Development and Characterization of Plantain (Musa paradisiaca) Flour-Based Biopolymer Films Reinforced with Plantain Fibers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemical Composition of Plantain Fibers
2.2.1. Ash Content
2.2.2. Lignin Content
2.2.3. Cellulose Content
2.2.4. Extractive Content
2.3. Film and Composite Preparation
2.4. Physical Characterization
2.5. Fourier-Transformed Infrared Spectroscopy (FTIR)
2.6. Thermo-Gravimetric Analysis (TGA)
2.7. Mechanical Characterization
2.8. Scanning Electron Microscopy (SEM)
2.9. Statistical Analysis
3. Results
3.1. Chemical Composition of Plantain Fibers
3.2. Physical Characterization
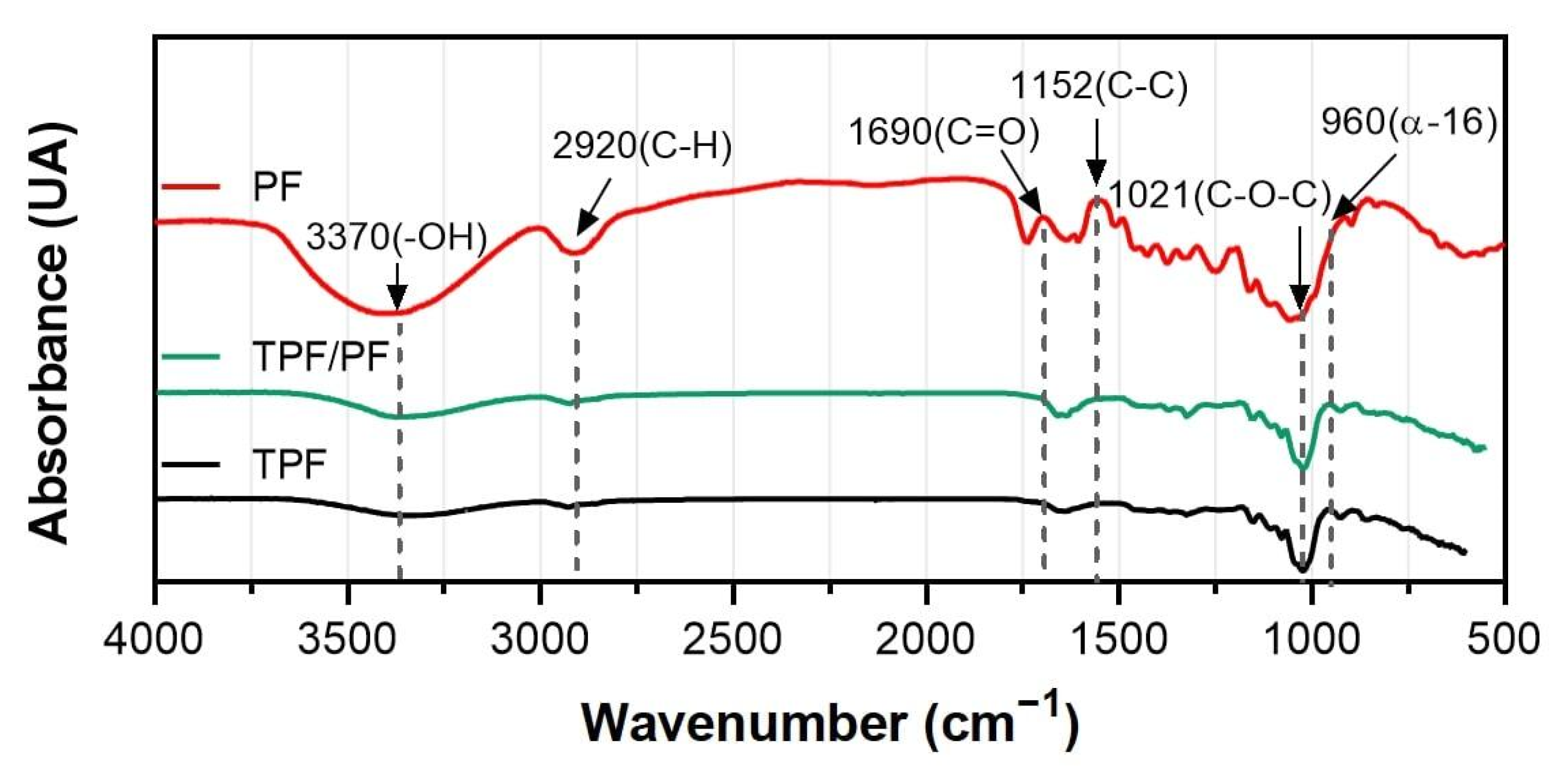
3.3. Fourier-Transformed Infrared Spectroscopy (FTIR)
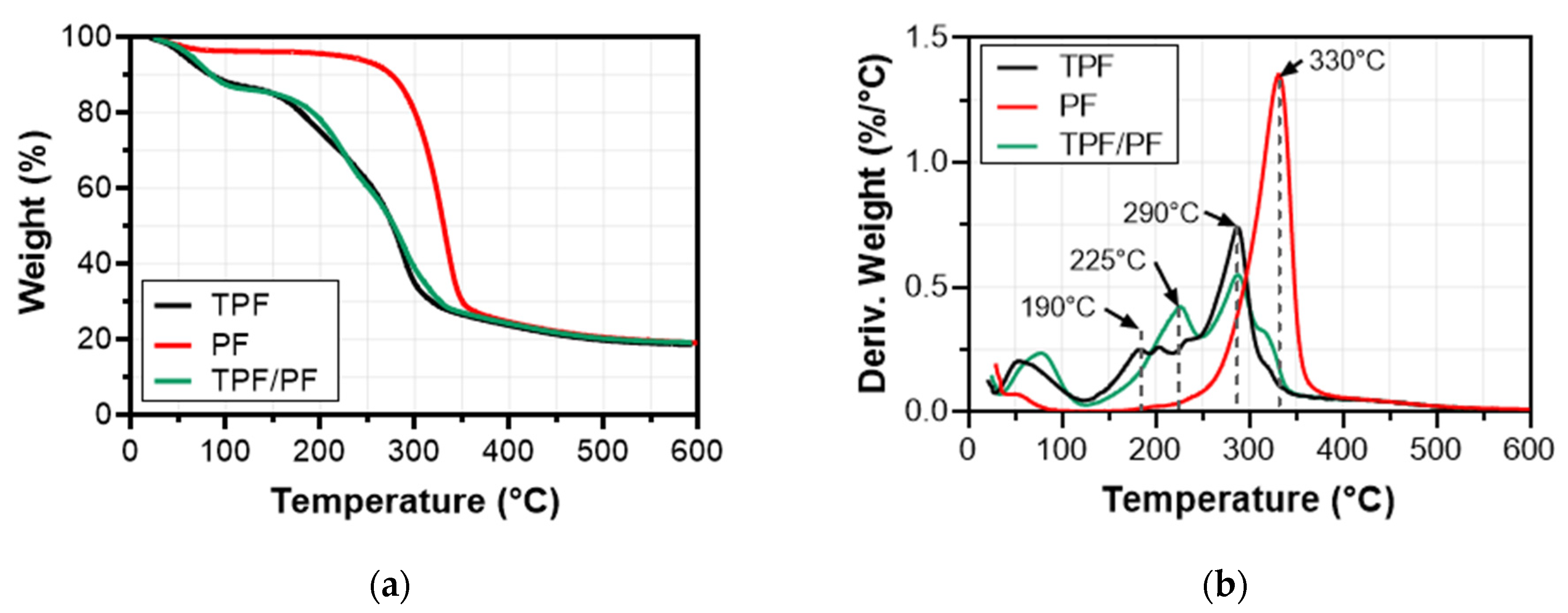
3.4. Thermo-Gravimetric Analysis (TGA)
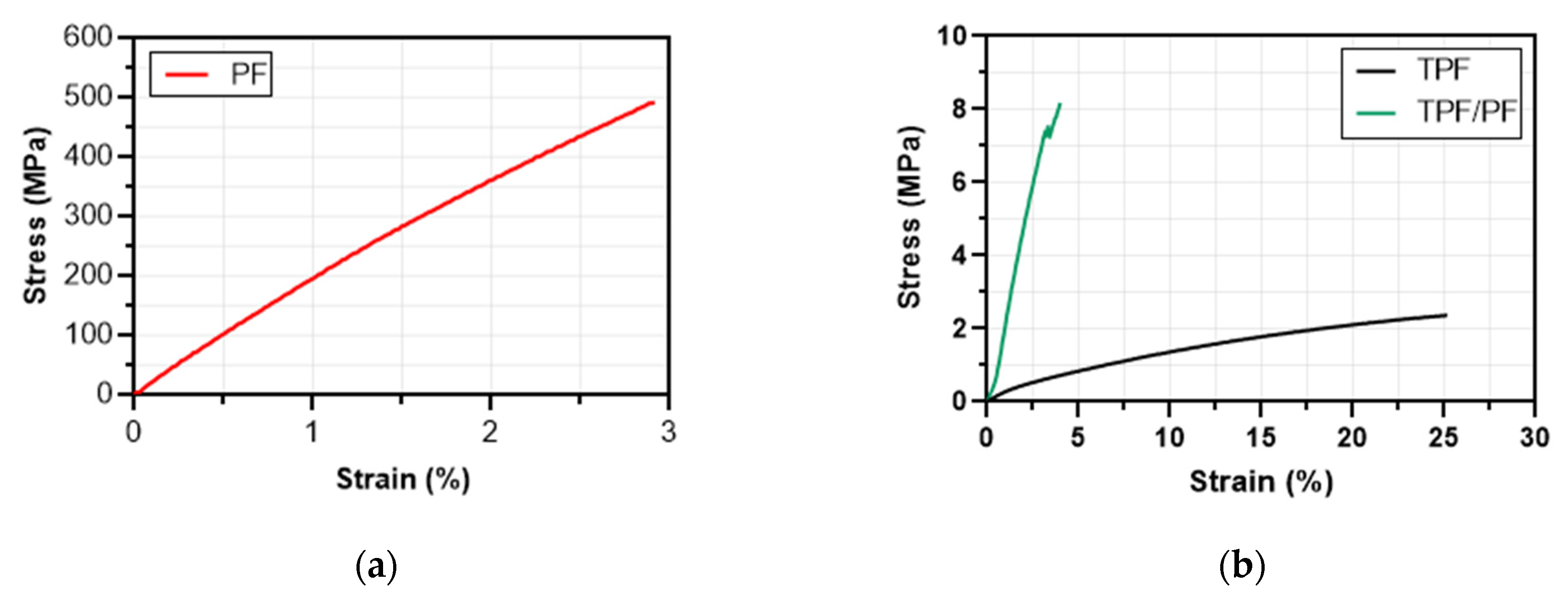
3.5. Mechanical Characterization
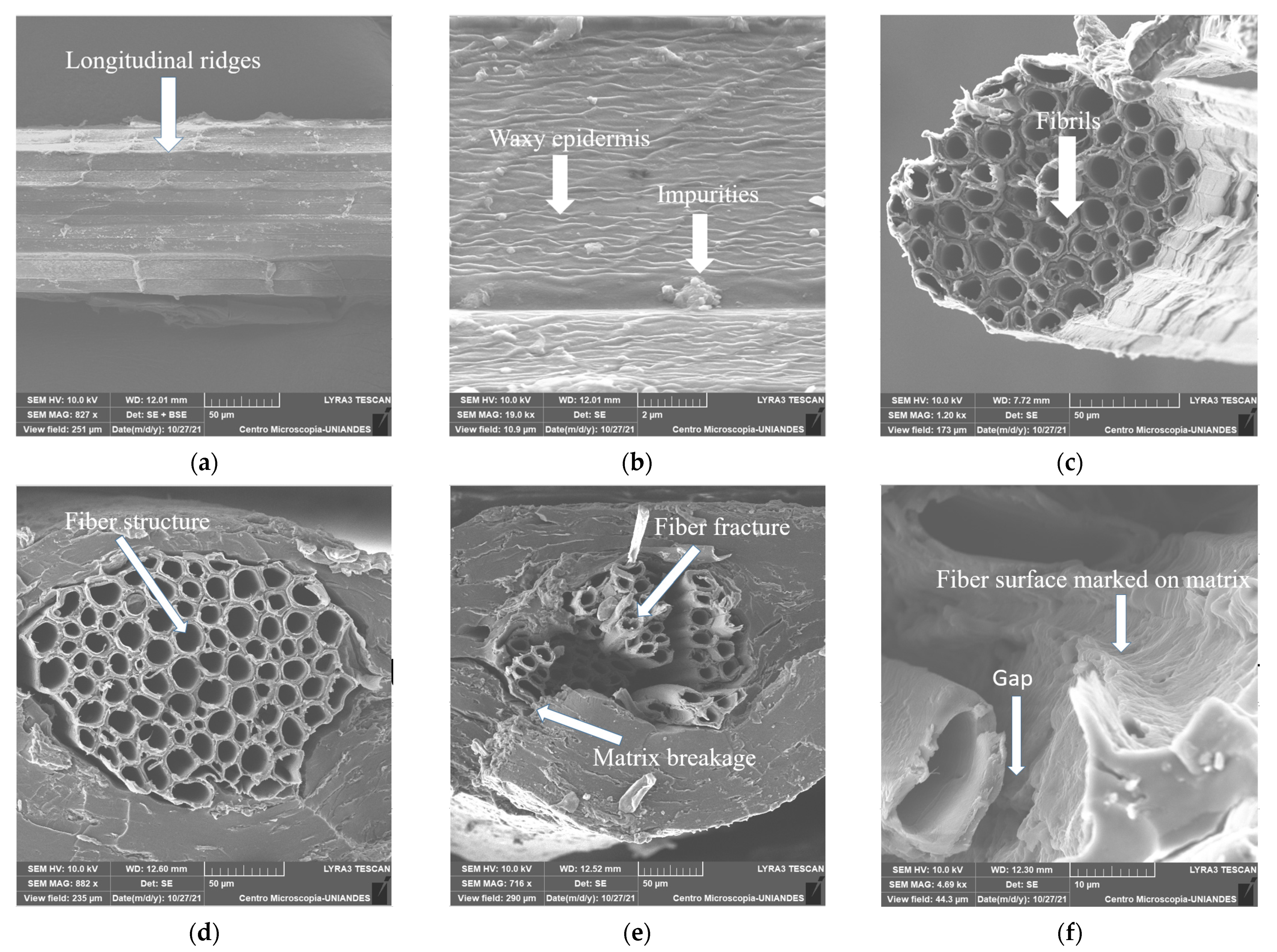
3.6. Scanning Electron Microscopy (SEM)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohanty, A.K.; Misra, M.; Drzal, L.T. Sustainable Bio-Composites from Renewable Resources: Opportunities and Challenges in the Green Materials World. J. Polym. Environ. 2002, 10, 19–26. [Google Scholar] [CrossRef]
- Haraguchi, K. Biocomposites. In Encyclopedia of Polymeric Nanomaterials; Kobayashi, S., Müllen, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 124–130. ISBN 978-3-642-29648-2. [Google Scholar]
- Masutani, K.; Kimura, Y. Biobased Polymers. In Encyclopedia of Polymeric Nanomaterials; Kobayashi, S., Müllen, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 118–124. ISBN 978-3-642-29648-2. [Google Scholar]
- Madsen, B.; Hoffmeyer, P.; Lilholt, H. Hemp Yarn Reinforced Composites—II. Tensile Properties. Compos. Part A Appl. Sci. Manuf. 2007, 38, 2204–2215. [Google Scholar] [CrossRef]
- Goutianos, S.; Peijs, T.; Nyström, B.; Skrifvars, M. Development of Flax Fibre Based Textile Reinforcements for Composite Applications. Appl. Compos. Mater. 2006, 13, 199–215. [Google Scholar] [CrossRef] [Green Version]
- Oksman, K.; Wallström, L.; Berglund, L.; Toledo Filho, R. Morphology and Mechanical Properties of Unidirectional Sisal—Epoxy Composites. J. Appl. Polym. Sci. 2002, 84, 2358–2365. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Bhat, I.U.H.; Jawaid, M.; Zaidon, A.; Hermawan, D.; Hadi, Y.S. Bamboo Fibre Reinforced Biocomposites: A Review. Mater. Des. 2012, 42, 353–368. [Google Scholar] [CrossRef]
- De Rosa, I.M.; Kenny, J.M.; Puglia, D.; Santulli, C.; Sarasini, F. Morphological, Thermal and Mechanical Characterization of Okra (Abelmoschus esculentus) Fibres as Potential Reinforcement in Polymer Composites. Compos. Sci. Technol. 2010, 70, 116–122. [Google Scholar] [CrossRef]
- Jayaramudu, J.; Guduri, B.R.; Varada Rajulu, A. Characterization of New Natural Cellulosic Fabric Grewia tilifolia. Carbohydr. Polym. 2010, 79, 847–851. [Google Scholar] [CrossRef]
- Jayaramudu, J.; Reddy, S.M.; Varaprasad, K.; Sadiku, R.; Sinha Ray, S.; Rajulu, A.V. Preparation and Properties of Biodegradable Films from Sterculia Urens Short Fiber/Cellulose Green Composites. Carbohydr. Polym. 2013, 93, 622–627. [Google Scholar] [CrossRef]
- Saravanakumar, S.S.; Kumaravel, A.; Nagarajan, T.; Sudhakar, P.; Baskaran, R. Characterization of a Novel Natural Cellulosic Fiber from Prosopis juliflora Bark. Carbohydr. Polym. 2013, 92, 1928–1933. [Google Scholar] [CrossRef]
- Adeniyi, A.; Ighalo, J.O.; Onifade, D. Banana and Plantain Fiber-Reinforced Polymer Composites. J. Polym. Eng. 2019, 39, 597–611. [Google Scholar] [CrossRef]
- Cadena Ch, E.M.; Vélez R, J.M.; Santa, J.F.; Otálvaro, G.V. Natural Fibers from Plantain Pseudostem (Musa paradisiaca) for Use in Fiber-Reinforced Composites. J. Nat. Fibers 2017, 14, 678–690. [Google Scholar] [CrossRef]
- Zarate, L.; Romero, A.; Bengoechea, C.; Partal, P.; Guerrero, A. Thermo-Mechanical and Hydrophilic Properties of Polysaccharide/Gluten-Based Bioplastics. Carbohydr. Polym. 2014, 112, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, T.J.; Guzmán, R.; Medina Jaramillo, C.; Famá, L. Effect of Beet Flour on Films Made from Biological Macromolecules: Native and Modified Plantain Flour. Int. J. Biol. Macromol. 2016, 82, 395–403. [Google Scholar] [CrossRef]
- Pelissari, F.M.; Andrade-Mahecha, M.M.; Sobral, P.J.d.A.; Menegalli, F.C. Optimization of Process Conditions for the Production of Films Based on the Flour from Plantain Bananas (Musa paradisiaca). LWT Food Sci. Technol. 2013, 52, 1–11. [Google Scholar] [CrossRef]
- Żołek-Tryznowska, Z.; Holica, J. Starch Films as an Environmentally Friendly Packaging Material: Printing Performance. J. Clean. Prod. 2020, 276, 124265. [Google Scholar] [CrossRef]
- Diyana, Z.N.; Jumaidin, R.; Selamat, M.Z.; Ghazali, I.; Julmohammad, N.; Huda, N.; Ilyas, R.A. Physical Properties of Thermoplastic Starch Derived from Natural Resources and Its Blends: A Review. Polymers 2021, 13, 1396. [Google Scholar] [CrossRef]
- Pelissari, F.M.; Andrade-Mahecha, M.M.; Sobral, P.J.d.A.; Menegalli, F.C. Comparative Study on the Properties of Flour and Starch Films of Plantain Bananas (Musa paradisiaca). Food Hydrocoll. 2013, 30, 681–690. [Google Scholar] [CrossRef]
- Andrade-Mahecha, M.; Tapia-Blácido, D.R.; Menegalli, F.C. Development and Optimization of Biodegradable Films Based on Achira Flour. Carbohydr. Polym. 2012, 88, 449–458. [Google Scholar] [CrossRef]
- Gañán, P.; Zuluaga, R.; Restrepo Osorio, A.; Labidi, J.; Mondragon, I. Plantain Fibre Bundles Isolated from Colombian Agro-Industrial Residues. Bioresour. Technol. 2008, 99, 486–491. [Google Scholar] [CrossRef]
- TAPPI. Ash in Wood, Pulp, Paper and Paperboard: Combustion at 525 °C (Five-Year Review of T 211 Om-02); TAPPI: Peachtree, GA, USA, 2007. [Google Scholar]
- TAPPI. Acid-Insoluble Lignin in Wood and Pulp (Reaffirmation of T 222 Om-02); TAPPI: Peachtree, GA, USA, 2006. [Google Scholar]
- TAPPI. Alpha-, Beta- and Gamma-Cellulose in Pulp; TAPPI: Peachtree, GA, USA, 1999. [Google Scholar]
- TAPPI. Solvent Extractives of Wood and Pulp (Proposed Revision of T 204 Cm-97); TAPPI: Peachtree, GA, USA, 2007. [Google Scholar]
- ASTM 4442. Standard Test Methods for Direct Moisture Content Measurement of Wood and Wood-Based Materials; ASTM: West Conshohocken, PA, USA, 2020. [Google Scholar]
- ASTM E1131-08. Standard Test Method for Compositional Analysis by Thermogravimetry; ASTM: West Conshohocken, PA, USA, 2014. [Google Scholar]
- ASTM D882-10. Standard Test Method for Tensile Properties of Thin Plastic Sheeting; ASTM: West Conshohocken, PA, USA, 2012. [Google Scholar]
- ASTM C1557. Standard Test Method for Tensile Strength and Young’s Modulus of Fibers; ASTM: West Conshohocken, PA, USA, 2017. [Google Scholar]
- Montgomery, D.C. Design and Analysis of Experiments, 2nd ed.; Wiley: New York, NY, USA, 1984. [Google Scholar]
- Komuraiah, A.; Kumar, N.S.; Prasad, B.D. Chemical Composition of Natural Fibers and Its Influence on Their Mechanical Properties. Mech. Compos. Mater. 2014, 50, 359–376. [Google Scholar] [CrossRef]
- Porras, A.; Maranon, A.; Ashcroft, I.A. Characterization of a Novel Natural Cellulose Fabric from Manicaria Saccifera Palm as Possible Reinforcement of Composite Materials. Compos. B Eng. 2015, 74, 66–73. [Google Scholar] [CrossRef]
- Bogoeva-Gaceva, G.; Avella, M.; Malinconico, M.; Bužarovska, A.; Grozdanov, A.; Gentile, G.; Errico, M. Natural Fiber Eco-Composites. Polym. Compos. 2007, 28, 98–107. [Google Scholar] [CrossRef]
- Williams, G.; Wool, R. Composites from Natural Fibers and Soy Oil Resins. Appl. Compos. Mater. 2000, 7, 421–432. [Google Scholar] [CrossRef]
- Mohanty, A.; Misra, M.; Drzal, L. Surface Modifications of Natural Fibers and Performance of the Resulting Biocomposites: An Overview. Compos. Interfaces 2001, 8, 313–343. [Google Scholar] [CrossRef]
- Chokshi, S.; Parmar, V.; Gohil, P.; Chaudhary, V. Chemical Composition and Mechanical Properties of Natural Fibers. J. Nat. Fibers 2020, 1–12. [Google Scholar] [CrossRef]
- Poletto, M.; Ornaghi, H.L.; Zattera, A.J. Native Cellulose: Structure, Characterization and Thermal Properties. Materials 2014, 7, 6105–6119. [Google Scholar] [CrossRef] [Green Version]
- Faruk, O.; Bledzki, A.K.; Fink, H.-P.; Sain, M. Biocomposites Reinforced with Natural Fibers: 2000–2010. Prog. Polym. Sci. 2012, 37, 1552–1596. [Google Scholar] [CrossRef]
- Bledzki, A.K.; Reihmane, S.; Gassan, J. Properties and Modification Methods for Vegetable Fibers for Natural Fiber Composites. J. Appl. Polym. Sci. 1996, 59, 1329–1336. [Google Scholar] [CrossRef]
- García-Ramón, J.; Carmona-García, R.; Valera-Zaragoza, M.; Aparicio-Saguilán, A.; Bello-Pérez, L.; Aguirre-Cruz, A.; Alvarez-Ramirez, J. Morphological, Barrier, and Mechanical Properties of Banana Starch Films Reinforced with Cellulose Nanoparticles from Plantain Rachis. Int. J. Biol. Macromol. 2021, 187, 35–42. [Google Scholar] [CrossRef]
- Godbillot, L.; Dole, P.; Joly, C.; Rogé, B.; Mathlouthi, M. Analysis of Water Binding in Starch Plasticized Films. Food Chem. 2006, 96, 380–386. [Google Scholar] [CrossRef]
- Bhatia, S.; Angra, S.; Khan, S. Mechanical and Wear Properties of Epoxy Matrix Composite Reinforced with Varying Ratios of Solid Glass Microspheres. J. Phys. Conf. Ser. 2019, 1240, 012080. [Google Scholar] [CrossRef]
- Brydson, J.A. 23—Phenolic Resins. In Plastics Materials (Seventh Edition); Brydson, J.A., Ed.; Butterworth-Heinemann: Oxford, UK, 1999; pp. 635–667. ISBN 978-0-7506-4132-6. [Google Scholar]
- Silverstein, R.; Webster, F.; Kiemle, D. Identificação Espectrofotométrica de Compostos Orgânicos, 7th ed.; LTC: Rio de Janeiro, Brazil, 2007; ISBN 978-85-216-1521-7. [Google Scholar]
- Kizil, R.; Irudayaraj, J.; Seetharaman, K. Characterization of Irradiated Starches by Using FT-Raman and FTIR Spectroscopy. J. Agric. Food Chem. 2002, 50, 3912–3918. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.R.; Vickers, T. Infrared Analysis of Peptides and Proteins. Appl. Spectrosc. 2001, 55, 108. [Google Scholar]
- Van Soest, J.J.G.; Tournois, H.; de Wit, D.; Vliegenthart, J.F.G. Short-Range Structure in (Partially) Crystalline Potato Starch Determined with Attenuated Total Reflectance Fourier-Transform IR Spectroscopy. Carbohydr. Res. 1995, 279, 201–214. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.; Jeng, R.; Sain, M.; Saville, B.; Hubbes, M. Production, Characterization, and Mechanical Properties of Starch Modified by Ophiostoma Spp. BioResources 2006, 1, 257–269. [Google Scholar] [CrossRef]
- Estrada-Monje, A.; Alonso-Romero, S.; Zitzumbo-Guzmán, R.; Estrada-Moreno, I.A.; Zaragoza-Contreras, E.A. Thermoplastic Starch-Based Blends with Improved Thermal and Thermomechanical Properties. Polymers 2021, 13, 4263. [Google Scholar] [CrossRef]
- Jiménez-Regalado, E.J.; Caicedo, C.; Fonseca-García, A.; Rivera-Vallejo, C.C.; Aguirre-Loredo, R.Y. Preparation and Physicochemical Properties of Modified Corn Starch–Chitosan Biodegradable Films. Polymers 2021, 13, 4431. [Google Scholar] [CrossRef]
- Dan-asabe, B.; Yaro, S.A.; Yawas, D.; Aku, S.; Samotu, I.; Abubakar Zaria, U.; Obada, D. Mechanical, Spectroscopic and Micro-Structural Characterization of Banana Particulate Reinforced PVC Composite as Piping Material. Tribol. Ind. 2016, 38, 255–266. [Google Scholar]
- Rodríguez, L.J.; Cardona, C.A.; Orrego, C.E. Water Uptake, Chemical Characterization, and Tensile Behavior of Modified Banana–Plantain Fiber and Their Polyester Composites. Polym. Compos. 2015, 37, 2960–2973. [Google Scholar] [CrossRef]
- Ayala Valencia, G.; Agudelo, A.; Zapata, R. Effect of Glycerol on the Electrical Properties and Phase Behavior of Cassava Starch Biopolymers. DYNA 2012, 79, 138–147. [Google Scholar]
- Nuwamanya, E.; Baguma, Y.; Wembabazi, E.; Rubaihayo, P. A Comparative Study of the Physicochemical Properties of Starches from Root, Tuber and Cereal Crops. Afr. J. Biotechnol. 2011, 10, 12018–12030. [Google Scholar] [CrossRef]
- Marques, P.T.; Lima, A.M.F.; Bianco, G.; Laurindo, J.B.; Borsali, R.; Le Meins, J.-F.; Soldi, V. Thermal Properties and Stability of Cassava Starch Films Cross-Linked with Tetraethylene Glycol Diacrylate. Polym. Degrad. Stab. 2006, 91, 726–732. [Google Scholar] [CrossRef]
- Liu, X.; Yu, L.; Liu, H.; Chen, L.; Li, L. In Situ Thermal Decomposition of Starch with Constant Moisture. Polym. Degrad. Stab. 2008, 93, 260–262. [Google Scholar] [CrossRef]
- Sanyang, M.L.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Sahari, J. Effect of Plasticizer Type and Concentration on Tensile, Thermal and Barrier Properties of Biodegradable Films Based on Sugar Palm (Arenga pinnata) Starch. Polymers 2015, 7, 1106–1124. [Google Scholar] [CrossRef]
- Shi, R.; Zhang, Z.; Liu, Q.; Han, Y.; Zhang, L.; Chen, D.; Tian, W. Characterization of Citric Acid/Glycerol Co-Plasticized Thermoplastic Starch Prepared by Melt Blending. Carbohydr. Polym. 2007, 69, 748–755. [Google Scholar] [CrossRef]
- Jimenez, A.; Betancourt, S.; Gañán, P.; Cruz, L. Degradación Térmica de Fibras Naturales Procedentes de La Calceta de Plátano. (Estudio Cinético). Rev. Latinoam. Metal. Mater. 2009, 29, 215–219. [Google Scholar]
- Kusić, D.; Božič, U.; Monzon, M.; Paz Hernández, R.; Bordón, P. Thermal and Mechanical Characterization of Banana Fiber Reinforced Composites for Its Application in Injection Molding. Materials 2020, 13, 3581. [Google Scholar] [CrossRef]
- Benítez, A.N.; Monzón, M.D.; Angulo, I.; Ortega, Z.; Hernández, P.M.; Marrero, M.D. Treatment of Banana Fiber for Use in the Reinforcement of Polymeric Matrices. Measurement 2013, 46, 1065–1073. [Google Scholar] [CrossRef]
- Song, H.; Choi, I.; Lee, J.-S.; Chung, M.-N.; Yoon, C.S.; Han, J. Comparative Study on Physicochemical Properties of Starch Films Prepared from Five Sweet Potato (Ipomoea batatas) Cultivars. Int. J. Biol. Macromol. 2021, 189, 758–767. [Google Scholar] [CrossRef]
- Alqahtani, N.; Alnemr, T.; Ali, S. Development of Low-Cost Biodegradable Films from Corn Starch and Date Palm Pits (Phoenix dactylifera). Food Biosci. 2021, 42, 101199. [Google Scholar] [CrossRef]
- Hasan, M.; Gopakumar, D.A.; Olaiya, N.G.; Zarlaida, F.; Alfian, A.; Aprinasari, C.; Alfatah, T.; Rizal, S.; Abdul Khalil, H.P.S. Evaluation of the Thermomechanical Properties and Biodegradation of Brown Rice Starch-Based Chitosan Biodegradable Composite Films. Int. J. Biol. Macromol. 2020, 156, 896–905. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Xie, F.; Yu, L.; Liu, H.; Meng, L.; Khalid, S.; Chen, L. Preparation and Characterization of Starch-Based Composite Films Reinfoced by Polysaccharide-Based Crystals. Compos. B Eng. 2018, 133, 122–128. [Google Scholar] [CrossRef]
- Bodirlau, R.; Teaca, C.-A.; Spiridon, I. Influence of Natural Fillers on the Properties of Starch-Based Biocomposite Films. Compos. B Eng. 2013, 44, 575–583. [Google Scholar] [CrossRef]
- Djafari Petroudy, S.R. 3—Physical and Mechanical Properties of Natural Fibers. In Advanced High Strength Natural Fibre Composites in Construction; Fan, M., Fu, F., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 59–83. ISBN 978-0-08-100411-1. [Google Scholar]
- Ochi, S. Development of High Strength Biodegradable Composites Using Manila Hemp Fiber and Starch-Based Biodegradable Resin. Compos. A Appl. Sci. Manuf. 2006, 37, 1879–1883. [Google Scholar] [CrossRef]
- Mahmud, S.; Hasan, K.M.; Jahid, M.; Mohiuddin, K.; Zhang, R.; Zhu, J. Comprehensive Review on Plant Fiber-Reinforced Polymeric Biocomposites. J. Mater. Sci. 2021, 56, 7231–7264. [Google Scholar] [CrossRef]
- Alawar, A.; Hamed, A.M.; Al-Kaabi, K. Characterization of Treated Date Palm Tree Fiber as Composite Reinforcement. Compos. B Eng. 2009, 40, 601–606. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Wibowo, A.; Misra, M.; Drzal, L.T. Effect of Process Engineering on the Performance of Natural Fiber Reinforced Cellulose Acetate Biocomposites. Compos. A Appl. Sci. Manuf. 2004, 35, 363–370. [Google Scholar] [CrossRef]
- Kabir, M.M.; Wang, H.; Lau, K.T.; Cardona, F. Chemical Treatments on Plant-Based Natural Fibre Reinforced Polymer Composites: An Overview. Compos. Part B Eng. 2012, 43, 2883–2892. [Google Scholar] [CrossRef]
- Yan, L.; Yuan, X. Improving the Mechanical Properties of Natural Fibre Fabric Reinforced Epoxy Composites by Alkali Treatment. J. Reinf. Plast. Compos. 2012, 31, 425–437. [Google Scholar] [CrossRef]
- Yan, L. Effect of Alkali Treatment on Vibration Characteristics and Mechanical Properties of Natural Fabric Reinforced Composites. J. Reinf. Plast. Compos. 2012, 31, 887–896. [Google Scholar] [CrossRef]
| Component | Percent (%) |
|---|---|
| Ash | 3.50 ± 0.32 |
| Extractives | 2.28 ± 0.86 |
| Lignin | 13.17 ± 1.25 |
| Cellulose a | 81.05 |
| α-cellulose | 69.09 ± 0.36 |
| β-cellulose | 0.40 ± 0.25 |
| γ-cellulose | 11.40 ± 0.75 |
| Fiber | Lignin (%) | Cellulose (%) |
|---|---|---|
| Jute | 12–13 | 61–71 |
| Hemp | 3 | 68 |
| Sisal | 8–10 | 65–78 |
| Flax | 3 | 71–81 |
| Piassava | 48.4 | 31.6 |
| Bamboo | 21–31 | 26–60 |
| Caraua | 7.5 | 81 |
| Oil palm | 29 | 40–50 |
| Coir | 40–45 | 32–43 |
| Bagasse | 25.3 | 52 |
| Plantain (Present study) | 13.2 | 81.1 |
| Property | TPF | PF | TPF/PF |
|---|---|---|---|
| Moisture (%) | 10.42 ± 0.01 | 8.39 ± 0.72 | 9.90 ± 0.01 |
| Density (g/cm3) | 1.14 ± 0.04 | 0.83 ± 0.05 | 1.10 ± 0.06 |
| Property | TPF | PF | TPF/PF |
|---|---|---|---|
| Tensile strength (MPa) | 2.36 ± 0.21 | 440.30 ± 146.95 | 8.16 ± 1.66 |
| Elongation (%) | 24.53 ± 2.82 | 2.94 ± 0.77 | 4.09 ± 0.53 |
| Young’s modulus (MPa) | 23.62 ± 3.10 | 15,668.81 ± 5534.94 | 281.34 ± 38.58 |
| Specific strength (MPa/g cm3) | 2.07 ± 0.21 | 530.12 ± 146.95 | 10.74 ± 1.66 |
| Specific modulus (MPa/g cm3) | 20.64 ± 2.82 | 18,878.08 ± 5534.94 | 370.18 ± 38.58 |
| Source | Tensile Strength (MPa) | Young’s Modulus (MPa) | Elongation at Break (%) |
|---|---|---|---|
| Corn starch | 1.23 ± 0.07 | 2.31 ± 0.06 | 53.13 ± 0.81 |
| Batata starch | 3.51 ± 0.16 | 25.09 ± 2.66 | 70.74 ± 5.67 |
| Rice starch | 3.70 ± 0.92 | 9.33 ± 0.47 | 39.50 ± 0.92 |
| Banana flour | 1.00 ± 0.10 | 2.70 ± 0.70 | 49 ± 2.00 |
| Banana/beet flour | 1.14 ± 0.03 | 3.3 ± 0.10 | 53 ± 1.00 |
| Chemically modified starch | 12.8 | 1169 | 8.3 |
| Commercial starch | 7.90 ± 0.07 | 73.10 ± 5.78 | 33.40 ± 0.41 |
| TPF (Present study) | 2.36 ± 0.21 | 23.62 ± 3.10 | 24.53 ± 2.82 |
| Fiber | Tensile Strength (MPa) | Young’s Modulus (GPa) | Elongation at Break (%) |
|---|---|---|---|
| Jute | 187–773 | 13–26.5 | 1.5–3.1 |
| Hemp | 580–1110 | 70 | 1.6–4.5 |
| Sisal | 507–885 | 9.4–22 | 1.9–3 |
| Flax | 343–1035 | 27.6 | 1.2–3 |
| Date | 58–203 | 2–7.5 | 5–10 |
| Bagasse | 20–290 | N/A | 1–3 |
| PF (Present study) | 211–599 | 10–21 | 2.2–3.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venegas, R.; Torres, A.; Rueda, A.M.; Morales, M.A.; Arias, M.J.; Porras, A. Development and Characterization of Plantain (Musa paradisiaca) Flour-Based Biopolymer Films Reinforced with Plantain Fibers. Polymers 2022, 14, 748. https://doi.org/10.3390/polym14040748
Venegas R, Torres A, Rueda AM, Morales MA, Arias MJ, Porras A. Development and Characterization of Plantain (Musa paradisiaca) Flour-Based Biopolymer Films Reinforced with Plantain Fibers. Polymers. 2022; 14(4):748. https://doi.org/10.3390/polym14040748
Chicago/Turabian StyleVenegas, Ramiro, Andres Torres, Ana M. Rueda, Maria A. Morales, Mary J. Arias, and Alicia Porras. 2022. "Development and Characterization of Plantain (Musa paradisiaca) Flour-Based Biopolymer Films Reinforced with Plantain Fibers" Polymers 14, no. 4: 748. https://doi.org/10.3390/polym14040748
APA StyleVenegas, R., Torres, A., Rueda, A. M., Morales, M. A., Arias, M. J., & Porras, A. (2022). Development and Characterization of Plantain (Musa paradisiaca) Flour-Based Biopolymer Films Reinforced with Plantain Fibers. Polymers, 14(4), 748. https://doi.org/10.3390/polym14040748






