Influence of Functional Bio-Based Coatings Including Chitin Nanofibrils or Polyphenols on Mechanical Properties of Paper Tissues
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Raw Materials and Substrates Characterization
2.3. Coating Application
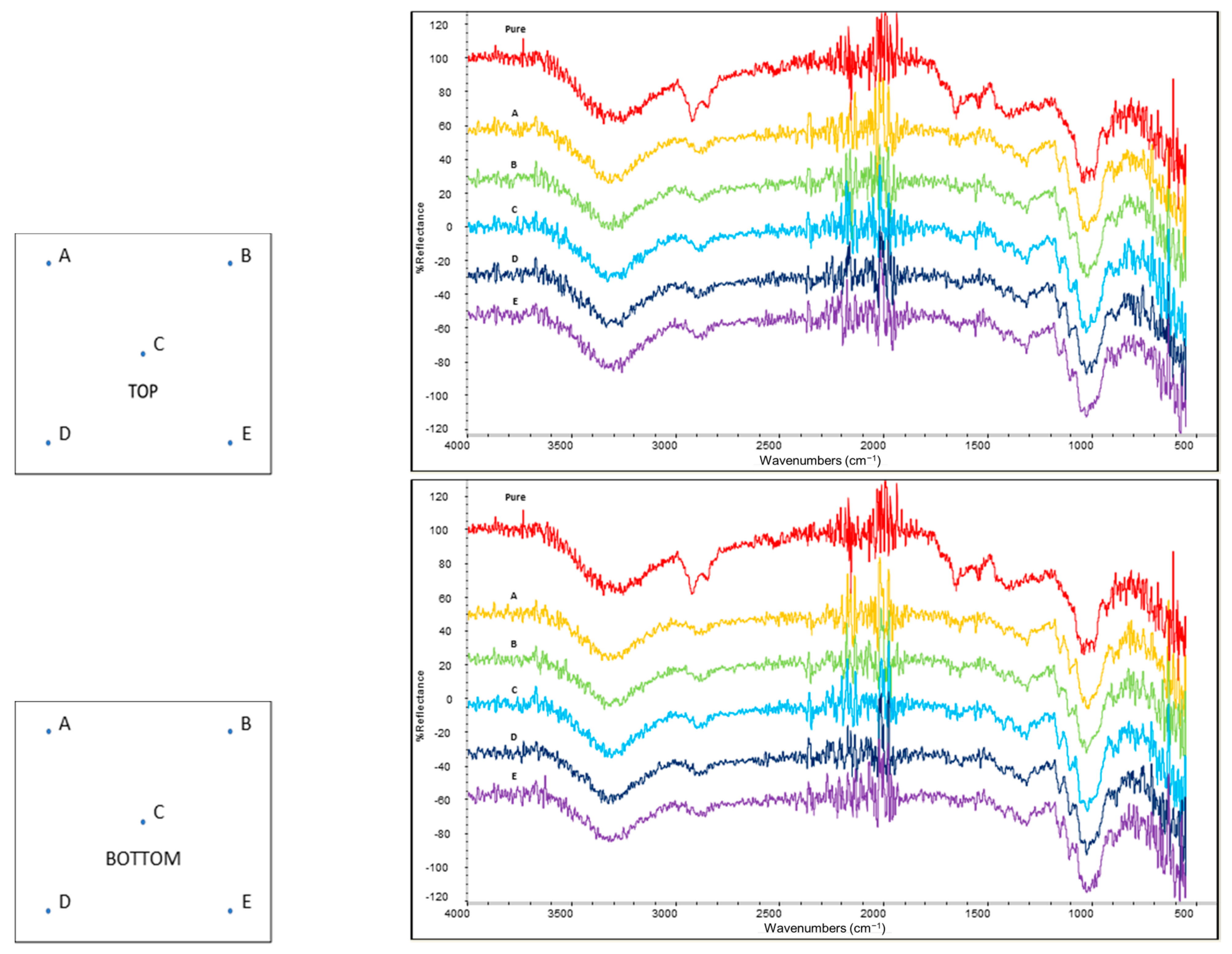
2.4. Coating Homogeneity Evaluation
2.5. Morphological Evaluation
2.6. Antioxidant and Antibacterial Assays
2.7. Mechanical Properties
2.8. Statistical Analysis
3. Results
3.1. Raw Materials Characterization
3.2. Homogeneity Evaluation
3.3. Antibacterial Test
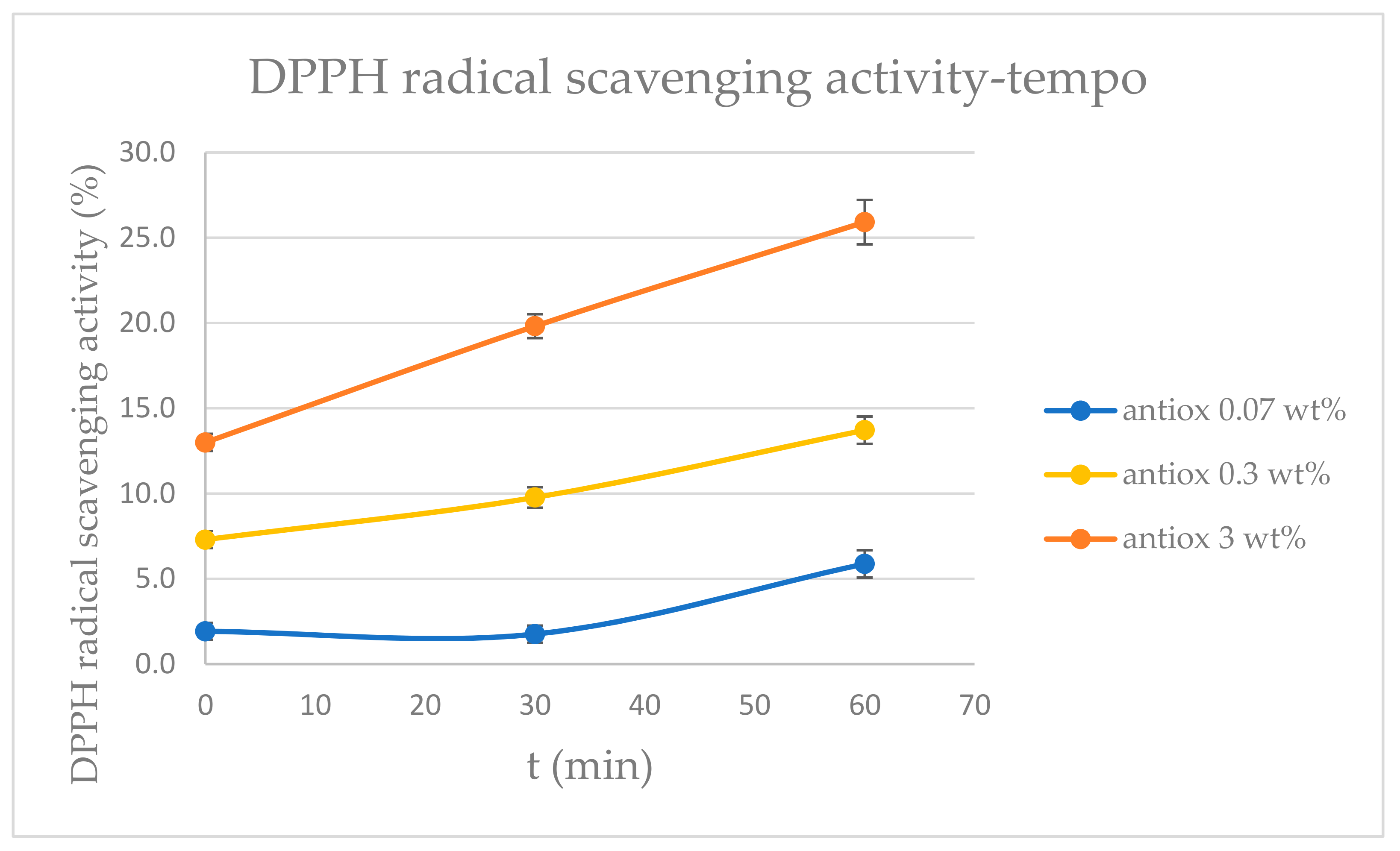
3.4. Antioxidant Test
3.5. Mechanical Properties
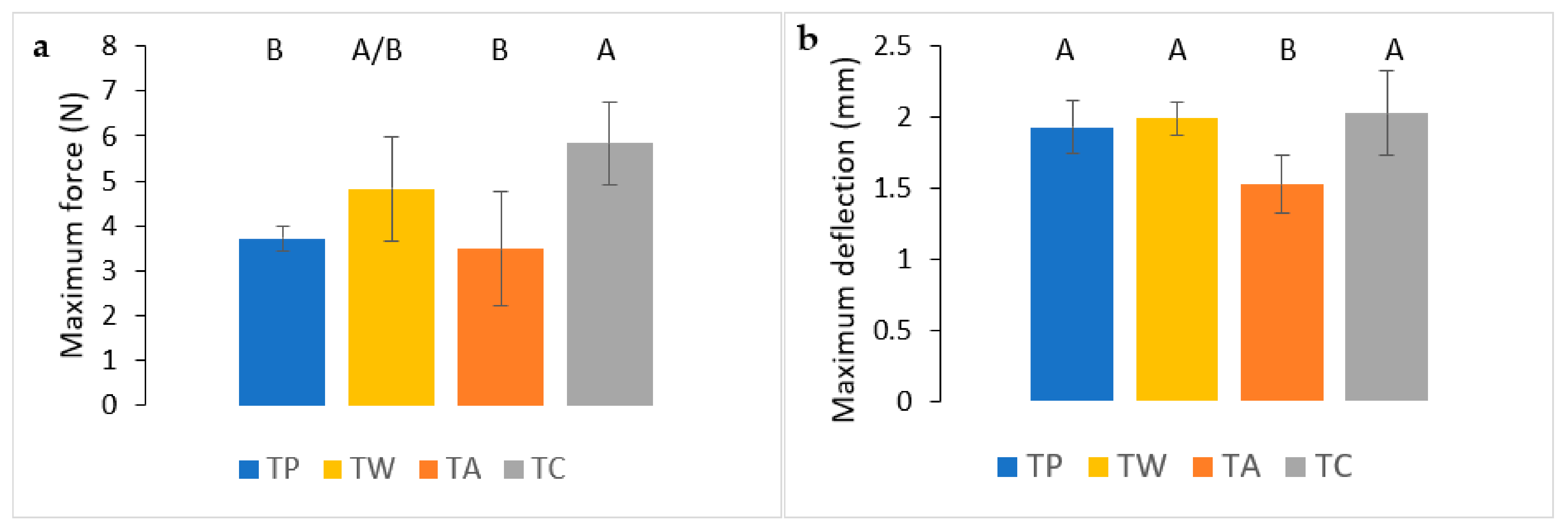
3.5.1. Puncture Resistance Test
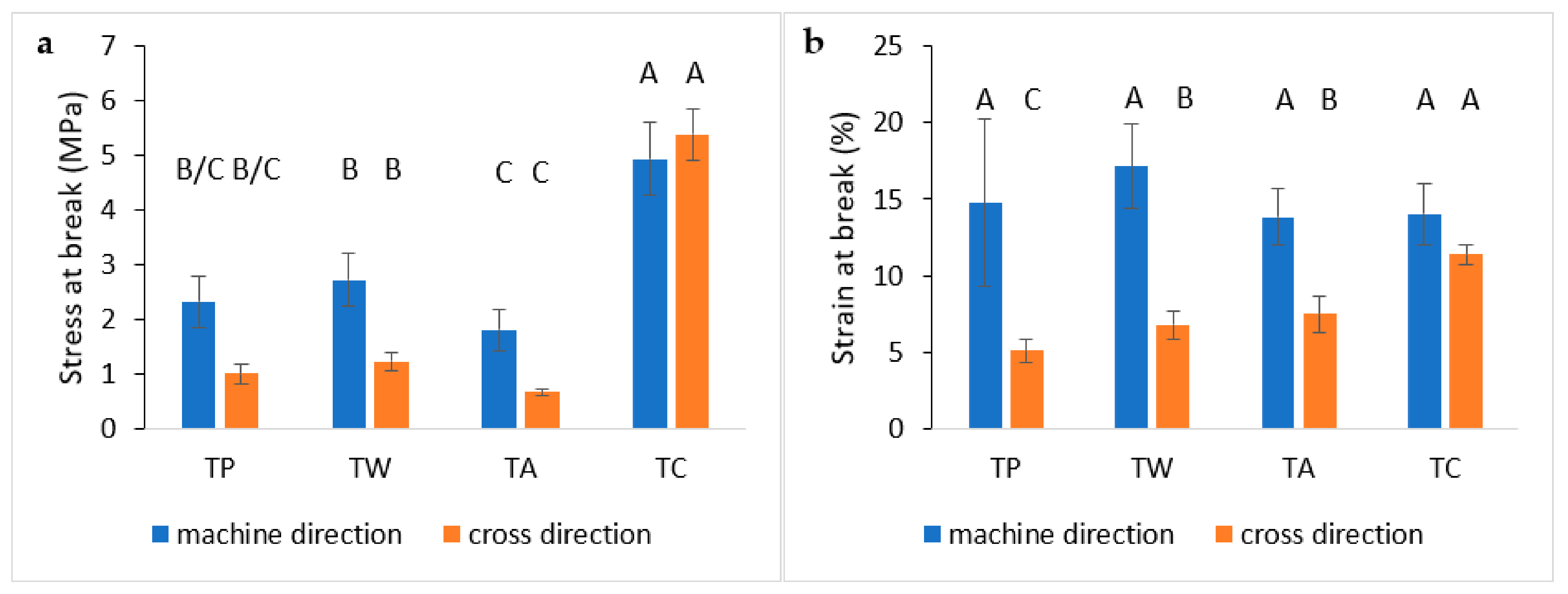
3.5.2. Tensile Test
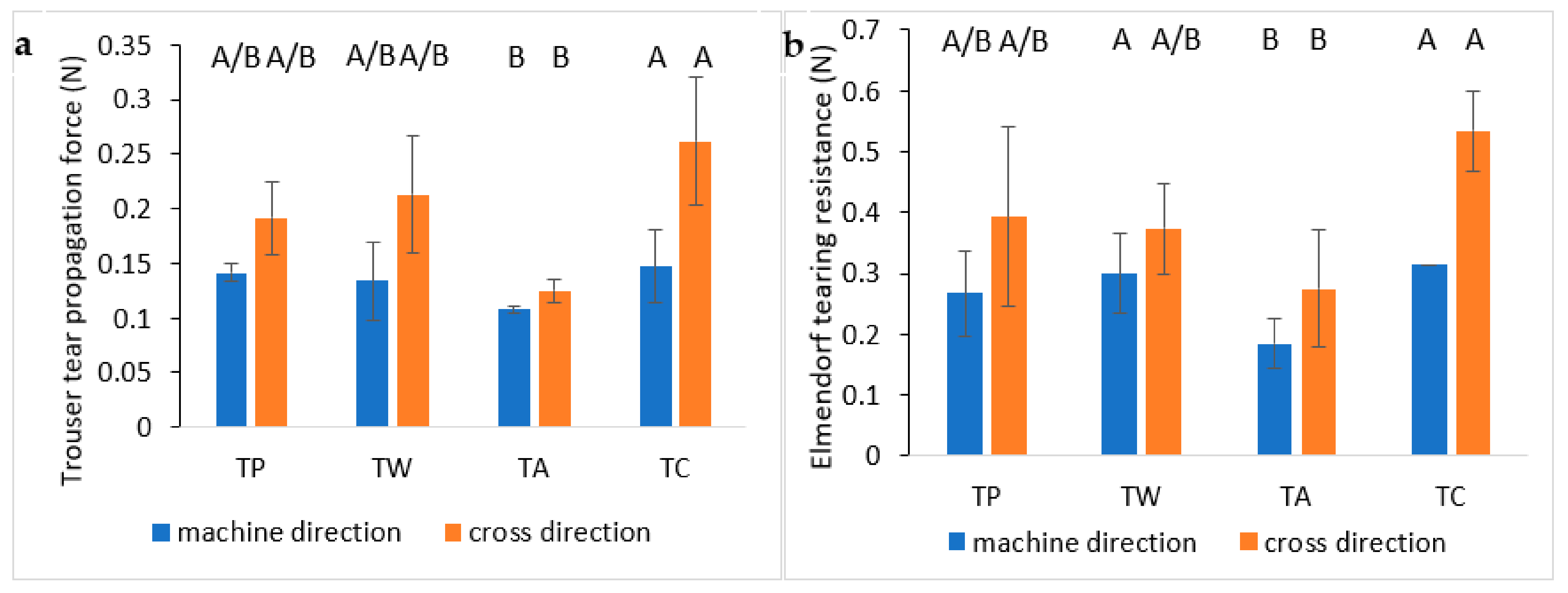
3.5.3. Tearing Tests
3.6. FESEM Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Xiong, S.J.; Pang, B.; Zhou, S.J.; Li, M.K.; Yang, S.; Wang, Y.Y.; Shi, Q.; Wang, S.F.; Yuan, T.Q.; Sun, R.C. Economically Competitive Biodegradable PBAT/Lignin Composites: Effect of Lignin Methylation and Compatibilizer. ACS Sustain. Chem. Eng. 2020, 8, 5338–5346. [Google Scholar] [CrossRef]
- Kjellgren, H.; Gällstedt, M.; Engström, G.; Järnström, L. Barrier and surface properties of chitosan-coated greaseproof paper. Carbohydr. Polym. 2006, 65, 453–460. [Google Scholar] [CrossRef]
- Zhang, W.; Xiao, H.; Qian, L. Enhanced water vapour barrier and grease resistance of paper bilayer-coated with chitosan and beeswax. Carbohydr. Polym. 2014, 101, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Xiao, H. Dual-Functional Beeswaxes on Enhancing Antimicrobial Activity and Water Vapor Barrier Property of Paper. ACS Appl. Mater. Interfaces 2013, 5, 3464–3468. [Google Scholar] [CrossRef] [PubMed]
- Cabañas-Romero, L.V.; Valls, C.; Valenzuela, S.V.; Roncero, M.B.; Pastor, F.I.J.; Diaz, P.; Martínez, J. Bacterial Cellulose–Chitosan Paper with Antimicrobial and Antioxidant Activities. Biomacromolecules 2020, 21, 1568–1577. [Google Scholar] [CrossRef]
- Wang, J.; Liu, X.; Milcovich, G.; Chen, T.-Y.; Durack, E.; Mallen, S.; Ruan, Y.; Weng, X.; Hudson, S.P. Co-reductive fabrication of carbon nanodots with high quantum yield for bioimaging of bacteria. Beilstein J. Nanotechnol. 2018, 9, 137–145. [Google Scholar] [CrossRef] [Green Version]
- Bartelmess, J.; Milcovich, G.; Maffeis, V.; d’Amora, M.; Bertozzi, S.M.; Giordani, S. Modulation of Efficient Diiodo-BODIPY in vitro Phototoxicity to Cancer Cells by Carbon Nano-Onions. Front. Chem. 2020, 8, 868. [Google Scholar] [CrossRef]
- Reichert, C.L.; Bugnicourt, E.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, A.; Canesi, I.; Braca, F.; Martínez, B.M.; Alonso, R.; Agostinis, L.; et al. Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability. Polymers 2020, 12, 1558. [Google Scholar] [CrossRef]
- De Barros, C.H.N.; Cruz, G.C.F.; Mayrink, W.; Tasic, L. Bio-based synthesis of silver nanoparticles from orange waste: Effects of distinct biomolecule coatings on size, morphology, and antimicrobial activity. Nanotechnol. Sci. Appl. 2018, 11, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Kocaman, S.; Karaman, M.; Gursoy, M.; Ahmetli, G. Chemical and plasma surface modification of lignocellulose coconut waste for the preparation of advanced biobased composite materials. Carbohydr. Polym. 2017, 159, 48–57. [Google Scholar] [CrossRef]
- Kuppusamy, S.; Thavamani, P.; Megharaj, M.; Naidu, R. Bioremediation potential of natural polyphenol rich green wastes: A review of current research and recommendations for future directions. Environ. Technol. Innov. 2015, 4, 17–28. [Google Scholar] [CrossRef]
- Castro-Muñoz, R.; Orozco-Álvarez, C.; Yáñez-Fernández, J. Recovery of bioactive compounds from food processing wastewaters by Ultra and Nanofiltration: A review. Adv. Bio Res. 2015, 6, 152–158. [Google Scholar]
- Mohamed Khalith, S.B.; Ramalingam, R.; Karuppannan, S.K.; Dowlath, M.J.H.; Kumar, R.; Vijayalakshmi, S.; Uma Maheshwari, R.; Arunachalam, K.D. Synthesis and characterization of polyphenols functionalized graphitic hematite nanocomposite adsorbent from an agro waste and its application for removal of Cs from aqueous solution. Chemosphere 2022, 286, 131493. [Google Scholar] [CrossRef] [PubMed]
- Di Donato, P.; Taurisano, V.; Tommonaro, G.; Pasquale, V.; Jiménez, J.M.S.; de Pascual-Teresa, S.; Poli, A.; Nicolaus, B. Biological Properties of Polyphenols Extracts from Agro Industry’s Wastes. Waste Biomass Valorization 2018, 9, 1567–1578. [Google Scholar] [CrossRef]
- Castaldo, L.; Izzo, L.; De Pascale, S.; Narváez, A.; Rodriguez-Carrasco, Y.; Ritieni, A. Chemical Composition, In Vitro Bioaccessibility and Antioxidant Activity of Polyphenolic Compounds from Nutraceutical Fennel Waste Extract. Molecules 2021, 26, 1968. [Google Scholar] [CrossRef]
- Prakash, A.; Vadivel, V.; Banu, S.F.; Nithyanand, P.; Lalitha, C.; Brindha, P. Evaluation of antioxidant and antimicrobial properties of solvent extracts of agro-food by-products (cashew nut shell, coconut shell and groundnut hull). Agric. Nat. Resour. 2018, 52, 451–459. [Google Scholar] [CrossRef]
- Pokhrel, S.; Yadav, P.N.; Adhikari, R. Applications of Chitin and Chitosan in Industry and Medical Science: A Review. Nepal J. Sci. Technol. 2016, 16, 99–104. [Google Scholar] [CrossRef] [Green Version]
- Abdou, E.S.; Nagy, K.S.A.; Elsabee, M.Z. Extraction and characterization of chitin and chitosan from local sources. Bioresour. Technol. 2008, 99, 1359–1367. [Google Scholar] [CrossRef]
- Abdelmalek, B.E.; Sila, A.; Haddar, A.; Bougatef, A.; Ayadi, M.A. β-Chitin and chitosan from squid gladius: Biological activities of chitosan and its application as clarifying agent for apple juice. Int. J. Biol. Macromol. 2017, 104, 953–962. [Google Scholar] [CrossRef]
- Triunfo, M.; Tafi, E.; Guarnieri, A.; Salvia, R.; Scieuzo, C.; Hahn, T.; Zibek, S.; Gagliardini, A.; Panariello, L.; Coltelli, M.B.; et al. Characterization of chitin and chitosan derived from Hermetia illucens, a further step in a circular economy process. Sci. Rep. 2022, 12, 6613. [Google Scholar] [CrossRef]
- Hahn, T.; Tafi, E.; Paul, A.; Salvia, R.; Falabella, P.; Zibek, S. Current state of chitin purification and chitosan production from insects. J. Chem. Technol. Biotechnol. 2020, 95, 2775–2795. [Google Scholar] [CrossRef]
- Percot, A.; Viton, C.; Domard, A. Optimization of Chitin Extraction from Shrimp Shells. Biomacromolecules 2003, 4, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Fazli Wan Nawawi, W.M.; Lee, K.-Y.; Kontturi, E.; Murphy, R.J.; Bismarck, A. Chitin Nanopaper from Mushroom Extract: Natural Composite of Nanofibers and Glucan from a Single Biobased Source. ACS Sustain. Chem. Eng. 2019, 7, 6492–6496. [Google Scholar] [CrossRef] [Green Version]
- Hassainia, A.; Satha, H.; Boufi, S. Chitin from Agaricus bisporus: Extraction and characterization. Int. J. Biol. Macromol. 2018, 117, 1334–1342. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, C.; Morganti, P. Preparation of Chitin and Derivatives Thereof for Cosmetic and Therapeutic Use. US Patent WO2006048829A2, 21 September 2006. [Google Scholar]
- Alexandru, L.; Binello, A.; Mantegna, S.; Boffa, L.; Chemat, F.; Cravotto, G. Efficient green extraction of polyphenols from post-harvested agro-industry vegetal sources in Piedmont. C. R. Chim. 2014, 17, 212–217. [Google Scholar] [CrossRef]
- Mellinas, A.C.; Jiménez, A.; Garrigós, M.C. Optimization of microwave-assisted extraction of cocoa bean shell waste and evaluation of its antioxidant, physicochemical and functional properties. LWT 2020, 127, 109361. [Google Scholar] [CrossRef]
- Zhao, D.; Huang, W.-C.; Guo, N.; Zhang, S.; Xue, C.; Mao, X. Two-Step Separation of Chitin from Shrimp Shells Using Citric Acid and Deep Eutectic Solvents with the Assistance of Microwave. Polymers 2019, 11, 409. [Google Scholar] [CrossRef] [Green Version]
- Cassano, A.; Conidi, C.; Ruby-Figueroa, R.; Castro-Muñoz, R. Nanofiltration and Tight Ultrafiltration Membranes for the Recovery of Polyphenols from Agro-Food By-Products. Int. J. Mol. Sci. 2018, 19, 351. [Google Scholar] [CrossRef] [Green Version]
- Cravotto, G.; Mariatti, F.; Gunjevic, V.; Secondo, M.; Villa, M.; Parolin, J.; Cavaglià, G. Pilot Scale Cavitational Reactors and Other Enabling Technologies to Design the Industrial Recovery of Polyphenols from Agro-Food By-Products, a Technical and Economical Overview. Foods 2018, 7, 130. [Google Scholar] [CrossRef] [Green Version]
- Hu, Z.; Berry, R.M.; Pelton, R.; Cranston, E.D. One-Pot Water-Based Hydrophobic Surface Modification of Cellulose Nanocrystals Using Plant Polyphenols. ACS Sustain. Chem. Eng. 2017, 5, 5018–5026. [Google Scholar] [CrossRef]
- Hai, L.; Choi, E.S.; Zhai, L.; Panicker, P.S.; Kim, J. Green nanocomposite made with chitin and bamboo nanofibers and its mechanical, thermal and biodegradable properties for food packaging. Int. J. Biol. Macromol. 2020, 144, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Danti, S.; Trombi, L.; Fusco, A.; Azimi, B.; Lazzeri, A.; Morganti, P.; Coltelli, M.-B.M.-B.; Donnarumma, G. Chitin Nanofibrils and Nanolignin as Functional Agents in Skin Regeneration. Int. J. Mol. Sci. 2019, 20, 2669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Andrade Arruda Fernandes, I.; Maciel, G.M.; Ribeiro, V.R.; Rossetto, R.; Pedro, A.C.; Haminiuk, C.W.I. The role of bacterial cellulose loaded with plant phenolics in prevention of UV-induced skin damage. Carbohydr. Polym. Technol. Appl. 2021, 2, 100122. [Google Scholar] [CrossRef]
- Rahman Liman, M.L.; Islam, M.T.; Repon, M.R.; Hossain, M.M.; Sarker, P. Comparative dyeing behavior and UV protective characteristics of cotton fabric treated with polyphenols enriched banana and watermelon biowaste. Sustain. Chem. Pharm. 2021, 21, 100417. [Google Scholar] [CrossRef]
- Sajadimajd, S.; Bahramsoltani, R.; Iranpanah, A.; Kumar Patra, J.; Das, G.; Gouda, S.; Rahimi, R.; Rezaeiamiri, E.; Cao, H.; Giampieri, F.; et al. Advances on Natural Polyphenols as Anticancer Agents for Skin Cancer. Pharmacol. Res. 2020, 151, 104584. [Google Scholar] [CrossRef] [PubMed]
- Casadidio, C.; Peregrina, D.V.; Gigliobianco, M.R.; Deng, S.; Censi, R.; Di Martino, P. Chitin and Chitosans: Characteristics, Eco-Friendly Processes, and Applications in Cosmetic Science. Mar. Drugs 2019, 17, 369. [Google Scholar] [CrossRef] [Green Version]
- Azimi, B.; Ricci, C.; Fusco, A.; Zavagna, L.; Linari, S.; Donnarumma, G.; Hadrich, A.; Cinelli, P.; Coltelli, M.-B.; Danti, S. Electrosprayed Shrimp and Mushroom Nanochitins on Cellulose Tissue for Skin Contact Application. Molecules 2021, 26, 4374. [Google Scholar] [CrossRef]
- Khan, M.K.; Paniwnyk, L.; Hassan, S. Polyphenols as Natural Antioxidants: Sources, Extraction and Applications in Food, Cosmetics and Drugs. In Plant Based ’’Green Chemistry 2.0’’: Moving from Evolutionary to Revolutionary; Li, Y., Chemat, F., Eds.; Springer Singapore: Singapore, 2019; pp. 197–235. ISBN 978-981-13-3810-6. [Google Scholar]
- JiangLian, D.; ShaoYing, Z. Application of chitosan based coating in fruit and vegetable preservation: A review. J. Food Process. Technol. 2013, 4, 227. [Google Scholar] [CrossRef] [Green Version]
- Hu, X.; Sun, Z.; Zhu, X.; Sun, Z. Montmorillonite-Synergized Water-Based Intumescent Flame Retardant Coating for Plywood. Coatings 2020, 10, 109. [Google Scholar] [CrossRef] [Green Version]
- Han, Y.; Chen, S.; Yang, M.; Zou, H.; Zhang, Y. Inorganic matter modified water-based copolymer prepared by chitosan-starch-CMC-Na-PVAL as an environment-friendly coating material. Carbohydr. Polym. 2020, 234, 115925. [Google Scholar] [CrossRef]
- LeCorre, D.; Dufresne, A.; Rueff, M.; Khelifi, B.; Bras, J. All starch nanocomposite coating for barrier material. J. Appl. Polym. Sci. 2014, 131, 39826. [Google Scholar] [CrossRef]
- Mates, J.E.; Schutzius, T.M.; Bayer, I.S.; Qin, J.; Waldroup, D.E.; Megaridis, C.M. Water-Based Superhydrophobic Coatings for Nonwoven and Cellulosic Substrates. Ind. Eng. Chem. Res. 2014, 53, 222–227. [Google Scholar] [CrossRef]
- Rentzhog, M.; Fogden, A. Print quality and resistance for water-based flexography on polymer-coated boards: Dependence on ink formulation and substrate pretreatment. Prog. Org. Coatings 2006, 57, 183–194. [Google Scholar] [CrossRef]
- Murthy, S.; Matschuk, M.; Huang, Q.; Mandsberg, N.K.; Feidenhans’l, N.A.; Johansen, P.; Christensen, L.; Pranov, H.; Kofod, G.; Pedersen, H.C.; et al. Fabrication of Nanostructures by Roll-to-Roll Extrusion Coating. Adv. Eng. Mater. 2016, 18, 484–489. [Google Scholar] [CrossRef] [Green Version]
- Gregory, B.H. Extrusion Coating: A Process Manual; Trafford Publishing: Bloomington, IN, USA, 2005; ISBN 9781412040723. [Google Scholar]
- Arulkumar, S.; Parthiban, S.; Goswami, A.; Varma, R.S.; Naushad, M.; Gawande, M.B. Low temperature processed titanium oxide thin-film using scalable wire-bar coating. Mater. Res. Express 2019, 6, 126427. [Google Scholar] [CrossRef]
- Kanwal, M.; Wang, X.; Shahzad, H.; Chen, Y.; Chai, H. Blade coating analysis of viscous nanofluid having Cu–water nanoparticles using flexible blade coater. J. Plast. Film Sheeting 2020, 36, 348–367. [Google Scholar] [CrossRef]
- Panariello, L.; Vannozzi, A.; Morganti, P.; Coltelli, M.-B.; Lazzeri, A. Biobased and Eco-Compatible Beauty Films Coated with Chitin Nanofibrils, Nanolignin and Vitamin E. Cosmetics 2021, 8, 27. [Google Scholar] [CrossRef]
- Kimpimäki, T.; Savolainen, A.V. Barrier dispersion coating of paper and board. In Surface Application of Paper Chemicals; Brander, J., Thorn, I., Eds.; Springer: Dordrecht, The Netherlands, 1997; pp. 208–228. ISBN 978-94-009-1457-5. [Google Scholar]
- Sun, Q.; Schork, F.J.; Deng, Y. Water-based polymer/clay nanocomposite suspension for improving water and moisture barrier in coating. Compos. Sci. Technol. 2007, 67, 1823–1829. [Google Scholar] [CrossRef]
- Tyagi, P.; Lucia, L.A.; Hubbe, M.A.; Pal, L. Nanocellulose-based multilayer barrier coatings for gas, oil, and grease resistance. Carbohydr. Polym. 2019, 206, 281–288. [Google Scholar] [CrossRef]
- Panariello, L.; Coltelli, M.-B.M.B.; Buchignani, M.; Lazzeri, A. Chitosan and nano-structured chitin for biobased anti-microbial treatments onto cellulose based materials. Eur. Polym. J. 2019, 113, 328–339. [Google Scholar] [CrossRef]
- Battisti, R.; Fronza, N.; Vargas Júnior, Á.; da Silveira, S.M.; Damas, M.S.P.; Quadri, M.G.N. Gelatin-coated paper with antimicrobial and antioxidant effect for beef packaging. Food Packag. Shelf Life 2017, 11, 115–124. [Google Scholar] [CrossRef]
- Kim, C.-K.; Lim, W.-S.; Lee, Y.K. Studies on the fold-ability of coated paperboard (I): Influence of latex on fold-ability during creasing/folding coated paperboard. J. Ind. Eng. Chem. 2010, 16, 842–847. [Google Scholar] [CrossRef]
- Nagasawa, S.; Fukuzawa, Y.; Yamaguchi, T.; Tsukatani, S.; Katayama, I. Effect of crease depth and crease deviation on folding deformation characteristics of coated paperboard. J. Mater. Process. Technol. 2003, 140, 157–162. [Google Scholar] [CrossRef]
- Sun, J.; Bi, H.; Jia, H.; Su, S.; Dong, H.; Xie, X.; Sun, L. A low cost paper tissue-based PDMS/SiO2 composite for both high efficient oil absorption and water-in-oil emulsion separation. J. Clean. Prod. 2020, 244, 118814. [Google Scholar] [CrossRef]
- Nhuapeng, W.; Thamjaree, W. Fabrication and Mechanical Properties of Hybrid Composites between Pineapple fiber/Styrofoam Particle/Paper Tissue. Mater. Today Proc. 2019, 17, 1444–1450. [Google Scholar] [CrossRef]
- Ferreira, A.C.S.; Aguado, R.; Bértolo, R.; Carta, A.M.M.S.; Murtinho, D.; Valente, A.J.M. Enhanced water absorption of tissue paper by cross-linking cellulose with poly(vinyl alcohol). Chem. Pap. 2022. [Google Scholar] [CrossRef]
- Spina, R.; Cavalcante, B. Characterizing materials and processes used on paper tissue converting lines. Mater. Today Commun. 2018, 17, 427–437. [Google Scholar] [CrossRef]
- De Assis, T.; Reisinger, L.W.; Pal, L.; Pawlak, J.; Jameel, H.; Gonzalez, R.W. Understanding the Effect of Machine Technology and Cellulosic Fibers on Tissue Properties—A Review. BioResources 2018, 13, 4593–4629. [Google Scholar] [CrossRef] [Green Version]
- Preston, J.; Elton, N.J.; Legrix, A.; Nutbeem, C.; Husband, J.C. The role of pore density in the setting of offset printing ink on coated paper. Tappi J. 2002, 1, 3–5. [Google Scholar]
- Schoelkopf, J.; Matthews, G.P. Influence of inertia on liquid absorption into paper coating structures. Nord. Pulp Pap. Res. J. 2000, 15, 422–430. [Google Scholar] [CrossRef]
- Lee, H.K.; Joyce, M.K.; Fleming, P.D. Influence of Pigment Particle Size and Pigment Ratio on Printability of Glossy Ink Jet Paper Coatings. J. Imaging Sci. Technol. 2005, 49, 54–60. [Google Scholar]
- Li, Y.; He, B. Characterization of Ink Pigment Penetration and Distribution Related to Surface Topography of Paper Using Confocal Laser Scanning Microscopy. BioResources 2011, 6, 2690–2702. [Google Scholar]
- Ginebreda, A.; Guillén, D.; Barceló, D.; Darbra, R.M. Additives in the Paper Industry. In Global Risk-Based Management of Chemical Additives I: Production, Usage and Environmental Occurrence; Bilitewski, B., Darbra, R.M., Barceló, D., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 11–34. ISBN 978-3-642-24876-4. [Google Scholar]
- Yong, H.; Liu, J. Active packaging films and edible coatings based on polyphenol-rich propolis extract: A review. Compr. Rev. Food Sci. Food Saf. 2021, 20, 2106–2145. [Google Scholar] [CrossRef] [PubMed]
- Kandirmaz, E.A. Fabrication of rosemary essential oil microcapsules and using in active packaging. Nord. Pulp Pap. Res. J. 2021, 36, 323–330. [Google Scholar] [CrossRef]
- Nechita, P.; Roman, M. Review on Polysaccharides Used in Coatings for Food Packaging Papers. Coatings 2020, 10, 566. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Wild, F.; Bugnicourt, E.; Cinelli, P.; Lindner, M.; Schmid, M.; Weckel, V.; Müller, K.; Rodriguez, P.; Staebler, A.; et al. State of the Art in the Development and Properties of Protein-Based Films and Coatings and Their Applicability to Cellulose Based Products: An Extensive Review. Coatings 2016, 6, 1. [Google Scholar] [CrossRef]
- Quiles-Carrillo, L.; Mellinas, C.; Garrigos, M.C.; Balart, R.; Torres-Giner, S. Optimization of Microwave-Assisted Extraction of Phenolic Compounds with Antioxidant Activity from Carob Pods. Food Anal. Methods 2019, 12, 2480–2490. [Google Scholar] [CrossRef]
- Sirivibulkovit, K.; Nouanthavong, S.; SameenoI, Y. Paper-based DPPH Assay for Antioxidant Activity Analysis. Anal. Sci. 2018, 34, 795–800. [Google Scholar] [CrossRef] [Green Version]
- Lange, J.; Mokdad, H.; Wysery, Y. Understanding Puncture Resistance and Perforation Behavior of Packaging Laminates. J. Plast. Film Sheeting 2002, 18, 231–244. [Google Scholar] [CrossRef]
- Irudayaraj, J.; Xu, F.; Tewari, J. Rapid determination of invert cane sugar adulteration in honey using FTIR spectroscopy and multivariate analysis. J. Food Sci. 2003, 68, 2040–2045. [Google Scholar] [CrossRef]
- Barakat, H.H.; Hussein, S.A.M.; Marzouk, M.S.; Merfort, I.; Linscheid, M.; Nawwar, M.A.M. Polyphenolic metabolites of Epilobium hirsutum. Phytochemistry 1997, 46, 935–941. [Google Scholar] [CrossRef]
- Kasaai, M.R. A review of several reported procedures to determine the degree of N-acetylation for chitin and chitosan using infrared spectroscopy. Carbohydr. Polym. 2008, 71, 497–508. [Google Scholar] [CrossRef]
- Kalogeropoulos, N.; Chiou, A.; Pyriochou, V.; Peristeraki, A.; Karathanos, V.T. Bioactive phytochemicals in industrial tomatoes and their processing byproducts. LWT—Food Sci. Technol. 2012, 49, 213–216. [Google Scholar] [CrossRef]
- Li, T.-T.; Wang, R.; Lou, C.W.; Lin, J.-H. Evaluation of high-modulus, puncture-resistance composite nonwoven fabrics by response surface methodology. J. Ind. Text. 2013, 43, 247–263. [Google Scholar] [CrossRef]
- Hassim, N.; Ahmad, M.R.; Ahmad, W.Y.W.; Samsuri, A.; Yahya, M.H.M. Puncture resistance of natural rubber latex unidirectional coated fabrics. J. Ind. Text. 2012, 42, 118–131. [Google Scholar] [CrossRef]
- Wang, Q.-S.; Sun, R.-J.; Tian, X.; Yao, M.; Feng, Y. Quasi-static puncture resistance behaviors of high-strength polyester fabric for soft body armor. Results Phys. 2016, 6, 554–560. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.; Vasita, R. Understanding the relation between structural and mechanical properties of electrospun fiber mesh through uniaxial tensile testing. J. Appl. Polym. Sci. 2017, 134, 45012. [Google Scholar] [CrossRef]
- Yamauchi, T.; Tanaka, A. Tearing test for paper using a tensile tester. J. Wood Sci. 2002, 48, 532–535. [Google Scholar] [CrossRef]
- Zhong, T.; Wolcott, M.P.; Liu, H.; Wang, J. Developing chitin nanocrystals for flexible packaging coatings. Carbohydr. Polym. 2019, 226, 115276. [Google Scholar] [CrossRef]
- Haslach, H.W. The Moisture and Rate-Dependent Mechanical Properties of Paper: A Review. Mech. Time-Dependent Mater. 2000, 4, 169–210. [Google Scholar] [CrossRef]

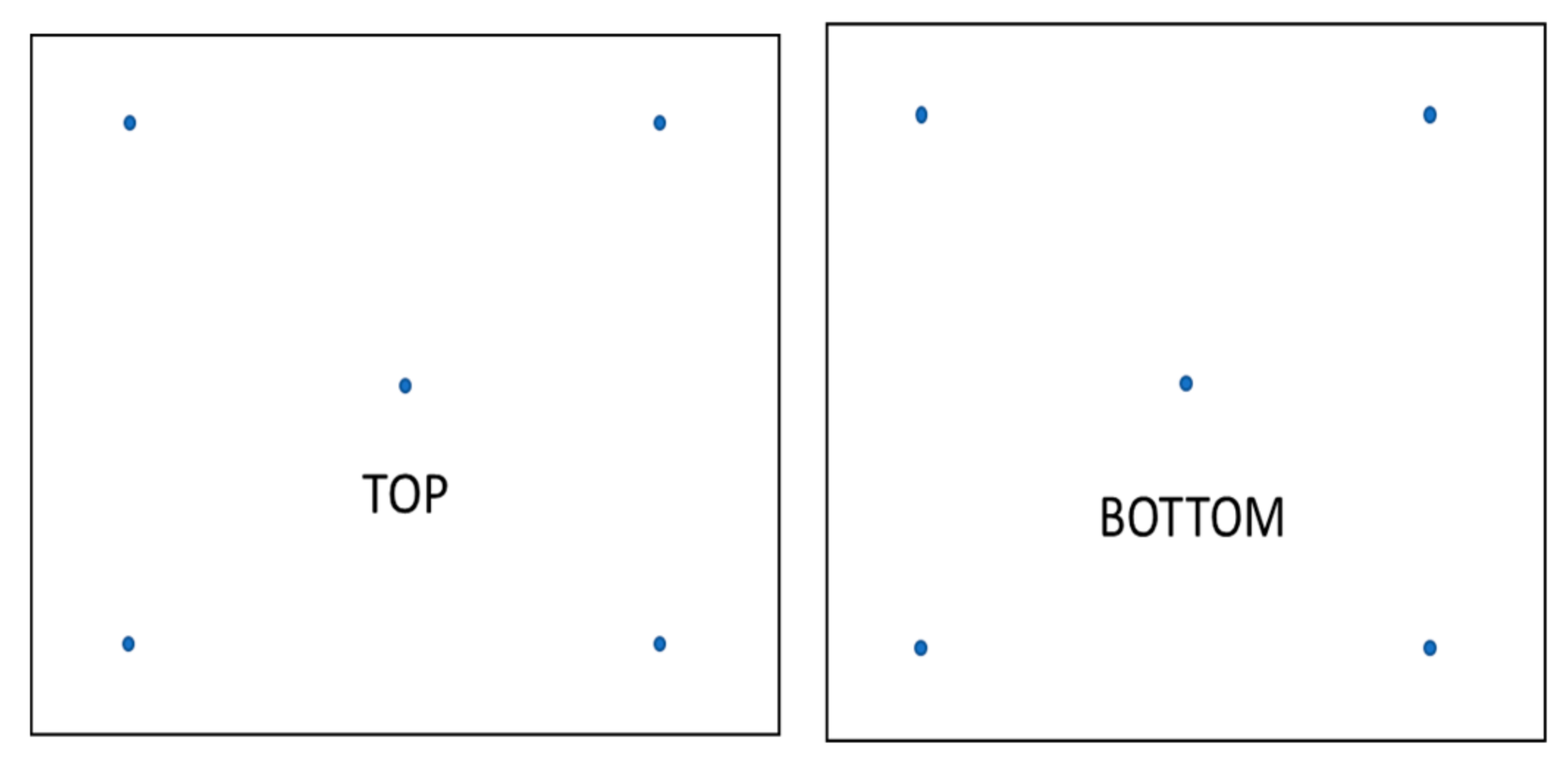
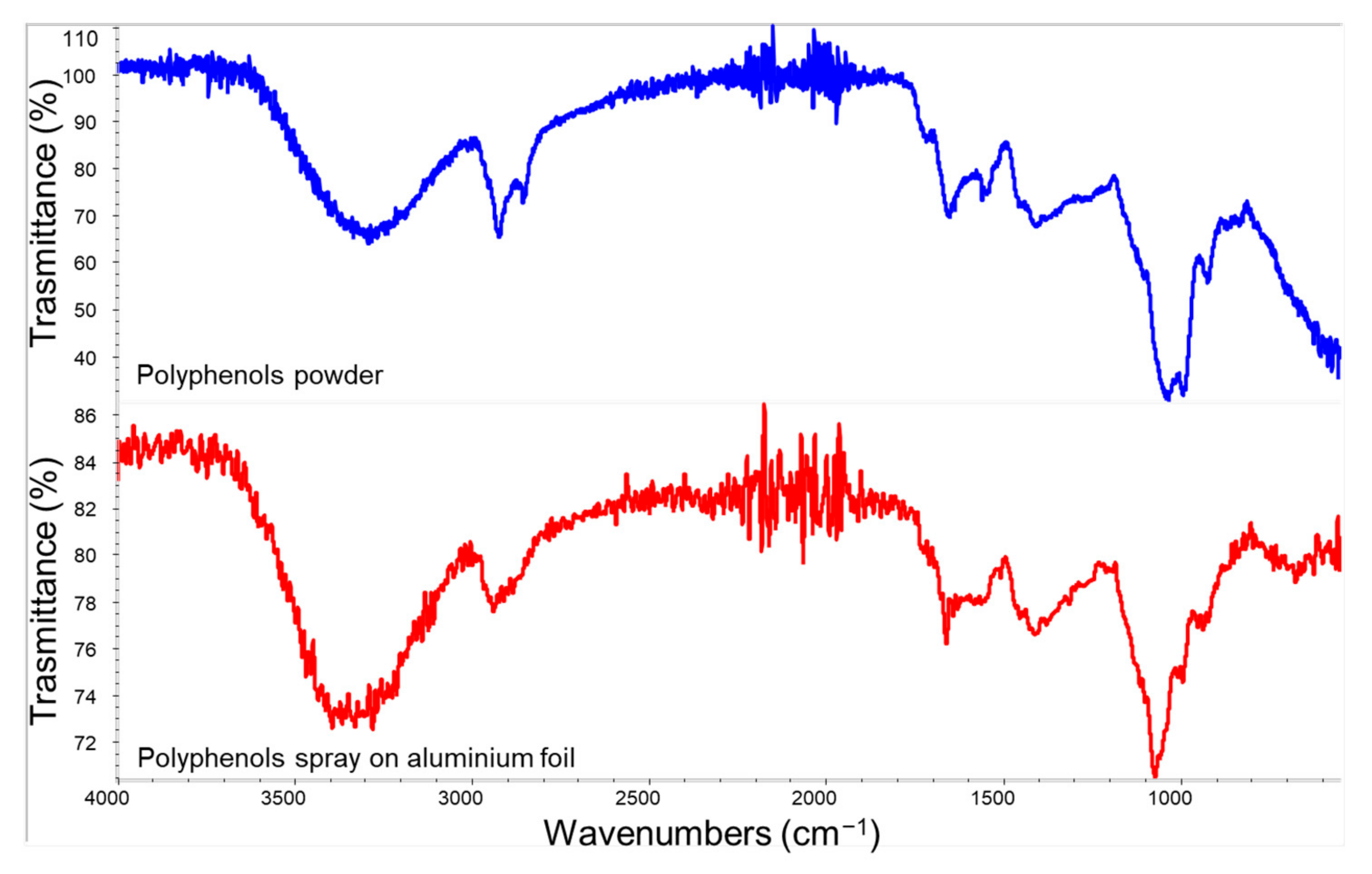
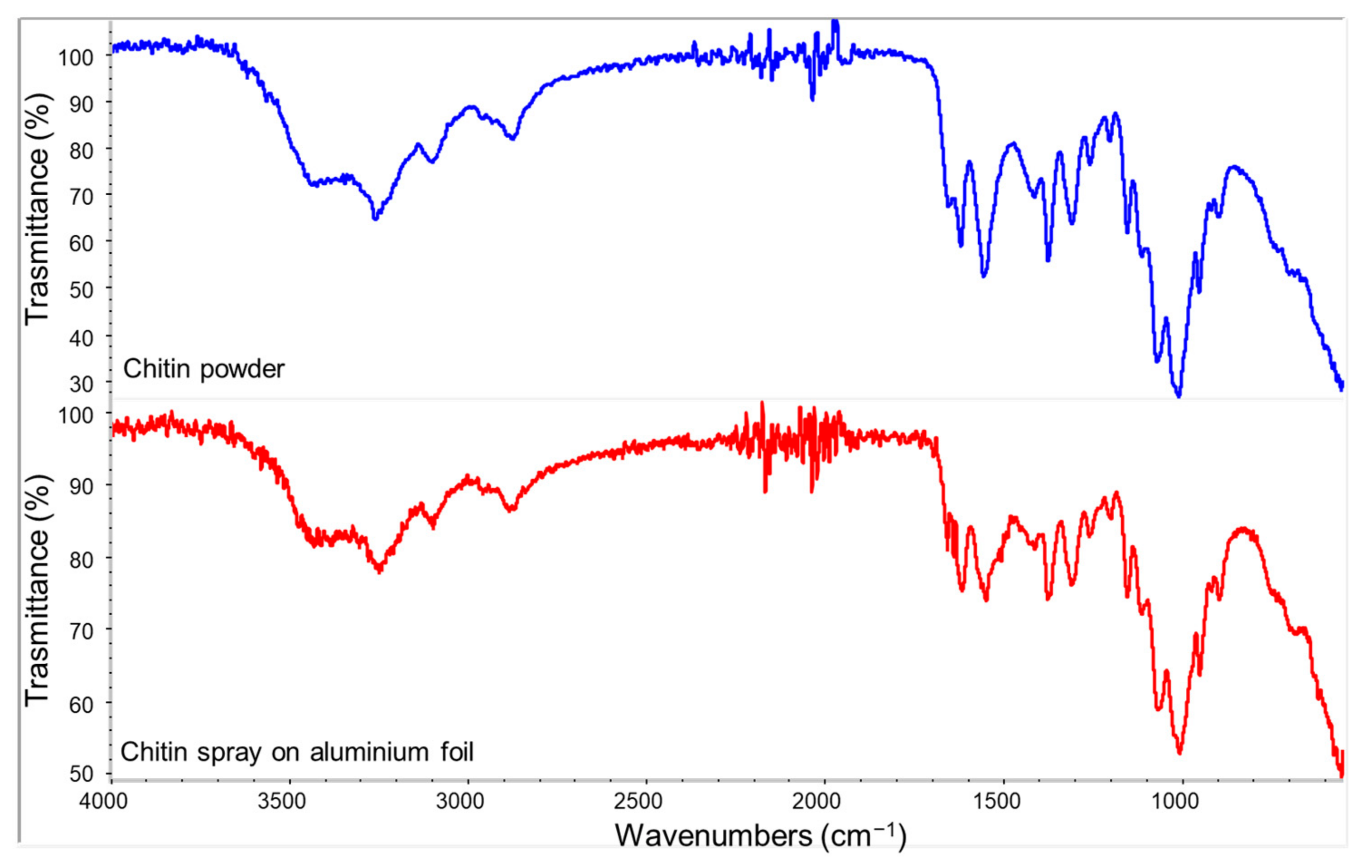
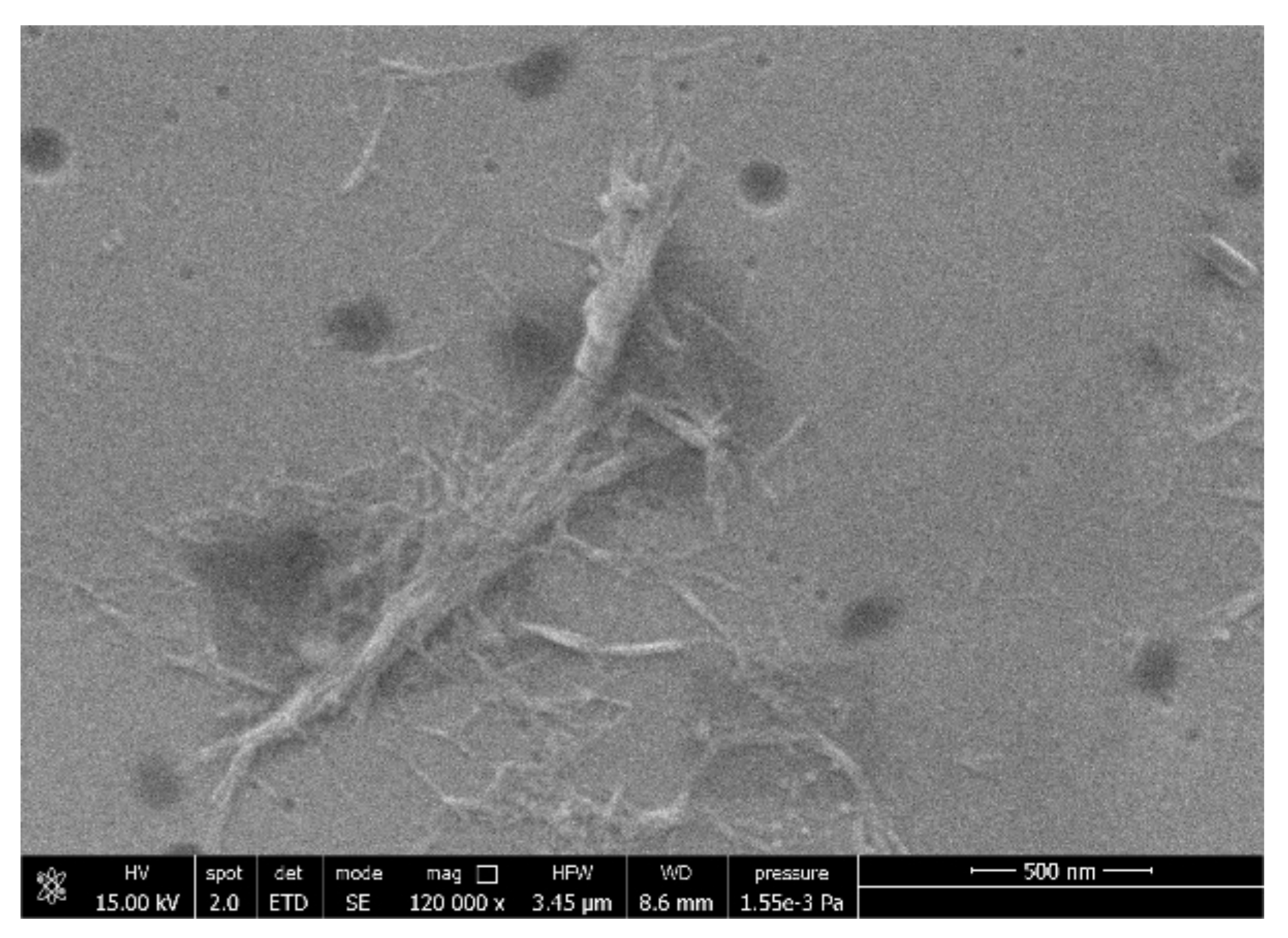



| Name | Composition |
|---|---|
| TP | Pure tissue |
| TW | Tissue treated with sole water |
| TA | Tissue treated with 10 wt% antioxidant polyphenols |
| TC | Tissue treated with 7.5 wt% ChNFs |
| Sample | Water Content (wt%) | Cellulose Degradation Onset Temperature (°C) | Cellulose Degradation (wt%) | Residue in N2 (900 °C) (wt%) | Residue in Air (900 °C) (wt%) |
|---|---|---|---|---|---|
| Tissue | 5.66 | 275 | 81.63 | 12.71 | 1.10 |
| Untreated | Tissue + 1 wt% Chitin | Tissue + 7.5 wt% Chitin | |
|---|---|---|---|
| Enumeration of bacteria on surface (CFU/g) | 1390 ± 120 | 867 ± 33 | 280 ± 17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panariello, L.; Coltelli, M.-B.; Giangrandi, S.; Garrigós, M.C.; Hadrich, A.; Lazzeri, A.; Cinelli, P. Influence of Functional Bio-Based Coatings Including Chitin Nanofibrils or Polyphenols on Mechanical Properties of Paper Tissues. Polymers 2022, 14, 2274. https://doi.org/10.3390/polym14112274
Panariello L, Coltelli M-B, Giangrandi S, Garrigós MC, Hadrich A, Lazzeri A, Cinelli P. Influence of Functional Bio-Based Coatings Including Chitin Nanofibrils or Polyphenols on Mechanical Properties of Paper Tissues. Polymers. 2022; 14(11):2274. https://doi.org/10.3390/polym14112274
Chicago/Turabian StylePanariello, Luca, Maria-Beatrice Coltelli, Simone Giangrandi, María Carmen Garrigós, Ahdi Hadrich, Andrea Lazzeri, and Patrizia Cinelli. 2022. "Influence of Functional Bio-Based Coatings Including Chitin Nanofibrils or Polyphenols on Mechanical Properties of Paper Tissues" Polymers 14, no. 11: 2274. https://doi.org/10.3390/polym14112274
APA StylePanariello, L., Coltelli, M.-B., Giangrandi, S., Garrigós, M. C., Hadrich, A., Lazzeri, A., & Cinelli, P. (2022). Influence of Functional Bio-Based Coatings Including Chitin Nanofibrils or Polyphenols on Mechanical Properties of Paper Tissues. Polymers, 14(11), 2274. https://doi.org/10.3390/polym14112274











