Gelatin/Na2Ti3O7 Nanocomposite Scaffolds: Mechanical Properties and Characterization for Tissue Engineering Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Mixture Design
2.3. Preparation of the Gelatin/Na2Ti3O7 Nanocomposite Scaffold Samples Using the Freeze-Drying Technique
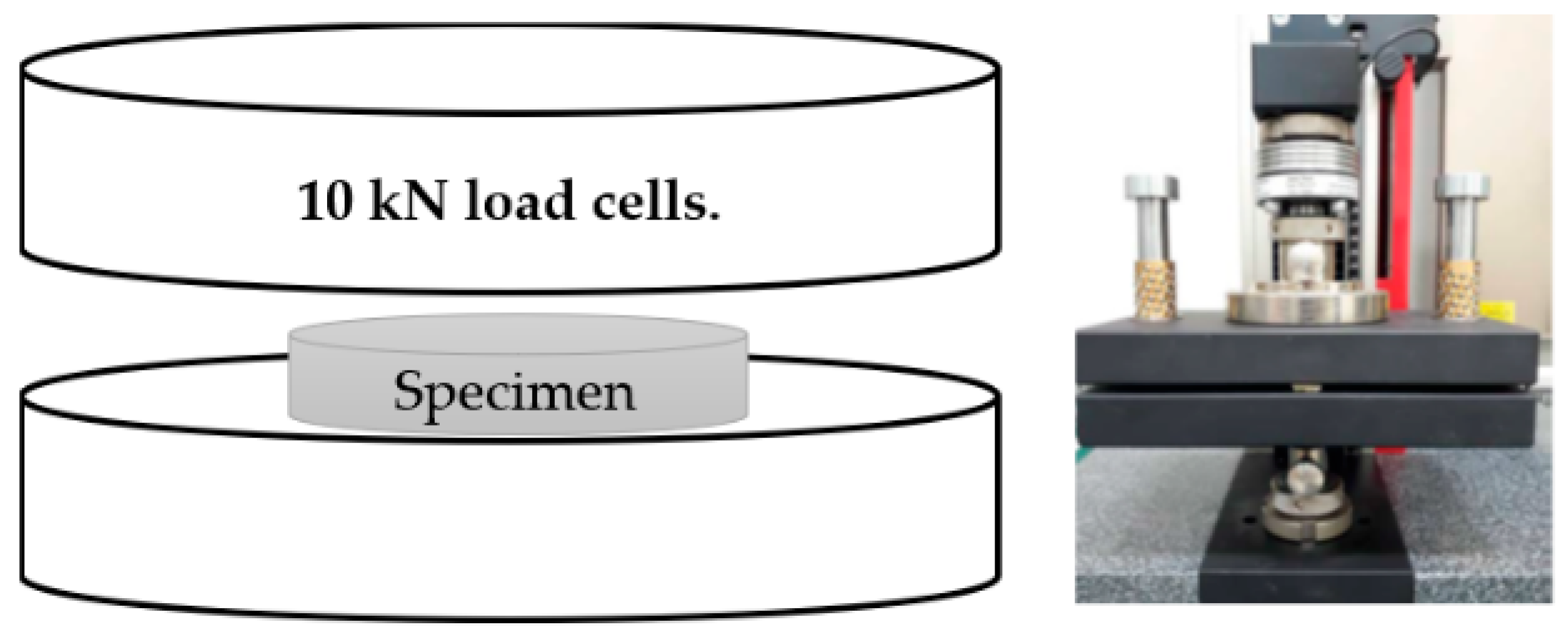
2.4. Compressive Test
2.5. Biodegradation of the Gelatin/Na2Ti3O7 Nanocomposite Scaffold Samples
- denotes the initial weight of the nanocomposite scaffold;
- denotes the final weight of the nanocomposite scaffold.
2.6. Swelling Test
- denotes the weight of the scaffold absorbing the water content;
- denotes the initial weight of the nanocomposite scaffold.
2.7. Statistical Analysis
3. Results and Discussion
3.1. Experimental Design and Results
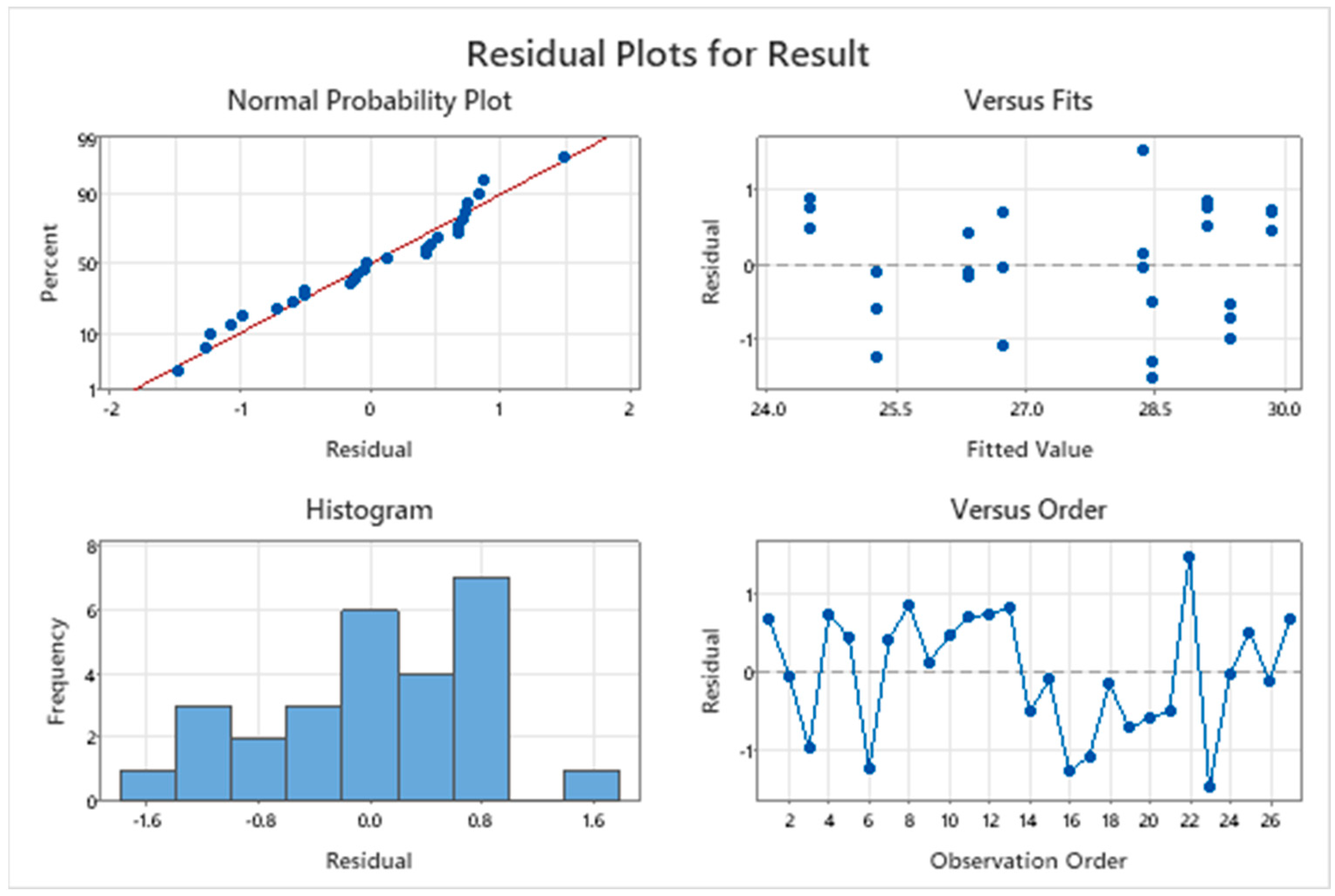
3.2. Residual Plots for Response
3.3. Analysis of Variance (ANOVA)
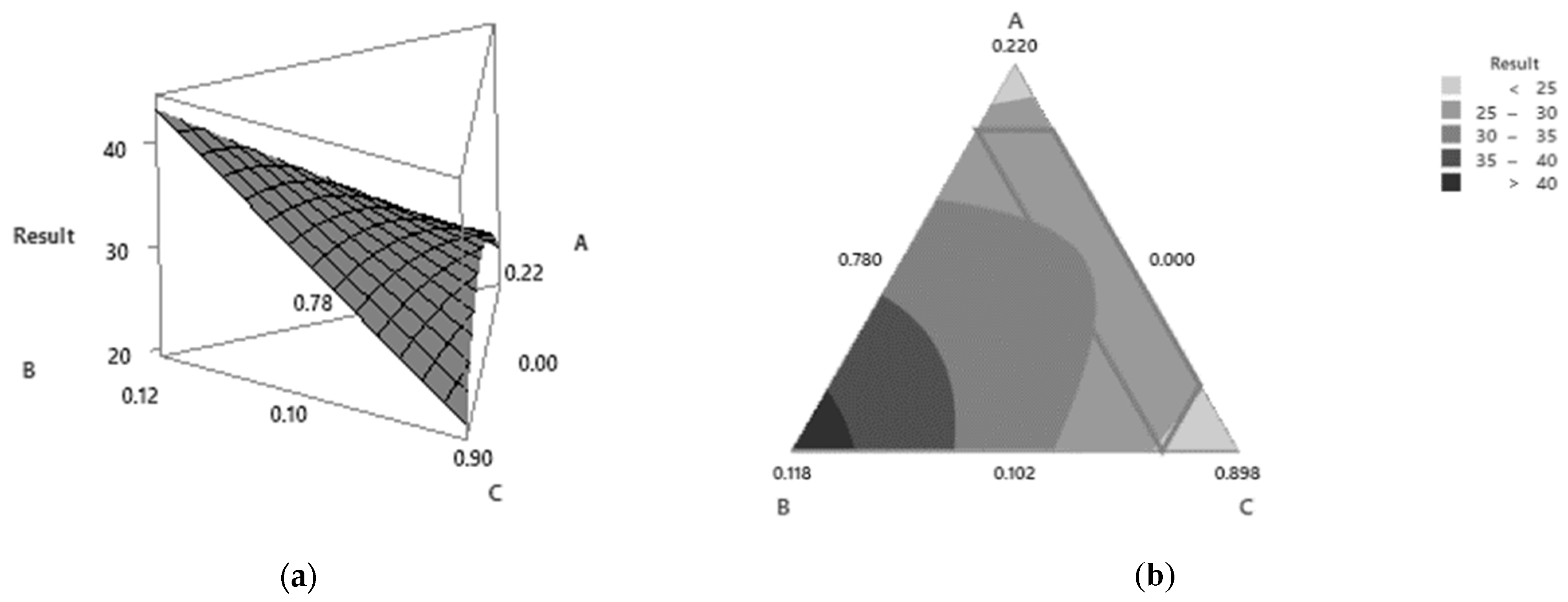
3.4. Results of the Response Surface Methodology Analysis
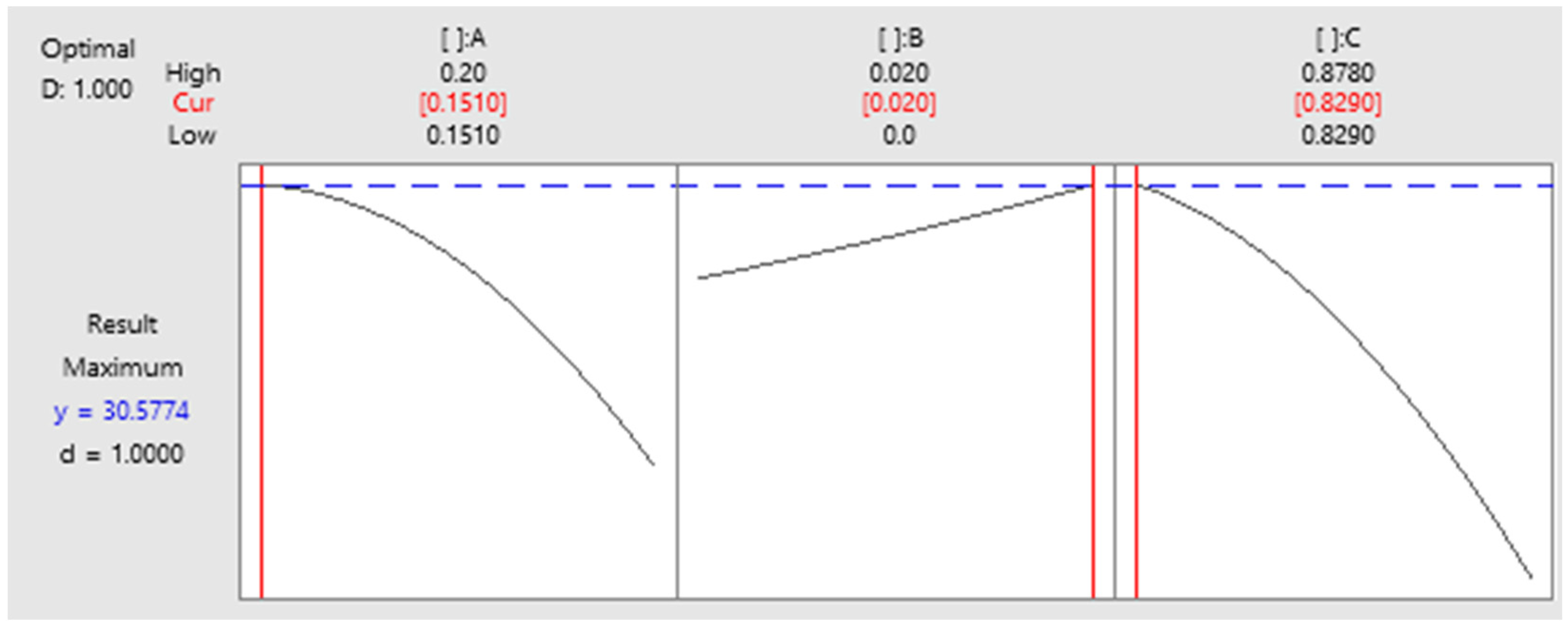
3.5. Optimization
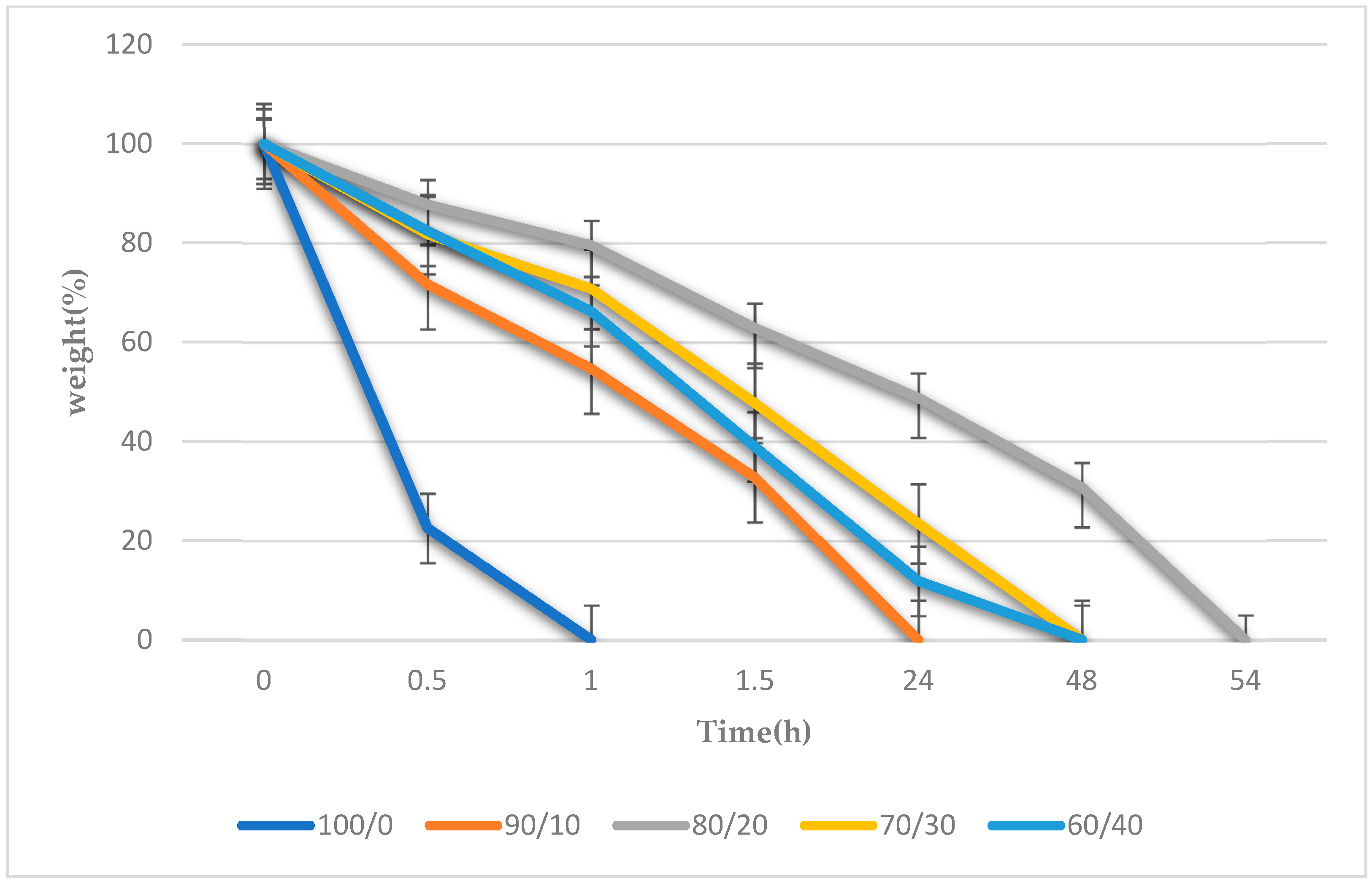
3.6. Biodegradation of the Gelatin/Na2Ti3O7 Nanocomposite Scaffold Samples Results
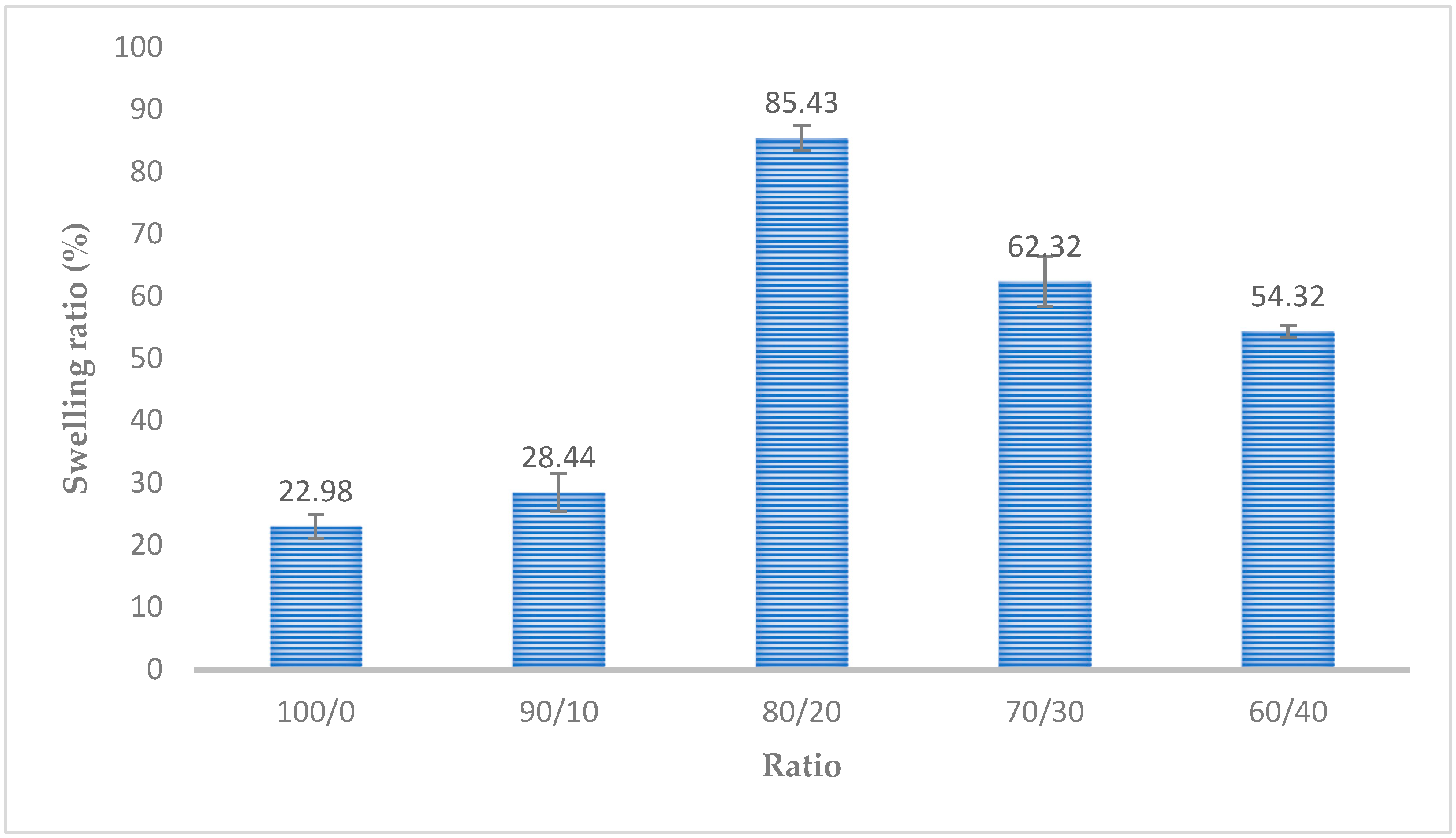
3.7. Swelling Test Results
3.8. Porosity Percent and Pore Size of the Gelatin/Na2Ti3O7 Nanocomposite Scaffolds Prepared Using the Freeze-Drying Technique
3.9. Surface Morphology of the Prepared Nanocomposite Scaffolds


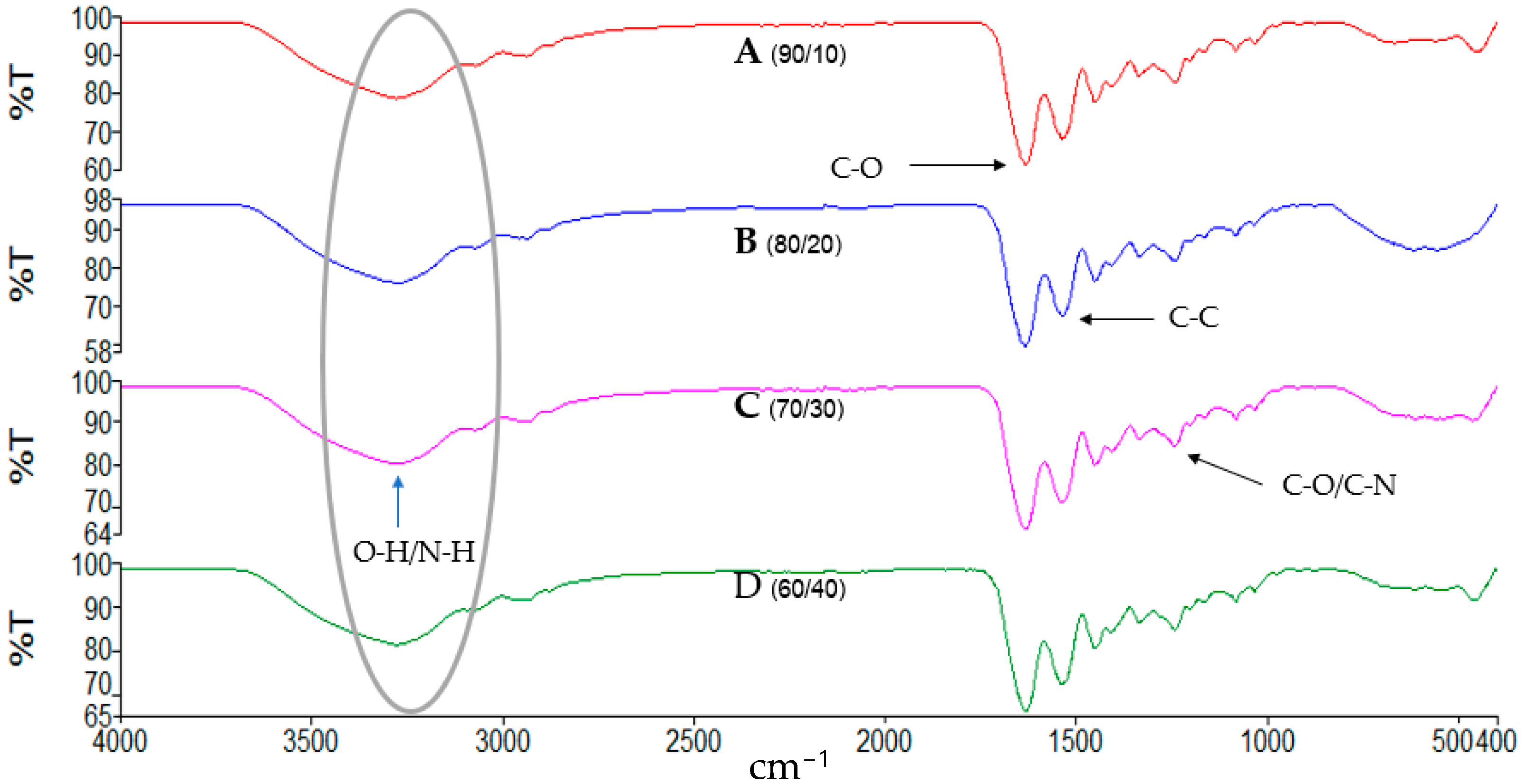
3.10. Characterization of the Gelatin/Na2Ti3O7 Nanocomposite Scaffolds
3.11. Effects of the Different Techniques Used for the Fabrication of a Scaffold: Freeze-Drying Technique and Salt-Leaching Technique
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, M.U.A.; Haider, S.; Shah, S.A.; Abd Razak, S.I.; Hassan, S.A.; Kadir, M.R.A.; Haider, A. Arabinoxylan-co-AA/HAp/TiO2 Nanocomposite Scaffold a Potential Material for Bone Tissue Engineering: An Invitro Study. Int. J. Biol. Macromol. 2020, 151, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.S.; Zhao, L.G.; Yin, Y.J.; De Yao, K. Structure and properties of bilayer chitosan–gelatin scaffolds. Biomaterials 2003, 24, 1067–1074. [Google Scholar] [CrossRef] [PubMed]
- Gomes, S.R.; Rodrigues, G.; Martins, G.G.; Henriques, C.M.R.; Silva, J.C. In vitro evaluation of crosslinked electrospun fish gelatin scaffolds. Mater. Sci. Eng. C 2013, 33, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Pezeshki-Modaress, M.; Mirzadeh, H. Gelatin–GAG electrospun nanofibrous scaffold for skin tissue engineering: Fabrication and modeling of process parameters. Mater. Sci. Eng. C 2015, 48, 704–712. [Google Scholar] [CrossRef]
- Zeugolis, D.I.; Khew, S.T.; Yew, E.S.Y.; Ekaputra, A.K.; Tong, Y.W.; Yung, L.-Y.L.; Hutmacher, D.W.; Sheppard, C.; Raghunath, M. Electrospinning of pure collagen nano-fibers Just an expensive way to make gelatin. Biomaterials 2008, 29, 2293–2305. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.Z.; Ramakrishna, S.; Lim, C.T. Electrospinning and mechanical characterization of gelatin nanofibers. Polymer 2004, 45, 5361–5368. [Google Scholar] [CrossRef]
- Pek, Y.S.; Spector, M.; Yannas, I.V.; Gibson, L.J. Degradation of a collagen–chondroitin-6-sulfate matrix by collagenase and by chondroitinase. Biomaterials 2004, 25, 473–482. [Google Scholar] [CrossRef]
- Genchi, G.; Nuhn, H.; Liakos, I.; Marino, A.; Marras, S.; Athanassiou, A.; Mattoli, V.; Desai, T. Titanium dioxide nanotube arrays coated with laminin enhance C2C12 skeletal myoblast adhesion and differentiation. RSC Adv. 2016, 22, 18502–18514. [Google Scholar] [CrossRef]
- Smith, B.S.; Yoriya, S.; Johnson, T.; Popat, K.C. Dermal fibroblast and epidermal keratinocyte functionality on titania nanotube arrays. Acta Biomater. 2011, 7, 2686–2696. [Google Scholar] [CrossRef]
- Fan, X.; Chen, K.; He, X.; Li, N.; Huang, J.; Tang, K.; Li, Y.; Wang, F. Nano-TiO2/collagen-chitosan porous scaffold for wound repairing. Int. J. Biol. Macromol. 2016, 91, 15–22. [Google Scholar] [CrossRef]
- Cordero-Arias, L.; Cabanas-Polo, S.; Gilabert, J.; Goudouri, O.M.; Sanchez, E.; Virtanen, S.; Boccaccini, A.R. Electrophoretic deposition of nanostructured TiO2/alginate and TiO2-bioactive glass/alginate composite coatings on stainless steel. Adv. Appl. Ceram. 2014, 113, 42–49. [Google Scholar] [CrossRef]
- Kulkarni, M.; Mazare, A.; Gongadze, E.; Perutkova, S.; Kralj-Iglic, V.; Milosev, I.; Schmuki, P.; Iglic, C.; Mozetic, M. Titanium nanostructures for biomedical applications. Nanotechnology 2015, 26, 062002. [Google Scholar] [CrossRef] [PubMed]
- Fu, S.-Y.; Lauke, B. Characterization of tensile behavior of hybrid short glass fiber/calcite particle/ABS composites. Compos. Part A: Appl. Sci. Manuf. 1998, 29, 575–583. [Google Scholar] [CrossRef]
- Lai, S.-M.; Li, H.-C.; Liao, Y.-C. Properties and preparation of compatibilized nylon 6 Nanocomposites/ABS blends: Part II—Physical and thermal properties. Eur. Polym. J. 2007, 43, 1660–1671. [Google Scholar] [CrossRef]
- Yousefi, M.; Salavati-Niasari, M.; Gholamian, F.; Ghanbari, D.; Aminifazl, A. Polymeric nanocomposite materials: Synthesis and thermal degradation of acrylonitrile–butadiene–styrene/tin sulfide (ABS/SnS). Inorg. Chim. Acta 2011, 371, 1–5. [Google Scholar] [CrossRef]
- Rezwan, K.; Chen, Q.Z.; Blaker, J.J.; Boccaccini, A.R. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials 2006, 27, 3413–3431. [Google Scholar] [CrossRef]
- Nam, Y.J.; Stöver, T.; Hartman, S.S.; Altschuler, R.A. Upregulation of glial cell line-derived neurotrophic factor (GDNF) in the rat cochlea following noise. Hear. Res. 2000, 146, 1–6. [Google Scholar] [CrossRef]
- Hou, Q.; Grijpma, D.W.; Feijen, J. Porous polymeric structures for tissue engineering prepared by coagulation, compression molding, and salt leaching technique. Biomaterials 2003, 24, 1937–1947. [Google Scholar] [CrossRef]
- Mikos, A.G.; Thorsen, A.J.; Czerwonka, L.A.; Bao, Y.; Langer, R.; Winslow, D.N.; Vacanti, J.P. Preparation and characterization of poly (L-lactic acid) foams. Polymer 1994, 35, 1068–1077. [Google Scholar] [CrossRef]
- Lo, H.; Ponticiello, M.S.; Leong, K.W. Fabrication of Controlled Release Biodegradable Foams by Phase Separation. Tissue Eng. 1995, 1, 18–25. [Google Scholar] [CrossRef]
- Mooney, D.J.; Baldwin, D.F.; Suh, N.P.; Vacanti, J.P.; Langer, R. A novel approach to fabricate porous sponges of poly (D, L-lactic-co-glycolic acid) without the use of organic solvents. Biomaterials 1996, 17, 1417–1422. [Google Scholar] [CrossRef]
- Leong, K.F.; Cheah, C.M.; Chua, C.K. Solid freeform fabrication of three-dimensional scaffolds for engineering replacement tissues and organs. Biomaterials 2003, 24, 2363–2378. [Google Scholar] [CrossRef]
- Whang, K.; Thomas, C.H.; Healy, K.E.; Nuber, G. A novel method to fabricate bioabsorbable scaffolds. Polymer 1995, 36, 837–842. [Google Scholar] [CrossRef]
- Hoque, M.E.; Nuge, T.; Yeow, T.K.; Nordin, N.; Prasad, R.G.S.V. Gelatin Based Scaffolds for Tissue Engineering—A review. Polym. Res. J. 2015, 9, 15–32. [Google Scholar]
- Cho, Y.S.; Kim, B.S.; You, H.K.; Cho, Y.S. A novel technique for scaffold fabrication: SLUP (salt leaching using powder). Curr. Appl. Phys. 2014, 14, 371–377. [Google Scholar] [CrossRef]
- Bruton, A.R. Manufacturing, and Performance of Titanium Dioxide Ultra-High Molecular Weight Polyethylene nanocomposite Materials. Master’s Thesis, University of Delaware, Newark, DE, USA, 2006. [Google Scholar]
- Giraldo, L.F.; Echeverri, M.; López, B.L. Reinforcement of Polyamide 6 with Nanoparticles. Macromol. Symp. 2007, 258, 119–128. [Google Scholar] [CrossRef]
- Zhang, Z.; Yang, J.L.; Friedrich, K. Creep resistant polymeric nanocomposites. Polymer 2004, 45, 3481–3485. [Google Scholar] [CrossRef]
- Matsuyama, T.; Tomoda, R.; Nakajima, T.; Wake, H. Photo electrochemical sterilization of microbial cells by semiconductor powders. FEMS Microbial. Lett. 1985, 29, 211–214. [Google Scholar] [CrossRef]
- Kiema, G.K.; Colgan, M.J.; Brett, M.J. Dye-sensitized solar cells incorporating obliquely deposited titanium oxide layers. Sol. Energy Mater. Sol. Cells 2005, 85, 321–331. [Google Scholar] [CrossRef]
- Walsh, F.C.; Bavykin, D.V.; Torrente-Murciano, L.; Lapkin, A.A.; Cressey, B.A. Synthesis of novel composite materials via the deposition of precious metals onto protonated titanate (TiO2) nanotubes. Trans. Inst. Met. Finish. 2006, 6, 293–299. [Google Scholar] [CrossRef]
- Bavykin, D.V.; Parmon, V.N.; Lapkin, A.A.; Walsh, F.C. The effect of hydrothermal condition on the mesoporous structure of TiO2 nanotube. J. Mater. Chem. 2004, 14, 3370–3377. [Google Scholar] [CrossRef]
- Fu, Y.; Wei, Z.D.; Chen, S.G.; Li, L.; Feng, Y.C.; Wang, Y.Q.; Ma, X.L.; Liao, M.J.; Shen, P.K.; Jiang, S.P. Synthesis of Pd/TiO2 nanotubes/Ti for oxygen reduction reaction in acidic solution. J. Power Sources 2009, 189, 982–987. [Google Scholar] [CrossRef]
- Dechakiatkrai, C.; Chen, J.; Lynam, C.; Phanichphant, S.; Wallace, G.G. Photocatalytic degradation of methanol using titanium dioxide/single walled carbon nanotube composite. J. Electrochem. Soc. 2007, 154, A407–A411. [Google Scholar] [CrossRef]
- Madhugiri, S.; Sun, B.; Smirniotis, P.G.; Ferraris, J.P.; Balkus, K.J., Jr. Electrospun mesoporous titanium dioxide fibers. Microporous Mesoporous Mater. 2004, 69, 77–83. [Google Scholar] [CrossRef]
- Saujanya, C.; Radhakrishnan, S. Structure development and crystallization behavior of PP/nanoparticulate composite. Polymer 2001, 42, 6723–6731. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhengb, Y.; Ning, R. Effects of nanoparticles SiO2 on the performance of nanocomposites. Mater. Lett. 2003, 57, 2940–2944. [Google Scholar] [CrossRef]
- Esthappan, S.K.; Kuttappan, S.K.; Joseph, R. Effect of titanium dioxide on the thermal aging of polypropylene. Polym. Degrad. Stab. 2012, 97, 615–620. [Google Scholar] [CrossRef]
- Akkapeddi, M.K. Glass fiber reinforced polyamide-6 nanocomposites. Polym. Compos. 2000, 21, 576–585. [Google Scholar] [CrossRef]
- Kasuga, T.; Hiramatsu, M.; Hoson, A.; Sekino, T.; Niihara, K. Formation of titanium oxide nanotube. Langmuir 1998, 14, 3160–3163. [Google Scholar] [CrossRef]
- Kasuga, T.; Hiramatsu, M.; Hoson, A.; Sekino, T.; Niihara, K. Titania nanotubes prepared by chemical processing. Adv. Mater. 1999, 11, 1307–1311. [Google Scholar] [CrossRef]
- Chen, Q.; Zhou, W.; Du, G.H.; Peng, L.M. Titanate nanotubes made via a single alkali treatment. Adv. Mater. 2002, 14, 1208–1211. [Google Scholar] [CrossRef]
- Kiatkittipong, K.; Scott, J.; Amal, R. Hydrothermally synthesized titanate nanostructures: Impact of heat treatment on particle characteristics and photocatalytic properties. ACS Appl. Mater. Interfaces 2011, 3, 3988–3996. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.Y.; Colomer, J.F.; Su, B.L. Titanium oxide nanoribbons. Chem. Phys. Lett. 2002, 363, 362–366. [Google Scholar] [CrossRef]
- Kiatkittipong, K.; Ye, C.; Scott, J.; Amal, R. Understanding Hydrothermal Titanate Nanoribbon Formation. Cryst. Growth Des. 2010, 10, 3618–3625. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Li, G.H.; Jin, Y.X.; Zhang, Y.; Zhang, J.; Zhang, L.D. Hydrothermal synthesis and photoluminescence of TiO2 nanowires. Chem. Phys. Lett. 2002, 365, 300–304. [Google Scholar] [CrossRef]
- Yoshida, R.; Suzuki, Y.; Yoshikawa, S. Syntheses of TiO2 (B) nanowires and TiO2 anatase nanowires by hydrothermal and post-heat treatments. J. Solid-State Chem. 2005, 178, 2179–2185. [Google Scholar] [CrossRef]
- Armstrong, A.R.; Armstrong, G.; Canales, J.; Bruce, P.G. TiO2 -B nanowires. Angew. Chem. Int. Ed. 2004, 43, 2286–2288. [Google Scholar] [CrossRef]
- Byrappa, K.; Yoshimura, M. Handbook of Hydrothermal Technology—A Technology for Crystal Growth and Materials Processing; William Andrew Publishing/Noyes: Norwich, NY, USA, 2001. [Google Scholar]
- Ma, R.; Bando, Y.; Sasaki, T. Nanotubes of lepidocrocite titanates. Chem. Phys. Lett. 2003, 380, 577–582. [Google Scholar] [CrossRef]
- Sangkatip, R.; Sriseubsai, W.; Kiatkittipong, K.; Jongwuttanaruk, K. Optimization of Salt-Leaching Parameters for Gelatin/Na2Ti3O7 Scaffolds Using a Mixture Design Experiment. Polymers 2022, 14, 559. [Google Scholar] [CrossRef]
- Sangkatip, R.; Sriseubsai, W.; Kiatkittipong, K. Preparation, and Characterization of the Na2Ti3O7: ABS/Na2Ti3O7 Composites. Key Eng. Mater. 2017, 753, 39–43. [Google Scholar] [CrossRef]
- Myers, R.H.; Montgomery, D.C. Response Surface Methodology Process and Product Optimization Using Designed Experiments, 2nd ed.; John Wiley and Son, Inc.: New York, NY, USA, 2002. [Google Scholar]
- Hajiali, H.; Karbasi, S.; Hosseinalipour, M.; Rezaie, H. Preparation of a novel biodegradable nanocomposite scaffold based on poly (3-hydroxybutyrate)/bioglass nanoparticles for bone tissue engineering. J. Mater. Sci. Mater. Med. 2010, 21, 2125–2132. [Google Scholar] [CrossRef]
- Chen, H.; Huang, J.; Yu, J.; Liu, S.; Gu, P. Electrospun chitosan-graft-poly (ε-caprolactone)/poly (ε-caprolactone) cationic nanofibrous mats as potential scaffolds for skin tissue engineering. Int. J. Biol. Macromol. 2011, 48, 13–19. [Google Scholar] [CrossRef]
- Mohd Amin, M.C.I.; Ahmad, N.; Halib, N.; Ahmad, I. Synthesis and characterization of thermo- and pH-responsive bacterial cellulose/acrylic acid hydrogels for drug delivery. Carbohydr. Polym. 2012, 88, 465–473. [Google Scholar] [CrossRef]
- Rokhade, A.P.; Agnihotri, S.A.; Patil, S.A.; Mallikarjuna, N.N.; Kulkarni, P.V.; Aminabhavi, T.M. Semi-interpenetrating polymer network microspheres of gelatin and sodium carboxymethyl cellulose for controlled release of ketorolac tromethamine. Carbohydr. Polym. 2006, 65, 243–252. [Google Scholar] [CrossRef]
- Ooi, S.Y.; Ahmad, I.; Amin, M.C.I.M. Cellulose nanocrystals extracted from rice husks as a reinforcing material in gelatin hydrogels for use in controlled drug delivery systems. Ind. Crops Prod. 2015, 93, 227–234. [Google Scholar] [CrossRef]
- Wiwatwongwana, F.; Promma, N. Characterization of Gelatin-Carboxymethylcellulose Scaffolds. In Proceedings of the 4th International Conference on Chemical Materials and Process (ICCMP 2018), 100th Building, Faculty of Engineering, Chulalongkorn University, Bangkok, Thailand, 23–25 May 2018. (Oral presentation). [Google Scholar]
- Jongwuttanaruk, K.; Surin, P.; Wiwatwongwana, F. Characterization of Gelatin/CMC Scaffolds by Electrospinning and Comparison with Freeze Dry Techniques. In Proceedings of the 4th International Conference on Chemical Materials and Process (ICCMP 2018), 100th Building, Faculty of Engineering, Chulalongkorn University, Bangkok, Thailand, 23–25 May 2018. (Oral presentation). [Google Scholar]
- Ma, J.; Wang, H.; He, B.; Chen, J. A preliminary in vitro study on the fabrication and tissue engineering applications of a novel chitosan bilayer material as a scaffold of human no fetal dermal fibroblasts. Biomaterials 2001, 22, 331–336. [Google Scholar] [CrossRef]
- Leea, S.B.; Kima, Y.H.; Chonga, M.S.; Hong, S.H.; Leea, Y.M. Study of gelatin-containing artificial skin V: Fabrication of gelatin scaffolds using a salt-leaching method. Biomaterials 2005, 26, 1961–1968. [Google Scholar] [CrossRef]

| Factors | Factor Levels | |
|---|---|---|
| Low Level | High Level | |
| A: Gelatin (g) | 0 | 20 |
| B: Na2Ti3O7 (g) | 0 | 2 |
| C: DI water (mL) | 0 | 87.8 |
| Run Order | Factors | Compressive Test (kPa.) | ||
|---|---|---|---|---|
| A: Gelatin | B: Na2Ti3O7 | C: DI Water | ||
| 01 | 0.200 | 0.000 | 0.800 | 27.407 |
| 02 | 0.200 | 0.002 | 0.798 | 26.764 |
| 03 | 0.122 | 0.000 | 0.878 | 24.678 |
| 04 | 0.120 | 0.002 | 0.878 | 25.240 |
| 05 | 0.161 | 0.001 | 0.839 | 30.306 |
| 06 | 0.180 | 0.001 | 0.819 | 29.861 |
| 07 | 0.180 | 0.002 | 0.818 | 28.396 |
| 08 | 0.141 | 0.001 | 0.858 | 28.511 |
| 09 | 0.140 | 0.002 | 0.858 | 27.186 |
| 10 | 0.200 | 0.000 | 0.800 | 26.687 |
| 11 | 0.200 | 0.002 | 0.798 | 26.222 |
| 12 | 0.122 | 0.000 | 0.878 | 24.038 |
| 13 | 0.120 | 0.002 | 0.878 | 25.370 |
| 14 | 0.161 | 0.001 | 0.839 | 30.586 |
| 15 | 0.180 | 0.001 | 0.819 | 29.621 |
| 16 | 0.180 | 0.002 | 0.818 | 28.866 |
| 17 | 0.141 | 0.001 | 0.858 | 28.337 |
| 18 | 0.140 | 0.002 | 0.858 | 26.974 |
| 19 | 0.200 | 0.000 | 0.800 | 25.647 |
| 20 | 0.200 | 0.002 | 0.798 | 26.182 |
| 21 | 0.122 | 0.000 | 0.878 | 25.170 |
| 22 | 0.120 | 0.002 | 0.878 | 24.970 |
| 23 | 0.161 | 0.001 | 0.839 | 30.546 |
| 24 | 0.180 | 0.001 | 0.819 | 29.941 |
| 25 | 0.180 | 0.002 | 0.818 | 28.659 |
| 26 | 0.141 | 0.001 | 0.858 | 29.871 |
| 27 | 0.140 | 0.002 | 0.858 | 27.953 |
| Source | DF | Seq SS | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|---|
| Regression | 3 | 87.933 | 87.933 | 29.3110 | 41.96 | 0.000 |
| Linear | 2 | 10.864 | 75.028 | 37.5141 | 53.70 | 0.000 |
| Quadratic | 1 | 77.069 | 77.069 | 77.0688 | 110.32 | 0.000 |
| A*C | 1 | 77.069 | 77.069 | 77.0688 | 110.32 | 0.000 |
| Residual Error | 23 | 16.067 | 16.067 | 0.6986 | ||
| Lack of Fit | 5 | 11.408 | 11.408 | 2.2817 | 8.82 | 0.000 |
| Pure Error | 18 | 4.659 | 4.659 | 0.2588 | ||
| Total | 26 | 104.000 | ||||
| S | R-sq | R-sq(adj) | PRESS | R-sq(pred) | ||
| 0.835807 | 84.55% | 82.54% | 21.6100 | 79.22% | ||
| Gelatin/Na2Ti3O7 | Pore Size (µm) | Porosity (%) |
|---|---|---|
| 100/0 | 124.23 ± 2.65 | 67.96 ± 2.77 |
| 90/10 | 195.32 ± 2.52 | 83.43 ± 0.82 |
| 80/20 | 278.71 ± 2.52 | 86.42 ± 1.08 |
| 70/30 | 257.79 ± 2.08 | 85.44 ± 1.20 |
| 60/40 | 234.56 ± 3.51 | 84.63 ± 2.30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sangkatip, R.; Jongwuttanaruk, K.; Sriseubsai, W. Gelatin/Na2Ti3O7 Nanocomposite Scaffolds: Mechanical Properties and Characterization for Tissue Engineering Applications. Polymers 2023, 15, 2322. https://doi.org/10.3390/polym15102322
Sangkatip R, Jongwuttanaruk K, Sriseubsai W. Gelatin/Na2Ti3O7 Nanocomposite Scaffolds: Mechanical Properties and Characterization for Tissue Engineering Applications. Polymers. 2023; 15(10):2322. https://doi.org/10.3390/polym15102322
Chicago/Turabian StyleSangkatip, Rittichai, Kaona Jongwuttanaruk, and Wipoo Sriseubsai. 2023. "Gelatin/Na2Ti3O7 Nanocomposite Scaffolds: Mechanical Properties and Characterization for Tissue Engineering Applications" Polymers 15, no. 10: 2322. https://doi.org/10.3390/polym15102322
APA StyleSangkatip, R., Jongwuttanaruk, K., & Sriseubsai, W. (2023). Gelatin/Na2Ti3O7 Nanocomposite Scaffolds: Mechanical Properties and Characterization for Tissue Engineering Applications. Polymers, 15(10), 2322. https://doi.org/10.3390/polym15102322










