Effects of Various Polymeric Films on the Pericarp Microstructure and Storability of Longan (cv. Shixia) Fruit Treated with Propyl Disulfide Essential Oil from the Neem (Azadirachta indica) Plant
Abstract
1. Introduction
2. Materials and Methods
2.1. Fumigation and Fruit Treatment
2.2. Quality Evaluation Tests
2.2.1. Weight Loss, Decay Incidence, and Pericarp Browning Index
2.2.2. Color, Firmness, Total Soluble Solids, Titratable Acidity, and pH
2.2.3. Extraction and Determination of Phenolic and Flavonoid Contents
2.2.4. Polyphenol Oxidase (PPO) and Peroxidase (POD) Activity Levels
2.3. Scanning Electron Microscope and Pericarp Microstructure
2.4. Statistical Analysis
3. Results
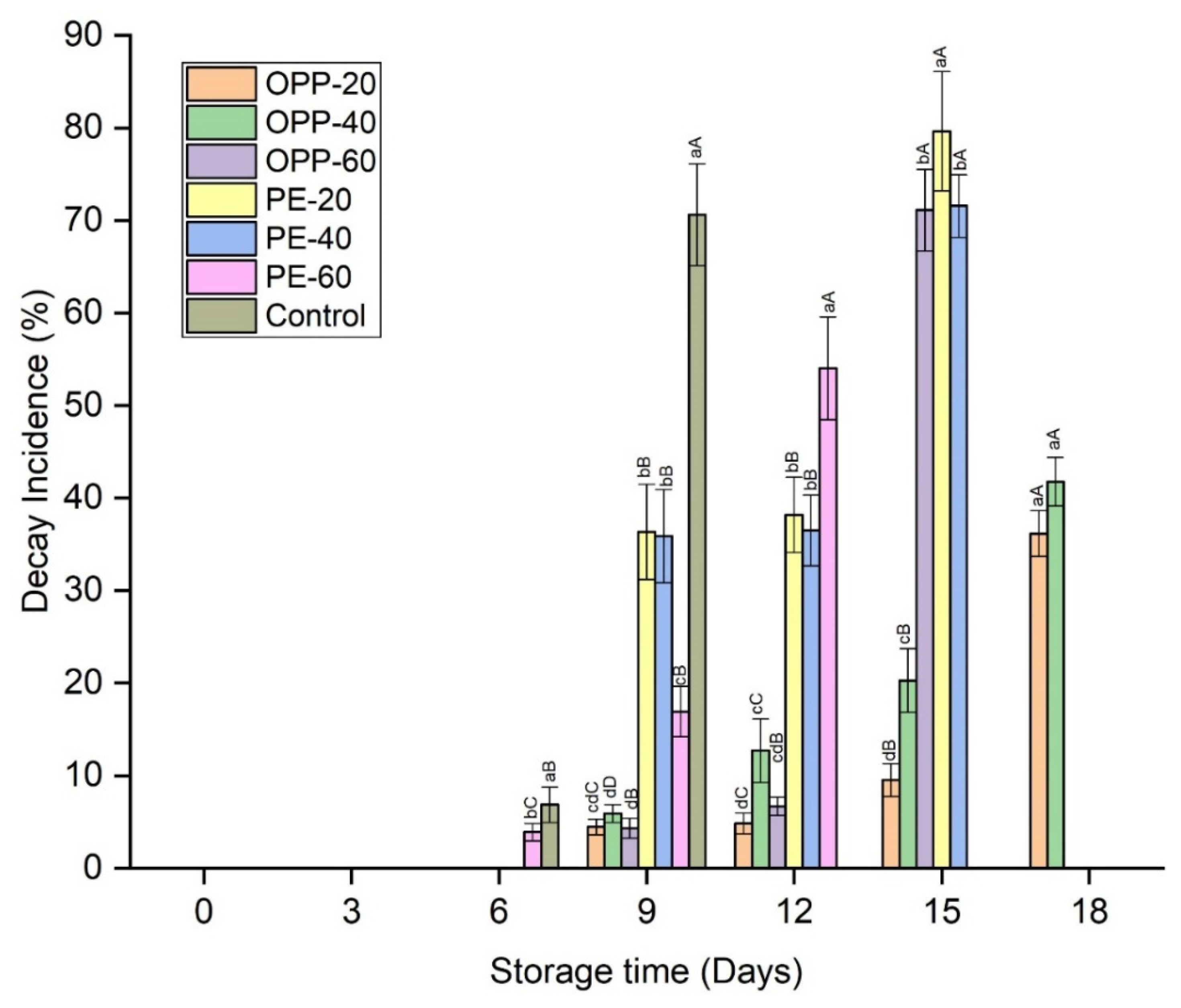
3.1. Weight Loss, Decay Incidence, and Pericarp Browning
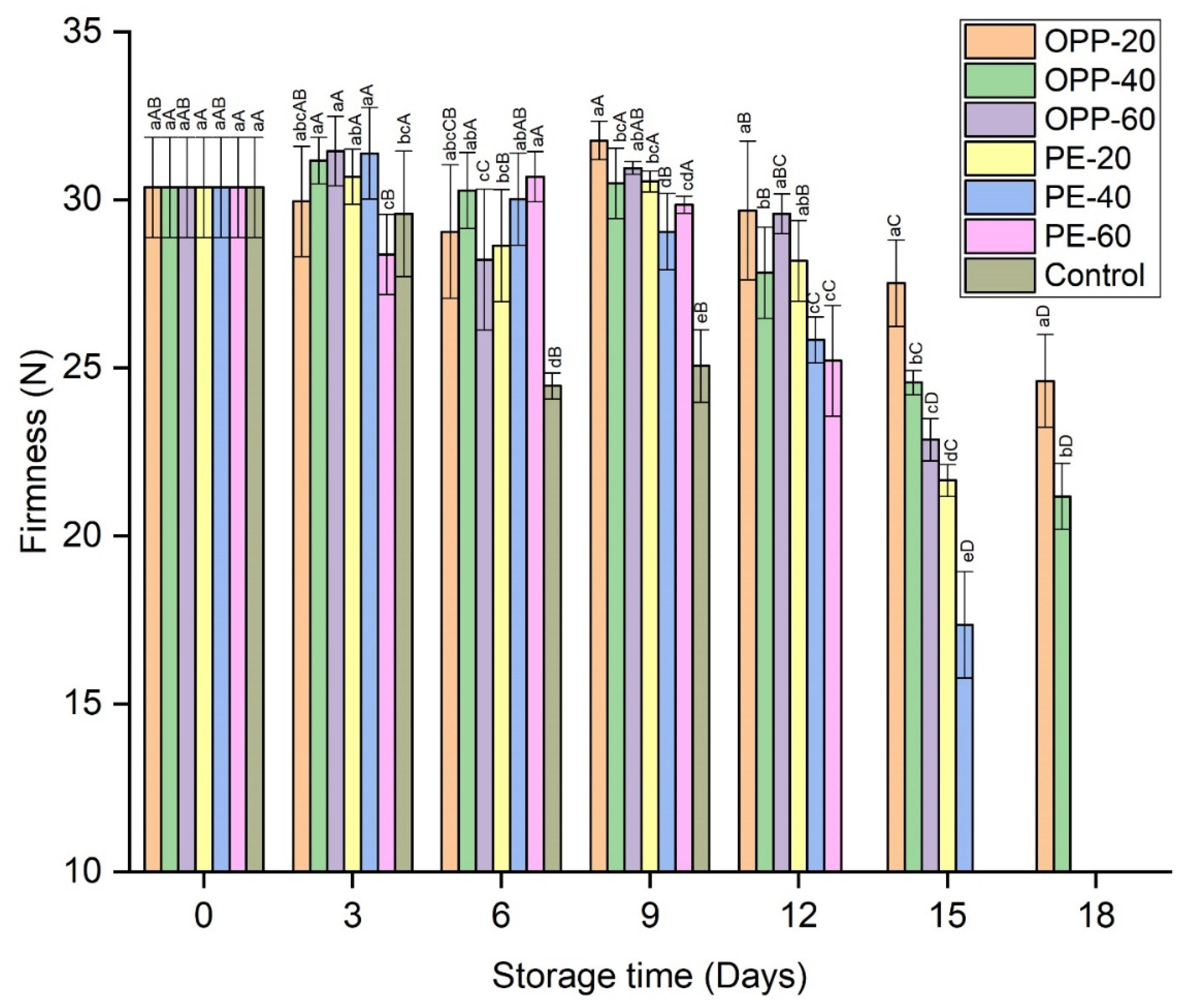
3.2. Firmness of Longan Fruit Samples
3.3. Color (L*, a*, and b*) Values
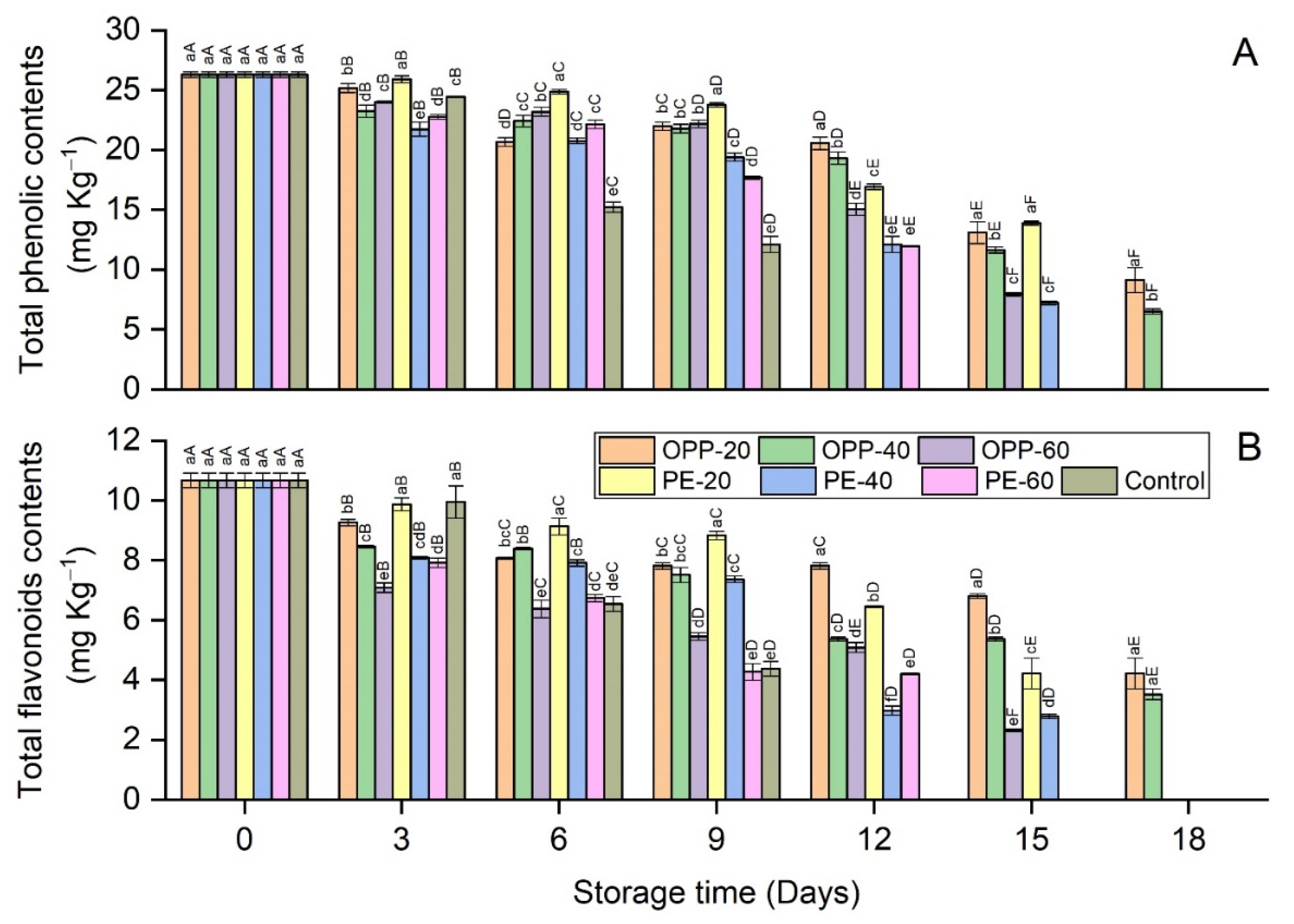
3.4. Total Phenolic (TPC) and Total Flavonoid Contents (TFC)
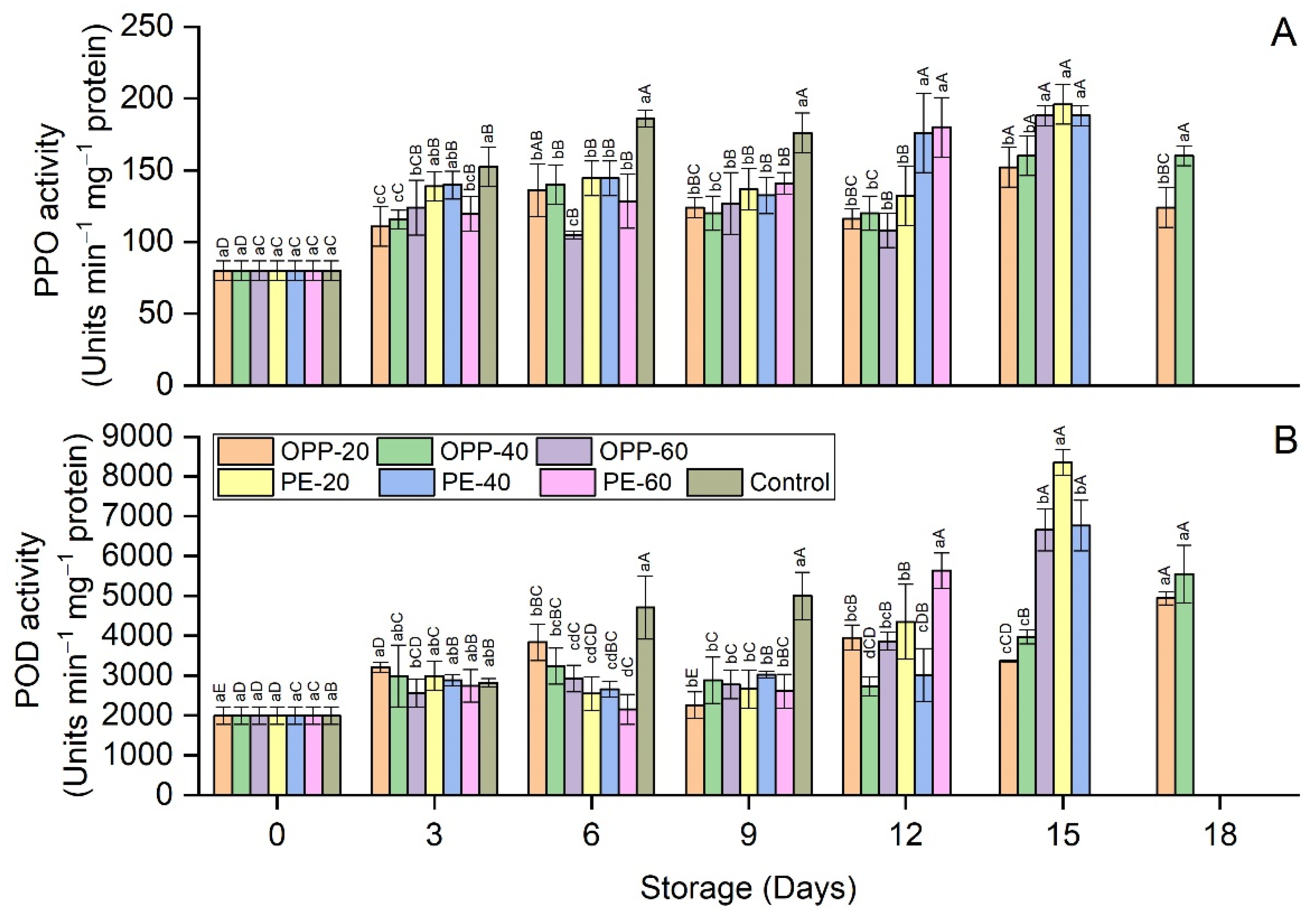
3.5. Enzymes Activity Levels
3.6. TSS, TA, and pH
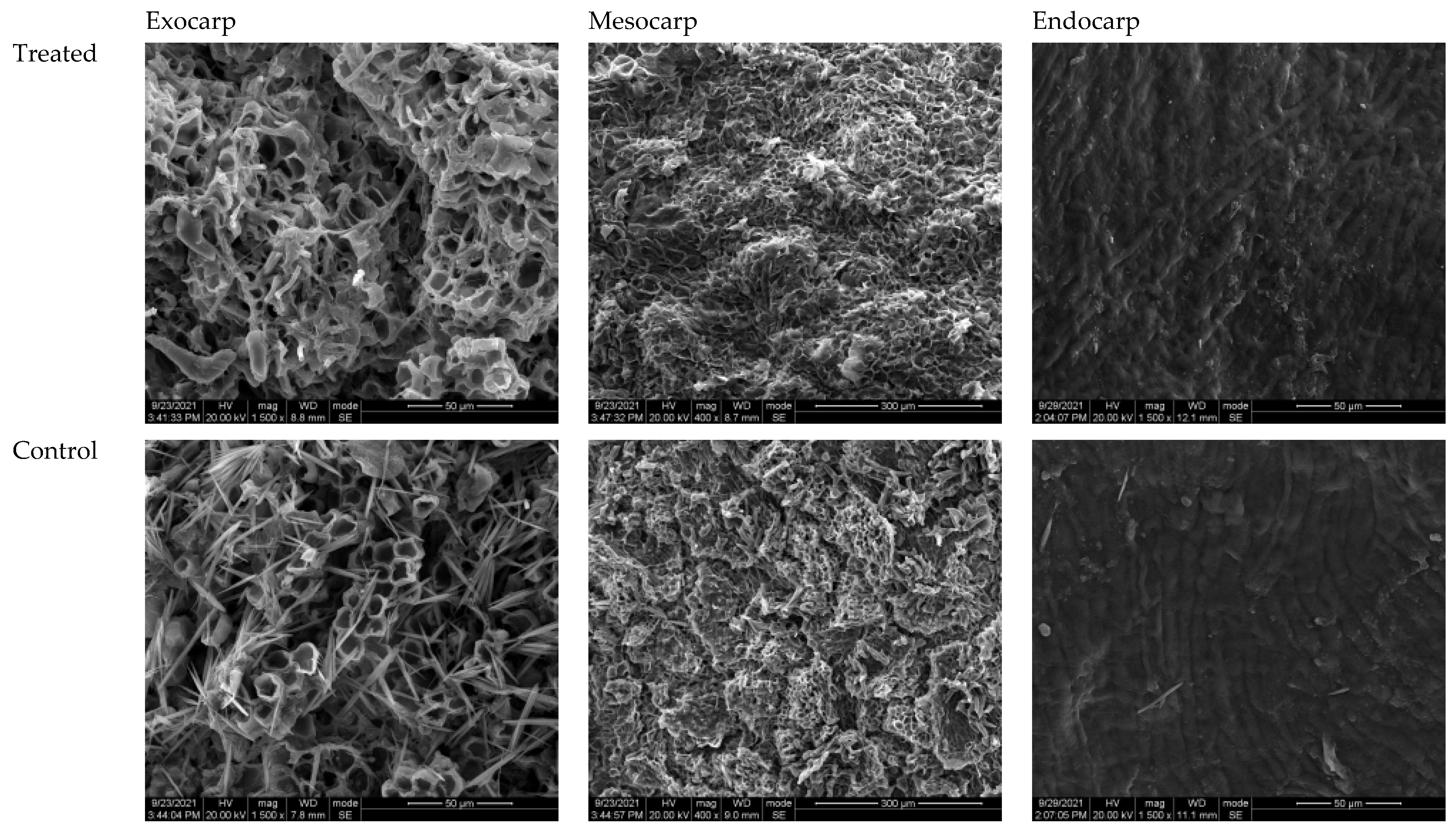
3.7. Scanning Electron Microscopy and Pericarp Microstructure
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yao, F.; Huang, Z.; Li, D.; Wang, H.; Xu, X.; Jiang, Y.; Qu, H. Phenolic components, antioxidant enzyme activities and anatomic structure of longan fruit pericarp following treatment with adenylatetriphosphate. Sci. Hortic. 2014, 180, 6–13. [Google Scholar] [CrossRef]
- Chumyam, A.; Shank, L.; Uthaibutra, J.; Saengnil, K. Effects of chlorine dioxide on mitochondrial energy levels and redox status of Daw longan pericarp during storage. Postharvest Biol. Technol. 2016, 116, 26–35. [Google Scholar] [CrossRef]
- Chen, M.; Lin, H.; Zhang, S.; Lin, Y.; Chen, Y.; Lin, Y. Effects of Adenosine Triphosphate (ATP) Treatment on Postharvest Physiology, Quality and Storage Behavior of Longan Fruit. Food Bioprocess Technol. 2015, 8, 971–982. [Google Scholar] [CrossRef]
- Lin, Y.; Lin, H.; Lin, Y.; Zhang, S.; Chen, Y.; Jiang, X. The roles of metabolism of membrane lipids and phenolics in hydrogen peroxide-induced pericarp browning of harvested longan fruit. Postharvest Biol. Technol. 2016, 111, 53–61. [Google Scholar] [CrossRef]
- Lin, Y.; Lin, Y.; Lin, Y.; Lin, M.; Chen, Y.; Wang, H.; Lin, H. A novel chitosan alleviates pulp breakdown of harvested longan fruit by suppressing disassembly of cell wall polysaccharides. Carbohydr. Polym. 2019, 217, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Joradol, A.; Uthaibutra, J.; Lithanatudom, P.; Saengnil, K. Induced expression of NOX and SOD by gaseous sulfur dioxide and chlorine dioxide enhances antioxidant capacity and maintains fruit quality of ‘Daw’ longan fruit during storage through H2O2 signaling. Postharvest Biol. Technol. 2019, 156, 110938. [Google Scholar] [CrossRef]
- Khan, M.R.; Huang, C.; Durrani, Y.; Muhammad, A. Chemistry of enzymatic browning in longan fruit as a function of pericarp pH and dehydration and its prevention by essential oil, an alternative approach to SO2 fumigation. PeerJ 2021, 9, e11539. [Google Scholar] [CrossRef]
- Khan, M.R.; Huang, C.; Zhao, H.; Huang, H.; Ren, L.; Faiq, M.; Hashmi, M.S.; Li, B.; Zheng, D.; Xu, Y.; et al. Antioxidant activity of thymol essential oil and inhibition of polyphenol oxidase enzyme: A case study on the enzymatic browning of harvested longan fruit. Chem. Biol. Technol. Agric. 2021, 8, 61. [Google Scholar] [CrossRef]
- Kumar, D.; Rahal, A.; Malik, K.J. Neem extract. In Nutraceuticals. Efficacy, Safety and Toxicity; Gupta, R.C., Ed.; Academic Press: Chennai, India, 2016; pp. 585–597. [Google Scholar] [CrossRef]
- Hossain, M.A.; Al-Toubi, W.A.S.; Weli, A.M.; Al-Riyami, Q.A.; Al-Sabahi, J.N. Identification and characterization of chemical compounds indifferent crude extracts from leaves of Omani neem. J. Taibah Univ. Sci. 2013, 7, 181–188. [Google Scholar] [CrossRef]
- Serrone, P.D.; Toniolo, C.; Nicoletti, M. Neem (Azadirachta indica A. Juss) oil: A natural preservative to control meat spoilage. Foods 2015, 4, 3–14. [Google Scholar] [CrossRef]
- VasudhaUdupa, A.; Gowda, B.; Kumarswammy, B.E.; Shivanna, M.B. The antimicrobial and antioxidant property, GC–MS analysis of non-edible oil-seed cakes of neem, madhuca, and simarouba. Bull. Natl. Res. Cent. 2021, 45, 41. [Google Scholar] [CrossRef]
- Pankaj, S.; Lokeshwar, T.; Mukesh, B.; Vishnu, B. Review on neem (Aazadirachta Indica): Thousand problems one solution. Int. Res. J. Pharm. 2011, 2, 97–102. [Google Scholar]
- Khan, M.R.; Chonhenchob, V.; Huang, C.; Suwanamornlert, P. Antifungal Activity of Propyl Disulfide from Neem (Azadirachta indica) in Vapor and Agar Diffusion Assays against Anthracnose Pathogens (Colletotrichum gloeosporioides and Colletotrichum acutatum) in Mango Fruit. Microorganisms 2021, 9, 839. [Google Scholar] [CrossRef]
- Khan, M.R.; Suwanamornlert, P.; Sangchote, S.; Chonhenchob, V. Antifungal activity of propyl disulphide from neem against Lasiodiplodia theobromae and Neofusicoccum parvum causing stem end rot in mango. J. Appl. Microbiol. 2020, 129, 1364–1373. [Google Scholar] [CrossRef] [PubMed]
- Zakawa, N.N.; Channya, K.F.; Magga, B.; Akesa, T.M. Antifungal effect of neem (Azadirachta indica) leaf extracts on mango fruit postharvest rot agents in Yola, Adamawa state. J. Pharmacogn. Phytochem. 2018, 7, 23–26. [Google Scholar]
- Suleiman, M.N. Antifungal properties of leaf extract of neem and tobacco on three fungal pathogens of tomato (Lycopersicon Esculentum Mill). Adv. Appl. Sci. Res. 2011, 2, 217–220. [Google Scholar]
- Serrone, P.D.; Nicoletti, M. Antimicrobial Activity of a Neem Cake Extract in a Broth Model Meat System. Int. J. Environ. Res. Public Health 2013, 10, 3282–3295. [Google Scholar] [CrossRef]
- Ouerfelli, M.; Villasante, J.; Kaâb, L.B.B.; Almajano, M. Effect of Neem (Azadirachta indica L.) on Lipid Oxidation in Raw Chilled Beef Patties. Antioxidants 2019, 8, 305. [Google Scholar] [CrossRef]
- Khan, M.R.; Suwanamornlert, P.; Leelaphiwat, P.; Chinsirikul, W.; Chonhenchob, V. Quality and biochemical changes in longan (Dimicarpus longan Lour cv. Daw) under different controlled atmosphere conditions. Int. J. Food Sci. Technol. 2017, 52, 2163–2170. [Google Scholar] [CrossRef]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal processing enhances the nutritional value of tomatoes by increasing total antioxidant activity. J. Agric. Food Chem. 2002, 50, 3010–3014. [Google Scholar] [CrossRef]
- Duan, X.; Su, X.; You, Y.; Qu, H.; Li, Y.; Jiang, Y. Effect of nitric oxide on pericarp browning of harvested longan fruit in relation to phenolic metabolism. Food Chem. 2007, 104, 571–576. [Google Scholar] [CrossRef]
- Jiang, Y.-M. Purification and some properties of polyphenol oxidase of longan fruit. Food Chem. 1999, 66, 75–79. [Google Scholar] [CrossRef]
- Zhang, Z.; Pang, X.; Xuewu, D.; Ji, Z.; Jiang, Y. Role of peroxidase in anthocyanin degradation in litchi fruit pericarp. Food Chem. 2005, 90, 47–52. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Chitbanchong, W.; Sardsud, V.; Whangchai, K.; Koslanund, R.; Thobunluepop, P. Minimally of polyphenol oxidase activity and controlling of rotting and browning of longan fruits cv. Daw by sulphur dioxide (SO2) treatment under cold storage conditions. Int. J. Agric. Res. 2009, 4, 349–361. [Google Scholar] [CrossRef][Green Version]
- Awad, A.H.R.; Parmar, A.; Ali, M.R.; El-Mogy, M.M.; Abdelgawad, K.F. Extending the Shelf-Life of Fresh-Cut Green Bean Pods by Ethanol, Ascorbic Acid, and Essential Oils. Foods 2021, 10, 1103. [Google Scholar] [CrossRef]
- Ramos De Resende, A.; Ludke Falcao, L.; Salviano Barbosa, G.; Helena Marcellino, L.; Silvano Gander, E. Neem (Azadirachta indica A. Juss) components: Candidates for control of Crinipellis perniciosa and Phytophthora spp. Microbiol. Res. 2007, 162, 238–243. [Google Scholar] [CrossRef]
- Koul, O. Biological activity of volatile di-n-propyl disulfide from seeds of neem, Azadirachta indica (Meliaceae), to two species of stored grain pests, Sitophilus oryzae (L.) and Tribolium castaneum (Herbst). J. Econ. Entomol. 2004, 97, 1142–1147. [Google Scholar] [CrossRef]
- Kumar, A.; Kudachikar, V. Efficacy of aroma compounds for postharvest management of mango Anthracnose. J. Plant Dis. Prot. 2019, 127, 245–256. [Google Scholar] [CrossRef]
- Valero, D.; Valverde, J.M.; Martínez-Romero, D.; Guillén, F.; Castillo, S.; Serrano, M. The combination of modified atmosphere packaging with eugenol or thymol to maintain quality, safety and functional properties of table grapes. Postharvest Biol. Technol. 2006, 41, 317–327. [Google Scholar] [CrossRef]
- Herrera-Calderon, O.; Chacaltana-Ramos, L.J.; Huayanca-Gutiérrez, I.C.; Algarni, M.A.; Alqarni, M.; Batiha, G.E.-S. Chemical Constituents, In Vitro Antioxidant Activity and In Silico Study on NADPH Oxidase of Allium sativum L. (Garlic) Essential Oil. Antioxidants 2021, 10, 1844. [Google Scholar] [CrossRef] [PubMed]




| Treatments | |||||||
|---|---|---|---|---|---|---|---|
| Days | OPP-20 | OPP-40 | OPP-60 | PE-20 | PE-40 | PE-60 | Control |
| L* | |||||||
| 0 | 69.44 aAB (2.44) | 69.44 aA (2.44) | 69.44 aA (2.44) | 69.44 aA (2.44) | 69.44 aA (2.44) | 69.44 aA (2.44) | 69.44 aA (2.44) |
| 3 | 70.68 aA (0.26) | 67.47 abAB (1.90) | 66.25 bAB (1.70) | 65.64 bB (0.89) | 67.13 abA (2.30) | 64.74 bA (0.78) | 56.59 cB (2.22) |
| 6 | 66.81 aBC (0.14) | 67.00 aAB (0.17) | 66.91 aAB (0.06) | 66.22 aB (0.87) | 66.61 aA (0.42) | 65.84 aA (1.10) | 48.44 bC (2.44) |
| 9 | 66.01 aCD (0.51) | 66.47 aB (0.61) | 64.33 bB (2.23) | 61.88 cC (2.07) | 62.16 cB (1.14) | 59.39 dB (0.86) | 35.50 eD (0.35) |
| 12 | 62.70 aDE (0.28) | 64.89 aB (0.82) | 56.91 bC (0.06) | 53.55 bD (0.13) | 53.27 bC (2.61) | 52.28 bC (4.16) | – |
| 15 | 59.37 aEF (1.15) | 60.61 aC (0.73) | 48.02 bD (1.13) | 46.88 bE (0.93) | 48.83 bD (2.82) | – | – |
| 18 | 57.94 aE (0.16) | 56.03 aD (1.02) | – | – | – | – | – |
| a* | |||||||
| 0 | 1.78 aC (0.24) | 1.78 aE (0.24) | 1.78 aD (0.24) | 1.78 aE (0.24) | 1.78 aD (0.24) | 1.78 aD (0.24) | 1.78 aD (0.24) |
| 3 | 2.36 bC (0.22) | 3.44 aD (0.30) | 2.64 bC (0.21) | 4.00 aD (0.15) | 3.38 aC (0.29) | 3.61 aBC (0.19) | 3.45 aC (0.13) |
| 6 | 4.02 abcB (0.08) | 3.65 bcCD (0.28) | 3.84 abcB (0.23) | 4.23 abCD (0.33) | 3.83 abcC (0.21) | 3.38 cC (0.26) | 4.51 aB (0.26) |
| 9 | 4.11 bcB (0.28) | 4.23 bcBCD (0.06) | 4.12 bcB (0.13) | 4.68 bBC (0.20) | 4.06 cBC (0.24) | 4.23 bcB (0.16) | 5.87 aA (0.35) |
| 12 | 4.43 bB (0.10) | 4.51 bBC (0.10) | 4.57 abB (0.16) | 4.91 abAB (0.18) | 4.57 abB (0.18) | 5.23 aA (0.17) | – |
| 15 | 4.68 bAB (0.03) | 5.07 abAB (0.24) | 6.02 aA (0.19) | 5.58 abA (0.43) | 5.35 abA (0.17) | – | – |
| 18 | 5.19 bA (0.19) | 5.84 aA (0.33) | – | – | – | – | – |
| b* | |||||||
| 0 | 35.23 aA (0.85) | 35.23 aA (0.85) | 35.23 aA (0.85) | 35.23 aA (0.85) | 35.23 aA (0.85) | 35.23 aA (0.85) | 35.23 aA (0.85) |
| 3 | 33.96 bA (0.10) | 35.04 aA (0.35) | 30.29 bB (1.82) | 28.66 bcB (0.75) | 35.15 aA (0.34) | 26.14 cdB (1.90) | 23.82 dB (2.14) |
| 6 | 31.51 aAB (0.62) | 32.15 aB (0.34) | 27.74 bBC (0.59) | 26.66 bcBC (1.14) | 32.37 aA (0.25) | 23.92 cdBC (0.67) | 20.38 dC (1.32) |
| 9 | 28.51 aBC (0.93) | 30.60 aB (0.91) | 25.40 bC (0.80) | 25.33 bC (0.22) | 28.71 aB (0.60) | 20.80 cCD (1.29) | 18.05 dC (0.57) |
| 12 | 25.74 aCD (0.70) | 25.71 aC (0.96) | 20.96 bD (1.33) | 22.55 abD (1.08) | 26.15 aB (0.09) | 18.92 bD (0.51) | – |
| 15 | 22.96 aDE (1.59) | 22.37 aD (0.23) | 18.51 bD (0.26) | 21.33 aD (0.59) | 21.37 aC (1.05) | – | – |
| 18 | 20.96 aE (0.37) | 19.71 bE (0.21) | – | – | – | – | – |
| Treatments | |||||||
|---|---|---|---|---|---|---|---|
| Days | OPP-20 | OPP-40 | OPP-60 | PE-20 | PE-40 | PE-60 | Control |
| TSS | |||||||
| 0 | 20.73 aA (0.12) | 20.73 aA (0.12) | 20.73 aA (0.12) | 20.73 aA (0.12) | 20.73 aA (0.12) | 20.73 aA (0.12) | 20.73 aA (0.12) |
| 3 | 18.77 bcC (0.15) | 18.53 cC (0.32) | 18.40 cC (0.10) | 18.80 bcB (0.40) | 18.60 cD (0.26) | 19.17 abC (0.29) | 19.43 aB (0.38) |
| 6 | 19.10 bB (0.10) | 18.10 cC (0.10) | 17.13 eE (0.15) | 17.73 dC (0.15) | 20.07 aB (0.06) | 18.23 cD (0.12) | 19.13 bBC (0.12) |
| 9 | 18.90 cBC (0.20) | 19.13 bcB (0.15) | 19.50 abB (0.17) | 17.93 dC (0.21) | 19.03 cC (0.06) | 19.90 aB (0.35) | 18.87 cC (0.35) |
| 12 | 18.97 aBC (0.23) | 17.13 cdD (0.47) | 13.63 bcD (0.31) | 16.97 dD (0.15) | 17.93 bE (0.35) | 17.90 bD (0.17) | – |
| 15 | 18.70 aC (0.30) | 17.50 bD (0.36) | 17.47 bD (0.15) | 16.83 cD (0.25) | 17.47 bF (0.21) | – | – |
| 18 | 17.53 aD (0.15) | 17.27 aD (0.55) | – | – | – | – | – |
| TA | |||||||
| 0 | 0.15 aD (0.01) | 0.15 aE (0.01) | 0.15 aE (0.01) | 0.15 aE (0.01) | 0.15 aC (0.01) | 0.15 aD (0.01) | 0.15 aB (0.01) |
| 3 | 0.16 bcC (0.01) | 0.16 bcD (0.01) | 0.17 abD (0.01) | 0.16 bcD (0.01) | 0.15 cC (0.01) | 0.17 abC (0.01) | 0.18 aA (0.01) |
| 6 | 0.19 aB (0.01) | 0.17 bD (0.01) | 0.19 aC (0.01) | 0.16 bD (0.01) | 0.18 aB (0.01) | 0.19 aB (0.01) | 0.18 aA (0.01) |
| 9 | 0.19 bB (0.01) | 0.19 bC (0.01) | 0.21 aB (0.01) | 0.18 bC (0.01) | 0.20 abB (0.01) | 0.20 abB (0.01) | 0.17 cA (0.01) |
| 12 | 0.20 bB (0.01) | 0.20 bBC (0.01) | 0.22 aAB (0.01) | 0.20 bB (0.01) | 0.22 aA (0.01) | 0.23 aA (0.01) | – |
| 15 | 0.23 abA (0.01) | 0.21 cAB (0.01) | 0.23 abA (0.01) | 0.22 bcA (0.01) | 0.24 aA (0.01) | – | – |
| 18 | 0.23 aA (0.01) | 0.22 aA (0.01) | – | – | – | – | – |
| pH | |||||||
| 0 | 6.88 aG (0.02) | 6.88 aE (0.02) | 6.88 aE (0.02) | 6.88 aE (0.02) | 6.88 aE (0.02) | 6.88 aD (0.02) | 6.88 aD (0.02) |
| 3 | 6.94 abcF (0.02) | 6.96 abcD (0.05) | 6.93 bcD (0.03) | 6.92 cE (0.05) | 6.98 aD (0.02) | 6.97 abC (0.02) | 6.99 aC (0.01) |
| 6 | 6.99 cE (0.01) | 6.99 cD (0.01) | 6.97 cC (0.02) | 6.98 cD (0.01) | 7.11 bC (0.02) | 7.11 bB (0.02) | 7.20 aB (0.02) |
| 9 | 7.13 cdD (0.04) | 7.18 bC (0.02) | 7.10 dB (0.01) | 7.14 cC (0.02) | 7.25 aB (0.02) | 7.10 cdB (0.01) | 7.29 aA (0.03) |
| 12 | 7.20 cC (0.01) | 7.26 bB (0.03) | 7.33 aA (0.03) | 7.21 cB (0.02) | 7.25 bcB (0.05) | 7.27 bA (0.02) | – |
| 15 | 7.31 bB (0.02) | 7.31 abA (0.03) | 7.32 abA (0.02) | 7.26 cA (0.03) | 7.35 aA (0.01) | – | – |
| 18 | 7.36 aA (0.03) | 7.31 bAB (0.02) | – | – | – | – | – |
| Wt. Loss | BI | DI | Texture | L* | a* | b* | TSS | TA | pH | TPC | TFC | PPO | POD | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Wt. loss | 1 | |||||||||||||
| BI | 0.87 | 1 | ||||||||||||
| DI | 0.68 | 0.94 | 1 | |||||||||||
| Texture | −0.47 | −0.79 | −0.90 | 1 | ||||||||||
| L* | −0.85 | −0.99 | −0.96 | 0.79 | 1 | |||||||||
| a* | 0.70 | 0.92 | 0.95 | −0.90 | −0.92 | 1 | ||||||||
| b* | −0.71 | −0.93 | −0.94 | 0.87 | 0.92 | −0.99 | 1 | |||||||
| TSS | −0.48 | −0.80 | −0.91 | 0.93 | 0.80 | −0.96 | 0.94 | 1 | ||||||
| TA | 0.31 | 0.70 | 0.83 | −0.91 | −0.69 | 0.89 | −0.89 | −0.97 | 1 | |||||
| pH | 0.63 | 0.89 | 0.92 | −0.91 | −0.87 | 0.98 | −0.98 | −0.96 | 0.93 | 1 | ||||
| TPC | −0.59 | −0.85 | −0.89 | 0.93 | 0.83 | −0.97 | 0.97 | 0.95 | −0.94 | −0.99 | 1 | |||
| TFC | −0.61 | −0.88 | −0.94 | 0.93 | 0.88 | −0.99 | 0.98 | 0.97 | −0.94 | −0.99 | 0.99 | 1 | ||
| PPO | 0.64 | 0.92 | 0.99 | −0.91 | −0.93 | 0.95 | −0.94 | −0.92 | 0.86 | 0.92 | −0.90 | −0.95 | 1 | |
| POD | 0.51 | 0.83 | 0.96 | −0.93 | −0.86 | 0.93 | −0.91 | −0.96 | 0.91 | 0.91 | −0.89 | −0.94 | 0.97 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.R.; Huang, C.; Ullah, R.; Ullah, H.; Qazi, I.M.; Nawaz, T.; Adnan, M.; Khan, A.; Su, H.; Ren, L. Effects of Various Polymeric Films on the Pericarp Microstructure and Storability of Longan (cv. Shixia) Fruit Treated with Propyl Disulfide Essential Oil from the Neem (Azadirachta indica) Plant. Polymers 2022, 14, 536. https://doi.org/10.3390/polym14030536
Khan MR, Huang C, Ullah R, Ullah H, Qazi IM, Nawaz T, Adnan M, Khan A, Su H, Ren L. Effects of Various Polymeric Films on the Pericarp Microstructure and Storability of Longan (cv. Shixia) Fruit Treated with Propyl Disulfide Essential Oil from the Neem (Azadirachta indica) Plant. Polymers. 2022; 14(3):536. https://doi.org/10.3390/polym14030536
Chicago/Turabian StyleKhan, Muhammad Rafiullah, Chongxing Huang, Rafi Ullah, Hakim Ullah, Ihsan Mabood Qazi, Taufiq Nawaz, Muhammad Adnan, Abdullah Khan, Hongxia Su, and Liu Ren. 2022. "Effects of Various Polymeric Films on the Pericarp Microstructure and Storability of Longan (cv. Shixia) Fruit Treated with Propyl Disulfide Essential Oil from the Neem (Azadirachta indica) Plant" Polymers 14, no. 3: 536. https://doi.org/10.3390/polym14030536
APA StyleKhan, M. R., Huang, C., Ullah, R., Ullah, H., Qazi, I. M., Nawaz, T., Adnan, M., Khan, A., Su, H., & Ren, L. (2022). Effects of Various Polymeric Films on the Pericarp Microstructure and Storability of Longan (cv. Shixia) Fruit Treated with Propyl Disulfide Essential Oil from the Neem (Azadirachta indica) Plant. Polymers, 14(3), 536. https://doi.org/10.3390/polym14030536









