A Molecular Dynamics Study of Cyanate Ester Monomer Melt Properties
Abstract
1. Introduction
2. Methods
2.1. Molecular Dynamics Simulations
2.2. Structural Characterization
2.2.1. Orientational Order Parameter
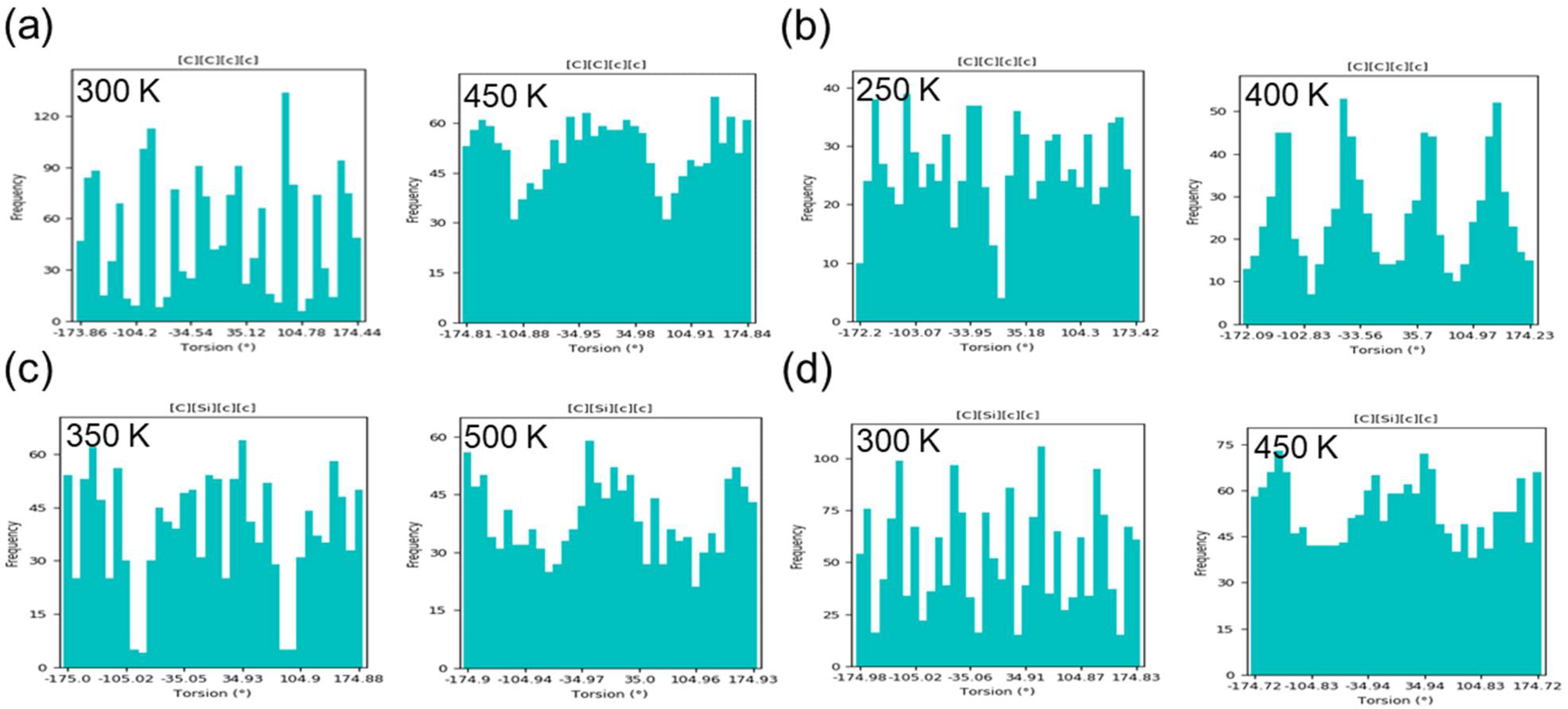
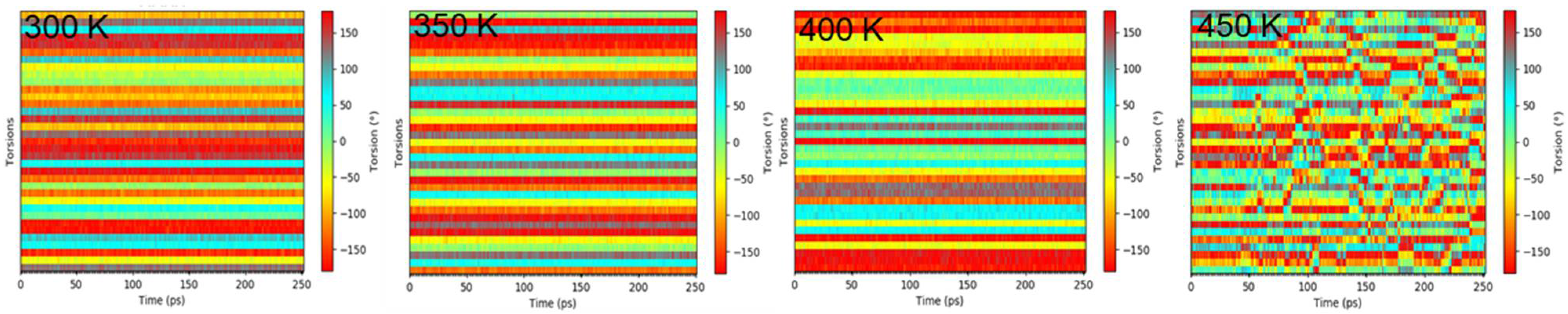
2.2.2. Torsion
3. Results and Discussion
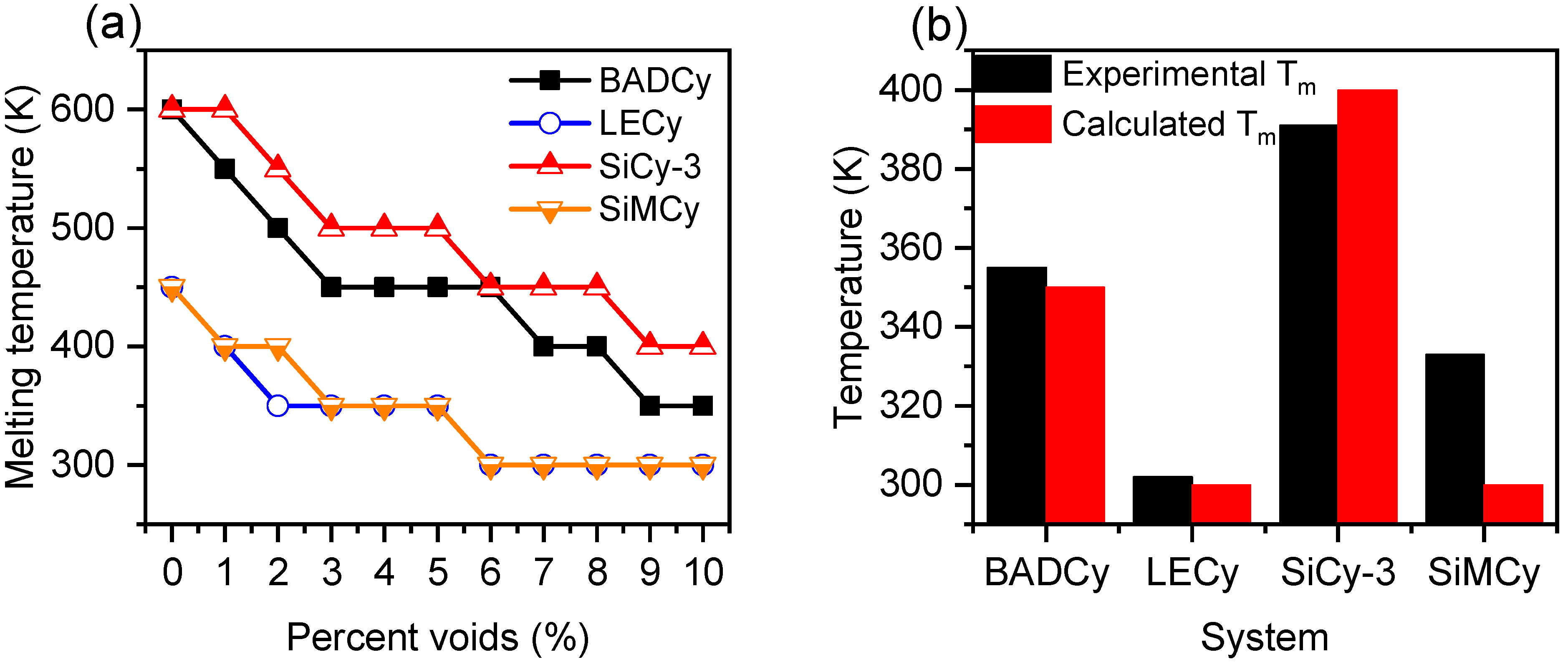
3.1. Melting of Monomers with 0% Voids
3.2. Dependence of Monomer Melting Temperature on Percentage of Voids
3.3. Structural Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ramdani, N.; Zaimeche, H.; Derradji, M. Biobased thermally-stable aromatic cyanate ester thermosets: A review. React. Funct. Polym. 2021, 168, 105037. [Google Scholar] [CrossRef]
- Crawford, A.O.; Hamerton, I.; Cavalli, G.; Howlin, B.J. Quantifying the Effect of Polymer Blending through Molecular Modelling of Cyanurate Polymers. PLoS ONE 2012, 7, e44487. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moore, L.M.J.; Redeker, N.D.; Browning, A.R.; Sanders, J.M.; Ghiassi, K.B. Polycyanurates via Molecular Dynamics: In Situ Crosslinking from Di(Cyanate Ester) Resins and Model Validation through Comparison to Experiment. Macromolecules 2021, 54, 6275–6284. [Google Scholar] [CrossRef]
- Laurenzi, S.; Marchetti, M. Advanced Composite Materials by Resin Transfer Molding for Aerospace Applications. In Composites and Their Properties; InTech: London, UK, 2012. [Google Scholar]
- Guenthner, A.J.; Ramirez, S.M.; Ford, M.D.; Soto, D.; Boatz, J.A.; Ghiassi, K.B.; Mabry, J.M. Organic Crystal Engineering of Thermosetting Cyanate Ester Monomers: Influence of Structure on Melting Point. Cryst. Growth Des. 2016, 16, 4082–4093. [Google Scholar] [CrossRef]
- Bamane, S.; Gaikwad, P.; Radue, M.; Gowtham, S.; Odegard, G. Wetting Simulations of High-Performance Polymer Resins on Carbon Surfaces as a Function of Temperature Using Molecular Dynamics. Polymers 2021, 13, 2162. [Google Scholar] [CrossRef] [PubMed]
- Ghiassi, K.B.; Guenthner, A.J.; Redeker, N.D.; Boatz, J.A.; Harvey, B.G.; Davis, M.C.; Chafin, A.P.; Groshens, T.J. Insights into Melting Behavior of Propyl-Bridged Di(cyanate ester) Monomers through Crystal Packing, Thermal Characterization, and Computational Analysis. Cryst. Growth Des. 2018, 18, 1030–1040. [Google Scholar] [CrossRef]
- Zheng, L.; Rice, B.M.; Thompson, D.L. Molecular Dynamics Simulations of the Melting Mechanisms of Perfect and Im-perfect Crystals of Dimethylnitramine. J. Phys. Chem. B 2007, 111, 2891–2895. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.-N.; Strachan, A.; Swift, D.C. Nonequilibrium melting and crystallization of a model Lennard-Jones system. J. Chem. Phys. 2004, 120, 11640–11649. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yoo, S.; Bai, J.; Morris, J.R.; Zeng, X.C. Melting temperature of ice Ih calculated from coexisting solid-liquid phases. J. Chem. Phys. 2005, 123, 036101. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, P.M.; Rice, B.M.; Thompson, D.L. Molecular dynamics study of the effects of voids and pressure in defect-nucleated melting simulations. J. Chem. Phys. 2003, 118, 9680–9688. [Google Scholar] [CrossRef]
- Zhang, Y.; Maginn, E. A comparison of methods for melting point calculation using molecular dynamics simulations. J. Chem. Phys. 2012, 136, 144116. [Google Scholar] [CrossRef] [PubMed]
- Alavi, S.; Thompson, D.L. Molecular dynamics studies of melting and some liquid-state properties of 1-ethyl-3-methylimidazolium hexafluorophosphate [emim][PF6]. J. Chem. Phys. 2005, 122, 154704. [Google Scholar] [CrossRef] [PubMed]
- Frenkel, D.; Ladd, A.J.C. New Monte Carlo method to compute the free energy of arbitrary solids. Application to the fcc and hcp phases of hard spheres. J. Chem. Phys. 1984, 81, 3188–3193. [Google Scholar] [CrossRef]
- Alfè, D.; Price, G.D.; Gillan, M.J. Iron under Earth’s core conditions: Liquid-state thermodynamics and high-pressure melting curve from ab initio calculations. Phys. Rev. B 2002, 65, 165118. [Google Scholar] [CrossRef]
- Eike, D.M.; Maginn, E. Atomistic simulation of solid-liquid coexistence for molecular systems: Application to triazole and benzene. J. Chem. Phys. 2006, 124, 164503. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, U.R. Direct calculation of the solid-liquid Gibbs free energy difference in a single equilibrium simulation. J. Chem. Phys. 2013, 139, 104102. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Xiang, S.; Dai, C. Investigation on the efficiency and accuracy of methods for calculating melting temperature by molecular dynamics simulation. Comput. Mater. Sci. 2020, 171, 109156. [Google Scholar] [CrossRef]
- Alavi, S.; Thompson, D.L. Simulations of melting of polyatomic solids and nanoparticles. Mol. Simul. 2006, 32, 999–1015. [Google Scholar] [CrossRef]
- Solca, J.; Dyson, A.J.; Steinebrunner, G.; Kirchner, B.; Huber, H. Melting curve for argon calculated from pure theory. Chem. Phys. 1997, 224, 253–261. [Google Scholar] [CrossRef]
- Materials Science Suite; Schrödinger, LLC: New York, NY, USA, 2021.
- Desmond Molecular Dynamics System, D.E. Shaw Research: Maestro-Desmond Interoperability Tools; Schrödinger: New York, NY, USA, 2021. [Google Scholar]
- Nosé, S. A unified formulation of the constant temperature molecular dynamics methods. J. Chem. Phys. 1984, 81, 511–519. [Google Scholar] [CrossRef]
- Hoover, W.G. Canonical dynamics: Equilibrium phase-space distributions. Phys. Rev. A 1985, 31, 1695–1697. [Google Scholar] [CrossRef]
- Martyna, G.J.; Tobias, D.J.; Klein, M.L. Constant pressure molecular dynamics algorithms. J. Chem. Phys. 1994, 101, 4177–4189. [Google Scholar] [CrossRef]
- Dubay, K.H.; Hall, M.L.; Hughes, T.F.; Wu, C.; Reichman, D.R.; Friesner, R.A. Accurate Force Field Development for Modeling Conjugated Polymers. J. Chem. Theory Comput. 2012, 8, 4556–4569. [Google Scholar] [CrossRef]
- Harder, E.; Damm, W.; Maple, J.; Wu, C.; Reboul, M.; Xiang, J.Y.; Wang, L.; Lupyan, D.; Dahlgren, M.K.; Knight, J.L.; et al. OPLS3: A Force Field Providing Broad Coverage of Drug-like Small Molecules and Proteins. J. Chem. Theory Comput. 2016, 12, 281–296. [Google Scholar] [CrossRef]
- Shivakumar, D.; Williams, J.; Wu, Y.; Damm, W.; Shelley, J.; Sherman, W. Prediction of Absolute Solvation Free Energies using Molecular Dynamics Free Energy Perturbation and the OPLS Force Field. J. Chem. Theory Comput. 2010, 6, 1509–1519. [Google Scholar] [CrossRef]
- Hamerton, I.; Hay, J.N. Recent developments in the chemistry of cyanate esters. Polym. Int. 1998, 47, 465–473. [Google Scholar] [CrossRef]
- Guenthner, A.J.; Reams, J.T.; Lamison, K.R.; Ramirez, S.M.; Swanson, D.D.; Yandek, G.R.; Sahagun, C.M.; Davis, M.C.; Mabry, J.M. Synergistic Physical Properties of Cocured Networks Formed from Di- and Tricyanate Esters. ACS Appl. Mater. Interfaces 2013, 5, 8772–8783. [Google Scholar] [CrossRef] [PubMed]
- Guenthner, A.J.; Yandek, G.R.; Wright, M.E.; Petteys, B.J.; Quintana, R.; Connor, D.; Gilardi, R.D.; Marchant, D. A New Silicon-Containing Bis(cyanate) Ester Resin with Improved Thermal Oxidation and Moisture Resistance. Macromolecules 2006, 39, 6046–6053. [Google Scholar] [CrossRef]
- Fyfe, C.A.; Niu, J.; Rettig, S.J.; Burlinson, N.E. High-resolution carbon-13 and nitrogen-15 NMR investigations of the mechanism of the curing reactions of cyanate-based polymer resins in solution and the solid state. Macromolecules 1992, 25, 6289–6301. [Google Scholar] [CrossRef]
- Cambridge Crystallographic Database. Available online: https://www.ccdc.cam.ac.uk/structures/ (accessed on 20 February 2022).
- Agrawal, P.M.; Rice, B.M.; Zheng, L.; Velardez, A.G.F.; Thompson, D.L. Molecular Dynamics Simulations of the Melting of 1,3,3-Trinitroazetidine. J. Phys. Chem. B 2006, 110, 5721–5726. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, N.; Chakrabarty, S.; Roy, S.; Kumar, R. A comparison of different water models for melting point calculation of methane hydrate using molecular dynamics simulations. Chem. Phys. 2019, 516, 6–14. [Google Scholar] [CrossRef]
- Ma, S.; Li, S.; Gao, T.; Shen, Y.; Chen, X.; Xiao, C.; Lu, T. Molecular dynamics simulations of structural and melting properties of Li2SiO. Ceram. Int. 2018, 44, 3381–3387. [Google Scholar] [CrossRef]
- Liu, Y.; Lai, W.; Yu, T.; Ma, Y.; Guo, W.; Ge, Z. Melting Point Prediction of Energetic Materials via Continuous Heating Simulation on Solid-to-Liquid Phase Transition. ACS Omega 2019, 4, 4320–4324. [Google Scholar] [CrossRef]
- Liu, N.; Zeman, S.; Shu, Y.-J.; Wu, Z.-K.; Wang, B.-Z.; Yin, S.-W. Comparative study of melting points of 3,4-bis(3-nitrofurazan-4-yl)furoxan (DNTF)/1,3,3-trinitroazetidine (TNAZ) eutectic compositions using molecular dynamic simulations. RSC Adv. 2016, 6, 59141–59149. [Google Scholar] [CrossRef]
- Solca, J.; Dyson, A.J.; Steinebrunner, G.; Kirchner, B.; Huber, H. Melting curves for neon calculated from pure theory. J. Chem. Phys. 1998, 108, 4107–4111. [Google Scholar] [CrossRef]
- Alvares, C.M.S.; Deffrennes, G.; Pisch, A.; Jakse, N. Thermodynamics and structural properties of CaO: A molecular dynamics simulation study. J. Chem. Phys. 2020, 152, 084503. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Liu, P.; Wang, M.; Liu, J. Molecular Dynamics Simulations of Melting Iron Nanoparticles with/without Defects Using a Reaxff Reactive Force Field. Sci. Rep. 2020, 10, 3408. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Stratt, R.M. Measuring order in disordered systems and disorder in ordered systems: Random matrix theory for isotropic and nematic liquid crystals and its perspective on pseudo-nematic domains. J. Chem. Phys. 2018, 148, 204501. [Google Scholar] [CrossRef]
- Steinhardt, P.J.; Nelson, D.R.; Ronchetti, M. Bond-orientational order in liquids and glasses. Phys. Rev. B 1983, 28, 784–805. [Google Scholar] [CrossRef]
- Daylight Chemical Information Systems, Inc. SMARTS—A Language for Describing Molecular Patterns. 2021. Available online: https://www.daylight.com/dayhtml/doc/theory/theory.smarts.html (accessed on 26 January 2022).
- Eike, D.M.; Brennecke, J.F.; Maginn, E. Toward a robust and general molecular simulation method for computing solid-liquid coexistence. J. Chem. Phys. 2005, 122, 14115. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.-N.; Ahrens, T.J.; Çağın, T.; Strachan, A.; Goddard, I.W.A.; Swift, D.C. Maximum superheating and undercooling: Systematics, molecular dynamics simulations, and dynamic experiments. Phys. Rev. B 2003, 68, 134206. [Google Scholar] [CrossRef]
- Li, C.; Strachan, A. Molecular scale simulations on thermoset polymers: A review. J. Polym. Sci. Part B Polym. Phys. 2015, 53, 103–122. [Google Scholar] [CrossRef]
- Ren, Y.; Zhang, Y.; Mao, Q.; Pitsch, H. Amorphous-to-Crystalline Transition during Sintering of Nascent TiO2 Nanopar-ticles in Gas-Phase Synthesis: A Molecular Dynamics Study. J. Phys. Chem. C 2020, 124, 27763–27771. [Google Scholar] [CrossRef]
- Sheng, X.; Akinc, M.; Kessler, M.R. Cure kinetics of thermosetting bisphenol E cyanate ester. J. Therm. Anal. 2008, 93, 77–85. [Google Scholar] [CrossRef]
- Fan, X.; Pan, D.; Li, M. Rethinking Lindemann criterion: A molecular dynamics simulation of surface mediated melting. Acta Mater. 2020, 193, 280–290. [Google Scholar] [CrossRef]
- Lavine, M.S.; Waheed, N.; Rutledge, G.C. Molecular dynamics simulation of orientation and crystallization of polyeth-ylene during uniaxial extension. Polymer 2003, 44, 1771–1779. [Google Scholar] [CrossRef]
- Tong, X.; Yang, P.; Zeng, M.; Wang, Q. Confinement Effect of Graphene Interface on Phase Transition of n-Eicosane: Molecular Dynamics Simulations. Langmuir 2020, 36, 8422–8434. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Thompson, D.L. Molecular dynamics simulations of melting of perfect crystalline hexahy-dro-1,3,5-trinitro-1,3,5-s-triazine. J. Chem. Phys. 2006, 125, 084505. [Google Scholar] [CrossRef]
- Pinal, R. Effect of molecular symmetry on melting temperature and solubility. Org. Biomol. Chem. 2004, 2, 2692–2699. [Google Scholar] [CrossRef] [PubMed]
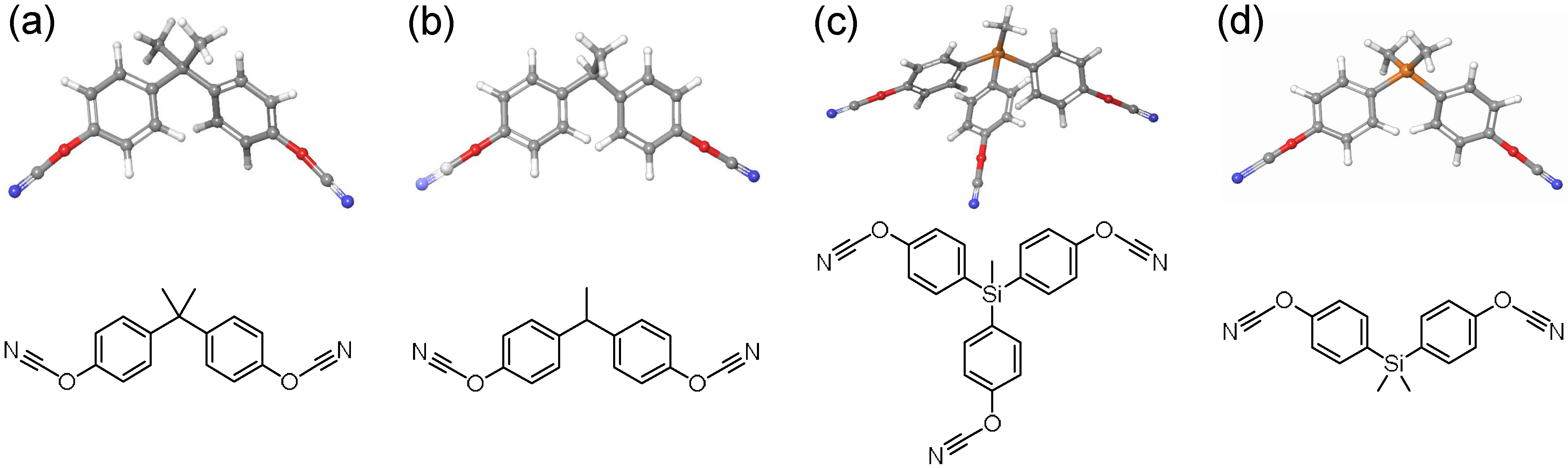





| Monomer | Order Parameter below Tm | Order Parameter above Tm |
|---|---|---|
| BADCy | 0.515 (300 K) | 0.09 (450 K) |
| LECy | 0.41 (250 K) | 0.06 (400 K) |
| SiCy-3 | 0.61 (350 K) | 0.06 (500 K) |
| SiMCy | 0.24 (300 K) | 0.03 (450 K) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haber, R.T.; Browning, A.R.; Graves, B.R.; Davis, W.P.; Wiggins, J.S. A Molecular Dynamics Study of Cyanate Ester Monomer Melt Properties. Polymers 2022, 14, 1219. https://doi.org/10.3390/polym14061219
Haber RT, Browning AR, Graves BR, Davis WP, Wiggins JS. A Molecular Dynamics Study of Cyanate Ester Monomer Melt Properties. Polymers. 2022; 14(6):1219. https://doi.org/10.3390/polym14061219
Chicago/Turabian StyleHaber, Rebecca T., Andrea R. Browning, Bayleigh R. Graves, William P. Davis, and Jeffrey S. Wiggins. 2022. "A Molecular Dynamics Study of Cyanate Ester Monomer Melt Properties" Polymers 14, no. 6: 1219. https://doi.org/10.3390/polym14061219
APA StyleHaber, R. T., Browning, A. R., Graves, B. R., Davis, W. P., & Wiggins, J. S. (2022). A Molecular Dynamics Study of Cyanate Ester Monomer Melt Properties. Polymers, 14(6), 1219. https://doi.org/10.3390/polym14061219







