Polymeric Membranes for Oil-Water Separation: A Review
Abstract
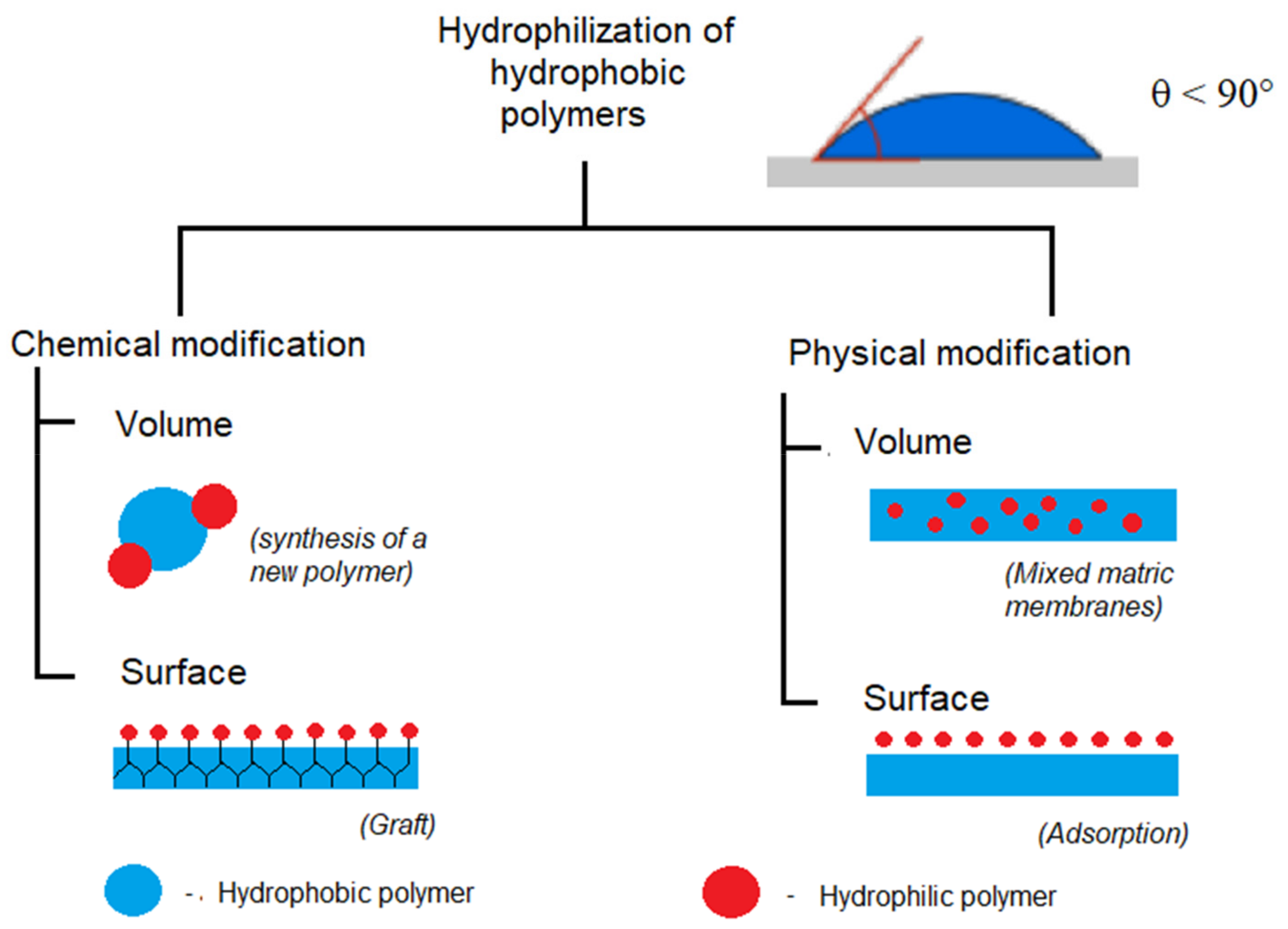
:1. Introduction
2. Commercial Polymer Membranes
- Ability to control the process and change the desired grafting density by changing the concentrations and time;
- Precise localization of functional groups on the surface;
- Long-term chemical stability, which is provided by the covalent attachment of functional groups.
2.1. Polysulfone, Polyethersulfone
- For the same pressure drop, permeate flow is higher for membranes with greater porosity;
- The optimal pressure drop is 100 kPa, since in this case the best combination of the permeate flow with the level of oil retention is achieved for all membrane options;
- The pore size and overall porosity of the membranes decreased with the increasing content of polysulfone in the casting composition.
2.2. Acrylonitrile Polyacrylonitrile
2.3. Polyvinylidene Fluoride
2.4. Other Commercial Polymers
2.5. Natural Polymers
2.6. Polymers as Thin Selective Layers on Inorganic Support
3. Membranes Formed by Electrospinning
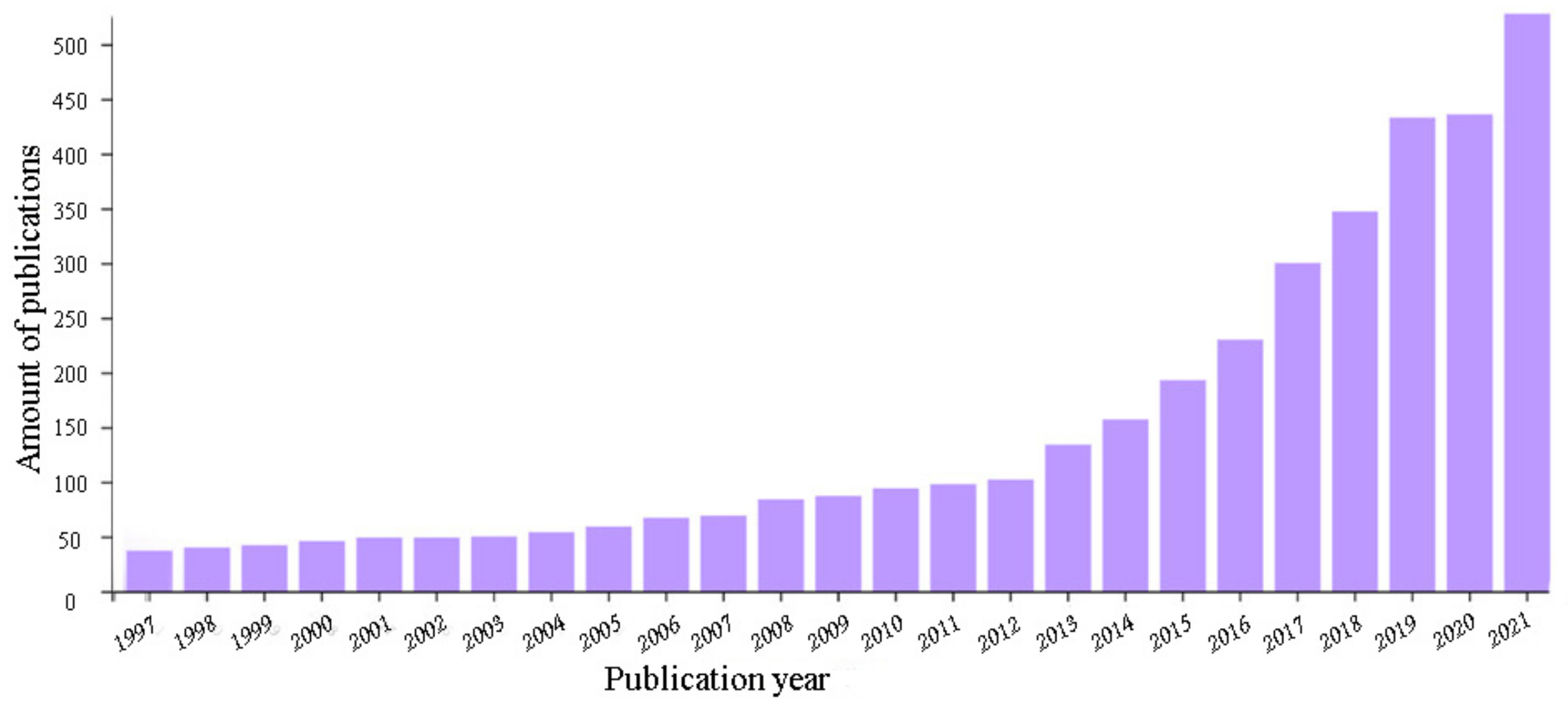
4. The Problem of Comparison of Literature Data
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xue, J.; Zhu, L.; Zhu, X.; Li, H.; Ma, C.; Yu, S.; Xue, Q. Tetradecylamine-MXene functionalized melamine sponge for effective oil/water separation and selective oil adsorption. Sep. Purif. Technol. 2021, 259, 118106. [Google Scholar] [CrossRef]
- Yang, S.; Chen, L.; Liu, S.; Hou, W.; Zhu, J.; Zhao, P.; Zhang, Q. Facile and sustainable fabrication of high-performance cellulose sponge from cotton for oil-in-water emulsion separation. J. Hazard. Mater. 2021, 408, 124408. [Google Scholar] [CrossRef] [PubMed]
- Hailan, S.M.; Ponnamma, D.; Krupa, I. The Separation of Oil/Water Mixtures by Modified Melamine and Polyurethane Foams: A Review. Polymers 2021, 13, 4142. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Jiang, X.; Goh, K.L.; Wang, K. The separation of oily water using low-cost natural materials: Review and development. Chemosphere 2021, 285, 131398. [Google Scholar] [CrossRef] [PubMed]
- Gafonova, O.V. Role of Asphaltenes and Resins in the Stabilization of Water-in-Hydrocarbon Emulsions; University of Calgary: Calgary, AB, Canada, 2001. [Google Scholar] [CrossRef]
- Mya, O.B.; Houga, S.; Chihouba, F.; Asla, B. Treatment of Algerian crude oil using REB09305 OS demulsifier. Egypt. J. Pet. 2018, 27, 769–773. [Google Scholar] [CrossRef]
- Liu, J.; Wang, H.; Li, X.; Jia, W.; Zhao, Y.; Ren, S. Recyclable magnetic graphene oxide for rapid and efficient demulsification of crude oil-in-water emulsion. Fuel 2017, 189, 79–87. [Google Scholar] [CrossRef] [Green Version]
- Roostaie, T.; Farsi, M.; Rahimpour, M.R.; Biniaz, P. Performance of biodegradable cellulose based agents for demulsification of crude oil: Dehydration capacity and rate. Sep. Purif. Technol. 2017, 179, 291–296. [Google Scholar] [CrossRef]
- Svarovskaya, L.I.; Manakov, A.Y.; Altunina, L.K.; Strelets, L.A. Bio-inert Interactions in an Oil—Microorganisms System. In Processes and Phenomena on the Boundary between Biogenic and Abiogenic Nature; Springer: Cham, Switzerland, 2020; pp. 195–205. [Google Scholar]
- Hao, M.; Bai, Z.; Wang, H.; Liu, W. Removal of oil from electric desalting wastewater using centrifugal contactors. J. Pet. Sci. Eng. 2013, 111, 37–41. [Google Scholar] [CrossRef]
- Adeyanju, O.A.; Ogundare, G. Experimental Investigation of the Centrifugal Effect on Demulsification of Water in Crude Oil Emulsion. J. Nat. Sci. Sustain. Technol. 2019, 13, 1–12. [Google Scholar]
- Issaka, S.A.; Nour, A.H.; Yunus, R.M. Review on the fundamental aspects of petroleum oil emulsions and techniques of demulsification. J. Pet. Environ. Biotechnol. 2015, 6, 1000214. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Palou, R.; Cerón-Camacho, R.; Chávez, B.; Vallejo, A.A.; Villanueva-Negrete, D.; Castellanos, J.; Aburto, J. Demulsification of heavy crude oil-in-water emulsions: A comparative study between microwave and thermal heating. Fuel 2013, 113, 407–414. [Google Scholar] [CrossRef]
- Binner, E.R.; Robinson, J.P.; Silvester, S.A.; Kingman, S.W.; Lester, E.H. Investigation into the mechanisms by which microwave heating enhances separation of water-in-oil emulsions. Fuel 2014, 116, 516–521. [Google Scholar] [CrossRef]
- Santos, D.; da Rocha, E.C.; Santos, R.L.; Cancelas, A.J.; Franceschi, E.; Santos, A.F.; Dariva, C. Demulsification of water-in-crude oil emulsions using single mode and multimode microwave irradiation. Sep. Purif. Technol. 2017, 189, 347–356. [Google Scholar] [CrossRef]
- Less, S.; Hannisdal, A.; Bjørklund, E.; Sjöblom, J. Electrostatic destabilization of water-in-crude oil emulsions: Application to a real case and evaluation of the Aibel VIEC technology. Fuel 2008, 87, 2572–2581. [Google Scholar] [CrossRef]
- Zhang, L.; He, L.; Ghadiri, M.; Hassanpour, A. Effect of surfactants on the deformation and break-up of an aqueous drop in oils under high electric field strengths. J. Pet. Sci. Eng. 2005, 125, 38–47. [Google Scholar] [CrossRef] [Green Version]
- Antes, F.G.; Diehl, L.O.; Pereira, J.S.; Guimarães, R.C.; Guarnieri, R.A.; Ferreira, B.M.; Flores, E.M. Feasibility of low frequency ultrasound for water removal from crude oil emulsions. Ultrason. Sonochem. 2015, 25, 70–75. [Google Scholar] [CrossRef]
- Xie, W.; Li, R.; Lu, X. Pulsed ultrasound assisted dehydration of waste oil. Ultrason. Sonochem. 2015, 26, 136–141. [Google Scholar] [CrossRef]
- Zhang, W.; Xiao, P.; Wang, D. Central treatment of different emulsion wastewaters by an integrated process of physicochemically enhanced ultrafiltration and anaerobic–aerobic biofilm reactor. Bioresour. Technol. 2014, 159, 150–156. [Google Scholar] [CrossRef]
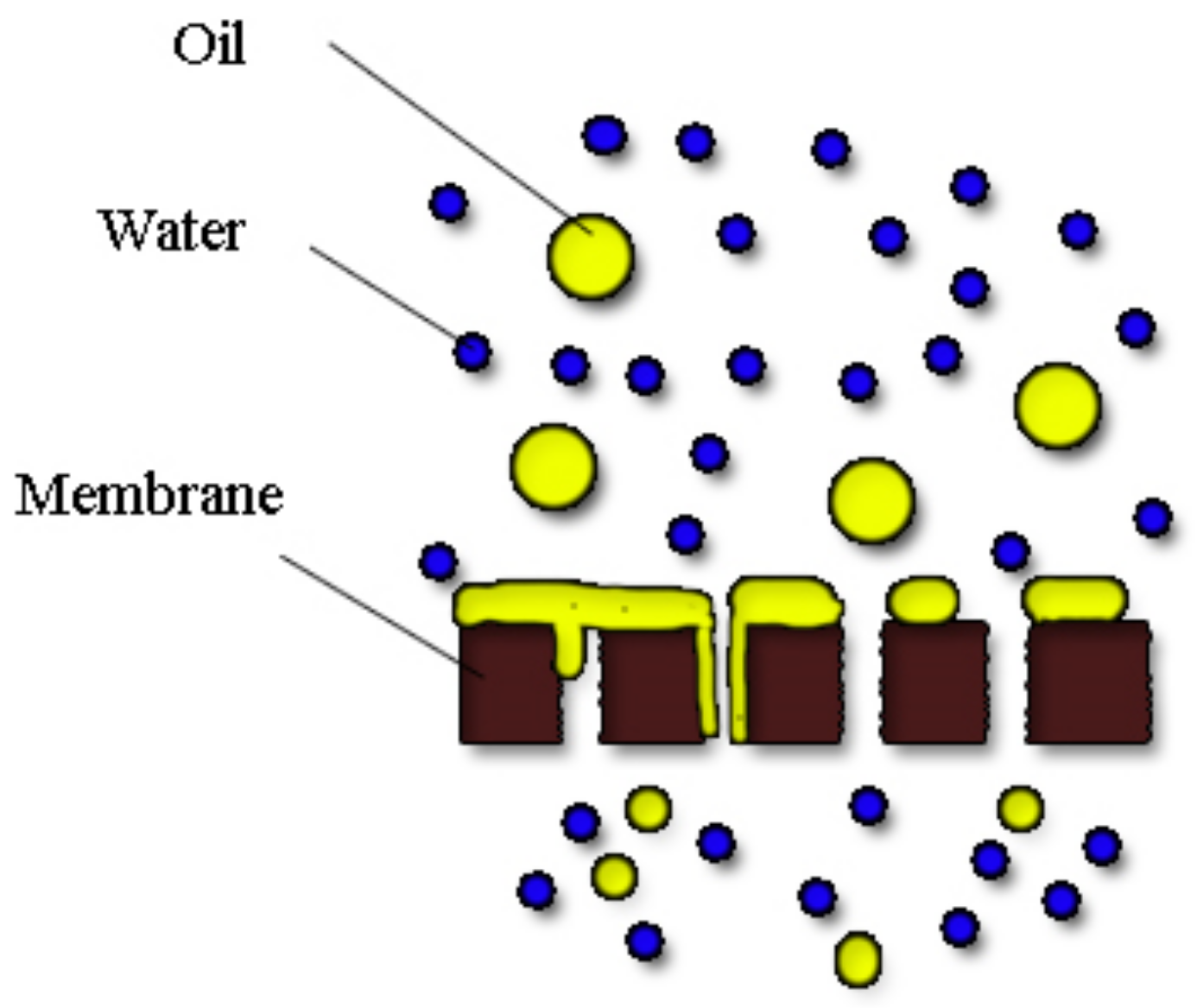
- Padaki, M.; Murali, R.S.; Abdullah, M.S.; Misdan, N.; Moslehyani, A.; Kassim, M.A.; Ismail, A.F. Membrane technology enhancement in oil–water separation. A review. Desalination 2015, 357, 197–207. [Google Scholar] [CrossRef]
- Cheryan, M.; Rajagopalan, N. Membrane processing of oily streams. Wastewater treatment and waste reduction. J. Membr. Sci. 1998, 151, 13–28. [Google Scholar] [CrossRef]
- Tanudjaja, H.J.; Hejase, C.A.; Tarabara, V.V.; Fane, A.G.; Chew, J.W. Membrane-based separation for oily wastewater: A practical perspective. Water Res. 2019, 156, 347–365. [Google Scholar] [CrossRef] [PubMed]
- Shalaby, M.S.; Sołowski, G.; Abbas, W. Recent Aspects in Membrane Separation for Oil/Water Emulsion. Adv. Mater. Interfaces 2021, 8, 2100448. [Google Scholar] [CrossRef]
- Lee, B.; Patel, R. Review on oil/water separation membrane technology. Membr. J. 2020, 30, 359–372. [Google Scholar] [CrossRef]
- Clark, C.E.; Veil, J.A. Produced Water Volumes and Management Practices in the United States, ANL/EVS/R-09/1, Prepared for the U.S. Department of Energy; National Energy Technology Laboratory: Lemont, IL, USA, 2009. [Google Scholar]
- Igunnu, E.T.; Chen, G.Z. Produced water treatment technologies. Int. J. Low Carbon Technol. 2012, 9, 157–177. [Google Scholar] [CrossRef] [Green Version]
- Baker, R.W. Membrane Technology and Applications, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2012; ISBN 0-470-85445-6. [Google Scholar]
- Hong, A.C.; Fane, A.G.; Burford, R.P. The effects of intermittent permeate flow and crossflow on membrane coalescence of oil-in-water emulsions. Desalination 2002, 144, 185–191. [Google Scholar] [CrossRef]
- Tummons, E.; Qi, H.; Tanudjaja, H.J.; Hejase, C.A.; Wei Chew, J.; Tarabara, V.V. Membrane fouling by emulsified oil: A review. Sep. Purif. Technol. 2020, 248, 116919. [Google Scholar] [CrossRef]
- Alent’ev, A.Y.; Volkov, A.V.; Vorotyntsev, I.V.; Maksimov, A.L.; Yaroslavtsev, A.B. Membrane Technologies for Decarbonization. Membr. Membr. Technol. 2021, 3, 255–273. [Google Scholar] [CrossRef]
- Fazullin, D.D.; Mavrin, G.V.; Shaikhiev, I.G.; Nizameev, I.R. Microwave Stabilization of a Dynamic Membrane Layer. Membr. Membr. Technol. 2019, 1, 6–13. [Google Scholar] [CrossRef] [Green Version]
- Osadchii, Y.P.; Nikiforova, T.E. A model of separation of wastewater containing vat dyes. Russ. J. Appl. Chem. 2007, 80, 1680–1682. [Google Scholar] [CrossRef]
- Hoek, E.M.V.; Tarabara, V.V. (Eds.) Encyclopedia of Membrane Science and Technology; Wiley: Hoboken, NJ, USA, 2013; Volume 3, ISBN 978-0-470-90687-3. [Google Scholar]
- Fane, A.G.; Wang, R.; Hu, M.X. Synthetic membranes for water purification: Status and future. Angew. Chem. Int. Ed. 2015, 54, 2–21. [Google Scholar] [CrossRef]
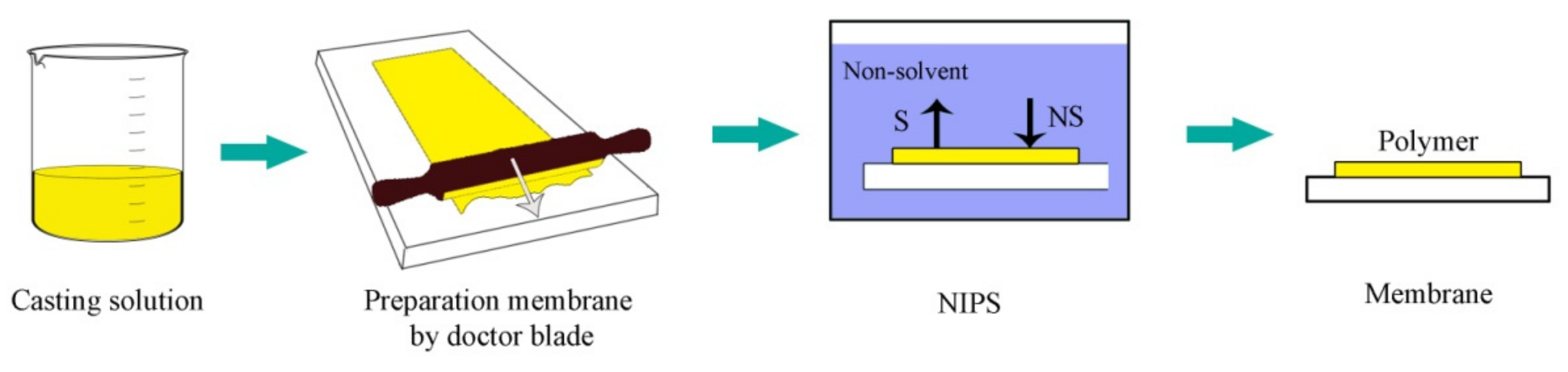
- Guillen, G.R.; Pan, Y.; Li, M.; Hoek, E.M. Preparation and characterization of membranes formed by nonsolvent induced phase separation: A review. Ind. Eng. Chem. Res. 2011, 50, 3798–3817. [Google Scholar] [CrossRef]
- Kim, J.F.; Kim, J.H.; Lee, Y.M.; Drioli, E. Thermally induced phase separation and electrospinning methods for emerging membrane applications: A review. AIChE J. 2016, 62, 461–490. [Google Scholar] [CrossRef]
- Liu, M.; Liu, S.; Xu, Z.; Wei, Y.; Yang, H. Formation of microporous polymeric membranes via thermally induced phase separation: A review. Front. Chem. Sci. Eng. 2016, 10, 57–75. [Google Scholar] [CrossRef]
- Venault, A.; Chang, Y.; Wang, D.M.; Bouyer, D. A review on polymeric membranes and hydrogels prepared by vapor-induced phase separation process. Polym. Rev. 2013, 53, 568–626. [Google Scholar] [CrossRef]
- Ismail, N.; Venault, A.; Mikkola, J.P.; Bouyer, D.; Drioli, E.; Kiadeh NT, H. Investigating the potential of membranes formed by the vapor induced phase separation process. J. Membr. Sci. 2020, 597, 117601. [Google Scholar] [CrossRef]
- Wang, D.M.; Lai, J.Y. Recent advances in preparation and morphology control of polymeric membranes formed by nonsolvent induced phase separation. Curr. Opin. Chem. Eng. 2013, 2, 229–237. [Google Scholar] [CrossRef]
- Matveev, D.; Vasilevsky, V.; Volkov, V.; Plisko, T.; Shustikov, A.; Volkov, A.; Bildyukevich, A. Fabrication of ultrafiltration membranes from non-toxic solvent dimethylsulfoxide: Benchmarking of commercially available acrylonitrile co-polymers. J. Environ. Chem. Eng. 2022, 10, 107061. [Google Scholar] [CrossRef]
- Bormashenko, E.; Pogreb, R.; Whyman, G.; Bormashenko, Y.; Jager, R.; Stein, T.; Aurbach, D. The reversible giant change in the contact angle on the polysulfone and polyethersulfone films exposed to UV irradiation. Langmuir 2008, 24, 5977–5980. [Google Scholar] [CrossRef]
- Wang, S.; Li, Y.; Fei, X.; Sun, M.; Zhang, C.; Li, Y.; Hong, X. Preparation of a durable superhydrophobic membrane by electrospinning poly (vinylidene fluoride)(PVDF) mixed with epoxy–siloxane modified SiO2 nanoparticles: A possible route to superhydrophobic surfaces with low water sliding angle and high water contact angle. J. Colloid Interface Sci. 2011, 359, 380–388. [Google Scholar] [CrossRef]
- Gao, S.J.; Zhu, Y.Z.; Zhang, F.; Jin, J. Superwetting polymer-decorated SWCNT composite ultrathin films for ultrafast separation of oil-in-water nanoemulsions. J. Mater. Chem. A 2015, 3, 2895–2902. [Google Scholar] [CrossRef]
- Wang, Z.; Jiang, X.; Cheng, X.; Lau, C.H.; Shao, L. Mussel-Inspired Hybrid Coatings that Transform Membrane Hydrophobicity into High Hydrophilicity and Underwater Superoleophobicity for Oil-in-Water Emulsion Separation. ACS Appl. Mater. Interfaces 2015, 7, 9534–9545. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Nandi, B.K.; Guria, C.; Mandal, A. Oil removal from produced water by ultrafiltration using polysulfone membrane. Braz. J. Chem. Eng. 2017, 34, 583–596. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Zhu, L.; Zhang, J.; Guo, T.; Li, X.; Xing, W.; Xue, Q. High-efficiency separation performance of oil-water emulsions of polyacrylonitrile nanofibrous membrane decorated with metal-organic frameworks. Appl. Surf. Sci. 2019, 476, 61–69. [Google Scholar] [CrossRef]
- Chen, W.; Su, Y.; Zheng, L.; Wang, L.; Jiang, Z. The improved oil/water separation performance of cellulose acetate-graft-polyacrylonitrile membranes. J. Membr. Sci. 2009, 337, 98–105. [Google Scholar] [CrossRef]
- Zhang, F.; Gao, S.; Zhu, Y.; Jin, J. Alkaline-induced superhydrophilic/underwater superoleophobic polyacrylonitrile membranes with ultralow oil-adhesion for high-efficient oil/water separation. J. Membr. Sci. 2016, 513, 67–73. [Google Scholar] [CrossRef]
- Malakhov, A.O.; Anokhina, T.S.; Petrova, D.A.; Vinokurov, V.A.; Volkov, A.V. Nanocellulose as a component of ultrafiltration membranes. Pet. Chem. 2018, 58, 923–933. [Google Scholar] [CrossRef]
- Minko, S. Grafting on solid surfaces:“grafting to” and “grafting from” methods. In Polymer Surfaces and Interfaces; Springer: Berlin/Heidelberg, Germany, 2008; pp. 215–234. [Google Scholar] [CrossRef]
- Mansourizadeh, A.; Javadi Azad, A. Preparation of blend polyethersulfone/cellulose acetate/polyethylene glycol asymmetric membranes for oil–water separation. J. Polym. Res. 2014, 21, 375. [Google Scholar] [CrossRef]
- Xu, Z.L.; Chung, T.S.; Loh, K.C.; Lim, B.C. Polymeric asymmetric membranes made from polyetherimide/polybenzimidazole/poly (ethylene glycol) (PEI/PBI/PEG) for oil–surfactant–water separation. J. Membr. Sci. 1999, 158, 41–53. [Google Scholar] [CrossRef]
- Meng, H.; Xu, T.; Gao, M.; Bai, J.; Li, C. An oil-contamination-resistant PVP/PAN electrospinning membrane for high-efficient oil–water mixture and emulsion separation. J. Appl. Polym. Sci. 2021, 138, 50043. [Google Scholar] [CrossRef]
- Ghandashtani, M.B.; Ashtiani, F.Z.; Karimi, M.; Fouladitajar, A. A novel approach to fabricate high performance nano-SiO2 embedded PES membranes for microfiltration of oil-in-water emulsion. Appl. Surf. Sci. 2015, 349, 393–402. [Google Scholar] [CrossRef]
- Mousa, H.M.; Alfadhel, H.; Ateia, M.; Abdel-Jaber, G.T. Polysulfone-iron acetate/polyamide nanocomposite membrane for oil-water separation. Environ. Nanotechnol. Monit. Manag. 2020, 14, 100314. [Google Scholar]
- Yang, M.; Zhao, C.; Zhang, S.; Li, P.; Hou, D. Preparation of graphene oxide modified poly(m-phenylene isophthalamide) nanofiltration membrane with improved water flux and antifouling property. Appl. Surf. Sci. 2017, 394, 149–159. [Google Scholar] [CrossRef]
- Li, S.; Huang, J.; Chen, Z.; Chen, G.; Lai, Y. A review on special wettability textiles: Theoretical models, fabrication technologies and multifunctional applications. J. Mater. Chem. A 2017, 5, 31–55. [Google Scholar] [CrossRef] [Green Version]
- Xu, D.; Zheng, X.; Xiao, R. Hydrophilic nanofibrous composite membrane prepared by melt-blending extrusion for effective separation of oil/water emulsion. RSC Adv. 2017, 7, 7108–7115. [Google Scholar] [CrossRef] [Green Version]
- Ao, C.; Yuan, W.; Zhao, J.; He, X.; Zhang, X.; Li, Q.; Lu, C. Superhydrophilic graphene oxide@ electrospun cellulose nanofiber hybrid membrane for high-efficiency oil/water separation. Carbohydr. Polym. 2017, 175, 216–222. [Google Scholar] [CrossRef]
- Bildyukevich, A.V.; Plisko, T.V.; Liubimova, A.S.; Volkov, V.V.; Usosky, V.V. Hydrophilization of polysulfone hollow fiber membranes via addition of polyvinylpyrrolidone to the bore fluid. J. Membr. Sci. 2017, 524, 537–549. [Google Scholar] [CrossRef]
- Anokhina, T.S.; Bazhenov, S.D.; Borisov, I.L.; Vasilevsky, V.P.; Vinokurov, V.A.; Volkov, A.V. Nanocellulose as modifier for hollow fiber ultrafiltration PSF membranes. In Key Engineering Materials; Trans Tech Publications Ltd.: Bäch, Switzerland, 2019; Volume 816, pp. 238–243. [Google Scholar] [CrossRef]
- Trupp, F.; Torasso, N.; Grondona, D.; Rubiolo, G.H.; Goyanes, S. Hierarchical selective membranes combining carbonaceous nanoparticles and commercial permeable substrates for oil/water separation. Sep. Purif. Technol. 2020, 234, 116053. [Google Scholar] [CrossRef]
- Ganwei, Z.; Renbi, B.; Shusu, S.; Xiaoji, Z.; Yongfu, G. Hydrophilic and photo-crosslinkable diblock copolymers employed for robust antifouling membrane coatings. Appl. Surf. Sci. 2019, 464, 429–439. [Google Scholar] [CrossRef]
- Zhu, Y.; Xie, W.; Zhang, F.; Xing, T.; Jin, J. Superhydrophilic in-situ-cross-linked zwitterionic polyelectrolyte/PVDF-blend membrane for highly efficient oil/water emulsion separation. ACS Appl. Mater. Interfaces 2017, 9, 9603–9613. [Google Scholar] [CrossRef]
- Zhang, G.; Yuan, S.; Cao, S.; Yan, G.; Wang, X.; Yang, J.; Van der Bruggen, B. Functionalized poly (arylene ether sulfone) containing hydroxyl units for the fabrication of durable, superhydrophobic oil/water separation membranes. Nanoscale 2019, 11, 7166–7175. [Google Scholar] [CrossRef]
- Serbanescu, O.S.; Voicu, S.I.; Thakur, V.K. Polysulfone functionalized membranes: Properties and challenges. Mater. Today Chem. 2020, 17, 100302. [Google Scholar] [CrossRef]
- Wang, H.H.; Jung, J.T.; Kim, J.F.; Kim, S.; Drioli, E.; Lee, Y.M. A novel green solvent alternative for polymeric membrane preparation via nonsolvent-induced phase separation (NIPS). J. Membr. Sci. 2019, 574, 44–54. [Google Scholar] [CrossRef]
- Kumar, S.; Guria, C.; Mandal, A. Synthesis, characterization and performance studies of polysulfone/bentonite nanoparticles mixed-matrix ultra-filtration membranes using oil field produced water. Sep. Purif. Technol. 2015, 150, 145–158. [Google Scholar] [CrossRef]
- Gohari, R.J.; Halakoo, E.; Lau, W.J.; Kassim, M.A.; Matsuura, T.; Ismail, A.F. Novel polyethersulfone (PES)/hydrous manganese dioxide (HMO) mixed matrix membranes with improved anti-fouling properties for oily wastewater treatment process. RSC Adv. 2014, 4, 17587–17596. [Google Scholar] [CrossRef]
- Nemade, P.R.; Ganjare, A.V.; Ramesh, K.; Rakte, D.M.; Vaishnavi, P.S.V.; Thapa, G. Low fouling sulphonated carbon soot-polysulphone membranes for rapid dehydration of stabilized oil-water emulsions. J. Water Process Eng. 2020, 38, 101590. [Google Scholar] [CrossRef]
- Anderson, G.K.; Saw, C.B.; Le, M.S. Oil/water separation with surface modified membranes. Environ. Technol. 1987, 8, 121–132. [Google Scholar] [CrossRef]
- McCloskey, B.D.; Park, H.B.; Ju, H.; Rowe, B.W.; Miller, D.J.; Freeman, B.D. A bioinspired fouling-resistant surface modification for water purification membranes. J. Membr. Sci. 2012, 413, 82–90. [Google Scholar] [CrossRef]
- Ju, H.; McCloskey, D.B.; Sagle, C.A.; Wu, H.Y.; Kusuma, A.V.; Freeman, D.B. Crosslinked poly (ethylene oxide) fouling resistant coating materials for oil/water separation. J. Membr. Sci. 2008, 307, 260–267. [Google Scholar] [CrossRef]
- Chen, W.; Peng, J.; Su, Y.; Zheng, L.; Wang, L.; Jiang, Z. Separation of oil/water emulsion using Pluronic F127 modified polyethersulfone ultrafiltration membranes. Sep. Purif. Technol. 2009, 66, 591–597. [Google Scholar] [CrossRef]
- Kappert, E.J.; Raaijmakers, M.J.; Tempelman, K.; Cuperus, F.P.; Ogieglo, W.; Benes, N.E. Swelling of 9 polymers commonly employed for solvent-resistant nanofiltration membranes: A comprehensive dataset. J. Membr. Sci. 2019, 569, 177–199. [Google Scholar] [CrossRef] [Green Version]
- Adegbola, T.A.; Agboola, O.; Fayomi, O.S.I. Review of polyacrylonitrile blends and application in manufacturing technology: Recycling and environmental impact. Results Eng. 2020, 7, 100144. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, S.; Yu, B.; Tan, Q.; Zhang, X.; Cong, H. Advanced modified polyacrylonitrile membrane with enhanced adsorption property for heavy metal ions. Sci. Rep. 2018, 8, 1260. [Google Scholar] [CrossRef] [Green Version]
- Onat, B.; Rosales-Solano, H.; Pawliszyn, J. Development of a Biocompatible Solid Phase Microextraction Thin Film Coating for the Sampling and Enrichment of Peptides. Anal. Chem. 2020, 92, 9379–9388. [Google Scholar] [CrossRef]
- Yushkin, A.A.; Balynin, A.V.; Efimov, M.E.; Karpacheva, G.; Volkov, A.V. Preparation of Fine Porous Ultrafiltration Membranes from Polyacrylonitrile. Key Eng. Mater. 2020, 869, 437–442. [Google Scholar] [CrossRef]
- Sun, Z.K.; Zhou, Y.; Jiao, Y.; Cheng, X.Q.; Zhang, Y.; Wang, P.; Shao, L. Multi-hydrophilic functional network enables porous membranes excellent anti-fouling performance for highly efficient water remediation. J. Membr. Sci. 2020, 608, 118191. [Google Scholar] [CrossRef]
- Wang, J.; Yue, Z.; Ince, J.S.; Economy, J. Preparation of nanofiltration membranes from polyacrylonitrile ultrafiltration membranes. J. Membr. Sci. 2006, 286, 333–341. [Google Scholar] [CrossRef]
- Tham, H.M.; Wang, K.Y.; Hua, D.; Japip, S.; Chung, T.S. From ultrafiltration to nanofiltration: Hydrazine cross-linked polyacrylonitrile hollow fiber membranes for organic solvent nanofiltration. J. Membr. Sci. 2017, 542, 289–299. [Google Scholar] [CrossRef]
- Yushkin, A.A.; Efimov, M.N.; Vasilev, A.A.; Karpacheva, G.P.; Volkov, A.V. PAN filtration membranes with extended solvent stability. J. Phys. Conf. Ser. 2018, 1099, 012031. [Google Scholar] [CrossRef]
- Yushkin, A.A.; Efimov, M.N.; Malakhov, A.O.; Karpacheva, G.P.; Bondarenko, G.; Marbelia, L.; Volkov, A.V. Creation of highly stable porous polyacrylonitrile membranes using infrared heating. React. Funct. Polym. 2021, 158, 104793. [Google Scholar] [CrossRef]
- Shami, Z.; Amininasab, S.M.; Katoorani, S.A.; Gharloghi, A.; Delbina, S. NaOH-Induced Fabrication of a Superhydrophilic and Underwater Superoleophobic Styrene-Acrylate Copolymer Filtration Membrane for Effective Separation of Emulsified Light Oil-Polluted Water Mixtures. Langmuir 2021, 37, 12304–12312. [Google Scholar] [CrossRef]
- Melbiah, J.B.; Nithya, D.; Mohan, D. Surface modification of polyacrylonitrile ultrafiltration membranes using amphiphilic Pluronic F127/CaCO3 nanoparticles for oil/water emulsion separation. Colloids Surf. A Physicochem. Eng. Asp. 2017, 516, 147–160. [Google Scholar] [CrossRef]
- Guan, Y.; Huang, J.; Zuo, L.; Xu, J.; Si, L.; Qiu, J.; Li, G. Effect of pluronic P123 and F127 block copolymer on P-glycoprotein transport and CYP3A metabolism. Arch. Pharmacal Res. 2011, 34, 1719–1728. [Google Scholar] [CrossRef]
- Naseeb, N.; Mohammed, A.A.; Laoui, T.; Khan, Z. A novel PAN-GO-SiO2 hybrid membrane for separating oil and water from emulsified mixture. Materials 2019, 12, 212. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Pan, X.; Xue, Q.; He, D.; Zhu, L.; Guo, Q. Antifouling hydrolyzed polyacrylonitrile/graphene oxide membrane with spindle-knotted structure for highly effective separation of oil-water emulsion. J. Membr. Sci. 2017, 532, 38–46. [Google Scholar] [CrossRef]
- Peng, Y.; Guo, F.; Wen, Q.; Yang, F.; Guo, Z. A novel polyacrylonitrile membrane with a high flux for emulsified oil/water separation. Sep. Purif. Technol. 2017, 184, 72–78. [Google Scholar] [CrossRef]
- Zhou, W.; Zhang, Y.; Du, S.; Chen, X.; Qi, K.; Wu, T.; He, J. Superwettable Amidoximed Polyacrylonitrile-Based Nanofibrous Nonwovens for Rapid and Highly Efficient Separation of Oil/Water Emulsions. ACS Appl. Polym. Mater. 2021, 3, 3093–3102. [Google Scholar] [CrossRef]
- Liu, F.; Hashim, N.A.; Liu, Y.; Abed, M.M.; Li, K. Progress in the production and modification of PVDF membranes. J. Membr. Sci. 2011, 375, 1–27. [Google Scholar] [CrossRef]
- Kang, G.D.; Cao, Y.M. Application and modification of poly (vinylidene fluoride) (PVDF) membranes–a review. J. Membr. Sci. 2014, 463, 145–165. [Google Scholar] [CrossRef]
- Boyraz, E.; Yalcinkaya, F.; Hruza, J.; Maryska, J. Surface-modified nanofibrous PVDF membranes for liquid separation technology. Materials 2019, 12, 2702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yalcinkaya, F.; Siekierka, A.; Bryjak, M. Preparation of fouling-resistant nanofibrous composite membranes for separation of oily wastewater. Polymers 2017, 9, 679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, C.; Liu, G.; Qu, Z.; Wang, W.; Yu, D. GO/TiO2-decorated electrospun polyvinylidene fluoride membrane prepared based on metal-polyphenol coordination network for oil–water separation and desalination. J. Mater. Sci. 2022, 57, 3452–3467. [Google Scholar] [CrossRef]
- Nayak, K.; Kumar, A.; Tripathi, B.P. Molecular grafting and zwitterionization based antifouling and underwater superoleophobic PVDF membranes for oil/water separation. J. Membr. Sci. 2022, 643, 120038. [Google Scholar] [CrossRef]
- He, Y.; Xu, K.; Feng, X.; Chen, L.; Jiang, Z. A nonionic polymer-brush-grafted PVDF membrane to analyse fouling during the filtration of oil/water emulsions. J. Membr. Sci. 2021, 637, 119644. [Google Scholar] [CrossRef]
- Nayak, K.; Tripathi, B.P. Molecularly grafted PVDF membranes with in-air superamphiphilicity and underwater superoleophobicity for oil/water separation. Sep. Purif. Technol. 2021, 259, 118068. [Google Scholar] [CrossRef]
- Cheng, B.; Li, Z.; Li, Q.; Ju, J.; Kang, W.; Naebe, M. Development of smart poly (vinylidene fluoride)-graft-poly (acrylic acid) tree-like nanofiber membrane for pH-responsive oil/water separation. J. Membr. Sci. 2017, 534, 1–8. [Google Scholar] [CrossRef]
- Gao, J.; Wang, J.; Xu, Q.; Wu, S.; Chen, Y. Regenerated cellulose strongly adhered by a supramolecular adhesive onto the PVDF membrane for a highly efficient oil/water separation. Green Chem. 2021, 23, 5633–5646. [Google Scholar] [CrossRef]
- Zhang, N.; Yang, N.; Zhang, L.; Jiang, B.; Sun, Y.; Ma, J.; Peng, F. Facile hydrophilic modification of PVDF membrane with Ag/EGCG decorated micro/nanostructural surface for efficient oil-in-water emulsion separation. Chem. Eng. J. 2020, 402, 126200. [Google Scholar] [CrossRef]
- Li, R.; Li, J.; Rao, L.; Lin, H.; Shen, L.; Xu, Y.; Liao, B.Q. Inkjet printing of dopamine followed by UV light irradiation to modify mussel-inspired PVDF membrane for efficient oil-water separation. J. Membr. Sci. 2021, 619, 118790. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, J.; Wang, Q.; Zhang, H.; Wang, Z.; Wu, Z. Evaluating of the performance of natural mineral vermiculite modified PVDF membrane for oil/water separation by membrane fouling model and XDLVO theory. J. Membr. Sci. 2022, 641, 119886. [Google Scholar] [CrossRef]
- Deng, W.; Fan, T.; Li, Y. In situ biomineralization-constructed superhydrophilic and underwater superoleophobic PVDF-TiO2 membranes for superior antifouling separation of oil-in-water emulsions. J. Membr. Sci. 2021, 622, 119030. [Google Scholar] [CrossRef]
- Zhang, X.; Wei, C.; Ma, S.; Zhang, C.; Li, Y.; Chen, D.; Huang, X. Janus poly (vinylidene fluoride)-graft-(TiO2 nanoparticles and PFDS) membranes with loose architecture and asymmetric wettability for efficient switchable separation of surfactant-stabilized oil/water emulsions. J. Membr. Sci. 2021, 640, 119837. [Google Scholar] [CrossRef]
- Alammar, A.; Park, S.H.; Williams, C.J.; Derby, B.; Szekely, G. Oil-in-water separation with graphene-based nanocomposite membranes for produced water treatment. J. Membr. Sci. 2020, 603, 118007. [Google Scholar] [CrossRef]
- Kim, K.Y.; Srivastava, R.P.; Khang, D.Y. Oleophilic to oleophobic wettability switching of isoporous through-hole membranes by surface structure control for low-voltage electrowetting-based oil-water separation. J. Membr. Sci. 2022, 646, 120281. [Google Scholar] [CrossRef]
- Li, R.; Rao, L.; Zhang, J.; Shen, L.; Xu, Y.; You, X.; Lin, H. Novel in-situ electroflotation driven by hydrogen evolution reaction (HER) with polypyrrole (PPy)-Ni-modified fabric membrane for efficient oil/water separation. J. Membr. Sci. 2021, 635, 119502. [Google Scholar] [CrossRef]
- Kozlov, S.V. Ultrafiltration of aqueous solutions of Bromocresol Purple in the presence of dispersed carbon. Russ. J. Appl. Chem. 2003, 76, 1104–1106. [Google Scholar] [CrossRef]
- Galdino, C.J.S., Jr.; Maia, A.D.; Meira, H.M.; Souza, T.C.; Amorim, J.D.; Almeida, F.C.; Sarubbo, L.A. Use of a bacterial cellulose filter for the removal of oil from wastewater. Process Biochem. 2020, 91, 288–296. [Google Scholar] [CrossRef]
- Wandera, D.; Wickramasinghe, S.R.; Husson, S.M. Modification and characterization of ultrafiltration membranes for treatment of produced water. J. Membr. Sci. 2011, 373, 178–188. [Google Scholar] [CrossRef]
- Zhang, M.; Yang, Q.; Gao, M.; Zhou, N.; Shi, J.; Jiang, W. Fabrication of Janus cellulose nanocomposite membrane for various water/oil separation and selective one-way transmission. J. Environ. Chem. Eng. 2021, 9, 106016. [Google Scholar] [CrossRef]
- Yin, X.; He, Y.; Wang, Y.; Yu, H.; Chen, J.; Gao, Y. Bio-inspired antifouling Cellulose nanofiber multifunctional filtration membrane for highly efficient emulsion separation and application in water purification. Korean J. Chem. Eng. 2020, 37, 1751–1760. [Google Scholar] [CrossRef]
- Lu, W.; Duan, C.; Zhang, Y.; Gao, K.; Dai, L.; Shen, M.; Ni, Y. Cellulose-based electrospun nanofiber membrane with core-sheath structure and robust photocatalytic activity for simultaneous and efficient oil emulsions separation, dye degradation and Cr (VI) reduction. Carbohydr. Polym. 2021, 258, 117676. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Shi, Y.; Wang, R.; Chen, K.; Zhou, N.; Yang, Q.; Shi, J. Triple-functional lignocellulose/chitosan/Ag@ TiO2 nanocomposite membrane for simultaneous sterilization, oil/water emulsion separation, and organic pollutant removal. J. Environ. Chem. Eng. 2021, 9, 106728. [Google Scholar] [CrossRef]
- Baggio, A.; Doan, H.N.; Vo, P.P.; Kinashi, K.; Sakai, W.; Tsutsumi, N.; Sangermano, M. Chitosan-Functionalized Recycled Polyethylene Terephthalate Nanofibrous Membrane for Sustainable On-Demand Oil-Water Separation. Glob. Chall. 2021, 5, 2000107. [Google Scholar] [CrossRef] [PubMed]
- Doan, H.N.; Vo, P.P.; Baggio, A.; Negoro, M.; Kinashi, K.; Fuse, Y.; Tsutsumi, N. Environmentally Friendly Chitosan-Modified Polycaprolactone Nanofiber/Nanonet Membrane for Controllable Oil/Water Separation. ACS Appl. Polym. Mater. 2021, 3, 3891–3901. [Google Scholar] [CrossRef]
- Mehranbod, N.; Khorram, M.; Azizi, S.; Khakinezhad, N. Modification and superhydrophilization of electrospun polyvinylidene fluoride membrane using graphene oxide-chitosan nanostructure and performance evaluation in oil/water separation. J. Environ. Chem. Eng. 2021, 9, 106245. [Google Scholar] [CrossRef]
- McCloskey, B.D.; Ju, H.; Freeman, B.D. Composite membranes based on a selective chitosan− poly (ethylene glycol) hybrid layer: Synthesis, characterization, and performance in oil− water purification. Ind. Eng. Chem. Res. 2010, 49, 366–373. [Google Scholar] [CrossRef]
- Li, F.; Gao, R.; Wu, T.; Li, Y. Role of layered materials in emulsified oil/water separation and anti-fouling performance of modified cellulose acetate membranes with hierarchical structure. J. Membr. Sci. 2017, 543, 163–171. [Google Scholar] [CrossRef]
- Kim, D.; Livazovic, S.; Falca, G.; Nunes, S.P. Oil–water separation using membranes manufactured from cellulose/ionic liquid solutions. ACS Sustain. Chem. Eng. 2018, 7, 5649–5659. [Google Scholar] [CrossRef] [Green Version]
- Sai, H.; Jin, Z.; Wang, Y.; Fu, R.; Wang, Y.; Ma, L. Facile and green route to fabricate bacterial cellulose membrane with superwettability for oil–water separation. Adv. Sustain. Syst. 2020, 4, 2000042. [Google Scholar] [CrossRef]
- Rohrbach, K.; Li, Y.; Zhu, H.; Liu, Z.; Dai, J.; Andreasen, J.; Hu, L. A cellulose based hydrophilic, oleophobic hydrated filter for water/oil separation. Chem. Commun. 2014, 50, 13296–13299. [Google Scholar] [CrossRef]
- Hong, S.K.; Bae, S.; Jeon, H.; Kim, M.; Cho, S.J.; Lim, G. An underwater superoleophobic nanofibrous cellulosic membrane for oil/water separation with high separation flux and high chemical stability. Nanoscale 2018, 10, 3037–3045. [Google Scholar] [CrossRef] [Green Version]
- Cheng, M.; He, H.; Zhu, H.; Guo, W.; Chen, W.; Xue, F.; Wang, S. Preparation and properties of pH-responsive reversible-wettability biomass cellulose-based material for controllable oil/water separation. Carbohydr. Polym. 2019, 203, 246–255. [Google Scholar] [CrossRef]
- Liu, R.; Chen, Q.; Cao, M.; Lin, J.; Lin, F.; Ye, W.; Zhao, S. Robust bio-inspired superhydrophilic and underwater superoleophobic membranes for simultaneously fast water and oil recovery. J. Membr. Sci. 2021, 623, 119041. [Google Scholar] [CrossRef]
- Kaur, H.; Bulasara, V.K.; Gupta, R.K. Influence of pH and temperature of dip-coating solution on the properties of cellulose acetate-ceramic composite membrane for ultrafiltration. Carbohydr. Polym. 2018, 195, 613–621. [Google Scholar] [CrossRef]
- Faibish, R.S.; Cohen, Y. Fouling-resistant ceramic-supported polymer membranes for ultrafiltration of oil-in-water microemulsions. J. Membr. Sci. 2001, 185, 129–143. [Google Scholar] [CrossRef]
- Yan, X.; Xiao, X.; Au, C.; Mathur, S.; Huang, L.; Wang, Y.; Chen, J. Electrospinning nanofibers and nanomembranes for oil/water separation. J. Mater. Chem. A 2021, 9, 21659–21684. [Google Scholar] [CrossRef]
- Figoli, A.; Ursino, C.; Sanchez Ramirez, D.O.; Carletto, R.A.; Tonetti, C.; Varesano, A.; Vineis, C. Fabrication of electrospun keratin nanofiber membranes for air and water treatment. Polym. Eng. Sci. 2019, 59, 1472–1478. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, J.; Zhang, F.; Gao, S.; Wang, A.; Fang, W.; Jin, J. Zwitterionic nanohydrogel grafted PVDF membranes with comprehensive antifouling property and superior cycle stability for oil-in-water emulsion separation. Adv. Funct. Mater. 2018, 28, 1804121. [Google Scholar] [CrossRef]
- Gangemi, C.M.A.; Iudici, M.; Spitaleri, L.; Randazzo, R.; Gaeta, M.; D’Urso, A.; Fragalà, M.E. Polyethersulfone mats functionalized with porphyrin for removal of para-nitroaniline from aqueous solution. Molecules 2019, 24, 3344. [Google Scholar] [CrossRef] [Green Version]
- Ognibene, G.; Gangemi, C.; Spitaleri, L.; Gulino, A.; Purrello, R.; Cicala, G.; Fragalà, M.E. Role of the surface composition of the polyethersulfone–TiiP–H2T4 fibers on lead removal: From electrostatic to coordinative binding. J. Mater. Sci. 2019, 54, 8023–8033. [Google Scholar] [CrossRef]
- Qing, W.; Li, X.; Wu, Y.; Shao, S.; Guo, H.; Yao, Z.; Tang, C.Y. In situ silica growth for superhydrophilic-underwater superoleophobic Silica/PVA nanofibrous membrane for gravity-driven oil-in-water emulsion separation. J. Membr. Sci. 2020, 612, 118476. [Google Scholar] [CrossRef]
- Hejase, C.A.; Tarabara, V.V. Nanofiltration of saline oil-water emulsions: Combined and individual effects of salt concentration polarization and fouling by oil. J. Membr. Sci. 2021, 617, 118607. [Google Scholar] [CrossRef]
- Washington, C. Stability of lipid emulsions for drug delivery. Adv. Drug Deliv. Rev. 1996, 20, 131–145. [Google Scholar] [CrossRef]
- Chen, G.; Tao, D. An experimental study of stability of oil–water emulsion. Fuel Processing Technol. 2005, 86, 499–508. [Google Scholar] [CrossRef]
- Chansiri, G.; Lyons, R.T.; Patel, M.V.; Hem, S.L. Effect of surface charge on the stability of oil/water emulsions during steam sterilization. J. Pharm. Sci. 1999, 88, 454–458. [Google Scholar] [CrossRef]
- Chen, C.M.; Lu, C.H.; Chang, C.H.; Yang, Y.M.; Maa, J.R. Influence of pH on the stability of oil-in-water emulsions stabilized by a splittable surfactant. Colloids Surf. A Physicochem. Eng. Asp. 2000, 170, 173–179. [Google Scholar] [CrossRef]

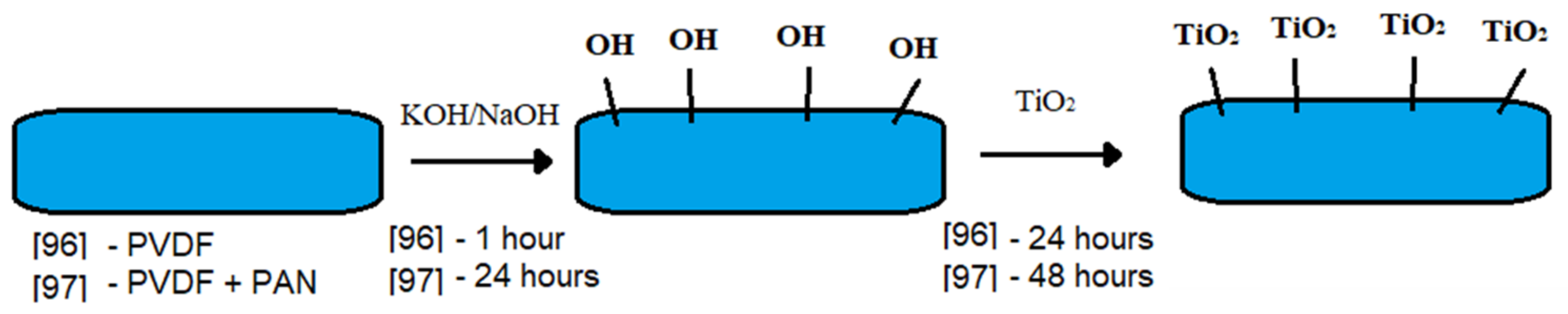
| Membrane | The Composition of the Casting Solution | The Composition of the Membranes % wt. | Average Pore Size, µm | PH2O | Pem | R | |||
|---|---|---|---|---|---|---|---|---|---|
| PSf | PVP | NMP | PSf | PVP | |||||
| M1 | 10 | 5 | 85 | 66.67 | 33.33 | 0.3 | 90 | 14–27 | 79 |
| M2 | 12 | 5 | 83 | 70.58 | 29.42 | 0.25 | 79 | 16–24 | 83 |
| M3 | 15 | 5 | 80 | 75.00 | 25.00 | 0.17 | 72 | 18–20 | 98 |
| Membrane Type | Equilibrium Water Content % wt. | Pure Water Flow, L ∙ m−2 ∙ h−1 bar−1 P = 300 kPa | Water Flow during Emulsion Cleaning m3 ∙ Pa−1 ∙ s−1 ∙ m−1 |
|---|---|---|---|
| PSf | 20.8 | 112 | 0.97 |
| PSf-Si 8% | 60.4 | 275 | 2.42 |
| PSf-Si 10% | 54.1 | 291 | 2.64 |
| PSf-bent. 8% | 62.2 | 612 | 5.25 |
| PSf-bent. 10% | 51.0 | 439 | 3.83 |
| Membrane Material | Oil | Co-p | SF | CSurf | Pex | R | P | No | FRR | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| PSf + PVP | Crude oil | 100–400 mg·L−1 | - | - | 1–4 | 79–98 | 10–70 | 1 | - | [47] |
| PES + PF 127 | Soybean oil | 900 mg·L−1 | SDS | 100 mg·L−1 | 1.5 | ≈100 | 43–83 | 1 | 78–90 | [76] |
| PSf + bentonite | Crude oil | 100–400 mg·L−1 | - | - | 3 | 90–95 | 112–612 | 1 | - | [70] |
| PES + SiO2 | Oil | 2000 mg·L−1 | - | - | 3 | >98 | 89–291 | 1 | - | [57] |
| PES + Mg(OH)2 | Crude oil | 100–1000 ppm | - | - | 1 | ≈100 | 60–573 | 1 | 40–75 | [71] |
| PSf + candle soot | Diesel | 2% wt. | - | - | 1 | ≈100 | 314 | 6 | 92 | [72] |
| PSf/dopamine | Soybean oil | 135 mg·L−1 | - | - | 10.2 | 98 | 65 | 1 | 44–99 | [74] |
| PSf/dopamine + PEG | 98 | 70 | ||||||||
| PSf/crosslinked polyethylene glycol diacrylate | Soybean/canola oil | 1500 ppm | DS 193 | 150 ppm | 10 | 98 | 6 | 1 | - | [75] |
| Membrane Material | Oil | Co-p | SF | CSurf. | Pex | R | P | No | FRR | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| PAN + CA | Vacuum oil | 300–1800 mg·L−1 | SDS | 30–180 mg·L−1 | 1.5 | ≈100 | 67 | 3 | 80–90 | [49] |
| PAN + styrene | Toluene | 1.0% vol | - | - | 0 | - | 2100 | 1 | - | [87] |
| Hexane | 1900 | |||||||||
| Petroleum ether | 1100 | |||||||||
| Toluene | SDS | 100 mg·L−1 | 1750 | |||||||
| Hexane | 1300 | |||||||||
| PAN + PF 127 + CaCO3 | Motor oil | 0.1% vol. | Tween 80 | 1000 mg·L−1 | - | 98 | 343 | 2 | 98 | [88] |
| PAN, Modified hydroxylamine | Diesel fuel | 1% vol. | SDS | 100 mg·L−1 | 0.1 | 96–98 | 3806 | 10 | 100 | [92] |
| Petroleum ether | 3000 | |||||||||
| Isooctane | 2100 | |||||||||
| Trichloromethane | 2200 | |||||||||
| PAN, Alkali Hydrolyzed | Hexane, petroleum ether, i-octane, hexadecane | 10% vol | Tween 80 | 0.1% wt. | 1 | 99 | 2000–2270 | 10 | 95 | [50] |
| Membrane Material | Oil | Co-p | SF | CSurf | Pex | R | P | No | FRR | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| PVFD + dofamine | Diesel | 10% vol. | SDS | 200 mg·L−1 | 0.4 | >99 | 243 | 3 | 82 | [105] |
| Gasoline | 331 | 90 | ||||||||
| Hexane | 558 | 87 | ||||||||
| PVDF + vermiculite NP | Compressor cleaning fluids | 79.5 mg·L−1 | - | - | 0.7–1.5 | 90.0 | ≈100 | 1 | - | [106] |
| 275.7 mg·L−1 | 93.4 | ≈50 | ||||||||
| PVDF + Cellulose | Lubricating oil | 0.2% wt. | CTAB | 0.002% wt. | 0.9 | >99 | 305 | 30 | 80–99 | [103] |
| Diesel oil | 310 | |||||||||
| Sunflower oil | 318 | |||||||||
| Pump oil | 313 | |||||||||
| PVDF + plant-derived epigallocatechin gallate (EGCG) and silver ion (Ag+) | Diesel fuel | 0.1% wt. | SDS | 0.01% wt. | 0.5 | 97 | 735 | 5 | ≈100 | [104] |
| Kerosene | 98 | 561 | ||||||||
| Soybean oil | 98 | 304 | ||||||||
| PVDF + grafted short chain alkylamines | Soybean oil | 1 mg·L−1 | SDS | 0.2 mg·L−1 | 0.5–1.5 | 60–98 | 3–100 | 10 | >96 | [99] |
| Silicone oil | 70–98 | 10–190 | ||||||||
| PVDF + grafted poly(N-acryloylmorpholine | Oil | 100 mg·L−1 | SDBS | 1 mg·L−1 | 1 | 91–99 | 50–500 | 5 | 25–30 | [100] |
| Tween-80 | 100–500 | 50–60 | ||||||||
| CTAB | 400–600 | ≈50 | ||||||||
| PVDF + grafted aminosilanes | Toluene, silicone oil, soybean oil | 10% wt. | - | - | 1–3 | 99 | 7–12 | 1 | - | [101] |
| Defluorinated PVDF + TiO2 | Oil | 50% vol. | Triton X-100 | 1% wt. | 0 | ≈100 | 4–25 | 3 | 60–99 | [96] |
| PVDF + PEI + TiO2 | Hexadecane | 7650 mg·L−1 | SDS | 50 mg·L−1 | 0.1 bar | ≈100 | 200–300 | 5 | ≈100 | [107] |
| PVDF + g-TiO2 + PFDS | Water-in-chloroform | 0.9% wt. | Span-80 | 0.4% wt. | 1 | >99 | 2421 | 5 | ≈100 | [108] |
| Water-in-toluene | 908 | |||||||||
| Water-in-rapeseed oil | 118 | |||||||||
| Water-in-soybean oil | 57 | |||||||||
| Dodecane in water | 0.15% wt | SDS | 0.02% wt. | 2542 | ||||||
| N-hexadecane in water | 398 | |||||||||
| Soybean oil in water | 314 | |||||||||
| Rapeseed oil in water | 443 |
| Polymer | Approximate Cost, $/kg | Manufacturer (Country) |
|---|---|---|
| PAN | 2 | Haihang industry Co (Jinan City, China) |
| PSf | 15 | Molan (dongguang) Plastic Technology (Guangdong, China) |
| PES | 16 | Molan (dongguang) Plastic Technology (Guangdong, China) |
| PVDF | 36 | AGRU KunststofftechnikGmbH (Bad Hall, Austria) |
| PBI | 2 | PBI Performance Products, Inc. (Charlotte, US) |
| PTFE | 17 | Hebei Luozheng Technology Co (Hebei, China) |
| PANI | 10 | Shandong Ningda Chemical Co (Shandong, China) |
| PUA | 15 | ANWIN TECHNOLOGY CO (Tapiei, Taiwan) |
| PPy | 2 | Dawn Groupe Co (Shandong, China) |
| Membrane Material | Oil | Co-p | SF | CSurf. | Pex | R | P | No | FRR | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Cellulose | Crude oil | 200–1000 ppm | SDBS | 2–10 ppm | 5 | 99 | 6–13 | 1 | 60–90 | [124] |
| CTAB | 1–20 | |||||||||
| Tween-80 | 1–2 | |||||||||
| Cellulose on stainless-steel mesh | Octane, hexane, dodecane, toluene | - | - | - | 0 | >99 | 15,200–46,500 | 10 | - | [125] |
| Cellulose | Oil | 10–230 ppm | Tween 20 | 0.5% wt. | 1 | ≈100 | 173 | 20 | - | [113] |
| Lignocellulosic biomass + dofamine | Oil | - | - | - | 30 | 68–97 | 6–28 | 1 | - | [126] |
| Cellulose paper + nanofibrillar cellulose hydrogel | Hexane | 50% vol. | SDS | 2500 mg·L−1 | 0 | 99 | 90 | 1 | - | [127] |
| Cellulose + grafted acrylic acid + acrylamide | Hexane | - | - | - | 0 | 98 | - | 1 | - | [128] |
| Cellulose acetate + dopamine + cellulose nanofibers | Diesel fuel, cyclohexane, tolulu, petroleum | 1% vol. | SDS | 100 mg·L−1 | 0.9 | 99 | 149–4525 | 5 | 90 | [116] |
| Cellulose + PVDF + SiO2 nanocomposite | 1,2-dichloroethane | 3.21% vol. | Tween 80 | 0.45% vol. | 0 | 95 | - | 10 | - | [115] |
| Toluene | 3.21% vol. | 0.45% vol. | 90 | |||||||
| Hexane | 3.21% vol. | 0.45% vol. | 92 | |||||||
| Chloroform | 1.64% vol. | 0.2% vol. | 90 | |||||||
| Lignocellulose + chitosan + Ag + TiO2 | Chloroform | 1.6% vol. | - | - | 0 | 98 | 765 | 7 | - | [118] |
| Cellulose acetate + GO + layered double hydroxides | Decane, Marcol 52 white oil | 1000 mg·L−1 | - | - | 1 | 98 | 200–1300 | 6 | 50–90 | [123] |
| Membrane Material | Oil | Co-p | SF | CSurf | Pex | R | P | No | FRR | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| PAN, Alkali Hydrolyzed + Graphene Oxide | Lubricating oil | 0.1% wt. | - | - | 0 | 99 | 1500–3500 | 4 | 99 | [91] |
| PAN+ amidoximated SiO2 | Chloroform | 1% vol. | SDS | 100 mg·L−1 | 0 | 98 | 1250 | 1 | 93 | [93] |
| Dichloroethane | 99 | 1500 | ||||||||
| n-hexane | 96 | 2750 | ||||||||
| Diesel | 97 | 2200 | ||||||||
| Nanofibers from deacetylated cellulose acetate + PVP + Fe compounds | Petroleum ether | 1% vol | - | - | 0 | 99 | 15,000 | 5 | - | [117] |
| Cyclohexane | 12,000 | |||||||||
| Toluene | 10,000 | |||||||||
| Silicone oil | 12,500 | |||||||||
| Colza oil | 7500 | |||||||||
| Petroleum ether | 1% vol | Tween 80 | 100 mg·L−1 | 0 | 99 | 3500 | 5 | - | ||
| Cyclohexane | 3000 | |||||||||
| Toluene | 3300 | |||||||||
| Silicone oil | 3500 | |||||||||
| Colza oil | 2800 | |||||||||
| PAN+ graphene oxide + silicon oxide | Lubricating oil | 10% wt. | SDS | 2000 ppm | 0 | 98–100 | 2600–3151 | 5 | 94 | [90] |
| PVDF + PAN, treated NaOH + TiO2 | Oil | 50% vol. | - | - | 0 | 73–100 | 10,000–30,000 | 10 | ≈100 | [97] |
| PVDF +PVP + TiO2 + graphene oxide | Hexadecane | 1% wt. | SDS | 0.015 wt. | 0 | 98 | 243 | 1 | - | [98] |
| PVDF + polyacrylic acid | Hexane | 50% vol. | - | - | 0 | 99 | 9600 | 5 | - | [102] |
| Cellulose nanofibers | Hexane, petroleum ether, kerosene | 50% vol. | - | - | 0 | >99 | 120,000 | 10 | - | [129] |
| Cellulose + graphene oxide | Hexane, toluene | 50% vol | - | - | 0 | ≈100 | 960 | 10 | 98 | [61] |
| PVS + SiO2 | Kerosene | 2% vol. | Tween 80 | 2000 mg·L−1 | 0 | 96 | 1450 | 5 | 99 | [137] |
| Surfactant Type | Size of Emulsified Oil Particles, nm | |
|---|---|---|
| Coil = 200 ppm, Surf/oil = 1:100 [125] | Coil = 100 ppm, Surf/oil = 1:10 [100] | |
| Anionic (SDBS) | 700–2500 | 260–300 |
| Cationic (CTAB) | 200–300 | 350–390 |
| Non-ionic (Tween 80) | 200–230 | 210–245 |
| Variable Parameter | Possible Options |
|---|---|
| Composition of emulsions | |
| Type of oil product | Crude oil, diesel, kerosene, soybean oil, canola oil, vacuum oil, pump oil, sunflower oil, lubricatting oil, petroleum ether, toluene, hexane, hexadecane, motor oil, isooctane, chloroform, dichliroethane, gasoline |
| Oil product concentration | 10–100,000 ppm |
| The presence of surfactants | SDS, SDBS, CTAB, Tween-80, Span 80, Trinon X100 |
| Surfactant concentration | 20–10,000 ppm |
| Presence of inorganic salts | NaCl, CaCl2, MgCl2, Na2SO4, NaHCO3 |
| Salt concentration | 1–20 g·L−1 |
| pH | 1–14 |
| Emulsified particle size | 200–1,000,000 nm |
| Emulsion preparation parameters | |
| Mixing intensity | 350–15,000 rpm |
| Mixing time | 5–360 min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dmitrieva, E.S.; Anokhina, T.S.; Novitsky, E.G.; Volkov, V.V.; Borisov, I.L.; Volkov, A.V. Polymeric Membranes for Oil-Water Separation: A Review. Polymers 2022, 14, 980. https://doi.org/10.3390/polym14050980
Dmitrieva ES, Anokhina TS, Novitsky EG, Volkov VV, Borisov IL, Volkov AV. Polymeric Membranes for Oil-Water Separation: A Review. Polymers. 2022; 14(5):980. https://doi.org/10.3390/polym14050980
Chicago/Turabian StyleDmitrieva, Evgenia S., Tatyana S. Anokhina, Eduard G. Novitsky, Vladimir V. Volkov, Ilya L. Borisov, and Alexey V. Volkov. 2022. "Polymeric Membranes for Oil-Water Separation: A Review" Polymers 14, no. 5: 980. https://doi.org/10.3390/polym14050980
APA StyleDmitrieva, E. S., Anokhina, T. S., Novitsky, E. G., Volkov, V. V., Borisov, I. L., & Volkov, A. V. (2022). Polymeric Membranes for Oil-Water Separation: A Review. Polymers, 14(5), 980. https://doi.org/10.3390/polym14050980