Norbornene as Key for a Possible Efficient Chemical Recycling in Structures Based on Ethylene
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Polyethylene
2.3. Synthesis of Copolymers Based on Ethylene and Norbornene
2.4. Materials Processing
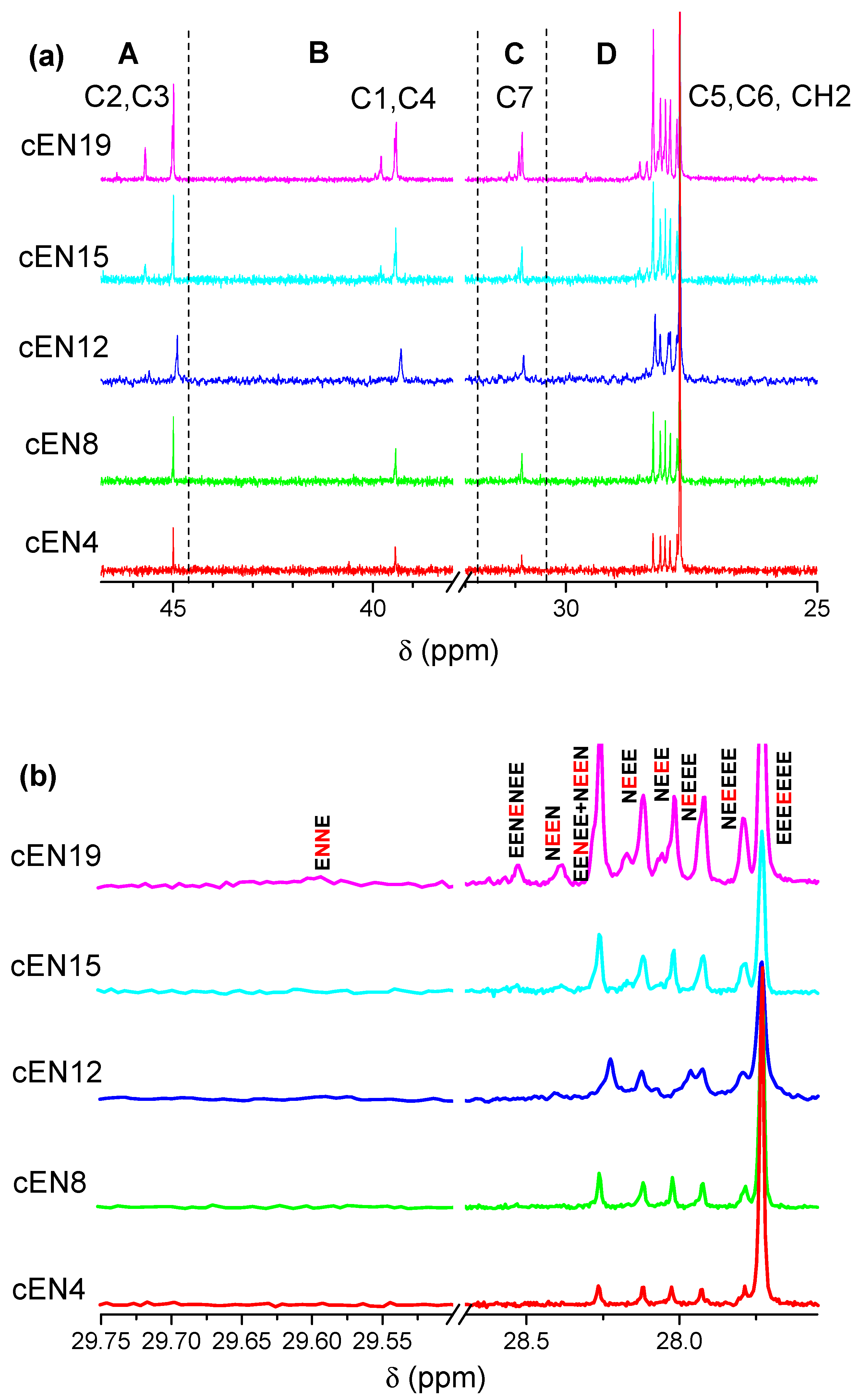
2.5. Nuclear Magnetic Resonance (13C NMR)
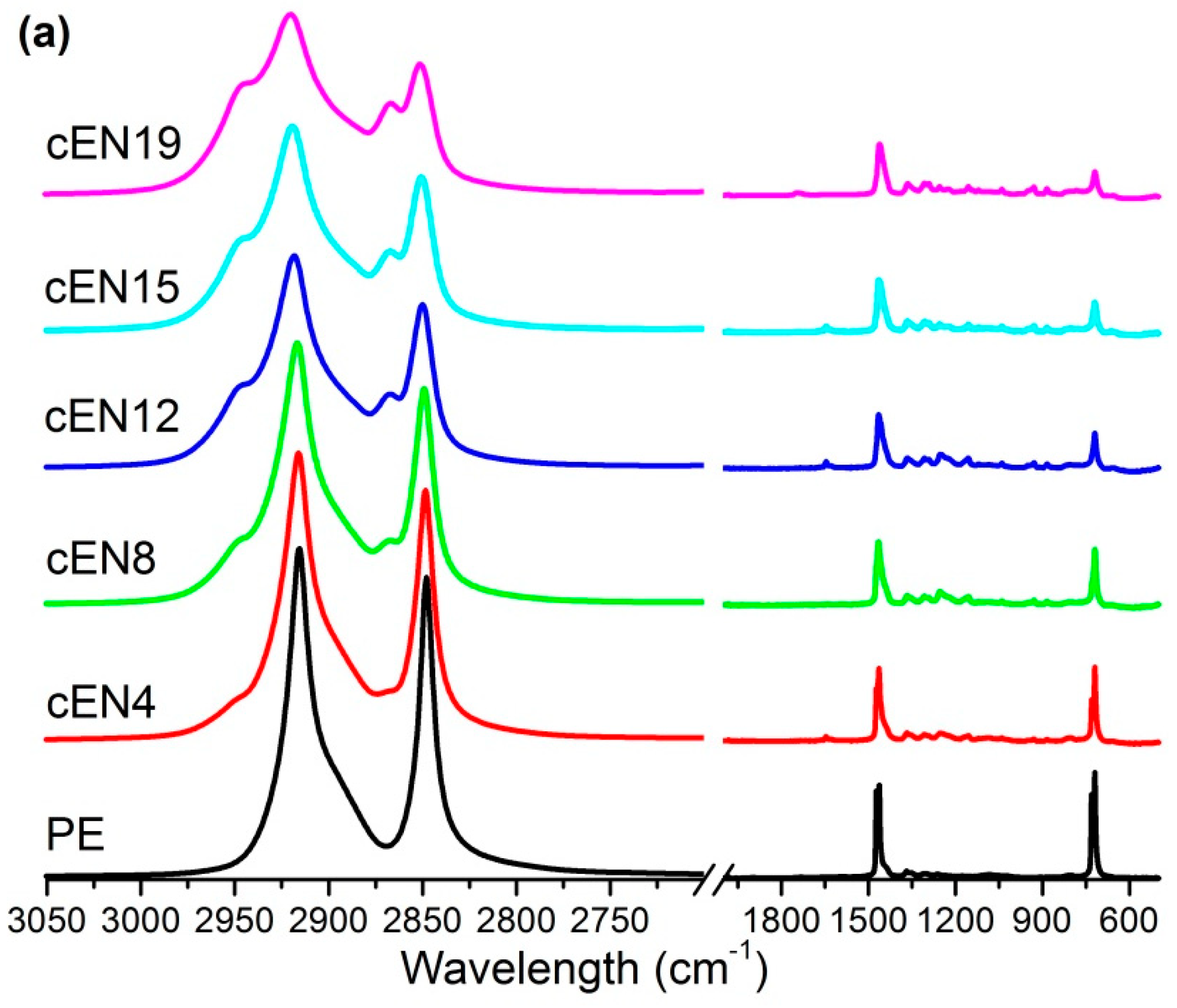
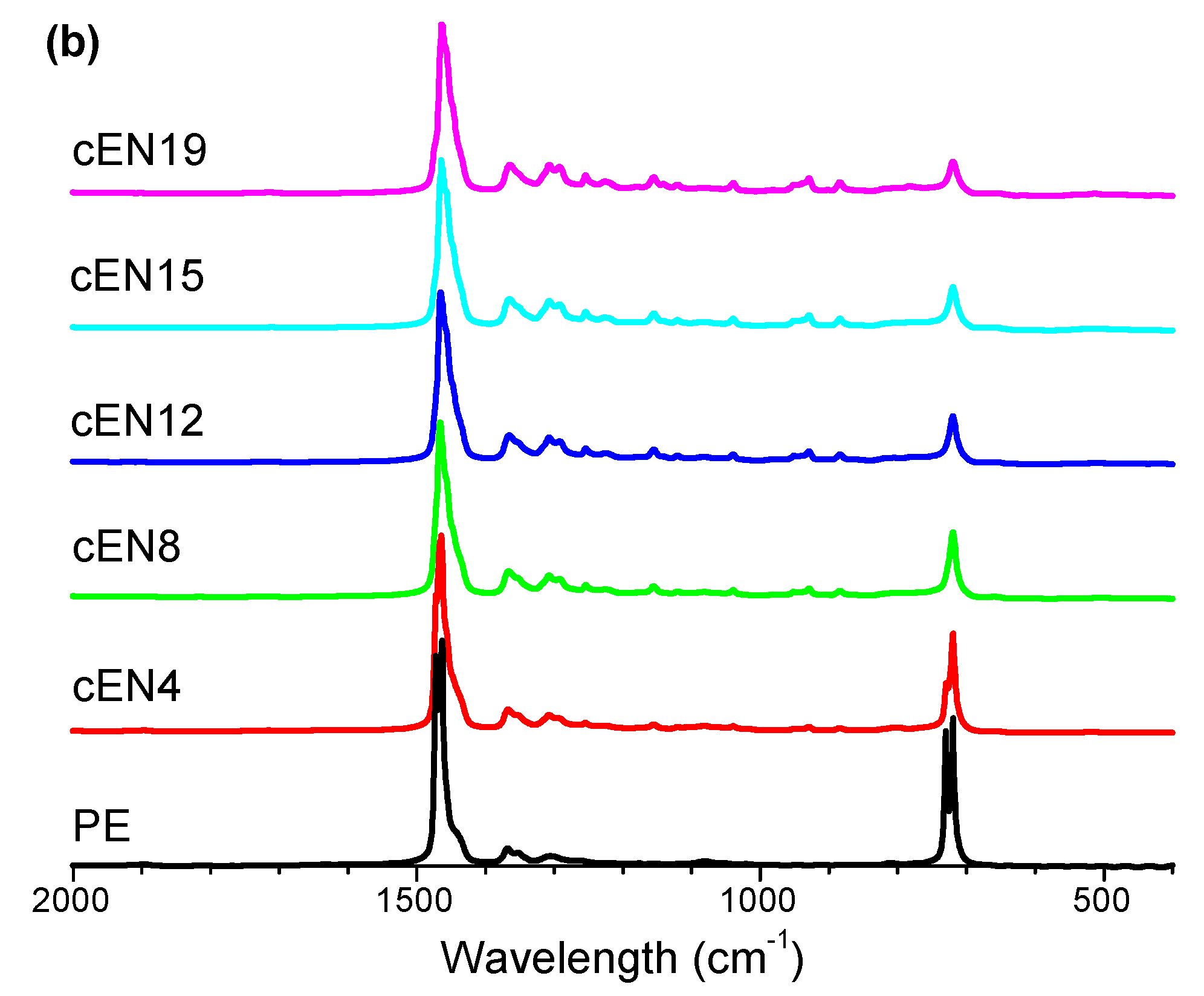
2.6. Fourier Transform Infrared Spectroscopy
2.7. Differential Scanning Calorimetry
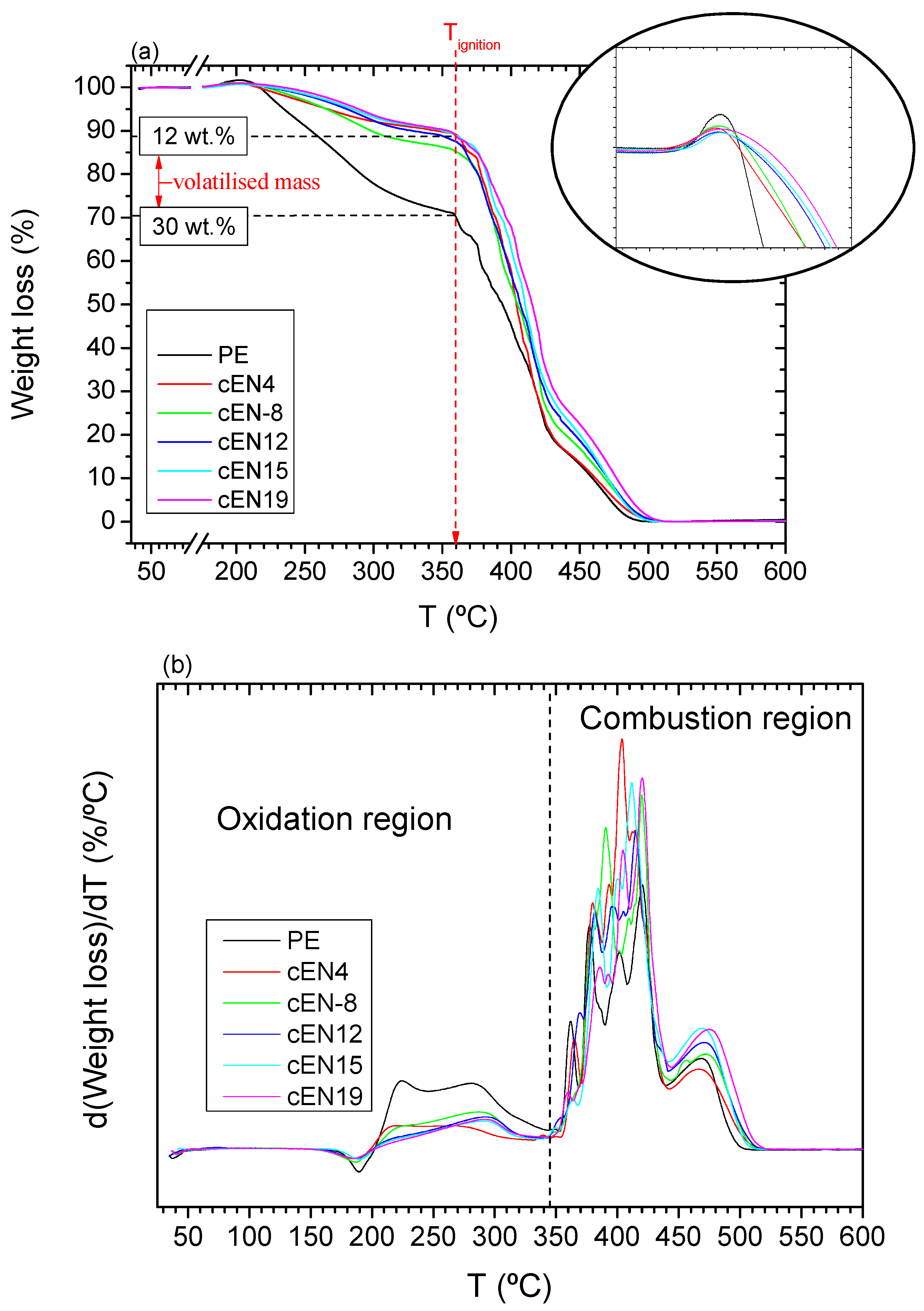
2.8. Thermogravimetry
2.9. Dynamic Mechanical Analysis (DMA)
3. Results and Discussion
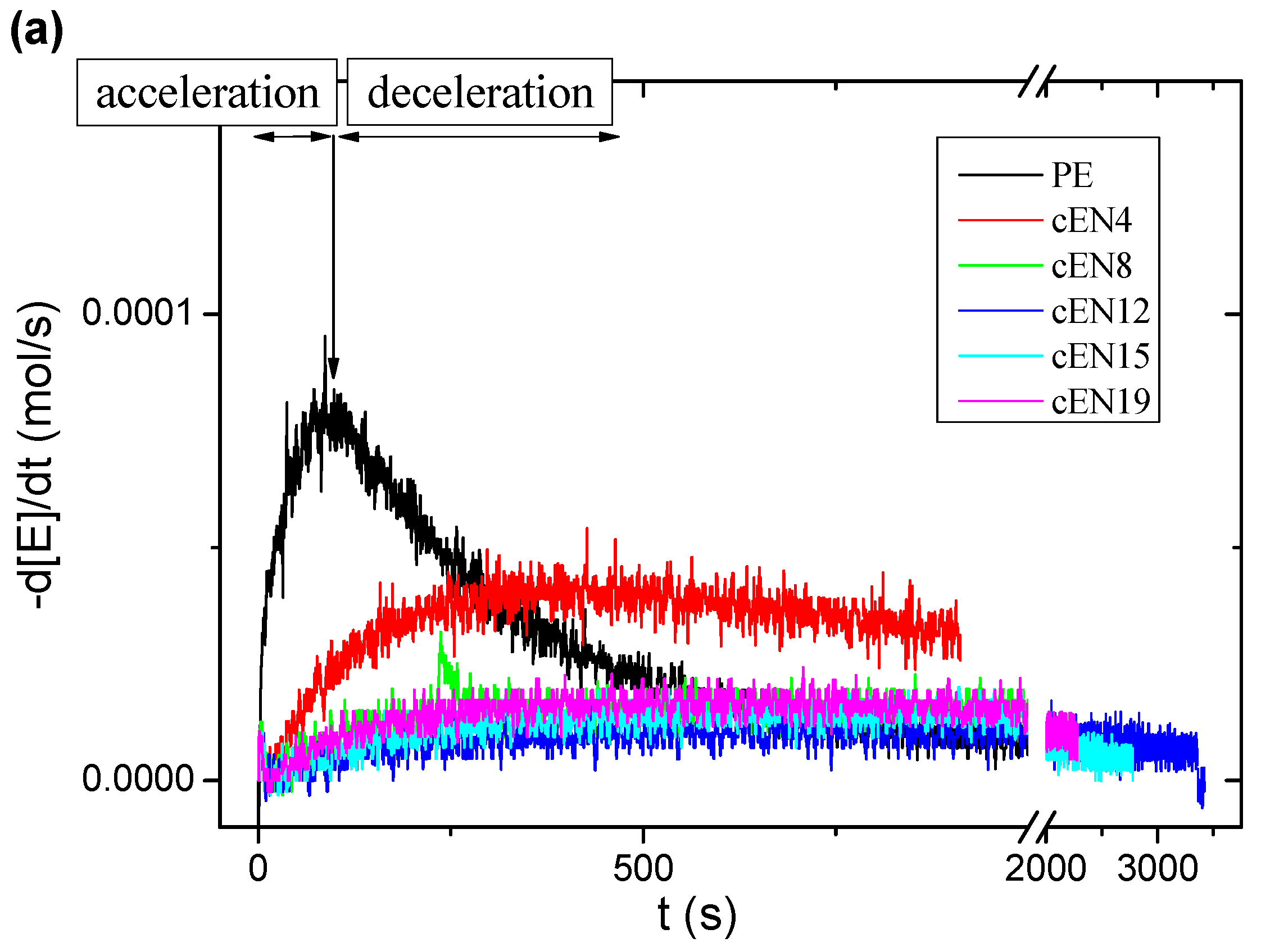
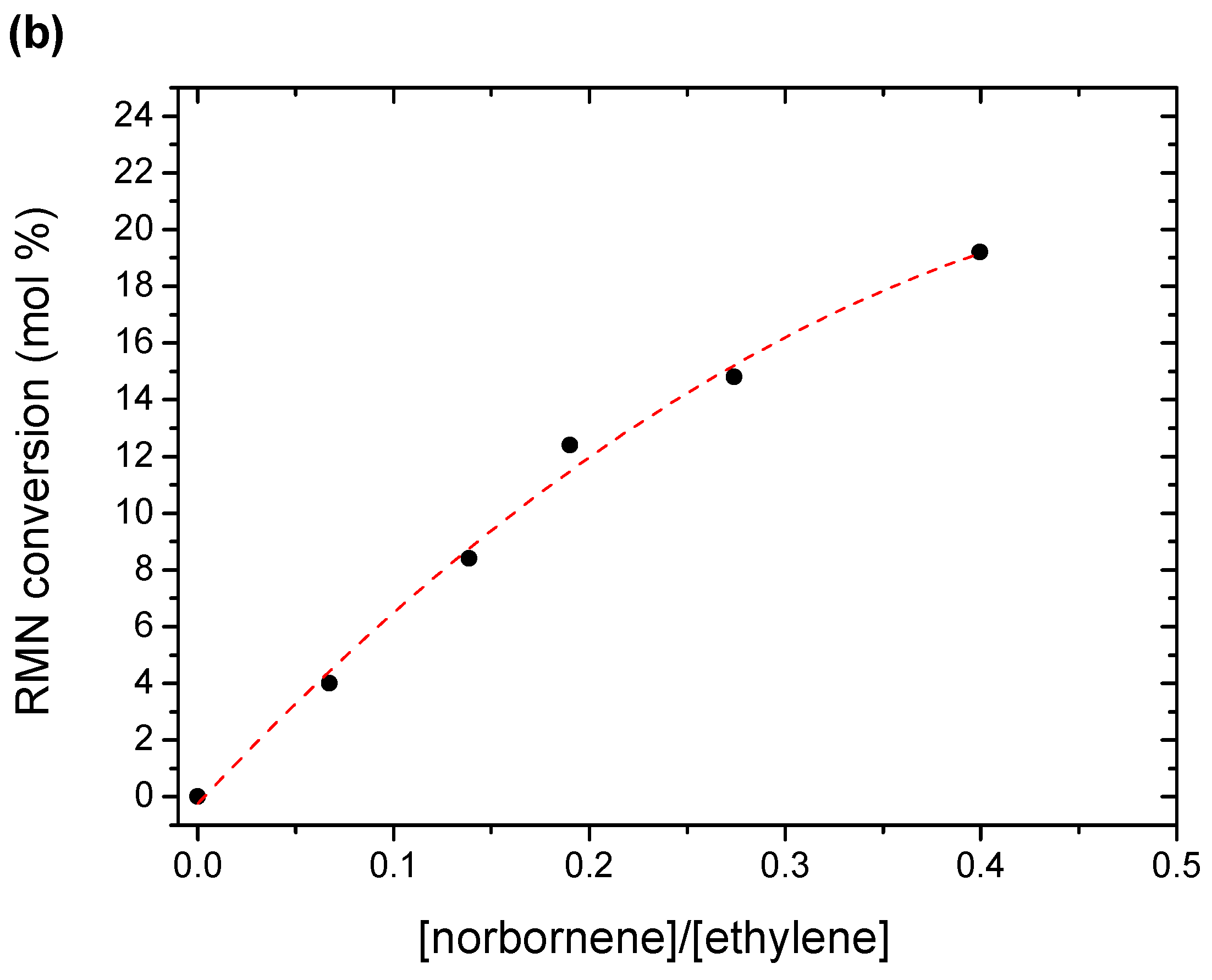
3.1. Kinetics and Catalytic Activity
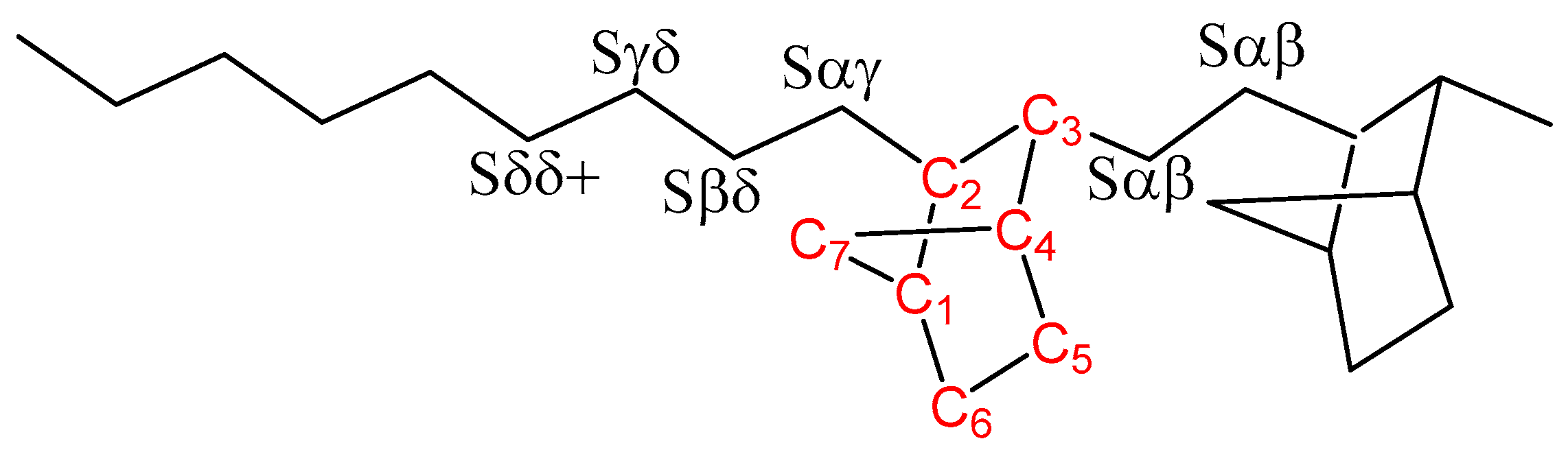
3.2. Molecular Characterization
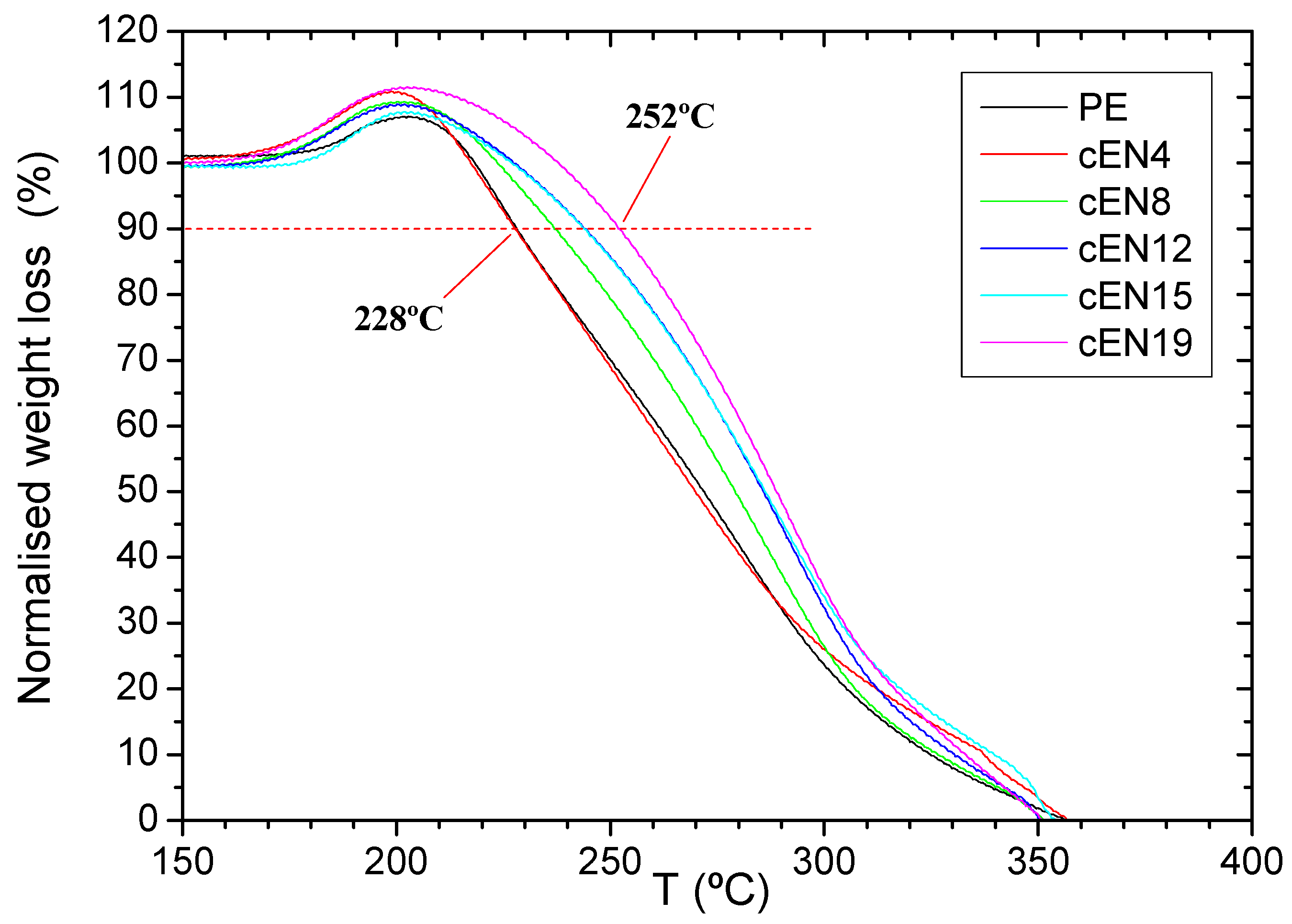
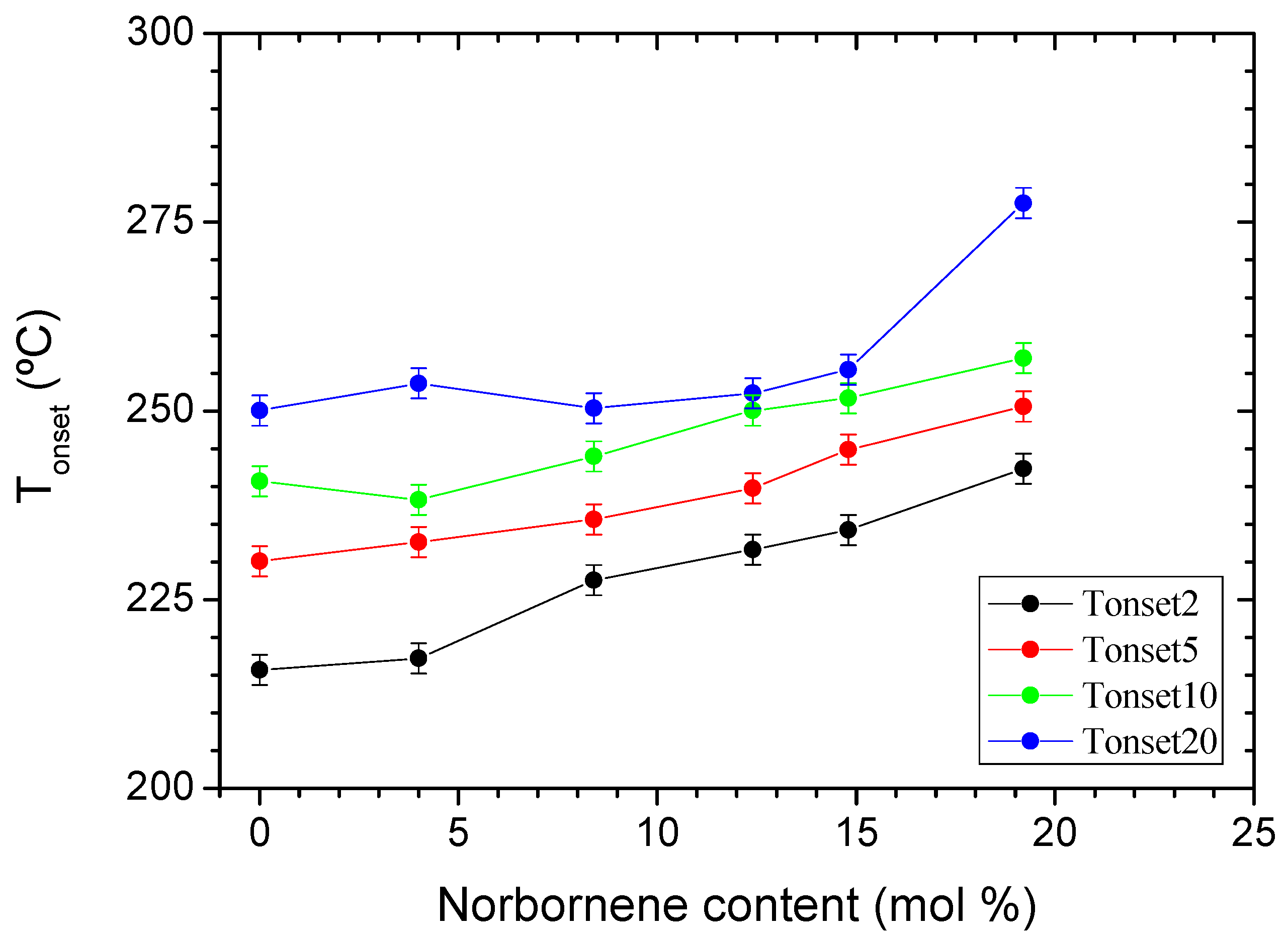
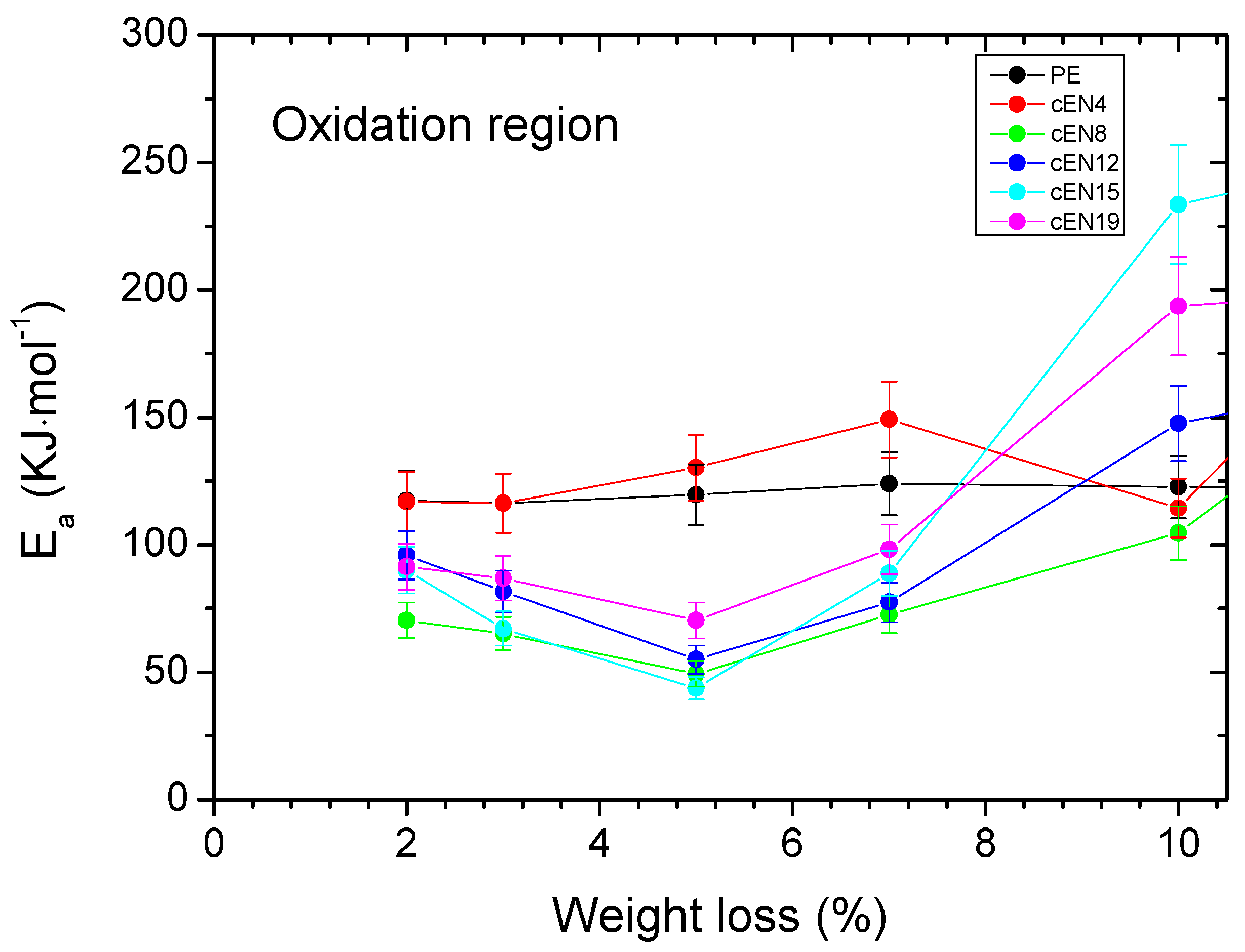
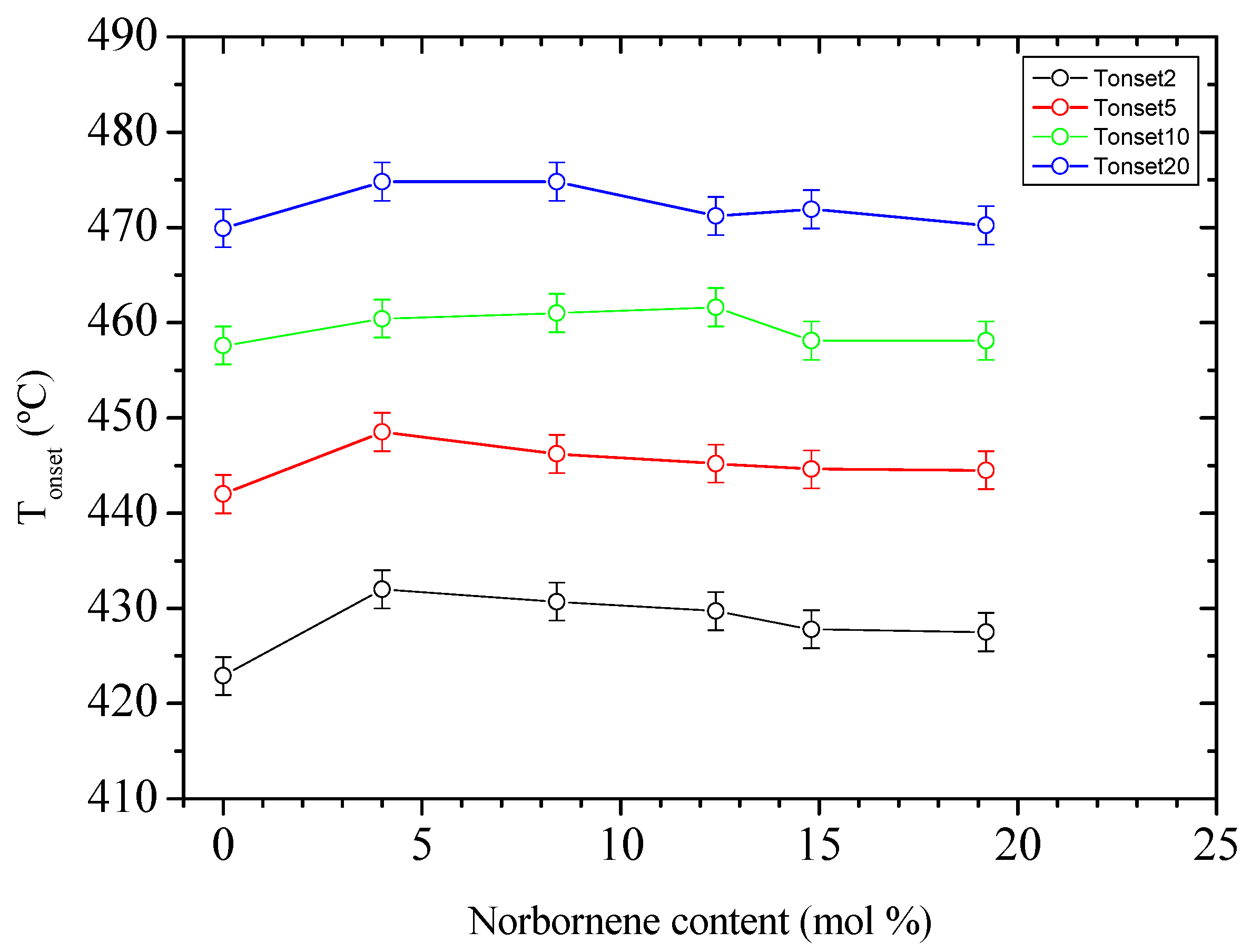
3.3. Thermo-Oxidative Stability
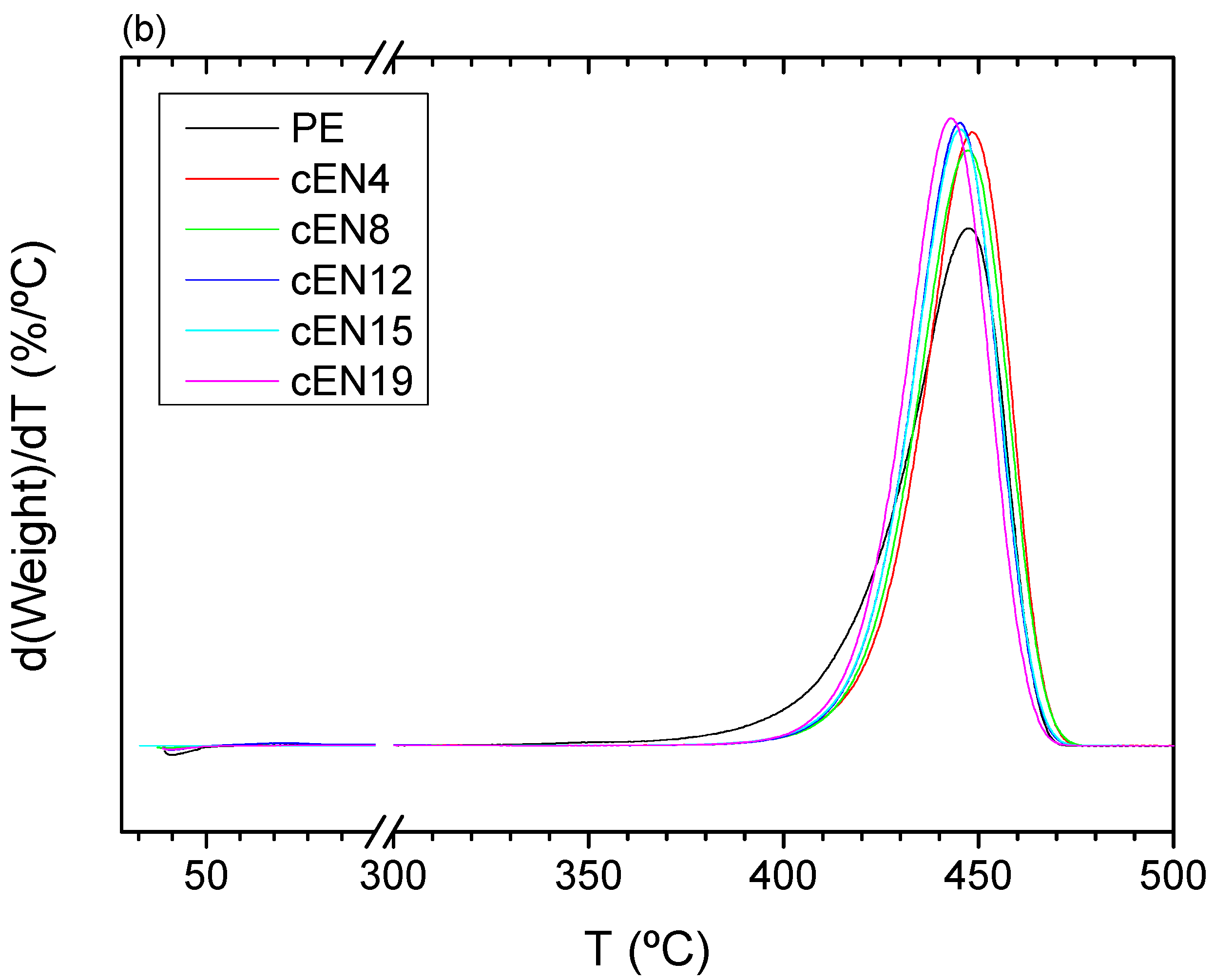
3.4. Thermal Stability under N2 Atmosphere
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Joo, J.; Lee, S.; Choi, H.; Lin, K.Y.A.; Lee, J. Single-Use Disposable Waste Upcycling via Thermochemical Conversion Pathway. Polymers 2021, 13, 2617. [Google Scholar] [CrossRef] [PubMed]
- Al-Salem, S.M.; Dutta, A. Wax Recovery from the Pyrolysis of Virgin and Waste Plastics. Ind. Eng. Chem. Res. 2021, 60, 8301–8309. [Google Scholar] [CrossRef]
- Anuar Sharuddin, S.D.; Abnisa, F.; Wan Daud, W.M.A.; Aroua, M.K. A review on pyrolysis of plastic wastes. Energy Convers. Manag. 2016, 115, 308–326. [Google Scholar] [CrossRef]
- Park, C.; Lee, J. Pyrolysis of polypropylene for production of fuel-range products: Effect of molecular weight of polypropylene. Int. J. Energy Res. 2021, 45, 13088–13097. [Google Scholar] [CrossRef]
- Sinfrônio, F.S.M.; Souza, A.G.; Santos, I.M.G.; Fernandes, V.J.; Novák, C.; Éhen, Z. Influence of H-ZSM-5, Al-MCM-41 and acid hybrid ZSM-5/MCM-41 on polyethylene decomposition. J. Therm. Anal. Calorim. 2006, 85, 391–399. [Google Scholar] [CrossRef]
- Kumar, S.; Panda, A.K.; Singh, R.K. A review on tertiary recycling of high-density polyethylene to fuel. Resour. Conserv. Recycl. 2011, 55, 893–910. [Google Scholar] [CrossRef]
- Beltramini, J.N. Catalytic conversion of municipal waste plastic into gasoline-range products over mesoporous materials. China Particuol. 2006, 4, 80–82. [Google Scholar] [CrossRef]
- Marcilla, A.; Gómez, A.; García, Á.N.; Mar Olaya, M. Kinetic study of the catalytic decomposition of different commercial polyethylenes over an MCM-41 catalyst. J. Anal. Appl. Pyrolysis 2002, 64, 85–101. [Google Scholar] [CrossRef]
- Kaminsky, W.; Zorriqueta, I.J.N. Catalytical and thermal pyrolysis of polyolefins. J. Anal. Appl. Pyrolysis 2007, 79, 368–374. [Google Scholar] [CrossRef]
- Ju Park, H.; Yim, J.H.; Jeon, J.K.; Man Kim, J.; Yoo, K.S.; Park, Y.K. Pyrolysis of polypropylene over mesoporous MCM-48 material. J. Phys. Chem. Solids 2008, 69, 1125–1128. [Google Scholar] [CrossRef]
- Gómez-Elvira, J.M.; Benavente, R.; Martínez, M.C. Unravelling the contribution of chain microstructure in the mechanism of the syndiotactic polypropylene pyrolysis. Polym. Degrad. Stab. 2013, 98, 1150–1163. [Google Scholar] [CrossRef]
- Martínez, M.C.; Benavente, R.; Gómez-Elvira, J.M. Molecular weight dependence and stereoselective chain cleavage during the early stages of the isotactic polypropylene pyrolysis. Polym. Degrad. Stab. 2017, 143, 26–34. [Google Scholar] [CrossRef]
- Gómez-Elvira, J.M.; Benavente, R.; Martínez, M.C. Correlation between chain microstructure and activation energy in the pyrolysis of a high molecular weight isotactic polypropylene. Polym. Degrad. Stab. 2015, 117, 46–57. [Google Scholar] [CrossRef]
- García-Peñas, A.; Cerrada, M.L.; Gómez-Elvira, J.M.; Pérez, E. Microstructure and thermal stability in metallocene iPP-materials: 1-pentene and 1-hexene copolymers. Polym. Degrad. Stab. 2016, 124, 77–86. [Google Scholar] [CrossRef]
- Forsyth, J.F.; Scrivani, T.; Benavente, R.; Marestin, C.; Pereña, J.M. Thermal and dynamic mechanical behavior of ethylene/norbornene copolymers with medium norbornene contents. J. Appl. Polym. Sci. 2001, 82, 2159–2165. [Google Scholar] [CrossRef]
- Tritto, I.; Marestin, C.; Boggioni, L.; Zetta, L.; Provasoli, A.; Ferro, D.R. Ethylene-norbornene copolymer microstructure. Assessment and advances based on assignments of 13C NMR spectra. Macromolecules 2000, 33, 8931–8944. [Google Scholar] [CrossRef]
- Liu, C.; Yu, J.; Sun, X.; Zhang, J.; He, J. Thermal degradation studies of cyclic olefin copolymers. Polym. Degrad. Stab. 2003, 81, 197–205. [Google Scholar] [CrossRef]
- Makrocka-Rydzyk, M.; Nowaczyk, G.; Głowinkowski, S.; Jurga, S. Dynamic mechanical study of molecular dynamics in ethylene-norbornene copolymers. Polymer 2010, 51, 908–912. [Google Scholar] [CrossRef]
- Lago, W.S.R.; Aymes-Chodur, C.; Ahoussou, A.P.; Yagoubi, N. Physico-chemical ageing of ethylene–norbornene copolymers: A review. J. Mater. Sci. 2017, 52, 6879–6904. [Google Scholar] [CrossRef]
- Villar, M.A.; Ferreira, M.L. Co-and Terpolymerization of Ethylene, Propylene, and Higher-Olefins with High Propylene Contents Using Metallocene Catalysts. J. Polym. Sci. Part A Polym. Chem. 2001, 39, 1136–1148. [Google Scholar] [CrossRef]
- Philippart, J.L.; Sinturel, C.; Arnaud, R.; Gardette, J.L. Influence of the exposure parameters on the mechanism of photooxidation of polypropylene. Polym. Degrad. Stab. 1999, 64, 213–225. [Google Scholar] [CrossRef]
- Randall, J. A Review of High Resolution Liquid Nuclear Magnetic Resonance Characterizations of Ethylene-Based Polymers. J. Macromol. Sci. Part C 1989, 29, 201–317. [Google Scholar] [CrossRef]
- Hill, G.T.H.; Almond, J.; Sugumaar, P.; Wenzel, M.N.; Hill, G.; Wallis, C. Determination of the carbonyl index of polyethylene and polypropylene using specified area under band methodology with ATR-FTIR spectroscopy. e-Polymers 2020, 20, 369–381. [Google Scholar] [CrossRef]
- Peterson, J.D.; Vyazovkin, S.; Wight, C.A. Kinetics of the Thermal and Thermo-Oxidative Degradation of Polystyrene, Polyethylene and Poly(propylene). Macromol. Chem. Phys 2001, 202, 775–784. [Google Scholar] [CrossRef]
- Lamnawar, K.; Vion-Loisel, F.; Maazouz, A. Rheological, morphological, and heat seal properties of linear low density polyethylene and cyclo olefine copolymer (LLDPE/COC) blends. J. Appl. Polym. Sci. 2010, 116, 2015–2022. [Google Scholar] [CrossRef]
- Bueche, F. Mechanical degradation of high polymers. J. Appl. Polym. Sci. 1960, 4, 101–106. [Google Scholar] [CrossRef]
- Nyden, M.R.; Stoliarov, S.I.; Westmoreland, P.R.; Guo, Z.X.; Jee, C. Applications of reactive molecular dynamics to the study of the thermal decomposition of polymers and nanoscale structures. Mater. Sci. Eng. A 2004, 365, 114–121. [Google Scholar] [CrossRef]
- Stoliarov, S.I.; Lyon, R.E.; Nyden, M.R. A reactive molecular dynamics model of thermal decomposition in polymers. II. Polyisobutylene. Polymer 2004, 45, 8613–8621. [Google Scholar] [CrossRef]
- Kruse, T.M.; Wong, H.W.; Broadbelt, L.J. Mechanistic Modeling of Polymer Pyrolysis: Polypropylene. Macromolecules 2003, 36, 9594–9607. [Google Scholar] [CrossRef]
- Levine, S.E.; Broadbelt, L.J. Detailed mechanistic modeling of high-density polyethylene pyrolysis: Low molecular weight product evolution. Polym. Degrad. Stab. 2009, 94, 810–822. [Google Scholar] [CrossRef]




| Sample | Initial Pressure (Bar) | Norbornene/Ethylene Molar Feeding Ratio | [Al]/[Zr] |
|---|---|---|---|
| PE | 1.58 | 0 | 1083 |
| cEN4 | 1.59 | 0.066 | 1083 |
| cEN8 | 1.53 | 0.137 | 1083 |
| cEN12 | 1.57 | 0.187 | 1083 |
| cEN15 | 1.56 | 0.269 | 1083 |
| cEN19 | 1.59 | 0.386 | 1064 |
| Sample | Processing Conditions | |
|---|---|---|
| T (°C) | P (Bar) | |
| PE | 190 | 110 |
| cEN4 | 160 | 110 |
| cEN8 | 160 | 100 |
| cEN12 | 150 | 50 |
| cEN15 | 140 | 25 |
| cEN19 | 170 | 160 |
| Sample | Yield (g) | Comonomer Content * (mol %) | Activity (kg/h·mol Zr) |
|---|---|---|---|
| PE | 0.84 | 0 | 685 |
| cEN4 | 1.10 | 4.0 | 1051 |
| cEN8 | 1.00 | 8.4 | 709 |
| cEN12 | 1.32 | 12.4 | 315 |
| cEN15 | 1.30 | 14.8 | 453 |
| cEN19 | 1.54 | 19.2 | 636 |
| Compositional Sequence | Signal (ppm) | Carbon Nucleus |
|---|---|---|
| EEEEEEE | 27.73 | Sδ+δ+ |
| NEEEEE | 27.79 | Sδδ+ |
| NEEEE | 27.92 | Sβδ+ |
| NEEE | 28.02 | Sγδ, Sγδ+ |
| NEEE | 28.12 | Sαδ+ |
| EENEE+NEEN | 28.26 | Sαδ |
| NEEN | 28.39 | Sβγ |
| EENENEE | 28.53 | Sαβ |
| ENNE | 29.68 | C6 |
| Sample | EEE | EEN | NEN | ENE | nE |
|---|---|---|---|---|---|
| cEN4 | 87.7 | 8.2 | 0 | 4.1 | 23 |
| cEN8 | 73.0 | 18.0 | 0 | 9 | 10 |
| cEN12 | 64.9 | 23.4 | 0 | 11.7 | 7 |
| cEN15 | 55.7 | 26.8 | 2.1 | 15.5 | 5 |
| cEN19 | 37.1 | 34.3 | 4.8 | 21.9 | 3 |
| Sample | CI |
|---|---|
| PE | 0.0148 |
| cEN4 | 0.0113 |
| cEN8 | 0.0084 |
| cEN12 | 0.0061 |
| cEN15 | 0.0106 |
| cEN19 | 0.0073 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calles-Valero, A.F.; García-Peñas, A.; Cerrada, M.L.; Gómez-Elvira, J.M. Norbornene as Key for a Possible Efficient Chemical Recycling in Structures Based on Ethylene. Polymers 2022, 14, 5052. https://doi.org/10.3390/polym14225052
Calles-Valero AF, García-Peñas A, Cerrada ML, Gómez-Elvira JM. Norbornene as Key for a Possible Efficient Chemical Recycling in Structures Based on Ethylene. Polymers. 2022; 14(22):5052. https://doi.org/10.3390/polym14225052
Chicago/Turabian StyleCalles-Valero, Antonio F., Alberto García-Peñas, María L. Cerrada, and José M. Gómez-Elvira. 2022. "Norbornene as Key for a Possible Efficient Chemical Recycling in Structures Based on Ethylene" Polymers 14, no. 22: 5052. https://doi.org/10.3390/polym14225052
APA StyleCalles-Valero, A. F., García-Peñas, A., Cerrada, M. L., & Gómez-Elvira, J. M. (2022). Norbornene as Key for a Possible Efficient Chemical Recycling in Structures Based on Ethylene. Polymers, 14(22), 5052. https://doi.org/10.3390/polym14225052







