Electrospinning of Biomedical Nanofibers/Nanomembranes: Effects of Process Parameters
Abstract
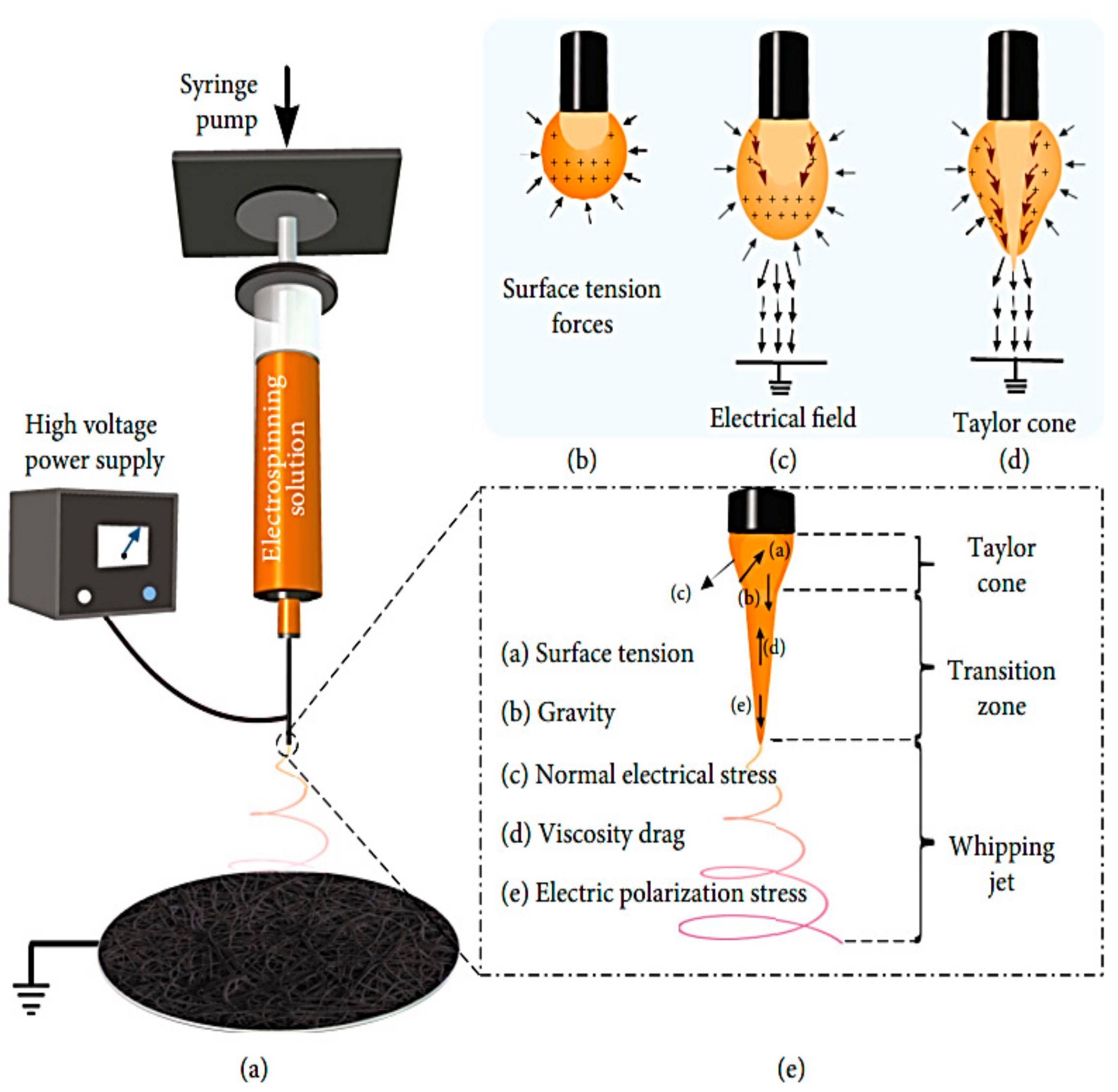
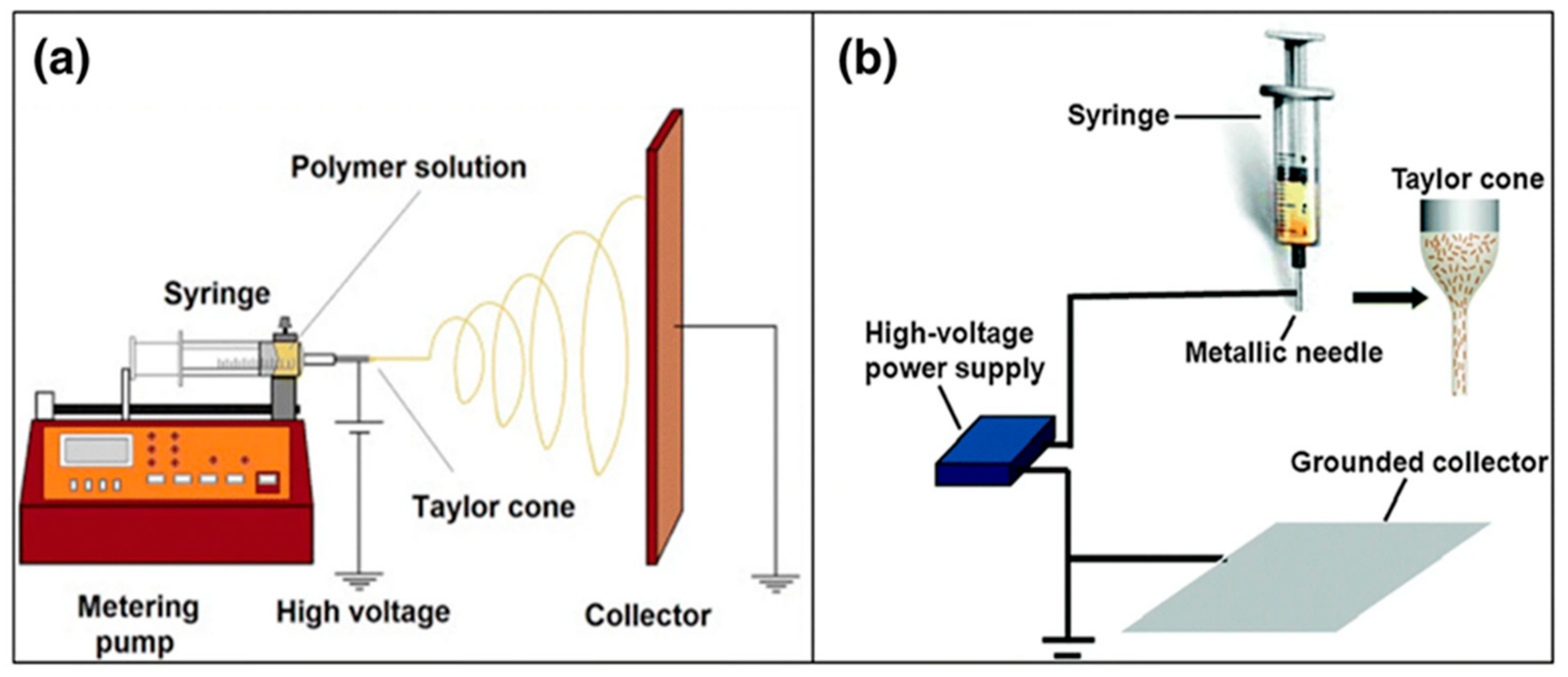
1. Introduction: Process Overview
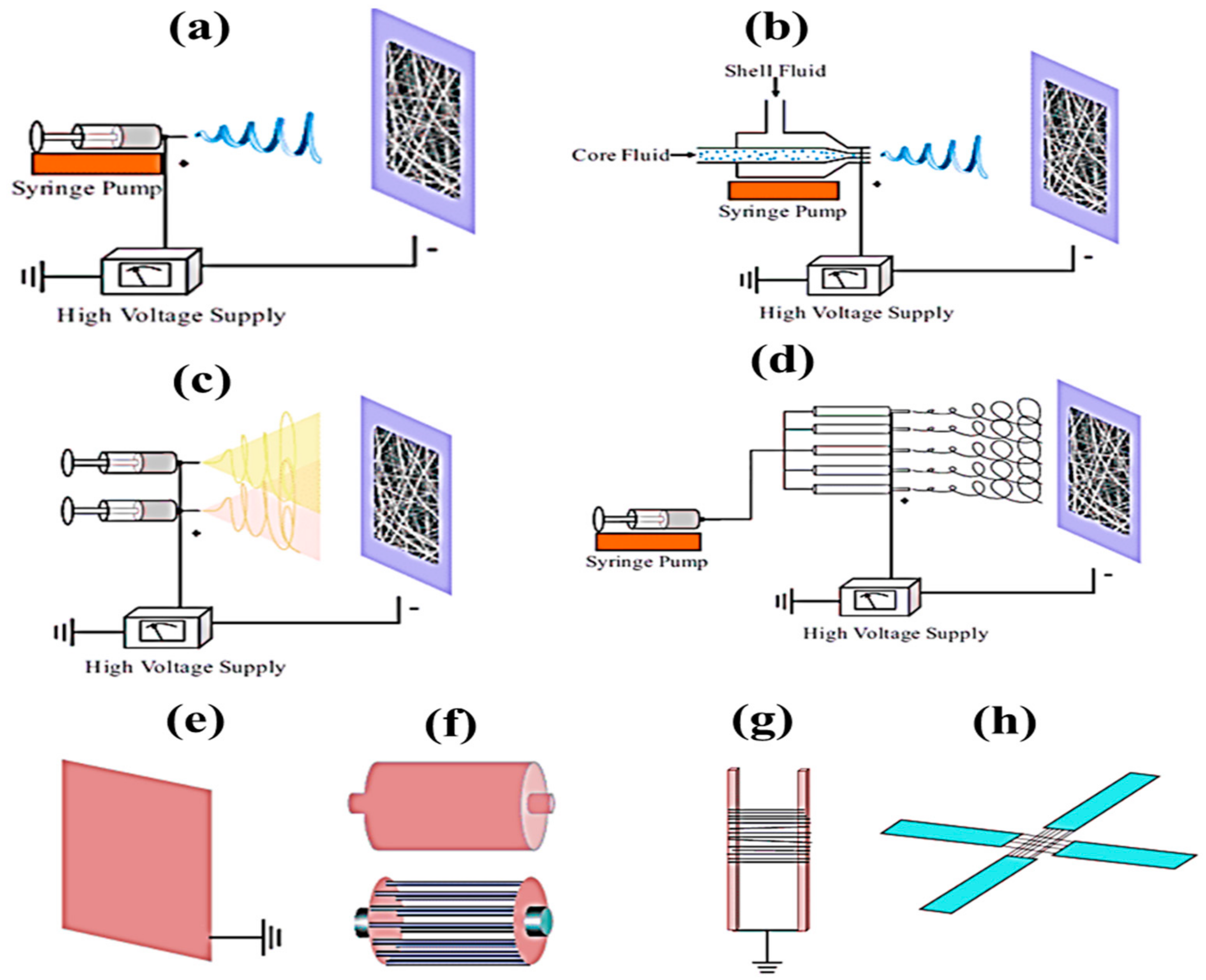
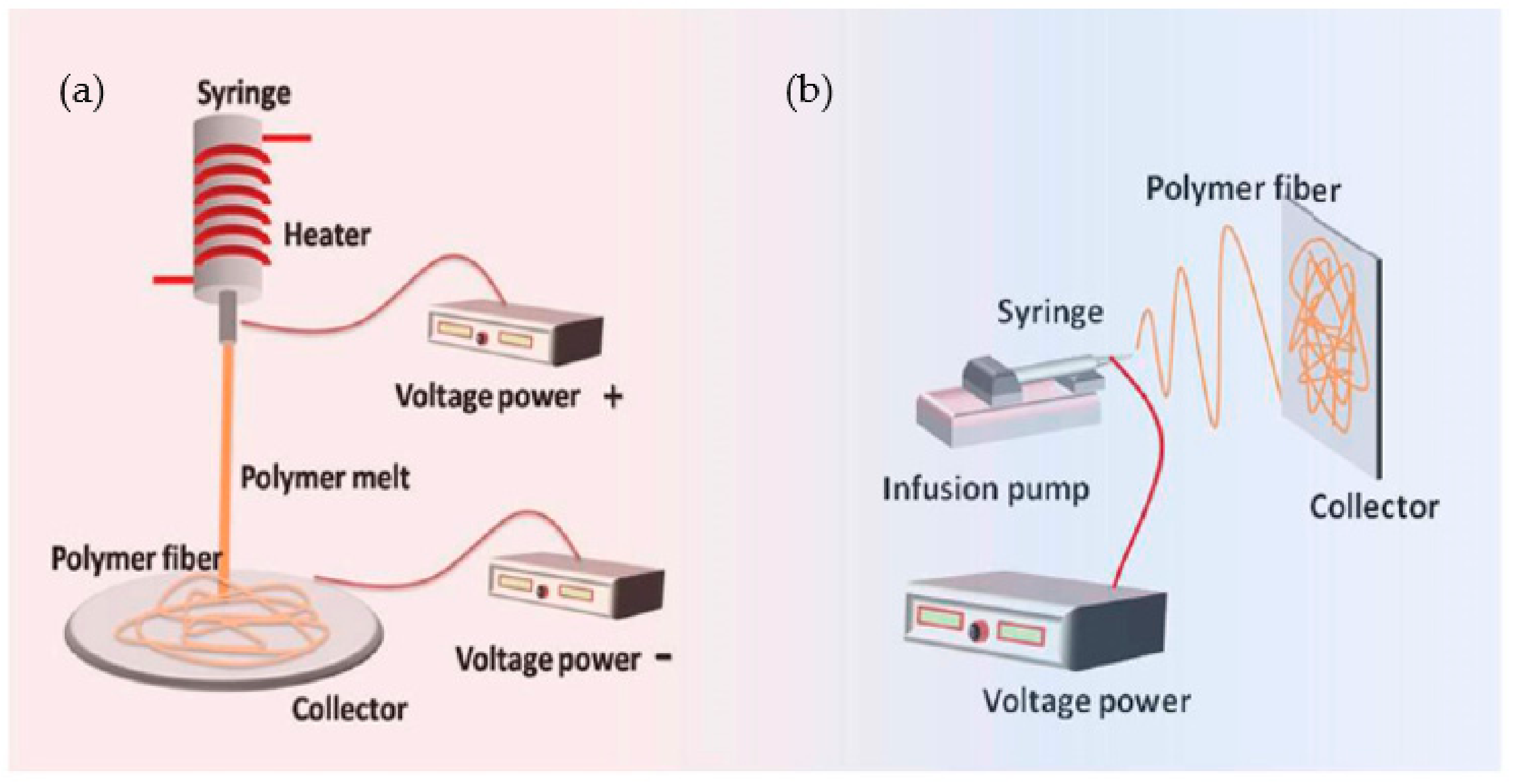
Methods of Electrospinning
2. Effects of Process Parameters
2.1. Electrospinning Solution Parameters
2.1.1. Electrospinning Polymeric Solution Concentrations
2.1.2. Molecular Weight of the Polymeric Solution
2.1.3. Polymeric Solution: Viscosity
2.1.4. Polymeric Solution Surface Tension
2.1.5. Polymeric Solution: Surface Charge Density/Conductivity
2.2. Process Parameters of Electrospinning
2.2.1. Polymeric Solution: Voltage
2.2.2. Solution’s Flow Rate
2.2.3. Role of Collectors
2.2.4. Role of Distance b/w Collector Tip of the Syringe
2.3. Ambient Parameters
2.3.1. Polymeric Solution: Temperature
2.3.2. Humidity
2.3.3. Air Flow
3. Surface Modification of the Electrospun Nanofiber
4. Electrospinning: Applications
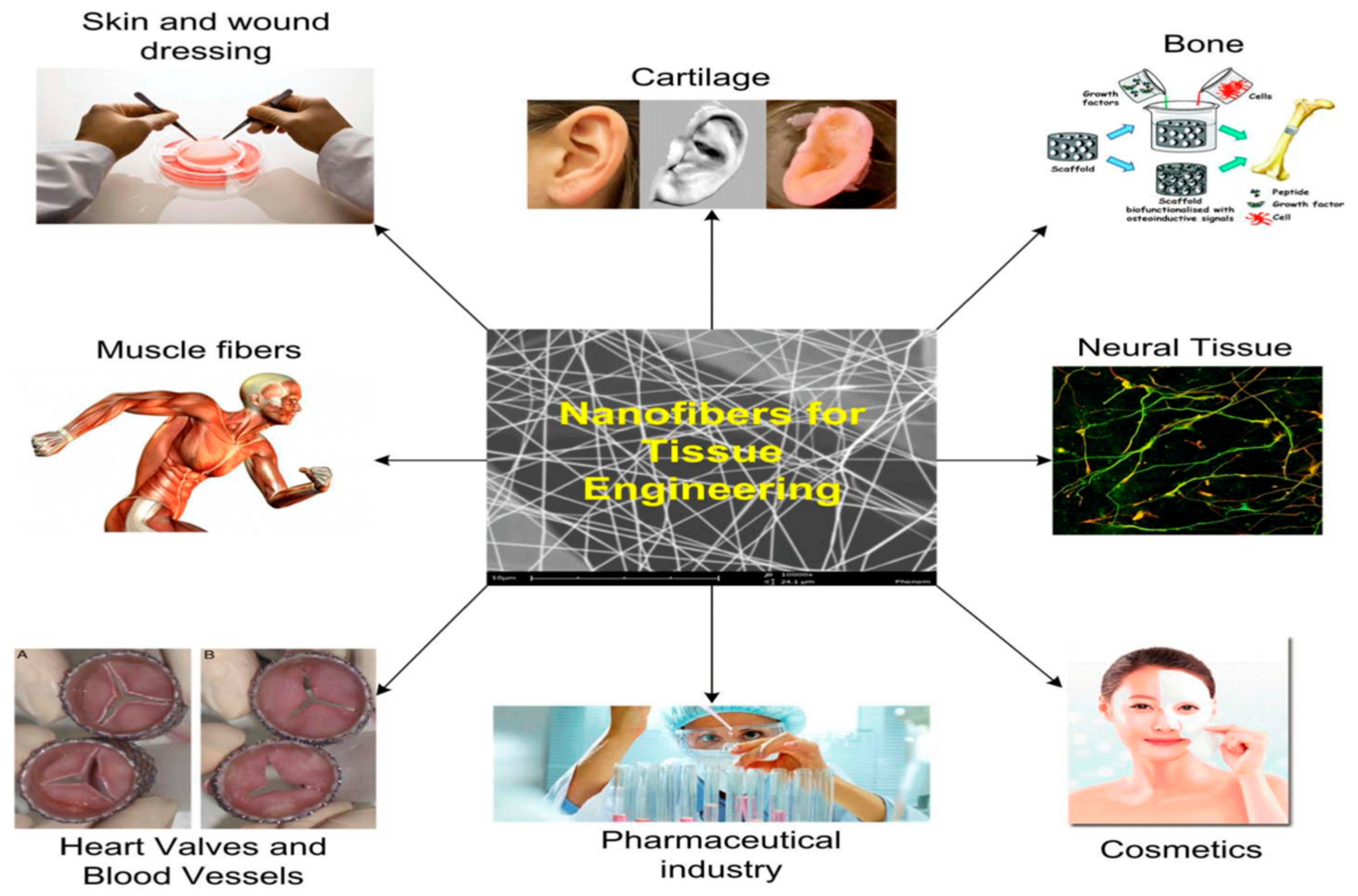
4.1. Medical: Tissue Engineering
4.2. Renewable Energy
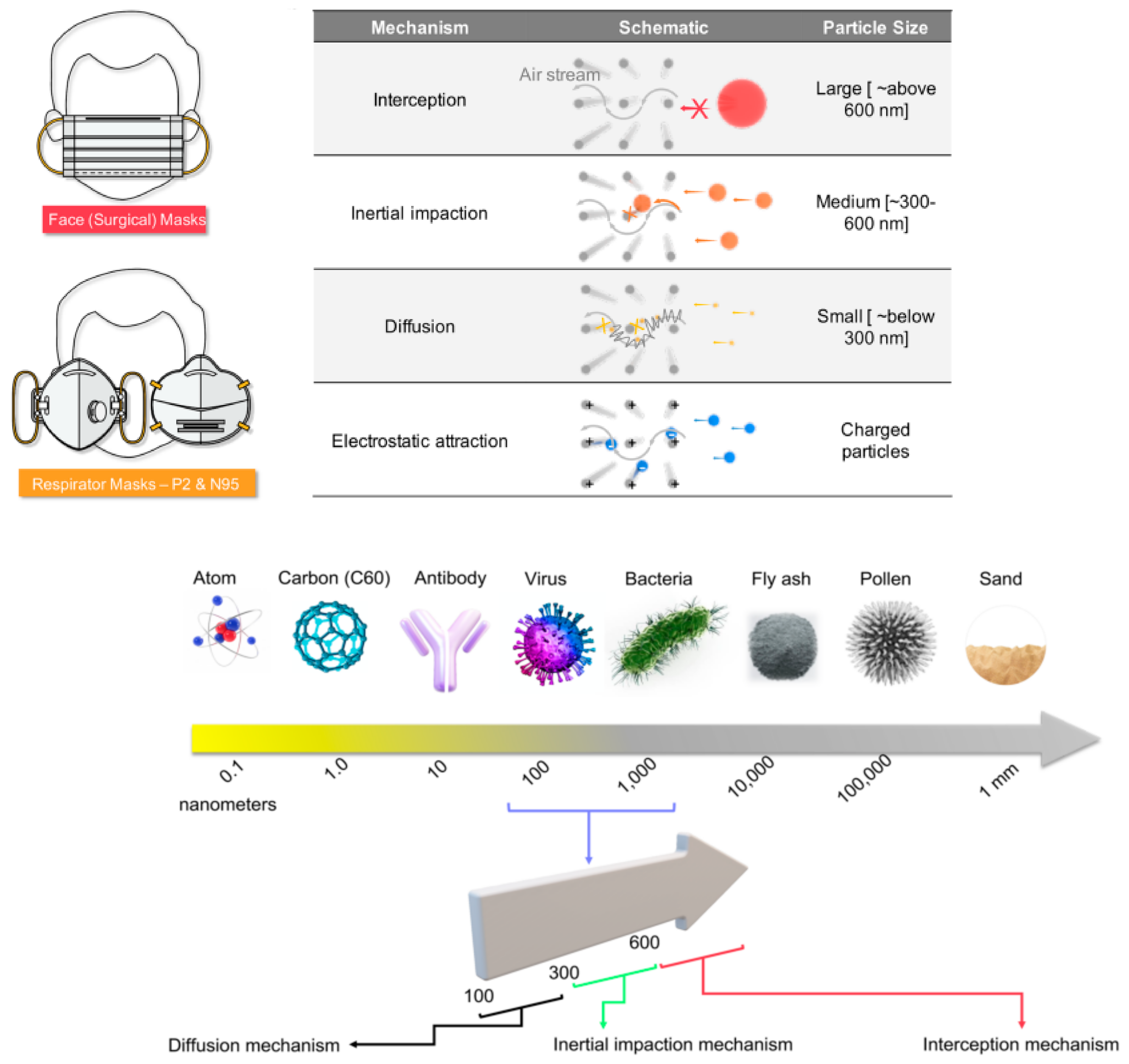
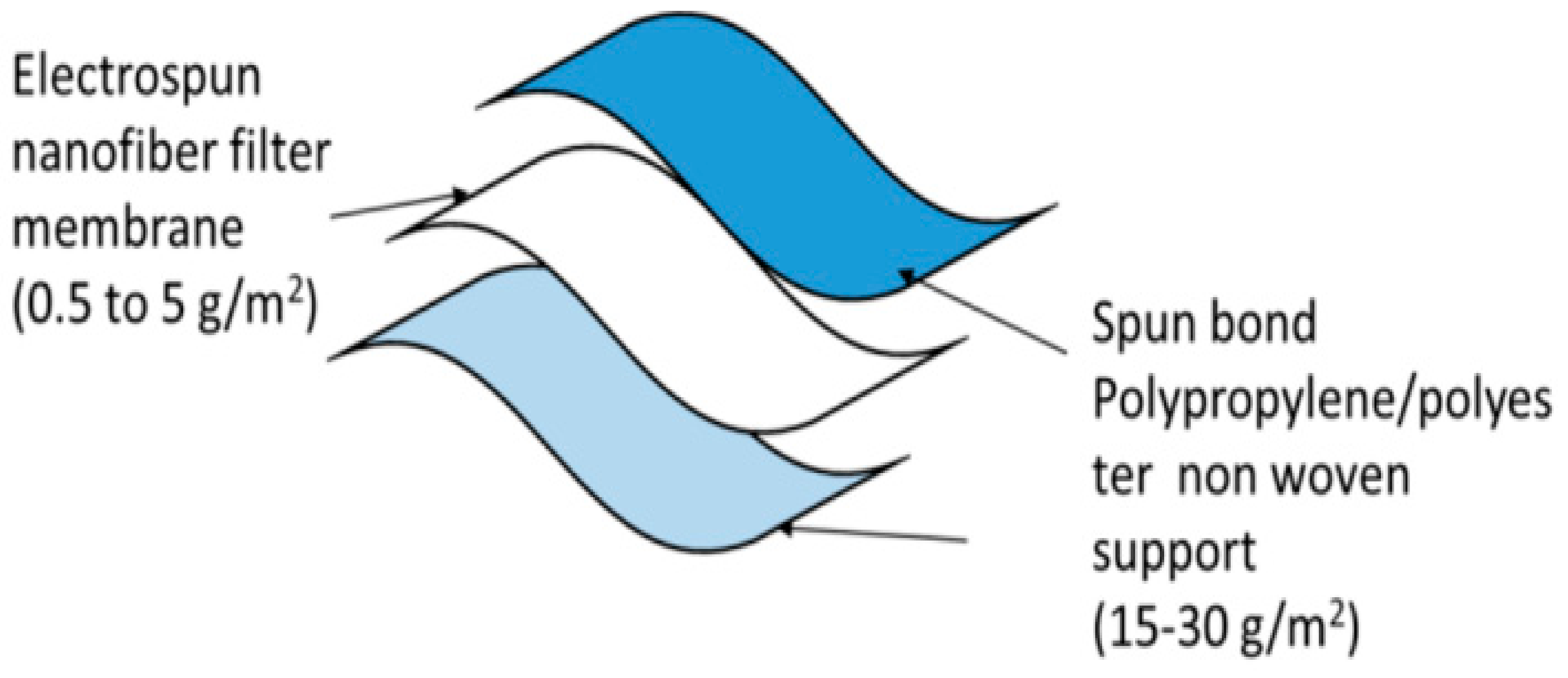
4.3. Face Mask
4.4. Melt Electrospinning
4.5. Melt Electrowriting
4.6. Wound Dressing
5. Conclusions and Future Directions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hoque, M.E.; Ramasamy, T.; Nuge, T. Hybrid Nanofibre Matrix for Regenerative Therapy Fabricated by Electrospinning: Effects of Process Parameters on the Fibre Efficacy. J. Nanomed. Biother. Discov. 2014, 4, 1. [Google Scholar]
- Min, L.L.; Yuan, Z.H.; Zhong, L.B.; Liu, Q.; Wu, R.X.; Zheng, Y.M. Preparation of chitosan based electrospun nanofiber membrane and its adsorptive removal of arsenate from aqueous solution. Chem. Eng. J. 2015, 267, 132–141. [Google Scholar] [CrossRef]
- Pradeep, S.A.; Prem Kumar, G.; Phani, A.R.; Prasad, R.G.S.V.; Enamul Hoque, M.; Raghavendra, H.L. Fabrication, characterization and in vitro osteogenic potential of polyvinyl pyrrolidone-titania (PVP-TiO) nanofibers. Anal. Chem. Lett. 2015, 5, 61–72. [Google Scholar] [CrossRef]
- Jiang, W.; Li, L.; Zhang, D.; Huang, S.; Jing, Z.; Wu, Y.; Zhao, Z.; Zhao, L.; Zhou, S. Incorporation of aligned PCL-PEG nanofibers into porous chitosan scaffolds improved the orientation of collagen fibers in regenerated periodontium. Acta Biomater. 2015, 25, 240–252. [Google Scholar] [CrossRef]
- Hoque, M.E.; Nuge, T.; Yeow, T.K.; Nordi, N. Electrospun matrices from natural polymers for skin regeneration. In Nanostructured Polymer Composites for Biomedical Applications; Elsevier: Amsterdam, The Netherlands, 2019; pp. 87–104. [Google Scholar] [CrossRef]
- Majumder, S.; Sharif, A.; Hoque, M.E. Electrospun cellulose acetate nanofiber: Characterization and applications. In Advanced Processing, Properties, and Applications of Starch and other Bio-Based Polymers; Elsevier: Amsterdam, The Netherlands, 2020; pp. 139–155. [Google Scholar]
- Nuge, T.; Tshai, K.Y.; Lim, S.S.; Nordin, N.; Hoque, E. Characterization and optimization of the mechanical properties of electrospun gelatin nanofibrous scaffolds. World J. Eng. 2020, 17, 12–20. [Google Scholar] [CrossRef]
- Ulubayram, K.; Calamak, S.; Shahbazi, R.; Eroglu, I. Nanofibers based antibacterial drug design, delivery and applications. Curr. Pharm. Des. 2015, 21, 1930–1943. [Google Scholar] [CrossRef]
- Topuz, F.; Uyar, T. Electrospinning of cyclodextrin nanofibers: The effect of process parameters. J. Nanomater. 2020, 2020, 7529306. [Google Scholar] [CrossRef]
- Kopp, A.; Smeets, R.; Kroger, N.; Fuest, S.; Kopf, M.; Kruse, M.; Krieger, J.; Rutkowski, R.; Henningsen, A.; Burg, S. Effect of process parameters on additive-free electrospinning of regenerated silk fibroin nonwovens. Bioact. Mater. 2020, 5, 241–252. [Google Scholar] [CrossRef]
- Jamnongkan, T.; Shirota, R.; Sukumaran, S.K.; Sugimoto, M.; Koyama, K. Effect of ZnO nanoparticles on the electrospinning of poly (vinyl alcohol) from aqueous solution: Influence of particle size. Polym. Eng. Sci. 2014, 54, 1969–1975. [Google Scholar] [CrossRef]
- Celebioglu, A.; Uyar, T. Design of polymer-free Vitamin-A acetate/cyclodextrin nanofibrous webs: Antioxidant and fast-dissolving properties. Food Funct. 2020, 11, 7626–7637. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Jiang, J.; Li, X.; Ye, F.; Fu, Y.; Zhao, L. Electrospun polymer-free nanofibers incorporating hydroxypropyl-β-cyclodextrin/Difenoconazole via supramolecular assembly for antifungal activity. J. Agric. Food Chem. 2021, 69, 5871–5881. [Google Scholar] [CrossRef] [PubMed]
- Rodoplu, D.; Mutlu, M. Effects of electrospinning setup and process parameters on nanofiber morphology intended for the modification of quartz crystal microbalance surfaces. J. Eng. Fibers Fabr. 2012, 7, 155892501200700217. [Google Scholar] [CrossRef]
- Theron, S.A.; Zussman, E.; Yarin, A.L. Experimental investigation of the governing parameters in the electrospinning of polymer solutions. Polymer 2004, 45, 2017–2030. [Google Scholar] [CrossRef]
- Jamnongkan, T.; Wattanakornsiri, A.; Pansila, P.P.; Migliaresi, C.; Kaewpirom, S. Effect of poly (vinyl alcohol)/chitosan ratio on electrospun-nanofiber morphologies. In Advanced Materials Research; Trans Tech Publications Ltd.: Bäch, Switzerland, 2012; Volume 463, pp. 734–738. [Google Scholar]
- Jamnongkan, T.; Sukumaran, S.K.; Sugimoto, M.; Hara, T.; Takatsuka, Y.; Koyama, K. Towards novel wound dressings: Antibacterial properties of zinc oxide nanoparticles and electrospun fiber mats of zinc oxide nanoparticle/poly (vinyl alcohol) hybrids. J. Polym. Eng. 2015, 35, 575–586. [Google Scholar] [CrossRef]
- Narayanan, G.; Shen, J.; Boy, R.; Gupta, B.S.; Tonelli, A.E. Aliphatic polyester nanofibers functionalized with cyclodextrins and cyclodextrin-guest inclusion complexes. Polymers 2018, 10, 428. [Google Scholar] [CrossRef]
- Ray, S.S.; Chen, S.-S.; Nguyen, N.C.; Nguyen, H.T. Electrospinning: A versatile fabrication technique for nanofibrous membranes for use in desalination. In Nanoscale Materials in Water Purification; Elsevier: Amsterdam, The Netherlands, 2019; pp. 247–273. [Google Scholar] [CrossRef]
- Lee, K.; Kim, H.; Bang, H.; Jung, Y.; Lee, S. The change of bead morphology formed on electrospun polystyrene fibers. Polymer 2003, 44, 4029–4034. [Google Scholar] [CrossRef]
- Li, W.; Danielson, K.G.; Alexander, P.G.; Tuan, R.S. Biological response of chondrocytes cultured in three-dimensional nanofibrous poly (ϵ-caprolactone) scaffolds. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2003, 67, 1105–1114. [Google Scholar] [CrossRef]
- Eda, G.; Shivkumar, S. Bead-to-fiber transition in electrospun polystyrene. J. Appl. Polym. Sci. 2007, 106, 475–487. [Google Scholar] [CrossRef]
- Koski, A.; Yim, K.; Shivkumar, S.J.M.L. Effect of molecular weight on fibrous PVA produced by electrospinning. Mater. Lett. 2004, 58, 493–497. [Google Scholar] [CrossRef]
- Haider, A.; Haider, S.; Kummara, M.R.; Kamal, T.; Alghyamah, A.-A.A.; Iftikhar, F.J.; Bano, B.; Khan, N.; Afridi, M.A.; Han, S.S.; et al. Advances in the scaffolds fabrication techniques using biocompatible polymers and their biomedical application: A technical and statistical review. J. Saudi Chem. Soc. 2020, 24, 186–215. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, J.; Cheng, H.; Li, G.; Cho, H.; Jiang, M.; Gao, Q.; Zhang, X. Developments of advanced electrospinning techniques: A critical review. Adv. Mater. Technol. 2021, 6, 2100410. [Google Scholar] [CrossRef]
- Lee, J.S.; Choi, K.H.; Ghim, H.D.; Kim, S.S.; Chun, D.H.; Kim, H.Y.; Lyoo, W.S. Role of molecular weight of atactic poly (vinyl alcohol) (PVA) in the structure and properties of PVA nanofabric prepared by electrospinning. J. Appl. Polym. Sci. 2004, 93, 1638–1646. [Google Scholar] [CrossRef]
- Mazoochin, T.; Hamadanian, M.; Ahmadi, M.; Jabbari, V. Investigation on the morphological characteristics of nanofiberous membrane as electrospun in the different processing parameters. Int. J. Ind. Chem. 2012, 3, 2. [Google Scholar] [CrossRef]
- Sill, T.J.; Von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef]
- Zargham, S.; Bazgir, S.; Tavakoli., A.; Rashidi, A.S.; Damerchely, R. The effect of flow rate on morphology and deposition area of electrospun nylon 6 nanofiber. J. Eng. Fibers Fabr. 2012, 7, 155892501200700414. [Google Scholar] [CrossRef]
- Milleret, V.; Simona, B.; Neuenschwander, P.; Hall, H. Tuning electrospinning parameters for production of 3D-fiber-fleeces with increased porosity for soft tissue engineering applications. Eur. Cell Mater. 2011, 21, 286–303. [Google Scholar] [CrossRef]
- Wang, C.; Chien, H.S.; Yan, K.W.; Hung, C.L.; Hung, K.L.; Tsai, S.J.; Jhang, H.J. Correlation between processing parameters and microstructure of electrospun poly (D, L-lactic acid) nanofibers. Polymer 2009, 50, 6100–6110. [Google Scholar] [CrossRef]
- Chowdhury, M.; Stylios, G. Effect of experimental parameters on the morphology of electrospun Nylon 6 fibres. Int. J. Basic Appl. Sci. 2010, 10, 70–78. [Google Scholar]
- De Vrieze, S.; Van Camp, T.; Nelvig, A.; Hagström, B.; Westbroek, P.; De Clerck, K. The effect of temperature and humidity on electrospinning. J. Mater. Sci. 2009, 44, 1357–1362. [Google Scholar] [CrossRef]
- Lin, K.; Chua, K.N.; Christopherson, G.T.; Lim, S.; Mao, H.Q. Reducing electrospun nanofiber diameter and variability using cationic amphiphiles. Polymer 2007, 48, 6384–6394. [Google Scholar] [CrossRef]
- Raksa, A.; Numpaisal, P.O.; Ruksakulpiwat, Y. The effect of humidity during electrospinning on morphology and mechanical properties of SF/PVA nanofibers. Mater. Today Proc. 2021, 47, 3458–3461. [Google Scholar] [CrossRef]
- Cui, W.; Li, X.; Zhou, S.; Weng, J. Investigation on process parameters of electrospinning system through orthogonal experimental design. J. Appl. Polym. Sci. 2007, 103, 3105–3112. [Google Scholar] [CrossRef]
- Park, K.; Ju, Y.M.; Son, J.S.; Ahn, K.; Han, D.K. Surface modification of biodegradable electrospun nanofiber scaffolds and their interaction with fibroblasts. J. Biomater. Sci. Polym. Ed. 2007, 18, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.C.; Thomas, V.; Schmidt, G.; Vohra, Y.K.; Chu, T.-M.G.; Kowolik, M.J.; Janowski, G.M. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—A materials perspective. Dent. Mater. 2012, 28, 703–721. [Google Scholar] [CrossRef]
- Oprea, A.E.; Ficai, A.; Andronescu, E. Electrospun nanofibers for tissue engineering applications. In Materials for Biomedical Engineering; Elsevier: Amsterdam, The Netherlands, 2019; pp. 77–95. [Google Scholar] [CrossRef]
- Han, D.; Gouma, P.I. Electrospun bioscaffolds that mimic the topology of extracellular matrix. In Nanomedicine in Cancer; Jenny Stanford Publishing: Singapore, 2017; pp. 159–169. [Google Scholar]
- Khorshidi, S.; Solouk, A.; Mazinani, S.; Mirzadeh, H. A Review on Different Approaches for Improving Cell Infiltration in Electrospun Nanofibrous Scaffolds. Pathobiol. Res. 2016, 18, 1–22. [Google Scholar]
- Lannutti, J.; Reneker, D.; Ma, T.; Tomasko, D.; Farson, D. Electrospinning for tissue engineering scaffolds. Mater. Sci. Eng. C 2007, 27, 504–509. [Google Scholar] [CrossRef]
- Liu, H.; Ding, X.; Zhou, G.; Li, P.; Wei, X.; Fan, Y. Electrospun nanofibers in tissue engineering. In Nanofibers—Production, Properties and Functional Applications; InTech Open: London, UK, 2011; pp. 347–373. [Google Scholar] [CrossRef]
- Li, Y.; BOU-AKL, T. Electrospinning in tissue engineering. Electrospinn.-Mater. Tech. Biomed. Appl. 2016, 3, 117–139. [Google Scholar]
- James, R.; Kumbar, S.G.; Laurencin, C.T.; Balian, G.; Chhabra, A.B. Tendon tissue engineering: Adipose-derived stem cell and GDF-5 mediated regeneration using electrospun matrix systems. Biomed. Mater. 2011, 6, 025011. [Google Scholar] [CrossRef] [PubMed]
- Laranjeira, M.; Domingues, R.; Costa-Almeida, R.; Reis, R.L.; Gomes, M.E. 3D mimicry of native-tissue-fiber architecture guides tendon-derived cells and adipose stem cells into artificial tendon constructs. Small 2017, 13, 1700689. [Google Scholar] [CrossRef]
- Taylor, E.D.; Nair, L.S.; Nukavarapu, S.P.; McLaughlin, S.; Laurencin, C.T. AAOS supplement selected scientific exhibits: Novel nanostructured scaffolds as therapeutic replacement options for rotator cuff disease. J. Bone Jt. Surgery. Am. Vol. 2010, 92 (Suppl. 2), 170–179. [Google Scholar]
- Hiremath, N.; Bhat, G. Melt blown polymeric nanofibers for medical applications-an overview. Nanosci. Technol. 2015, 2, 1–9. [Google Scholar]
- Kim, K.; Luu, Y.K.; Chang, C.; Fang, D.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Incorporation and controlled release of a hydrophilic antibiotic using poly (lactide-co-glycolide)-based electrospun nanofibrous scaffolds. J. Control. Release 2004, 98, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Thavasi, V.; Singh, G.; Ramakrishna, S. Electrospun nanofibers in energy and environmental applications. Energy Environ. Sci. 2008, 1, 205–221. [Google Scholar] [CrossRef]
- Bazilevsky, A.V.; Yarin, A.L.; Megaridis, C.M. Co-electrospinning of core—Shell fibers using a single-nozzle technique. Langmuir 2007, 23, 2311–2314. [Google Scholar] [CrossRef]
- Xu, X.; Chen, X.; Xu, X.; Lu, T.; Wang, X.; Yang, L.; Jing, X. BCNU-loaded PEG-PLLA ultrafine fibers and their in vitro antitumor activity against Glioma C6 cells. J. Control. Release 2006, 114, 307–316. [Google Scholar] [CrossRef]
- Zeleny, J. The electrical discharge from liquid points, and a hydrostatic method of measuring the electric intensity at their surfaces. Phys. Rev. 1914, 3, 69. [Google Scholar] [CrossRef]
- Kumar, P.S.; Sundaramurthy, J.; Sundarrajan, S.; Babu, V.J.; Singh, G.; Allakhverdiev, S.I.; Ramakrishna, S. Hierarchical electrospun nanofibers for energy harvesting, production and environmental remediation. Energy Environ. Sci. 2014, 7, 3192–3222. [Google Scholar] [CrossRef]
- Chapin, D.M.; Fuller, C.S.; Pearson, G.L. A new silicon pn junction photocell for converting solar radiation into electrical power. In Semiconductor Devices: Pioneering Papers; World Scientific: London, UK, 1991; pp. 969–970. [Google Scholar] [CrossRef]
- Tebyetekerwa, M.; Xu, Z.; Yang, S.; Ramakrishna, S. Electrospun nanofibers-based face masks. Adv. Fiber Mater. 2020, 2, 161–166. [Google Scholar] [CrossRef]
- Chua, M.H.; Cheng, W.; Goh, S.S.; Kong, J.; Li, B.; Lim, J.Y.C.; Mao, L.; Wang, S.; Xue, K.; Yang, L.; et al. Face masks in the new COVID-19 normal: Materials, testing, and perspectives. Research 2020, 2020, 7286735. [Google Scholar] [CrossRef] [PubMed]
- Naragund, V.S.; Panda, P.K. Electrospun nanofiber-based respiratory face masks—A review. Emerg. Mater. 2022, 5, 261–278. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, H.; Xiong, J.; Li, J.; Miao, X.; Lan, X.; Liu, X.; Wang, W.; Cai, N.; Tang, Y. Fabrication and in vitro evaluation of PCL/gelatin hierarchical scaffolds based on melt electrospinning writing and solution electrospinning for bone regeneration. Mater. Sci. Eng. C 2021, 128, 112287. [Google Scholar] [CrossRef]
- Dalton, P.D. Melt Electrowriting with additive manufacturing principles. Curr. Opin. Biomed. Eng. 2017, 2, 49–57. [Google Scholar] [CrossRef]
- Robinson, T.M.; Hutmacher, D.W.; Dalton, P.D. The next frontier in melt electrospinning: Taming the jet. Adv. Funct. Mater. 2019, 29, 1904664. [Google Scholar] [CrossRef]
- Sarwar, Z.; Krugly, E.; Danilovas, P.P.; Ciuzas, D.; Kauneliene, V.; Martuzevicius, D. Fabrication and characterization of PEBA fibers by melt and solution electrospinning. J. Mater. Res. Technol. 2019, 8, 6074–6085. [Google Scholar] [CrossRef]
- Lian, H.; Zhaoxu, M. Melt electrospinning vs. solution electrospinning: A comparative study of drug-loaded poly (ε-caprolactone) fibres. Mater. Sci. Eng. C 2017, 74, 117–123. [Google Scholar] [CrossRef]
- Buivydiene, D.; Todea, A.M.; Asbach, C.; Krugly, E.; Martuzevicius, D.; Kliucininkas, L. Composite micro/nano fibrous air filter by simultaneous melt and solution electrospinning. J. Aerosol Sci. 2021, 154, 105754. [Google Scholar] [CrossRef]
- Semjonov, K.; Lust, A.; Kogermann, K.; Laidmäe, I.; Maunu, S.L.; Hirvonen, S.-P.; Yliruusi, J.; Nurk, G.; Lust, E.; Heinämäki, J. Melt-electrospinning as a method to improve the dissolution and physical stability of a poorly water-soluble drug. Eur. J. Pharm. Sci. 2018, 121, 260–268. [Google Scholar] [CrossRef]
- Govinna, N.D.; Keller, T.; Schick, C.; Cebe, P. Melt-electrospinning of poly (ether ether ketone) fibers to avoid sulfonation. Polymer 2019, 171, 50–57. [Google Scholar] [CrossRef]
- Daneshfar, A.; Dumée, L.F.; Hughes, T.C.; Kong, L. Thermally-Stable Photo-Curing Chemistry for Additive Manufacturing by Direct Melt Electrowriting. Addit. Manuf. 2022, 51, 102623. [Google Scholar] [CrossRef]
- Brooks-Richards, T.L.; Paxton, N.C.; Allenby, M.C.; Woodruff, M.A. Dissolvable 3D printed PVA moulds for melt electrowriting tubular scaffolds with patient-specific geometry. Mater. Des. 2022, 215, 110466. [Google Scholar] [CrossRef]
- Liu, Y.; Li, T.; Han, Y.; Li, F.; Liu, Y. Recent development of electrospun wound dressing. Curr. Opin. Biomed. Eng. 2021, 17, 100247. [Google Scholar] [CrossRef]
- Arampatzis, A.S.; Kontogiannopoulos, K.N.; Theodoridis, K.; Aggelidou, E.; Rat, A.; Willems, A.; Tsivintzelis, I.; Papageorgiou, V.P.; Kritis, A.; Assimopoulou, A.N. Electrospun wound dressings containing bioactive natural products: Physico-chemical characterization and biological assessment. Biomater. Res. 2021, 25, 23. [Google Scholar] [CrossRef]
- Kozior, T.; Mamun, A.; Trabelsi, M.; Wortmann, M.; Lilia, S.; Ehrmann, A. Electrospinning on 3D Printed Polymers for Mechanically Stabilized Filter Composites. Polymers 2019, 11, 2034. [Google Scholar] [CrossRef]
- Jamnongkan, T.; Jaroensuk, O.; Khankhuean, A.; Laobuthee, A.; Srisawat, N.; Pangon, A.; Mongkholrattanasit, R.; Phuengphai, P.; Wattanakornsiri, A.; Huang, C.-F. A Comprehensive Evaluation of Mechanical, Thermal, and Antibacterial Properties of PLA/ZnO Nanoflower Biocomposite Filaments for 3D Printing Application. Polymers 2022, 14, 600. [Google Scholar] [CrossRef]
- Siva, S.; Li, C.; Cui, H.; Meenatchi, V.; Lin, L. Encapsulation of essential oil components with methyl-β-cyclodextrin using ultrasonication: Solubility, characterization, DPPH and antibacterial assay. Ultrason. Sonochem. 2020, 64, 104997. [Google Scholar] [CrossRef]

| Polymers | Solvents | Characterization Methods |
|---|---|---|
| PEO/Lignin | Methanol and DCM | FTIR, SEM, SEM, XRD, DSC, mechanical test, TGA, in vitro and in vivo |
| CS/PEO | Acetic acid/DI water | |
| PEO | DI water | |
| PVA | DI water | |
| PAN/PMMA | DMF | |
| PMMA | DMF | |
| PCL | Acetone DCM/methanol HFP | |
| Gelatin/PCl | TFE | |
| PVP, PLA | Ethanol DCM and DMF | |
| PLGA | Chloroform/DMF | |
| Nylon-6 | FA | |
| PDLLA | Chloroform | |
| PVDF | DMAc DMSO/acetone | |
| PVDF-HFP | DMF/acetone | |
| PPY | DMF | |
| PS | DCM/ethanol DMF/THF | |
| PLI | DMF | |
| PAA | DMAc/DMSO DMEA | |
| Collagen | HFP | |
| CA | HFP acetone/DMAc |
| Solution Parameters | Process Parameters | Ambient Parameters |
|---|---|---|
|
|
|
| Sl. No | Concentration | Formation of Fibrous Structure |
|---|---|---|
| 1 | Concentration level is very low | (i) Micro- or (ii) nanopolymeric fibers |
| 2 | Concentration is a little higher | Combinations of beads and fibers would be formed on macro- and nanofibers |
| 3 | Optimum and most suitable | Smooth macro- and nanofibers |
| 4 | Concentration is very high | Non-nanoscaled and helix-shaped microribbons |
| Sl. No | Molecular Weight (g/mol) | Fibrous Structure | Diameter | Cross section of the Fibers |
|---|---|---|---|---|
| 1. | 9000–13,000 | Unstable and with a bead-on-string structure | 250 nm and 1 Am | Circular |
| 2. | 13,000–23,000 | Fibrous structure is stabilized | 500 nm and 1.25 Am | Circular |
| 3. | 31,000–50,000 | Flat fibers | 1–2 Am | The progressive transition from circular to flat strands |
| 4 | Too high a molecular weight with the low concentration | Microribbon | - | Microribbon |
| 5 | Very high molecular weight | Helical fibers | - | - |
| 6 | Low concentration | Zigzag ribbon | - | - |
| Sl. No | Flow Rate | Fibers’ Shapes | Operating Parameter |
|---|---|---|---|
| 1. | Lower flow rate | Smooth fiber | Flow rates of 0.40 mL/h |
| 2. | Very high flow rate | Thick diameter of bead fibers | Flow rates 0.66 mL/h |
| Sl. No | Solution Parameters | Effects on Fiber | Mechanism |
|---|---|---|---|
| 1 | Conductivity of the Solution | It has been demonstrated that increasing the solution conductivity improves the fiber quality, as seen by fewer beads and a smaller fiber diameter. | Increased solution conductivity results in increased stretching of the solution jet due to the solution’s larger charge-carrying capacity. Additionally, increasing solution conductivity results in an increase in bending instability and a longer jet path. |
| Solution viscosity | Fiber diameter will increase as the viscosity of the fluid increases. | There is a possibility that the charges used to start spinning will not be enough to stretch the solution to reach the target when viscosity increases. | |
| 2 | Solvent volatility | The location with the greatest solvent volatility must be identified. Ribbon/flat fibers and fibers with surface pores may be produced by using a more volatile solvent. | Wet fibers, fused fibers, or no fiber collection can all occur when a solution is made using a solvent with a low volatility. The solidification of the new polymeric solution at the spinneret tip might cause intermittent spinning when the polymer has a high volatility. |
| 3 | Humidity | Larger fiber diameters have been linked to both higher and lower relative humidity levels. | There are two factors that contribute to this: high humidity causes polymer precipitation, preventing fiber elongation; and lower relative humidity causes faster solvent vaporization, which causes an increase in solidification rate that results in larger fiber diameter, thus resulting in thicker fibers. |
| 4 | Temperature | The diameter of the fiber will be smaller at higher temperatures. | Higher temperatures result in quicker solvent evaporation and a lower viscosity of the polymer solution. |
| Sl. No | Present Scenario | Different Suggested Mask | Mask Requirements |
|---|---|---|---|
| 1 | Pedestrians | Cloth/FFP 1,2 with carbon fabric (reusable)/dust mask | Respiratory protection; decrease in particulate matter and volatile organic compounds (VOCs) |
| 2 | Workers in industry | P95/N95 respirator mask with carbon fabric | Respiratory protection is required to reduce aerosols, emissions/VOCs, gaseous |
| 3 | Pandemic | 3-ply fabric mask | speaking or breathing without a valve, reduce the ejection or aerosol, inhalation or cough droplets |
| 4 | Infected persons | Medical/N95 mask | PM, cough droplets, biological aerosol, no valve, respiratory protection, and source management are all addressed in this treatment |
| 5 | Healthcare | Medical mask | Cough droplets and blood are filtered out by biological aerosol |
| 6 | Doctors | Medical N95/surgical respirator mask | Filter cough droplets |
| Sl. No | Melt Electrospinning | Solution Electrospinning | References |
|---|---|---|---|
| 1 | High crystalline and without a burst phase. | Low crystallinity, porous structure, rough, high drug release rate with a burst | [63] |
| 2 | Diameter of fiber is 1.92 ± 3.31 m when TTCD is 60 mm | Diameter fiber of 0.37 ± 0.34 m when TTCD is 200 mm | [62] |
| 3 | With lower filtration efficiencies | Higher filtration efficiencies | [64] |
| 4 | Microfibrous with grid structure | Morphology of random nanofiber | [59] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chinnappan, B.A.; Krishnaswamy, M.; Xu, H.; Hoque, M.E. Electrospinning of Biomedical Nanofibers/Nanomembranes: Effects of Process Parameters. Polymers 2022, 14, 3719. https://doi.org/10.3390/polym14183719
Chinnappan BA, Krishnaswamy M, Xu H, Hoque ME. Electrospinning of Biomedical Nanofibers/Nanomembranes: Effects of Process Parameters. Polymers. 2022; 14(18):3719. https://doi.org/10.3390/polym14183719
Chicago/Turabian StyleChinnappan, Balaji Ayyanar, Marimuthu Krishnaswamy, Huaizhong Xu, and Md Enamul Hoque. 2022. "Electrospinning of Biomedical Nanofibers/Nanomembranes: Effects of Process Parameters" Polymers 14, no. 18: 3719. https://doi.org/10.3390/polym14183719
APA StyleChinnappan, B. A., Krishnaswamy, M., Xu, H., & Hoque, M. E. (2022). Electrospinning of Biomedical Nanofibers/Nanomembranes: Effects of Process Parameters. Polymers, 14(18), 3719. https://doi.org/10.3390/polym14183719







