Fabrication, Characterization, and Microbial Biodegradation of Transparent Nanodehydrated Bioplastic (NDB) Membranes Using Novel Casting, Dehydration, and Peeling Techniques
Abstract
1. Introduction
2. Materials and Methods
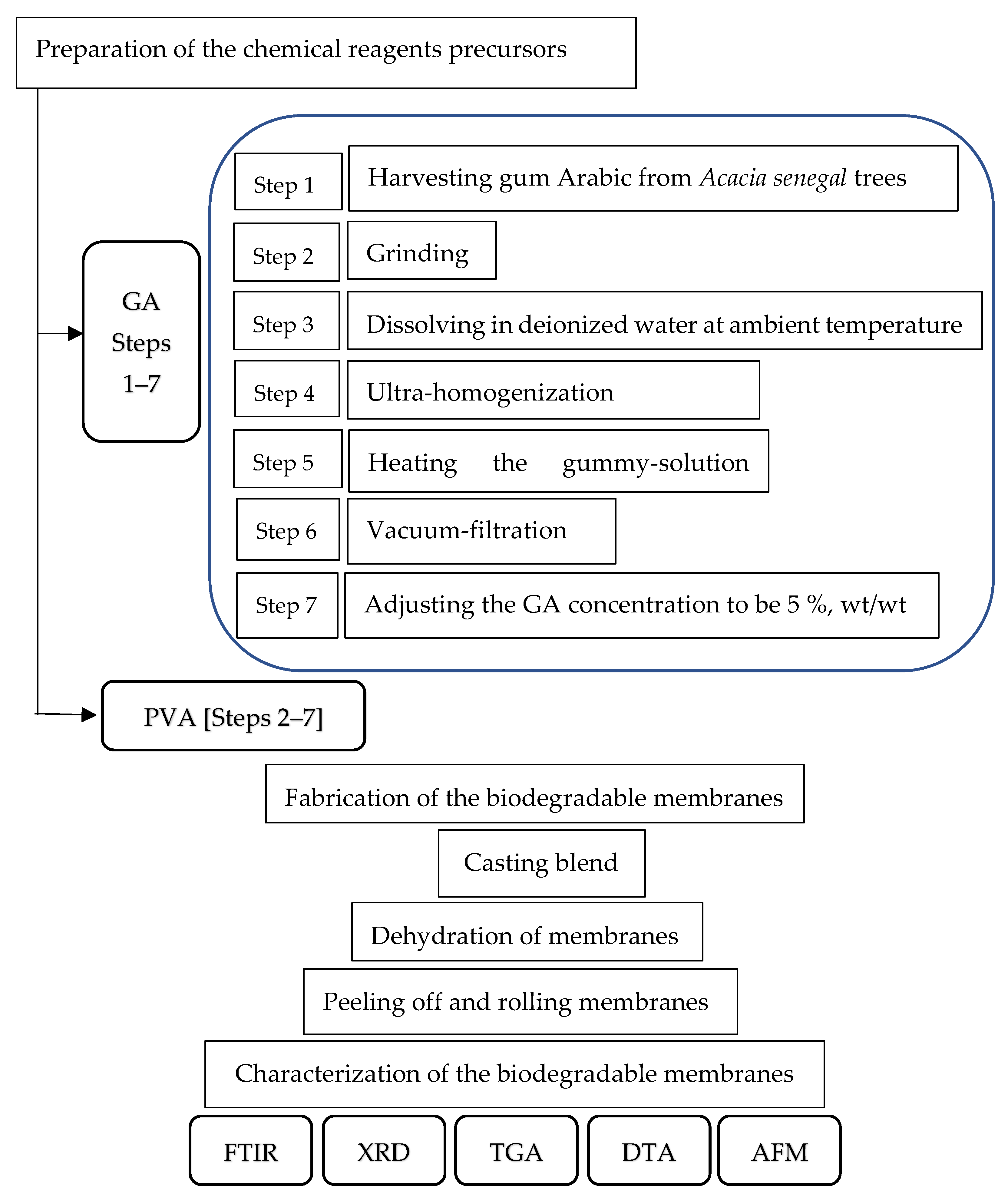
2.1. Raw Material
2.1.1. GA
2.1.2. PVA
2.2. Preparation of the Bioplastic Blends
2.3. The Casting Platform
2.4. Casting the Bioplastic Blends
2.5. Drying the NDBs
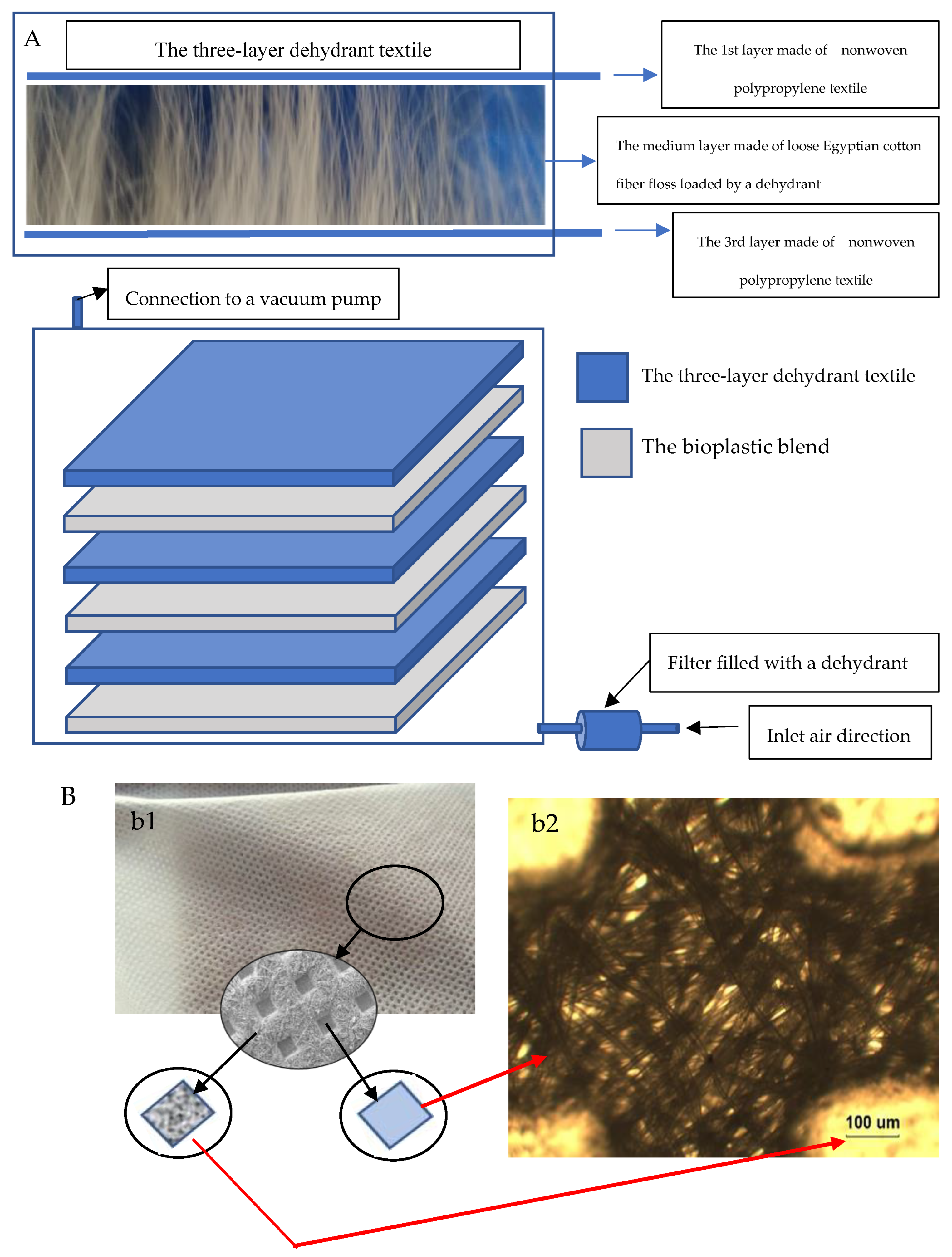
The Stratified Nano-Dehydrator (SND)
2.6. Peeling off the Bioplastic Membranes
2.7. Characterization of the Bioplastic Membranes
2.7.1. FTIR
2.7.2. X-ray Diffraction (XRD)
2.7.3. Thermal Analysis
2.7.4. Surface Topography (ST)
2.8. Mechanical Properties of the Bioplastic Membranes
2.8.1. Ultimate Tensile Strength (UTS)
2.8.2. Modulus of Elasticity (MoE)
2.8.3. Elongation at Failure (EaF)
2.9. Microbial Biodegradation
2.9.1. Sample Preparation and Soil Burial Studies
2.9.2. Isolation and Counting of Microbial Communities
2.10. Statistical Design and Analysis
3. Results
3.1. Chemical and Physical Properties of the Bioplastic Membranes
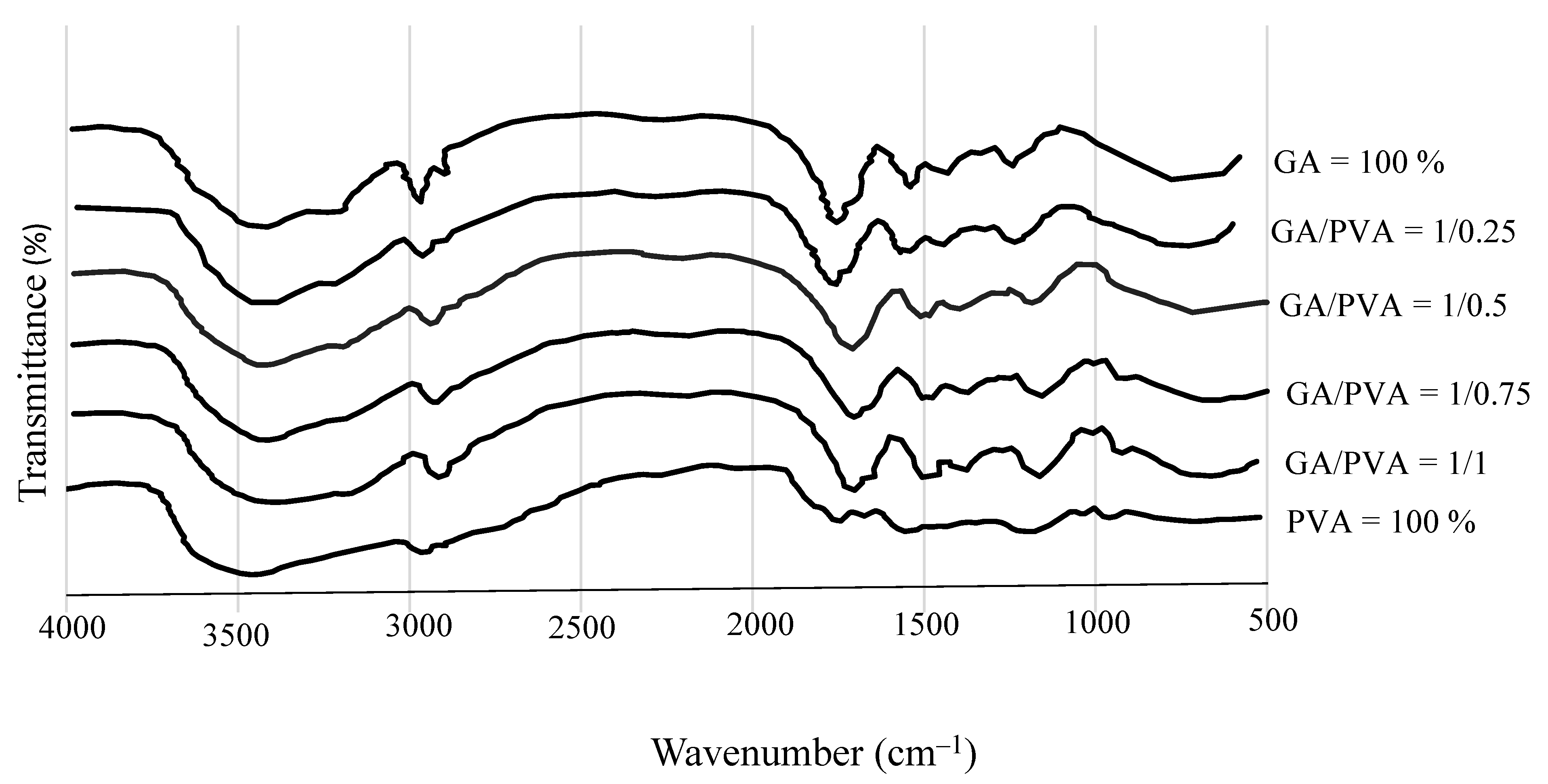
3.1.1. FTIR
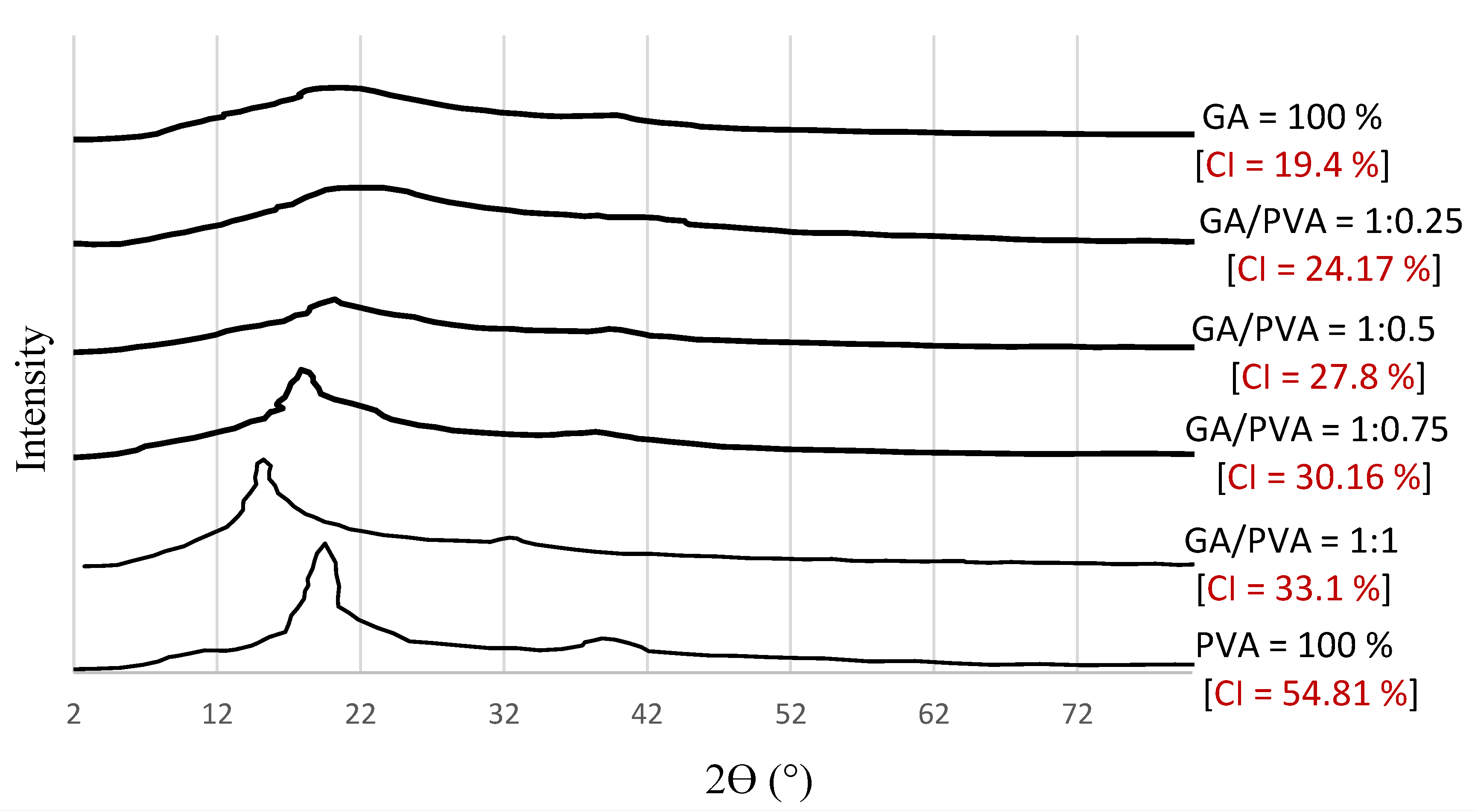
3.1.2. XRD
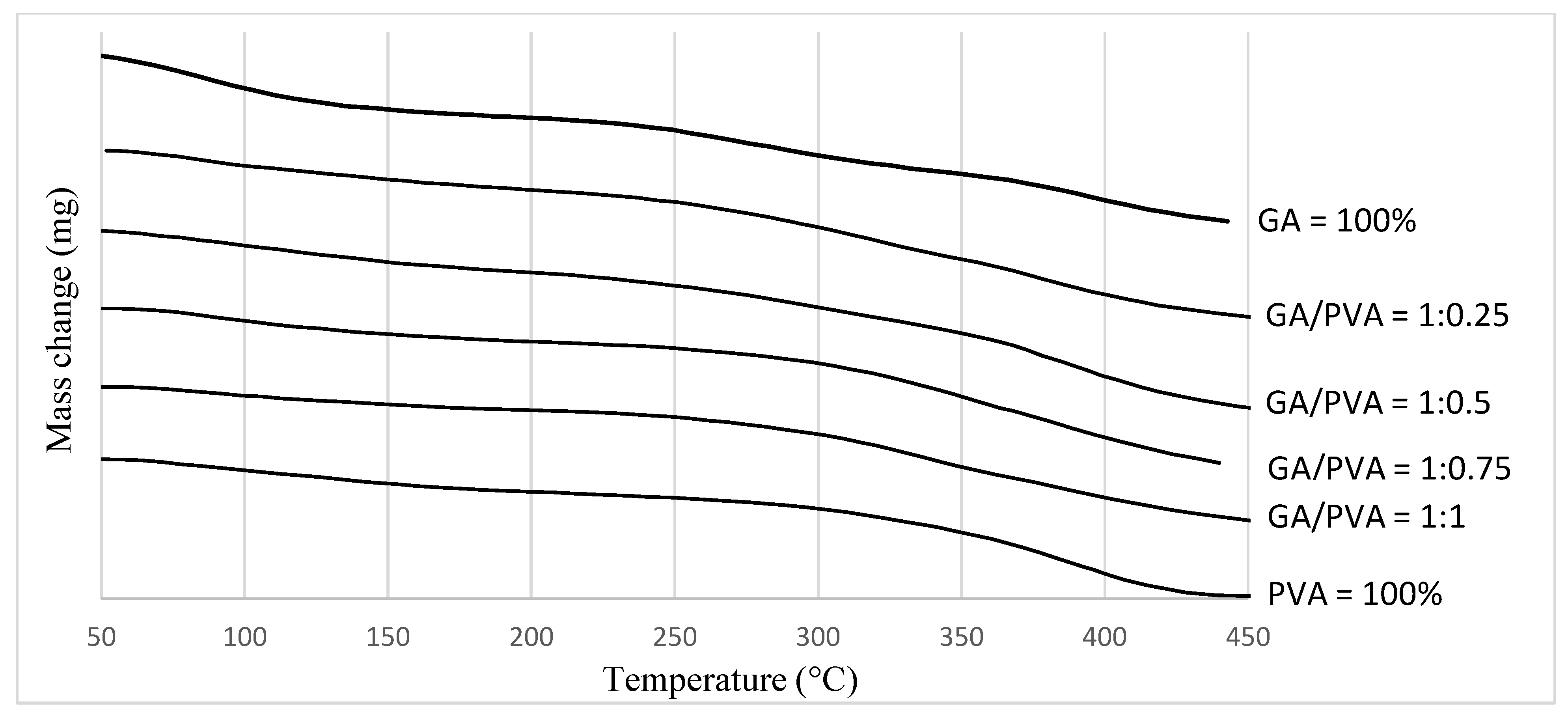
3.1.3. TGA
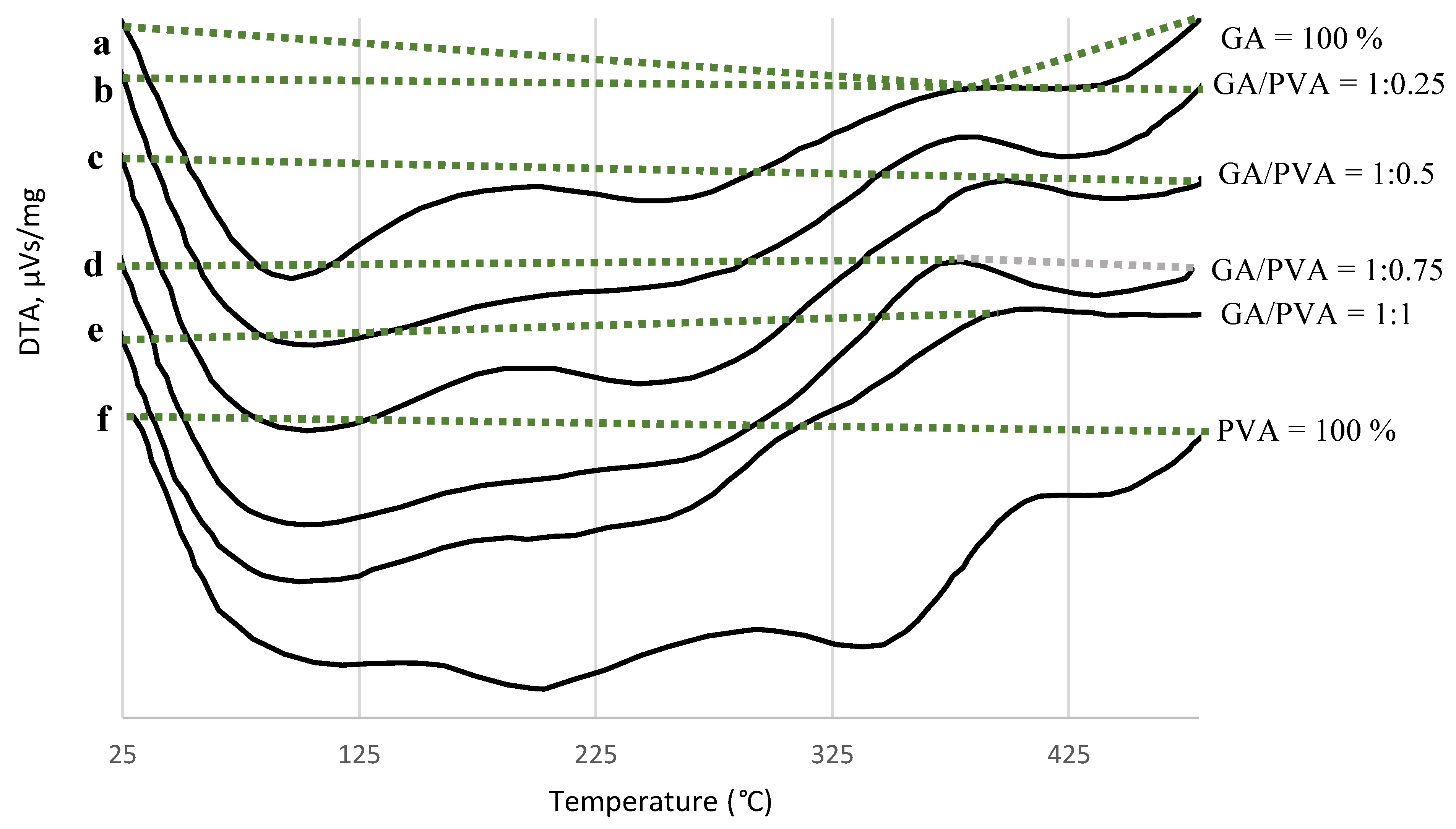
3.1.4. DTA
3.2. Ultrastructure of the Bioplastic Membrane
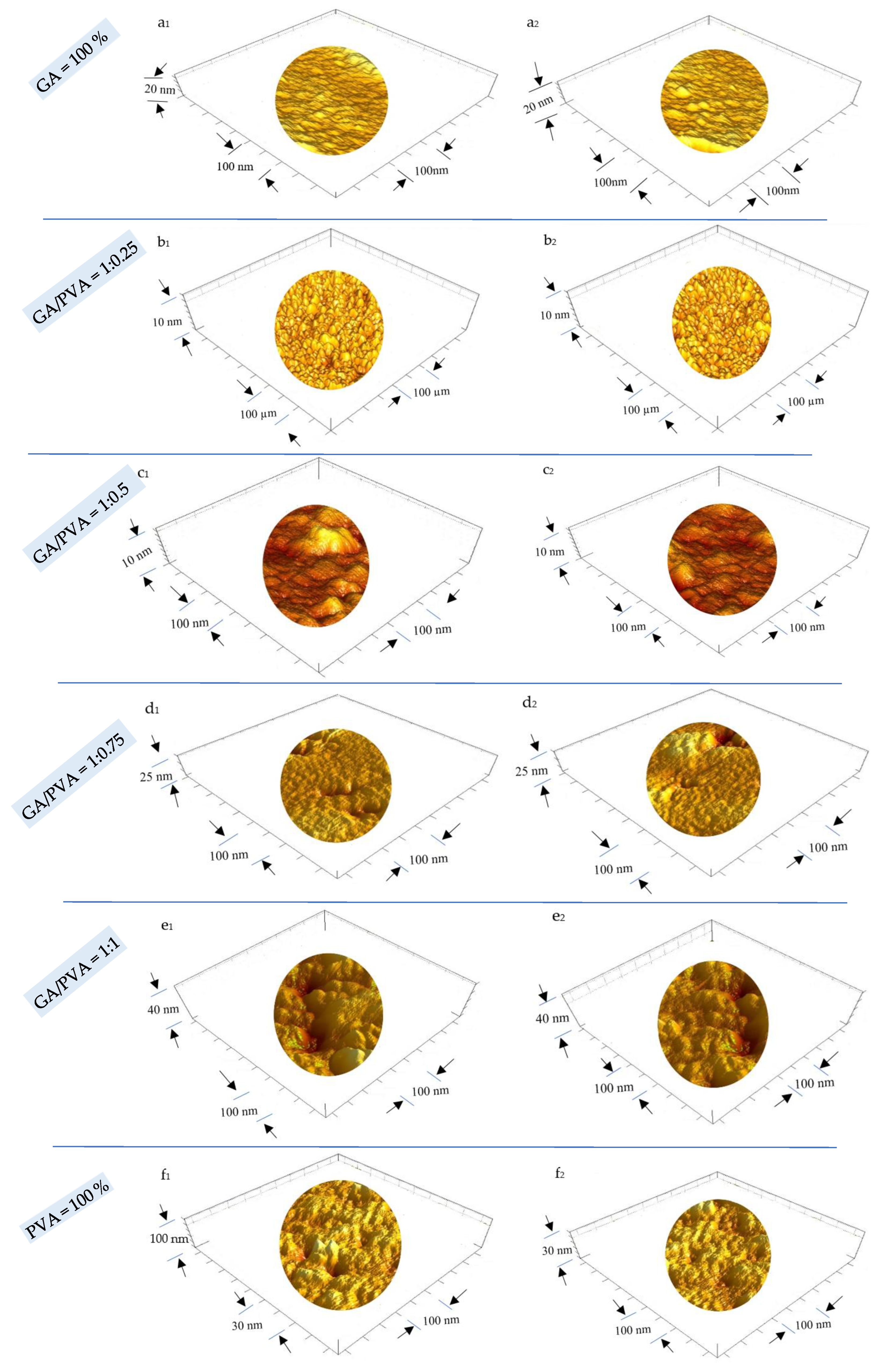
3.2.1. Surface Roughness (SR) and Particle Size (PS)
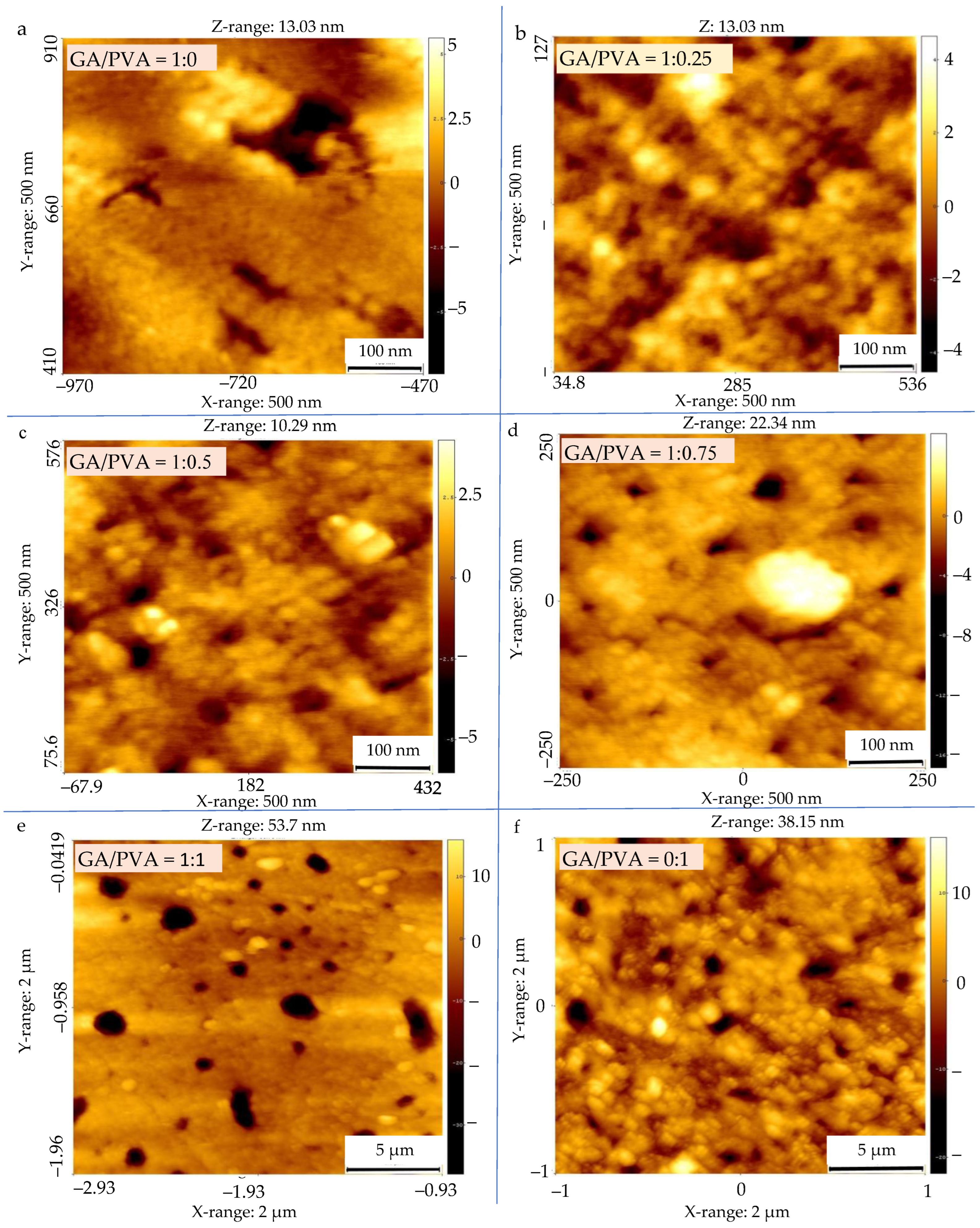
3.2.2. Pore Diameter (PD) and Void Volume (VV) of the NDB Membranes
3.3. Mechanical Properties of the Bioplastic Membranes
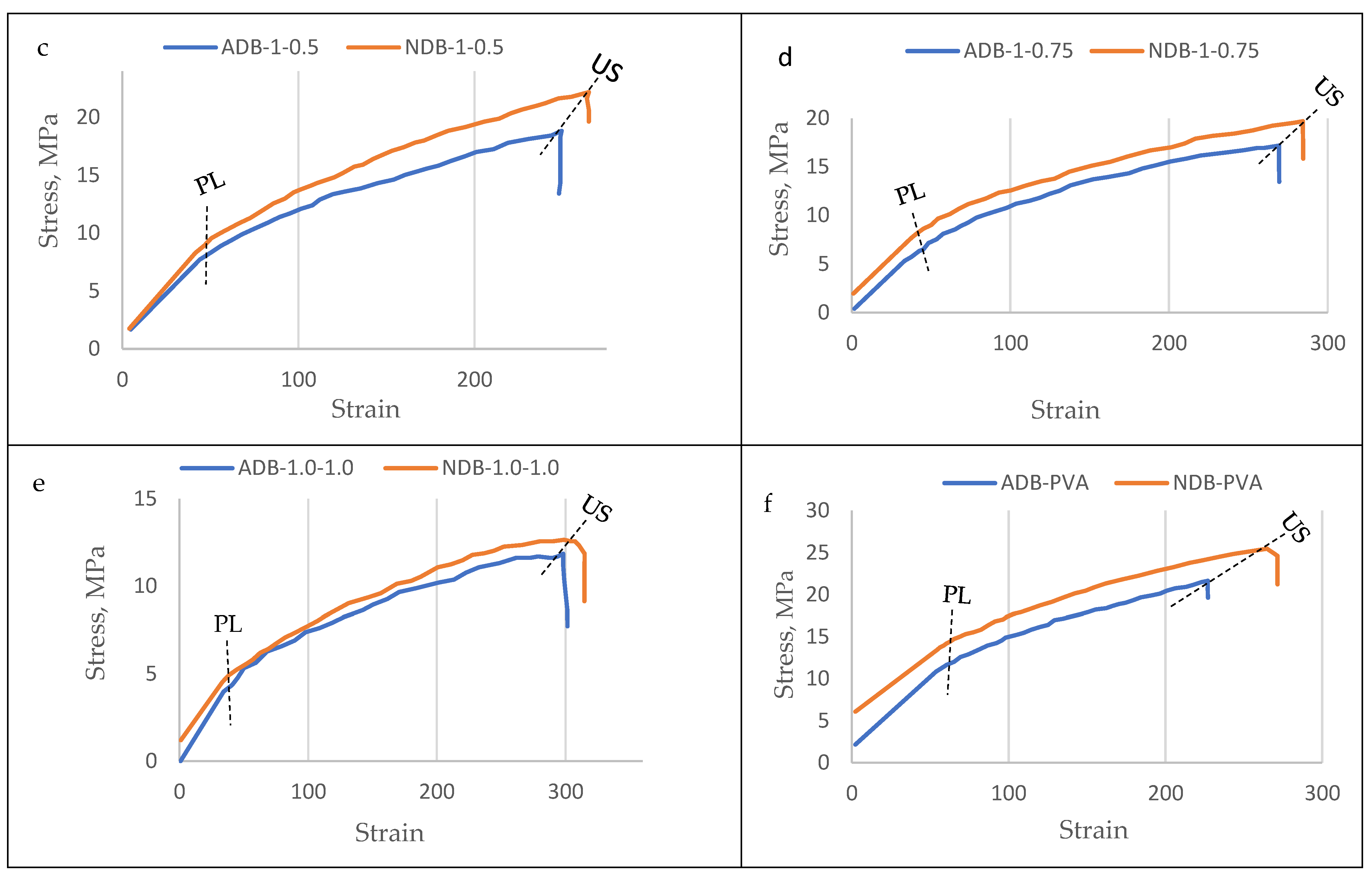
3.3.1. Ultimate Tensile Strength (UTS)
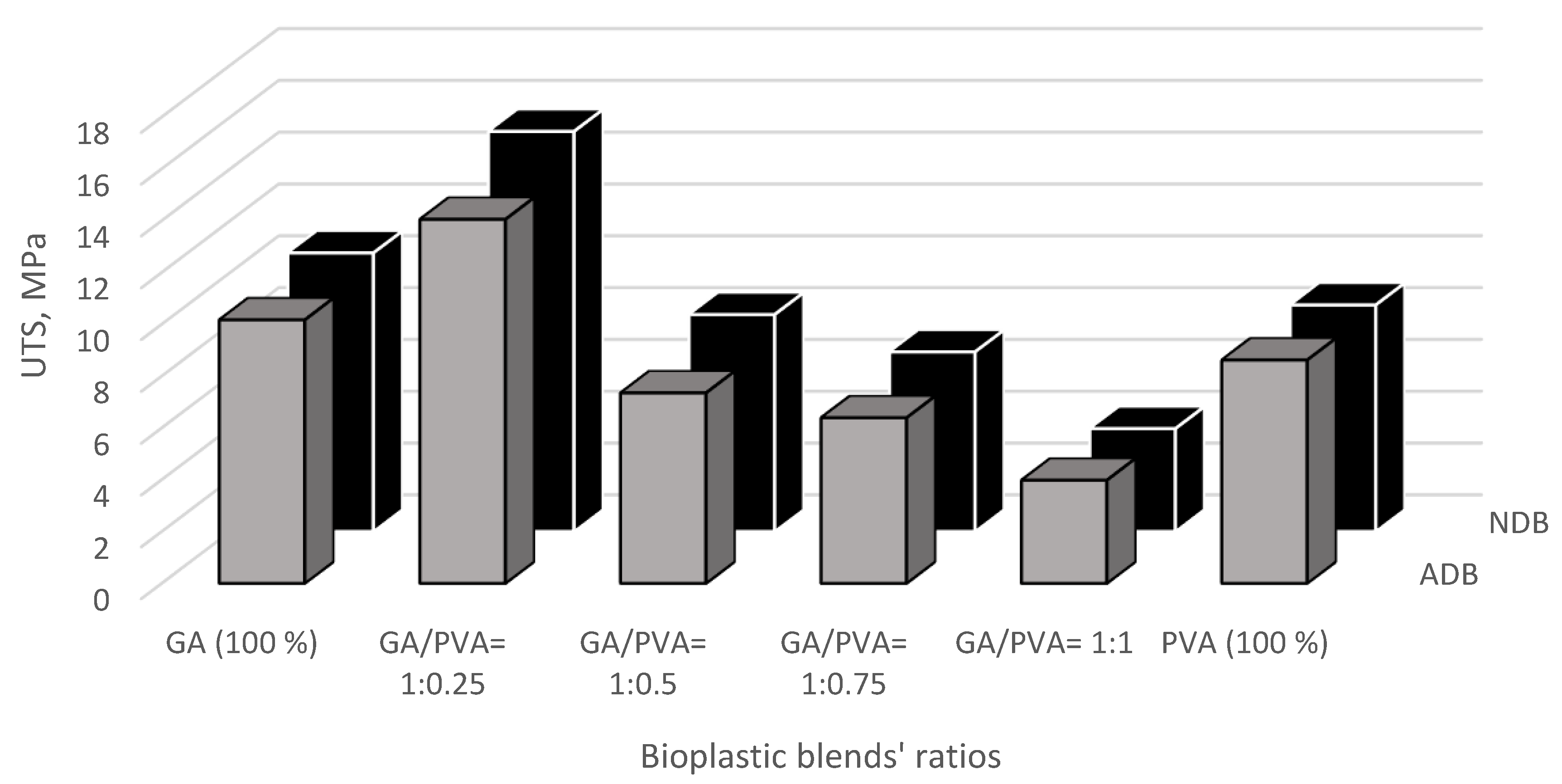
3.3.2. Modulus of Elasticity (MoE)
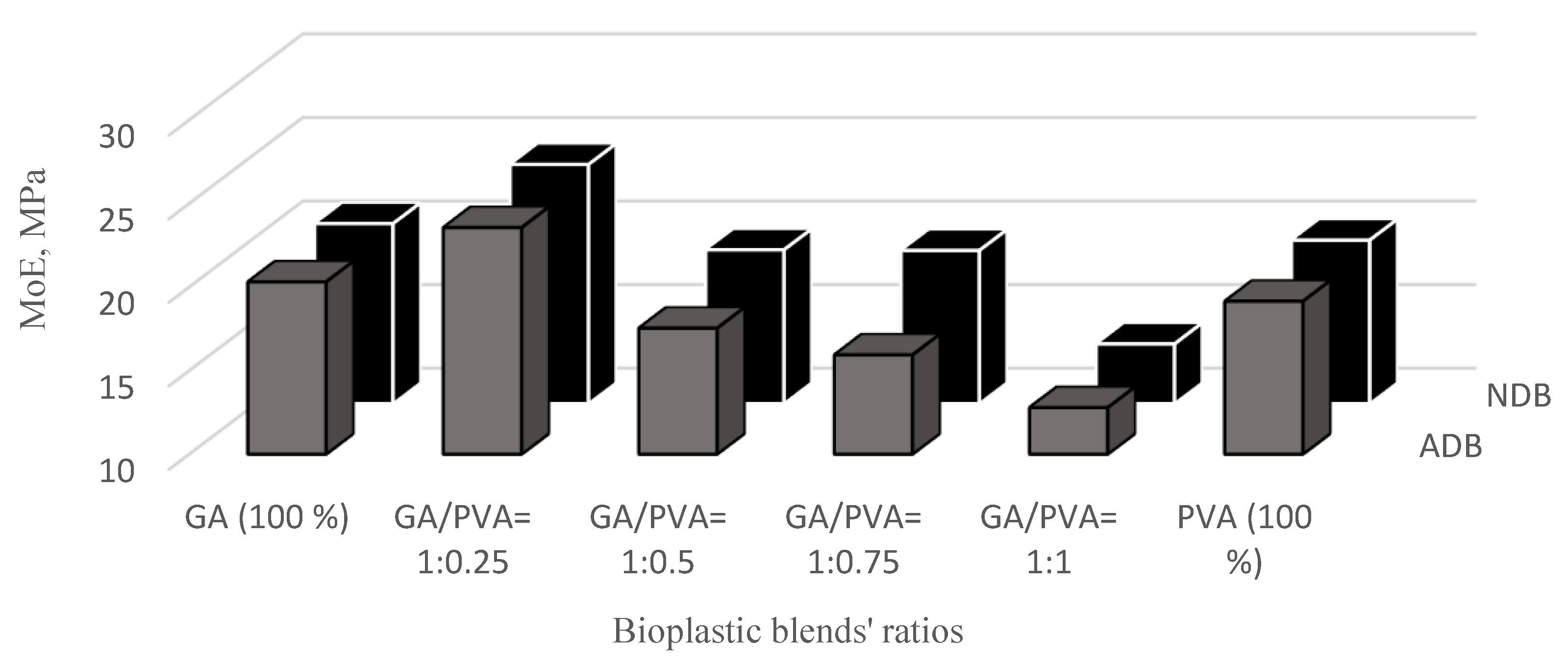
3.3.3. Elongation at Failure (EaF)
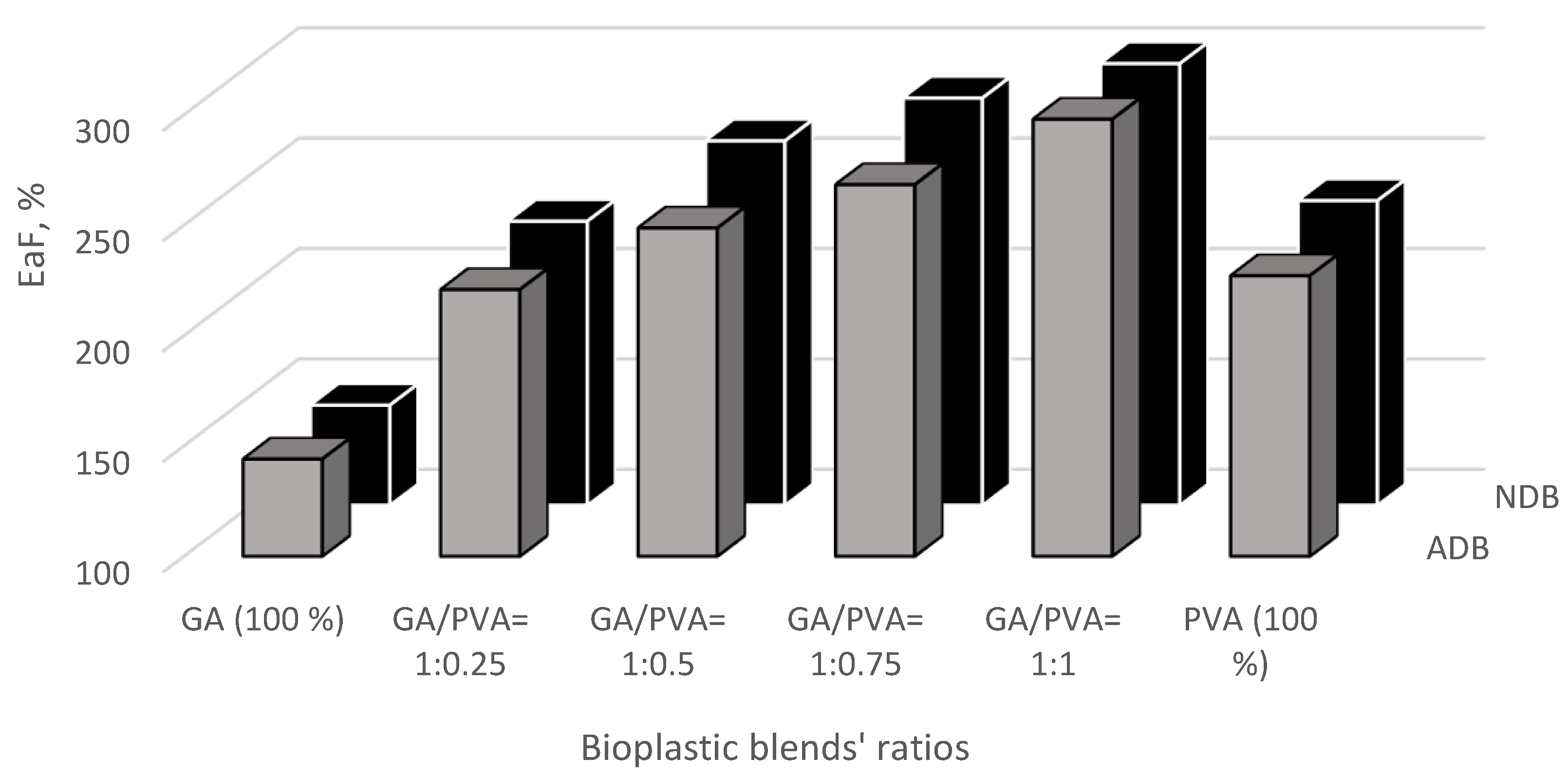
3.4. Bacterial and Fungal Biodegradation
4. Discussion
4.1. Scientific Illustration of the Ease of Peeling the Bioplastic Membranes Away from the Acrylic Platform
- Acrylic is a powerful static generator in terms of electrostatic charge. When its surface is wiped back and forth, positive and negative surficial charges arise that draw and hold microscopic particles. Surficial charge variations have the potential to cause agglomerated particles to discharge in an unanticipated manner, endangering contamination-sensitive materials [127]. PMMA is positioned close to the middle of this empirical series for the surface potential and is regarded as a tribo-positive electron-donating material [119,127].
4.2. Scientific Illustration of the Nanodehydration of the Bioplastic Membranes
4.3. Chemical and Physical Properties of the Bioplastic Membranes
4.3.1. FTIR
4.3.2. XRD
4.3.3. TGA
4.3.4. DTA
4.4. Anatomical Ultrastructure of the Bioplastic Membranes
4.4.1. Surface Roughness and Nanometric Particle Size
4.4.2. Membrane Permeability
4.5. Mechanical Properties of the Bioplastic Membranes
4.6. Microbial Biodegradation
5. Conclusions and Future Perspectives
6. Patent
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Nomenclature
| ADB | Air-dried bioplastic | ||
| ACS | The American Chemical Society | ||
| AFM | Atomic force microscopy | ||
| CI | Crystallinity index | ||
| CFU | Colony-forming unit of microbial populations | ||
| DSC | Differential scanning calorimetry | ||
| DTA | Differential thermal analysis | ||
| EaF | Elongation at failure | ||
| EC | Enthalpy change | ||
| FTIR | Fourier transform infrared spectroscopy | ||
| GA | Gum Arabic | ||
| HC | Heat change in µVs/mg | ||
| HD | Hourly duplication | ||
| MoE | Modulus of elasticity | ||
| µVs/mg | Microvolts per milligram | ||
| NDB | Nanodehydrated bioplastic | ||
| NPS | Nanometric particle size | ||
| PubChem | Open chemistry database managed by the National Institutes of Health (NHI) | ||
| PVA | Polyvinyl alcohol | ||
| SD | Standard deviation | ||
| SECT | Self-electrostatic charged-template | ||
| SP | Statistical parameters | ||
| SR | Surface roughness | ||
| PD | Pore diameter | ||
| PS | Particle size | ||
| TGA | Thermogravimetric analysis | ||
| TR | Temperature range (°C) | ||
| UTS | Ultimate tensile strength | ||
| XRD | X-ray diffraction | ||
| VFHF | Vibrated-free horizontal flow | ||
| VV | Void volume |
References
- Mergaert, J.; Anderson, C.; Wouters, A.; Swings, J.; Kersters, K. Biodegradation of polyhydroxyalkanoates. FEMS Microbiol. Lett. 1992, 9, 317–321. [Google Scholar] [CrossRef] [PubMed]
- Nampoothiri, K.M.; Nair, N.R.; John, R.P. An overview of the recent developments in polylactide (PLA) research. Bioresour. Technol. 2010, 101, 8493–8501. [Google Scholar] [CrossRef]
- Boyandin, A.N.; Prudnikova, S.V.; Filipenko, M.L.; Khrapov, E.A.; Vasil’ev, A.D.; Volova, T.G. Biodegradation of polyhydroxyalkanoates by soil microbial communities of different structures and detection of PHA degrading microorganisms. Appl. Biochem. Microbiol. 2012, 48, 28–36. [Google Scholar] [CrossRef]
- Merugu, R. Studies on PHB (Polyhydroxy butyrate) degradation by some species of Aspergillus. Studies 2012, 4, 1111–1113. [Google Scholar]
- Gautam, N.; Kaur, I. Soil burial biodegradation studies of starch grafted polyethylene and identification of Rhizobium meliloti therefrom. J. Environ. Chem. Ecotoxicol. 2013, 5, 147–158. [Google Scholar]
- Karamanlioglu, M. Environmental Degradation of the Compostable Plastic Packaging Material Poly (lactic) acid and Its Impact on Fungal Communities in Compost. Ph.D. Thesis, Manchester University, Manchester, UK, 2013; p. 198. [Google Scholar]
- Badreldin, H.A.; Al-Husseni, I.; Beegam, S.; Al-Shukaili, A.; Nemmar, A.; Schierling, S.; Queisser, N.; Schupp, N. Effect of gum Arabic on oxidative stress and inflammation in adenine–induced chronic renal failure in rats. PLoS ONE 2013, 8, e55242. [Google Scholar]
- Feddersen, R.L.; Thorp, S.N. Sodium carboxymethyl cellulose. In Industrial Gums, Polysaccharides and Their Derivatives; Whistler, R.L., Bemiller, J.N., Eds.; Academic Press: New York, NY, USA, 1993; pp. 537–578. [Google Scholar]
- Verbeken, D.; Dierckx, S.; Dewettinck, K. Exudate gums: Occurrence, production, and applications. Appl. Microbiol. Biotechnol. 2003, 63, 10–21. [Google Scholar] [CrossRef]
- Williams, P.A.; Phillips, G.O. Handbook of Hydrocolloids; Williams, P.A., Phillips, G.O., Eds.; CRC Press: Cambridge, UK; Elsevier: Amsterdam, The Netherlands, 2000; pp. 155–168. [Google Scholar]
- Anonymous; FAO. Food and Nutrition; Food and Agriculture Organization: Rome, Italy, 1990; p. 49. [Google Scholar]
- Buffo, R.A.; Reineccius, G.A.; Oehlert, G.W. Factors affecting the emulsifying and rheological properties of gum acacia in beverage emulsions. Food Hydrocoll. 2001, 15, 53–66. [Google Scholar] [CrossRef]
- Almuslet, N.A.; Hassan, E.A.; Al-Sherbini, A.A.M.; Muhgoub, M.G.A. Diode laser (532 nm) induced grafting of polyacrylamide onto gum Arabic. J. Phys. Sci. 2012, 23, 43–53. [Google Scholar]
- Anonymous. Production and Marketing of Gum Arabic; Network for Natural Gums and Resins in Africa (NGARA): Nairobi, Kenya, 2016. [Google Scholar]
- Krempel, M.; Griffin, K.; Khouryieh, H. Hydrocolloids as emulsifiers and stabilizers in beverage preservation. In Preservatives and Preservation Approaches in Beverages; The Science of Beverages; Grumezescu, A.M., Holban, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2019; Volume 15, pp. 427–465. [Google Scholar]
- Rinsky, L.H.; Rinsky, G. The Pastry Chef’s Companion: A Comprehensive Resource Guide for the Baking and Pastry Professional; John Wiley & Sons: Chichester, UK, 2009; Volume 1, p. 134. [Google Scholar]
- Maqbool, M.; Ali, A.; Alderson, P.G.; Zahid, N. Exploring the new applications of gum Arabic obtained from acacia species to preserve fresh fruits and vegetables. In Proceedings of the II International Symposium on Underutilized Plant Species: Crops for the Future—Beyond Food Security, Kuala Lumpur, Malaysia, 27 June–1 July 2013; Volume 2, pp. 2406–6168. [Google Scholar]
- McEachran, R. Gum Arabic: The Invisible Ingredient in Soft Drink Supply Chains. Guardian 2013. Available online: www.theguardian.com (accessed on 16 February 2023).
- Anderson, D.M.W.; Farquhar, J.G.K. Gum exudates from the genus Prosopis. Int. Tree Crops J. 1982, 2, 15–24. [Google Scholar] [CrossRef]
- Anderson, D.M.W.; McNab, C.G.A.; Anderson, C.G.; Brown, P.M.; Pringuer, M.A. Studies of uronic acid materials, Part 58: Gum exudates from the genus Sterculia (gum karaya). Int. Tree Crops J. 1983, 2, 147–154. [Google Scholar] [CrossRef]
- Suliman, S.M.; Hamdouk, M.I.; Elfaki, M.B. Gum Arabic fibre as a supplement to low protein diet in chronic renal failure patients. Sudan Association of Physicians. In Proceedings of the 17th Conference, Friendship Hall, Khartoum, Sudan, 21–23 March 2000. [Google Scholar]
- Gamal el-din, A.M.; Mostafa, A.M.; Al-Shabanah, O.A.; Al-Bekairi, A.M.; Nagi, M.N. Protective effect of Arabic gum against acetaminophen–induced hepatotoxicity in mice. Pharmacol. Res. 2003, 48, 631–635. [Google Scholar] [CrossRef]
- Eltayeb, I.B.; Awad, A.I.; Elderbi, M.A.; Shadad, S.A. Effect of gum Arabic on the absorption of a single oral dose of amoxicillin in healthy Sudanese volunteers. J. Antimicrob. Chemother. 2004, 54, 577–578. [Google Scholar] [CrossRef]
- Ali, A.A.; Ali, K.E.; Fadlalla, A.; Khalid, K.E. The effects of gum Arabic oral treatment on the metabolic profile of chronic renal failure patients under regular haemodialysis in central Sudan. Nat. Prod. Res. 2008, 22, 12–21. [Google Scholar] [CrossRef]
- Hills, S. Gum Arabic Caloric Value Lowered. 2008. Available online: www.foodnavigator-usa.com (accessed on 24 January 2023).
- Ali, B.H.; Ziada, A.; Blunden, G. Biological effects of gum Arabic: A review of some recent research. Food Chem. Toxicol. 2009, 47, 1–8. [Google Scholar] [CrossRef]
- Omer, A.E.; Ayed, I.A.M.; El Badwi, S.M.A. Effect of gum Arabic on nephrotoxicity induced by Aristolochia bracteolata in rats. Sch. Acad. J. Biosci. 2013, 1, 377–380. [Google Scholar]
- Luo, Y.; Zhang, Y.; Pan, K.; Critzer, F.; Davidson, P.M.; Zhong, Q. Self-emulsification of alkaline–dissolved clove bud oil by whey protein, gum Arabic, lecithin, and their combinations. J. Agric. Food. Chem. 2014, 62, 4417–4424. [Google Scholar] [CrossRef]
- Wang, H.; Williams, P.A.; Senan, C. Synthesis, characterization and emulsification properties of dodecenyl succinic anhydride derivatives of gum Arabic. Food Hydrocoll. 2014, 37, 143–148. [Google Scholar] [CrossRef]
- Lawrence, R.; Jeyakumar, E.; Gupta, A. Antibacterial activity of Acacia Arabica (Bark) extract against selected multi drug resistant pathogenic bacteria. Int. J. Curr. Microbiol. Appl. Sci. 2015, 1, 213–222. [Google Scholar]
- Hadavi, M.; Hasannia, S.; Faghihi, S.; Mashayekhi, F.; Zadeh, H.; Mostofi, S. Novel calcified gum Arabic porous nano–composite scaffold for bone tissue regeneration. Biochem. Biophys. Res. Commun. 2017, 488, 671–678. [Google Scholar] [CrossRef]
- Salih, N.K. Applications of gum Arabic in medical and health benefits. In Gum Arabic; Academic Press: Cambridge, MA, USA, 2018; pp. 269–281. [Google Scholar]
- Kraaijpoel, D.; Herenius, C. Het Kunstschilderboek–Handboek voor Materialen en Technieken, Cantecleer; Tirion Art: Utrecht, The Netherlands, 2007; p. 183. [Google Scholar]
- Banerjee, S.S.; Chen, D.-H. Magnetic nanoparticles grafted with cyclodextrin for hydrophobic drug delivery. Chem. Mater. 2007, 19, 6345–6349. [Google Scholar] [CrossRef]
- Wilson, O.C., Jr.; Blair, E.; Kennedy, S.; Rivera, G.; Mehl, P. Surface modification of magnetic nanoparticles with oleylamine and gum Arabic. Mater. Sci. Eng. C 2008, 28, 438–442. [Google Scholar] [CrossRef]
- Kattumuri, V.; Katti, K.; Bhaskaran, S.; Boote, E.J.; Casteel, S.W.; Fent, G.M.; Robert-son, D.J.; Chandrasekhar, M.; Kannan, R.; Katti, K.V. Gum Arabic as a photochemical construct for the stabilization of gold nanoparticles: In vivo pharmacokinetics and X-ray-contrast-imaging studies. Small 2007, 3, 333–341. [Google Scholar] [CrossRef]
- Kumar, M.K.; Reddy, A.L.M.; Ramaprabhu, S. Exfoliated single-walled carbonnnanotube-based hydrogen sensor. Sens. Actuators B 2008, 130, 653–660. [Google Scholar] [CrossRef]
- Park, C.; Lim, K.H.; Kwon, D.; Yoon, T.H. Biocompatible quantum dot nanocolloids stabilized by gum Arabic. Bull. Kor. Chem. Soc. 2008, 29, 1277–1279. [Google Scholar]
- Razzak, M.T.; Darwis, D. Irradiation of polyvinyl alcohol and polyvinyl pyrrolidone blended hydrogel for wound dressing. Radiat. Phys. Chem. 2001, 62, 107–113. [Google Scholar] [CrossRef]
- DeMerlis, C.C.; Schoneker, D.R. Review of the oral toxicity of polyvinyl alcohol (PVA). Food Chem. Toxicol. 2003, 41, 319–326. [Google Scholar] [CrossRef]
- Dos Reis, E.F.; Campos, F.S.; Lage, A.P.; Leite, R.C.; Heneine, L.G.; Vasconcelosc, W.L.; Portela Lobato, Z.I.; Mansur, H.S. Synthesis and characterization of Poly (Vinyl Alcohol) Hydrogels and Hybrids for rMPB70 Protein Adsorption. Mat. Res. 2006, 9, 185–191. [Google Scholar] [CrossRef]
- Nair, N.R.; Nampoothiri, K.M.; Pandey, A. Preparation of poly (L-lactide) blends and biodegradation by Lentzea waywayandensis. Biotechnol. Lett. 2012, 34, 2031–2035. [Google Scholar] [CrossRef]
- Liu, M.; Guo, B.; Du, M.; Jia, D. Drying induced aggregation of halloysite nanotubes in polyvinyl alcohol/halloysite nanotubes solution and its effect on properties of composite film. Appl. Phys. A 2007, 88, 391–395. [Google Scholar] [CrossRef]
- Masti, S.P.; Chougale, R.B. Influence of Poly (Vinylpyrrolidone) on Binary Blend Films Made from Poly(Vinyl Alcohol)/Chitosan. Inter. Res. J. Env. Sci. 2014, 3, 11–13. [Google Scholar]
- Limpan, N.; Prodpran, T.; Benjakul, S.; Prasarpran, S. Influences of degree of hydrolysis and molecular weight of poly (vinyl alcohol), PVA on properties of fish myofibrillar protein/PVA blend films. Food Hydrocoll. 2012, 29, 226–233. [Google Scholar] [CrossRef]
- Qiu, K.; Netravali, A.N. Fabrication and characterization of biodegradable composites based on microfibrillated cellulose and polyvinyl alcohol. Compos. Sci. Technol. 2012, 72, 1588–1594. [Google Scholar] [CrossRef]
- Qiu, K.; Netravali, A.N. A Composting study of membrane–like polyvinyl alcohol based resins and nanocomposites. J. Polym. Environ. 2013, 21, 658–674. [Google Scholar] [CrossRef]
- Qiu, K.; Netravali, A.N. Halloysite nanotube reinforced biodegradable nanocomposites using noncrosslinked and malonic acid crosslinked polyvinyl alcohol. Polym. Compos. 2013, 34, 799–809. [Google Scholar] [CrossRef]
- Mudigoudra, B.S.; Masti, S.P.; Chougale, R.B. Thermal behavior of poly (vinyl alcohol). Poly (vinyl pyrrolidone)/chitosan ternary polymer blend films. Res. J. Recent Sci. 2012, 1, 83–86. [Google Scholar]
- Onyari, J.M.; Mulaa, F.; Muia, J.; Shiundu, P. Biodegradability of poly (lactic acid), preparation and characterization of PLA/gum Arabic blends. J. Polym. Environ. 2008, 16, 205–212. [Google Scholar] [CrossRef]
- Cozic, C.; Picton, L.; Garda, M.R.; Marlhoux, F.; Le Cerf, D. Analysis of Arabic gum: Study of degradation and water desorption processes. Food Hydrocoll. 2009, 23, 1930–1934. [Google Scholar] [CrossRef]
- Tiwari, A.; Terada, D.; Kobayash, H. Polyvinyl modified guar–gum bioplastics for packaging applications. Handb. Bioplastics Biocomposites Eng. Appl. 2011, 24, 177. [Google Scholar]
- Nakashima, T.; Xu, C.; Bin, Y.; Matsuo, M. Morphology and mechanical properties of poly (vinyl alcohol) and starch blends prepared by gelation/crystallization from solutions. Colloid. Polym. Sci. 2001, 279, 646–654. [Google Scholar] [CrossRef]
- Huang, X.; Netravali, A. Biodegradable green composites made using bamboo micro/nano–fibrils and chemically modified soy protein resin. Compos. Sci. Technol. 2009, 69, 1009–1015. [Google Scholar] [CrossRef]
- Padil, V.V.T.; Nguyen, N.H.; Ševců, A.; Černík, M. Fabrication, characterization, and antibacterial properties of electrospun membrane composed of gum karaya, polyvinyl alcohol, and silver nanoparticles. J. Nanomater. 2015, 271, 32–38. [Google Scholar]
- Padil, V.V.T.; Cernik, M.; Vellora, V. Tree gum based electrospun nanofibre sheets: Process optimization, characterization and environmental application. In Proceedings of the Nanocon 2014, 6th International Conference, Brno, Czech Republic, 4–7 November 2014. [Google Scholar]
- Hindi, S.S.; Albureikan, M.O.I. System, Apparatus, and Methods for Manufacturing Biodegradable Biopolymeric Materials. U.S. Patent No. 11548192, 1 October 2023. [Google Scholar]
- Volova, T.G.; Boyandin, A.N.; Vasil’ev, A.D.; Karpov, V.A.; Kozhevnikov, I.V.; Prudnikova, S.V.; Gitel’Zon, I.I. Biodegradation of polyhydroxyalkanoates (PHAs) in the South China Sea and identification of PHA–degrading bacteria. Microbiology 2011, 80, 252. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, Y.; Du, G.C.; Hua, Z.Z.; Zhu, Y. Biodegradation of polyvinyl alcohol by a mixed microbial culture. Enzym. Microb. Technol. 2007, 40, 1686–1691. [Google Scholar] [CrossRef]
- Corti, A.; Solaro, R.; Chiellini, E. Biodegradation of poly (vinyl alcohol) in selected mixed microbial culture and relevant culture filtrate. Polym. Degrad. Stab. 2002, 75, 447–458. [Google Scholar] [CrossRef]
- Rong, D.; Usui, K.; Morohoshi, T.; Kato, N.; Zhou, M.; Ikeda, T. Symbiotic degradation of polyvinyl alcohol by Novosphingobium sp. and Xanthobacter flavus. J. Environ. Biotechnol. 2009, 9, 131–134. [Google Scholar]
- Abd Alla, F.A.A. The Effects of Microbiological Biodegradation on Gum Arabic Structure and Molecular Mass. Ph.D. Thesis, Sudan University of Science and Technology, Khartoum, Sudan, 2012. [Google Scholar]
- Wail, F.; Sabir, A.; Jacob, K.I. Novel reverse osmosis membranes composed of modified PVA/gum Arabic conjugates: Biofouling mitigation and chlorine resistance enhancement. Carbohydr. Polym. 2017, 155, 28–39. [Google Scholar]
- Solomon, M.M.; Gerengi, H.; Umoren, S.A.; Essien, N.B.; Essien, U.B.; Kaya, E. Gum Arabic-silver nanoparticles composite as a green anticorrosive formulation for steel corrosion in strong acid media. Carbohydr. Polym. 2018, 181, 43–55. [Google Scholar] [CrossRef]
- Tahsiri, Z.; Mirzaei, H.; Hosseini, S.M.H.; Khalesi, M. Gum Arabic improves the mechanical properties of wild almond protein film. Carbohydr. Polym. 2019, 222, 114994. [Google Scholar] [CrossRef]
- Ling, M.; Xu, Y.; Zhao, H.; Gu, X.; Qiu, J.; Li, S.; Wu, M.; Song, X.; Yan, C.; Liu, G.; et al. Dual-functional gum Arabic binder for silicon anodes in lithium ion batteries. Nano Energy 2015, 12, 178–185. [Google Scholar] [CrossRef]
- Silvestri, D.; Mikšíček, J.; Wacławek, S.; Torres-Mendieta, R.; Padil, V.V.; Černík, M. Production of electrospun nanofibers based on graphene oxide/gum Arabic. Int. J. Biol. Macromol. 2019, 124, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Lubambo, A.F.; de Freitas, R.A.; Sierakowski, M.R.; Lucyszyn, N.; Sassaki, G.L.; Serafim, B.M.; Saul, C.K. Electrospinning of commercial guar–gum: Effects of purification and filtration. Carbohydr. Polym. 2013, 93, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Tieguhong, J.C.; Ndoye, O. Development of trade and marketing of non-wood forest products for poverty alleviation Africa. In Proceedings of the Lessons Learnt on SFM in Africa, Uppsala, Sweden, 18–22 October 2004. [Google Scholar]
- Anonymous. Policy Note: Export Marketing of Gum Arabic from Sudan. Washington, D.C. World Bank Group. Available online: http://documents.worldbank.org/curated/en/736741468334873447/Policy-note-export-marketing-of-gum-arabic-from-Sudan (accessed on 2 March 2023).
- ElKhawad, H.; ElBagher, M.A. Gum Arabic Processing and Arketing in the Sudan. Master’s Thesis, Chemical Engineering, Khartoum, Sudan, 2008. [Google Scholar]
- Naili, D.; Wenzhi, D. Technology for Producing Gum Arabic Powder. Chinese Patent Application No. CN 101143995A, 19 March 2008. [Google Scholar]
- Chikamai, B.N.; Banks, W.B.; Anderson, D.M.W.; Weiping, W. Processing of gum Arabic and some new opportunities. Food Hydrocoll. 1996, 10, 309–316. [Google Scholar] [CrossRef]
- Masti, S.; Kasai, D.; Mudigoudra, B.; Chougale, R. Effect of gum acacia (GA) on tensile properties of biodegradable chitosan (CS)/poly (vinyl alcohol) (PVA) polymer blend films. J. Mater. Sci. 2016, 4, 5–8. [Google Scholar]
- Chougale, S.I.; Mamza, P.A.P.; Ja’o, A.M. Effect of pure and modified gum Arabic on the mechanical properties of poly (vinyl chloride). Int. I Sci. Res. Publ. 2015, 5, 7. [Google Scholar]
- Chougale, R.B.; P Masti, S.P.; Kasai, D.R.; Mudigoudra, B.S. Influence of gum ghatti on morphological and mechanical properties of poly(vinyl alcohol)/poly(vinyl pyrrolidone) blend films. Pharma Chem. 2018, 10, 1–6. [Google Scholar]
- Gomaa, M.M.; El Fadly, E.; Salama, M.A.; Abdin, M. Production of bio composite films from gum Arabic and galangal extract to prolong the shelf life of Agaricus bisporus. J. Polym. Environ. 2022, 30, 4787–4799. [Google Scholar] [CrossRef]
- Ibrahim, M.; Krejčík, M.; Havlíček, K.; Petrík, S.; Eldessouki, M. Evaluation of chemical and physical properties of biodegradable gum Arabic/PVA/Ag nanofibrous membranes as a potential wrapping material. J. Eng. Fibers Fabr. 2020, 15, 1–9. [Google Scholar] [CrossRef]
- Jain, N.; Singh, V.K.; Chauhan, S. A review on mechanical and water absorption properties of polyvinyl alcohol based composites/films. J. Mech. Behav. Mater. 2017, 26, 213–222. [Google Scholar] [CrossRef]
- Schmidt, M.W.; Torn, M.S.; Abiven, S.; Dittmar, T.; Guggenberger, G.; Janssens, I.A.; Trumbore, S.E. Persistence of soil organic matter as an ecosystem property. Nature 2011, 478, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, I.G.E. Enzymatic Degradation and Analysis of Gum Arabic. Ph.D. Thesis, UOFK, Lexington, KY, USA, 2015. [Google Scholar]
- Freedman, Z.; Zak, D.R. Soil bacterial communities are shaped by temporal and environmental filtering: Evidence from a long-term chronosequence. Environ. Microbiol. 2015, 17, 3208–3218. [Google Scholar] [CrossRef]
- Turner, S.; Mikutta, R.; Meyer-Stüve, S.; Guggenberger, G.; Schaarschmidt, F.; Lazar, C.S.; Schippers, A. Microbial community dynamics in soil depth profiles over 120,000 years of ecosystem development. Front. Microbiol. 2017, 8, 874. [Google Scholar] [CrossRef]
- Adam, F.A.; Abdellah, A.M.; Abdel–Magid, H.M.; Osman, M.E.; Al-Aassaf, S.; Phillips, G.O. Effect of some isolated bacterial species on the physicochemical aspects and main components of gum arabic (Acacia senegal var. senegal). Int. J. Dev. Res. 2018, 8, 18436–18442. [Google Scholar]
- Wu, H.F.; Yue, L.Z.; Jiang, S.L.; Lu, Y.Q.; Wu, Y.X.; Wan, Z.Y. Biodegradation of polyvinyl alcohol by different dominant degrading bacterial strains in a baffled anaerobic bioreactor. Int. J. Dev. Res. 2019, 79, 2005–2012. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Chai, Y.N.; Lopes, L.D.; Ordóñez, R.A.; Wright, E.E.; Archontoulis, S.; Schachtman, D.P. The effects of soil depth on the structure of microbial communities in agricultural soils in Iowa, USA. Appl. Environ. Microbiol. 2021, 87, e02673-20. [Google Scholar] [CrossRef] [PubMed]
- Sichert, A.; Cordero, O.X. Polysaccharide–bacteria interactions from the lens of evolutionary ecology. Front. Microbiol. 2021, 12, 705082. [Google Scholar] [CrossRef]
- Polman, E.M.; Gruter, G.J.M.; Parsons, J.R.; Tietema, A. Comparison of the aerobic biodegradation of biopolymers and the corresponding bioplastics: A review. Sci. Total Environ. 2021, 753, 141953. [Google Scholar] [CrossRef]
- Villa-Rivera, M.G.; Cano–Camacho, H.; López-Romero, E.; Zavala-Páramo, M.G. The role of arabinogalactan type II degradation in plant–microbe interactions. Front. Microbiol. 2021, 12, 730543. [Google Scholar] [CrossRef]
- Sasaki, Y.; Uchimura, Y.; Kitahara, K.; Fujita, K. Characterization of a GH36 α-D-galactosidase associated with assimilation of gum Arabic in Bifidobacterium longum subsp. longum JCM7052. J. Appl. Glycosci. 2021, 68, 47–52. [Google Scholar] [CrossRef]
- Chien, H.-L.; Tsai, Y.-T.; Tseng, W.-S.; Wu, J.-A.; Kuo, S.-L.; Chang, S.-L.; Huang, S.-J.; Liu, C.-T. Biodegradation of PBSA films by Elite Aspergillus isolates and farmland soil. Polymer 2022, 14, 1320. [Google Scholar] [CrossRef]
- Sasaki, Y.; Komeno, M.; Ishiwata, A.; Horigome, A.; Odamaki, T.; Xiao, J.Z.; Fujita, K. Mechanism of cooperative degradation of gum arabic arabinogalactan protein by bifidobacterium longum surface enzymes. Appl. Environ. Microbiol. 2022, 88, e02187-21. [Google Scholar] [CrossRef]
- Santos-Beneit, F.; Chen, L.M.; Bordel, S.; Frutos de la Flor, R.; García-Depraect, O.; Lebrero, R.; Rodriguez–Vega, S.; Muñoz, R.; Börner, R.A.; Börner, T. Screening enzymes that can depolymerize commercial biodegradable polymers: Heterologous expression of Fusarium solani cutinase in Escherichia coli. Microorganisms 2023, 11, 328. [Google Scholar] [CrossRef]
- Tang, Y.; Zhou, D.; Zhang, J. Novel polyvinyl alcohol/styrene butadiene rubber latex/carboxymethyl cellulose nanocomposites reinforced with modified halloysite nanotubes. J. Nanomater. 2013, 2013, 1–8. [Google Scholar] [CrossRef]
- Chiellini, E.; Corti, A.; Solaro, R. Biodegradation of poly (vinyl alcohol) based blown films under different environmental conditions. Polym. Degrad. Stab. 1999, 64, 305–312. [Google Scholar] [CrossRef]
- Jayasekara, R.; Harding, I.; Bowater, I.; Christie, G.B.; Lonergan, G.T. Biodegradation by composting of surface modified starch and PVA blended films. J. Polym. Environ. 2003, 11, 49–56. [Google Scholar] [CrossRef]
- Matsumura, S.; Tanaka, T. Novel malonate-type copolymers containing vinyl alcohol blocks as biodegradable segments and their builder performance in detergent formulations. J. Appl. Polym. Sci. 1994, 2, 89–97. [Google Scholar] [CrossRef]
- Hindi, S.S.; Albureikan, M.; Othman, I.; Al-ghamdy, A.A.; Alhummiany, H.; Ansari, M.S. Synthesis and characterization of gum Arabic based bioplastic membranes. J. Nanosci. Nanotechnol. Res. 2017, 4, 32–42. [Google Scholar]
- Hindi, S.S.; Albureikan, M.O.I.; Attieh, A.; Al-ghamdy, A.A.; Alhummiany, H.; Al-Sharabi, S.M. Effect of potassium dichromate on properties and biodegradation of gum Arabic based bioplastic membranes. Nanosci. Nanotechnol. Res. 2017, 4, 49–58. [Google Scholar] [CrossRef]
- Hindi, S.S. Some crystallographic properties of cellulose I as affected by cellulosic resource, smoothing, and computation methods. Int. J. Innov. Res. Sci. 2017, 6, 732–752. [Google Scholar]
- Hindi, S.S. Suitability of date palm leaflets for sulphated cellulose nanocrystals synthesis. J. Nanosci. Nanotechnol. Res. 2017, 4, 7–16. [Google Scholar] [CrossRef]
- Hindi, S.S. Nanocrystalline cellulose: Synthesis from pruning waste of Zizyphus spina christi and characterization. J. Nanosci. Nanotechnol. 2017, 4, 106–114. [Google Scholar]
- Fortunati, E.; Puglia, D.; Monti, M.; Peponi, L.; Santulli, C.; Kenny, J.M.; Torre, L. Extraction of cellulose nanocrystals from Phormium tenax fibres. J. Polym. Environ. 2013, 21, 319–328. [Google Scholar] [CrossRef]
- Al-Solaimani, S.G. Chemical properties of soils and underground water of Hada Al-Sham Research Station, Kingdom of Saudi Arabia. J. Environ. Sci. Ain Shams Univ. 2003, 6, 257–284. [Google Scholar]
- Al-Solaimani, S.G.; Al-Toukhy, A.; Al-Zahrani, S. Mineral characteristics, classification and evaluation of soils of Hada Al-Sham Res. Station, Kingdom of Saudi Arabia. J. Environ. Sci. Ain Shams Univ. 2003, 6, 285–322. [Google Scholar]
- Mostafa, H.M.; Sourell, H.; Bockisch, F.J. Mechanical properties of some bioplastics under different soil types used as biodegradable drip tubes. Agric. Eng. Int. CIGR J. 2010, 12, 12–21. [Google Scholar]
- El-Nakhlawy, F.S. Principles of Statistics, Biostatistical Experimental Design and Analysis; KAU Pub. Center KSA: Jeddah, Saudi Arabia, 2008. [Google Scholar]
- Şişmanoğlu, T.; Karakuş, S.; Birer, Ö.; Soylu, G.S.P.; Kolan, A.; Tan, E.; Ürk, Ö.; Akdut, G.; Kilislioglu, A. Preparation and characterization of antibacterial Senegalia (acacia) Senegal/iron—Silica bio-nanocomposites. Appl. Surf. Sci. 2015, 354, 250–255. [Google Scholar] [CrossRef]
- Bouaziz, F.; Koubaa, M.; Barba, F.J.; Roohinejad, S.; Chaabouni, S.E. Antioxidant properties of water–soluble gum from flaxseed hulls. Antioxidants 2016, 5, 26. [Google Scholar] [CrossRef]
- Anicuta, S.-G.; Dobre, L.; Stroescu, M.; Jipa, I. Fourier transform infrared (FTIR) spectroscopy for characterization of antimicrobial films containing chitosan. Analele Univ. Din Oradea Fasc. Ecotoxicologie Zooteh. Si Tehnol. Ind. Aliment. 2010, 1234–1240. [Google Scholar]
- Rathna, G.V.N.; Jog, J.P.; Gaikwad, A.B. Development of non-woven nanofibers of egg albumen-poly (vinyl alcohol) blends: Influence of solution properties on morphology of nanofibers. Polym. J. 2011, 43, 654–661. [Google Scholar] [CrossRef]
- Mori, T.; Sakimoto, M.; Kagi, T.; Sakai, T. Isolation and characterization of a stra108–in of Bacillus megaterium that degrades poly (vinyl alcohol). Biosci. Biotechnol. Biochem. 1996, 60, 330–332. [Google Scholar] [CrossRef] [PubMed]
- Patil, R.; Bagde, U.S. Enrichment and isolation of microbial strains degrading bioplastic polyvinyl alcohol and time course study of their degradation potential. Afr. J. Biotechnol. 2015, 14, 2216–2226. [Google Scholar]
- Jecu, L.; Gheorghe, A.; Rosu, A.; Raut, I.; Grosu, E.; Ghiurea, M. Ability of fungal strains to degrade PVA based materials. J. Polym. Environ. 2010, 18, 284–290. [Google Scholar] [CrossRef]
- Kawai, F.; Hu, X. Biochemistry of microbial polyvinyl alcohol degradation. Appl. Microbiol. Biotechnol. 2009, 84, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Cadmus, M.C.; Jackson, L.K.; Burton, K.A.; Plattner, R.D.; Slodki, M.E. Biodegradation of xanthan gum by Bacillus sp. Appl. Environ. Microbiol. 1982, 44, 5–11. [Google Scholar] [CrossRef]
- Hossain, I.; Sarafat, A. Fungi grow faster than Gram-negative bacteria in Emb media and also deterring the growth of these bacteria by reducing the pH of that media. J. Pharmacogenomics 2020, 12, 1–10. [Google Scholar]
- Johannes, R.; Bååth, E. Growth of saprotrophic fungi and bacteria in soil. FEMS Microbiol. Ecol. 2011, 78, 17–30. [Google Scholar]
- Busoloa, T.; Urab, D.P.; Kima, S.K.; Marzecc, M.M.; Bernasik, A.; Stachewicz, U.; Kar-Narayan, S. Surface potential tailoring of PMMA fibers by electrospinning for enhanced triboelectric performance. Nano Energy 2019, 57, 500–506. [Google Scholar] [CrossRef]
- Anonymous. National Center for Biotechnology Information. PubChem Compound Summary for CID 11199, Polyvinyl Alcohol. 2023. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Vinyl–alcohol (accessed on 11 March 2023).
- Anonymous. National Center for Biotechnology Information. PubChem Substance Record for SID 50019090, 2-Methyl-2-Propenoic Acid Methyl Ester Homopolymer, Source: LeadScope. 2023. Available online: https://pubchem.ncbi.nlm.nih.gov/substance/50019090 (accessed on 12 March 2023).
- Anonymous. PubChem, Compound Summary for CID 24847856, Galactoarabinan. 2023. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Galactoarabinan (accessed on 8 March 2023).
- Anonymous. National Center for Biotechnology Information. PubChem, compound Summary for CID 439212, Glycoprotein. 2023. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Glycoprotein (accessed on 13 March 2023).
- Anonymous. National Center for Biotechnology Information. PubChem, Substance Record for SID 405234199, a Plant Arabinogalactan-[Protein], Source: BioCyc. 2023. Available online: https://pubchem.ncbi.nlm.nih.gov/substance/405234199 (accessed on 22 March 2023).
- Schmidt, D.L.; Coburn, C.E.; DeKoven, B.M.; Potter, G.E.; Meyers, G.F.; Fischer, D.A. Water-based non-stick hydrophobic coatings. Nature 1994, 368, 39–41. [Google Scholar] [CrossRef]
- Hild, F. Surface Energy of Plastics. 2009. Available online: https://www.tstar.com/blog/bid/33845/surface-energy-of-plastics (accessed on 22 March 2023).
- Zi, Y.; Wang, Z.L. Nanogenerators: An emerging technology towards nanoenergy. APL Mater. 2017, 5, 074103. [Google Scholar] [CrossRef]
- Randall, R.C.; Phillips, G.O.; Williams, P.A. The role of the proteinaceous component on the emulsifying properties of gum arabic. Food Hydrocoll. 1988, 2, 131–140. [Google Scholar] [CrossRef]
- Randall, R.C.; Phillips, G.O.; Williams, P.A. Fractionation and characterization of gum from Acacia senegal. Food Hydrocoll. 1989, 3, 65–75. [Google Scholar] [CrossRef]
- Azzaoui, K.; Hammouti, B.; Lamhamdi, A.; Mejdoubi, E.; Berrabah, M. The gum Arabic in the southern region of Morocco. Mor. J. Chem. 2014, 3, 99–107. [Google Scholar]
- Elsabee, M.Z.; Naguib, H.F.; Morsi, R.E. Chitosan based nanofibers, review. Mater. Sci. Eng. C 2012, 32, 1711–1726. [Google Scholar] [CrossRef] [PubMed]
- Pakravan, M.; Heuzey, M.C.; Ajji, A. A fundamental study of chitosan/PEO electrospinning. Polym. J. 2011, 52, 4813–4824. [Google Scholar] [CrossRef]
- Wiśniewska, M.; Bogatyrov, V.; Ostolska, I.; Szewczuk-Karpisz, K.; Terpiłowski, K.; Nosal-Wiercińska, A. Impact of poly(vinyl alcohol) adsorption on the surface characteristics of mixed oxide MnxOy–SiO2. Adsorption 2016, 22, 417–423. [Google Scholar] [CrossRef]
- Chibowski, S.; Paszkiewicz, M.; Krupa, M. Investigation of the influence of the polyvinyl alcohol adsorption on the electrical properties of Al2O3–solution interface, thickness of the adsorption layers of PVA. Powder Technol. 2000, 107, 251–255. [Google Scholar] [CrossRef]
- Nuraje, N.; Khan, W.S.; Lei, Y.; Ceylan, M.; Asmatulu, R. Superhydrophobic electrospum nanofibers. J. Mater. Chem. A.1 2013, 1, 1929–1946. [Google Scholar] [CrossRef]
- Osti, G.B.F.; Wolf, F.G.; Philippi, P.C. Spreading of liquid drops on acrylic surfaces. In Proceedings of the 20th International Congress of Mechanical Engineering, Gramado, Brazil, 15–20 November 2009. [Google Scholar]
- Johnson, R.E.; Dettre, R.H. Wetting of Low-Energy Surfaces; Marcel Dekker: New York, NY, USA, 1993. [Google Scholar]
- Pavluchkov, V.; Shefer, I.; Peer-Haim, O.; Blotevogel, J.; Epsztein, R. Indications of ion dehydration in diffusion-only and pressure-driven nanofiltration. J. Membr. Sci. 2022, 648, 120358. [Google Scholar] [CrossRef]
- Walker, J.A. Physics for Introductory Biology. 2020. Available online: https://www.middleprofessor.com/files/physics_for_biologists/_book/index.html (accessed on 11 July 2023).
- Ibekwe, C.A.; Oyatogun, G.M.; Esan, T.A.; Oluwasegun, K.M. Synthesis and characterization of chitosan/gum Arabic nanoparticles for bone regeneration. Am. J. Mater. Sci. Eng. 2017, 5, 28–36. [Google Scholar]
- Mir, M.B.; Haripriya, S. Assessment of physical and structural characteristics of almond gum. Int. J. Biol. Macromol. 2016, 93, 476–482. [Google Scholar]
- Stuart, B.H. Infrared Spectroscopy: Fundamentals and Applications, 1st ed.; John Wiley & Sons Ltd.: West Sussex, UK, 2004. [Google Scholar]
- Kumar, A.; Negi, Y.S.; Choudhary, V.; Bhardwaj, N.K. Characterization of cellulose nanocrystals produced by acid-hydrolysis from sugarcane bagasse as agro-waste. Mater. Chem. Phys. 2014, 2, 1–8. [Google Scholar] [CrossRef]
- Yoshimi, Y.; Yaguchi, K.; Kaneko, S.; Tsumuraya, Y.; Kotake, T. Properties of two fungal endo-beta-1,3-galactanases and their synergistic action with an exo–beta–1,3–galactanase in degrading arabinogalactan-proteins. Carbohydr. Res. 2017, 45, 26–35. [Google Scholar] [CrossRef]
- Yoshimi, Y.; Hara, K.; Yoshimura, M.; Tanaka, N.; Higaki, T.; Tsumuraya, Y.; Kotake, T. Expression of a fungal exo-beta-1,3-galactanase in Arabidopsis reveals a role of type II arabinogalactans in the regulation of cell shape. J. Exp. Bot. 2020, 71, 5414–5424. [Google Scholar] [CrossRef]
- Gao, X.; Fu, C.; Li, M.; Qi, X.; Jia, X. Effects of biodegradation of corn-starch-sodium alginate–biobased liquid mulch film on soil microbial functions. Int. J. Environ. Res. Public Health 2022, 19, 8631. [Google Scholar] [CrossRef]
- Augustine, R.; Kalarikkal, N.; Thomas, S. Effect of zinc oxide nanoparticles on the invitro degradation of electrospun polycaprolactone membranes in simulated body fluid. Int. J. Polym. Mater. Polym. Biomater. 2016, 65, 28–37. [Google Scholar] [CrossRef]



| Equation | Definitions |
|---|---|
| 1 CI = (App/At) × 100 | App: Total planar area (mm2) of the XRD’s diffractogram. |
| At: The planar area (mm2) of the principle peaks arises around 2θ° of 20°. | |
| 2 ML = [(W2 − W1)/W1] × 100 | Wi: Initial weight of the bioplastic sample at a temperature zone, estimated from the TGA curves. Wf: Final weight of the bioplastic sample at the same temperature zone, estimated from the TGA curves. |
| 3 TR = Ti − Tf | Ti: Initial temperature of a certain thermogram. Tf: Final temperature of the same thermogram. |
| 4 EC = Ef − Ei | Ef: Initial enthalpy of a certain thermogram. Ei: Final enthalpy of the same thermogram. |
| 5 TS = Ff/A 6 MoE = σ/ε 7 ε = [∆L/Lo] = [(Lf − L○)/L○] 8 EaF = ∆Lf = [(Lf − L○)/L○] × 100 | Ff: Force at failure in Newton (N). |
| A: Cross-section area (m2) of the bioplastic sample. | |
| σ: Tensile stress (Pa). | |
| Lf: The length of the bioplastic sample at failure. | |
| L○: The initial length of the bioplastic sample at failure. | |
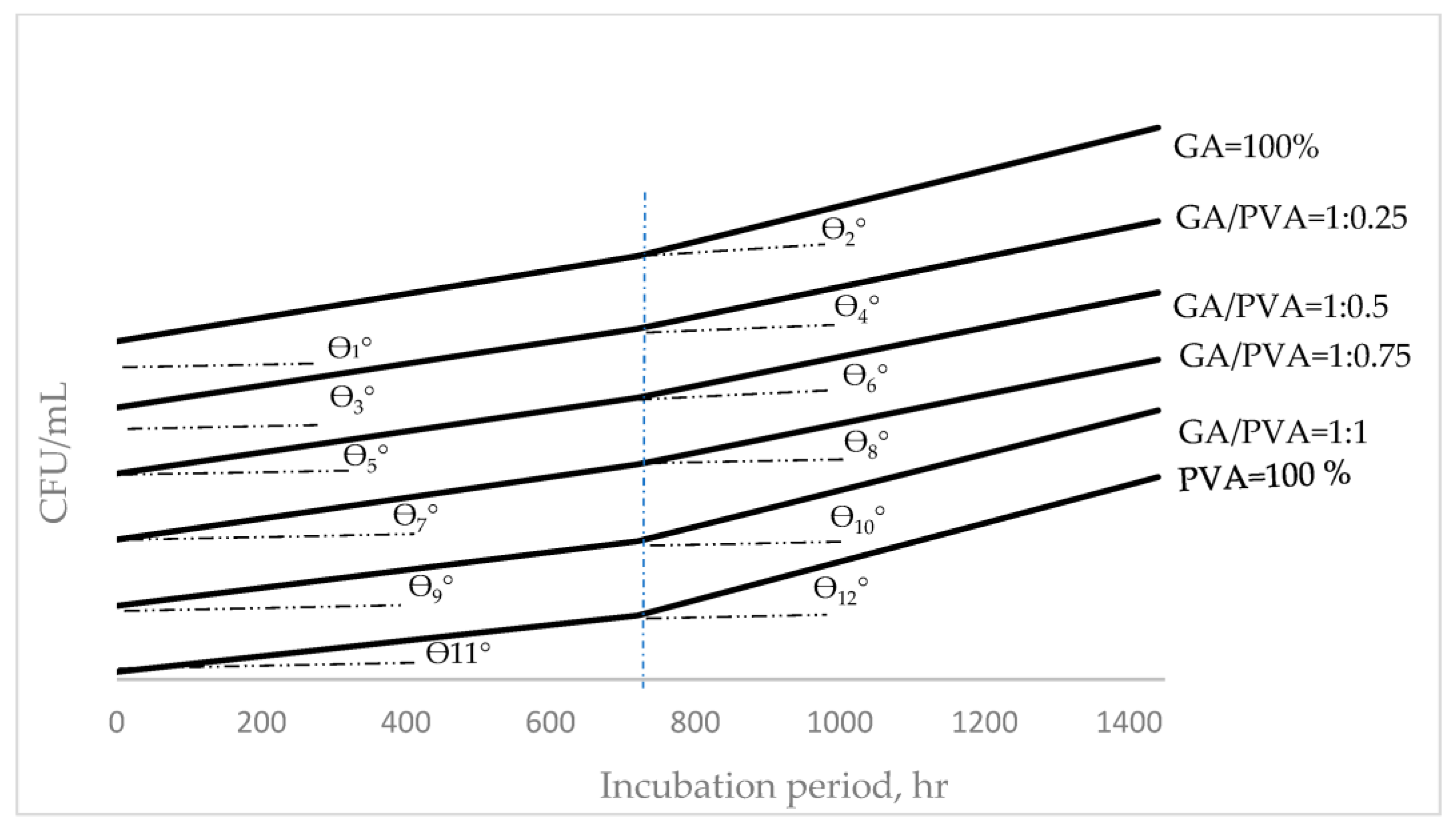
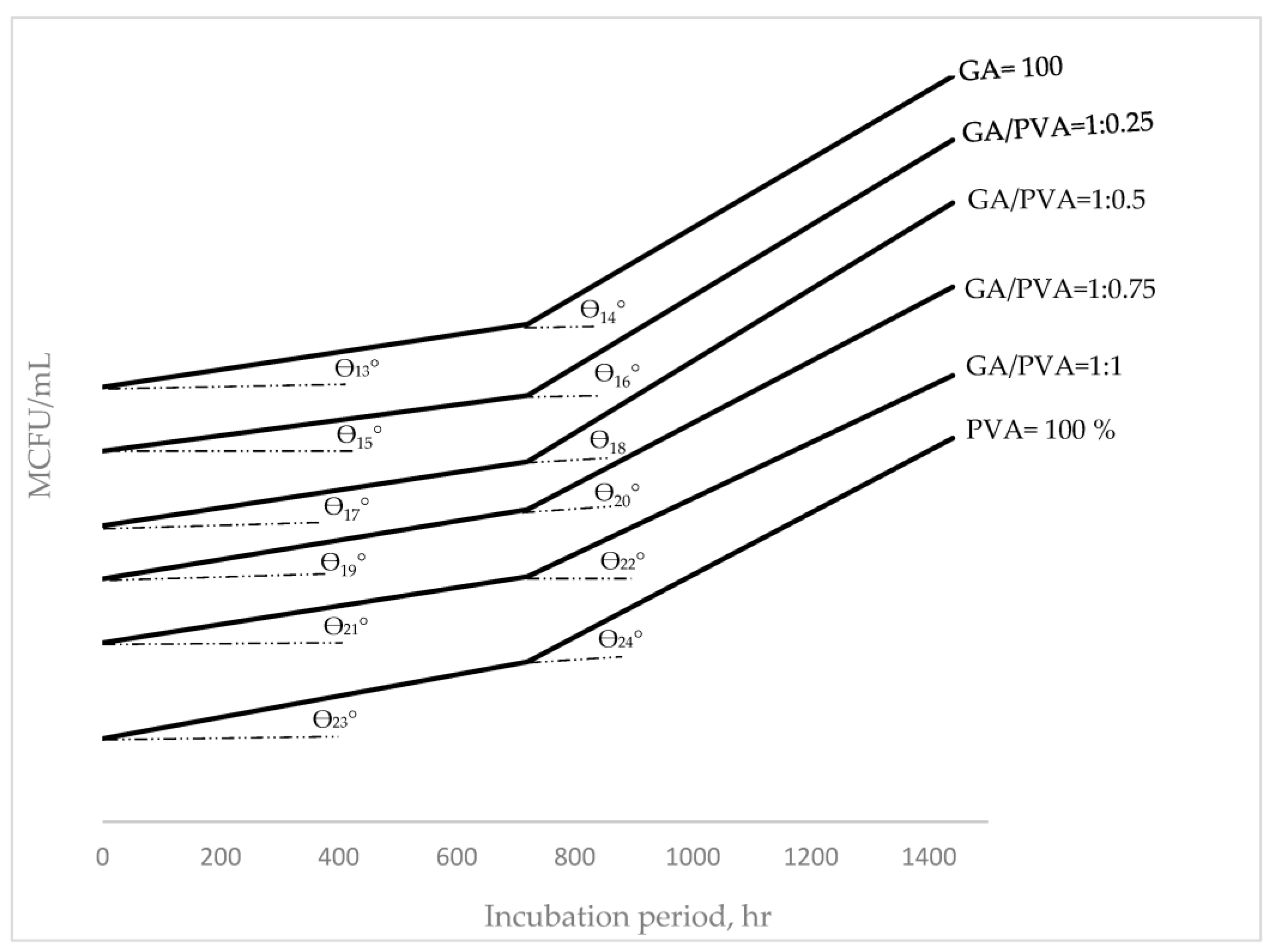
| 9 Tan (θn°) = (y2 − y1)/(x2 − x1) y = mx + C m = Tan (θn°) | θ°: The incline angle (θ) in degrees of the hourly duplication (HD) curve for microbial populations. x1: The 1st incubation period (hr.) for the HD curve. x2: The 2nd incubation period (hr.) for the HD curve. y1: The initial HD of the colony-forming units (CFUs) in mega units (MCFU). y2: The final HD of the colony-forming units (CFUs) in mega units (MCFU). m: The slope of the curve C: The intersecting section of the y-coordinate for the HD’s curve. |
| T-Zones °C | GA/PVA Ratio | |||||
|---|---|---|---|---|---|---|
| GA 100% | 1:0.25 | 1:0.5 | 1:0.75 | 1:1 | PVA 100% | |
| 50°–100° | 13.68 Ad | 4.57 Bg | 4.85 Be | 5.02 Bef | 5.23 Bef | 5.69 Bf |
| 100°–150° | 11.11 Ae | 6.32 ABf | 6.37 ABe | 6.93 ABe | 6.47 ABef | 8.98 Bef |
| 150°–200° | 4.18 BCg | 4.88 BCg | 6.46 Be | 4.26 BCef | 3.23 Cf | 8.07 Aef |
| 00°–250° | 6.96 Bf | 8.18 Ae | 6.18 Be | 3.7 Cf | 5.71 Bef | 3.9 Cg |
| 250°–300° | 7.48 Cf | 14.9 Ad | 7.36 Ce | 10.77 ABd | 11.62 Bd | 10.15 ABe |
| 300°–350° | 22.2 Bb | 12.62 Ede | 15.93 Dd | 22.41 Bc | 28.57 Ac | 18.08 Cd |
| 350°–400° | 18.2 Eb | 21.13 Db | 26.32 Cb | 33.3 Bb | 34.4 Bb | 44.14 Ac |
| 400°–450° | 28.6 Da | 27.68 Da | 41.43 Ca | 39.17 BCa | 46.34 Ba | 79.0 Aa |
| 450°–500° | 15.6 Ec | 18.52 Dc | 21.95 CDc | 24.66 Cc | 45.5 Ba | 58.8 Ab |
| Points of Reaction | GA/PVA Ratio | Thermogram Type | TR °C | EC µVs/mg |
|---|---|---|---|---|
| a | GA = 100% | Endotherm Exotherm | 25–265 265–435 | −1017.25 +52.39 |
| b | 1:0.25 | Endotherm | 25–475 | −2268.77 |
| c | 1.0.5 | Endotherm Exotherm | 25–397 397–480 | −1127.7 −16.67 |
| d | 1:0.75 | Endotherm Exotherm | 25–375 375–475 | −1276.04 −20.89 |
| e | 1:1 | Endotherm | 25–475 | −1467.19 |
| f | PVA = 100% | Endotherm | 25–475 | −2119.72 |
| GA/PVA Ratio | GA Amount % | PVA Amount % | SPs | Particle Size nm | Permeability | ||||
|---|---|---|---|---|---|---|---|---|---|
| Pore Diameter nm | Void Volume nm3 | ||||||||
| ADB | NDB | ADB | NDB | ADB | NDB | ||||
| 1/0 | 0 | 100 | Mean 1,2 | 13.57 | 14.77 | 0.91 | 0.953 | 83.24 | 84.29 |
| Max. 3 | 55.44 | 56.68 | 3.905 | 3.948 | 1397.9 | 1398.91 | |||
| Min. 4 | 4.24 | 5.49 | 0.002 | 0.045 | 0.007 | 1.057 | |||
| SD 5 | 7.66 | 7.66 | 0.904 | 0.904 | 160.68 | 160.68 | |||
| 1:0.25 | 20 | 80 | Mean 1,2 | 14.17 | 15.42 | 0.553 | 0.606 | 105.74 | 106.74 |
| Max. 2,3 | 76.94 | 78.19 | 3.54 | 3.593 | 1374.8 | 1375.47 | |||
| Min. 2,4 | 4.24 | 5.49 | 0.001 | 0.055 | 0.005 | 1.008 | |||
| SD | 8.93 | 8.93 | 0.457 | 0.457 | 156.17 | 156.17 | |||
| 1:0.5 | 66.7 | 33.3 | Mean 1,2 | 15.15 | 16.4 | 0.608 | 0.671 | 120.66 | 121.87 |
| Max. 2,3 | 67.01 | 68.26 | 2.38 | 2.443 | 8009 | 8010.22 | |||
| Min. 2,4 | 4.24 | 5.49 | 0.001 | 0.064 | 0.002 | 1.219 | |||
| SD 5 | 8.51 | 8.51 | 0.469 | 0.469 | 309.6 | 309.6 | |||
| 1:0.75 | 57.1 | 42.9 | Mean 1,2 | 17.01 | 18.07 | 0.714 | 0.788 | 226.98 | 228.18 |
| Max. 2,3 | 72.32 | 73.38 | 3.608 | 3.683 | 8411.8 | 8412.98 | |||
| Min. 2,4 | 4.24 | 5.3 | 0.007 | 0.082 | 0.007 | 1.215 | |||
| SD 5 | 9.26 | 9.26 | 0.615 | 0.615 | 631.41 | 631.41 | |||
| 1:1 | 50 | 50 | Mean 1,2 | 18.42 | 19.58 | 1.145 | 1.23 | 460.18 | 461.5 |
| Max. 2,3 | 89.75 | 90.91 | 4.75 | 4.839 | 8411.8 | 8413.1 | |||
| Min. 2,4 | 4.24 | 5.4 | 0.019 | 0.093 | 0.007 | 1.34 | |||
| SD 5 | 12.69 | 12.69 | 2.342 | 1.002 | 1062.04 | 1062.04 | |||
| 0/1 | 100 | 0 | Mean 1,2 | 20.34 | 21.35 | 1.485 | 1.58 | 548.95 | 552.41 |
| Max. 2,3 | 89.75 | 90.76 | 14.851 | 14.946 | 9315 | 9318.46 | |||
| Min. 2,4 | 4.24 | 5.25 | 0.019 | 0.114 | 0.001 | 3.46 | |||
| SD 5 | 14.58 | 14.58 | 2.342 | 2.342 | 1198.36 | 1198.36 | |||
| GA/PVA Ratio | GA Amount % | PVA Amount % | Stress Type | ADB | NDB | ||
|---|---|---|---|---|---|---|---|
| Stress MPa | Strain | Stress MPa | Strain | ||||
| 1/0 | 0 | 100 | PL | 8.1 | 29.69 | 11.34 | 37.42 |
| US | 20.42 | 145.24 | 21.42 | 145.25 | |||
| 1:0.25 | 20 | 80 | PL | 14.24 | 41.22 | 17.61 | 39.73 |
| US | 28.91 | 319.13 | 31.05 | 300.9 | |||
| 1:0.5 | 66.7 | 33.3 | PL | 8.92 | 46.47 | 9.56 | 50.56 |
| US | 18.84 | 249.5 | 20.64 | 264.98 | |||
| 1:0.75 | 57.1 | 42.9 | PL | 7.15 | 48.4 | 10.04 | 56.3 |
| US | 15.92 | 269.39 | 19.7 | 284.39 | |||
| 1:1 | 50 | 50 | PL | 4.92 | 38.41 | 4.92 | 38.41 |
| US | 11.86 | 297.99 | 12.33 | 310.55 | |||
| 0/1 | 100 | 0 | PL | 12.03 | 65.26 | 15.3 | 72.61 |
| US | 21.64 | 227.09 | 25.47 | 264.91 | |||
| AG/PVA Ratio | After 30 Days | After 60 Days | ||
|---|---|---|---|---|
| Bacteria CFU/mL | Fungi CFU/mL | Bacteria CFU/mL | Fungi CFU/mL | |
| GA = 100% | 2.8 × 106 1 [0.032] | 1.77 × 103 [0.008] | 6.69 × 106 [0.086] | 4.32 × 103 [0.077] |
| 1:0.25 | 2.6 × 106 [0.07] | 1.8 × 103 [0.042] | 5.86 × 106 [0.074] | 3.8 × 103 [0.093] |
| 1:0.5 | 2.52 × 106 [0.028] | 1.88 × 103 [0.094] | 5.7 × 106 [0.064] | 4.21 × 103 [0.086] |
| 1:0.75 | 2.5 × 106 [0.031] | 1.93 × 103 [0.095] | 5.67 × 106 [0.095] | 4.02 × 103 [0.086] |
| 1:1 | 2.17 × 106 [0.088] | 1.9 × 103 [0.012] | 6.14 × 106 [0.088] | 3.79 × 103 [0.044] |
| PVA = 100% | 1.93 × 106 [0.008] | 2.1 × 103 [0.083] | 6.12 × 106 [0.093] | 4.83 × 103 [0.046] |
| Soil control sample | Bacteria: 2.28 × 105 CFU/mL [0.058] | |||
| Fungi: 1.28 × 103 CFU/mL [0.022] | ||||
| Microbial Type | AG/PVA Ratio | HD-Equation | |||
|---|---|---|---|---|---|
| 1 IA | The 1st Stage (0–720 h) | IAS | The 2nd Stage (720–1400 h) | ||
| Bacteria | GA = 100% | θ1° | y = 3572.2x + 228 × 103 | θ2° | y = 8975x + 228 × 103 |
| 1:0.25 | θ3° | y = 3294.4x + 228 × 103 | θ4° | y = 7822.2x + 228 × 103 | |
| 1:0.5 | θ5° | y = 3183.3x + 228 × 103 | θ6° | y = 7600x + 228 × 103 | |
| 1:0.75 | θ7° | y = 3155.56x + 228 × 103 | θ8° | y = 7558.3x + 228 × 103 | |
| 1:1 | θ9° | y = 2697.2x + 228 × 103 | θ10° | y = 7100x + 228 × 103 | |
| PVA = 100% | θ11° | y = 2363.89x + 228 × 103 | θ12° | y = 6794.4x + 228 × 103 | |
| Fungi | GA = 100% | θ13° | y = 0.8194x + 1.28 × 103 | θ14° | y = 3.2361x + 1.87 × 103 |
| 1:0.25 | θ15° | y = 0.7222x + 1.28 × 103 | θ16° | y = 3.3333x + 1.8 × 103 | |
| 1:0.5 | θ17° | y = 0.8333x + 1.28 × 103 | θ18° | y = 3.375x + 1.88 × 103 | |
| 1:0.75 | θ19° | y = 0.9028x + 1.28 × 103 | θ20° | y = 2.90278x + 1.93 × 103 | |
| 1:1 | θ21° | y = 0.8611x + 1.48 × 103 | θ22° | y = 2.625x + 2.1 × 103 | |
| PVA = 100% | θ23° | y = x + 1.78 × 103 | θ24° | y = 2.9167x + 2.5 × 103 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hindi, S.S.; Albureikan, M.O.I. Fabrication, Characterization, and Microbial Biodegradation of Transparent Nanodehydrated Bioplastic (NDB) Membranes Using Novel Casting, Dehydration, and Peeling Techniques. Polymers 2023, 15, 3303. https://doi.org/10.3390/polym15153303
Hindi SS, Albureikan MOI. Fabrication, Characterization, and Microbial Biodegradation of Transparent Nanodehydrated Bioplastic (NDB) Membranes Using Novel Casting, Dehydration, and Peeling Techniques. Polymers. 2023; 15(15):3303. https://doi.org/10.3390/polym15153303
Chicago/Turabian StyleHindi, Sherif S., and Mona Othman I. Albureikan. 2023. "Fabrication, Characterization, and Microbial Biodegradation of Transparent Nanodehydrated Bioplastic (NDB) Membranes Using Novel Casting, Dehydration, and Peeling Techniques" Polymers 15, no. 15: 3303. https://doi.org/10.3390/polym15153303
APA StyleHindi, S. S., & Albureikan, M. O. I. (2023). Fabrication, Characterization, and Microbial Biodegradation of Transparent Nanodehydrated Bioplastic (NDB) Membranes Using Novel Casting, Dehydration, and Peeling Techniques. Polymers, 15(15), 3303. https://doi.org/10.3390/polym15153303







