lan, H.; Ge, Q. Fractal-Based Stretchable Circuits via Electric-Field-Driven Microscale 3D Printing for Localized Heating of Shape Memory Polymers in 4D Printing. ACS Appl. Mater. Interfaces 2021, 13, 41414–41423. [Google Scholar] [CrossRef]
- Sonatkar, J.; Kandasubramanian, B.; Ismail, S.O. 4D printing: Pragmatic progression in biofabrication. Eur. Polym. J. 2022, 169, 111128. [Google Scholar] [CrossRef]
- Grigsby, W.J.; Scott, S.M.; Plowman-Holmes, M.I.; Middlewood, P.G.; Recabar, K. Combination and processing keratin with lignin as biocomposite materials for additive manufacturing technology. Acta Biomater. 2020, 104, 95–103. [Google Scholar] [CrossRef]
- Kirillova, A.; Maxson, R.; Stoychev, G.; Gomillion, C.T.; Ionov, L. 4D Biofabrication Using Shape-Morphing Hydrogels. Adv. Mater. 2017, 29, 1703443. [Google Scholar] [CrossRef] [PubMed]
- Berman, B. 3-D printing: The new industrial revolution. Bus. Horiz. 2012, 55, 155–162. [Google Scholar] [CrossRef]
- Lee, Y.-B.; Polio, S.; Lee, W.; Dai, G.; Menon, L.; Carroll, R.S.; Yoo, S.-S. Bio-printing of collagen and VEGF-releasing fibrin gel scaffolds for neural stem cell culture. Exp. Neurol. 2010, 223, 645–652. [Google Scholar] [CrossRef]
- Agarwal, T.; Chiesa, I.; Presutti, D.; Irawan, V.; Vajanthri, K.Y.; Costantini, M.; Nakagawa, Y.; Tan, S.A.; Makvandi, P.; Zare, E.N.; et al. Recent advances in bioprinting technologies for engineering different cartilage-based tissues. Mater. Sci. Eng. C 2021, 123, 112005. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Wang, S. Regulating MC3T3-E1 cells on deformable poly(ε-caprolactone) honeycomb films prepared using a surfactant-free breath figure method in a water-miscible solvent. ACS Appl. Mater. Interfaces 2012, 4, 4966–4975. [Google Scholar] [CrossRef] [PubMed]
- Jones, N. Three-Dimensional Printers Are Opening Up New Worlds to Research. Nature 2012, 487, 22–23. [Google Scholar] [CrossRef] [PubMed]
- Stewart, S.A.; Domínguez-Robles, J.; McIlorum, V.J.; Mancuso, E.; Lamprou, D.A.; Donnelly, R.F.; Larrañeta, E. Development of a biodegradable subcutaneous implant for prolonged drug delivery using 3D printing. Pharmaceutics 2020, 12, 105. [Google Scholar] [CrossRef]
- Jiang, R.; Wang, G.; Zhang, J.; Zhang, X.; Zhou, L.; Xu, T. Three-dimensional bioprinting of auricular cartilage: A review. Med. Drug Discov. 2019, 3, 100016. [Google Scholar] [CrossRef]
- Huang, J.; Chen, Z.; Wen, C.; Ling, T.; Chen, Z. Thermally assisted 3D printing of bio-polymer with high solute loading with improved mechanical properties. Addit. Manuf. 2022, 59, 103088. [Google Scholar] [CrossRef]
- Yu, Y.; Moncal, K.K.; Li, J.; Peng, W.; Rivero, I.; Martin, J.A.; Ozbolat, I.T. Three-dimensional bioprinting using self-Assembling scalable scaffold-free “tissue strands” as a new bioink. Sci. Rep. 2016, 6, 28714. [Google Scholar] [CrossRef]
- Murata, D.; Arai, K.; Nakayama, K. Scaffold-Free Bio-3D Printing Using Spheroids as “Bio-Inks” for Tissue (Re-)Construction and Drug Response Tests. Adv. Healthc. Mater. 2020, 9, 1901831. [Google Scholar] [CrossRef]
- Nadine, S.; Chung, A.; Diltemiz, S.E.; Yasuda, B.; Lee, C.; Hosseini, V.; Karamikamkar, S.; de Barros, N.R.; Mandal, K.; Advani, S.; et al. Advances in microfabrication technologies in tissue engineering and regenerative medicine. Artif. Organs 2022, 46, 211–243. [Google Scholar] [CrossRef]
- Qu, M.; Wang, C.; Zhou, X.; Libanori, A.; Jiang, X.; Xu, W.; Zhu, S.; Chen, Q.; Sun, W.; Khademhosseini, A. Multi-Dimensional Printing for Bone Tissue Engineering. Adv. Healthc. Mater. 2021, 10, 2001986. [Google Scholar] [CrossRef] [PubMed]
- Pieralli, S.; Spies, B.C.; Hromadnik, V.; Nicic, R.; Beuer, F.; Wesemann, C. How accurate is oral implant installation using surgical guides printed from a degradable and steam-sterilized biopolymer? J. Clin. Med. 2020, 9, 2322. [Google Scholar] [CrossRef] [PubMed]
- Raddatz, L.; de Vries, I.; Austerjost, J.; Lavrentieva, A.; Geier, D.; Becker, T.; Beutel, S.; Scheper, T. Additive manufactured customizable labware for biotechnological purposes. Eng. Life Sci. 2017, 17, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Aida, H.J.; Nadlene, R.; Mastura, M.T.; Yusriah, L.; Sivakumar, D.; Ilyas, R.A. Natural fibre filament for Fused Deposition Modelling (FDM): A review. Int. J. Sustain. Eng. 2021, 14, 1988–2008. [Google Scholar] [CrossRef]
- Bevis, J.B.; Dunlavey, S.; Martinez-Duarte, R. Comparing the performance of different extruders in the Robocasting of biopolymer-nanoparticle composites towards the fabrication of complex geometries of porous Tungsten Carbide. Procedia Manuf. 2021, 53, 338–342. [Google Scholar] [CrossRef]
- Fang, Y.; Sun, W.; Zhang, T.; Xiong, Z. Recent advances on bioengineering approaches for fabrication of functional engineered cardiac pumps: A review. Biomaterials 2022, 280, 121298. [Google Scholar] [CrossRef]
- Adel, I.M.; Elmeligy, M.F.; Elkasabgy, N.A. Conventional and Recent Trends of Scaffolds Fabrication: A Superior Mode for Tissue Engineering. Pharmaceutics 2022, 14, 306. [Google Scholar] [CrossRef]
- Momeni, F.; Mehdi, M.; Hassani, N.S.; Liu, X.; Ni, J. A review of 4D printing. Mater. Des. 2017, 122, 42–79. [Google Scholar] [CrossRef]
- Ge, Q.; Dunn, C.K.; Qi, H.J.; Dunn, M.L. Active origami by 4D printing. Smart Mater. Struct. 2014, 23, 094007. [Google Scholar] [CrossRef]
- Arif, Z.U.; Khalid, M.Y.; Zolfagharian, A.; Bodaghi, M. 4D bioprinting of smart polymers for biomedical applications: Recent progress, challenges, and future perspectives. React. Funct. Polym. 2022, 179, 105374. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, D.; Cheng, S.; Huang, X. Combined effects of runoff and soil erodibility on available nitrogen losses from sloping farmland affected by agricultural practices. Agric. Water Manag. 2016, 176, 1–8. [Google Scholar] [CrossRef]
- Kuksenok, O.; Balazs, A.C. Stimuli-responsive behavior of composites integrating thermo-responsive gels with photo-responsive fibers. Mater. Horiz. 2016, 3, 53–62. [Google Scholar] [CrossRef]
- Bakarich, S.E.; Gorkin, R.; Panhuis, M.; Spinks, G.M. 4D printing with mechanically robust, thermally actuating hydrogels. Macromol. Rapid Commun. 2015, 36, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Yarali, E.; Baniasadi, M.; Zolfagharian, A.; Chavoshi, M.; Arefi, F.; Hossain, M.; Bastola, A.; Ansari, M.; Foyouzat, A.; Dabbagh, A.; et al. Magneto-/electro-responsive polymers toward manufacturing, characterization, and biomedical/soft robotic applications. Appl. Mater. Today 2022, 26, 101306. [Google Scholar] [CrossRef]
- Lukin, I.; Musquiz, S.; Erezuma, I.; Al-Tel, T.H.; Golafshan, N.; Dolatshahi-Pirouz, A.; Orive, G. Can 4D bioprinting revolutionize drug development? Expert. Opin. Drug Discov. 2019, 14, 953–956. [Google Scholar] [CrossRef]
- Falahati, M.; Ahmadvand, P.; Safaee, S.; Chang, Y.C.; Lyu, Z.; Chen, R.; Li, L.; Lin, Y. Smart polymers and nanocomposites for 3D and 4D printing. Mater. Today 2020, 40, 215–245. [Google Scholar] [CrossRef]
- Khare, V.; Sonkaria, S.; Lee, G.Y.; Ahn, S.H.; Chu, W.S. From 3D to 4D printing—design, material and fabrication for multi-functional multi-materials. Int. J. Precis. Eng. Manuf. 2017, 4, 291–299. [Google Scholar] [CrossRef]
- Khalid, M.Y.; Arif, Z.U.; Ahmed, W.; Umer, R.; Zolfagharian, A.; Bodaghi, M. 4D printing: Technological developments in robotics applications. Sens. Actuators A Phys. 2022, 343, 113670. [Google Scholar] [CrossRef]
- Wan, X.; He, Y.; Liu, Y.; Leng, J. 4D printing of multiple shape memory polymer and nanocomposites with biocompatible, programmable and selectively actuated properties. Addit. Manuf. 2022, 53, 102689. [Google Scholar] [CrossRef]
- Wiberg, A.; Persson, J.; Ölvander, J. Design for additive manufacturing–a review of available design methods and software. Rapid Prototyp. J. 2019, 25, 1080–1094. [Google Scholar] [CrossRef]
- Ureña, J.; Blasco, J.R.; Jordá, O.; Martínez, M.; Portolés, L.; Gonzalez-Gutierrez, J.; Schuschnigg, S. Development of Material and Processing Parameters for AM. In A Guide to Additive Manufacturing; Godec, D., Gonzalez-Gutierrez, J., Nordin, A., Pei, E., Ureña Alcázar, J., Eds.; Springer Tracts in Additive Manufacturing; Springer: Cham, Switzerland, 2022; pp. 231–306. [Google Scholar] [CrossRef]
- Jin, Y.; Du, J.; He, Y. Optimization of process planning for reducing material consumption in additive manufacturing. J. Manuf. Syst. 2017, 44, 65–78. [Google Scholar] [CrossRef]
- Schaub, D.A.; Chu, K.R.; Montgomery, D.C. Optimizing stereolithography throughput. J. Manuf. Syst. 1997, 16, 290–303. [Google Scholar] [CrossRef]
- Cho, H.S.; Park, W.S.; Choi, B.W.; Leu, M.C. Determining optimal parameters for stereolithography processes via genetic algorithm. J. Manuf. Syst. 2000, 19, 18–27. [Google Scholar] [CrossRef]
- Gowda, R.B.S.; Udayagiri, C.S.; Narendra, D.D. Studies on the Process Parameters of Rapid Prototyping Technique (Stereolithography) for the Betterment of Part Quality. Int. J. Manuf. Eng. 2014, 2014, 804705. [Google Scholar] [CrossRef]
- Jing, Z.; Yang, Y.; Li, L. A comprehensive evaluation for different post-curing methods used in stereolithography additive manufacturing. J. Manuf. Process. 2000, 56, 867–877. [Google Scholar] [CrossRef]
- Pazhamannil, R.V.; Rajeev, A.; Govindan, P.; Edacherian, A. Experimental investigations into the effects of process parameters and UV curing on the tensile strength of projection based stereolithography. Strength Mater. 2022, 54, 483–492. [Google Scholar] [CrossRef]
- Rahmati, S.; Ghadami, F. Process parameters optimization to improve dimensional accuracy of stereolithography parts. Int. J. Adv. Des. Manuf. 2014, 7, 59–65. [Google Scholar]
- Romli, D.S.; Seprianto, D.; Putra, D.P.; Zamheri, R.; Rasyid, M. Effect of object making process parameters with digital light processing rapid prototyping technology on bending stress. J. Phys. Conf. Ser. 2020, 1, 012027. [Google Scholar] [CrossRef]
- Pagac, M.; Hajnys, J.; Ma, Q.-P.; Jancar, L.; Jansa, J.; Stefek, P.; Mesicek, J. A Review of Vat Photopolymerization Technology: Materials, Applications, Challenges, and Future Trends of 3D Printing. Polymers 2021, 13, 598. [Google Scholar] [CrossRef]
- Piedra-Cascón, W.; Krishnamurthy, V.R.; Att, W.; Revilla-León, M. 3D printing parameters, supporting structures, slicing, and post-processing procedures of vat-polymerization additive manufacturing technologies: A narrative review. J. Dent. 2021, 109, 103630. [Google Scholar] [CrossRef]
- Zakeri, S.; Vippola, M.; Levänen, E. A comprehensive review of the photopolymerization of ceramic resins used instereolithography. Addit. Manuf. 2020, 35, 101177. [Google Scholar] [CrossRef]
- El-Sherif, L.M. Modeling Stereolithography Process Parameters Using System Dynamics. Master’s Thesis, Arab Academy for Science, Technology & Maritime Transport, Cairo, Egypt, 2015. [Google Scholar]
- Hu, G.; Cao, Z.; Hopkins, M.; Hayes, C.; Daly, M.; Zhou, H.; Devinel, D.M. Optimizing the hardness of SLA printed objects by using the neural network and genetic algorithm. Procedia Manuf. 2019, 38, 117–124. [Google Scholar] [CrossRef]
- Ferreira, P.; Coelho, J.F.J.; Almeida, J.F.; Gil, M.H. Photocrosslinkable Polymers for Biomedical Applications, Biomedical Engineering-Frontiers and Challenges; Fazel, R., Ed.; IntechOpen: London, UK, 2011. [Google Scholar] [CrossRef]
- Raman, R.; Bashir, R. Stereolithographic 3D Bioprinting for Biomedical Applications. In Essentials of 3D Biofabrication and Translation; Atala, A., Yoo, J.J., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 89–121. [Google Scholar] [CrossRef]
- Bagheri, A.; Jin, J. Photopolymerization in 3D Printing. ACS Appl. Polym. Mater. 2019, 1, 593–611. [Google Scholar] [CrossRef]
- Yu, C.; Schimelman, J.; Wang, P.; Miller, K.L.; Ma, X.; You, S.; Guan, J.; Sun, B.; Zhu, W.; Chen, S. Photopolymerizable Biomaterials and Light-Based 3D Printing Strategies for Biomedical Applications. Chem. Rev. 2020, 120, 10695–10743. [Google Scholar] [CrossRef] [PubMed]
- Tomal, W.; Ortyl, J. Water-Soluble Photoinitiators in Biomedical Applications. Polymers 2020, 12, 1073. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Ke, D.; Sahasrabudhe, H.; Bandyopadhyay, A. Additive manufacturing of biomaterials. Prog. Mater. Sci. 2018, 93, 45–111. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Chen, J.; Wang, J. The selection of photoinitiators for photopolymerization of biodegradable polymers and its application in digital light processing additive manufacturing. J. Biomed. Mater. Res. 2022, 110, 204–216. [Google Scholar] [CrossRef]
- Choi, J.R.; Yong, K.W.; Choi, J.W.; Cowie, A.C. Recent advances in photo-crosslinkable hydrogels for biomedical applications. BioTechniques 2019, 66, 40–53. [Google Scholar] [CrossRef]
- Arifin, N.; Sudin, I.; Ngadiman, N.H.A.; Ishak, M.S.A. A Comprehensive Review of Biopolymer Fabrication in Additive Manufacturing Processing for 3D-Tissue-Engineering Scaffolds. Polymers 2022, 14, 2119. [Google Scholar] [CrossRef] [PubMed]
- Bártolo, P.J. Stereolithographic Processes; Stereolithography: Materials, Processes and Applications; Springer: New York, NY, USA, 2011; pp. 1–36. [Google Scholar] [CrossRef]
- Bahraminasab, M. Challenges on optimization of 3D-printed bone scaffolds. BioMed Eng. Online 2020, 19, 69. [Google Scholar] [CrossRef]
- Wanga, C.; Huang, W.; Zhou, Y.; He, L.; Hea, Z.; Chen, Z.; He, X.; Tian, S.; Liao, J.; Lu, B.; et al. 3D printing of bone tissue engineering scaffolds. Bioact. Mater. 2020, 5, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Punia, U.; Kaushik, A.; Garg, R.K.; Chhabra, D.; Sharma, A. 3D printable biomaterials for dental restoration: A systematic review. Mater. Today Proc. 2022, 63, 566–572. [Google Scholar] [CrossRef]
- Oladapo, B.I.; Zahedi, S.A.; Ismail, S.O.; Omigbodun, F.T.; Bowoto, O.K.; Olawumi, M.A.; Muhammad, M.A. 3D printing of PEEK–cHAp scaffold for medical bone implant. Bio Des. Manuf. 2021, 4, 44–59. [Google Scholar] [CrossRef]
- Luzanin, O.; Movrin, D.; Stathopoulos, V.; Pandis, P.; Radusin, T.; Guduric, V. Impact of processing parameters on tensile strength, in-process crystallinity and mesostructure in FDM-fabricated PLA specimens. Rapid Prot. J. 2019, 25, 1398–1410. [Google Scholar] [CrossRef]
- Yang, C.; Tian, X.; Li, D.; Cao, Y.; Zhao, F.; Shi, C. Influence of thermal processing conditions in 3D printing on the crystallinity and mechanical properties of PEEK material. J. Mat. Proc. Tech. 2017, 248, 1–7. [Google Scholar] [CrossRef]
- Elkasabgy, N.A.; Mahmoud, A.A.; Maged, A. 3D printing: An appealing route for customized drug delivery systems. Int. J. Pharm. 2020, 588, 119732. [Google Scholar] [CrossRef]
- Kumar, P.; Rajak, K.D.; Abubakar, M.; Mustafa Ali, S.G.M.; Hussain, M. 3D Printing Technology for Biomedical Practice: A Review. J. Mat. Eng. Perform. 2021, 30, 5342–5355. [Google Scholar] [CrossRef]
- Tripathy, C.R.; Sharma, R.K.; Rattan, V.K. Effect of printing parameters on the mechanical behaviour of the thermoplastic polymer processed by FDM technique: A research review. Adv. Prod. Eng. Manag. 2022, 17, 279–294. [Google Scholar] [CrossRef]
- Alama, F.; Shukla, V.R.; Varadarajan, K.M.; Kumar, S. Microarchitected 3D printed polylactic acid (PLA) nanocomposite scaffolds for biomedical applications. J. Mech. Beh. Biomed. Mat. 2020, 103, 103576. [Google Scholar] [CrossRef]
- Mohd Pu’ad, N.A.S.; Abdul Haq, R.H.; Mohd Noh, H.; Abdullah, H.Z.; Idris, M.I.; Lee, T.C. Review on the fabrication of fused deposition modelling (FDM) composite filament for biomedical applications. Mat. Today Proc. 2020, 29, 228–232. [Google Scholar] [CrossRef]
- Abeykoon, C.; Sri-Amphorn, P.; Fernando, A. Optimization of fused deposition modeling parameters for improved PLA and ABS 3D printed structures. Int. J. Lightweight Mat. Manuf. 2020, 3, 284–297. [Google Scholar] [CrossRef]
- Hikmat, M.; Rostam, S.; Ahmed, Y.M. Investigation of tensile property-based Taguchi method of PLA parts fabricated by FDM 3D printing technology. Res. Eng. 2021, 11, 100264. [Google Scholar] [CrossRef]
- Sarasua, J.R.; López-Rodríguez, N.; Zuza, E.; Petisco, S.; Castro, B.; del Olmo, M.; Palomares, T.; Alonso-Varona, A. Crystallinity assessment and in vitro cytotoxicity of polylactide scaffolds for biomedical applications. J. Mater. Sci. Mater. Med. 2011, 22, 2513–2523. [Google Scholar] [CrossRef]
- Honigmann, P.; Sharma, N.; Okolo, B.; Popp, U.; Msallem, B.; Thieringer, F.M. Patient-Specific Surgical Implants Made of 3D Printed PEEK: Material, Technology, and Scope of Surgical Application. BioMed Res. Int. 2018, 8, 4520636. [Google Scholar] [CrossRef] [PubMed]
- Dinga, S.; Zoua, B.; Wang, P.; Dinga, H. Effects of nozzle temperature and building orientation on mechanical properties and microstructure of PEEK and PEI printed by 3D-FDM. Polym. Test. 2019, 78, 105948. [Google Scholar] [CrossRef]
- Vaezi, M.; Yang, S. Extrusion-based additive manufacturing of PEEK for biomedical applications. Virtual Phys. Prototyp. 2015, 10, 123–135. [Google Scholar] [CrossRef]
- Sharma, N.; Aghlmandi, S.; Cao, S.; Kunz, C.; Honigmann, P.; Thieringer, F.M. Quality Characteristics and Clinical Relevance of In-House 3D-Printed Customized Polyetheretherketone (PEEK) Implants for Craniofacial Reconstruction. J. Clin. Med. 2020, 9, 2818. [Google Scholar] [CrossRef]
- Wu, W.; Geng, P.; Li, G.; Zhao, D.; Zhang, H.; Zhao, J. Influence of Layer Thickness and Raster Angle on the Mechanical Properties of 3D-Printed PEEK and a Comparative Mechanical Study between PEEK and ABS. Materials 2015, 8, 5834–5846. [Google Scholar] [CrossRef]
- Han, W.; Kong, L.; Xu, M. Advances in selective laser sintering of polymers. Int. J. Extrem. Manuf. 2022, 4, 042002. [Google Scholar] [CrossRef]
- Riza, S.H.; Masood, S.H.; Rashid, R.A.R.; Chandra, S. Selective Laser Sintering in Biomedical Manufacturing; Wen, C., Ed.; Woodhead Publishing Series in Biomaterials; Elsevier Ltd.: Amsterdam, The Netherlands, 2020; pp. 193–233. [Google Scholar] [CrossRef]
- El Magri, A.; Bencaid, S.E.; Vanaei, H.R.; Vaudreuil, S. Effects of Laser Power and Hatch Orientation on Final Properties of PA12 Parts Produced by Selective Laser Sintering. Polymers 2022, 14, 3674. [Google Scholar] [CrossRef]
- Wu, L.; Koen, W.; Singh, A.; Sharma, A. Investigation on the suitability of polymers for selective laser sintering using novel midir lasers. In Proceedings of the 19th Annual Conference Additive Manufacturing as a Key Driver of the 4th Industrial Revolution, Protea Parktown, Braamfontein, Johannesburg, South Africa, 6–9 November 2018; pp. 71–76. [Google Scholar]
- Gueche, Y.A.; Sanchez-Ballester, N.M.; Cailleaux, S.; Bataille, B.; Soulairol, I. Selective Laser Sintering (SLS), a New Chapter in the Production of Solid Oral Forms (SOFs) by 3D Printing. Pharmaceutics 2021, 13, 1212. [Google Scholar] [CrossRef]
- Bae, S.W.; Kim, J.S.; Kim, D.S.; Yoo, S.Y. An experimental study for rising manufacturing time and accuracy on SLS process. In Proceedings of the 2009 IEEE International Symposium on Assembly and Manufacturing, Seoul, Republic of Korea, 17–20 November 2009; pp. 83–87. [Google Scholar] [CrossRef]
- Wang, R.J.; Wang, L.; Zhao, L.; Liu, Z.J. Influence of process parameters on part shrinkage in SLS. Int. J. Adv. Manuf. Technol. 2007, 33, 498–504. [Google Scholar] [CrossRef]
- Liu, X.; Wang, J.; Zhu, J.; Liew, P.J.; Li, C.; Huang, C. Ultrasonic abrasive polishing of additive manufactured parts: An experimental study on the effects of process parameters on polishing performance. Adv. Prod. Eng. Manag. 2022, 17, 193–204. [Google Scholar] [CrossRef]
- Dadbakhsh, S.; Verbelen, L.; Vandeputte, T.; Strobbe, D.; Van Puyvelde, P.; Kruth, J.P. Effect of Powder Size and Shape on the SLS Processability and Mechanical Properties of a TPU Elastomer. Phys. Procedia 2016, 83, 971–980. [Google Scholar] [CrossRef]
- Drummer, D.; Rietzel, D.; Kühnlein, F. Development of a characterization approach for the sintering behavior of new thermoplastics for selective laser sintering. Phys. Procedia 2010, 5 Pt B, 533–542. [Google Scholar] [CrossRef]
- Beal, V.E.; Paggi, R.A.; Salmoria, G.V.; Lago, A. Statistical evaluation of laser energy density effect on mechanical properties of polyamide parts manufactured by selective laser sintering. J. Appl. Polym. Sci. 2009, 113, 2910–2919. [Google Scholar] [CrossRef]
- Erdal, M.; Dag, S.; Jande, Y.; Tekin, C.M. Manufacturing of functionally graded porous products by selective laser sintering. Mater. Sci. Forum 2010, 631–632, 253–258. [Google Scholar] [CrossRef]
- Zeng, Z.; Deng, X.; Cui, J.; Jiang, H.; Yan, S.; Peng, B. Improvement on Selective Laser Sintering and Post-Processing of Polystyrene. Polymers 2019, 11, 956. [Google Scholar] [CrossRef]
- Caulfield, B.; McHugh, P.E.; Lohfeld, S. Dependence of mechanical properties of polyamide components on build parameters in the SLS process. J. Mater. Process Technol. 2007, 182, 477–488. [Google Scholar] [CrossRef]
- Berretta, S.; Evans, K.E.; Ghita, O. Processability of PEEK, a new polymer for High Temperature Laser Sintering (HT-LS). Eur. Polym. J. 2015, 68, 243–266. [Google Scholar] [CrossRef]
- Toth-Taşcău, M.; Raduta, A.; Stoia, D.I.; Locovei, C. Influence of the energy density on the porosity of polyamide parts in SLS process. Solid State Phenom. 2012, 188, 400–405. [Google Scholar] [CrossRef]
- Madžarević, M.; Medarević, Đ.; Pavlović, S.; Ivković, B.; Đuriš, J.; Ibrić, S. Understanding the Effect of Energy Density and Formulation Factors on the Printability and Characteristics of SLS Irbesartan Tablets—Application of the Decision Tree Model. Pharmaceutics 2021, 13, 1969. [Google Scholar] [CrossRef]
- Razaviye, M.K.; Tafti, R.A.; Khajehmohammadi, M. An investigation on mechanical properties of PA12 parts produced by a SLS 3D printer: An experimental approach. CIRP J. Manuf. Sci. Technol. 2022, 38, 760–768. [Google Scholar] [CrossRef]
- Pilipović, A.; Brajlih, T.; Drstvenšek, I. Influence of Processing Parameters on Tensile Properties of SLS Polymer Product. Polymers 2018, 10, 1208. [Google Scholar] [CrossRef]
- Bierwisch, C.; Mohseni-Mofidi, S.; Dietemann, B.; Grünewald, M.; Rudloff, J.; Lang, M. Universal process diagrams for laser sintering of polymers. Mater. Des. 2021, 199, 109432. [Google Scholar] [CrossRef]
- Chung, H.; Jee, H.; Das, S. Selective laser sintering of PCL/TCP composites for tissue engineering scaffolds. J. Mech. Sci. Technol. 2010, 24, 241–244. [Google Scholar] [CrossRef]
- Partee, B.; Hollister, S.J.; Das, S. Selective laser sintering process optimization for layered manufacturing of CAPA_6501 polycaprolactone bone tissue engineering scaffolds. J. Manuf. Sci. Eng. Trans. ASME 2006, 128, 531–540. [Google Scholar] [CrossRef]
- Torres, J.; Cole, M.; Owji, A.; DeMastry, Z.; Gordon, A.P. An approach for mechanical property optimization of fused deposition modeling with polylactic acid via design of experiments. Rapid Prototyp. J. 2019, 22, 387–404. [Google Scholar] [CrossRef]
- Cerda-Avila, S.; Medellin-Castillo, N.; Hugo, I.; Lim, T. An experimental methodology to analyse the structural behaviour of FDM parts with variable process parameters. Rapid Prototyp. J. 2020, 26, 1615–1625. [Google Scholar] [CrossRef]
- Harpool, T.D.; Alarifi, I.M.; Alshammari, B.A.; Aabid, A.; Baig, M.; Malik, R.A.; Mohamed Sayed, A.; Asmatulu, R.; EL-Bagory, T.M.A.A. Evaluation of the Infill Design on the Tensile Response of 3D Printed Polylactic Acid Polymer. Materials 2021, 14, 2195. [Google Scholar] [CrossRef]
- Aloyaydi, B.; Sivasankaran, S.; Mustafa, A. Investigation of infill-patterns on mechanical response of 3D printed poly-lactic-acid. Polym. Test. 2020, 87, 106557. [Google Scholar] [CrossRef]
- Akhoundi, B.; Behravesh, A.H. Effect of Filling Pattern on the Tensile and Flexural Mechanical Properties of FDM 3D Printed Products. Exp. Mech. 2019, 59, 883–897. [Google Scholar] [CrossRef]
- Lalegani Dezaki, M.; Mohd Ariffin, M.K.A. The Effects of Combined Infill Patterns on Mechanical Properties in FDM Process. Polymers 2020, 12, 2792. [Google Scholar] [CrossRef] [PubMed]
- Martín, M.J.; Auñón, J.A.; Martín, F. Influence of Infill Pattern on Mechanical Behavior of Polymeric and Composites Specimens Manufactured Using Fused Filament Fabrication Technology. Polymers 2021, 13, 2934. [Google Scholar] [CrossRef] [PubMed]
- Ehrmann, G.; Ehrmann, A. Investigation of the Shape-Memory Properties of 3D Printed PLA Structures with Different Infills. Polymers 2021, 13, 164. [Google Scholar] [CrossRef]
- Ehrmann, G.; Ehrmann, A. Pressure Orientation-Dependent Recovery of 3D-Printed PLA Objects with Varying Infill Degree. Polymers 2021, 13, 1275. [Google Scholar] [CrossRef]
- Jamadi, A.H.; Razali, N.; Petrů, M.; Taha, M.M.; Muhammad, N.; Ilyas, R.A. Effect of Chemically Treated Kenaf Fibre on Mechanical and Thermal Properties of PLA Composites Prepared through Fused Deposition Modeling (FDM). Polymers 2021, 13, 3299. [Google Scholar] [CrossRef]
- Cuan-Urquizo, E.; Álvarez-Trejo, A.; Robles Gil, A.; Tejada-Ortigoza, V.; Camposeco-Negrete, C.; Uribe-Lam, E.; Treviño-Quintanilla, C.D. Effective Stiffness of Fused Deposition Modeling Infill Lattice Patterns Made of PLA-Wood Material. Polymers 2022, 14, 337. [Google Scholar] [CrossRef]
- Ayrilmis, N.; Nagarajan, R.; Kuzman, M.K. Effects of the Face/Core Layer Ratio on the Mechanical Properties of 3D Printed Wood/Polylactic Acid (PLA) Green Biocomposite Panels with a Gyroid Core. Polymers 2020, 12, 2929. [Google Scholar] [CrossRef]
- Guessasma, S.; Belhabib, S. Infill Strategy in 3D Printed PLA Carbon Composites: Effect on Tensile Performance. Polymers 2022, 14, 4221. [Google Scholar] [CrossRef]
- Saleh, M.; Anwar, S.; Al-Ahmari, A.M.; Alfaify, A. Compression Performance and Failure Analysis of 3D-Printed Carbon Fiber/PLA Composite TPMS Lattice Structures. Polymers 2022, 14, 4595. [Google Scholar] [CrossRef] [PubMed]
- Schiavone, N.; Verney, V.; Askanian, H. High-Density Bio-PE and Pozzolan Based Composites: Formulation and Prototype Design for Control of Low Water Flow. Polymers 2021, 13, 1908. [Google Scholar] [CrossRef] [PubMed]
- Tarrés, Q.; Melbø, J.K.; Delgado-Aguilar, M.; Espinach, F.X.; Mutjé, P.; Chinga-Carrasco, G. Bio-polyethylene reinforced with thermomechanical pulp fibers: Mechanical and micromechanical characterization and its application in 3D-printing by fused deposition modelling. Compos. B Eng. 2018, 153, 70–77. [Google Scholar] [CrossRef]
- Tian, J.; Zhang, R.; Wu, Y.; Xue, P. Additive manufacturing of wood flour/polyhydroxyalkanoates (PHA) fully bio-based composites based on micro-screw extrusion system. Mater. Des. 2021, 199, 109418. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.; Heer, B. Additive manufacturing of multi-material structures. Mater. Sci. Eng. R Rep. 2018, 129, 1–16. [Google Scholar] [CrossRef]
- Chung, J.J.; Im, H.; Kim, S.H.; Park, J.W.; Jung, Y. Toward Biomimetic Scaffolds for Tissue Engineering: 3D Printing Techniques in Regenerative Medicine. Front. Bioeng. Biotechnol. 2020, 8, 586406. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; He, J.; Cai, Z.; Wang, F.; Zhang, J.; Wang, L.; Ling, R.; Li, D. Design and additive manufacturing of flexible polycaprolactone scaffolds with highly-tunable mechanical properties for soft tissue engineering. Mater. Des. 2020, 189, 108508. [Google Scholar] [CrossRef]
- Feng, P.; Wu, P.; Gao, C.; Yang, Y.; Guo, W.; Yang, W.; Shuai, C. A Multimaterial Scaffold with Tunable Properties: Toward Bone Tissue Repair. Adv. Sci. 2018, 5, 1700817. [Google Scholar] [CrossRef]
- Ovsianikov, A.; Farsari, M.; Chichkov, B.N. Photonic and biomedical applications of the Two-photon polymerization technique. In Stereolithographic Processes, Stereolithography: Materials, Processes, Applications; Bartolo, P.J., Ed.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef]
- Wu, Y.; Lu, Y.; Zhao, M.; Bosiakov, S.; Li, L. A critical review of additive manufacturing techniques and associated biomaterials used in bone tissue engineering. Polymers 2022, 14, 2117. [Google Scholar] [CrossRef]
- Do, A.V.; Khorsand, B.; Geary, S.M.; Salem, A.K. 3D Printing of Scaffolds for Tissue Regeneration Applications. Adv. Healthc. Mater. 2015, 4, 1742–1762. [Google Scholar] [CrossRef] [PubMed]
- Zerankeshi, M.M.; Bakhshi, R.; Alizadeh, R. Polymer/metal composite 3D porous bone tissue engineering scaffolds fabricated by additive manufacturing techniques: A review. Bioprinting 2022, 25, e00191. [Google Scholar] [CrossRef]
- Li, X.; Heidari, V.; Nourbakhsh, S.M.; Mohammadi, R.; Semiromi, D. Design and fabrication of elastic two-component polymer-metal disks using a 3D printer under different loads for the lumbar spine. Polym. Test. 2022, 112, 107633. [Google Scholar] [CrossRef]
- Shuai, C.; Li, Y.; Feng, P.; Guo, W.; Yang, W.; Peng, S. Positive feedback effects of Mg on the hydrolysis of poly-l-lactic acid (PLLA): Promoted degradation of PLLA scaffolds. Polym. Test. 2018, 68, 27–33. [Google Scholar] [CrossRef]
- Tan, K.H.; Chua, C.K.; Leong, K.F.; Cheah, C.M.; Cheang, P.; Abu Bakar, M.S.; Cha, S.W. Scaffold development using selective laser sintering of polyetheretherketone–hydroxyapatite biocomposite blends. Biomaterials 2003, 24, 3115–3123. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh-Osgouei, M.; Li, Y.; Wen, C. A comprehensive review of biodegradable synthetic polymer-ceramic composites and their manufacture for biomedical applications. Bioact. Mater. 2019, 4, 22–36. [Google Scholar] [CrossRef]
- Zarek, M.; Layani, M.; Cooperstein, I.; Sachyani, E.; Cohn, D.; Magdassi, S. 3D Printing of Shape Memory Polymers for Flexible Electronic Devices. Adv. Mater. 2016, 28, 4449–4454. [Google Scholar] [CrossRef]
- Available online: https://prezi.com/dhwrp5ivfgmy/electronic-heart-sleeve/ (accessed on 5 November 2022).
- Zarek, M.; Mansour, N.; Shapira, S.; Cohn, D. 4D printing of shape memory-based personalized endoluminal medical devices. Macromol. Rapid Commun. 2017, 38, 1600628. [Google Scholar] [CrossRef]
- Zhu, Z.; Park, H.S.; McAlpine, M.C. 3D printed deformable sensors. Sci. Adv. 2020, 6, eaba5575. [Google Scholar] [CrossRef]
- Patel, P.; Gohil, P. Custom orthotics development process based on additive manufacturing. Mater. Today Proc. 2022, 59, A52–A63. [Google Scholar] [CrossRef]
- Lokesh, M.; Kumar Babu, D.; Sherwin, R.; Veezhinatha, M. Evaluation of 3D printed customizable hand orthosis for forearm fractures based on Finite Element Modelling. Mater. Today Proc. 2022, 62, 4707–4713. [Google Scholar] [CrossRef]
- Boolos, M.; Corbin, S.; Herrmann, A.; Regez, B. 3D printed orthotic leg brace with movement assist. Ann. 3D Print. Med. 2022, 7, 100062. [Google Scholar] [CrossRef]
- Hudak, Y.F.; Li, J.S.; Cullum, S.; Strzelecki, B.M.; Richburg, C.; Kaufman, G.E.; Abrahamson, D.; Heckman, J.T.; Ripley, B.; Telfer, S.; et al. A novel workflow to fabricate a patient-specific 3D printed accommodative foot orthosis with personalized latticed metamaterial. Med. Eng. Phys. 2022, 1, 104. [Google Scholar] [CrossRef] [PubMed]
- Hale, L.; Linley, E.; Kalaskar, D.M. A digital workfow for design and fabrication of bespoke orthoses using 3D scanning and 3D printing, a patient-based case study. Sci. Rep. 2020, 10, 7028. [Google Scholar] [CrossRef]
- Walbran, M.; Turner, K.; McDaid, A.J. Customized 3D printed ankle-foot orthosis with adaptable carbon fibre composite spring joint. Cogent Eng. 2016, 3, 1227022. [Google Scholar] [CrossRef]
- Tabaković, S.; Konstantinović, V.; Radosavljević, R.; Movrin, D.; Hadžistević, M.; Hatab, N. Application of Computer-Aided Designing and Rapid Prototyping Technologies in Reconstruction of Blowout Fractures of the Orbital Floor. J. Craniofac. Surg. 2015, 26, 1558–1563. [Google Scholar] [CrossRef]
- Hay, J.A.; Smayra, T.; Moussa, R. Customized Polymethylmethacrylate Cranioplasty Implants Using 3-Dimensional Printed Polylactic Acid Molds: Technical Note with 2 Illustrative Cases. World Neurosurg. 2017, 105, 971–979. [Google Scholar] [CrossRef]
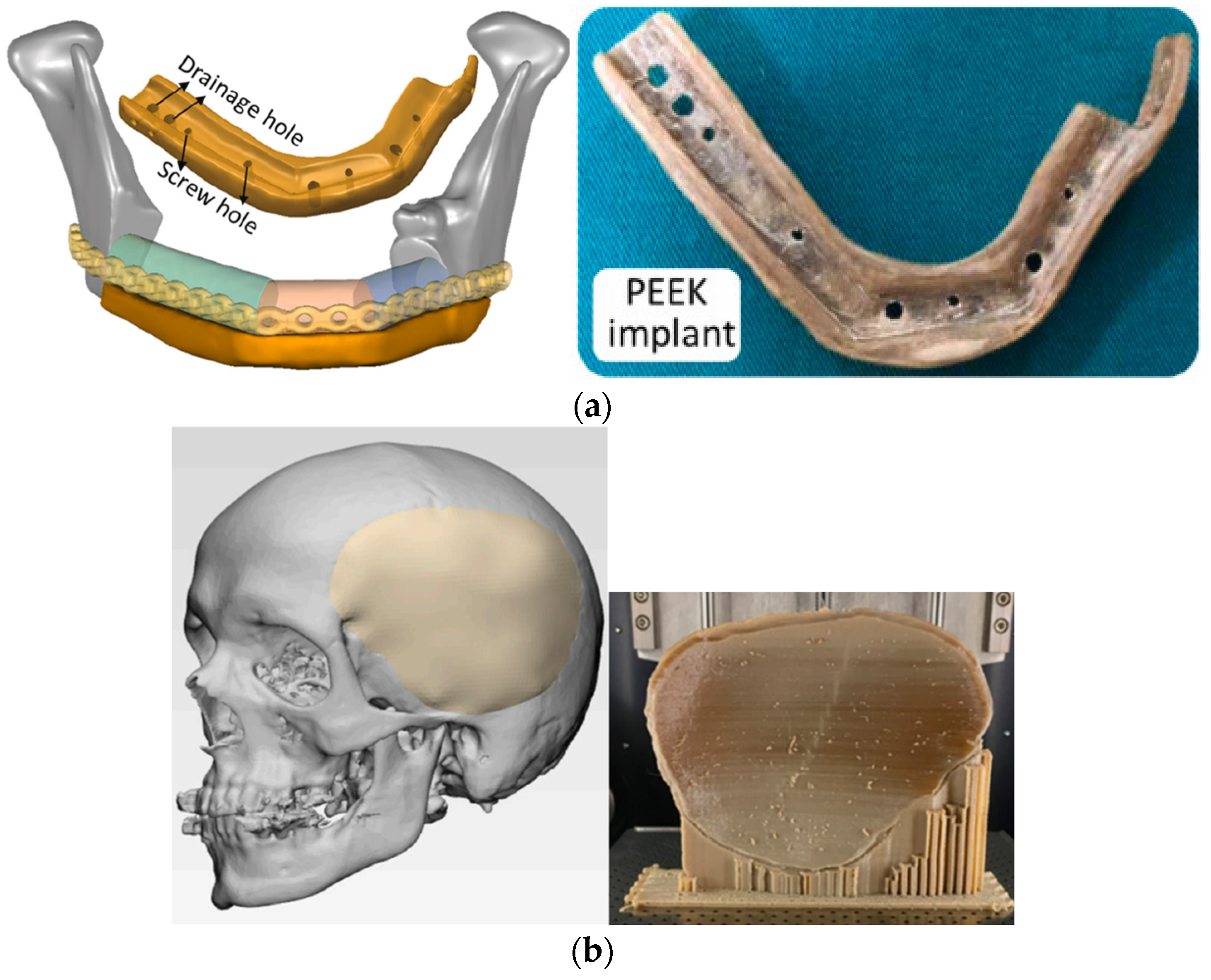
- Kang, J.; Zhang, J.; Zheng, J.; Wang, L.; Li, D.; Liu, S. 3D-printed PEEK implant for mandibular defects repair—A new method. J. Mech. Behav. Biomed. Mater. 2021, 116, 104335. [Google Scholar] [CrossRef]
- Sharma, N.; Aghlmandi, S.; Dalcanale, F.; Seiler, D.; Zeilhofer, H.-F.; Honigmann, P.; Thieringer, F.M. Quantitative Assessment of Point-of-Care 3D-Printed Patient-Specific Polyetheretherketone (PEEK) Cranial Implants. Int. J. Mol. Sci. 2021, 22, 8521. [Google Scholar] [CrossRef]



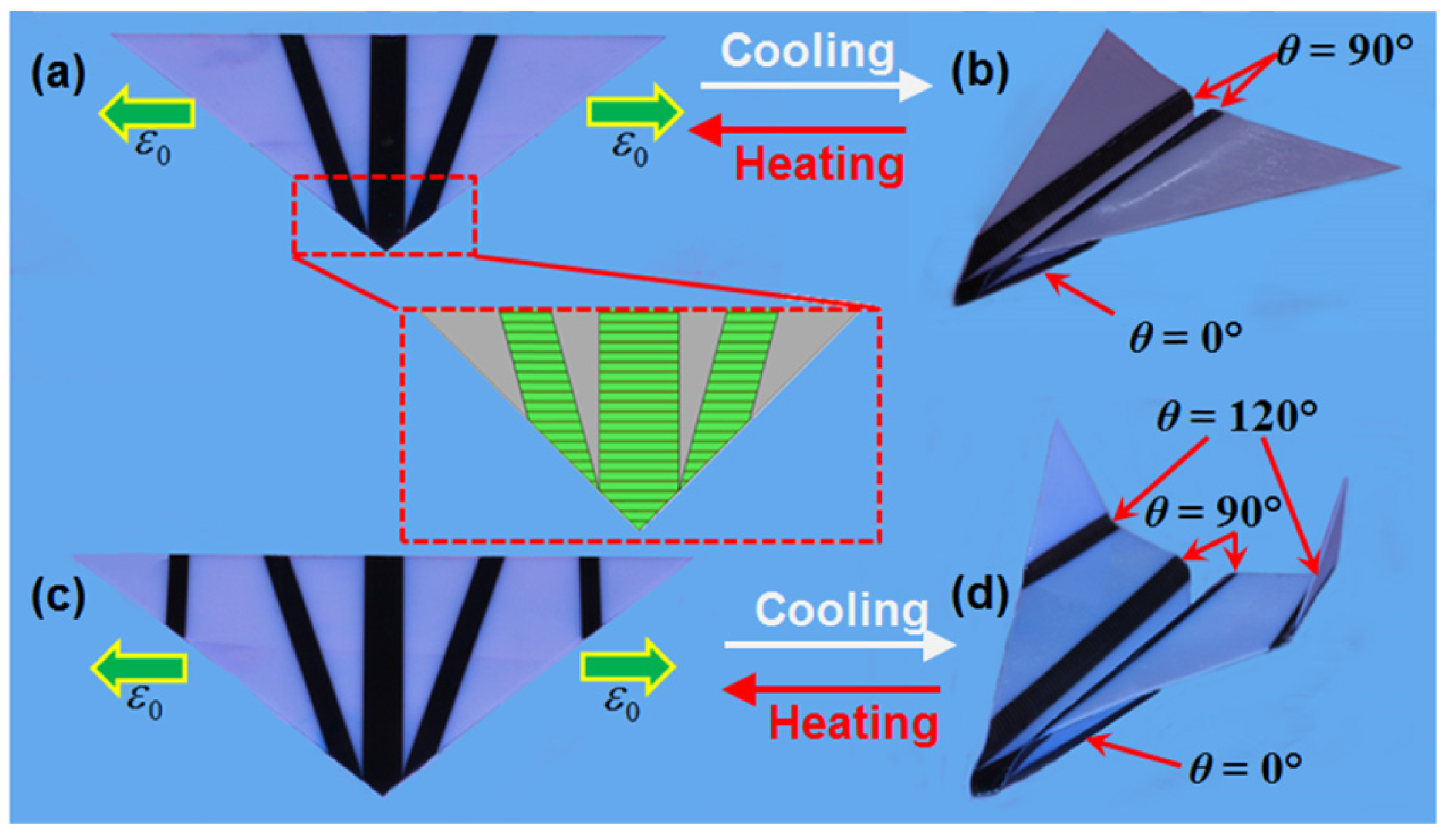
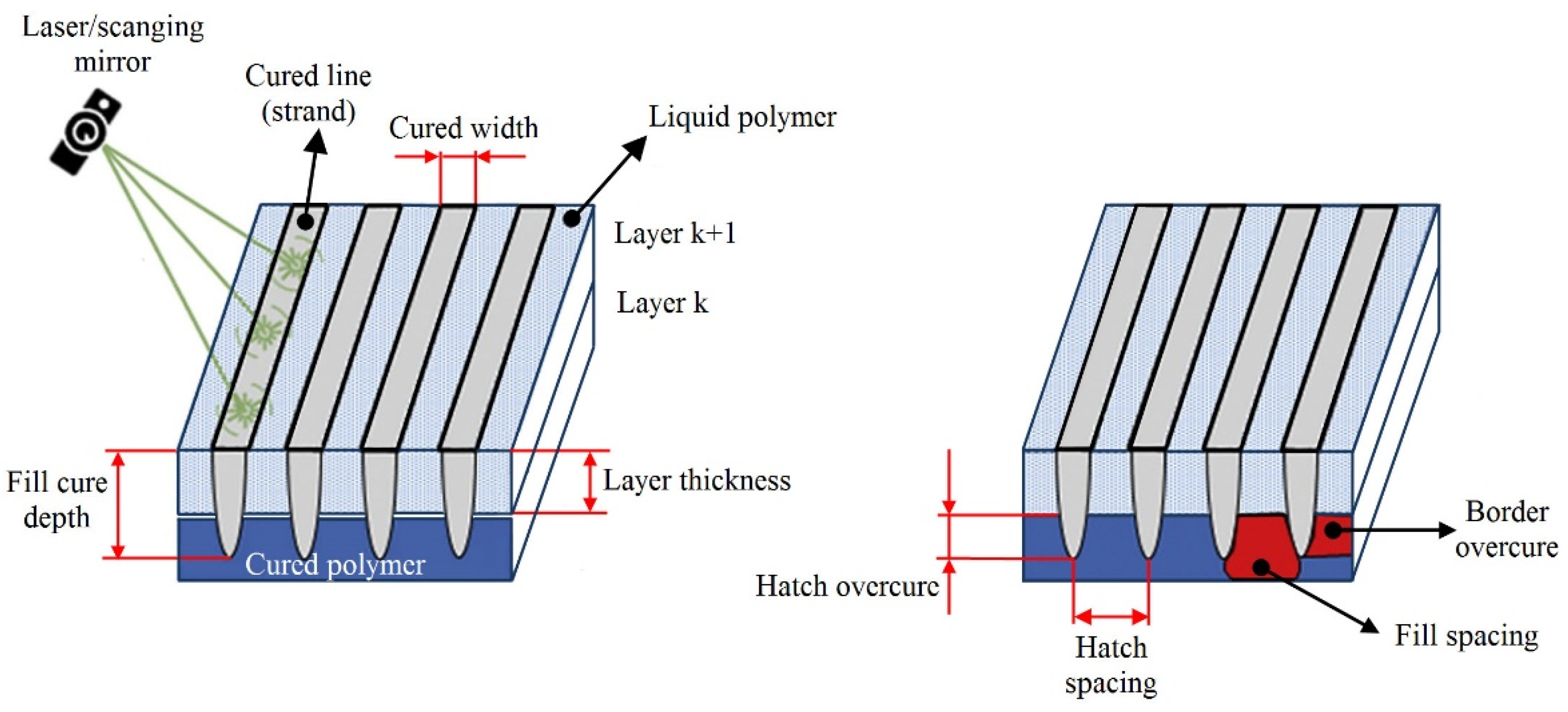

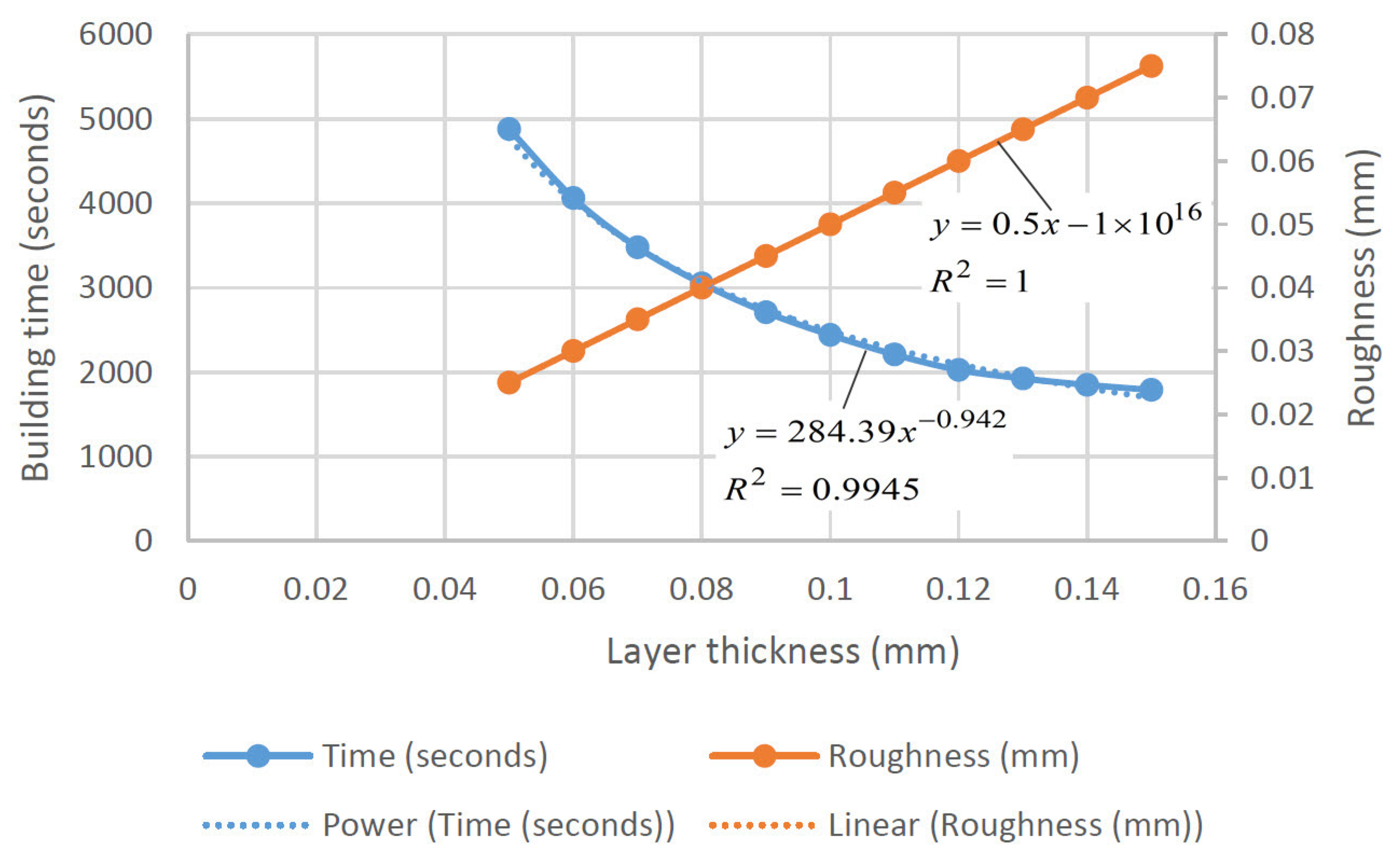

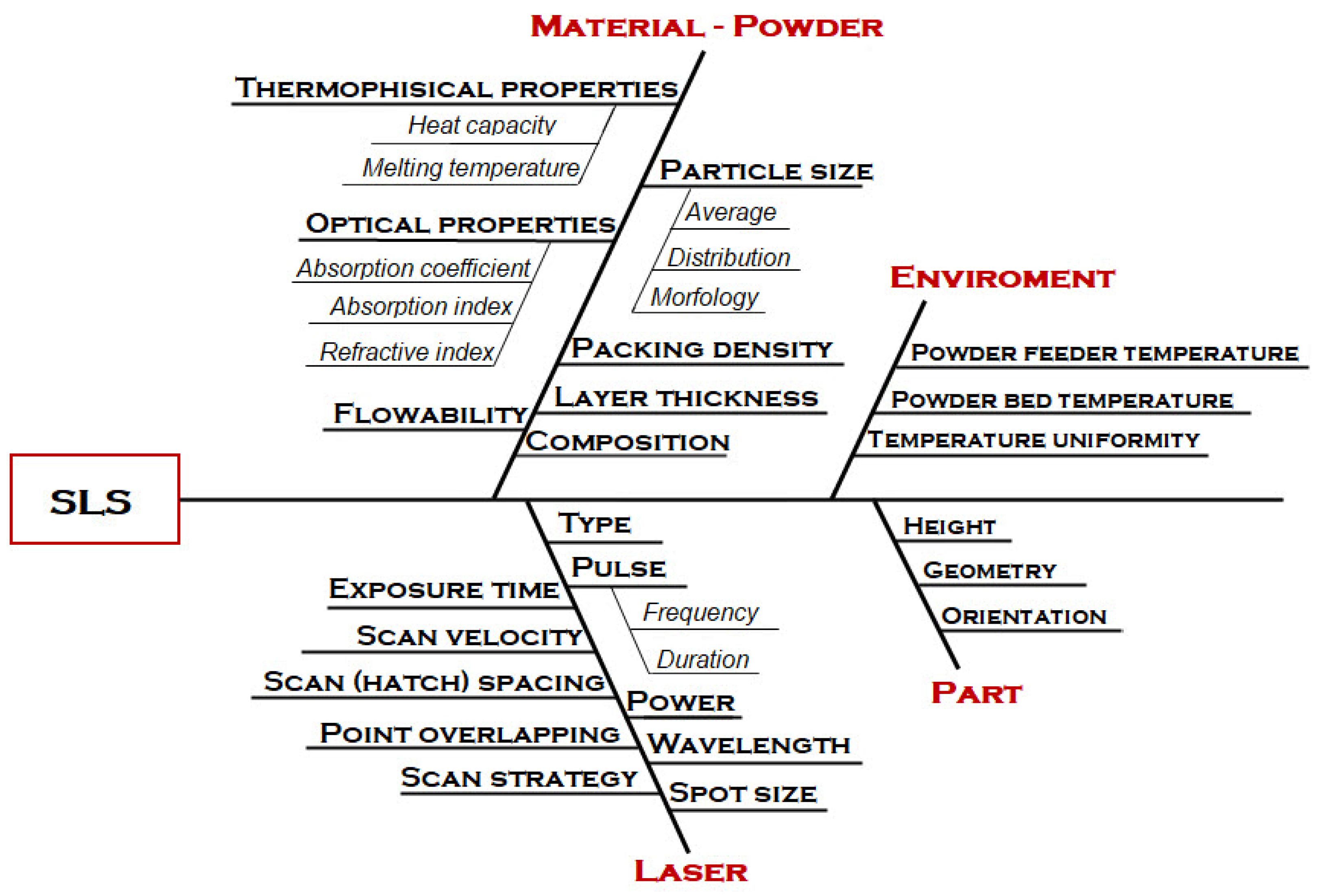


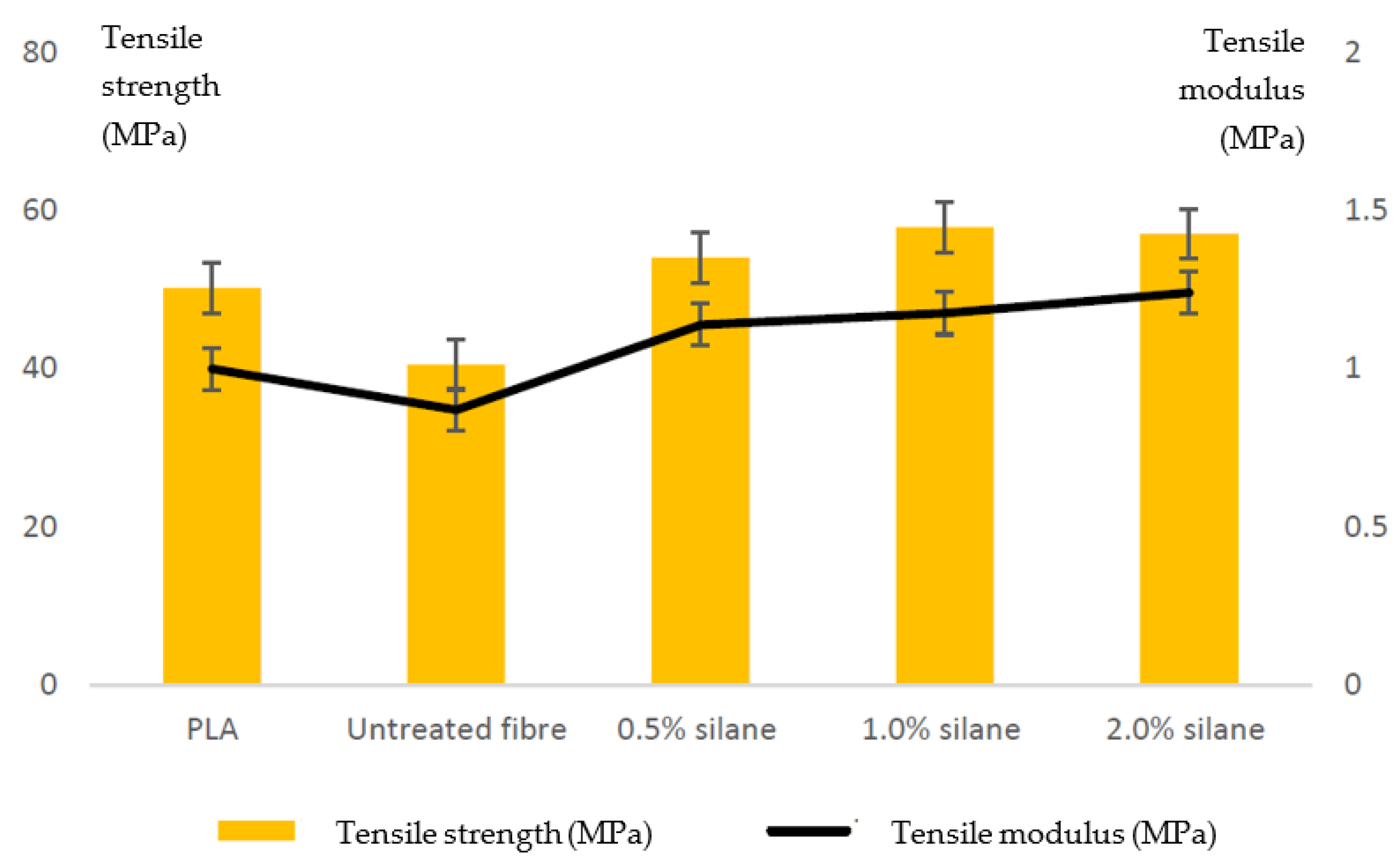
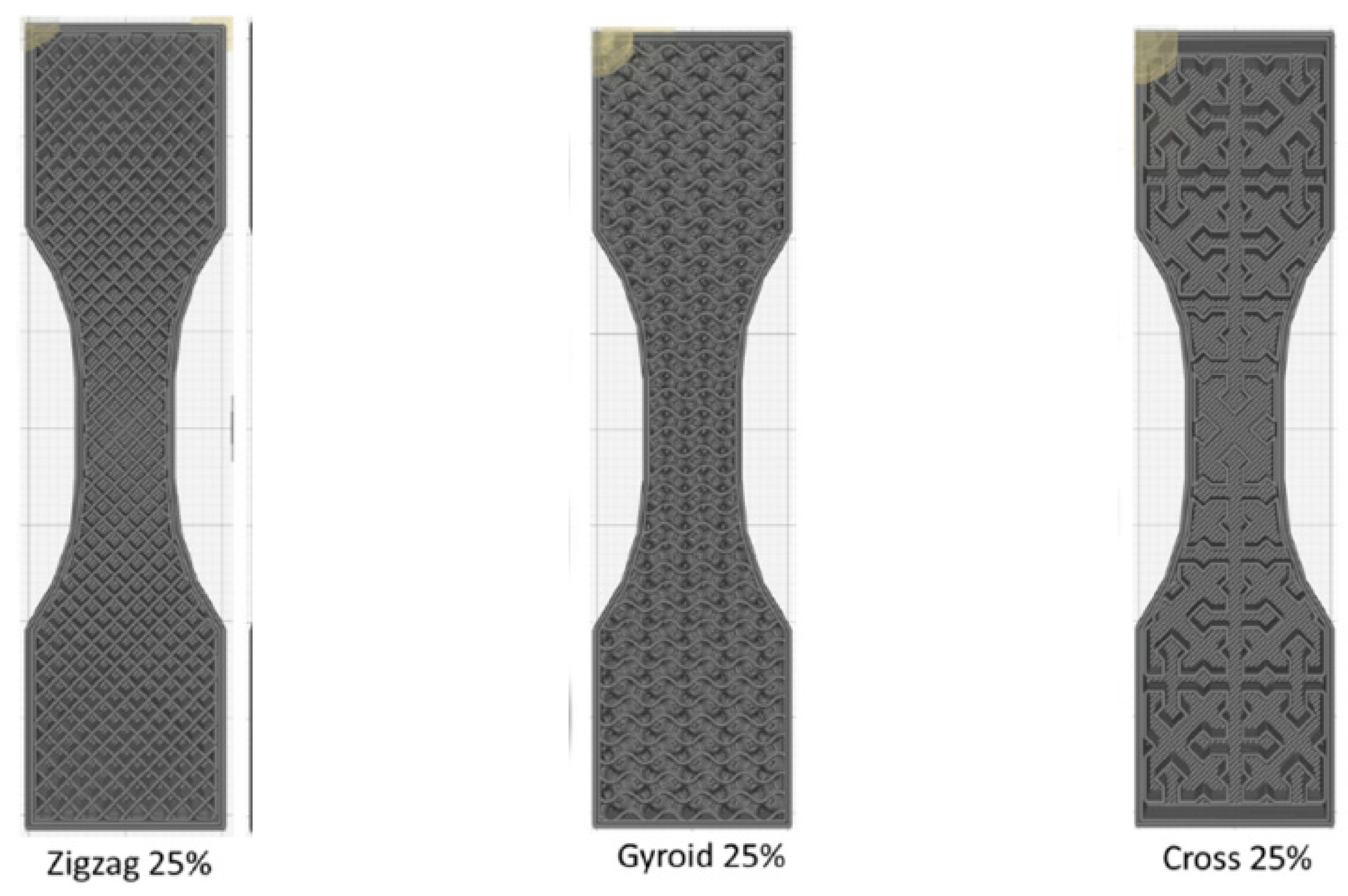
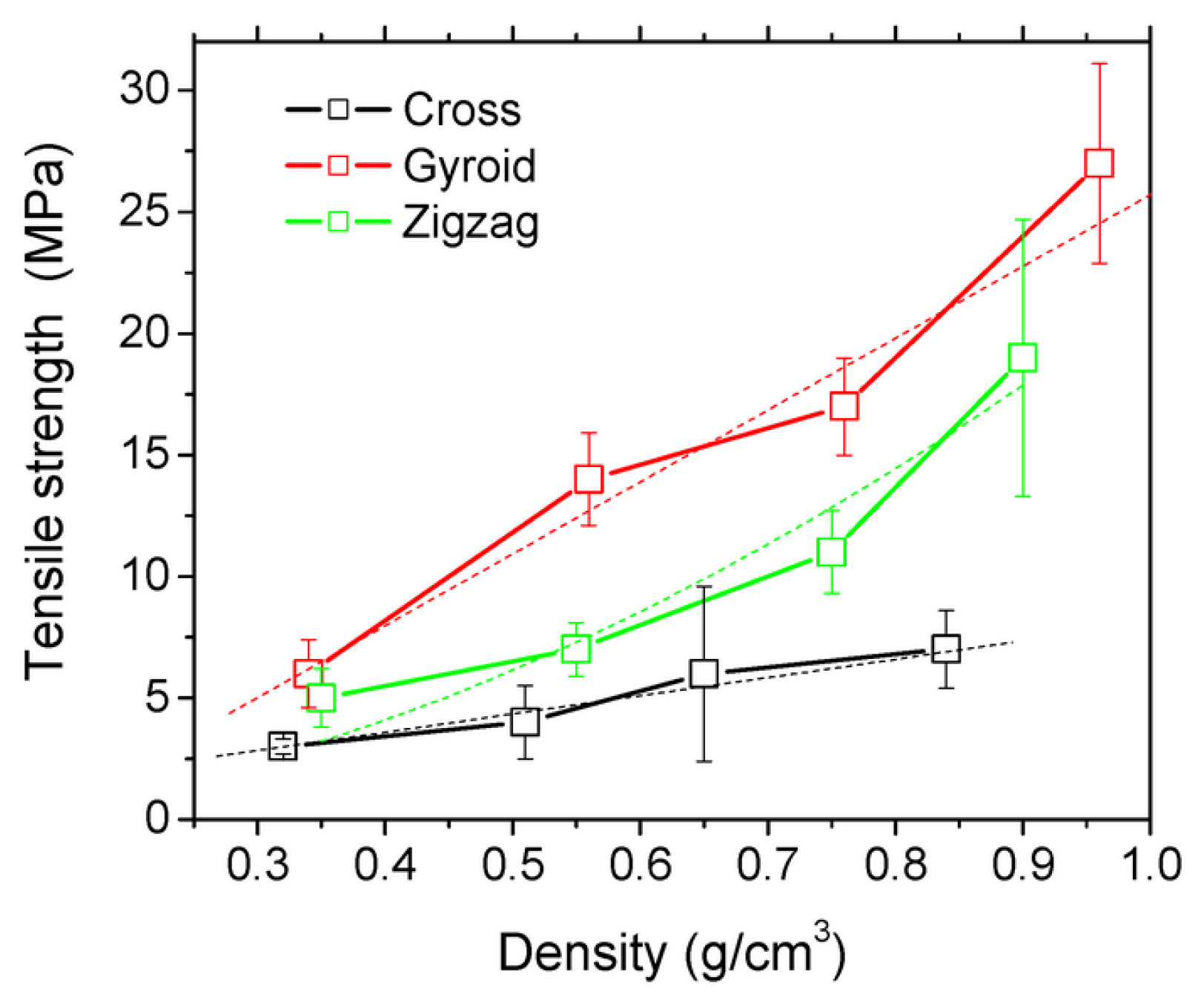
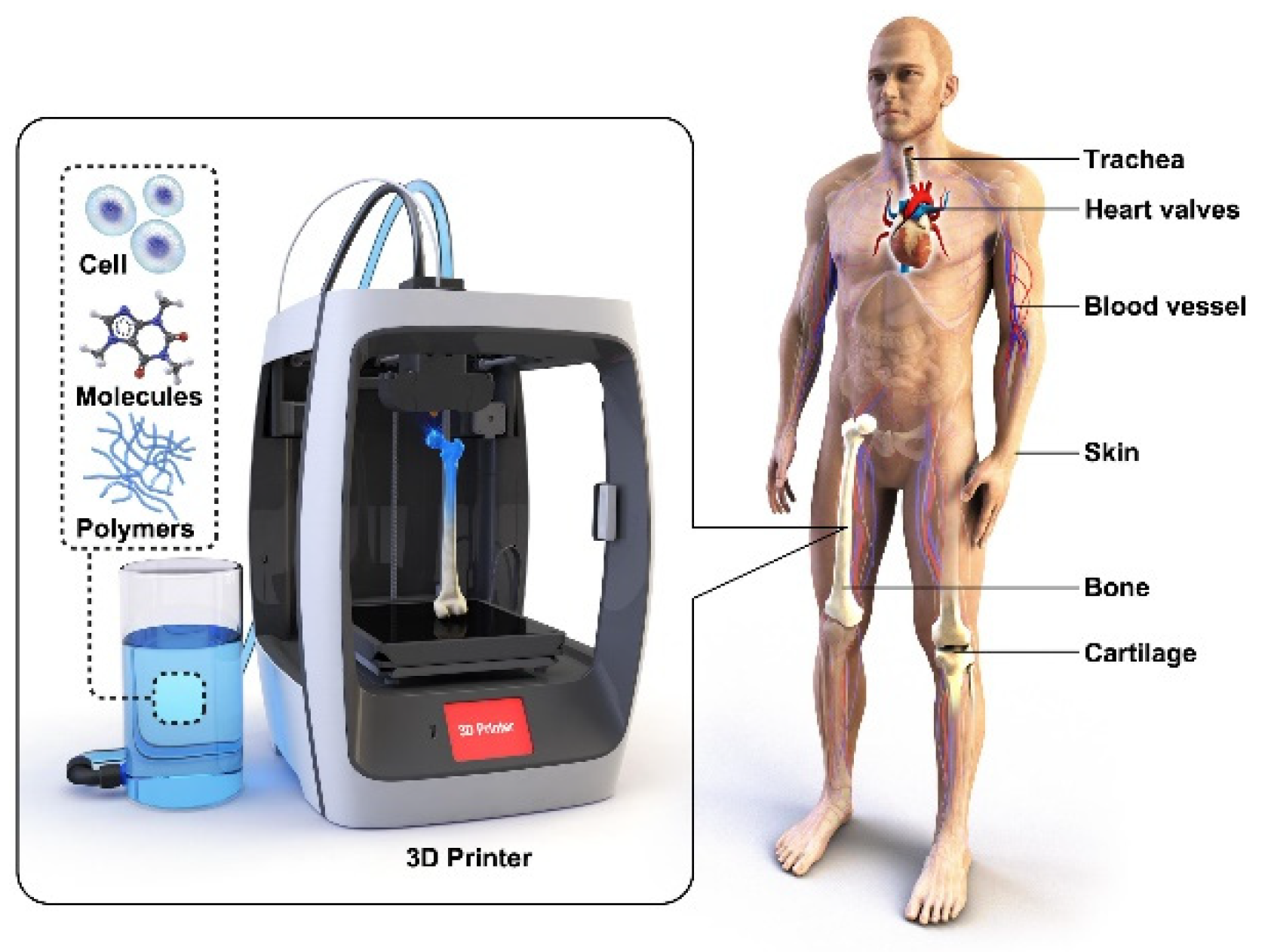




| Keywords | Number of Scientific Works |
|---|---|
| “additive manufacturing” or “AM” and “3D printing” | 69,341 |
| stereolithography (SLA) | 17,699 |
| selective laser sintering (SLS) | 17,366 |
| direct metal laser sintering (DMLS) | 1123 |
| electron-beam melting (EBM) | 8538 |
| selective heat sintering (SHS) | 16,220 |
| selective laser melting (SLM) | 16,474 |
| fused deposition modelling (FDM) + fused filament fabrication (FFF) | 16,645 |
| laminated object manufacturing (LOM) | 99 |
| laser-engineered net shaping | 232 |
| Summary | 147,263 |
| Printing Parameter | Mechanical Properties | Surface Quality | Morphological Changes |
|---|---|---|---|
| Layer thickness | + | + | − |
| Printing head temperature | + | − | + |
| Printing bed temperature | + | − | + |
| Printing speed | + | + | − |
| Infill percent | + | − | − |
| Infill shape | + | − | − |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pepelnjak, T.; Stojšić, J.; Sevšek, L.; Movrin, D.; Milutinović, M. Influence of Process Parameters on the Characteristics of Additively Manufactured Parts Made from Advanced Biopolymers. Polymers 2023, 15, 716. https://doi.org/10.3390/polym15030716
Pepelnjak T, Stojšić J, Sevšek L, Movrin D, Milutinović M. Influence of Process Parameters on the Characteristics of Additively Manufactured Parts Made from Advanced Biopolymers. Polymers. 2023; 15(3):716. https://doi.org/10.3390/polym15030716
Chicago/Turabian StylePepelnjak, Tomaž, Josip Stojšić, Luka Sevšek, Dejan Movrin, and Mladomir Milutinović. 2023. "Influence of Process Parameters on the Characteristics of Additively Manufactured Parts Made from Advanced Biopolymers" Polymers 15, no. 3: 716. https://doi.org/10.3390/polym15030716
APA StylePepelnjak, T., Stojšić, J., Sevšek, L., Movrin, D., & Milutinović, M. (2023). Influence of Process Parameters on the Characteristics of Additively Manufactured Parts Made from Advanced Biopolymers. Polymers, 15(3), 716. https://doi.org/10.3390/polym15030716








