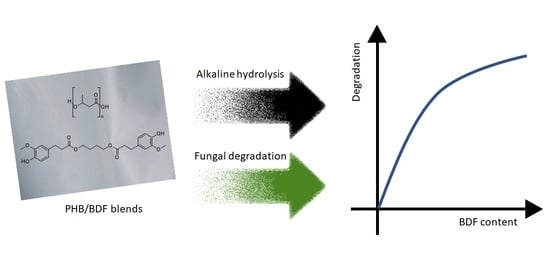
Impact of Bis-O-dihydroferuloyl-1,4-butanediol Content on the Chemical, Enzymatic and Fungal Degradation Processes of Poly(3-hydroxybutyrate)
Abstract
1. Introduction
2. Materials and Methods
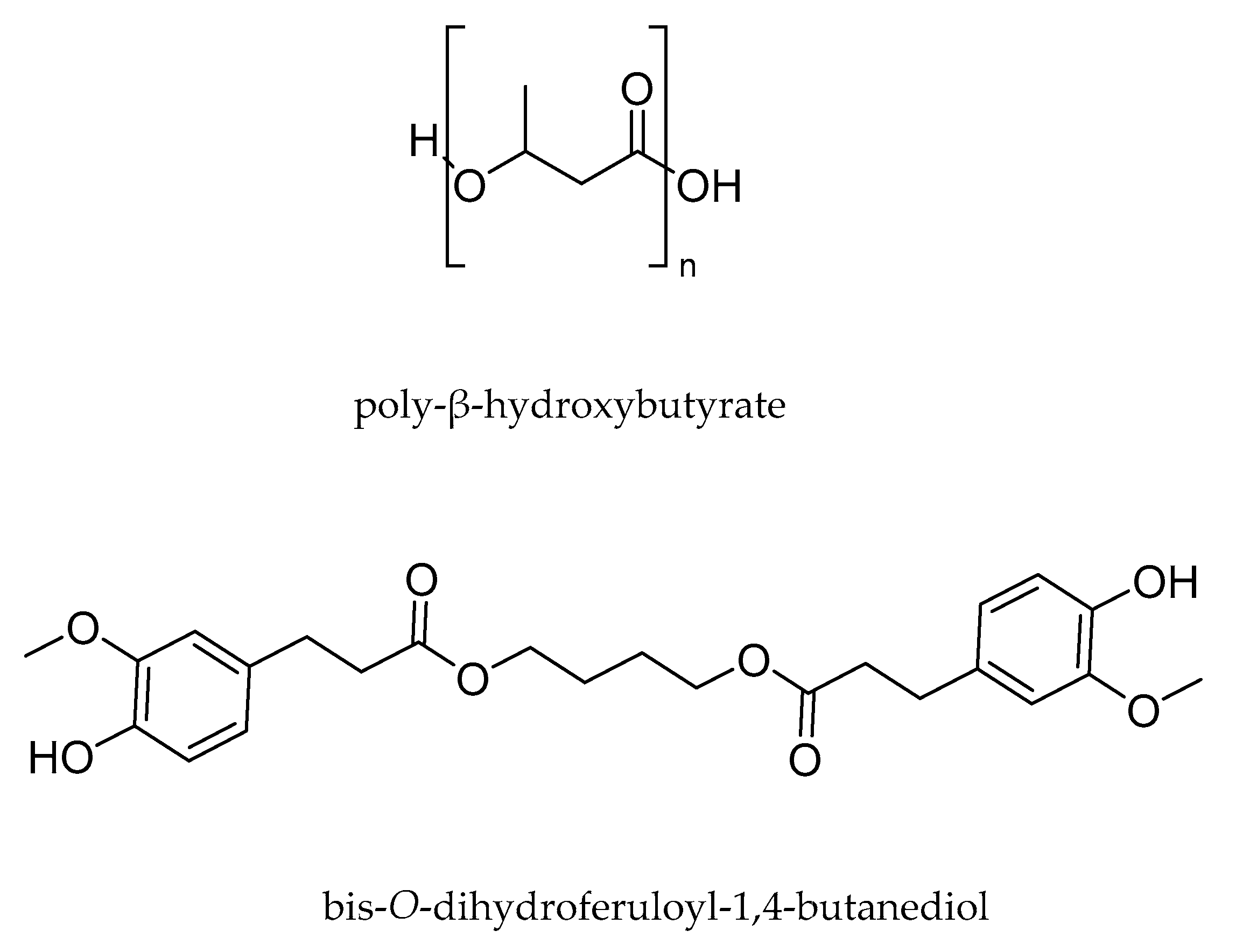
2.1. Polymers
2.1.1. Preparation of PHB/BDF Specimens
2.1.2. Preparation of PHB/BDF Films
2.1.3. Preparation of PHB/BDF Powders
2.1.4. Characterization of the Material
Differential Scanning Calorimetry
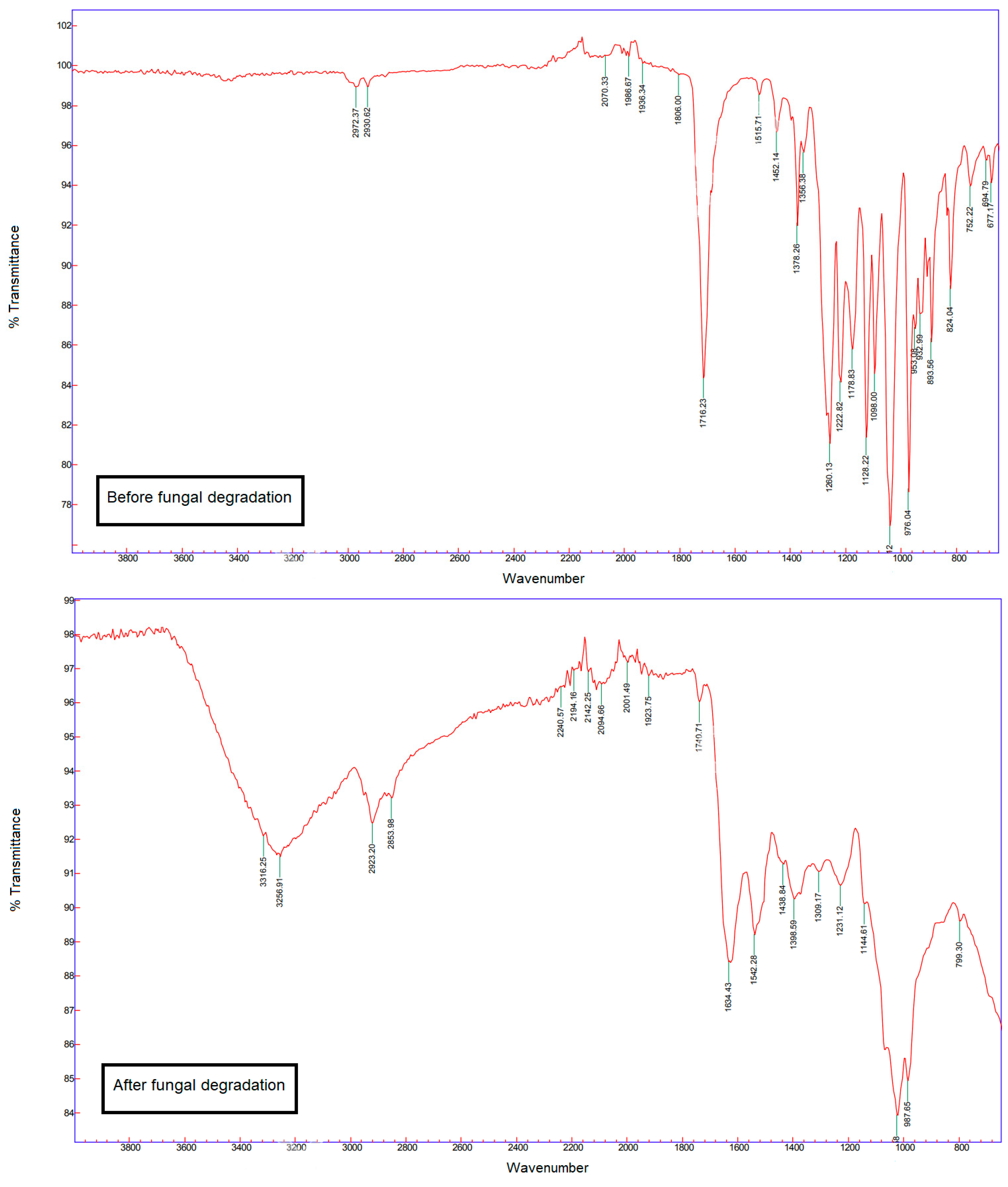
Fourier-Transform Infrared Spectroscopy
2.2. Enzymatic and Chemical Degradation
2.2.1. Chemical Degradation
2.2.2. Enzymatic Degradation
2.2.3. Evaluation of the Degradation Potential through Nuclear Magnetic Resonance (NMR) Spectroscopy
2.3. Fungal Degradation
2.3.1. Sample Collection
2.3.2. Isolation of PHB-Degrading Fungi
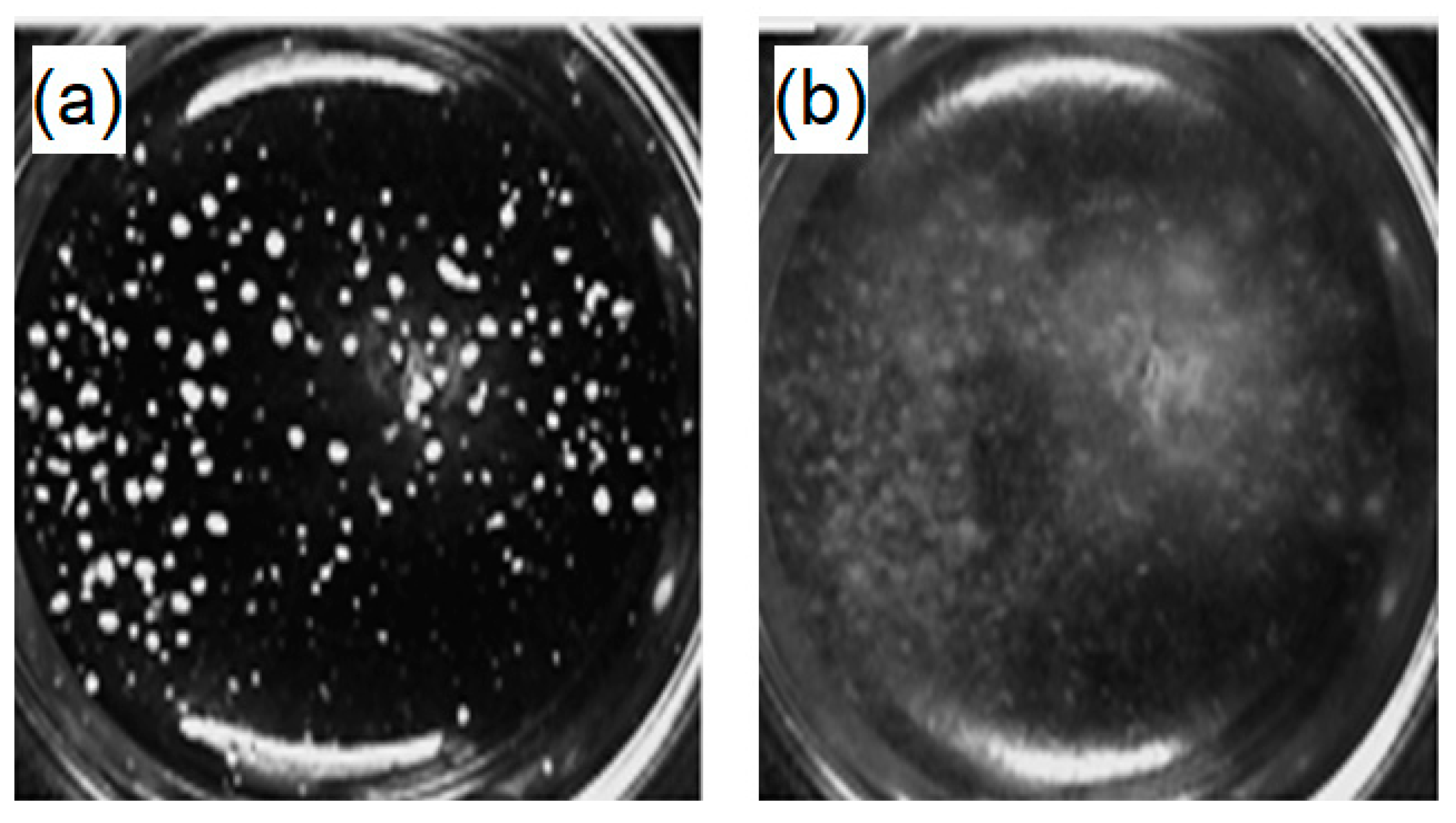
2.3.3. Evaluation of the Degradation Potential of PHB
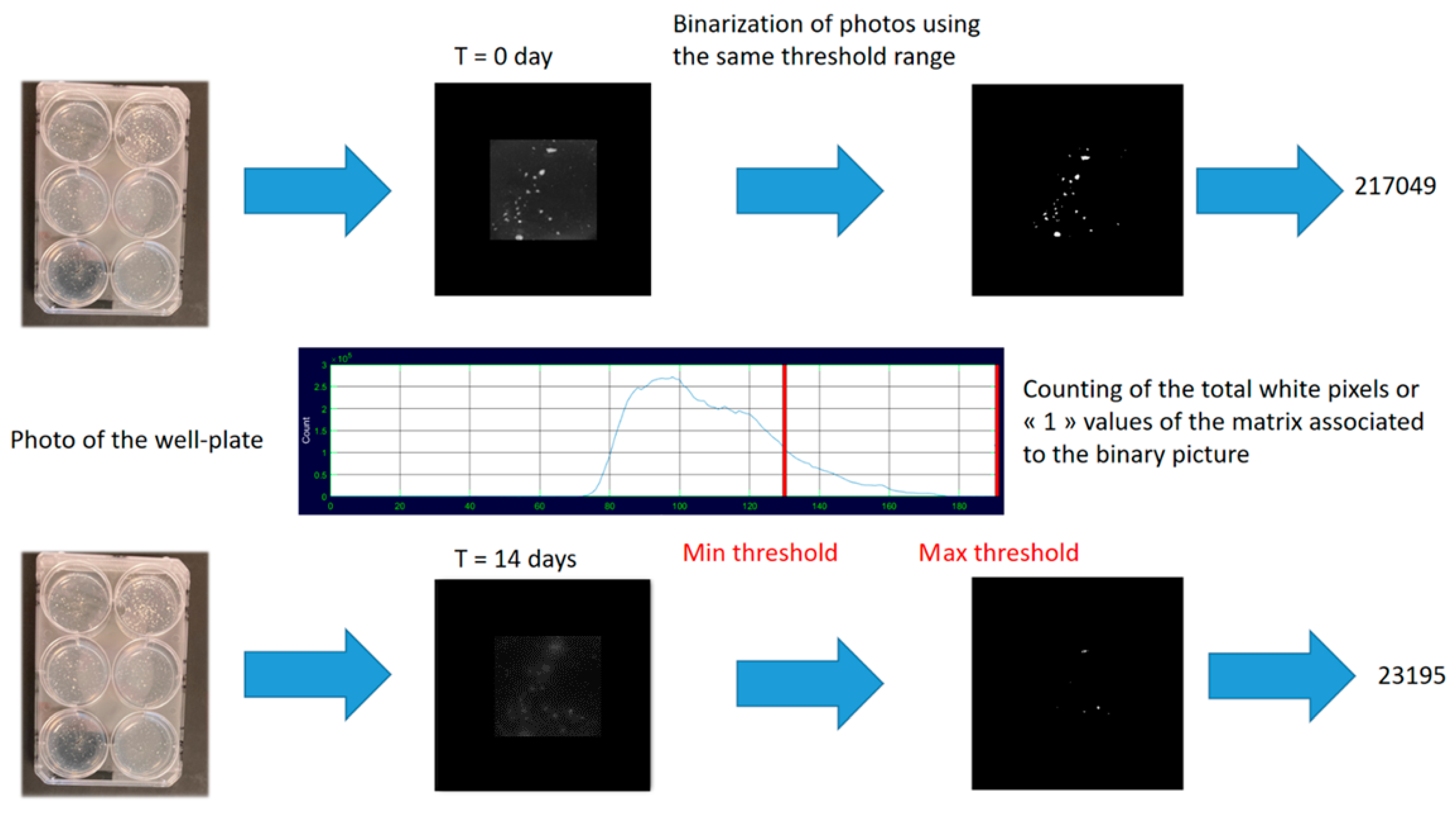
2.3.4. Evaluation of the Degradation Potentials of the PHB Blends through Image Analysis
2.3.5. Statistical Analysis
3. Results and Discussion
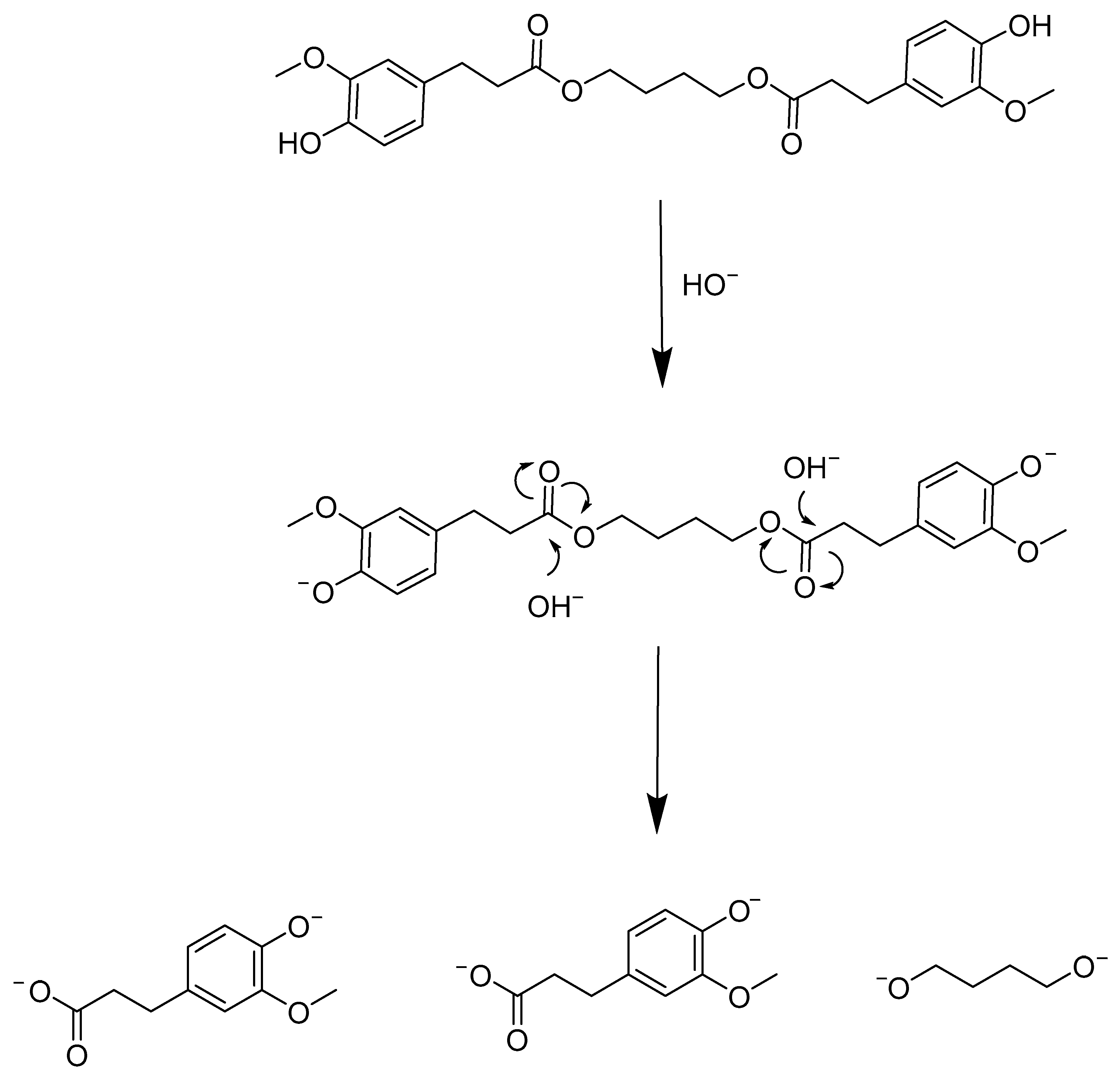
3.1. Chemical Degradation
3.2. Enzymatic Degradation
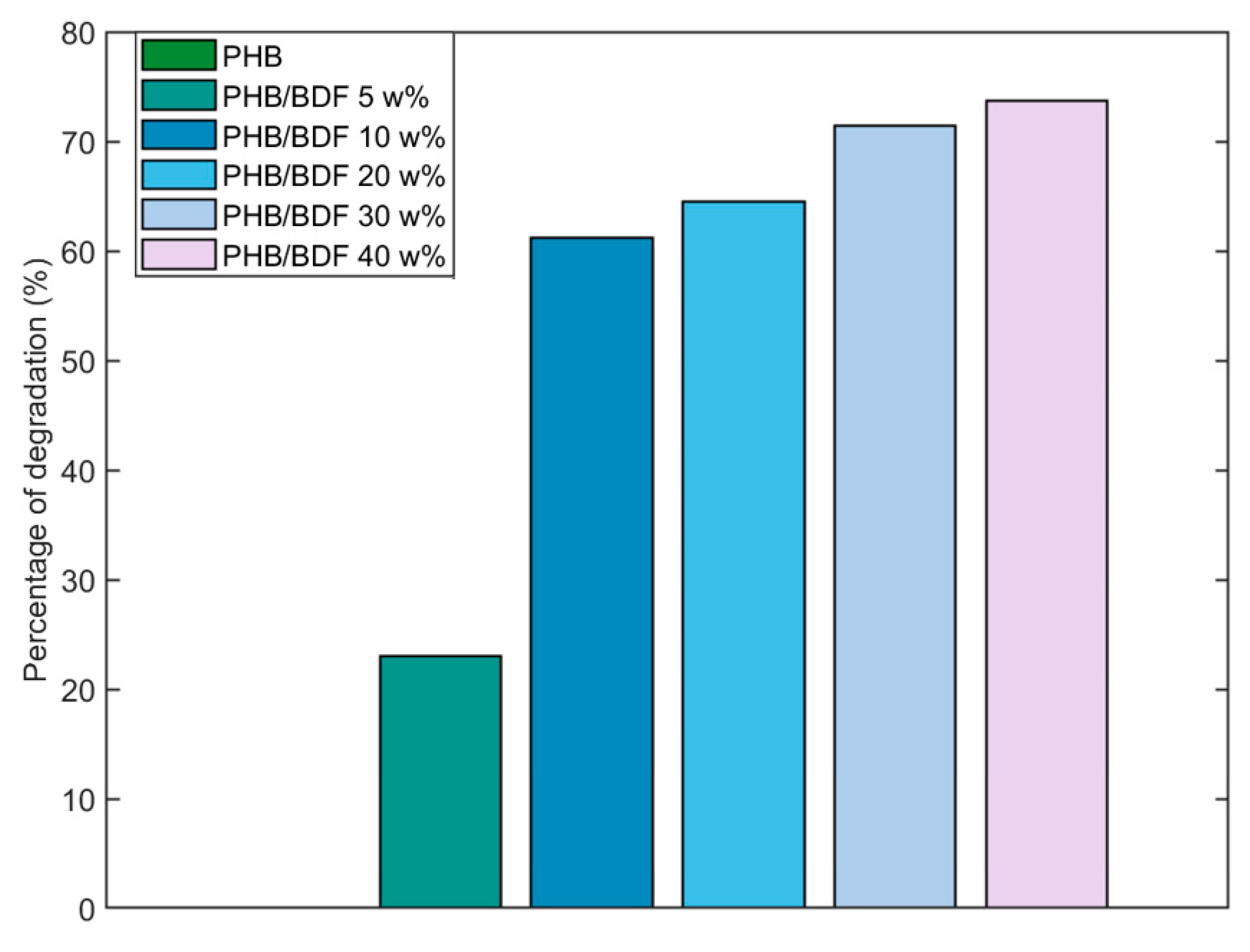
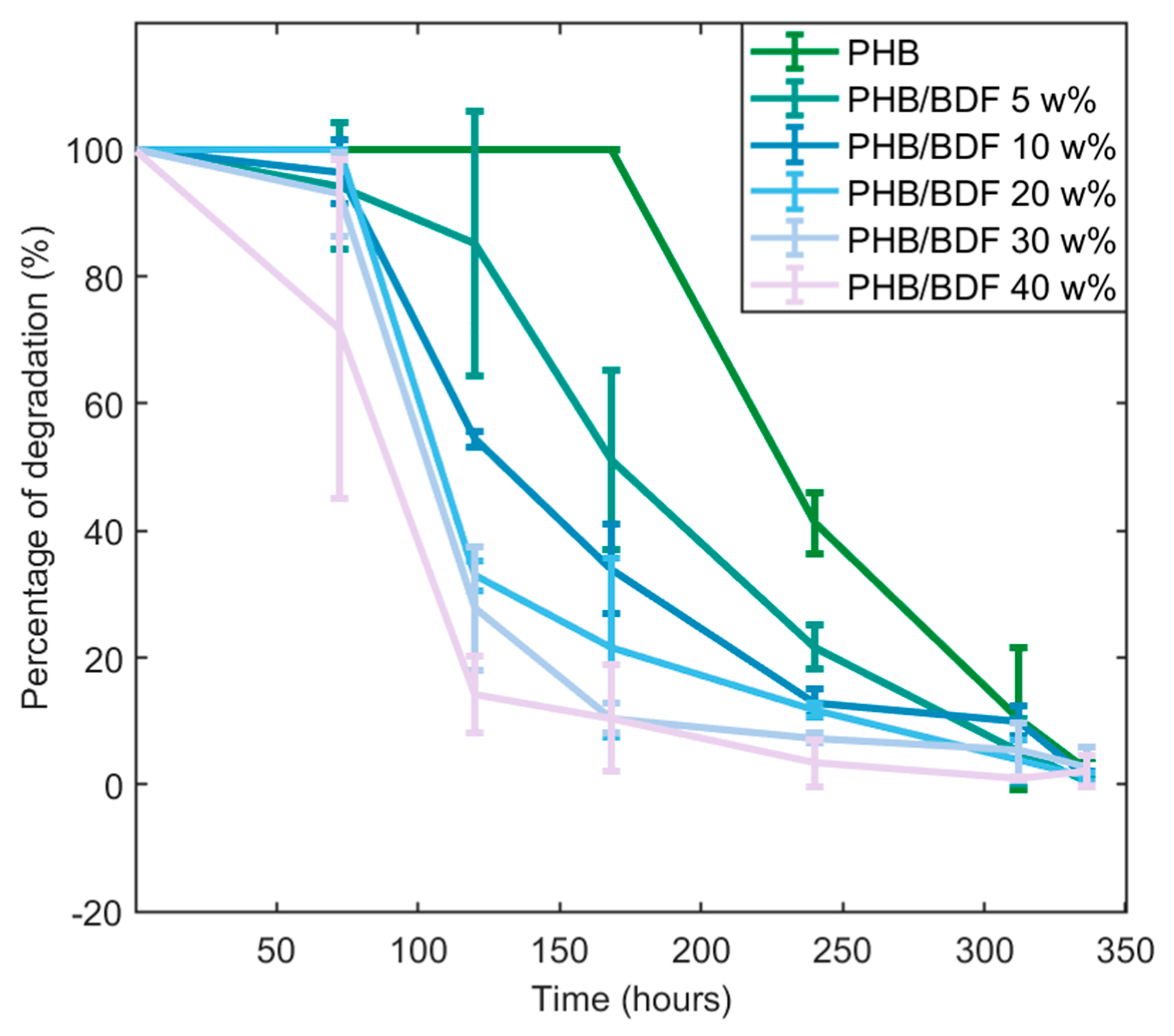
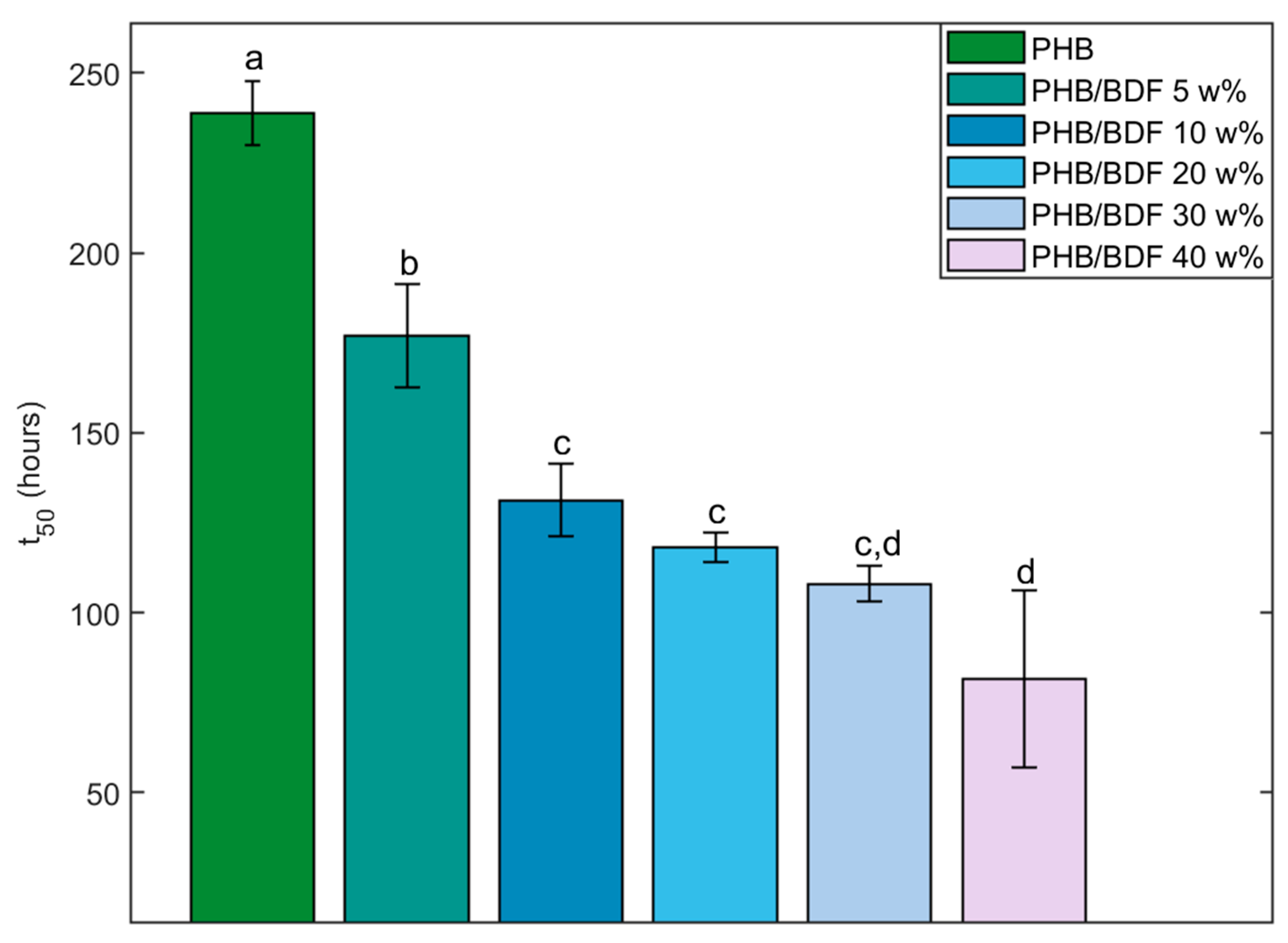
3.3. Fungal Degradation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Avella, M.; Buzarovska, A.; Errico, M.; Gentile, G.; Grozdanov, A. Eco-Challenges of Bio-Based Polymer Composites. Materials 2009, 2, 911–925. [Google Scholar] [CrossRef]
- Fadlallah, S.; Sinha Roy, P.; Garnier, G.; Saito, K.; Allais, F. Are Lignin-Derived Monomers and Polymers Truly Sustainable? An in-Depth Green Metrics Calculations Approach. Green Chem. 2021, 23, 1495–1535. [Google Scholar] [CrossRef]
- Koncar, V. Composites and Hybrid Structures. In Smart Textiles for In Situ Monitoring of Composites; Koncar, V., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 153–215. ISBN 978-0-08-102308-2. [Google Scholar]
- Sevim, K.; Pan, J. A Model for Hydrolytic Degradation and Erosion of Biodegradable Polymers. Acta Biomater. 2018, 66, 192–199. [Google Scholar] [CrossRef]
- Huang, Y.; Chen, F.; Pan, Y.; Chen, C.; Jiang, L.; Dan, Y. Effect of Hydrophobic Fluoropolymer and Crystallinity on the Hydrolytic Degradation of Poly(Lactic Acid). Eur. Polym. J. 2017, 97, 308–318. [Google Scholar] [CrossRef]
- Khakbaz, M.; Hejazi, I.; Seyfi, J.; Jafari, S.-H.; Khonakdar, H.A.; Davachi, S.M. A Novel Method to Control Hydrolytic Degradation of Nanocomposite Biocompatible Materials via Imparting Superhydrophobicity. Appl. Surf. Sci. 2015, 357, 880–886. [Google Scholar] [CrossRef]
- Lizundia, E.; Sáenz-Pérez, M.; Patrocinio, D.; Aurrekoetxea, I.; dM Vivanco, M.; Vilas, J.L. Nanopatterned Polystyrene-b-Poly(Acrylic Acid) Surfaces to Modulate Cell-Material Interaction. Mater. Sci. Eng. C 2017, 75, 229–236. [Google Scholar] [CrossRef]
- Siparsky, G.L.; Voorhees, K.J.; Miao, F. Hydrolysis of Polylactic Acid (PLA) and Polycaprolactone (PCL) in Aqueous Acetonitrile Solutions: Autocatalysis. J. Polym. Environ. 1998, 6, 31–41. [Google Scholar] [CrossRef]
- Garrison, T.; Murawski, A.; Quirino, R. Bio-Based Polymers with Potential for Biodegradability. Polymers 2016, 8, 262. [Google Scholar] [CrossRef]
- Jendrossek, D. Polyhydroxyalkanoate Granules Are Complex Subcellular Organelles (Carbonosomes). J. Bacteriol. 2009, 191, 3195–3202. [Google Scholar] [CrossRef]
- McAdam, B.; Brennan Fournet, M.; McDonald, P.; Mojicevic, M. Production of Polyhydroxybutyrate (PHB) and Factors Impacting Its Chemical and Mechanical Characteristics. Polymers 2020, 12, 2908. [Google Scholar] [CrossRef]
- Sen, K.Y.; Baidurah, S. Renewable Biomass Feedstocks for Production of Sustainable Biodegradable Polymer. Curr. Opin. Green Sustain. Chem. 2021, 27, 100412. [Google Scholar] [CrossRef]
- Saratale, R.G.; Cho, S.K.; Saratale, G.D.; Ghodake, G.S.; Bharagava, R.N.; Kim, D.S.; Nair, S.; Shin, H.S. Efficient Bioconversion of Sugarcane Bagasse into Polyhydroxybutyrate (PHB) by Lysinibacillus sp. and Its Characterization. Bioresour. Technol. 2021, 324, 124673. [Google Scholar] [CrossRef]
- Yang, X.; Zhao, K.; Chen, G.-Q. Effect of Surface Treatment on the Biocompatibility of Microbial Polyhydroxyalkanoates. Biomaterials 2002, 23, 1391–1397. [Google Scholar] [CrossRef]
- El-Hadi, A.; Schnabel, R.; Straube, E.; Müller, G.; Henning, S. Correlation between Degree of Crystallinity, Morphology, Glass Temperature, Mechanical Properties and Biodegradation of Poly (3-Hydroxyalkanoate) PHAs and Their Blends. Polym. Test. 2002, 21, 665–674. [Google Scholar] [CrossRef]
- Roohi; Zaheer, M.R.; Kuddus, M. PHB (Poly-β-Hydroxybutyrate) and Its Enzymatic Degradation. Polym. Adv. Technol. 2018, 29, 30–40. [Google Scholar] [CrossRef]
- Smith, M.K.M.; Paleri, D.M.; Abdelwahab, M.; Mielewski, D.F.; Misra, M.; Mohanty, A.K. Sustainable Composites from Poly(3-Hydroxybutyrate) (PHB) Bioplastic and Agave Natural Fibre. Green Chem. 2020, 22, 3906–3916. [Google Scholar] [CrossRef]
- Omura, T.; Goto, T.; Maehara, A.; Kimura, S.; Abe, H.; Iwata, T. Thermal Degradation Behavior of Poly[(R)-3-Hydroxybutyrate-Co-4-Hydroxybutyrate]. Polym. Degrad. Stab. 2021, 183, 109460. [Google Scholar] [CrossRef]
- Kai, D.; Zhang, K.; Liow, S.S.; Loh, X.J. New Dual Functional PHB-Grafted Lignin Copolymer: Synthesis, Mechanical Properties, and Biocompatibility Studies. ACS Appl. Bio Mater. 2019, 2, 127–134. [Google Scholar] [CrossRef]
- Pion, F.; Reano, A.F.; Ducrot, P.-H.; Allais, F. Chemo-Enzymatic Preparation of New Bio-Based Bis- and Trisphenols: New Versatile Building Blocks for Polymer Chemistry. RSC Adv. 2013, 3, 8988. [Google Scholar] [CrossRef]
- Gallos, A.; Allais, F. Shape Memory Polymer Based on Poly(Hydroxyalkanoates). French Patent WO2020165075A1, 20 August 2020. [Google Scholar]
- Michalska-Pożoga, I.; Tomkowski, R.; Rydzkowski, T.; Thakur, V.K. Towards the Usage of Image Analysis Technique to Measure Particles Size and Composition in Wood-Polymer Composites. Ind. Crops Prod. 2016, 92, 149–156. [Google Scholar] [CrossRef]
- Ley, A.; Altschuh, P.; Thom, V.; Selzer, M.; Nestler, B.; Vana, P. Characterization of a Macro Porous Polymer Membrane at Micron-Scale by Confocal-Laser-Scanning Microscopy and 3D Image Analysis. J. Membr. Sci. 2018, 564, 543–551. [Google Scholar] [CrossRef]
- Jelcic, Z.; Holjevac-Grguric, T.; Rek, V. Mechanical Properties and Fractal Morphology of High-Impact Polystyrene/Poly(Styrene-b-Butadiene-b-Styrene) Blends. Polym. Degrad. Stab. 2005, 90, 295–302. [Google Scholar] [CrossRef]
- Rodríguez-Contreras, A.; Calafell-Monfort, M.; Marqués-Calvo, M.S. Enzymatic Degradation of Poly(3-Hydroxybutyrate) by a Commercial Lipase. Polym. Degrad. Stab. 2012, 97, 2473–2476. [Google Scholar] [CrossRef]
- Image Analyst. Thresholding an Image. 2021. Available online: https://fr.mathworks.com/matlabcentral/fileexchange/29372-thresholding-an-image (accessed on 6 April 2022).
- Farias, R.F.; Canedo, E.L.; Wellen, R.M.R.; Rabello, M.S. Environmental Stress Cracking of Poly(3-Hydroxibutyrate) Under Contact with Sodium Hydroxide. Mater. Res. 2015, 18, 258–266. [Google Scholar] [CrossRef][Green Version]
- Tsuji, H.; Suzuyoshi, K.; Tezuka, Y.; Ishida, T. Environmental Degradation of Biodegradable Polyesters: 3. Effects of Alkali Treatment on Biodegradation of Poly(ε-Caprolactone) and Poly[(R)-3-Hydroxybutyrate) Films in Controled Soil. J. Polym. Environ. 2003, 11, 57–65. [Google Scholar] [CrossRef]
- Zhao, K.; Yang, X.; Chen, G.-Q.; Chen, J.-C. Effect of Lipase Treatment on the Biocompatibility of Microbial Polyhydroxyalkanoates. J. Mater. Sci. Mater. Med. 2002, 13, 849–854. [Google Scholar] [CrossRef]
- Gowda U.S., V.; Shivakumar, S. Poly(-β-Hydroxybutyrate) (PHB) Depolymerase PHAZ Pen from Penicillium Expansum: Purification, Characterization and Kinetic Studies. 3 Biotech 2015, 5, 901–909. [Google Scholar] [CrossRef]
- Tarazona, N.A.; Machatschek, R.; Lendlein, A. Influence of Depolymerases and Lipases on the Degradation of Polyhydroxyalkanoates Determined in Langmuir Degradation Studies. Adv. Mater. Interfaces 2020, 7, 2000872. [Google Scholar] [CrossRef]
- Lee, K.-M.; Gimore, D.F.; Huss, M.J. Fungal Degradation of the Bioplastic PHB (Poly-3-Hydroxy- Butyric Acid). J. Polym. Environ. 2005, 13, 213–219. [Google Scholar] [CrossRef]
- Oda, Y.; Asari, H.; Urakami, T.; Tonomura, K. Microbial Degradation of Poly(3-Hydroxybutyrate) and Polycaprolactone by Filamentous Fungi. J. Ferment. Bioeng. 1995, 80, 265–269. [Google Scholar] [CrossRef]
- Aburas, M.M.A. Degradation of Poly (3-Hydroxybuthyrate) Using Aspergillus oryzae Obtained from Uncultivated Soil. Life Sci. J. 2016, 13, 51–56. [Google Scholar] [CrossRef]
- Satti, S.M.; Shah, Z.; Luqman, A.; Hasan, F.; Osman, M.; Shah, A.A. Biodegradation of Poly(3-Hydroxybutyrate) and Poly(3-Hydroxybutyrate-Co-3-Hydroxyvalerate) by Newly Isolated Penicillium Oxalicum SS2 in Soil Microcosms and Partial Characterization of Extracellular Depolymerase. Curr. Microbiol. 2020, 77, 1622–1636. [Google Scholar] [CrossRef] [PubMed]
- Jeszeová, L.; Puškárová, A.; Bučková, M.; Kraková, L.; Grivalský, T.; Danko, M.; Mosnáčková, K.; Chmela, Š.; Pangallo, D. Microbial Communities Responsible for the Degradation of Poly(Lactic Acid)/Poly(3-Hydroxybutyrate) Blend Mulches in Soil Burial Respirometric Tests. World J. Microbiol. Biotechnol. 2018, 34, 101. [Google Scholar] [CrossRef] [PubMed]
- Arunprasath, T.; Sudalai, S.; Meenatchi, R.; Jeyavishnu, K.; Arumugam, A. Biodegradation of Triphenylmethane Dye Malachite Green by a Newly Isolated Fungus Strain. Biocatal. Agric. Biotechnol. 2019, 17, 672–679. [Google Scholar] [CrossRef]
- Sauvola, J.; Pietikäinen, M. Adaptive Document Image Binarization. Pattern Recognit. 2000, 33, 225–236. [Google Scholar] [CrossRef]
- Ghosh, S.; Sachan, A.; Mitra, A. Degradation of Ferulic Acid by a White Rot Fungus Schizophyllum commune. World J. Microbiol. Biotechnol. 2005, 21, 385–388. [Google Scholar] [CrossRef]
- Carboué, Q.; Rébufa, C.; Hamrouni, R.; Roussos, S.; Bombarda, I. Statistical Approach to Evaluate Effect of Temperature and Moisture Content on the Production of Antioxidant Naphtho-Gamma-Pyrones and Hydroxycinnamic Acids by Aspergillus Tubingensis in Solid-State Fermentation. Bioprocess Biosyst. Eng. 2020, 43, 2283–2294. [Google Scholar] [CrossRef]
- Mousavioun, P.; George, G.A.; Doherty, W.O.S. Environmental Degradation of Lignin/Poly(Hydroxybutyrate) Blends. Polym. Degrad. Stab. 2012, 97, 1114–1122. [Google Scholar] [CrossRef]

| Type | Specimen | Film | Powder | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Composition | Tg (°C) | Tm (°C) | Tc (°C) | Tcc (°C) | Tg (°C) | Tm (°C) | Tc (°C) | Tcc (°C) | Tg (°C) | Tm (°C) | Tc (°C) | Tcc (°C) |
| PHB | 0.7 | 162.9 | 90.4 | 35.8 | 0.1 | 159.8 | - | 36.5 | - | 170.0 | 92.2 | - |
| PHB/BDF 5 w% | 0.0 | 162.0 | 64.1 | 36.2 | −4.7 | 157.7 | - | 32.5 | 0.75 | 160.7 | 66.6 | 31.7 |
| PHB/BDF 10 w% | −1.5 | 156.7 | - | 37.2 | 0.8 | 160.1 | - | 39.5 | 0.7 | 158.5 | 58.7 | 34.5 |
| PHB/BDF 20 w% | −5.8 | 151.4 | - | 38.0 | −2.5 | 152.7 | - | 39.8 | −4.9 | 150.6 | - | 35.4 |
| PHB/BDF 30 w% | −8.6 | 146.7 | - | 44.3 | −11.7 | 140.0 | - | 42.4 | −8.4 | 143.3 | - | 35.9 |
| PHB/BDF 40 w% | −9.4 | 144.0 | - | 45.7 | −10.3 | 145.5 | - | 37.4 | −9.1 | 142.2 | - | 38.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carboué, Q.; Fadlallah, S.; Werghi, Y.; Longé, L.; Gallos, A.; Allais, F.; Lopez, M. Impact of Bis-O-dihydroferuloyl-1,4-butanediol Content on the Chemical, Enzymatic and Fungal Degradation Processes of Poly(3-hydroxybutyrate). Polymers 2022, 14, 1564. https://doi.org/10.3390/polym14081564
Carboué Q, Fadlallah S, Werghi Y, Longé L, Gallos A, Allais F, Lopez M. Impact of Bis-O-dihydroferuloyl-1,4-butanediol Content on the Chemical, Enzymatic and Fungal Degradation Processes of Poly(3-hydroxybutyrate). Polymers. 2022; 14(8):1564. https://doi.org/10.3390/polym14081564
Chicago/Turabian StyleCarboué, Quentin, Sami Fadlallah, Yasmine Werghi, Lionel Longé, Antoine Gallos, Florent Allais, and Michel Lopez. 2022. "Impact of Bis-O-dihydroferuloyl-1,4-butanediol Content on the Chemical, Enzymatic and Fungal Degradation Processes of Poly(3-hydroxybutyrate)" Polymers 14, no. 8: 1564. https://doi.org/10.3390/polym14081564
APA StyleCarboué, Q., Fadlallah, S., Werghi, Y., Longé, L., Gallos, A., Allais, F., & Lopez, M. (2022). Impact of Bis-O-dihydroferuloyl-1,4-butanediol Content on the Chemical, Enzymatic and Fungal Degradation Processes of Poly(3-hydroxybutyrate). Polymers, 14(8), 1564. https://doi.org/10.3390/polym14081564