Comparison of Various Antimicrobial Agents for Thermoplastic Polymeric Retainers
Abstract
:1. Introduction
2. Materials and Methods
Statistical Analysis
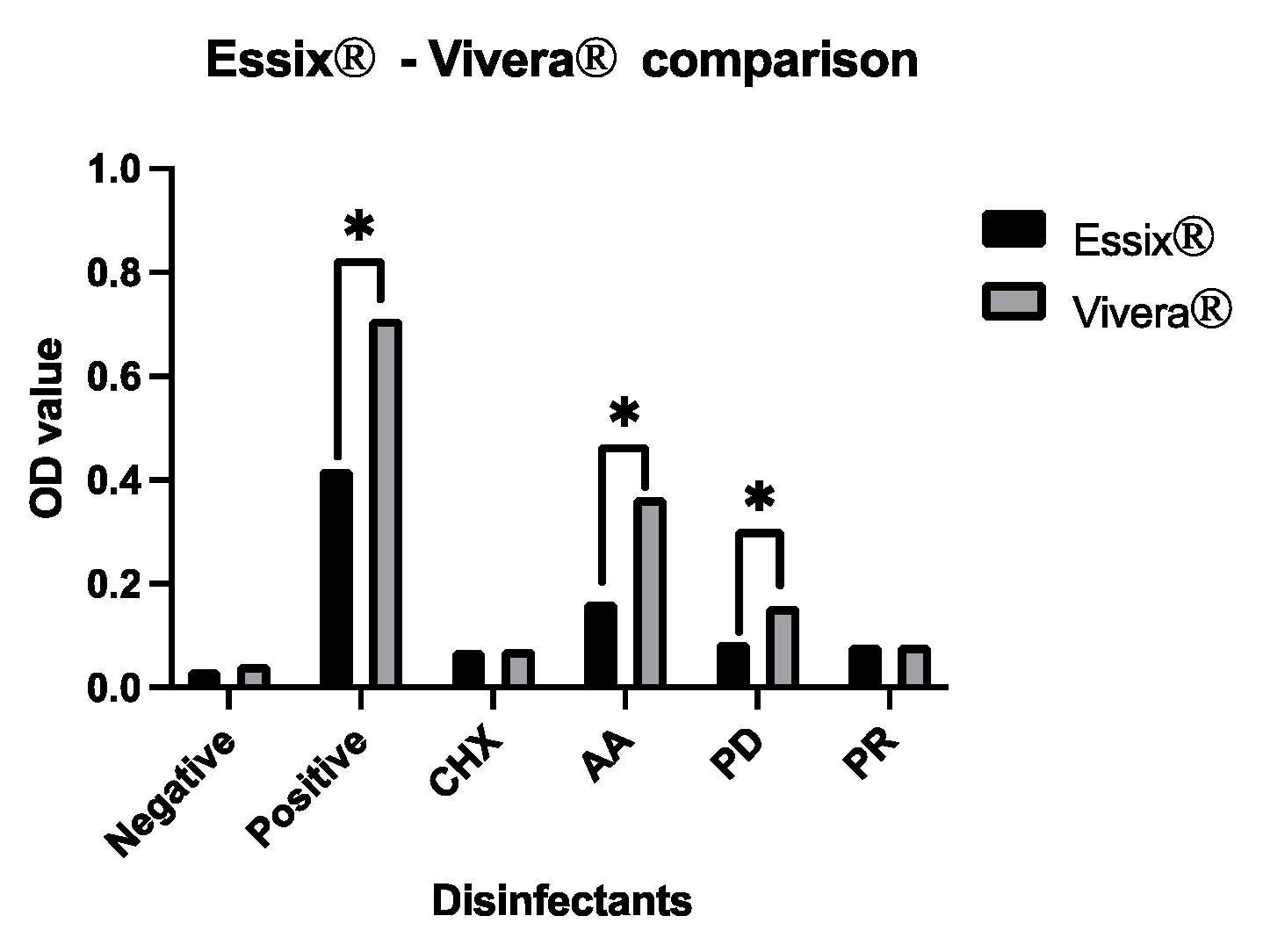
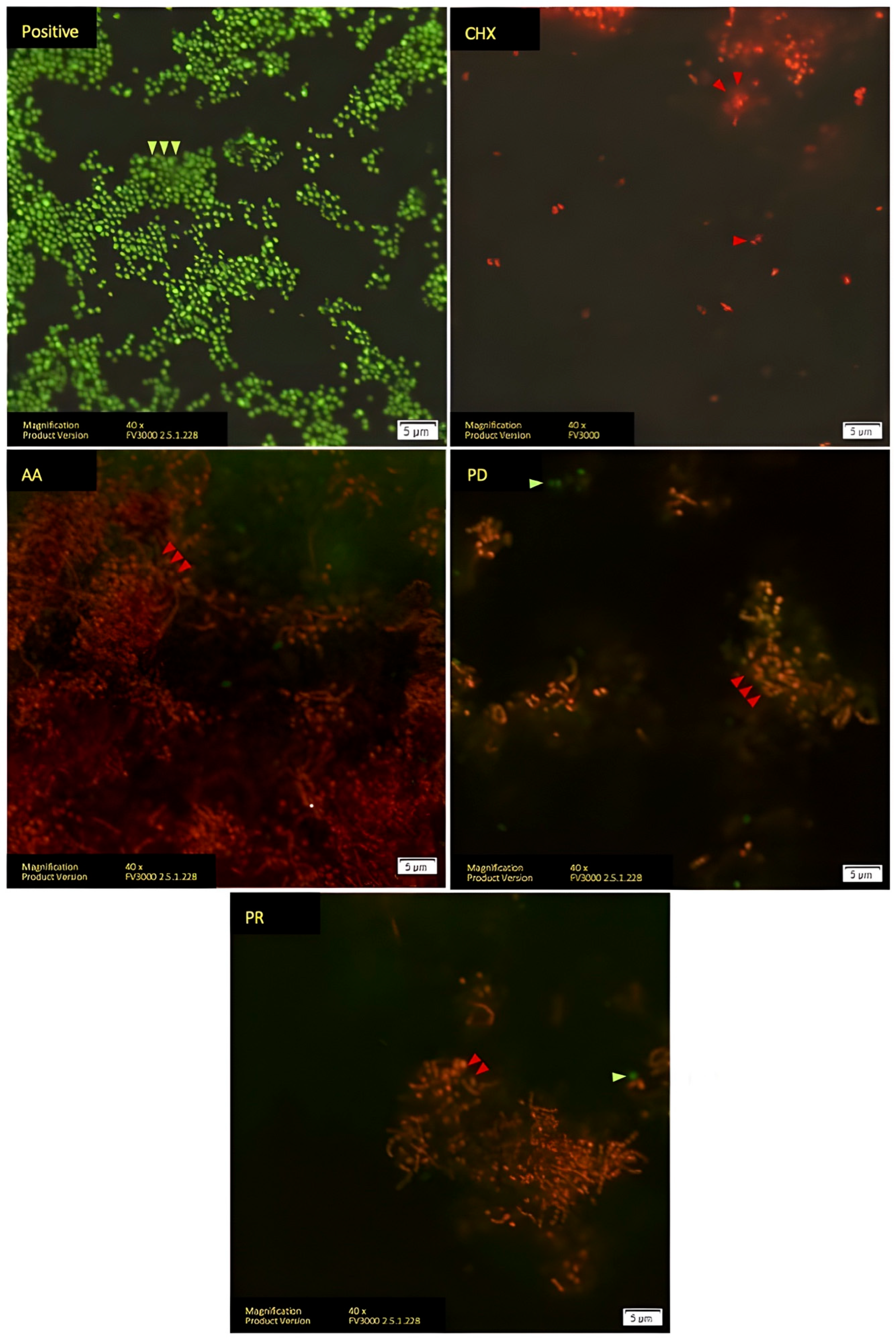
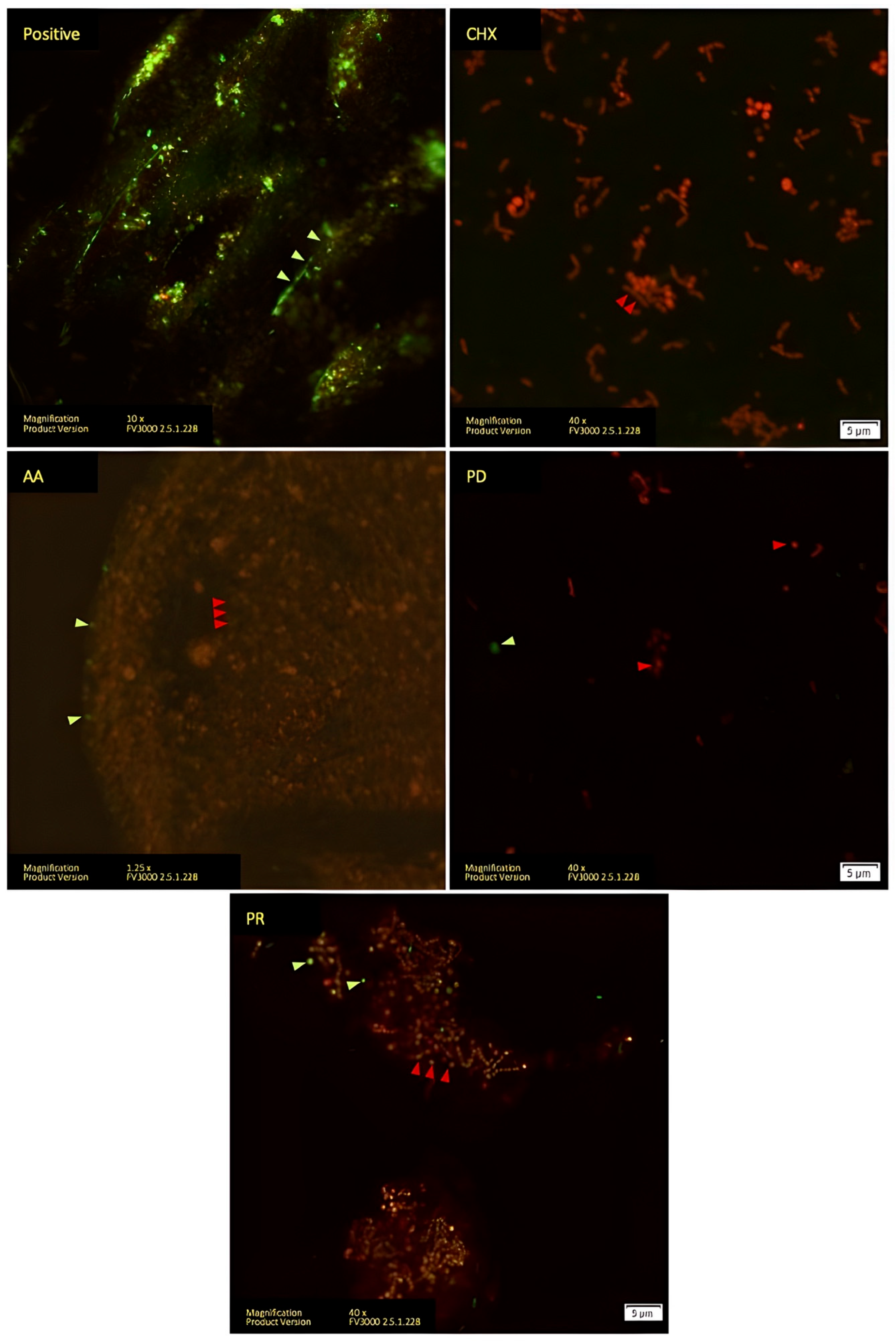
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kiatwarawut, K.; Rokaya, D.; Sirisoontorn, I. Antimicrobial Activity of Various Disinfectants to Clean Thermoplastic Polymeric Appliances in Orthodontics. Polymers 2022, 14, 2256. [Google Scholar] [CrossRef] [PubMed]
- Türköz, Ç.; Canigür Bavbek, N.; Kale Varlik, S.; Akça, G. Influence of thermoplastic retainers on Streptococcus mutans and Lactobacillus adhesion. Am. J. Orthod. Dentofac. Orthop. 2012, 141, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Alshatti, H. Comparison of White Spot Lesions among Clear Aligners, Self-Ligating Brackets and Conventional Brackets-A Randomized Controlled Clinical Trial. Master’s Thesis, University of Connecticut, Storrs, CT, USA, 2017. [Google Scholar]
- Moshiri, M.; Eckhart, J.E.; Mcshane, P.; German, D.S. Consequences of poor oral hygiene during clear aligner therapy. J. Clin. Orthod. JCO 2013, 8, 494–498. [Google Scholar]
- Sadowsky, C.; Sakols, E.I. Long-term assessment of orthodontic relapse. Am. J. Orthod. 1982, 82, 456–463. [Google Scholar] [CrossRef]
- Sheridan, J.; LeDoux, W.; McMinn, R. Essix retainers: Fabrication and supervision for permanent retention. J. Clin. Orthod. JCO 1993, 27, 37–45. [Google Scholar]
- Barth, E.; Myrvik, Q.M.; Wagner, W.; Gristina, A.G. In vitro and in vivo comparative colonization of Staphylococcus aureus and Staphylococcus epidermidis on orthopaedic implant materials. Biomaterials 1989, 10, 325–328. [Google Scholar] [CrossRef]
- Mai, W.; He, J.A.; Meng, H.; Jiang, Y.; Huang, C.; Li, M.; Yuan, K.; Kang, N. Comparison of vacuum-formed and Hawley retainers: A systematic review. Am. J. Orthod. Dentofac. Orthop. 2014, 145, 720–727. [Google Scholar] [CrossRef]
- Hichens, L.; Rowland, H.; Williams, A.; Hollinghurst, S.; Ewings, P.; Clark, S.; Ireland, A.; Sandy, J. Cost-effectiveness and patient satisfaction: Hawley and vacuum-formed retainers. Eur. J. Orthod. 2007, 29, 372–378. [Google Scholar] [CrossRef]
- Edman Tynelius, G.; Bondemark, L.; Lilja-Karlander, E. Evaluation of orthodontic treatment after 1 year of retention—A randomized controlled trial. Eur. J. Orthod. 2010, 32, 542–547. [Google Scholar] [CrossRef]
- Oga, M.; Sugioka, Y.; Hobgood, C.; Gristina, A.; Myrvik, Q. Surgical biomaterials and differential colonization by Staphylococcus epidermidis. Biomaterials 1988, 9, 285–289. [Google Scholar] [CrossRef]
- Hogt, A.; Dankert, J.; De Vries, J.; Feijen, J. Adhesion of coagulase-negative staphylococci to biomaterials. Microbiology 1983, 129, 2959–2968. [Google Scholar] [CrossRef]
- Pringle, J.; Fletcher, M. Influence of substratum hydration and adsorbed macromolecules on bacterial attachment to surfaces. Appl. Environ. Microbiol. 1986, 51, 1321–1325. [Google Scholar] [CrossRef] [Green Version]
- Locci, R.; Peters, G.; Pulverer, G. Microbial colonization of prosthetic devices. I. Microtopographical characteristics of intravenous catheters as detected by scanning electron microscopy. Zentralbl. Bakteriol. Mikrobiol. Hyg. B 1981, 173, 285–292. [Google Scholar]
- Emilson, C. Susceptibility of various microorganisms to chlorhexidine. Eur. J. Oral Sci. 1977, 85, 255–265. [Google Scholar] [CrossRef]
- Shpack, N.; Greenstein, R.B.-N.; Gazit, D.; Sarig, R.; Vardimon, A.D. Efficacy of three hygienic protocols in reducing biofilm adherence to removable thermoplastic appliance. Angle Orthod. 2014, 84, 161–170. [Google Scholar] [CrossRef]
- Lessa, F.C.; Enoki, C.; Ito, I.Y.; Faria, G.; Matsumoto, M.A.; Nelson-Filho, P. In-vivo evaluation of the bacterial contamination and disinfection of acrylic baseplates of removable orthodontic appliances. Am. J. Orthod. Dentofacial Orthop. 2007, 131, 705.e11–705.e17. [Google Scholar] [CrossRef]
- Pilloni, J.A. Human Saliva Biofilm Reduction by Thermoplastic Retainer Cleaners: An In Vitro Study. Master’s Thesis, State University of New York at Stony Brook, Stony Brook, NY, USA, 2019. [Google Scholar]
- Albanna, R.H.; Farawanah, H.M.; Aldrees, A.M. Microbial evaluation of the effectiveness of different methods for cleansing clear orthodontic retainers: A randomized clinical trial. Angle Orthod. 2017, 87, 460–465. [Google Scholar] [CrossRef]
- Agarwal, M.; Wible, E.; Ramir, T.; Altun, S.; Viana, G.; Evans, C.; Lukic, H.; Megremis, S.; Atsawasuwan, P. Long-term effects of seven cleaning methods on light transmittance, surface roughness, and flexural modulus of polyurethane retainer material. Angle Orthod. 2018, 88, 355–362. [Google Scholar] [CrossRef]
- Wible, E.; Agarwal, M.; Altun, S.; Ramir, T.; Viana, G.; Evans, C.; Lukic, H.; Megremis, S.; Atsawasuwan, P. Long-term effects of different cleaning methods on copolyester retainer properties. Angle Orthod. 2019, 89, 221–227. [Google Scholar] [CrossRef]
- Wible, E.; Agarwal, M.; Altun, S.; Ramir, T.; Viana, G.; Evans, C.; Lukic, H.; Megremis, S.; Atsawasuwan, P. Long-term effects of various cleaning methods on polypropylene/ethylene copolymer retainer material. Angle Orthod. 2019, 89, 432–437. [Google Scholar] [CrossRef]
- Katara, G.; Hemvani, N.; Chitnis, S.; Chitnis, V.; Chitnis, D. Surface disinfection by exposure to germicidal UV light. Indian J. Med. Microbiol. 2008, 26, 241–242. [Google Scholar] [CrossRef]
- Stepanovic, S.; Vukovic, D.; Dakic, I.; Savic, B.; Svabic-Vlahovic, M. A modified microtiter-plate test for quantification of staphylococcal biofilm formation. J. Microbiol. Methods 2000, 40, 175–179. [Google Scholar] [CrossRef]
- Zheng, Y.; Yanful, E.K.; Bassi, A.S. A review of plastic waste biodegradation. Crit. Rev. Biotechnol. 2005, 25, 243–250. [Google Scholar] [CrossRef]
- Allen, A.B.; Hilliard, N.P.; Howard, G.T. Purification and characterization of a solublepolyurethane degrading enzyme from Comamonasacidovorans. Int. Biodeterior. Biodegrad. 1999, 43, 37–41. [Google Scholar] [CrossRef]
- Kleeberg, I.; Hetz, C.; Kroppenstedt, R.M.; Müller, R.-J.; Deckwer, W.-D. Biodegradation of aliphatic-aromatic copolyesters by Thermomonospora fusca and other thermophilic compost isolates. Appl. Environ. Microbiol. 1998, 64, 1731–1735. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.-J.; Kleeberg, I.; Deckwer, W.-D. Biodegradation of polyesters containing aromatic constituents. J. Biotechnol. 2001, 86, 87–95. [Google Scholar] [CrossRef]
- Nakajima-Kambe, T.; Shigeno-Akutsu, Y.; Nomura, N.; Onuma, F.; Nakahara, T. Microbial degradation of polyurethane, polyester polyurethanes and polyether polyurethanes. Appl. Microbiol. Biotechnol. 1999, 51, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Leikin, J.B.P.; Frank, P. (Eds.) Chlorhexidine gluconate. In Poisoning and Toxicology Handbook; Taylor & Francis: Abingdon-on-Thames, UK, 2008. [Google Scholar]
- Russell, A.; Day, M. Antibacterial activity of chlorhexidine. J. Hosp. Infect. 1993, 25, 229–238. [Google Scholar] [CrossRef]
- Hugo, W.; Longworth, A. Some aspects of the mode of action of chlorhexidine. J. Pharm. Pharmacol. 1964, 16, 655–662. [Google Scholar] [CrossRef]
- Pantanella, F.; Berlutti, F.; Passariello, C.; Sarli, S.; Morea, C.; Schippa, S. Violacein and biofilm production in Janthinobacterium lividum. J. Appl. Microbiol. 2007, 102, 992–999. [Google Scholar] [CrossRef]
- Brindle, E.R.; Miller, D.A.; Stewart, P.S. Hydrodynamic deformation and removal of Staphylococcus epidermidis biofilms treated with urea, chlorhexidine, iron chloride, or DispersinB. Biotechnol. Bioeng. 2011, 108, 2968–2977. [Google Scholar] [CrossRef]
- Sun, P.-F.; Kim, T.-S.; Kim, H.-S.; Ham, S.-Y.; Jang, Y.; Park, Y.-G.; Tang, C.Y.; Park, H.-D. Improved anti-biofouling performance of pressure retarded osmosis (PRO) by dosing with chlorhexidine gluconate. Desalination 2020, 481, 114376. [Google Scholar] [CrossRef]
- Salmond, C.V.; Kroll, R.G.; Booth, I.R. The effect of food preservatives on pH homeostasis in Escherichia coli. Microbiology 1984, 130, 2845–2850. [Google Scholar] [CrossRef] [Green Version]
- Walter, A.; Gutknecht, J. Monocarboxylic acid permeation through lipid bilayer membranes. J. Membr. Biol. 1984, 77, 255–264. [Google Scholar] [CrossRef]
- Slonczewski, J.L.; Rosen, B.P.; Alger, J.R.; Macnab, R.M. pH homeostasis in Escherichia coli: Measurement by 31P nuclear magnetic resonance of methylphosphonate and phosphate. Proc. Natl. Acad. Sci. USA 1981, 78, 6271–6275. [Google Scholar] [CrossRef]
- Slonczewski, J.L.; Fujisawa, M.; Dopson, M.; Krulwich, T.A. Cytoplasmic pH measurement and homeostasis in bacteria and archaea. Adv. Microb. Physiol. 2009, 55, 1–317. [Google Scholar]
- Hirshfield, I.N.; Terzulli, S.; O’Byrne, C. Weak organic acids: A panoply of effects on bacteria. Sci. Prog. 2003, 86, 245–270. [Google Scholar] [CrossRef]
- Budtz-Jørgensen, E. Materials and methods for cleaning dentures. J. Prosthet. Dent. 1979, 42, 619–623. [Google Scholar] [CrossRef]
- Kasibut, P.; Kuvatanasuchati, J.; Thaweboon, B.; Sirisoontorn, I. Oral Microbiome in Orthodontic Acrylic Retainer. Polymers 2022, 14, 3583. [Google Scholar] [CrossRef]
- Low, B.; Lee, W.; Seneviratne, C.J.; Samaranayake, L.P.; Hägg, U. Ultrastructure and morphology of biofilms on thermoplastic orthodontic appliances in ‘fast’ and ‘slow’ plaque formers. Eur. J. Orthod. 2011, 33, 577–583. [Google Scholar] [CrossRef]
- Zafeiriadis, A.A.; Karamouzos, A.; Athanasiou, A.E.; Eliades, T.; Palaghias, G. An in vivo spectrophotometric evaluation of Vivera and Essix clear thermoplastic retainer discolouration. Australas. Orthod. J. 2018, 34, 3–10. [Google Scholar] [CrossRef]
- Proffit, W.; Fields, H.; Larson, B.; Sarver, D. Contemporary Orthodontics, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Boulos, L.; Prevost, M.; Barbeau, B.; Coallier, J.; Desjardins, R. LIVE/DEAD®® BacLight™: Application of a new rapid staining method for direct enumeration of viable and total bacteria in drinking water. J. Microbiol. Methods 1999, 37, 77–86. [Google Scholar] [CrossRef]
- Jin, Y.; Zhang, T.; Samaranayake, Y.; Fang, H.; Yip, H.; Samaranayake, L. The use of new probes and stains for improved assessment of cell viability and extracellular polymeric substances in Candida albicans biofilms. Mycopathologia 2005, 159, 353–360. [Google Scholar] [CrossRef] [PubMed]

| Bacterial adherence classification of Essix® specimens | |||||
|---|---|---|---|---|---|
| OD | ≤ | 0.0417 | Non-adherent (−) | ||
| 0.0417 | < | OD | ≤ | 0.0833 | Weakly adhearent (+) |
| 0.0883 | < | OD | ≤ | 0.1668 | Moderately adhearent (++) |
| 0.1668 | < | OD | Strongly adhearent (+++) | ||
| Bacterial adherence classification of Vivera® specimens | |||||
| OD | ≤ | 0.0664 | Non-adherent (−) | ||
| 0.0664 | < | OD | ≤ | 0.1328 | Weakly adhearent (+) |
| 0.1328 | < | OD | ≤ | 0.2656 | Moderately adhearent (++) |
| 0.2656 | < | OD | Strongly adhearent (+++) | ||
| Group (Essix®) | Mean OD | Level | Group (Vivera®) | Mean OD | Level |
|---|---|---|---|---|---|
| Neg | 0.0345 | - | Neg | 0.0454 | - |
| Pos | 0.4295 | +++ | Pos | 0.7799 | +++ |
| CHX | 0.0672 | + | CHX | 0.0817 | + |
| AA | 0.1626 | ++ | AA | 0.3736 | +++ |
| PD | 0.0830 | + | PD | 0.1649 | ++ |
| PR | 0.0834 | + | PR | 0.1086 | + |
| Agent | Neg | Pos | CHX | AA | PD | PR |
|---|---|---|---|---|---|---|
| Neg | 1.000 | 0.000 a | 0.010 a | 0.000 a | 0.000 a | 0.000 a |
| Pos | 0.000 a | 1.000 | 0.000 a | 0.027 a | 0.000 a | 0.000 a |
| CHX | 0.010 a | 0.000 a | 1.000 | 0.000 a | 0.048 a | 0.077 |
| AA | 0.000 a | 0.027 a | 0.000 a | 1.000 | 0.007 a | 0.005 a |
| PD | 0.000 a | 0.000 a | 0.048 a | 0.007 a | 1.000 | 0.847 |
| PR | 0.000 a | 0.000 a | 0.077 | 0.005 a | 0.847 | 1.000 |
| Agent | Neg | Pos | CHX | AA | PD | PR |
|---|---|---|---|---|---|---|
| Neg | 1.000 | 0.000 a | 0.004 a | 0.000 a | 0.000 a | 0.000 a |
| Pos | 0.000 a | 1.000 | 0.000 a | 0.107 | 0.000 a | 0.000 a |
| CHX | 0.004 a | 0.000 a | 1.000 | 0.000 a | 0.095 | 0.449 |
| AA | 0.000 a | 0.107 | 0.000 a | 1.000 | 0.021 a | 0.001 a |
| PD | 0.000 a | 0.000 a | 0.095 | 0.021 a | 1.000 | 0.353 |
| PR | 0.000 a | 0.000 a | 0.449 | 0.001 a | 0.353 | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kiatwarawut, K.; Kuvatanasuchati, J.; Thaweboon, B.; Sirisoontorn, I. Comparison of Various Antimicrobial Agents for Thermoplastic Polymeric Retainers. Polymers 2022, 14, 3753. https://doi.org/10.3390/polym14183753
Kiatwarawut K, Kuvatanasuchati J, Thaweboon B, Sirisoontorn I. Comparison of Various Antimicrobial Agents for Thermoplastic Polymeric Retainers. Polymers. 2022; 14(18):3753. https://doi.org/10.3390/polym14183753
Chicago/Turabian StyleKiatwarawut, Kanket, Jintakorn Kuvatanasuchati, Boonyanit Thaweboon, and Irin Sirisoontorn. 2022. "Comparison of Various Antimicrobial Agents for Thermoplastic Polymeric Retainers" Polymers 14, no. 18: 3753. https://doi.org/10.3390/polym14183753
APA StyleKiatwarawut, K., Kuvatanasuchati, J., Thaweboon, B., & Sirisoontorn, I. (2022). Comparison of Various Antimicrobial Agents for Thermoplastic Polymeric Retainers. Polymers, 14(18), 3753. https://doi.org/10.3390/polym14183753








