Synthesis Metakaolin-Based Geopolymer Incorporated with SiC Sludge Using Design of Experiment Method
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design of Experiments (DOEs)
2.2. Application of Experimental Design Method in Optimizing Design Parameters
2.3. Materials and Sample Preparation
2.4. Test Items and Methods
3. Results and Discussion
3.1. Factor Parameter and Physical Property Test
3.2. One-Way ANOVA
3.3. Application of Optimization Design Factors by the Multivariate Statistical Analysis
3.3.1. Raw Material Analysis
3.3.2. Dimensionless Ratio Analysis
3.4. Microstructural Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Davidovits, J. Geopolymer Chemistry and Applications, 3rd ed.; Geopolymer Institute: Saint-Quentin, France, 2008. [Google Scholar]
- Si, R.Z.; Guo, S.C.; Dai, Q.L.; Wang, J.Q. Atomic-structure, microstructure, and mechanical properties of glass powder modified metakaolin-based geopolymer. Constr. Build. Mater. 2020, 254, 119303. [Google Scholar] [CrossRef]
- Chougan, M.; Ghaffar, S.H.; Jahanzat, M.; Albar, A.; Mujaddedi, N.; Swash, R. The influence of nano-additives in strengthening mechanical performance of 3D printed multi-binder geopolymer composites. Constr. Build. Mater. 2020, 250, 118928. [Google Scholar] [CrossRef]
- Top, S.; Vapur, H.; Altiner, M.; Kaya, D.; Ekicibil, A. Properties of fly ash-based lightweight geopolymer concrete prepared using pumice and expanded perlite as aggregates. J. Mol. Struct. 2020, 1202, 127236. [Google Scholar] [CrossRef]
- Assaedi, H.; Alomayri, T.; Kaze, C.R.; Jindal, B.B.; Subaer, S.; Shaikh, F.; Alraddadi, S. Characterization and properties of geopolymer nanocomposites with different contents of nano-CaCO3. Constr. Build. Mater. 2020, 252, 119137. [Google Scholar] [CrossRef]
- Mesgari, S.; Akbarnezhad, A.; Xiao, J.Z. Recycled geopolymer aggregates as coarse aggregates for Portland cement concrete and geopolymer concrete: Effects on mechanical properties. Constr. Build. Mater. 2020, 236, 117571. [Google Scholar] [CrossRef]
- Jiang, X.; Xiao, R.; Zhang, M.M.; Hu, W.; Bai, Y.; Huang, B.S. A laboratory investigation of steel to fly ash-based geopolymer paste bonding behavior after exposure to elevated temperatures. Constr. Build. Mater. 2020, 254, 119267. [Google Scholar] [CrossRef]
- Wan, Q.; Zhang, Y.M.; Zhang, R.B. Using mechanical activation of quartz to enhance the compressive strength of metakaolin based geopolymers. Cem. Concr. Compos. 2020, 111, 103635. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Bui, H.H.; Ngo, T.D.; Nguyen, G.D.; Kreher, M.U.; Darve, F. A micromechanical investigation for the effects of pore size and its distribution on geopolymer foam concrete under uniaxial compression. Eng. Fract. Mech. 2019, 209, 228–244. [Google Scholar] [CrossRef]
- Łach, M.; Korniejenko, K.; Mikuła, J. Thermal insulation and thermally resistant materials made of geopolymer foams. Procedia Eng. 2016, 151, 410–416. [Google Scholar] [CrossRef]
- Sakkas, K.; Sofianos, A.; Nomikos, P.; Panias, D. Behaviour of Passive Fire Protection K-Geopolymer under Successive Severe Fire Incidents. Materials 2015, 8, 6096–6104. [Google Scholar] [CrossRef]
- Ghanbari, M.; Hadian, A.M.; Nourbakhsh, A.A. Effect of Processing Parameters on Compressive Strength of Metakaolinite Based Geopolymers: Using DOE Approach. Procedia Mater. Sci. 2015, 11, 711–716. [Google Scholar] [CrossRef]
- Nur, A.J.; Liew, Y.M.; Heah, C.Y.; Al Bakri Abdullah, M.M.; Hussin, K. Correlation between pore structure, compressive strength and thermal conductivity of porous metakaolin geopolymer. Constr. Build. Mater. 2020, 247, 118641. [Google Scholar]
- Dong, M.; Elchalakani, M.; Karrech, A. Development of high strength one-part geopolymer mortar using sodium metasilicate. Constr. Build. Mater. 2020, 236, 117611. [Google Scholar] [CrossRef]
- Xu, H.; van Deventer, J.S.J. The effect of alkali metals on the formation of geopolymeric gels from alkali-feldspars. Colloids Surf. A Physicochem. Eng. Asp. 2003, 216, 27–44. [Google Scholar] [CrossRef]
- Varanda, C.; Portugal, I.; Ribeiro, J.; Silva, A.M.S.; Silva, C.M. Optimization of bitumen formulations using mixture design of experiments (MDOE). Constr. Build. Mater. 2017, 156, 611–620. [Google Scholar] [CrossRef]
- Ohno, M.; Li, V.C. An integrated design method of Engineered Geopolymer Composite. Cem. Concr. Compos. 2018, 88, 73–85. [Google Scholar] [CrossRef]
- Askarian, M.; Tao, Z.; Samali, B.; Adam, G.; Shuaibu, R. Mix composition and characterisation of one-part geopolymers with different activators. Constr. Build. Mater. 2019, 225, 526–537. [Google Scholar] [CrossRef]
- Zhang, M.; El-Korchi, T.; Zhang, G.; Liang, J.; Tao, M. Synthesis factors affecting mechanical properties, microstructure, and chemical composition of red mud–fly ash based geopolymers. Fuel 2014, 134, 315–325. [Google Scholar] [CrossRef]
- Reddy, M.S.; Dinakar, P.; Rao, B.H. A review of the influence of source material’s oxide composition on the compressive strength of geopolymer concrete. Microporous Mesoporous Mater. 2016, 234, 12–23. [Google Scholar] [CrossRef]
- Natassia, B.S.N.; Danila, F.N.; Adriano, M.B. Valorization of rice husk ash and aluminum anodizing sludge as precursors for the synthesis of geopolymers. J. Clean. Prod. 2021, 298, 126770. [Google Scholar]
- Mendes, J.P.; Elyseu, F.; Jaramillo Nieves, L.J.; Zaccaron, A.; Bernardin, A.M.; Angioletto, E. Synthesis and characterization of geopolymers using clay ceramic waste as source of aluminosilicate. Sustain. Mater. Technol. 2021, 28, e00264. [Google Scholar] [CrossRef]
- Tailby, J.; MacKenzie, K.J.D. Structure and mechanical properties of aluminosilicate geopolymer composites with Portland cement and its constituent minerals. Cem. Concr. Res. 2010, 40, 787–794. [Google Scholar] [CrossRef]
- Rovnaník, P. Effect of curing temperature on the development of hard structure of metakaolin-based geopolymer. Constr. Build. Mater. 2010, 24, 1176–1183. [Google Scholar] [CrossRef]
- Xiao, R.; Ma, Y.T.; Jiang, X.; Zhang, M.M.; Zhang, Y.Y.; Wang, Y.H.; Huang, B.S.; He, Q. Strength, microstructure, efflorescence behavior and environmental impacts of waste glass geopolymers cured at ambient temperature. J. Clean. Prod. 2020, 252, 119610. [Google Scholar] [CrossRef]
- Sourav, K.D.; Sandeep, S. Influence of molarity and alkali mixture ratio on ambient temperature cured waste cement concrete based geopolymer mortar. Constr. Build. Mater. 2021, 301, 124380. [Google Scholar]
- Iman, S.L.; Mohammad, M.T.; Vahid, T. Improvement of poorly graded sandy soil by using copper mine tailing dam sediments-based geopolymer and silica fume. Constr. Build. Mater. 2021, 281, 122591. [Google Scholar]
- Ko, M.S.; Chen, H.Y.; Lyu, S.J.; Wang, T.T.; Ueng, T.H. Permeation characteristics and impact factors of geopolymers made of kaolin. Constr. Build. Mater. 2015, 93, 301–308. [Google Scholar] [CrossRef]
- ASTM C109/C109M; Standard Test Method for Compressive Strength of Hydraulic Cement Mortars. ASTM International: West Conshohocken, PA, USA, 2017.
- Wan, Q.; Rao, F.; Song, S.X. Reexamining calcination of kaolinite for the synthesis of metakaolin geopolymers—Roles of dehydroxylation and recrystallization. J. Non-Cryst. Solids 2017, 460, 74–80. [Google Scholar] [CrossRef]
- Cho, Y.K.; Yoo, S.W.; Jung, S.H.; Lee, K.M.; Kwon, S.J. Effect of Na2O content, SiO2/Na2O molar ratio, and curing conditions on the compressive strength of FA-based geopolymer. Constr. Build. Mater. 2017, 145, 253–260. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, Y.J.; He, P.Y.; Li, C.J.; Liu, L.C. Novel activated carbon route to low-cost geopolymer based porous composite with high mechanical resistance and enhanced CO2 capacity. Microporous Mesoporous Mater. 2020, 305, 110282. [Google Scholar] [CrossRef]
- Alvi, M.A.A.; Khalifeh, M.; Agonafir, M.B. Effect of nanoparticles on properties of geopolymers designed for well cementing applications. J. Pet. Sci. Eng. 2020, 191, 107128. [Google Scholar] [CrossRef]
- Fernández-Jiménez, A.; Zibouche, F.; Boudissa, N.; García-Lodeiro, I.; Abadlia, M.T.; Palomo, A. “Metakaolin-Slag-Clinker Blends.” The Role of Na+ or K+ as Alkaline Activators of Theses Ternary Blends. J. Am. Ceram. Soc. 2013, 96, 1991–1998. [Google Scholar] [CrossRef]
- Fletcher, R.A.; MacKenzie, K.J.D.; Nicholson, C.L.; Shimada, S. The composition range of aluminosilicate geopolymers. J. Eur. Ceram. Soc. 2005, 25, 1471–1477. [Google Scholar] [CrossRef]
- He, P.G.; Wang, M.R.; Fu, S.A.; Jia, D.C.; Yan, S.; Yuan, J.K.; Xu, J.H.; Wang, P.F.; Zhou, Y. Effects of Si/Al ratio on the structure and properties of metakaolin based geopolymer. Ceram. Int. 2016, 42, 14416–14422. [Google Scholar] [CrossRef]
- Fiset, J.; Cellier, M.; Vuillaume, P.Y. Macroporous geopolymers designed for facile polymers post-infusion. Cem. Concr. Compos. 2020, 110, 103591. [Google Scholar] [CrossRef]
- Lahoti, M.; Wong, K.K.; Tan, K.H.; Yang, E.H. Effect of alkali cation type on strength endurance of fly ash geopolymers subject to high temperature exposure. Mater. Des. 2018, 154, 8–19. [Google Scholar] [CrossRef]
- Zhang, F.; Zhang, L.Y.; Liu, M.; Mu, C.Z.; Liang, Y.N.; Hu, X. Role of alkali cation in compressive strength of metakaolin based geopolymers. Ceram. Int. 2017, 43, 3811–3817. [Google Scholar] [CrossRef]
- Zheng, Z.; Ma, X.; Zhang, Z.H.; Li, Y.X. In-situ transition of amorphous gels to Na-P1 zeolite in geopolymer: Mechanical and adsorption properties. Constr. Build. Mater. 2019, 202, 851–860. [Google Scholar] [CrossRef]
- Hu, Y.; Liang, S.; Yang, J.K.; Chen, Y.; Ye, N.; Ke, Y.; Tao, S.Y.; Xiao, K.K.; Hu, J.P.; Hou, H.J.; et al. Role of Fe species in geopolymer synthesized from alkali-thermal pretreated Fe-rich Bayer red mud. Constr. Build. Mater. 2019, 200, 398–407. [Google Scholar] [CrossRef]


| No. | Na2SiO3 | MK | Water | NaOH | Si/Na | S/L | OW | OH− (M) | Compressive Strength (MPa) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Low | Low | Low | Low | 0.50 | 0.20 | 0.02 | 0.67 | 2.04 ± 1.56 |
| 2 | Low | Medium | Medium | Medium | 0.80 | 0.80 | 0.03 | 3.05 | 43.40 ± 2.30 |
| 3 | Low | High | High | High | 1.00 | 1.20 | 0.05 | 5.40 | 30.82 ± 3.21 |
| 4 | Medium | Low | Medium | High | 1.50 | 0.40 | 0.03 | 9.67 | 13.80 ± 1.82 |
| 5 | Medium | Medium | High | Low | 1.60 | 1.00 | 0.04 | 1.60 | 49.44 ± 14.04 |
| 6 | Medium | High | Low | Medium | 1.60 | 1.20 | 0.02 | 4.52 | 22.55 ± 2.06 |
| 7 | High | Low | High | Medium | 2.00 | 0.60 | 0.04 | 4.96 | 40.08 ± 6.01 |
| 8 | High | Medium | Low | High | 2.50 | 1.00 | 0.02 | 12.46 | 0.47 ± 0.07 |
| 9 | High | High | Medium | Low | 3.00 | 2.20 | 0.03 | 2.84 | N/A |
| No. | Si/Na | S/L | MK (%) | SCS (%) | Strength (MPa) |
|---|---|---|---|---|---|
| 1 | 0.8 | 1.0 | 100 | 0 | 50.07 ± 3.25 |
| 2 | 0.8 | 1.0 | 90 | 10 | 47.16 ± 5.13 |
| 3 | 0.8 | 1.0 | 80 | 20 | 48.43 ± 6.77 |
| 4 | 0.8 | 1.0 | 70 | 30 | 37.47 ± 1.60 |
| 5 | 0.8 | 1.0 | 60 | 40 | 37.00 ± 3.37 |
| 6 | 1.2 | 1.0 | 100 | 0 | 57.67 ± 7.57 |
| 7 | 1.2 | 1.0 | 90 | 10 | 63.64 ± 4.20 |
| 8 | 1.2 | 1.0 | 80 | 20 | 63.05 ± 9.81 |
| 9 | 1.2 | 1.0 | 70 | 30 | 59.24 ± 8.79 |
| 10 | 1.2 | 1.0 | 60 | 40 | 54.47 ± 3.36 |
| 11 | 1.6 | 0.4 | 100 | 0 | 17.18 ± 0.84 |
| 12 | 1.6 | 0.4 | 90 | 10 | 12.23 ± 1.09 |
| 13 | 1.6 | 0.4 | 80 | 20 | 9.71 ± 0.47 |
| 14 | 1.6 | 0.4 | 70 | 30 | 7.28 ± 0.62 |
| 15 | 1.6 | 0.4 | 60 | 40 | 3.52 ± 0.22 |
| 16 | 1.6 | 0.6 | 100 | 0 | 43.08 ± 9.02 |
| 17 | 1.6 | 0.6 | 90 | 10 | 36.26 ± 3.58 |
| 18 | 1.6 | 0.6 | 80 | 20 | 28.43 ± 4.83 |
| 19 | 1.6 | 0.6 | 70 | 30 | 24.61 ± 1.72 |
| 20 | 1.6 | 0.6 | 60 | 40 | 15.53 ± 1.72 |
| 21 | 1.6 | 0.8 | 100 | 0 | 61.76 ± 4.40 |
| 22 | 1.6 | 0.8 | 90 | 10 | 59.18 ± 5.50 |
| 23 | 1.6 | 0.8 | 80 | 20 | 57.71 ± 9.43 |
| 24 | 1.6 | 0.8 | 70 | 30 | 51.00 ± 2.81 |
| 25 | 1.6 | 0.8 | 60 | 40 | 26.88 ± 1.92 |
| 26 | 1.6 | 1.0 | 100 | 0 | 64.37 ± 7.71 |
| 27 | 1.6 | 1.0 | 90 | 10 | 66.08 ± 9.13 |
| 28 | 1.6 | 1.0 | 80 | 20 | 64.59 ± 6.77 |
| 29 | 1.6 | 1.0 | 70 | 30 | 60.34 ± 8.86 |
| 30 | 1.6 | 1.0 | 60 | 40 | 46.90 ± 2.94 |
| 31 | 2.0 | 1.0 | 100 | 0 | 61.51 ± 3.61 |
| 32 | 2.0 | 1.0 | 90 | 10 | 54.43 ± 5.82 |
| 33 | 2.0 | 1.0 | 80 | 20 | 31.09 ± 5.11 |
| 34 | 2.0 | 1.0 | 70 | 30 | 17.35 ± 1.83 |
| 35 | 2.0 | 1.0 | 60 | 40 | 14.05 ± 3.05 |
| Source | Sum of Squares | Degree of Freedom | Mean Square | F | p-Value |
|---|---|---|---|---|---|
| Si/Na | 2.517 | 7 | 0.36 | 18.776 | 5.14 × 10-4 |
| Error | 0.005 | 1 | 0.005 | ||
| Total | 2.522 | 8 | |||
| Solid/Liquid | 0.7 | 7 | 0.1 | 19.737 | 4.09 × 10-4 |
| Error | 0.02 | 1 | 0.02 | ||
| Total | 0.72 | 8 | |||
| Si/Al | 0.135 | 7 | 0.019 | 19.381 | 4.45 × 10-4 |
| Error | 0.054 | 1 | 0.054 | ||
| Total | 0.189 | 8 | |||
| OH− (M) | 114.379 | 7 | 16.34 | 13.727 | 1.92 × 10−3 |
| Error | 2.247 | 1 | 2.247 | ||
| Total | 116.626 | 8 |
| Factor | Coefficient | Standard Error | t (35) | p-Value (<0.05) |
|---|---|---|---|---|
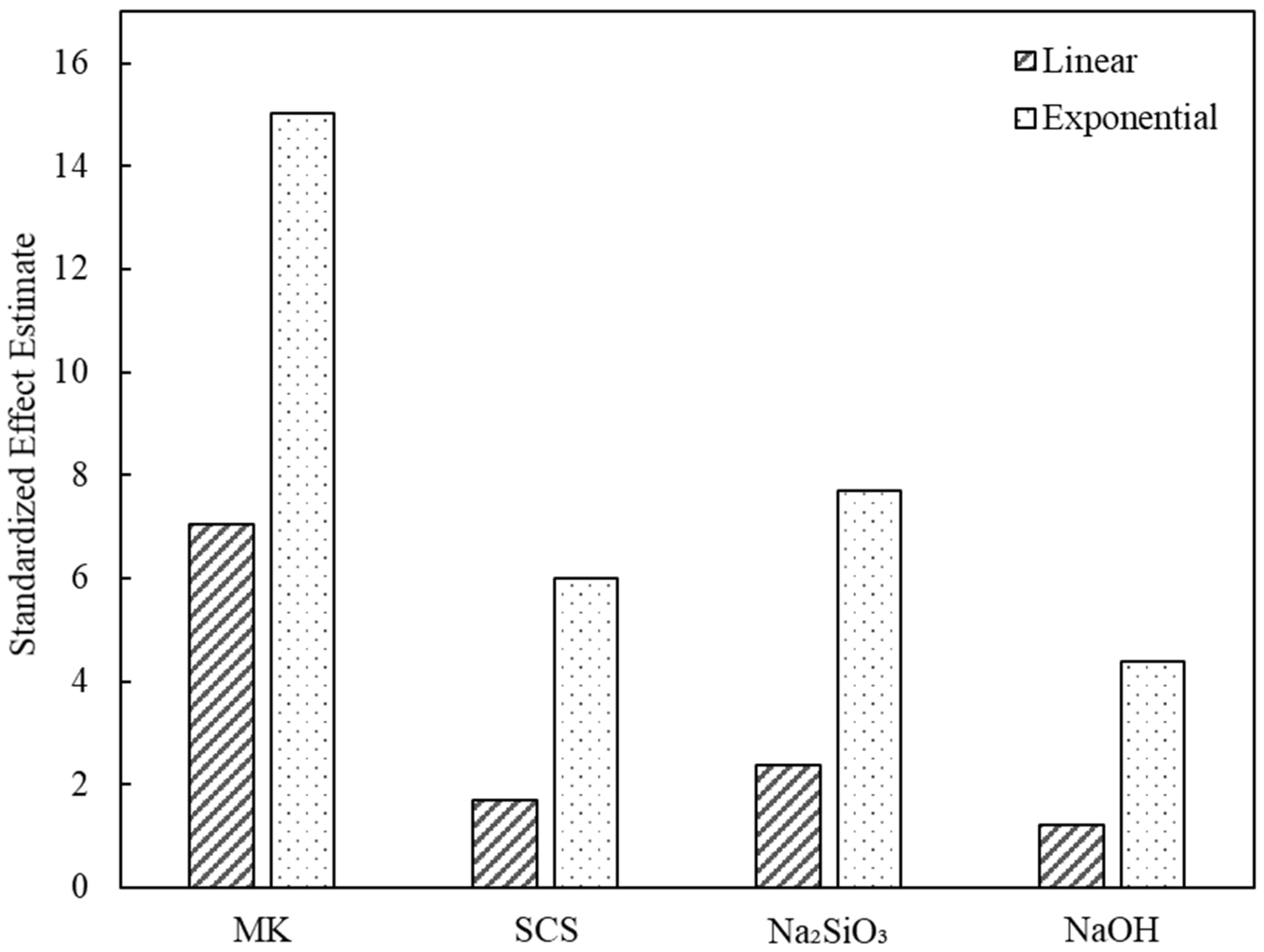
| (1) MK | 29.960 | 1.996 | 15.013 | 0.000 |
| (2) SCS | −25.540 | 4.186 | −6.101 | 0.000 |
| (3) Na2SiO3 | 39.785 | 5.180 | 7.681 | 0.000 |
| (4) NaOH | −15.365 | 3.512 | −4.375 | 0.000 |
| (1) by (2) | 55.500 | 4.537 | 12.233 | 0.000 |
| (1) by (3) | −9.825 | 4.743 | −2.072 | 0.045 |
| (1) by (4) | 45.325 | 3.336 | 13.588 | 0.000 |
| (2) by (3) | −65.325 | 3.824 | −17.085 | 0.000 |
| (2) by (4) | −10.175 | 2.624 | −3.878 | 0.000 |
| (3) by (4) | 55.150 | 3.637 | 15.163 | 0.000 |
| Factor | Coefficient | Standard Error | t (35) | p-Value (<0.05) |
|---|---|---|---|---|
| Si/Na | −42.540 | 3.142 | −13.538 | 0.000 |
| S/L | −43.190 | 3.106 | −13.905 | 0.000 |
| OW | −43.896 | 3.131 | −14.020 | 0.000 |
| Factor | Effect | Std. Err. | t (40) | p-Level (<0.05) |
|---|---|---|---|---|
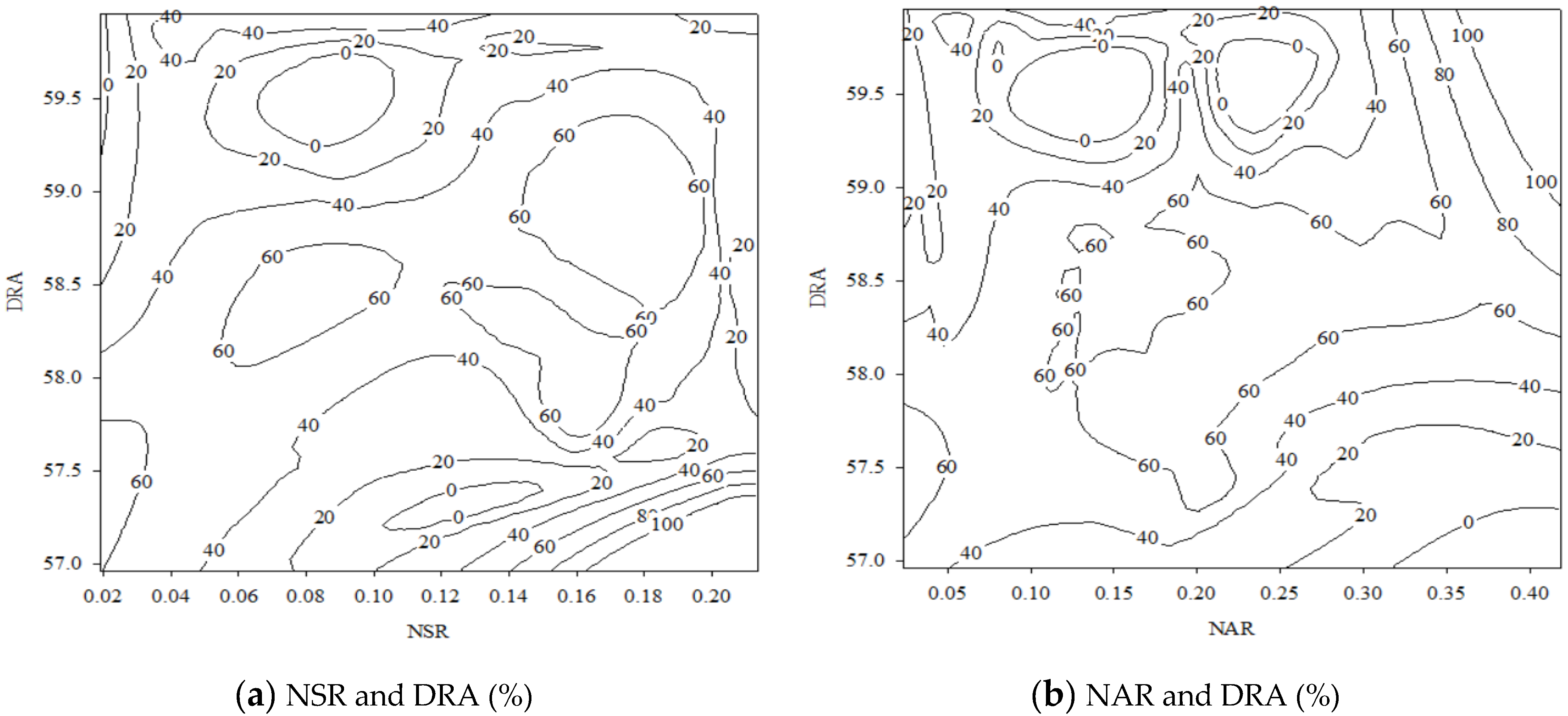
| (1) NSR | −43.918 | 3.129 | −14.038 | 0.000 |
| (2) NAR | −43.845 | 3.130 | −14.008 | 0.000 |
| (3) DRS | −1.487 | 2.329 | −0.639 | 0.527 |
| (4) DRA | 14.651 | 3.187 | 4.597 | 0.000 |
| (5) OH− | −39.612 | 3.159 | −12.539 | 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo, K.-W.; Lin, W.-T.; Lin, Y.-W.; Cheng, T.-W.; Lin, K.-L. Synthesis Metakaolin-Based Geopolymer Incorporated with SiC Sludge Using Design of Experiment Method. Polymers 2022, 14, 3395. https://doi.org/10.3390/polym14163395
Lo K-W, Lin W-T, Lin Y-W, Cheng T-W, Lin K-L. Synthesis Metakaolin-Based Geopolymer Incorporated with SiC Sludge Using Design of Experiment Method. Polymers. 2022; 14(16):3395. https://doi.org/10.3390/polym14163395
Chicago/Turabian StyleLo, Kang-Wei, Wei-Ting Lin, Ya-Wen Lin, Ta-Wui Cheng, and Kae-Long Lin. 2022. "Synthesis Metakaolin-Based Geopolymer Incorporated with SiC Sludge Using Design of Experiment Method" Polymers 14, no. 16: 3395. https://doi.org/10.3390/polym14163395
APA StyleLo, K.-W., Lin, W.-T., Lin, Y.-W., Cheng, T.-W., & Lin, K.-L. (2022). Synthesis Metakaolin-Based Geopolymer Incorporated with SiC Sludge Using Design of Experiment Method. Polymers, 14(16), 3395. https://doi.org/10.3390/polym14163395









