Synthesis and Properties of Fully Biobased Crosslinked Starch Oleate Films
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Methods
2.2.1. Synthesis of Starch Oleate
2.2.2. HC Films
2.2.3. UVC Films
2.3. Characterization
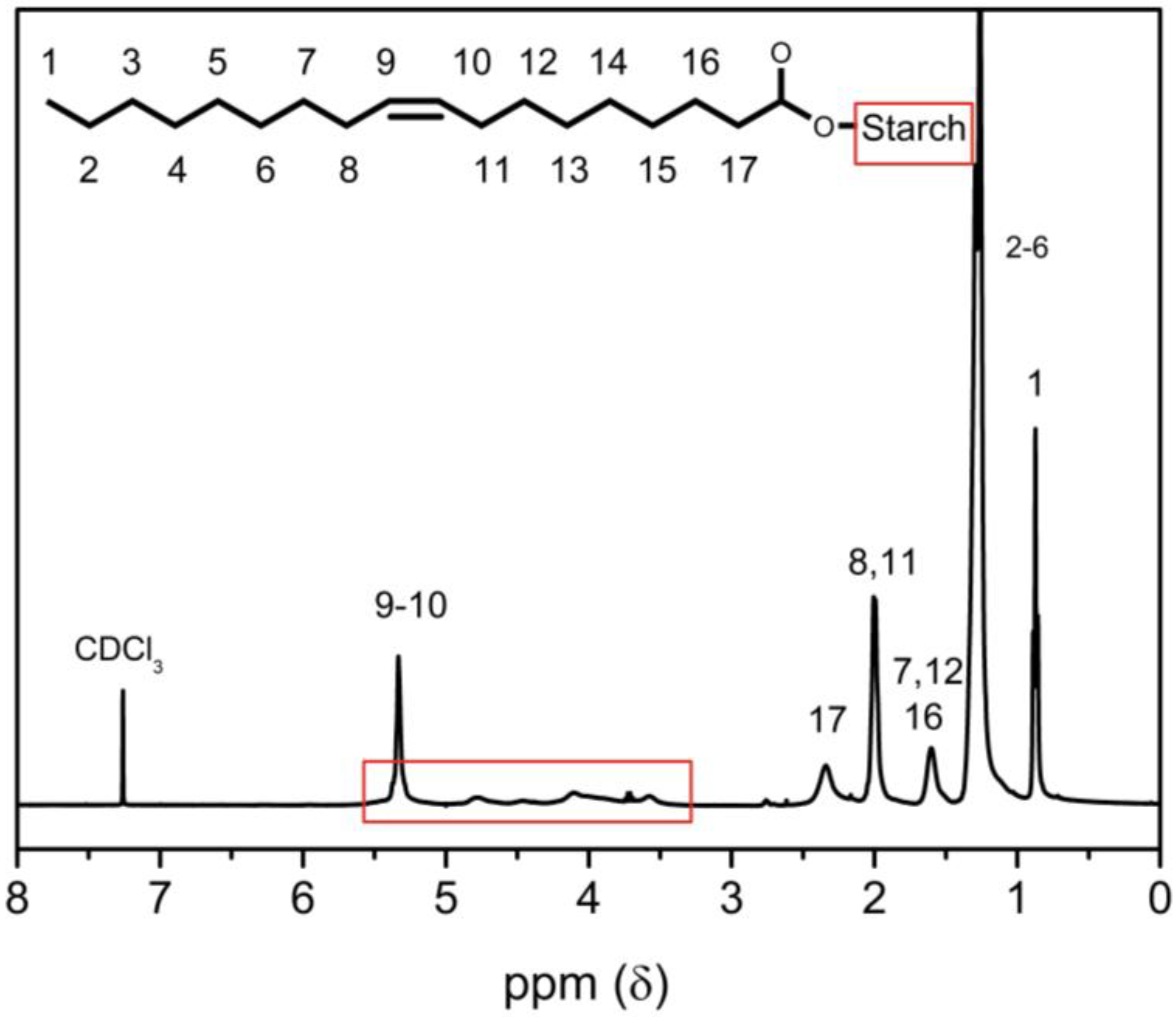
2.3.1. 1H-NMR Spectroscopy
2.3.2. Fourier Transform Infrared Spectroscopy (FTIR)
2.3.3. Film Gel Contents
2.3.4. Tensile Testing of Films
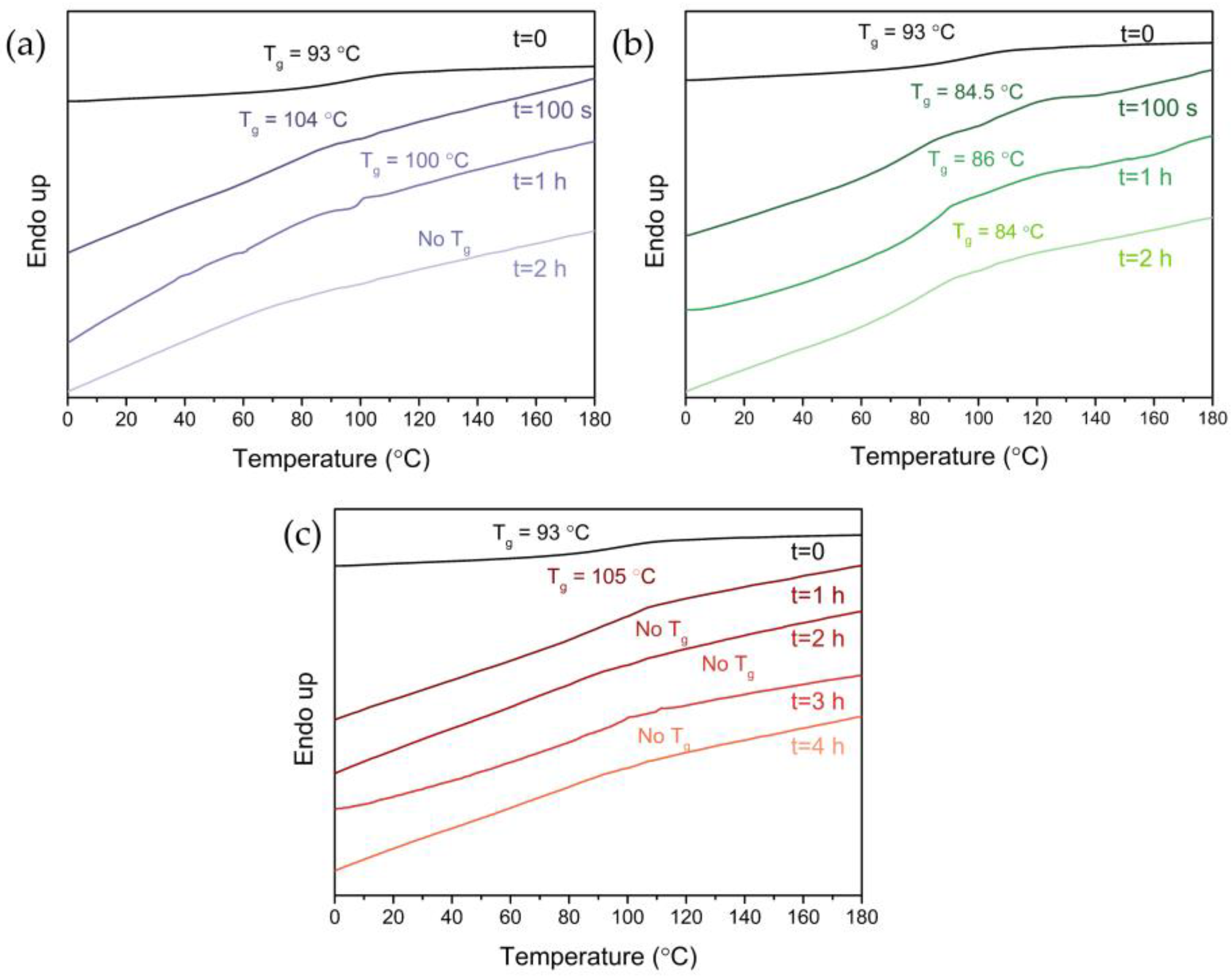
2.3.5. Differential Scanning Calorimetry (DSC)
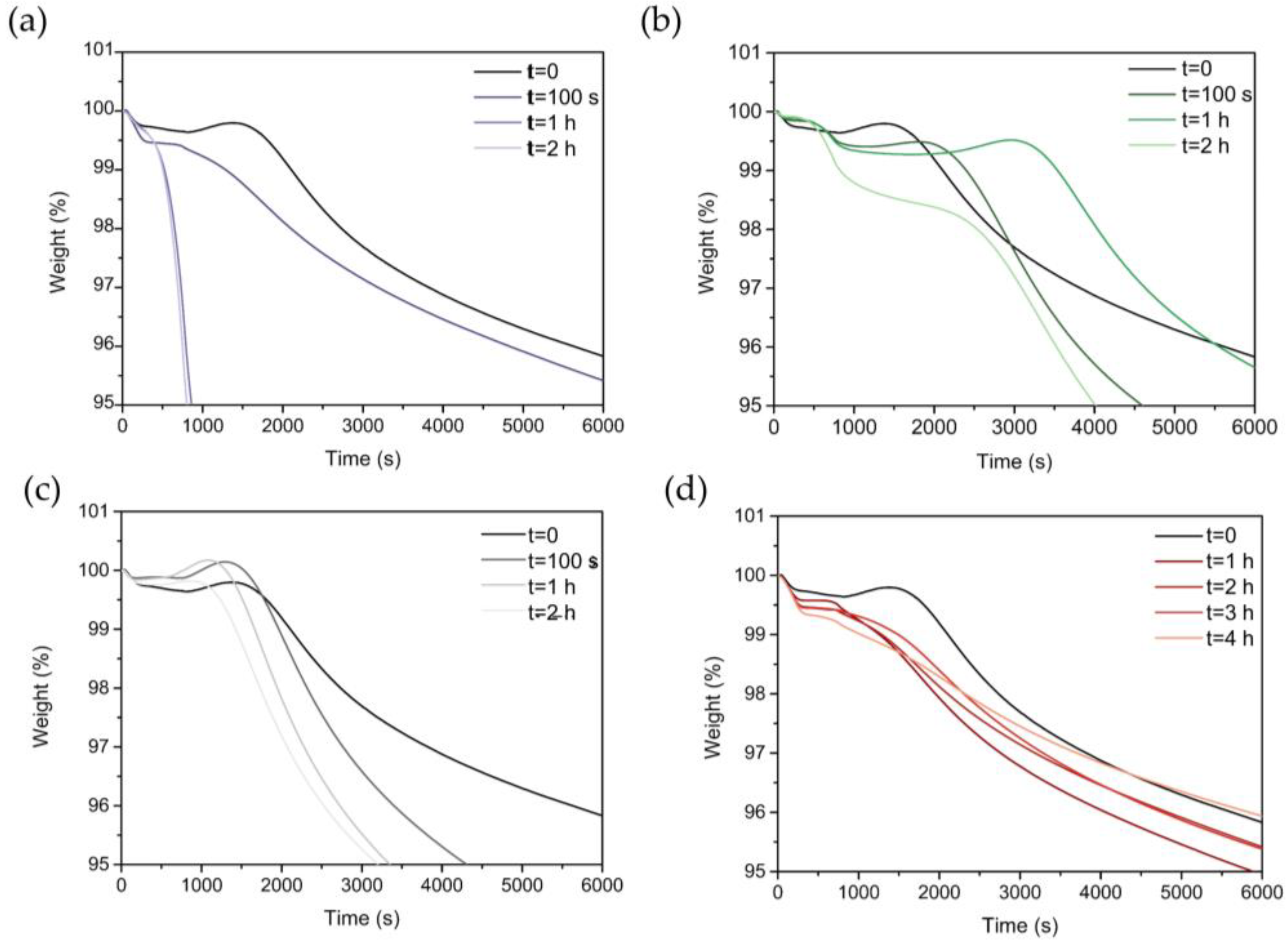
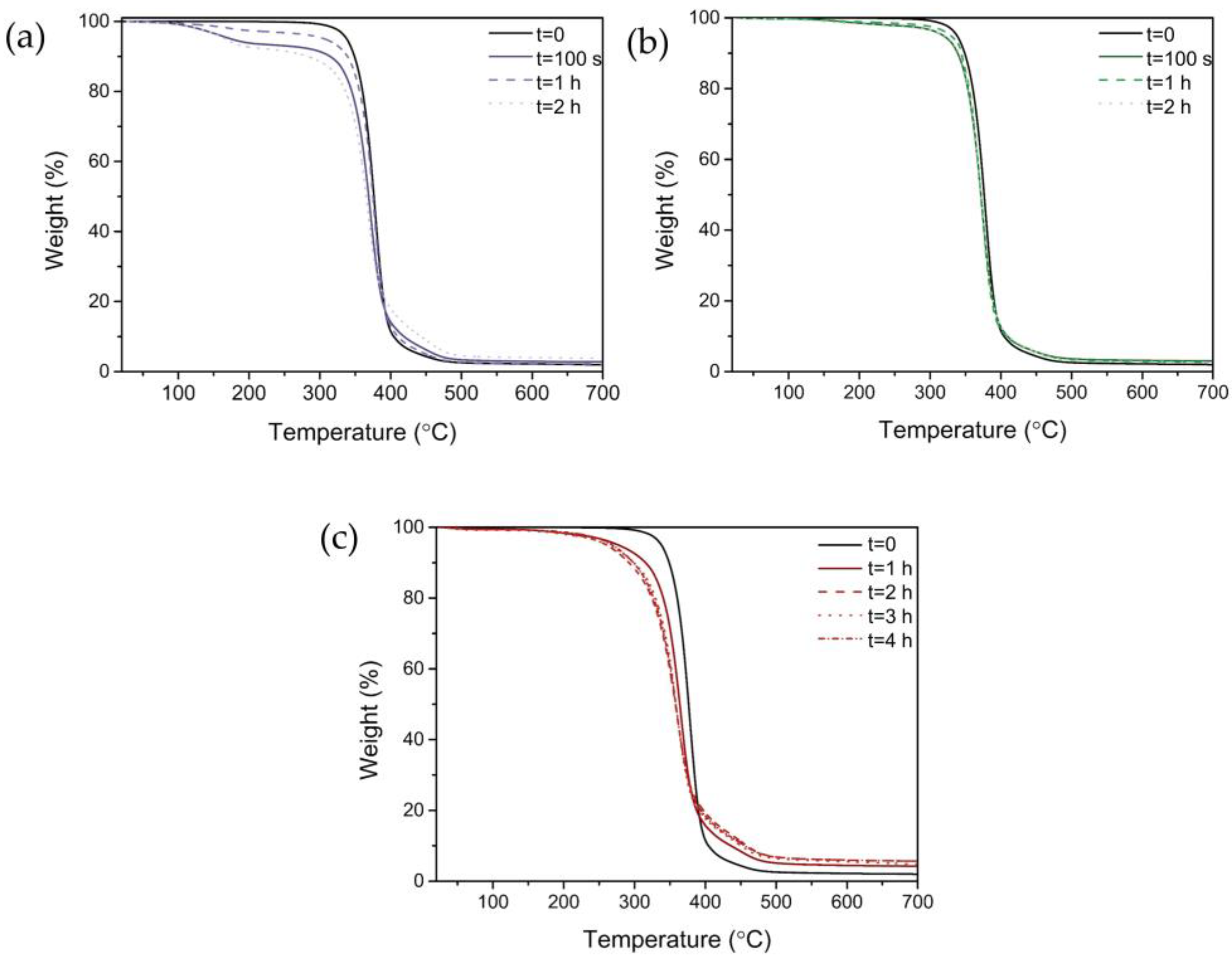
2.3.6. Thermogravimetric Analyses (TGA)
2.3.7. Contact Angle
2.3.8. Statistical Analyses
3. Results and Discussion
3.1. Starch Oleate Analysis
3.2. Film Appearance
3.3. Verification of Film Crosslinking
3.4. Mechanical Properties
3.5. Thermal Properties
3.6. Hydrophobicity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rosenboom, J.G.; Langer, R.; Traverso, G. Bioplastics for a Circular Economy. Nat. Rev. Mater. 2022, 7, 117–137. [Google Scholar] [CrossRef]
- Stevens, E.S. Plastics and the Environment. In Green Plastics: An introduction to the New Science of Biodegradable Plastics; Princeton Univerisity Press: Princeton, NJ, USA, 2002; pp. 15–30. [Google Scholar]
- Boetje, L.; Lan, X.; van Dijken, J.; Woortman, A.J.J.; Popken, T.; Polhuis, M.; Loos, K. The Effect of Casting Temperature and Starch Source on Starch Ester Film Properties. Zernike Institute for Advanced Materials, University of Groningen, Groningen, The Netherlands. Carbohydr. Polym. 2023. submitted. [Google Scholar]
- Imre, B.; Vilaplana, F. Organocatalytic Esterification of Corn Starches towards Enhanced Thermal Stability and Moisture Resistance. Green Chem. 2020, 22, 5017–5031. [Google Scholar] [CrossRef]
- Vanmarcke, A.; Leroy, L.; Stoclet, G.; Duchatel-Crépy, L.; Lefebvre, J.M.; Joly, N.; Gaucher, V. Influence of Fatty Chain Length and Starch Composition on Structure and Properties of Fully Substituted Fatty Acid Starch Esters. Carbohydr. Polym. 2017, 164, 249–257. [Google Scholar] [CrossRef]
- Winkler, H.; Vorwerg, W.; Rihm, R. Thermal and Mechanical Properties of Fatty Acid Starch Esters. Carbohydr. Polym. 2014, 102, 941–949. [Google Scholar] [CrossRef]
- Gan, Y.; Jiang, X. Photo-Cured Materials from Vegetable Oils. RSC Green Chem. 2015, 29, 1–27. [Google Scholar]
- Kalita, D.J.; Tarnavchyk, I.; Kalita, H.; Chisholm, B.J.; Webster, D.C. Bio-Based Coating Resins Derived from Cardanol Using Carbocationic Polymerization and Their Evaluation as One-Component Alkyd-Type Coatings. Prog. Org. Coat. 2023, 174, 107252. [Google Scholar] [CrossRef]
- Kalita, D.J.; Tarnavchyk, I.; Sibi, M.; Moser, B.R.; Webster, D.C.; Chisholm, B.J. Biobased Poly(Vinyl Ether)s Derived from Soybean Oil, Linseed Oil, and Camelina Oil: Synthesis, Characterization, and Properties of Crosslinked Networks and Surface Coatings. Prog. Org. Coat. 2018, 125, 453–462. [Google Scholar] [CrossRef]
- Frankel, E.N. Chemistry of Free Radical and Singlet Oxidation of Lipids. Prog. Lipid Res. 1984, 23, 197–221. [Google Scholar] [CrossRef]
- Frankel, E.N. Review. Recent Advances in Lipid Oxidation. J. Sci. Food Agric. 1991, 54, 495–511. [Google Scholar] [CrossRef]
- Laguerre, M.; Lecomte, J.; Villeneuve, P. Evaluation of the Ability of Antioxidants to Counteract Lipid Oxidation: Existing Methods, New Trends and Challenges. Prog. Lipid Res. 2007, 46, 244–282. [Google Scholar] [CrossRef]
- Mosiewicki, M.A.; Rojas, O.; Sibaja, R.; Borrajo, J.; Aranguren, M.I. Aging Study of Linseed Oil Resin/Styrene Thermosets and Their Composites with Wood Flour. Polym. Int. 2007, 881, 875–881. [Google Scholar] [CrossRef]
- Peng, X.; Zhu, D.; Xiao, P. Naphthoquinone Derivatives: Naturally Derived Molecules as Blue-Light-Sensitive Photoinitiators of Photopolymerization. Eur. Polym. J. 2020, 127, 109569. [Google Scholar] [CrossRef]
- Alsulami, Q.A.; Albukhari, S.M.; Hussein, M.A.; Tay, G.S.; Rozman, H.D. Biodegradable Lignin as a Reactive Raw Material in UV Curable Systems. Polym. Technol. Mater. 2020, 59, 1387–1406. [Google Scholar] [CrossRef]
- Decker, C. Photoinitiated Curing of Multifunctional Monomers. Acta Polym. 1994, 347, 333–347. [Google Scholar] [CrossRef]
- Ohtake, K.; Onose, Y.; Kuwabara, J.; Kanbara, T. Synthesis and Characterization of a Thermally Crosslinkable Polyolefin from Oleic Acid. J. Polym. Sci. Part A Polym. Chem. 2018, 57, 85–89. [Google Scholar] [CrossRef]
- Boetje, L.; Lan, X.; Silvianti, F.; van Dijken, J.; Polhuis, M.; Loos, K. A More Efficient Synthesis and Properties of Saturated and Unsaturated Starch Esters. Carbohydr. Polym. 2022, 292, 119649. [Google Scholar] [CrossRef]
- Mousawi, A.A.; Garra, P.; Schmitt, M.; Toufaily, J.; Hamieh, T.; Gra, B.; Fouassier, J.P.; Dumur, F.; Laleve, J. 3-Hydroxy Flavone and N-Phenylglycine in High Performance Photoinitiating Systems for 3D Printing and Photocomposites Synthesis. Macromolecules 2018, 51, 4633–4641. [Google Scholar] [CrossRef]
- Blohm, S.; Heinze, T. Synthesis and Properties of Thermoplastic Starch Laurates. Carbohydr. Res. 2019, 486, 107833. [Google Scholar] [CrossRef]
- Durand, P.; Chollet, G.; Cramail, H. Versatile Cross-Linked Fatty Acids-Based Polycarbonate Networks Obtained by Thiol–Ene Coupling Reaction. RSC Adv. 2018, 9, 145–150. [Google Scholar] [CrossRef]
- Kumarathasan, R.; Rajkumar, A.B.; Hunter, N.R.; Gesser, H.D. Autoxidation and Yellowing of Methyl Linolenate. Prog. Lipid Res. 1992, 31, 109–126. [Google Scholar] [CrossRef]
- Mallégol, J.; Lemaire, J.; Gardette, J.-L. Yellowing of Oil-Based Paints. Stud. Conserv. 2001, 46, 121–131. [Google Scholar]
- Bassas, M.; Marqués, A.M.; Manresa, A. Study of the Crosslinking Reaction (Natural and UV Induced) in Polyunsaturated PHA from Linseed Oil. Biochem. Eng. J. 2008, 40, 275–283. [Google Scholar] [CrossRef]
- Su, Y.; Lin, H.; Zhang, S.; Yang, Z.; Yuan, T. One-Step Synthesis of Novel Renewable Vegetable Oil-Based Acrylate Prepolymers and Their Application in UV-Curable Coatings. Polymers 2020, 12, 1165. [Google Scholar] [CrossRef]
- Winkler, H.; Vorwerg, W.; Wetzel, H. Synthesis and Properties of Fatty Acid Starch Esters. Carbohydr. Polym. 2013, 98, 208–216. [Google Scholar] [CrossRef]
- Shanti, R.; Hadi, A.N.; Salim, Y.S.; Chee, S.Y.; Ramesh, S.; Ramesh, K. Degradation of Ultrahigh Molecular Weight Poly(Methyl Methacrylate-Co-Butyl Acrylate-Co-Acrylic Acid) under Ultra Violet Irradiation. RSC Adv. 2017, 7, 112–120. [Google Scholar] [CrossRef]
- Kalita, D.J.; Tarnavchyk, I.; Selvakumar, S.; Chisholm, B.J.; Sibi, M.; Webster, D.C. Biobased Poly (Vinyl Ethers) Based on the Biomass-Derived Compound, Eugenol, and Their One-Component, Ambient-Cured Surface Coatings. Prog. Org. Coat. 2022, 170, 106996. [Google Scholar] [CrossRef]
- Frankel, E.N. Volatile Lipid Oxidation Products. Prog. Lipid Res. 1983, 22, 1–33. [Google Scholar] [CrossRef]
- Saad, B.; Wai, W.T.; Lim, B.P. Comparative Study on Oxidative Decomposition Behavior of Vegetable Oils and Its Correlation with Iodine Value Using Thermogravimetric Analysis. J. Oleo Sci. 2008, 57, 257–261. [Google Scholar] [CrossRef]
- Rathouský, J.; Kalousek, V.; Kolář, M.; Jirkovský, J.; Barták, P. A Study into the Self-Cleaning Surface Properties—The Photocatalytic Decomposition of Oleic Acid. Catal. Today 2011, 161, 202–208. [Google Scholar] [CrossRef]
- Manaila, E.; Craciun, G.; Ighigeanu, D.; Lungu, I.B.; Dumitru, M.; Stelescu, M.D. Electron Beam Irradiation: A Method for Degradation of Composites Based on Natural Rubber and Plasticized Starch. Polymers 2021, 13, 1950. [Google Scholar] [CrossRef]
- Zhou, X.; Ye, X.; He, J.; Wang, R.; Jin, Z. Effects of Electron Beam Irradiation on the Properties of Waxy Maize Starch and Its Films. Int. J. Biol. Macromol. 2020, 151, 239–246. [Google Scholar] [CrossRef]
- Hirschl, C.; Biebl-Rydlo, M.; Debiasio, M.; Mühleisen, W.; Neumaier, L.; Scherf, W.; Oreski, G.; Eder, G.; Chernev, B.; Schwab, W.; et al. Determining the Degree of Crosslinking of Ethylene Vinyl Acetate Photovoltaic Module Encapsulants—A Comparative Study. Sol. Energy Mater. Sol. Cells 2013, 116, 203–218. [Google Scholar] [CrossRef]
- Huang, J.; Fu, P.; Li, W.; Xiao, L.; Chen, J.; Nie, X. Influence of Crosslinking Density on the Mechanical and Thermal Properties of Plant Oil-Based Epoxy Resin. RSC Adv. 2022, 12, 23048–23056. [Google Scholar] [CrossRef]
- Huang, M.; Liu, Y.; Klier, J.; Schi, J.D. High-Performance, UV-Curable Cross-Linked Films via Grafting of Hydroxyethyl Methacrylate Methylene Malonate. Ind. Eng. Chem. Res. 2020, 59, 4542–4548. [Google Scholar] [CrossRef]
- Costas, G.B. Structures and Phase Transitions of Starch Polymers. In Polysaccharide Association Structures in Food, 1st ed.; Walter, R.H., Ed.; Marcel Dekker: New York, NY, USA, 1998; pp. 57–140. [Google Scholar]
- Bauer, F.; Flyunt, R.; Czihal, K.; Langguth, H.; Mehnert, R.; Schubert, R.; Buchmeiser, M.R. UV Curing and Matting of Acrylate Coatings Reinforced by Nano-Silica and Micro-Corundum Particles. Prog. Org. Coat. 2007, 60, 121–126. [Google Scholar] [CrossRef]
- NO Fang, J.M.; Fowler, P.A.; Tomkinson, J.; Hill, C.A.S. The Preparation and Characterisation of a Series of Chemically Modified Potato Starches. Carbohydr. Polym. 2002, 47, 245–252. [Google Scholar] [CrossRef]
- Majder-Łopatka, M.; Węsierski, T.; Ankowski, A.; Ratajczak, K.; Duralski, D.; Piechota-Polanczyk, A.; Polanczyk, A. Thermal Analysis of Plastics Used in the Food Industry. Materials 2022, 15, 248. [Google Scholar] [CrossRef]
- Haynes, W.M. CRC Handbook of Chemistry and Physics, 97th ed.; CRC Press: Boca Raton, FL, USA, 2016; pp. 15–21. [Google Scholar]
- Lease, J.; Kawano, T.; Andou, Y. Esterification of Cellulose with Long Fatty Acid Chain through Mechanochemical Method. Polymers 2021, 13, 4397. [Google Scholar] [CrossRef]




| Curing Method | Curing Time | Gel Content (%) |
|---|---|---|
| UV CPI | 1 h | 99.11 ± 0.80 a |
| HC | 4 h | 98.86 ± 1.98 a |
| HC | 3 h | 97.47 ± 2.41 a |
| HC | 2 h | 97.14 ± 3.74 a |
| UV CPI | 100 s | 97.03 ± 3.40 a |
| HC | 1 h | 96.06 ± 6.20 a |
| UV CPI | 2 h | 95.14 ± 3.25 a |
| UV NPI | 2 h | 85.30 ± 1.33 ab |
| UV NPI | 100 s | 74.66 ± 12.33 b |
| UV NPI | 1 h | 66.49 ± 3.23 bd |
| UV no PI | 2 h | 34.99 ± 3.43 c |
| UV no PI | 1 h | 16.19 ± 15.05 cd |
| UV no PI | 100 s | 13.04 ± 9.85 da |
| Non-crosslinked | - | 0 |
| Curing Method | Curing Time | Young’s Modulus (MPa) | Maximum Strength (MPa) | Maximum Elongation (%) |
|---|---|---|---|---|
| HC | 3 h | 130.00 ± 13.65 a | 7.37 ± 0.62 a | 17.01 ± 3.59 ab |
| HC | 4 h | 122.36 ± 3.75 a | 6.95 ± 0.39 a | 13.14 ± 1.71 b |
| HC | 1 h | 99.36 ± 2.88 b | 6.23 ± 0.19 b | 17.72 ± 1.34 ab |
| HC | 2 h | 98.37 ± 3.13 b | 6.26 ± 0.10 b | 19.18 ± 2.28 ab |
| UV NPI | 1 h | 74.05 ± 2.20 c | 4.96 ± 0.13 c | 24.21 ± 2.84 ab |
| UV CPI | 1 h | 72.90 ± 2.90 c | 4.76 ± 0.08 c | 22.24 ± 4.46 ab |
| UV NPI | 2 h | 72.54 ± 3.07 c | 4.64 ± 0.14 cde | 20.33 ± 4.36 ab |
| UV CPI | 100 s | 51.96 ± 1.90 d | 3.89 ± 0.06 e | 15.25 ± 1.28 ab |
| Non-crosslinked | - | 45.42 ± 0.98 de | 4.15 ± 0.10 de | 22.47 ± 9.29 ab |
| UV CPI | 2 h | 40.54 ± 1.54 e | 2.89 ± 0.14 f | 15.93 ± 5.67 ab |
| UV NPI | 100 s | 38.62 ± 4.72 e | 2.95 ± 0.08 f | 25.06 ± 10.59 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boetje, L.; Lan, X.; van Dijken, J.; Polhuis, M.; Loos, K. Synthesis and Properties of Fully Biobased Crosslinked Starch Oleate Films. Polymers 2023, 15, 2467. https://doi.org/10.3390/polym15112467
Boetje L, Lan X, van Dijken J, Polhuis M, Loos K. Synthesis and Properties of Fully Biobased Crosslinked Starch Oleate Films. Polymers. 2023; 15(11):2467. https://doi.org/10.3390/polym15112467
Chicago/Turabian StyleBoetje, Laura, Xiaohong Lan, Jur van Dijken, Michael Polhuis, and Katja Loos. 2023. "Synthesis and Properties of Fully Biobased Crosslinked Starch Oleate Films" Polymers 15, no. 11: 2467. https://doi.org/10.3390/polym15112467
APA StyleBoetje, L., Lan, X., van Dijken, J., Polhuis, M., & Loos, K. (2023). Synthesis and Properties of Fully Biobased Crosslinked Starch Oleate Films. Polymers, 15(11), 2467. https://doi.org/10.3390/polym15112467








