Effect of the Addition of Different Natural Waxes on the Mechanical and Rheological Behavior of PLA—A Comparative Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Mixtures
2.3. Film Preparation
2.4. Characterization
3. Discussion and Results
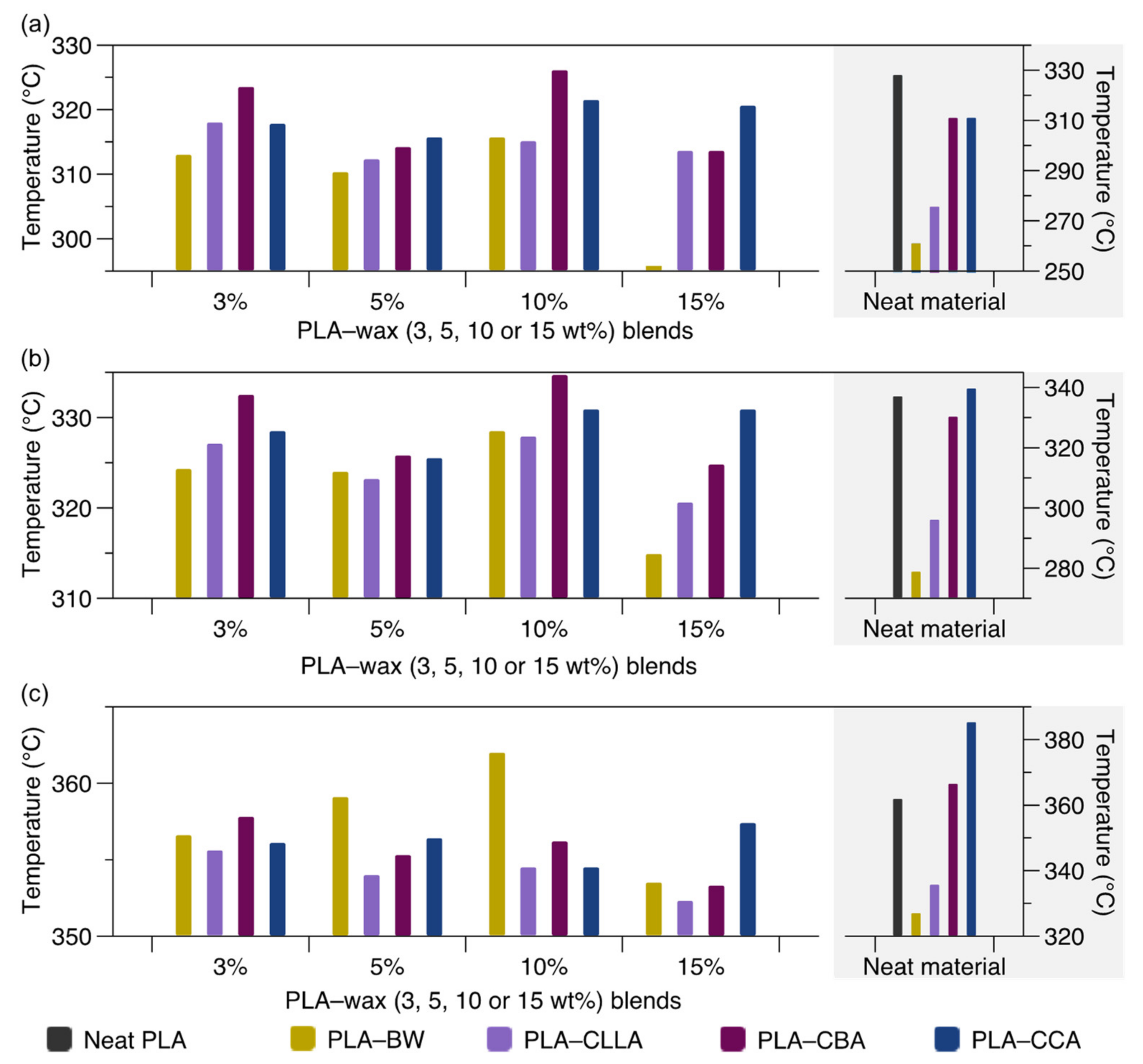
3.1. Thermogravimetric Analysis
3.2. Differential Scanning Calorimetry
3.3. Rheological Properties
3.4. Viscoelastic Properties
3.5. Dynamic Mechanical Analysis (Tensile Tests)
3.6. Contact Angle Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gálvez, J.; Correa Aguirre, J.P.; Hidalgo Salazar, M.A.; Mondragón, B.V.; Wagner, E.; Caicedo, C. Effect of Extrusion Screw Speed and Plasticizer Proportions on the Rheological, Thermal, Mechanical, Morphological and Superficial Properties of PLA. Polymers 2020, 12, 2111. [Google Scholar] [CrossRef]
- Murariu, M.; Da Silva Ferreira, A.; Alexandre, M.; Dubois, P. Polylactide (PLA) Designed with Desired End-Use Properties: 1. PLA Compositions with Low Molecular Weight Ester-like Plasticizers and Related Performances. Polym. Adv. Technol. 2008, 19, 636–646. [Google Scholar] [CrossRef]
- Hassouna, F.; Raquez, J.M.; Addiego, F.; Toniazzo, V.; Dubois, P.; Ruch, D. New Development on Plasticized Poly(Lactide): Chemical Grafting of Citrate on PLA by Reactive Extrusion. Eur. Polym. J. 2012, 48, 404–415. [Google Scholar] [CrossRef]
- Tripathi, N.; Misra, M.; Mohanty, A.K. Durable Polylactic Acid (PLA)-Based Sustainable Engineered Blends and Biocomposites: Recent Developments, Challenges, and Opportunities. ACS Eng. Au 2021, 1, 7–38. [Google Scholar] [CrossRef]
- Ljungberg, N.; Wesslén, B. Preparation and Properties of Plasticized Poly(Lactic Acid) Films. Biomacromolecules 2005, 6, 1789–1796. [Google Scholar] [CrossRef] [PubMed]
- Baiardo, M.; Frisoni, G.; Scandola, M.; Rimelen, M.; Lips, D.; Ruffieux, K.; Wintermantel, E. Thermal and Mechanical Properties of Plasticized Poly(L-Lactic Acid). J. Appl. Polym. Sci. 2003, 90, 1731–1738. [Google Scholar] [CrossRef]
- Martin, O.; Avérous, L. Poly(Lactic Acid): Plasticization and Properties of Biodegradable Multiphase Systems. Polymer 2001, 42, 6209–6219. [Google Scholar] [CrossRef]
- Shellhammer, T.H.; Rumsey, T.R.; Krochta, J.M. Viscoelastic Properties of Edible Lipids. J. Food Eng. 1997, 33, 305–320. [Google Scholar] [CrossRef]
- Talens, P.; Krochta, J.M. Plasticizing Effects of Beeswax and Carnauba Wax on Tensile and Water Vapor Permeability Properties of Whey Protein Films. J. Food Sci. 2005, 70, E239–E243. [Google Scholar] [CrossRef]
- Zhang, Y.; Adams, M.J.; Zhang, Z.; Vidoni, O.; Leuenberger, B.H.; Achkar, J. Plasticisation of Carnauba Wax with Generally Recognised as Safe (GRAS) Additives. Polymer 2016, 86, 208–219. [Google Scholar] [CrossRef]
- Elvers, B.; Ha, S. Ullmann’s Encyclopedia of Industrial Chemistry, 5th ed.; Elvers, B., Hawkin, S., Eds.; Verlag Chemie: Weinheim, Germany, 1996; Volume 1. [Google Scholar]
- Asperger, A.; Engewald, W.; Fabian, G. Analytical Characterization of Natural Waxes Employing Pyrolysis-Gas Chromatography-Mass Spectrometry. J. Anal. Appl. Pyrolysis 1999, 50, 103–115. [Google Scholar] [CrossRef]
- Ribas Garriga, M. Evaluation of Natural Wax for Green Packaging Applications; Lulea University of Technology: Lulea, Sweden, 2019; Volume 1. [Google Scholar]
- Jana, S.; Martini, S. Phase Behavior of Binary Blends of Four Different Waxes. JAOCS J. Am. Oil Chem. Soc. 2016, 93, 543–554. [Google Scholar] [CrossRef]
- Endlein, E.; Peleikis, K.-H. Natural Waxes Properties, Compositions and Applications. Int. J. Appl. Sci. 2011, 4, 1–8. [Google Scholar]
- Reis, M.O.; Olivato, J.B.; Bilck, A.P.; Zanela, J.; Grossmann, M.V.E.; Yamashita, F. Biodegradable Trays of Thermoplastic Starch/Poly (Lactic Acid) Coated with Beeswax. Ind. Crops Prod. 2018, 112, 481–487. [Google Scholar] [CrossRef]
- Cuq, B.; Gontard, N.; Guilbert, S. Edible Films and Coatings as Active Layers. In Active Food Packaging; Rooney, M.L., Ed.; Springer: Boston, MA, USA, 1995; pp. 111–142. [Google Scholar]
- Fabra, M.J.; Talens, P.; Chiralt, A. Tensile Properties and Water Vapor Permeability of Sodium Caseinate Films Containing Oleic Acid-Beeswax Mixtures. J. Food Eng. 2008, 85, 393–400. [Google Scholar] [CrossRef]
- Diyana, Z.N.; Jumaidin, R.; Selamat, M.Z.; Suan, M.S.M. Thermoplastic Starch/Beeswax Blend: Characterization on Thermal Mechanical and Moisture Absorption Properties. Int. J. Biol. Macromol. 2021, 190, 224–232. [Google Scholar] [CrossRef]
- Zou, J.; Lu, M.; Chen, S.; Cai, C.; Yao, Z.; Cui, W.; Fan, C.; Liu, S. Beeswax-Inspired Superhydrophobic Electrospun Membranes for Peritendinous Anti-Adhesion. Mater. Sci. Eng. C 2020, 116, 111166–111175. [Google Scholar] [CrossRef]
- De Oliveira, T.A.; De Oliveira Mota, I.; De Carvalho, L.H.; Barbosa, R.; Alves, T.S. Influence of Carnauba Wax on Films of Poly (Butylene Adipate Co-Terephthalate) and Sugarcane Residue for Application in Soil Cover (Mulching). Mater. Res. 2019, 22, 1–10. [Google Scholar] [CrossRef]
- Kulinski, Z.; Piorkowska, E. Crystallization, Structure and Properties of Plasticized Poly(L-Lactide). Polymer 2005, 46, 10290–10300. [Google Scholar] [CrossRef]
- Li, H.; Huneault, M.A. Effect of Nucleation and Plasticization on the Crystallization of Poly(Lactic Acid). Polymer 2007, 48, 6855–6866. [Google Scholar] [CrossRef]
- Janjarasskul, T.; Rauch, D.J.; McCarthy, K.L.; Krochta, J.M. Barrier and Tensile Properties of Whey Protein-Candelilla Wax Film/Sheet. LWT 2014, 56, 377–382. [Google Scholar] [CrossRef]
- Bennet, H. Industrial Waxes; Chemical Publishing Company: New York, NY, USA, 1975. [Google Scholar]
- Molina, P.; Silva, M.; Valle, L. Estudio Preliminar de Compatibilizantes En Mezclas. Preliminary Study of Compatibilizers in PLA-Achira (Canna Edulis) Starch Blends. Rev. Cumbres 2017, 3, 33–40. [Google Scholar]
- Silva, M.; Encalada, K.; Valle, V. Estudio de La Cera Carnauba Como Compatibilizante En Mezclas Poli (Ácido Láctico)-Almidón de Achira (Canna Edulis). Rev. Politécnica 2017, 39, 13–18. [Google Scholar]
- El Achaby, M.; Qaiss, A. Processing and Properties of Polyethylene Reinforced by Graphene Nanosheets and Carbon Nanotubes. Mater. Des. 2013, 44, 81–89. [Google Scholar] [CrossRef]
- Lim, J.H.; Kim, J.A.; Ko, J.A.; Park, H.J. Preparation and Characterization of Composites Based on Polylactic Acid and Beeswax with Improved Water Vapor Barrier Properties. J. Food Sci. 2015, 80, E2471–E2477. [Google Scholar] [CrossRef]
- Santos, E.F.; Oliveira, R.V.B.; Reiznautt, Q.B.; Samios, D.; Onia, S.; Nachtigall, M.B. Sunflower-Oil Biodiesel-Oligoesters/Polylactide Blends: Plasticizing Effect and Ageing. Polym. Test. 2014, 39, 23–29. [Google Scholar] [CrossRef]
- Chen, Y.J.; Yu, E.; Ellingham, T.; Chung, C.; Turng, L.S. Improving the Processibility and Mechanical Properties of Poly(Lactic Acid)/Linear Low-Density Polyethylene/Paraffin Wax Blends by Subcritical Gas-Assisted Processing. Polym. Eng. Sci. 2018, 58, 2320–2331. [Google Scholar] [CrossRef]
- Zou, Z.M.; Sun, Z.Y.; An, L.J. Effect of Fumed Silica Nanoparticles on the Morphology and Rheology of Immiscible Polymer Blends. Rheol. Acta 2014, 53, 43–53. [Google Scholar] [CrossRef]
- Nakayama, D.; Wu, F.; Mohanty, A.K.; Hirai, S.; Misra, M. Biodegradable Composites Developed from PBAT/PLA Binary Blends and Silk Powder: Compatibilization and Performance Evaluation. ACS Omega 2018, 3, 12412–12421. [Google Scholar] [CrossRef]
- Lafranche, E.; Krawczak, P. Toughness Improvement of Novel Biobased Aromatic Polyamide/SEBS/Organoclay Ternary Hybrids. J. Appl. Polym. Sci. 2020, 137, 15–24. [Google Scholar] [CrossRef]
- Kang, H.; Li, Y.; Gong, M.; Guo, Y.; Guo, Z.; Fang, Q.; Li, X. An Environmentally Sustainable Plasticizer Toughened Polylactide. RSC Adv. 2018, 8, 11643–11651. [Google Scholar] [CrossRef] [PubMed]
- Mezger, T. The Rheology Handbook, 4th ed.; Network, V., Ed.; Vincentz Network: Hanover, Germany, 2020; ISBN 978-3-86630-650-9. [Google Scholar]
- Carbonell-Verdu, A.; Garcia-Garcia, D.; Dominici, F.; Torre, L.; Sanchez-Nacher, L.; Balart, R. PLA Films with Improved Flexibility Properties by Using Maleinized Cottonseed Oil. Eur. Polym. J. 2017, 91, 248–259. [Google Scholar] [CrossRef]
- Joshi, B.L.; Graf, R.; Gindra, S.; Vilgis, T.A. Effect of Different Derivatives of Paraffin Waxes on Crystallization of Eutectic Mixture of Cocoa Butter-Coconut Oil. Curr. Res. Food Sci. 2021, 4, 784–799. [Google Scholar] [CrossRef] [PubMed]
- Ferri, J.M.; Samper, M.D.; García-Sanoguera, D.; Reig, M.J.; Fenollar, O.; Balart, R. Plasticizing Effect of Biobased Epoxidized Fatty Acid Esters on Mechanical and Thermal Properties of Poly(Lactic Acid). J. Mater. Sci. 2016, 51, 5356–5366. [Google Scholar] [CrossRef]






| Neat PLA | BW | CLLA | CBA | CCA | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T5% | T10% | Tmax | T5% | T10% | Tmax | T5% | T10% | Tmax | T5% | T10% | Tmax | T5% | T10% | Tmax | |
| Neat material | 328.1 | 337.1 | 361.9 | 261.0 | 278.9 | 327.0 | 275.6 | 296.1 | 335.7 | 311.0 | 330.3 | 366.5 | 311.0 | 339.7 | 385.3 |
| PLA–Wax % | |||||||||||||||
| 3 | – | – | – | 313.0 | 324.3 | 356.6 | 318.0 | 327.1 | 355.6 | 323.5 | 332.5 | 357.8 | 317.8 | 328.5 | 356.1 |
| 5 | – | – | – | 310.3 | 324.0 | 359.1 | 312.3 | 323.2 | 354.0 | 314.2 | 325.8 | 355.3 | 315.7 | 325.5 | 356.4 |
| 10 | – | – | – | 315.7 | 328.5 | 362.0 | 315.1 | 327.9 | 354.5 | 326.1 | 334.7 | 356.2 | 321.5 | 330.9 | 354.5 |
| 15 | – | – | – | 295.8 | 314.9 | 353.5 | 305.6 | 320.6 | 352.3 | 313.6 | 324.8 | 353.3 | 320.6 | 330.9 | 357.4 |
| Sample | Tg 1 (°C) | Tcc 2 | ΔHcc 3 | Tm 4 (°C) | ΔHm 5 | ΔHm − ΔHcc |
|---|---|---|---|---|---|---|
| BW | -- | -- | -- | 68.3 | 102.3 | -- |
| CLLA | -- | -- | -- | 77.4 | 117.4 | -- |
| CBA | -- | -- | -- | 87.5 | 161.1 | -- |
| CCA | -- | -- | -- | 42.5 | 62.1 | -- |
| neat PLA | 65.6 | 111.1 | 20.3 | 176.5 | 24.8 | 4.5 |
| BW | ||||||
| PLA–BW 3% | 65.4 | 106.0 | 21.4 | 173.0 | 31.5 | 10.1 |
| PLA–BW 5% | 64.9 | 106.8 | 23.0 | 175.0 | 32.6 | 9.6 |
| PLA–BW 10% | 64.7 | 108.4 | 18.9 | 176.5 | 28.2 | 9.3 |
| PLA–BW 15% | 65.6 | 109.2 | 22.3 | 175.0 | 28.3 | 6.0 |
| CLLA | ||||||
| PLA–CLLA 3% | 67.7 | 115.3 | 17.1 | 175.3 | 31.1 | 14.0 |
| PLA–CLLA 5% | 64.6 | 114.9 | 25.2 | 173.6 | 32.6 | 7.4 |
| PLA–CLLA 10% | 64.7 | 116.9 | 14.9 | 172.7 | 27.4 | 12.5 |
| PLA–CLLA 15% | 67.5 | 122.0 | 20.4 | 174.0 | 29.7 | 9.3 |
| CBA | ||||||
| PLA–CBA 3% | 67.0 | 112.7 | 11.4 | 175.5 | 33.8 | 22.5 |
| PLA–CBA 5% | 63.8 | 109.4 | 8.3 | 174.1 | 32.8 | 24.6 |
| PLA–CBA 10% | 68.8 | 112.9 | 17.8 | 175.9 | 31.6 | 13.8 |
| PLA–CBA 15% | 67.5 | 109.2 | 21.2 | 172.1 | 31.2 | 10.0 |
| CCA | ||||||
| PLA–CCA 3% | 69.4 | 108.7 | 28.0 | 176.4 | 47.9 | 19.9 |
| PLA–CCA 5% | 63.0 | 107.2 | 29.3 | 170.9 | 31.5 | 2.2 |
| PLA–CCA 10% | 65.1 | 110.7 | 22.3 | 173.7 | 27.4 | 5.1 |
| PLA–CCA 15% | 69.4 | 111.3 | 25.8 | 173.2 | 25.9 | 0.1 |
| |η*| (Pa∙s) | 3 wt% LF * | 5 wt% LF * | 10 wt% LF * | 15 wt% LF * |
|---|---|---|---|---|
| Highest | CCA | Neat PLA | Neat PLA | Neat PLA |
 | CLLA | CCA | CCA | CCA |
| Neat PLA | CBA | CLLA | BW | |
| CBA | CLLA | BW | CLLA | |
| Lowest | BW | BW | CBA | CBA |
| |η*| (Pa∙s) | 3 wt% HF * | 5 wt% HF * | 10 wt% HF * | 15 wt% HF * |
|---|---|---|---|---|
| Highest | Neat PLA | Neat PLA | Neat PLA | Neat PLA |
 | CCA | CCA | CCA | CCA |
| CLLA | CBA | BW | BW | |
| BW | CLLA | CLLA | CLLA | |
| Lowest | CBA | BW | CBA | CBA |
| G′ (Pa) | 3 wt% LF * | 5 wt% LF * | 10 wt% LF * | 15 wt% LF * |
|---|---|---|---|---|
| Highest | BW | similar modulus | CCA | CCA |
 | CCA | CBA | BW | |
| CBA | CLLA | CLLA | ||
| CLLA | BW | CBA | ||
| Lowest | Neat PLA | Neat PLA | Neat PLA |
| G′ (Pa) | 3 wt% HF * | 5 wt% HF * | 10 wt% HF * | 15 wt% HF * |
|---|---|---|---|---|
| Highest | Neat PLA | Neat PLA | Neat PLA | Neat PLA |
 | CCA | CCA | CCA | CCA |
| CLLA | CLLA | BW | BW | |
| CBA | BW | CLLA | CBA | |
| Lowest | BW | CBA | CBA | CLLA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendoza-Duarte, M.E.; Estrada-Moreno, I.A.; López-Martínez, E.I.; Vega-Rios, A. Effect of the Addition of Different Natural Waxes on the Mechanical and Rheological Behavior of PLA—A Comparative Study. Polymers 2023, 15, 305. https://doi.org/10.3390/polym15020305
Mendoza-Duarte ME, Estrada-Moreno IA, López-Martínez EI, Vega-Rios A. Effect of the Addition of Different Natural Waxes on the Mechanical and Rheological Behavior of PLA—A Comparative Study. Polymers. 2023; 15(2):305. https://doi.org/10.3390/polym15020305
Chicago/Turabian StyleMendoza-Duarte, Mónica Elvira, Iván Alziri Estrada-Moreno, Erika Ivonne López-Martínez, and Alejandro Vega-Rios. 2023. "Effect of the Addition of Different Natural Waxes on the Mechanical and Rheological Behavior of PLA—A Comparative Study" Polymers 15, no. 2: 305. https://doi.org/10.3390/polym15020305
APA StyleMendoza-Duarte, M. E., Estrada-Moreno, I. A., López-Martínez, E. I., & Vega-Rios, A. (2023). Effect of the Addition of Different Natural Waxes on the Mechanical and Rheological Behavior of PLA—A Comparative Study. Polymers, 15(2), 305. https://doi.org/10.3390/polym15020305