Applications of the ROS-Responsive Thioketal Linker for the Production of Smart Nanomedicines
Abstract
:1. Introduction
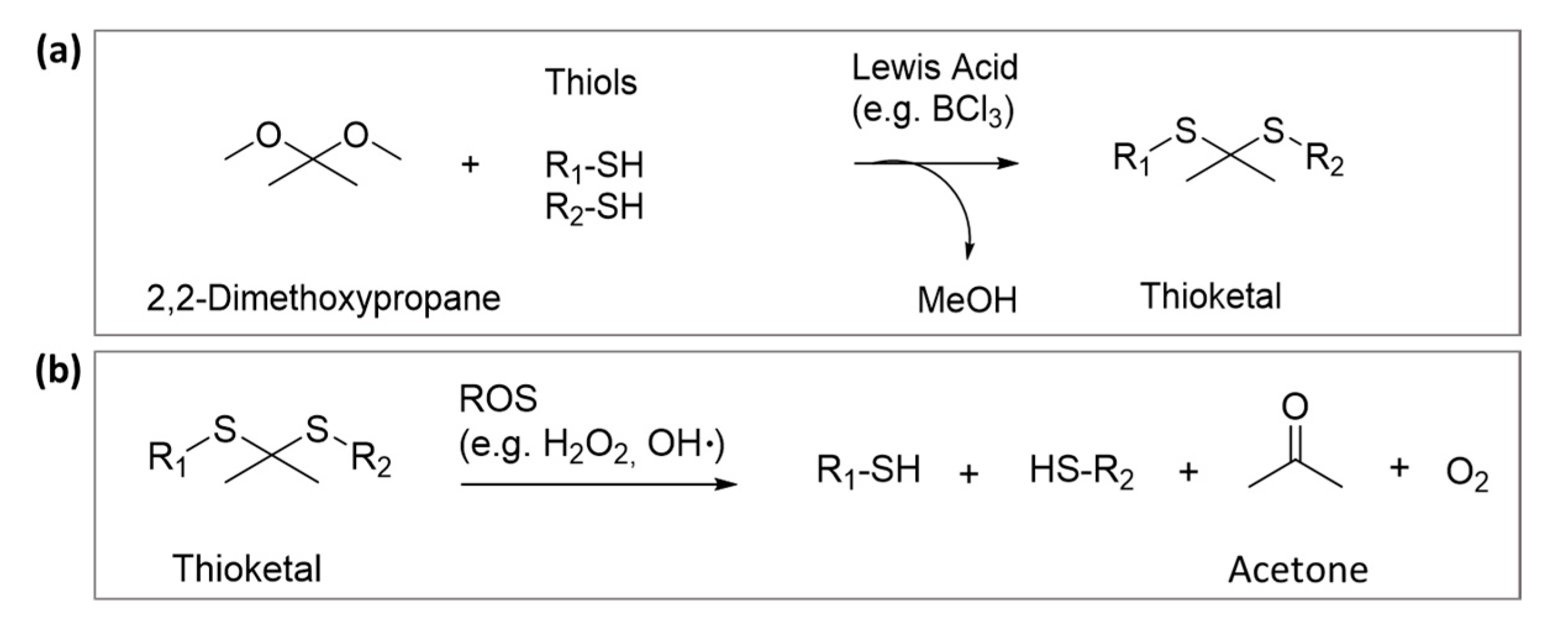
2. Thioketal
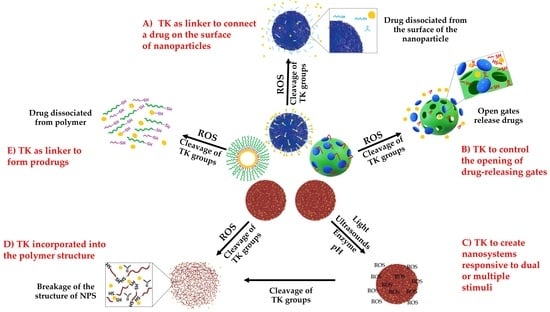
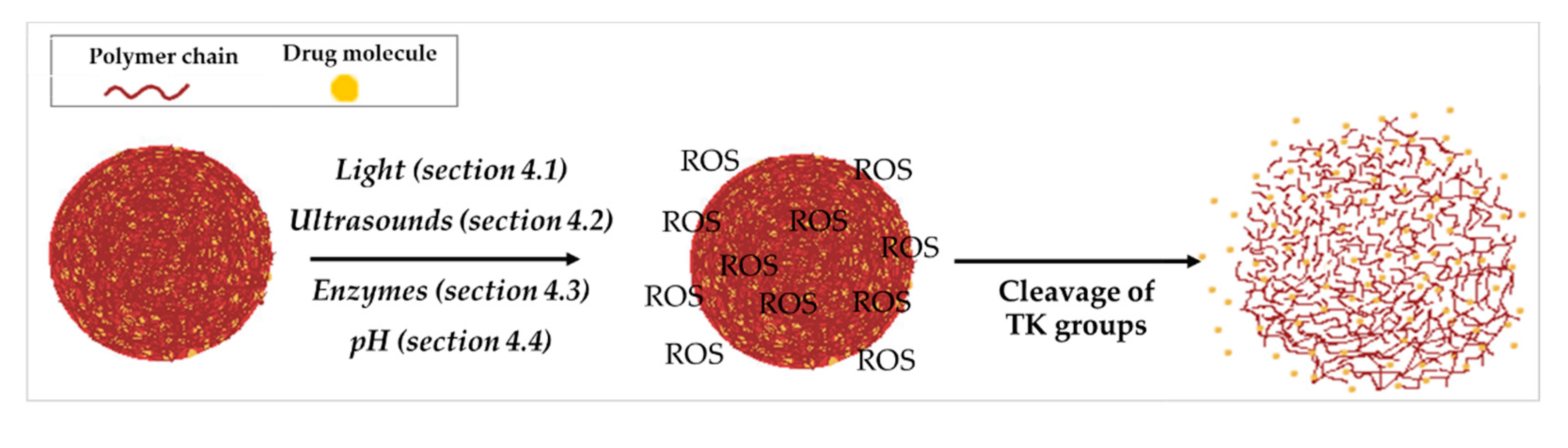
3. Different Technological Uses of TK in Drug Delivery Systems
3.1. TK Used as a Linker in DDS Conjugation for On-Demand Drug Release
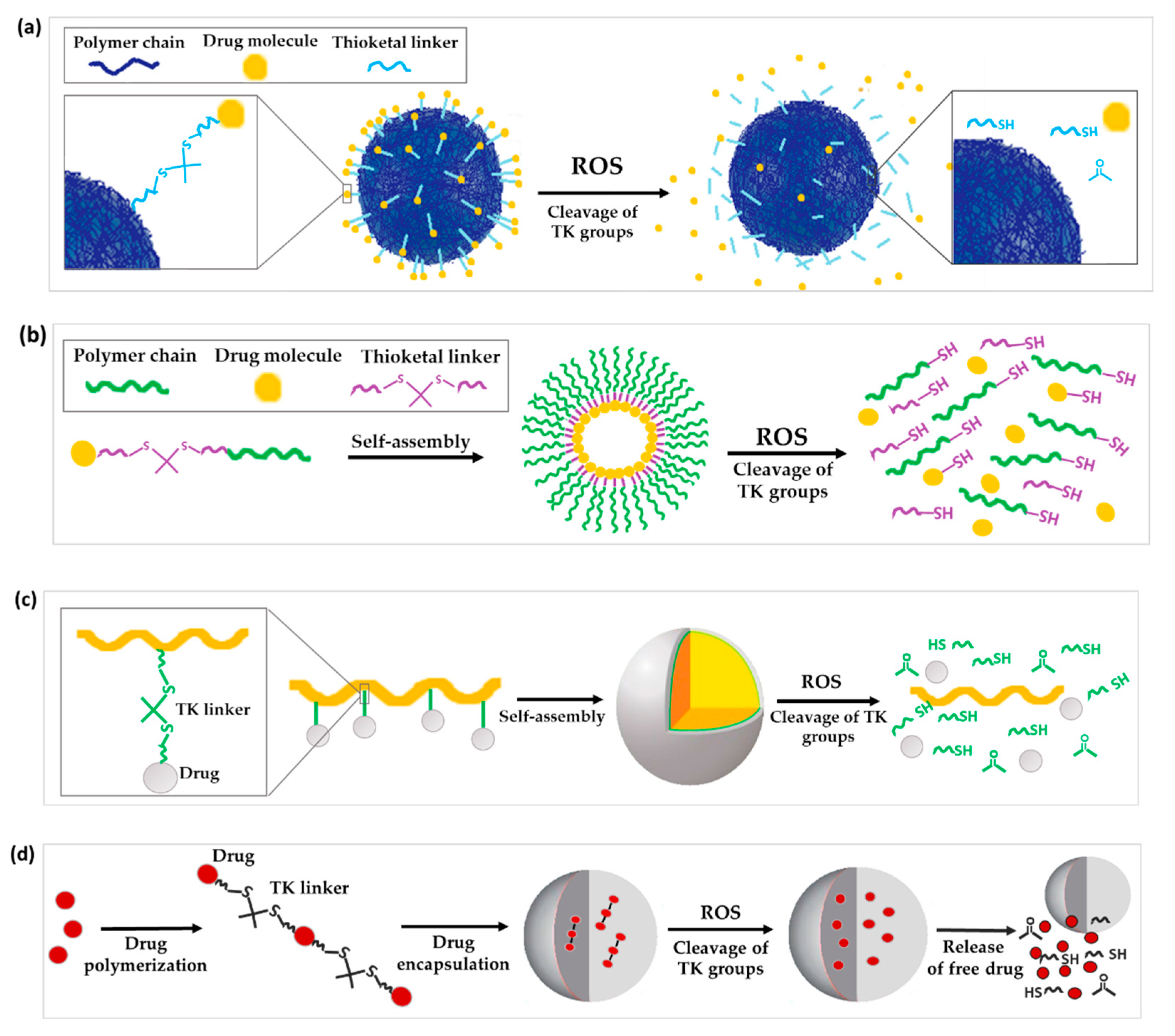
3.1.1. TK as Linker to Attach Drugs to the Surface of Nanoparticles
3.1.2. TK as a Linker for Polymer-Dye Conjugation
3.1.3. TK as Linkage to Form Prodrug or Polyprodrugs
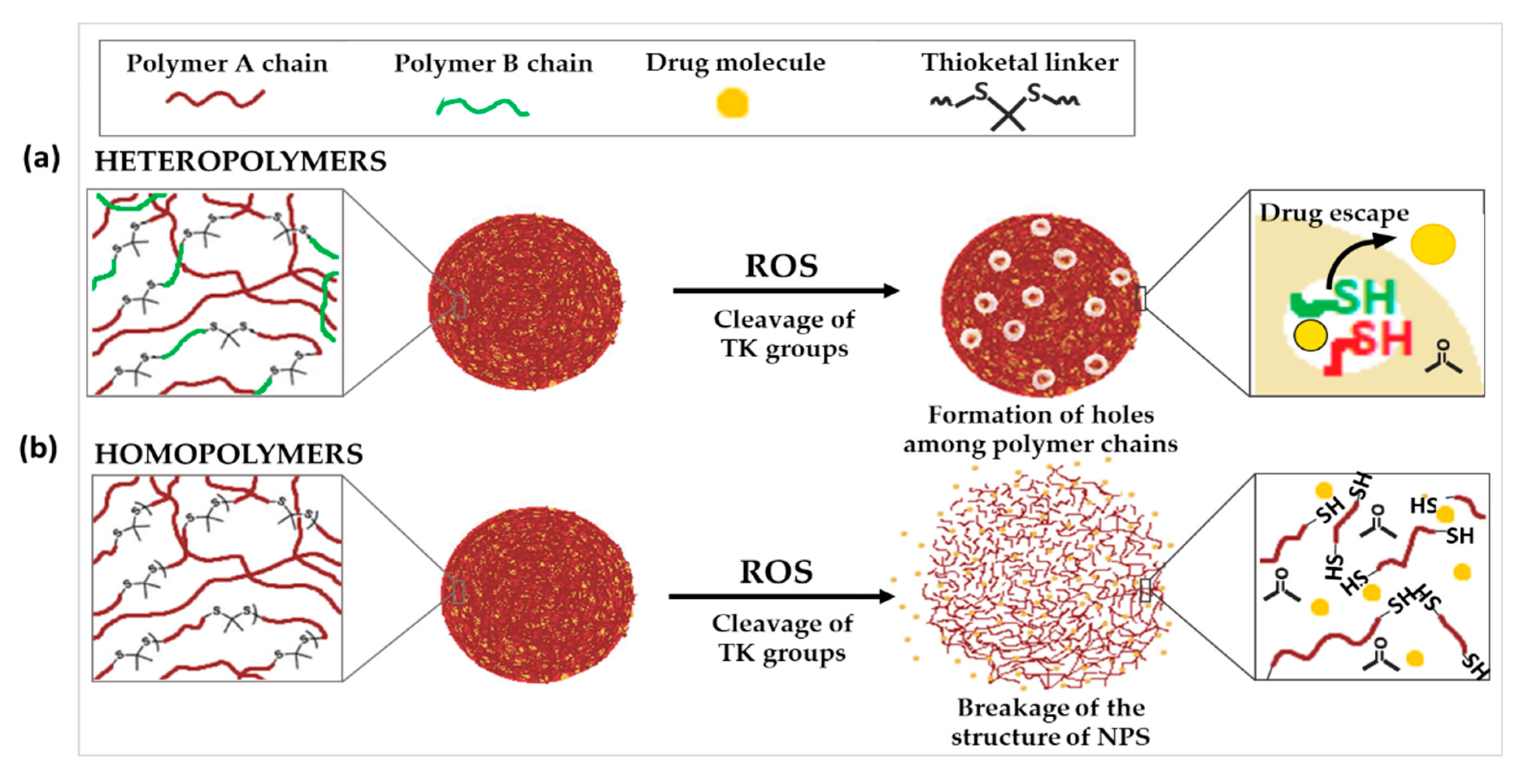
3.2. TK Incorporated into the Polymer Structure
3.3. Other Technological Uses of TK in DDS Structure
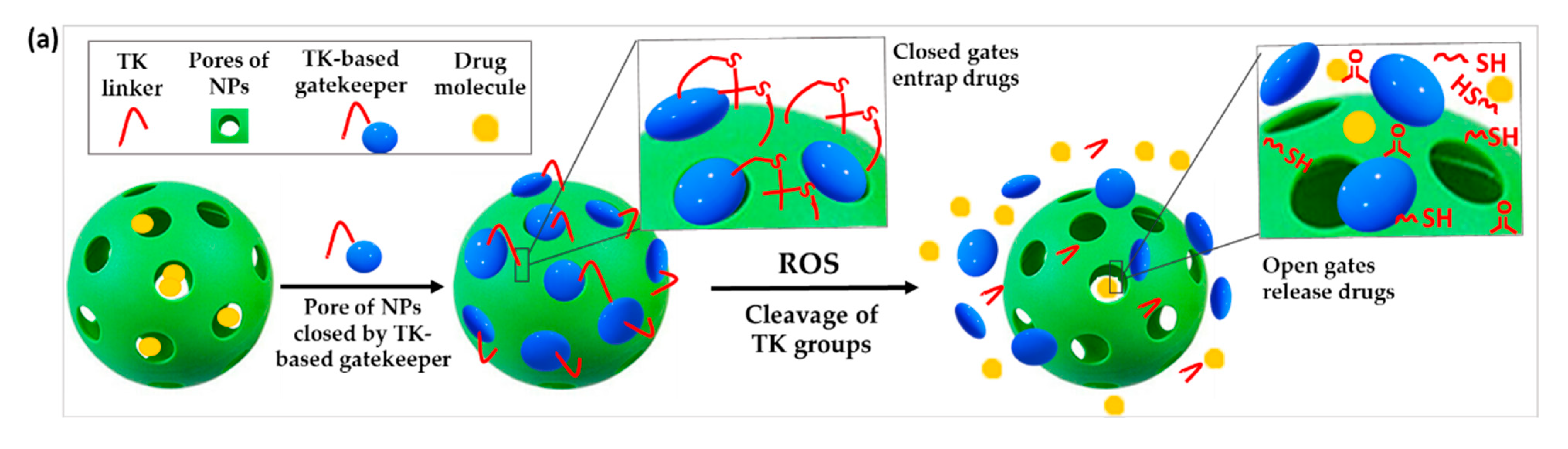
3.3.1. TK-Based ROS-Responsive Gates to Prevent Premature Drug Release
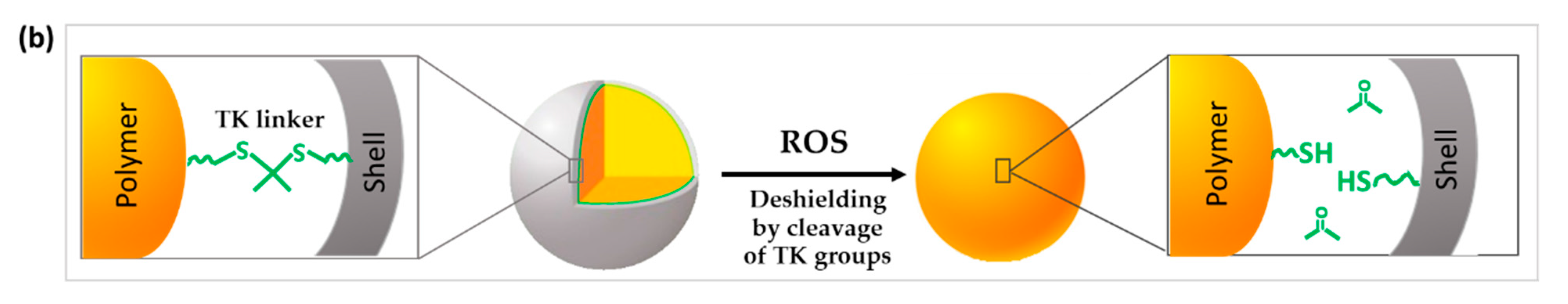
3.3.2. TK Used to Change the Surface Characteristics or the Conformation of the DDS
4. TK-Based ROS-Responsive DDS in Association with Other Stimuli
4.1. Light
4.1.1. TK as Linker in DDS Conjugation and Light
4.1.2. TK Incorporated into Polymers and PDT
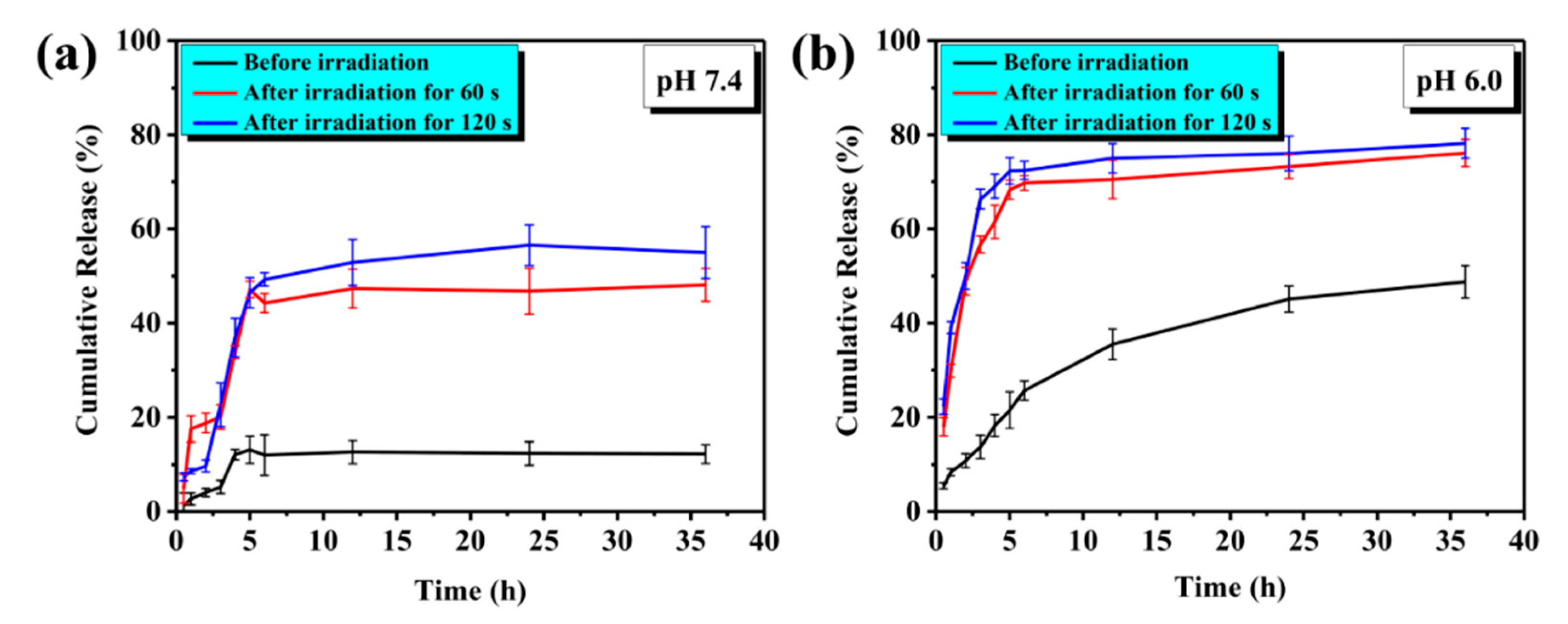
4.1.3. TK-Based ROS-Responsive Gates and Light
4.1.4. TK Used to Change the Surface Characteristics of the DDS and Light
4.1.5. TK-Containing Nanocomplexes for Gene Delivery and Light
4.2. TK and Ultrasounds
4.3. TK and Enzymes
4.4. pH
4.5. Multistimuli
5. Current Limitations in the Application of TK-Based DDS
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.d.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano Based Drug Delivery Systems: Recent Developments and Future Prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, G.; Yang, L.; Chen, G.; Xu, F.; Yang, F.; Yu, H.; Li, L.; Dong, X.; Han, J.; Cao, C.; et al. A Review on Drug Delivery System for Tumor Therapy. Front. Pharmacol. 2021, 12, 2294. [Google Scholar] [CrossRef] [PubMed]
- Simonazzi, A.; Cid, A.G.; Villegas, M.; Romero, A.I.; Palma, S.D.; Bermúdez, J.M. Nanotechnology Applications in Drug Controlled Release. In Drug Targeting and Stimuli Sensitive Drug Delivery Systems; Elsevier: Amsterdam, The Netherlands, 2018; pp. 81–116. ISBN 978-0-12-813689-8. [Google Scholar]
- Li, C.; Wang, J.; Wang, Y.; Gao, H.; Wei, G.; Huang, Y.; Yu, H.; Gan, Y.; Wang, Y.; Mei, L.; et al. Recent Progress in Drug Delivery. Acta Pharm. Sin. B 2019, 9, 1145–1162. [Google Scholar] [CrossRef] [PubMed]
- Duskey, J.T.; Ottonelli, I.; Rinaldi, A.; Parmeggiani, I.; Zambelli, B.; Wang, L.Z.; Prud’homme, R.K.; Vandelli, M.A.; Tosi, G.; Ruozi, B. Tween® Preserves Enzyme Activity and Stability in PLGA Nanoparticles. Nanomaterials 2021, 11, 2946. [Google Scholar] [CrossRef]
- Duskey, J.T.; Ottonelli, I.; Da Ros, F.; Vilella, A.; Zoli, M.; Kovachka, S.; Spyrakis, F.; Vandelli, M.A.; Tosi, G.; Ruozi, B. Novel Peptide-Conjugated Nanomedicines for Brain Targeting: In Vivo Evidence. Nanomedicine 2020, 28, 102226. [Google Scholar] [CrossRef]
- Mulvihill, J.J.; Cunnane, E.M.; Ross, A.M.; Duskey, J.T.; Tosi, G.; Grabrucker, A.M. Drug Delivery across the Blood–Brain Barrier: Recent Advances in the Use of Nanocarriers. Nanomedicine 2020, 15, 205–214. [Google Scholar] [CrossRef]
- Edgar, J.Y.C.; Wang, H. Introduction for Design of Nanoparticle Based Drug Delivery Systems. Curr. Pharm. Des. 2017, 23, 2108–2112. [Google Scholar] [CrossRef]
- Liu, D.; Yang, F.; Xiong, F.; Gu, N. The Smart Drug Delivery System and Its Clinical Potential. Theranostics 2016, 6, 1306–1323. [Google Scholar] [CrossRef]
- Lombardo, D.; Kiselev, M.A.; Caccamo, M.T. Smart Nanoparticles for Drug Delivery Application: Development of Versatile Nanocarrier Platforms in Biotechnology and Nanomedicine. J. Nanomater. 2019, 2019, e3702518. [Google Scholar] [CrossRef]
- Martin, J.R.; Gupta, M.K.; Page, J.M.; Yu, F.; Davidson, J.M.; Guelcher, S.A.; Duvall, C.L. A Porous Tissue Engineering Scaffold Selectively Degraded by Cell-Generated Reactive Oxygen Species. Biomaterials 2014, 35, 3766–3776. [Google Scholar] [CrossRef] [Green Version]
- Ramasamy, T.; Ruttala, H.B.; Gupta, B.; Poudel, B.K.; Choi, H.-G.; Yong, C.S.; Kim, J.O. Smart Chemistry-Based Nanosized Drug Delivery Systems for Systemic Applications: A Comprehensive Review. J. Control. Release 2017, 258, 226–253. [Google Scholar] [CrossRef] [PubMed]
- Hossen, S.; Hossain, M.K.; Basher, M.K.; Mia, M.N.H.; Rahman, M.T.; Uddin, M.J. Smart Nanocarrier-Based Drug Delivery Systems for Cancer Therapy and Toxicity Studies: A Review. J. Adv. Res. 2019, 15, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Kwon, E.J.; Lo, J.H.; Bhatia, S.N. Smart Nanosystems: Bio-Inspired Technologies That Interact with the Host Environment. Proc. Natl. Acad. Sci. USA 2015, 112, 14460–14466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, P.; Zhu, F.; Zhang, Z.; Cheng, Y.; Wang, Z.; Li, Y. Stimuli-Responsive Polydopamine-Based Smart Materials. Chem. Soc. Rev. 2021, 50, 8319–8343. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wang, C.; Li, L.; Zhu, F.; Ren, X.; Huang, Q.; Cheng, Y.; Li, Y. Bioinspired Integration of Naturally Occurring Molecules towards Universal and Smart Antibacterial Coatings. Adv. Funct. Mater. 2022, 32, 2108749. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative Stress: An Essential Factor in the Pathogenesis of Gastrointestinal Mucosal Diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef] [Green Version]
- Ballance, W.C.; Qin, E.C.; Chung, H.J.; Gillette, M.U.; Kong, H. Reactive Oxygen Species-Responsive Drug Delivery Systems for the Treatment of Neurodegenerative Diseases. Biomaterials 2019, 217, 119292. [Google Scholar] [CrossRef]
- Zahra, K.F.; Lefter, R.; Ali, A.; Abdellah, E.-C.; Trus, C.; Ciobica, A.; Timofte, D. The Involvement of the Oxidative Stress Status in Cancer Pathology: A Double View on the Role of the Antioxidants. Oxid. Med. Cell. Longev. 2021, 2021, 9965916. [Google Scholar] [CrossRef]
- Barnham, K.J.; Masters, C.L.; Bush, A.I. Neurodegenerative Diseases and Oxidative Stress. Nat. Rev. Drug Discov. 2004, 3, 205–214. [Google Scholar] [CrossRef]
- Joshi-Barr, S.; de Gracia Lux, C.; Mahmoud, E.; Almutairi, A. Exploiting Oxidative Microenvironments in the Body as Triggers for Drug Delivery Systems. Antioxid. Redox Signal. 2014, 21, 730–754. [Google Scholar] [CrossRef] [Green Version]
- Saravanakumar, G.; Kim, J.; Kim, W.J. Reactive-Oxygen-Species-Responsive Drug Delivery Systems: Promises and Challenges. Adv. Sci. 2017, 4, 1600124. [Google Scholar] [CrossRef] [PubMed]
- Huo, M.; Yuan, J.; Tao, L.; Wei, Y. Redox-Responsive Polymers for Drug Delivery: From Molecular Design to Applications. Polym. Chem. 2014, 5, 1519–1528. [Google Scholar] [CrossRef]
- Lee, S.H.; Gupta, M.K.; Bang, J.B.; Bae, H.; Sung, H.-J. Current Progress in Reactive Oxygen Species (ROS)-Responsive Materials for Biomedical Applications. Adv. Healthc. Mater. 2013, 2, 908–915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, B.; Thayumanavan, S. Mechanistic Investigation on Oxidative Degradation of ROS-Responsive Thioacetal/Thioketal Moieties and Their Implications. Cell Rep. Phys. Sci. 2020, 1, 100271. [Google Scholar] [CrossRef]
- El-Mohtadi, F.; d’Arcy, R.; Tirelli, N. Oxidation-Responsive Materials: Biological Rationale, State of the Art, Multiple Responsiveness, and Open Issues. Macromol. Rapid Commun. 2019, 40, 1800699. [Google Scholar] [CrossRef] [PubMed]
- Shim, M.S.; Xia, Y. A Reactive Oxygen Species (ROS)-Responsive Polymer for Safe, Efficient, and Targeted Gene Delivery in Cancer Cells. Angew. Chem. Int. Ed. 2013, 52, 6926–6929. [Google Scholar] [CrossRef] [PubMed]
- Geven, M.; d’Arcy, R.; Turhan, Z.Y.; El-Mohtadi, F.; Alshamsan, A.; Tirelli, N. Sulfur-Based Oxidation-Responsive Polymers. Chemistry, (Chemically Selective) Responsiveness and Biomedical Applications. Eur. Polym. J. 2021, 149, 110387. [Google Scholar] [CrossRef]
- Liang, J.; Liu, B. ROS-responsive Drug Delivery Systems. Bioeng. Transl. Med. 2016, 1, 239–251. [Google Scholar] [CrossRef]
- McEnery, M.A.P.; Lu, S.; Gupta, M.K.; Zienkiewicz, K.J.; Wenke, J.C.; Kalpakci, K.N.; Shimko, D.A.; Duvall, C.L.; Guelcher, S.A. Oxidatively Degradable Poly(Thioketal Urethane)/Ceramic Composite Bone Cements with Bone-like Strength. RSC Adv. 2016, 6, 109414–109424. [Google Scholar] [CrossRef] [Green Version]
- Ling, X.; Zhang, S.; Shao, P.; Wang, P.; Ma, X.; Bai, M. Synthesis of a Reactive Oxygen Species Responsive Heterobifunctional Thioketal Linker. Tetrahedron Lett. 2015, 56, 5242–5244. [Google Scholar] [CrossRef] [Green Version]
- Zheng, N.; Xie, D.; Zhang, Z.; Kuang, J.; Zheng, Y.; Wang, Q.; Li, Y. Thioketal-Crosslinked: ROS-Degradable Polycations for Enhanced in Vitro and in Vivo Gene Delivery with Self-Diminished Cytotoxicity. J. Biomater. Appl. 2019, 34, 326–338. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Jo, S.D.; Seah, G.L.; Kim, I.; Nam, Y.S. ROS-Induced Biodegradable Polythioketal Nanoparticles for Intracellular Delivery of Anti-Cancer Therapeutics. J. Ind. Eng. Chem. 2015, 21, 1137–1142. [Google Scholar] [CrossRef]
- Khan, F.; Tanaka, M. Designing Smart Biomaterials for Tissue Engineering. Int. J. Mol. Sci. 2018, 19, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGough, M.A.P.; Shiels, S.M.; Boller, L.A.; Zienkiewicz, K.J.; Duvall, C.L.; Wenke, J.C.; Guelcher, S.A. Poly(Thioketal Urethane) Autograft Extenders in an Intertransverse Process Model of Bone Formation. Tissue Eng. Part A 2019, 25, 949–963. [Google Scholar] [CrossRef]
- Wilson, D.S.; Dalmasso, G.; Wang, L.; Sitaraman, S.V.; Merlin, D.; Murthy, N. Orally Delivered Thioketal Nanoparticles Loaded with TNF-α–SiRNA Target Inflammation and Inhibit Gene Expression in the Intestines. Nat. Mater. 2010, 9, 923–928. [Google Scholar] [CrossRef]
- Tao, W.; He, Z. ROS-Responsive Drug Delivery Systems for Biomedical Applications. Asian J. Pharm. Sci. 2018, 13, 101–112. [Google Scholar] [CrossRef]
- Shi, J.; Wang, B.; Wang, L.; Lu, T.; Fu, Y.; Zhang, H.; Zhang, Z. Fullerene (C 60 )-Based Tumor-Targeting Nanoparticles with “off-on” State for Enhanced Treatment of Cancer. J. Control. Release 2016, 235, 245–258. [Google Scholar] [CrossRef]
- Oddone, N.; Pederzoli, F.; Duskey, J.T.; De Benedictis, C.A.; Grabrucker, A.M.; Forni, F.; Angela Vandelli, M.; Ruozi, B.; Tosi, G. ROS-Responsive “Smart” Polymeric Conjugate: Synthesis, Characterization and Proof-of-Concept Study. Int. J. Pharm. 2019, 570, 118655. [Google Scholar] [CrossRef]
- Redasani, V.K.; Bari, S.B. (Eds.) Chapter 2—Concept of Prodrug. In Prodrug Design; Academic Press: Cambridge, MA, USA, 2015; pp. 7–20. ISBN 978-0-12-803519-1. [Google Scholar]
- Rautio, J.; Kumpulainen, H.; Heimbach, T.; Oliyai, R.; Oh, D.; Järvinen, T.; Savolainen, J. Prodrugs: Design and Clinical Applications. Nat. Rev. Drug. Discov. 2008, 7, 255–270. [Google Scholar] [CrossRef]
- Taresco, V.; Alexander, C.; Singh, N.; Pearce, A.K. Stimuli-Responsive Prodrug Chemistries for Drug Delivery. Adv. Ther. 2018, 1, 1800030. [Google Scholar] [CrossRef] [Green Version]
- Oddone, N.; Boury, F.; Garcion, E.; Grabrucker, A.M.; Martinez, M.C.; Da Ros, F.; Janaszewska, A.; Forni, F.; Vandelli, M.A.; Tosi, G.; et al. Synthesis, Characterization, and In Vitro Studies of an Reactive Oxygen Species (ROS)-Responsive Methoxy Polyethylene Glycol-Thioketal-Melphalan Prodrug for Glioblastoma Treatment. Front. Pharmacol. 2020, 11, 574. [Google Scholar] [CrossRef] [PubMed]
- Ryrfeldt, A.; Andersson, P.; Edsbäcker, S.; Tönnesson, M.; Davies, D.; Pauwels, R. Pharmacokinetics and Metabolism of Budesonide, a Selective Glucocorticoid. Eur. J. Respir. Dis. Suppl. 1982, 122, 86–95. [Google Scholar] [PubMed]
- Li, S.; Xie, A.; Li, H.; Zou, X.; Zhang, Q. A Self-Assembled, ROS-Responsive Janus-Prodrug for Targeted Therapy of Inflammatory Bowel Disease. J. Control. Release 2019, 316, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Swierczewska, M.; Lee, K.C.; Lee, S. What Is the Future of PEGylated Therapies? Expert Opin. Emerg. Drugs 2015, 20, 531–536. [Google Scholar] [CrossRef]
- Pan, Q.; Deng, X.; Gao, W.; Chang, J.; Pu, Y.; He, B. ROS Triggered Cleavage of Thioketal Moiety to Dissociate Prodrug Nanoparticles for Chemotherapy. Colloids Surf. B Biointerfaces 2020, 194, 111223. [Google Scholar] [CrossRef]
- Lin, C.; Tao, Y.; Saw, P.E.; Cao, M.; Huang, H.; Xu, X. A Polyprodrug-Based Nanoplatform for Cisplatin Prodrug Delivery and Combination Cancer Therapy. Chem. Commun. 2019, 55, 13987–13990. [Google Scholar] [CrossRef]
- Li, M.; Jiang, S.; Haller, A.; Wirsching, S.; Fichter, M.; Simon, J.; Wagner, M.; Mailänder, V.; Gehring, S.; Crespy, D.; et al. Encapsulation of Polyprodrugs Enables an Efficient and Controlled Release of Dexamethasone. Nanoscale Horiz. 2021, 6, 791–800. [Google Scholar] [CrossRef]
- Yang, K.; Yang, Z.; Yu, G.; Nie, Z.; Wang, R.; Chen, X. Polyprodrug Nanomedicines: An Emerging Paradigm for Cancer Therapy. Adv. Mater. 2021, 34, 2107434. [Google Scholar] [CrossRef]
- Yin, W.; Ke, W.; Chen, W.; Xi, L.; Zhou, Q.; Mukerabigwi, J.F.; Ge, Z. Integrated Block Copolymer Prodrug Nanoparticles for Combination of Tumor Oxidative Stress Amplification and ROS-Responsive Drug Release. Biomaterials 2019, 195, 63–74. [Google Scholar] [CrossRef]
- Xu, X.; Saw, P.E.; Tao, W.; Li, Y.; Ji, X.; Bhasin, S.; Liu, Y.; Ayyash, D.; Rasmussen, J.; Huo, M.; et al. ROS-Responsive Polyprodrug Nanoparticles for Triggered Drug Delivery and Effective Cancer Therapy. Adv. Mater. 2017, 29, 1700141. [Google Scholar] [CrossRef]
- Pei, P.; Sun, C.; Tao, W.; Li, J.; Yang, X.; Wang, J. ROS-Sensitive Thioketal-Linked Polyphosphoester-Doxorubicin Conjugate for Precise Phototriggered Locoregional Chemotherapy. Biomaterials 2019, 188, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.-Y.; Jiang, H.; Yu, Y.; He, F.; Liu, Y.; Wang, Z.; Xiao, S.; Tang, C.; Xia, Z.; Ji, S.; et al. A New Method of Wound Treatment: Targeted Therapy of Skin Wounds with Reactive Oxygen Species-Responsive Nanoparticles Containing SDF-1α. Int. J. Nanomed. 2015, 10, 6571–6585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Wen, Y.; Wen, J.; Zhang, Y.-P.; Xu, X.-D.; Victorious, A.; Zavitz, R.; Xu, X. A New Biosafe Reactive Oxygen Species (ROS)-Responsive Nanoplatform for Drug Delivery. RSC Adv. 2016, 6, 38984–38989. [Google Scholar] [CrossRef]
- Li, Q.; Wen, Y.; You, X.; Zhang, F.; Shah, V.; Chen, X.; Tong, D.; Wei, X.; Yin, L.; Wu, J.; et al. Development of a Reactive Oxygen Species (ROS)-Responsive Nanoplatform for Targeted Oral Cancer Therapy. J. Mater. Chem. B 2016, 4, 4675–4682. [Google Scholar] [CrossRef]
- Sun, C.; Liang, Y.; Hao, N.; Xu, L.; Cheng, F.; Su, T.; Cao, J.; Gao, W.; Pu, Y.; He, B. A ROS-Responsive Polymeric Micelle with a π-Conjugated Thioketal Moiety for Enhanced Drug Loading and Efficient Drug Delivery. Org. Biomol. Chem. 2017, 15, 9176–9185. [Google Scholar] [CrossRef]
- Gisbert-Garzarán, M.; Vallet-Regí, M. Influence of the Surface Functionalization on the Fate and Performance of Mesoporous Silica Nanoparticles. Nanomaterials 2020, 10, 916. [Google Scholar] [CrossRef]
- Vijayakameswara, R.N.; Han, H.S.; Lee, H.; Nguyen, V.Q.; Jeon, S.; Jung, D.-W.; Lee, J.; Yi, G.-R.; Park, J.H. ROS-Responsive Mesoporous Silica Nanoparticles for MR Imaging-Guided Photodynamically Maneuvered Chemotherapy. Nanoscale 2018, 10, 9616–9627. [Google Scholar] [CrossRef]
- Shi, J.; Chen, Z.; Wang, B.; Wang, L.; Lu, T.; Zhang, Z. Reactive Oxygen Species-Manipulated Drug Release from a Smart Envelope-Type Mesoporous Titanium Nanovehicle for Tumor Sonodynamic-Chemotherapy. ACS Appl. Mater. Interfaces 2015, 7, 28554–28565. [Google Scholar] [CrossRef]
- Hu, J.-J.; Lei, Q.; Peng, M.-Y.; Zheng, D.-W.; Chen, Y.-X.; Zhang, X.-Z. A Positive Feedback Strategy for Enhanced Chemotherapy Based on ROS-Triggered Self-Accelerating Drug Release Nanosystem. Biomaterials 2017, 128, 136–146. [Google Scholar] [CrossRef]
- Li, J.; Sun, C.; Tao, W.; Cao, Z.; Qian, H.; Yang, X.; Wang, J. Photoinduced PEG Deshielding from ROS-Sensitive Linkage-Bridged Block Copolymer-Based Nanocarriers for on-Demand Drug Delivery. Biomaterials 2018, 170, 147–155. [Google Scholar] [CrossRef]
- Cheng, D.-B.; Zhang, X.-H.; Gao, Y.-J.; Ji, L.; Hou, D.; Wang, Z.; Xu, W.; Qiao, Z.-Y.; Wang, H. Endogenous Reactive Oxygen Species-Triggered Morphology Transformation for Enhanced Cooperative Interaction with Mitochondria. J. Am. Chem. Soc. 2019, 141, 7235–7239. [Google Scholar] [CrossRef] [PubMed]
- Abrahamse, H.; Hamblin, M.R. New Photosensitizers for Photodynamic Therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Master, A.; Livingston, M.; Gupta, A.S. Photodynamic Nanomedicine in the Treatment of Solid Tumors: Perspectives and Challenges. J. Control Release 2013, 168, 88–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raza, A.; Hayat, U.; Rasheed, T.; Bilal, M.; Iqbal, H.M.N. “Smart” Materials-Based near-Infrared Light-Responsive Drug Delivery Systems for Cancer Treatment: A Review. J. Mater. Res. Technol. 2019, 8, 1497–1509. [Google Scholar] [CrossRef]
- Zhang, T.; Lin, H.; Cui, L.; An, N.; Tong, R.; Chen, Y.; Yang, C.; Li, X.; Liu, J.; Qu, F. Near Infrared Light Triggered Reactive Oxygen Species Responsive Upconversion Nanoplatform for Drug Delivery and Photodynamic Therapy. Eur. J. Inorg. Chem. 2016, 2016, 1206–1213. [Google Scholar] [CrossRef]
- Cho, H.J.; Chung, M.; Shim, M.S. Engineered Photo-Responsive Materials for near-Infrared-Triggered Drug Delivery. J. Ind. Eng. Chem. 2015, 31, 15–25. [Google Scholar] [CrossRef]
- Ilhami, F.B.; Peng, K.-C.; Chang, Y.-S.; Alemayehu, Y.A.; Tsai, H.-C.; Lai, J.-Y.; Chiao, Y.-H.; Kao, C.-Y.; Cheng, C.-C. Photo-Responsive Supramolecular Micelles for Controlled Drug Release and Improved Chemotherapy. Int. J. Mol. Sci. 2020, 22, 154. [Google Scholar] [CrossRef]
- Yue, C.; Zhang, C.; Alfranca, G.; Yang, Y.; Jiang, X.; Yang, Y.; Pan, F.; de la Fuente, J.M.; Cui, D. Near-Infrared Light Triggered ROS-Activated Theranostic Platform Based on Ce6-CPT-UCNPs for Simultaneous Fluorescence Imaging and Chemo-Photodynamic Combined Therapy. Theranostics 2016, 6, 456–469. [Google Scholar] [CrossRef]
- Li, J.; Wei, K.; Zuo, S.; Xu, Y.; Zha, Z.; Ke, W.; Chen, H.; Ge, Z. Light-Triggered Clustered Vesicles with Self-Supplied Oxygen and Tissue Penetrability for Photodynamic Therapy against Hypoxic Tumor. Adv. Funct. Mater. 2017, 27, 1702108. [Google Scholar] [CrossRef]
- Montaseri, H.; Kruger, C.A.; Abrahamse, H. Recent Advances in Porphyrin-Based Inorganic Nanoparticles for Cancer Treatment. Int. J. Mol. Sci. 2020, 21, 3358. [Google Scholar] [CrossRef]
- Yue, C.; Yang, Y.; Zhang, C.; Alfranca, G.; Cheng, S.; Ma, L.; Liu, Y.; Zhi, X.; Ni, J.; Jiang, W.; et al. ROS-Responsive Mitochondria-Targeting Blended Nanoparticles: Chemo- and Photodynamic Synergistic Therapy for Lung Cancer with On-Demand Drug Release upon Irradiation with a Single Light Source. Theranostics 2016, 6, 2352–2366. [Google Scholar] [CrossRef] [PubMed]
- Phua, S.Z.F.; Xue, C.; Lim, W.Q.; Yang, G.; Chen, H.; Zhang, Y.; Wijaya, C.F.; Luo, Z.; Zhao, Y. Light-Responsive Prodrug-Based Supramolecular Nanosystems for Site-Specific Combination Therapy of Cancer. Chem. Mater. 2019, 31, 3349–3358. [Google Scholar] [CrossRef]
- Shi, S.; Zhang, L.; Zhu, M.; Wan, G.; Li, C.; Zhang, J.; Wang, Y.; Wang, Y. Reactive Oxygen Species-Responsive Nanoparticles Based on PEGlated Prodrug for Targeted Treatment of Oral Tongue Squamous Cell Carcinoma by Combining Photodynamic Therapy and Chemotherapy. ACS Appl. Mater. Interfaces 2018, 10, 29260–29272. [Google Scholar] [CrossRef] [PubMed]
- Seah, G.L.; Yu, J.H.; Koo, B.I.; Lee, D.J.; Nam, Y.S. Cancer-Targeted Reactive Oxygen Species-Degradable Polymer Nanoparticles for near Infrared Light-Induced Drug Release. J. Mater. Chem. B 2018, 6, 7737–7749. [Google Scholar] [CrossRef]
- Seah, G.L.; Yu, J.H.; Yang, M.Y.; Kim, W.J.; Kim, J.-H.; Park, K.; Cho, J.-W.; Kim, J.S.; Nam, Y.S. Low-Power and Low-Drug-Dose Photodynamic Chemotherapy via the Breakdown of Tumor-Targeted Micelles by Reactive Oxygen Species. J. Control. Release 2018, 286, 240–253. [Google Scholar] [CrossRef]
- Sun, C.-Y.; Cao, Z.; Zhang, X.-J.; Sun, R.; Yu, C.-S.; Yang, X. Cascade-Amplifying Synergistic Effects of Chemo-Photodynamic Therapy Using ROS-Responsive Polymeric Nanocarriers. Theranostics 2018, 8, 2939–2953. [Google Scholar] [CrossRef]
- Li, Y.; Hu, J.; Liu, X.; Liu, Y.; Lv, S.; Dang, J.; Ji, Y.; He, J.; Yin, L. Photodynamic Therapy-Triggered on-Demand Drug Release from ROS-Responsive Core-Cross-Linked Micelles toward Synergistic Anti-Cancer Treatment. Nano Res. 2019, 12, 999–1008. [Google Scholar] [CrossRef] [Green Version]
- Jin, H.; Zhu, T.; Huang, X.; Sun, M.; Li, H.; Zhu, X.; Liu, M.; Xie, Y.; Huang, W.; Yan, D. ROS-Responsive Nanoparticles Based on Amphiphilic Hyperbranched Polyphosphoester for Drug Delivery: Light-Triggered Size-Reducing and Enhanced Tumor Penetration. Biomaterials 2019, 211, 68–80. [Google Scholar] [CrossRef]
- Mohammed, F.; Ke, W.; Mukerabigwi, J.F.; Japir, A.A.-W.M.M.; Ibrahim, A.; Wang, Y.; Zha, Z.; Lu, N.; Zhou, M.; Ge, Z. ROS-Responsive Polymeric Nanocarriers with Photoinduced Exposure of Cell-Penetrating Moieties for Specific Intracellular Drug Delivery. ACS Appl. Mater. Interfaces 2019, 11, 31681–31692. [Google Scholar] [CrossRef]
- Wang, J.; He, X.; Shen, S.; Cao, Z.; Yang, X. ROS-Sensitive Cross-Linked Polyethylenimine for Red-Light-Activated SiRNA Therapy. ACS Appl. Mater. Interfaces 2019, 11, 1855–1863. [Google Scholar] [CrossRef]
- Wang, J.; He, H.; Xu, X.; Wang, X.; Chen, Y.; Yin, L. Far-Red Light-Mediated Programmable Anti-Cancer Gene Delivery in Cooperation with Photodynamic Therapy. Biomaterials 2018, 171, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Wan, G.-Y.; Liu, Y.; Chen, B.-W.; Liu, Y.-Y.; Wang, Y.-S.; Zhang, N. Recent Advances of Sonodynamic Therapy in Cancer Treatment. Cancer Biol. Med. 2016, 13, 325–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, Y.; Ye, J.; Li, Z.; Chen, H.; Gao, Y. Recent Progress in Sono-Photodynamic Cancer Therapy: From Developed New Sensitizers to Nanotechnology-Based Efficacy-Enhancing Strategies. Acta Pharm. Sin. B 2021, 11, 2197–2219. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Dong, W.; Guo, X.; Qiao, X.; Guo, S.; Zhang, L.; Wan, M.; Zong, Y. ROS-Responsive Blended Nanoparticles: Cascade-Amplifying Synergistic Effects of Sonochemotherapy with On-demand Boosted Drug Release During SDT Process. Adv. Healthc. Mater. 2019, 8, 1900720. [Google Scholar] [CrossRef]
- Chen, W.-H.; Luo, G.-F.; Qiu, W.-X.; Lei, Q.; Hong, S.; Wang, S.-B.; Zheng, D.-W.; Zhu, C.-H.; Zeng, X.; Feng, J.; et al. Programmed Nanococktail for Intracellular Cascade Reaction Regulating Self-Synergistic Tumor Targeting Therapy. Small 2016, 12, 733–744. [Google Scholar] [CrossRef]
- Rajamäki, K.; Nordström, T.; Nurmi, K.; Åkerman, K.E.O.; Kovanen, P.T.; Öörni, K.; Eklund, K.K. Extracellular Acidosis Is a Novel Danger Signal Alerting Innate Immunity via the NLRP3 Inflammasome. J. Biol. Chem. 2013, 288, 13410–13419. [Google Scholar] [CrossRef] [Green Version]
- Swietach, P.; Vaughan-Jones, R.D.; Harris, A.L.; Hulikova, A. The Chemistry, Physiology and Pathology of PH in Cancer. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2014, 369, 20130099. [Google Scholar] [CrossRef] [Green Version]
- Bami, M.S.; Raeisi Estabragh, M.A.; Khazaeli, P.; Ohadi, M.; Dehghannoudeh, G. PH-Responsive Drug Delivery Systems as Intelligent Carriers for Targeted Drug Therapy: Brief History, Properties, Synthesis, Mechanism and Application. J. Drug Deliv. Sci. Technol. 2021, 102987. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, Q.; An, S.; Lu, Y.; Li, J.; He, X.; Liu, L.; Zhang, Y.; Sun, T.; Jiang, C. ROS-Switchable Polymeric Nanoplatform with Stimuli-Responsive Release for Active Targeted Drug Delivery to Breast Cancer. ACS Appl. Mater. Interfaces 2017, 9, 12227–12240. [Google Scholar] [CrossRef]
- Wang, S.; Yu, G.; Wang, Z.; Jacobson, O.; Lin, L.; Yang, W.; Deng, H.; He, Z.; Liu, Y.; Chen, Z.; et al. Enhanced Antitumor Efficacy by a Cascade of Reactive Oxygen Species Generation and Drug Release. Angew. Chem. 2019, 131, 14900–14905. [Google Scholar] [CrossRef]
- Bao, Y.; Yin, L.; Liu, L.; Chen, L. Acid-sensitive ROS-triggered Dextran-based Drug Delivery System for Advanced Chemo-photodynamic Synergistic Therapy. J. Biomed. Mater. Res. 2020, 108, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Nomura, S.; Morimoto, Y.; Tsujimoto, H.; Arake, M.; Harada, M.; Saitoh, D.; Hara, I.; Ozeki, E.; Satoh, A.; Takayama, E.; et al. Highly Reliable, Targeted Photothermal Cancer Therapy Combined with Thermal Dosimetry Using a near-Infrared Absorbent. Sci. Rep. 2020, 10, 9765. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Zhang, X.; Wang, X.; Guan, X.; Zhang, W.; Ma, J. Recent Advances in Selective Photothermal Therapy of Tumor. J. Nanobiotechnology 2021, 19, 335. [Google Scholar] [CrossRef] [PubMed]
- Bastiancich, C.; Da Silva, A.; Estève, M.-A. Photothermal Therapy for the Treatment of Glioblastoma: Potential and Preclinical Challenges. Front. Oncol. 2021, 10, 3095. [Google Scholar] [CrossRef]
- Li, C.; Lin, W.; Liu, S.; Zhang, W.; Xie, Z. Self-Destructive PEG–BODIPY Nanomaterials for Photodynamic and Photothermal Therapy. J. Mater. Chem. B 2019, 7, 4655–4660. [Google Scholar] [CrossRef]
- Yang, G.-G.; Zhang, H.; Zhang, D.-Y.; Cao, Q.; Yang, J.; Ji, L.-N.; Mao, Z.-W. Cancer-Specific Chemotherapeutic Strategy Based on the Vitamin K3 Mediated ROS Regenerative Feedback and Visualized Drug Release In Vivo. Biomaterials 2018, 185, 73–85. [Google Scholar] [CrossRef]
- de Gracia Lux, C.; Joshi-Barr, S.; Nguyen, T.; Mahmoud, E.; Schopf, E.; Fomina, N.; Almutairi, A. Biocompatible Polymeric Nanoparticles Degrade and Release Cargo in Response to Biologically Relevant Levels of Hydrogen Peroxide. J. Am. Chem. Soc. 2012, 134, 15758–15764. [Google Scholar] [CrossRef] [Green Version]
- Chen, B.; Zhang, Y.; Ran, R.; Wang, B.; Qin, F.; Zhang, T.; Wan, G.; Chen, H.; Wang, Y. Reactive Oxygen Species-Responsive Nanoparticles Based on a Thioketal-Containing Poly(β-Amino Ester) for Combining Photothermal/Photodynamic Therapy and Chemotherapy. Polym. Chem. 2019, 10, 4746–4757. [Google Scholar] [CrossRef]
- Yao, Y.; Zhang, H.; Wang, Z.; Ding, J.; Wang, S.; Huang, B.; Ke, S.; Gao, C. Reactive Oxygen Species (ROS)-Responsive Biomaterials Mediate Tissue Microenvironments and Tissue Regeneration. J. Mater. Chem. B 2019, 7, 5019–5037. [Google Scholar] [CrossRef]
- Song, C.-C.; Du, F.-S.; Li, Z.-C. Oxidation-Responsive Polymers for Biomedical Applications. J. Mater. Chem. B 2014, 2, 3413–3426. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rinaldi, A.; Caraffi, R.; Grazioli, M.V.; Oddone, N.; Giardino, L.; Tosi, G.; Vandelli, M.A.; Calzà, L.; Ruozi, B.; Duskey, J.T. Applications of the ROS-Responsive Thioketal Linker for the Production of Smart Nanomedicines. Polymers 2022, 14, 687. https://doi.org/10.3390/polym14040687
Rinaldi A, Caraffi R, Grazioli MV, Oddone N, Giardino L, Tosi G, Vandelli MA, Calzà L, Ruozi B, Duskey JT. Applications of the ROS-Responsive Thioketal Linker for the Production of Smart Nanomedicines. Polymers. 2022; 14(4):687. https://doi.org/10.3390/polym14040687
Chicago/Turabian StyleRinaldi, Arianna, Riccardo Caraffi, Maria Vittoria Grazioli, Natalia Oddone, Luciana Giardino, Giovanni Tosi, Maria Angela Vandelli, Laura Calzà, Barbara Ruozi, and Jason Thomas Duskey. 2022. "Applications of the ROS-Responsive Thioketal Linker for the Production of Smart Nanomedicines" Polymers 14, no. 4: 687. https://doi.org/10.3390/polym14040687
APA StyleRinaldi, A., Caraffi, R., Grazioli, M. V., Oddone, N., Giardino, L., Tosi, G., Vandelli, M. A., Calzà, L., Ruozi, B., & Duskey, J. T. (2022). Applications of the ROS-Responsive Thioketal Linker for the Production of Smart Nanomedicines. Polymers, 14(4), 687. https://doi.org/10.3390/polym14040687