A Review on Biomedical Application of Polysaccharide-Based Hydrogels with a Focus on Drug Delivery Systems
Abstract
1. Introduction
2. Materials and Methods
3. Technical Features of Polysaccharide-Based Hydrogels in Drug Delivery
3.1. Biochemical Characteristics of Polysaccharide-Based Hydrogels
3.2. Chemically and Physical Crosslinked Polysaccharide-Based Hydrogels
3.3. Swelling Properties of Polysaccharide-Based Hydrogels
3.4. The Elasticity of Polysaccharide-Based Hydrogels
3.5. Mechanical Properties of Polysaccharide-Based Hydrogels
3.6. Biocompatibility and Bioactivity of Polysaccharide-Based Hydrogels
3.7. Inhomogeneity of Polysaccharide-Based Hydrogels
3.8. Absorption under Load (AUL)
3.9. Molding Time
3.10. Self-Assembled Supramolecular Polysaccharide-Based Hydrogels
3.11. pH Sensitivity of Polysaccharide-Based Hydrogels
3.12. Polysaccharide-Based Hydrogels with Temperature-Sensitivity Feature
3.13. Affinity of Polysaccharide-Based Hydrogels
4. Hydrogel-Based Drug Delivery Systems
4.1. Polysaccharide-Based Hydrogels
4.2. Hydrogel-Based Controlled and Extended-Release Systems
4.3. Nano-Systems for Polysacharid-Based Hydrogels
4.4. Dual-DDS Based on Hydrogel/Micelle Composites
5. Biomedical Applications of Polysaccharide-Based Hydrogels
5.1. Topical Drug Administration
5.2. Ocular Drug Delivery
5.3. Hydrogels in Colonic Drug Delivery
5.4. Hydrogels for Sustained Delivery of Proteins and DNA
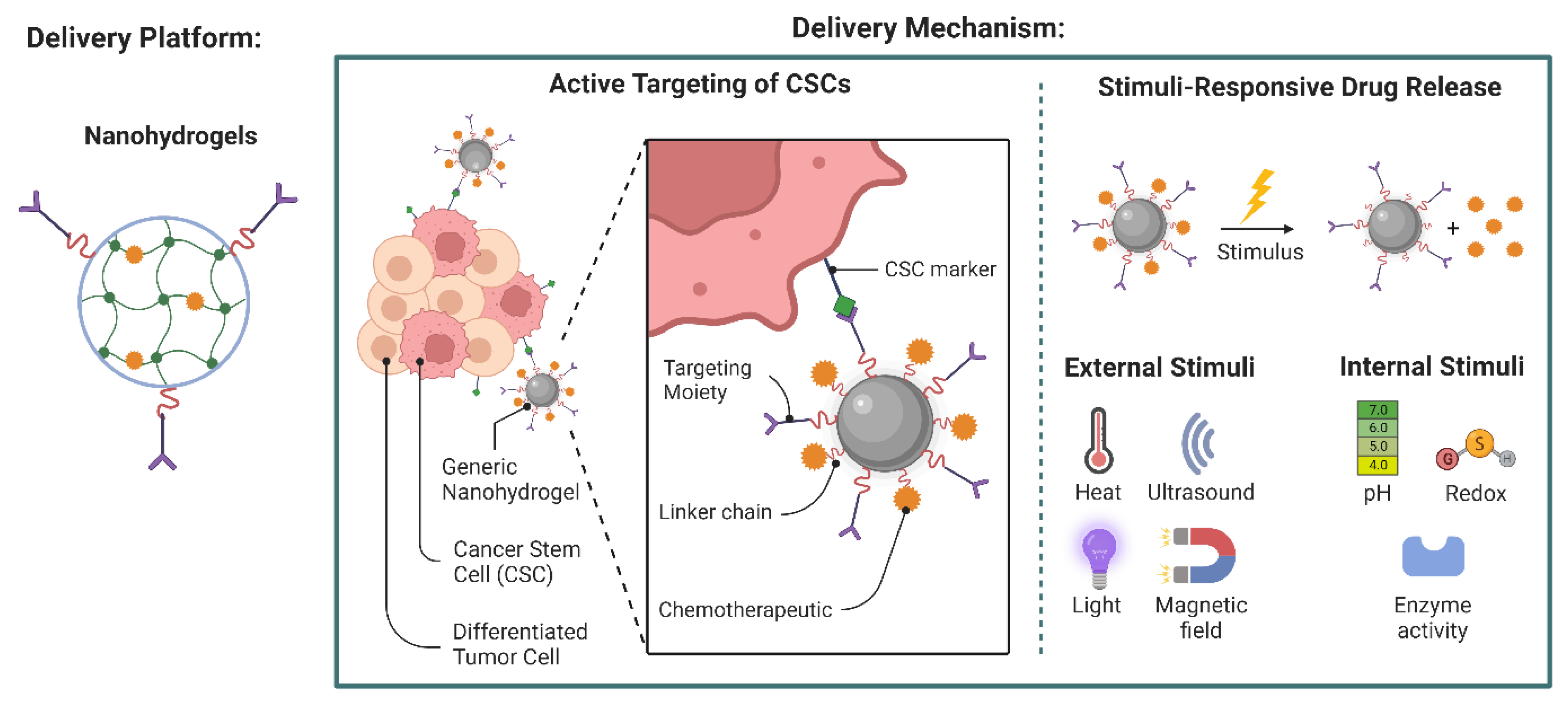
5.5. Hydrogels in Cancer Therapy
5.6. Applications of Hydrogels in Gynecology
5.7. Hydrogels for Buccal Delivery
6. Conclusions and Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patel, P.; Mandal, A.; Gote, V.; Pal, D.; Mitra, A.K. Thermosensitive hydrogel-based drug delivery system for sustained drug release. J. Polym. Res. 2019, 26, 131. [Google Scholar] [CrossRef]
- Heragh, B.K.; Javanshir, S.; Mahdavinia, G.R.; Jamal, M.R.N. Hydroxyapatite grafted chitosan/laponite RD hydrogel: Evaluation of the encapsulation capacity, pH-responsivity, and controlled release behavior. Int. J. Biol. Macromol. 2021, 190, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Vahed, T.A.; Naimi-Jamal, M.R.; Panahi, L. Alginate-coated ZIF-8 metal-organic framework as a green and bioactive platform for controlled drug release. J. Drug Deliv. Sci. Technol. 2019, 49, 570–576. [Google Scholar] [CrossRef]
- Gooneh-Farahani, S.; Naghib, S.M.; Naimi-Jamal, M.R.; Seyfoori, A. A pH-sensitive nanocarrier based on BSA-stabilized graphene-chitosan nanocomposite for sustained and prolonged release of anticancer agents. Sci. Rep. 2021, 11, 17404. [Google Scholar] [CrossRef]
- Sohrabi, M.; Yekta, B.E.; Rezaie, H.R.; Naimi-Jamal, M.R. Rheology, injectability, and bioactivity of bioactive glass containing chitosan/gelatin, nano pastes. J. Appl. Polym. Sci. 2020, 137, 49240. [Google Scholar] [CrossRef]
- Sohrabi, M.; Yekta, B.; Rezaie, H.; Naimi-Jamal, M.; Kumar, A.; Cochis, A.; Miola, M.; Rimondini, L. Enhancing Mechanical Properties and Biological Performances of Injectable Bioactive Glass by Gelatin and Chitosan for Bone Small Defect Repair. Biomedicines 2020, 8, 616. [Google Scholar] [CrossRef]
- Amirnejat, S.; Nosrati, A.; Javanshir, S.; Naimi-Jamal, M.R. Superparamagnetic alginate-based nanocomposite modified by L-arginine: An eco-friendly bifunctional catalysts and an efficient antibacterial agent. Int. J. Biol. Macromol. 2020, 152, 834–845. [Google Scholar] [CrossRef]
- Gu, X.; Xu, Y.; Li, S.; Wang, Z.; Meng, Q.; Yu, J. Preparation of a Photocured Biocompatible Hydrogel for Urethral Tissue Engineering. ACS Appl. Polym. Mater. 2021, 3, 3519–3527. [Google Scholar] [CrossRef]
- Dheer, D.; Arora, D.; Jaglan, S.; Rawal, R.K.; Shankar, R. Polysaccharides based nanomaterials for targeted anti-cancer drug delivery. J. Drug Target. 2017, 25, 1–16. [Google Scholar] [CrossRef]
- Bashir, S.; Hina, M.; Iqbal, J.; Rajpar, A.H.; Mujtaba, M.A.; Alghamdi, N.A.; Wageh, S.; Ramesh, K.; Ramesh, S. Fundamental Concepts of Hydrogels: Synthesis, Properties, and Their Applications. Polymers 2020, 12, 2702. [Google Scholar] [CrossRef]
- Chai, Q.; Jiao, Y.; Yu, X. Hydrogels for Biomedical Applications: Their Characteristics and the Mechanisms behind Them. Gels 2017, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Gooneh-Farahani, S.; Naimi-Jamal, M.R.; Naghib, S.M. Stimuli-responsive graphene-incorporated multifunctional chitosan for drug delivery applications: A review. Expert Opin. Drug Deliv. 2019, 16, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Tiwari, S. A review on biomacromolecular hydrogel classification and its applications. Int. J. Biol. Macromol. 2020, 162, 737–747. [Google Scholar] [CrossRef]
- Ansuja Pulickal Mathew, S.U.; Cho, K.Y.; Cho, C.S.; Park, I.K. Injectable hydrogels for delivering biotherapeutic molecules. Int. J. Bio. Macromol. 2018, 110, 7–29. [Google Scholar]
- Kai Wang, Z.H. Injectable hydrogels for ophthalmic applications. J. Control. Release 2017, 268, 212–224. [Google Scholar] [CrossRef]
- Ravani, L.; Esposito, E.; Bories, C.; Moal, V.L.-L.; Loiseau, P.M.; Djabourov, M.; Cortesi, R.; Bouchemal, K. Clotrimazole-loaded nanostructured lipid carrier hydrogels: Thermal analysis and in vitro studies. Int. J. Pharm. 2013, 454, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Pushpamalar, J.; Meganathan, P.; Tan, H.L.; Dahlan, N.A.; Ooi, L.-T.; Neerooa, B.N.H.M.; Essa, R.Z.; Shameli, K.; Teow, S.-Y. Development of a Polysaccharide-Based Hydrogel Drug Delivery System (DDS): An Update. Gels 2021, 7, 153. [Google Scholar] [CrossRef]
- Rostami, J.; Gordeyeva, K.; Benselfelt, T.; Lahchaichi, E.; Hall, S.A.; Riazanova, A.V.; Larsson, P.A.; Ciftci, G.C.; Wågberg, L. Hierarchical build-up of bio-based nanofibrous materials with tunable metal–organic framework biofunctionality. Mater. Today 2021, 48, 47–58. [Google Scholar] [CrossRef]
- Bastola, A.K.; Rodriguez, N.; Behl, M.; Soffiatti, P.; Rowe, N.P.; Lendlein, A. Cactus-inspired design principles for soft robotics based on 3D printed hydrogel-elastomer systems. Mater. Des. 2021, 202, 109515. [Google Scholar] [CrossRef]
- Varghese, S.A.; Rangappa, S.M.; Siengchin, S.; Parameswaranpillai, J. Natural polymers and the hydrogels prepared from them. In Hydrogels Based on Natural Polymers; Elsevier: Amsterdam, The Netherlands, 2020; pp. 17–47. [Google Scholar]
- Ahmad, Z.; Salman, S.; Khan, S.A.; Amin, A.; Rahman, Z.U.; Al-Ghamdi, Y.O.; Akhtar, K.; Bakhsh, E.M.; Khan, S.B. Versatility of Hydrogels: From Synthetic Strategies, Classification, and Properties to Biomedical Applications. Gels 2022, 8, 167. [Google Scholar] [CrossRef]
- Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity. Signal Transduct. Target. Ther. 2021, 6, 426. [Google Scholar] [CrossRef] [PubMed]
- Parhi, A. Cross-Linked Hydrogel for Pharmaceutical Applications: A Review. Adv. Pharm. Bull. 2017, 7, 515–530. [Google Scholar] [CrossRef]
- Mayorova, O.A.; Jolly, B.C.; Verkhovskii, R.A.; Plastun, V.O.; Sindeeva, O.A.; Douglas, T.E. pH-Sensitive dairy-derived hydrogels with a prolonged drug release profile for cancer treatment. Materials 2021, 14, 749. [Google Scholar] [CrossRef] [PubMed]
- Suflet, D.M.; Popescu, I.; Pelin, I.M.; Ichim, D.L.; Daraba, O.M.; Constantin, M.; Fundueanu, G. Dual Cross-Linked Chitosan/PVA Hydrogels Containing Silver Nanoparticles with Antimicrobial Properties. Pharmaceutics 2021, 13, 1461. [Google Scholar] [CrossRef] [PubMed]
- Hamdy, D.M.; Hassabo, A.G. Ph and temperature thermosensitive for modification of cotton fabric (A review). Biointerface Res. Appl. Chem. 2022, 12, 2216–2228. [Google Scholar]
- Das, S.K.; Parandhaman, T.; Dey, M.D. Biomolecule-assisted synthesis of biomimetic nanocomposite hydrogel for hemostatic and wound healing applications. Green Chem. 2021, 23, 629–669. [Google Scholar] [CrossRef]
- Qian, C.; Zhang, T.; Gravesande, J.; Baysah, C.; Song, X.; Xing, J. Injectable and self-healing polysaccharide-based hydrogel for pH-responsive drug release. Int. J. Biol. Macromol. 2019, 123, 140–148. [Google Scholar] [CrossRef]
- Niemczyk-Soczynska, B.; Zaszczyńska, A.; Zabielski, K.; Sajkiewicz, P. Hydrogel, electrospun and composite materials for bone/cartilage and neural tissue engineering. Materials 2021, 14, 6899. [Google Scholar] [CrossRef]
- Singh, B.; Sharma, S.; Dhiman, A. Acacia gum polysaccharide based hydrogel wound dressings: Synthesis, characterization, drug delivery and biomedical properties. Carbohydr. Polym. 2017, 165, 294–303. [Google Scholar] [CrossRef]
- Weng, T.; Zhang, W.; Xia, Y.; Wu, P.; Yang, M.; Jin, R.; Xia, S.; Wang, J.; You, C.; Han, C.; et al. 3D bioprinting for skin tissue engineering: Current status and perspectives. J. Tissue Eng. 2021, 12, 20417314211028574. [Google Scholar] [CrossRef]
- Ali, A.; Hussain, M.A.; Haseeb, M.T.; Bukhari, S.N.A.; Tabassum, T.; Farid-Ul-Haq, M.; Sheikh, F.A. A pH-responsive, biocompatible, and non-toxic citric acid cross-linked polysaccharide-based hydrogel from Salvia spinosa L. offering zero-order drug release. J. Drug Deliv. Sci. Technol. 2022, 69, 103144. [Google Scholar] [CrossRef]
- Cao, J.; Wu, P.; Cheng, Q.; He, C.; Chen, Y.; Zhou, J. Ultrafast Fabrication of Self-Healing and Injectable Carboxymethyl Chitosan Hydrogel Dressing for Wound Healing. ACS Appl. Mater. Interfaces 2021, 13, 24095–24105. [Google Scholar] [CrossRef] [PubMed]
- Kopač, T.; Abrami, M.; Grassi, M.; Ručigaj, A.; Krajnc, M. Polysaccharide-based hydrogels crosslink density equation: A rheological and LF-NMR study of polymer-polymer interactions. Carbohydr. Polym. 2021, 277, 118895. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Kim, D.H.; Lee, Y.S. The influence of monomer composition and surface-crosslinking condition on biodegradation and gel strength of super absorbent polymer. Polymers 2021, 13, 663. [Google Scholar] [CrossRef] [PubMed]
- Zakerikhoob, M.; Abbasi, S.; Yousefi, G.; Mokhtari, M.; Noorbakhsh, M.S. Curcumin-incorporated crosslinked sodium alginate-g-poly (N-isopropyl acrylamide) thermo-responsive hydrogel as an in-situ forming injectable dressing for wound healing: In vitro characterization and in vivo evaluation. Carbohydr. Polym. 2021, 271, 118434. [Google Scholar] [CrossRef]
- Hoque, J.; Sangaj, N.; Varghese, S. Stimuli-Responsive Supramolecular Hydrogels and Their Applications in Regenerative Medicine. Macromol. Biosci. 2019, 19, 1800259. [Google Scholar] [CrossRef]
- Naghieh, S.; Chen, X. Printability–A key issue in extrusion-based bioprinting. J. Pharm. Anal. 2021, 11, 564–579. [Google Scholar] [CrossRef]
- Zhuo, S.; Zhang, F.; Yu, J.; Zhang, X.; Yang, G.; Liu, X. pH-Sensitive Biomaterials for Drug Delivery. Molecules 2020, 25, 5649. [Google Scholar] [CrossRef]
- Rizwan, M.; Yahya, R.; Hassan, A.; Yar, M.; Azzahari, A.D.; Selvanathan, V.; Sonsudin, F.; Abouloula, C.N. PH Sensitive Hydrogels in Drug Delivery: Brief History, Properties, Swelling, and Release Mechanism, Material Selection and Applications. Polymers 2017, 9, 137. [Google Scholar] [CrossRef]
- Huang, H.; Qi, X.; Chen, Y.; Wu, Z. Thermo-sensitive hydrogels for delivering biotherapeutic molecules: A review. Saudi Pharm. J. 2019, 27, 990–999. [Google Scholar] [CrossRef]
- Saha, S.; Banskota, S.; Roberts, S.; Kirmani, N.; Chilkoti, A. Engineering the Architecture of Elastin-Like Polypeptides: From Unimers to Hierarchical Self-Assembly. Adv. Ther. 2020, 3, 1900164. [Google Scholar] [CrossRef] [PubMed]
- Abune, L.; Wang, Y. Affinity Hydrogels for Protein Delivery. Trends Pharmacol. Sci. 2021, 42, 300–312. [Google Scholar] [CrossRef] [PubMed]
- Rial-Hermida, M.I.; Rey-Rico, A.; Blanco-Fernandez, B.; Carballo-Pedrares, N.; Byrne, E.M.; Mano, J.F. Recent Progress on Polysaccharide-Based Hydrogels for Controlled Delivery of Therapeutic Biomolecules. ACS Biomater. Sci. Eng. 2021, 7, 4102–4127. [Google Scholar] [CrossRef] [PubMed]
- Bordbar-Khiabani, A.; Gasik, M. Smart Hydrogels for Advanced Drug Delivery Systems. Int. J. Mol. Sci. 2022, 23, 3665. [Google Scholar] [CrossRef]
- Zhang, J.; Li, S.; Yao, L.; Yi, Y.; Shen, L.; Li, Z.; Qiu, H. Responsive switchable deep eutectic solvents: A review. Chin. Chem. Lett. 2022, 107750. [Google Scholar] [CrossRef]
- Bian, Q.; Fu, L.; Li, H. Engineering shape memory and morphing protein hydrogels based on protein unfolding and folding. Nat. Commun. 2022, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Balan, K.E.; Boztepe, C.; Künkül, A. Modeling the effect of physical crosslinking degree of pH and temperature responsive poly(NIPAAm-co-VSA)/alginate IPN hydrogels on drug release behavior. J. Drug Deliv. Sci. Technol. 2022, 75, 103671. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, Y.; Yang, G.; Kong, H.; Guo, L.; Wei, G. Recent advances in protein hydrogels: From design, structural and functional regulations to healthcare applications. Chem. Eng. J. 2022, 451, 138494. [Google Scholar] [CrossRef]
- Saeedi, M.; Vahidi, O.; Moghbeli, M.R.; Ahmadi, S.; Asadnia, M.; Akhavan, O.; Seidi, F.; Rabiee, M.; Saeb, M.R.; Webster, T.J.; et al. Customizing nano-chitosan for sustainable drug delivery. J. Control. Release 2022, 350, 175–192. [Google Scholar] [CrossRef]
- Yu, A.; Hu, Y.; Ma, X.; Mo, L.; Pan, M.; Bi, X.; Wu, Y.; Wang, J.; Li, X. Sequential drug release of co-assembled supramolecular hydrogel as synergistic therapy against Staphylococcus aureus endophthalmitis. Chem. Eng. J. 2021, 427, 130979. [Google Scholar] [CrossRef]
- Danielsen, S.P.O.; Beech, H.K.; Wang, S.; El-Zaatari, B.M.; Wang, X.; Sapir, L.; Ouchi, T.; Wang, Z.; Johnson, P.N.; Hu, Y.; et al. Molecular Characterization of Polymer Networks. Chem. Rev. 2021, 121, 5042–5092. [Google Scholar] [CrossRef] [PubMed]
- Ghaedamini, H.; Amiri, M. Effects of temperature and surfactant concentration on the structure and morphology of calcium carbonate nanoparticles synthesized in a colloidal gas aphrons system. J. Mol. Liq. 2019, 282, 213–220. [Google Scholar] [CrossRef]
- Fang, K.; Zhang, Y.; Yin, J.; Yang, T.; Li, K.; Wei, L.; Li, J.; He, W. Hydrogel beads based on carboxymethyl cassava starch/alginate enriched with MgFe2O4 nanoparticles for controlling drug release. Int. J. Biol. Macromol. 2022, 220, 573–588. [Google Scholar] [CrossRef] [PubMed]
- Andrade, F.; Roca-Melendres, M.M.; Llaguno, M.; Hide, D.; Raurell, I.; Martell, M.; Vijayakumar, S.; Oliva, M.; Schwartz, S.; Durán-Lara, E.F.; et al. Smart and eco-friendly N-isopropylacrylamide and cellulose hydrogels as a safe dual-drug local cancer therapy approach. Carbohydr. Polym. 2022, 295, 119589. [Google Scholar] [CrossRef] [PubMed]
- Suhail, M.; Shih, C.-M.; Liu, J.-Y.; Hsieh, W.-C.; Lin, Y.-W.; Lin, I.-L.; Wu, P.-C. Synthesis of glutamic acid/polyvinyl alcohol based hydrogels for controlled drug release: In-vitro characterization and in-vivo evaluation. J. Drug Deliv. Sci. Technol. 2022, 75, 103715. [Google Scholar] [CrossRef]
- Corredor-Chaparro, M.Y.; Vargas-Riveros, D.; Mora-Huertas, C.E. Hypromellose–Collagen hydrogels/sesame oil organogel based bigels as controlled drug delivery systems. J. Drug Deliv. Sci. Technol. 2022, 75, 103637. [Google Scholar] [CrossRef]
- Balasubramanian, R.; Kim, S.S.; Lee, J. Novel synergistic transparent k-Carrageenan/Xanthan gum/Gellan gum hydrogel film: Mechanical, thermal and water barrier properties. Int. J. Biol. Macromol. 2018, 118, 561–568. [Google Scholar] [CrossRef]
- Zheng, W.; Zhang, H.; Wang, J.; Wang, J.; Yan, L.; Liu, C.; Zheng, L. Pickering emulsion hydrogel based on alginate-gellan gum with carboxymethyl chitosan as a pH-responsive controlled release delivery system. Int. J. Biol. Macromol. 2022, 216, 850–859. [Google Scholar] [CrossRef]
- Chyzy, A.; Tomczykowa, M.; Plonska-Brzezinska, M.E. Hydrogels as potential nano-, micro-and macro-scale systems for controlled drug delivery. Materials 2020, 13, 188. [Google Scholar] [CrossRef]
- Gupta, M.K.; Bajpai, J.; Bajpai, A.K. Inverse emulsion polymerization-assisted designing of superparamagnetic poly (2-hydroxyethyl methacrylate) nanoparticles and magnetically triggered release of cisplatin. Polym. Eng. Sci. 2021, 61, 1427–1439. [Google Scholar] [CrossRef]
- Li, W.; Wang, C.; Wang, Z.; Gou, L.; Zhou, Y.; Peng, G.; Zhu, M.; Zhang, J.; Li, R.; Ni, H.; et al. Physically Cross-Linked DNA Hydrogel-Based Sustained Cytokine Delivery for In Situ Diabetic Alveolar Bone Rebuilding. ACS Appl. Mater. Interfaces 2022, 14, 25173–25182. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Huang, W.; Shi, C.; Atkinson, S.T.; Luo, J. Reversibly crosslinked nanocarriers for on-demand drug delivery in cancer treatment. Ther. Deliv. 2012, 3, 1409–1427. [Google Scholar] [CrossRef]
- Shah, S.A.; Sohail, M.; Khan, S.A.; Kousar, M. Improved drug delivery and accelerated diabetic wound healing by chondroitin sulfate grafted alginate-based thermoreversible hydrogels. Mater. Sci. Eng. C 2021, 126, 112169. [Google Scholar] [CrossRef] [PubMed]
- Das, R.; Vecitis, C.D.; Schulze, A.; Cao, B.; Ismail, A.F.; Lu, X.; Chen, J.; Ramakrishna, S. Recent advances in nanomaterials for water protection and monitoring. Chem. Soc. Rev. 2017, 46, 6946–7020. [Google Scholar] [CrossRef] [PubMed]
- Andrade, F.; Roca-Melendres, M.M.; Durán-Lara, E.F.; Rafael, D.; Schwartz, S., Jr. Stimuli-responsive hydrogels for cancer treatment: The role of pH, light, ionic strength and magnetic field. Cancers 2021, 13, 1164. [Google Scholar] [CrossRef]
- Ko, J.; Park, K.; Kim, Y.-S.; Kim, M.S.; Han, J.K.; Kim, K.; Park, R.-W.; Song, H.K.; Lee, D.S.; Kwon, I.C. Tumoral acidic extracellular pH targeting of pH-responsive MPEG-poly(β-amino ester) block copolymer micelles for cancer therapy. J. Control. Release 2007, 123, 109–115. [Google Scholar] [CrossRef]
- Lee, B.-J.; Ryu, S.-G.; Cui, J.-H. Controlled release of dual drug-loaded hydroxypropyl methylcellulose matrix tablet using drug-containing polymeric coatings. Int. J. Pharm. 1999, 188, 71–80. [Google Scholar] [CrossRef]
- Alibak, A.H.; Khodarahmi, M.; Fayyazsanavi, P.; Alizadeh, S.M.; Hadi, A.J.; Aminzadehsarikhanbeglou, E. Simulation the adsorption capacity of polyvinyl alcohol/carboxymethyl cellulose based hydrogels towards methylene blue in aqueous solutions using cascade correlation neural network (CCNN) technique. J. Clean. Prod. 2022, 337, 130509. [Google Scholar] [CrossRef]
- Wei, L.; Cai, C.; Lin, J.; Chen, T. Dual-drug delivery system based on hydrogel/micelle composites. Biomaterials 2009, 30, 2606–2613. [Google Scholar] [CrossRef]
- Cong, Z.; Shi, Y.; Wang, Y.; Wang, Y.; Niu, J.; Chen, N.; Xue, H. A novel controlled drug delivery system based on alginate hydrogel/chitosan micelle composites. Int. J. Biol. Macromol. 2018, 107, 855–864. [Google Scholar] [CrossRef]
- Ma, D.; Zhang, H.-B.; Tu, K.; Zhang, L.-M. Novel supramolecular hydrogel/micelle composite for co-delivery of anticancer drug and growth factor. Soft Matter 2012, 8, 3665–3672. [Google Scholar] [CrossRef]
- Anirudhan, T.; Parvathy, J.; Nair, A.S. A novel composite matrix based on polymeric micelle and hydrogel as a drug carrier for the controlled release of dual drugs. Carbohydr. Polym. 2016, 136, 1118–1127. [Google Scholar] [CrossRef] [PubMed]
- Pourmanouchehri, Z.; Ebrahimi, S.; Limoee, M.; Jalilian, F.; Janfaza, S.; Vosoughi, A.; Behbood, L. Controlled release of 5-fluorouracil to melanoma cells using a hydrogel/micelle composites based on deoxycholic acid and carboxymethyl chitosan. Int. J. Biol. Macromol. 2022, 206, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Khorsandi, Z.; Afshinpour, M.; Molaei, F.; Askandar, R.H.; Keshavarzipour, F.; Abbasi, M.; Sadeghi-Aliabadi, H. Design and synthesis of novel phe-phe hydroxyethylene derivatives as potential coronavirus main protease inhibitors. J. Biomol. Struct. Dyn. 2021, 40, 7940–7948. [Google Scholar] [CrossRef]
- Anirudhan, T.; Suriya, R.; Anoop, S. Polymeric micelle/nano hydrogel composite matrix as a novel multi-drug carrier. J. Mol. Struct. 2022, 1264, 133265. [Google Scholar] [CrossRef]
- Lv, J.; Wu, G.; Liu, Y.; Li, C.; Huang, F.; Zhang, Y.; Liu, J.; An, Y.; Ma, R.; Shi, L. Injectable dual glucose-responsive hydrogel-micelle composite for mimicking physiological basal and prandial insulin delivery. Sci. China Ser. B Chem. 2019, 62, 637–648. [Google Scholar] [CrossRef]
- Kazemi, M.; Emami, J.; Hasanzadeh, F.; Minaiyan, M.; Mirian, M.; Lavasanifar, A.; Mokhtari, M. In Vitro and In Vivo Evaluation of Novel DTX-Loaded Multifunctional Heparin-Based Polymeric Micelles Targeting Folate Receptors and Endosomes. Recent Pat. Anti-Cancer Drug Discov. 2020, 15, 341–359. [Google Scholar] [CrossRef]
- dos Santos, A.M.; Carvalho, S.G.; Araujo, V.H.S.; Carvalho, G.C.; Gremião, M.P.D.; Chorilli, M. Recent advances in hydrogels as strategy for drug delivery intended to vaginal infections. Int. J. Pharm. 2020, 590, 119867. [Google Scholar] [CrossRef]
- Faress, F.; Yari, A.; Kouchi, F.R.; Nezhad, A.S.; Hadizadeh, A.; Bakhtiar, L.S.; Naserzadeh, Y.; Mahmoudi, N. Developing an accurate empirical correlation for predicting anti-cancer drugs’ dissolution in supercritical carbon dioxide. Sci. Rep. 2022, 12, 1–17. [Google Scholar] [CrossRef]
- Shoukat, H.; Buksh, K.; Noreen, S.; Pervaiz, F.; Maqbool, I. Hydrogels as potential drug-delivery systems: Network design and applications. Ther. Deliv. 2021, 12, 375–396. [Google Scholar] [CrossRef]
- Kumar, S.; Prasad, M.; Rao, R. Topical delivery of clobetasol propionate loaded nanosponge hydrogel for effective treatment of psoriasis: Formulation, physicochemical characterization, antipsoriatic potential and biochemical estimation. Mater. Sci. Eng. C 2020, 119, 111605. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Ullah, A.; Xu, G.; Xu, Z.; Wang, F.; Liu, T.; Su, Y.; Zhang, T.; Wang, K. Topically applied liposome-in-hydrogels for systematically targeted tumor photothermal therapy. Drug Deliv. 2021, 28, 1923–1931. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, M.-Y.; Lin, A.-C.; Liao, W.-H.; Wang, T.-G.; Hsu, C.-H.; Chen, W.-S.; Lin, F.-H. Drug-loaded hyaluronic acid hydrogel as a sustained-release regimen with dual effects in early intervention of tendinopathy. Sci. Rep. 2019, 9, 4784. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Vogus, D.; Zhao, Z.; He, W.; Krishnan, V.; Kim, J.; Shi, Y.; Sarode, A.; Ukidve, A.; Mitragotri, S. Injectable hyaluronic acid hydrogels encapsulating drug nanocrystals for long-term treatment of inflammatory arthritis. Bioeng. Transl. Med. 2022, 7, e10245. [Google Scholar] [CrossRef]
- Li, Z.; Cheng, H.; Ke, L.; Liu, M.; Wang, C.; Loh, X.J.; Li, Z.; Wu, Y. Recent Advances in New Copolymer Hydrogel-Formed Contact Lenses for Ophthalmic Drug Delivery. ChemNanoMat 2021, 7, 564–579. [Google Scholar] [CrossRef]
- Ilochonwu, B.C.; Mihajlovic, M.; Maas-Bakker, R.F.; Rousou, C.; Tang, M.; Chen, M.; Hennink, W.E.; Vermonden, T. Hyaluronic Acid-PEG-Based Diels–Alder In Situ Forming Hydrogels for Sustained Intraocular Delivery of Bevacizumab. Biomacromolecules 2022, 23, 2914–2929. [Google Scholar] [CrossRef]
- Das, S.; Subuddhi, U. Potential of guar gum hydrogels in drug delivery. In Plant and Algal Hydrogels for Drug Delivery and Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2021; pp. 143–180. [Google Scholar]
- Koev, T.T.; Harris, H.C.; Kiamehr, S.; Khimyak, Y.Z.; Warren, F.J. Starch hydrogels as targeted colonic drug delivery vehicles. Carbohydr. Polym. 2022, 289, 119413. [Google Scholar] [CrossRef]
- Suhail, M.; Shao, Y.-F.; Vu, Q.L.; Wu, P.-C. Designing of pH-Sensitive Hydrogels for Colon Targeted Drug Delivery; Characterization and In Vitro Evaluation. Gels 2022, 8, 155. [Google Scholar] [CrossRef]
- Jo, Y.-J.; Gulfam, M.; Jo, S.-H.; Gal, Y.-S.; Oh, C.-W.; Park, S.-H.; Lim, K.T. Multi-stimuli responsive hydrogels derived from hyaluronic acid for cancer therapy application. Carbohydr. Polym. 2022, 286, 119303. [Google Scholar] [CrossRef]
- Osouli-Bostanabad, K.; Masalehdan, T.; Kapsa, R.M.I.; Quigley, A.; Lalatsa, A.; Bruggeman, K.F.; Franks, S.J.; Williams, R.J.; Nisbet, D.R. Traction of 3D and 4D Printing in the Healthcare Industry: From Drug Delivery and Analysis to Regenerative Medicine. ACS Biomater. Sci. Eng. 2022, 8, 2764–2797. [Google Scholar] [CrossRef]
- Dedeloudi, A.; Siamidi, A.; Pavlou, P.; Vlachou, M. Recent Advances in the Excipients Used in Modified Release Vaginal Formulations. Materials 2022, 15, 327. [Google Scholar] [CrossRef] [PubMed]
- Buckenmeyer, M.J.; Sukhwani, M.; Iftikhar, A.; Nolfi, A.L.; Xian, Z.; Dadi, S.; Case, Z.W.; Steimer, S.R.; D’Amore, A.; Orwig, K.E.; et al. Bioengineering an in situ ovary (ISO) for fertility preservation. bioRxiv 2020. bioRxiv:2020.2001.2003.893941. [Google Scholar] [CrossRef]
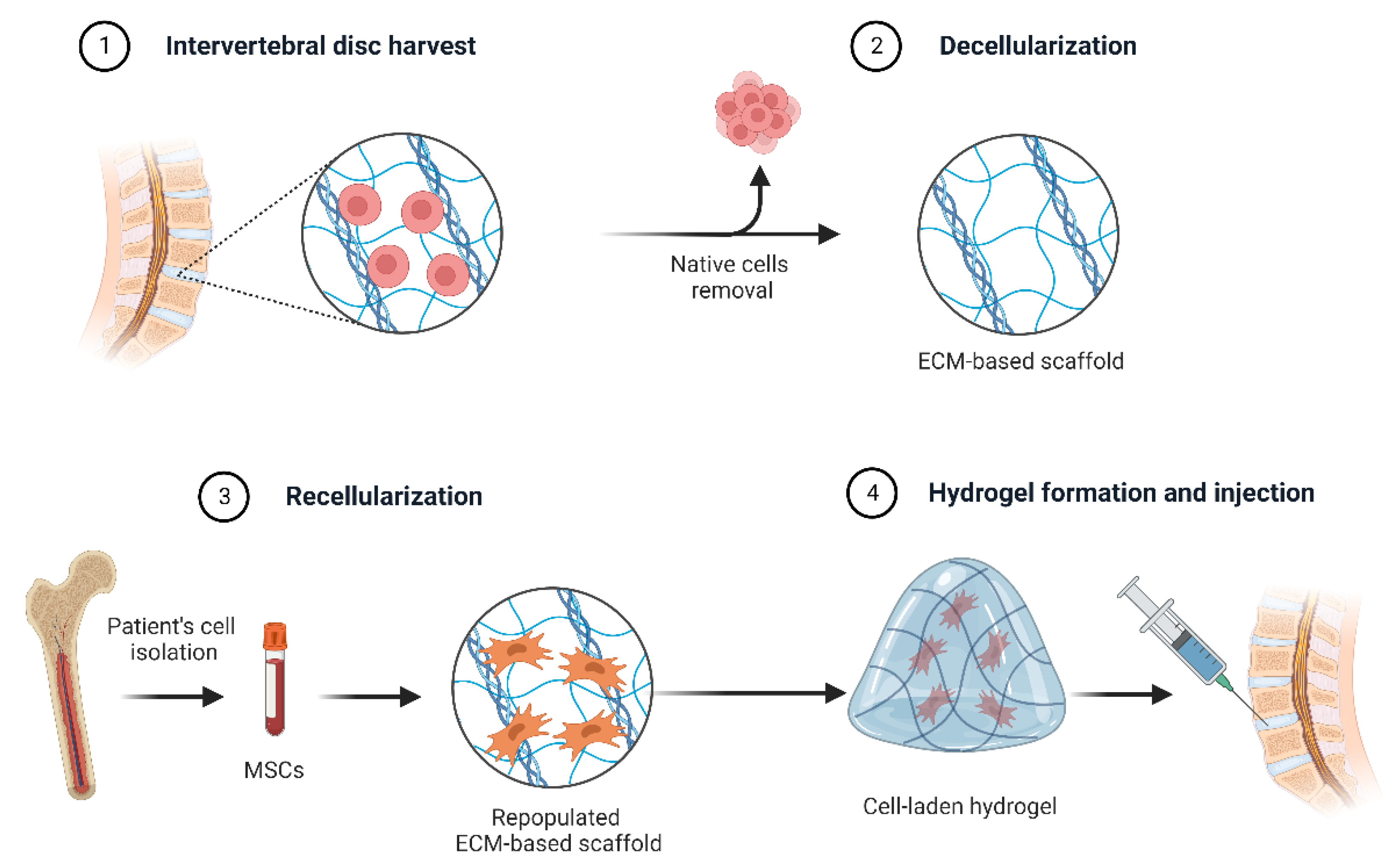
- Francés-Herrero, E.; Rodríguez-Eguren, A.; Gómez-Álvarez, M.; de Miguel-Gómez, L.; Ferrero, H.; Cervelló, I. Future Challenges and Opportunities of Extracellular Matrix Hydrogels in Female Reproductive Medicine. Int. J. Mol. Sci. 2022, 23, 3765. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.; Zheng, J.; Li, Z.; Qi, X.; Tian, Y.; Zhang, M.; Zhang, J.; Huang, X. Printing 3D vagina tissue analogues with vagina decellularized extracellular matrix bioink. Int. J. Biol. Macromol. 2021, 180, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Cascone, S.; Lamberti, G. Hydrogel-based commercial products for biomedical applications: A review. Int. J. Pharm. 2020, 573, 118803. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.bandagesplus.com/wound-care/wound-dressings/hydrogel-dressings/suprasorb-g (accessed on 1 November 2022).
- Available online: http://dermarite.com/product/aquaderm/ (accessed on 1 November 2022).
- Available online: http://www.smith-nephew.com/professional/products/advanced-wound-management/other-wound-care-products/solosite-gel/ (accessed on 1 November 2022).
- Available online: https://www.moirabeauty.com/products/rose-soothing-hydrogel-mask (accessed on 1 November 2022).
- Available online: https://www.lancome-usa.com/skin-care/masks/advanced-genifique-hydrogel-melting-sheet-mask/LAN192.html (accessed on 1 November 2022).
- Chen, H.; Cheng, R.; Zhao, X.; Zhang, Y.; Tam, A.; Yan, Y.; Shen, H.; Zhang, Y.S.; Qi, J.; Feng, Y.; et al. An injectable self-healing coordinative hydrogel with antibacterial and angiogenic properties for diabetic skin wound repair. NPG Asia Mater. 2019, 11, 3. [Google Scholar] [CrossRef]
- Available online: https://www.maxvuevision.com/index.php/products/clear-lens/airsoft (accessed on 1 November 2022).
- Nedunchezian, S.; Banerjee, P.; Lee, C.-Y.; Lee, S.-S.; Lin, C.-W.; Wu, C.-W.; Wu, S.-C.; Chang, J.-K.; Wang, C.-K. Generating adipose stem cell-laden hyaluronic acid-based scaffolds using 3D bioprinting via the double crosslinked strategy for chondrogenesis. Mater. Sci. Eng. C 2021, 124, 112072. [Google Scholar] [CrossRef]
- Hauptstein, J.; Forster, L.; Nadernezhad, A.; Groll, J.; Teßmar, J.; Blunk, T. Tethered TGF-β1 in a Hyaluronic Acid-Based Bioink for Bioprinting Cartilaginous Tissues. Int. J. Mol. Sci. 2022, 23, 924. [Google Scholar] [CrossRef]
- Tsanaktsidou, E.; Kammona, O.; Kiparissides, C. Recent Developments in Hyaluronic Acid-Based Hydrogels for Cartilage Tissue Engineering Applications. Polymers 2022, 14, 839. [Google Scholar] [CrossRef]
- Später, T.; Mariyanats, A.O.; Syachina, M.A.; Mironov, A.V.; Savelyev, A.G.; Sochilina, A.V.; Menger, M.D.; Vishnyakova, P.A.; Kananykhina, E.Y.; Fatkhudinov, T.K.; et al. In Vitro and in Vivo Analysis of Adhesive, Anti-Inflammatory, and Proangiogenic Properties of Novel 3D Printed Hyaluronic Acid Glycidyl Methacrylate Hydrogel Scaffolds for Tissue Engineering. ACS Biomater. Sci. Eng. 2020, 6, 5744–5757. [Google Scholar] [CrossRef]
- Ngo, T.B.; Spearman, B.S.; Hlavac, N.; Schmidt, C.E. Three-Dimensional Bioprinted Hyaluronic Acid Hydrogel Test Beds for Assessing Neural Cell Responses to Competitive Growth Stimuli. ACS Biomater. Sci. Eng. 2020, 6, 6819–6830. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.-W.; Zhang, X.-W.; Mi, C.-H.; Qi, X.-Y.; Zhou, J.; Wei, D.-X. Recent advances in hyaluronic acid-based hydrogels for 3D bioprinting in tissue engineering applications. Smart Mater. Med. 2022, 4, 59–68. [Google Scholar] [CrossRef]
- Duan, B.; Hockaday, L.A.; Kapetanovic, E.; Kang, K.H.; Butcher, J.T. Stiffness and adhesivity control aortic valve interstitial cell behavior within hyaluronic acid based hydrogels. Acta Biomater. 2013, 9, 7640–7650. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, N.S.; Chauhan, G.; Goyal, M.; Sarvepalli, S.; Gupta, V. Development of gelatin methacrylate (GelMa) hydrogels for versatile intracavitary applications. Biomater. Sci. 2022, 10, 4492–4507. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Lin, Z.; Yang, Y.; Jiang, T.; Shang, J.; Luo, Z. Biocompatible Conductive Hydrogels: Applications in the Field of Biomedicine. Int. J. Mol. Sci. 2022, 23, 4578. [Google Scholar] [CrossRef]
- Tan, R.Y.H.; Lee, C.S.; Pichika, M.R.; Cheng, S.F.; Lam, K.Y. PH Responsive Polyurethane for the Advancement of Biomedical and Drug Delivery. Polymers 2022, 14, 1672. [Google Scholar] [CrossRef]
- Tong, M.-Q.; Luo, L.-Z.; Xue, P.-P.; Han, Y.-H.; Wang, L.-F.; Zhuge, D.-L.; Yao, Q.; Chen, B.; Zhao, Y.-Z.; Xu, H.-L. Glucose-responsive hydrogel enhances the preventive effect of insulin and liraglutide on diabetic nephropathy of rats. Acta Biomater. 2021, 122, 111–132. [Google Scholar] [CrossRef]
- Hamcerencu, M.; Desbrieres, J.; Popa, M.; Riess, G. Thermo-sensitive gellan maleate/N-isopropylacrylamide hydrogels: Initial “in vitro” and “in vivo” evaluation as ocular inserts. Polym. Bull. 2020, 77, 741–755. [Google Scholar] [CrossRef]
- El-Husseiny, H.M.; Mady, E.A.; Hamabe, L.; Abugomaa, A.; Shimada, K.; Yoshida, T.; Tanaka, T.; Yokoi, A.; Elbadawy, M.; Tanaka, R. Smart/stimuli-responsive hydrogels: Cutting-edge platforms for tissue engineering and other biomedical applications. Mater. Today Bio. 2022, 13, 100186. [Google Scholar] [CrossRef]
- Silva, E.; Babo, P.; Costa-Almeida, R.; Domingues, R.; Mendes, B.; Paz, E.; Freitas, P.; Rodrigues, M.; Granja, P.; Gomes, M. P037 Multifunctional magnetic-responsive hydrogels modulate platelet lysate-derived growth factor release and guide cell fate. In Proceedings of the TERMIS-EU 2017, Davos, Switzerland, 26–30 June 2017; Volume 33 (Suppl. S2). [Google Scholar]
- Chen, C.; Song, J.; Qiu, J.; Zhao, J. Repair of a Meniscal Defect in a Rabbit Model Through Use of a Thermosensitive, Injectable, In Situ Crosslinked Hydrogel With Encapsulated Bone Mesenchymal Stromal Cells and Transforming Growth Factor β1. Am. J. Sports Med. 2020, 48, 884–894. [Google Scholar] [CrossRef]

| No | Category | Product Name | Agent | Medical Application | Type/Form of Hydrogel | Polymer Name | Reference |
|---|---|---|---|---|---|---|---|
| 1 | Wound dressings | Suprasorb® G | * | Adjusting moisture and the pain of the wound, removing necrotic tissue, relieving pain via cooling and soothing effect | Hydrogel film | Acrylic polymers, polyethylene, and phenoxyethanol with 70% water content | [98] |
| 2 | Wound dressings | AquaDerm™ | 2-Acrylamido-2 methyl-1 propanesulfonic acid sodium | Pressure ulcers, minor burns, radiation tissue damage | Hydrogel sheet | Propylene Glycol, poly (ethylene glycol) dimethacrylate, 2-Hydroxy-2-methylpropiophenone with 38–55% water | [99] |
| 3 | Wound dressings | SOLOSITE® Gel | * | With creation of a moist wound environment: Minor burns Superficial lacerations Cuts and abrasions (partial thickness wounds) Skin tears with the creation of a moist wound environment Venous ulcers (leg ulcers) Surgical incisions Diabetic foot ulcers Pressure ulcers (including stage IV) Assistance in autolytic debridement of wounds covered with necrotic tissues | Gel form of hydrogels | Sodium salt of carboxymethyl cellulose and glycerol with above 60% water | [100] |
| 4 | Cosmetic products | Rose soothing hydrogel mask | * | Face skin (it soothes and rejuvenates the skin) | Face mask | Sodium polyacrylate, glycerin, cellulose gum, water, etc | [101] |
| 5 | Cosmetic products | Advanced Genifique hydrogel Mask | * | Face skin (skin will become moisturized, radiant, smoother) | Face mask | Water, etc., enriched with Bifidus extract | [102] |
| 6 | Contact lenses | Focus® DAILIES® with AquaRelease™ | * | Astigmatism | Lubricates the eye for every blink | * | [103] |
| 7 | Contact lenses | Airsoft™ | * | Astigmatism | High water content and oxygen permeability | Silicone hydrogel material | [104] |
| 8 | Cartilage | * | * | Chondrogenesis | Visible pores, 3D structural integrity, good biocompatibility and proliferative activity | Biotinylated-HA, sodium alginate | [105] |
| 9 | Cartilage | * | TGF-β1 | Cartilage regeneration | 3D bioprinting HA-based hydrogels in tissues engineering | Thiol-modified HA | [106] |
| 10 | Bone | * | Maleimide | tissue engineered bone substitutes | 3D bioprinting HA-based hydrogels | HA | [107] |
| 11 | Vascular | * | * | Angiogenesis | 3D bioprinting HA-based hydrogels | HA glycidyl methacrylate, polylactic-co-glycolic acid (PLGA) | [108] |
| 12 | Vascular | * | collagen-I | Boosting neuronal development, improving peripheral nerve regeneration | 3D bioprinting HA-based hydrogels | Methacrylated HA | [109] |
| 13 | Nervous | * | Dopamine-conjugated HA, dopamine-conjugated gelatin, thiolated Pluronic F-127 | Nerve tissue regeneration | 3D bioprinting HA-based hydrogels | HA, conjugated gelatin | [110] |
| 14 | Cardiac | * | * | Cardiac tissue engineering | 3D bioprinting HA-based hydrogels | Methacrylated HA, methacrylated gelatin | [111] |
| 15 | Wound dressings | * | * | Dressing to enhance skin wound healing | Temperature- responsive hydrogels | Methylacrylate gelatin | [112] |
| 16 | Tissue engineering | * | * | Neural tissue engineering | Light/photo- responsive hydrogel | Conducting polymer hydrogel (CPH) based on copolymerized PANI and PAM (PAM/PANI CPH) | [113] |
| 17 | Drug delivery | * | * | Controlled drug delivery | PH-responsive hydrogels | Poly(methacryloyloxyethyl phosphorylcholine-co-4- formylbenzoate ethyl methacrylate) P(MPC-co-FBEMA) copolym | [114] |
| 18 | Drug delivery | * | * | Drug Delivery | Glucose-responsive hydrogels | Phenylboronic acid-grafted γ-Polyglutamic acid (PBA-PGA) | [115] |
| 19 | Corneal tissue engineering | * | Adrenaline and Chloramphenicol | Ocular drug delivery system | Temperature- responsive | Gellan maleate (MA-G) | [116] |
| 20 | Osseous tissue engineering | * | NaF, BSA, and BMP-2 | Cargo for delivery of different therapeutics | Temperature and Ultrasound- responsive | Alginate (Alg.) | [117] |
| 21 | Tendinous tissue engineering | * | PL | Delivery of PL for release of PL-derived growth factors | Magnetic- Responsive | Methacrylated chondroitin sulfate (MA- CS) | [118] |
| 22 | Meniscal tissue engineering | * | TGF-β1 | Release of TGF-β1 to manage the fibrochondrogenic differentiation of BMSCs and develop meniscal defects in rabbit model | Temperature- responsive | Glycol Chitosan GC/4- Arm PEG-CHO Hydrogel | [119] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farasati Far, B.; Naimi-Jamal, M.R.; Safaei, M.; Zarei, K.; Moradi, M.; Yazdani Nezhad, H. A Review on Biomedical Application of Polysaccharide-Based Hydrogels with a Focus on Drug Delivery Systems. Polymers 2022, 14, 5432. https://doi.org/10.3390/polym14245432
Farasati Far B, Naimi-Jamal MR, Safaei M, Zarei K, Moradi M, Yazdani Nezhad H. A Review on Biomedical Application of Polysaccharide-Based Hydrogels with a Focus on Drug Delivery Systems. Polymers. 2022; 14(24):5432. https://doi.org/10.3390/polym14245432
Chicago/Turabian StyleFarasati Far, Bahareh, Mohammad Reza Naimi-Jamal, Maryam Safaei, Kimia Zarei, Marzieh Moradi, and Hamed Yazdani Nezhad. 2022. "A Review on Biomedical Application of Polysaccharide-Based Hydrogels with a Focus on Drug Delivery Systems" Polymers 14, no. 24: 5432. https://doi.org/10.3390/polym14245432
APA StyleFarasati Far, B., Naimi-Jamal, M. R., Safaei, M., Zarei, K., Moradi, M., & Yazdani Nezhad, H. (2022). A Review on Biomedical Application of Polysaccharide-Based Hydrogels with a Focus on Drug Delivery Systems. Polymers, 14(24), 5432. https://doi.org/10.3390/polym14245432








