On the Mechanism of the Ionizing Radiation-Induced Degradation and Recycling of Cellulose
Abstract
1. Introduction
1.1. Cellulose Sources
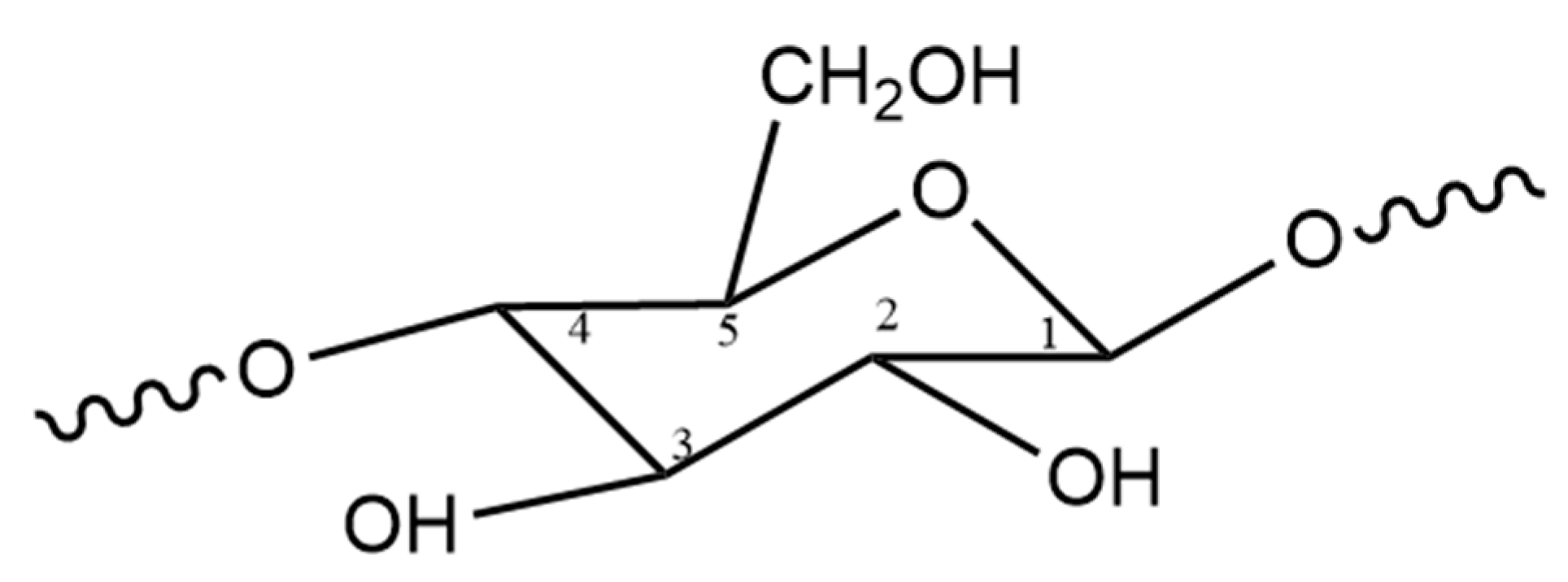
1.2. Cellulose Structure
1.3. Cellulose Extraction
1.4. Morphological Forms of Cellulose
1.5. Cellulose Fibers
1.6. Cellulose Filaments
1.7. Cellulose Nanofibrils
1.8. Crystalline Cellulose
1.9. Regenerated Cellulose
1.10. N-Methylmorpholine-N-Oxide (NMMO)
1.11. Ionic Liquids (ILs)
1.12. LiCl/N,N-Dimethylacetamide (LiCl/DMAc)
1.13. NaOH Aqueous Solutions
1.14. Alkali/Urea and NaOH/Thiourea Aqueous Solution
1.15. Tetra Butyl Ammonium Fluoride/Dimethyl Sulfoxide System
1.16. Metal Complex Solutions
1.17. Molten Inorganic Salt Hydrates
1.18. Functionalization of Cellulose
1.19. Cellulose Ethers
- Methyl cellulose—Methyl cellulose is the main commercial cellulose ether. It is the simplest alkyl ether and is synthesized in an alkaline medium with methylating agents like dimethyl sulfate or methyl chloride [70];
- Carboxymethyl cellulose—Carboxymethyl cellulose is synthesized by the reaction of cellulose with monochloroacetic acid. The C2, C3, and C6 hydroxyl groups are substituted by carboxymethyl groups. The substitution at the C2 position is favored slightly. There are no secondary OH groups formed in this process [71];
- Hydroxyethyl cellulose—Hydroxyethyl cellulose, which is prepared from the reaction of alkali cellulose and ethylene oxide. The chemical structure of hydroxyethyl cellulose can be easily further modified due to its reactive hydroxyl groups [51];
- Hydroxypropyl cellulose—Hydroxypropyl cellulose is formed in a reaction with 1,2-propylene oxide. The secondary OH groups can further react to form other novel compounds [74].
1.20. Cellulose Esters
- Cellulose nitrate, also referred to as nitrocellulose or celluloid, holds the distinction of being the inaugural semi-synthetic polymer in the realm of plastics. This substance is produced by subjecting cellulose to a chemical reaction with nitric acid. Within its structure, cellulose nitrate exhibits a polynitrate ester configuration, typically containing 2.2–2.8 nitrate groups per glucose unit [78];
- Cellulose sulfate—The sulfation of cellulose is done by the utilization of acids such as chlorosulfonic acid, sulfuric acid, or even sulfur trioxide [79].
1.21. Application of Cellulose and Its Derivatives
1.22. Principles of Ionization Radiation
- A.
- Physical stage (Ionization and excitation):
- B.
- Physico-chemical stage (Free radical formation):
- C.
- Chemical stage (Modifications in organic materials):
1.23. Reaction of Organic Free Radicals
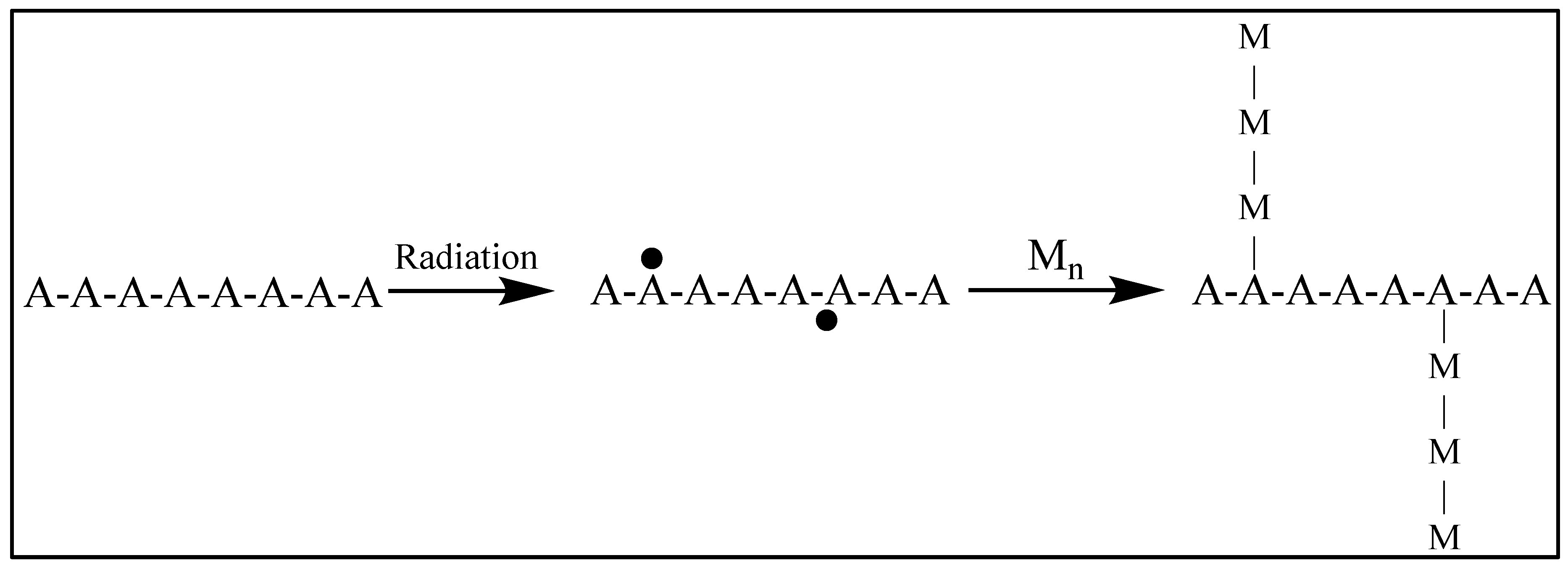
1.23.1. Crosslinking and Polymerization
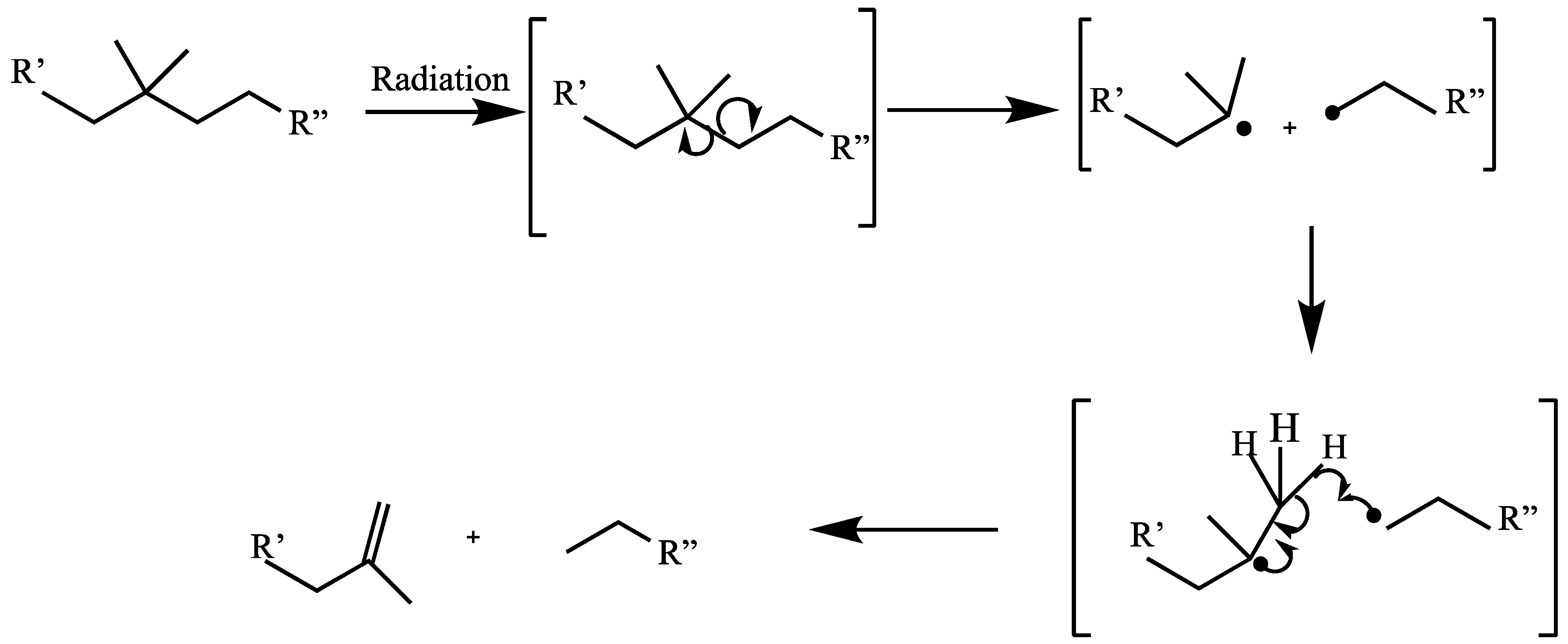
1.23.2. Scissioning
1.23.3. Crosslinking versus Main-Chain Scission
1.23.4. Curing
1.23.5. Grafting
1.23.6. Unsaturation
1.23.7. Gas Evolution
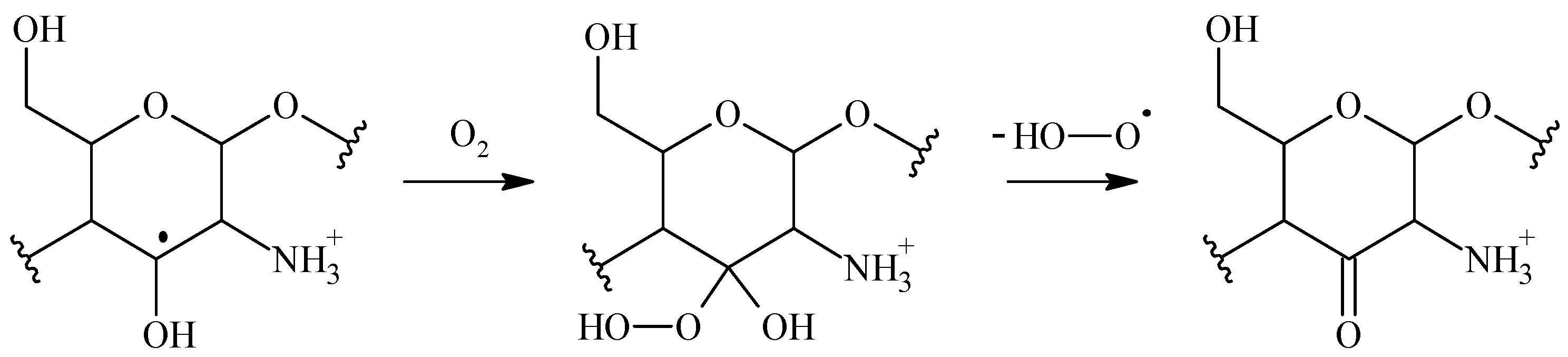
1.23.8. Influence of Atmospheric Oxygen
1.24. Radiation-Chemical Reactions in Aqueous Solutions
1.25. Properties of the Primary Species: The Hydrated Electron
1.26. Hydrogen Radicals
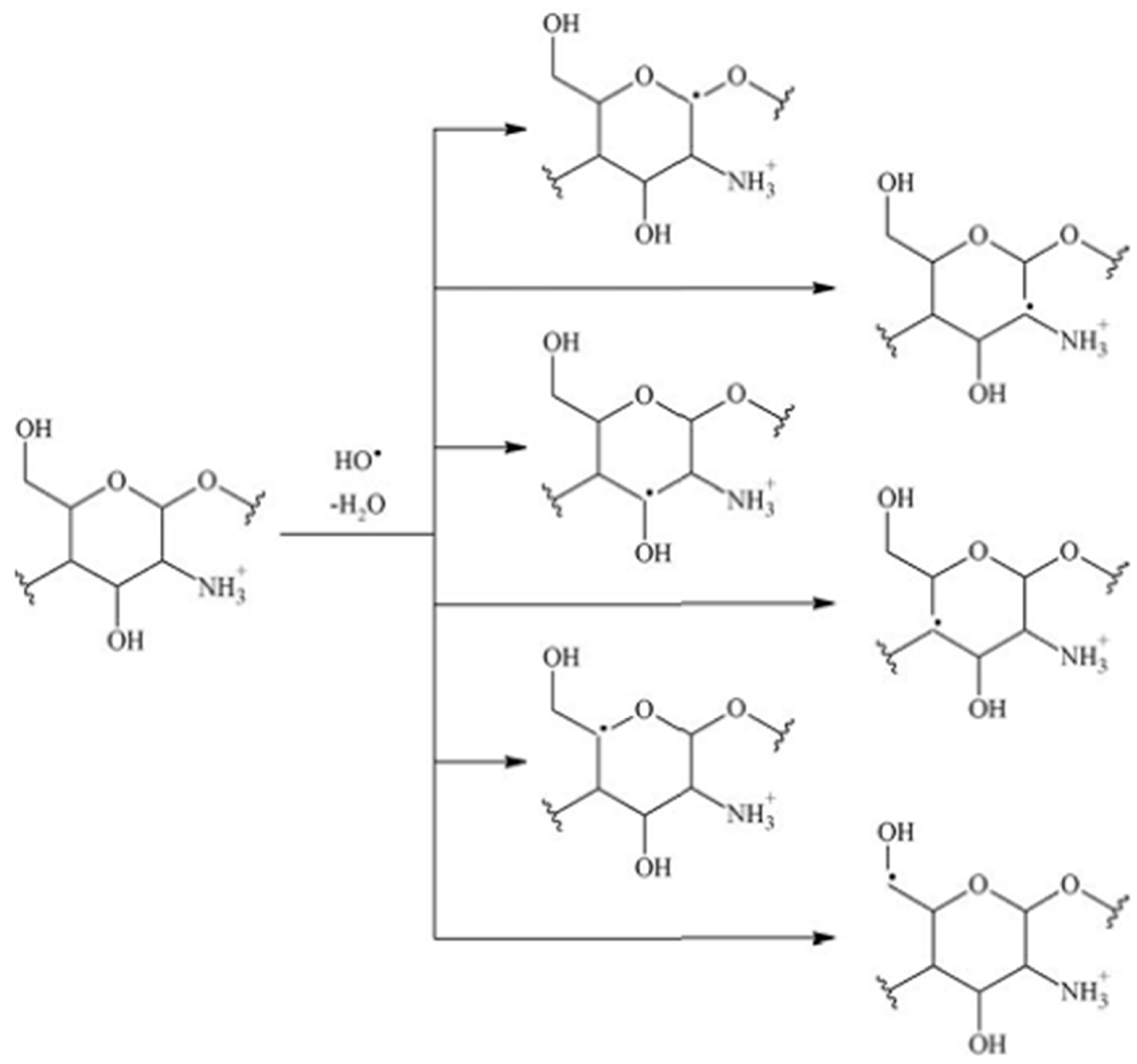
1.27. Hydroxyl Radical
Trapped Species
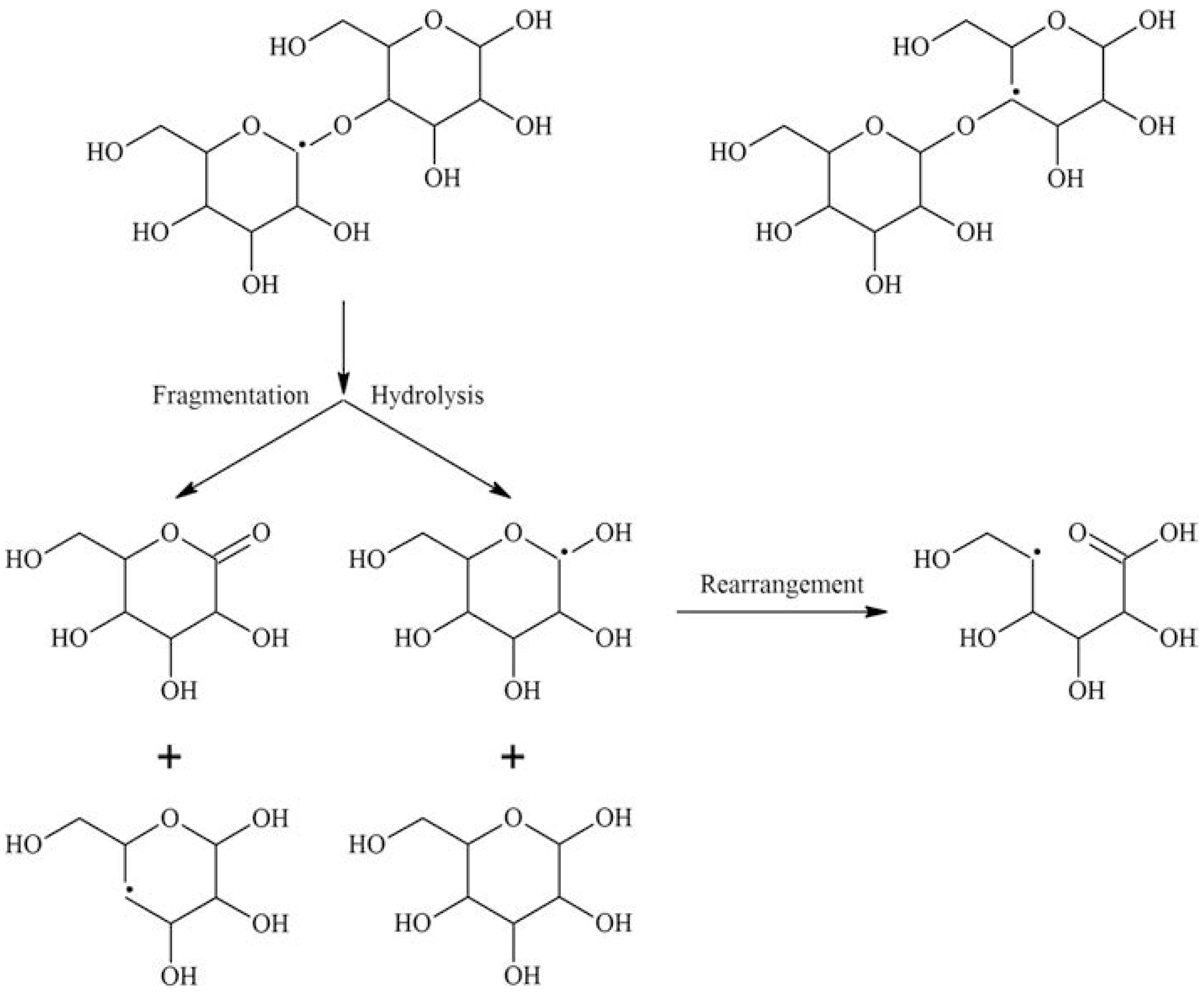
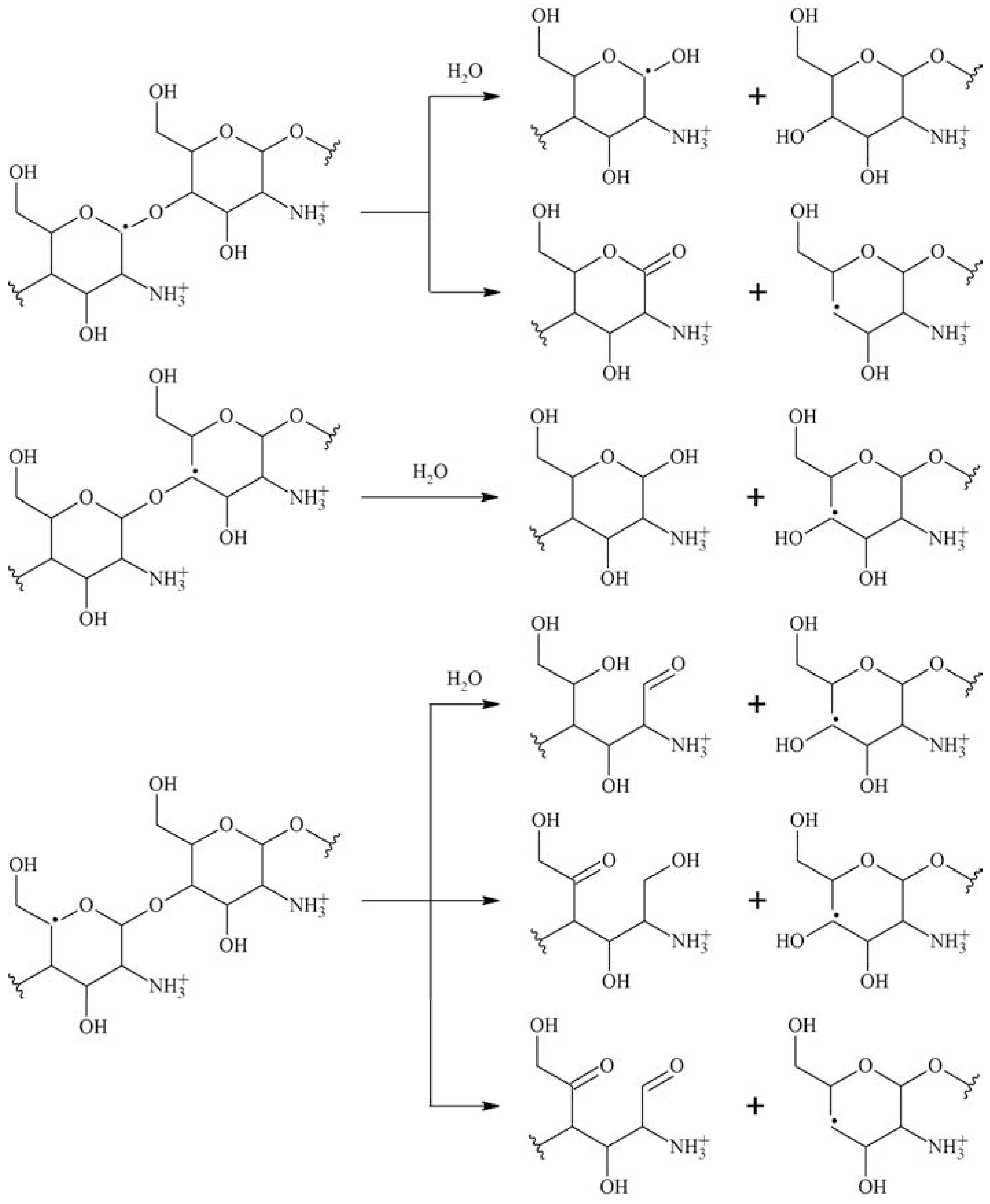
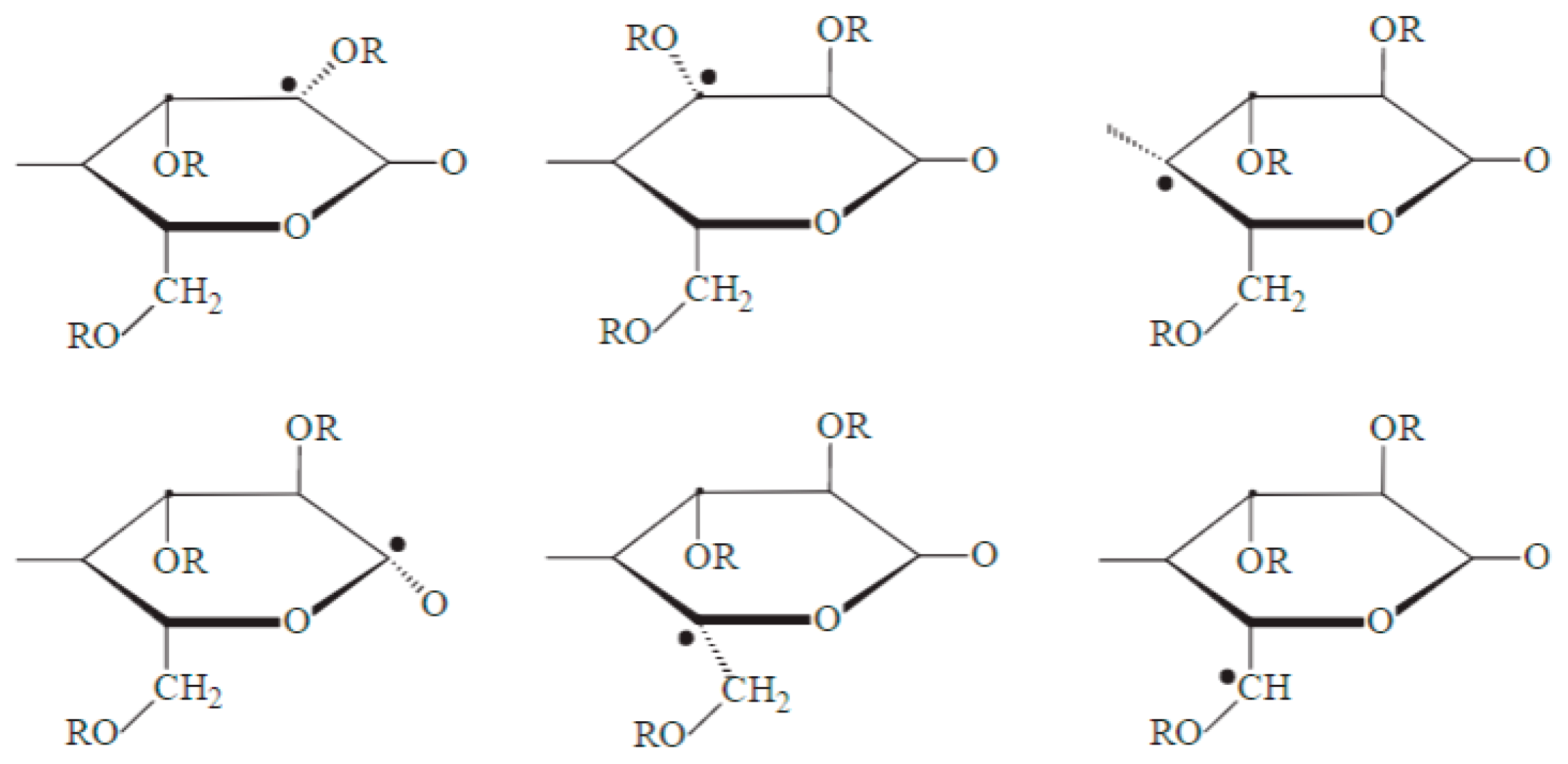
1.28. Irradiation of Polysaccharides
Scission
1.29. Crosslinking
1.30. Post Radiation Effects
1.31. Role of Ionizing Radiation in Cellulose
1.32. Ionizing Radiation in the Preparation of Cellulosic Feedstocks
2. Techniques to Identify Irradiation Effects on Cellulose
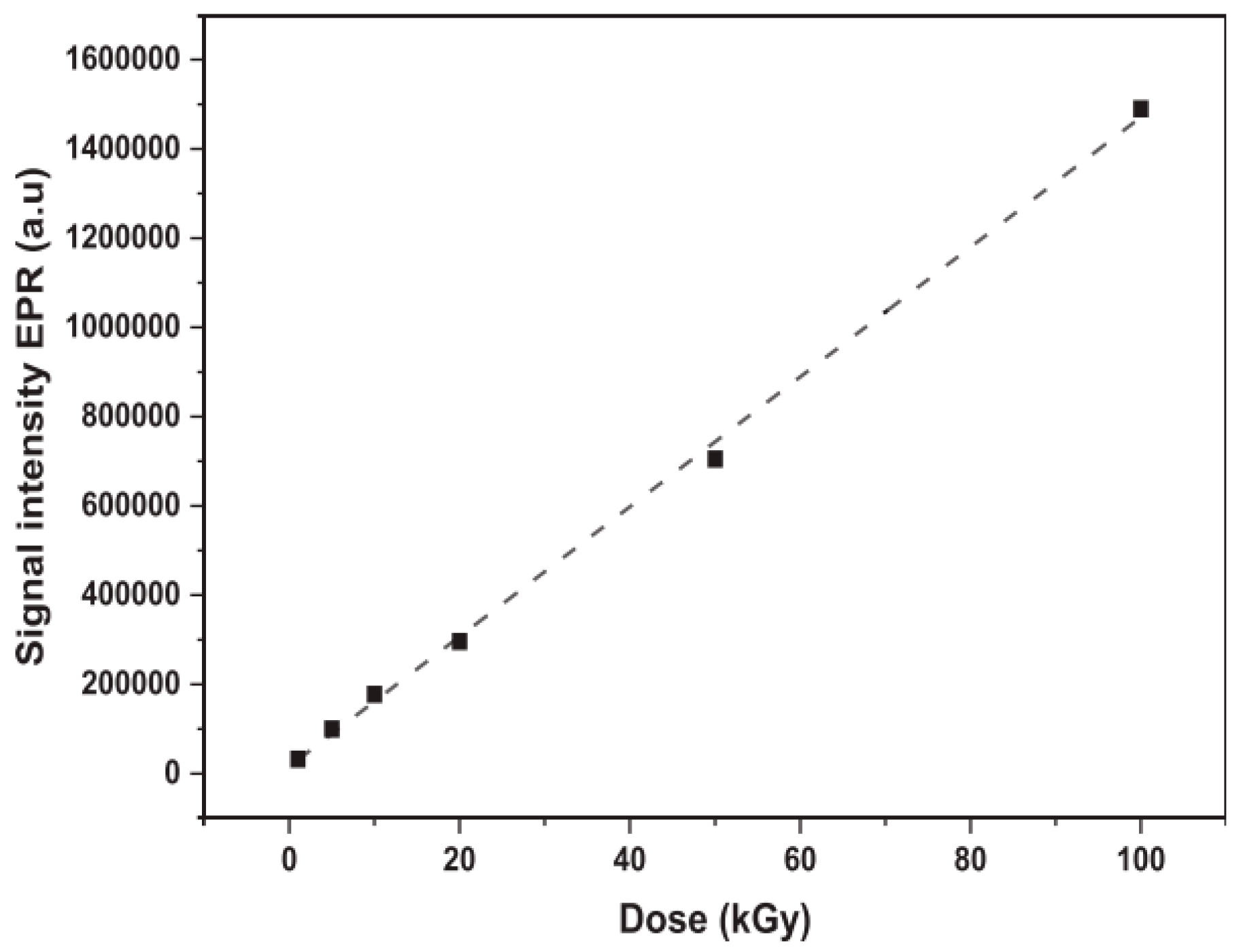
2.1. Electron Paramagnetic Resonance (EPR)
2.2. Gas Permeation Chromatography (GPC)
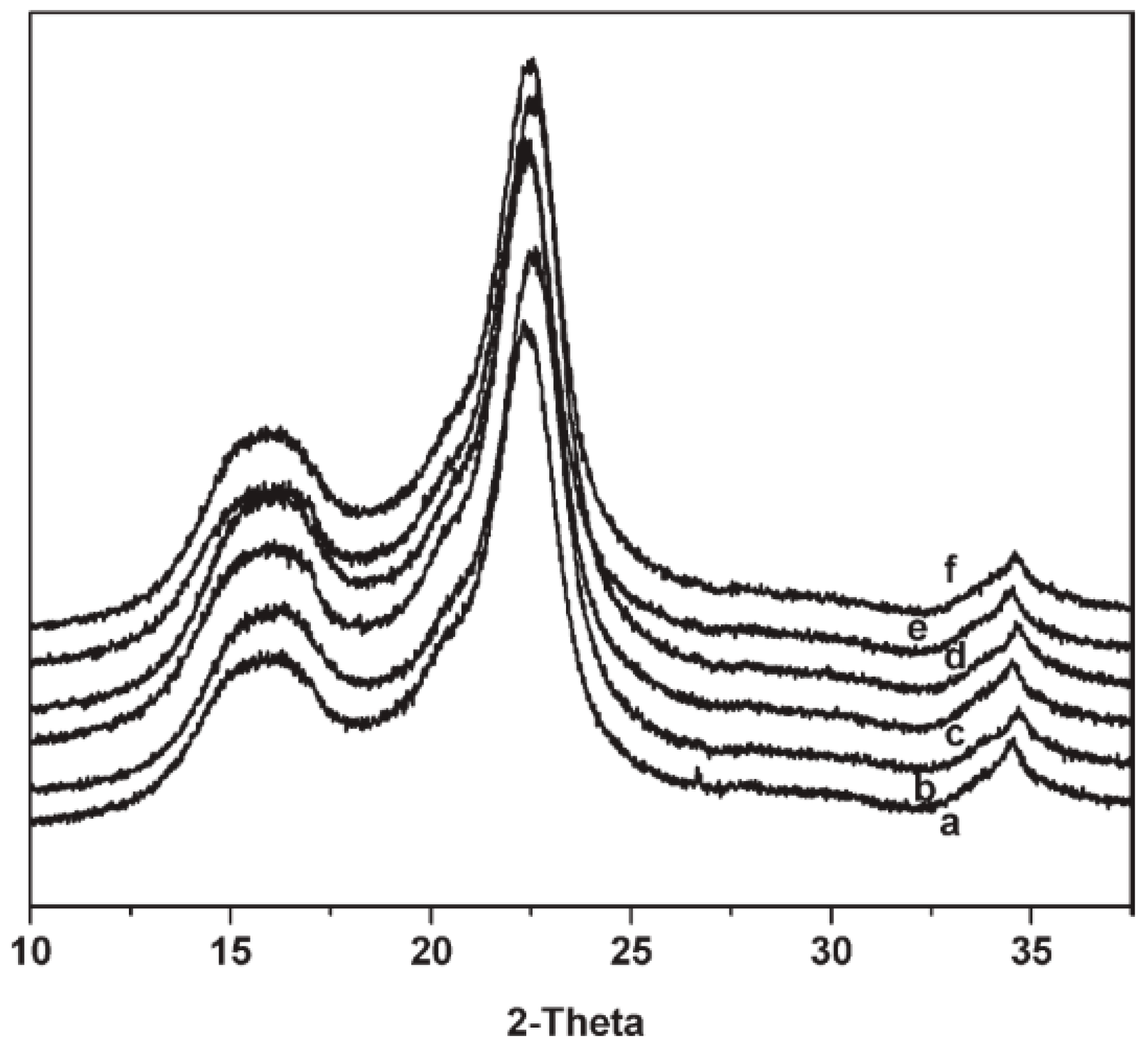
2.3. X-ray Diffraction (XRD)
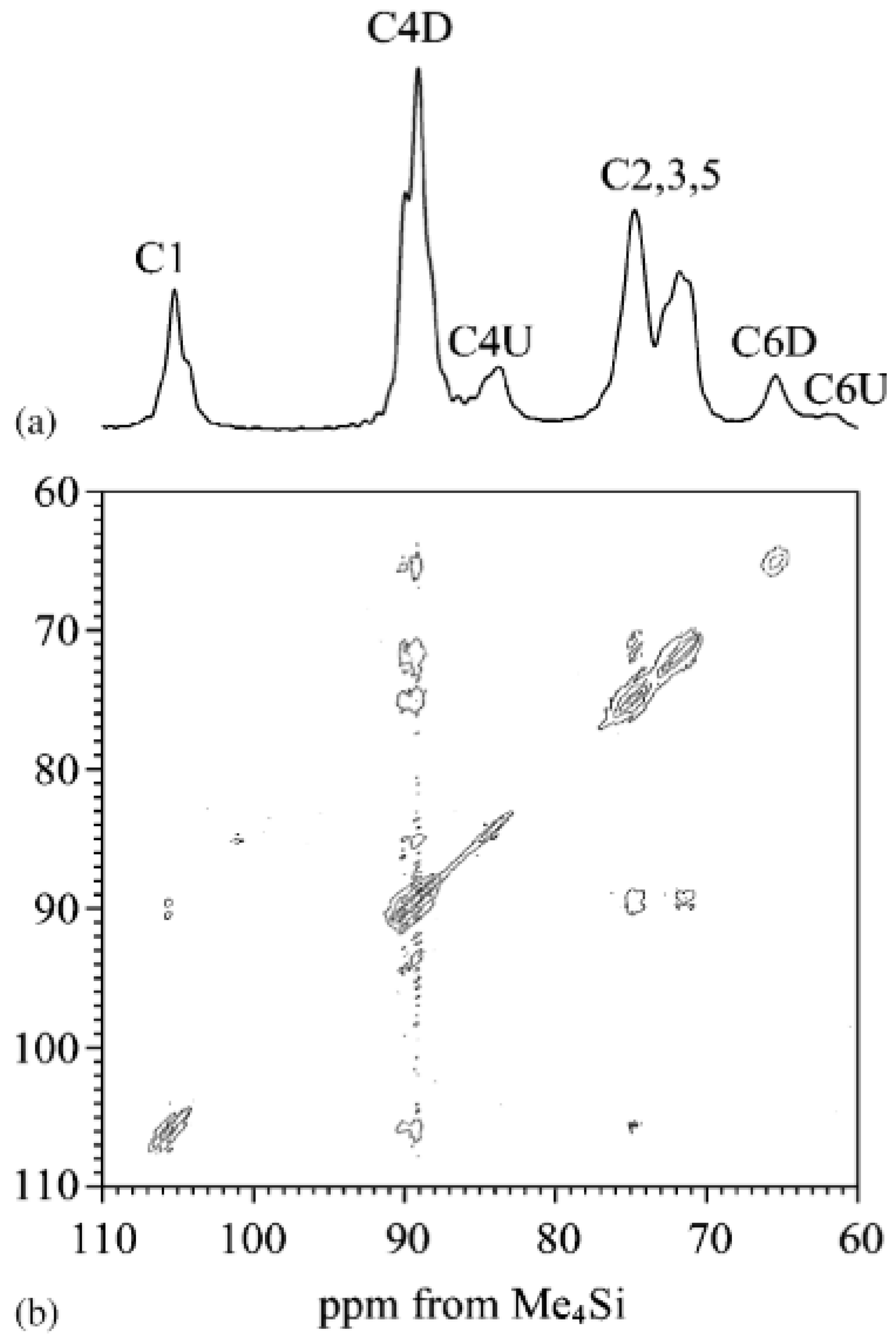
2.4. Solid-State Nuclear Magnetic Resonance (ss-NMR)
2.5. 1H and 13C Nuclear Magnetic Resonance (NMR) Spectra
2.6. Fourier Transform Infrared (FT-IR) Spectra
2.7. Raman Spectroscopy
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Peng, B.L.; Dhar, N.; Liu, H.L.; Tam, K.C. Chemistry and Applications of Nanocrystalline Cellulose and Its Derivatives: A Nanotechnology Perspective. Can. J. Chem. Eng. 2011, 89, 1191–1206. [Google Scholar] [CrossRef]
- Keshk, S.; Sameshima, K. Influence of Lignosulfonate on Crystal Structure and Productivity of Bacterial Cellulose in a Static Culture. Enzym. Microb. Technol. 2006, 40, 4–8. [Google Scholar] [CrossRef]
- Klemm, D.; Heublein, B.; Fink, H.-P.; Bohn, A. Cellulose: Fascinating Biopolymer and Sustainable Raw Material. Angew. Chem. Int. Ed. 2005, 44, 3358–3393. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.M. The Biosynthesis of Cellulose. J. Macromol. Sci. Part A 1996, 33, 1345–1373. [Google Scholar] [CrossRef]
- Ross, P.; Mayer, R.; Benziman, M. Cellulose Biosynthesis and Function in Bacteria. Microbiol. Rev. 1991, 55, 35–58. [Google Scholar] [CrossRef]
- Blanton, R.L.; Fuller, D.; Iranfar, N.; Grimson, M.J.; Loomis, W.F. The Cellulose Synthase Gene of Dictyostelium. Proc. Natl. Acad. Sci. USA 2000, 97, 2391–2396. [Google Scholar] [CrossRef]
- Kimura, S.; Chen, H.P.; Saxena, I.M.; Brown, R.M.; Itoh, T. Localization of C-Di-GMP-Binding Protein with the Linear Terminal Complexes of Acetobacter Xylinum. J. Bacteriol. 2001, 183, 5668–5674. [Google Scholar] [CrossRef]
- Römling, U. Molecular Biology of Cellulose Production in Bacteria. Res. Microbiol. 2002, 153, 205–212. [Google Scholar] [CrossRef]
- Iguchi, M.; Yamanaka, S.; Budhiono, A. Bacterial Cellulose-a Masterpiece of Nature’s Arts. J. Mater. Sci. 2000, 35, 261–270. [Google Scholar] [CrossRef]
- Moon, R.J.; Martini, A.; Nairn, J.; Simonsen, J.; Youngblood, J. Cellulose Nanomaterials Review: Structure, Properties and Nanocomposites. Chem. Soc. Rev. 2011, 40, 3941. [Google Scholar] [CrossRef]
- Pogorelova, N.; Rogachev, E.; Digel, I.; Chernigova, S.; Nardin, D. Bacterial Cellulose Nanocomposites: Morphology and Mechanical Properties. Materials 2020, 13, 2849. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yu, H.; Liu, Y.; Chen, P.; Zhang, M.; Hai, Y. Individualization of Cellulose Nanofibers from Wood Using High-Intensity Ultrasonication Combined with Chemical Pretreatments. Carbohydr. Polym. 2011, 83, 1804–1811. [Google Scholar] [CrossRef]
- Klemm, D.; Cranston, E.D.; Fischer, D.; Gama, M.; Kedzior, S.A.; Kralisch, D.; Kramer, F.; Kondo, T.; Lindström, T.; Nietzsche, S.; et al. Nanocellulose as a Natural Source for Groundbreaking Applications in Materials Science: Today’s State. Mater. Today 2018, 21, 720–748. [Google Scholar] [CrossRef]
- Zhong, C. Industrial-Scale Production and Applications of Bacterial Cellulose. Front. Bioeng. Biotechnol. 2020, 8, 605374. [Google Scholar] [CrossRef] [PubMed]
- Global Cellulosic Materials Market Report 2021-Nano-cellulose and Cellulose Hydrogel/Aerogels to Gain Commercial Adoption; PR Newswire: Chicago, IL, USA, 2022; Available online: https://www.prnewswire.com/news-releases/global-cellulosic-materials-market-report-2021---nano-cellulose-and-cellulose-hydrogelaerogels-to-gain-commercial-adoption-301460780.html (accessed on 15 April 2023).
- McNaughy, A.D.; Wilkinson, A. The IUPAC Compendium of Chemical Terminology; Gold, V., Ed.; International Union of Pure and Applied Chemistry (IUPAC): Research Triangle Park, NC, USA, 2019. [Google Scholar] [CrossRef]
- Web, N. Ionizing Radiation. Available online: https://www.nrc.gov/reading-rm/basic-ref/glossary/ionizing-radiation.html (accessed on 15 April 2023).
- Seddiqi, H.; Oliaei, E.; Honarkar, H.; Jin, J.; Geonzon, L.C.; Bacabac, R.G.; Klein-Nulend, J. Cellulose and Its Derivatives: Towards Biomedical Applications. Cellulose 2021, 28, 1893–1931. [Google Scholar] [CrossRef]
- Madsen, B.; Gamstedt, E.K. Wood versus Plant Fibers: Similarities and Differences in Composite Applications. Adv. Mater. Sci. Eng. 2013, 2013, 564346. [Google Scholar] [CrossRef]
- Hallac, B.B.; Ragauskas, A.J. Analyzing Cellulose Degree of Polymerization and Its Relevancy to Cellulosic Ethanol. Biofuels Bioprod. Biorefining 2011, 5, 215–225. [Google Scholar] [CrossRef]
- Brown, A.J. XLIII.—On an Acetic Ferment Which Forms Cellulose. J. Chem. Soc. Trans. 1886, 49, 432–439. [Google Scholar] [CrossRef]
- Moon, R.J.; Schueneman, G.T.; Simonsen, J. Overview of Cellulose Nanomaterials, Their Capabilities and Applications. JOM 2016, 68, 2383–2394. [Google Scholar] [CrossRef]
- Rabemanolontsoa, H.; Saka, S. Comparative Study on Chemical Composition of Various Biomass Species. RSC Adv. 2013, 3, 3946. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, J. Excellent Chemical and Material Cellulose from Tunicates: Diversity in Cellulose Production Yield and Chemical and Morphological Structures from Different Tunicate Species. Cellulose 2014, 21, 3427–3441. [Google Scholar] [CrossRef]
- Zhu, G.; Xu, H.; Dufresne, A.; Lin, N. High-Adsorption, Self-Extinguishing, Thermal, and Acoustic-Resistance Aerogels Based on Organic and Inorganic Waste Valorization from Cellulose Nanocrystals and Red Mud. ACS Sustain. Chem. Eng. 2018, 6, 7168–7180. [Google Scholar] [CrossRef]
- French, A.D.; Santiago Cintrón, M. Cellulose Polymorphy, Crystallite Size, and the Segal Crystallinity Index. Cellulose 2013, 20, 583–588. [Google Scholar] [CrossRef]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose Nanocrystals: Chemistry, Self-Assembly, and Applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Sabapathi, S.N. Cellulose Nanocrystals: Synthesis, Functional Properties, and Applications. Nanotechnol. Sci. Appl. 2015, 8, 45–54. [Google Scholar] [CrossRef]
- Qing, Y.; Sabo, R.; Zhu, J.Y.; Agarwal, U.; Cai, Z.; Wu, Y. A Comparative Study of Cellulose Nanofibrils Disintegrated via Multiple Processing Approaches. Carbohydr. Polym. 2013, 97, 226–234. [Google Scholar] [CrossRef]
- Peterlin, A.; Ingram, P. Morphology of Secondary Wall Fibrils in Cotton. Text. Res. J. 1970, 40, 345–354. [Google Scholar] [CrossRef]
- Agarwal, U.P.; Ralph, S.A.; Reiner, R.S.; Baez, C. Probing Crystallinity of Never-Dried Wood Cellulose with Raman Spectroscopy. Cellulose 2016, 23, 125–144. [Google Scholar] [CrossRef]
- Mazeau, K.; Heux, L. Molecular Dynamics Simulations of Bulk Native Crystalline and Amorphous Structures of Cellulose. J. Phys. Chem. B 2003, 107, 2394–2403. [Google Scholar] [CrossRef]
- O’sullvian, A.C. Cellulose: The Structure Slowly Unravels. Cellulose 1997, 4, 173–207. [Google Scholar] [CrossRef]
- Ioelovich, M. Cellulose: Nanostructured Natural Polymer; LAP Lambert Academic Publishing: Sunnyvale, CA, USA, 2014. [Google Scholar]
- Avolio, R.; Bonadies, I.; Capitani, D.; Errico, M.E.; Gentile, G.; Avella, M. A Multitechnique Approach to Assess the Effect of Ball Milling on Cellulose. Carbohydr. Polym. 2012, 87, 265–273. [Google Scholar] [CrossRef]
- Brinchi, L.; Cotana, F.; Fortunati, E.; Kenny, J.M. Production of Nanocrystalline Cellulose from Lignocellulosic Biomass: Technology and Applications. Carbohydr. Polym. 2013, 94, 154–169. [Google Scholar] [CrossRef] [PubMed]
- Atalla, R.H.; Atalla, R.S.; Agarwal, U.P. The Nanostructures of Native Celluloses, Their Transformations upon Isolation, and Their Implications for Production of Nanocelluloses. In Nanocelluloses: Their Preparation, Properties, and Applications; American Chemical Society: Washington, DC, USA, 2017. [Google Scholar] [CrossRef]
- Wada, M.; Ike, M.; Tokuyasu, K. Enzymatic Hydrolysis of Cellulose I Is Greatly Accelerated via Its Conversion to the Cellulose II Hydrate Form. Polym. Degrad. Stab. 2010, 95, 543–548. [Google Scholar] [CrossRef]
- Mercer, J. Improvements in the Preparation of Cotton and Other Fabrics and Other Fibrous Materials. Br. Pat. 1850, 13, 1850. [Google Scholar]
- Nishiyama, Y. Structure and Properties of the Cellulose Microfibril. J. Wood Sci. 2009, 55, 241–249. [Google Scholar] [CrossRef]
- Bajpai, P. Biermann’s Handbook of Pulp and Paper, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Brown, F.B.H. The Preparation and Treatment of Woods for Microscopic Study. Bull. Torrey Bot. Club 1919, 46, 127–150. [Google Scholar] [CrossRef]
- Berlyn, G.; Miksche, J.; Sass, J. Botanical Microtechnique and Cytochemistry, 1st ed.; Iowa State University Press: Ames, IA, USA, 1976. [Google Scholar]
- Kryn, J.M. Annotated List of References On the Preparation of Wood for Microscopic Study; U.S. Dept. of Agriculture, Forest Service, Forest Products Laboratory: Madison, WI, USA, 1953.
- Mahesh, S.; Kumar, P.; Ansari, S.A. A Rapid and Economical Method for the Maceration of Wood Fibers in Boswellia Serrata Roxb. Trop. Plant Res. 2015, 2, 108–111. [Google Scholar]
- Ardanuy, M.; Claramunt, J.; Toledo Filho, R.D. Cellulosic Fiber Reinforced Cement-Based Composites: A Review of Recent Research. Constr. Build. Mater. 2015, 79, 115–128. [Google Scholar] [CrossRef]
- Walther, A.; Timonen, J.V.I.; Díez, I.; Laukkanen, A.; Ikkala, O. Multifunctional High-Performance Biofibers Based on Wet-Extrusion of Renewable Native Cellulose Nanofibrils. Adv. Mater. 2011, 23, 2924–2928. [Google Scholar] [CrossRef]
- Håkansson, K.M.O.; Fall, A.B.; Lundell, F.; Yu, S.; Krywka, C.; Roth, S.V.; Santoro, G.; Kvick, M.; Prahl Wittberg, L.; Wågberg, L.; et al. Hydrodynamic Alignment and Assembly of Nanofibrils Resulting in Strong Cellulose Filaments. Nat. Commun. 2014, 5, 4018. [Google Scholar] [CrossRef]
- Vuoriluoto, M.; Orelma, H.; Lundahl, M.; Borghei, M.; Rojas, O.J. Filaments with Affinity Binding and Wet Strength Can Be Achieved by Spinning Bifunctional Cellulose Nanofibrils. Biomacromolecules 2017, 18, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Tibolla, H.; Pelissari, F.M.; Martins, J.T.; Vicente, A.A.; Menegalli, F.C. Cellulose Nanofibers Produced from Banana Peel by Chemical and Mechanical Treatments: Characterization and Cytotoxicity Assessment. Food Hydrocoll. 2018, 75, 192–201. [Google Scholar] [CrossRef]
- Wang, S.; Lu, A.; Zhang, L. Recent Advances in Regenerated Cellulose Materials. Prog. Polym. Sci. 2016, 53, 169–206. [Google Scholar] [CrossRef]
- Wang, H.; Gurau, G.; Rogers, R.D. Ionic Liquid Processing of Cellulose. Chem. Soc. Rev. 2012, 41, 1519–1537. [Google Scholar] [CrossRef]
- Rosenau, T.; Potthast, A.; Sixta, H.; Kosma, P. The Chemistry of Side Reactions and Byproduct Formation in the System NMMO/Cellulose (Lyocell Process). Prog. Polym. Sci. 2001, 26, 1763–1837. [Google Scholar] [CrossRef]
- Gagnaire, D.; Mancier, D.; Vincendon, M. Cellulose Organic Solutions: A Nuclear Magnetic Resonance Investigation. J. Polym. Sci. Polym. Chem. Ed. 1980, 18, 13–25. [Google Scholar] [CrossRef]
- Pinkert, A.; Marsh, K.N.; Pang, S.; Staiger, M.P. Ionic Liquids and Their Interaction with Cellulose. Chem. Rev. 2009, 109, 6712–6728. [Google Scholar] [CrossRef]
- Swatloski, R.P.; Spear, S.K.; Holbrey, J.D.; Rogers, R.D. Dissolution of Cellose with Ionic Liquids. J. Am. Chem. Soc. 2002, 124, 4974–4975. [Google Scholar] [CrossRef]
- Zhu, S.; Wu, Y.; Chen, Q.; Yu, Z.; Wang, C.; Jin, S.; Ding, Y.; Wu, G. Dissolution of Cellulose with Ionic Liquids and Its Application: A Mini-Review. Green Chem. 2006, 8, 325–327. [Google Scholar] [CrossRef]
- Henniges, U.; Schiehser, S.; Rosenau, T.; Potthast, A. Cellulose Solubility: Dissolution and Analysis of “Problematic” Cellulose Pulps in the Solvent System DMAc/LiCl. ACS Symp. Ser. 2010, 1033, 165–177. [Google Scholar] [CrossRef]
- Cuissinat, C.; Navard, P. Swelling and Dissolution of Cellulose Part 1: Free Floating Cotton and Wood Fibres in N-Methylmorpholine-N-Oxide–Water Mixtures. Macromol. Symp. 2006, 244, 1–18. [Google Scholar] [CrossRef]
- Cuissinat, C.; Navard, P. Swelling and Dissolution of Cellulose Part II: Free Floating Cotton and Wood Fibres in NaOH–Water–Additives Systems. Macromol. Symp. 2006, 244, 19–30. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, W.C.; Li, F.X.; Yu, J.Y. Swelling and Dissolution of Cellulose in NaOH Aqueous Solvent Systems. Cellul. Chem. Technol. 2013, 47, 671–679. [Google Scholar]
- Qi, H.; Chang, C.; Zhang, L. Effects of Temperature and Molecular Weight on Dissolution of Cellulose in NaOH/Urea Aqueous Solution. Cellulose 2008, 15, 779–787. [Google Scholar] [CrossRef]
- Jiang, Z.; Fang, Y.; Xiang, J.; Ma, Y.; Lu, A.; Kang, H.; Huang, Y.; Guo, H.; Liu, R.; Zhang, L. Intermolecular Interactions and 3D Structure in Cellulose-NaOH-Urea Aqueous System. J. Phys. Chem. B 2014, 118, 10250–10257. [Google Scholar] [CrossRef]
- Lue, A.; Zhang, L. Advances in Aqueous Cellulose Solvents. In Cellulose Solvents: For Analysis, Shaping and Chemical Modification; American Chemical Society: Washington, DC, USA, 2010. [Google Scholar] [CrossRef]
- Heinze, T.; Dicke, R.; Koschella, A.; Kull, A.H.; Klohr, E.-A.; Koch, W. Effective Preparation of Cellulose Derivatives in a New Simple Cellulose Solvent. Macromol. Chem. Phys. 2000, 201, 627–631. [Google Scholar] [CrossRef]
- Heinze, T.; Koschella, A. Solvents Applied in the Field of Cellulose Chemistry: A Mini Review. Polímeros 2005, 15, 84–90. [Google Scholar] [CrossRef]
- Yang, Y.J.; Shin, J.M.; Kang, T.H.; Kimura, S.; Wada, M.; Kim, U.J. Cellulose Dissolution in Aqueous Lithium Bromide Solutions. Cellulose 2014, 21, 1175–1181. [Google Scholar] [CrossRef]
- Zheng, X.; Gandour, R.D.; Edgar, K.J. Remarkably Regioselective Deacylation of Cellulose Esters Using Tetraalkylammonium Salts of the Strongly Basic Hydroxide Ion. Carbohydr. Polym. 2014, 111, 25–32. [Google Scholar] [CrossRef]
- Kamel, S.; Ali, N.; Jahangir, K.; Shah, S.M.; El-Gendy, A.A. Pharmaceutical Significance of Cellulose: A Review. Express Polym. Lett. 2008, 2, 758–778. [Google Scholar] [CrossRef]
- Viera, R.G.P.; Filho, G.R.; de Assunção, R.M.N.; da Carla, C.; Vieira, J.G.; de Oliveira, G.S. Synthesis and Characterization of Methylcellulose from Sugar Cane Bagasse Cellulose. Carbohydr. Polym. 2007, 67, 182–189. [Google Scholar] [CrossRef]
- Heinze, T.; Pfeiffer, K. Studies on the Synthesis and Characterization of Carboxymethylcellulose. Die Angew. Makromol. Chem. 1999, 266, 37–45. [Google Scholar] [CrossRef]
- Rekhi, G.S.; Jambhekar, S.S. Ethylcellulose-A Polymer Review. Drug Dev. Ind. Pharm. 1995, 21, 61–77. [Google Scholar] [CrossRef]
- Adeleke, O.A. Premium Ethylcellulose Polymer Based Architectures at Work in Drug Delivery. Int. J. Pharm. X 2019, 1, 100023. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, J.; Sun, H.; Hong, D.; Li, L.; Yang, Y.; Yong, X.; Zhang, C.; Cui, J. An Optical Fiber Relative Humidity Sensor Based on Hollow-Core Fiber and Hydroxypropyl Methylcellulose Hydrogel Film. Optik 2019, 195, 163172. [Google Scholar] [CrossRef]
- Edgar, K.J.; Buchanan, C.M.; Debenham, J.S.; Rundquist, P.A.; Seiler, B.D.; Shelton, M.C.; Tindall, D. Advances in Cellulose Ester Performance and Applicaton.Pdf.Crdownload. Prog. Polym. Sci. 2001, 26, 1605–1688. [Google Scholar] [CrossRef]
- Kulterer, M.R.; Reischl, M.; Reichel, V.E.; Hribernik, S.; Wu, M.; Köstler, S.; Kargl, R.; Ribitsch, V. Nanoprecipitation of Cellulose Acetate Using Solvent/Nonsolvent Mixtures as Dispersive Media. Colloids Surf. A Physicochem. Eng. Asp. 2011, 375, 23–29. [Google Scholar] [CrossRef]
- Cheng, K.; Shannon, S.S. An Investigation of Lignocellulosic Biomass Including: Compositional Modeling, Novel Pretreatments and Extraction of Cellulose Nanocrystals. Ph.D. Thesis, State University of New York College of Environmental Science and Forestry, Syracuse, NY, USA, 2010. [Google Scholar]
- Neves, A.; Angelin, E.M.; Roldão, É.; Melo, M.J. New Insights into the Degradation Mechanism of Cellulose Nitrate in Cinematographic Films by Raman Microscopy. J. Raman Spectrosc. 2019, 50, 202–212. [Google Scholar] [CrossRef]
- Schweiger, R.G. Polysaccharide Sulfates. I. Cellulose Sulfate with a High Degree of Substitution. Carbohydr. Res. 1972, 21, 219–228. [Google Scholar] [CrossRef]
- Li, T.; Chen, C.; Brozena, A.H.; Zhu, J.Y.; Xu, L.; Driemeier, C.; Dai, J.; Rojas, O.J.; Isogai, A.; Wågberg, L.; et al. Developing Fibrillated Cellulose as a Sustainable Technological Material. Nature 2021, 590, 47–56. [Google Scholar] [CrossRef]
- Azimi, B.; Ricci, C.; Fusco, A.; Zavagna, L.; Linari, S.; Donnarumma, G.; Hadrich, A.; Cinelli, P.; Coltelli, M.B.; Danti, S.; et al. Electrosprayed Shrimp and Mushroom Nanochitins on Cellulose Tissue for Skin Contact Application. Molecules 2021, 26, 4374. [Google Scholar] [CrossRef] [PubMed]
- Costa, E.M.; Pereira, C.F.; Ribeiro, A.A.; Casanova, F.; Freixo, R.; Pintado, M.; Ramos, O.L. Characterization and Evaluation of Commercial Carboxymethyl Cellulose Potential as an Active Ingredient for Cosmetics. Appl. Sci. 2022, 12, 6560. [Google Scholar] [CrossRef]
- Mbituyimana, B.; Liu, L.; Ye, W.; Ode Boni, B.O.; Zhang, K.; Chen, J.; Thomas, S.; Vasilievich, R.V.; Shi, Z.; Yang, G. Bacterial Cellulose-Based Composites for Biomedical and Cosmetic Applications: Research Progress and Existing Products. Carbohydr. Polym. 2021, 273, 118565. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.S.; Hasan, M.S.; Nitai, A.S.; Nam, S.; Karmakar, A.K.; Ahsan, M.S.; Shiddiky, M.J.A.; Ahmed, M.B. Recent Developments of Carboxymethyl Cellulose. Polymers 2021, 13, 1345. [Google Scholar] [CrossRef] [PubMed]
- Martins, D.; Rocha, C.; Dourado, F.; Gama, M. Bacterial Cellulose-Carboxymethyl Cellulose (BC:CMC) Dry Formulation as Stabilizer and Texturizing Agent for Surfactant-Free Cosmetic Formulations. Colloids Surf. A Physicochem. Eng. Asp. 2021, 617, 126380. [Google Scholar] [CrossRef]
- Reimer, M.; Zollfrank, C. Cellulose for Light Manipulation: Methods, Applications, and Prospects. Adv. Energy Mater. 2021, 11, 2003866. [Google Scholar] [CrossRef]
- Zhu, H.; Fang, Z.; Wang, Z.; Dai, J.; Yao, Y.; Shen, F.; Preston, C.; Wu, W.; Peng, P.; Jang, N.; et al. Extreme Light Management in Mesoporous Wood Cellulose Paper for Optoelectronics. ACS Nano 2016, 10, 1369–1377. [Google Scholar] [CrossRef]
- Chen, Q.; Ying, D.; Chen, Y.; Xie, H.; Zhang, H.; Chang, C. Highly Transparent, Hydrophobic, and Durable Anisotropic Cellulose Films as Electronic Screen Protectors. SSRN Electron. J. 2022. [Google Scholar] [CrossRef]
- Sabharwal, S.; Products, R.; Atomic, I.; Agency, E. Electron Beam Irradiation Applications. In Proceedings of the 25th Particle Accelerator Conference, Pasadena, CA, USA, 29 September–4 October 2013; pp. 745–748. [Google Scholar]
- Cleland, M.R.; Parks, L.A.; Cheng, S. Applications for Radiation Processing of Materials. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2003, 208, 66–73. [Google Scholar] [CrossRef]
- Chaudhary, N.; Singh, A.; Debnath, A.K.; Acharya, S.; Aswal, D.K. Electron Beam Modified Organic Materials and Their Applications. Solid State Phenom. 2015, 239, 72–97. [Google Scholar] [CrossRef]
- Krumeich, F. Properties of Electrons, Their Interactions with Matter. ETH Zur. 2015, 1–24. [Google Scholar]
- Raja, S.N.; Basu, S.; Limaye, A.M.; Anderson, T.J.; Hyland, C.M.; Lin, L.; Alivisatos, A.P.; Ritchie, R.O. Strain-Dependent Dynamic Mechanical Properties of Kevlar to Failure: Structural Correlations and Comparisons to Other Polymers. Mater. Today Commun. 2015, 2, e33–e37. [Google Scholar] [CrossRef]
- Woods, R.; Pikaev, A. Applied Radiation Chemistry: Radiation Processing, 1st ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1993. [Google Scholar]
- Kónya, J.; Nagy, N. Nuclear and Radiochemistry; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar] [CrossRef]
- Goldstein, J.I.; Newbury, D.E.; Michael, J.R.; Ritchie, N.W.M.; Scott, J.H.J.; Joy, D.C. Scanning Electron Microscopy and X-ray Microanalysis; Springer New York: New York, NY, USA, 2018. [Google Scholar] [CrossRef]
- Navarro, R.; Burillo, G.; Adem, E.; Marcos-Fernández, A. Effect of Ionizing Radiation on the Chemical Structure and the Physical Properties of Polycaprolactones of Different Molecular Weight. Polymers 2018, 10, 397. [Google Scholar] [CrossRef] [PubMed]
- Titchmarsh, J. Handbook of Surface and Interface Analysis; Riviere, J.C., Myhra, S., Eds.; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar] [CrossRef]
- Williams, D.B.; Carter, C.B. Transmission Electron Microscopy; Springer: Boston, MA, USA, 2009. [Google Scholar] [CrossRef]
- Dapor, M. (Ed.) Electron-Beam Interactions with Solids; Springer: Berlin/Heidelberg, Germany, 2003. [Google Scholar] [CrossRef]
- Ciesla, K.A. Radiation Modification of Polysaccharides and Their Composites/Nanocomposites. Appl. Ioniz. Radiat. Mater. Process. 2017, 2, 327–354. [Google Scholar]
- Abad, L.V.; Al-Assaf, S.; Coqueret, X.; Duarte, C.; Kume, T.; Lacroix, M.; Zaman, K.; Sáfrány, Á.; Sen, M.; Tahtat, D.; et al. The Radiation Chemistry of Polysaccharides; International Atomic Energy Agency: Vienna, Austria, 2016. [Google Scholar]
- Chapiro, A. Radiation Chemistry of Polymers. Radiat. Res. Suppl. 1964, 4, 179. [Google Scholar] [CrossRef]
- Al-Assaf, S.; Gulrez, S.K.H.; Czechowska-Biskup, R.; Wach, R.A.; Rosiak, J.M.; Ulanski, P. Radiation Modification of Polysaccharides. In The Radiation Chemistry of Polysaccharides; Al-Assaf, S., Coqueret, X., Zaman, K., Sen, M., Ulanski, P., Eds.; International Atomic Energy Agency: Vienna, Austria, 2006; pp. 77–115. [Google Scholar]
- Lin, N.; Huang, J.; Dufresne, A. Preparation, Properties and Applications of Polysaccharide Nanocrystals in Advanced Functional Nanomaterials: A Review. Nanoscale 2012, 4, 3274. [Google Scholar] [CrossRef]
- Buxton, G.V.; Greenstock, C.L.; Helman, W.P.; Ross, A.B. Critical Review of Rate Constants for Reactions of Hydrated Electrons, Hydrogen Atoms and Hydroxyl Radicals (⋅OH/⋅O− in Aqueous Solution. J. Phys. Chem. Ref. Data 1988, 17, 513–886. [Google Scholar] [CrossRef]
- Gulrez, S.K.H.; Al-Assaf, S.; Phillips, G.O. Hydrogels: Methods of Preparation, Characterisation and Applications. In Progress in Molecular and Environmental Bioengineering-From Analysis and Modeling to Technology Applications; InTech: London, UK, 2011. [Google Scholar] [CrossRef]
- Al-Assaf, S.; Phillips, G.O.; Williams, P.A.; du Plessis, T.A. Application of Ionizing Radiations to Produce New Polysaccharides and Proteins with Enhanced Functionality. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms. 2007, 265, 37–43. [Google Scholar] [CrossRef]
- Augsten, C.; Knolle, W.; Mäder, K. Characterizing the Influence of Electron Irradiation on Scleroglucan. Carbohydr. Polym. 2008, 72, 707–718. [Google Scholar] [CrossRef]
- Ashfaq, A.; Clochard, M.C.; Coqueret, X.; Dispenza, C.; Driscoll, M.S.; Ulański, P.; Al-Sheikhly, M. Polymerization Reactions and Modifications of Polymers by Ionizing Radiation. Polymers 2020, 12, 2877. [Google Scholar] [CrossRef]
- Tissot, C.; Grdanovska, S.; Barkatt, A.; Silverman, J.; Al-Sheikhly, M. On the Mechanisms of the Radiation-Induced Degradation of Cellulosic Substances. Radiat. Phys. Chem. 2013, 84, 185–190. [Google Scholar] [CrossRef]
- Madrid, J.F.; Abad, L.V. Modification of Microcrystalline Cellulose by Gamma Radiation-Induced Grafting. Radiat. Phys. Chem. 2015, 115, 143–147. [Google Scholar] [CrossRef]
- Laitinen, O.; Suopajärvi, T.; Liimatainen, H. Enhancing Packaging Board Properties Using Micro- and Nanofibers Prepared from Recycled Board. Cellulose 2020, 27, 7215–7225. [Google Scholar] [CrossRef]
- Martínez-Barrera, G.; Martínez-López, M.; González-Rivas, N.; del Coz-Diaz, J.J.; Ávila-Córdoba, L.; dos Reis, J.M.L.; Gencel, O. Recycled Cellulose from Tetra Pak Packaging as Reinforcement of Polyester Based Composites. Constr. Build. Mater. 2017, 157, 1018–1023. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Processing and Characterization of Natural Cellulose Fibers/Thermoset Polymer Composites. Carbohydr. Polym. 2014, 109, 102–117. [Google Scholar] [CrossRef]
- Henniges, U.; Okubayashi, S.; Rosenau, T.; Potthast, A. Irradiation of Cellulosic Pulps: Understanding Its Impact on Cellulose Oxidation. Biomacromolecules 2012, 13, 4171–4178. [Google Scholar] [CrossRef]
- Schnabel, T.; Huber, H.; Grünewald, T.A.; Petutschnigg, A. Changes in Mechanical and Chemical Wood Properties by Electron Beam Irradiation. Appl. Surf. Sci. 2015, 332, 704–709. [Google Scholar] [CrossRef]
- Sarosi, O.P.; Bischof, R.H.; Potthast, A. Tailoring Pulp Cellulose with Electron Beam Irradiation: Effects of Lignin and Hemicellulose. ACS Sustain. Chem. Eng. 2020, 8, 7235–7243. [Google Scholar] [CrossRef]
- Ma, X.; Zheng, X.; Zhang, M.; Yang, X.; Chen, L.; Huang, L.; Cao, S. Electron Beam Irradiation of Bamboo Chips: Degradation of Cellulose and Hemicelluloses. Cellulose 2014, 21, 3865–3870. [Google Scholar] [CrossRef]
- Ma, Y.; Hummel, M.; Kontro, I.; Sixta, H. High Performance Man-Made Cellulosic Fibres from Recycled Newsprint. Green Chem. 2018, 20, 160–169. [Google Scholar] [CrossRef]
- Nemţanu, M.R.; Minea, R.; Mitru, E. Electron Beam Influence on Microcrystalline Cellulose. In AIP Conference Proceedings; AIP: Long Island, NY, USA, 2007. [Google Scholar] [CrossRef]
- Kumakura, M.; Kaetsu, I. Effect of Radiation Pretreatment of Bagasse on Enzymatic and Acid Hydrolysis. Biomass 1983, 3, 199–208. [Google Scholar] [CrossRef]
- Xin, L.Z.; Kumakura, M. Effect of Radiation Pretreatment on Enzymatic Hydrolysis of Rice Straw with Low Concentrations of Alkali Solution. Bioresour. Technol. 1993, 43, 13–17. [Google Scholar] [CrossRef]
- Yang, S.J.; Yoo, H.Y.; Choi, H.S.; Lee, J.H.; Park, C.; Kim, S.W. Enhancement of Enzymatic Digestibility of Miscanthus by Electron Beam Irradiation and Chemical Combined Treatments for Bioethanol Production. Chem. Eng. J. 2015, 275, 227–234. [Google Scholar] [CrossRef]
- Sung, Y.J.; Shin, S.-J. Compositional Changes in Industrial Hemp Biomass (Cannabis sativa L.) Induced by Electron Beam Irradiation Pretreatment. Biomass Bioenergy 2011, 35, 3267–3270. [Google Scholar] [CrossRef]
- Foston, M. Advances in Solid-State NMR of Cellulose. Curr. Opin. Biotechnol. 2014, 27, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sanz, M.; Gidley, M.J.; Gilbert, E.P. Application of X-ray and Neutron Small Angle Scattering Techniques to Study the Hierarchical Structure of Plant Cell Walls: A Review. Carbohydr. Polym. 2015, 125, 120–134. [Google Scholar] [CrossRef] [PubMed]
- Hon, D.N.-S. Cellulose: A Random Walk along Its Historical Path. Cellulose 1994, 1, 1–25. [Google Scholar] [CrossRef]
- Zhao, H.; Kwak, J.; Conradzhang, Z.; Brown, H.; Arey, B.; Holladay, J. Studying Cellulose Fiber Structure by SEM, XRD, NMR and Acid Hydrolysis. Carbohydr. Polym. 2007, 68, 235–241. [Google Scholar] [CrossRef]
- Eichhorn, S.J.; Dufresne, A.; Aranguren, M.; Marcovich, N.E.; Capadona, J.R.; Rowan, S.J.; Weder, C.; Thielemans, W.; Roman, M.; Renneckar, S.; et al. Review: Current International Research into Cellulose Nanofibres and Nanocomposites. J. Mater. Sci. 2010, 45, 1–33. [Google Scholar] [CrossRef]
- Trache, D.; Hussin, M.H.; Haafiz, M.K.M.; Thakur, V.K. Recent Progress in Cellulose Nanocrystals: Sources and Production. Nanoscale 2017, 9, 1763–1786. [Google Scholar] [CrossRef]
- Rongpipi, S.; Ye, D.; Gomez, E.D.; Gomez, E.W. Progress and Opportunities in the Characterization of Cellulose—An Important Regulator of Cell Wall Growth and Mechanics. Front. Plant Sci. 2019, 9, 1894. [Google Scholar] [CrossRef]
- Toumpanaki, E.; Shah, D.U.; Eichhorn, S.J. Beyond What Meets the Eye: Imaging and Imagining Wood Mechanical–Structural Properties. Adv. Mater. 2021, 33, 2001613. [Google Scholar] [CrossRef]
- Harris, D.M.; Corbin, K.; Wang, T.; Gutierrez, R.; Bertolo, A.L.; Petti, C.; Smilgies, D.-M.; Estevez, J.M.; Bonetta, D.; Urbanowicz, B.R.; et al. Cellulose Microfibril Crystallinity Is Reduced by Mutating C-Terminal Transmembrane Region Residues CESA1 A903V and CESA3 T942I of Cellulose Synthase. Proc. Natl. Acad. Sci. USA 2012, 109, 4098–4103. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Mishra, L.; Carr, P.; Pilling, M.; Gardner, P.; Mansfield, S.D.; Turner, S. Exploiting CELLULOSE SYNTHASE (CESA) Class Specificity to Probe Cellulose Microfibril Biosynthesis. Plant Physiol. 2018, 177, 151–167. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, C.M.; Kafle, K. Characterization of Crystalline Cellulose in Biomass: Basic Principles, Applications, and Limitations of XRD, NMR, IR, Raman, and SFG. Korean J. Chem. Eng. 2013, 30, 2127–2141. [Google Scholar] [CrossRef]
- Pönni, R.; Vuorinen, T.; Kontturi, E. Proposed Nano-Scale Coalescence of Cellulose in Chemical Pulp Fibers during Technical Treatments. Bioresources 2012, 7, 6077–6108. [Google Scholar] [CrossRef]
- Driscoll, M.; Stipanovic, A.; Winter, W.; Cheng, K.; Manning, M.; Spiese, J.; Galloway, R.A.; Cleland, M.R. Electron Beam Irradiation of Cellulose. Radiat. Phys. Chem. 2009, 78, 539–542. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, J.; Wu, X.; Wang, K.; Su, X.; Chen, L.; Zhou, H.; Xiong, X. Insights into the Effects of γ-Irradiation on the Microstructure, Thermal Stability and Irradiation-Derived Degradation Components of Microcrystalline Cellulose (MCC). RSC Adv. 2015, 5, 34353–34363. [Google Scholar] [CrossRef]
- Eaton, G.R.; Eaton, S.S.; Barr, D.P.; Weber, R.T. Quantitative EPR; Springer: Vienna, Austria, 2010. [Google Scholar] [CrossRef]
- Jeschke, G. Physical Chemistry IV Part 2: Electron Paramagnetic Resonance; Gunnar Jeschke: Zurich, Switzerland, 2016. [Google Scholar]
- Rånby, B.; Rabek, J.F. ESR Spectroscopy in Polymer Research; Polymers Properties and Applications; Springer: Berlin/Heidelberg, Germany, 1977; Volume 1. [Google Scholar] [CrossRef]
- Baugh, P.J.; Hinojosa, O.; Arthur, J.C. ESR Study of Post-Irradiation Reactions of Cellulose and Acrylonitrile. J. Appl. Polym. Sci. 1967, 11, 1139–1153. [Google Scholar] [CrossRef]
- Reine, A.H.; Hinojosa, O.; Arthur, J.C. ESR Studies of Free Radicals in Photo-Initiated Graft Polymerization Reactions with Cotton Cellulose. J. Appl. Polym. Sci. 1973, 17, 3337–3343. [Google Scholar] [CrossRef]
- Arthur, J.C.; Hinojosa, O.; Bains, M.S. ESR Study of Reactions of Cellulose with ·OH Generated by Fe+2/H2O2. J. Appl. Polym. Sci. 1968, 12, 1411–1421. [Google Scholar] [CrossRef]
- Nakamura, Y.; Hinojosa, O.; Arthur, J.C. Cellulose Graft Copolymers. II. Graft Copolymerization of Ethyl Acrylate with γ-Irradiated Cellulose from Acetone–Water Systems. J. Appl. Polym. Sci. 1970, 14, 789–797. [Google Scholar] [CrossRef]
- Hinojosa, O.; Nakamura, Y.; Arthur, J.C. ESR Study of Interactions of γ-Irradiated Cellulose I and Cellulose II with Ammonia, Water, and Sodium Hydroxide Solutions. J. Polym. Sci. Part C Polym. Symp. 1972, 37, 27–46. [Google Scholar] [CrossRef]
- Dubey, K.A.; Pujari, P.K.; Ramnani, S.P.; Kadam, R.M.; Sabharwal, S. Microstructural Studies of Electron Beam-Irradiated Cellulose Pulp. Radiat. Phys. Chem. 2004, 69, 395–400. [Google Scholar] [CrossRef]
- Takács, E.; Wojnárovits, L.; Borsa, J.; Papp, J.; Hargittai, P.; Korecz, L. Modification of Cotton-Cellulose by Preirradiation Grafting. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2005, 236, 259–265. [Google Scholar] [CrossRef]
- Wach, R.A.; Mitomo, H.; Nagasawa, N.; Yoshii, F. Radiation Crosslinking of Carboxymethylcellulose of Various Degree of Substitution at High Concentration in Aqueous Solutions of Natural PH. Radiat. Phys. Chem. 2003, 68, 771–779. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Singh, B.C. Radiation-Induced and Photoinduced Grafting onto Cellulose and Cellulosic Materials. Polym.-Plast. Technol. Eng. 1988, 27, 435–466. [Google Scholar] [CrossRef]
- Margolin, A.L.; Menendes Tomassevich, M.A.; Shlyapintokh, V.Y. Kinetics of the Accumulation and Decay of Radicals on Photo-Oxidation of Cellulose. Polym. Sci. USSR 1990, 32, 194–201. [Google Scholar] [CrossRef]
- Stefanovic, B.; Pirker, K.F.; Rosenau, T.; Potthast, A. Effects of Tribochemical Treatments on the Integrity of Cellulose. Carbohydr. Polym. 2014, 111, 688–699. [Google Scholar] [CrossRef]
- Yordanov, N.D.; Gancheva, V. A New Approach for Extension of the Identification Period of Irradiated Cellulose-Containing Foodstuffs by EPR Spectroscopy. Appl. Radiat. Isot. 2000, 52, 195–198. [Google Scholar] [CrossRef]
- Raffi, J.; Yordanov, N.D.; Chabane, S.; Douifi, L.; Gancheva, V.; Ivanova, S. Identification of Irradiation Treatment of Aromatic Herbs, Spices and Fruits by Electron Paramagnetic Resonance and Thermoluminescence. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2000, 56, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Aleksieva, K.; Georgieva, L.; Tzvetkova, E.; Yordanov, N.D. EPR Study on Tomatoes before and after Gamma-Irradiation. Radiat. Phys. Chem. 2009, 78, 823–825. [Google Scholar] [CrossRef]
- Ershov, B.G. Radiation-Chemical Degradation of Cellulose and Other Polysaccharides. Russ. Chem. Rev. 1998, 67, 315–334. [Google Scholar] [CrossRef]
- Ershov, B.G.; Isakova, O.V. Formation and Thermal Transformations of Free Radicals in Gamma Radiation of Cellulose. Bull. Acad. Sci. USSR Div. Chem. Sci. 1984, 33, 1171–1175. [Google Scholar] [CrossRef]
- Iller, E.; Kukiełka, A.; Stupińska, H.; Mikołajczyk, W. Electron-Beam Stimulation of the Reactivity of Cellulose Pulps for Production of Derivatives. Radiat. Phys. Chem. 2002, 63, 253–257. [Google Scholar] [CrossRef]
- Mondal, R.K.; Dubey, K.A.; Kumar, J.; Jagannath; Bhardwaj, Y.K.; Melo, J.S.; Varshney, L. Carbon Nanotube Functionalization and Radiation Induced Enhancements in the Sensitivity of Standalone Chemiresistors for Sensing Volatile Organic Compounds. ACS Appl. Nano Mater. 2018, 1, 5470–5482. [Google Scholar] [CrossRef]
- Polovka, M.; Brezová, V.; Staško, A.; Mazúr, M.; Suhaj, M.; Šimko, P. EPR Investigations of Gamma-Irradiated Ground Black Pepper. Radiat. Phys. Chem. 2006, 75, 309–321. [Google Scholar] [CrossRef]
- Taibi, J.; Rouif, S.; Clément, J.L.; Ameduri, B.; Sonnier, R.; Otazaghine, B. Flame Retardancy of Flax Fibers by Pre-Irradiation Grafting of a Phosphonate Monomer. Ind. Crops Prod. 2022, 176, 114334. [Google Scholar] [CrossRef]
- Bayram, G.; Delincée, H. Identification of Irradiated Turkish Foodstuffs Combining Various Physical Detection Methods. Food Control 2004, 15, 81–91. [Google Scholar] [CrossRef]
- Komolprasert, V.; Morehouse, K.M. (Eds.) Irradiation of Food and Packaging; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2004; Volume 875. [Google Scholar] [CrossRef]
- Tonyali, B.; Yucel, U. Novel Deconvolution Approach for the Analysis of Complex Electron Paramagnetic Resonance Spectra of Irradiated Sweet Potatoes. ACS Food Sci. Technol. 2022, 2, 1525–1534. [Google Scholar] [CrossRef]
- Khan, A.A.; Shahid, M.K. Identification of Radiation Processing of Different Plant Foods of Pakistan Origin Using the Rapid Technique of Electron Spin Resonance (ESR) Spectrometry. Radiat. Phys. Chem. 2023, 204, 110667. [Google Scholar] [CrossRef]
- Tonyali, B.; Sommers, C.; Ceric, O.; Smith, J.S.; Yucel, U. An Analysis of Cellulose- and Dextrose-based Radicals in Sweet Potatoes as Irradiation Markers. J. Food Sci. 2020, 85, 2745–2753. [Google Scholar] [CrossRef] [PubMed]
- Aleksieva, K.I.; Yordanov, N.D. Various Approaches in EPR Identification of Gamma-Irradiated Plant Foodstuffs: A Review. Food Res. Int. 2018, 105, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Sudprasert, W.; Insuan, P.; Khamkhrongmee, S. EPR Study of Free Radicals in Cotton Fiber for Its Potential Use as a Fortuitous Dosimeter in Radiological Accidents. J. Phys. Conf. Ser. 2015, 611, 012012. [Google Scholar] [CrossRef]
- Hassoun, A.; Carpena, M.; Prieto, M.A.; Simal-Gandara, J.; Özogul, F.; Özogul, Y.; Çoban, Ö.E.; Guðjónsdóttir, M.; Barba, F.J.; Marti-Quijal, F.J.; et al. Use of Spectroscopic Techniques to Monitor Changes in Food Quality during Application of Natural Preservatives: A Review. Antioxidants 2020, 9, 882. [Google Scholar] [CrossRef] [PubMed]
- Aleksieva, K.; Yordanov, N.D. EPR Investigation of Some Gamma-Irradiated Excipients. Radiat. Eff. Defects Solids 2012, 167, 685–689. [Google Scholar] [CrossRef]
- Damian, G.; Teodor, A.; Popescu, I.A.; Creanga, E.D. Electron Paramagnetic Resonance Investigations of Ultraviolet Irradiated Prednisone. In Proceedings of the 2017 E-Health and Bioengineering Conference (EHB), Sinaia, Romania, 22–24 June 2017; IEEE: Piscataway, NJ, USA, 2017; pp. 737–740. [Google Scholar] [CrossRef]
- AbouZid, S.; Ahmed, H. Brief Review on Applications of Continuous-Wave Electron Paramagnetic Resonance Spectroscopy in Natural Product Free Radical Research. Stud. Nat. Prod. Chem. 2020, 66, 355–369. [Google Scholar] [CrossRef]
- Wang, K.; Xiong, X.; Chen, J.; Chen, L.; Su, X.; Liu, Y. Comparison of Gamma Irradiation and Steam Explosion Pretreatment for Ethanol Production from Agricultural Residues. Biomass Bioenergy 2012, 46, 301–308. [Google Scholar] [CrossRef]
- Dyer, J.; Phifer, L.H. Application of Gpc to Studies of the Viscose Process. III. Effect of Various Treatments of Rayon Print Cloth on the Molecular Weight Distribution of Cellulose. J. Polym. Sci. Part C Polym. Symp. 1971, 36, 103–119. [Google Scholar] [CrossRef]
- Chen, Q.-Y.; Ma, X.-J.; Li, J.-G.; Miao, Q.-X.; Huang, L.-L. Effect of the Utilization of Electron Beam Irradiation on the Reactivity of Bamboo Dissolving Pulp. BioResources 2017, 12, 6251–6261. [Google Scholar] [CrossRef]
- Bouchard, J.; Méthot, M.; Jordan, B. The Effects of Ionizing Radiation on the Cellulose of Woodfree Paper. Cellulose 2006, 13, 601–610. [Google Scholar] [CrossRef]
- Kumar, S.; Gupta, R.; Lee, Y.Y.; Gupta, R.B. Cellulose Pretreatment in Subcritical Water: Effect of Temperature on Molecular Structure and Enzymatic Reactivity. Bioresour. Technol. 2010, 101, 1337–1347. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Xu, L.; Ge, M.; Zhai, M. Radiation Degradation of Microcrystalline Cellulose in Solid Status. J. Appl. Polym. Sci. 2013, 127, 1630–1636. [Google Scholar] [CrossRef]
- Su, X.-J.; Zhang, C.-Y.; Li, W.-J.; Wang, F.; Wang, K.-Q.; Liu, Y.; Li, Q.-M. Radiation-Induced Structural Changes of Miscanthus Biomass. Appl. Sci. 2020, 10, 1130. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, Y.; Wei, M.; Shao, H.; Hu, X. Influence of γ-Ray Radiation on the Structure and Properties of Paper Grade Bamboo Pulp. Carbohydr. Polym. 2010, 81, 114–119. [Google Scholar] [CrossRef]
- French, A.D. Idealized Powder Diffraction Patterns for Cellulose Polymorphs. Cellulose 2014, 21, 885–896. [Google Scholar] [CrossRef]
- El Hariri El Nokab, M.; Habib, M.H.; Alassmy, Y.A.; Abduljawad, M.M.; Alshamrani, K.M.; Sebakhy, K.O. Solid State NMR a Powerful Technique for Investigating Sustainable/Renewable Cellulose-Based Materials. Polymers 2022, 14, 1049. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Hong, M. Solid-State NMR Investigations of Cellulose Structure and Interactions with Matrix Polysaccharides in Plant Primary Cell Walls. J. Exp. Bot. 2016, 67, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Munson, C.R.; Gao, Y.; Mortimer, J.C.; Murray, D.T. Solid-State Nuclear Magnetic Resonance as a Tool to Probe the Impact of Mechanical Preprocessing on the Structure and Arrangement of Plant Cell Wall Polymers. Front. Plant Sci. 2022, 12, 766506. [Google Scholar] [CrossRef]
- Werner, M.A. Applications of Solid State Nuclear Magnetic Resonance Spectroscopy for Non-Destructive Analysis of Plant Cell Walls and Engineered Soil Surrogates. Master’s Thesis, The Pennsylvania State University, State College, PA, USA, 2019. [Google Scholar]
- Brouwer, D.H.; Mikolajewski, J.G. Resolving the Discrepancies in Reported 13C Solid State NMR Chemical Shifts for Native Celluloses. Cellulose 2023, 30, 4827–4839. [Google Scholar] [CrossRef]
- Ren, W.; Guo, F.; Zhu, J.; Cao, M.; Wang, H.; Yu, Y. A Comparative Study on the Crystalline Structure of Cellulose Isolated from Bamboo Fibers and Parenchyma Cells. Cellulose 2021, 28, 5993–6005. [Google Scholar] [CrossRef]
- Foston, M.; Katahira, R.; Gjersing, E.; Davis, M.F.; Ragauskas, A.J. Solid-State Selective 13 C Excitation and Spin Diffusion NMR To Resolve Spatial Dimensions in Plant Cell Walls. J. Agric. Food Chem. 2012, 60, 1419–1427. [Google Scholar] [CrossRef]
- Masuda, K.; Adachi, M.; Hirai, A.; Yamamoto, H.; Kaji, H.; Horii, F. Solid-State 13C and 1H Spin Diffusion NMR Analyses of the Microfibril Structure for Bacterial Cellulose. Solid State Nucl. Magn. Reson. 2003, 23, 198–212. [Google Scholar] [CrossRef] [PubMed]
- Kirui, A.; Ling, Z.; Kang, X.; Dickwella Widanage, M.C.; Mentink-Vigier, F.; French, A.D.; Wang, T. Atomic Resolution of Cotton Cellulose Structure Enabled by Dynamic Nuclear Polarization Solid-State NMR. Cellulose 2019, 26, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Li, W.; Plog, A.; Xu, Y.; Buntkowsky, G.; Gutmann, T.; Zhang, K. Multi-Responsive Cellulose Nanocrystal–Rhodamine Conjugates: An Advanced Structure Study by Solid-State Dynamic Nuclear Polarization (DNP) NMR. Phys. Chem. Chem. Phys. 2014, 16, 26322–26329. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Deligey, F.; Chandra Shekar, S.; Mentink-Vigier, F.; Wang, T. Current Limitations of Solid-State NMR in Carbohydrate and Cell Wall Research. J. Magn. Reson. 2022, 341, 107263. [Google Scholar] [CrossRef]
- Aguilera-Sáez, L.M.; Arrabal-Campos, F.M.; Callejón-Ferre, Á.J.; Suárez Medina, M.D.; Fernández, I. Use of Multivariate NMR Analysis in the Content Prediction of Hemicellulose, Cellulose and Lignin in Greenhouse Crop Residues. Phytochemistry 2019, 158, 110–119. [Google Scholar] [CrossRef]
- Carnahan, S.L.; Chen, Y.; Wishart, J.F.; Lubach, J.W.; Rossini, A.J. Magic Angle Spinning Dynamic Nuclear Polarization Solid-State NMR Spectroscopy of γ-Irradiated Molecular Organic Solids. Solid State Nucl. Magn. Reson. 2022, 119, 101785. [Google Scholar] [CrossRef]
- Ciolacu, D.; Ciolacu, F.; Popa, V. Amorphous Cellulose-Structure and Characterization. Cellul. Chem. Technol. 2011, 45, 13–21. [Google Scholar]
- Sun, F.; Jiang, Z.; Sun, Q.; Lu, F. Changes in Chemical Composition and Microstructure of Bamboo after Gamma Ray Irradiation. Bioresources 2014, 9, 5794–5800. [Google Scholar] [CrossRef]
- Liu, Y. Recent Progress in Fourier Transform Infrared (FTIR) Spectroscopy Study of Compositional, Structural and Physical Attributes of Developmental Cotton Fibers. Materials 2013, 6, 299–313. [Google Scholar] [CrossRef]
- Oh, S.Y.; Yoo, D.I.; Shin, Y.; Kim, H.C.; Kim, H.Y.; Chung, Y.S.; Park, W.H.; Youk, J.H. Crystalline Structure Analysis of Cellulose Treated with Sodium Hydroxide and Carbon Dioxide by Means of X-ray Diffraction and FTIR Spectroscopy. Carbohydr. Res. 2005, 340, 2376–2391. [Google Scholar] [CrossRef]
- Fujisawa, S.; Okita, Y.; Fukuzumi, H.; Saito, T.; Isogai, A. Preparation and Characterization of TEMPO-Oxidized Cellulose Nanofibril Films with Free Carboxyl Groups. Carbohydr. Polym. 2011, 84, 579–583. [Google Scholar] [CrossRef]
- Schwanninger, M.; Rodrigues, J.C.; Pereira, H.; Hinterstoisser, B. Effects of Short-Time Vibratory Ball Milling on the Shape of FT-IR Spectra of Wood and Cellulose. Vib. Spectrosc. 2004, 36, 23–40. [Google Scholar] [CrossRef]
- Schenzel, K.; Fischer, S.; Brendler, E. New Method for Determining the Degree of Cellulose I Crystallinity by Means of FT Raman Spectroscopy. Cellulose 2005, 12, 223–231. [Google Scholar] [CrossRef]
- Agarwal, U.P.; Reiner, R.S.; Ralph, S.A. Cellulose I Crystallinity Determination Using FT-Raman Spectroscopy: Univariate and Multivariate Methods. Cellulose 2010, 17, 721–733. [Google Scholar] [CrossRef]
- Agarwal, U.P.; Ralph, S.A.; Reiner, R.S.; Baez, C. New Cellulose Crystallinity Estimation Method That Differentiates between Organized and Crystalline Phases. Carbohydr. Polym. 2018, 190, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, U.P. Analysis of Cellulose and Lignocellulose Materials by Raman Spectroscopy: A Review of the Current Status. Molecules 2019, 24, 1659. [Google Scholar] [CrossRef] [PubMed]
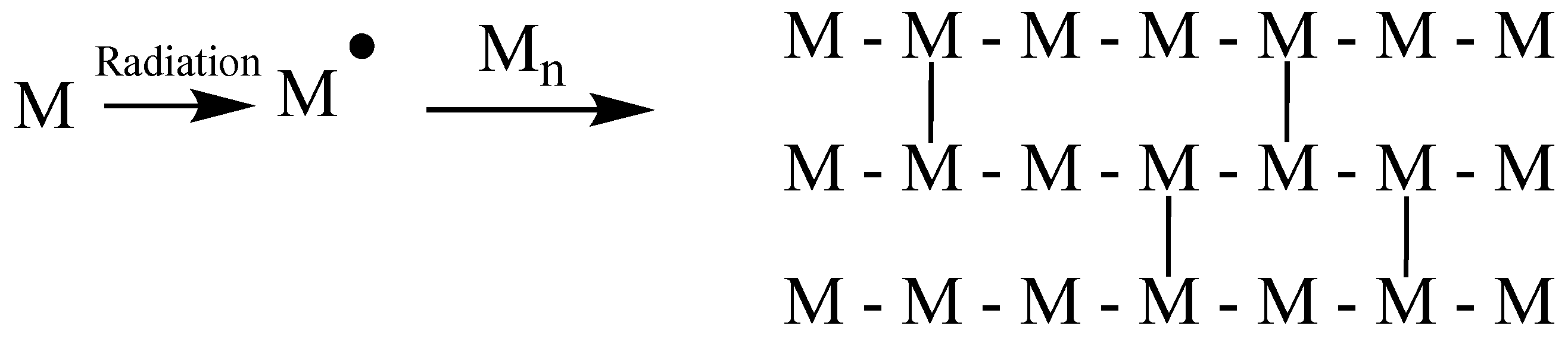



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
List, R.; Gonzalez-Lopez, L.; Ashfaq, A.; Zaouak, A.; Driscoll, M.; Al-Sheikhly, M. On the Mechanism of the Ionizing Radiation-Induced Degradation and Recycling of Cellulose. Polymers 2023, 15, 4483. https://doi.org/10.3390/polym15234483
List R, Gonzalez-Lopez L, Ashfaq A, Zaouak A, Driscoll M, Al-Sheikhly M. On the Mechanism of the Ionizing Radiation-Induced Degradation and Recycling of Cellulose. Polymers. 2023; 15(23):4483. https://doi.org/10.3390/polym15234483
Chicago/Turabian StyleList, Richard, Lorelis Gonzalez-Lopez, Aiysha Ashfaq, Amira Zaouak, Mark Driscoll, and Mohamad Al-Sheikhly. 2023. "On the Mechanism of the Ionizing Radiation-Induced Degradation and Recycling of Cellulose" Polymers 15, no. 23: 4483. https://doi.org/10.3390/polym15234483
APA StyleList, R., Gonzalez-Lopez, L., Ashfaq, A., Zaouak, A., Driscoll, M., & Al-Sheikhly, M. (2023). On the Mechanism of the Ionizing Radiation-Induced Degradation and Recycling of Cellulose. Polymers, 15(23), 4483. https://doi.org/10.3390/polym15234483






