Novel Fucoidan Pharmaceutical Formulations and Their Potential Application in Oncology—A Review
Abstract
:1. Introduction
2. Materials and Methods
3. Fucoidan Sources and Chemical Structure
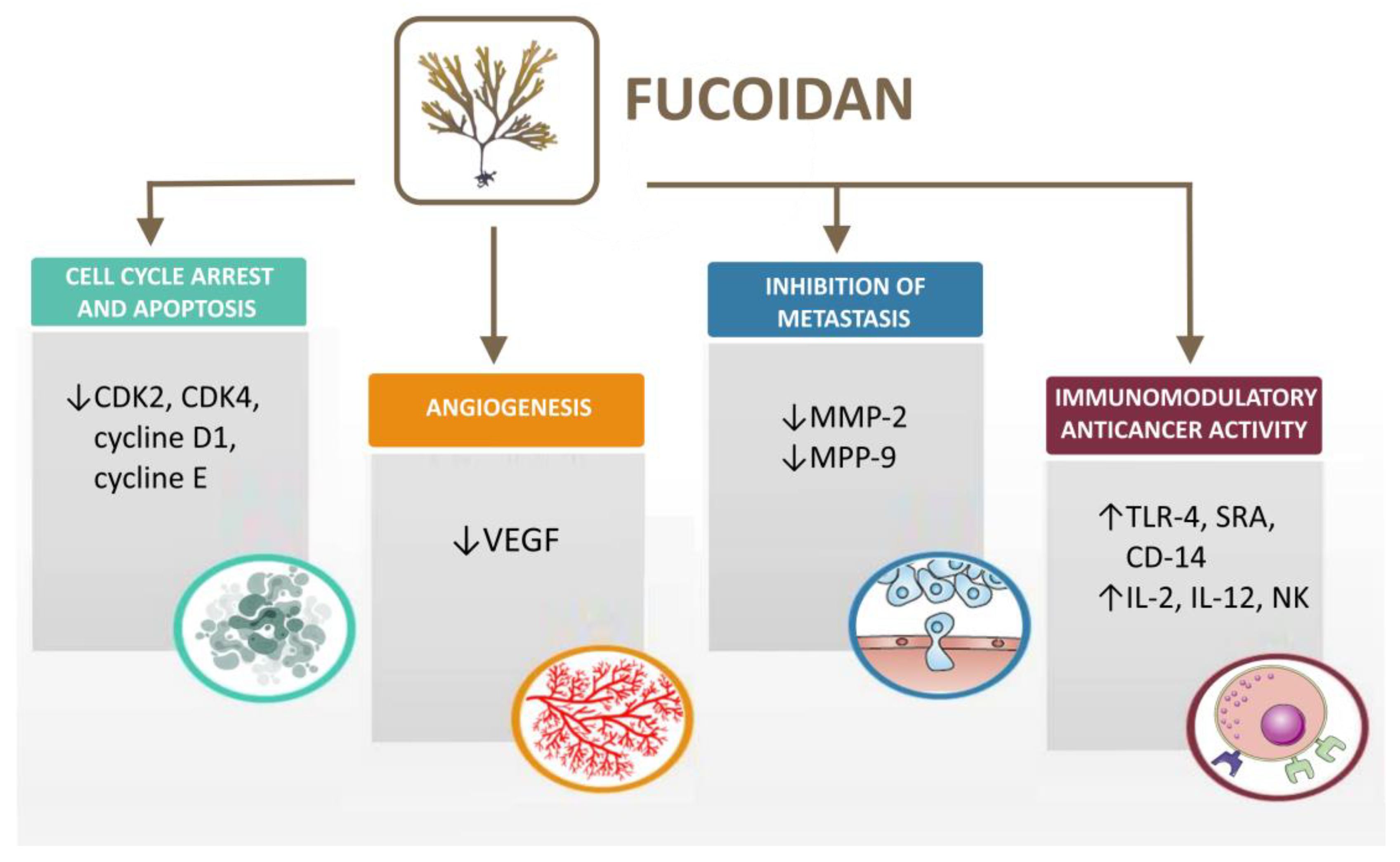
4. Fucoidan Biological Activities and Anticancer Mechanisms
4.1. Cell Cycle Arrest and Apoptosis
4.2. Fucoidan Effects on Angiogenesis
4.3. Inhibition of Cell Migration and Metastasis
4.4. Immunomodulatory Anticancer Activity
| Fucoidan Source | Dose µg/mL | Cancer Type/ Cell Line | Data Obtained | Reference |
|---|---|---|---|---|
| Saccharina | 50 | Colon cancer, DLD-1; Brest cancer, T-47D | Cell proliferation inhibition; Inhibition of EGF receptor binding. | [124] |
| Sargassum | 200 | Hepatic carcinoma, Huh6, Huh7, SK-Hep1 and HepG2 | Cell proliferation inhibition via TGF-β R1, 2 ↓ Phospho-Smad2/3↓ Smad 4 protein ↓ | [37] |
| Sargassum | 100 | Colon cancer, DLD-1 | Cell proliferation inhibition; Inhibition of the formation of cytotoxic colony. | [125] |
| F. vesiculosus | 20 | Colon cancer, HT-29 HCT-116 | Induced apoptosis via caspase-8, 9, 7, 3 activation PARP, Bak, Bid, Fas ↑, survivin, XIAP ↓. | [126] |
| F. vesiculosus | 1000 | Colon cancer, HT-29 | Cell proliferation inhibition and induced cell apoptosis (↓Ras/Raf/ERK proteins). | [127] |
| F. vesiculosus | 300 | Brest cancer, MCF-7 | Induced cell apoptosis via Caspase-8 activation Cytochrome C, Bax ↑ Bcl-2 ↓ Release of APAf-1 ↑. | [32] |
| F. vesiculosus | 90–120 | Brest cancer, MDA-MB-231 | Cell proliferation inhibition via expression of phosphorylated Smad2,3 and Smad4 inhibition. | [128] |
| F. vesiculosus | 50–400 | Lung cancer, Lewis lung carcinoma cells | Inhibition of metastasis via inhibition of VEGF and MMPs. | [129] |
| F. vesiculosus | 400 | Lung cancer, A549 | Cell proliferation inhibition and induced cell apoptosis via Caspase-3 ↑ and PARP cleavage. | [130] |
| F. vesiculosus | 1000 | Hepatic carcinoma, Huh-7, SNU-761, SNU-3085 | Cell proliferation inhibition via Caspase-7, -8, -9 ↑. | [131] |
| F. vesiculosus | 100–1000 | Hepatic carcinoma, Huh-BAT, Huh-7, SNU-761 | Induced cell apoptosis via Bax, Bid, Fas ↑ Caspase-7, -8, -9 cleavage Phosphorylatedp42/44↑. | [132] |
| F. vesiculosus | 150 | Leucemia, HL-60 NB4 THP-1 | Induced cell apoptosis via PARP cleavage Caspase-8, 9, 3 ↑ Mcl-1, Bid ↓. | [133] |
| F. vesiculosus | 50–200 | Leucemia, SUDHL-4, OCI-LY8, NU-DUL-1, TMD8, U293, DB | Induced cell apoptosis via PARP cleavage and cleaved caspase-8,9, 3 ↑. | [134] |
| F. vesiculosus | 12.5–100 | Leucemia, NB4, HL60 | Cell proliferation inhibition and induced cell apoptosis via Caspase-3, 8, 9, ↑ PARP cleavage and Bax ↑. | [135] |
| F. vesiculosus | 20–100 | Leucemia, U937 | Cell proliferation inhibition and induced cell apoptosis via Caspase-3, 8, 9 ↑, PARP cleavage, Bax↑. | [136] |
| U. pinnatifida | 200–1000 | Colon cancer, WiDr LoVo Brest cancer, MCF-7 | Cell proliferation inhibition. | [108] |
| U. pinnatifida | 10–200 | Lung cancer, A549 | Cell proliferation inhibition and induced cell apoptosis via Bcl 2, p38, Phospho-PI3K/Akt, procaspase- 3↓ Bax, caspase-9, Phospho-ERK1/2 ↑ PARP cleavage. | [137] |
| U. pinnatifida | 65.2–1000 | Hepatic carcinoma, SMMC-7721 | Cell proliferation inhibition and induced cell apoptosis via Livin, XIAP mRNA ↓ Caspase-3, -8, -9 ↑ Bax-to-Bcl-2 ratio↑ Cytochrome C ↑. | [138] |
| Cladosiphon | 1000 | Brest cancer, MCF-7 | Induced cell apoptosis via PARP cleavage Caspase-7,8,9 ↑ Cytochrome C, Bax, Bid↑. | [139] |
| B. bifurcata | 2–9 | Lung cancer, NSCLC-N6 | Cell proliferation inhibition via irreversible growth arrest. | [119] |
5. Fucoidan-Based Pharmaceutical Formulations
5.1. Fucoidan Microparticles
5.2. Fucoidan Nanoparticles
5.3. Fucoidan Liposomes
| Active Substance | Dosage Form | Therapeutic Application | Data Obtained | Reference |
|---|---|---|---|---|
| Fucoidan | Microparticles | Skin burns | Enhanced skin regeneration resulted in an increase in the epithelial thickness in vivo on rabbits. | [155] |
| Perfluorooctyl bromide | Microparticles | Thrombosis | High affinity to P-selectin and ability to detect thrombosis in vivo on rats with induced aortic aneurysm. | [156] |
| Isoniazid, Rifabutin | Microparticles | Tuberculosis | Effective inhibition of mycobacterial growth. Low toxicity on human alveolar epithelum and monocytic cell line (HTP-1). | [159] |
| Doxorubicin | Microparticles | Breast cancer | Significant antiproliferative activity on MCF-7 breast cancer cell line. Low toxicity on mouse myoblasts (concentration 400 µg/mL). | [162] |
| N-methyl anthranilic acid | Nanoparticles | Inflammation | Significant reduction in carrageenan-induced inflammation in rats by decreasing the cyclooxygenase-2, (TNF)-α E2, NO, IL-1β, IL-6. | [163] |
| Docetaxel | Nanoparticles | Anticancer | High cellular uptake efficiency and subcellular localization. Low cytotoxicity on non-tumor cell lines. | [164] |
| Oversulfated fucoidan | Nanoparticles | Anticancer | Inhibition of human umbilical vein endothelial cells HUVECs via competitive binding to bFGF receptors (bFGFRs). | [168] |
| Amoxicillin | Nanogels | H. pylori infection | pH-dependent sustained drug release, which can improve the therapy of Helicobacter pylori. | [169] |
| Piperlongumine | Nanoparticles | Prostate cancer | Higher cytotoxicity in the PC-3 cells than in the non-cancerous hDFB cells; increased the intracellular ROS levels in the PC-3 cells. | [176] |
| Usnic acid | Liposomes | M. tuberculosis | Higher cellular uptake and cellular internalization. | [174] |
6. Fucoidan Nanoparticles for Cancer Therapy
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cormode, D.P.; Jarzyna, P.A.; Mulder, W.J.M.; Fayad, Z.A. Modified natural nanoparticles as contrast agents for medical imaging. Adv. Drug Delivery. Rev. 2010, 62, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Fernando, I.P.S.; Kim, D.; Nah, J.W.; Jeon, Y.J. Advances in functionalizing fucoidansand alginates (bio)polymers by structural modifications: A review. Chem. Eng. J. 2019, 355, 33–48. [Google Scholar] [CrossRef]
- Morya, V.; Kim, J.; Kim, E.K. Algal fucoidan: Structural and size-dependent bioactivities and their perspectives. Appl. Microbiol. Biotechnol. 2012, 93, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, W.A.J.P.; Jeon, Y.J. Biological activities and potential industrial applications of fucose 2rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Citkowska, A.; Szekalska, M.; Winnicka, K. Possibilities of Fucoidan Utilization in the Development of Pharmaceutical Dosage Forms. Mar. Drugs 2019, 17, 458. [Google Scholar] [CrossRef]
- Chollet, L.; Saboural, P.; Chauvierre, C.; Villemin, J.N.; Letourneur, D.; Chaubet, F. Fucoidans in Nanomedicine. Mar. Drugs 2016, 14, 145. [Google Scholar] [CrossRef]
- Hentati, F.; Tounsi, L.; Djomdi, D.; Pierre, G.; Delattre, C.; Ursu, A.V.; Fendri, I.; Abdelkafi, S.; Michaud, P. Bioactive Polysaccharides from Seaweeds. Molecules 2020, 25, 3152. [Google Scholar] [CrossRef]
- Bilan, M.I.; Grachev, A.A.; Ustuzhanina, N.E.; Shashkov, A.S.; Nifantiev, N.E.; Usov, A.I. A highly regular fraction of a fucoidan from the brown seaweed Fucus distichus L. Carbohydr. Res. 2004, 339, 511–517. [Google Scholar] [CrossRef]
- Vishchuk, O.S.; Ermakova, S.P.; Zvyagintseva, T.N. Sulfated polysaccharides from brown seaweeds Saccharina japonica and Undaria pinnatifida: Isolation, structural characteristics, and antitumor activity. Carbohydr. Res. 2011, 346, 2769–2776. [Google Scholar] [CrossRef]
- Chevolot, L.; Mulloy, B.; Ratiskol, J.; Foucault, A.; Colliec-Jouault, S. A disaccharide repeat unit is the major structure in fucoidans from two species of brown algae. Carbohydr. Res. 2001, 330, 529–535. [Google Scholar] [CrossRef]
- Zayed, A.; El-Aasr, M.; Ibrahim, A.S.; Ulber, R. Fucoidan Characterization: Determination of Purity and Physicochemical and Chemical Properties. Mar. Drugs 2020, 18, 571. [Google Scholar] [CrossRef] [PubMed]
- Luthuli, S.; Siya, W.; Cheng, Y.; Zheng, X.; Wu, M.; Tong, H. Therapeutic Effects of Fucoidan: A Review on Recent Studies. Mar. Drugs 2019, 17, 487. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, Q.; Zhang, Z.; Li, Z. Antioxidant activity of sulfated polysaccharide fractions extracted from Laminaria japonica. Int. J. Biol. Macromol. 2008, 42, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Li, Q.; Wu, J.; Yang, X.; Yang, S.; Zhu, W.; Liu, Y. Fucoidan Extracted From Sporophyll of Undaria pinnatifida Grown in Weihai, China—Chemical Composition and Comparison of Antioxidant Activity of Different Molecular Weight Fractions. Front. Nutr. 2021, 8, 636930. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Xu, J.; Xu, X. Bioactivity of fucoidan extracted from Laminaria japonica using a novel procedure with high yield. Food Chem. 2018, 245, 911–918. [Google Scholar] [CrossRef]
- Fletcher, H.R.; Biller, P.; Ross, A.B.; Adams, J. The Seasonal Variation of Fucoidan within Three Species of Brown Macroalgae. Algal Res. 2017, 22, 79–86. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Manivasagan, P.; Venkatesan, J.; Kim, S.K. Brown Seaweed Fucoidan: Biological Activity and Apoptosis, Growth Signaling Mechanism in Cancer. Int. J. Biol. Macromol. 2013, 60, 366–374. [Google Scholar] [CrossRef]
- Anisha, G.S.; Padmakumari, S.; Patel, A.K.; Pandey, A.; Singhania, R.R. Fucoidan from Marine Macroalgae: Biological Actions and Applications in Regenerative Medicine, Drug Delivery Systems and Food Industry. Bioengineering 2022, 9, 472. [Google Scholar] [CrossRef]
- van Weelden, G.; Bobiński, M.; Okła, K.; van Weelden, W.J.; Romano, A.; Pijnenborg, J.M.A. Fucoidan Structure and Activity in Relation to Anti-Cancer Mechanisms. Mar. Drugs 2019, 17, 32. [Google Scholar] [CrossRef] [PubMed]
- Nishimoto, S. Clinical improvement in cancer patients through integrated medicine, mainly using low molecular weight fucoidan supplements. J. Int. Soc. Life Inf. Sci. 2015, 33, 25–47. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Teruya, K.; Eto, H.; Shirahata, S. Induction of Apoptosis by Low-Molecular-Weight Fucoidan through Calcium- and Caspase-Dependent Mitochondrial Pathways in MDA-MB-231 Breast Cancer Cells. Biosci. Biotechnol. Biochem. 2013, 77, 235–242. [Google Scholar] [CrossRef]
- Chen, M.C.; Hsu, W.L.; Hwang, P.A.; Chou, T.C. Low Molecular Weight Fucoidan Inhibits Tumor Angiogenesis through Downregulation of HIF-1/VEGF Signaling under Hypoxia. Mar. Drugs 2015, 13, 4436–4451. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.C.; Hsu, W.L.; Hwang, P.A.; Chen, Y.L.; Chou, T.C. Combined administration of fucoidan ameliorates tumor and chemotherapy-induced skeletal muscle atrophy in bladder cancer-bearing mice. Oncotarget 2016, 7, 51608–51618. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, K.; Xue, C.; Zhao, X.; Mori, M.; Sugawara, T.; Hirata, T. Effects of middle molecular weight fucoidans on in vitro and ex vivo angiogenesis of endothelial cells. Int. J. Mol. Med. 2005, 15, 695–699. [Google Scholar] [CrossRef]
- Matou, S.; Helley, D.; Chabut, D.; Bros, A.; Fischer, A.M. Effect of fucoidan on fibroblast growth factor-2-induced angiogenesis in vitro. Thromb. Res. 2002, 106, 213–221. [Google Scholar] [CrossRef]
- Chabut, D.; Fischer, A.M.; Hettey, D.; Colliec, S. Low molecular weight fucoidan promotes FGF-2-induced vascular tube formation by human endothelial cells, with decreased PAI-1 release and ICAM-1 downregulation. Thromb. Res. 2004, 113, 93–95. [Google Scholar] [CrossRef]
- Park, H.Y.; Park, S.H.; Jeong, J.W.; Yoon, D.; Han, M.H.; Lee, D.S. Induction of p53-independent apoptosis and G1 cell cycle arrest by fucoidan in HCT116 human colorectal carcinoma cells. Mar. Drugs 2017, 15, 154. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Kim, G.Y.; Nam, T.J.; Kim, N.D.; Choi, Y.H. Antiproliferative activity of fucoidan was associated with the induction of apoptosis and autophagy in AGS human gastric cancer cells. J. Food Sci. 2011, 76, T77–T83. [Google Scholar] [CrossRef] [PubMed]
- Sanjeewa, K.K.A.; Lee, J.S.; Kim, W.S.; Jeon, Y.J. The potential of brown-algae polysaccharides for the development of anticancer agents: An update on anticancer effects reported for fucoidan and laminaran. Carbohydr. Polym. 2017, 177, 451–459. [Google Scholar] [CrossRef]
- Li, B.; Lu, F.; Wei, X.J.; Zhao, R.X. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef]
- Li, B.; Zhao, R.X.; Wei, X.J. Anticoagulant activity of fucoidan from Hizikia fusiforme. Agro Food Ind. Hi-Tech 2008, 19, 22–24. [Google Scholar]
- Koyanagi, S.; Tanigawa, N.; Nakagawa, H.; Soeda, S.; Shimeno, H. Oversulfation of fucoidan enhances its anti-angiogenic and antitumor activities. Biochem. Pharmacol. 2003, 65, 173–179. [Google Scholar] [CrossRef]
- Oliveira, C.; Ferreira, A.S.; Novoa-Carballal, R.; Nunes, C.; Pashkuleva, I.; Neves, N.M.; Coimbra, M.A.; Reis, R.L.; Martins, A.; Silva, T.H. The Key Role of Sulfation and Branching on Fucoidan Antitumor Activity. Macromol. Biosci. 2017, 17, 1600340. [Google Scholar] [CrossRef]
- Irhimeh, M.R.; Fitton, J.H.; Lowenthal, R.M.; Kongtawelert, P. A quantitative method to detect fucoidan in human plasma using a novel antibody. Methods Find. Exp. Clin. Pharmacol. 2005, 27, 705–710. [Google Scholar] [CrossRef]
- Zhang, W.; Sun, D.; Zhao, X.; Jin, W.; Wang, J.; Zhang, Q. Microanalysis and preliminary pharmacokinetic studies of a sulfated polysaccharide from Laminaria japonica. Chin. J. Oceanol. Limnol. 2016, 34, 177–185. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Shikov, A.N.; Faustova, N.M.; Obluchinskaya, E.D.; Kosman, V.M.; Vuorela, H.; Makarov, V.G. Pharmacokinetic and Tissue Distribution of Fucoidan from Fucus vesiculosus after Oral Administration to Rats. Mar. Drugs 2018, 16, 132. [Google Scholar] [CrossRef] [PubMed]
- Tokita, Y.; Nakajima, K.; Mochida, H.; Iha, M.; Nagamine, T. Development of a fucoidan-specific antibody and measurement of fucoidan in serum and urine by sandwich ELISA. Biosci. Biotechnol. Biochem. 2010, 74, 350–357. [Google Scholar] [CrossRef]
- Nagamine, T.; Nakazato, K.; Tomioka, S.; Iha, M.; Nakajima, K. Intestinal Absorption of Fucoidan Extracted from the Brown Seaweed, Cladosiphon okamuranus. Mar. Drugs 2015, 13, 48–64. [Google Scholar] [CrossRef]
- Pozharitskaya, O.N.; Shikov, A.N.; Obluchinskaya, E.D.; Vuorela, H. The Pharmacokinetics of Fucoidan after Topical Application to Rats. Mar. Drugs 2019, 17, 687. [Google Scholar] [CrossRef] [PubMed]
- Kimura, R.; Rokkaku, T.; Takeda, S.; Senba, M.; Mori, N. Cytotoxic Effects of Fucoidan Nanoparticles against Osteosarcoma. Mar. Drugs 2013, 11, 4267–4278. [Google Scholar] [CrossRef] [PubMed]
- Koh, H.; Lu, J.; Zhou, W. Structure characterization and antioxidant activity of fucoidan isolated from Undaria pinnatifida grown in New Zealand. Carbohydr. Polym. 2019, 212, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.M.C.L.; dos Santos Lisboa, L.; Paiva, W.S.; Batista, L.A.N.C.; Luchiari, A.C.; Rocha, H.A.O.; Camara, R.B.G. Comparison of in vitro and in vivo antioxidant activities of commercial fucoidans from Macrocystis pyrifera, Undaria pinnatifida, and Fucus vesiculosus. Int. J. Biol. Macromol. 2022, 216, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Choi, J.; Park, H. Antioxidant activities of fucoidan degraded by gamma irradiation and acidic hydrolysis. Radiat. Phys. Chem. 2015, 109, 23–26. [Google Scholar] [CrossRef]
- Lim, S.J.; Wan Aida, W.M.; Maskat, M.Y.; Mamot, S.; Ropien, J.; Mazita Mohd, D. Isolation and antioxidant capacity of fucoidan from selected Malaysian seaweeds. Food Hydrocoll. 2014, 42, 280–288. [Google Scholar] [CrossRef]
- Kim, K.J.; Yoon, K.Y.; Lee, B.Y. Low molecular weight fucoidan from the sporophyll of Undaria pinnatifida suppresses inflammation by promoting the inhibition of mitogen-activated protein kinases and oxidative stress in RAW264.7 cells. Fitoterapia 2012, 83, 1628–1635. [Google Scholar] [CrossRef] [PubMed]
- Pérez, M.J.; Falqué, E.; Domínguez, H. Antimicrobial Action of Compounds from Marine seaweed. Mar. Drugs 2016, 14, 52. [Google Scholar] [CrossRef]
- Silva, A.; Silva, S.A.; Carpena, M.; Gull, P.; Barroso, M.F.; Prieto, M.A. Macroalgae as a Source of Valuable Antimicrobial Compounds: Extraction and Applications. Antibiotics 2020, 9, 642. [Google Scholar] [CrossRef]
- Liu, M.; Liu, Y.; Cao, M.J.; Liu, G.-M.; Chen, Q.; Sun, L.; Chen, H. Antibacterial activity and mechanisms of depolymerized fucoidans isolated from Laminaria japonica. Carbohydr. Polym. 2017, 172, 294–305. [Google Scholar] [CrossRef]
- Shannon, E.; Abu-Ghannam, N. Antibacterial Derivatives of Marine Algae: An Overview of Pharmacological Mechanisms and Applications. Mar. Drugs 2016, 14, 81. [Google Scholar] [CrossRef]
- Lee, K.Y.; Jeong, M.R.; Choi, S.M.; Na, S.S.; Cha, J.D. Synergistic effect of fucoidan with antibiotics against oral pathogenic bacteria. Arch. Oral Biol. 2013, 58, 482–492. [Google Scholar] [CrossRef]
- Hans, N.; Malik, A.; Naik, S. Antiviral activity of sulfated polysaccharides from marine algae and its application in combating COVID-19: Mini review. Bioresour. Technol. Rep. 2021, 13, 100623. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, K.; Lee, J.-B.; Nakano, T.; Hayashi, T. Anti-influenza A virus characteristics of a fucoidan from sporophyll of Undaria pinnatifida in mice with normal and compromised immunity. Microbes Infect. 2013, 15, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, K.C.S.; Medeiros, V.P.; Queiroz, L.S.; Abreu, L.R.D.; Rocha, H.A.O.; Ferreira, C.V.; Jucá, M.B.; Aoyama, H.; Leite, E.L. Inhibition of reverse transcriptase activity of HIV by polysaccharides of brown algae. Biomed. Pharmacother. 2008, 62, 303–307. [Google Scholar] [CrossRef]
- Thuy, T.T.T.; Ly, B.M.; Van, T.T.T.; Quang, N.V.; Tu, H.C.; Zheng, Y.; Seguin-Devaux, C.; Mi, B.; Ai, U. Anti-HIV activity of fucoidans from three brown seaweed species. Carbohydr. Polym. 2015, 115, 122–128. [Google Scholar] [CrossRef]
- Springer, G.F.; Wurzel, H.A.; McNeal, G.M., Jr.; Ansell, N.J.; Doughty, M.F. Isolation of anncoagulant fractions from crude fucoidin. Proc. Sot. Exptl. Biol. Med. 1957, 94, 404–409. [Google Scholar] [CrossRef]
- Grauffel, V.; Kloareg, B.; Mabeau, S.; Durand, P.; Jozefonvicz, J. New natural polysaccharides with potent antithrombic activity: Fucans from brown algae. Biomaterials 1989, 10, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Wang, Q.; Yang, Y.; Hu, Y.; Song, Y.; He, Y.; Liu, S.; Wu, L. Hypolipidemic effects of fucoidan fractions from Saccharina sculpera (Laminariales, Phaeophyceae). Int. J. Biol. Macromol. 2019, 140, 188–195. [Google Scholar] [CrossRef]
- Oliveira, R.M.; Câmara, R.B.G.; Monte, J.F.S.; Viana, R.L.S.; Melo, K.R.T.; Queiroz, M.F.; Filgueira, L.G.A.; Oyama, L.M.; Rocha, H.A.O. Commercial Fucoidans from Fucus vesiculosus Can Be Grouped into Antiadipogenic and Adipogenic Agents. Mar. Drugs 2018, 16, 193. [Google Scholar] [CrossRef]
- Sim, S.Y.; Shin, Y.E.; Kim, H.K. Fucoidan from Undaria pinnatifida has anti-diabetic effects by stimulation of glucose uptake and reduction of basal lipolysis in 3T3-L1 adipocytes. Nutr. Res. 2019, 65, 54–62. [Google Scholar] [CrossRef]
- Narayani, S.S.; Saravanan, S.; Ravindran, J.; Ramasamy, M.S.; Chitra, J. In vitro anticancer activity of fucoidan extracted from Sargassum cinereum against Caco-2 cells. Int. J. Biol. Macromol. 2019, 138, 618–628. [Google Scholar] [CrossRef]
- Chantree, P.; Na-Bangchang, K.; Martviset, P. Anticancer activity of fucoidan via apoptosis and cell cycle arrest on cholangiocarcinoma cell. Asian Pac. J. Cancer Prev. 2021, 22, 209–217. [Google Scholar] [CrossRef]
- Banafa, A.M.; Roshan, S.; Liu, Y.Y.; Chen, H.J.; Chen, M.J.; Yang, G.X.; He, G.Y. Fucoidan induces G1 phase arrest and apoptosis through caspases-dependent pathway and ROS induction in human breast cancer MCF-7 cells. J. Huazhong Univ. Sci. Technolog. Med. Sci. 2013, 33, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Teruya, K.; Yoshida, T.; Eto, H.; Shirahata, S. Fucoidan extract enhances the anti-cancer activity of chemotherapeutic agents in MDA-MB-231 and MCF-7 breast cancer cells. Mar. Drugs 2013, 11, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wang, P.; Wang, H.; Li, Q.; Teng, H.; Liu, Z.; Yang, W.; Hou, L.; Zou, X. Fucoidan derived from Undaria pinnatifida induces apoptosis in human hepatocellular carcinoma SMMC-7721 cells via the ROS-mediated mitochondrial pathway. Mar. Drugs 2013, 11, 1961–1976. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhao, Y.; Zhang, Y.; Zhang, D. Fucoidan induces cancer cell apoptosis by modulating the endoplasmic reticulum stress cascades. PLoS ONE 2014, 9, e108157. [Google Scholar] [CrossRef]
- Ma, D.; Wei, J.; Chen, S.; Wang, H.; Ning, L.; Luo, S.-H.; Liu, C.-L.; Song, G.; Yao, Q. Fucoidan Inhibits the Progression of Hepatocellular Carcinoma via Causing lncRNA LINC00261 Overexpression. Front. Oncol. 2021, 11, 653902. [Google Scholar] [CrossRef]
- Takahashi, H.; Kawaguchi, M.; Kitamura, K.; Narumiya, S.; Kawamura, M.; Tengan, I.; Nishimoto, S.; Hanamure, Y.; Majima, Y.; Tsubura, S.; et al. An Exploratory Study on the Anti-inflammatory Effects of Fucoidan in Relation to Quality of Life in Advanced Cancer Patients. Integr. Cancer Ther. 2018, 17, 282–291. [Google Scholar] [CrossRef]
- Hwang, P.A.; Phan, N.N.; Lu, W.J.; Ngoc Hieu, B.T.; Lin, Y.C. Low-molecular-weight fucoidan and high-stability fucoxanthin from brown seaweed exert prebiotics and anti-inflammatory activities in Caco-2 cells. J. Food Nutr. Res. 2016, 60, 32033. [Google Scholar] [CrossRef]
- Cui, M.; Li, X.; Geng, L.; Wu, N.; Wang, J.; Deng, Z.; Li, Z.; Zhang, Q. Comparative study of the immunomodulatory effects of different fucoidans from Saccharina japonica mediated by scavenger receptors on RAW264.7 macrophages. Int. J. Biol. Macromol. 2022, 215, 253–261. [Google Scholar] [CrossRef]
- Nakamura, T.; Suzuki, H.; Wada, Y.; Kodama, T.; Doi, T. Fucoidan induces nitric oxide production via p38 mitogen-activated protein kinase and NF-_B-dependent signaling pathways through macrophage scavenger receptors. Biochem. Biophys. Res. Commun. 2006, 343, 286–294. [Google Scholar] [CrossRef]
- Saravana, P.S.; Cho, Y.J.; Park, Y.B.; Woo, H.C.; Chun, B.S. Structural, antioxidant, and emulsifying activities of fucoidan from Saccharina japonica using pressurized liquid extraction. Carbohydr. Polym. 2016, 153, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Kim, K.B.W.R.; Jeong, D.H.; Ahn, D.H. Anti-inflammatory activity of ethanolic extract of Sargassum sagamianum in RAW 264.7 cells. Food Sci. Biotechnol. 2013, 22, 1113–1120. [Google Scholar] [CrossRef]
- Cui, M.; Wu, J.; Wang, S.; Shu, H.; Zhang, M.; Liu, K.; Liu, K. Characterization and anti-inflammatory effects of sulfated polysaccharide from the red seaweed Gelidium pacificum Okamura. Int. J. Biol. Macromol. 2019, 129, 377–385. [Google Scholar] [CrossRef]
- Ni, L.; Wang, L.; Fu, X.; Duan, D.; Jeon, Y.; Xu, J.; Gao, X. In vitro and in vivo anti-inflammatory activities of a fucose-rich fucoidan isolated from Saccharina japonica. Int. J. Biol. Macromol. 2020, 156, 717–729. [Google Scholar] [CrossRef] [PubMed]
- Fernando, I.P.S.; Sanjeewa, K.K.A.; Samarakoon, K.W.; Lee, W.W.; Kim, H.S.; Kang, N.; Ranasinghe, P.; Lee, H.S.; Jeon, Y.J. A fucoidan fraction purified from Chnoospora minima; a potential inhibitor of LPS-induced inflammatory responses. Int. J. Biol. Macromol. 2017, 104, 1185–1193. [Google Scholar] [CrossRef]
- Pavliga, S.N.; Kompanets, G.G.; Tsygankov, V.Y. The experimental research in vitro of carrageenans and fucoidans to decrease activity of Hantavirus. Food Environ. Virol. 2017, 8, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Santoyo, S.; Plaza, M.; Jaime, L.; Ibañez, E.; Reglero, G.; Señorans, J. Pressurized liquids as an alternative Green process to extract antiviral agents from the edible seaweed Himanthalia elongata. J. Appl. Phycol. 2011, 23, 909–917. [Google Scholar] [CrossRef]
- Wan, P.; Yang, X.M.; Cai, B.N.; Chen, H.; Sun, H.L.; Chen, D.K.; Pan, J.Y. Ultrasonic extraction of polysaccharides from Laminaria japonica and their antioxidative and glycosidase inhibitory activities. J. Ocean Univ. China 2015, 14, 651–662. [Google Scholar] [CrossRef]
- Fu, Z.F.; Li, H.H.; Liu, H.B.; Hu, S.M.; Li, Y.J.; Wang, M.X.; Guan, H.S. Bioassay-guided extraction of crude fucose-containing sulphated polysaccharides from Sargassum fusiforme with response surface methodology. J. Ocean Univ. China 2016, 15, 533–540. [Google Scholar] [CrossRef]
- Ko, S.C.; Lee, S.H.; Ahn, G.; Kim, K.N.; Cha, S.H.; Kim, S.K.; Jeon, B.T.; Park, P.J.; Lee, K.W.; Jeon, Y.J. Effect of enzyme-assisted extract of Sargassum coreanum on induction of apoptosis in HL-60 tumor cells. J. Appl. Phycol. 2012, 24, 675–684. [Google Scholar] [CrossRef]
- Zhurishkina, E.V.; Stepanov, S.I.; Shvetsova, S.V.; Kulminskaya, A.A.; Lapina, I.M. A comparison of the effect of fucoidan from alga Fucus vesiculosus and its fractions obtained by anion-exchange chromatography on HeLa G-63, Hep G2, and Chang liver cells. Cell Tissue Biol. 2017, 11, 242–249. [Google Scholar] [CrossRef]
- Bobiński, M.; Okła, K.; Bednarek, W.; Wawruszak, A.; Dmoszyńska-Graniczka, M.; Garcia-Sanz, P.; Wertel, I.; Kotarski, J. The effect of fucoidan, a potential new, natural, anti-neoplastic agent on uterine sarcomas and carcinosarcoma cell lines: ENITEC collaborative study. Arch. Immunol. Ther. Exp. 2019, 67, 125–131. [Google Scholar] [CrossRef]
- Wu, J.; Chen, X.; Qiao, K.; Su, Y.; Liu, Z. Purification, structural elucidation, and in vitro antitumor effects of novel polysaccharides from Bangia fuscopurpurea. Food Sci. Hum. Wellness 2021, 10, 63–71. [Google Scholar] [CrossRef]
- Azuma, K.; Ishihara, T.; Nakamoto, H.; Amaha, T.; Osaki, T.; Tsuka, T.; Imagawa, T.; Minami, S.; Takashima, O.; Ifuku, S.; et al. Effects of oral administration of fucoidan extracted from Cladosiphon okamuranus on tumor growth and survival time in a tumor-bearing mouse model. Mar. Drugs 2012, 10, 2337–2348. [Google Scholar] [CrossRef]
- Xue, M.; Ge, Y.; Zhang, J.; Wang, Q.; Hou, L.; Liu, Y.; Sun, L.; Li, Q. Anticancer properties and mechanisms of fucoidan on mouse breast cancer in vitro and in vivo. PLoS ONE 2012, 7, e43483. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Cao, R.; Zhang, S.X.; Man, Y.N.; Wu, X.Z. Fucoidan inhibits the growth of hepatocellular carcinoma independent of angiogenesis. Evid. Based Complement. Alternat. Med. 2013, 2013, 692549. [Google Scholar] [CrossRef]
- Hsu, H.Y.; Lin, T.Y.; Hwang, P.A.; Tseng, L.M.; Chen, R.H.; Tsao, S.M.; Hsu, J. Fucoidan induces changes in the epithelial to mesenchymal transition and decreases metastasis by enhancing ubiquitin-dependent TGFβ receptor degradation in breast cancer. Carcinogenesis 2013, 34, 874–884. [Google Scholar] [CrossRef]
- Suresh, V.; Anbazhagan, C.; Thangam, R.; Senthilkumar, D.; Senthilkumar, N.; Kannan, S.; Rengasamy, R.; Palani, P. Stabilization of mitochondrial and microsomal function of fucoidan from Sargassum plagiophyllum in diethylnitrosamine induced hepatocarcinogenesis. Carbohydr. Polym. 2013, 92, 1377–1385. [Google Scholar] [CrossRef]
- Takeda, K.; Tomimori, K.; Kimura, R.; Ishikawa, C.; Nowling, T.K.; Mori, N. Anti-tumor activity of fucoidan is mediated by nitric oxide released from macrophages. Int. J. Oncol. 2012, 40, 251–260. [Google Scholar]
- Granert, C.; Raud, J.; Waage, A.; Lindquist, L. Effects of polysaccharide fucoidin on cerebrospinal fluid interleukin-1 and tumor necrosis factor a in pneumococcal meningitis in the rabbit. Infect. Immun. 1999, 67, 2071–2074. [Google Scholar] [CrossRef]
- Hidalgo, A.; Peired, A.J.; Weiss, L.A.; Katayama, Y.; Frenette, P.S. The integrin αMβ2 anchors hematopoietic progenitors in the bone marrow during enforced mobilization. Blood 2004, 104, 993–1001. [Google Scholar] [CrossRef]
- Tanaka, K.; Ito, M.; Kodama, M.; Tomita, M.; Kimura, S.; Hoyano, M.; Mitsuma, W.; Hirono, S.; Hanawa, H.; Aizawa, Y. Sulfated polysaccharide fucoidan ameliorates experimental autoimmune myocarditis in rats. J. Cardiovasc. Pharmacol. Ther. 2011, 16, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Sharma, G.; Das, P.K. Fucoidan cures infection with both antimony-susceptible and -resistant strains of Leishmania donovani through Th1 response and macrophage-derived oxidants. Antimicrob. Chemother. 2011, 66, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.O.; Zhang, W.; Du, J.Y.; Wong, K.W.; Oda, T.; Yu, Q. Fucoidan can function as an adjuvant in vivo to enhance dendritic cell maturation and function and promote antigen-specific t cell immune responses. PLoS ONE 2014, 9, e99396. [Google Scholar] [CrossRef]
- Hayashi, K.; Nakano, T.; Hashimoto, M.; Kanekiyo, K.; Hayashi, T. Defensive effects of a fucoidan from brown alga Undaria pinnatifida against herpes simplex virus infection. Int. Immunopharmacol. 2008, 8, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Oda, T.; Yu, Q.; Jin, J.O. Fucoidan from Macrocystis pyrifera has powerful immune-modulatory effects compared to three other fucoidans. Mar. Drugs 2015, 13, 1084–1104. [Google Scholar] [CrossRef]
- Maruyama, H.; Tamauchi, H.; Kawakami, F.; Yoshinaga, K.; Nakano, T. Suppressive effect of dietary fucoidan on proinflammatory immune response and MMP-1 expression in UVB-irradiated mouse skin. Planta Med. 2015, 81, 1370–1374. [Google Scholar] [CrossRef]
- Carvalho, A.C.; Sousa, R.B.; Franco, A.X.; Costa, J.V.; Neves, L.M.; Ribeiro, R.A.; Sutton, R.; Criddle, D.N.; Soares, P.M.; de Souza, M.H. Protective effects of fucoidan, a p- and l-selectin inhibitor, in murine acute pancreatitis. Pancreas 2014, 43, 82–87. [Google Scholar] [CrossRef]
- Lean, Q.Y.; Eri, R.D.; Fitton, J.H.; Patel, R.P.; Gueven, N. Fucoidan extracts ameliorate acute colitis. PLoS ONE 2015, 10, e0128453. [Google Scholar] [CrossRef] [PubMed]
- AlKahtane, A.A.; Abushouk, A.I.; Mohammed, E.T.; ALNasser, M.; Alarifi, S.; Ali, D.; Alessia, M.S.; Almeer, R.S.; AlBasher, G.; Alkahtani, S. Fucoidan alleviates microcystin-LR-induced hepatic, renal, and cardiac oxidative stress and inflammatory injuries in mice. Environ. Sci. Pollut. Res. 2020, 27, 2935–2944. [Google Scholar] [CrossRef]
- Aleissa, M.S.; Alkahtani, S.; Abd Eldaim, M.A.; Ahmed, A.M.; Bungău, S.G.; Almutairi, B.; Bin-Jumah, M.; AlKahtane, A.A.; Alyousif, M.S.; Abdel-Daim, M.M. Fucoidan ameliorates oxidative stress, inflammation, DNA damage, and hepatorenal injuries in diabetic rats intoxicated with aflatoxin B1. Oxidative Med. Cell. Longev. 2020, 2020, 9316751. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.; Yoon, W.J.; Ham, Y.M.; Yoon, S.A.; Kang, S.C. Anti-arthritis effect through the anti-inflammatory effect of Sargassum muticum extract in collagen-induced arthritic (CIA) Mice. Molecules 2019, 24, 276. [Google Scholar] [CrossRef]
- Matsumoto, S.; Nagaoka, M.; Hara, T.; Kimura-Takagi, I.; Mistuyama, K.; Ueyama, S. Fucoidan derived from Cladosiphon okamuranus Tokida ameliorates murine chronic colitis through the down-regulation of interleukin 6 production on colonic epithelial cells. Clin. Exp. Immunol. 2004, 136, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Ko, C.I.; Jee, Y.; Jeong, Y.; Kim, M.; Kim, J.S.; Jeon, Y.J. Anti-inflammatory effect of fucoidan extracted from Ecklonia cava in zebrafish model. Carbohydr. Polym. 2013, 92, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Park, S.B.; Chun, K.R.; Kim, J.K.; Suk, K.; Jung, Y.M.; Lee, W.H. The differential effect of high and low molecular weight fucoidans on the severity of collagen-induced arthritis in mice. Phytother. Res. 2010, 24, 1384–1391. [Google Scholar] [CrossRef] [PubMed]
- Ananthi, S.; Gayathri, V.; Malarvizhi, R.; Bhardwaj, M.; Vasanthi, H.R. Anti-arthritic potential of marine macroalgae Turbinaria ornata in complete freund’s adjuvant induced rats. Exp. Toxicol. Pathol. 2017, 69, 672–680. [Google Scholar] [CrossRef]
- Manikandan, R.; Parimalanandhini, D.; Mahalakshmi, K.; Beulaja, M.; Arumugam, M.; Janarthanan, S.; Palanisamy, S.; You, S.; Prabhu, N.M. Studies on isolation, characterization of fucoidan from brown algae Turbinaria decurrens and evaluation of it’s in vivo and in vitro anti-inflammatory activities. Int. J. Biol. Macromol. 2020, 160, 1263–1276. [Google Scholar] [CrossRef]
- Atashrazm, F.; Lowenthal, R.M.; Woods, G.M.; Holloway, A.F.; Dickinson, J.L. Fucoidanand cancer: A multifunctional molecule with anti-tumor potential. Mar. Drugs 2015, 13, 2327–2346. [Google Scholar] [CrossRef]
- Yan, M.D.; Yao, C.J.; Chow, J.M.; Chang, C.L.; Hwang, P.A.; Chuang, S.E.; Peng, J.W.; Lai, G.M. Fucoidan elevates MicroRNA-29b to regulate DNMT3B-MTSS1 Axis and inhibit EMT in human hepatocellular carcinoma cells. Mar. Drugs 2015, 13, 6099. [Google Scholar] [CrossRef]
- Marinval, N.; Saboural, P.; Haddad, O.; Maire, M.; Bassand, K.; Geinguenaud, F.; Djaker, N.; Ben Akrout, K.; Lamy de la Chapelle, M.; Robert, R.; et al. Identification of a pro-angiogenic potential and cellular uptake mechanism of a LMW highly sulfated fraction of Fucoidan from Ascophyllum nodosum. Mar. Drugs 2016, 14, 185. [Google Scholar] [CrossRef]
- Han, Y.S.; Lee, J.H.; Lee, S.H. Fucoidan inhibits themigration and proliferation of HT- 29 human colon cancer cells via the phosphoinositide-3 kinase/Akt/mechanistic target of rapamycin pathways. Mol. Med. Rep. 2015, 12, 3446–3452. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Liang, H.; Tang, Q.; Xue, C.; He, X.; Zhang, L.; Zhang, Z.; Liang, Z.; Bian, K.; Zhang, L.; et al. The protective and Immunomodulatory effects of Fucoidan against 7,12-dimethyl benz[a]anthracene-induced experimental mammary carcinogenesis through the PD1/PDL1 signaling pathway in rats. Nutr. Cancer 2017, 69, 1234–1244. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Zhang, Q.; Kong, Y.; Xie, B.; Gao, M.; Tao, Y.; Xu, H.; Zhan, F.; Dai, B.; Shi, J.; et al. Antitumor activity of fucoidan against diffuse large B cell lymphoma in vitro and in vivo. Acta Biochim. Biophys. Sin. Shanghai 2015, 47, 9. [Google Scholar] [CrossRef]
- Yoshimoto, M.; Higaki, K.; Nanba, E.; Ikeguchi, M. Anti-proliferation activity of Fucoidan in MKN45 gastric cancer cells and downregulation of phosphorylated ASK1, a cell cycle-regulated kinase. Yonago Acta Med. 2015, 58, 1–7. [Google Scholar]
- Etman, S.M.; Elnaggar, Y.S.R.; Abdallah, O.Y. Fucoidan, a natural biopolymer in cancer combating: From edible algae to nanocarrier tailoring. Int. J. Biol. Macromol. 2020, 147, 799–808. [Google Scholar] [CrossRef]
- Zhang, Z.; Teruya, K.; Eto, H.; Shirahata, S. Fucoidan extract induces apoptosis inMCF-7 cells via a mechanism involving the ROS-dependent JNK activation and mitochondria-mediated pathways. PLoS ONE 2011, 6, e27441. [Google Scholar] [CrossRef]
- Li, J.; Chen, K.; Li, S.; Liu, T.; Wang, F.; Xia, Y.; Lu, J.; Zhou, Y.; Guo, C. Pretreatment with Fucoidan from Fucus vesiculosus protected against ConA-induced acute liver injury by inhibiting both intrinsic and extrinsic apoptosis. PLoS ONE 2016, 11, e0152570. [Google Scholar] [CrossRef]
- Ustyuzhanina, N.E.; Bilan, M.I.; Ushakova, N.A.; Usov, A.I.; Kiselevskiy, M.V.; Nifantiev, N.E. Fucoidans: Pro- or antiangiogenic agents? Glycobiology 2014, 24, 1265–1274. [Google Scholar] [CrossRef]
- Rui, X.; Pan, H.F.; Shao, S.L.; Xu, X.M. Anti-tumor and anti-angiogenic effects of Fucoidan on prostate cancer: Possible JAK-STAT3 pathway. BMC Complement. Altern. Med. 2017, 17, 378. [Google Scholar] [CrossRef]
- Chen, H.; Cong, Q.; Du, Z.; Liao, W.; Zhang, L.; Yao, Y.; Ding, K. Sulfated fucoidan FP08S2 inhibits lung cancer cell growth in vivo by disrupting angiogenesis via targeting VEGFR2/VEGF and blocking VEGFR2/Erk/VEGF signaling. Cancer Lett. 2016, 382, 44–52. [Google Scholar] [CrossRef]
- Lee, H.; Kim, J.S.; Kim, E. Fucoidan from seaweed Fucus vesiculosus inhibits migration and invasion of human lung Cancer cell via PI3K-Akt-mTOR pathways. PLoS ONE 2012, 7, e50624. [Google Scholar] [CrossRef] [PubMed]
- Cho, T.M.; Kim, W.J.; Moon, S.K. AKT signaling is involved in fucoidan-induced inhibition of growth and migration of human bladder cancer cells. Food Chem. Toxicol. 2014, 64, 344–352. [Google Scholar] [CrossRef]
- Teruya, T.; Tatemoto, H.; Konishi, T.; Tako, M. Structural characteristics and in vitro macrophage activation of acetyl fucoidan from Cladosiphon okamuranus. Glycoconj. J. 2009, 26, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wang, J.; Jin, W.; Zhang, H.; Zhang, Q. Degradation of Laminaria japonica fucoidan by hydrogen peroxide and antioxidant activities of the degradation products of different molecular weights. Carbohydr. Polym. 2012, 87, 153–159. [Google Scholar] [CrossRef]
- Imbs, T.I.; Skriptsova, A.V.; Zvyagintseva, T.N. Antioxidant activity of fucose-containing sulfated polysaccharides obtained from Fucus evanescens by different extraction methods. J. Appl. Phycol. 2015, 27, 545–553. [Google Scholar] [CrossRef]
- Park, H.Y.; Kim, G.Y.; Moon, S.K.; Kim, W.J.; Yoo, Y.H.; Choi, Y.H. Fucoidan inhibits the proliferation of human urinary bladder cancer T24 cells by blocking cell cycle progression and inducing apoptosis. Molecules 2014, 19, 5981–5998. [Google Scholar] [CrossRef]
- Kim, E.J.; Park, S.Y.; Lee, J.Y.; Park, J.H. Fucoidan present in brown algae induces apoptosis of human colon cancer cells. BMC Gastroenterol. 2010, 10, 96. [Google Scholar] [CrossRef]
- Hsu, H.Y.; Lin, T.Y.; Hu, C.H.; Shu, D.T.F.; Lu, M.K. Fucoidan upregulates TLR4/CHOP-mediated caspase-3 and PARP activation to enhance cisplatin-induced cytotoxicity in human lung cancer cells. Cancer Lett. 2018, 432, 112–120. [Google Scholar] [CrossRef]
- Teng, H.; Yang, Y.; Wei, H. Fucoidan Suppresses Hypoxia-Induced Lymphangiogenesis and Lymphatic Metastasis in Mouse Hepatocarcinoma. Mar. Drugs 2015, 13, 3514–3530. [Google Scholar] [CrossRef]
- Mabeau, S.; Kloareg, B.; Joseleau, J.P. Fractionation and analysis of fucans from brown algae. Phytochemistry 1990, 29, 2441–2445. [Google Scholar] [CrossRef]
- Alwarsamy, M.; Gooneratne, R.; Ravichandran, R. Effect of fucoidan from Turbinaria conoides on human lung adenocarcinoma epithelial (A549) cells. Carbohydr. Polym. 2016, 152, 207–213. [Google Scholar] [CrossRef]
- Holtkamp, A.D.; Kelly, S.; Ulber, R.; Lang, S. Fucoidans and fucoidanases--focus on techniques for molecular structure elucidation and modification of marine polysaccharides. Appl. Microbiol. Biotechnol. 2009, 82, 1–11. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fedewa, S.A. Colorectal cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Thinh, P.D.; Menshova, R.V.; Ermakova, S.P.; Anastyuk, S.D.; Ly, B.M.; Zvyagintseva, T.N. Structural characteristics and anticancer activity of fucoidan from the brown alga Sargassum mcclurei. Mar. Drugs 2013, 11, 1456–1476. [Google Scholar] [CrossRef]
- Back, H.I.; Kim, S.Y.; Park, S.H.; Oh, M.R.; Kim, M.G.; Jeon, J.Y.; Chae, S.W.; Bae, J.S.; Kim, Y.S.; Hwang, Y.J.; et al. Effects of Fucoidan supplementation on Helicobacter pylori in humans. FASEB J. 2010, 24, lb347. [Google Scholar] [CrossRef]
- Vishchuk, O.S.; Ermakova, S.P.; Zvyagintseva, T.N. The fucoidans from brown algae of Far-Eastern seas: Anti-tumor activity and structure-function relationship. Food Chem. 2013, 141, 1211–1217. [Google Scholar] [CrossRef]
- Jin, W.; Cai, X.F.; Na, M.; Lee, J.J.; Bae, K. Triterpenoids and diarylheptanoids from Alnus hirsuta inhibit HIF-1 in AGS cells. Arch. Pharm. Res. 2007, 30, 412–418. [Google Scholar] [CrossRef]
- Ale, M.T.; Mikkelsen, J.D.; Meyer, A.S. Important determinants for fucoidan bioactivity: A critical review of structure-function relations and extraction methods for fucose-containing sulfated polysaccharides from brown seaweeds. Mar. Drugs 2011, 9, 2106–2130. [Google Scholar] [CrossRef]
- Huang, T.H.; Chiu, Y.H.; Chan, Y.L. Prophylactic administration of fucoidan represses cancer metastasis by inhibiting vascular endothelial growth factor (VEGF) and matrix metalloproteinases (MMPs) in Lewis tumor-bearing mice. Mar. Drugs 2015, 13, 1882–1900. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.W.; Riaz, T.; Mahmood, S.; Bilal, M.; Manzoor, M.F.; Qamar, S.A.; Qi, X. Fucoidan-based nanomaterial and its multifunctional role for pharmaceutical and biomedical applications. Crit. Rev. Food Sci. Nutr. 2022, 2022, 1–27. [Google Scholar] [CrossRef]
- Cunha, L.; Grenha, A. Sulfated seaweed polysaccharides as multifunctional materials in drug delivery applications. Mar. Drugs 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.J.; Costa, R.R.; Mano, J.F. Marine origin polysaccharides in drug delivery systems. Mar. Drugs 2016, 14, 34. [Google Scholar] [CrossRef]
- Sezer, A.D.; Cevher, E. Fucoidan: A versatile biopolymer for biomedical applications. In Active Implants and Scafolds for Tissue Regeneration; Zilberman, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 8, pp. 377–406. [Google Scholar]
- Huang, Y.C.; Yang, Y.T. Effect of basic fibroblast growth factor released from chitosan–fucoidan nanoparticles on neurite extension. J. Tissue Eng. Regen. Med. 2016, 10, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.M.; Kim, J.K.; Cho, T.S. Applications of ophthalmic biomaterials embedded with fucoidan. Ind. Eng. Chem. 2012, 18, 1197–1201. [Google Scholar] [CrossRef]
- Fan, L.; Lu, Y.; Ouyang, X.K.; Ling, J. Development and characterization of soybean protein isolate and fucoidan nanoparticles for curcumin encapsulation. Int. J. Biol. Macromol. 2021, 169, 194–205. [Google Scholar] [CrossRef]
- Park, H.W.; Kim, D.Y.; Shin, W.S. Fucoidan improves the structural integrity and the molecular stability of β-lactoglobulin. Food Sci. Biotechnol. 2018, 27, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Nguyen, V.P.; Manivasagan, P.; Jung, M.J.; Kim, S.W.; Oh, J.; Kang, H.W. Doxorubicin-fucoidan-gold nanoparticles composite for dual-chemo-photothermal treatment on eye tumors. Oncotarget 2017, 8, 113719–113733. [Google Scholar] [CrossRef]
- Wang, P.; Kankala, R.K.; Chen, B.; Long, R.; Cai, D.; Liu, Y.; Wang, S. Poly-allylamine hydrochloride and fucoidan-based self-assembled polyelectrolyte complex nanoparticles for cancer therapeutics. J. Biomed. Mater. Res. A 2019, 107, 339–347. [Google Scholar] [CrossRef]
- Huang, Y.C.; Kuo, T.H. O-carboxylmethyl chitosan/fucoidan nanoparticles increase cellular curcumin uptake. Food Hydrocoll. 2016, 53, 261–269. [Google Scholar] [CrossRef]
- Hwang, P.A.; Lin, X.Z.; Kuo, K.L.; Hsu, F.Y. Fabrication and cytotoxicity of fucoidan-cisplatin nanoparticles for macrophage and tumor cells. Materials 2017, 10, 291. [Google Scholar] [CrossRef]
- Hussain, N.; Jaitley, V.; Florence, A.T. Recent advances in the understanding of uptake of microparticulates across the gastrointestinal lymphatics. Adv. Drug Deliv. Rev. 2001, 50, 107–142. [Google Scholar] [CrossRef]
- Gänger, S.; Schindowski, K. Tailoring Formulations for Intranasal Nose-to-Brain Delivery: A Review on Architecture, Physico-Chemical Characteristics and Mucociliary Clearance of the Nasal Olfactory Mucosa. Pharmaceutics 2018, 10, 116. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Akbuǧa, J. The design of biodegradable ofloxacin-based core-shell microspheres: Influence of the formulation parameters on in vitro characterization. Pharm. Dev. Technol. 2012, 17, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Sezer, A.D.; Cevher, E.; Hatipoğlu, F.; Oğurtan, Z.; Baş, A.L.; Akbuğa, J. The use of fucosphere in the treatment of dermal burns in rabbits. Eur. J. Pharm. Biopharm. 2008, 69, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Juenet, M.; Aid-Launais, R.; Maire, M.; Ollivier, V.; Letourneur, D.; Chauvierre, C. Development of polymer microcapsules functionalized with fucoidan to target P-selectin overexpressed in cardiovascular diseases. Adv. Health Mater. 2017, 6, 160. [Google Scholar] [CrossRef]
- Szekalska, M.; Citkowska, A.; Wróblewska, M.; Winnicka, K. The Impact of Gelatin on the Pharmaceutical Characteristics of Fucoidan Microspheres with Posaconazole. Materials 2021, 14, 4087. [Google Scholar] [CrossRef]
- Valente, S.A.; Silva, L.M.; Lopes, G.R.; Sarmento, B.; Coimbra, M.A.; Passos, C.P. Polysaccharide-based formulations as potential carriers for pulmonary delivery—A review of their properties and fates. Carbohydr. Polym. 2022, 277, 118784. [Google Scholar] [CrossRef]
- Cunha, L.; Rodrigues, S.; Rosa da Costa, A.M.; Faleiro, M.L.; Buttini, F.; Grenha, A. Inhalable fucoidan microparticles combining two antitubercular drugs with potential application in pulmonary tuberculosis therapy. Polymers 2018, 10, 636. [Google Scholar] [CrossRef]
- Deepika, M.S.; Thangam, R.; Sheena, T.S.; Sasirekha, R.; Sivasubramanian, S.; Babu, M.D.; Jeganathan, K.; Thirumurugan, R. A novel rutin-fucoidan complex based phytotherapy for cervical cancer through achieving enhanced bioavailability and cancer cell apoptosis. Biomed. Pharmacother 2019, 109, 1181–1195. [Google Scholar] [CrossRef]
- Moustafa, M.A.; El-Refaie, W.M.; Elnaggar, Y.S.; El-Mezayen, N.S.; Awaad, A.K.; Abdallah, O.Y. Fucoidan/hyaluronic acid cross-linked zein nanoparticles loaded with fisetin as a novel targeted nanotherapy for oral cancer. Int. J. Biol. Macromol. 2023, 241, 124528. [Google Scholar] [CrossRef]
- Wang, P.; Kankala, R.K.; Fan, J.; Long, R.; Liu, Y.; Wang, S. Poly-L-ornithine/fucoidan-coated calcium carbonate microparticles by layer-by-layer self-assembly technique for cancer theranostics. J. Mater. Sci. Mater. Med. 2018, 29, 68. [Google Scholar] [CrossRef] [PubMed]
- Abdelkader, D.H.; Elekhnawy, E.; Negm, W.A.; El-Masry, T.A.; Almukainzi, M.; Zayed, A.; Ulber, R. Insight into Fucoidan-Based PEGylated PLGA Nanoparticles Encapsulating Methyl Anthranilic Acid: In Vitro Evaluation and In Vivo Anti-Inflammatory Study. Mar. Drugs 2022, 20, 694. [Google Scholar] [CrossRef]
- Lai, Y.H.; Chiang, C.S.; Hsu, C.H.; Cheng, H.W.; Chen, S.Y. Development and Characterization of a Fucoidan-Based Drug Delivery System by Using Hydrophilic Anticancer Polysaccharides to Simultaneously Deliver Hydrophobic Anticancer Drugs. Biomolecules 2020, 10, 970. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Chen, J.K.; Lam, U.I.; Chen, S.Y. Preparing, characterizing, and evaluating chitosan/fucoidan nanoparticles as oral delivery carriers. J. Polym. Res. 2014, 21, 415. [Google Scholar] [CrossRef]
- Tsai, L.C.; Chen, C.H.; Lin, C.W.; Ho, Y.C.; Mi, F.L. Development of mutlifunctional nanoparticles self-assembled from trimethyl chitosan and fucoidan for enhanced oral delivery of insulin. Int. J. Biol. Macromol. 2019, 126, 141–150. [Google Scholar] [CrossRef]
- Chen, C.H.; Lin, Y.S.; Wu, S.J.; Mi, F.L. Mutlifunctional nanoparticles prepared from arginine-modified chitosan and thiolated fucoidan for oral delivery of hydrophobic and hydrophilic drugs. Carbohydr. Polym. 2018, 193, 163–172. [Google Scholar] [CrossRef]
- Yu, S.H.; Tang, D.W.; Hsieh, H.Y.; Wu, W.S.; Lin, B.X.; Chuang, E.Y.; Sung, H.W.; Mi, F.L. Nanoparticle-induced tight-junction opening for the transport of an anti-angiogenic sulfated polysaccharide across Caco-2 cell monolayers. Acta Biomater. 2013, 9, 7449–7459. [Google Scholar] [CrossRef]
- Lin, Y.H.; Lu, K.Y.; Tseng, C.L.; Wu, J.Y.; Chen, C.H.; Mi, F.L. Development of genipin-crosslinked fucoidan/chitosan-N-arginine nanogels for preventing Helicobacter infection. Nanomed 2017, 12, 1491–1510. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Huang, Y.C. Soluble eggshell membrane protein-loaded chitosan/fucoidan nanoparticles for treatment of defective intestinal epithelial cells. Int. J. Biol. Macromol. 2019, 131, 949–958. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, J.; Qin, Y.; Jiang, B.; Zhang, T. Zein/fucoidan-based composite nanoparticles for the encapsulation of pterostilbene: Preparation, characterization, physicochemical stability, and formation mechanism. Int. J. Biol. Macromol. 2020, 158, 461–470. [Google Scholar] [CrossRef]
- Haggag, Y.; Abu Ras, B.; El-Tanani, Y.; Tambuwala, M.M.; McCarron, P.; Isreb, M.; El-Tanani, M. Co-delivery of a RanGTP inhibitory peptide and doxorubicin using dual-loaded liposomal carriers to combat chemotherapeutic resistance in breast cancer cells. Expert Opin. Drug Deliv. 2020, 17, 1655–1669. [Google Scholar] [CrossRef] [PubMed]
- Qadir, S.A.; Kwon, M.C.; Han, J.G.; Ha, J.H.; Jin, L.; Jeong, H.S.; Kim, J.C.; You, S.G.; Lee, H.Y. Enhancement of immunomodulatory and anticancer activity of fucoidan by nano encapsulation. Food Sci. Biotechnol. 2008, 17, 1254–1260. [Google Scholar]
- Lima Salviano, T.; dos Santos, M.; Charles, D.; de Siqueira Ferraz Carvalho, R.; Pereira, M.A.; de Arruda, B.; Santos, V.; dos Santos Aguiar, J.; Souto, F.O.; da Silva, C.; et al. Fucoidan-Coated Liposomes: A Target System to Deliver the Antimicrobial Drug Usnic Acid to Macrophages Infected with Mycobacterium tuberculosis. J. Biomed. Nanotechnol. 2021, 17, 1699–1710. [Google Scholar] [CrossRef]
- Zhang, X.; Wei, Z.; Xue, C. Physicochemical properties of fucoidan and its applications as building blocks of nutraceutical delivery systems. Crit. Rev. Food Sci. Nutr. 2021, 62, 8935–8953. [Google Scholar] [CrossRef]
- Choi, D.G.; Venkatesan, J.; Shim, M.S. Selective Anticancer Therapy Using Pro-Oxidant Drug-Loaded Chitosan-Fucoidan Nanoparticles. Int. J. Mol. Sci. 2019, 20, 3220. [Google Scholar] [CrossRef] [PubMed]
- Rioux, L.; Turgeon, S.L.; Beaulieu, M. Rheological characterisation of polysaccharides extracted from brown seaweeds. J. Sci. Food Agric. 2007, 87, 1630–1638. [Google Scholar] [CrossRef]
- Lu, K.Y.; Li, R.; Hsu, C.H.; Lin, C.W.; Chou, S.C.; Tsai, M.L.; Mi, F.L. Development of a new type of multifunctional fucoidan-based nanoparticles for anticancer drug delivery. Carbohydr. Polym. 2017, 165, 410–420. [Google Scholar] [CrossRef]
- Pawar, V.K.; Singh, Y.; Sharma, K.; Shrivastav, A.; Sharma, A.; Singh, A.; Meher, J.G.; Singh, P.; Raval, K.; Kumar, A.; et al. Improved chemotherapy against breast cancer through immunotherapeutic activity of fucoidan decorated electrostatically assembled nanoparticles bearing doxorubicin. Int. J. Biol. Macromol. 2019, 122, 1100–1114. [Google Scholar] [CrossRef]
- Jang, B.; Moorthy, M.S.; Manivasagan, P.; Xu, L.; Song, K.; Lee, K.D.; Kwak, M.; Oh, J.; Jin, J.O. Fucoidan-coated CuS nanoparticles for chemo- and photothermal therapy against cancer. Oncotarget 2018, 9, 12649–12661. [Google Scholar] [CrossRef]
- da Silva, L.C.; Garcia, T.; Mori, M.; Sandri, G.; Bonferoni, M.C.; Finotelli, P.V.; Cinelli, L.P.; Caramella, C.; Cabral, L.M. Preparation and characterization of polysaccharide-based nanoparticles with anticoagulant activity. Int. J. Nanomed. 2012, 7, 2975–2986. [Google Scholar] [CrossRef]
- Barbosa, A.I.; Costa Lima, S.S.; Reis, S. Development of methotrexate loaded fucoidan/chitosan nanoparticles with anti-inflammatory potential and enhanced skin permeation. Int. J. Biol. Macromol. 2019, 124, 1115–1122. [Google Scholar] [CrossRef]
- Huang, Y.C.; Lam, U.I. Chitosan/fucoidan pH sensitive nanoparticles for oral delivery system. J. Chin. Chem. Soc. 2011, 58, 779–785. [Google Scholar] [CrossRef]
- Guo, R.; Deng, M.; He, X.; Li, M.; Li, J.; He, P.; Liu, H.; Li, M.; Zhang, Z.; He, Q. Fucoidan-functionalized activated platelet-hitchhiking micelles simultaneously track tumor cells and remodel the immunosuppressive microenvironment for efficient metastatic cancer treatment. Acta Pharm. Sin. B 2022, 12, 467–482. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Kang, K.; Chae, A.; Kim, Y.K.; Jang, H.; Min, D.H. Fucoidan-coated coral-like Pt nanoparticles for computed tomography-guided highly enhanced synergistic anticancer effect against drug-resistant breast cancer cells. Nanoscale 2019, 11, 15173–15183. [Google Scholar] [CrossRef] [PubMed]
- Jafari, M.; Sriram, V.; Xu, Z.; Harris, G.M.; Lee, J.Y. Fucoidan-Doxorubicin Nanoparticles Targeting P-Selectin for Effective Breast Cancer Therapy. Carbohydr. Polym. 2020, 249, 116837. [Google Scholar] [CrossRef] [PubMed]
- DuRoss, A.N.; Landry, M.R.; Thomas, C.R.; Neufeld, M.J.; Sun, C. Fucoidan-coated nanoparticles target radiation-induced P-selectin to enhance chemoradiotherapy in murine colorectal cancer. Cancer Lett. 2021, 500, 208–219. [Google Scholar] [CrossRef]
- Chen, M.L.; Lai, C.J.; Lin, Y.N.; Huang, C.M.; Lin, Y.H. Multifunctional nanoparticles for targeting the tumor microenvironment to improve synergistic drug combinations and cancer treatment effects. J. Mater. Chem. 2020, 8, 10416–10427. [Google Scholar] [CrossRef]
- Oliveira, C.; Gonçalves, C.S.; Martins, E.P.; Neves, N.M.; Reis, R.L.; Costa, B.M.; Silva, T.H.; Martins, A. Fucoidan/chitosan nanoparticles functionalized with anti-ErbB-2 target breast cancer cells and impair tumor growth in vivo. Int. J. Pharm. 2021, 600, 120548. [Google Scholar] [CrossRef]
- Cavalcanti, I.D.L.; Ximenes, R.M.; Pessoa, O.D.L.; Magalhães, N.S.S.; Lira-Nogueira, M.C.B. Fucoidan-coated PIBCA nanoparticles containing oncocalyxone A: Activity against metastatic breast cancer cells. J. Drug Deliv. Sci. Technol. 2021, 65, 102698. [Google Scholar] [CrossRef]
- Shamay, Y.; Elkabets, M.; Li, H.; Shah, J.; Brook, S.; Wang, F.; Adler, K.; Baut, E.; Scaltriti, M.; Jena, P.V. P-selectin is a nanotherapeutic delivery target in the tumor microenvironment. Sci. Transl. Med. 2016, 8, 345. [Google Scholar] [CrossRef]
- Tsai, H.L.; Tai, C.J.; Huang, C.W.; Chang, F.R.; Yuan, J.; Wang, J.Y. Efficacy of low-molecular-weight fucoidan as a supplemental therapy in metastatic colorectal cancer patients: A double-blind randomized controlled trial. Mar. Drugs 2017, 15, 122. [Google Scholar] [CrossRef]
- Tomori, M.; Nagamine, T.; Miyamoto, T.; Iha, M. Effects of Ingesting Fucoidan Derived from Cladosiphon okamuranus Tokida on Human NK Cells: A Randomized, Double-Blind, Parallel-Group, Placebo-Controlled Pilot Study. Mar. Drugs 2021, 19, 340. [Google Scholar] [CrossRef]
- Ikeguchi, M.; Yamamoto, M.; Arai, Y.; Maeta, Y.; Ashida, K.; Katano, K.; Miki, Y.; Kimura, T. Fucoidan reduces the toxicities of chemotherapy for patients with unresectable advanced or recurrent colorectal cancer. Oncol. Lett. 2011, 2, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.; Yeh, Y.; Chen, P.; Chang, Y.; Chen, Y.; Li, C.; Su, W.; Chang, T.; Huang, C.; Wang, J. The auxiliary effects of low-molecular-weight fucoidan on the quality of life for locally advanced rectal cancer patients receiving neoadjuvant concurrent chemoradiotherapy before surgery: A double-blind, randomized, placebo- controlled study. Res. Sq. 2023, 2023, 1–25. [Google Scholar] [CrossRef]
- A Randomized, Double-Blind Study to Evaluate the Clinical Effect and Safety of Fucoidan in Patients with Squamous Cell Carcinomas of the Head and Neck. ClinicalTrials.gov Identifier: NCT04597476, Taiwan; 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04597476#contacts (accessed on 1 June 2023).
- Study of Oligo-Fucoidan in Advanced Hepatocellular Carcinoma. ClinicalTrials.gov Identifier: NCT04066660, China; 2019. Available online: https://clinicaltrials.gov/ct2/show/NCT04066 (accessed on 1 June 2023).
- To Evaluate the Quality of Life Improvement of Oral Oligo Fucoidan in Subjects Receiving Platinum-Based Chemotherapy with Non-Small Cell Lung Cancer. ClinicalTrials.gov Identifier: NCT03130829, Taiwan; 2019. Available online: https://clinicaltrials.gov/ct2/show/NCT03130829 (accessed on 1 June 2023).
- Tocaciu, S.; Oliver, L.J.; Lowenthal, R.M.; Peterson, G.M.; Patel, R.; Shastri, M.; McGuinness, G.; Olesen, I.; Fitton, J.H. The Effect of Undaria pinnatifida Fucoidan on the Pharmacokinetics of Letrozole and Tamoxifen in Patients with Breast Cancer. Integr. Cancer Ther. 2018, 17, 99–105. [Google Scholar] [CrossRef] [PubMed]
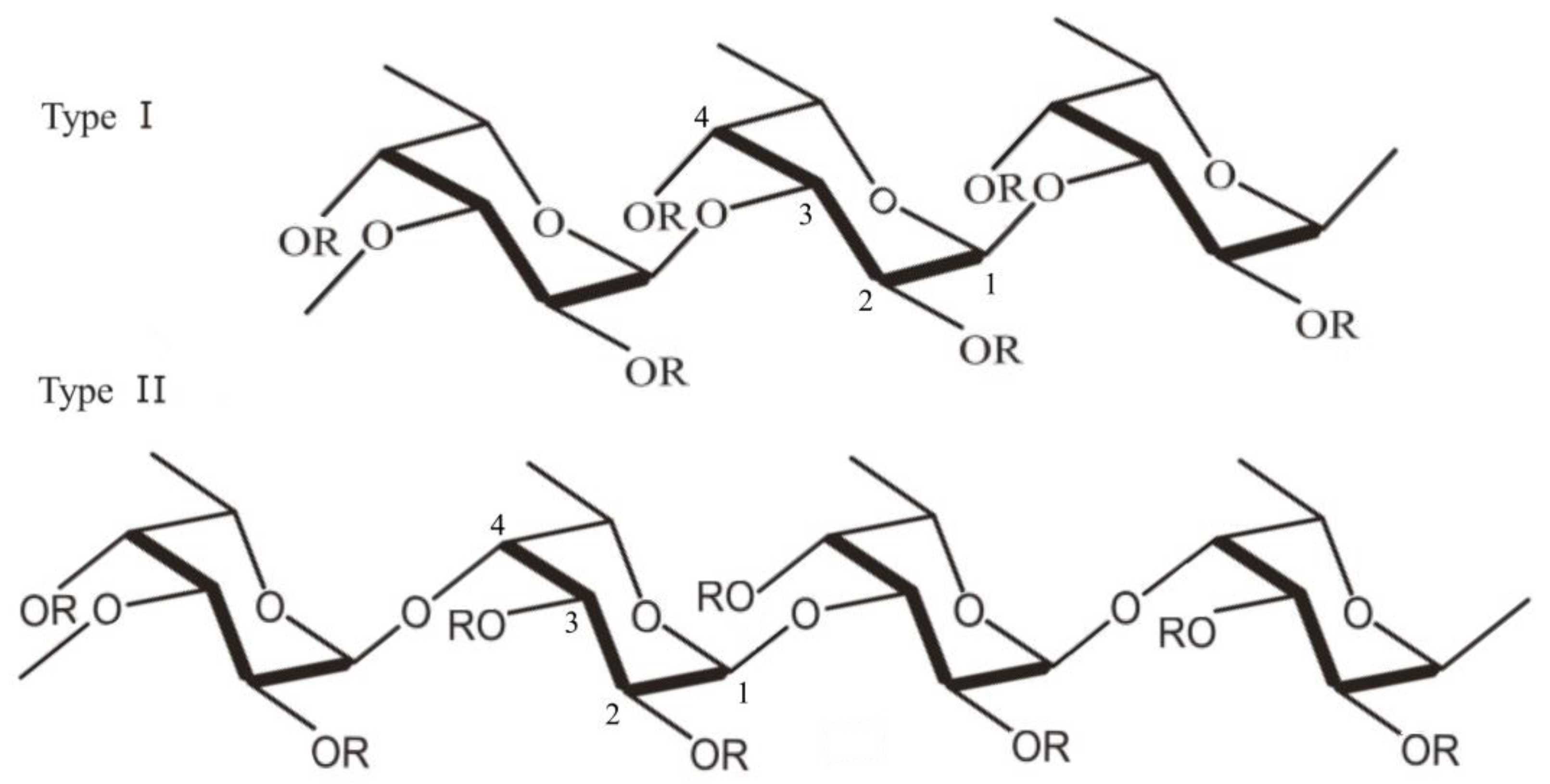



| Brown Seaweed | Structure | Reference |
|---|---|---|
| Fucus evanescens | Main chain of (1→3)-and (1→4)-α-L-Fucp highly substituted by sulfate groups at O2 and/or O3 positions | [6] |
| Fucus seratus | Main chain of (1→3)-and (1→4)-α-L-Fucp with short branches of α-L-Fucp-(1→4)-α-L-Fucp and α-L-Fucp-(1→3)-α-L-Fucp in O4 of α-(1→3)-L-Fucp and sulfate groups in O2 and/or O4 positions | [7] |
| Chorda filum | Main chain of (1→3)-α-L-Fucp highly ramified at O2 by terminal residues and substituted by sulfate groups at O2 and/or O4 positions. | [7] |
| Fucus distichus | [(1→3)-α-L-Fucp-(2,4-di-SO3-)-(1→4)- α-L-Fucp -(2SO3-)-(1→]n | [8] |
| Undaria pinnatifida | Backbone structure of (1→3):(1→4)-O-glycosidic bonds | [9] |
| Ascophyllum nodosum | [(1→3)-α-L-Fuc-(2SO3-)-(1→4)- α-L-Fuc-(2,3-di-SO3-)-(1→]n | [10] |
| Laminaria saccharina | Main chain of (1→3)-α-L-Fucp branched at O2 and O4 of α-L-Fucp by terminal residues and sulfate groups | [11] |
| Activity | Dose, µg/mL | Source | Cell Lines | Data Obtained | Reference |
|---|---|---|---|---|---|
| Antioxidant | 1 | S. japonica | - | DPPH radical and ABTS+ radical scavenging activity | [71] |
| Anti- inflammatory | 0.1–100 | S. sagamianum | LPS-induced RAW 264.7 cells | Inhibition of NO, IL-6, IL-1β, TNF-α, iNOS, COX-2, NF-κB p65 | [72] |
| Anti- inflammatory | 1–300 | G. pacificum | LPS-induced THP-1 cells | Protected the THP-1 cells against LPS-stimulated cytotoxicity | [73] |
| Anti- inflammatory | 3–25 | S. japonica | LPS-induced RAW 264.7 cells | Decreased production of NO and TNF-α, IL-1β and IL-6. | [74] |
| Anti- inflammatory | 25–100 | C. minima | LPS-induced RAW 264.7 cells | Inhibition of NO production and expression of PGE2 | [75] |
| Antiviral | 10–1000 | L. japonica | Vero E6 cell line from mouse macrophage | Reduced number of infected cells | [76] |
| Antiviral | 50–100 | H. elongata | Vero cell line from African green monkey | Inhibition of Herpes simplex virus type 1 intracellular replication | [77] |
| Hypoglycemic | 0.5–5 | L. japonica | Cell α-glucosidase | α-glucosidase inhibitory activity | [78] |
| Immuno- modulatory | 5–125 | S. fusiforme | B lymphocytes | Promote LPS-induced spleen lymphocyte proliferation | [79] |
| Anticancer | 25–100 | S. coreanum | HL-60, CT-26, B-16 and HeLa cell model | Cell apoptosis and activation of caspase-3 and cleavage of poly(ADP-ribose) polymerase | [80] |
| Anticancer | 120 | F. vesiculosus | HeLa G-63, Hep G2 and Chang liver cells model | Inhibition of cell proliferation | [81] |
| Anticancer | 10–500 | U. pinnatifida | Sarcomas and carcinosarcoma cell lines | Induced apoptosis | [82] |
| Anticancer | 0.05–100 | B. fuscopurpurea | Ovarian cancer cells | Inhibition of A2780, COC1, SKOV3, HO-8910 and OVCAR3 ovarian cancer cells proliferation | [83] |
| Effect | Dose | Source | Tested Model | Reported Results | Reference |
|---|---|---|---|---|---|
| Anticancer | 5 g/kg p.o. | C. okamuranus | Mice with induced colon cancer | Significantly suppressed tumor growth | [84] |
| Anticancer | 5 mg/kg i.p. | F. vesicolosus | Mice with induced breast cancer | Inhibition of angiogenesis and induction of apoptosis | [85] |
| Anticancer | 200 mg/kg i.p. | - | Mice with induced hepatocellular carcinoma | Inhibition of cancer cells proliferation | [86] |
| Anticancer | 0.25 mg/day i.v. | F. vesicolosus | Mice with induced breast cancer | Cancer metastasis prevention | [87] |
| Anticancer | 75 mg/kg p.o. | S. plagiophyllum | Mice with induced hepatocellular carcinoma | Inhibition of carcinogen metabolism | [88] |
| Anticancer | 100 mg/kg p.o. | C. okamuranus | Mice with induced sarcoma | Tumor growth reduction by NO produced by macrophages | [89] |
| Immuno- modulatory | 10 mg/kg i.v. | - | Rabbits with induced bacterial meningitis | Decreased influx of leukocytes into the cerebrospinal fluid | [90] |
| Immuno- modulatory | 50 mg/kg i.v. | - | Mice with L-selectin deficient | Mobilization of hematopoietic progenitor stem cells | [91] |
| Immuno- modulatory | 25 mg/kg i.v. | F. vesiculosus | Rats with induced myocarditis | Inhibition of extravasation of macrophages and CD4+ T cells | [92] |
| Immuno- modulatory | 200 mg/kg p.o. | - | Mice with induced leishmania infection | Th1 switch in Leishmania infection | [93] |
| Immuno- modulatory | 20 mg/kg i.p. | F. vesiculosus | Mice | Increased levels of TNF-α and IL-6 in spleens and blood serum | [94] |
| Immuno- modulatory | 20 mg/kg p.o. | U. pinnatifida | Mice with induced herpes simplex | Increased activity of NK cells | [95] |
| Immuno- modulatory | 50 mg/kg i.p. | M. pyrifera | Mice | Increased maturation and activation of NK cells | [96] |
| Immuno- modulatory | 500 mg/kg p.o. | U. pinnatifida | UVB-irradiated mouse skin | Lowered IFN-γ levels after irradiation; reduced skin edema | [97] |
| Anti- inflammantory | 25 mg/kg i.v. | F. vesiculosus | Mice with induced pancreatitis | Decreased levels of IL-1β, TNF-α and myeloperoxidase | [98] |
| Anti- inflammantory | 10–400 mg/kg p.o./i.p. | F. vesiculosus | Mice with induced acute colitis | Decreased levels of IL-1α, IL-1β and IL-10 | [99] |
| Anti- inflammantory | 50–100 mg/kg p.o. | L. japonica | Mice with induced liver damage | Reduced levels of TNF-α, IL-1β and IL-6 | [100] |
| Anti- inflammantory | 100 mg/kg p.o. | L. japonica | Mice with induced diabetes melitus | Reduced blood glucose level and serum levels of IL-1β, IL-6, TNF-α | [101] |
| Anti- inflammantory | 50–200 mg/kg p.o. | L. japonica | Mice with induced myocarditis | Decreased levels of TNF- α and IL-6; increased levels of IL-10 | [102] |
| Anti- inflammantory | 50–200 mg/kg p.o. | S. muticum | Mice with induced rheumatoid arthritis | Decreased levels of TNF-α, IFN-γ and IL-6 | [103] |
| Anti- inflammantory | - | C. okamuranus | Mice with induced chronic colitis | Decreased levels of IL-6 and increased levels of IL-10 | [75] |
| Anti- inflammantory | 12.5–50 μg/mL | C. minima | Zebrafish embryos | Decreased production of NO, ROS, COX-2, iNOS | [104] |
| Anti- inflammantory | 300 mg/mL p.o. | U. pinnatifida | Mice with induced rheumatoid arthritis | Reduced cartilage and bone destruction and inflammation | [105] |
| Anti- inflammantory | 2.5–300 mg/kg p.o. | T. ornata | Rats with induced arthritis | Decreased levels of TNF-α, IL-6 and PGE2 | [106] |
| Anti- inflammantory | 50 mg/kg p.o. | T. ornata | Mice with paw edema | Reduced the expression of genes of COX-2, IL-1β, the NF-κB signaling pathway | [107] |
| Source, Mw | Copolymer | Drug | Preparation Method | Administration Route | Reference |
|---|---|---|---|---|---|
| F. vesiculosus, 200–400 kDa | Gold nanoparticles | Doxorubicin | Electrostatic complexation | Ocular | [148] |
| L. japonica, 80 kDa | Protamine | Doxorubicin | Self-assembly | Intravenous | [178] |
| F. vesiculosus, 200–400 kDa | Polyethyleneimine | Doxorubicin | Coacervation | Intravenous | [179] |
| F. vesiculosus, 200–400 kDa | Polyallylamine HCl | Copper sulfide | Layer-by-layer | Intratumoral | [180] |
| F. vesiculosus, 200–400 kDa | Polyallylamine HCl | Methotrexate | Self-assembly | NA * | [149] |
| F. vesiculosus, 200–400 kDa | Chitosan/Chondroitin | Fucoidan | Coacervation | Oral | [181] |
| F. vesiculosus, 50–190 kDa | Chitosan | Methotrexate | Self-assembly | Topical | [182] |
| F. vesiculosus, 50–190 kDa | Chitosan | Curcumin | Self-assembly | Oral | [183] |
| Fucoidan Source | Cancer Type | Dose Applied | Study Type | Number of Patients | Clinical Results | Reference |
|---|---|---|---|---|---|---|
| LMW fucoidan from S. hemiphyllum | Metastatic colorectal cancer | 4 g/day | Randomized, double-blind, controlled trial | 54 | Improved disease control | [192] |
| LMW fucoidan from C. okamuranus | Advanced metastatic cancer | 400 mL/day | Open-label clinical study | 20 | Improved therapy | [67] |
| LMW fucoidan from C. okamuranus | NK cells from patients in remission | 1.5 g twice a day | Randomized double-blind placebo-controlled study | 39 | Enhanced NK cell activity | [193] |
| HMW fucoidan from C. okamuranus | Colorectal cancer | 150 mL/day | A randomized trial | 20 | Decreased general fatigue | [194] |
| Fucoidan (without clarification). | Rectal cancer | N/A | A double-blind randomized placebo-controlled study | 100 | N/A | [195] |
| Fucoidan as a dietary supplement | Squamous cell carcinoma | 4.4 g/day | A randomized double-blind study | 119 | Study not completed. | [196] |
| Fucoidan as a dietary supplement | Hepatocellular carcinoma | 4.4 g/day | A randomized double-blind study | 100 | Study not completed. | [197] |
| LMW fucoidan as a dietary supplement | Lung cancer | 4.4 g twice a day | A double-blind randomized controlled trial | N/A | Withdrawn. | [198] |
| LMW fucoidan from Undaria pinnatifida | Brest cancer | 500 mg twice a day | Open label non-crossover study | 20 | Co-administration with letrozole, tamoxifen. | [199] |
| LMW fucoidan from Nemacystis decipiens | Prostate, liver, breast, pancreatic cancer | 60–300 mL/day | Case report | 10 | Decreased tumor markers | [42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zahariev, N.; Katsarov, P.; Lukova, P.; Pilicheva, B. Novel Fucoidan Pharmaceutical Formulations and Their Potential Application in Oncology—A Review. Polymers 2023, 15, 3242. https://doi.org/10.3390/polym15153242
Zahariev N, Katsarov P, Lukova P, Pilicheva B. Novel Fucoidan Pharmaceutical Formulations and Their Potential Application in Oncology—A Review. Polymers. 2023; 15(15):3242. https://doi.org/10.3390/polym15153242
Chicago/Turabian StyleZahariev, Nikolay, Plamen Katsarov, Paolina Lukova, and Bissera Pilicheva. 2023. "Novel Fucoidan Pharmaceutical Formulations and Their Potential Application in Oncology—A Review" Polymers 15, no. 15: 3242. https://doi.org/10.3390/polym15153242






