Microstructural Characterization of Alkali-Activated Composites of Lightweight Aggregates (LWAs) Embedded in Alkali-Activated Foam (AAF) Matrices
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
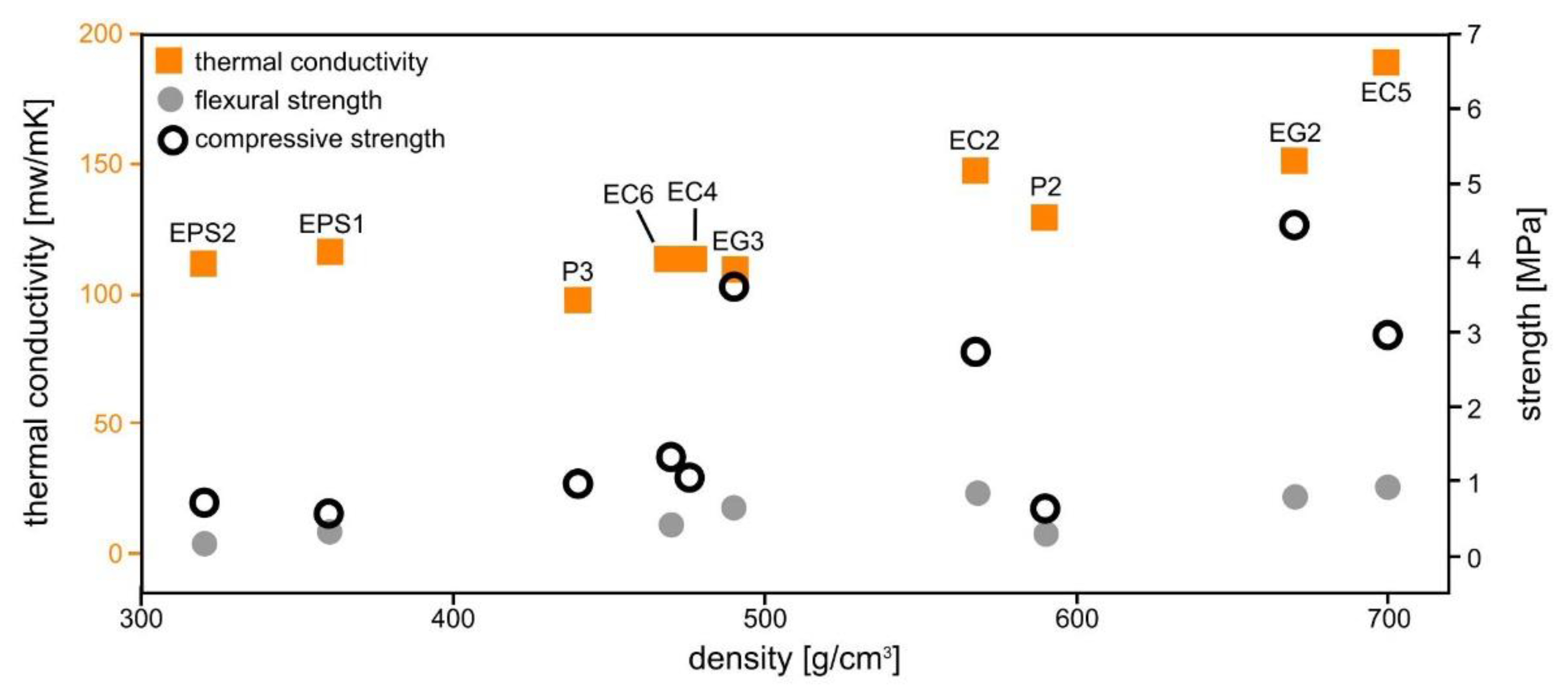
3.1. Mechanical Properties
3.2. SEM Analysis
3.3. Thermal Conductivity
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Provis, J.L.; van Deventer, J.S.J. (Eds.) Alkali Activated Materials: State-of-the-Art Report; RILEM TC 224-AAM; Springer/RILEM: Berlin, Germany, 2013. [Google Scholar]
- Bai, C.Y.; Colombo, P. Processing, properties and applications of highly porous geopolymers: A review. Ceram. Int. 2018, 44, 16103–16118. [Google Scholar] [CrossRef]
- Papa, E.; Landi, E.; Miccio, F.; Medri, V. K2O-Metakaolin-Based Geopolymer Foams: Production, Porosity Characterization and Permeability Test. Materials 2022, 15, 1008. [Google Scholar] [CrossRef] [PubMed]
- Prud’homme, E.; Joussein, E.; Rossignol, S. Use of silicon carbide sludge to form porous alkali-activated materials for insulating application. Eur. Phys. J. Spec. Top. 2015, 224, 1725–1735. [Google Scholar] [CrossRef]
- Ducman, V.; Korat, L. Characterization of geopolymer fly-ash based foams obtained with the addition of Al powder or H2O2 as foaming agents. Mater. Charact. 2016, 113, 207–213. [Google Scholar] [CrossRef]
- Rao, P.R.; Muralidharan, K.; Momayez, M.; Runge, K.A.; Loy, D.A. Direct foaming driven synthesis and thermophysical characterization of silica-alumina foams: Applications for thermal insulation. Ceram. Int. 2020, 46, 10431–10441. [Google Scholar] [CrossRef]
- Korat, L.; Ducman, V. The influence of the stabilizing agent SDS on porosity development in alkali-activated fly-ash based foams. Cem. Concr. Comp. 2017, 80, 168–174. [Google Scholar] [CrossRef]
- Traven, K.; Češnovar, M.; Škapin, S.D.; Ducman, V. Mechanical and Microstructural Properties of Fly Ash-Based Alkali-Activated Foams Exposed to Room and Elevated Temperatures. In Sheffield, United Kingdom, Book of abstracts of the 74th RILEM Annual Week & the 40th Cement and Concrete Science Conference [Elektronski vir]; The University of Sheffield: Sheffield, UK, 2020. [Google Scholar]
- Zhang, Z.H.; Provis, J.L.; Reid, A.; Wang, H. Geopolymer foam concrete: An emerging material for sustainable construction. Constr. Build. Mater. 2014, 56, 113–127. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; Ngo, T.; Mendis, P. Enhancing the strength of pre-made foams for foam concrete applications. Cem. Concr. Compos. 2018, 87, 164–171. [Google Scholar] [CrossRef]
- Chandra, S.; Berntsson, L. Lightweight Aggregate Concrete; Elsevier: London, UK, 2002. [Google Scholar] [CrossRef]
- Cheeseman, C.R.; Makinde, A.; Bethanis, S. Properties of lightweight aggregate produced by rapid sintering of incinerator bottom ash. Resour. Conserv. Recycl. 2005, 43, 147–162. [Google Scholar] [CrossRef]
- Frankovič, A.; Bokan-Bosiljkov, V.; Ducman, V. Lightweight aggregates made from fly ash using the cold-bond process and their use in lightweight concrete. Mater. Tehnol. 2017, 51, 267–274. [Google Scholar] [CrossRef]
- Korat, L.; Ducman, V.; Legat, A.; Mirtič, B. Characterisation of the pore-forming process in lightweight aggregate based on silica sludge by means of X-ray micro-tomography (micro-CT) and mercury intrusion porosimetry (MIP). Ceram. Int. 2013, 39, 6997–7005. [Google Scholar] [CrossRef]
- Howard, K. Method for Manufacturing Expanded Polystyrene Foam Components from Used Polystyrene Materials. USO05217660A. 1993. Available online: https://patentimages.storage.googleapis.com/38/74/c0/e31dc824c43b22/US5217660.pdf (accessed on 22 April 2022).
- Sulong, N.H.R.; Mustapa, S.A.S.; Rashid, M.K.A. Application of expanded polystyrene (EPS) in buildings and constructions: A review. J. Appl. Polym. Sci. 2019, 136, 47529. [Google Scholar] [CrossRef] [Green Version]
- Bui, L.A.; Hwang, C.; Chen, C.; Lin, K.; Hsieh, M. Manufacture and performance of cold bonded lightweight aggregate using alkaline activators for high performance concrete. Constr. Build. Mater. 2012, 35, 1056–1062. [Google Scholar] [CrossRef]
- Uttaravalli, A.N.; Dinda, S.; Gidla, B.R. Scientific and engineering aspects of potential applications of post-consumer (waste) expanded polystyrene: A review. Process Saf. Environ. Prot. 2020, 137, 140–148. [Google Scholar] [CrossRef]
- Posi, P.; Teerachanwit, C.; Tanutong, C.; Limkamoltip, S.; Lertnimoolchai, S.; Sata, V.; Chindaprasirt, P. Lightweight geopolymer concrete containing aggregate from recycle lightweight block. Mater. Des. 2013, 52, 580–586. [Google Scholar] [CrossRef]
- Mastali, M.; Kinnunen, P.; Isomoisio, H.; Karhu, M.; Illikainen, M. Mechanical and acoustic properties of fiber-reinforced alkali-activated slag foam concretes containing lightweight structural aggregates. Constr. Build. Mater. 2018, 187, 371–381. [Google Scholar] [CrossRef]
- Wongsa, A.; Sata, V.; Nuakiong, P.; Chindaprasirt, P. Use of crushed clay brick and pumice aggregates in lightweight geopolymer concrete. Constr. Build. Mater. 2018, 188, 1025–1034. [Google Scholar] [CrossRef]
- Medri, V.; Papa, E.; Mazzocchi, M.; Laghi, L.; Morganti, M.; Francisconi, J.; Landi, E. Production and characterization of lightweight vermiculite/geopolymer-based panels. Mater. Des. 2015, 85, 266–274. [Google Scholar] [CrossRef]
- Novais, R.M.; Senff, L.; Carvalheiras, J.; Seabra, M.P.; Pullar, R.C.; Labrincha, J.A. Sustainable and efficient cork-inorganic polymer composites: An innovative and eco-friendly approach to produce ultra-lightweight and low thermal conductivity materials. Cem. Concr. Compos. 2019, 97, 107–117. [Google Scholar] [CrossRef]
- Contrafatto, L.; Danzuso, C.L.; Gazzo, S.; Greco, L. Physical, mechanical and thermal properties of lightweight insulating mortar with recycled Etna volcanic aggregates. Constr. Build. Mater. 2020, 240, 117917. [Google Scholar] [CrossRef]
- Liao, Y.C.; Huang, C.Y.; Chen, Y.M. Lightweight aggregates from water reservoir sediment with added sodium hydroxide. Constr. Build. Mater. 2013, 46, 79–85. [Google Scholar] [CrossRef]
- Gao, H.; Liu, H.; Liao, L.B.; Mei, L.F.; Lv, G.C.; Liang, L.M.; Zhu, G.D.; Wang, Z.J.; Huang, D.L. Improvement of performance of foam perlite thermal insulation material by the design of a triple-hierarchical porous structure. Energy Build. 2019, 200, 21–30. [Google Scholar] [CrossRef]
- Vaou, V.; Panias, D. Thermal insulating foamy geopolymers from perlite. Miner. Eng. 2010, 23, 1146–1151. [Google Scholar] [CrossRef]
- Papa, E.; Medri, V.; Murri, A.N.; Laghi, L.; De Aloysio, G.; Bandini, S.; Landi, E. Characterization of alkali bonded expanded perlite. Constr. Build. Mater. 2018, 191, 1139–1147. [Google Scholar] [CrossRef]
- Duan, P.; Song, L.X.; Yan, C.J.; Ren, D.M.; Li, Z. Novel thermal insulating and lightweight composites from metakaolin geopolymer and polystyrene particles. Ceram. Int. 2017, 43, 5115–5120. [Google Scholar] [CrossRef]
- Shi, J.Y.; Liu, B.J.; Liu, Y.C.; Wang, E.L.; He, Z.H.; Xu, H.J.; Ren, X.D. Preparation and characterization of lightweight aggregate foamed geopolymer concretes aerated using hydrogen peroxide. Constr. Build. Mater. 2020, 256, 119442. [Google Scholar] [CrossRef]
- Rickard, W.D.A.; Gluth, G.J.G.; Pistol, K. In-situ thermo-mechanical testing of fly ash geopolymer concretes made with quartz and expanded clay aggregates. Cem. Concr. Res. 2016, 80, 33–43. [Google Scholar] [CrossRef]
- Rozek, P.; Krol, M.; Mozgawa, W. Lightweight geopolymer-expanded glass composites for removal of methylene blue from aqueous solutions. Ceram. Int. 2020, 46, 19785–19791. [Google Scholar] [CrossRef]
- Schumacher, K.; Saßmannshausen, N.; Pritzel, C.; Trettin, R. Lightweight aggregate concrete with an open structure and a porous matrix with an improved ratio of compressive strength to dry density. Constr. Build. Mater. 2020, 264, 120167. [Google Scholar] [CrossRef]
- Nicolas, R.S.; Provis, J.L. The interfacial Transition Zone in alkali-activated slag Mortars. Front. Mater. 2015, 2, 70. [Google Scholar] [CrossRef] [Green Version]
- Lee, W.K.W.; van Deventer, J.S.J. Chemical interactions between siliceous aggregates and low-Ca alkali-activated cements. Cem. Conc. Res. 2007, 37, 844–855. [Google Scholar] [CrossRef]
- Fang, G.; Zhang, M. The evolution of interfacial transition zone in alkali-activated fly ash-slag Concrete. Cem. Conc. Res. 2020, 129, 105963. [Google Scholar] [CrossRef]
- Traven, K.; Češnovar, M.; Škapin, S.D.; Ducman, V. Evaluation of Fly Ash Based Alkali-Activated Foams at Room and Elevated Temperatures; Bogataj, M., Kravanja, Z., Novak-Pintarič, Z., Eds.; University of Maribor Press: Portorož, Slovenia, 2020; pp. 75–84. [Google Scholar] [CrossRef]
- Ducman, V.; Traven, K.; Češnovar, M. Alkali-Activated Lightweight Composites Based on Alkali-Activated Foams and Manufacturing Process. Slovenian Patent SI 26042 (A), 28 February 2022. Available online: www3.uil-sipo.si/PublicationServer/documentpdf.jsp?iDocId=50296&iepatch=.pdf (accessed on 22 April 2022).
- Češnovar, M.; Traven, K.; Horvat, B.; Ducman, V. The Potential of Ladle Slag and Electric Arc Furnace Slag use in Synthesizing Alkali Activated Materials; The Influence of Curing on Mechanical Properties. Materials 2019, 12, 1173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Method of Testing Cement—Part 2: Chemical Analysis of Cement. EN 196-2; European Committee for Standardization: Bruxelles, Belgium, 2013.
- Masonry and Masonry Products—Methods for Determining Thermal Properties. EN 1745; European Committee for Standardization: Bruxelles, Belgium, 2020.
- Petrella, A.; de Mundo, R.; Notarnicola, M. Recycled Expanded Polystyrene as Lightweight Aggregate for Environmentally Sustainable Cement Conglomerates. Materials 2020, 13, 988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maaroufi, M.; Belarbi, R.; Abahri, K.; Benmahiddine, F. Full characterization of hygrothermal, mechanical and morphological properties of a recycled expanded polystyrene-based mortar. Constr. Build. Mater. 2021, 301, 124310. [Google Scholar] [CrossRef]



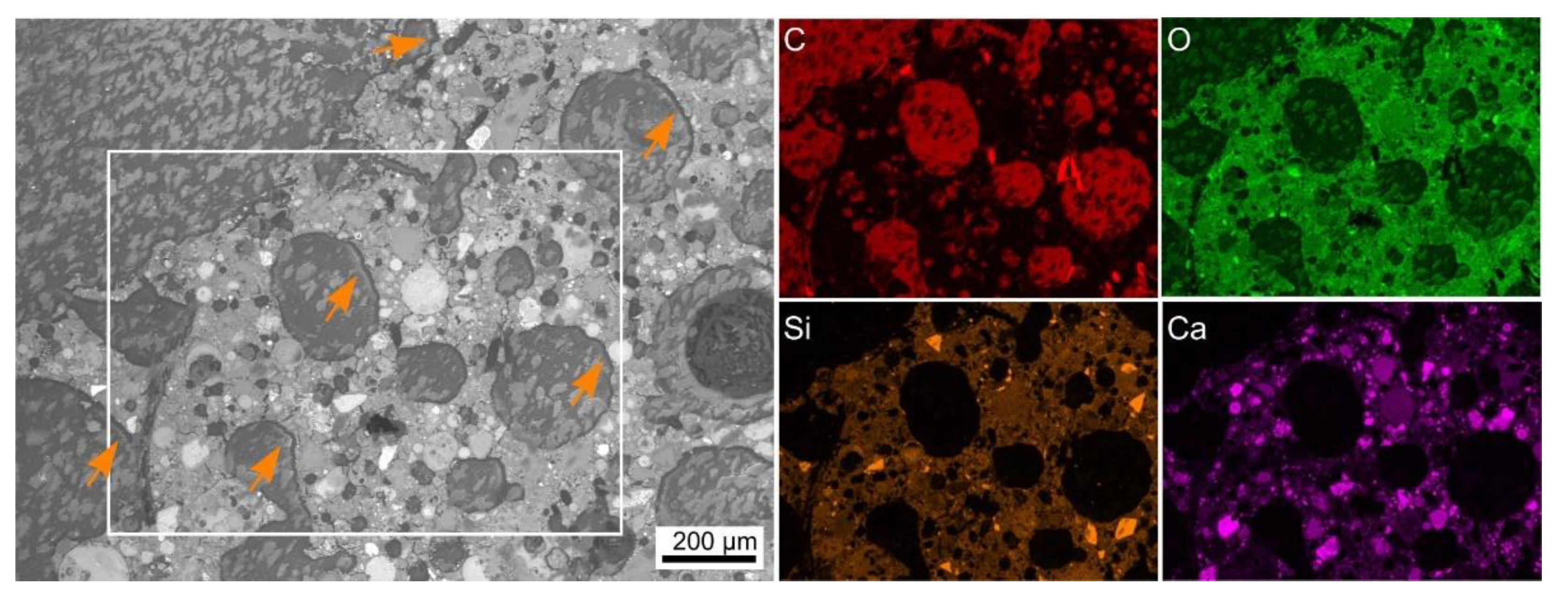

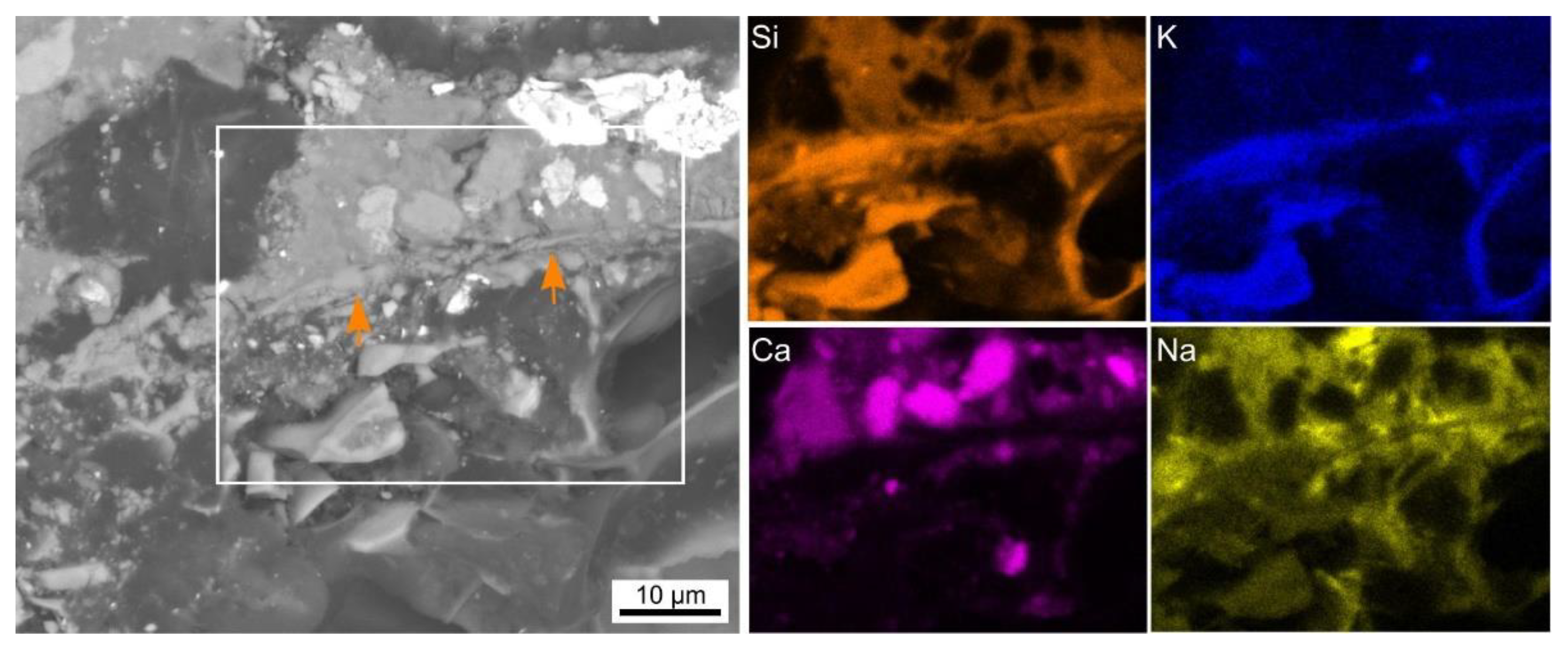
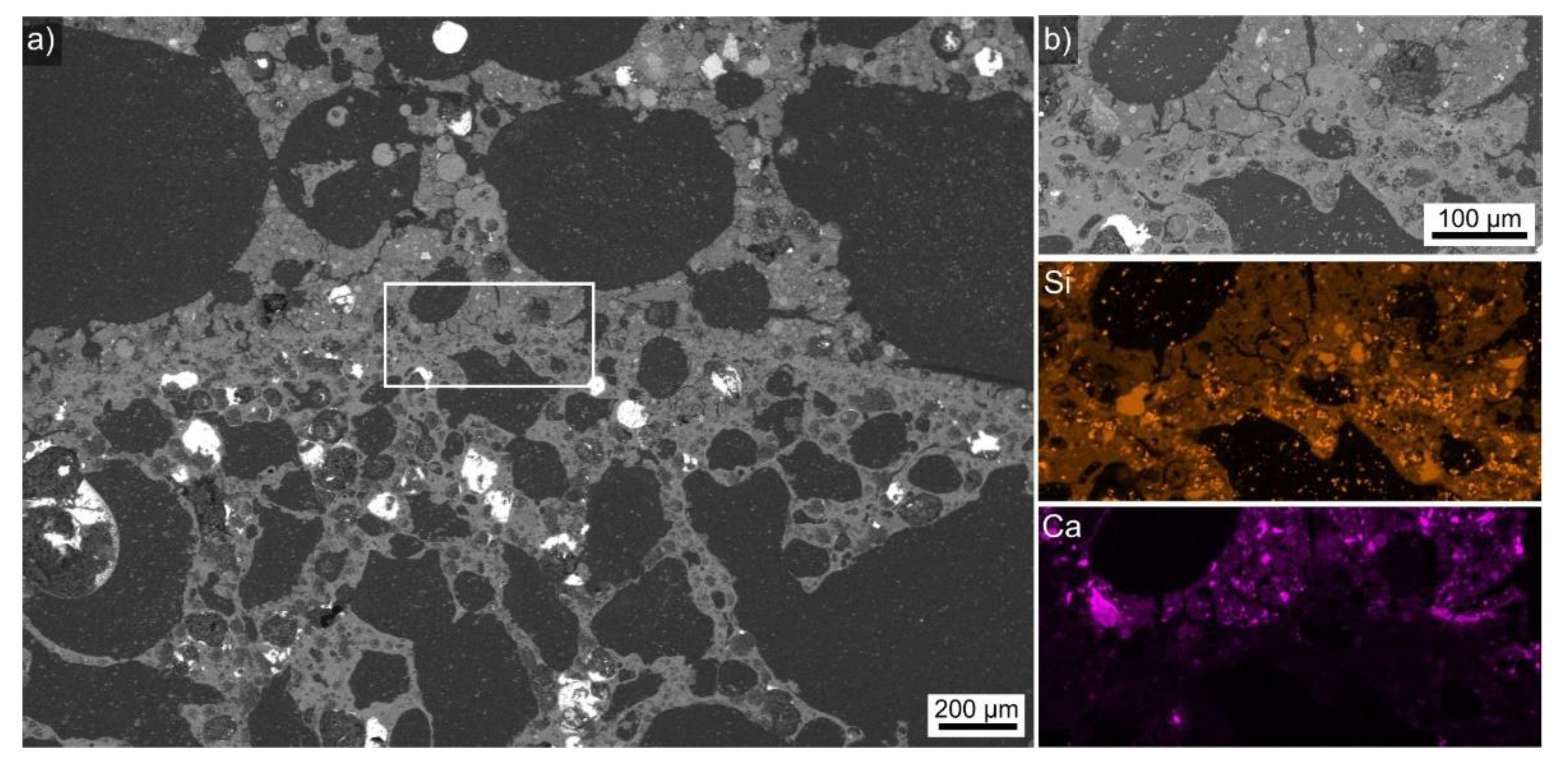
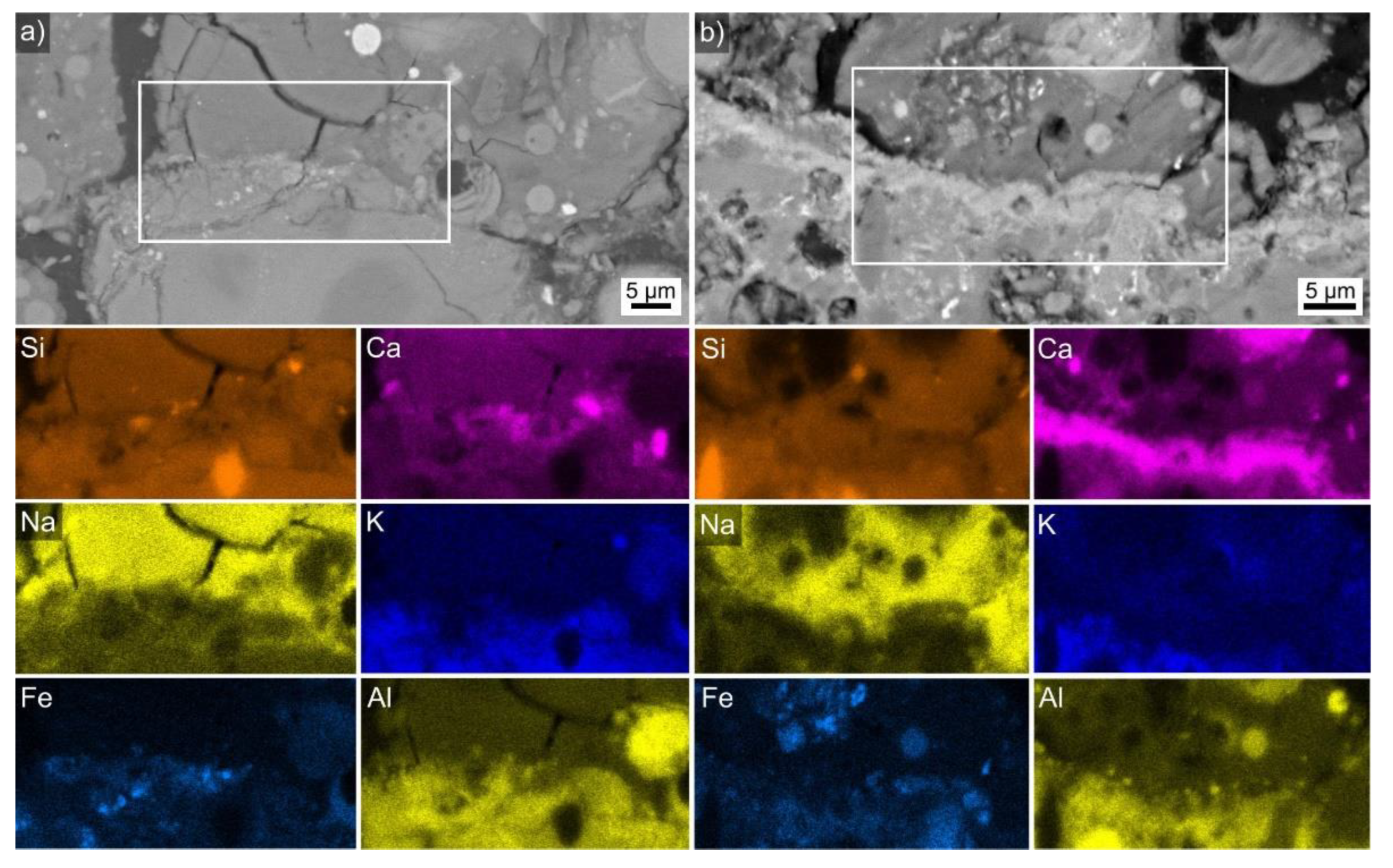

| Sample | FA-p | Slag Precursor Mix | Na2SiO3 | NaOH | Triton | Foaming Agent/Type | PP-Fibers | LWA |
|---|---|---|---|---|---|---|---|---|
| EC1 | 110.0 | / | 37.4 | 4.0 | 1.5 | 1.5/H | / | 58.0 |
| EC2 | 110.0 | / | 37.4 | 4.0 | 1.5 | 1.5/H | 0.5 | 58.0 |
| EC3 | / | 132.0 | 72.0 | 2.0 | 2.0 | 4.6/H | / | 58.0 |
| EC4 | / | 132.0 | 72.0 | 2.0 | 2.0 | 4.6/H | 0.5 | 58.0 |
| EC5 | 50.0 | 50.0 | 54.0 | 0.6 | 0.8 | 0.5/PB | 0.5 | 70.0 |
| EC6 | 50.0 | 50.0 | 54.0 | 0.6 | 0.8 | 2.0/H | 0.5 | 100.0 |
| P1 | / | 132.0 | 72.0 | 2.0 | 2.0 | 4.6/H | / | 11.8 |
| P2 | 50.0 | 50.0 | 54.0 | 0.6 | 0.8 | 0.7/PB | 0.5 | 30.0 |
| P3 | 50.0 | 50.0 | 54.0 | 0.6 | 0.8 | 2.0/H | 0.5 | 40.0 |
| EPS1 | 50.0 | 50.0 | 55.0 | 0.6 | 0.8 | 1.5/H | 0.5 | 4.0 |
| EPS2 | 50.0 | 50.0 | 55.0 | 0.6 | 0.8 | 1.0/PB | 0.5 | 4.0 |
| EPS3 | 50.0 | 50.0 | 55.0 | 0.6 | 0.8 | 1.0/PB | 0.5 | 4.0 |
| EG1 | / | 132.0 | 72.0 | 2.0 | 2.0 | 4.6/H | / | 10.0 |
| EG2 | 50.0 | 50.0 | 54.0 | 0.6 | 0.8 | 0.5/PB | 0.5 | 60.0 |
| EG3 | 50.0 | 50.0 | 54.0 | 0.6 | 0.8 | 2.0/H | 0.5 | 80.0 |
| Component | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | Cr2O3 | MnO | Others |
|---|---|---|---|---|---|---|---|---|---|---|
| slag A-p | 24.53 | 9.95 | 13.25 | 24.32 | 17.33 | 0.15 | 0.20 | 4.38 | 2.61 | 3.26 |
| slag R-p | 17.05 | 6.48 | 5.78 | 34.69 | 28.96 | 0.35 | 0.17 | 0.22 | 0.77 | 5.52 |
| FA | 44.83 | 22.98 | 10.65 | 12.38 | 2.80 | 1.19 | 2.20 | 0.02 | 0.26 | 2.68 |
| Sample | σFS [MPa] (std) | σCS [MPa] (std) |
|---|---|---|
| EC2 | 0.82 * | 2.72 (0.00) |
| EC4 | b.d.l. | 1.04 (0.12) |
| EC5 | 0.90 * | 2.95 (0.26) |
| EC6 | 0.39 * | 1.31 (0.01) |
| P2 | 0.29 * | 0.61 (0.05) |
| P3 | b.d.l. | 0.95 (0.07) |
| EPS1 | 0.34 (0.10) | 0.55 (0.09) |
| EPS2 | 0.15 (0.01) | 0.69 (0.42) |
| EG1 | b.d.l. | 0.85 (0.01) |
| EG2 | 0.77 * | 4.44 (0.28) |
| EG3 | 0.62 * | 3.62 (1.08) |
| in wt % | O | Na | Mg | Al | Si | K | Ca | Fe | Si/Ca |
|---|---|---|---|---|---|---|---|---|---|
| interface 1 | 48 | 12 | 1 | 5 | 29 | 2 | 3 | 1 | 10:1 |
| interface 2 | 50 | 5 | 0 | 6 | 34 | 3 | 2 | 1 | 20:1 |
| interface 3 | 50 | 5 | 0 | 6 | 33 | 2 | 1 | 1 | 27:1 |
| in wt % | O | Na | Mg | Al | Si | K | Ca | Fe | Si/Ca |
|---|---|---|---|---|---|---|---|---|---|
| Figure 9a | 48 | 3 | 4 | 7 | 28 | 2 | 5 | 3 | 6:1 |
| Figure 9b | 45 | 3 | 5 | 6 | 21 | 1 | 14 | 6 | 3:2 |
| Comparable interface | 44 | 1 | 4 | 6 | 20 | 0 | 20 | 5 | 1:1 |
| Sample | Width [mm] | Length [mm] | Height [mm] | Weight [g] | Density [kg/m³] | Thermal Conductivity [mW/mK] |
|---|---|---|---|---|---|---|
| EC1 | 90.80 | 90.60 | 28.49 | 128.87 | 549.8 | 140 (134) |
| EC2 | 70.50 | 76.30 | 21.03 | 64.23 | 567.8 | 148 (140) |
| EC3 | 86.60 | 84.00 | 35.95 | 100.45 | 384.1 | 96 (123) |
| EC4 | 91.80 | 68.30 | 27.21 | 81.20 | 475.9 | 114 (130) |
| EC5 | 101.95 | 21.50 | 20.13 | 30.79 | 700 | 190 |
| EC6 | 100.50 | 21.70 | 29.23 | 30.19 | 470 | 114 |
| P2 | 102.00 | 26.00 | 21.43 | 33.62 | 590 | 129 |
| P3 | 103.80 | 21.35 | 25.77 | 25.10 | 440 | 98 |
| EPS1 | 99.70 | 18.23 | 25.65 | 17.01 | 360 | 116 |
| EPS2 | 97.44 | 22.83 | 20.86 | 14.88 | 320 | 112 |
| EG2 | 101.95 | 20.40 | 21.60 | 29.93 | 670 | 151 |
| EG3 | 100.25 | 21.30 | 22.37 | 23.38 | 490 | 108 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Traven, K.; Wisniewski, W.; Češnovar, M.; Ducman, V. Microstructural Characterization of Alkali-Activated Composites of Lightweight Aggregates (LWAs) Embedded in Alkali-Activated Foam (AAF) Matrices. Polymers 2022, 14, 1729. https://doi.org/10.3390/polym14091729
Traven K, Wisniewski W, Češnovar M, Ducman V. Microstructural Characterization of Alkali-Activated Composites of Lightweight Aggregates (LWAs) Embedded in Alkali-Activated Foam (AAF) Matrices. Polymers. 2022; 14(9):1729. https://doi.org/10.3390/polym14091729
Chicago/Turabian StyleTraven, Katja, Wolfgang Wisniewski, Mark Češnovar, and Vilma Ducman. 2022. "Microstructural Characterization of Alkali-Activated Composites of Lightweight Aggregates (LWAs) Embedded in Alkali-Activated Foam (AAF) Matrices" Polymers 14, no. 9: 1729. https://doi.org/10.3390/polym14091729
APA StyleTraven, K., Wisniewski, W., Češnovar, M., & Ducman, V. (2022). Microstructural Characterization of Alkali-Activated Composites of Lightweight Aggregates (LWAs) Embedded in Alkali-Activated Foam (AAF) Matrices. Polymers, 14(9), 1729. https://doi.org/10.3390/polym14091729







