Thermal Analysis of Parylene Thin Films for Barrier Layer Applications
Abstract
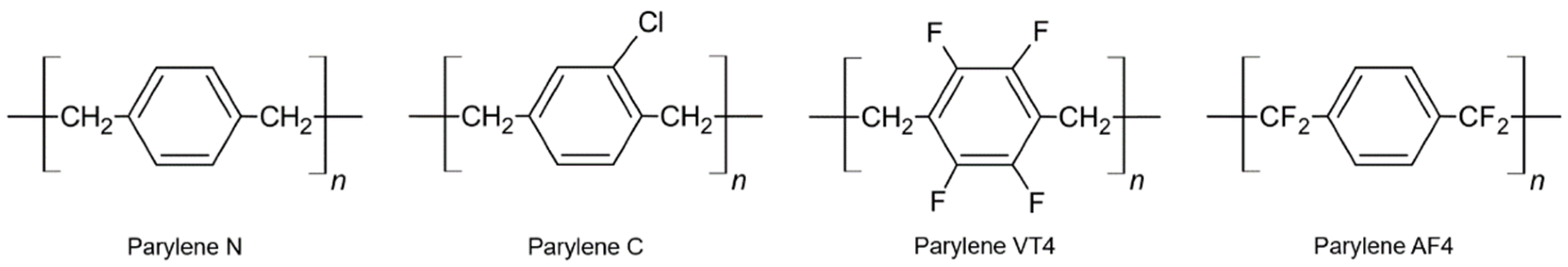
:1. Introduction
2. Materials and Methods
2.1. Parylene Film Deposition
2.2. Heat Treatments
2.3. Differential Scanning Calorimetry
2.4. Thermogravimetric Analysis
2.5. X-ray Diffraction
2.6. Helium Gas Permeation
2.7. Water Vapor Permeation
3. Results
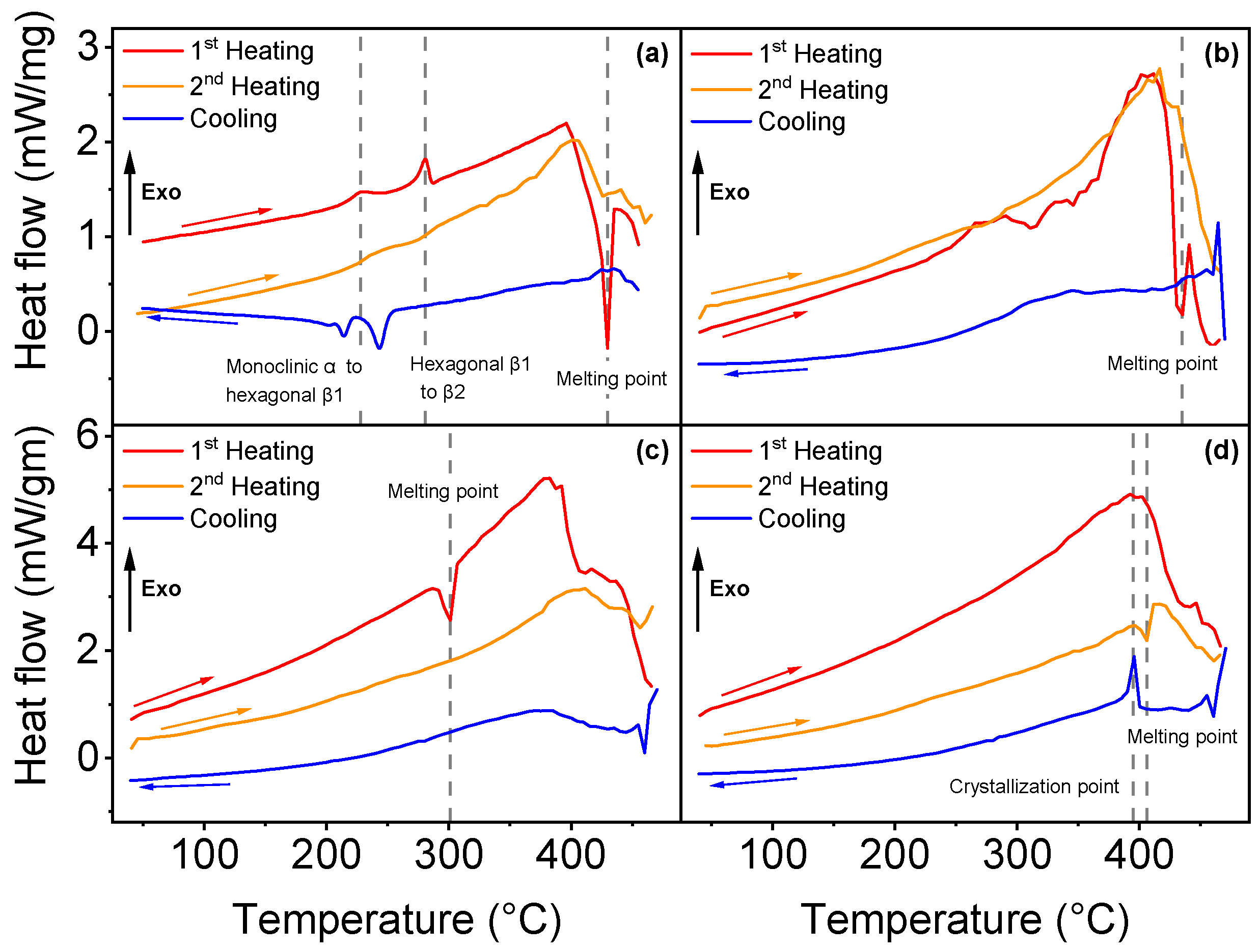
3.1. Differential Scanning Calorimetry
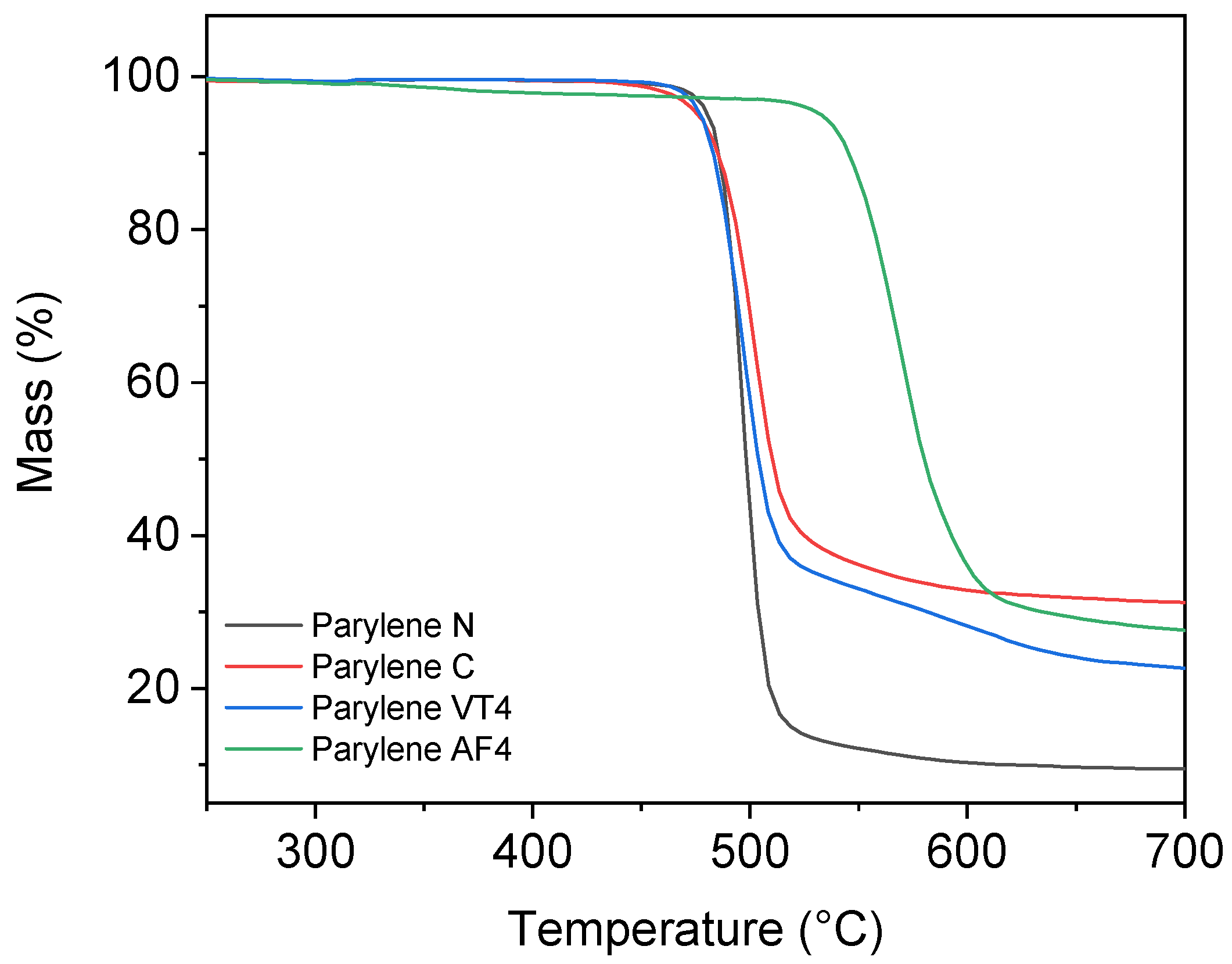
3.2. Thermogravimetric Analysis
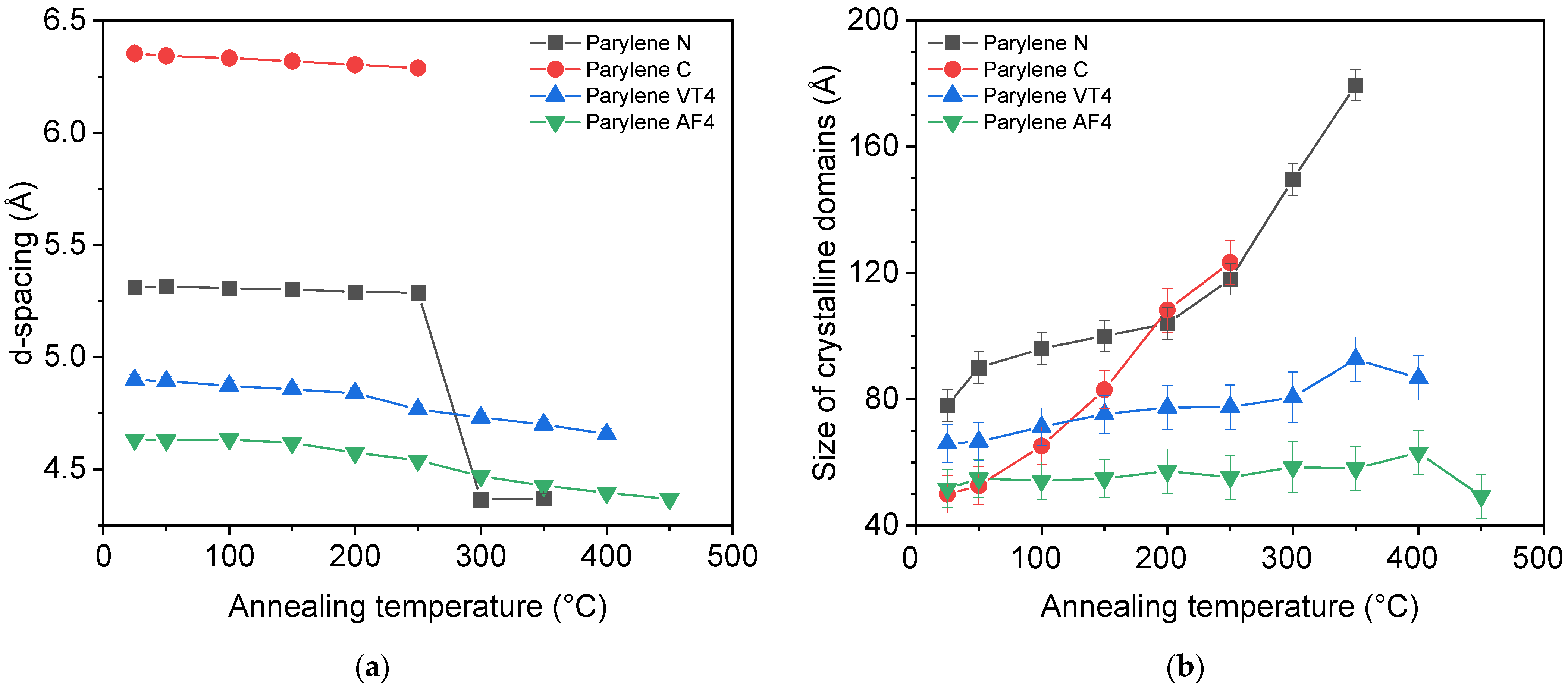
3.3. X-ray Diffraction
3.4. Permeation Measurements
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Licari, J.J. Coating Materials for Electronic Applications Polymers, Processes, Reliability, Testing; Noyes Publications/William Andrew, Inc.: Norwich, NY, USA, 2003; ISBN 0815514921. [Google Scholar]
- Hogg, A.; Uhl, S.; Feuvrier, F.; Girardet, Y.; Graf, B.; Aellen, T.; Keppner, H.; Tardy, Y.; Burger, J. Protective Multilayer Packaging for Long-Term Implantable Medical Devices. Surf. Coat. Technol. 2014, 255, 124–129. [Google Scholar] [CrossRef]
- Kim, B.J.; Meng, E. Micromachining of Parylene C for BioMEMS. Polym. Adv. Technol. 2016, 27, 564–576. [Google Scholar] [CrossRef]
- Dong, X.; Zhang, M.; Lei, Y.; Li, Z.; Jin, Y.; Wang, W. Parylene-MEMS Technique-Based Flexible Electronics. Sci. China Inf. Sci. 2018, 61, 1–9. [Google Scholar] [CrossRef]
- Ortigoza-Diaz, J.; Scholten, K.; Larson, C.; Cobo, A.; Hudson, T.; Yoo, J.; Baldwin, A.; Weltman Hirschberg, A.; Meng, E. Techniques and Considerations in the Microfabrication of Parylene C Microelectromechanical Systems. Micromachines 2018, 9, 422. [Google Scholar] [CrossRef]
- Lin, Y.-W.; Efimovskaya, A.; Shkel, A.M. Folded MEMS Platform Based on Polymeric Flexible Hinges for 3D Integration of Spatially-Distributed Sensors. J. Microelectromech. Syst. 2021, 30, 907–914. [Google Scholar] [CrossRef]
- Golda-Cepa, M.; Engvall, K.; Hakkarainen, M.; Kotarba, A. Recent Progress on Parylene C Polymer for Biomedical Applications: A Review. Prog. Org. Coat. 2020, 140, 105493. [Google Scholar] [CrossRef]
- Chang, J.H.C.; Liu, Y.; Tai, Y.C. Long Term Glass-Encapsulated Packaging for Implant Electronics. In Proceedings of the 2014 IEEE 27th International Conference on Micro Electro Mechanical Systems (MEMS), Francisco, CA, USA, 26–30 January 2014; pp. 1127–1130. [Google Scholar] [CrossRef]
- Seymour, J.P.; Elkasabi, Y.M.; Chen, H.-Y.; Lahann, J.; Kipke, D.R. The Insulation Performance of Reactive Parylene Films in Implantable Electronic Devices. Biomaterials 2009, 30, 6158–6167. [Google Scholar] [CrossRef]
- Jackson, N.; Olszewski, O.Z.; O’Murchu, C.; Mathewson, A. Ultralow-Frequency PiezoMEMS Energy Harvester Using Thin-Film Silicon and Parylene Substrates. J. Micro Nanolithogr. MEMS MOEMS 2018, 17, 015005. [Google Scholar] [CrossRef]
- Lagomarsini, C.; Jean-Mistral, C.; Kachroudi, A.; Monfray, S.; Sylvestre, A. Outstanding Performance of Parylene Polymers as Electrets for Energy Harvesting and High-Temperature Applications. J. Appl. Polym. Sci. 2020, 137, 48790. [Google Scholar] [CrossRef]
- Lo, H.W.; Tai, Y.C. Parylene-HT-Based Electret Rotor Generator. In Proceedings of the 2008 IEEE 21st International Conference on Micro Electro Mechanical Systems, Tucson, AZ, USA, 13–17 January 2008; pp. 984–987. [Google Scholar] [CrossRef]
- Ribeiro, L.; Saotome, O.; d’Amore, R.; de Oliveira Hansen, R. High-Speed and High-Temperature Calorimetric Solid-State Thermal Mass Flow Sensor for Aerospace Application: A Sensitivity Analysis. Sensors 2022, 22, 3484. [Google Scholar] [CrossRef]
- Lin, M.; Chen, Q.; Wang, Z.; Fang, Y.; Liu, J.; Yang, Y.; Wang, W.; Cai, Y.; Huang, R. Flexible Polymer Device Based on Parylene-C with Memory and Temperature Sensing Functionalities. Polymers 2017, 9, 310. [Google Scholar] [CrossRef]
- Kan, T.; Aoki, H.; Binh-Khiem, N.; Matsumoto, K.; Shimoyama, I. Ratiometric Optical Temperature Sensor Using Two Fluorescent Dyes Dissolved in an Ionic Liquid Encapsulated by Parylene Film. Sensors 2013, 13, 4138–4145. [Google Scholar] [CrossRef]
- Xie, X.; Rieth, L.; Caldwell, R.; Diwekar, M.; Tathireddy, P.; Sharma, R.; Solzbacher, F. Long-Term Bilayer Encapsulation Performance of Atomic Layer Deposited Al2O3 and Parylene C for Biomedical Implantable Devices. In IEEE Transactions on Biomedical Engineering; IEEE: Piscataway, NJ, USA, 2013; Volume 60, pp. 2943–2951. [Google Scholar] [CrossRef]
- Selbmann, F.; Baum, M.; Wiemer, M.; Gessner, T. Deposition of Parylene C and Characterization of Its Hermeticity for the Encapsulation of MEMS and Medical Devices. In Proceedings of the 2016 IEEE 11th Annual International Conference on Nano/Micro Engineered and Molecular Systems (NEMS), Sendai, Japan, 17–20 April 2016; pp. 427–432. [Google Scholar] [CrossRef]
- Park, H.; Choi, W.; Oh, S.; Kim, Y.-J.; Seok, S.; Kim, J. A Study on Biocompatible Polymer-Based Packaging of Neural Interface for Chronic Implantation. Micromachines 2022, 13, 516. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.; Oh, S.; Lee, T.; Park, M. Thermo-Mechanical Analyses of Printed Board Assembly during Reflow Process for Warpage Prediction. In Proceedings of the 2014 15th International Conference on Thermal, Mechanical and Mulit-Physics Simulation and Experiments in Microelectronics and Microsystems (EuroSimE), Ghent, Belgium, 7–9 April 2014; pp. 1–5. [Google Scholar]
- Association Connecting Electronics Industries. IPC-7530A Guidelines for Temperature Profiling for Mass Soldering Processes (Reflow & Wave); 2016. [Google Scholar]
- Hassler, C.; Von Metzen, R.P.; Ruther, P.; Stieglitz, T. Characterization of Parylene C as an Encapsulation Material for Implanted Neural Prostheses. J. Biomed. Mater. Res. Part B Appl. Biomater. Off. J. Soc. Biomater. 2010, 93, 266–274. [Google Scholar] [CrossRef] [PubMed]
- von Metzen, R.; Stieglitz, T. Impact of Sterilization Procedures on the Stability of Parylene Based Flexible Multilayer Structures. Biomed. Eng./Biomed. Tech. 2013, 58, 4089. [Google Scholar] [CrossRef]
- Gorham, W.F. A New, General Synthetic Method for the Preparation of Linear Poly-p-Xylylenes. J. Polym. Sci. Part A-1 Polym. Chem. 1966, 4, 3027–3039. [Google Scholar] [CrossRef]
- Kahouli, A.; Sylvestre, A.; Pairis, S.; Laithier, J.-F. Effect of ClH Aromatic Substitution on Structural and Dielectric Properties of Poly(p-Xylylene). Polymer 2012, 53, 3001–3007. [Google Scholar] [CrossRef]
- Kachroudi, A.; Kahouli, A.; Legrand, J.; Jomni, F. Dielectric and Conduction Mechanisms of Parylene N at High Temperature: Phase-Transition Effect. J. Phys. Chem. A 2015, 119, 6428–6435. [Google Scholar] [CrossRef]
- Menon, P.R.; Li, W.; Tooker, A.; Tai, Y.C. Characterization of Water Vapor Permeation through Thin Film Parylene C. In Proceedings of the TRANSDUCERS 2009-2009 International Solid-State Sensors, Actuators and Microsystems Conference, Denver, CO, USA, 21–25 June 2009; pp. 1892–1895. [Google Scholar] [CrossRef]
- Kahouli, A.; Sylvestre, A.; Laithier, J.F.; Lutsen, L.; Pairis, S.; André, E.; Garden, J.L. Structural and Dielectric Properties of Parylene-VT4 Thin Films. Mater. Chem. Phys. 2014, 143, 908–914. [Google Scholar] [CrossRef]
- Nitti, A.; Debattista, F.; Abbondanza, L.; Bianchi, G.; Po, R.; Pasini, D. Donor-Acceptor Conjugated Copolymers Incorporating Tetrafluorobenzene as the π-Electron Deficient Unit. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 1601–1610. [Google Scholar] [CrossRef]
- Corsini, F.; Nitti, A.; Tatsi, E.; Mattioli, G.; Botta, C.; Pasini, D.; Griffini, G. Large-Area Semi-Transparent Luminescent Solar Concentrators Based on Large Stokes Shift Aggregation-Induced Fluorinated Emitters Obtained Through a Sustainable Synthetic Approach. Adv. Opt. Mater. 2021, 9, 2100182. [Google Scholar] [CrossRef]
- Babudri, F.; Farinola, G.M.; Naso, F.; Ragni, R. Fluorinated Organic Materials for Electronic and Optoelectronic Applications: The Role of the Fluorine Atom. Chem. Commun. 2007, 1003–1022. [Google Scholar] [CrossRef] [PubMed]
- Dolbier, W.R.; Beach, W.F. Parylene-AF4: A Polymer with Exceptional Dielectric and Thermal Properties. J. Fluor. Chem. 2003, 122, 97–104. [Google Scholar] [CrossRef]
- Yang, N. Parylene AF4 Its Preparations, Characteristics and Applications. Invit. Talk IMID2017 Mater. Transparent Flex. Disp. 2017, 4–5. [Google Scholar] [CrossRef]
- Degen, T.; Sadki, M.; Bron, E.; König, U.; Nénert, G. The HighScore Suite. Powder Diffr. 2014, 29, S13–S18. [Google Scholar] [CrossRef] [Green Version]
- Hogg, A.; Aellen, T.; Uhl, S.; Graf, B.; Keppner, H.; Tardy, Y.; Burger, J. Ultra-Thin Layer Packaging for Implantable Electronic Devices. J. Micromech. Microeng. 2013, 23, 075001. [Google Scholar] [CrossRef]
- Yoshida, H.; Ebina, T.; Arai, K.; Kobata, T.; Ishii, R.; Aizawa, T.; Suzuki, A. Development of Water Vapor Transmission Rate Measuring Device Using a Quadrupole Mass Spectrometer and Standard Gas Barrier Films down to the 10−6 g M−2 Day−1 Level. Rev. Sci. Instrum. 2017, 88, 043301. [Google Scholar] [CrossRef]
- Yoshida, H.; Arai, K.; Hirata, M.; Akimichi, H. New Leak Element Using Sintered Stainless Steel Filter for In-Situ Calibration of Ionization Gauges and Quadrupole Mass Spectrometers. Vacuum 2012, 86, 838–842. [Google Scholar] [CrossRef]
- Yoshida, H.; Arai, K.; Kobata, T. In-Situ Calibration Method for Ionization Gauges and Quadrupole Mass Spectrometers by Combining the Standard Conductance Element and the Conductance Modulation Method (SCEeCM Method). Vacuum 2014, 101, 433–439. [Google Scholar] [CrossRef]
- Yoshida, H.; Arai, K.; Suzuki, A.; Aizawa, T.; Ishii, R.; Ebina, T. Development of a Gas Permeation Measuring Device and the Evaluation of Gas Barrier Property of Clay-Polymer Nanocomposite Films. Clay Sci. 2018, 22, 95–102. [Google Scholar] [CrossRef]
- Miller, K.J.; Hollinger, H.B.; Wunderlich, B. On the Conformations of Poly(p-Xylylene) and Its Mesophase Transitions. Macromolecules 1990, 23, 3855–3859. [Google Scholar] [CrossRef]
- Morgen, M.; Rhee, S.H.; Zhao, J.H.; Malik, I.; Ryan, T.; Ho, H.M.; Piano, M.A.; Ho, P. Comparison of Crystalline Phase Transitions in Fluorniated vs Nonfluorinated Parylene Thin Films. Macromolecules 1999, 32, 7555–7561. [Google Scholar] [CrossRef]
- Jackson, N.; Stam, F.; O’Brien, J.; Kailas, L.; Mathewson, A.; O’Murchu, C. Crystallinity and Mechanical Effects from Annealing Parylene Thin Films. Thin Solid Film. 2016, 603, 371–376. [Google Scholar] [CrossRef]
- Senkevich, J.J.; Desu, S.B.; Simkovic, V. Temperature Studies of Optical Birefringence and X-Ray Diffraction with Poly(p-Xylylene), Poly(Chloro-p-Xylylene) and Poly(Tetrafluoro-p-Xylylene) CVD Thin Films. Polymer 2000, 41, 2379–2390. [Google Scholar] [CrossRef]
- Von Metzen, R.P.; Stieglitz, T. The Effects of Annealing on Mechanical, Chemical, and Physical Properties and Structural Stability of Parylene C. Biomed. Microdevices 2013, 15, 727–735. [Google Scholar] [CrossRef]
- Wu, P.K.; Yang, G.R.; McDonald, J.F.; Lu, T.M. Surface Reaction and Stability of Parylene N and F Thin Films at Elevated Temperatures. J. Electron. Mater. 1995, 24, 53–58. [Google Scholar] [CrossRef]
- Hsu, J.M.; Rieth, L.; Kammer, S.; Orthner, M.; Solzbacher, F. Effect of Thermal and Deposition Processes on Surface Morphology, Crystallinity, and Adhesion of Parylene-C. Sensors Mater. 2008, 20, 87–102. [Google Scholar] [CrossRef]
- Harder, T.A.; Yao, T.-J.; He, Q.; Shih, C.-Y.; Tai, Y.-C. Residual Stress in Thin-Film Parylene-C. In Proceedings of the Technical Digest. MEMS 2002 IEEE International Conference. Fifteenth IEEE International Conference on Micro Electro Mechanical Systems (Cat. No.02CH37266), Las Vegas, NV, USA, 24 January 2002; pp. 435–438. [Google Scholar]
- Lua, A.C.; Su, J. Isothermal and Non-Isothermal Pyrolysis Kinetics of Kapton® Polyimide. Polym. Degrad. Stab. 2006, 91, 144–153. [Google Scholar] [CrossRef]
- McKeen, L.W. 7-Polyimides. In Permeability Properties of Plastics and Elastomers, 4th ed.; McKeen, L.W., Ed.; Plastics Design Library; William Andrew Publishing: Oxford, UK, 2010; pp. 149–173. ISBN 978-0-323-50859-9. [Google Scholar]
- Kumar, R. New Developments in Parylene Technology for Medical Electronics Advancement. In Proceedings of the SMTA Medical Electronics Symposium, Minneapolis, MN, USA, May 2006; p. 9. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buchwalder, S.; Borzì, A.; Diaz Leon, J.J.; Bourgeois, F.; Nicolier, C.; Nicolay, S.; Neels, A.; Zywitzki, O.; Hogg, A.; Burger, J. Thermal Analysis of Parylene Thin Films for Barrier Layer Applications. Polymers 2022, 14, 3677. https://doi.org/10.3390/polym14173677
Buchwalder S, Borzì A, Diaz Leon JJ, Bourgeois F, Nicolier C, Nicolay S, Neels A, Zywitzki O, Hogg A, Burger J. Thermal Analysis of Parylene Thin Films for Barrier Layer Applications. Polymers. 2022; 14(17):3677. https://doi.org/10.3390/polym14173677
Chicago/Turabian StyleBuchwalder, Sébastien, Aurelio Borzì, Juan J. Diaz Leon, Florian Bourgeois, Cléo Nicolier, Sylvain Nicolay, Antonia Neels, Olaf Zywitzki, Andreas Hogg, and Jürgen Burger. 2022. "Thermal Analysis of Parylene Thin Films for Barrier Layer Applications" Polymers 14, no. 17: 3677. https://doi.org/10.3390/polym14173677
APA StyleBuchwalder, S., Borzì, A., Diaz Leon, J. J., Bourgeois, F., Nicolier, C., Nicolay, S., Neels, A., Zywitzki, O., Hogg, A., & Burger, J. (2022). Thermal Analysis of Parylene Thin Films for Barrier Layer Applications. Polymers, 14(17), 3677. https://doi.org/10.3390/polym14173677








