Synthesis and Performance of a Novel Cotton Linter Based Cellulose Derivatives Dispersant for Coal–Water Slurries
Abstract
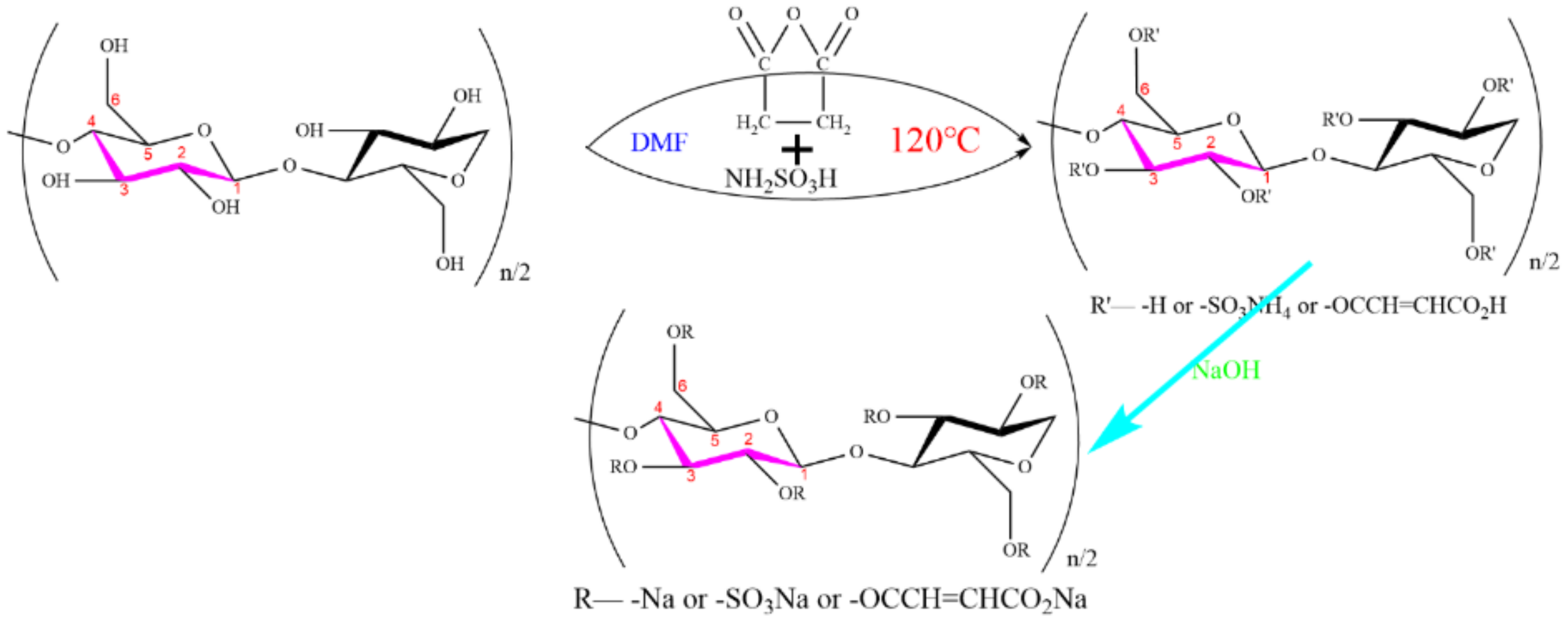
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Determination of Degree of Substitution (DS)
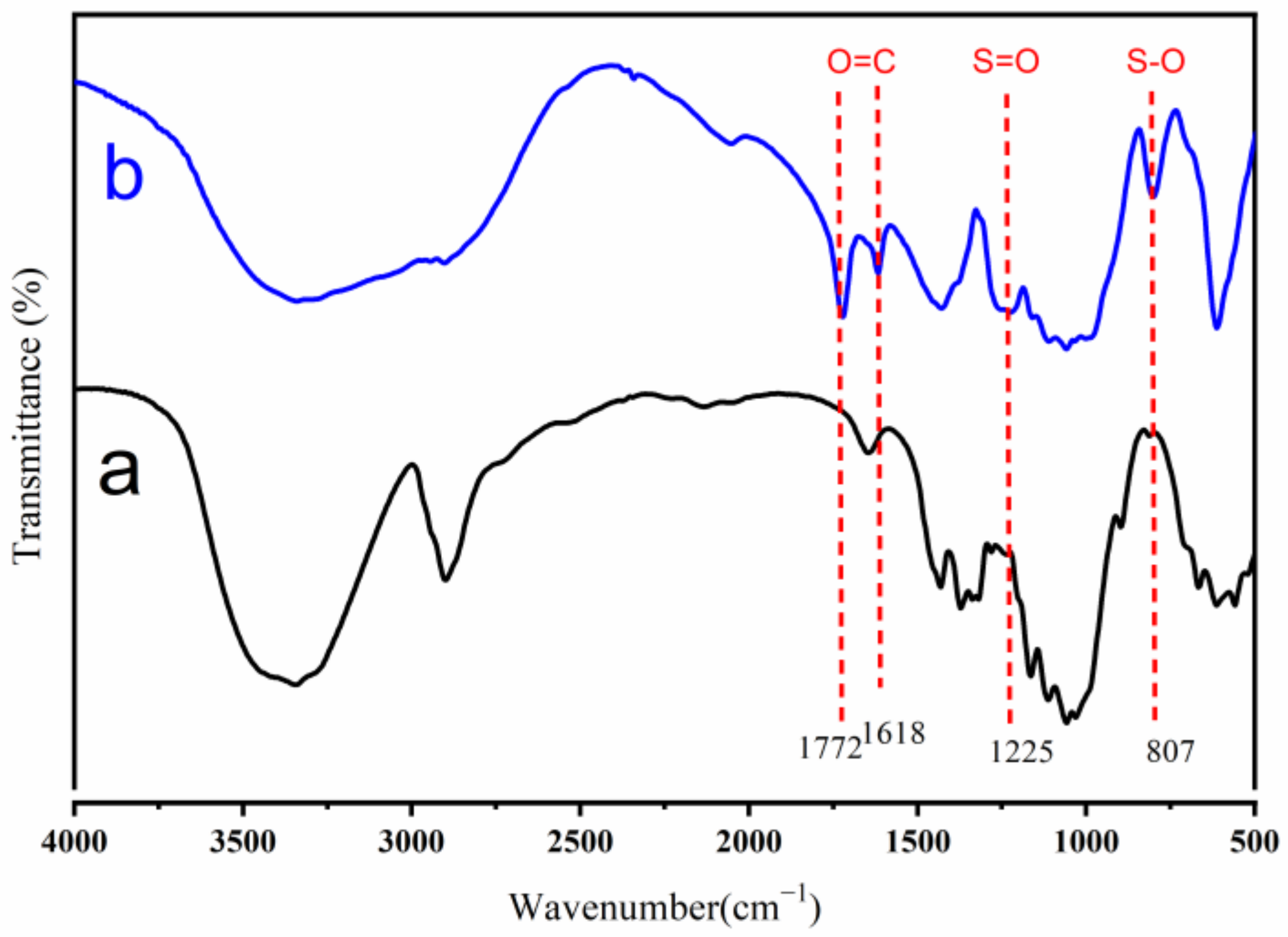
2.2.2. Fourier Transform-Infrared (FT-IR) Spectroscopy
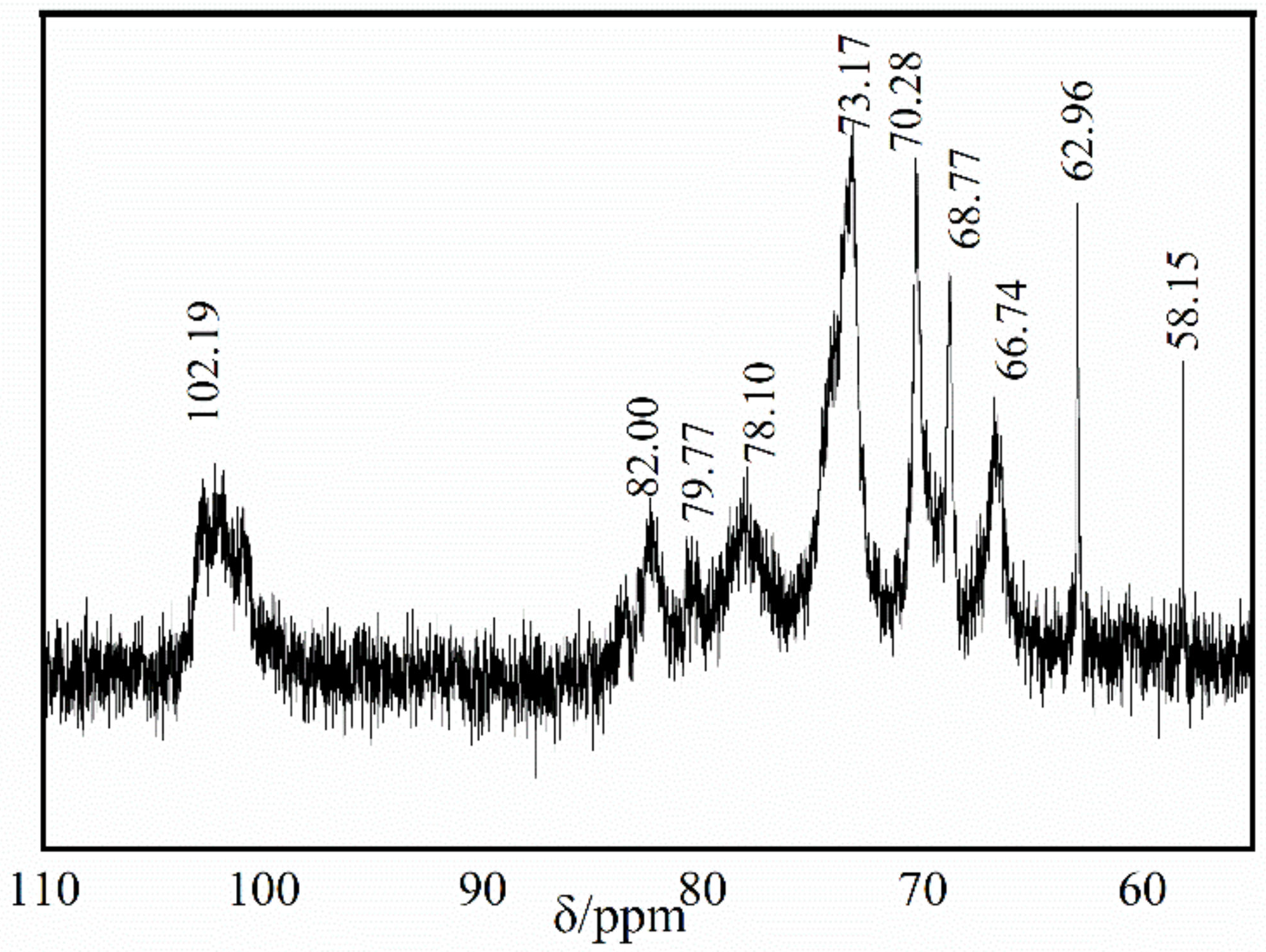
2.2.3. Nuclear Magnetic Resonance Spectroscopy (NMR)
2.2.4. Scanning Electron Microscopy (SEM)
2.2.5. Molecular Weight Distribution
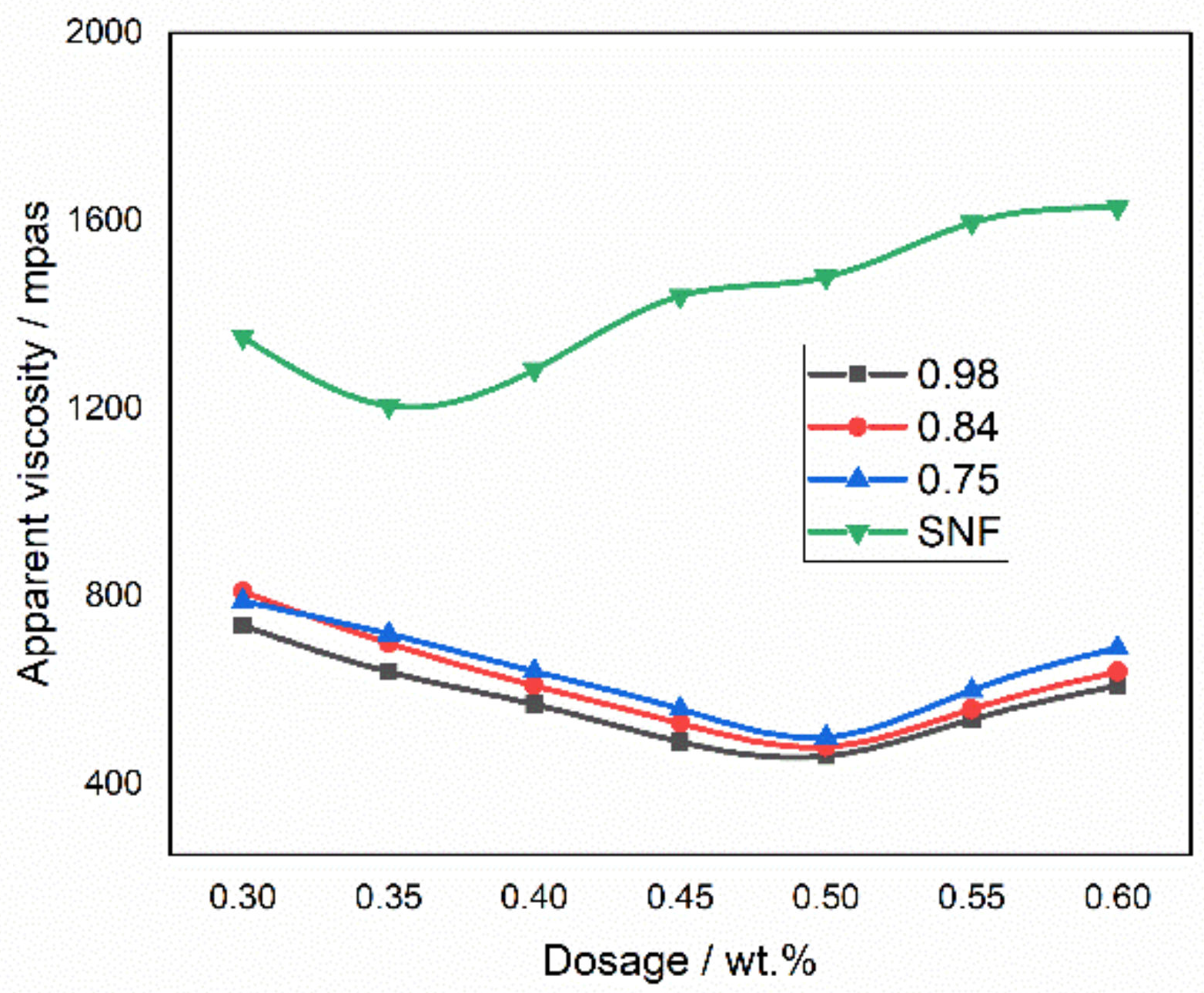
2.2.6. Viscosity Measurement and Rheological Property Determination
2.2.7. Adsorption Amount Measurement
3. Results and Discussion
3.1. FT-IR Analyses
3.2. Morphological Analysis by SEM
3.3. 13C-NMR Characterization
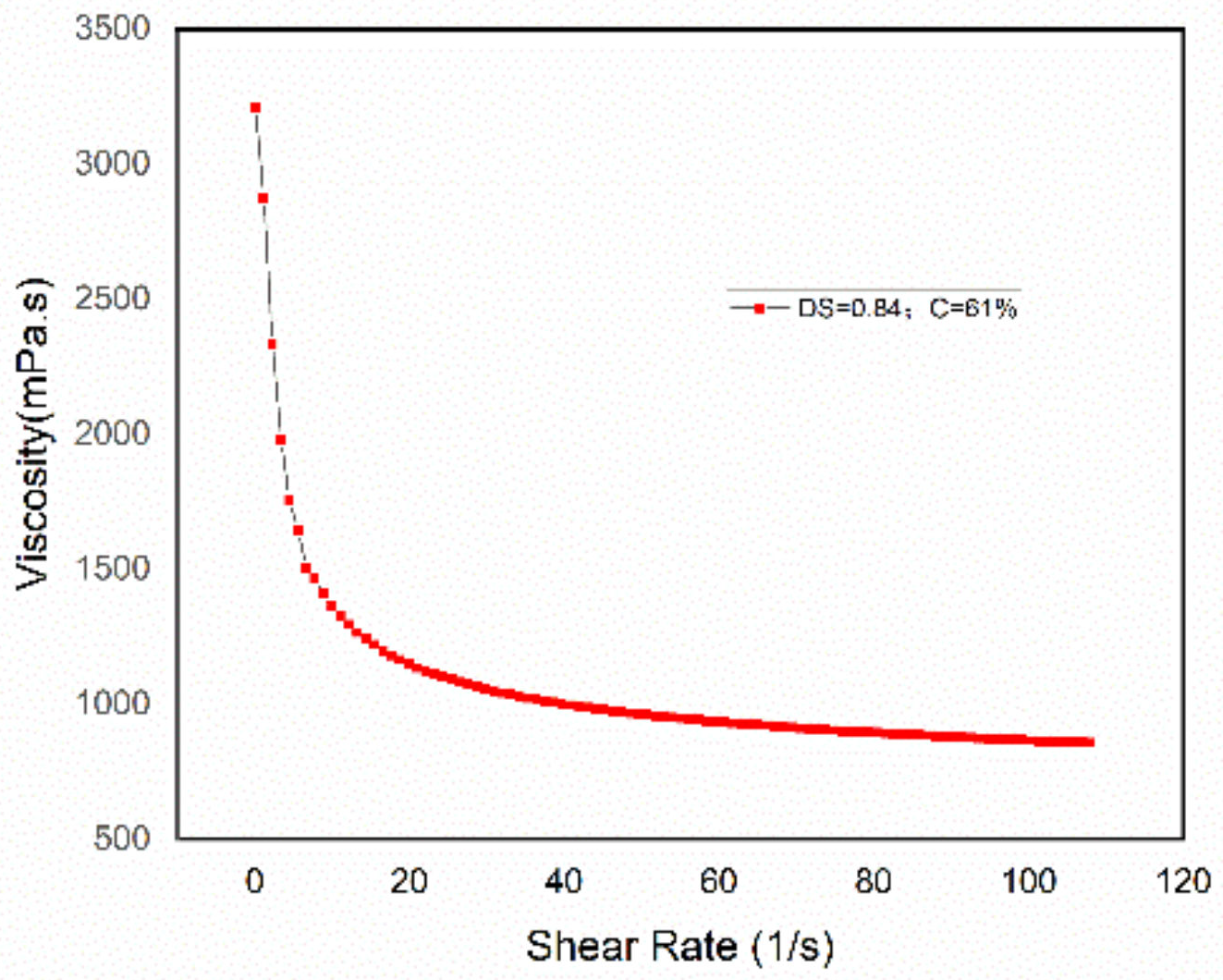
3.4. Viscosity and Rheological Characteristics of CWS
3.5. Static Stability of CWS
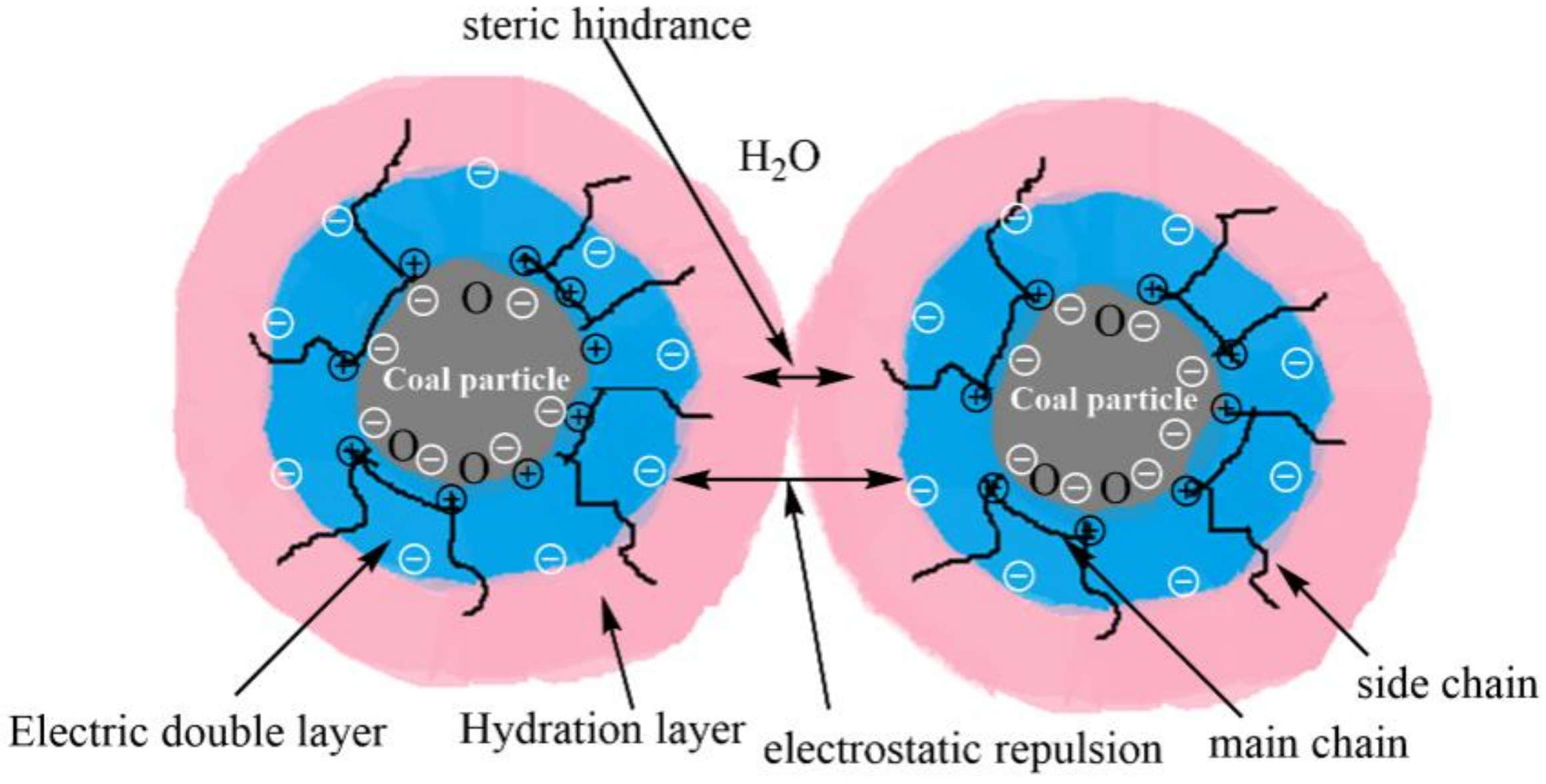
3.6. Adsorption and Dispersion Mechanism of S-CSHE
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| SA | Sulfamic acid: |
| S-CSHE | Sulfonic-cellulose succinate half ester; |
| DS | degree of substitution; |
| FT-IR | Fourier translation infrared spectrum; |
| GPC | Gel Permeation Chromatography; |
| DMF | N,N-Dimethylformamide |
References
- Glushkov, D.O.; Lyrshchikov, S.Y.; Shevyrev, S.A.; Strizhak, P.A. Burning Properties of Slurry Based on Coal and Oil Processing Waste. Energy Fuels 2016, 30, 3441–3450. [Google Scholar] [CrossRef]
- Qiu, X.; Zhou, M.; Yang, D.; Lou, H.; Ouyang, X.; Pang, Y. Evaluation of sulphonated acetone–formaldehyde (SAF) used in coal water slurries prepared from different coals. Fuel 2007, 86, 1439–1445. [Google Scholar] [CrossRef]
- Chen, D.; Jiang, Y.; Jiang, X.; Ma, Z.; Yan, J.; Cen, K.; Yu, X.; Liao, H.; Zhao, H. The effect of anionic dispersants on the moisture distribution of a coal water slurry. Fuel Process. Technol. 2014, 126, 122–130. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Guo, G.Z.; La Zhang, L.; Song, J.H. Synthesis, analysis and application of naphthalene sulfonic acid formaldehyde condensate. Earth Environ. Sci. 2019, 237, 022029. [Google Scholar] [CrossRef]
- Pei, M.; Yang, Y.; Zhang, X.; Zhang, J.; Li, Y. Synthesis and properties of water-soluble sulfonated acetone-formaldehyde resin. J. Appl. Polym. Sci. 2010, 91, 3248–3250. [Google Scholar] [CrossRef]
- Xu, Y.; Chen, H.; Chen, C.; Chu, J.; Zeng, X. Synthesis of sulfonated melamine-p-hydroxybenzoic acid-formaldehyde resin superplasticizer. China Adhes. 2016, 12, 24–26. [Google Scholar]
- Li, L.; Ma, C.; Hu, S.; He, M.; Yu, H.; Wang, Q.; Cao, X.; You, X. Effect of the benzene ring of the dispersant on the rheological characteristics of coal-water slurry: Experiments and theoretical calculations. Int. J. Min. Sci. Technol. 2021, 37, 1283. [Google Scholar] [CrossRef]
- Du, L.; Zhang, G.; Yang, D.; Luo, J.; Liu, Y.; Zhang, W.; Zhang, C.; Li, J.; Zhu, J. Synthesis of a novel amphoteric copolymer and its application as a dispersant for coal water slurry preparation. R. Soc. Open Sci. 2021, 8, 201480. [Google Scholar] [CrossRef]
- Shen, J.; Fatehi, P.; Ni, Y. Biopolymers for surface engineering of paper-based products. Cellulose 2014, 21, 3145–3160. [Google Scholar] [CrossRef]
- Kontturi, E.; Vuorinen, T. Indirect evidence of supramolecular changes within cellulose microfibrils of chemical pulp fibers upon drying. Cellulose 2009, 16, 65–74. [Google Scholar] [CrossRef]
- Omran, A.A.B.; Mohammed, A.A.B.A.; Sapuan, S.M.; Ilyas, R.A.; Asyraf, M.R.M.; Koloor, S.S.R.; Petrů, M. Micro- and Nanocellulose in Polymer Composite Materials: A Review. Polymers 2021, 13, 231. [Google Scholar] [CrossRef]
- Kumar, B.; Agumba, D.; Pham, D.; Latif, M.; Dinesh; Kim, H.; Alrobei, H.; Kim, J. Recent Research Progress on Lignin-Derived Resins for Natural Fiber Composite Applications. Polymers 2021, 13, 1162. [Google Scholar] [CrossRef]
- Al-Rajabi, M.M.; Haan, T.Y. Green Synthesis of Thermo-Responsive Hydrogel from Oil Palm Empty Fruit Bunches Cellulose for Sustained Drug Delivery. Polymers 2021, 13, 2153. [Google Scholar] [CrossRef]
- Chen, T.-H. Mechanical Properties and Tribological Behavior of MoS2-Enhanced Cellulose-Based Biocomposites for Food Packaging. Polymers 2021, 13, 1362. [Google Scholar]
- Ashour, R.M.; Abdel-Magied, A.F.; Wu, Q.; Olsson, R.T.; Forsberg, K. Green Synthesis of Metal-Organic Framework Bacterial Cellulose Nanocomposites for Separation Applications. Polymers 2020, 12, 1104. [Google Scholar] [CrossRef]
- Rouzi, K.; Mamuti, M. Performance of sulfonated cellulose based water-reducing agent for cement. Concrete 2012, 29, 74–76. [Google Scholar]
- Rouzi, K.; Maihemuti, M.; Mamuti, M. Application performance of oxidized and sulfonated cellulose based concrete high range water-reducing agent. New Build. Mater. 2015, 35, 17–21. [Google Scholar]
- Wang, L.J.; Huang, F.Y.; Ma, X.C.; Zhang, H. Synthesis and properties of water-soluble cellulose ether based concrete water-reducing agent. J. Build. Mater. 2008, 11, 52–57. [Google Scholar]
- Kumar, R.S.; Nagarajan, R.; Vijay, K.; Perumal, P.T. Sulfamic Acid: An Efficient Catalyst for the Diastereoselective, One-Pot Synthesis of Pyrano Benzopyrans, Furano Benzopyrans and Ethoxy Benzopyrans. Lett. Org. Chem. 2005, 2, 458–460. [Google Scholar] [CrossRef]
- Shetty, M.R.; Samant, S.D. Sulfamic Acid (H2NSO3H): A Low-Cost, Mild, and Efficient Catalyst for the Synthesis of Substituted N-Phenylpyrazoles Under Solvent-Free Conditions. Synth. Commun. 2012, 42, 1411–1418. [Google Scholar] [CrossRef]
- Xia, M.; Wu, B.; Xiang, G.F. Sulfamic Acid as an Effective Catalyst in Solvent-Free Synthesis of β-Enaminoketone Derivatives and X-ray Crystallography of Their Representatives. Synth. Commun. 2008, 39, 1268–1278. [Google Scholar] [CrossRef]
- Zhang, Z.; Ma, B.; Zhu, Q.; Ding, Y.; Wang, C.; Song, W. ChemInform Abstract: Sulfamic Acid as a Cost-Effective Catalyst for Synthesis of α-Acyloxyacrylate Esters as Candidate Monomers for Biobased Polymers by Acylation of Pyruvate Esters. Synth. Commun. 2012, 43, 3053–3060. [Google Scholar] [CrossRef]
- Wang, B.; Gu, Y.; Song, G.; Yang, T.; Yang, L.; Suo, J. An efficient procedure for protection of carbonyls catalyzed by sulfamic acid. J. Mol. Catal. A Chem. 2005, 233, 121–126. [Google Scholar] [CrossRef]
- Upadhyaya, D.J.; Barge, A.; Stefania, R.; Cravotto, G. Efficient, solventless N-Boc protection of amines carried out at room temperature using sulfamic acid as recyclable catalyst. ChemInform 2008, 39, 8318–8322. [Google Scholar]
- Abdul Khalil, H.P.S.; Davoudpour, Y.; Islam, M.N.; Mustapha, A.; Sudesh, K.; Dungani, R.; Jawaid, M. Production and modification of nanofibrillated cellulose using various mechanical processes: A review. Carbohydr. Polym. 2014, 99, 649–665. [Google Scholar] [CrossRef]
- Wang, L.-F.; Pan, S.-Y.; Hu, H.; Miao, W.-H.; Xu, X.-Y. Synthesis and properties of carboxymethyl kudzu root starch. Carbohydr. Polym. 2010, 80, 174–179. [Google Scholar] [CrossRef]
- Ibrahim, M.M.E.-Z.; Waleed, K. Preparation characterization and properties of fast-responding bulk hydrogel and treated bleached banana pulp. Polym. Adv. Technol. 2004, 15, 275–283. [Google Scholar] [CrossRef]
- El-Sukkary, M.M.A.; Syed, N.A.; Aiad, I.; El-Azab, W. Synthesis and Characterization of some Alkyl Polyglycosides Surfactants. J. Surfactants Deterg. 2008, 11, 129–137. [Google Scholar] [CrossRef]
- Nehls, I.; Wagenknecht, W.; Philipp, B.; Stscherbina, D. Characterization of cellulose and cellulose derivatives in solution by high resolution 13C-NMR spectroscopy. Prog. Polym. Sci. 1994, 19, 29–78. [Google Scholar] [CrossRef]
- Philipp, B.; Nehls, I.; Wagenknecht, W.; Schnabelrauch, M. 13C-NMR spectroscopic study of the homogeneous sulphation of cellulose and xylan in the N2O4-DMF system. Carbohydr. Res. 1987, 164, 107–116. [Google Scholar] [CrossRef]
- Zhang, G.; Li, J.; Zhu, J.; Qu, Q.; Xiong, W. Syntheses and evaluations of three sulfonated polycondensate dispersants for coal–water slurries. Powder Technol. 2014, 254, 572–578. [Google Scholar] [CrossRef]
- Qin, Y.; Yang, D.; Guo, W.; Qiu, X. Investigation of grafted sulfonated alkali lignin polymer as dispersant in coal-water slurry. J. Ind. Eng. Chem. 2015, 27, 192–200. [Google Scholar] [CrossRef]
- Gibis, M.; Schuh, V.; Allard, K.; Weiss, J. Influence of molecular weight and degree of substitution of various carboxymethyl celluloses on unheated and heated emulsion-type sausage models. Carbohydr. Polym. 2017, 159, 76–85. [Google Scholar] [CrossRef]
- Zhang, K.; Jin, L.; Cao, Q. Evaluation of modified used engine oil acting as a dispersant for concentrated coal–water slurry. Fuel 2016, 175, 202–209. [Google Scholar] [CrossRef]
- Wang, R.; Liu, J.; Yu, Y.; Hu, Y.; Zhou, J.; Cen, K. The Slurrying Properties of Coal Water Slurries Containing Raw Sewage Sludge. Energy Fuels 2011, 25, 747–752. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, G.; Miao, Z.; Shang, T. Synthesis and performance of a comblike amphoteric polycarboxylate dispersant for coal–water slurry. Colloids Surf. A Physicochem. Eng. Asp. 2012, 412, 101–107. [Google Scholar] [CrossRef]
- Huang, J.; Xu, J.; Wang, D.; Li, L.; Guo, X. Effects of Amphiphilic Copolymer Dispersants on Rheology and Stability of Coal Water Slurry. Ind. Eng. Chem. Res. 2013, 52, 8427–8435. [Google Scholar] [CrossRef]
- Phulkerd, P.; Thongchul, N.; Bunyakiat, K.; Petsom, A. Coal water slurry using dispersant synthesized from cashew nut shell liquid (CNSL). Fuel Process. Technol. 2014, 119, 256–262. [Google Scholar] [CrossRef]
- Zhang, G.-H.; Zhu, N.; Li, Y.-B.; Zhu, J.-F.; Jia, Y.-R.; Ge, L. Influence of side-chain structure of polycarboxylate dispersant on the performance of coal water slurry. Fuel Process. Technol. 2017, 161, 1–7. [Google Scholar] [CrossRef]
- Zhu, J.; Wang, P.; Li, Y.; Li, J.; Zhang, G. Dispersion performance and mechanism of polycarboxylates bearing side chains of moderate length in coal-water slurries. Fuel 2016, 190, 221–228. [Google Scholar] [CrossRef]


| Sample | Proximate Analysis (wt.%) | Ultimate Analysis (wt.%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Inherent Moisture | Ash | Volatile Matter | Carbon | Hydrogen | Oxygen | Nitrogen | Sulfur | |
| ZhunDong coal | 9.14 | 5.86 | 37.21 | 67.71 | 5.27 | 16.84 | 1.24 | 0.57 |
| Coal Particle Size (Mesh) | 20–40 | 40–120 | 120–200 | 200–300 | <325 |
|---|---|---|---|---|---|
| Coal particle size distribution (wt.%) | 10 | 39 | 11 | 9 | 31 |
| No. | DS | Mw |
|---|---|---|
| S-CSHE1 | 0.75 | 1.2 × 104 |
| S-CSHE2 | 0.84 | 6.5 × 103 |
| S-CSHE3 | 0.98 | 2.2 × 103 |
| Sample | Herschel–Bulkley | |||
|---|---|---|---|---|
| τ0 (Pa) | K (Pa.sn) | n | R2 | |
| Zhundong | 3.21 | 0.48 | 0.86 | 0.9938 |
| Coal Content (wt.%) | Water Separation Ratio (%) | ||
|---|---|---|---|
| 0.75 | 0.84 | 0.98 | |
| 58.51 | 9.7 | 10.4 | 12.3 |
| 59.45 | 9.3 | 9.8 | 11.8 |
| 61.32 | 8.2 | 9.2 | 10.2 |
| 63.03 | 7.6 | 8.8 | 9.8 |
| 63.94 | 7.2 | 8.2 | 9.5 |
| 65.02 | 7.1 | 8.0 | 9.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, C.; Zhu, X.; Ma, X.; Yang, H. Synthesis and Performance of a Novel Cotton Linter Based Cellulose Derivatives Dispersant for Coal–Water Slurries. Polymers 2022, 14, 1103. https://doi.org/10.3390/polym14061103
Ding C, Zhu X, Ma X, Yang H. Synthesis and Performance of a Novel Cotton Linter Based Cellulose Derivatives Dispersant for Coal–Water Slurries. Polymers. 2022; 14(6):1103. https://doi.org/10.3390/polym14061103
Chicago/Turabian StyleDing, Chengli, Xiao Zhu, Xue Ma, and Hongsheng Yang. 2022. "Synthesis and Performance of a Novel Cotton Linter Based Cellulose Derivatives Dispersant for Coal–Water Slurries" Polymers 14, no. 6: 1103. https://doi.org/10.3390/polym14061103
APA StyleDing, C., Zhu, X., Ma, X., & Yang, H. (2022). Synthesis and Performance of a Novel Cotton Linter Based Cellulose Derivatives Dispersant for Coal–Water Slurries. Polymers, 14(6), 1103. https://doi.org/10.3390/polym14061103





